Effects of Marine Microalgae on the Developmental Growth of the Sea Urchin Larviculture Anthocidaris crassipina
Abstract
1. Introduction
2. Materials and Methods
2.1. Sea Urchin Samples
2.2. Insemination and Hatchery
2.3. Embryonic and Larval Development
2.4. Microalgal Diets
2.5. Experimental Grouping
2.6. Body Length Measurement and Morphology
2.7. Statistical Analysis
3. Results
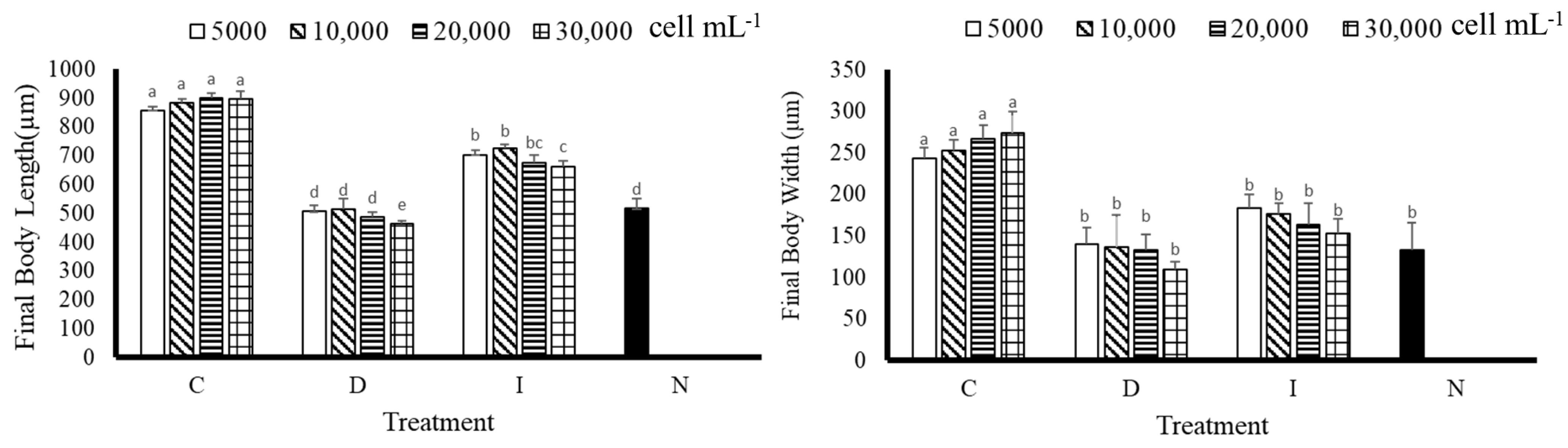
3.1. Body Length and Body Width
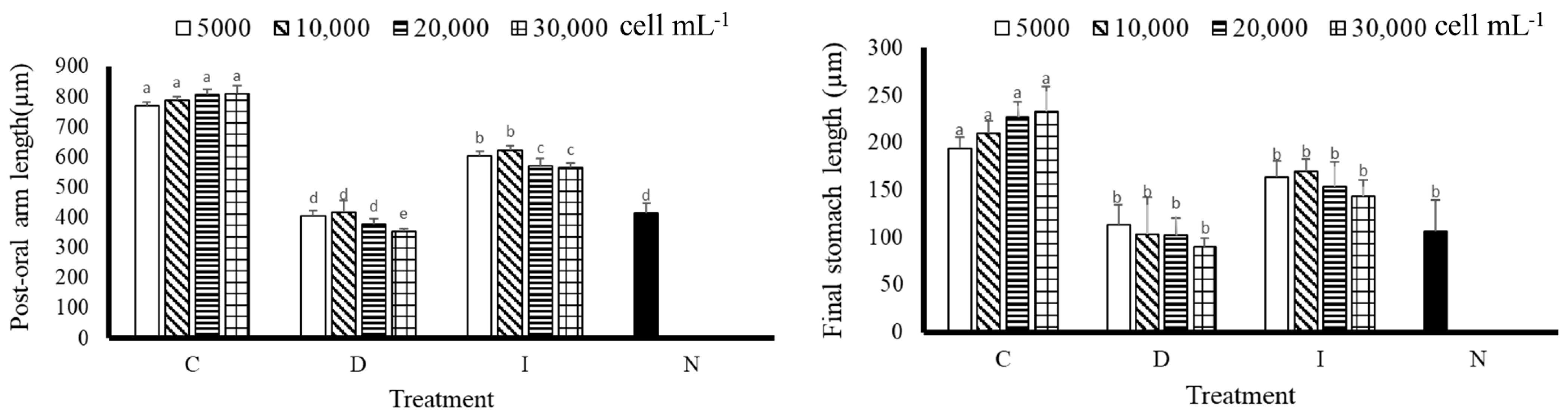
3.2. Postoral Arm Length and Stomach Length
3.3. Rudiment and Metamorphosis
3.4. Survival
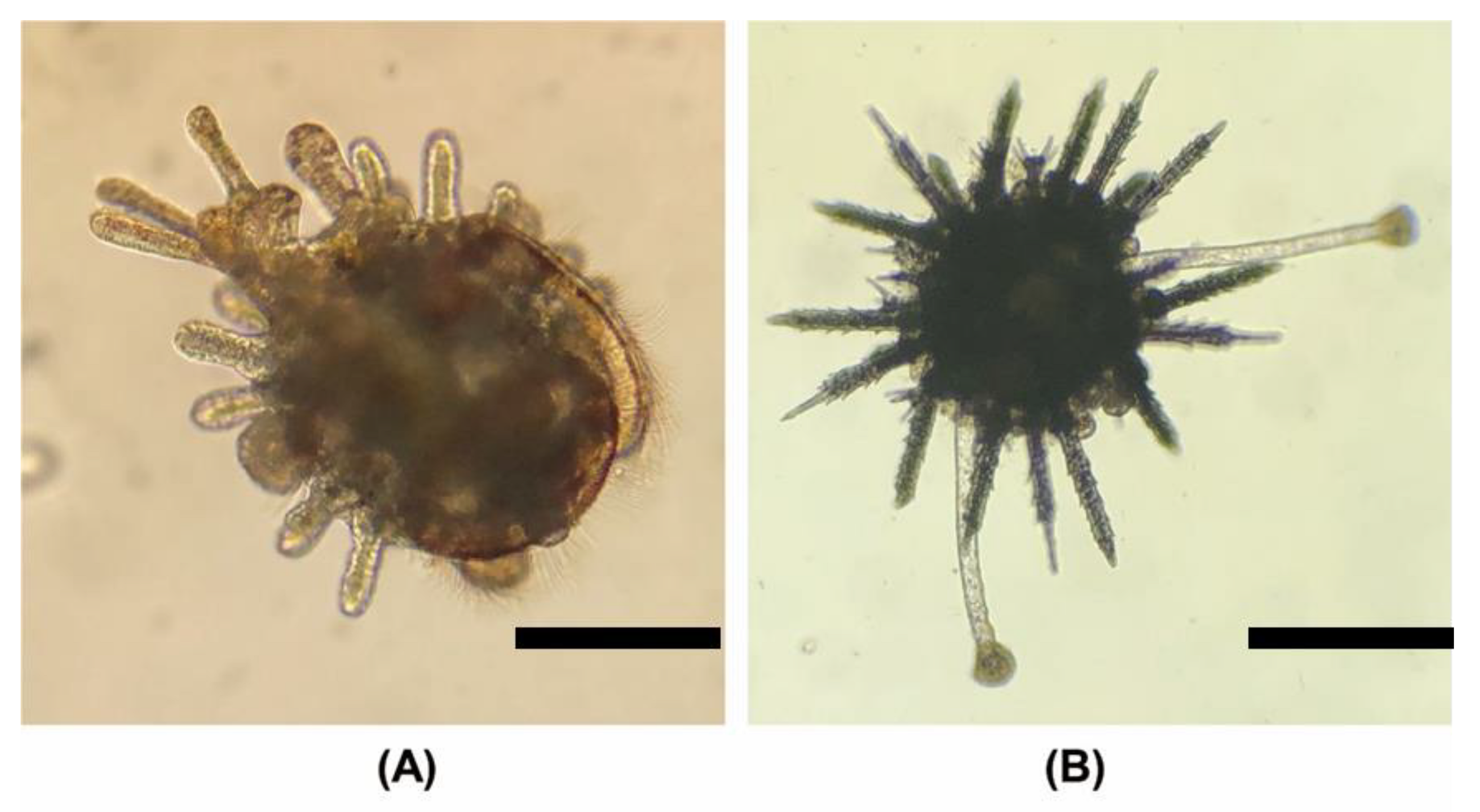
3.5. Developmental Morphology
3.5.1. Cleavages and Blastula
3.5.2. Larval and Early Juvenile
4. Discussion
4.1. Growth and Survival
4.2. Larval Development
4.3. Morphology
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Grosjean, P. Growth Model of the Reared Sea Urchin Paracentrotus lividus (Lamarck, 1816). Ph.D. Thesis, Université de Mons, Mons, Belgium, 2001. [Google Scholar]
- Pais, A.; Chessa, L.A.; Serra, S.; Ruiu, A.; Meloni, G.; Donno, Y. The impact of commercial and recreational harvesting for Paracentrotus lividus on shallow rocky reef sea urchin communities in North-western Sardinia, Italy. Estuar. Coast. Shelf Sci. 2007, 73, 589–597. [Google Scholar] [CrossRef]
- Kirkfeldt, T.S.; Frazão Santos, C. A review of sustainability concepts in marine spatial planning and the potential to supporting the UN sustainable development goal 14. Front. Mar. Sci. 2021, 8, 713980. [Google Scholar] [CrossRef]
- Yatsuya, K.; Nakahara, H. Density, growth and reproduction of the sea urchin Anthocidaris crassispina (A. Agassiz) in two different adjacent habitats, the Sargassum area and Corallina area. Fish. Sci. 2004, 70, 233–240. [Google Scholar] [CrossRef]
- Agnetta, D.; Badalamenti, F.; Ceccherelli, G.; Di Trapani, F.; Bonaviri, C.; Gianguzza, P. Role of two co-occurring Mediterranean sea urchins in the formation of barren from Cystoseira canopy. Estuar. Coast. Shelf Sci. 2015, 152, 73–77. [Google Scholar] [CrossRef]
- Kurihara, H.; Shirayama, Y. Effects of increased atmospheric CO2 on sea urchin early development. Mar. Ecol. Prog. Ser. 2004, 274, 161–169. [Google Scholar] [CrossRef]
- Lu, X.; Wu, R. Ultraviolet damages sperm mitochondrial function and membrane integrity in the sea urchin Anthocidaris crassispina. Ecotoxicol. Environ. Saf. 2005, 61, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.; Uehara, T. Induction of metamorphosis and substratum preference in four sympatric and closely related species of sea urchins (Genus Echinometra) in Okinawa. Zool. Stud. 2001, 40, 29–43. [Google Scholar]
- Brundu, G.; Vallainc, D.; Baroli, M.; Figus, A.M.; Pinna, A.; Carboni, S. Effects of on-demand feeding on sea urchin larvae (Paracentrotus lividus; Lamarck, 1816), development, survival and microalgae utilization. Aquac. Res. 2017, 48, 1550–1560. [Google Scholar] [CrossRef]
- Kalam Azad, A.; McKinley, S.; Pearce, C.M. Factors influencing the growth and survival of larval and juvenile echinoids. Rev. Aquac. 2010, 2, 121–137. [Google Scholar] [CrossRef]
- Castilla-Gavilán, M.; Buzin, F.; Cognie, B.; Dumay, J.; Turpin, V.; Decottignies, P. Optimising microalgae diets in sea urchin Paracentrotus lividus larviculture to promote aquaculture diversification. Aquaculture 2018, 490, 251–259. [Google Scholar] [CrossRef]
- Kelly, M.S.; Hunter, A.J.; Scholfield, C.L.; McKenzie, J.D. Morphology and survivorship of larval Psammechinus miliaris (Gmelin) (Echinodermata: Echinoidea) in response to varying food quantity and quality. Aquaculture 2000, 183, 223–240. [Google Scholar] [CrossRef]
- Ding, D.-S.; Sun, W.-T.; Pan, C.-H. Feeding of a Scleractinian Coral, Goniopora columna, on Microalgae, Yeast, and Artificial Feed in Captivity. Animals 2021, 11, 3009. [Google Scholar] [CrossRef] [PubMed]
- Miller, B.A.; Emlet, R.B. Development of newly metamorphosed juvenile sea urchins (Strongylocentrotus franciscanus and S. purpuratus): Morphology, the effects of temperature and larval food ration, and a method for determining age. J. Exp. Mar. Biol. Ecol. 1999, 235, 67–90. [Google Scholar] [CrossRef]
- Dworjanyn, S.A.; Pirozzi, I. Induction of settlement in the sea urchin Tripneustes gratilla by macroalgae, biofilms and conspecifics: A role for bacteria? Aquaculture 2008, 274, 268–274. [Google Scholar] [CrossRef]
- Meyer, E.; Green, A.; Moore, M.; Manahan, D. Food availability and physiological state of sea urchin larvae (Strongylocentrotus purpuratus). Mar. Biol. 2007, 152, 179–191. [Google Scholar] [CrossRef]
- Burdett-Coutts, V.; Metaxas, A. The effect of the quality of food patches on larval vertical distribution of the sea urchins Lytechinus variegatus (Lamarck) and Strongylocentrotus droebachiensis (Mueller). J. Exp. Mar. Biol. Ecol. 2004, 308, 221–236. [Google Scholar] [CrossRef]
- Rahman, M.A.; Yusoff, F.M.; Arshad, A.; Shamsudin, M.N.; Amin, S. Embryonic, larval, and early juvenile development of the tropical sea urchin, Salmacis sphaeroides (Echinodermata: Echinoidea). Sci. World J. 2012, 2012, 938482. [Google Scholar] [CrossRef]





| Water Quality Conditions | |
|---|---|
| Temperature | 26.00 ± 0.5 °C |
| pH | 8.0 ± 0.5 |
| Dissolved oxygen | 5.00 ± 0.05 ppm |
| Nitrous acid | 0.01 ± 0.05 ppm |
| Nitric acid | 0.05 ± 0.05 ppm |
| Calcium | 425 ± 30.12 ppm |
| Magnesium | 1345 ± 40.25 ppm |
| Ammonia nitrogen | 0.01 ± 0.05 ppm |
| Phosphate | 0.01 ± 0.01 ppm |
| Treatment | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 5000 Cell mL−1 | 10,000 Cell mL−1 | 20,000 Cell mL−1 | 30,000 Cell mL−1 | N | |||||||||
| C | D | I | C | D | I | C | D | I | C | D | I | ||
| Final rudiment length (µm) | 153.33 b (12.47) | 0.00 (0.00) | 0.00 (0.00) | 173.33 b (9.43) | 0.00 (0.00) | 0.00 (0.00) | 193.33 a (4.71) | 0.00 (0.00) | 0.00 (0.00) | 203.33 a (12.47) | 0.00 (0.00) | 0.00 (0.00) | 0.00 (0.00) |
| Final survival Rate (%) | 21.67 b (2.36) | 3.23 c (0.17) | 1.67 d (0.18) | 25.00 ab (4.08) | 2.63 c (0.19) | 2.00 d (0.16) | 40.00 a (4.08) | 0.00 (0.00) | 0.00 (0.00) | 31.67 a (3.12) | 0.00 (0.00) | 0.00 (0.00) | 0.00 (0.00) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chu, Y.; Ding, D.-S.; Sun, W.-T.; Satuito, C.G.; Pan, C.-H. Effects of Marine Microalgae on the Developmental Growth of the Sea Urchin Larviculture Anthocidaris crassipina. Fishes 2023, 8, 278. https://doi.org/10.3390/fishes8060278
Chu Y, Ding D-S, Sun W-T, Satuito CG, Pan C-H. Effects of Marine Microalgae on the Developmental Growth of the Sea Urchin Larviculture Anthocidaris crassipina. Fishes. 2023; 8(6):278. https://doi.org/10.3390/fishes8060278
Chicago/Turabian StyleChu, Yiru, De-Sing Ding, Wei-Ting Sun, Cyril Glenn Satuito, and Chih-Hung Pan. 2023. "Effects of Marine Microalgae on the Developmental Growth of the Sea Urchin Larviculture Anthocidaris crassipina" Fishes 8, no. 6: 278. https://doi.org/10.3390/fishes8060278
APA StyleChu, Y., Ding, D.-S., Sun, W.-T., Satuito, C. G., & Pan, C.-H. (2023). Effects of Marine Microalgae on the Developmental Growth of the Sea Urchin Larviculture Anthocidaris crassipina. Fishes, 8(6), 278. https://doi.org/10.3390/fishes8060278









