Genetic Diversity of Jinshaia sinensis (Cypriniformes, Balitoridae) Distributed Upstream of the Yangtze River
Abstract
1. Introduction
2. Materials and Methods
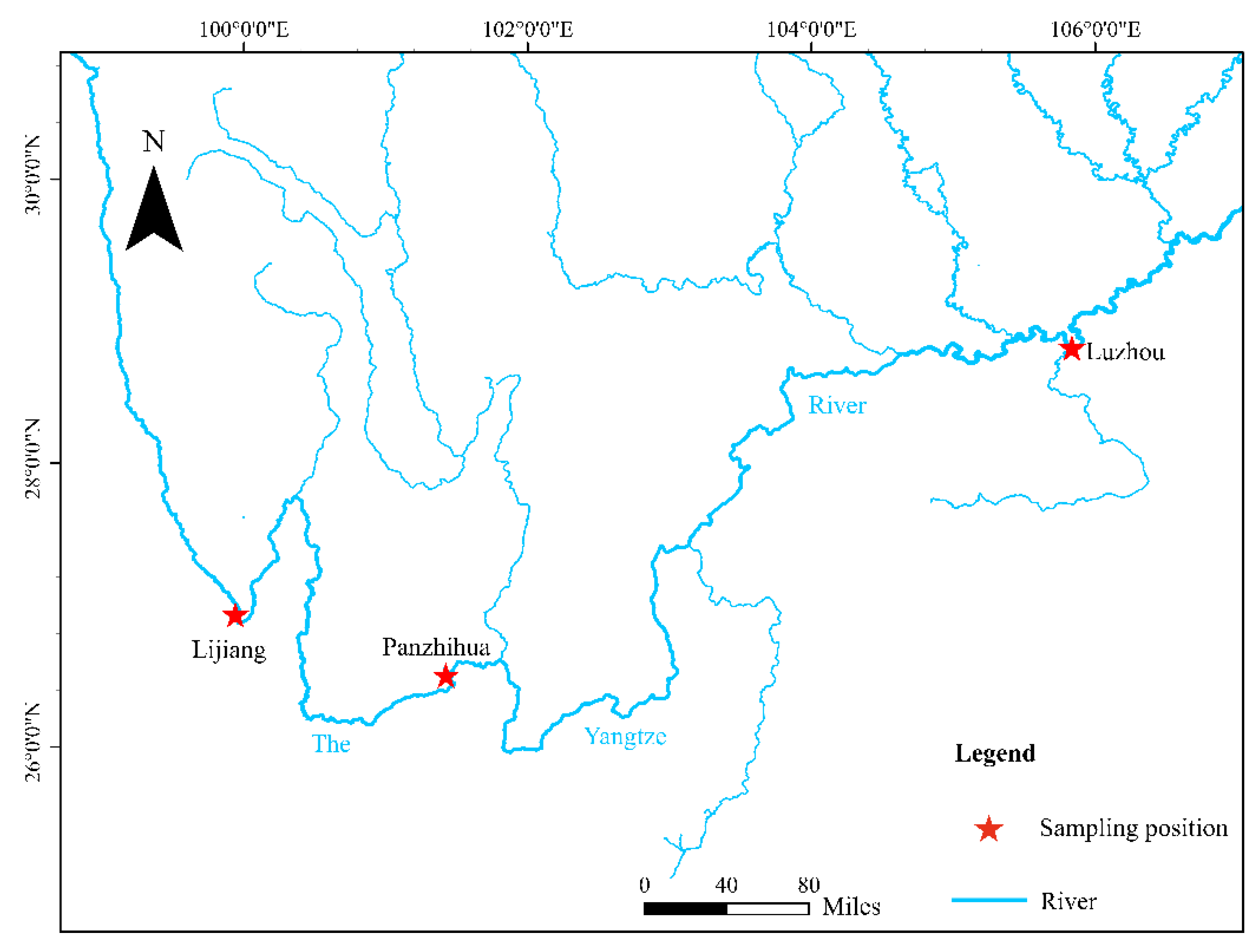
2.1. Sampling and DNA Extraction
2.2. PCR Amplification and Sequencing
2.3. Sequences Information
2.4. Data Analysis
3. Results
3.1. Genetic Diversity
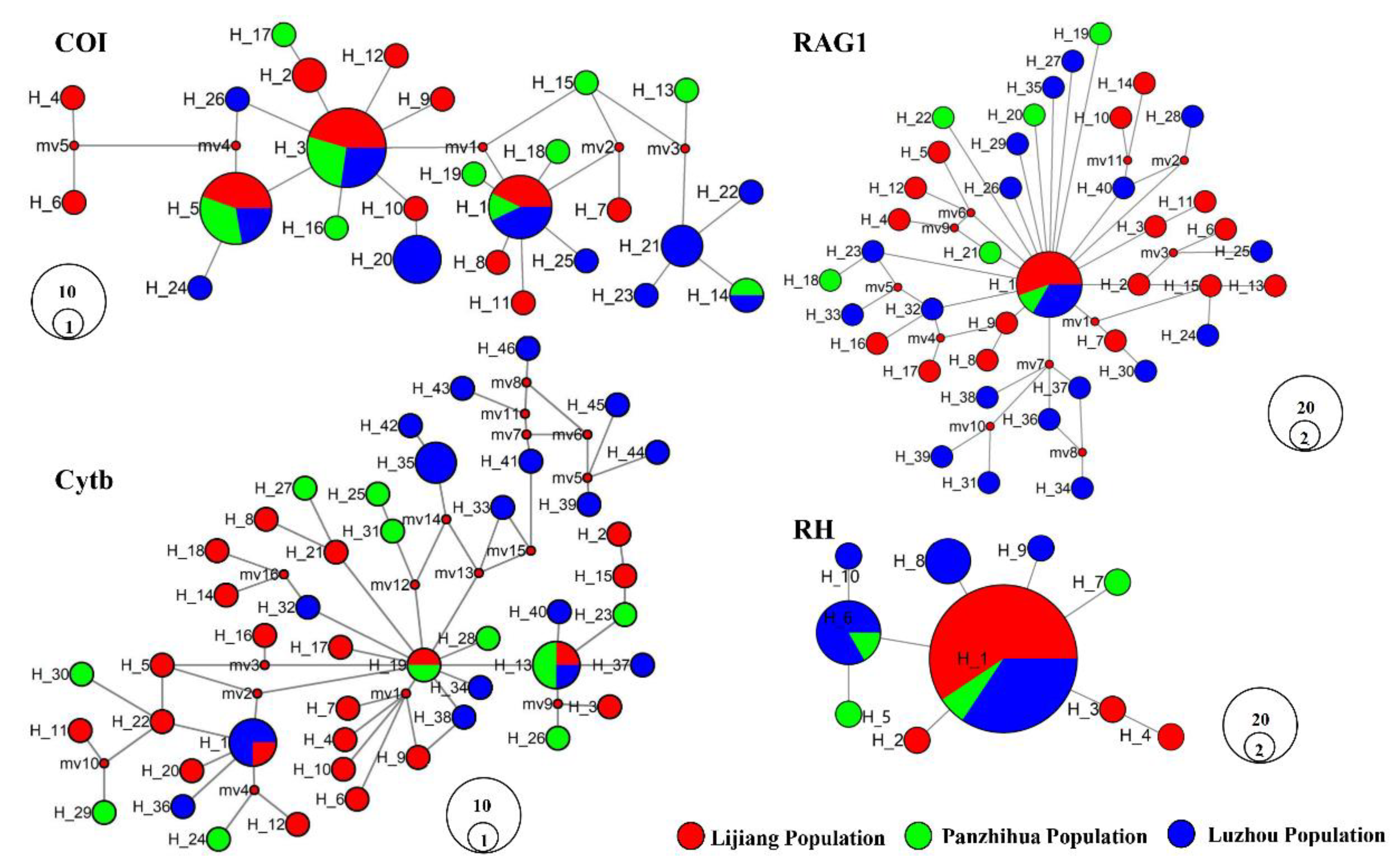
3.2. Haplotype Networks
3.3. Population Structure
3.4. Population History Demography
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Liu, Z.G.; Bao, W.K.; Wu, N.; Liu, Q.; Pan, K.W.; Chen, Q.H.; Yin, K.P. Ecological reconstruction in the upper reaches of the Yangtze River: Environmental problems, degraded mechanism and rehabiltation methodology. World Sci-Tech R D 2000, 22, 32–34. [Google Scholar] [CrossRef]
- Liu, J.K.; Cao, W.X. Fish resources in the Yangtze basin and the strategy for their conservation. Resour. Environ. Yangtze Basin 1992, 1, 17–23. [Google Scholar]
- Gao, T.H.; Tian, H.W.; Ye, C.; Duan, X.B. Diversity and composition of fish in the mainstream of national nature reserve of rare and endemic fish in the upper Yangtze River. Freshw. Fish. 2013, 43, 36–42. [Google Scholar] [CrossRef]
- Wu, J.; Li, L.; Du, H.; Zhang, H.; Wang, C.; Wei, Q. Length-weight relations of 14 endemic fish species from the upper Yangtze River Basin, China. Acta Ichthyol. Piscat. 2013, 43, 163. [Google Scholar] [CrossRef]
- Liu, M.; Wang, D.; Gao, L.; Tian, H.; Liu, S.; Chen, D.; Duan, X. Species diversity of drifting fish eggs in the Yangtze River using molecular identification. PeerJ 2018, 6, e5807. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Yang, J.; Chen, X. Discovery and its significance of spawning grounds of Jinshaia sinensis from upper and middle Jinshajiang River. Zool. Res. 2013, 34, 626–630. [Google Scholar]
- Zhou, C.S.; Liang, Y.S.; Huang, H.N. Ecological features of the spawing of certain fishes in the Hanjiang River after the construction of dams. Acta Hydrobiol. Sin. 1980, 2, 175–188. [Google Scholar]
- Duan, X.B.; Chen, D.Q.; Li, Z.H.; Wang, K.; Huang, M.J.; Liu, S.P. Current status of spawning grounds of fishes with pelagic eggs in the middle reaches of the Yangtze River after impoundment of the Three Gorges Reservior. J. Fish. Sci. China 2008, 4, 523–532. [Google Scholar] [CrossRef]
- Li, C. A preliminary Analysis of the Impacts of the Cascade Hydropower Development on the Fish Biodiversity in the Upper Reach of the Yangtze River; Huazhong University of Science & Technology: Wuhan, China, 2012. [Google Scholar]
- Chen, D.; Xiong, F.; Wang, K.; Chang, Y. Status of research on Yangtze fish biology and fisheries. Environ. Biol. Fishes 2009, 85, 337–357. [Google Scholar] [CrossRef]
- Shao, K.; Shi, F.; Tang, H.; Xiong, M.; Li, W.; Zhu, B. Isolation and characterization of microsatellite loci in Jinshaia sinensis. Conserv. Genet. Resour. 2012, 4, 1075–1077. [Google Scholar] [CrossRef]
- Nazir, A.; Khan, M.A.; Ghosh, P. Assessment of variations in metal concentrations of the Ganges River water by using multivariate statistical techniques. Limnologica 2022, 95, 125989. [Google Scholar] [CrossRef]
- Huang, S.L.; Wang, S.W. Conservation status and prospects of endangered aquatic wildlife in Yangtze River basin. J. Shanghai Ocean. Univ. 2020, 29, 128–138. [Google Scholar] [CrossRef]
- Shen, Y.; Guan, L.; Wang, D.; Gan, X. DNA barcoding and evaluation of genetic diversity in Cyprinidae fish in the midstream of the Yangtze River. Ecol. Evol. 2016, 6, 2702–2713. [Google Scholar] [CrossRef] [PubMed]
- Ding, X.M.; Yan, Y.H.; Li, Q.; Luo, X.; Tang, L.Z. Genetic diversity of goby Rhinogobius giurinus in the fragmented habitat in Nanpan River. Fish. Sci. 2020, 39, 852–862. [Google Scholar] [CrossRef]
- Ferguson, A.; Taggart, J.; Prodöhl, P.; McMeel, O.; Thompson, C.; Stone, C.; Mcginnity, P.; Hynes, R. The application of molecular markers to the study and conservation of fish populations, with special reference to Salmo. J. Fish Biol. 1995, 47, 103–126. [Google Scholar] [CrossRef]
- Nazir, A.; Khan, M.A. Stock discrimination of Sperata aor from river Ganga using microsatellite markers: Implications for conservation and management. Aquat. Living Resour. 2017, 30, 33. [Google Scholar] [CrossRef]
- Chen, W.; Shen, Y.; Gan, X.; Wang, X.; He, S. Genetic diversity and evolutionary history of the Schizothorax species complex in the Lancang River (upper Mekong). Ecol. Evol. 2016, 6, 6023–6036. [Google Scholar] [CrossRef]
- Shen, Y.; Kou, Q.; Chen, W.; He, S.; Yang, M.; Li, X.; Gan, X. Comparative population structure of two dominant species, Shinkaia crosnieri (Munidopsidae: Shinkaia) and Bathymodiolus platifrons (Mytilidae: Bathymodiolus), inhabiting both deep-sea vent and cold seep inferred from mitochondrial multi-genes. Ecol. Evol. 2016, 6, 3571–3582. [Google Scholar] [CrossRef]
- Chen, W.; Li, Y.; Cai, X.; Xiang, D.; Gao, S.; Li, C.; Lan, C.; Zhu, S.; Yang, J.; Li, X. Genetic Structure of an East Asian Minnow (Toxabramis houdemeri) in Southern China, with Implications for Conservation. Biology 2022, 11, 1641. [Google Scholar] [CrossRef]
- Lin, P.; Liu, F.; Li, M.; Gao, X.; Liu, H. Spatial pattern of fish assemblages along the river-reservoir gradient caused by the Three Gorge Reservoir (TGR). Acta Hydrobiol. Sin. 2018, 42, 1124–1134. [Google Scholar]
- Zhu, T.; Yang, D. Length–weight relationships of two fish species from the middle reaches of the Jinsha River, China. J. Appl. Ichthyol. 2016, 32, 747–748. [Google Scholar] [CrossRef]
- Duan, Y.J.; Zhang, F.T.; Cao, S.M.; Wang, J.W.; Tan, D.Q. Isolation and characterization of polymorphic microsatellite loci in Jinshaia sinensis. Acta Hydrobiol. Sin. 2012, 36, 148–151. [Google Scholar]
- Long, A.Y.; Tian, H.W.; Wang, D.Q.; Chen, D.Q.; Zhou, H.H.; Duan, X.B. Genetic diversity between Jinshaia sinensis and J. abbreviata in the upper reaches of Yangtze River based on mitochondrial DNA. Freshw. Fish. 2020, 50, 34–41. [Google Scholar] [CrossRef]
- Tang, Q.Y.; Liu, S.Q.; Yu, D.; Liu, H.Z.; Danley, P.D. Mitochondrial capture and incomplete lineage sorting in the diversification of balitorine loaches (Cypriniformes, Balitoridae) revealed by mitochondrial and nuclear genes. Zool. Scr. 2012, 41, 233–247. [Google Scholar] [CrossRef]
- Li, M.Q. Genetic diversity of Lepturichthys fimbriata in the upper reaches of the Yangtze River; Southwest University: Chongqing, China, 2020. [Google Scholar]
- Yang, L.; Mayden, R.L. Phylogenetic relationships, subdivision, and biogeography of the cyprinid tribe Labeonini (sensu) (Teleostei: Cypriniformes), with comments on the implications of lips and associated structures in the labeonin classification. Mol. Phylogenetics Evol. 2010, 54, 254–265. [Google Scholar] [CrossRef]
- Xiao, W.H.; Zhang, Y.P.; Liu, H.Z. Molecular systematics of Xenocyprinae (Teleostei: Cyprinidae): Taxonomy, biogeography, and coevolution of a special group restricted in east Asia. Mol. Phylogenetics Evol. 2001, 18, 163–173. [Google Scholar] [CrossRef]
- Ward, R.D.; Zemlak, T.S.; Innes, B.H.; Last, P.R.; Hebert, P.D.N. DNA barcoding Australia’s fish species. Philos. Trans. R. Soc. B Biol. Sci. 2005, 360, 1847–1857. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef]
- Nei, M. Molecular Evolutionary Genetics; Columbia University Press: New York, NY, USA, 1987. [Google Scholar]
- Tamura, K.; Nei, M.; Kumar, S. Prospects for inferring very large phylogenies by using the neighbor-joining method. Proc. Natl. Acad. Sci. USA 2004, 101, 11030–11035. [Google Scholar] [CrossRef]
- Excoffier, L.; Lischer, H. Arlequin suite ver 3.5: A new series of programs to perform population genetics analyses under Linux and Windows. Mol. Ecol. Resour. 2010, 10, 564–567. [Google Scholar] [CrossRef]
- Wright, S. The interpretation of population structure by F-Statistics with special regard to systems of mating. Evolution 1965, 19, 395–420. [Google Scholar] [CrossRef]
- Wright, S. Variability Within and Among Natural Populations; University of Chicago Press: Chicago, IL, USA, 1978. [Google Scholar]
- Leigh, J.W.; Bryant, D. Popart: Full-feature software for haplotype network construction. Methods Ecol. Evol. 2015, 6, 1110–1116. [Google Scholar] [CrossRef]
- Excoffier, L.; Smouse, P.E.; Quattro, J.J.G. Analysis of molecular variance inferred from metric distances among DNA haplotypes: Application to human mitochondrial DNA restriction data. Genetics 1992, 131, 479–491. [Google Scholar] [CrossRef] [PubMed]
- Tajima, F. Statistical method for testing the neutral mutation hypothesis by DNA polymorphism. Genetics 1989, 123, 585–595. [Google Scholar] [CrossRef]
- Librado, P.; Rozas, J. DnaSP v5: A software for comprehensive analysis of DNA polymorphism data. Bioinformatics 2009, 25, 1451–1452. [Google Scholar] [CrossRef]
- Nei, M. Part A: The unfolding genome. In Evolution of Human Races at the Gene Level; Progress in Clinical and Biological Research: New York, NY, USA, 1982; pp. 167–181. [Google Scholar]
- Fu, Y.X. Statistical tests of neutrality of mutations against population growth, hitchhiking and background selection. Genetics 1997, 147, 915–925. [Google Scholar] [CrossRef]
- Grant, W.; Bowen, B.W. Shallow population histories in deep evolutionary lineages of marine fishes: Insights from sardines and anchovies and lessons for conservation. J. Hered. 1998, 89, 415–426. [Google Scholar] [CrossRef]
- Long, A.Y. Population Genetics of Jinshaia Sinensis and J. abbreviata in the Upper Yangtze River; Shanghai Ocean University: Shanghai, China, 2020. [Google Scholar]
- Rogers, A.R.; Harpending, H. Population growth makes waves in the distribution of pairwise genetic differences. Mol. Biol. Evol. 1992, 9, 552–569. [Google Scholar] [CrossRef] [PubMed]
- Schneider, S.; Excoffier, L. Estimation of past demographic parameters from the distribution of pairwise differences when the mutation rates very among sites: Application to human mitochondrial DNA. Genetics 1999, 152, 1079–1089. [Google Scholar] [CrossRef]
- Avise, J.C.; Arnold, J.; Ball, R.M.; Bermingham, E.; Lamb, T.; Neigel, J.E.; Reeb, C.A.; Saunders, N.C. Intraspecific phylogeography: The mitochondrial DNA bridge between population genetics and systematics. Annu. Rev. Ecol. Syst. 1987, 18, 489–522. [Google Scholar] [CrossRef]
- Chen, D.Q.; Liu, S.P.; Duan, X.B. Genetic diversity of Botia superciliaris in the upper Yangtze River. Freshw. Fish. 2009, 39, 9–14. [Google Scholar] [CrossRef]
- Zhao, G.; Zhou, J.; Du, J.; Liu, G.; Chen, X.; Zhu, J.; Li, J. Genetic diversity of Elongate loach (Leptobotia elongata) inferred from mitochondrial DNA control region. Southwest China J. Agric. Sci. 2010, 23, 8. [Google Scholar]
- Shen, S.Y.; Tian, H.W.; Wang, D.Q.; Chen, D.Q.; Liu, S.P. Genetic diversity of Leptobotia rubrilabris in the upper Yangtze River inferred from mitochondrial control region. Freshw. Fish. 2017, 47, 83–90. [Google Scholar] [CrossRef]
- Lange, K.; Meier, P.; Trautwein, C.; Schmid, M.; Robinson, C.T.; Weber, C.; Brodersen, J. Basin-scale effects of small hydropower on biodiversity dynamics. Front. Ecol. Environ. 2018, 16, 397–404. [Google Scholar] [CrossRef]
- Ellis, L.E.; Jones, N.E. Longitudinal trends in regulated rivers: A review and synthesis within the context of the serial discontinuity concept. Environ. Rev. 2013, 21, 136–148. [Google Scholar] [CrossRef]
- Heggenes, J.; Røed, K. Do dams increase genetic diversity in brown trout (Salmo trutta)? Microgeographic differentiation in a fragmented river. Ecol. Freshw. Fish 2006, 15, 366–375. [Google Scholar] [CrossRef]
- Shao, K.; Yan, S.; Li, W.; Xiong, M.; Tang, H.; Shi, F. Genetic structure and diversity of Rhinogobio ventralis in the upper Yangtze River obtained by analysis of the mitochondrial DNA control region. J. Hydroecology 2018, 39, 7. [Google Scholar]
- Dong, W.; Wang, D.; Tian, H.; Yan, L.; Duan, X. Genetic structure of two sympatric gudgeon fishes (Xenophysogobio boulengeri and X. nudicorpa) in the upper reaches of Yangtze River Basin. PeerJ 2019, 7, e7393. [Google Scholar] [CrossRef]
- Bickham, J.W.; Sandhu, S.; Hebert, P.D.; Chikhi, L.; Athwal, R. Effects of chemical contaminants on genetic diversity in natural populations: Implications for biomonitoring and ecotoxicology. Mutat. Res. 2000, 463, 33–51. [Google Scholar] [CrossRef]
- Dale, J. Intraspecific variation in coloration. Bird Color. 2006, 2, 36–86. [Google Scholar]
- Fan, Q.; He, S. The parttern of upper and middle Yangtze drainages shaped the genetic structure and diversity of Hemiculter leucisculus revealed by mitochondrial DNA locus. Acta Hydrobiol. Sin. 2014, 38, 627–635. [Google Scholar]
- Pu, Y.; Tian, H.; Chen, D.; Duan, X.; Liu, S.; Wang, D. Genetic diversity of mitochondrial Cytb sequence in Rhinogobio cylindricus from the middle and upper Yangtze River. Freshw. Fish. 2019, 49, 14–19. [Google Scholar]
- Yuan, J.; Zhang, Q.; Li, F.; Zhu, C.; Luo, F. MtDNA control region sequence variation and genetic diversity of Coreius heterodon (Bleeker) in the upper and middle sections of the Yangtze River. Acta Hydrobiol. Sin. 2010, 34, 9–19. [Google Scholar] [CrossRef]

| Population | Gene | N | H | h | Pi | Tajima’s D |
|---|---|---|---|---|---|---|
| Lijiang | COI | 22 | 12 | 0.913 ± 0.038 | 0.0183 ± 0.0083 | −1.523 |
| Cytb | 22 | 22 | 1.000 ± 0.014 | 0.0203 ± 0.0084 | −1.455 | |
| RAG1 | 21 | 17 | 0.929 ± 0.027 | 0.0060 ± 0.0014 | −1.310 | |
| RH | 22 | 4 | 0.254 ± 0.085 | 0.0022 ± 0.0008 | −1.069 | |
| Panzhihua | COI | 14 | 10 | 0.934 ± 0.051 | 0.0079 ± 0.0020 | −1.025 |
| Cytb | 12 | 11 | 0.985 ± 0.040 | 0.0063 ± 0.0005 | −1.387 | |
| RAG1 | 6 | 6 | 0.909 ± 0.046 | 0.0019 ± 0.0003 | −0.339 | |
| RH | 5 | 4 | 0.800 ± 0.089 | 0.0046 ± 0.0015 | 0.249 | |
| Luzhou | COI | 21 | 11 | 0.924 ± 0.032 | 0.0126 ± 0.0012 | 0.526 |
| Cytb | 21 | 17 | 0.971 ± 0.026 | 0.0147 ± 0.0016 | 0.067 | |
| RAG1 | 21 | 19 | 0.962 ± 0.013 | 0.0064 ± 0.0007 | −0.377 | |
| RH | 21 | 5 | 0.660 ± 0.058 | 0.0011 ± 0.0002 | −0.190 |
| Genes | Populations | Lijiang | Panzhihua | Luzhou |
|---|---|---|---|---|
| COI | Lijiang | 0.0196 ± 0.0025 | 0.0140 ± 0.0019 | 0.0181 ± 0.0025 |
| Panzhihua | 0.0069 | 0.0080 ± 0.0018 | 0.0110 ± 0.0022 | |
| Luzhou | 0.1043 ** | 0.0509 | 0.0128 ± 0.0027 | |
| Cytb | Lijiang | 0.0217 ± 0.0021 | 0.0144 ± 0.0014 | 0.0203 ± 0.0019 |
| Panzhihua | 0.0051 | 0.0063 ± 0.0014 | 0.0125 ± 0.0017 | |
| Luzhou | 0.0963 ** | 0.1201 ** | 0.0150 ± 0.0021 | |
| RAG1 | Lijiang | 0.0062 ± 0.0009 | 0.0043 ± 0.0006 | 0.0069 ± 0.0009 |
| Panzhihua | 0.0346 | 0.0021 ± 0.0007 | 0.0049 ± 0.0009 | |
| Luzhou | 0.0962 ** | 0.1018 * | 0.0066 ± 0.0013 | |
| RH | Lijiang | 0.0023 ± 0.0007 | 0.0039 ± 0.0009 | 0.0018 ± 0.0005 |
| Panzhihua | 0.1821 * | 0.0052 ± 0.0015 | 0.0031 ± 0.0009 | |
| Luzhou | 0.1063 ** | 0.1857 ** | 0.0011 ± 0.0006 |
| Source of Variation | COI | Cytb | RAG1 | RH | ||||
|---|---|---|---|---|---|---|---|---|
| Percentage | FST | Percentage | FST | Percentage | FST | Percentage | FST | |
| Among populations | 6.64 | 0.066 * | 7.98 | 0.080 ** | 9.04 | 0.090 ** | 12.99 | 0.130 ** |
| Within populations | 93.36 | 92.02 | 90.96 | 87.01 | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luo, Y.; Zhang, Y.; Cheng, R.; Li, Q.; Zhang, Y.; Li, Y.; Shen, Y. Genetic Diversity of Jinshaia sinensis (Cypriniformes, Balitoridae) Distributed Upstream of the Yangtze River. Fishes 2023, 8, 75. https://doi.org/10.3390/fishes8020075
Luo Y, Zhang Y, Cheng R, Li Q, Zhang Y, Li Y, Shen Y. Genetic Diversity of Jinshaia sinensis (Cypriniformes, Balitoridae) Distributed Upstream of the Yangtze River. Fishes. 2023; 8(2):75. https://doi.org/10.3390/fishes8020075
Chicago/Turabian StyleLuo, Yang, Yufeng Zhang, Ruli Cheng, Qinghua Li, Yu Zhang, Yingwen Li, and Yanjun Shen. 2023. "Genetic Diversity of Jinshaia sinensis (Cypriniformes, Balitoridae) Distributed Upstream of the Yangtze River" Fishes 8, no. 2: 75. https://doi.org/10.3390/fishes8020075
APA StyleLuo, Y., Zhang, Y., Cheng, R., Li, Q., Zhang, Y., Li, Y., & Shen, Y. (2023). Genetic Diversity of Jinshaia sinensis (Cypriniformes, Balitoridae) Distributed Upstream of the Yangtze River. Fishes, 8(2), 75. https://doi.org/10.3390/fishes8020075






