Effect of Microalgal Diets on Sunray Venus Clam (Macrocallista nimbosa) Production and Fatty Acid Profile
Abstract
1. Introduction
2. Methods
2.1. Spawning and Larval Production
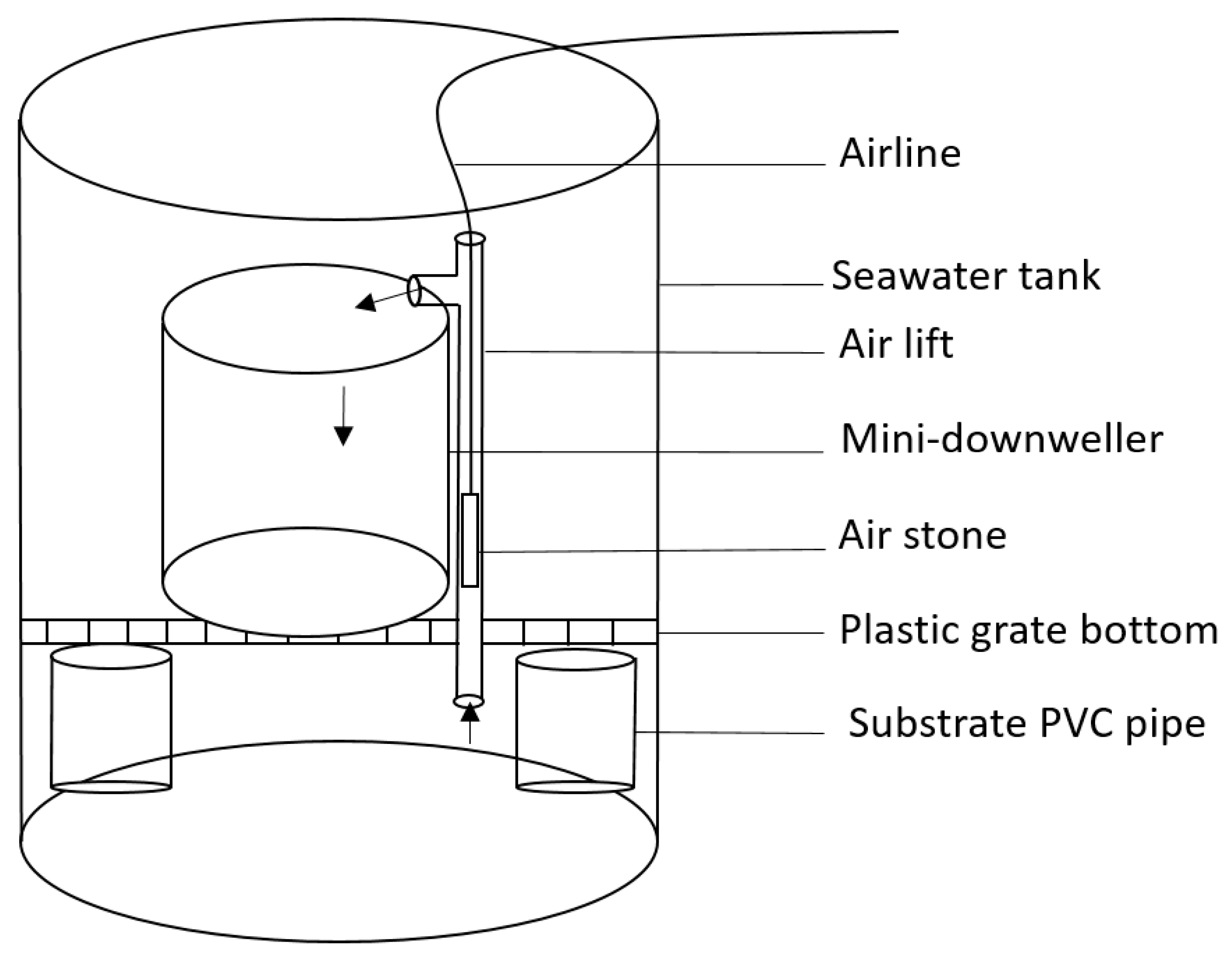
2.2. Experimental System
2.3. Algae Culture and Standardization
2.4. Experimental Design
2.5. Clam Growth and Survival
2.6. Clam and Microalgae Fatty Acid Profile
2.7. Statistical Analysis
3. Results
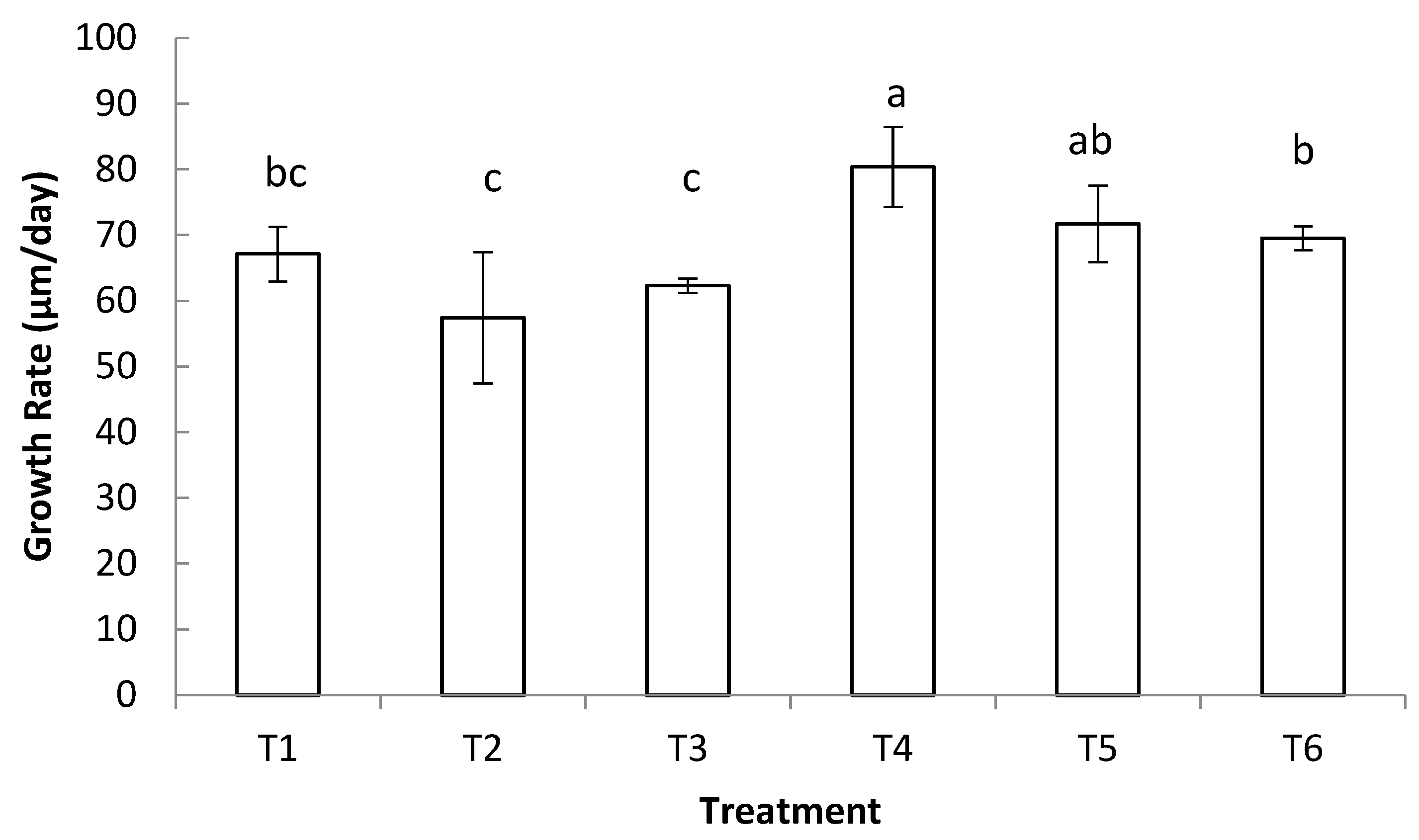
3.1. Growth
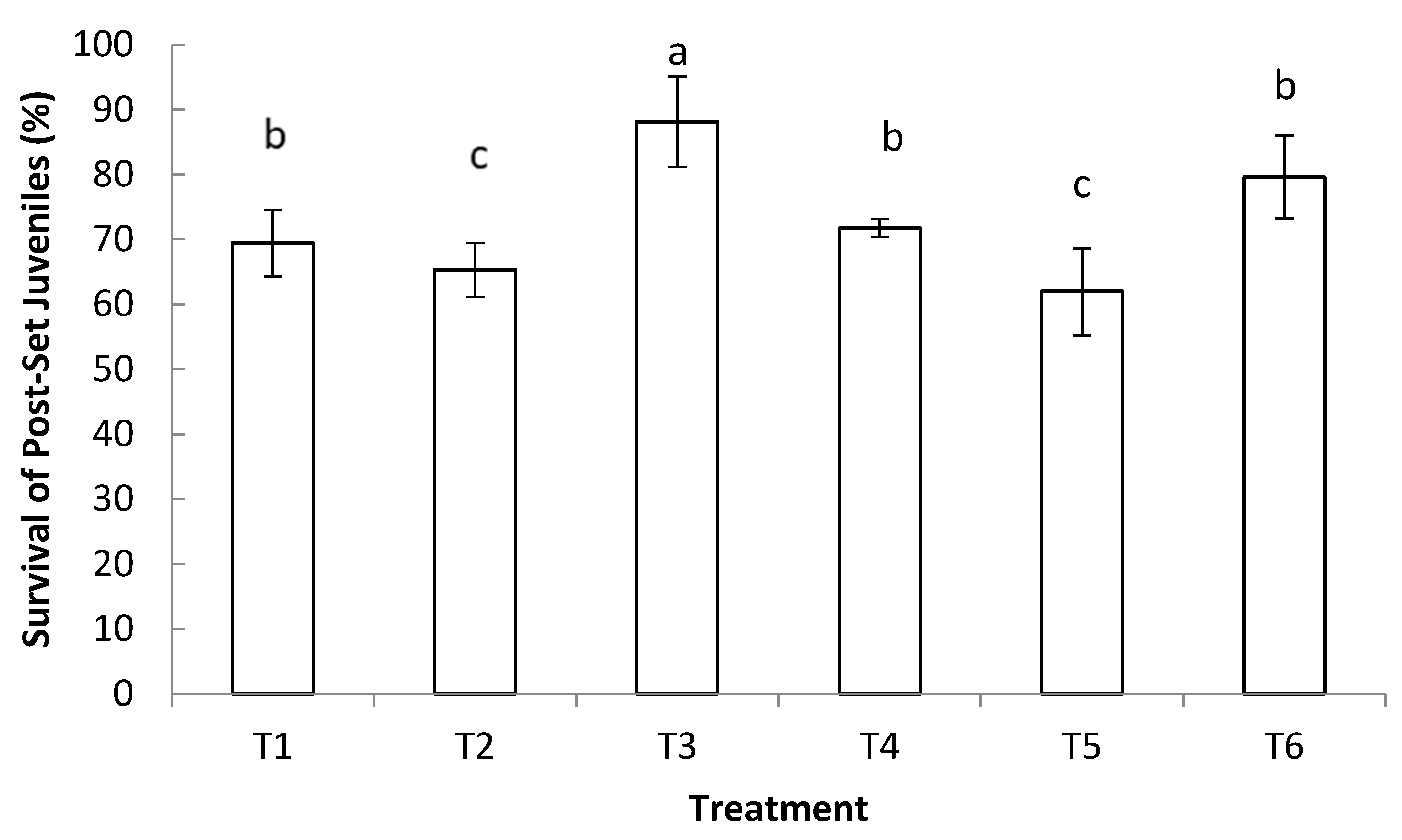
3.2. Survival
3.3. Clam Fatty Acid Profile
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- USDA. Florida Aquaculture Industry Overview. United States Department of Agriculture. 2020. Available online: https://www.fdacs.gov/content/download/91723/file/FDACS-P-02145-2020FLAquacultureIndustryOverview.pdf (accessed on 30 October 2021).
- Gustafson, R.G.; Creswell, R.L.; Jacobsen, T.R.; Vaughan, D.E. Larval biology and mariculture of the angelwing clam, Cyrtopleura costata. Aquaculture 1991, 95, 257–279. [Google Scholar] [CrossRef]
- Blake, N.J.; Adams, C.; Degner, R.; Sweat, D. Aquaculture and Marketing of the Florida Bay Scallop in Crystal River, Florida; Technical Paper; Gainesville, F.L., Ed.; Florida Sea Grant College Program: Gainesville, FL, USA, 2000; Volume 106. [Google Scholar]
- Power, A.J.; Sturmer, L.; Lucas, C.; Walker, R.L.; Manley, J. Gametogenic cycle of the ponderous ark, Noetia ponderosa (Say, 1822), from Cedar Kay, Florida. J. Shellfish Res. 2005, 24, 69–73. [Google Scholar]
- Sturmer, L.N.; Nunez, J.; Creswell, R.; Baker, S.M. The Potential of Blood ark and Ponderous Ark Aquaculture in Florida, Florida Sea Grant Technical Paper, TP-169; Florida Sea Grant College Program: Gainesville, FL, USA, 2009; 81p.
- Scarpa, J.; Sturmer, L.N.; Nuñez, J.; Creswell, R.L. Evaluation of the sunray venus clam Macrocallista nimbosa for aquaculture in Florida. J. Shellfish Res. 2008, 27, 1051. [Google Scholar]
- Scarpa, J.; Sturmer, L.N.; Adams, C.M.; Creswell, R.L.; Nuñez, J. Sunray Venus Clam: A New Species to Diversify the Florida Aquaculture Hard Clam Industry; Florida Sea Grant Final Technical Report, Project R/LR-A-44; Florida Sea Grant College Program: Gainesville, FL, USA, 2009; p. 23. [Google Scholar]
- Abbott, R.T. American Seashells: The Marine Mollusca of the Atlantic and Pacific Coasts of North America, 2nd ed.; Van Nostrand Reinhold: New York, NY, USA, 1974. [Google Scholar]
- Sturmer, L.N.; Scarpa, J.; Laramore, S.E.; Creswell, L. Evaluation of the sunray venus clam Macrocallista nimbosa under field nursery and growout culture conditions in Florida. J. Shellfish Res. 2009, 28, 734. [Google Scholar]
- Loosanoff, V.L.; Davis, H.C. Rearing of bivalve mollusks. Adv. Mar. Biol. 1963, 1, 1–136. [Google Scholar]
- Epifanio, C. Growth in bivalve molluscs: Nutritional effects of two or more species of algae in diets fed to the American oyster Crassostrea virginica (Gmelin) and the hard clam Mercenaria mercenaria (L.). Aquaculture 1979, 18, 1–12. [Google Scholar] [CrossRef]
- Brown, M.R.; Jeffery, S.W.; Volkman, J.K.; Dunstan, G.A. Nutritional properties of microalgae for mariculture. Aquaculture 1997, 151, 315–331. [Google Scholar] [CrossRef]
- Whetstone, J.M.; Sturmer, L.N.; Oesterling, M.J. Biology and Culture of the Hard Clam (Mercenaria mercenaria); SRAC Publication No. 433; Southern Regional Aquaculture Center: Stoneville, MS, USA, 2005. [Google Scholar]
- Marshall, R.; McKinley, S.; Pearce, C.M. Effects of nutrition on larval growth and survival in bivalves. Rev. Aquac. 2010, 2, 33–55. [Google Scholar] [CrossRef]
- Bendif, E.M.; Probert, I.; Schroeder, D.C.; de Vargas, C. On the description of Tisochrysis lutea gen. nov. sp. nov. and Isochrysis nuda sp. nov. in the Isochrysidales, and the transfer of Dicrateria to the Prymnesiales (Haptophyta). J. App. Phycol. 2013, 25, 1763–1776. [Google Scholar] [CrossRef]
- Brown, M.R.; Blackburn, S.I. Live microalgae as feeds in aquaculture hatcheries. In Advances in Aquaculture Hatchery Technology; Allan, G., Burnell, G., Eds.; Elsevier: Amsterdam, The Netherlands, 2013; pp. 117–156, 157e–158e. [Google Scholar]
- Wikfors, G.H.; Ferris, G.E.; Smith, B.C. The relationship between gross biochemical composition of cultured algal foods and growth of the hard clam, Mercenaria mercenaria (L.). Aquaculture 1992, 108, 135–154. [Google Scholar] [CrossRef]
- Gallagher, S.M.; Mann, R.; Sasaki, G.C. Lipid as an index of growth and viability in three species of bivalve larvae. Aquaculture 1986, 56, 81–103. [Google Scholar] [CrossRef]
- Whyte, J.N.C.; Bourne, N.; Ginther, N.G. Biochemical changes during embryogenesis in the rock scallop Crassadoma gigantea. Mar. Biol. 1990, 106, 239–244. [Google Scholar] [CrossRef]
- Pernet, F. Effect of varying dietary levels of ω6 polyunsaturated fatty acids during the early ontogeny of the sea scallop, Placopecten magellanicus. J. Exp. Mar. Biol. Ecol. 2005, 327, 115–133. [Google Scholar] [CrossRef]
- Fernandez-Reiriz, M.J.; Perez-Camacho, A.; Peteiro, L.G.; Labarta, U. Growth and kinetics of lipids and fatty acids of the clam Venerupis pullastra during larval development and postlarvae. Aquac. Nutr. 2011, 17, 13–23. [Google Scholar] [CrossRef]
- Matias, D.; Joaquim, S.; Ramos, M.; Sobral, P.; Leitao, A. Biochemical compounds’ dynamics during larval development of the carpet-shell clam Ruditapes decussatus (Linnaeus, 1758): Effects of mono-specific diets and starvation. Helgol. Mar. Res. 2011, 65, 369–379. [Google Scholar] [CrossRef]
- Reis Batista, I.; Kamermans, P.; Verdegem, M.C.J.; Smaal, A.C. Growth and fatty acid composition of juvenile Cerastoderma edule (L.) fed live microalgae diets with different fatty acid profiles. Aquac. Nutr. 2014, 20, 132–142. [Google Scholar] [CrossRef]
- Liu, W.; Pearce, C.M.; McKinley, R.S.; Forster, I.P. Nutritional value of selected species of microalgae for larvae and early post-set juveniles of the Pacific geoduck clam, Panopea generosa. Aquaculture 2016, 452, 326–341. [Google Scholar] [CrossRef]
- Soudant, P.; Marty, Y.; Moal, J.; Masski, H.; Samain, J.F. Fatty acid composition of polar lipid classes during larval development of scallop Pecten maximus (L.) Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 1998, 121, 279–288. [Google Scholar] [CrossRef]
- Helm, M.M.; Bourne, N.; Lovatelli, A. Hatchery Culture of Bivalves, a Practical Manual; FAO Fisheries Technical Paper 471; Food and Agriculture Organization of the United Nations: Rome, Italy, 2004; Available online: https://www.fao.org/3/y5720e/y5720e00.htm (accessed on 30 October 2021).
- Pronker, A.E.; Nevejan, N.M.; Peene, F.; Geijsen, P.; Sorgeloos, P. Hatchery broodstock conditioning of the blue mussel Mytilus edulis (Linnaeus 1758). Part I. Impact of different micro-algae mixtures on broodstock performance. Aquac. Int. 2008, 16, 297–307. [Google Scholar] [CrossRef]
- Martínez-Fernández, E.; Southgate, P.C. Use of tropical microalgae as food for larvae of the black-lip pearl oyster Pinctada margaritifera. Aquaculture 2007, 263, 220–226. [Google Scholar] [CrossRef]
- Mourente, G.; Lubián, L.M.; Odriozola, J.M. Total fatty acid composition as a taxonomic index of some marine microalgae used as food in marine aquaculture. Hydrobiologia 1990, 203, 147–154. [Google Scholar] [CrossRef]
- Renaud, S.M.; Parry, D.L.; Thinh, L. Microalgae for use in tropical aquaculture I: Gross chemical and fatty acid composition of twelve species of microalgae from the Northern Territory, Australia. J. App. Phycol. 1994, 6, 337–345. [Google Scholar] [CrossRef]
- Patil, V.; Källqvist, T.; Olsen, E. Fatty acid composition of 12 microalgae for possible use in aquaculture feed. Aquac. Int. 2007, 15, 1–9. [Google Scholar] [CrossRef]
- Myers, J.A.; Boisvert, R.N. The economics of producing algae and bivalve seed in hatcheries. Aquaculture 1990, 86, 163–179. [Google Scholar] [CrossRef]
- Benemann, J. Microalgae aquaculture feeds. J. App. Phycol. 1992, 4, 233–245. [Google Scholar] [CrossRef]
- Coutteau, P.; Sorgeloos, P. The use of algal substitutes and the requirement for live algae in the hatchery and nursery of bivalve molluscs: An international survey. J. Shellfish Res. 1992, 11, 467–476. [Google Scholar]
- Hassan, M.M.; Parks, V.; Laramore, S. Optimizing microalgae diets for hard clam, Mercenaria mercenaria, larvae culture. Aquac. Rep. 2021, 20, 100716. [Google Scholar] [CrossRef]
- Guillard, R.R.L.; Ryther, J.H. Studies of marine planktonic diatoms. I. Cyclotella nana Hustedt and Detonula confervacea. Can. J. Microbiol. 1962, 8, 229–239. [Google Scholar] [CrossRef]
- Mamat, N.Z.; Alfaro, A.C. Evaluation of microalgal and formulated diets for the culture of the New Zealand pipi clam Paphiesaustralis. Int. Aquat. Res. 2014, 6, 57–69. [Google Scholar] [CrossRef]
- Epifanio, C. Comparison of yeast and algal diets for bivalve molluscs. Aquaculture 1979, 16, 187–192. [Google Scholar] [CrossRef]
- Milke, L.M.; Bricelj, V.M.; Parrish, C.C. Growth of postlarval sea scallops, Placopecten magellanicus, on microalgal diets, with emphasis on the nutritional role of lipids and fatty acids. Aquaculture 2004, 234, 293–317. [Google Scholar] [CrossRef]
- Ronquillo, J.D.; Fraser, J.; McConkey, A.J. Effect of mixed microalgal diets on growth and polyunsaturated fatty acid profile of European oyster (Ostrea edulis) juveniles. Aquaculture 2012, 360, 64–68. [Google Scholar] [CrossRef]
- Ren, Y.; Liu, W.; Pearce, C.M.; Forster, I.; McKinley, R.S. Effects of selected mixed-algal diets on growth and survival of early postset juveniles of the Pacific geoduck clam, Panopea generosa (Gould, 1850). Aquac. Nutr. 2015, 21, 152–161. [Google Scholar] [CrossRef]
- Helm, M.M. Mixed algal feeding of Ostrea edulis larvae with Isochrysis galbana and Tetraselmis suecica. J. Mar. Biol. Assoc. UK 1977, 57, 1019–1029. [Google Scholar] [CrossRef]
- Pettersen, A.K.; Turchini, G.M.; Jahangard, S.; Ingram, B.A.; Sherman, C.D.H. Effects of different dietary microalgae on survival, growth, settlement and fatty acid composition of blue mussel (Mytilus galloprovincialis) larvae. Aquaculture 2010, 309, 115–124. [Google Scholar] [CrossRef]
- Wong, W.; Levinton, J. Culture of the blue mussel Mytilus edulis (Linnaeus, 1758) fed both phytoplankton and zooplankton: A microcosm experiment. Aquac. Res. 2004, 35, 965–969. [Google Scholar] [CrossRef]
- Galley, T.H.; Batista, F.M.; Braithwaite, R.; King, J.; Beaumont, A.R. Optimisation of larval culture of the mussel Mytilus edulis (L.). Aquac. Int. 2010, 18, 315–325. [Google Scholar] [CrossRef]
- Tang, B.; Liu, B.; Wang, G.; Zhang, T.; Xiang, J. Effects of various algal diets and starvation on larval growth and survival of Meretrix meretrix. Aquaculture 2006, 254, 526–533. [Google Scholar] [CrossRef]
- Hassan, M.M.; Perri, E.; Parks, V.; Laramore, S. Growth, survival, and fatty acid profile of hard clam, Mercenaria mercenaria, juveniles fed live microalgae diets. Aquac. Fish Fisheries 2022, in press. [Google Scholar] [CrossRef]
- Hemaiswarya, S.; Raja, R.; Ravi Kumar, R.; Ganesan, V.; Anbazhagan, C. Microalgae: A sustainable feed source for aquaculture. World J. Microbiol. Biotechnol. 2011, 27, 1737–1746. [Google Scholar] [CrossRef]
- Saucedo, P.E.; González-Jiménez, A.; Acosta-Salmón, H.; Mazón-Suástegui, J.M.; Ronsón-Paulín, J.A. Nutritional value of microalgae-based diets for lions-paw scallop (Nodipecten subnodosus) juveniles reared at different temperatures. Aquaculture 2013, 392–395, 113–119. [Google Scholar] [CrossRef]
- Rico-Villa, B.; Le Coz, J.R.; Mingant, C.; Robert, R. Influence of phytoplankton diet mixtures on microalgae consumption, larval development and settlement of the Pacific oyster Crassostrea gigas (Thunberg). Aquaculture 2006, 256, 377–388. [Google Scholar] [CrossRef]
- Kapranova, L.L.; Nekhoroshev, M.V.; Malakhova, L.V.; Ryabushko, V.I.; Kapranov, S.V.; Kuznetsova, T.V. Fatty acid composition of gonads and gametes in the Black Sea bivalve mollusk Mytilus galloprovincialis Lam. at different stages of sexual maturation. J. Evol. Biochem. Physiol. 2019, 55, 448–455. [Google Scholar] [CrossRef]
- Marty, Y.; Delaunay, F.; Moal, J.; Samain, J.F. Changes in the fatty acid composition of Pecten maximus (L.) during larval development. J. Exp. Mar. Biol. Ecol. 1992, 163, 221–234. [Google Scholar] [CrossRef]
- Gagné, R.; Tremblay, R.; Pernet, F.; Miner, P.; Samain, J.F.; Olivier, F. Lipid requirements of the scallop Pecten maximus (L.) during larval and post-larval development in relation to addition of Rhodomonas salina in diet. Aquaculture 2010, 309, 212–221. [Google Scholar] [CrossRef]
- Soudant, P.; Chu, F.L.E.; Samain, J.F. Lipid requirements in some economically important marine bivalves. J. Shellfish Res. 2000, 19, 605–612. [Google Scholar]
- Geng, S.; Zhou, C.; Chen, W.; Yu, S.; Huang, W.; Huan, T.; Xu, J.; Yan, S. Fatty acid and sterol composition reveal food selectivity of juvenile ark shell Tegillarca granosa Linnaeus after feeding with mixed microalgae. Aquaculture 2016, 455, 109–117. [Google Scholar] [CrossRef]
- Emata, A.C.; Ogata, H.Y.; Garibay, E.S.; Furuita, H. Advanced broodstock diets for the mangrove red snapper and a potential importance of arachidonic acid in eggs and fry. Fish Physiol. Biochem. 2003, 28, 489–491. [Google Scholar] [CrossRef]
- Carrier, J.K., III; Watanabe, W.O.; Harel, M.; Rezek, T.C.; Seaton, P.J.; Shafer, T.H. Effects of dietary arachidonic acid on larval performance, fatty acid profiles, stress resistance and expression of Na+/K+ ATPase mRNA in black sea bass Centropristis striata. Aquaculture 2011, 319, 111–121. [Google Scholar] [CrossRef]
- Torrecillas, S.; Betancor, M.B.; Caballero, M.J.; Rivero, F.; Robaina, L.; Izquierdo, M.; Montero, D. Supplementation of arachidonic acid rich oil in European sea bass juveniles (Dicentrarchus labrax) diets: Effects on growth performance, tissue fatty acid profile and lipid metabolism. Fish Physiol. Biochem. 2018, 44, 283–300. [Google Scholar] [CrossRef]
- Delaporte, M.; Soudant, P.; Moal, J.; Giudicelli, E.; Lambert, C.; Seguineau, C.; Samain, J.F. Impact of 20,4n-6 supplementation of the fatty acid composition and hemocyte parameters of the Pacific oyster Crassostrea gigas. Lipids 2006, 6, 567–576. [Google Scholar] [CrossRef]
- Hurtado, M.A.; Reza, M.; Ibarra, A.M.; Wille, M.; Sorgeloos, P.; Soudant, P.; Palacios, E. Arachidonic acid (20,4 n-6) effect on reproduction, immunology, and prostaglandin E2 levels in Crassostrea corteziensis (Hertlein, 1951). Aquaculture 2009, 294, 300–305. [Google Scholar] [CrossRef]
- Volkman, J.; Jeffery, S.; Nichols, P.; Rogers, G.; Garland, C. Fatty-Acid and Lipid-Composition of 10 Species of Microalgae Used in Mariculture. J. Exp. Mar. Biol. Ecol. 1989, 128, 219–240. [Google Scholar] [CrossRef]
- Yang, Y.; Du, L.; Hosokawa, M.; Miyashita, K. Total lipids content, lipid class and fatty acid composition of ten species of microalgae. J. Oleo Sci. 2020, 69, 1181–1189. [Google Scholar] [CrossRef] [PubMed]
- DeMoreno, J.E.A.; Moreno, V.J.; Brenner, R.R. Lipid metabolism of the yellow clam, Mesodesma mactroides: 2-polyunsaturated fatty acid metabolism. Lipids 1976, 11, 561–566. [Google Scholar] [CrossRef] [PubMed]
- Schiopu, D.; George, S.B.; Castell, J. Ingestion rates and dietary lipids affect growth and fatty acid composition of Dendraster excentricus larvae. J. Exp. Mar. Biol. Ecol. 2006, 328, 47–75. [Google Scholar] [CrossRef]
- Liu, H.; Kelly, M.S.; Cook, E.J.; Black, K.; Orr, H.; Zhu, J.X.; Dong, S.L. The effect of diet type on growth and fatty-acid composition of sea urchin larvae, I. Paracentrotus lividus (Lamarck, 1816) (Echinodermata). Aquaculture 2007, 264, 247–262. [Google Scholar] [CrossRef]
- Sargent, J.R.; McEvoy, L.A.; Bell, J.G. Requirements, presentation and sources of polyunsaturated fatty acids in marine fish larval feeds. Aquaculture 1997, 155, 117–127. [Google Scholar] [CrossRef]
- Carboni, S.; Vignier, J.; Chiantore, M.; Tocher, D.R.; Migaud, H. Effects of dietary microalgae on growth, survival and fatty acid composition of sea urchin Paracentrotus lividus throughout larval development. Aquaculture 2012, 324, 250–258. [Google Scholar] [CrossRef]
- Laramore, S.; Sturmer, L.; Baptiste, R.; Yang, H.; Sinacore, C.; Urban-Gedmadke, E. Fatty acid composition of adult and larval Sunray Venus clams Macrocallista nimbosa: Environmental and gametogenic impacts. J. Shellfish Res. 2017, 36, 403–416. [Google Scholar] [CrossRef]
- Enright, C.T.; Newkirk, G.F.; Craigie, J.S.; Castell, J.D. Evaluation of phytoplankton as diets for juvenile Ostrea edulis L. J. Exp. Mar. Biol. Ecol. 1986, 96, 1–13. [Google Scholar] [CrossRef]
- Reitan, K.I. Digestion of lipids and carbohydrates from microalgae (Chaetoceros muelleri Lemmermann and Isochrysis aff. galbana clone T-ISO) in juvenile scallops (Pecten maximus L.). Aquac. Res. 2011, 42, 1530–1538. [Google Scholar] [CrossRef]
- Kreeger, D.A.; Langdon, C.J. Effect of dietary protein content on growth of juvenile mussels, Mytilus trossulus (Gould 1850). Biol. Bull. 1993, 185, 123–139. [Google Scholar] [CrossRef] [PubMed]
- Orban, E.; Di Lena, G.; Nevigato, T.; Casini, I.; Caproni, R.; Santaroni, G.; Giulini, G. Nutritional and commercial quality of the striped venus clam, Chamelea gallina, from the Adriatic sea. Food Chem. 2006, 101, 1063–1070. [Google Scholar] [CrossRef]
- Kim-Kabari, D.B.; Hart, A.D.; Nyeche, P.T. Nutritional composition of selected shellfish consumed in Rivers State, Nigeria. Amer. J. Food. Nutri. 2017, 5, 142–146. [Google Scholar]
- Sturmer, L.N.; Morgan, K.L.; Degner, R.L. Nutritional Composition and Marketable Shelf-Life of Blood Ark Clams and Ponderous Ark Clams; EDIS document FE568; The Department of Food and Resource Economics, Florida Cooperative Extension Service, Institute of Food and Agricultural Sciences, University of Florida: Gainesville, FL, USA, 2005; p. 6. [Google Scholar]
| Treatment | Live Microalgae Diet (% Inclusion) |
|---|---|
| T1 (TL + DL) | Tisochrysis lutea (50%) + Diacronema lutheri (50%) |
| T2 (CN + TW) | Chaetoceros neogracile (50%) + Thalassiosira weissflogii (50%) |
| T3 (TL + CN) | T. lutea (50%) + C. neogracile (50%) |
| T4 (TL + DL + CN + TW) | T. lutea (25%) + D. lutheri (25%) + C. neogracile (25%) + T. weissflogii (25%) |
| T5 (TL + DL + CN) | T. lutea (33.3%) + D. lutheri (33.3%) + C. neogracile (33.3%) |
| T6 (TL + CN + TW) | T. lutea (33.3%) + C. neogracile (33.3%) + T. weissflogii (33.3%) |
| Fatty Acid | Tisochrysis lutea | Diacronema lutheri | Chaetoceros neogracile | Thalassiosira weissflogii |
|---|---|---|---|---|
| 12:0 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.14 ± 0.0 | ND |
| 14:0 | 25.4 ± 0.2 | 12.3 ± 0.1 | 20.9 ± 0.7 | 17.4 ± 0.2 |
| 15:0 | 0.7 ± 0.0 | 1.0 ± 0.1 | 1.0 ± 0.1 | 1.4 ± 0.1 |
| 16:0 | 10.6 ± 1.3 | 21.3 ± 1.0 | 18.3 ± 0.4 | 13.4 ± 0.1 |
| 17:0 | 2.3 ± 0.0 | 3.3 ± 0.5 | 3.4 ± 1.6 | 4.5 ± 0.3 |
| 20:0 | 0.2 ± 0.0 | 1.8 ± 0.6 | 0.9 ± 0.0 | 2.8 ± 0.1 |
| Total SFA | 39.2 ± 1.1 | 39.7 ± 0.1 | 44.6 ± 0.4 | 39.6 ± 0.2 |
| 14:1n-5 | 0.4 ± 0.0 | 0.1 ± 0.0 | 0.4 ± 0.0 | 0.3 ± 0.0 |
| 14:1n-7 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 | ND |
| 15:1n-6 | ND | ND | 0.2 ± 0.0 | 0.3 ± 0.0 |
| 16:1n-5 | 0.1 ± 0.0 | 0.2 ± 0.0 | 0.6 ± 0.1 | 0.9 ± 0.0 |
| 16:1n-6 | 1.0 ± 0.0 | 0.7 ± 0.0 | 3.2 ± 0.1 | 7.4 ± 0.1 |
| 16:1n-7 | 6.3 ± 0.1 | 20.9 ± 0.0 | 37.7 ± 0.3 | 21.9 ± 0.9 |
| 17:1n-8 | 0.5 ± 0.0 | ND | 0.1 ± 0.0 | 0.2 ± 0.1 |
| 18:1n-7 | 1.4 ± 0.0 | 8.2 ± 0.5 | 2.4 ± 0.0 | 3.4 ± 0.3 |
| 18:1n-8 | 9.7 ± 0.3 | ND | ND | ND |
| 18:1n-9 | 12.6 ± 1.1 | ND | 0.8 ± 0.0 | 0.5 ± 0.0 |
| Total MUFA | 32.2 ± 1.0 | 30.1 ± 0.4 | 45.6 ± 0.5 | 34.9 ± 1.2 |
| 18:2n-6 | 4.7 ± 0.1 | 0.5 ± 0.0 | 0.9 ± 0.0 | 1.6 ± 0.0 |
| 18:3n-6 | 0.3 ± 0.0 | 0.2 ± 0.0 | 0.9 ± 0.1 | ND |
| 20:3n-6 | ND | 0.04 ± 0.0 | 0.1 ± 0.1 | 0.2 ± 0.0 |
| 20:4n-6 ARA | 0.2 ± 0.1 | 0.2 ± 0.0 | 0.7 ± 0.00 | 0.9 ± 0.0 |
| 22:5n-6 | 0.8 ± 0.0 | 0.3 ± 0.0 | ND | ND |
| Total PUFA (n-6) | 6.1 ± 0.0 | 1.2 ± 0.0 | 2.6 ± 0.2 | 2.8 ± 0.1 |
| 18:4n-3 | 13.4 ± 0.8 | 5.2 ± 0.0 | 0.5 ± 0.0 | 0.5 ± 0.0 |
| 20:5n-3 EPA | 0.7 ± 0.0 | 17.8 ± 0.2 | 5.5 ± 0.2 | 13.9 ± 0.0 |
| 22:6n-3 DHA | 6.8 ± 0.2 | 5.1 ± 0.1 | 0.2 ± 0.0 | 2.5 ± 0.0 |
| Total PUFA (n-3) | 21.0 ± 1.0 | 28.2 ± 0.3 | 6.1 ± 0.2 | 16.9 ± 0.1 |
| Total Combined PUFA (n-3, n-6) | 26.3 ± 1.0 | 29.4 ± 0.4 | 8.7 ± 0.4 | 19.7 ± 0.1 |
| Diet Treatment | Duration of Feeding | ||
|---|---|---|---|
| 2 Weeks (Age: 44 DPF) | 4 Weeks (Age: 58 DPF) | 6 Weeks (Age: 72 DPF) | |
| T1(TL + DL) | 2.57 ± 0.68 a | 3.53 ± 0.75 a | 3.8 ± 0.91 b |
| T2 (CN + TW) | 2.12 ± 0.67 c | 3.22 ± 0.86 ab | 3.38 ± 1.46 bc |
| T3 (TL + CN) | 2.36 ± 0.64 ab | 2.84 ± 0.50 bc | 3.59 ± 0.72 bc |
| T4 (TL + DL + CN + TW) | 2.30 ± 0.68 abc | 3.41 ± 0.70 ab | 4.37 ± 0.97 a |
| T5 (TL + DL + CN) | 2.44 ± 0.74 a | 3.29 ± 0.73 ab | 4.0 ± 0.95 ab |
| T6 (TL + CN + TW) | 2.28 ± 0.85 bc | 2.98 ± 0.87 b | 3.9 ± 0.79 b |
| Treatment Group | ||||||
|---|---|---|---|---|---|---|
| Fatty Acid | T1 | T2 | T3 | T4 | T5 | T6 |
| 14:0 | 5.9 ± 0.2 a | 2.0 ± 0.1 d | 4.1 ± 0.2 b | 3.0 ± 0.1 c | 3.8 ± 0.2 b | 3.3 ± 0.1 c |
| 15:0 | 0.4 ± 0.01 d | 0.6 ± 0.03 ab | 0.5 ± 0.01 cd | 0.5 ± 0.03 bc | 0.4 ± 0.02 d | 0.6 ± 0.01 a |
| 16:0 | 25.2 ± 0.2 ab | 23.6 ± 0.7 c | 24.7 ± 0.5 abc | 25.7 ± 0.2 a | 25.9 ± 0.7 a | 24.2 ± 0.3 bc |
| 17:0 | 6.3 ± 0.3 | 7.0 ± 3.0 | 6.8 ± 0.5 | 8.1 ± 0.4 | 7.4 ± 0.1 | 9.0 ± 0.1 |
| 20:0 | 0.9 ± 0.1 b | 2.3 ± 0.1 a | 1.1 ± 0.03 b | 1.2 ± 0.4 b | 1.4 ± 0.4 b | 1.6 ± 0.1 b |
| 21:0 | 0.7 ± 0.2 a | 0.1 ± 0.1 b | 0.8 ± 0.2 a | 0.7 ± 0.05 a | 0.8 ± 0.1 a | 0.6 ± 0.1 a |
| 22:0 | 0.1 ± 0.1 c | 0.8 ± 0.3 a | 0.3 ± 0.02 ab | 0.4 ± 0.02 ab | 0.3 ± 0.04 ab | 0.4 ± 0.04 b |
| Total SFA | 39.5 ± 0.3 ab | 36.4 ± 2.6 b | 38.2 ± 0.8 ab | 39.6 ± 0.1 a | 40.0 ± 0.5 a | 39.6 ± 0.5 a |
| 16:1n-6 | 0.2 ± 0.04 | 0.2 ± 0.2 | 0.3 ± 0.02 | 0.2 ± 0.05 | 0.2 ± 0.04 | 0.3 ± 0.02 |
| 16:1n-7 | 3.8 ± 0.3 d | 9.3 ± 0.7 a | 4.8 ± 0.2 bc | 4.8 ± 0.1 bc | 4.4 ± 0.4 cd | 5.5 ± 0.1 b |
| 17:1n-7 | 0.3 ± 0.02 a | 0.0 ± 0.0 c | 0.04 ± 0.07 c | 0.0 ± 0.0 c | 0.2 ± 0.02 b | 0.0 ± 0.0 c |
| 18:1n-6 | 0.1 ± 0.1 c | 0.4 ± 0.1 a | 0.3 ± 0.03 abc | 0.3 ± 0.03 abc | 0.2 ± 0.1 bc | 0.3 ± 0.04 ab |
| 18:1n-7 | 5.7 ± 0.4 a | 4.0 ± 0.3 bc | 3.7 ± 0.1 c | 4.4 ± 0.02 b | 4.5 ± 0.3 b | 3.6 ± 0.1 c |
| 18:1n-9 | 9.9 ± 0.5 a | 3.5 ± 0.2 d | 9.1 ± 0.2 b | 7.4 ± 0.2 c | 8.6 ± 0.2 b | 7.1 ± 0.1 c |
| 20:1n-6 | 1.1 ± 0.1 e | 3.0 ± 0.04 a | 1.3 ± 0.1 d | 2.1 ± 0.06 b | 1.6 ± 0.02 c | 1.9 ± 0.1 b |
| 20:1n-9 | 3.4 ± 0.4 a | 0.0 ± 0.0 b | 3.6 ± 0.1 a | 0.0 ± 0.0 b | 0.0 ± 0.0 b | 0.2 ± 0.02 b |
| Total MUFA | 24.5 ± 1.9 a | 20.4 ± 1.5 b | 23.1 ± 0.9 a | 19.2 ± 0.4 ab | 19.5 ± 1.2 bc | 18.9 ± 0.4 c |
| 18:2n-6 | 1.9 ± 0.4 a | 1.1 ± 0.1 bc | 1.5 ± 0.04 ab | 1.0 ± 0.06 c | 1.1 ± 0.1 c | 1.0 ± 0.02 c |
| 18:3n-6 | 0.03 ± 0.1 e | 0.5 ± 0.1 a | 0.2 ± 0.02 cd | 0.2 ± 0.02 bc | 0.1 ± 0.1 de | 0.3 ± 0.02 b |
| 20:2n-6 | 5.9 ± 0.2 d | 7.2 ± 0.2 c | 7.3 ± 0.2 c | 10.0 ± 0.06 a | 10.4 ± 0.6 a | 9.1 ± 0.3 b |
| 20:3n-6 | 0.5 ± 0.0 d | 1.7 ± 0.1 a | 0.6 ± 0.01 d | 0.8 ± 0.02 c | 0.5 ± 0.1 d | 0.9 ± 0.1 b |
| 20:4n-6 ARA | 2.8 ± 0.2 d | 6.0 ± 0.1 a | 4.3 ± 0.2 bc | 4.4 ± 0.02 b | 4.0 ± 0.1 c | 4.4 ± 0.1 bc |
| 22:4n-6 | 0.4 ± 0.1 d | 2.3 ± 0.1 a | 1.0 ± 0.1 b | 1.0 ± 0.06 b | 0.7 ± 0.1 c | 1.0 ± 0.03 b |
| 22:5n-6 | 3.9 ± 0.1 a | 0.5 ± 0.02 e | 3.4 ± 0.2 b | 3.0 ± 0.1 c | 3.7 ± 0.2 ab | 2.2 ± 0.1 d |
| 24:4n-6 | 0.3 ± 0.04 | 0.1 ± 0.1 | 0.2 ± 0.2 | 0.3 ± 0.04 | 0.3 ± 0.03 | 0.1 ± 0.1 |
| Total PUFA (n-6) | 15.7 ± 1.1 c | 19.4 ± 1.0 a | 18.5 ± 0.9 b | 20.7 ± 0.7 a | 20.8 ± 1.3 a | 19.0 ± 0.7 a |
| 15:4n-3 | 0.6 ± 0.02 d | 2.5 ± 0.1 a | 0.8 ± 0.02 c | 1.4 ± 0.1 b | 1.0 ± 0.06 c | 1.5 ± 0.02 b |
| 18:4n-3 | 2.1 ± 0.1 a | 0.0 ± 0.0 d | 1.8 ± 0.1 ab | 1.4 ± 0.3 bc | 1.5 ± 0.2 bc | 1.3 ± 0.03 c |
| 20:5n-3 EPA | 3.6 ± 0.04 c | 10.0 ± 0.2 a | 4.8 ± 0.2 c | 6.0 ± 0.01 b | 4.8 ± 0.2 c | 6.4 ± 0.1 b |
| 22:1n-3 | 0.3 ± 0.02 a | 0.1 ± 0.1 b | 0.3 ± 0.02 a | 0.3 ± 0.02 a | 0.4 ± 0.03 a | 0.3 ± 0.01 a |
| 22:5n-3 | 0.4 ± 0.1 c | 3.4 ± 0.1 a | 0.9 ± 0.2 b | 0.9 ± 0.1 b | 0.7 ± 0.2 b | 0.9 ± 0.1 b |
| 22:6n-3 DHA | 12.0 ± 0.3 a | 6.3 ± 0.1 d | 10.7 ± 0.6 b | 10.3 ± 0.1 bc | 10.8 ± 0.6 b | 9.4 ± 0.04 c |
| Total PUFA (n-3) | 19.1 ± 0.5 b | 22.2 ± 0.6 a | 19.3 ± 1.1 b | 20.3 ± 0.6 b | 19.1 ± 1.1 b | 19.7 ± 0.2 b |
| Total Combined PUFA (n-3, n-6) | 34.8 ± 0.3 c | 41.6 ± 0.8 a | 37.8 ± 1.4 b | 41.0 ± 0.2 a | 39.9 ± 0.4 b | 38.7 ± 0.3 b |
| EPA/ARA | 1.3 ± 0.1 cd | 1.7 ± 0.02 a | 1.1 ± 0.01 e | 1.4 ± 0.01 bc | 1.2 ± 0.04 de | 1.5 ± 0.05 b |
| EPA/DHA | 0.3 ± 0.01 d | 1.6 ± 0.04 a | 0.4 ± 0.01 c | 0.6 ± 0.01 b | 0.4 ± 0.04 c | 0.3 ± 0.02 d |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perri, E.; Sturmer, L.; Wills, P.S.; Baldwin, J.; Laramore, S. Effect of Microalgal Diets on Sunray Venus Clam (Macrocallista nimbosa) Production and Fatty Acid Profile. Fishes 2023, 8, 72. https://doi.org/10.3390/fishes8020072
Perri E, Sturmer L, Wills PS, Baldwin J, Laramore S. Effect of Microalgal Diets on Sunray Venus Clam (Macrocallista nimbosa) Production and Fatty Acid Profile. Fishes. 2023; 8(2):72. https://doi.org/10.3390/fishes8020072
Chicago/Turabian StylePerri, Edward, Leslie Sturmer, Paul S. Wills, John Baldwin, and Susan Laramore. 2023. "Effect of Microalgal Diets on Sunray Venus Clam (Macrocallista nimbosa) Production and Fatty Acid Profile" Fishes 8, no. 2: 72. https://doi.org/10.3390/fishes8020072
APA StylePerri, E., Sturmer, L., Wills, P. S., Baldwin, J., & Laramore, S. (2023). Effect of Microalgal Diets on Sunray Venus Clam (Macrocallista nimbosa) Production and Fatty Acid Profile. Fishes, 8(2), 72. https://doi.org/10.3390/fishes8020072






