3.2.1. Cambeva Beta-Clade
Cambeva cf. Cambeva botuvera Costa, Feltrin & Katz, 2021
Trichomycterus cubataonis (non
Trichomycterus cubataonis Bizerril, 1994): Katz & Barbosa [
55] (pp. 4–6, Figures 2 and 3 in Ref. [
55]) (misidentification).
Remarks. The taxon here identified as
C. cf.
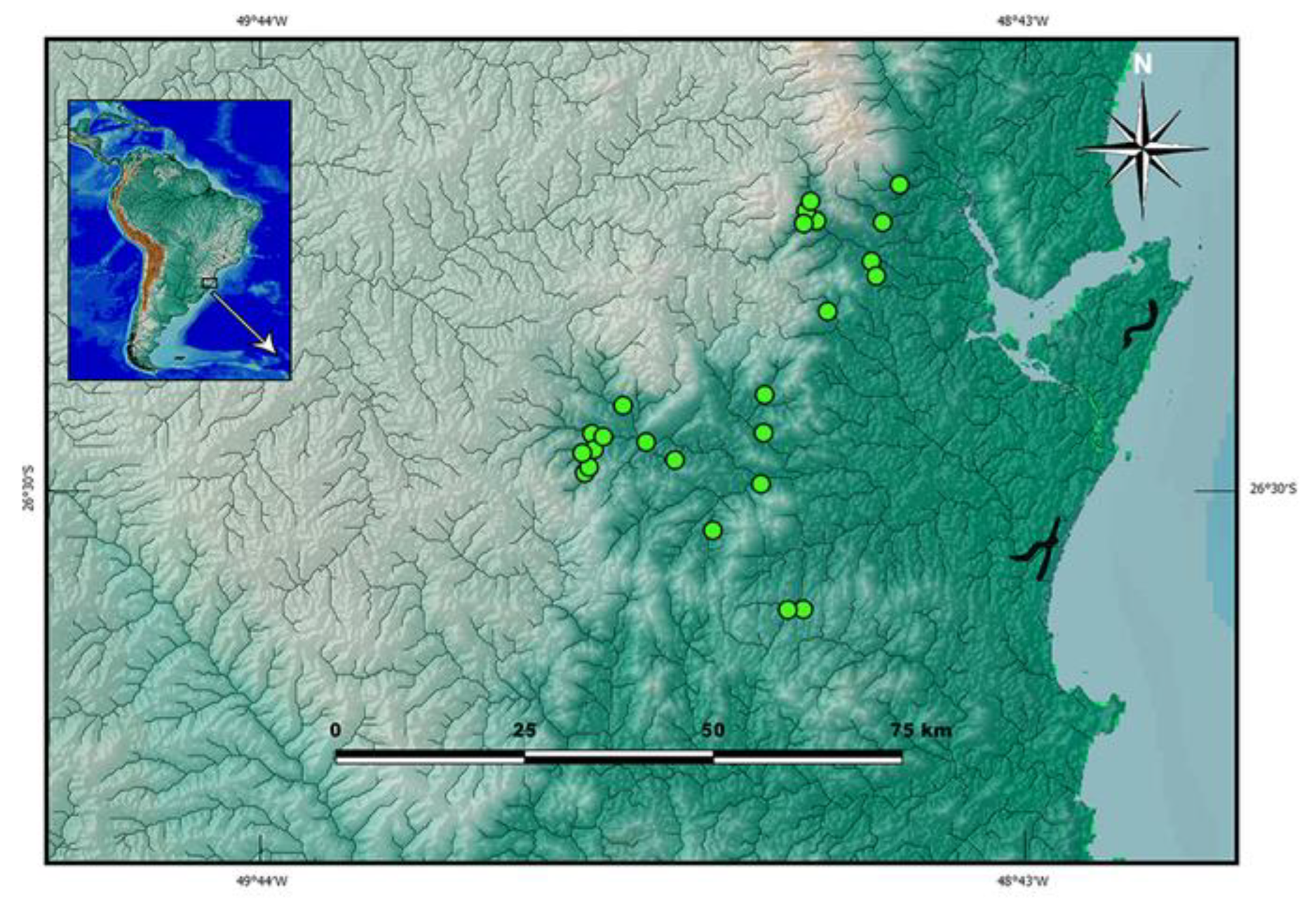
botuvera is broadly distributed in GBIA (
Figure 3). It is still unclear whether it represents a morphologically polymorphic species or a species complex. Some colour morphs are not distinguishable from some colour morphs described for
C. barbosae and
C. botuvera [
6] (p. 5, Figure 3 in Ref. [
6]), whereas different populations from GBIA may exhibit distinct morphological traits. On the other hand, our molecular studies in progress indicate low genetic differentiation among GBIA populations. Like
C. botuvera,
C. cf.
botuvera has eight rays in the pectoral fin, instead of seven as in
C. barbosae. This complex group is presently the focus of a specific study on species delimitation that is in progress by the authors, encompassing a broader geographical area much beyond the limits of GBIA.
Populations of
C. cf.
botuvera from GBIA were formerly misidentified as
C. cubataonis by Katz & Barbosa [
55]. In the Rio Cubatão do Norte basin, both species occur in sympatry and can be misidentified because of the similar colour pattern and fin morphology, although
C. cf.
botuvera usually has paler colours. In this basin,
C. cf.
botuvera is distinguishable from
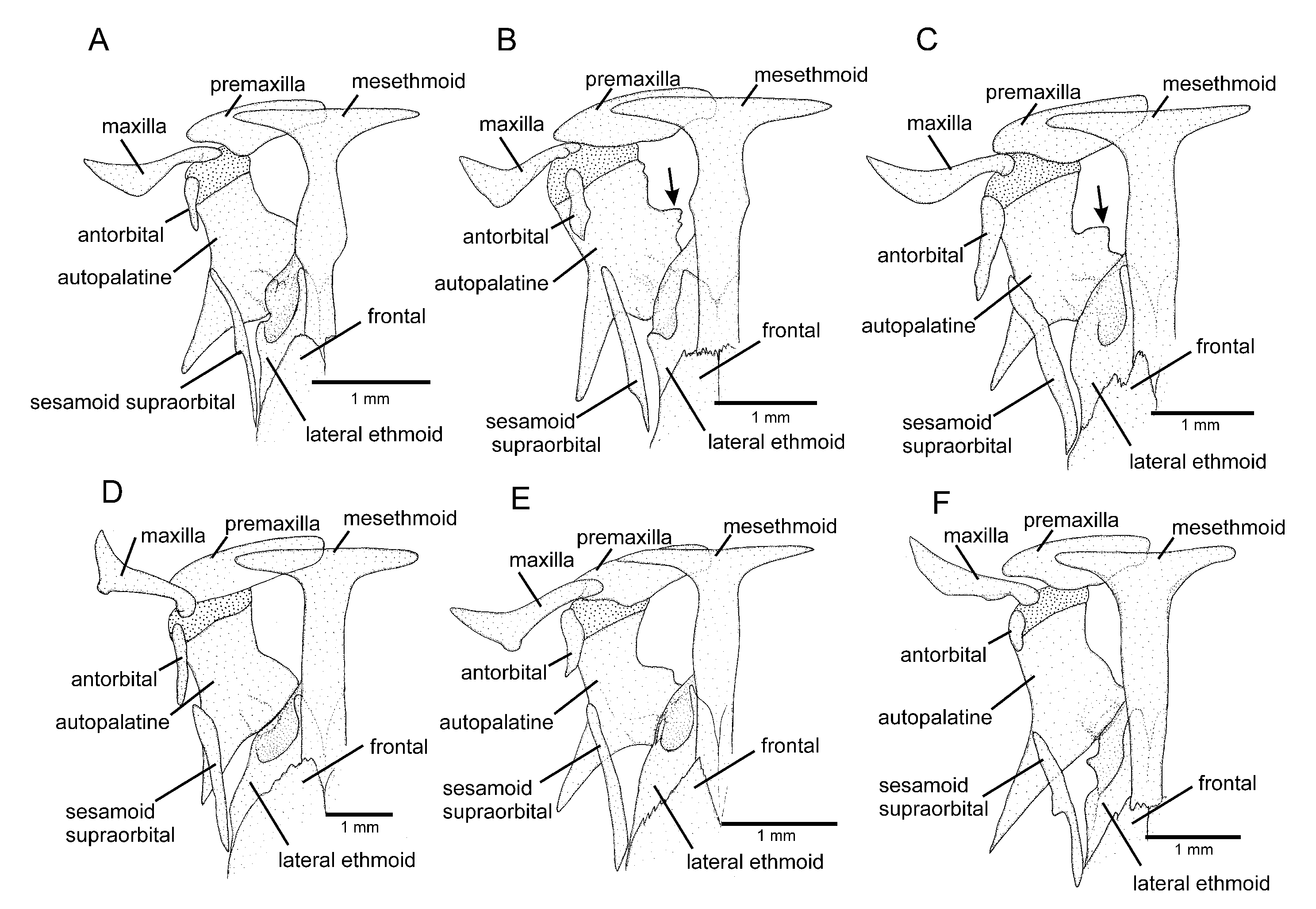
C. cubataonis by the absence of the anterior infraorbital canal (vs. presence) and by having fewer vertebrae (36 or 37 vs. 40 or 41), fewer ribs (12 or 13 vs. 15 or 16), dorsal-fin origin at a vertical through the centrum of the 18th or 19th vertebra (vs. 21st); anal-fin origin at a vertical through the centrum of the 22nd or 23rd vertebra (vs. 25th or 26th); the autopalatine with a weakly concave medial margin (
Figure 5A; vs. deeply concave,
Figure 5B); and the lateral process of the parurohyal with straight posterior margin and distally terminating in sharply pointed tip (
Figure 6A; vs. curved posterior margin and truncate distal extremity,
Figure 6B).
Distribution.
Cambeva cf.
botuvera was found in river basins draining into the Baía de Babitonga and Baía de Guaratuba, and in the Rio Itapocu basin (
Figure 3).
Material examined. All from Brazil: Santa Catarina State. Baía de Guaratuba system: Rio São João basin: Garuva Municipality: UFRJ 13298, 1 ex. (C&S); UFRJ 13299, 3 exs.; Rio São João, 26°01′07″ S 48°50′56″ W, about 20 m asl; C. R. M. Feltrin, 25 July 2020. UFRJ 12908, 1 ex.; Rio Braço, 26°01′24″ S 48°51′44″ W, about 75 m asl; C. R. M. Feltrin & R. Dalcin, 16 April 2022. Baía de Babitonga system: Rio Três Barras basin: Garuva Municipality: UFRJ 13006, 8 ex.; Rio Três Barras, 26°05′40″ S 48°53′04″ W, about 20 m asl; C. R. M. Feltrin, 26 June 2022. Rio Cubatão do Norte basin: Joinville Municipality: UFRJ 8736, 16 ex.; UFRJ 8871, 3 ex. (C&S); Rio Lindo, tributary of the Rio Cubatão do Norte, 26°11′48″ S 48°55′19″ W, about 30 m asl; A. M. Katz et al., 29 May 2012. UFRJ 12886, 1 ex; middle course of the Rio Cubatão do Norte, Quiriri de Baixo, 26°08′35″ S 48°59′43″ W, about 35 m; C. R. M. Feltrin, 17 April 2022. UFRJ 12891, 1 ex.; UFRJ 12914, 1 ex; Rio Alandaf, Pirabeiraba, 26°13′00″ S 48°54′56″ W, about 35 m asl; C. R. M. Feltrin, 17 April 2022. UFRJ 12882, 11 ex.; UFRJ 12873, 5 ex.; Rio Lindo, Pirabeiraba, 26°11′49″ S 48°55′18″ W, about 30 m asl; C. R. M. Feltrin, 15 April 2022. UFRJ 10631, 2 ex; Rio Pirabeiraba, 26°08′42″ S 48°54′25″ W, about 30 m asl; A. Katz et al., 11 June 2015. UFRJ 12889, 2 ex; Rio Cubatão do Norte, Quiriri de Baixo, 26°08′35″ S 48°59′44″ W, about 55 m asl; C. R. M. Feltrin, 17 April 2022. UFRJ 12971, 1 ex; Rio Quiriri, 26°07′48″ S 49°00′32″ W, about 95 m asl; R. Dalcin, 15 April 2021. UFRJ 13008, 13 ex; UFRJ 13010, 1 ex.; stream tributary of Rio Quiriri, Quiriri de Baixo, 26°06′59″ S 49°00′11″ W, about 110 m asl; C. R. M. Feltrin, 26 July 2022. UFRJ 13214, 4 ex.; middle course of Rio Cubatão do Norte, Quiriri de Baixo, 26°08′35″ S 48°59′43″ W, about 35 m; C. R. M. Feltrin, 6 November 2020. UFRJ 13268, 2 ex.; Rio Quiriri, 26°15′50″ S 48°58′50″ W, about 85 m asl; R. Dalcin, 15 April 2021. UFRJ 12880, 3 ex.; same locality and collector as UFRJ 13214, 15 April 2019. UFRJ 13269, 18 ex.; Rio Cubatão do Norte, 26°08′48″ S 49°00′44″ W, about 95 m asl; C. R. M. Feltrin, 26 July 2022. MHNCI 11110, 2 ex.; Rio da Serra, a tributary of Rio Quiriri, Alto Quiriri, 49°00′33″ W 26°06′01″ S, V. Abilhoa, L.F. Duboc, L.P. Bastos & G. Otto, 20 April 2004. MHNCI 11057, 2 ex.; Rio Quiriri, Alto Quiriri, 49°00′12″ W 26°06′08″ S, V. Abilhoa, L.F. Duboc, L.P. Bastos, G. Otto, 20 April 2004. Rio Itapocu basin: Schroeder Municipality: UFRJ 12885, 8 ex; UFRJ 12870, 2 ex.; Rio Hern, tributary of Rio Itapocuzinho, Schroeder city, 26°25′33″ S 49°03′57″ W, about 35 m asl; C. R. M. Feltrin, 15 April 2022. UFRJ 12884, 1 ex; Rio Braço do Sul, tributary of Rio Itapocuzinho, 26°22′29″ S 49°03′52″ W, about 50 m asl; C. R. M. Feltrin, 19 January 2022. Corupá Municipality: UFRJ 12627, 7 ex.; Rio Novo, 26°25′37″ S 49°17′42″ W, about 120 m asl; C. R. M. Feltrin, 20 December 2019. UFRJ 13195, 2 ex; UFRJ 13196, 2 ex; Rio Isabel, tributary of Rio Novo, 26°28′45″ S 49°18′16″ W, about 165 m asl; C. R. M. Feltrin, 15 April 2022. UFRJ 13191, 4 ex.; UFRJ 13190, 2 ex.; Rio Paulo Grande, tributary to Rio Isabel, tributary of Rio Novo, 26°26′52″ S 49°17′31″ W, about 110 m; C. R. M. Feltrin, 30 August 2022. UFRJ 13193, 4 ex.; UFRJ 13194, 10 ex.; stream tributary of Rio Isabel, tributary of Rio Novo, 26°28′18″ S 49°17′56″ W, about 150 m asl; C. R. M. Feltrin, 17 April 2022. UFRJ 13201, 5 ex.; UFRJ 13202, 2 ex.; Rio Humboldt, Osvaldo Amaral, 26°23′21″ S 49°15′12″ W, about 90 m; C. R. M. Feltrin, 29 December 2022. UFRJ 13192, 2 ex.; Rio Paulo Pequeno, tributary of Rio Paulo Grande, tributary of Rio Isabel, tributary of Rio Novo, 26°27′11″ S 49°18′29″ W, about 115 m asl; C. R. M. Feltrin, 30 August 2022. UFRJ 12861, 4 ex.; upper Rio Itapocu, João Tozini, 26°26′17″ S 49°13′21″ W, about 35 m asl; C. R. M. Feltrin, 15 April 2022. UFRJ 13218, 3 ex. (C&S); UFRJ 13219, 15 ex.; UFRJ 13216, 2 ex. (C&S); same locality and collector as UFRJ 12861, 26 November 2020. UFRJ 12865, 8 ex; Rio Novo, 26°25′51″ S 49°16′47″ W, about 95 m SL; C. R. M. Feltrin, 15 April 2022. Jaraguá do Sul Municipality: UFRJ 12907, 5 ex., stream at the city of Jaraguá do Sul, SC, 26°29′38″ S 49°04′10″ W, about 30 m asl; C. R. M. Feltrin, 15 April 2022. UFRJ 12903, 1 ex; UFRJ 12864, 1 ex.; Ribeirão Cavalo, tributary of the upper Rio Itapocu, 26°27′43″ S 49°11′03″ W, about 80 m asl; C. R. M. Feltrin, 15 April 2022. UFRJ 12862, 2 ex.; UFRJ 12888, 3 ex.; Rio Cerro, 26°33′21″ S 49°08′01″ W, about 50 m asl; C. R. M. Feltrin, 15 April 2022. Massaranduba Municipality: UFRJ 12902, 2 ex; UFRJ 12874, 2 ex.; Rio Sete de Janeiro, 26°39′40″ S 49°00′46″ W, about 80 m asl; C. R. M. Feltrin, 15 April 2022. UFRJ 13209, 11 ex.; Ribeirão Treze de Maio, tributary of Rio Massaranduba; 26°39′42″ S 49°02′01″ W, about 70 m asl; 15 April 2022.
Cambeva chrysornata sp. nov.
LSID:urn:lsid:zoobank.org:act:B69BBE4B-8FD5-4878-BFDC-45EF4F28C627
Cambeva cubataonis (non
Trichomycterus cubataonis Bizerril, 1994): Donin, Ferrer & Carvalho [
30] (Figure 6C–E in Ref. [
30]) (misidentification).
Holotype. UFRJ 13011, 94.5 mm SL; Brazil: Santa Catarina State: Garuva Municipality: a stream tributary of Rio Palmital, Baía de Babitonga system, near the centre of the town of Garuva, 26°02′05″ S 48°51′28″ W, about 35 m asl; C. R. M. Feltrin, 25 June 2022.
Paratypes. UFRJ 12899, 3 ex., 30.0–71.4 mm SL; UFRJ 13017, 2 ex., 42.7–65.1 mm SL (C&S); UFRJ 12860, 1 ex., 38.2 mm SL (DNA); CICCAA 07549, 1 ex., 42.9 mm SL; same locality as holotype; C. R. M. Feltrin and R. H. Dalcin, 16 April 2022.
Diagnosis. Cambeva chrysornata is distinguished from all other congeners, except C. cubataonis and C. guaratuba, by adult specimens above about 40 mm SL, having flank and dorsum predominantly dark grey to black contrasting with bright yellow marks (vs. never a similar colour pattern) and the presence of odontodes with a rounded extremity at least on the internal-most series of the interopercle in specimens above about 60 mm SL (vs. pointed odontodes). Cambeva chrysornata is distinguished from C. cubataonis and C. guaratuba by having shorter barbels, the tip of the rictal barbel posteriorly reaching the anterior margin of the orbit or an area anterior to it (vs. reaching area posterior to the orbit) and maxillary and rictal barbels not reaching interopercular patch of odontodes (vs. reaching interopercular patch of odontodes), a slender dorsal fin with short rays, with the longest ray shorter than the dorsal-fin base (vs. longest ray longer than the dorsal-fin base), pectoral fin rounded in dorsal view (vs. subtriangular), pelvic fin rounded, its posterior extremity reaching a vertical anterior to the dorsal-fin origin (vs. truncate, its posterior extremity reaching a vertical through the anterior half of the dorsal-fin base), fewer procurrent caudal-fin rays (total of 16 or 17 dorsally and 11 or 12 ventrally, vs. 20 or 21 dorsally and 13 or 14 ventrally), fewer vertebrae (38 or 39, vs. 40 or 41), and the presence of an irregularly shaped golden longitudinal line on the medio-dorsal portion of the flank (vs. no distinctive golden longitudinal line on the medio-dorsal portion of the flank). Cambeva chrysornata is similar to C. cubataonis and is distinguished from C. guaratuba by the medial margin of the autopalatine exhibiting a deep concavity in its middle portion, posteriorly followed by a distinctive projection towards the mesethmoid. Cambeva chrysornata is also distinguished from C. cubataonis by having 14 vertebrae (vs. 15–16).
Description. General morphology: Morphometric data are presented in
Table 1. Body relatively slender, subcylindrical on anterior region, compressed on posterior region. Greatest body depth in area immediately anterior to pelvic-fin base. Dorsal profile slightly convex between snout and dorsal-fin base end, nearly straight on caudal peduncle; ventral profile convex on head, approximately straight on trunk. Anus and urogenital papilla opening at vertical through middle portion of dorsal-fin base. Head sub-trapezoidal, with anterior profile of snout slightly convex in dorsal view. Eye small, dorsally positioned on head, nearer snout tip than posterior margin of opercle. Distance between anterior and posterior nostrils about half distance between posterior nostril and orbital rim. Barbels narrow and short, tip of nasal barbel posteriorly reaching orbit, tip of maxillary and rictal barbels reaching area anterior to interopercular patch of odontodes. Mouth subterminal. Lateral fleshy lobe of the mouth small, its largest length about one-fourth of lower jaw length excluding lobes; ventral surface of lobe flat. Jaw teeth irregularly arranged, more external teeth longer, incisiform in specimens above about 50 mm SL, with rounded extremity in smaller specimens, more internal teeth pointed, 40–54 on premaxilla, 41–66 on dentary. Minute skin papillae on ventral surface of head, including lateral lobe of mouth, and dorso-lateral surface of snout. Branchial membrane attached to isthmus only at its anterior-most point, in ventral midline.
Dorsal and anal fins subtriangular, margin rounded, dorsal fin slender, rays short, longest ray shorter than fin base. Total dorsal-fin rays 11 (ii + II + 7), total anal-fin rays 9 (ii + II + 5). Anal-fin origin at posterior portion to dorsal-fin base. Dorsal-fin origin at vertical through centrum of 20th or 21st vertebra; anal-fin origin at vertical through centrum of 24th or 25th vertebra. Pectoral fin rounded in dorsal view, first pectoral-fin ray shorter than second ray, not forming terminal filament. Total pectoral-fin rays 7 (I + 6). Pelvic fin rounded, its posterior extremity in vertical anterior to dorsal-fin origin. Pelvic-fin bases medially separated by minute interspace. Total pelvic-fin rays 5 (I + 4). Caudal fin subtruncate, posterior corners rounded. Total principal caudal-fin rays 13 (I + 11 + I), total dorsal procurrent rays 16 or 17 (xv–xvi + I), total ventral procurrent rays 10 or 11 (ix–x + I).
Laterosensory system: Supraorbital, posterior section of infraorbital canal and postorbital canal continuous. Supraorbital sensory canal pores 3: s1, adjacent to medial margin of anterior nostril; s3, adjacent and just posterior to medial margin of posterior nostril; s6, in transverse line through posterior half of orbit; pore s6 slightly nearer orbit than its paired s6 homologous pore. Anterior infraorbital sensory canal pores 2: i1, at transverse line through anterior nostril, and i3, at transverse line just anterior to posterior nostril. Posterior infraorbital sensory canal pores 2: pore i10, adjacent to ventral margin of orbit, and pore i11, posterior to orbit. Postorbital canal pores 2: po1, at vertical through posterior portion of interopercular patch of odontodes, and po2, at vertical through posterior portion of opercular patch of odontodes. Lateral line pores 2; posterior-most pore at vertical just posterior to pectoral-fin base.
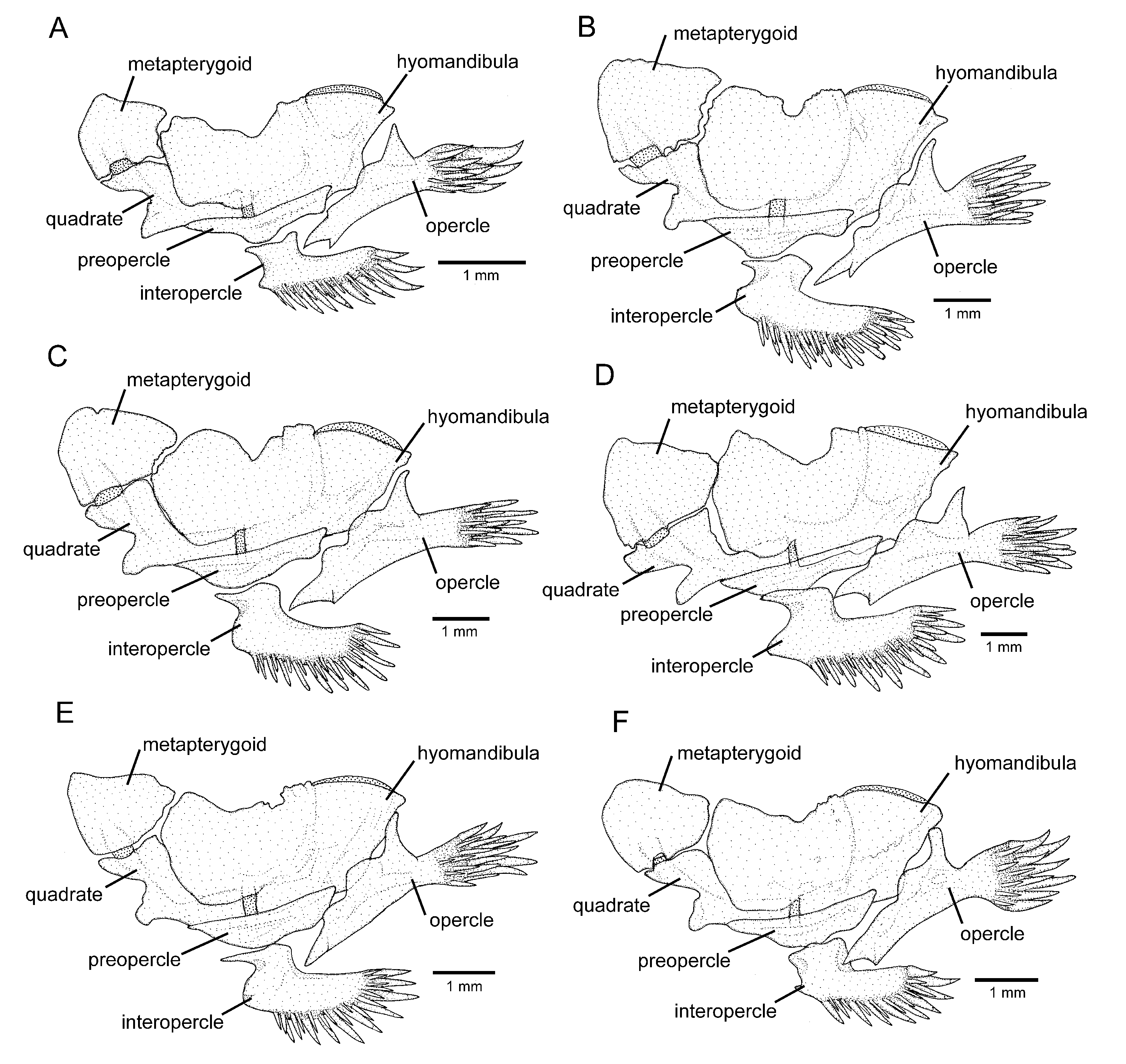
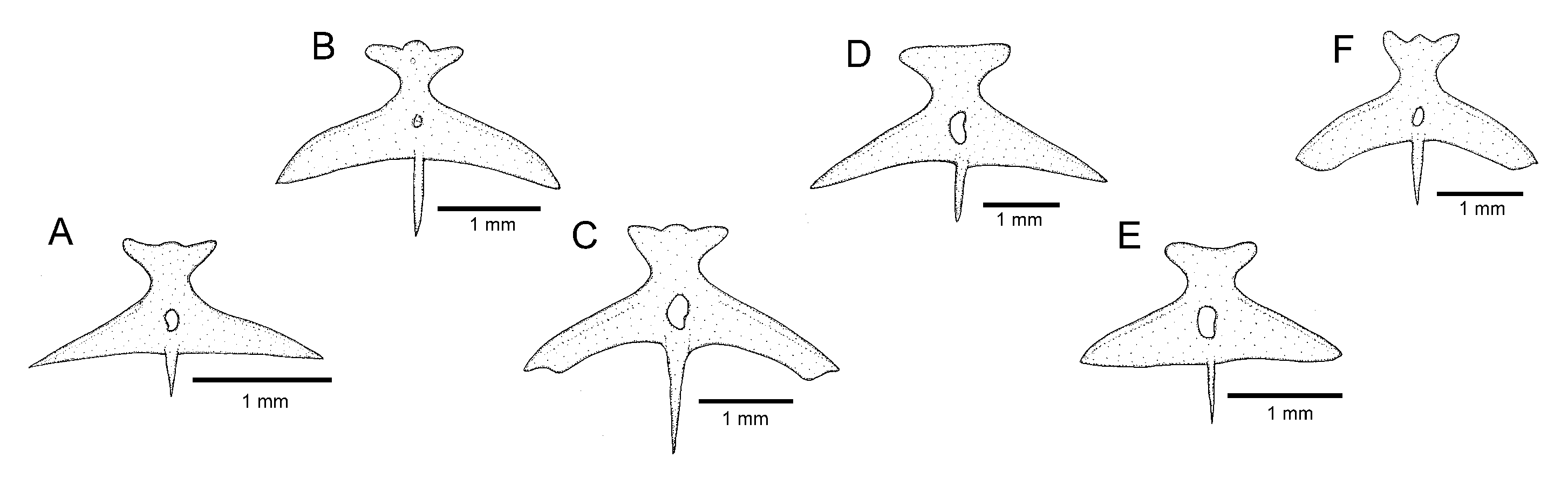
Osteology: Mesethmoid thin, anterior margin straight to slightly convex, lateral margin with small projection just anterior to lateral ethmoid. Mesethmoid cornu narrow, tip rounded. Postero-lateral area of lateral ethmoid with pronounced projection. Antorbital thin, sub-elliptical, about half length of premaxilla. Sesamoid supraorbital slender, flat, with small postero-lateral projection, its length about two and half times antorbital length. Premaxilla sub-trapezoidal in dorsal view, laterally narrowing, longer than maxilla. Maxilla curved, about L-shaped. Autopalatine sub-rectangular in dorsal view when excluding its postero-lateral process, its largest width about two-thirds of its length, including anterior cartilage; medial margin sinuous, with deep concavity in middle portion, posteriorly followed by distinctive projection towards mesethmoid. Autopalatine posterolateral process well-developed, its length about equal to autopalatine length without anterior cartilage. Metapterygoid thin, longer than deep. Quadrate slender, dorsal process with constricted base, dorsoposterior margin in contact with hyomandibula outgrowth. Hyomandibula long, with well-developed anterior outgrowth; dorsal margin of hyomandibula outgrowth with shallow concavity. Opercle long, longer than interopercle; opercular odontode patch robust, its width about three-fourths of dorsal hyomandibula articular facet. Opercular odontodes 18, irregularly arranged, narrow, nearly straight. Dorsal process of opercle short, slightly curved, extremity rounded. Opercular articular facet for hyomandibula with rounded flap, articular facet for preopercle small, about half size of articular facet for hyomandibula, rounded. Interopercle moderate in length, about equal to hyomandibula outgrow horizontal length, with rounded anterior margin. Interopercular odontodes 30 or 31, nearly straight, larger odontodes as long as larger opercular odontodes, irregularly arranged; in specimens above about 50 mm SL, posterior odontodes with rounded tip, anterior odontodes pointed, in smaller specimens, odontodes always pointed. Preopercle compact, with minute ventral projection. Parurohyal robust, lateral process slightly curved. Parurohyal head well-developed, with prominent anterolateral paired process. Middle parurohyal foramen minute. Posterior process of parurohyal moderately long, about three-fourths distance between anterior margin of parurohyal and anterior insertion of posterior process. Branchiostegal rays 8 or 9. Vertebrae 38 or 39. Ribs 14. Two dorsal hypural plates corresponding to hypurals 3 + 4 + 5; single ventral hypural plate corresponding to hypurals 1 + 2 and parhypural.
Colouration in alcohol: Flank, dorsum and head side dark grey to brown, darker on dorsal portion, with irregularly shaped yellow reticulation, more diffused ventrally on flank and head, usually forming pale golden longitudinal line along medio-dorsal portion of flank, between area just posterior to head and area below dorsal-fin base, sometimes interrupted or with dorsal extensions. Venter and ventral surface of head white. Barbels grey. Fins hyaline with white basal portion, sometimes with dark chromatophores forming pale grey dots. Smallest specimen (30.0 mm SL), body homogeneously pale grey, with broad longitudinal dark grey band along dorsal portion of flank.
Etymology. From the Greek chrysos (gold) and the Latin ornata (ornate), an allusion to the characteristic bright yellow marks on the flank of the new species.
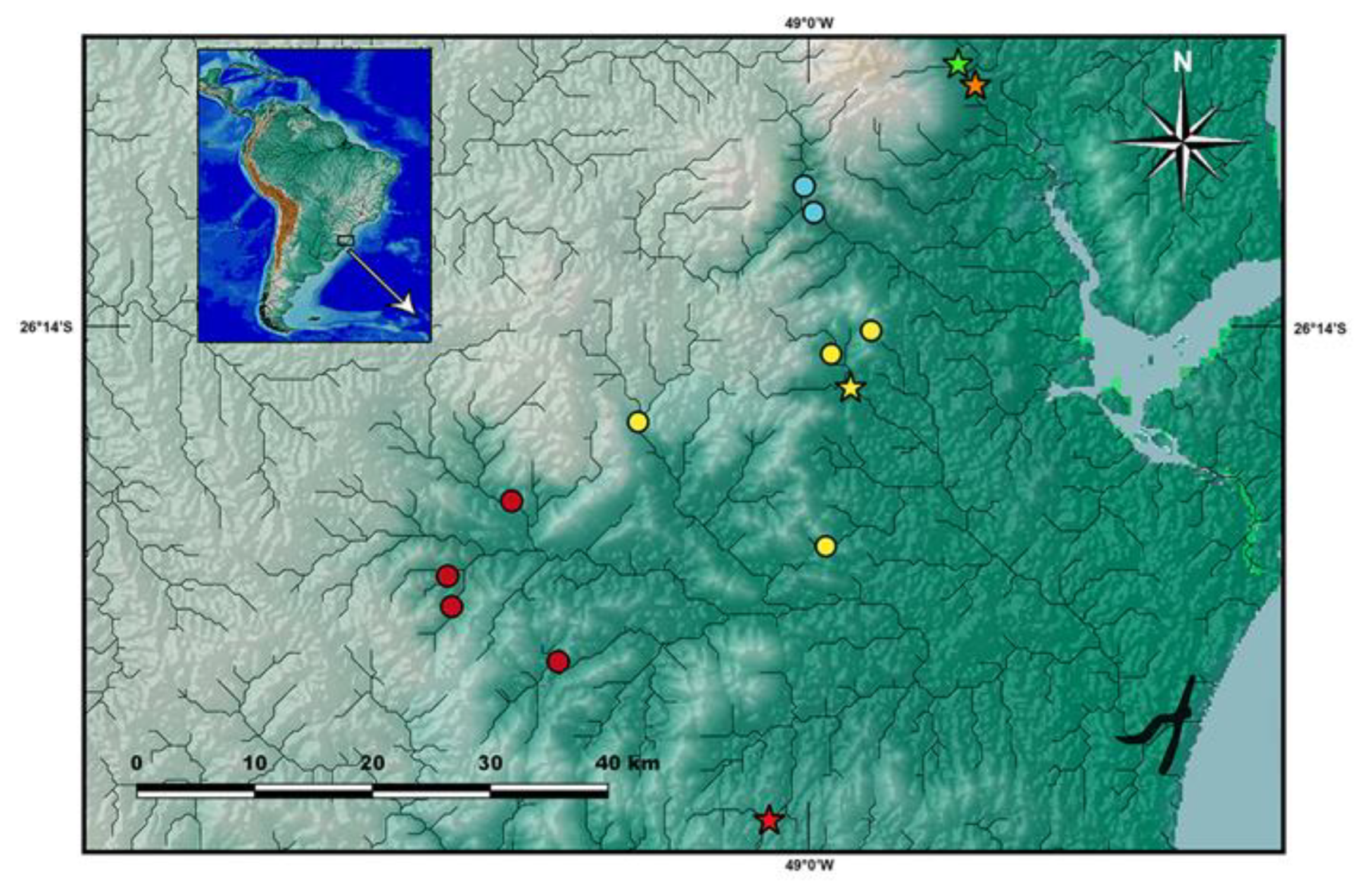
Distribution and habitat notes.
Cambeva chrysornata is only known from a single locality, a stream tributary of the upper Rio Palmital basin, which is the northern-most basin of the Baía de Babitonga basin, altitude about 35 m asl (
Figure 4). Different sections of the basin were sampled, including the Rio da Anta, Rio da Onça and Rio Três Barras, but
C. chrysornata was only found at the type locality site, where the species was rare. The type locality is situated within the urban area of the city of Garuva. While sampling the stream along about 2 km above the type locality, discharges of domestic effluents such as detergents, coliforms, oils and hydrocarbons were recorded. Below the type locality, the hydrographic basin is highly modified, crossing the city. Marginal vegetation comprises a narrow zone of degraded riparian forest. At the type locality, the stream is about 1.5 to 5 m wide and about 0.05 to 1 m deep. Most type specimens were found below median to large sized stones, about 0.4 m of diameter, but smaller specimens were found below smaller pebbles, about 0.05 to 0.15 m of diameter, and the two largest specimens under leaf litter below a large stone about 0.70 m of diameter.
Cambeva cubataonis (Bizerril, 1994)
Trichomycterus cubataonis Bizerril, 1994 [
23] (p. 618).
Cambeva cubataonis (Bizerril): Katz et al. [
21] (p. 563) (new combination).
Diagnosis. Cambeva cubataonis is distinguished from all other congeners, except C. chrysornata and C. guaratuba, by adult specimens above about 40 mm SL, having flank and dorsum predominantly dark grey to black contrasting with bright yellow marks (vs. never a similar colour pattern) and the presence of odontodes with a rounded extremity at least on the internal-most series of the interopercle in larger specimens, above about 60 mm SL (vs. always pointed odontodes). Cambeva cubataonis is distinguished from C. chrysornata and C. guaratuba by having 15 or 16 ribs (vs. 14) and a relatively longer posterior process of the parurohyal, its length longer that the length between the anterior margin of the parurohyal and the anterior insertion of the posterior process. Cambeva cubataonis is also distinguished from C. chrysornata by having longer barbels, the tip of the rictal barbel posteriorly reaching area posterior to the orbit (vs. reaching the anterior margin of the orbit or an area anterior to it) and maxillary and rictal barbels reaching interopercular patch of odontodes (vs. not reaching), a relatively deep dorsal fin, with the longest ray longer than the dorsal-fin base (vs. longest ray shorter than the dorsal-fin base), pectoral fin subtriangular in dorsal view (vs. rounded), pelvic fin truncate, its posterior extremity reaching a vertical at the anterior half of the dorsal-fin base (vs. rounded, its posterior extremity reaching a vertical anterior to the dorsal-fin origin), more procurrent caudal-fin rays (total of 20 or 21 dorsally and 13 or 14 ventrally, vs. 16 or 17 dorsally and 11 or 12 ventrally), more vertebrae (40 or 41, vs. 38 or 39), and the absence of an irregularly shaped golden longitudinal line on the medio-dorsal portion of the flank (vs. presence). Cambeva cubataonis is also distinguished from C. guaratuba by having jaw teeth with pointed to rounded tip, irregularly arranged (vs. truncate to slightly bilobed teeth, arranged in three rows), more opercular odontodes (18–20, vs. 13–16), and presence of a distinctive projection on the posterior portion of the medial margin of the autopalatine (vs. absence).
Distribution and ecological notes. Cambeva cubataonis presently is known only from the type locality area, in the Rio Cubatão do Norte basin, Baía de Babitonga system, in altitudes between about 45 and 110 m asl (
Figure 3). It was mostly found both in the middle main channel of the Rio Cubatão do Norte and in the Rio Quiriri, rarely being collected in tributary streams. Their preference for microhabitats follows that described for most congeners, with smaller specimens found below small pebbles, between about 0.01 and 0.15 m, and larger specimens in pebbles of greater diameters, about 0.25 m or more. The Rio Cubatão do Norte river, in the stretches where
C. cubataonis occurs, has an average width of about 35 m and deepest areas with 1.2 m. In the Rio Quiriri, the locality where
C. cubataonis was found is about 20 m wide and about 0.8 m at the deepest points. At both areas, the marginal forest is still relatively well preserved.
Material examined. All localities in Brazil: Santa Catarina State: Joinville Municipality: Rio Cubatão do Norte basin, Baía de Babitonga system: MNRJ 12490, holotype; middle Rio Cubatão do Norte; C. R. S. F. Bizerril & P. M. C. Araújo, 10 November 1991. UFRJ 13212, 1; UFRJ 13213, 1 (C&S); small tributary of the middle Rio Cubatão do Norte, Baía de Babitonga system, Quiriri de Baixo, 26°08′35″ S 48°59′44″ W, about 75 m asl; C. R. M. Feltrin, 6 November 2020. UFRJ 13189, 1; same locality and collector, 26 July 2022. UFRJ 13167, 8 (DNA); same locality and collector, 24 September 2022. UFRJ 13267, 2 ex., Rio Quiriri, 26°07′14″ S 49°00′14″ W, about 110 m asl; R. Dalcin, 15 April 2015. UFRJ 12978, 4; UFRJ 12972, 2 (C&S); same locality and collector as UFRJ 13267, 15 April 2021.
Cambeva guaratuba sp. nov.
LSID:urn:lsid:zoobank.org:act:DF384344-8118-4CD5-8965-6C1728CE209C
Cambeva cubataonis (non
Trichomycterus cubataonis Bizerril, 1994): Donin et al. [
30] (Figure 6A,B in Ref. [
30]) (misidentification).
Holotype. UFRJ 13296, 63.6 mm SL; Brazil: Paraná State: Guaratuba Municipality: Rio Imbira, a tributary of Rio São João, Baía de Guaratuba system, 25°57′31″ S 48°53′58″ W, about 105 m asl; C. R. M. Feltrin, 25 June 2022.
Paratypes. All from Brazil: UFRJ 13007, 6 ex., 28.1–63.0 mm SL; collected with the holotype. Santa Catarina State: Garuva Municipality: UFRJ 12302, 1 ex. (DNA), 49.6 mm SL; UFRJ 12303, 2 ex. (C&S), 27.5–82.2 mm SL; Rio São João, 26°01′22″ S 48°51′41″ W, about 70 m asl; R. Dalcin, 19 April 2014. CICCAA 07550, 1 ex., 67.9 mm SL; same locality as UFRJ 12302; R. Dalcin, 15 September 2019.
Diagnosis: Cambeva guaratuba is distinguished from all other congeners, except C. chrysornata and C. cubataonis, by adult specimens, above about 40 mm SL, having flank and dorsum predominantly dark grey to black contrasting with bright yellow marks (vs. never a similar colour pattern) and the presence of odontodes with a rounded extremity at least on the internal-most series of the interopercle in larger specimens, above about 60 mm SL (vs. pointed odontodes). Cambeva guaratuba is distinguished from C. chrysornata and C. cubataonis by having truncate to slightly bilobed jaw teeth, arranged in three rows (vs. teeth with pointed to rounded tip, irregularly arranged), fewer opercular odontodes (13–16, vs. 18–20), and absence of a distinctive projection on the posterior portion of the medial margin of the autopalatine (vs. presence). Cambeva guaratuba is also distinguished from C. chrysornata by having longer barbels, the tip of the rictal barbel posteriorly reaching area posterior to the orbit (vs. reaching the anterior margin of the orbit or an area anterior to it) and maxillary and rictal barbels reaching interopercular patch of odontodes (vs. not reaching), a relatively deep dorsal fin, with the longest ray longer than the dorsal-fin base (vs. longest ray shorter than the dorsal-fin base), pectoral fin subtriangular in dorsal view (vs. rounded), pelvic fin truncate, its posterior extremity reaching a vertical at the anterior half of the dorsal-fin base (vs. rounded, its posterior extremity reaching a vertical anterior to the dorsal-fin origin), more procurrent caudal-fin rays (total of 20 or 21 dorsally and 13 or 14 ventrally, vs. 16 or 17 dorsally and 11 or 12 ventrally), more vertebrae (40 or 41, vs. 38 or 39), and the absence of an irregularly shaped golden longitudinal line on the medio-dorsal portion of the flank (vs. presence). Cambeva guaratuba is also distinguished from C. cubataonis by having 14 vertebrae (vs. 15–16).
Description. General morphology: Morphometric data are presented in
Table 2. Body relatively slender, subcylindrical on anterior region, compressed on posterior region. Greatest body depth in area immediately anterior to pelvic-fin base. Dorsal profile slightly convex between snout and dorsal-fin base end, nearly straight on caudal peduncle; ventral profile convex on head, approximately straight on trunk. Anus and urogenital papilla opening at vertical through area just anterior to middle of dorsal-fin base. Head sub-trapezoidal, with anterior profile of snout slightly convex in dorsal view. Eye small, dorsally positioned on head, nearer snout tip than posterior margin of opercle. Distance between anterior and posterior nostrils about half distance between posterior nostril and orbital rim. Barbels narrow, moderate in length, tip of nasal barbel reaching area between orbit and opercular patch of odontodes, tip of maxillary barbel reaching posterior portion of interopercular patch of odontodes, rictal barbel reaching middle portion of interopercular patch of odontodes. Mouth subterminal. Lateral fleshy lobe of the mouth small, its largest length about one fourth of lower jaw length excluding lobes; ventral surface of lobe flat. Jaw teeth incisiform, sometimes slightly bilobed in specimens above about 50 mm SL, pointed in smaller specimens, 38–40 on premaxilla, 38–43 on dentary. Minute skin papillae on ventral surface of head, including lateral lobe of mouth, and dorso-lateral surface of snout. Branchial membrane attached to isthmus only at its anterior-most point, in ventral midline.
Dorsal and anal fins subtriangular, anterior margin straight, posterior margin slightly convex, longest ray longer than fin base. Total dorsal-fin rays 11 (ii + II + 7), total anal-fin rays 9 (ii + II + 5). Anal-fin origin at posterior portion to dorsal-fin base, at vertical through base of fifth bifid ray. Dorsal-fin origin at vertical through centrum of 20th vertebra; anal-fin origin at vertical through centrum of 24th vertebra. Pectoral fin subtriangular in dorsal view, first pectoral-fin ray shorter than second ray, not forming terminal filament. Total pectoral-fin rays 7 (I + 6). Pelvic fin truncate, its posterior extremity at vertical through anterior portion of dorsal-fin base. Pelvic-fin bases medially in close proximity. Total pelvic-fin rays 5 (I + 4). Caudal fin subtruncate, posterior corners rounded. Total principal caudal-fin rays 13 (I + 11 + I), total dorsal procurrent rays 20 (xix + I), total ventral procurrent rays 13 or 14 (xii–xiii + I)
Laterosensory system: Supraorbital, posterior section of infraorbital canal and postorbital canal continuous. Supraorbital sensory canal pores 3: s1, adjacent to medial margin of anterior nostril; s3, adjacent and just posterior to medial margin of posterior nostril; s6, in transverse line through posterior half of orbit; pore s6 slightly nearer orbit than its paired s6 homologous pore. Anterior infraorbital sensory canal pores 2: i1, at transverse line through anterior nostril, and i3, at transverse line just anterior to posterior nostril. Posterior infraorbital sensory canal pores 2: pore i10, adjacent to ventral margin of orbit, and pore i11, posterior to orbit. Postorbital canal pores 2: po1, at vertical through posterior portion of interopercular patch of odontodes, and po2, at vertical through posterior portion of opercular patch of odontodes. Lateral line pores 2; posterior-most pore at vertical just posterior to pectoral-fin base.
Osteology: Mesethmoid thin, anterior margin approximately straight, lateral margin without projections. Mesethmoid cornu narrow, tip rounded. No distinctive projection on lateral surface of lateral ethmoid. Antorbital thin, elongate, about half length of premaxilla. Sesamoid supraorbital slender, flat, without lateral projections, its length about two times antorbital length. Premaxilla sub-trapezoidal in dorsal view, longer than maxilla. Maxilla curved, boomerang-shaped. Autopalatine sub-rectangular in dorsal view when excluding its postero-lateral process, its largest width about three fourths of its length, including anterior cartilage; medial margin with pronounced concavity, lateral margin slightly concave. Autopalatine posterolateral process well-developed, triangular in dorsal view, its length about two-thirds of autopalatine length. Metapterygoid thin, longer than deep. Quadrate slender, dorsal process with constricted base, dorsoposterior margin in contact with hyomandibula outgrowth. Hyomandibula long, with well-developed anterior outgrowth; dorsal margin of hyomandibula outgrowth with shallow concavity. Opercle long, longer than interopercle; opercular odontode patch robust, its width about three fourths of dorsal hyomandibula articular facet. Opercular odontodes 13–16, irregularly arranged, narrow, nearly straight, with rounded to slightly pointed extremity. Dorsal process of opercle short, extremity pointed. Opercular articular facet for hyomandibula with rounded flap, articular facet for preopercle slightly smaller than articular facet for hyomandibula, rounded. Interopercle moderate in length, about equal to hyomandibula outgrow horizontal length, with prominent expansion on anterior margin. Interopercular odontodes 28–32, nearly straight, larger odontodes as long as larger opercular odontodes, irregularly arranged; in specimens above about 50 mm SL, posterior odontodes with rounded tip, anterior odontodes pointed, in smaller specimens, odontodes always pointed. Preopercle compact, with minute ventral projection. Parurohyal robust, lateral process triangular, posterior margin about straight. Parurohyal head well-developed, with prominent anterolateral paired process. Middle parurohyal oval. Posterior process of parurohyal short, about half distance between anterior margin of parurohyal and anterior insertion of posterior process. Branchiostegal rays 8. Vertebrae 40. Ribs 14. Two dorsal hypural plates corresponding to hypurals 3 + 4 + 5; single ventral hypural plate corresponding to hypurals 1 + 2 and parhypural.
Colouration in alcohol: Flank, dorsum and head side dark grey to brown, with irregularly shaped yellow reticulation. Venter and ventral surface of head white. Barbels grey. Fins hyaline with dark chromatophores forming dark grey spots on basal region. In smallest specimens, about 30.0 mm SL, body pale grey with small brown spots.
Etymology. The name guaratuba is an allusion to the occurrence of the new species in the Baía de Guaratuba system, here used as a noun in apposition. From the Tupi-Guarani, the name guaratuba means guara (popular name of the bird Eudocimus ruber Linnaeus, 1758) and tuba (numerous), designating a place inhabited by numerous birds of this species.
Distribution and habitat notes.
Cambeva guaratuba was found in localities of the Rio São João basin, which is connected to the Baía de Guaratuba system, in altitudes between about 70 and 105 m asl (
Figure 3). At the type locality area, the Rio Imbira is a fast-flowing stream, between about 7 and 15 m wide, and about 1 m deep in the deepest areas, with the bottom substrate mostly comprising pebbles and stones, between about 0.01 and 0.6 of diameter. Larger specimens were found below larger stones, as well as, within tangles of marginal plant roots. The marginal forest is relatively well-preserved, and the water does not present vestiges of pollution. However, frequent accidents with freight trucks on the road adjacent to this drainage area, the BR-376 road, can have negative effects on water quality. In the last 12 years, 89 accidents with spillage of dangerous products occurred in the region. For example, in February 2022, a truck carrying 31,970 kg of residual 69% sulfuric acid overturned, spilling its entire load into a tributary of the São João River. The rarity of this species may be related to these accidents.
Cambeva ventropapillata sp. nov.
LSID:urn:lsid:zoobank.org:act:82C632F7-3A94-4DE4-96AE-8053DD857349
Trichomycterus nigricans (non
Trichomycterus nigricans Valenciennes, 1832): de Pinna [
56] (p. 227).
Holotype. UFRJ 13013, 75.3 mm SL; Brazil: Santa Catarina State: Massaranduba Municipality: Ribeirão Treze de Maio, a tributary of Rio Massaranduba, itself a tributary of Rio Pitanga, Rio Itapocu basin, 26°39′42″ S 49°02′01″ W, about 75 m asl; C. R. M. Feltrin, 15 April 2022.
Paratypes. All from Brazil: Estado de Santa Catarina: Rio Itapocu basin. Massaranduba Municipality: UFRJ 12910, 5 ex., 35.7–39.5 mm SL; UFRJ 13016, 3 ex. (C&S), 40.8–54.7 mm SL; UFRJ 12859, 2 ex. (DNA), 34.6–38.4 mm SL; collected with holotype. Corupá Municipality: UFRJ 13295, 1 ex., 79.9 mm SL; Rio Novo, tributary of Rio Itapocu; C. R. M. Feltrin, 20 December 2019. UFRJ 13203, 2 ex., 63.3–72.7 mm SL; Rio Paulo Pequeno, tributary of Rio Paulo Grande, tributary of Rio Isabel, tributary of Rio Novo, 26°27′11″ S 49°18′29″ W, about 115 m asl; C. R. M. Feltrin, 8 December 2021. UFRJ 13220, 5 ex., 57.4–82.8 mm SL; UFRJ 13217, 1 ex. (C&S), 56.1 mm SL; upper Rio Itapocu, João Tozini, 26°26′17″ S 49°13′21″ W, about 35 m asl; C. R. M. Feltrin, 26 November 2020. UFRJ 13207, 1 ex., 56.9 mm SL; Rio Paulo Grande, tributary to Rio Isabel, tributary of Rio Novo, 26°26′52″ S 49°17′31″ W, about 110 m; C. R. M. Feltrin, 16 August 2022. UFRJ 13199, 4 ex., 24.4–46.7 mm SL; Rio Humboldt, Osvaldo Amaral, 26°23′21″ S 49°15′12″ W, about 90 m; C. R. M. Feltrin, 31 August 2022. UFRJ 13206, 2 ex., 50.0–75.2 mm SL; Rio Isabel, tributary of Rio Novo, 26°28′45″ S 49°18′16″ W, about 165 m asl; C. R. M. Feltrin, 30 August 2022. Jaraguá do Sul Municipality: CICCAA 07551; 2 ex., 36.0–46.3 mm SL; Rio Jaraguá, about 1 km SE from the village of Garibaldi, 26°31′34″ S 49°12′49″ W, about 75 m asl; C. R. M. Feltrin, 15 April 2022. São Bento do Sul Municipality: MHNCI 11949, 9 ex., 31.1–77.5 mm SL; Rio Vermelho, a tributary of Rio Humboldt, 49°19′12″ W 26°18′35″ S, about 730 m asl; V. Abilhoa, L.F. Duboc and P. Pinheiro, 8 August 2006.
Diagnosis. Cambeva ventropapillata is distinguished from all congeners, except C. papillifera (Wosiacki & Garavello, 2004), by having broad laminar, ribbon-shaped nasal barbels (vs. narrow, wire-shaped), maxillary and rictal barbels broad at their basal portion, abruptly narrowing distally (vs. not distinctively widened at the proximal portion, gradually narrowing distally), and large skin papillae on the ventral surface of the head (vs. papillae minute or rudimentary). Cambeva ventropapillata differs from C. papillifera by having longer maxillary and rictal barbels, their tips posteriorly reaching the anterior region of the interopercular patch of odontodes (vs. reaching area about midway between barbel base and interopercular patch of odontodes), mouth subterminal (vs. ventral), and the anal-fin origin at a vertical through the posterior portion of the dorsal-fin base (vs. through the middle of the dorsal-fin base).
Description. General morphology: Morphometric data are presented in
Table 3. Body slender to moderately deep, subcylindrical on anterior region, compressed on posterior region. Greatest body depth in area immediately anterior to pelvic-fin base. Dorsal profile slightly convex between snout and dorsal-fin base end, nearly straight on caudal peduncle; ventral profile convex on head, approximately straight on trunk. Anus and urogenital papilla opening at vertical through area just posterior to dorsal-fin origin. Head sub-trapezoidal, with anterior profile of snout slightly convex in dorsal view. Eye small, dorsally positioned on head, nearer snout tip than posterior margin of opercle. Distance between anterior and posterior nostrils about half distance between posterior nostril and orbital rim. Barbels broad, short, nasal barbel ribbon-shaped, often with waved margin, maxillary and rictal barbels abruptly narrowing distally. Tip of nasal barbel posteriorly reaching posterior margin of orbit or area immediately posterior to it, tip of maxillary and rictal barbels reaching anterior region of interopercular patch of odontodes. Mouth subterminal. Lateral fleshy lobe of the mouth moderate in size, its largest length about one-third of lower jaw length excluding lobes; ventral surface of lobe flat. Jaw teeth irregularly arranged, more external teeth longer, tip pointed to slightly rounded, 44–48 on premaxilla, 39–48 on dentary. Well-developed skin papillae on ventral surface of head, diameter of larger papilla about one-fifth of orbital diameter. Skin papillae extending over ventral and dorsal surfaces of snout and lateral lobe of mouth. Branchial membrane attached to isthmus only at its anterior-most point, in ventral midline.
Dorsal and anal fins subtriangular, margins about straight, dorsal fin moderately deep, longest ray longer than fin base. Total dorsal-fin rays 12–13 (iii–iv + II + 7), total anal-fin rays 10 (iii + II + 5). Anal-fin origin at vertical through posterior portion to dorsal-fin base, between bases of 5th and 6th bifid rays. Dorsal-fin origin at vertical through centrum of 20th or 21st vertebra; anal-fin origin at vertical through centrum of 25th or 26th vertebra. Pectoral fin subtriangular in dorsal view, margins rounded. First pectoral-fin ray shorter than second ray, not forming terminal filament. Total pectoral-fin rays 7 (I + 6). Pelvic fin subtruncate, its posterior extremity in vertical at dorsal-fin origin. Pelvic-fin bases medially separated by minute interspace. Total pelvic-fin rays 5 (I + 4). Caudal fin subtruncate, posterior corners rounded. Total principal caudal-fin rays 13 (I + 11 + I), total dorsal procurrent rays 17–20 (xvi–xix + I), total ventral procurrent rays 10–13 (ix–xii + I).
Laterosensory system: Supraorbital, posterior section of infraorbital canal and postorbital canal continuous. Supraorbital sensory canal pores 3: s1, adjacent to medial margin of anterior nostril; s3, adjacent and just posterior to medial margin of posterior nostril; s6, in transverse line through posterior half of orbit; pore s6 slightly nearer orbit than its paired s6 homologous pore. Anterior infraorbital sensory canal usually closed with pores 2: i1, at transverse line through anterior nostril, and i3, at transverse line just anterior to posterior nostril; sometimes canal opened. Posterior infraorbital sensory canal pores 2: pore i10, adjacent to ventral margin of orbit, and pore i11, posterior to orbit. Postorbital canal pores 2: po1, at vertical through posterior portion of interopercular patch of odontodes, and po2, at vertical through posterior portion of opercular patch of odontodes. Lateral line pores 2; posterior-most pore at vertical just posterior to pectoral-fin base.
Osteology: Mesethmoid thin, anterior margin straight to slightly convex. Mesethmoid cornu narrow, tip rounded. Lateral margin of lateral ethmoid without projections. Antorbital thin, sub-elliptical, about half length of premaxilla. Sesamoid supraorbital slender, flat, without lateral projections, its length about two and half times antorbital length. Premaxilla sub-trapezoidal in dorsal view, laterally narrowing, approximately equal to maxilla in length. Maxilla slightly curved. Autopalatine sub-rectangular in dorsal view when excluding its postero-lateral process, its largest width about half of its length when excluding anterior cartilage; medial margin, with deep concavity in middle portion. Autopalatine posterolateral process well-developed, its length about two-thirds of autopalatine length. Metapterygoid thin, longer than deep. Quadrate slender, dorsal process with constricted base, dorsoposterior margin in contact with hyomandibula outgrowth. Hyomandibula long, with well-developed anterior outgrowth; dorsal margin of hyomandibula outgrowth without pronounced concavities. Opercle long, longer than interopercle; opercular odontode patch robust, its width about three-fourths of dorsal hyomandibula articular facet. Opercular odontodes 13–24, straight and pointed, irregularly arranged. Dorsal process of opercle short, nearly straight; opercular articular facet for hyomandibula with rounded flap, articular facet for preopercle slightly smaller than articular facet for hyomandibula, rounded. Interopercle moderate in length, about equal to hyomandibula outgrow horizontal length, with about straight anterior margin and prominent anterior process on its dorsal limit. Interopercular odontodes 16–24, nearly straight, pointed, longer odontodes about as long as larger opercular odontodes, arranged in irregular longitudinal rows. Preopercle compact, with minute ventral projection. Parurohyal robust, lateral process triangular, posterior margin straight. Parurohyal head well-developed, with prominent anterolateral paired process. Middle parurohyal foramen large, elliptical. Posterior process of parurohyal moderately long, about half distance between anterior margin of parurohyal and anterior insertion of posterior process. Branchiostegal rays 8 or 9. Vertebrae 39–41. Ribs 13 or 14. Two dorsal hypural plates corresponding to hypurals 3 + 4 + 5; single ventral hypural plate corresponding to hypurals 1 + 2 and parhypural.
Colouration in alcohol: Head and trunk light grey to brown, slightly darker on dorsum, humeral region and area around opercular and interopercular patches of odontodes. Venter white. Fins hyaline with basal portion dark grey to brown. In specimens below about 45 mm SL, dorsal part of flank and dorsum with faint grey dots.
Etymology. From the Latin, ventro (venter) and papillata (with papillae), an allusion to the well-developed papillae on the ventral surface of the head.
Distribution and habitat notes. Cambeva ventropapillata is known from the Rio Itapocu basin, occurring in the upper section of the western and south-western part of the basin (
Figure 3), in altitudes between about 35 and 165 m asl. Habitats were streams with moderate current, with deepest areas reaching about 0.8 m. Specimens of
Cambeva ventropapillata were found among small and medium pebbles (grain sizes ranging from 0.01 to 0.5 m). Larger specimens of the type series were found in association with marginal plants and ravines, in a mixture of pebbles, clayey soil, macrophytes and margin grasses. Larger specimens also showed preference for larger pebbles, when compared to small specimens. Specimens were also often found at the middle of the river channel. In the Rio Treze de Maio, the water was cloudy, indicating a high degree of pollution. In this area, the riparian forests were preserved at some points, anthropized in others, and absent in certain places. Downstream of the type locality, the water was being used for the irrigation of small and medium-sized plantations. Even considering that the basin is under different environmental impacts, the species was not rare, being easily recorded in different areas of the basin.
Remarks. Cambeva ventropapillata was formerly misidentified as T. nigricans by de Pinna (1992), based on four specimens deposited in the Museu de Ciências e Tecnologia, Pontificia Universidade Católica do Rio Grande do Sul, Porto Alegre, Brazil (MCP 10649), which were examined by one of us (WJEMC, 1992). See Costa et al. (2020b) for a historical review about equivocal localities and misidentification of T. nigricans.
3.2.2. Cambeva Gama-Clade
Cambeva biseriata sp. nov.
LSID:urn:lsid:zoobank.org:act:3524D6F0-BD72-49EC-B6F9-AEB7FA6D03C6
Holotype. UFRJ 13265, 70.8 mm SL; Brazil: Santa Catarina State: Joinville Municipality: Rio Piraí, Rio Itapocu basin, Vila Nova, 26°17′34″ S 48°57′51″ W, about 30 m asl; C. R. M. Feltrin, 14 August 2020.
Paratypes. All from Brazil: Santa Catarina State: Rio Itapocu basin. Joinville Municipality. UFRJ 13263, 3 ex. (C&S), 42.5–55.4 mm SL; UFRJ 13264, 2 ex., 39.5–55.4 mm SL; collected with holotype. CICCAA 07548, 3 ex., 34.9–70.8 mm SL; UFRJ 11320, 3 ex. (C&S), 51.7–68.1 mm SL; Rio Mutucas, Rio Piraí drainage, 26°14′38″ S 48°56′48″ W, about 95 m asl; R. Dalcin, 23 January 2016. UFRJ 12305, 8, 61.3–86.4 mm SL; Rio Tateto, 26°15′50″ S 48°58′51″ W, about 90 m asl; R. Dalcin, 18 April 2014. UFRJ 12878, 4, 67.3–75.7 mm SL; same locality and collector as UFRJ 12305, 15 April 2014. UFRJ 12979, 6, 46.1–74.5 mm SL; Jaraguá do Sul Municipality: Rio Itapocuzinho, 26°19′18″ S 49°08′44″ W, about 290 m asl; R. Dalcin, 7 March 2021. Guaramirim Municipality: UFRJ 13266, 6 ex., 36.3–57.1 mm SL; stream tributary to Rio Piraí, Bruderthal, near Pedra Sapo, 26°25′40″ S 48°59′07″ W, about 40 m asl; C. R. M. Feltrin, 6 November 2020. UFRJ 12872, 2 ex. (DNA), 40.6–59.2 mm SL; same locality as UFRJ 13266; C. R. M. Feltrin, 15 April 2022.
Diagnosis. Cambeva biseriata is distinguished from all other congeners of the C. balios-C. tropeira clade by having a unique colour pattern, consisting of two longitudinal rows of small brown rounded spots, one along the lateral midline of body and the other on the dorsal part of flank, overlapped by minute pale brown dots scattered over the entire flank and dorsum (vs. never a similar colour pattern), jaw teeth incisiform (vs. pointed in C. balios, C. duplimaculata, C. longipalata, C. notabilis, C. tropeira, and C. urubici, pointed to sub-incisiform, with rounded extremity, in C. diffusa and C. pericoh), and more dorsal procurrent rays in the caudal fin (18 or 19, vs. 12–17). Cambeva biseriata also differs from C. balios, C. diffusa, C. duplimaculata, C. longipalata, C. notabilis, C. pericoh, C. tropeira and C. urubici by having a large lateral fleshy lobe of the mouth, its largest length occupying about three-fourths of the lower jaw length excluding lobes (vs. about one fourth to one third), and from C. duplimaculata, C. longipalata, C. notabilis, C. tropeira and C. urubici by the absence of the anterior section of the infraorbital canal absent (vs. presence). Cambeva biseriata is also distinguished from all congeners, except C. longipalata, by having a long postero-lateral process of the autopalatine, its length about equal to the autopalatine length (vs. about two thirds). Cambeva biseriata is further distinguished from C. longipalata by having more ventral procurrent rays in the caudal fin (13–15 vs. 10–12) and fewer vertebrae (37–39 vs. 41–42).
Description. General morphology: Morphometric data are presented in
Table 4. Body moderately slender, subcylindrical on anterior region, compressed on posterior region. Greatest body depth in area just anterior to pelvic-fin base. Dorsal and ventral profiles slightly convex between snout and anterior portion of caudal peduncle, nearly straight on caudal peduncle. Anus and urogenital papilla opening at vertical just posterior to dorsal-fin origin. Head sub-trapezoidal in dorsal view, with anterior profile of snout nearly straight. Eye small, dorsally positioned on head, nearer snout tip than posterior margin of opercle. Distance between anterior and posterior nostrils about one-third distance between posterior nostril and orbital rim. Barbels moderate in length, tip of nasal barbel posteriorly reaching area between orbit and preopercle, tip of maxillary and rictal barbels reaching interopercular patch of odontodes, or sometimes rictal barbel reaching area just anterior to interopercular patch of odontodes. Mouth subterminal. Lateral fleshy lobe of the mouth large, its largest length about three-fourths of lower jaw length excluding lobes; ventral surface of lobe slightly concave. Jaw teeth incisiform, irregularly arranged, 48–50 on premaxilla, 47–62 on dentary. Minute skin papillae on ventral surface of head, conspicuous only on area just anterior to branchiostegal region. Branchial membrane attached to isthmus only at its anterior-most point, in ventral midline.
Dorsal and anal fins subtriangular, anterior margin about straight, posterior margin weakly convex, longest ray about equal fin base in length. Total dorsal-fin rays 12 (iii + II + 7), total anal-fin rays 10 (iii + II + 5). Anal-fin origin at vertical through middle portion of dorsal-fin base, at vertical of 4th bifid ray. Dorsal-fin origin at vertical through centrum of 20th or 21st vertebra; anal-fin origin at vertical through centrum of 24th vertebra. Pectoral fin subtriangular in dorsal view, posterior margin convex. First pectoral-fin ray about equal second ray in length, not forming terminal filament. Total pectoral-fin rays 7 (I + 6). Pelvic fin rounded, its posterior extremity at vertical through dorsal-fin origin. Pelvic-fin bases medially in close proximity, almost in contact. Total pelvic-fin rays 5 (I + 4). Caudal fin truncate. Total principal caudal-fin rays 13 (I + 11 + I), total dorsal procurrent rays 17–19 (xvi–xviii + I), total ventral procurrent rays 13–15 (xii–xiv + I).
Laterosensory system: Supraorbital, posterior section of infraorbital canal and postorbital canal continuous. Supraorbital sensory canal pores 3: s1, adjacent to medial margin of anterior nostril; s3, adjacent and just posterior to medial margin of posterior nostril; s6, in transverse line through posterior half of orbit; pore s6 slightly nearer orbit than its paired s6 homologous pore. Anterior infraorbital sensory canal absent. Posterior infraorbital sensory canal pores 2: pore i10, adjacent to ventral margin of orbit, and pore i11, posterior to orbit. Postorbital canal pores 2: po1, at vertical through posterior portion of interopercular patch of odontodes, and po2, at vertical through posterior portion of opercular patch of odontodes. Lateral line pores 2; posterior-most pore at vertical just posterior to pectoral-fin base.
Osteology: Mesethmoid thin, anterior margin straight to slightly convex, main axis tapering posteriorly. Mesethmoid cornu narrow, tip rounded. Lateral surface of lateral ethmoid with pronounced projection posterior to articular connection to autopalatine. Antorbital thin, sub-elliptical, small, about one-fourth of half length of sesamoid supraorbital. Sesamoid supraorbital relatively slender, flat, with small postero-lateral projection, its length about equal autopalatine length. Premaxilla sub-trapezoidal in dorsal view, laterally narrowing, slightly longer than maxilla. Maxilla gently curved. Autopalatine sub-rectangular in dorsal view when excluding its postero-lateral process, its largest width about two three-fourths its length excluding anterior cartilage; medial margin slightly sinuous. Autopalatine posterolateral process long, its length about equal to autopalatine length. Metapterygoid thin, longer than deep. Quadrate slender, dorsal process with constricted base, dorsoposterior margin in contact with hyomandibula outgrowth. Hyomandibula long, with well-developed anterior outgrowth; dorsal margin of hyomandibula outgrowth with shallow concavity. Opercle moderately long, longer than interopercle; opercular odontode patch robust, its width about slightly smaller than dorsal hyomandibula articular facet. Opercular odontodes 15, irregularly arranged, narrow, largest odontodes slightly curved. Dorsal process of opercle short, extremity rounded. Opercular articular facet for hyomandibula with rounded flap, articular facet for preopercle indistinct. Interopercle relatively short, its longitudinal length smaller than hyomandibula outgrow horizontal length; anterior margin of interopercle about straight. Interopercular odontodes 23, nearly straight to slightly curved, pointed, arranged in irregular longitudinal rows, largest odontodes slightly smaller than largest opercular odontodes, irregularly arranged. Preopercle compact, with minute ventral projection. Parurohyal robust, lateral process slightly curved, with subtruncate distal extremity. Parurohyal head well-developed, with prominent anterolateral paired process. Middle parurohyal small, elliptical. Posterior process of parurohyal moderately long, about three-fifths distance between anterior margin of parurohyal and anterior insertion of posterior process. Branchiostegal rays 8 or 9. Vertebrae 37–39. Ribs 13. Two dorsal hypural plates corresponding to hypurals 3 + 4 + 5; single ventral hypural plate corresponding to hypurals 1 + 2 and parhypural.
Colouration in alcohol: Flank and dorsum pale yellow; longitudinal row of small brown rounded spots along lateral midline of body, between humeral region and caudal peduncle, spot diameter about equal opercular patch of odontodes or slightly smaller; longitudinal row of dark brown rounded spots on dorsal part of flank, between nape region and caudal peduncle, spot diameter about four times opercular patch of odontodes or slightly larger; minute pale brown dots scattered over whole flank and dorsum. Dorsal and lateral portion of head pale yellow with dark brown blotches. Venter and ventral surface of head white. Barbels pale yellow with brown margins on dorsal surface, light grey to white in ventral surface. Fins hyaline with brown blotches on basal portion.
Etymology. From de Latin biseriata (with two series), referring to the two longitudinal series of brown spots on the flank that is diagnostic for this new species.
Distribution and ecological notes.
Cambeva biseriata is only known from the northern portion of the Rio Itapocu basin (
Figure 3), in altitudes between about 30 and 290 m asl.
Cambeva biseriata occurs in lotic/semi-lotic environments, closely associated with stony substrate, with granulometries ranging from 0.01 to 0.6 m, and low depths, between 0.5 and 0.8 m. The channel of the Rio Piraí, in Vila Nova, Joinville, has an average width of 21 m, and depths ranging from 5 cm to more than 2 m. The tributary creek of the Rio Piraí has widths that vary between 1.5 and 8.5 m, with depths not exceeding 1 m, prevailing small and medium rolled pebbles. Like other congeners, smaller individuals were distributed preferably in shallow places, with fine gravel and small pebbles about 0.01 to 0.20 in diameter, sometimes associated with marginal vegetation and/or leaf litter deposits. Larger specimens were found below large pebbles, about 0.4 m or more in diameter. In the tributary creek of the Rio Piraí, in Bruderthal, this species is still abundant in places without human intervention, mainly in those with well-preserved riparian forests. Irrigated rice monoculture activities represent, at the sub-basin level of the Rio Piraí, the main threat to
C. biseriata. Such activity promotes the diversion of natural waters from rivers and streams, in addition to an immeasurable load of silting up of clay fines, agricultural defensives, deconfiguration of streams or even total suppression.