Immune and Oxidative Stress Response of the Fish Xyrichthys novacula Infected with the Trematode Ectoparasite Scaphanocephalus sp. in the Balearic Islands
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Recollection
2.2. Molecular Analysis
2.3. Biochemical Analysis
2.4. Statistical Analysis
3. Results
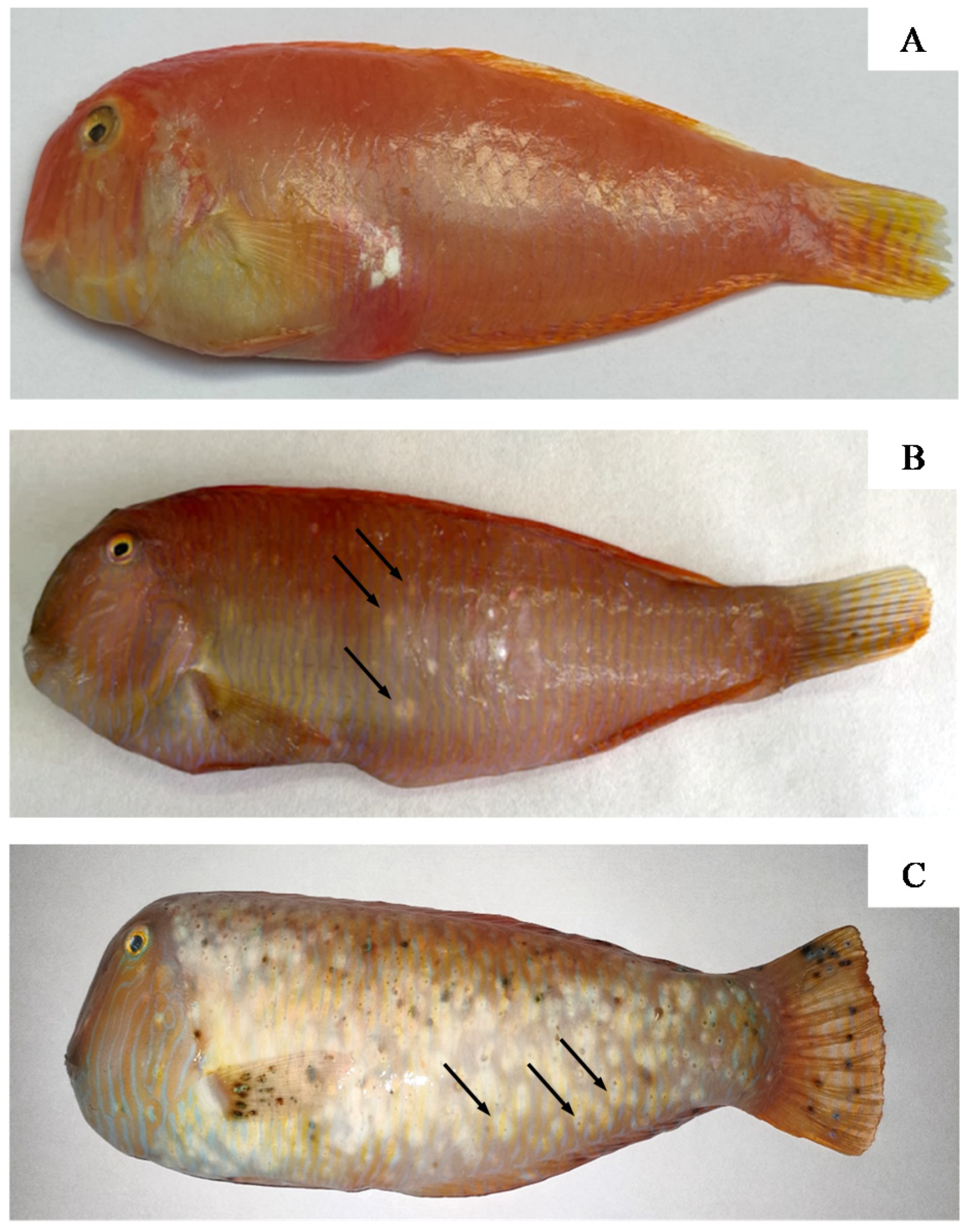
3.1. Fish and Parasite Characteristics
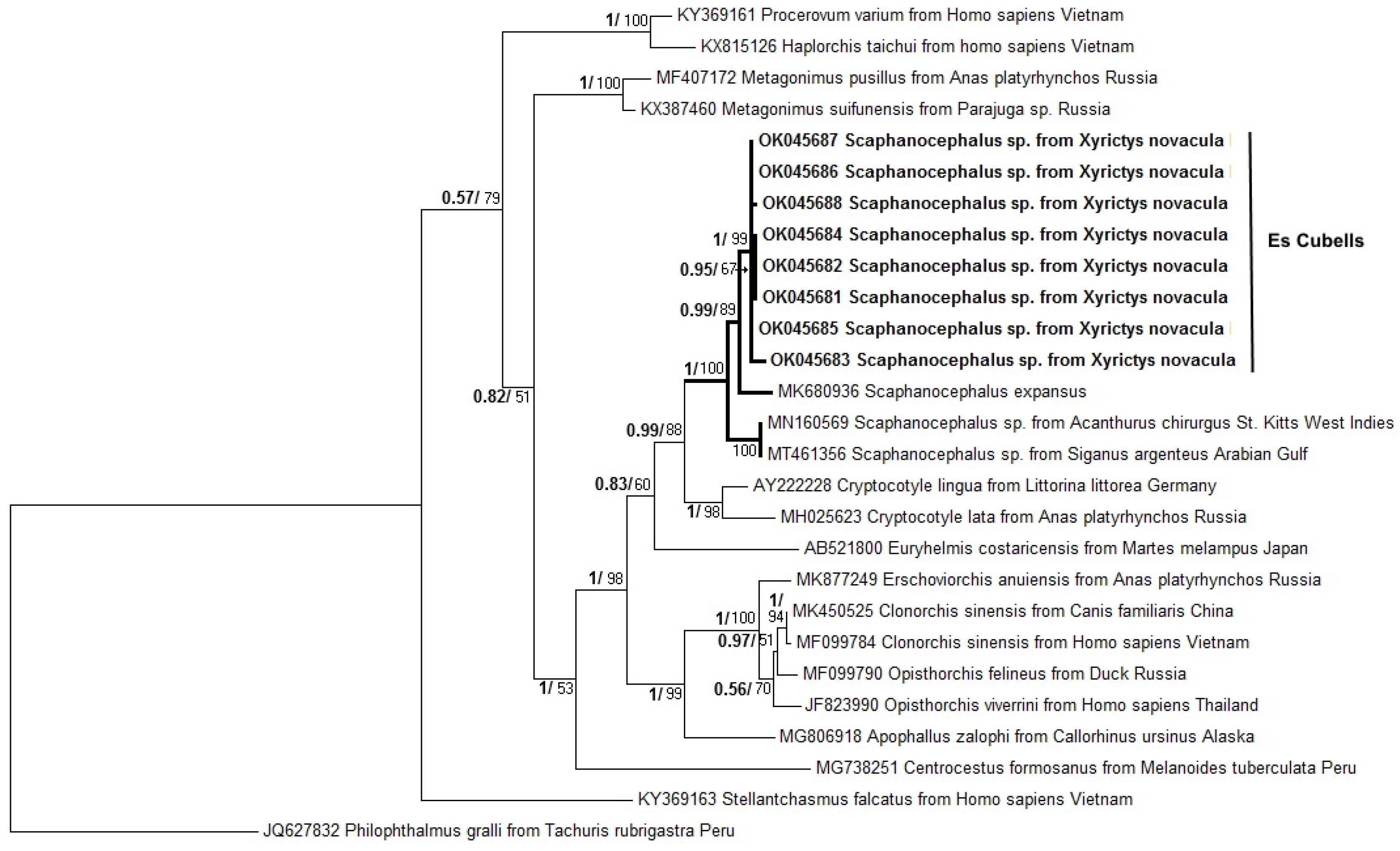
3.2. Molecular Analysis and Phylogenetic Analyses
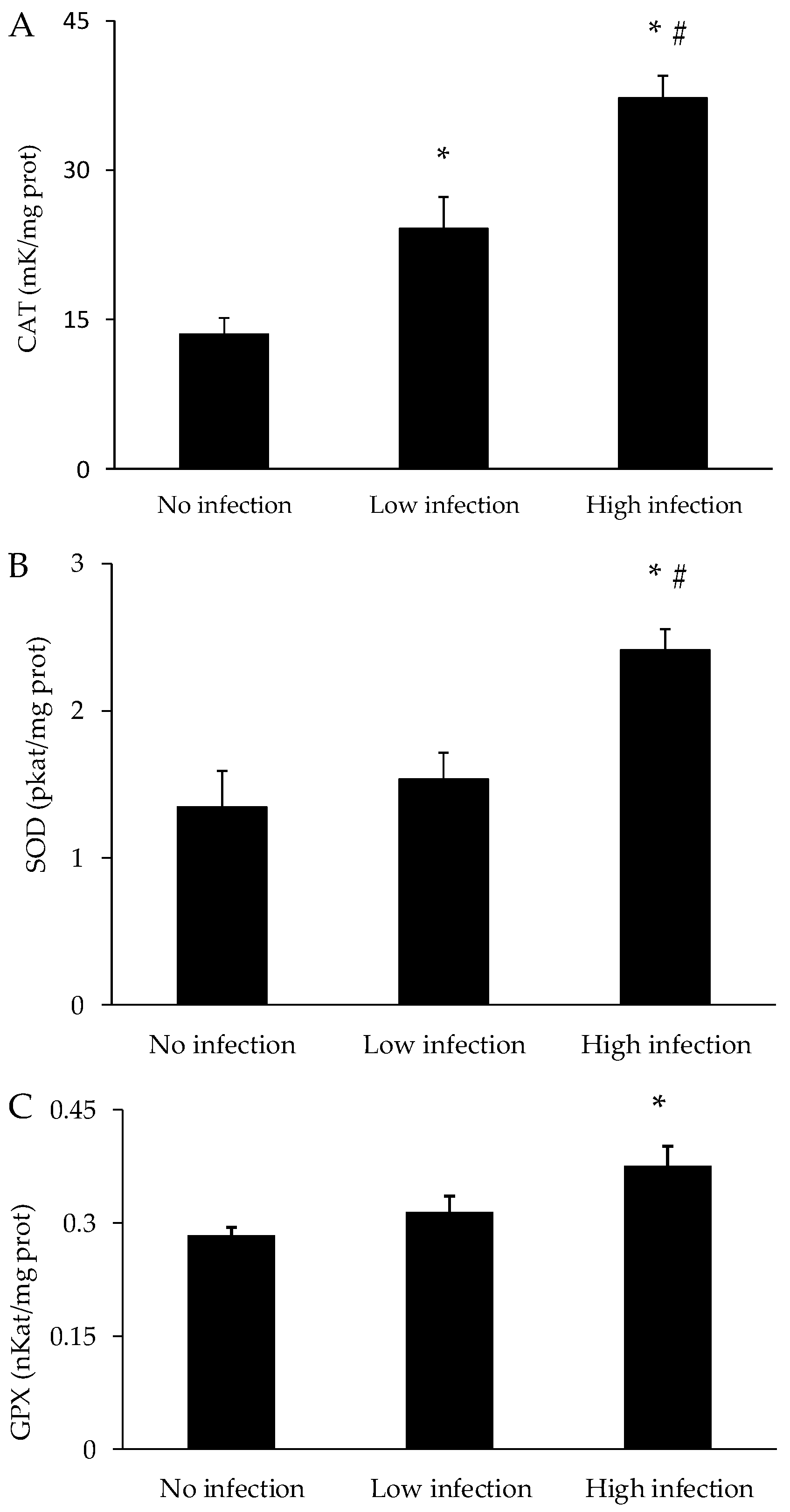
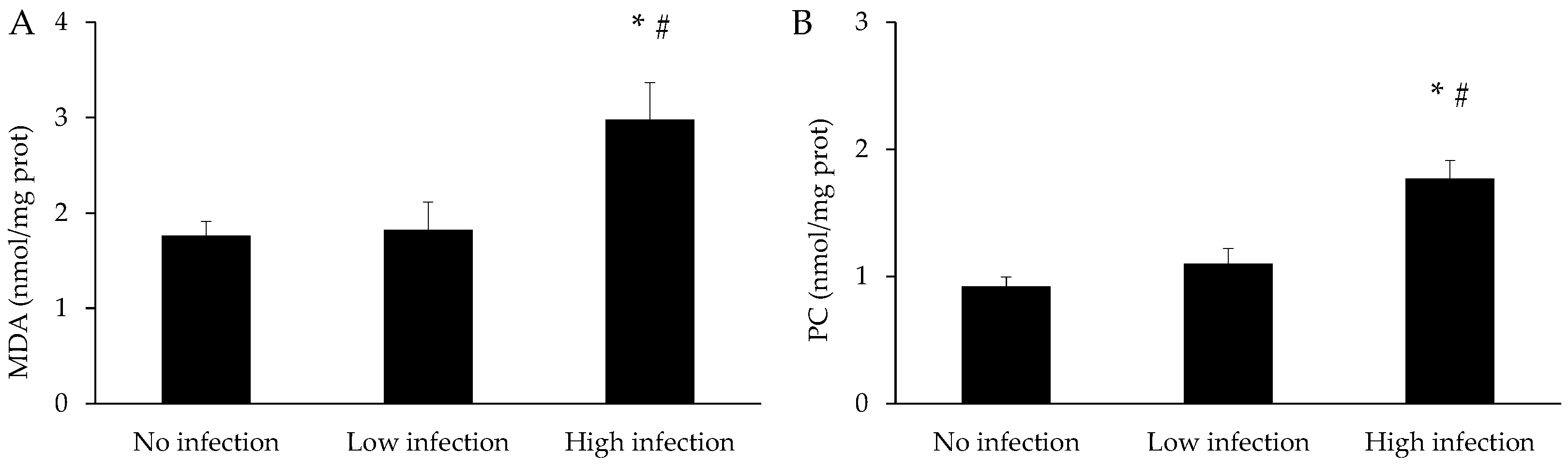
3.3. Oxidative Stress Parameters
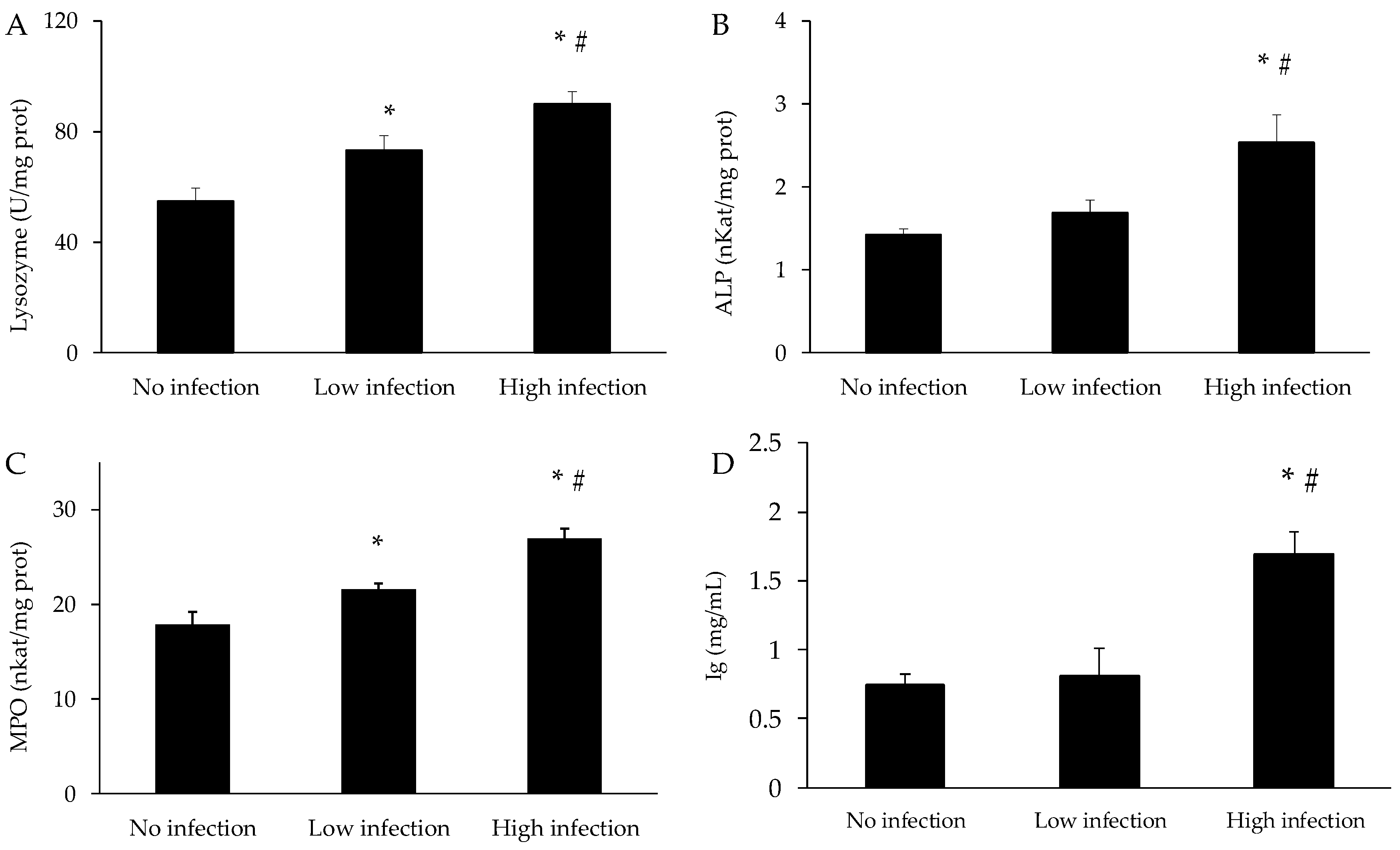
3.4. Immune Parameters
3.5. Bivariate Correlations
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Pyšek, P.; Hulme, P.E.; Simberloff, D.; Bacher, S.; Blackburn, T.M.; Carlton, J.T.; Dawson, W.; Essl, F.; Foxcroft, L.C.; Genovesi, P.; et al. Scientists’ Warning on Invasive Alien Species. Biol. Rev. 2020, 95, 1511–1534. [Google Scholar] [CrossRef]
- Turbelin, A.J.; Malamud, B.D.; Francis, R.A. Mapping the global state of invasive alien species: Patterns of invasion and policy responses. Glob. Ecol. Biogeogr. 2017, 26, 78–92. [Google Scholar] [CrossRef]
- Ulman, A.; Ferrario, J.; Forcada, A.; Seebens, H.; Arvanitidis, C.; Occhipinti-Ambrogi, A.; Marchini, A. Alien Species Spreading via Biofouling on Recreational Vessels in the Mediterranean Sea. J. Appl. Ecol. 2019, 56, 2620–2629. [Google Scholar] [CrossRef]
- Fèvre, E.M.; Bronsvoort, B.M.D.C.; Hamilton, K.A.; Cleaveland, S. Animal movements and the spread of infectious diseases. Trends Microbiol. 2006, 14, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Hulme, P.E. Trade, Transport and Trouble: Managing Invasive Species Pathways in an Era of Globalization. J. Appl. Ecol. 2009, 46, 10–18. [Google Scholar] [CrossRef]
- Pinsky, M.L.; Selden, R.L.; Kitchel, Z.J. Climate-Driven Shifts in Marine Species Ranges: Scaling from Organisms to Communities. Ann. Rev. Mar. Sci. 2020, 12, 153–179. [Google Scholar] [CrossRef] [PubMed]
- Byers, J.E. Marine Parasites and Disease in the Era of Global Climate Change. Ann. Rev. Mar. Sci. 2021, 13, 397–420. [Google Scholar] [CrossRef]
- Marcogliese, D.J. The Impact of Climate Change on the Parasites and Infectious Diseases of Aquatic Animals. OIE Rev. Sci. Tech. 2008, 27, 467–484. [Google Scholar] [CrossRef]
- Pravdová, M.; Kolářová, J.; Grabicová, K.; Randák, T.; Janáč, M.; Kvach, Y.; Jurajda, P.; Ondračková, M. Pharmaceutical Contamination and Biotic Factors Affecting Parasitism in Common Carp (Cyprinus carpio). Aquac. Res. 2022, 53, 4116–4127. [Google Scholar] [CrossRef]
- MacKenzie, K. Parasites as Pollution Indicators in Marine Ecosystems: A Proposed Early Warning System. Mar. Pollut. Bull. 1999, 38, 955–959. [Google Scholar] [CrossRef]
- Nachev, M.; Sures, B. Environmental Parasitology: Parasites as Accumulation Bioindicators in the Marine Environment. J. Sea Res. 2016, 113, 45–50. [Google Scholar] [CrossRef]
- Iu, V.; Kurochkin, L.I.B. The Etiology and Diagnosis of “Black Spot Disease” of Fish. Available online: https://pubmed.ncbi.nlm.nih.gov/8984434/ (accessed on 21 July 2023).
- Foronda, P.; Santana-Morales, M.A.; Feliu, C.; Valladares, B. New record of Scaphanocephalus expansus from the Canary Islands (Spain). Helminthologia 2009, 46, 198–200. [Google Scholar] [CrossRef]
- Kohl, Z.F.; Calhoun, D.M.; Elmer, F.; Peachey, R.B.J.; Leslie, K.L.; Tkach, V.; Kinsella, J.M.; Johnson, P.T.J. Black-spot syndrome in Caribbean fishes linked to trematode parasite infection (Scaphanocephalus expansus). Coral Reefs 2019, 38, 917–930. [Google Scholar] [CrossRef]
- Yao, L.; Wang, C.; Li, G.; Xie, G.; Jia, Y.; Wang, W.; Liu, S.; Xu, T.; Luo, K.; Zhang, Q.; et al. Identification of Fusarium Solani as a Causal Agent of Black Spot Disease (BSD) of Pacific White Shrimp, Penaeus vannamei. Aquaculture 2022, 548, 737602. [Google Scholar] [CrossRef]
- Shimose, T.; Katahira, H.; Kanaiwa, M. Interspecific Variation of Prevalence by Scaphanocephalus (Platyhelminthes: Trematoda: Heterophyidae) Metacercariae in Parrotfishes (Labridae: Scarini) from an Okinawan Coral Reef. Int. J. Parasitol. Parasites Wildl. 2020, 12, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Harrison, E.J.; Hadley, W.F. Possible Effects of Black-Spot Disease on Northern Pike. Available online: https://afspubs.onlinelibrary.wiley.com/doi/abs/10.1577/1548-8659%281982%29111%3C106%3APEOBDO%3E2.0.CO%3B2 (accessed on 21 July 2023).
- Timi, J.T.; Poulin, R. Why ignoring parasites in fish ecology is a mistake. Int. J. Parasitol. 2020, 50, 755–761. [Google Scholar] [CrossRef] [PubMed]
- Clarkson, P.M.; Thompson, H.S. Antioxidants: What role do they play in physical activity and health? Am. J. Clin. Nutr. 2000, 72, 637S–646S. [Google Scholar] [CrossRef]
- Sridhar, A.; Krishnasamy Sekar, R.; Manikandan, D.B.; Arumugam, M.; Veeran, S.; Ramasamy, T. Activity profile of innate immune-related enzymes and bactericidal of freshwater fish epidermal mucus extract at different pH. Environ. Sci. Pollut. Res. 2021, 28, 33914–33926. [Google Scholar] [CrossRef]
- Uribe, C.; Folch, H.; Enriquez, R.; Moran, G. Innate and adaptive immunity in teleost fish: A review. Vet. Med. 2011, 56, 486–503. [Google Scholar] [CrossRef]
- Morand, S.; Harvey, P.H. Mammalian metabolism, longevity and parasite species richness. Proc. Biol. Sci. 2000, 267, 1999–2003. [Google Scholar] [CrossRef]
- Vaquer-Sunyer, R.; Barrientos, N.; Alós, P.; Vivó, T. Abundancia Relativa de Raors (Xyrichthys novacula). Informe Mar Balear 2020. Available online: https://www.informemarbalear.org/es/pesca/imb-raor-esp.pdf (accessed on 3 December 2023).
- Box, A.; Grau, A.M.; Blanco, A.; Riera, F. Els Raors (Xyrichthys novacula) a La Reserva Dels Freus d’Eivissa i Formentera; Efecte de La Protecció Espacial. Bolleti La Soc. D’historia Nat. Les Balear. 2009, 52, 193–201. [Google Scholar]
- Alós, J.; Cabanellas-Reboredo, M.; Lowerre-Barbieri, S. Diel Behaviour and Habitat Utilisation by the Pearly Razorfish during the Spawning Season. Mar. Ecol. Prog. Ser. 2012, 460, 207–220. [Google Scholar] [CrossRef]
- Cohen-Sánchez, A.; Valencia, J.M.; Box, A.; Solomando, A.; Tejada, S.; Pinya, S.; Catanese, G.; Sureda, A. Black Spot Disease Related to a Trematode Ectoparasite Causes Oxidative Stress in Xyrichtys novacula. J. Exp. Mar. Biol. Ecol. 2023, 560, 151854. [Google Scholar] [CrossRef]
- Capó, X.; Alomar, C.; Compa, M.; Sole, M.; Sanahuja, I.; Soliz Rojas, D.L.; González, G.P.; Garcinuño Martínez, R.M.; Deudero, S. Quantification of differential tissue biomarker responses to microplastic ingestion and plasticizer bioaccumulation in aquaculture reared sea bream Sparus aurata. Environ. Res. 2022, 211, 113063. [Google Scholar] [CrossRef]
- Littlewood, D.T.J.; Curini-Galletti, M.; Herniou, E.A. The interrelationships of Proseriata (Platyhelminthes: Seriata) tested with molecules and morphology. Mol. Phylogenet. Evol. 2000, 16, 449–466. [Google Scholar] [CrossRef]
- Olson, P.D.; Cribb, T.H.; Tkach, V.V.; Bray, R.A.; Littlewood, D.T.J. Phylogeny and classification of the Digenea (Platyhelminthes: Trematoda). Int. J. Parasitol. 2003, 33, 733–755. [Google Scholar] [CrossRef] [PubMed]
- Hall, T.A. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Aebi, H. Catalase in Vitro. In Methods in Enzymology; Academic Press: Cambridge, MA, USA, 1984; Volume 105, pp. 121–126. [Google Scholar] [CrossRef]
- Flohé, L.; Ötting, F. Superoxide Dismutase Assays. In Methods in Enzymology; Academic Press: Cambridge, MA, USA, 1984; Volume 105, pp. 93–104. [Google Scholar] [CrossRef]
- Flohé, L.; Günzler, W.A. Assays of Glutathione Peroxidase. In Methods in Enzymology; Academic Press: Cambridge, MA, USA, 1984; Volume 105, pp. 114–120. [Google Scholar] [CrossRef]
- Capeillère-Blandin, C. Oxidation of Guaiacol by Myeloperoxidase: A Two-Electron-Oxidized Guaiacol Transient Species as a Mediator of NADPH Oxidation. Biochem. J. 1998, 336 Pt 2, 395–404. [Google Scholar] [CrossRef]
- Ross, N.W.; Firth, K.J.; Wang, A.; Burka, J.F.; Johnson, S.C. Changes in Hydrolytic Enzyme Activities of Naive Atlantic Salmon Salmo Salar Skin Mucus Due to Infection with the Salmon Louse Lepeophtheirus Salmonis and Cortisol Implantation. Dis. Aquat. Organ. 2000, 41, 43–51. [Google Scholar] [CrossRef]
- Lee, Y.C.; Yang, D. Determination of Lysozyme Activities in a Microplate Format. Anal. Biochem. 2002, 310, 223–224. [Google Scholar] [CrossRef] [PubMed]
- Milla, S.; Mathieu, C.; Wang, N.; Lambert, S.; Nadzialek, S.; Massart, S.; Henrotte, E.; Douxfils, J.; Mélard, C.; Mandiki, S.N.M.; et al. Spleen Immune Status Is Affected after Acute Handling Stress but Not Regulated by Cortisol in Eurasian Perch, Perca fluviatilis. Fish Shellfish Immunol. 2010, 28, 931–941. [Google Scholar] [CrossRef] [PubMed]
- Levine, R.L.; Williams, J.A.; Stadtman, E.P.; Shacter, E. Carbonyl assay for determination of oxidatively modified proteins. Methods. Enzymol. 1994, 233, 246–257. [Google Scholar] [CrossRef]
- Mcmichael, A.J.; Campbell-Lendrum, D.H.; Corvalán, C.F.; Ebi, K.L.; Githeko, A.; Scheraga, J.D.; Woodward, A. (Eds.) Climate Change and Human Health—Risks and Responses; World Health Organization: Geneva, Switzerland, 2003. [Google Scholar]
- Pastor, F.; Valiente, J.A.; Khodayar, S. A warming Mediterranean: 38 years of increasing sea surface temperature. Remote Sens. 2020, 12, 2687. [Google Scholar] [CrossRef]
- Juza, M.; Fernández-Mora, À.; Tintoré, J. Sub-Regional marine heat waves in the Mediterranean Sea from observations: Long-term surface changes, Sub-surface and coastal responses. Front. Mar. Sci. 2022, 9, 785771. [Google Scholar] [CrossRef]
- Cabanellas-Reboredo, M.; Vázquez-Luis, M.; Mourre, B.; Álvarez, E.; Deudero, S.; Amores, Á.; Addis, P.; Ballesteros, E.; Barrajón, A.; Coppa, S.; et al. Tracking a mass mortality outbreak of pen shell Pinna nobilis populations: A collaborative effort of scientists and citizens. Sci. Rep. 2019, 9, 13355. [Google Scholar] [CrossRef] [PubMed]
- Kaukas, A.; Rollinson, D. Interspecific Variation within the “hypervariable” Region of the 18S Ribosomal RNA Gene among Species of Schistosoma Weinland, 1858 (Digenea). Syst. Parasitol. 1997, 36, 157–160. [Google Scholar] [CrossRef]
- Noguera, P.A.; Feist, S.W.; Bateman, K.S.; Lang, T.; Grütjen, F.; Bruno, D.W. Hyperpigmentation in North Sea dab Limanda limanda. II. Macroscopic and microscopic characteristics and pathogen screening. Dis. Aquat. Organ. 2013, 103, 25–34. [Google Scholar] [CrossRef]
- Al-Salem, A.A.M.; Baghdadi, H.B.; Mahmoud, M.A.; Ibrahim, M.; Bayoumy, E.M. Morphomolecular and Pathological Study of Scaphanocephalus Sp. in New Host Siganus Argenteus in the Arabian Gulf. Dis. Aquat. Organ. 2021, 144, 221–230. [Google Scholar] [CrossRef]
- Culurgioni, J.; De Murtas, R.; Cannella, S.; Figus, V. Parasites of Wild European Sea Bass Dicentrarchus labrax (Linnaeus, 1758) from St. Gilla Lagoon (Sardinia, South Western Mediterranean). Ittiopatologia 2010, 7, 123–133. [Google Scholar]
- Châari, M.; Neifar, L. Parasitic Infections in the Mediterranean Needlefish Tylosurus acus imperialis (Teleostei: Belonidae) off Tunisian Coast. J. Coast. Life Med. 2015, 3, 673–676. [Google Scholar] [CrossRef]
- Lymbery, A.J.; Morine, M.; Kanani, H.G.; Beatty, S.J.; Morgan, D.L. Co-Invaders: The Effects of Alien Parasites on Native Hosts. Int. J. Parasitol. Parasites Wildl. 2014, 3, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Sures, B.; Nachev, M. Effects of Multiple Stressors in Fish: How Parasites and Contaminants Interact. Parasitology 2022, 149, 1822–1828. [Google Scholar] [CrossRef] [PubMed]
- Espinosa-Ruíz, C.; Esteban, M.Á. Wound-Induced Changes in Antioxidant Enzyme Activities in Skin Mucus and in Gene Expression in the Skin of Gilthead Seabream (Sparus aurata L.). Fishes 2021, 6, 15. [Google Scholar] [CrossRef]
- Dzul-Caamal, R.; Salazar-Coria, L.; Olivares-Rubio, H.F.; Rocha-Gómez, M.A.; Girón-Pérez, M.I.; Vega-López, A. Oxidative Stress Response in the Skin Mucus Layer of Goodea Gracilis (Hubbs and Turner, 1939) Exposed to Crude Oil: A Non-Invasive Approach. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2016, 200, 9–20. [Google Scholar] [CrossRef]
- Kar, B.; Mohanty, J.; Hemaprasanth, K.P.; Sahoo, P.K. The Immune Response in Rohu, Labeo rohita (Actinopterygii: Cyprinidae) to Argulus siamensis (Branchiura: Argulidae) Infection: Kinetics of Immune Gene Expression and Innate Immune Response. Aquac. Res. 2015, 46, 1292–1308. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhou, X.Q.; Jiang, W.D.; Wu, P.; Liu, Y.; Ren, H.M.; Jin, X.W.; Feng, L. Vitamin D Promotes Mucosal Barrier System of Fish Skin Infected with Aeromonas hydrophila through Multiple Modulation of Physical and Immune Protective Capacity. Int. J. Mol. Sci. 2023, 24, 11243. [Google Scholar] [CrossRef]
- Shephard, K.L. Functions for Fish Mucus. Rev. Fish Biol. Fish. 1994, 4, 401–429. [Google Scholar] [CrossRef]
- Guardiola, F.A.; Cuesta, A.; Arizcun, M.; Meseguer, J.; Esteban, M.A. Comparative Skin Mucus and Serum Humoral Defence Mechanisms in the Teleost Gilthead Seabream (Sparus aurata). Fish Shellfish Immunol. 2014, 36, 545–551. [Google Scholar] [CrossRef]
- Kumar, S.; Raman, R.P.; Prasad, K.P.; Srivastava, P.P.; Kumar, S.; Rajendran, K.V. Modulation of Innate Immune Responses and Induction of Oxidative Stress Biomarkers in Pangasianodon Hypophthalmus Following an Experimental Infection with Dactylogyrid monogeneans. Fish Shellfish Immunol. 2017, 63, 334–343. [Google Scholar] [CrossRef]
- Kaur, N.; Kumar, R.; Kamilya, D. Modulation of Systemic and Mucosal Immune Responses of Catla catla (Hamilton, 1822) Experimentally Challenged with Gill Monogeneans. Fish Shellfish Immunol. 2018, 74, 567–572. [Google Scholar] [CrossRef] [PubMed]
- Yin, F.; Dan, X.M.; Sun, P.; Shi, Z.H.; Gao, Q.X.; Peng, S.M.; Li, A.X. Growth, Feed Intake and Immune Responses of Orange-Spotted Grouper (Epinephelus coioides) Exposed to Low Infectious Doses of Ectoparasite (Cryptocaryon irritans). Fish Shellfish Immunol. 2014, 36, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Yin, F.; Gong, H.; Ke, Q.; Li, A. Stress, Antioxidant Defence and Mucosal Immune Responses of the Large Yellow Croaker Pseudosciaena crocea Challenged with Cryptocaryon irritans. Fish Shellfish Immunol. 2015, 47, 344–351. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.; Yi, M.; Xiao, P.; Meng, L.; Li, X.; Sun, G.; Liu, Y. The Impact of Aeromonas salmonicida Infection on Innate Immune Parameters of Atlantic Salmon (Salmo salar L). Fish Shellfish Immunol. 2015, 44, 307–315. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, D.; Xie, J.; Chang, O.; Wang, Q.; Shi, C.; Zhao, F.; Gong, H.; Ren, Y.; Musa, N.; et al. Do ectoparasites on fish gills “talk” with gut microbiota far away? Aquaculture 2023, 562, 738880. [Google Scholar] [CrossRef]
- De Vico, G.; Cataldi, M.; Carella, F.; Marino, F.; Passantino, A. Histological, histochemical and morphometric changes of splenic melanomacrophage centers (SMMCs) in Sparicotyle-infected cultured sea breams (Sparus aurata). Immunopharmacol. Immunotoxicol. 2008, 30, 27–35. [Google Scholar] [CrossRef]



| Kc | CAT | SOD | GPX | MDA | PC | LYZ | ALP | MPO | Ig | ||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Spots | r | −0.472 ** | 0.630 ** | 0.635 ** | 0.575 ** | 0.524 ** | 0.809 ** | 0.589 ** | 0.379 * | 0.684 ** | 0.529 ** |
| p | 0.010 | 0.000 | 0.000 | 0.001 | 0.004 | 0.000 | 0.001 | 0.043 | 0.000 | 0.003 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cohen-Sánchez, A.; Sánchez-Mairata, A.G.; Valencia, J.M.; Box, A.; Pinya, S.; Tejada, S.; Sureda, A. Immune and Oxidative Stress Response of the Fish Xyrichthys novacula Infected with the Trematode Ectoparasite Scaphanocephalus sp. in the Balearic Islands. Fishes 2023, 8, 600. https://doi.org/10.3390/fishes8120600
Cohen-Sánchez A, Sánchez-Mairata AG, Valencia JM, Box A, Pinya S, Tejada S, Sureda A. Immune and Oxidative Stress Response of the Fish Xyrichthys novacula Infected with the Trematode Ectoparasite Scaphanocephalus sp. in the Balearic Islands. Fishes. 2023; 8(12):600. https://doi.org/10.3390/fishes8120600
Chicago/Turabian StyleCohen-Sánchez, Amanda, Antoni Gabriel Sánchez-Mairata, José María Valencia, Antonio Box, Samuel Pinya, Silvia Tejada, and Antoni Sureda. 2023. "Immune and Oxidative Stress Response of the Fish Xyrichthys novacula Infected with the Trematode Ectoparasite Scaphanocephalus sp. in the Balearic Islands" Fishes 8, no. 12: 600. https://doi.org/10.3390/fishes8120600
APA StyleCohen-Sánchez, A., Sánchez-Mairata, A. G., Valencia, J. M., Box, A., Pinya, S., Tejada, S., & Sureda, A. (2023). Immune and Oxidative Stress Response of the Fish Xyrichthys novacula Infected with the Trematode Ectoparasite Scaphanocephalus sp. in the Balearic Islands. Fishes, 8(12), 600. https://doi.org/10.3390/fishes8120600







