Genetic Diversity and Population Structure of the Giant Mottled Eel, Anguilla marmorata Quoy & Gaimard, 1824 in Central Vietnam
Abstract
1. Introduction
2. Materials and Methods
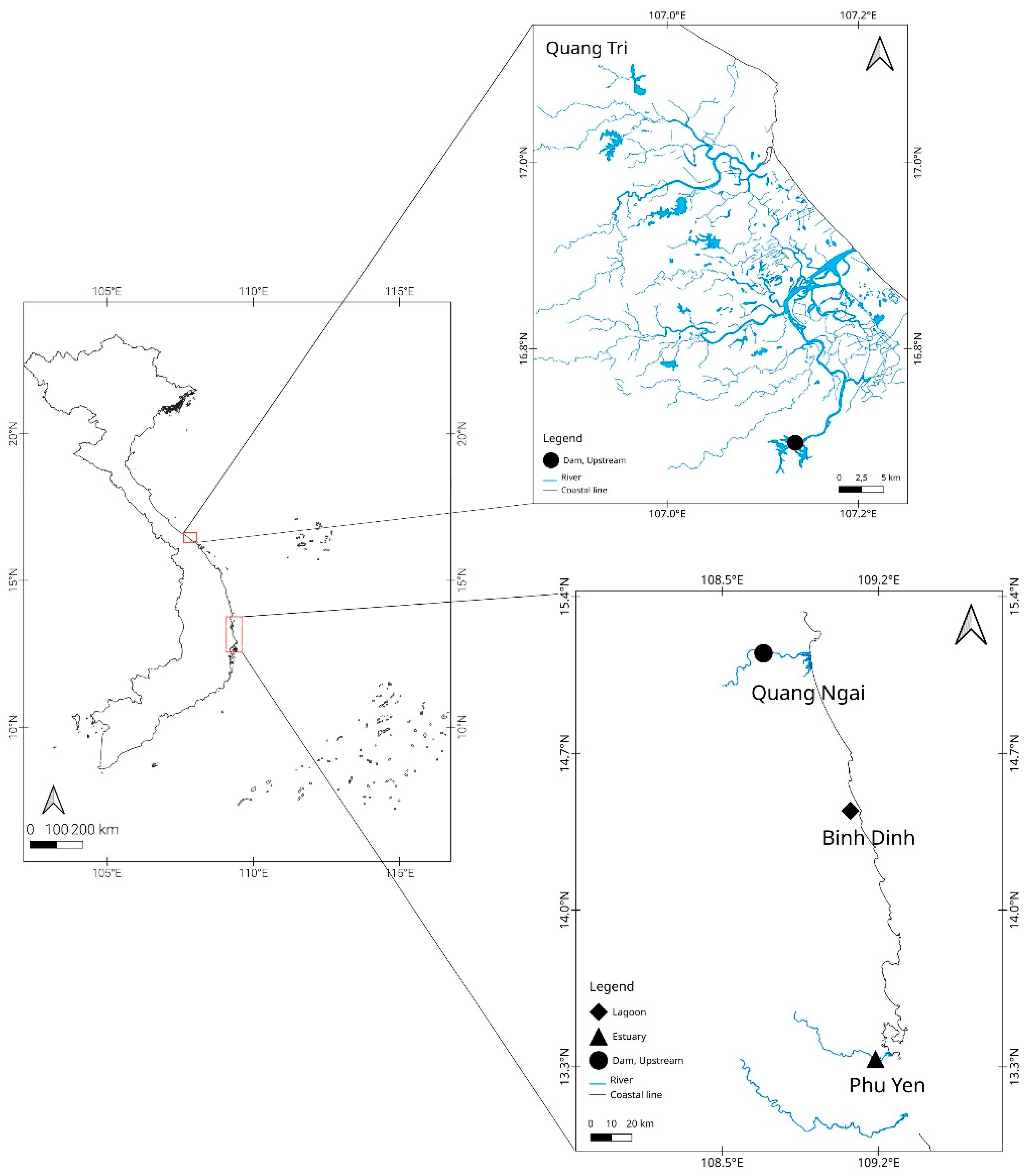
2.1. Sampling Location
2.2. Sampling Protocols
2.3. DNA Extraction and Amplification
2.4. Sequencing Analysis
3. Results
3.1. Genetic Diversity
3.2. Population Genetic Structure
3.3. The Genetic Relationship between the A. marmorata Population in Vietnam and Other Populations
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ege, V. A revision of the genus Anguilla Shaw: A systematic, phylogenetic and geographical study. Dana Rep. 1939, 16, 1–256. [Google Scholar]
- Kuroki, M.; Aoyama, J.; Miller, M.; Yoshinaga, T.; Shinoda, A.; Hagihara, S.; Tsukamoto, K. Sympatric spawning of Anguilla marmorata and Anguilla japonica in the western North Pacific Ocean. J. Fish Biol. 2009, 74, 1853–1865. [Google Scholar] [CrossRef]
- Ishikawa, S.; Tsukamoto, K.; Nishida, M. Genetic evidence for multiple geographic populations of the giant mottled eel Anguilla marmorata in the Pacific and Indian oceans. Ichthyol. Res. 2004, 51, 343–353. [Google Scholar] [CrossRef]
- Minegishi, Y.; Aoyama, J.; Tsukamoto, K. Multiple population structure of the giant mottled eel, Anguilla marmorata. Mol. Ecol. 2008, 17, 3109–3122. [Google Scholar] [CrossRef] [PubMed]
- Tabeta, O.; Tanimoto, T.; Takai, T.; Matsui, I.; Imamura, T. Seasonal occurrence of anguillid elvers in Cagayan River, Luzon Island, the Philippines. Bull. Jpn. Soc. Sci. Fish 1976, 42, 421–426. [Google Scholar] [CrossRef]
- Budimawan. The early life history of the tropical eel Anguilla marmorata (Quog & Gaimard, 1824) from four Pacific estuaries, as revealed from otolith microstructural analysis. J. Appl. Ichthyol. 1997, 13, 57–62. [Google Scholar]
- Arai, T.; Aoyama, J.; Limbong, D.; Tsukamoto, K. Species composition and inshore migration of the tropical eels Anguilla spp. recruiting to the estuary of the Poigar River, Sulawesi Island. Mar. Ecol. Prog. Ser. 1999, 188, 299–303. [Google Scholar] [CrossRef]
- Arai, T.; Limbong, D.; Otake, T.; Tsukamoto, K. Metamorphosis and inshore migration of tropical eels Anguilla spp. in the Indo-Pacific. Mar. Ecol. Prog. Ser. 1999, 182, 283–293. [Google Scholar] [CrossRef]
- Arai, T.; Limbong, D.; Otake, T.; Tsukamoto, K. Recruitment mechanisms of tropical eels Anguilla spp. and implications for the evolution of oceanic migration in the genus Anguilla. Mar. Ecol. Prog. Ser. 2001, 216, 253–264. [Google Scholar] [CrossRef]
- Marui, M.; Arai, T.; Miller, M.J.; Jellyman, D.J.; Tsukamoto, K. Comparison of early life history between New Zealand temperate eels and Pacific tropical eels revealed by otolith microstructure and microchemistry. Mar. Ecol. Prog. Ser. 2001, 213, 273–284. [Google Scholar] [CrossRef]
- Arai, T. Do we protect freshwater eels or do we drive them to extinction? SpringerPlus 2014, 3, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Arai, T. How have spawning ground investigations of the Japanese eel Anguilla japonica contributed to the stock enhancement? Rev. Fish Biol. Fish 2014, 24, 75–88. [Google Scholar] [CrossRef]
- Dekker, W. Worldwide decline of eel resources necessitates immediate action: Quebec declaration of concern. Fisheries 2003, 28, 28–30. [Google Scholar]
- Jacoby, D.M.; Casselman, J.M.; Crook, V.; DeLucia, M.B.; Ahn, H.; Kaifu, K.; Kurwie, T.; Sasal, P.; Silfvergrip, A.M.C.; Smith, K.G.; et al. Synergistic patterns of threat and the challenges facing global anguillid eel conservation. Glob. Ecol. Conserv. 2015, 4, 321–333. [Google Scholar] [CrossRef]
- Marini, M.; Pedrosa-Gerasmio, I.R.; Santos, M.D.; Shibuno, T.; Daryani, A.; Romana-Eguia, M.R.R.; Wibowo, A. Genetic diversity, population structure and demographic history of the tropical eel Anguilla bicolor pacifica in Southeast Asia using mitochondrial DNA control region sequences. Glob. Ecol. ConserV. 2021, 26, e01493. [Google Scholar] [CrossRef]
- Robinet, T.; Guyet, S.; Marquet, G.; Mounaix, B.; Olivier, J.M.; Tsukamoto, K.; Pierre, V.; Feunteun, E. Elver invasion, population structure and growth of marbled eels Anguilla marmorata in a tropical river on Réunion Island in the Indian Ocean. Environ. Biol. Fishes 2003, 68, 339–348. [Google Scholar] [CrossRef]
- Maes, G.E.; Pujolar, J.; Hellemans, B.; Volckaert, F.A. Evidence for isolation by time in the European eel (Anguilla anguilla L.). Mol. Ecol. 2006, 15, 2095–2107. [Google Scholar] [CrossRef] [PubMed]
- Gagnaire, P.A.; Minegishi, Y.; Zenboudji, S.; Valade, P.; Aoyama, J.; Berrebi, P. Within-population structure highlighted by differential introgression across semipermeable barriers to gene flow in Anguilla marmorata. Evol. Int. J. Org. Evol. 2011, 65, 3413–3427. [Google Scholar] [CrossRef] [PubMed]
- Minegishi, Y.; Gagnaire, P.A.; Aoyama, J.; Bosc, P.; Feunteun, E.; Tsukamoto, K.; Berrebi, P. Present and past genetic connectivity of the Indo-Pacific tropical eel Anguilla bicolor. J. Biogeogr. 2012, 39, 408–420. [Google Scholar] [CrossRef]
- Fahmi, M.R.; Solihin, D.D.; Shao, Z.; Pouyaud, L.; Berrebi, P. Population genetic structure of the tropical eel Anguilla bicolor in Indonesian waters based on microsatellite markers. Folia Zool. 2015, 64, 87–96. [Google Scholar] [CrossRef]
- Watanabe, S.; Aoyama, J.; Nishida, M.; Tsukamoto, K. Evaluation of the population structure of Anguilla bicolor bicolor using total number of vertebrae and the mtDNA control region. Coast. Mar. Sci. 2005, 29, 165–169. [Google Scholar]
- Gubili, C.; Schabetsberger, R.; Poellabauer, C.; Bates, B.; Wagstaff, R.M.; Woodward, L.M.; Sichrowsky, U.; Scheck, A.; Boseto, D.T.; Feunteun, E.; et al. High genetic diversity and lack of pronounced population structure in five species of sympatric Pacific eels. Fish. Manag. Ecol. 2019, 26, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Aoyama, J. Life history and evolution of migration in catadromous eels (Genus Anguilla). Aqua-BioSci. Monogr. 2009, 2, 1–42. [Google Scholar] [CrossRef]
- Lutaenko, K.A. Coastal marine biodiversity of Vietnam (South China Sea), its current problems and major threats. In Proceedings of the Russia-China Bilateral Workshop, Valadivostok, Russia, 10–11 October 2019. [Google Scholar]
- Nguyen, A.T.; Tsukamoto, K.; Lokman, P.M. Composition and distribution of freshwater eels Anguilla spp. in Vietnam. Fish Sci. 2018, 84, 987–994. [Google Scholar] [CrossRef]
- Tuan, N.T.; Duat, H.V. An overview of the Anguillid eel culture in Vietnam. J. Aquac. Mar. Biol. 2021, 10, 96–101. [Google Scholar] [CrossRef]
- Chen, C.A.; Ablan, M.C.A.; McManus, J.W.; Diepernk Bell, J.; Tuan, V.S.; Cabanban, A.S.; Shao, K.T. Population structure and genetic variability of six bar wrasse (Thallasoma hardwicki) in northern South China Sea revealed by mitochondrial control region sequences. Mar. Biotechnol. 2004, 6, 312–326. [Google Scholar] [CrossRef]
- Adamkewicz, S.; Harasewych, M. Systematics and biogeography of the genus Donax (Bivalvia: Donacidae) in eastern North America. Am. Malacol. Bull. 1996, 13, 97–103. [Google Scholar]
- Hall, T. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Rozas, J.; Ferrer-Mata, A.; Sánchez-DelBarrio, J.C.; Guirao-Rico, S.; Librado, P.; Ramos-Onsins, S.E.; Sánchez-Gracia, A. DnaSP 6: DNA sequence polymorphism analysis of large data sets. Mol. Biol. Evol. 2017, 34, 3299–3302. [Google Scholar] [CrossRef]
- Fu, Y.X. Statistical tests of neutrality of mutations against population growth, hitchhiking and background selection. Genetics 1997, 147, 915–925. [Google Scholar] [CrossRef]
- Tajima, F. Statistical method for testing the neutral mutation hypothesis by DNA polymorphism. Genetics 1989, 123, 585–595. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Soriano, A.; Ramos-Onsins, S.E.; Rozas, J.; Calafell, F.; Navarro, A. Statistical power analysis of neutrality tests under demographic expansions, contractions and bottlenecks with recombination. Genetics 2008, 179, 555–567. [Google Scholar] [CrossRef] [PubMed]
- Excoffier, L.; Lischer, H.E. Arlequin suite ver 3.5: A new series of programs to perform population genetics analyses under Linux and Windows. Mol. Ecol. Resour. 2010, 10, 564–567. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C. MUSCLE: A multiple sequence alignment method with reduced time and space complexity. BMC Bioinform. 2004, 5, 1–19. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547. [Google Scholar] [CrossRef]
- Honda, S.; Muthmainnah, D.; Suryati, N.K. Exchanging Information on Catadromous Eels in South East Asia. SEAFDEC-Newsletter 2016, 39, 8–9. [Google Scholar]
- Moritz, C.; Dowling, T.; Brown, W. Evolution of animal mitochondrial DNA: Relevance for population biology and systematics. Annu. Rev. Ecol. Evol. Syst. 1987, 18, 269–292. [Google Scholar] [CrossRef]
- Stoneking, M.; Hedgecock, D.; Higuchi, R.G.; Vigilant, L.; Erlich, H.A. Population variation of human mtDNA control region sequences detected by enzymatic amplification and sequence-specific oligonucleotide probes. Am. J. Hum. Genet. 1991, 48, 370. [Google Scholar]
- Avise, J. Molecular Markers, Natural History and Evolution, 2nd ed.; Sinauer & Associates: Sunderland, MA, USA, 2004. [Google Scholar]
- Frankham, R.; Ballou, S.E.J.D.; Briscoe, D.A.; Ballou, J.D. Introduction to Conservation Genetics; Cambridge University Press: Cambridge, UK, 2002. [Google Scholar]
- Zhang, D.X.; Hewitt, G.M. Nuclear DNA analyses in genetic studies of populations: Practice, problems and prospects. Mol. Ecol. 2003, 12, 563–584. [Google Scholar] [CrossRef]
- Miller, M.J.; Mochioka, N.; Otake, T.; Tsukamoto, K. Evidence of a spawning area of Anguilla marmorata in the western North Pacific. Mar. Biol. 2002, 140, 809–814. [Google Scholar]
- Tsukamoto, K.; Chow, S.; Otake, T.; Kurogi, H.; Mochioka, N.; Miller, M.J.; Aoyama, J.; Kimura, S.; Watanabe, S.; Yoshinaga, T.; et al. Oceanic spawning ecology of freshwater eels in the western North Pacific. Nat. Commun. 2011, 2, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.S.; Yambot, A.V.; Zhang, H.; Hung, C.L. Sympatric spawning but allopatric distribution of Anguilla japonica and Anguilla marmorata: Temperature-and oceanic current-dependent sieving. PLoS ONE 2012, 7, e37484. [Google Scholar] [CrossRef] [PubMed]
- Wakiya, R.; Itakura, H.; Kaifu, K. Age, growth and sex ratios of the giant mottled eel, Anguilla marmorata, in freshwater habitats near its northern geographic limit: A comparison to tropical regions. Zool. Stud. 2019, 58, 34. [Google Scholar]
- Hu, J.; Kawamura, H.; Hong, H.; Qi, Y. A review on the currents in the South China Sea: Seasonal circulation, South China Sea warm current and Kuroshio intrusion. J. Oceanogr. 2000, 56, 607–624. [Google Scholar] [CrossRef]
- Xue, H.; Chai, F.; Pettigrew, N.; Xu, D.; Shi, M.; Xu, J. Kuroshio intrusion and the circulation in the South China Sea. J. Geophys. Res. Oceans 2004, 109, 1978–2012. [Google Scholar] [CrossRef]
- Arai, T.; Taha, H.; Mohd-Riduan, M.N.; Mokti, S.S.A. Molecular and morphological evidence for the identity of the giant mottled eel, Anguilla marmorata in Southeast Asia. Trop. Ecol. 2020, 61, 429–436. [Google Scholar] [CrossRef]
- Kuroki, M.; Miller, I.J.; Feunteun, E.; Sasal, P.; Pikering, T.; Han, Y.S.; Faliex, E.; Acou, A.; Dessier, A.; Schabetsberger, R.; et al. Distribution of anguillid leptocephali and possible spawning areas in the South Pacific Ocean. Prog. Oceanogr. 2020, 180, 102234. [Google Scholar] [CrossRef]
- Schabetsberger, R.; Økland, F.; Aarestrup, K.; Kalfatak, D.; Sichrowsky, U.; Tambets, M.; Dall’Olmo, G.; Kaiser, R.; Miller, P.I. Oceanic migration behaviour of tropical Pacific eels from Vanuatu. Mar. Ecol. Prog. Ser. 2013, 475, 177–190. [Google Scholar] [CrossRef]
- Lee, T.; Fournier, S.; Gordon, A.L.; Sprintall, J. Maritime Continent water cycle regulates low-latitude chokepoint of global ocean circulation. Nat. Commun. 2019, 10, 2103. [Google Scholar] [CrossRef]
- Pattiaratchi, C.B.; Siji, P. Variability in Ocean Current Around Australia. 2020. Available online: https://www.imosoceanreport.org.au/time-series/environment/current-variability/ (accessed on 29 June 2022).
- Ganachaud, A.; Cravatte, S.; Melet, A.; Schiller, A.; Holbrook, N.J.; Sloyan, B.M.; Widlansky, M.J.; Bowen, M.J.; Verron, J.; Wiles, P.; et al. The Southwest Pacific Ocean circulation and climate experiment (SPICE). J. Geophys. Res. Oceans 2014, 119, 7660–7686. [Google Scholar] [CrossRef]
- Kessler, W.S.; Cravatte, S. Mean circulation of Coral Sea. J. Geophys. Res. Oceans 2013, 118, 6385–6410. [Google Scholar] [CrossRef]
- Gordon, A.L.; Susanto, R.D.; Vranes, K. Cool Indonesian Throughflow as a consequence of restricted surface layer flow. Nature 2003, 425, 824–828. [Google Scholar] [CrossRef] [PubMed]
- Susanto, R.D.; Fang, G.; Soesilo, I.; Zheng, Q.; Qiao, F.; Wei, Z.; Sulistyo, B. New surveys of a branch of the Indonesian throughflow. Eos 2010, 91, 261–263. [Google Scholar] [CrossRef]
- Susanto, R.D.; Wei, Z.; Adi, A.T.; Fan, B.; Li, S.; Fang, G. Karimata throughflow from December 2007 to November 2008. Acta Oceanol. Sin. 2013, 32, 1–6. [Google Scholar] [CrossRef]
- Susanto, R.D.Z.; Wei, T.R.; Adi, Q.; Zheng, G.; Fang, B.; Fan, A.; Supangat, T.; Agustiadi, S.; Li, M.; Trenggono, M.; et al. Oceanography Surrounding Krakatau Volcano. Oceanography 2016, I29, 264–272. [Google Scholar]
- Dao, H.T.; Smith-Keune, C.; Wolanski, E.; Jones, C.M.; Jerry, D.R. Oceanographic currents and local ecological knowledge indicate and genetics does not refute, a contemporary pattern of larval dispersal for the ornate spiny lobster, Panulirus ornatus in the south-east Asian archipelago. PLoS ONE 2015, 10, e0124568. [Google Scholar] [CrossRef]
- Williams, K.C. Spiny lobster ecology and exploitation in the South China Sea region. In Proceedings of the Australian Centre for International Agriculture Research, the Institue of Oceanography, Nha Trang, Vietnam, 16 November 2004. [Google Scholar]
- Wyrtki, K. Physical oceanography of the Indian Ocean. In The Biology of the Indian Ocean; NAGA Report: San Diego, CA, USA, 1961. [Google Scholar]
- Arai, T.; Taha, H. Contrasting patterns of genetic population structure in tropical freshwater eels of genus Anguilla in the Indo-Pacific. Heliyon 2021, 7, e07097. [Google Scholar] [CrossRef]
| Locality | Collected Month | Total Length (millimeters) | Habitat |
|---|---|---|---|
| Quang Tri | October | 131–265 | Dam, Upstream |
| Quang Ngai | May | 115–251 | Dam, Upstream |
| Binh Dinh | March and December | 45–47 | Lagoon |
| Phu Yen | March and December | 44–46 | Estuary |
| Geographical Area | Locality | Number of Samples | DDBJ/EMBL/GenBank Accession Nos |
|---|---|---|---|
| Western Indian | Madagascar | 23 | AB279256–AB279278 |
| Reunion | 24 | AB279232–AB279255 | |
| Eastern Indian | Sumatra | 36 | AB279196–AB279231 |
| North Pacific | Sulawesi | 14 | AB279119–AB279132 |
| Philippines | 12 | AB279107–AB279118 | |
| Taiwan | 39 | AB279068–AB279106 | |
| Japan | 13 | AB279055–AB279067 | |
| Guam | 7 | AB279315–AB279321 | |
| Ambon | 36 | AB279279–AB279314 | |
| South Pacific | Papua New Guinea | 15 | AB279181–AB279195 |
| New Caledonia | 9 | AB279172–AB279180 | |
| Fiji | 20 | AB279152–AB279171 | |
| Tahiti | 19 | AB279133–AB279151 |
| Population | Number of Samples | Number of Haplotypes | Haplotype Diversity (hd ± SD) | Nucleotide Diversity (π ± SD) | Number of Polymorphic sites (S) | Number of Mutations (η) | Average Nucleotide Differences (k) |
|---|---|---|---|---|---|---|---|
| BD1 | 29 | 29 | 1.000 ± 0.009 | 0.035 ± 0.004 | 192 | 200 | 34.998 |
| PY1 | 28 | 27 | 0.997 ± 0.010 | 0.026 ± 0.004 | 167 | 174 | 25.966 |
| QN | 30 | 29 | 0.998 ± 0.009 | 0.021 ± 0.002 | 143 | 149 | 20.786 |
| QT | 30 | 30 | 1.000 ± 0.009 | 0.029 ± 0.004 | 194 | 200 | 29.085 |
| BD2 | 31 | 29 | 0.996 ± 0.009 | 0.019 ± 0.003 | 148 | 151 | 19.523 |
| PY2 | 28 | 28 | 1.000 ± 0.009 | 0.026 ± 0.003 | 163 | 165 | 26.521 |
| VN (total) | 176 | 165 | 0.999 ± 0.001 | 0.026 ± 0.002 | 306 | 327 | 25.148 |
| Source | Degree of Freedom | Sum of Squares | Variance Components | Percentage of Total Variance (%) |
|---|---|---|---|---|
| Among populations | 5 | 80.062 | 0.098 Va | 0.740 |
| Within populations | 170 | 2234.523 | 13.144 Vb | 99.260 |
| Fixation index (FST) | 0.007 (p value = 0.096 ± 0.007) | |||
| Sampling Sites | BD1 | BD2 | PY1 | PY2 | QN | QT |
|---|---|---|---|---|---|---|
| BD1 | 0.04418 * | 0.01090 | 0.00719 | 0.05235 * | 0.01104 | |
| BD2 | 0.01802 | 0.00058 | 0.00085 | −0.00463 | −0.00054 | |
| PY1 | 0.20721 | 0.34234 | −0.02356 | 0.00553 | −0.01199 | |
| PY2 | 0.23423 | 0.42342 | 0.99099 | 0.00686 | −0.00956 | |
| QN | 0.00000 | 0.63964 | 0.16216 | 0.18018 | 0.00577 | |
| QT | 0.15315 | 0.54054 | 0.90090 | 0.82883 | 0.17117 |
| Population | Tajima’s D | Fu’s Fs |
|---|---|---|
| BD1 | −1.099 (p = 0.133) | −9.428 (p < 0.01) |
| PY1 | −1.835 (p = 0.019) | −16.367 (p < 0.01) |
| QN | −1.531 (p = 0.037) | −10.816 (p < 0.01) |
| QT | −1.422 (p = 0.060) | −10.844 (p < 0.01) |
| BD2 | −1.707 (p = 0.025) | −15.140 (p < 0.01) |
| PY2 | −1.559 (p = 0.044) | −11.244 (p < 0.01) |
| VN (total) | −1.525 (p = 0.049) | −12.308 (p < 0.01) |
| VN | QT-219 | BD1-007 and BD1-013 | |
|---|---|---|---|
| QT-219 | 93.32 | ||
| BD1-007 and BD1-013 | 94.20 | 96.91 | |
| GU | 94.31 | 95.66 | 96.39 |
| AB | 95.13 | 94.87 | 95.68 |
| MD | 93.43 | 95.43 | 95.45 |
| RU | 93.48 | 95.37 | 95.51 |
| SM | 93.82 | 96.79 | 97.31 |
| PNG | 93.61 | 96.47 | 97.19 |
| NC | 93.87 | 96.77 | 97.64 |
| FJ | 93.96 | 96.88 | 98.08 |
| TH | 93.98 | 96.70 | 98.12 |
| SL | 95.22 | 95.13 | 95.84 |
| PH | 97.38 | 93.24 | 94.14 |
| TW | 96.99 | 93.71 | 94.40 |
| JP | 97.43 | 93.56 | 94.30 |
| Locations | JP | TW | PH | SL | TH | FJ | NC | PNG | SM | RU | MD | AB | GU | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Parameters | ||||||||||||||
| FST | −0.0068 | 0.0083 | −0.0141 | 0.2979 | 0.5548 | 0.5651 | 0.5547 | 0.5513 | 0.6065 | 0.5735 | 0.5925 | 0.24543 | 0.4784 | |
| p value | 0.6139 | 0.0854 | 0.8472 | 0.0000 * | 0.0000 * | 0.0000 * | 0.0000 * | 0.0000 * | 0.0000 * | 0.0000 * | 0.0000 * | 0.0000 * | 0.0000 * | |
| Genetic identity (%) | 97.37 | 96.94 | 97.32 | 95.22 | 94.04 | 94.02 | 93.93 | 93.66 | 93.87 | 93.52 | 93.46 | 95.13 | 94.34 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen, A.T.; Dao, H.T.; Quang, H.T.; Hagihara, S.; Lokman, P.M.; Damsteegt, E.L. Genetic Diversity and Population Structure of the Giant Mottled Eel, Anguilla marmorata Quoy & Gaimard, 1824 in Central Vietnam. Fishes 2022, 7, 286. https://doi.org/10.3390/fishes7050286
Nguyen AT, Dao HT, Quang HT, Hagihara S, Lokman PM, Damsteegt EL. Genetic Diversity and Population Structure of the Giant Mottled Eel, Anguilla marmorata Quoy & Gaimard, 1824 in Central Vietnam. Fishes. 2022; 7(5):286. https://doi.org/10.3390/fishes7050286
Chicago/Turabian StyleNguyen, Anh Tuan, Hoc Tan Dao, Hoang Tan Quang, Seishi Hagihara, P. Mark Lokman, and Erin L. Damsteegt. 2022. "Genetic Diversity and Population Structure of the Giant Mottled Eel, Anguilla marmorata Quoy & Gaimard, 1824 in Central Vietnam" Fishes 7, no. 5: 286. https://doi.org/10.3390/fishes7050286
APA StyleNguyen, A. T., Dao, H. T., Quang, H. T., Hagihara, S., Lokman, P. M., & Damsteegt, E. L. (2022). Genetic Diversity and Population Structure of the Giant Mottled Eel, Anguilla marmorata Quoy & Gaimard, 1824 in Central Vietnam. Fishes, 7(5), 286. https://doi.org/10.3390/fishes7050286







