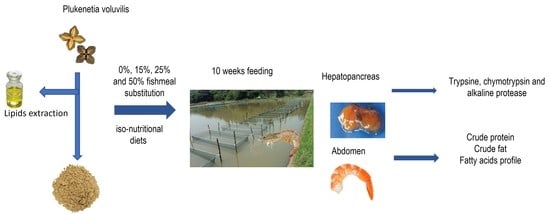
Effect of Replacing Fishmeal with Plukenetia volubilis Cake on Growth, Digestive Enzymes, and Body Composition in Whiteleg Shrimp (Litopenaeus vannamei)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sacha Inchi Cake
2.2. Experimental Diets
2.3. Feeding Trial and Sampling
2.4. Animals Ethics
2.5. Growth Performance and Nutrient Utilization
2.6. Proximate Composition and Fatty Acids Analysis
2.7. Digestive Enzymes Activity
2.8. Statistical Analysis
3. Results
3.1. Nutrition and Growth Indices
3.2. Proximal Composition and Fatty Acid Profile
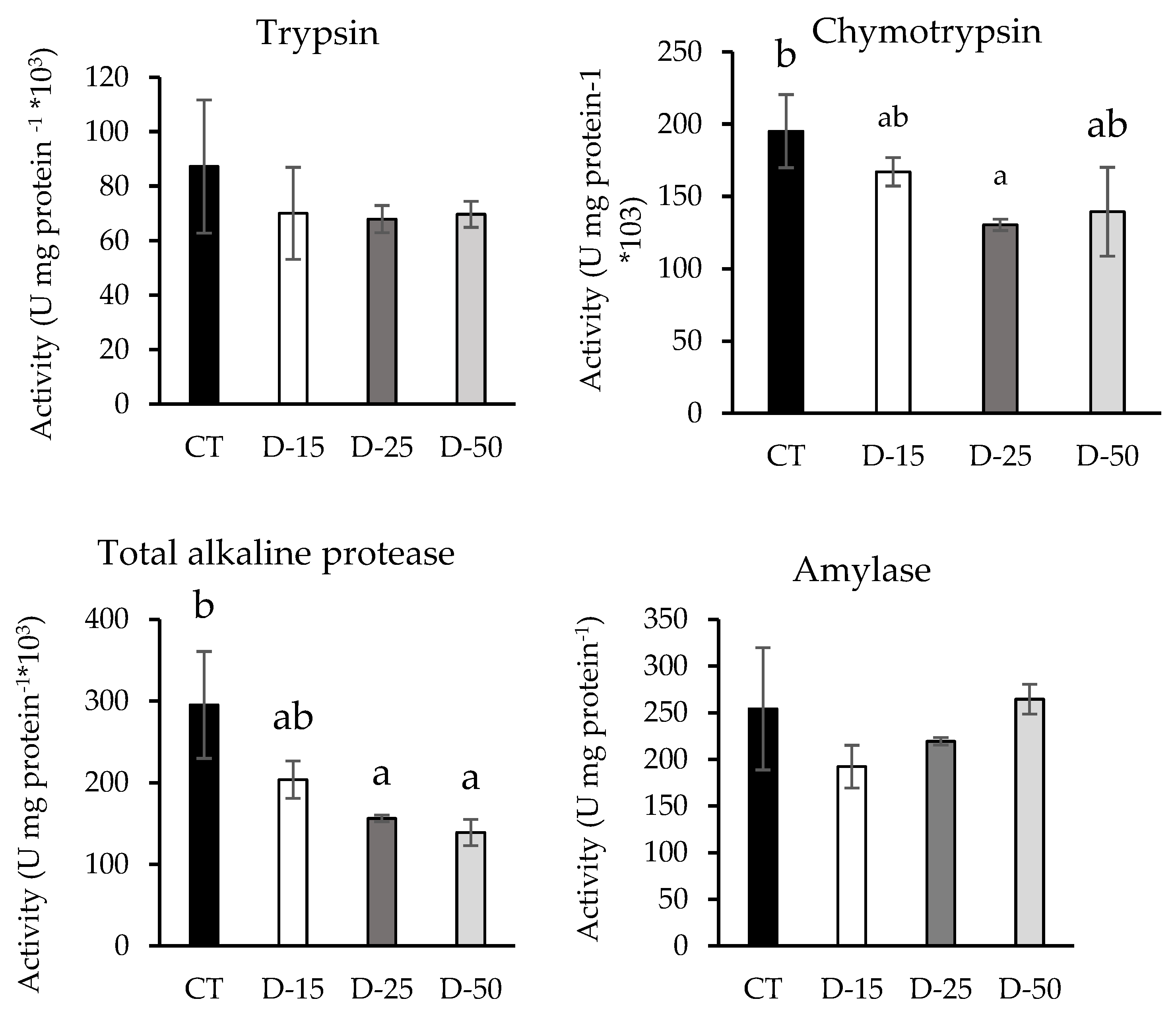
3.3. Digestive Enzymes
4. Discussion
4.1. Growth Performance
4.2. Proximal Composition and Fatty Acid Profile
4.3. Digestive Enzymes
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAO. El Estado Mundial de la Pesca y la Acuicultura 2018. Cumplir los Objetivos de Desarrollo Sostenible; Licencia: CC BY-NC-SA 3.0 IGO; FAO: Rome, Italy, 2018; ISBN 978-92-5-130562-1. [Google Scholar]
- Ayisi, C.L.; Hua, X.M.; Apraku, A.; Afriyie, G.; Kyei, B.A. Recent studies towards the development of practical diets for Shrimp and their nutritional requirements. Hayati J. Biosci. 2017, 24, 109–117. [Google Scholar] [CrossRef]
- Suárez, J.A.; Gaxiola, G.; Mendoza, R.; Cadavid, S.; Garcia, G.; Alanis, G.; Súares, A.; Faillace, J.; Cuzon, G. Substitution of fish meal with plant protein source and energy budget for shrimp Litopenaeus vannamei (Boone, 1931). Aquaculture 2009, 289, 118–123. [Google Scholar] [CrossRef]
- Sánchez-Muros, M.J.; Renteria, P.; Vizcaino, A.; Barroso, F.G. Innovative protein sources in shrimp (Litopenaeus vannamei) feeding. Rev. Aquac. 2020, 12, 186–203. [Google Scholar] [CrossRef]
- Tacon, A.G.; Metian, M. Global overview on the use of fish meal and fish oil in industrially compounded aquafeeds: Trends and future prospects. Aquaculture 2008, 285, 146–158. [Google Scholar] [CrossRef]
- IFFO. 2018. Available online: http://www.iffo.net/fish-fish-out-fifo-ratios-conversion-wild-feed (accessed on 18 April 2022).
- Ayadi, F.Y.; Rosentrater, K.A.; Muthukumarappan, K. Alternative protein sources for aquaculture feeds. J. Aquacult Feed Sci. Nutr. 2012, 4, 1–26. [Google Scholar] [CrossRef]
- Daniel, N. A review on replacing fish meal in aqua feeds using plant protein sources. Int. J. Fish Aquat. Stud. 2018, 6, 164–179. [Google Scholar]
- Lim, C.; Dominy, W. Evaluation of soybean meal as a replacement for marine animal protein in diets for shrimp Penaeus vannamei. Aquaculture 1990, 87, 53–64. [Google Scholar] [CrossRef]
- Sánchez-Muros, M.J.; Barroso, F.G.; Manzano-Agugliaro, F. Insect meal as renewable source of food for animal feeding: A review. J. Clean Prod. 2014, 65, 16–27. [Google Scholar] [CrossRef]
- Guillen, M.D.; Ruiz, A.; Cabo, N.; Chirinos, R.; Pascual, G. Characterization of Sacha Inchi (Plukenetia volubilis L.) oil by FTIR spectroscopy and 1H NMR. Comparison with linseed oil. J. Am. Oil. Chem. Soc. 2003, 80, 755–762. [Google Scholar] [CrossRef]
- Hamaker, B.R.; Valles, C.; Gilman, R.; Hardmeier, R.M.; Clark, D.; García, H.H.; Gonzales, A.E.; Kohlstad, I.; Castro, M.; Valdivia, R.; et al. Amino acid and acid fatty profiles of the inca peanut (Plukenetia volubilis). Note Cereal Chem. 1992, 69, 461–463. [Google Scholar]
- Gutiérrez, L.F.; Rosada, L.M.; Jiménez, A. Chemical composition of Sacha Inchi (Plukenetia volubilis L.) seeds and characteristics of their lipid fraction. Grasas. Aceites 2011, 62, 76–83. [Google Scholar] [CrossRef]
- Fernandes, G.; Venkatraman, J.T. Role of omega-3 fatty acids in health and disease. Nut. Res. 1993, 13, S19–S45. [Google Scholar] [CrossRef]
- Chirinos, R.; Zuloeta, G.; Pedreschi, R.; Mignolet, E.; Larondelle, Y.; Campos, D. Sacha inchi (Plukenetia volubilis): A seed source of polyunsaturated fatty acids, tocopherols, phytosterols, phenolic compounds and antioxidant capacity. Food Chem. 2013, 141, 1732–1739. [Google Scholar] [CrossRef] [PubMed]
- Fanali, C.; Dugo, L.; Cacciola, F.; Beccaria, M.; Grasso, S.; Dachà, M.; Dugo, P.; Mondello, L. Chemical characterisation of Sacha inchi (Plukenetia volubilis L.) oil. J. Agric. Food Chem. 2011, 59, 13043–13049. [Google Scholar] [CrossRef]
- Follegatti-Romero, L.A.; Piantino, C.R.; Grimaldi, R.; Cabral, F.A. Supercritical CO2 extraction of omega-3 rich oil from Sacha inchi (Plukenetia volubilis L.) seeds. J. Supercrit Fluids 2009, 49, 323–329. [Google Scholar] [CrossRef]
- Hanssen, H.P.; Schmitz-Hübsch, M. Sacha inchi (Plukenetia volubilis L.) nut oil and its therapeutic and nutritional uses. In Nuts and Seeds in Health and Disease Prevention; Victor, R.P., Ronald Ross, W., Vinood, B.P., Eds.; Academic Press: San Diego, CA, USA, 2011; pp. 991–994. [Google Scholar]
- Chirinos, R.; Aquino, M.; Pedreschi, R.; Campos, D. Optimized Methodology for Alkaline and Enzyme-Assisted Extraction of Protein from Sacha Inchi (Plukenetia volubilis) Kernel Cake. J. Food Process Eng. 2017, 40, 12412. [Google Scholar] [CrossRef]
- Ferchau, E. Equipment for Decentralised Cold Pressing of Oil Seeds. Folkecenter for Renewable Energy. 2000. Available online: http://www.folkecenter.net/mediafiles/folkecenter/pdf/dk/efdcpos_ef.pdf (accessed on 13 April 2022).
- Miranda-Gelvez, R.A.; Guerrero-Alvarado, C.E. Efecto de la torta de Sacha Inchi (Plukenetia volubilis) sobre el desempeño productivo de juveniles de tilapia roja (Oreochromis sp.). Respuestas 2015, 20, 83–94. [Google Scholar] [CrossRef]
- Araújo-Dairiki, T.B.; Chaves, F.C.M.; Dairiki, J.K. Seeds of sacha inchi (Plukenetia volubilis, Euphorbiaceae) as a feed ingredient for juvenile tambaqui, Colossoma macropomum, and matrinxã, Brycon amazonicus (Characidae). Acta Amaz 2018, 48, 32–37. Available online: http://old.scielo.br/scielo.php?pid=S0044-59672018000100032&script=sci_arttext&tlng=en (accessed on 24 March 2022). [CrossRef]
- Ortiz-Chura, A.; Pari-Puma, R.M.; Huanca, F.H.R.; Cerón-Cucchi, M.E.; Araníbar, M.J.A. Apparent digestibility of dry matter, organic matter, protein and energy of native Peruvian feedstuffs in juvenile rainbow trout (Oncorhynchus mykiss). Fish Aquat. Sci. 2018, 21, 32. [Google Scholar] [CrossRef]
- Spackman, D.H.; Stein, W.H.; Moore, S. Aparato de registro automático para su uso en cromatografía de aminoácidos. Quím. Anal. 1958, 30, 1190–1206. [Google Scholar]
- Bradford, M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Directiva 2010/63/UE del Parlamento Europeo y del Consejo, de 22 de Septiembre de 2010, Relativa a la Protección de los Animales Utilizados Para Fines Científicos; Diario Oficial de la Unión Europea Estrasburgo 2010. Available online: https://eur-lex.europa.eu/legal-content/ES/ALL/?uri=celex%3A32010L0063 (accessed on 17 September 2022).
- Association of Analytical Chemists. Official Methods of Analysis of Official Analytical Chemists International, 17th ed.; Association of Analytical Chemists (AOAC): Wasington, DC, USA, 2000. [Google Scholar]
- Lepage, G.; Roy, C.C. Improved recovery of fatty acid through direct transesterification without prior extraction or purification. J. Lipid Res. 1984, 25, 1391–1396. [Google Scholar] [CrossRef]
- Venegas-Venegas, E.; Rincón-Cervera, M.A.; Guil-Guerrero, J.L. Triarachidonoyl glycerol purification process. J. Am. Oil. Chem. Soc. 2011, 88, 323–328. [Google Scholar] [CrossRef]
- Alarcón, F.J.; Díaz, M.; Moyano, F.J.; Abellán, E. Characterization and functional properties of digestive proteases in two sparids: Gilthead sea bream (Sparus aurata) and common dentex (Dentex dentex). Fish Physiol. Biochem. 1998, 19, 257–267. [Google Scholar] [CrossRef]
- Erlanger, B.F.; Kokowsky, N.; Cohen, W. The preparation and properties of two new chromogenic substrates of trypsin. Arch. Biochem. Biophys. 2011, 95, 271–278. [Google Scholar] [CrossRef]
- Del Mar, E.G.; Largman, C.; Broderick, J.W.; Geokas, M.C. A sensitive new substrate for chymotrypsin. Anal. Biochem. 1979, 99, 316–320. [Google Scholar] [CrossRef]
- Robyt, J.F.; Whelan, W.J. The b-amylases. In Starch and Its Derivatives; Radley, J.A., Ed.; Academic Press: London, UK, 1968; pp. 477–497. [Google Scholar]
- Hurtado-Ramírez, L.; Paredes-López, D.; Robles-Huaynate, R. Efecto de la torta de Sacha inchi (Plukenetia volubilis) en el perfil bioquímico sanguíneo e histopatología del hígado de aves de postura. Cienc. Amaz. 2015, 4, 60–66. [Google Scholar] [CrossRef]
- Inga, R.R.; López, D.P.; Huaynate, R.R. Determinación del efecto del consumo de la torta de sacha inchi (Plukenetia volubilis) sobre el perfil bioquímico sanguíneo de pollos de carne. Folia Amaz. 2015, 24, 31–38. [Google Scholar] [CrossRef]
- Winsor, M.L. Available online: https://www.fao.org/3/x5926e/x5926e01.htm#:~:text=A%20whole%20meal%20made%20from,per%20cent%20water%20and%2021 (accessed on 17 September 2022).
- González-Félix, M.L.; da Silva, F.S.D.; Davis, D.A.; Samocha, T.M.; Morris, T.C.; Wilkenfeld, J.S.; Perez-Velazquez, M. Replacement of fish oil in plant based diets for Pacific white shrimp (Litopenaeus vannamei). Aquaculture 2010, 309, 152–158. [Google Scholar] [CrossRef]
- Suprayudi, M.A.; Takeuchi, T.; Hamasaki, K. Essential fatty acids for larval mud crab Scylla serrata: Implications of lack of the ability to bioconvert C18 unsaturated fatty acids to highly unsaturated fatty acids. Aquaculture 2004, 231, 403–416. [Google Scholar] [CrossRef]
- Gonzalez-Felix, M.L.; Gatlin, D.M.; Lawrence, A.L.; Perez-Velazquez, M. Effect of dietary phospholipid on essential fatty acid requirements and tissue lipid composition of Litopenaeus vannamei juveniles. Aquaculture 2002, 207, 151–167. [Google Scholar] [CrossRef]
- Turchini, G.M.; Torstensen, B.E.; Ng, W.K. Fish oil replacement in finfish nutrition. Rev. Aquac. 2009, 1, 10–57. [Google Scholar] [CrossRef]
- Córdova-Murueta, J.H.; García-Carreño, F.L. Nutritive value of squid and hydrolyzed protein supplement in shrimp feed. Aquaculture 2002, 210, 371–384. [Google Scholar] [CrossRef]
- Kristinsson, H.G.; Rasco, B.A. Hydrolysis of salmon muscle proteins by an enzyme mixture extracted from Atlantic salmon (Salmo salar) pyloric caeca. J. Food Biochem. 2000, 24, 177–187. [Google Scholar] [CrossRef]
- Sunde, J.; Taranger, G.L.; Rungruangsak-Torrissen, K. Digestive protease activities and free amino acids in white muscle as indicators for feed conversion efficiency and growth rate in Atlantic salmon (Salmo salar L.). J. Fish Physiol. Biochem. 2001, 25, 335–345. [Google Scholar] [CrossRef]
- Bueno-Borges, L.B.; Sartim, M.A.; Gil, C.C.; Sampaio, S.V.; Rodrigues, P.H.V.; Regitano-d’Arce, M.A.B. Sacha inchi seeds from sub-tropical cultivation: Effects of roasting on antinutrients, antioxidant capacity and oxidative stability. J. Food Sci. Technol. 2018, 55, 4159–4166. [Google Scholar] [CrossRef]
- Lázaro Aguilar, R.D. Determinación de la presencia de sustancias antinutricionales y alérgenos en semilla y torta de sacha inchi (Plukenetia volubilis L.). Tesis doctoral, Universidad Nacional Agraria la Molina, Perú, 2015. Available online: http://repositorio.lamolina.edu.pe/handle/UNALM/20.500.12996/1822 (accessed on 17 September 2022).
- Ruiz, C.; Díaz, C.; Anaya, J.; Rojas, R. Análisis proximal, antinutrientes, perfil de ácidos grasos y de aminoácidos de semillas y tortas de 2 especies de sacha inchi (Plukenetia volubilis y Plukenetia huayllabambana). Rev. Soc. Quím. Perú 2013, 79, 29–36. [Google Scholar]
- Rojo-Arreola, L.; Choquet, C.; Cordova-Murueta, J.; García-Carreño, F. The protease-based compensatory mechanism to minimize the effect of dietary Soybean Trypsin Inhibitor in Litopenaeus vannamei. Aquaculture 2019, 500, 18–23. [Google Scholar] [CrossRef]
- Schwarzenberger, A.; Fink, P. Gene expression and activity of digestive enzymes of Daphnia pulex in response to food quality differences. Comp. Biochem. Physiol. Part-B Biochem. Mol. Biol. 2018, 218, 23–29. [Google Scholar] [CrossRef]
- Santamaria, M.E.; Arnaiz, A.; Diaz-Mendoza, M.; Martinez, M.; Diaz, I. Inhibitory properties of cysteine protease pro-peptides from barley confer resistance to spider mite feeding. PLoS ONE 2015, 10, e0128323. [Google Scholar] [CrossRef]
- Lazarević, J.; Janković-Tomanić, M. Dietary and phylogenetic correlates of digestive trypsin activity in insect pests. Entomol. Exp. Appl. 2015, 157, 123–151. [Google Scholar] [CrossRef]
- Le Moullac, G.; Klein, B.; Sellos, D.; Van wormhoudt, A. Adaptation of trypsin, chymotrypsin and α-amylase to casein level and protein source in Penaeus vannamei (Crustacea Decapoda). J. Exp. Marine Biol. Ecol. 1996, 208, 107–125. [Google Scholar] [CrossRef]
| Sacha Inchi Cake | Fish Meal | |
|---|---|---|
| Non-essential amino acids | ||
| Asp | 3.62 | 3.81 |
| Tyr | 1.34 | 1.33 |
| Ser | 1.98 | 1.69 |
| Glu | 4.34 | 5.19 |
| Pro | 1.29 | 1.79 |
| Gly | 3.01 | 2.74 |
| Ala | 1.19 | 2.98 |
| Cys | 0.66 | 0 |
| Essential amino acids | ||
| Thr | 1.23 | 1.57 |
| Val | 1.18 | 1.71 |
| Met | 0.33 | 1.39 |
| Ile | 0.91 | 1.38 |
| Leu | 1.97 | 2.94 |
| Phe | 0.73 | 0.72 |
| His | 0.75 | 1.39 |
| Lys | 1.48 | 3.12 |
| Arg | 2.86 | 2.3 |
| Total | 28.85 | 37.15 |
| CT | D-15 | D-25 | D-50 | |
|---|---|---|---|---|
| Ingredients (g kg dry matter−1) | ||||
| Fish meal | 186.9 | 158.9 | 140.2 | 93.5 |
| Sacha inchi | 0.00 | 28.0 | 46.7 | 93.5 |
| Soy meal | 313.1 | 313.1 | 303.7 | 294.4 |
| Wheat meal | 414.6 | 414.6 | 414.6 | 414.6 |
| Squid meal | 18.7 | 18.7 | 28.0 | 37.4 |
| Vitamin | 10.0 | 10.0 | 10.0 | 10.0 |
| Mineral | 10.0 | 10.0 | 10.0 | 10.0 |
| Fish oil | 46.7 | 46.7 | 46.7 | 46.7 |
| Proximate composition (g kg dry matter−1) | ||||
| Crude protein | 359.6 | 359.7 | 356.6 | 356.5 |
| Total lipid | 87.8 | 87.9 | 84.7 | 83.8 |
| NDF | 189.3 | 128.1 | 189.7 | 195.8 |
| Ash | 86.7 | 86.7 | 82.1 | 83.6 |
| MELN | 465.9 | 465.7 | 476.6 | 477.1 |
| Sacha Inchi Cake | CT | D-15 | D-25 | D-50 | |
|---|---|---|---|---|---|
| 14:0 | 4.20 ± 0.03 | 4.46 ± 0.08 | 3.91 ± 0.23 | 4.31 ± 0.02 | |
| 16:0 | 3.79 ± 0.22 | 23.21 ± 0.15 | 24.32 ± 0.49 | 23.08 ± 0.07 | 23.17 ± 0.20 |
| 16:1n7 | 4.71 ± 0.01 | 4.94 ± 0.01 | 4.52 ± 0.18 | 4.80 ± 0.04 | |
| 18:0 | 2.42 ± 0.08 | 5.07 ± 0.04 | 5.26 ± 0.30 | 5.33 ± 0.41 | 5.10 ± 0.09 |
| 18:1n9 | 9.28 ± 0.19 | 14.29 ± 0.22 | 14.57 ± 0.04 | 15.00 ± 0.40 | 14.52 ± 0.14 |
| 18:1n7 | 0.33 ± 0.04 | 2.54 ± 0.04 | 2.66 ± 0.03 | 2.63 ± 0.05 | 2.63 ± 0.01 |
| 18:2n6 | 38.83 ± 0.16 | 13.21 ± 0.01 | 14.15 ± 0.08 | 15.18 ± 0.43 | 13.58 ± 0.13 |
| 18:3n3 | 44.42 ± 0.15 | 3.11 ± 0.01 | 1.98 ± 0.05 | 5.04 ± 0.39 | 3.63 ± 0.00 |
| 20:4n6 | 1.70 ± 0.03 | 1.95 ± 0.01 | 1.79 ± 0.01 | 1.87 ± 0.02 | |
| 20:5n3 | 6.91 ± 0.12 | 8.01 ± 0.40 | 6.81 ± 0.22 | 7.73 ± 0.10 | |
| 22:5n3 | 1.23 ± 0.01 | 1.49 ± 0.03 | 1.30 ± 0.05 | 1.42 ± 0.02 | |
| 22:6n3 | 14.48 ± 0.52 | 16.56 ± 0.86 | 15.36 ± 0.45 | 16.75 ± 0.02 |
| CT | D-15 | D-25 | D-50 | p Value | |
|---|---|---|---|---|---|
| Final body weight | 14.25 ± 1.02 a | 14.57 ± 1.34 ab | 16.04 ± 0.25 ab | 16.72 ± 0.67 b | 0.031 |
| Cephalothorax weight | 3.99 ± 0.59 ab | 3.65 ± 0.36 a | 4.48 ± 0.41 b | 4.40 ± 0.82 b | 0.009 |
| Abdomen weight | 8.74 ± 1.17 ab | 8.86 ± 1.04 ab | 9.75 ± 0.99 ab | 10.32 ± 1.71 b | 0.024 |
| Abdomen + exoskeleton | 10.20 ± 1.77 ab | 10.24 ± 0.95 a | 11.34 ± 1.07 ab | 12.21 ± 1.98 b | 0.013 |
| Hepatopancreas weight | 0.42 ± 0.13 ab | 0.45 ± 0.11 a | 0.60 ± 0.09 b | 0.48 ± 0.15 ab | 0.011 |
| Total length | 12.76 ± 1.40 | 13.10 ± 0.39 | 13.30 ± 0.59 | 13.65 ± 0.53 | 0.127 |
| Cephalothorax length | 5.65 ± 0.30 | 5.65 ± 0.30 | 5.49 ± 0.31 | 5.30 ± 0.42 | 0.079 |
| Abdomen length | 8.96 ± 0.61 | 8.96 ± 0.61 | 8.95 ± 0.28 | 9.13 ± 0.52 | 0.843 |
| DG | 0.15 ± 0.01 a | 0.16 ± 0.02 ab | 0.17 ± 0.01 ab | 0.19 ± 0.01 b | 0.028 |
| SGR | 1.95 ± 0.10 a | 2.13 ± 0.19 ab | 2.03 ± 0.09 a | 2.36 ± 0.06 b | 0.015 |
| FCR | 4.72 ± 0.46 b | 4.45 ± 0.54 ab | 4.09 ± 0.14 ab | 3.69 ± 0.15 a | 0.042 |
| Dietary Treatment | Crude Protein | Total Lipids |
|---|---|---|
| CT | 86.14 ± 5.40 b | 3.86 ± 0.55 |
| D-15 | 73.98 ± 2.61 a | 3.65 ± 0.40 |
| D-25 | 74.11 ± 1.29 a | 3.86 ± 0.50 |
| D-50 | 75.05 ± 1.88 a | 3.37 ± 0.28 |
| p value | < 0.001 | 0.055 |
| CT | D-15 | D-25 | D-50 | p Value | |
|---|---|---|---|---|---|
| 14:0 | 0.72 ± 0.06 | 0.86 ± 0.08 | 0.78 ± 0.20 | 0.94 ± 0.10 | 0.430 |
| 15:0 | 0.66 ± 0.06 | 0.76 ± 0.01 | 0.75 ± 0.14 | 0.83 ± 0.16 | 0.536 |
| 16:0 | 18.04 ± 0.39 | 16.77 ± 0.84 | 16.98 ± 1.14 | 16.65 ± 0.42 | 0.376 |
| 16:1n7 | 1.98 ± 0.36 | 2.12 ± 0.09 | 1.99 ± 0.34 | 2.22 ± 0.67 | 0.939 |
| 17:0 | 2.05 ± 0.19 | 2.17 ± 0.16 | 2.04 ± 0.22 | 1.50 ± 0.48 | 0.252 |
| 18:0 | 10.70 ± 0.05 c | 10.11 ± 0.02 b | 9.96 ± 0.10 b | 9.23 ± 0.20 a | 0.001 |
| 18:1n9 | 11.23 ± 0.64 | 9.50 ± 0.07 | 9.96 ± 1.31 | 8.81 ± 0.22 | 0.114 |
| 18:1n7 | 2.69 ± 0.20 | 2.75 ± 0.12 | 2.66 ± 0.21 | 2.56 ± 0.26 | 0.826 |
| 18:2n6 | 8.11 ± 0.17 | 7.63 ± 0.15 | 7.87 ± 0.27 | 8.14 ± 0.16 | 0.158 |
| 18:3n6 | 0.57 ± 0.05 | 0.55 ± 0.02 | 0.53 ± 0.07 | 0.56 ± 0.02 | 0.799 |
| 18:3n3 | 1.12 ± 0.21 | 1.31 ± 0.13 | 1.25 ± 0.29 | 1.74 ± 0.35 | 0.238 |
| 20:1n9 | 0.63 ± 0.20 | 0.44 ± 0.03 | 0.40 ± 0.00 | 0.37 ± 0.05 | 0.159 |
| 20:4n6 | 4.53 ± 0.51 | 4.20 ± 0.12 | 4.01 ± 0.17 | 4.05 ± 0.01 | 0.344 |
| 20:5n3 | 12.06 ± 0.34 | 12.22 ± 1.38 | 11.35 ± 0.80 | 11.00 ± 0.36 | 0.489 |
| 22:5n3 | 0.68 ± 0.01 | 0.73 ± 0.07 | 0.73 ± 0.01 | 0.71 ± 0.06 | 0.847 |
| 22:6n3 | 14.84 ± 1.41 | 12.97 ± 0.69 | 13.37 ± 0.96 | 12.31 ± 0.09 | 0.175 |
| SFA | 32.17 ± 0.74 b | 30.67 ± 0.62 ab | 30.51 ± 0.47 ab | 29.15 ± 0.39 a | 0.027 |
| MUFA | 16.53 ± 0.12 b | 14.81 ± 0.13 ab | 15.01 ± 0.76 ab | 13.96 ± 1.09 a | 0.044 |
| PUFA | 41.57 ± 0.41 b | 39.60 ± 1.61 a | 39.12 ± 1.50 a | 38.52 ± 0.73 a | 0.019 |
| n-3 | 28.36 ± 0.80 | 27.22 ± 1.86 | 26.70 ± 1.47 | 25.76 ± 0.86 | 0.380 |
| n-6 | 13.21 ± 0.39 | 12.39 ± 0.25 | 12.41 ± 0.03 | 12.75 ± 0.13 | 0.071 |
| n-6/n-3 | 0.47 ± 0.03 | 0.46 ± 0.04 | 0.47 ± 0.02 | 0.50 ± 0.02 | 0.617 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Renteria, P.; Vizcaíno, A.J.; Sánchez-Muros, M.J.; Santacruz-Reyes, R.A.; Saez, M.I.; Fabrikov, D.; Barroso, F.G.; Vargas-García, M.d.C. Effect of Replacing Fishmeal with Plukenetia volubilis Cake on Growth, Digestive Enzymes, and Body Composition in Whiteleg Shrimp (Litopenaeus vannamei). Fishes 2022, 7, 244. https://doi.org/10.3390/fishes7050244
Renteria P, Vizcaíno AJ, Sánchez-Muros MJ, Santacruz-Reyes RA, Saez MI, Fabrikov D, Barroso FG, Vargas-García MdC. Effect of Replacing Fishmeal with Plukenetia volubilis Cake on Growth, Digestive Enzymes, and Body Composition in Whiteleg Shrimp (Litopenaeus vannamei). Fishes. 2022; 7(5):244. https://doi.org/10.3390/fishes7050244
Chicago/Turabian StyleRenteria, Patricio, Antonio Jesús Vizcaíno, María José Sánchez-Muros, Roberto A. Santacruz-Reyes, María Isabel Saez, Dmitri Fabrikov, Fernando G. Barroso, and María del Carmen Vargas-García. 2022. "Effect of Replacing Fishmeal with Plukenetia volubilis Cake on Growth, Digestive Enzymes, and Body Composition in Whiteleg Shrimp (Litopenaeus vannamei)" Fishes 7, no. 5: 244. https://doi.org/10.3390/fishes7050244
APA StyleRenteria, P., Vizcaíno, A. J., Sánchez-Muros, M. J., Santacruz-Reyes, R. A., Saez, M. I., Fabrikov, D., Barroso, F. G., & Vargas-García, M. d. C. (2022). Effect of Replacing Fishmeal with Plukenetia volubilis Cake on Growth, Digestive Enzymes, and Body Composition in Whiteleg Shrimp (Litopenaeus vannamei). Fishes, 7(5), 244. https://doi.org/10.3390/fishes7050244