Length-Based Assessment Methods for the Conservation of a Pelagic Shark, Carcharhinus falciformis from the Tropical Pacific Ocean
Abstract
:1. Introduction
2. Materials and Methods
2.1. Data Collection
2.2. Stock Assessment Indicators
2.2.1. Weight-Length Relationship
2.2.2. Estimation of Growth Parameters
2.2.3. Natural Mortality Estimate (M)
2.2.4. Estimating Size at Maturity L50% and L95%
2.3. The Length-Based Spawning Potential Ratio (LBSPR) Model
2.4. The Length-Based Bayesian Biomass (LBB) Model
3. Results
3.1. Weight-Length Relationship
3.2. Estimates of Growth Parameters, Natural Mortality, and Size at 50% and 95% Sexual Maturity
3.2.1. Growth Parameters
3.2.2. Natural Mortality Estimates (M)
3.2.3. Estimating Size at-L50%-and-L95% Maturity
3.3. Estimation of Biological Reference Points (BRP) and Stock Status
3.3.1. Estimates Provided by the LBSPR Model
3.3.2. Estimates Provided by the LBB Model
4. Discussion
4.1. Growth, Mortality, and Sexual Maturity
4.2. Diagnosis of the Stock-Based on LBSPR and LBB
4.2.1. LBSPR
4.2.2. Stock Condition Analysis Based on LBB
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Prince, J.; Creech, S.; Madduppa, H.; Hordyk, A. Length-based assessment of spawning potential ratio in data-poor fisheries for blue swimming crab (Portunus spp.) in Sri Lanka and Indonesia: Implications for sustainable management. Reg. Stud. Mar. Sci. 2020, 36, 101309. [Google Scholar] [CrossRef]
- FAO. FAO Yearbook; Fishery and Aquaculture Statistics: Rome, Italy, 2019. [Google Scholar]
- Methot, R.D.; Wetzel, C.R. Stock synthesis: A biological and statistical framework for fish stock assessment and fishery management. Fish. Res. 2013, 142, 86–99. [Google Scholar] [CrossRef]
- Last, P.R.; Stevens, J.D. Sharks and Rays of Australia; CSIRO Division of Fisheries: Hobart, Australia, 2009. [Google Scholar]
- Rigby, C.L.; Sherman, C.S.; Chin, A.; Simpfendorfer, C. Carcharhinus falciformis. The IUCN Red List of Threatened Species. 2017: E.T39370A117721799. Available online: https://doi.org/10.2305/IUCN.UK.2017RLTS.T39370A117721799.en (accessed on 26 September 2021).
- Hutchinson, M.; Coffey, D.M.; Holland, K.; Itano, D.; Leroy, B.; Kohin, S.; Vetter, R.; Williams, A.J.; Wren, J. Movements and habitat use of juvenile silky sharks in the Pacific Ocean inform conservation strategies. Fish. Res. 2019, 210, 131–142. [Google Scholar] [CrossRef]
- Bonfil, R. The Biology and Ecology of the Silky Shark, Carcharhinus falciformis. In Sharks of the Open Ocean: Biology, Fisheries and Conservation; Camhi, M., Pikitch, E.K., Babcock, E.A., Eds.; Blackwell Science: Hoboken, NJ, USA, 2008; pp. 114–127. [Google Scholar]
- Clarke, S.; Harley, S.; Hoyle, S.; Rice, J. An indicator-based analysis of key shark species based on data held by SPC-OFP. In Proceedings of the Seventh Regular Session of the Scientific Committee of the Western Central Pacific Fisheries Commission, Pohnpei, Federated States of Micronesia, 9–17 August 2011. [Google Scholar]
- Oliver, S.; Braccini, M.; Newman, S.J.; Harvey, E.S. Global patterns in the bycatch of sharks and rays. Mar. Pol. 2015, 54, 86–97. [Google Scholar] [CrossRef]
- Dulvy, N.K.; Baum, J.K.; Clarke, S.; Compagno, L.J.V.; Corte’s, E.; Domingo, A.S.; Fordham, S.; Fowler, S.; Francis, M.P.; Gibson, C.; et al. You can swim but you can’t hide: The global status and conservation of oceanic pelagic sharks and rays. Aquat. Conserv. 2008, 18, 459–482. [Google Scholar] [CrossRef]
- Lopez, J.; Alvarez-Berastegui, D.; Soto, M.; Murua, H. Using fisheries data to model the oceanic habitats of juvenile silky shark (Carcharhinus falciformis) in the tropical eastern Atlantic Ocean. Biodivers. Conserv. 2020, 29, 2377–2397. [Google Scholar] [CrossRef]
- Pacoureau, N.; Rigby, C.L.; Kyne, P.M.; Sherley, R.B.; Winker, H.; Carlson, J.K.; Fordham, S.V.; Barreto, R.; Fernando, D.; Francis, M.P.; et al. Half a century of global decline in oceanic sharks and rays. Nature 2021, 589, 567–571. [Google Scholar] [CrossRef]
- Dulvy, N.K.; Pacoureau, N.; Rigby, C.L.; Pollom, R.A.; Jabado, R.W.; Ebert, D.A.; Finucci, B.; Pollock, C.M.; Cheok, J.; Derrick, D.H.; et al. Overfishing drives over one-third of all sharks and rays toward a global extinction crisis. Curr. Biol. 2021, 31, 4773–4787.e8. [Google Scholar] [CrossRef]
- Simeon, B.M.; Muttaqin, E.; Mardhiah, U.; Ichsan, M.; Dharmadi; Prasetyo, A.P.; Fahmi; Yulianto, I. Increasing abundance of silky sharks in the Eastern Indian Ocean: Good news or a reason to be cautious? Fishes 2018, 3, 29. [Google Scholar] [CrossRef] [Green Version]
- Cardeñosa, D.; Fields, A.T.; Babcock, E.; Shea, K.H.; Feldheim, K.A.; Kraft, D.W.; Hutchinson, M.; Herrera, M.A.; Caballero, S.; Chapman, D.D. Indo-Pacific origins of silky shark fins in major shark fin markets highlights supply chains and management bodies key for conservation. Conserv. Lett. 2021, 14, e12780. [Google Scholar] [CrossRef]
- Quinn, T.J., II; Deriso, R.B. Quantitative Fish Dynamics; Oxford University Press: Oxford, UK, 1999. [Google Scholar]
- Costello, C.; Ovando, D.; Hilborn, R.; Gaines, S.D.; Deschenes, O.; Lester, S.E. Status and solutions for the world’s unassessed fisheries. Science 2012, 338, 517–520. [Google Scholar] [CrossRef] [Green Version]
- Froese, R. Keep it simple: Three indicators to deal with overfishing. Fish Fish. 2004, 5, 86–91. [Google Scholar] [CrossRef] [Green Version]
- Pons, M.; Kell, L.; Rudd, M.B.; Cope, J.M.; Frédou, F.L. Performance of length-based data-limited methods in a multi-fleet context: Application to small tunas, mackerels, and boni-tos in the Atlantic Ocean. ICES J. Mar. Sci. 2019, 76, 960–973. [Google Scholar] [CrossRef]
- Chrysafi, A.; Kuparinen, A. Assessing abundance of populations with limited data: Lessons learned from data-poor fisheries stock assessment. Environ. Rev. 2016, 24, 25–38. [Google Scholar] [CrossRef]
- Dowling, N.A.; Dichmont, C.M.; Haddon, M.; Smith, D.C.; Smith, A.D.M.; Sainsbury, K. Empirical harvest strategies for data-poor fisheries: A review of the literature. Fish Res. 2015, 171, 141–153. [Google Scholar] [CrossRef]
- Froese, R.; Winker, H.; Coro, G.; Demirel, N.; Tsikliras, A.C.; Dimarchopoulou, D.; Scarcella, G.; Probst, W.N.; Dureuil, M.; Pauly, D. A new approach for estimating stock status from length frequency data. ICES J. Mar. Sci. 2018, 75, 2004–2015. [Google Scholar] [CrossRef]
- Hordyk, A.; Ono, K.; Valencia, S.; Loneragan, N.; Prince, J. A novel length-based empirical estimation method of spawning potential ratio (SPR), and tests of its performance, for small-scale, data-poor fisheries. ICES J. Mar. Sci. 2015, 72, 217–231. [Google Scholar] [CrossRef] [Green Version]
- Hordyk, A.; Ono, K.; Prince, J.D.; Walters, C.J. A simple length-structured model based on life history ratios and incorporating size-dependent selectivity: Application to spawning potential ratios for data-poor stocks. Can. J. Fish. Aquat. Sci. 2016, 73, 1787–1799. [Google Scholar] [CrossRef]
- Aires-da-Silva, A.; Lennert-Cody, C.; Maunder, M.N.; Román-Verdesoto, M. Updated stock status indicators for silky sharks in the eastern Pacific Ocean (1994–2014). In Proceedings of the Inter-American Tropical Tuna Commission Scientific Advisory Committee Sixth Meeting, La Jolla, CA, USA, 11–15 May 2015. [Google Scholar]
- Sparre, P.; Venema, S.C. Introduction to Tropical Fish Stock Assessment—Part 1: Manual; FAO Fisheries Technical Paper 306/1 Rev. 2; FAO: Rome, Italy, 1998; pp. 185–214. [Google Scholar]
- Mildenberger, T.K. Single-Species Fish Stock Assessment with TropFishR. Available online: https://cran%20r-project%20org/web/packages/TropFishR/vignettes/tutorial%20html (accessed on 27 April 2022).
- Von Bertalanffy, L. A quantitative theory of organic growth (inquiries on growth laws II). Hum. Biol. 1983, 10, 181–213. [Google Scholar]
- Pauly, D. Studying Single-Species Dynamics in a Tropical Multispecies Context. In Theory and Management of Tropical Fisheries, Proceedings of the ICLARM/CSIRO Workshop on the Theory and Management of Tropical Multispecies Stocks, ICLARM, Cronulla, Australia, 12–21 January 1982; International Center for Living Aquatic Resources Management: Makati, Philippines, 1982. [Google Scholar]
- Schwamborn, R.; Mildenberger, T.K.; Taylor, M.H. Assessing source of uncertainty in length-based estimates of body growth in populations of fishes and macro-invertebrates with bootstrapped ELEFAN. Ecol. Model. 2019, 293, 37–51. [Google Scholar] [CrossRef] [Green Version]
- Kenchington, T.J. Natural mortality estimators for information-limited fisheries. Fish Fish. 2014, 15, 533–562. [Google Scholar] [CrossRef]
- Alverson, D.L.; Carney, M.J. A graphic review of the growth and decay of population cohorts. ICES J. Mar. Sci. 1975, 36, 133–143. [Google Scholar] [CrossRef]
- Pauly, D. On the interrelationships between natural mortality, growth parameters, and mean environmental temperature in 175 fish stocks. ICES J. Mar. Sci. 1980, 39, 175–192. [Google Scholar] [CrossRef]
- Hoenig, J.M. Empirical Use of Longevity Data to Estimate Mortality Rates. Fish. Bull. 1983, 82, 898–903. [Google Scholar]
- Hewitt, D.A.; Hoenig, J.M. Comparison of two approaches for estimating natural mortality based on longevity. Fish. Bull. 2005, 103, 433–437. [Google Scholar]
- Then, A.Y.; Hoenig, J.M.; Hall, N.G.; Hewitt, D.A.; Jardim, H.E.E. Evaluating the predictive performance of empirical estimators of natural mortality rate using information on over 200 fish species. ICES J. Mar. Sci. 2015, 72, 82–92. [Google Scholar] [CrossRef]
- Mildenberger, T.; Marc, H.; Wolff, T.M. Tropical Fisheries Analysis Version: 1.6.3. 2021. Available online: https://github.com/tokami/TropFishR (accessed on 17 April 2022).
- Wu, F.; Kindong, R.; Dai, X.; Sarr, O.; Jiangfeng, Z.; Siquan, T.; Yunkai, L.; Bruno, T.N.N. Aspects of the reproductive biology of two pelagic sharks in the eastern Atlantic Ocean. J. Fish Biol. 2020, 97, 1651–1661. [Google Scholar] [CrossRef]
- King, M.G. Fisheries Biology, Assessment and Management, 2nd ed.; Blackwell Scientific Publications: Oxford, UK, 2007; pp. 211–219. [Google Scholar]
- Kindong, R.; Wu, F.; Sarr, O.; Dai, L.; Tian, S.; Dai, X. Life history of wahoo, Acanthocybium solandri, in the Tropical Eastern Atlantic Ocean–the importance of applying a suite of methods for fisheries assessment in data-limited situations. Oceano Hydrobiol. Stud. 2022, 51, 115–132. [Google Scholar] [CrossRef]
- Prince, J.D.; Hordyk, A.R.; Valencia, S.R.; Loneragan, N.R.; Sainsbury, K.J. Revisiting the concept of Beverton-Holt life-history invariants with the aim of informing data-poor fisheries assessment. ICES J. Mar. Sci. 2015, 72, 194–203. [Google Scholar] [CrossRef] [Green Version]
- Goodyear, C.P. Spawning Stock Biomass per Recruit in Fisheries Management: Foundation and Current Use. In Risk Evaluation and Biological Reference Points for Fisheries Management; Smith, S.J., Hunt, J.J., Rivard, D., Eds.; NRC Research Press: Ottawa, ON, Canada, 1993; pp. 67–81. [Google Scholar]
- Hordyk, A. LBSPR: Length-Based Spawning Potential Ratio. R Package Version 0.1.6. Available online: https://github.com/AdrianHordyk/LBSPR (accessed on 17 April 2022).
- Palomares, M.L.D.; Froese, R.; Derrick, B.; Nöel, S.-L.; Tsui, G.; Woroniak, J.; Pauly, D. A preliminary global assessment of the status of exploited marine fish and invertebrate populations. In A Report Prepared by the Sea Around Us for OCEANA; OCEANA: Washington, DC, USA, 2018; p. 64. [Google Scholar]
- R Development Core Team. R: A Language and Environment for Statistical Computing; Version 4.1.3; R Foundation for Statistical Computing: Vienna, Austria, 2022; ISBN 3-900051-07-0. Available online: http://www.R-project.org (accessed on 10 June 2022).
- Froese, R.; Pauly, D. FishBase. World Wide Web Electronic Publication. Available online: www.fishbase.se (accessed on 19 April 2022).
- Clarke, S.; Hoyle, S. Development of limit reference points for elasmobranchs. In Proceedings of the Scientific Committee Tenth Regular Session, Majuro, Marshall Islands, 6–14 August 2014; p. 43. [Google Scholar]
- Tsai, W.-P.; Huang, C.-H. Data-limited approach to the management and conservation of the pelagic thresher shark in the Northwest Pacific. Conserv. Sci. Pract. 2022, 5, e12682. [Google Scholar] [CrossRef]
- Sousa, F.; Isidro, E.; Erzini, K. Semi-pelagic longline selectivity for two demersal species from the Azores: The black spot sea bream (Pagellus bogaraveo) and the bluemouth rockfish (Helicolenus dactylopterus dactylopterus). Fish. Res. 1999, 41, 25–35. [Google Scholar] [CrossRef]
- Clarke, M.W.; Borges, L.; Officer, R.A. Comparisons of Trawl and Longline Catches of Deepwater Elasmobranchs West and North of Ireland. J. Northw. Atl. Fish. Sci. 2005, 35, 429–442. [Google Scholar] [CrossRef]
- Herrón, P.; Mildenberger, T.K.; Diaz, J.M.; Wolff, M. Assessment of the stock status of small scale and multi-gear fisheries resources in the tropical Eastern Pacific region. Reg. Stud. Mar. Sci. 2018, 24, 311–323. [Google Scholar] [CrossRef]
- Amande, M.; Chassot, E.; Chavance, P.; Pianet, R. Silky shark (Carcharhinus falciformis) bycatch in the French tuna purse-seine fishery of the Indian Ocean. In Proceedings of the IOTC-2008-WPEB-16, Bangkok, Thailand, 20–22 October 2008; 22p. [Google Scholar]
- Molony, B. Fisheries biology and ecology of highly migratory species that commonly interact with industrialized longline and purse-seine fisheries in the western and central Pacific Ocean. In Proceedings of the Fourth Scientific Committee Meeting of the Western and Central Pacific Fisheries Commission, WCPFC, Port Moresby, Papua New Guinea, 11–18 August 2008. [Google Scholar]
- Hutchinson, M.; Coffey, D.M.; Holland, K.; Itano, D.; Leroy, B.; Kohin, S.; Vetter, R.; Williams, A.; Wren, J. Life history characteristics of the silky shark Carcharhinus falciformis from the central west Pacific. Mar. Fresh. Res. 2018, 69, 562–573. [Google Scholar]
- Kindong, R.; Zhu, J.F.; Dai, X.J.; Tian, S.Q. Life history parameters and yield per recruit analysis for Tachysurus nitidus and Plagiognathops microlepis in lake Dianshan and their management implications. Turk. J. Fish. Aqua. Sci. 2019, 19, 1025–1038. [Google Scholar] [CrossRef]
- Kindong, R.; Gao, C.; Pandong, N.A.; Ma, Q.; Tian, S.; Wu, F.; Sarr, O. Stock status assessments of five small pelagic species in the Atlantic and Pacific Oceans using the Length-Based Bayesian Estimation (LBB) Method. Fron. Mar. Sci. 2020, 7, 592082. [Google Scholar] [CrossRef]
- Cousido-Rocha, M.; Cerviño, S.; Alonso-Fernández, A.; Gil, J.; Herraiz, I.G.; Rincón, M.M.; Ramos, F.; Rodríguez-Cabello, C.; Sampedro, P.; Vila, Y.; et al. Applying length-based assessment methods to fishery resources in the Bay of Biscay and Iberian Coast ecoregion: Stock status and parameter sensitivity. Fish. Res. 2022, 248, 106197. [Google Scholar] [CrossRef]
- Clarke, S.; Langley, A.; Lennert-Cody, C.; Alexandre, A.; Mark, M. Pacific-wide Silky Shark (Carcharhinus falciformis) Stock Status Assessment. In Proceedings of the WCPFC Scientific Committee 14th Regular Session, Busan, Korea, 8–16 August 2018; p. 137. [Google Scholar]
- Ortiz de Urbina, J.; Thomas, B.; Rui, C.; Gorka, M.; Catarina, S.; Hilario, M.; Pascal, B.; Sámar, S.; David, M. A preliminary stock assessment for the silky shark in the Indian Ocean using a data-limited approach. In Proceedings of the IOTC—14th Working Party on Ecosystems and Bycatch, IOTC-WPEB14-2018-033, Cape Town, South Africa, 10–14 September 2018; p. 14. [Google Scholar]
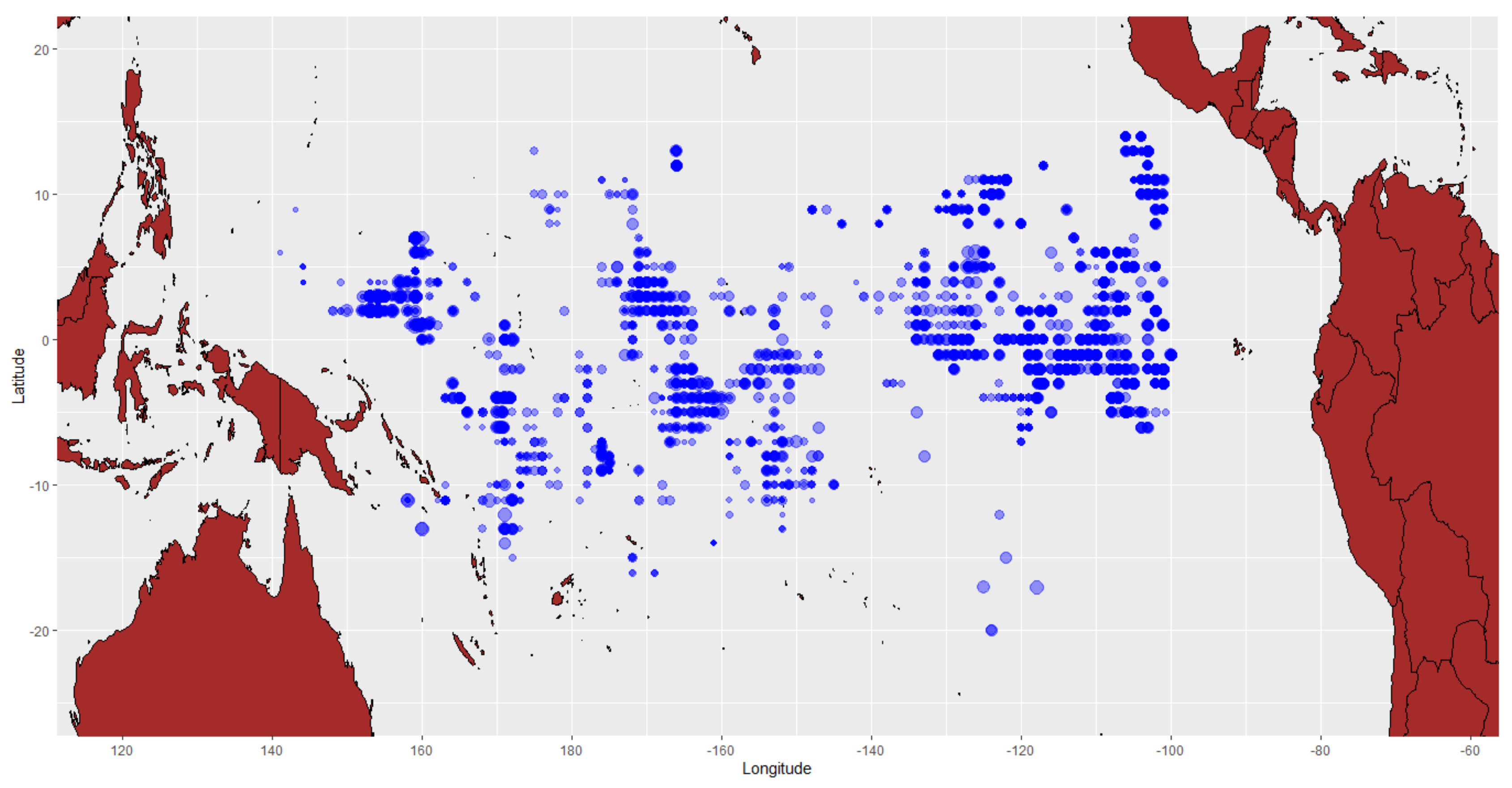
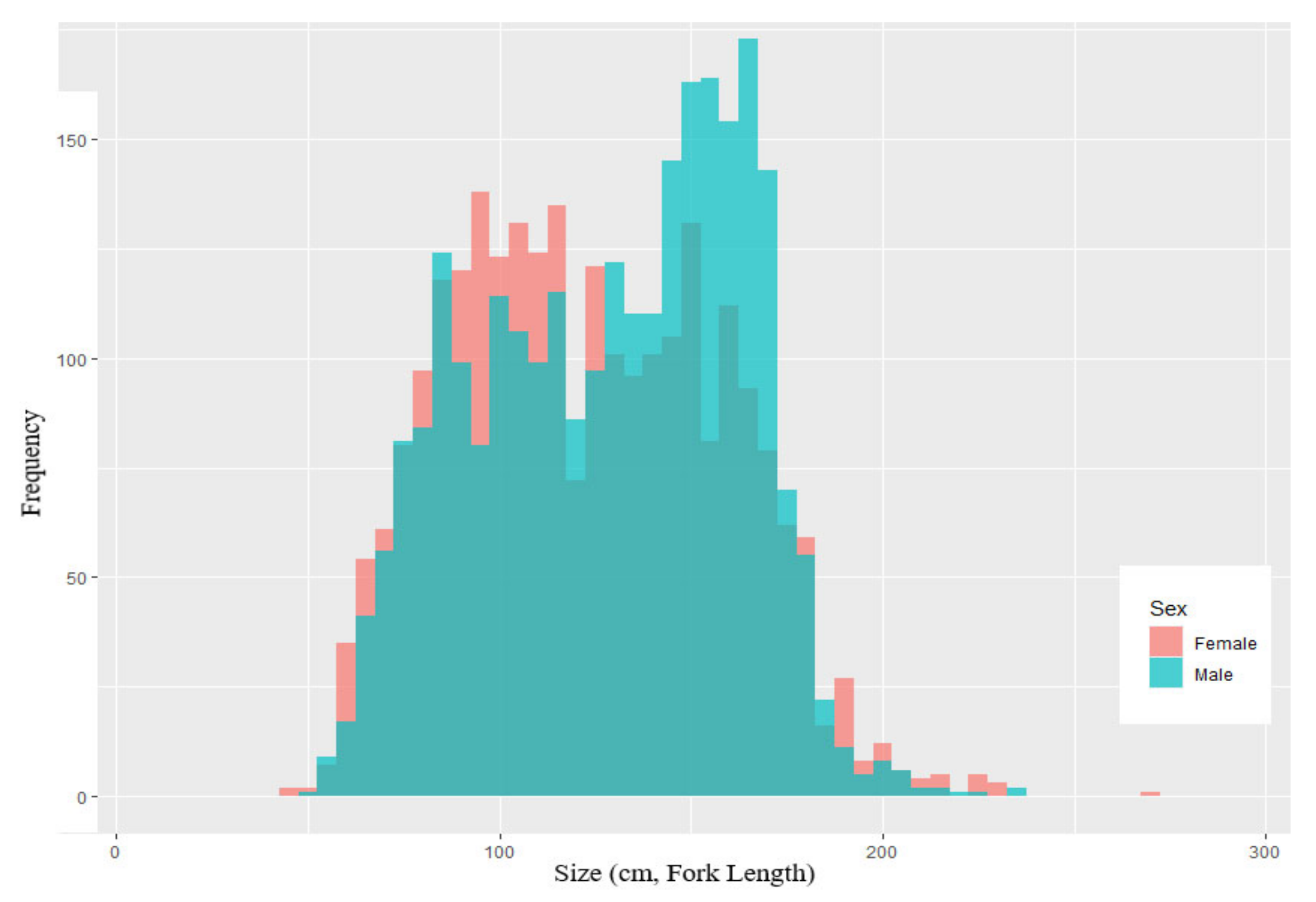
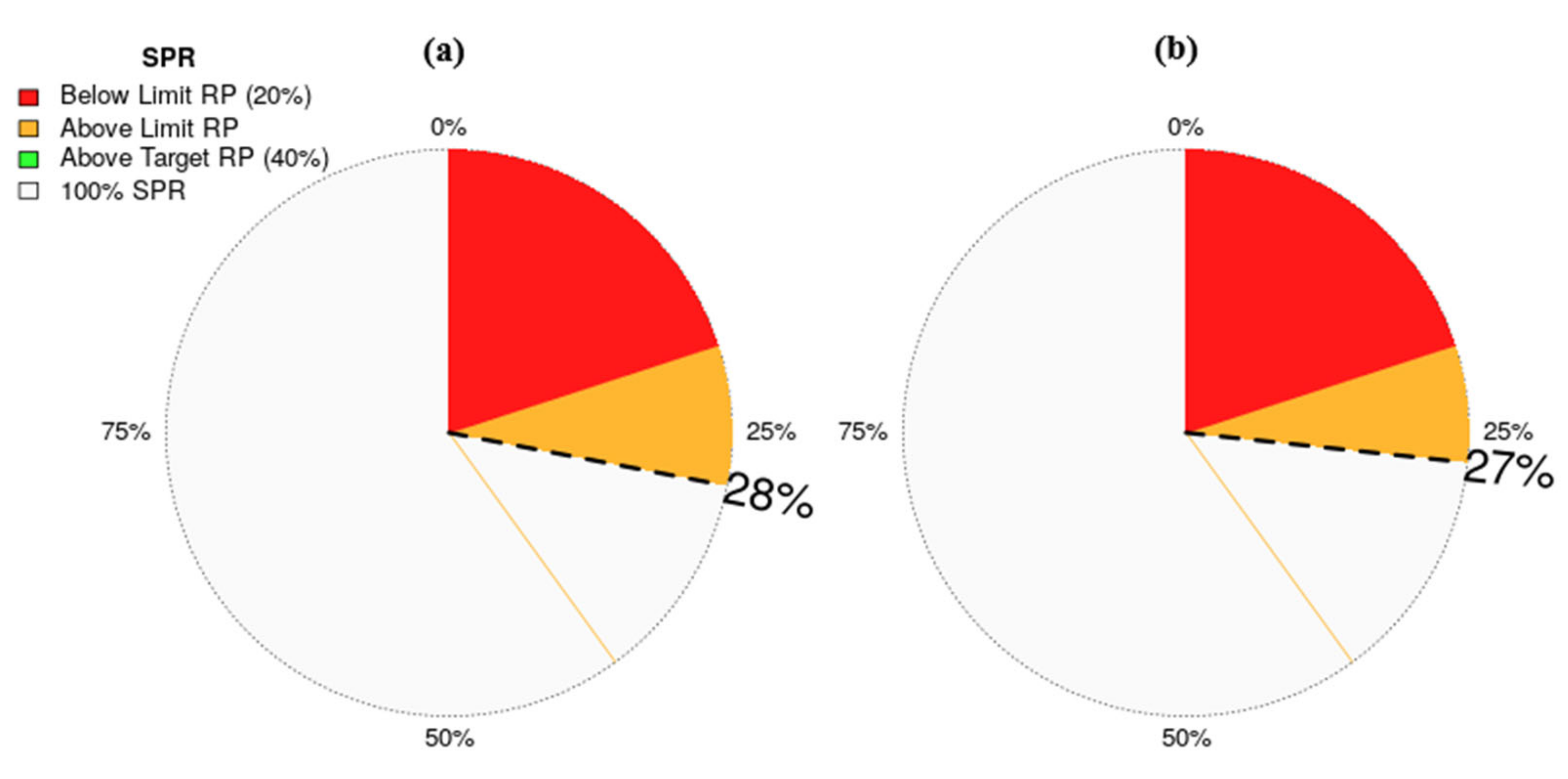
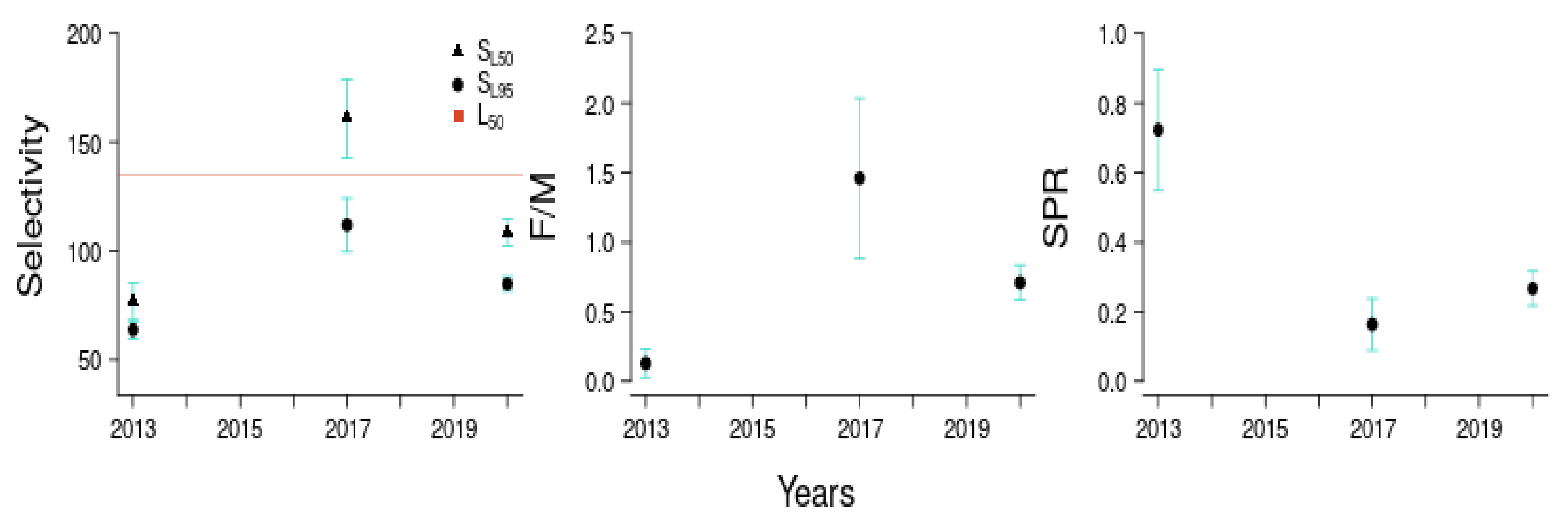
| Parameters | Symbol | Parameter Estimates | LBSPR | LBB |
|---|---|---|---|---|
| Length-Frequency Data | LFD | |||
| von Bertalanffy asymptotic Length | Linf (cm, FL) (cm, TL) | 318 (396.5) | ||
| von Bertalanffy growth parameter | K | 0.058 | ||
| Length where 50% of the fish are mature | L50% (cm, FL) | 135 | ||
| Length where 95% of the fish are mature | L95% (cm, FL) | 148.5 | ||
| Length–weight relationship parameter a | a | 0.00944 | ||
| Length–weight relationship parameter b | b | 3.094 | ||
| Natural mortality (median) | M (year−1) | 0.173 (0.077–0.257) | ||
| M/K invariant | M/K | 2.983 | ||
| Coefficient of variation of von Bertalanffy asymptotic length (Linf) | CV Linf | 0.1 |
| Acronym | Equations | Estimated | References |
|---|---|---|---|
| Pauly_Linf | 0.120 | [33] | |
| Alverson_Carney | 0.273 | [32] | |
| Then_1 | 0.257 | [36] | |
| Then_2 | 0.077 | [36] | |
| Hewitt Hoenig | 0.180 | [35] | |
| Hoenig | 0.167 | [34] |
| Length Composition Aggregated into One Year (2010–2020) | ||||
| Aggregated Year | SL50 (CI) | SL95 (CI) | F/M (CI) | SPR (CI) |
| 2010–2020 | 88.72 (84.11–93.33) | 122.23 (114.29–130.17) | 0.7 (0.57–0.83) | 0.28 (0.23–0.33) |
| Length Composition Aggregated into 3-Year Dataset | ||||
| Aggregated Years | SL50 (CI) | SL95 (CI) | F/M (CI) | SPR (CI) |
| 2010–2013 | 63.72 (59.67–67.77) | 76.76 (68.01–85.51) | 0.13 (0.03–0.23) | 0.72 (0.55–0.9) |
| 2014–2017 | 111.93 (99.65–124.21) | 160.86 (142.76–178.96) | 1.46 (0.88–2.04) | 0.16 (0.09–0.24) |
| 2018–2020 | 84.83 (81.53–88.13) | 108.49 (102.36–114.62) | 0.71 (0.58–0.84) | 0.27 (0.22–0.32) |
| Species Scientific Name | Linf (CI) | Lc/Lc_opt | F/M (CI) | Lc (CI) | B/BMSY (CI) | B/B0 (CI) | BMSY/B0 (CI) | Stock Status |
|---|---|---|---|---|---|---|---|---|
| Carcharhinus falciformis | 316 (309–324) | 0.77 | 2.1 (1.3–3.2) | 113 (107–130) | 0.54 (0.42–0.61) | 0.2 (0.14–0.36) | 0.44 (0.24–0.6) | Overfished |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kindong, R.; Sarr, O.; Wu, F.; Tian, S. Length-Based Assessment Methods for the Conservation of a Pelagic Shark, Carcharhinus falciformis from the Tropical Pacific Ocean. Fishes 2022, 7, 184. https://doi.org/10.3390/fishes7040184
Kindong R, Sarr O, Wu F, Tian S. Length-Based Assessment Methods for the Conservation of a Pelagic Shark, Carcharhinus falciformis from the Tropical Pacific Ocean. Fishes. 2022; 7(4):184. https://doi.org/10.3390/fishes7040184
Chicago/Turabian StyleKindong, Richard, Ousmane Sarr, Feng Wu, and Siquan Tian. 2022. "Length-Based Assessment Methods for the Conservation of a Pelagic Shark, Carcharhinus falciformis from the Tropical Pacific Ocean" Fishes 7, no. 4: 184. https://doi.org/10.3390/fishes7040184
APA StyleKindong, R., Sarr, O., Wu, F., & Tian, S. (2022). Length-Based Assessment Methods for the Conservation of a Pelagic Shark, Carcharhinus falciformis from the Tropical Pacific Ocean. Fishes, 7(4), 184. https://doi.org/10.3390/fishes7040184







