Identification and Expression Characterization of the Smad3 Gene and SNPs Associated with Growth Traits in the Hard Clam (Meretrix meretrix)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Collection and Preparation
2.2. Cloning of Full-Length cDNA and Promoter
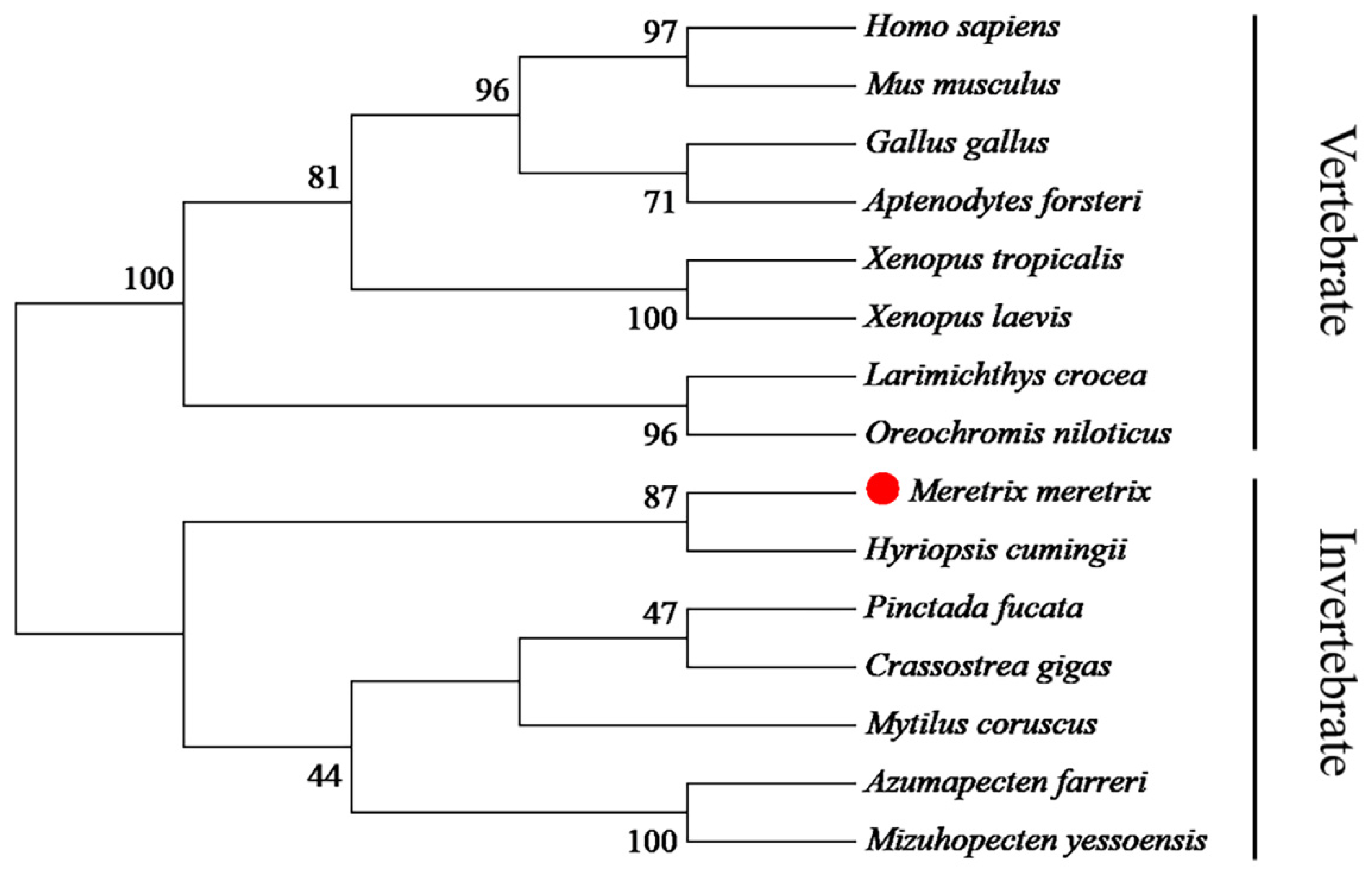
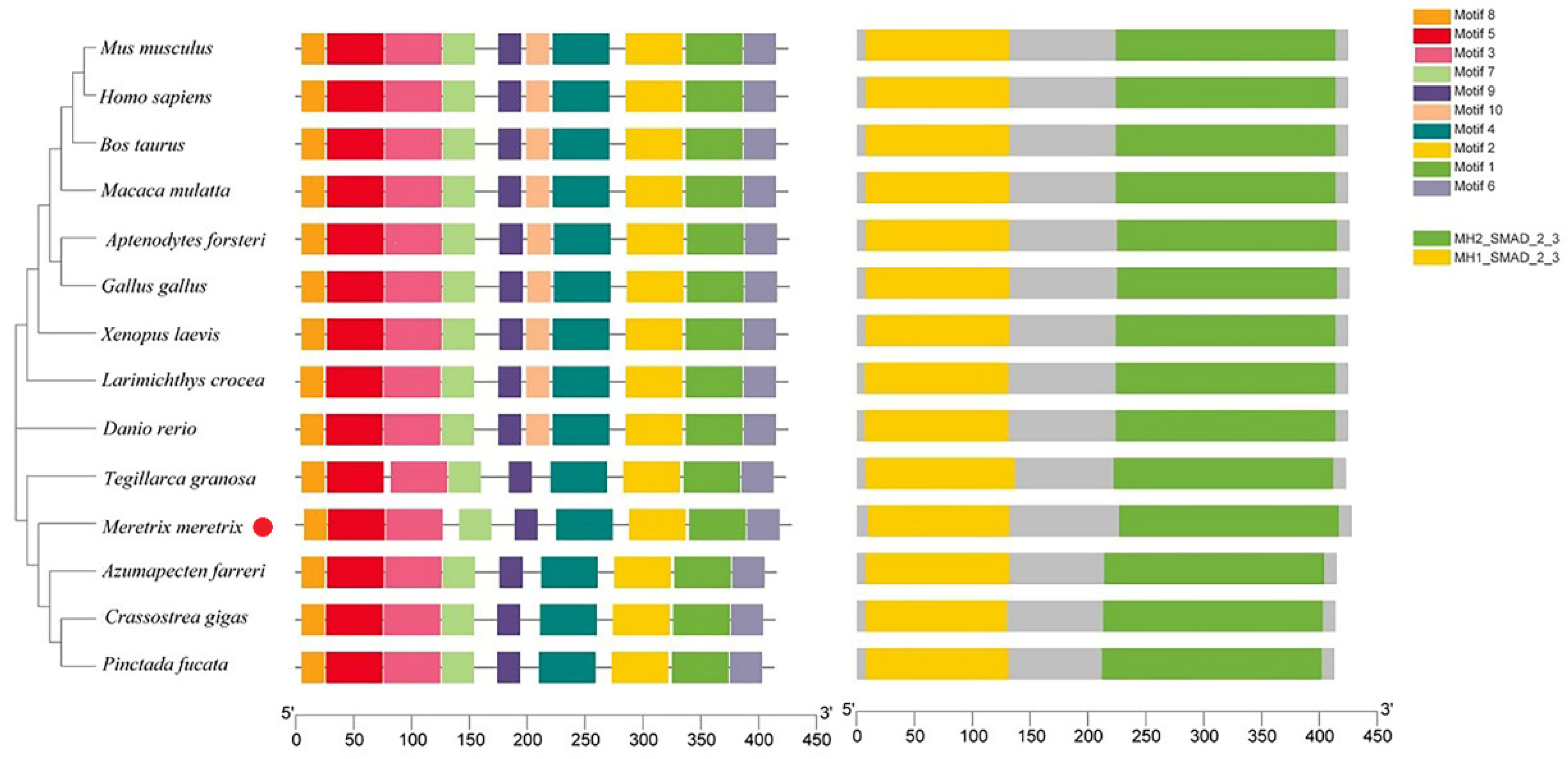
2.3. Sequence and Phylogenetic Analysis
2.4. Quantitative Expression Analysis
2.5. Mantle Repair Experiment
2.6. SNPs and Their Association with Growth Traits
2.7. Statistical Analysis
3. Results
3.1. cDNA and Promoter Sequence Analysis of Mm-Smad3
3.2. Tissue and Developmental Stage Quantitative Expression of Mm-Smad3
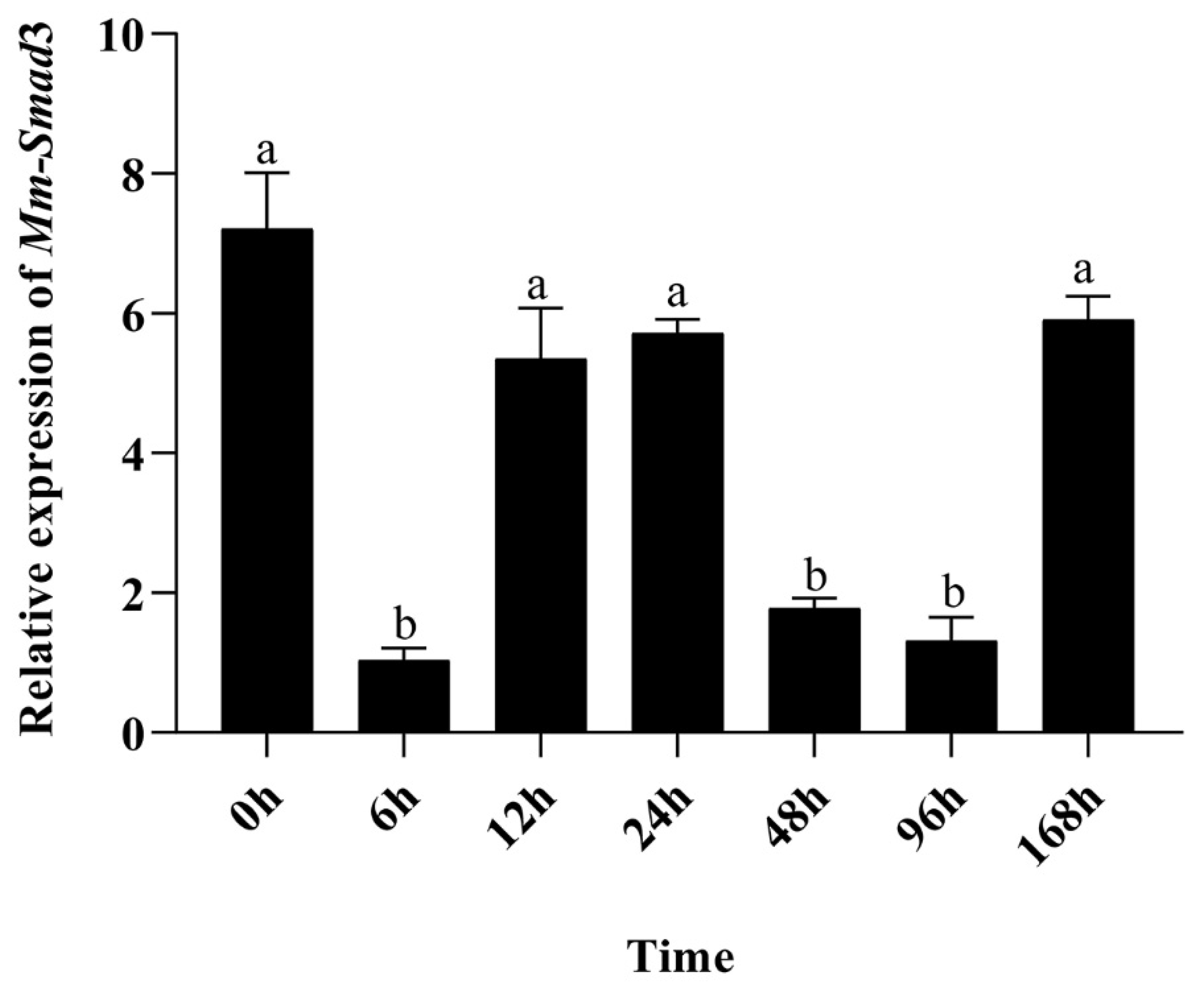
3.3. Time-Course Quantitative Expression of Mm-Smad3 after the Mantle Repair Experiment
3.4. Growth-Related SNPs in Mm-Smad3 Exons
4. Discussion
4.1. The Mm-Smad3 Gene and Amino Acid Sequence Features
4.2. Quantitative Expression Analysis of Mm-Smad3
4.3. Analysis of the Effect of Mm-Smad3 Gene on Tissue Repair
4.4. Association Analysis of SNPs with Growth Traits in the Mm-Smad3 Gene
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Moustakas, A.; Heldin, C.H. The regulation of TGF-β signal transduction. Development 2009, 136, 3699. [Google Scholar] [CrossRef] [Green Version]
- Shi, Y.; Massagué, J. Mechanisms of TGF-β signaling from cell membrane to the nucleus. Cell 2003, 113, 685–700. [Google Scholar] [CrossRef] [Green Version]
- Hata, A.; Chen, Y.G. TGF-β signaling from receptors to Smads. Cold Spring Harb. Perspect. Biol. 2011, 8, a022061. [Google Scholar] [CrossRef]
- Kjellman, C.; Honeth, G.; Järnum, S.; Lindvall, M.; Darabi, A.; Nilsson, I.; Edvardsen, K.; Salford, L.G.; Widegren, B. Identification and characterization of a human smad3 splicing variant lacking part of the linker region. Gene 2004, 327, 141–152. [Google Scholar] [CrossRef] [PubMed]
- Savage, C.; Das, P.; Finelli, A.L. Caenorhabditis elegans genes sma-2, sma-3, and sma-4 define a conserved family of transforming growth factor β pathway components. Proc. Natl. Acad. Sci USA 1996, 93, 790–794. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dick, A.; Mayr, T.; Bauer, H.; Meier, A.; Hammerschmidt, M. Cloning and characterization of zebrafish smad2, smad3 and smad4. Gene 2000, 246, 69–80. [Google Scholar] [CrossRef]
- Shi, T.; Xu, Y.; Yang, M.J.; Zhou, Y.; Liu, M.; Lan, X.Y.; Lei, C.Z.; Qi, X.L.; Lin, F.P.; Bai, Y.Y.; et al. Genetic variation, association analysis, and expression pattern of SMAD3 gene in Chinese cattle. Czech J. Anim. Sci. 2016, 61, 209–216. [Google Scholar] [CrossRef] [Green Version]
- Tian, Z.H.; Jiao, C.Z.; Cheng, Y.X. Molecular cloning, sequence analysis, and tissue expression of Smad3-like protein from Eriocheir sinensis. J. Fish Sci. China 2018, 25, 316–324. [Google Scholar] [CrossRef]
- Fu, L.G.; Liu, H.W.; Lei, W.J. MiR-596 inhibits osteoblastic differentiation and cell proliferation by targeting Smad3 in steroid-induced osteonecrosis of femoral head. J. Orthop. Surg. Res. 2020, 15, 1–10. [Google Scholar] [CrossRef]
- Zhang, K.; Rajput, S.K.; Lee, K.B.; Wang, D.; Huang, J.; Folger, J.K.; Knott, J.G.; Zhang, J.; Smith, G.W. Evidence supporting a role for SMAD2/3 in bovine early embryonic development: Potential implications for embryotropic actions of follistatin. Biol. Reprod. 2015, 93, 86. [Google Scholar] [CrossRef] [Green Version]
- Roberts, A.B.; Tian, F.; Byfield, S.D.; Stuelten, C.; Ooshima, A.; Saika, S.; Flanders, K.C. Smad3 is key to TGF-β-mediated epithelial-to-mesenchymal transition, fibrosis, tumor suppression and metastasis. Cytokine Growth Factor Rev. 2006, 17, 19–27. [Google Scholar] [CrossRef] [PubMed]
- de Kroon, L.M.G.; Narcisi, R.; Van den Akker, G.G.H.; Vitters, E.L.; Blaney Davidson, E.N.; Van Osch, G.J.V.M.; van der Kraan, P.M. SMAD3 and SMAD4 have a more dominant role than SMAD2 in TGFβ-induced chondrogenic differentiation of bone marrow-derived mesenchymal stem cells. Sci. Rep. 2017, 7, 43164. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pluijm, I.; Vliet, N.; Thusen, J.H.; Robertus, J.L.; Ridwan, Y.; Heijningen, P.M.; Thiel, B.S.; Vermeij, M.; Hoeks, S.; Buijs-Offerman, R.; et al. Defective connective tissue remodeling in Smad3 mice leads to accelerated aneurysmal growth through disturbed downstream TGF-β signaling. Ebiomedicine 2016, 12, 280–294. [Google Scholar] [CrossRef] [Green Version]
- Amanda, B.; Anna, R.P.; Kimberlee, T.; Craig, F.; Charlotte, P. Transforming growth factor-β1/Smad3-independent epithelial–mesenchymal transition in type I collagen glomerulopathy. Int. J. Nephrol. Renovasc. Dis. 2017, 10, 251. [Google Scholar]
- Turner, A.W.; Martinuk, A.; Silva, A.; Lau, P.; Nikpay, M.; Eriksson, P.; Folkersen, L.; Perisic, L.; Hedin, U.; Soubeyrand, S.; et al. Functional analysis of a novel genome-wide association study signal in SMAD3 that confers protection from coronary artery disease. Thromb. Vasc. Biol. 2016, 36, 972–983. [Google Scholar] [CrossRef] [Green Version]
- Patel, P.; Sekiguchi, Y.; Oh, K.H.; Patterson, S.E.; Kolb, M.R.J.; Margetts, P.J. Smad3-dependent and -independent pathways are involved in peritoneal membrane injury. Kidney. Int. 2010, 77, 319–328. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Y.J.; He, Z.X.; Li, Q.; Xie, L.P.; Zhang, R.Q. Cloning and expression pattern of a Smad3 homolog from the pearl oyster, Pinctada fucata. Acta. Biochim. Biophys. Sin. 2008, 40, 244–252. [Google Scholar] [CrossRef] [Green Version]
- Guo, H. Cloning and Expression Analysis of TGF-β/Smad Signaling Pathway Genes in Azumapecten farreri and Screening of SNP Sites Related to Growth Traits. Ph.D. Thesis, Ocean University of China, Qingdao, China, 2012. [Google Scholar]
- Dong, Y.H. High-Throughput Transcriptome Analysis of Tegillarca granosa and the Cloning and Expression of Growth-Related Genes. Ph.D. Thesis, Ocean University of China, Qingdao, China, 2012. [Google Scholar]
- Awaji, M.; Machii, A. Fundamental studies on in vivo and in vitro pearl formation—Contribution of outer epithelial cells of pearl oyster mantle and pearl sacs. Aquat. Biosci. Monogr. 2011, 4, 1–39. [Google Scholar] [CrossRef] [Green Version]
- Lin, Z.H. Biology and Culture Technology of Meretrix meretrix; Science Press: Beijing, China, 2015; pp. 1–10. [Google Scholar]
- Wang, H.; Chai, X.; Liu, B. Estimation of genetic parameters for growth traits in cultured clam Meretrix meretrix (Bivalvia: Veneridae) using the Bayesian method based on Gibbs sampling. Aquacult. Res. 2011, 42, 240–247. [Google Scholar] [CrossRef]
- Moreau, L.; Charcosset, A.; Hospital, F.; Gallais, A. Marker-assisted selection efficiency in populations of finite size. Genetics 1998, 148, 1353. [Google Scholar] [CrossRef]
- Hospital, F.; Moreau, L.; Lacoudre, F.; Charcosset, A.; Gallais, A. More on the efficiency of marker-assisted selection. Theor. Appl. Genet. 1997, 95, 1181–1189. [Google Scholar] [CrossRef]
- Gao, X.Y.; Dong, Y.H.; Shi, S.J.; Yao, H.H.; Ran, W.B.; Zhao, J.X.; Lin, Z.H. Cloning, spatiotemporal expression and SNPs identification of GRB2 gene in hard clam Meretrix meretrix. J. Fish China 2015, 39, 55–63. [Google Scholar]
- Wang, C.; Yao, Y.; Wang, H.X.; Liu, B.Z. Genetic diversity of the sulfotransferase-like gene and one nonsynonymous SNP associated with growth traits of clam, Meretrix meretrix. Mol. Biol. Rep. 2012, 39, 1323–1331. [Google Scholar] [CrossRef]
- Zou, L.; Liu, B.Z. Identification of a serum amyloid A gene and the association of SNPs with vibrio-resistance and growth traits in the clam Meretrix meretrix. Fish Shellfish. Immunol. 2015, 43, 301–309. [Google Scholar] [CrossRef]
- Tan, B.; Yang, S.L.; Yang, R.C.; Wang, X.P.; Zhou, M.D.; Zhou, X.; Yang, S.H. Application research progress of SMAD3 gene in livestock production. China Anim. Husb. Vet. Med. 2019, 46, 185–193. [Google Scholar]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Gjedrem, T. Trygve Selection and breeding programs in aquaculture. Aquacult. Res. 2005, 37, 287–299. [Google Scholar]
- Parmentier, I.; Portetelle, D.; Gengler, N.; Prandi, A.; Renaville, R. Candidate gene markers associated with somatotropic axis and milk selection. Domest. Anim. Endocrinol. 1999, 17, 139–148. [Google Scholar] [CrossRef]
- Parviz, M.; Hu, L.; Zhu, N.; Zea, B.; Saverio, B.; John, G.; Dimitris, K.; Li, C. SMAD3 prevents binding of NKX2.1 and FOXA1 to the SpB promoter through its MH1 and MH2 domains. Nucleic Acids Res. 2008, 36, 179–188. [Google Scholar]
- Wu, G.; Chen, Y.G.; Ozdamar, B.; Gyuricza, C.A.; Chong, P.A.; Wrana, J.L.; Massagué, J.; Shi, Y. Structural basis of Smad2 recognition by the smad anchor for receptor activation. Science 2000, 287, 92–97. [Google Scholar] [CrossRef]
- Randall, R.A.; Germain, S.; Inman, G.J.; Bates, P.A.; Hill, C. Different Smad2 partners bind a common hydrophobic pocket in Smad2 via a defined proline-rich motif. EMBO J. 2014, 21, 145–156. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dong, L.; Kang, J.; Derynck, R. TGF-β-activated Smad3 represses MEF2-dependent transcription in myogenic differentiation. EMBO J. 2004, 23, 1557–1566. [Google Scholar]
- Chai, N.; Li, W.X.; Wang, J.; Wang, Z.X.; Yang, S.M.; Wu, J.W. Structural basis for the Smad5 MH1 domain to recognize different DNA sequences. Nucleic Acids. Res. 2015, 43, 9051–9064. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Budd, A.; Mcdougall, C.; Green, K.; Degnan, B.M. Control of shell pigmentation by secretory tubules in the abalone mantle. Front. Zool. 2014, 11, 62–71. [Google Scholar] [CrossRef]
- Sandhya, S.; Subha, S.; Kumar, J.P.; Xiaojia, G.; Sudarsanareddy, L.; Desmond, M.F.C.; Walter, W.; Ravi, K.; Mridula, S. Myostatin augments muscle-specific ring finger protein-1 expression through an NF-kB independent mechanism in SMAD3 null muscle. Mol. Endocrinol. 2014, 28, 317–330. [Google Scholar]
- Qian, L.W.; Fourcaudot, A.B.; Yamane, K.; You, T.; Chan, R.K.; Leung, K.P. Exacerbated and prolonged inflammation impairs wound healing and increases scarring. Wound Repair Regen. 2016, 24, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.T.; Chen, J.S.; Wu, M.H.; Hsieh, I.S.; Liang, C.H.; Hsu, C.L.; Hong, T.M.; Chen, Y.L. Galectin-1 accelerates wound healing by regulating the neuropilin-1/smad3/nox4 pathway and ros production in myofibroblasts. J. Investig. Dermatol. 2015, 135, 258–268. [Google Scholar] [CrossRef] [Green Version]
- Tan, N.S.; Michalik, L.; Di-Poi, N.; Ng, C.Y.; Wahli, W. Essential role of Smad3 in the inhibition of inflammation-induced PPARβ/δ expression. EMBO J. 2014, 23, 4211–4221. [Google Scholar] [CrossRef] [Green Version]
- Yoneda, N.; Yasue, A.; Watanabe, T.; Tanaka, E. Down-regulation of Smad3 accelerates palatal wound repair. J. Dent. Res. 2013, 92, 716–720. [Google Scholar] [CrossRef] [PubMed]
- Nolte, M.; Margadant, C. Controlling immunity and inflammation through Integrin-dependent regulation of TGF-β. Trends Cell Biol. 2019, 30, 49–59. [Google Scholar] [CrossRef]
- Falanga, V.; Schrayer, D.; Cha, J.; Butmarc, J.; Roberts, A.B. Full-thickness wounding of the mouse tail as a model for delayed wound healing: Accelerated wound closure in Smad3 knock-out mice. Wound Repair Regen. 2010, 12, 320–326. [Google Scholar] [CrossRef]
- Nong, X.L.; Rajbanshi, G.; Chen, L.; Li, J.Q.; Li, Z.; Liu, T.T.; Chen, S.H.; Wei, G.; Li, J.S. Effect of artesunate and relation with TGF-β1 and SMAD3 signaling on experimental hypertrophic scar model in rabbit ear. Arch. Dermatol. Res. 2019, 311, 761–772. [Google Scholar] [CrossRef] [Green Version]
- Ulloa, P.E.; Rincón, G.; Islas-Trejo, A.; Araneda, C.; Iturra, P.; Neira, R.; Medrano, J.F. RNA sequencing to study gene expression and SNP variations associated with growth in zebrafish fed a plant protein-based diet. Mar. Biotechnol. 2015, 17, 353–363. [Google Scholar] [CrossRef] [PubMed]
- Yue, X.; Wang, H.X.; Huang, X.H.; Wang, C.; Chai, X.L.; Wang, C.D.; Liu, B.Z. Single nucleotide polymorphisms in i-type lysozyme gene and their correlation with vibrio-resistance and growth of clam Meretrix meretrix based on the selected resistance stocks. Fish Shellfish Immunol. 2012, 33, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Guo, M.; Wang, H.H.; Shao, Y.; Xing, R.L.; Zhao, X.L.; Zhang, W.W.; Li, C.H. Gene identification and antimicrobial activity analysis of a novel lysozyme from razor clam Sinonovacula constricta. Fish Shellfish Immunol. 2019, 89, 198–206. [Google Scholar] [CrossRef] [PubMed]
- Thongda, W.; Zhao, H.; Zhang, D.; Jescovitch, L.N.; Liu, M.; Guo, X.; Schrandt, M.; Powers, S.P.; Peatman, E. Development of SNP panels as a new tool to assess the genetic diversity, population structure, and parentage analysis of the Eastern Oyster (Crassostrea virginica). Mar. Biotechnol. 2018, 20, 385–395. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, L.M.; Zhang, H.Q.; Wang, W.J.; Zhao, Y. Association between SMAD3 gene rs12901499 polymorphism and knee osteoarthritis in a Chinese population. J. Clin. Lab. Anal. 2018, 32, e22383. [Google Scholar] [CrossRef] [Green Version]
- Mcknight, A.J.; Woodman, A.M.; Parkkonen, M.; Patterson, C.C.; Savage, D.A.; Forsblom, C.; Pettigrew, K.A.; Sadlier, D.; Groop, P.H.; Maxwell, A.P. Investigation of DNA polymorphisms in SMAD genes for genetic predisposition to diabetic nephropathy in patients with type 1 diabetes mellitus. Diabetologia 2009, 52, 844–849. [Google Scholar] [CrossRef] [Green Version]
- Greenwood, T.A.; Kelsoe, J.R. Promoter and intronic variants affect the transcriptional regulation of the human dopamine transporter gene. Genomics 2003, 82, 511–520. [Google Scholar] [CrossRef]
- Sarfaty, C.K.; Oh, J.M.; Kim, I.W.; Sauna, Z.E.; Calcagno, A.M.; Ambudkar, S.V.; Gottesman, M.M. A “silent” polymorphism in the MDR1 gene changes substrate specificity. Science 2007, 315, 525–528. [Google Scholar] [CrossRef] [Green Version]


| Primer | Primer Sequence 5′–3′ | Applications | Tm | Product Length |
|---|---|---|---|---|
| Mm-Smad3-GSP1 | ACAGAACACAGCAAGTAGAGATGACGC | 3′ RACE | 65.4 | 1019 bp |
| Mm-Smad3-GSP2 | CGTCATCTCTACTTGCTGTGTTCTGT | 5′ RACE | 65.4 | 954 bp |
| Mm-Smad3-F | AAATGGAGCGAAAAGGCAG | cDNA identification | 58 | 918 bp |
| Mm-Smad3-R | ACAGACAGTTGCTGGGTGCC | cDNA identification | 60.5 | |
| Mm-Smad3-qRT-F | CAAATGGAGCGAAAAGGCAG | qRT-PCR | 60.9 | 197 bp |
| Mm-Smad3-qRT-R | ATCGCCAAAGTCGGCAGTA | qRT-PCR | 59 | |
| 18s-F | CTATCAAATGTCTGCCCTATCACAC | Internal reference primer | 60 | 196 bp |
| 18s-R | TCCCGTATTGTTATTTTTCGTCACT | Internal reference primer | 61.9 | |
| Mm-Smad3-GSP3 | TTCACAAGGCTTTTCACTGCCTT | Promoter amplification | 63.1 | 646 bp |
| Mm-Smad3-GSP4 | TGTTCGGTTAGTTCCGTCTCTTCACG | Promoter amplification | 64.7 | |
| Mm-Smad3-GSP5 | GCATTTGGTGCTTGAATCTTGTTGTG | Nested amplification | 65.5 | 2761 bp |
| Mm-Smad3-GSP6 | CGTGAAGAGACGGAACTAACCGAACA | Nested amplification | 64.7 | 2175 bp |
| Mm-Smad3-esF1 | GACACAACAAGATTCAAGCACC | SNP detection | 57.2 | 838 bp |
| Mm-Smad3-esR1 | ACAGACAGTTGCTGGGTGCC | SNP detection | 60.5 | |
| Mm-Smad3-esF2 | AAATGGAGCGAAAAGGCAG | SNP detection | 58 | 1635 bp |
| Mm-Smad3-esR2 | TTGATGGACCAGGGAGGAC | SNP detection | 57.3 |
| Locus | Genotype | Sample | Percent (%) | Shell Length (mm) | Shell Height (mm) | Shell Width (mm) | Total Weight (g) |
|---|---|---|---|---|---|---|---|
| c.597 C > T | CC | 14 | 17.50 | 35.10 ± 2.99 a | 29.71 ± 2.46 a | 17.48 ± 1.99 a | 12.54 ± 3.96 a |
| CT | 37 | 46.25 | 36.29 ± 3.15 ab | 30.34 ± 2.78 ab | 18.08 ± 1.99 ab | 13.04 ± 3.83 ab | |
| TT | 29 | 36.25 | 38.52 ± 3.47 b | 32.29 ± 2.78 b | 19.32 ± 1.83 b | 15.59 ± 4.24 b | |
| c.660 C > T | CC | 64 | 80.00 | 37.34 ± 3.47 a | 31.29 ± 2.94 a | 18.74 ± 2.03 a | 14.39 ± 4.32 a |
| CT | 16 | 20.00 | 35.00 ± 2.83 b | 29.41 ± 2.26 b | 17.09 ± 1.54 ab | 11.55 ± 2.51 ab | |
| c.792 A > T | AA | 46 | 57.50 | 36.95 ± 3.35 ab | 31.00 ± 2.81 | 18.46 ± 1.87 ab | 13.75 ± 4.05 ab |
| AT | 23 | 28.75 | 38.12 ± 3.38 a | 31.98 ± 2.91 | 19.16 ± 2.14 a | 15.75 ± 4.34 a | |
| TT | 11 | 13.75 | 35.33 ± 2.69 b | 29.29 ± 2.08 | 17.20 ± 1.45 b | 11.34 ± 1.68 b |
| Locus | Allele Frequency | Ho | He | Ne | PIC |
|---|---|---|---|---|---|
| c.597 C > T | C 0.4062 T 0.5938 | 0.4625 | 0.4855 | 1.9321 | 0.3660 |
| c.660 C > T | C 0.9000 T 0.1000 | 0.2000 | 0.1811 | 1.2195 | 0.1638 |
| c.792 A > T | A 0.7188 T 0.2812 | 0.2875 | 0.4068 | 1.6787 | 0.3225 |
| Locus | c.597 C > T | c.660 C > T | c.792 A > T |
|---|---|---|---|
| c.597 C > T | 1.000 | 1.000 | |
| c.660 C > T | 0.076 | 1.000 | |
| c.792 A > T | 0.268 | 0.284 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fu, L.; Chi, Q.; Bao, Y.; Yao, H.; Lin, Z.; Dong, Y. Identification and Expression Characterization of the Smad3 Gene and SNPs Associated with Growth Traits in the Hard Clam (Meretrix meretrix). Fishes 2021, 6, 83. https://doi.org/10.3390/fishes6040083
Fu L, Chi Q, Bao Y, Yao H, Lin Z, Dong Y. Identification and Expression Characterization of the Smad3 Gene and SNPs Associated with Growth Traits in the Hard Clam (Meretrix meretrix). Fishes. 2021; 6(4):83. https://doi.org/10.3390/fishes6040083
Chicago/Turabian StyleFu, Lulu, Qiudie Chi, Yongbo Bao, Hanhan Yao, Zhihua Lin, and Yinghui Dong. 2021. "Identification and Expression Characterization of the Smad3 Gene and SNPs Associated with Growth Traits in the Hard Clam (Meretrix meretrix)" Fishes 6, no. 4: 83. https://doi.org/10.3390/fishes6040083
APA StyleFu, L., Chi, Q., Bao, Y., Yao, H., Lin, Z., & Dong, Y. (2021). Identification and Expression Characterization of the Smad3 Gene and SNPs Associated with Growth Traits in the Hard Clam (Meretrix meretrix). Fishes, 6(4), 83. https://doi.org/10.3390/fishes6040083






