Determining Stingray Movement Patterns in a Wave-Swept Coastal Zone Using a Blimp for Continuous Aerial Video Surveillance
Abstract
1. Introduction
2. Results
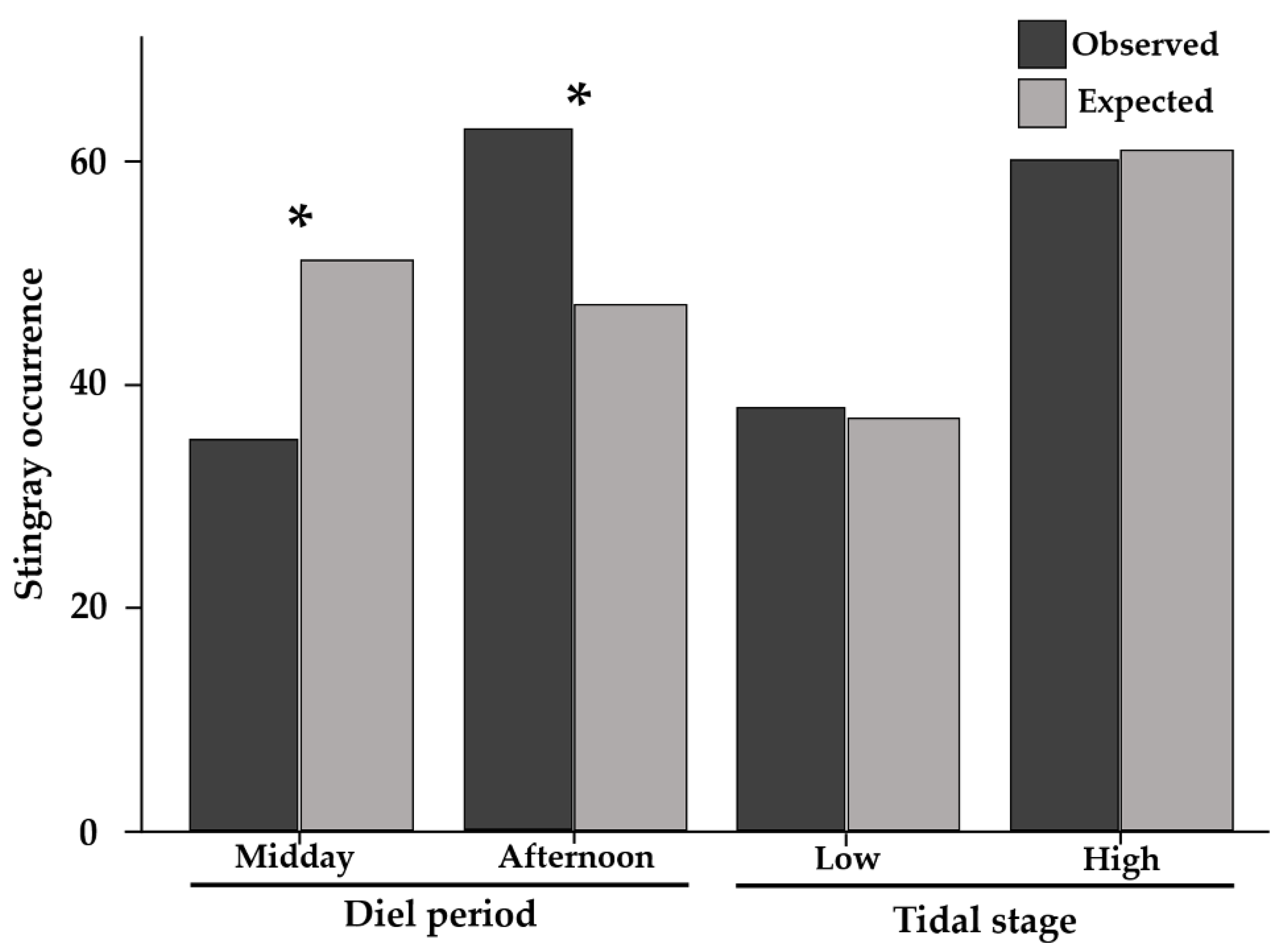
2.1. Stingray Occurence
2.2. Patterns of Movement
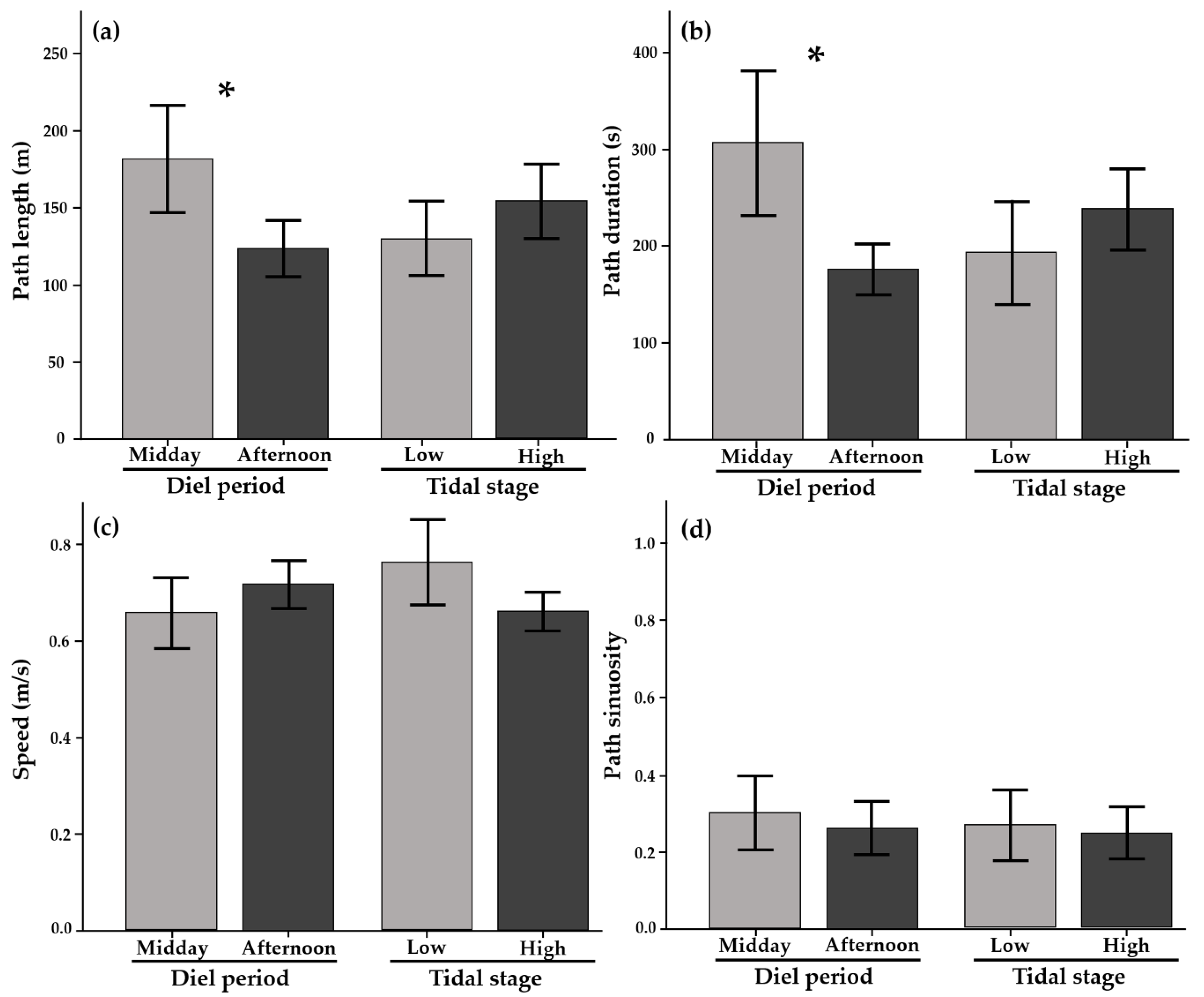
2.2.1. Movement Metrics
2.2.2. Route Fidelity
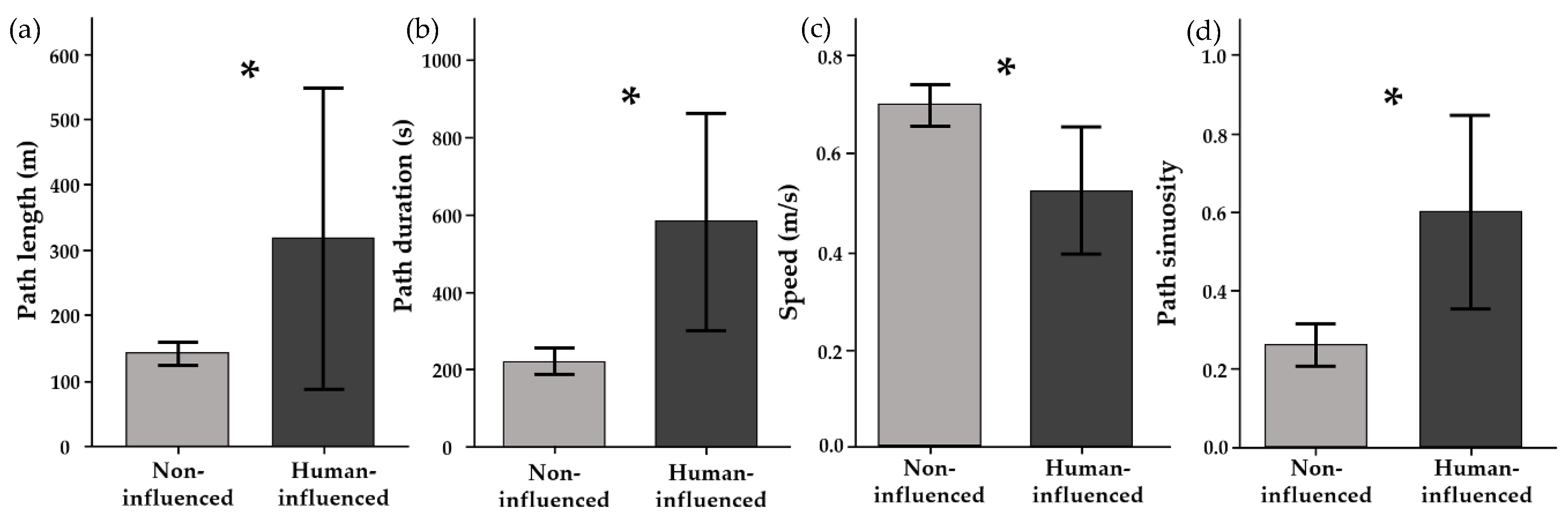
2.3. Human Influence on Stingray Movement Paths
3. Discussion
4. Materials and Methods
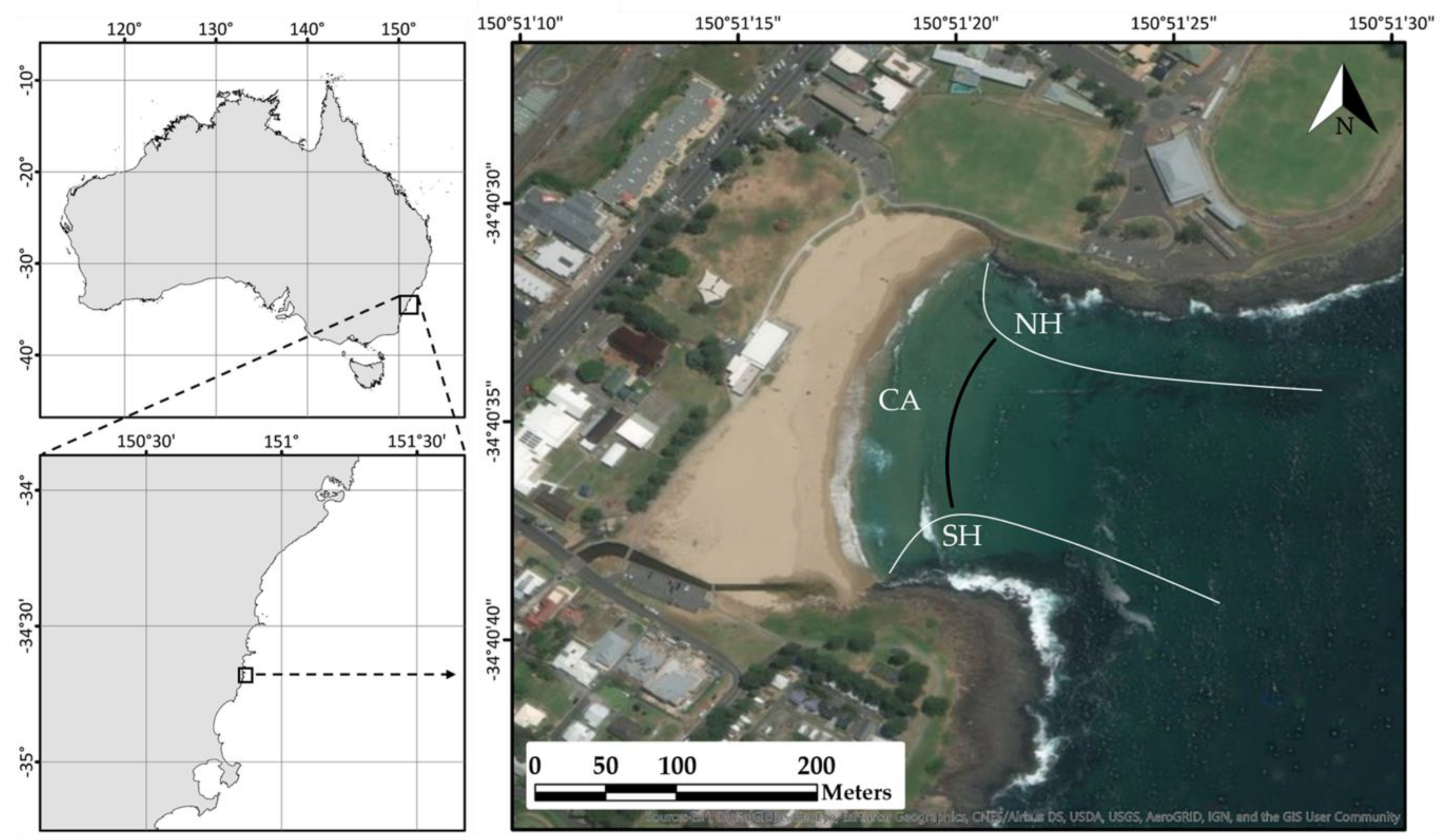
4.1. Study Area
4.2. Data Collection
4.3. Stingray Occurrence
4.4. Movement Metrics
4.5. Route Fidelity
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Navia, A.F.; Mejía-Falla, P.A.; López-García, J.; Giraldo, A.; Cruz-Escalona, V. How many trophic roles can elasmobranchs play in a marine tropical network? Mar. Freshw. Res. 2017, 68, 1342–1353. [Google Scholar] [CrossRef]
- Martins, A.P.B.; Heupel, M.R.; Chin, A.; Simpfendorfer, C.A. Batoid nurseries: Definition, use and importance. Mar. Ecol. Prog. Ser. 2018, 595, 253–267. [Google Scholar] [CrossRef]
- O’Shea, O.R.; Thums, M.; van Keulen, M.; Meekan, M. Bioturbation by stingrays at Ningaloo Reef, Western Australia. Mar. Freshw. Res. 2012, 63, 189–197. [Google Scholar] [CrossRef]
- Hammerschlag, N.; Schmitz, O.J.; Flecker, A.S.; Lafferty, K.D.; Sih, A.; Atwood, T.B.; Gallagher, A.J.; Irschick, D.J.; Skubel, R.; Cooke, S.J. Ecosystem Function and Services of Aquatic Predators in the Anthropocene. Trends Ecol. Evol. 2019, 34, 369–383. [Google Scholar] [CrossRef] [PubMed]
- Sheaves, M. Consequences of ecological connectivity: The coastal ecosystem mosaic. Mar. Ecol. Prog. Ser. 2009, 391, 107–115. [Google Scholar] [CrossRef]
- Dulvy, N.K.; Fowler, S.L.; Musick, J.A.; Cavanagh, R.D.; Kyne, P.M.; Harrison, L.R.; Carlson, J.K.; Davidson, L.N.K.; Fordham, S.V.; White, W.T.; et al. Extinction risk and conservation of the world’s sharks and rays. eLife 2014, 3, e00590. [Google Scholar] [CrossRef] [PubMed]
- Cortés, E. Life History Patterns and Correlations in Sharks. Rev. Fish Sci. 2000, 8, 299–344. [Google Scholar] [CrossRef]
- Knip, D.M.; Heupel, M.R.; Simpfendorfer, C.A. Sharks in nearshore environments: Models, importance, and consequences. Mar. Ecol. Prog. Ser. 2010, 402, 1–11. [Google Scholar] [CrossRef]
- Davy, L.E.; Simpfendorfer, C.A.; Heupel, M.R. Movement patterns and habitat use of juvenile mangrove whiprays (Himantura granulata). Mar. Freshw. Res. 2015, 66, 481–492. [Google Scholar] [CrossRef]
- Owoeye, K.; Musolesi, M.; Hailles, S. Characterizing animal movement patterns across different scales and habitats using information theory. bioRxiv 2018, 311241. [Google Scholar] [CrossRef]
- Sequeira, A.M.M.; Rodríguez, J.P.; Eguíluz, V.M.; Harcourt, R.; Hindell, M.; Sims, D.W.; Duarte, C.M.; Costa, D.P.; Fernández-Gracia, J.; Thums, M.; et al. Convergence of marine megafauna movement patterns in coastal and open oceans. Proc. Natl. Acad. Sci. USA 2018, 115, 3072–3077. [Google Scholar] [CrossRef] [PubMed]
- Chapman, D.D.; Feldheim, K.A.; Papastamatiou, Y.P.; Hueter, R.E. There and Back Again: A Review of Residency and Return Migrations in Sharks, with Implications for Population Structure and Management. Annu. Rev. Mar. Sci. 2015, 7, 547–570. [Google Scholar] [CrossRef] [PubMed]
- Legare, B.; Skomal, G.; DeAngelis, B. Diel movements of the blacktip shark (Carcharhinus limbatus) in a Caribbean nursery. Environ. Biol. Fishes 2018, 101, 1011–1023. [Google Scholar] [CrossRef]
- Morales, J.M.; Moorcroft, P.R.; Matthiopoulos, J.; Frair, J.L.; Kie, J.G.; Powell, R.A.; Merrill, E.H.; Haydon, D.T. Building the bridge between animal movement and population dynamics. Philos. Trans. R. Soc. B 2010, 365, 2289–2301. [Google Scholar] [CrossRef]
- Signer, J.; Ovaskainen, O. Detecting the influence of environmental covariates on animal movement: A semivariance approach. Methods Ecol. Evol. 2017, 8, 561–570. [Google Scholar] [CrossRef]
- Hays, G.C.; Ferreira, L.C.; Sequeira, A.M.M.; Meekan, M.G.; Duarte, C.M.; Bailey, H.; Bailleul, F.; Bowen, W.D.; Caley, M.J.; Thums, M.; et al. Key Questions in Marine Megafauna Movement Ecology. Trends Ecol. Evol. 2016, 31, 463–475. [Google Scholar] [CrossRef]
- Le Port, A.; Sippel, T.; Montgomery, J.C. Observations of mesoscale movements in the short-tailed stingray, Dasyatis brevicaudata from New Zealand using a novel PSAT tag attachment method. J. Exp. Mar. Biol. Ecol. 2008, 359, 110–117. [Google Scholar] [CrossRef]
- Brinton, C.P.; Curran, M.C. Tidal and diel movement patterns of the Atlantic stingray (Dasyatis sabina) along a stream-order gradient. Mar. Freshw. Res. 2017, 68, 1716–1725. [Google Scholar] [CrossRef]
- Martins, A.P.B.; Heupel, M.R.; Bierwagen, S.L.; Chin, A.; Simpfendorfer, C. Diurnal activity patterns and habitat use of juvenile Pastinachus ater in a coral reef flat environment. PLoS ONE 2020, 15, e0228280. [Google Scholar] [CrossRef]
- Vaudo, J.J.; Heithaus, M.R. Diel and seasonal variation in the use of a nearshore sandflat by a ray community in a near pristine system. Mar. Freshw. Res. 2012, 63, 1077–1084. [Google Scholar] [CrossRef]
- Huveneers, C.; Simpfendorfer, C.A.; Kim, S.; Semmens, J.M.; Hobday, A.J.; Pederson, H.; Stieglitz, T.; Vallee, R.; Webber, D.; Harcourt, R.G.; et al. The influence of environmental parameters on the performance and detection range of acoustic receivers. Methods Ecol. Evol. 2016, 7, 825–835. [Google Scholar] [CrossRef]
- Martins, A.P.B.; Heupel, M.R.; Oakley-Cogan, A.; Chin, A.; Simpfendorfer, C.A. Towed-float GPS telemetry: A tool to assess movement patterns and habitat use of juvenile stingrays. Mar. Freshw. Res. 2020, 71, 89–98. [Google Scholar] [CrossRef]
- Raoult, V.; Tosetto, L.; Williamson, J.E. Drone-based high-resolution tracking of aquatic vertebrates. Drones 2018, 2, 37. [Google Scholar] [CrossRef]
- Wilson, R.P.; Liebsch, N.; Davies, I.M.; Quintana, F.; Weimerskirch, H.; Storch, S.; Lucke, K.; Siebert, U.; Zankl, S.; McMahon, C.R.; et al. All at sea with animal tracks; methodological and analytical solutions for the resolution of movement. Deep Sea Res. Part II 2007, 54, 193–210. [Google Scholar] [CrossRef]
- Wilson, A.D.M.; Brownscombe, J.W.; Krause, J.; Krause, S.; Gutowsky, L.F.G.; Brooks, E.J.; Cooke, S.J. Integrating network analysis, sensor tags, and observation to understand shark ecology and behavior. Behav. Ecol. 2015, 26, 1577–1586. [Google Scholar] [CrossRef]
- Bicknell, A.W.J.; Godley, B.J.; Sheehan, E.V.; Votier, S.C.; Witt, M.J. Camera technology for monitoring marine biodiversity and human impact. Front. Ecol. Environ. 2016, 14, 424–432. [Google Scholar] [CrossRef]
- Adams, K.; Broad, A.; Ruiz-García, D.; Davis, A.R. Continuous wildlife monitoring using blimps as an aerial platform: A case study observing marine megafauna. Aust. Zool. 2020, 40, 407–415. [Google Scholar] [CrossRef]
- Colefax, A.P.; Butcher, P.A.; Kelaher, B.P. The potential for unmanned aerial vehicles (UAVs) to conduct marine fauna surveys in place of manned aircraft. ICES J. Mar. Sci. 2017, 75, 1–8. [Google Scholar] [CrossRef]
- Rieucau, G.; Kiszka, J.J.; Castillo, J.C.; Mourier, J.; Boswell, K.M.; Heithaus, M.R. Using unmanned aerial vehicle (UAV) surveys and image analysis in the study of large surface-associated marine species: A case study on reef sharks Carcharhinus melanopterus shoaling behaviour. J. Fish Biol. 2018, 93, 119–127. [Google Scholar] [CrossRef]
- Bevan, E.; Whiting, S.; Tucker, T.; Guinea, M.; Raith, A.; Douglas, R. Measuring behavioral responses of sea turtles, saltwater crocodiles, and crested terns to drone disturbance to define ethical operating thresholds. PLoS ONE 2018, 13, e0194460. [Google Scholar] [CrossRef]
- Kanno, S.; Schlaff, A.M.; Heupel, M.R.; Simpfendorfer, C.A. Stationary video monitoring reveals habitat use of stingrays in mangroves. Mar. Ecol. Prog. Ser. 2019, 621, 155–168. [Google Scholar] [CrossRef]
- Mulero-Pázmány, M.; Jenni-Eiermann, S.; Strebel, N.; Sattler, T.; Negro, J.J.; Tablado, Z. Unmanned aircraft systems as a new source of disturbance for wildlife: A systematic review. PLoS ONE 2017, 12, e0178448. [Google Scholar] [CrossRef] [PubMed]
- Erbe, C.; Williams, R.; Parsons, M.; Parsons, S.K.; Hendrawan, I.G.; Dewantama, I.M.I. Underwater noise from airplanes: An overlooked source of ocean noise. Mar. Pollut. Bull. 2018, 137, 656–661. [Google Scholar] [CrossRef] [PubMed]
- Hassanalian, M.; Abdelkefi, A. Classifications, applications, and design challenges of drones: A review. Prog. Aerosp. Sci. 2017, 91, 99–131. [Google Scholar] [CrossRef]
- Hain, J.H.; Harris, L.E. Aerostats for oceanographic and atmospheric research. Sea Technol. 2004, 45, 75–80. [Google Scholar]
- Hodgson, A. “BLIMP-CAM”: Aerial Video Observations of Marine Animals. Mar. Technol. Soc. J. 2007, 41, 39–43. [Google Scholar] [CrossRef]
- Nosal, A.P.; Cartamil, D.C.; Long, J.W.; Lührmann, M.; Wegner, N.C.; Graham, J.B. Demography and movement patterns of leopard sharks (Triakis semifasciata) aggregating near the head of a submarine canyon along the open coast of southern California, USA. Environ. Biol. Fishes 2013, 96, 865–878. [Google Scholar] [CrossRef]
- Tilley, A.; López-Angarita, J.; Turner, J.R. Effects of scale and habitat distribution on the movement of the southern stingray Dasyatis americana on a Caribbean atoll. Mar. Ecol. Prog. Ser. 2013, 482, 169–179. [Google Scholar] [CrossRef]
- Zollner, P.A.; Lima, S.L. Search strategies for landscape-level interpatch movements. Ecology 1999, 80, 1019–1030. [Google Scholar] [CrossRef]
- Benhamou, S. How to reliably estimate the tortuosity of an animal’s path: Straightness, sinuosity, or fractal dimension? J. Theor. Biol. 2004, 229, 209–220. [Google Scholar] [CrossRef]
- Van Moorter, B.; Visscher, D.; Benhamou, S.; Börger, L.; Boyce, M.S.; Gaillard, J. Memory keeps you at home: A mechanistic model for home range emergence. Oikos 2009, 118, 641–652. [Google Scholar] [CrossRef]
- Papastamatiou, Y.P.; Cartamil, D.P.; Lowe, C.G.; Meyer, C.G.; Wetherbee, B.M.; Holland, K.N. Scales of orientation, directed walks and movement path structure in sharks. J. Anim. Ecol. 2011, 80, 864–874. [Google Scholar] [CrossRef] [PubMed]
- Hollensead, L.D.; Grubbs, R.D.; Carlson, J.K.; Bethea, D.M. Analysis of fine-scale daily movement patterns of juvenile Pristis pectinata within a nursery habitat. Aquat. Conserv. Mar. Freshw. Ecosyst. 2016, 26, 492–505. [Google Scholar] [CrossRef]
- Bartumeus, F.; da Luz, M.G.E.; Viswanathan, G.M.; Catalan, J. Animal search strategies: A quantitative random-walk analysis. Ecology 2005, 86, 3078–3087. [Google Scholar] [CrossRef]
- Papastamatiou, Y.P.; De Salles, P.A.; McCauley, D.J. Area-restricted searching by manta rays and their response to spatial scale in lagoon habitats. Mar. Ecol. Prog. Ser. 2012, 456, 233–244. [Google Scholar] [CrossRef]
- Meese, E.N.; Lowe, C.G. Active acoustic telemetry tracking and tri-axial accelerometers reveal fine-scale movement strategies of a non-obligate ram ventilator. Mov. Ecol. 2020, 8, 8. [Google Scholar] [CrossRef]
- Cartamil, D.P.; Vaudo, J.J.; Lowe, C.G.; Wetherbee, B.M.; Holland, K.N. Diel movement patterns of the Hawaiian stingray, Dasyatis lata: Implications for ecological interactions between sympatric elasmobranch species. Mar. Biol. 2003, 142, 841–847. [Google Scholar] [CrossRef]
- Ward, C.R.E.; Bouyoucos, I.A.; Brooks, E.J.; O’Shea, O.R. Novel attachment methods for assessing activity patterns using triaxial accelerometers on stingrays in the Bahamas. Mar. Biol. 2019, 166, 53. [Google Scholar] [CrossRef]
- Rizzari, J.R.; Semmens, J.M.; Fox, A.; Huveneers, C. Observations of marine wildlife tourism effects on a non-focal species. J. Fish Biol. 2017, 91, 981–988. [Google Scholar] [CrossRef]
- Gilliam, D.S.; Sullivan, K.M. Diet and feeding habits of the southern stingray Dasyatis americana in the central Bahamas. Bull. Mar. Sci. 1993, 52, 1007. Available online: https://nsuworks.nova.edu/occ_facarticles/560 (accessed on 25 September 2020).
- Newsome, D.; Lewis, A.; Moncrieff, D. Impacts and risks associated with developing, but unsupervised, stingray tourism at Hamelin Bay, Western Australia. Int. J. Tour. Res. 2004, 6, 305–323. [Google Scholar] [CrossRef]
- Pini-Fitzsimmons, J.; Knott, N.A.; Brown, C. Effects of food provisioning on site use in the short-tail stingray Bathytoshia brevicaudata. Mar. Ecol. Prog. Ser. 2018, 600, 99–110. [Google Scholar] [CrossRef]
- NSW DPI. NSW Recreational Saltwater Fishing Guide 2020–2021; New South Wales Department of Primary Industries: Nowra, NSW, Australia, 2020. Available online: https://www.dpi.nsw.gov.au/__data/assets/pdf_file/0012/600222/16359-NSW-Recreational-Fishing-Guides-Saltwater-2020-07Winter.pdf (accessed on 22 July 2020).
- Fist, R. Stingray Show a Hit in Kiama; Kiama Independent: Kiama, NSW, Australia, 2018; Available online: https://www.kiamaindependent.com.au/story/5165842/stingray-show-a-hit-in-kiama/ (accessed on 25 September 2020).
- Gorkin, R.; Adams, K.; Berryman, M.J.; Aubin, S.; Li, W.; Davis, A.R.; Barthelemy, J. Sharkeye: Real-Time Autonomous Personal Shark Alerting via Aerial Surveillance. Drones 2020, 4, 18. [Google Scholar] [CrossRef]
- Lemos, C.; Floc’h, F.; Yates, M.; Le Dantec, N.; Marieu, V.; Hamon, K.; Cuq, V.; Suanez, S.; Delacourt, C. Equilibrium modeling of the beach profile on a macrotidal embayed low tide terrace beach. Ocean Dyn. 2018, 68, 1207–1220. [Google Scholar] [CrossRef]
- Last, P.R.; Naylor, G.J.P.; Manjaji-Matsumoto, B. A revised classification of the family Dasyatidae (Chondrichthyes: Myliobatiformes) based on new morphological and molecular insights. Zootaxa 2016, 4139, 368. [Google Scholar] [CrossRef]
- R Development Core Team. R: A Language and Environment for Statistical Computing, R version 3.6.1; R Development Core Team: Vienna, Austria, 2019; Available online: https://www.r-project.org/ (accessed on 7 August 2019).
- Mangiafico, S.S. An R Companion for the Handbook of Biological Statistics, version 1.3.3; Rutgers Cooperative Extension: New Brunswick, NJ, USA, 2015; Available online: https://rcompanion.org/rcompanion/ (accessed on 25 September 2020).
- Australia’s Official Weather Forecasts & Weather Radar: Bureau of Meteorology. Available online: www.bom.gov.au (accessed on 7 August 2019).
- Compute Sunrise, Sunset & Twilight Times: Geoscience Australia. Available online: www.ga.gov.au/geodesy/astro/sunrise.jsp (accessed on 7 August 2019).


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruiz-García, D.; Adams, K.; Brown, H.; Davis, A.R. Determining Stingray Movement Patterns in a Wave-Swept Coastal Zone Using a Blimp for Continuous Aerial Video Surveillance. Fishes 2020, 5, 31. https://doi.org/10.3390/fishes5040031
Ruiz-García D, Adams K, Brown H, Davis AR. Determining Stingray Movement Patterns in a Wave-Swept Coastal Zone Using a Blimp for Continuous Aerial Video Surveillance. Fishes. 2020; 5(4):31. https://doi.org/10.3390/fishes5040031
Chicago/Turabian StyleRuiz-García, David, Kye Adams, Heidi Brown, and Andrew R. Davis. 2020. "Determining Stingray Movement Patterns in a Wave-Swept Coastal Zone Using a Blimp for Continuous Aerial Video Surveillance" Fishes 5, no. 4: 31. https://doi.org/10.3390/fishes5040031
APA StyleRuiz-García, D., Adams, K., Brown, H., & Davis, A. R. (2020). Determining Stingray Movement Patterns in a Wave-Swept Coastal Zone Using a Blimp for Continuous Aerial Video Surveillance. Fishes, 5(4), 31. https://doi.org/10.3390/fishes5040031





