Salmon Louse (Lepeophtheirus salmonis (Krøyer)) Control Methods and Efficacy in Atlantic Salmon (Salmo salar (Linnaeus)) Aquaculture: A Literature Review
Abstract
1. Introduction
2. Results
2.1. Chemical Treatments
2.2. Cleaner Fish
2.3. Other Non-Chemical Treatments and Preventive Measures
3. Discussion
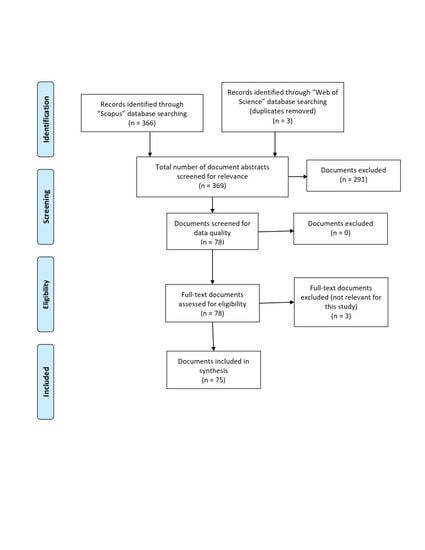
4. Materials and Methods
4.1. Data Collection
4.2. Data Extraction and Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Abolofia, J.; Asche, F.; Wilen, J.E. The cost of lice: Quantifying the impact of parasitic sea lice on farmed salmon. Mar. Resour. 2017, 32, 329–349. [Google Scholar] [CrossRef]
- Grave, K.; Horsberg, T.E.; Lunestad, B.T.; Litleskare, I. Consumption of drugs for sea lice infestations in Norwegian fish farms: Methods for assessment of treatment patterns and treatment rate. Dis. Aquat. Organ. 2004, 60, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Hamre, L.A.; Eichner, C.; Caipang, C.M.A.; Dalvin, S.T.; Bron, J.E.; Nilsen, F.; Boxshall, G.; Skern-Mauritzen, R. The salmon louse Lepeophtheirus salmonis (Copepoda: Caligidae) life cycle has only two chalimus stages. PLoS ONE 2013, 8, e73539. [Google Scholar] [CrossRef] [PubMed]
- Macken, A.; Lillicrap, A.; Langford, K. Benzoylurea pesticides used as veterinary medicines in aquaculture: Risks and developmental effects on nontarget crustaceans. Environ. Toxicol. Chem. 2015, 34, 1533–1542. [Google Scholar] [CrossRef]
- Thorstad, E.B.; Todd, C.D.; Uglem, I.; Bjørn, P.A.; Gargan, P.G.; Vollset, K.W.; Halttunen, E.; Kls, S.; Berg, M.; Finstad, B. Effects of salmon lice Lepeophtheirus salmonis on wild sea trout Salmo trutta—A literature review. Aquacult. Environ. Interact. 2015, 7, 91–113. [Google Scholar] [CrossRef]
- Overton, K.; Dempster, T.; Oppedal, F.; Kristiansen, T.S.; Gismervik, K.; Stien, L.H. Salmon lice treatments and salmon mortality in Norwegian aquaculture: A review. Rev. Aquacult. 2019. [Google Scholar] [CrossRef]
- Helgesen, K.O.; Horsberg, T.E.; Tarpai, A. The Surveillance Programme for Resistance to Chemoterapeutants in Salmon Lice (Lepeophtheirus salmonis) in Norway 2018; Norwegian Veterinary Institute: Oslo, Norway, 2018; pp. 1–18. [Google Scholar]
- Gharbi, K.; Matthews, L.; Bron, J.; Roberts, R.; Tinch, A.; Stear, M. The control of sea lice in Atlantic salmon by selective breeding. J. R. Soc. Interface 2015, 12, 1–8. [Google Scholar] [CrossRef]
- Sutherland, B.J.G.; Covello, J.M.; Friend, S.E.; Poley, J.D.; Koczka, K.W.; Purcell, S.L. Host-parasite transcriptomics during immunostimulant-enhanced rejection of salmon lice (Lepeophtheirus salmonis) by Atlantic salmon (Salmo salar). Facets 2017, 2, 477–495. [Google Scholar] [CrossRef]
- Jensen, L.B.; Provan, F.; Larssen, E.; Bron, J.E.; Obach, A. Reducing sea lice (Lepeophtheirus salmonis) infestation of farmed Atlantic salmon (Salmo salar L.) through functional feeds. Aquac. Nutr. 2015, 21, 983–993. [Google Scholar] [CrossRef]
- Raynard, R.S.; Bricknell, I.R.; Billingsley, P.F.; Nisbet, A.J.; Vigneau, A.; Sommerville, C. Development of vaccines against sea lice. Pest. Manag. Sci. 2002, 58, 569–575. [Google Scholar] [CrossRef]
- Jansen, P.A.; Grøtvedt, R.N.; Tarpai, A.; Helgesen, K.O.; Horsberg, T.E. Surveillance of the sensitivity towards antiparasitic bath-treatments in the salmon louse (Lepeophtheirus salmonis). PLoS ONE 2016, 11, e0149006. [Google Scholar] [CrossRef] [PubMed]
- Aaen, S.M.; Helgesen, K.O.; Jørgensen Bakke, M.; Kaur, K.; Horsberg, T.E. Drug resistance in sea lice: A threat to salmon aquaculture. Trends Parasitol. 2015, 31, 72–81. [Google Scholar] [CrossRef] [PubMed]
- Jevne, L.S.; Reitan, K.I. How are the salmon lice (Lepeophtheirus salmonis Krøyer, 1837) in Atlantic salmon farming affected by different control efforts: A case study of an intensive production area with coordinated production cycles and changing delousing practices in 2013–2018. J. Fish. Dis. 2019, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Bui, S.; Oppedal, F.; Sievers, M.; Dempster, T. Behaviour in the toolbox to outsmart parasites and improve fish welfare in aquaculture. Rev. Aquacult. 2019, 11, 168–186. [Google Scholar] [CrossRef]
- Urbina, M.A.; Cumillaf, J.P.; Paschke, K.; Gebauer, P. Effects of pharmaceuticals used to treat salmon lice on non-target species: Evidence from a systematic review. Sci. Total Environ. 2019, 649, 1124–1136. [Google Scholar] [CrossRef]
- Powell, A.; Treasurer, J.W.; Pooley, C.L.; Keay, A.J.; Lloyd, R.; Imsland, A.K.; Garcia de Leaniz, C. Use of lumpfish for sea-lice control in salmon farming: Challenges and opportunities. Rev. Aquacult. 2018, 10, 683–702. [Google Scholar] [CrossRef]
- Haugland, G.T.; Olsen, A.-B.; Rønneseth, A.; Andersen, L. Lumpfish (Cyclopterus lumpus L.) develop amoebic gill disease (AGD) after experimental challenge with Paramoeba perurans and can transfer amoebae to Atlantic salmon (Salmo salar L.). Aquaculture 2017, 478, 48–55. [Google Scholar] [CrossRef]
- Halvorsen, K.T.; Larsen, T.; Sørdalen, T.K.; Vøllestad, L.A.; Knutsen, H.; Olsen, E.M. Impact of harvesting cleaner fish for salmonid aquaculture assessed from replicated coastal marine protected areas. Mar. Biol. Res. 2017, 13, 359–369. [Google Scholar] [CrossRef]
- Stien, L.H.; Lind, M.B.; Oppedal, F.; Wright, D.; Seternes, T. Skirts on salmon production cages reduced salmon lice infestations without affecting fish welfare. Aquaculture 2018, 490, 281–287. [Google Scholar] [CrossRef]
- Stien, L.H.; Dempster, T.; Bui, S.; Glaropoulus, A.; Fosseidengen, J.E. ‘Snorkel’ sea lice barrier technology reduces sea lice loads on harvest-sized Atlantic salmon with minimal welfare impacts. Aquaculture 2016, 458, 29–37. [Google Scholar] [CrossRef]
- Ugelvik, M.S.; Skorping, A.; Moberg, O.; Mennerat, A. Evolution of virulence under intensive farming: Salmon lice increase skin lesions and reduce host growth in salmon farms. J. Evol. Biol. 2017, 30, 1136–1142.18. [Google Scholar] [CrossRef] [PubMed]
- Johnson, S.C.; Constible, J.M.; Richard, J. Laboratory investigations on the efficacy of hydrogen peroxide against salmon louse Lepeophtheirus salmonis and its toxicological and histopathological effects on Atlantic salmon Salmo salar and chinook salmon Oncorhynchus tshawytscha. Dis. Aquat. Organ. 1993, 17, 197–204. [Google Scholar] [CrossRef]
- Overton, K.; Samsing, F.; Oppedal, F.; Stien, L.H.; Dempster, T. Lowering treatment temperature reduces salmon mortality: A new way to treat with hydrogen peroxide in aquaculture. Pest. Manag. Sci. 2017, 74, 535–540. [Google Scholar] [CrossRef] [PubMed]
- Sevatdal, S.; Copley, L.; Wallace, C.; Jackson, D.; Horsberg, T.E. Monitoring of the sensitivity of sea lice (Lepeophtheirus salmonis) to pyrethroids in Norway, Ireland and Scotland using bioassays and probit modelling. Aquaculture 2015, 244, 19–27. [Google Scholar] [CrossRef]
- Jensen, E.M.; Sevatdal, S.; Bakke, M.J.; Kaur, K.; Horsberg, T.E. A selection study on a laboratory designed population of salmon lice (Lepeophtheirus salmonis) using organophosphate and pyrethroid pesticides. PLoS ONE 2017, 11, e0149264. [Google Scholar] [CrossRef]
- Carmona-Antoñanzas, G.; Bekaert, M.; Humble, J.L.; Boyd, S.; Roy, W.; Bassett, D.I.; Houston, R.D.; Gharbi, K.; Bron, J.E.; Sturm, A. Maternal inheritance of deltamethrin resistance in the salmon louse Lepeophtheirus salmonis (Krøyer) is associated with unique mtDNA haplotypes. PLoS ONE 2017, 12, e0180625. [Google Scholar] [CrossRef]
- Ritchie, G.; Rønsberg, S.S.; Hoff, K.A.; Branson, E.J. Clinical efficacy of teflubenzuron (Calicide®) for the treatment of Lepeophtheirus salmonis infestations of farmed Atlantic salmon Salmo salar at low water temperatures. Dis. Aquat. Organ. 2002, 51, 101–106. [Google Scholar] [CrossRef][Green Version]
- Poley, J.D.; Braden, L.M.; Messmer, A.M.; Igboeli, O.O.; Whyte, S.K.; Macdonald, A. High efficacy of lufenuron against sea lice (Lepeophtheirus salmonis) linked to rapid impact on moulting processes. Int. J. Parasitol. Drugs Drug Resist. 2018, 8, 174–188. [Google Scholar] [CrossRef]
- O’Halloran, J.; Hogans, W.E. First use in North America of azamethiphos to treat Atlantic salmon for sea lice infestation: Procedures and efficacy. Can. Vet. J. 1996, 37, 610–611. [Google Scholar]
- Stone, J.; Sutherland, I.H.; Sommerville, C.; Richards, R.H.; Varma, K.J. Field trials to evaluate the efficacy of emamectin benzoate in the control of sea lice, Lepeophtheirus salmonis (Krøyer) and Caligus elongatus Nordmann, infestations in Atlantic salmon Salmo salar L. Aquaculture 2000, 186, 205–219. [Google Scholar] [CrossRef]
- Whyte, S.K.; Poley, J.D.; Mueller, A.; Van Iderstine, C.; Fitzpatrick, K.E.; Purcell, S.L. Avermectin treatment for Lepeophtheirus salmonis: Impacts on host (Salmo salar) and parasite immunophysiology. Aquaculture 2019, 501, 488–501. [Google Scholar] [CrossRef]
- Aaen, S.M.; Horsberg, T.E. A screening of multiple classes of pharmaceutical compounds for effect on preadult salmon lice Lepeophtheirus salmonis. J. Fish. Dis 2016, 39, 1213–1223. [Google Scholar] [CrossRef] [PubMed]
- Kvenseth, P.G. Use of wrasse to control salmon lice. In Fish. Farming Technology; Reinertsen, H., Dahle, L.A., Jørgensen, L., Tvinnereim, K., Eds.; A.A. Balkema: Rotterdam, The Netherlands, 1993; pp. 227–232. [Google Scholar]
- Treasurer, J. Prey selection and daily food consumption by cleaner fish, Ctenolabrus rupestris (L.), on a farmed Atlantic salmon, Salmo salar L. Aquaculture 1994, 122, 269–277. [Google Scholar] [CrossRef]
- Imsland, A.K.; Reynolds, P.; Eliassen, G.; Hangstad, T.A.; Nytrø, A.V.; Foss, A.; Vikingstad, E.; Elvegrd, T.A. Feeding preferences of lumpfish (Cyclopterus lumpus L.) maintained in open net-pens with Atlantic salmon (Salmo salar L.). Aquaculture 2015, 436, 47–51. [Google Scholar] [CrossRef]
- Imsland, A.K.; Reynolds, P.; Eliassen, G.; Hangstad, T.A.; Foss, A.; Vikingstad, E.; Elvegård, T.A. The use of lumpfish (Cyclopterus lumpus L.) to control sea lice (Lepeophtheirus salmonis Krøyer) infestations in intensively farmed Atlantic salmon (Salmo salar L.). Aquaculture 2014, 424, 18–23. [Google Scholar] [CrossRef]
- Imsland, A.K.; Reynolds, P.; Eliassen, G.; Hangstad, T.A.; Nytrø, A.V.; Foss, A. Assessment of growth and sea lice infestation levels in Atlantic salmon stocked in small-scale cages with lumpfish. Aquaculture 2014, 433, 137–142. [Google Scholar] [CrossRef]
- Deady, S.; Varian, S.J.A.; Fives, J.M. The use of cleaner-fish to control sea lice in two Irish salmon (Salmo salar) farms with particular reference to wrasse behavior in salmon cages. Aquaculture 1995, 131, 73–90. [Google Scholar] [CrossRef]
- Leclercq, E.; Davie, A.; Migaud, H. Delousing efficiency of farmed ballan wrasse (Labrus bergylta) against Lepeophtheirus salmonis infesting Atlantic salmon (Salmo salar) post-smolts. Pest. Manag. Sci. 2013, 70, 1274–1282. [Google Scholar] [CrossRef]
- Imsland, A.K.; Reynolds, P.; Eliassen, G.; Hangstad, T.A.; Nytrø, A.V.; Foss, A.; Vikingstad, E.; Elvegrd, T.A. Notes on the behaviour of lumpfish in sea pens with and without Atlantic salmon present. J. Ethol. 2014, 32, 117–122. [Google Scholar] [CrossRef]
- Imsland, A.K.; Reynolds, P.; Eliassen, G.; Morten, A.; Hansen, Ø.J.; Puvanendran, V.; Hangstad, T.A.; Jnsdttir, I.; Emaus, P.-A.; Elvegrd, T.A. Is cleaning behaviour in lumpfish (Cyclopterus lumpus) parentally controlled? Aquaculture 2016, 459, 156–165. [Google Scholar] [CrossRef]
- Holm, H.; Santi, N.; Kjøglum, S.; Perisic, N.; Skugor, S. Difference in skin immune responses to infection with salmon louse (Lepeophtheirus salmonis) in Atlantic salmon (Salmo salar L.) of families selected for resistance and susceptibility. Fish. Shellfish Immun. 2015, 42, 384–394. [Google Scholar] [CrossRef] [PubMed]
- Tsai, H.-Y.; Hamilton, A.; Tinch, A.E.; Guy, D.R.; Bron, J.E.; Taggart, J.B. Genomic prediction of host resistance to sea lice in farmed salmon populations. Genet. Sel. Evol. 2016, 48, 47. [Google Scholar] [CrossRef] [PubMed]
- Grøntvedt, R.N.; Kristoffersen, A.B.; Jansen, P.A. Reduced exposure of farmed salmon to salmon louse (Lepeophtheirus salmonis L.) infestation by use of plankton nets: Estimating the shielding effect. Aquaculture 2018, 495, 865–872. [Google Scholar] [CrossRef]
- Wright, D.W.; Stien, L.H.; Dempster, T.; Vågseth, T.; Nola, V.; Fosseidengen, J.-E. ‘Snorkel’ lice barrier technology reduced two co-occurring parasites, the salmon louse (Lepeophtheirus salmonis) and the amoebic gill disease causing agent (Neoparamoeba perunans), in commercial salmon sea cages. Prev. Vet. Med. 2017, 140, 97–105. [Google Scholar] [CrossRef]
- Ljungfeldt, L.E.R.; Quintela, M.; Besnier, F.; Nilsen, F. A pedigree-based experiment reveals variation in salinity and thermal tolerance in the salmon louse, Lepeophtheirus salmonis. Evol. Appl. 2017, 10, 1007–1019. [Google Scholar] [CrossRef] [PubMed]
- Hjeltnes, B.; Bang-Jensen, B.; Bornø, G.; Haukaas, A.; Walde, C.S. The Health Situation in Norwegian Aquaculture 2018; Norwegian Veterinary Institute: Oslo, Norway, 2019; pp. 1–127. [Google Scholar]
- Sevatdal, S.; Horsberg, T.E. Determination of reduced sensitivity in sea lice (Lepeophtheirus salmonis Krøyer) against the pyrethroid deltamethrin using bioassays and probit modelling. Aquaculure 2003, 218, 21–31. [Google Scholar] [CrossRef]
- Imsland, A.K.; Frogg, N.; Stefansson, S.O.; Reynolds, P. Improving sea lice grazing of lumpfish (Cyclopterus lumpus L.) by feeding live feeds prior to transfer to Atlantic salmon (Salmo salar L.) net-pens. Aquaculture 2019, 511, 734224. [Google Scholar] [CrossRef]
- Goater, T.M.; Goater, C.P.; Esch, G.W. Parasitism; Cambridge University Press: Cambridge, UK, 2014; pp. 1–10; 263–334; 411–427. [Google Scholar]
- Burridge, L.; Weis, J.S.; Cabello, F.; Pazarro, J.; Bostick, K. Chemical use in salmon aquaculture: A review of current practices and possible environmental effects. Aquaculture 2010, 306, 7–23. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; The PRISMA Group. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Downs, S.H.; Black, N. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J. Epidemiol. Community Health 1998, 52, 377–384. [Google Scholar] [CrossRef]
- O’Connor, S.R.; Tully, M.A.; Byan, B.; Bradley, J.M.; Baxter, G.D.; McDonough, S.M. Failure of numerical quality assessment scale to identify potential risk of bias in a systematic literature review: A comparison study. BMC Res. Notes 2015, 8, 224. [Google Scholar] [CrossRef] [PubMed]
| Chemical Class | Active Ingredients | Effect on Salmon Louse |
|---|---|---|
| Organophosphates | Azamethiphos | Paralysis |
| Pyrethroids | Cypermethrin and deltamethrin | Paralysis |
| Avermectin | Emamectin benzoate | Paralysis |
| Hydrogen peroxide | Hydrogen peroxide | Creation of gas bubbles within the body of the salmon louse, making it unable to grip a surface |
| Benzoylurea | Teflubenzuron and diflubenzuron | Chitin synthesis inhibitor—the louse cannot moult successfully |
| Treatment Group | Date of First Publication Describing Treatment | Efficacy of Treatment Reported in the First Publication (Concentration) | Year When Resistance to Treatment First Documented | Efficacy Reported in Most Recent Publication (conc.) |
|---|---|---|---|---|
| Hydrogen peroxide | 1993 | 20% pre-adult survival (1.5 g/L bath) [23] | 1994 | 16%–50% pre-adult survival (1.5 g/L bath) [24] |
| Pyrethroids | 1998 | 50% immobilization of lice (1.03 μg/L bath) [25] | 2001 | immobilization for resistant lice strains 13.2%–20%, and 70.3%–80% for sensitive lice strains (2 μg/L bath) [26,27] |
| Benzoylurea | 1995 | 69.4%–77.5% effective (10 mg/kg fish biomass) [28] | 2015 | 96% reduction (700 μg/L bath) [29] |
| Organophosphates | 1996 | 100% gravid female reduction; 98.3% pre-adult reduction; 68% chalimus reduction (100 μg/L bath) [30] | 2012 | 19.1% immobilization of resistant strain (100 μg/L bath) [26] |
| Avermectins | 1999 | 68%–98% immobilization of lice (50 μg/kg fish biomass) [31] | 2019 | no significant difference between the control and treatment groups (50 μg/kg fish biomass); significant difference (1.2 in lice/salmon compared with 3.9) between the control and treatment group (150 μg/fish biomass) [32] |
| Chemical Treatment | Resistance Recorded (Number of Publications) | No Resistance (Number of Publications) | Not Mentioned | Percentage of Publications Reporting Resistance |
|---|---|---|---|---|
| Hydrogen peroxide | 5 | 3 | 1 | 55.5% |
| Avermectins | 19 | 8 | 1 | 67.8% |
| Pyrethroids | 5 | 1 | 2 | 62.5% |
| Organophosphates | 4 | 1 | 0 | 80.0% |
| Benzoylurea | 1 | 5 | 0 | 16.7% |
| Method | Number of Trials Reported in the Peer-Reviewed Literature | Significant Reduction in the Percentage of Lice Numbers Reported (Yes/No) |
|---|---|---|
| Preventive Measures | ||
| Selective breeding | 3 | Yes [43,44] (e.g., no treatment needed after 10 generations of selection) [8] |
| Skirt/plankton net | 2 | Yes—30%–80% reduction [20,45] |
| Functional feeds | 1 | Yes—20%–50% reduction [10] |
| Snorkel cage | 2 | Yes—72%–84% reduction [21,46] |
| Non-Chemical Bath Treatments | ||
| Warm water | 1 | Yes—18.6%–42% reduction [47] |
| Fresh water | 1 | Yes—81.9% reduction [47] |
| 1. Population examined | Farmed Atlantic salmon (S. salar) and salmon louse (L. salmonis) |
| 2. Intervention method (at least one of listed) | Chemical treatment, cleaner fish, non-chemical treatment, or preventive measures |
| 3. Language | English |
| 4. Time period | 1 January 1991—1 September 2019 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cerbule, K.; Godfroid, J. Salmon Louse (Lepeophtheirus salmonis (Krøyer)) Control Methods and Efficacy in Atlantic Salmon (Salmo salar (Linnaeus)) Aquaculture: A Literature Review. Fishes 2020, 5, 11. https://doi.org/10.3390/fishes5020011
Cerbule K, Godfroid J. Salmon Louse (Lepeophtheirus salmonis (Krøyer)) Control Methods and Efficacy in Atlantic Salmon (Salmo salar (Linnaeus)) Aquaculture: A Literature Review. Fishes. 2020; 5(2):11. https://doi.org/10.3390/fishes5020011
Chicago/Turabian StyleCerbule, Kristine, and Jacques Godfroid. 2020. "Salmon Louse (Lepeophtheirus salmonis (Krøyer)) Control Methods and Efficacy in Atlantic Salmon (Salmo salar (Linnaeus)) Aquaculture: A Literature Review" Fishes 5, no. 2: 11. https://doi.org/10.3390/fishes5020011
APA StyleCerbule, K., & Godfroid, J. (2020). Salmon Louse (Lepeophtheirus salmonis (Krøyer)) Control Methods and Efficacy in Atlantic Salmon (Salmo salar (Linnaeus)) Aquaculture: A Literature Review. Fishes, 5(2), 11. https://doi.org/10.3390/fishes5020011