Effect of Light–Dark Cycle on Skin Mucosal Immune Activities of Gilthead Seabream (Sparus aurata) and European Sea Bass (Dicentrarchus labrax)
Abstract
1. Introduction
2. Results
2.1. Total IgM Levels
2.2. Enzyme Activities in Skin Mucus
2.3. Bactericidal Activity
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Experimental Design and Sampling
4.3. Total Immunoglobulin M Levels
4.4. Protease Activity
4.5. Antiprotease Activity
4.6. Peroxidase Activity
4.7. Lysozyme Activity
4.8. Bactericidal Activity
4.9. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Valero, Y.; García-Alcázar, A.; Esteban, M.Á.; Cuesta, A.; Chaves-Pozo, E. Seasonal variations of the humoral immune parameters of European sea bass (Dicentrarchus labrax L.). Fish Shellfish Immunol. 2014, 39, 185–187. [Google Scholar] [CrossRef]
- Bowden, T.J.; Thompson, K.D.; Morgan, A.L.; Gratacap, R.M.; Nikoskelainen, S. Seasonal variation and the immune response: A fish perspective. Fish Shellfish Immunol. 2007, 22, 695–706. [Google Scholar] [CrossRef]
- Kalra, S.P.; Dube, M.G.; Pu, S.; Xu, B.; Horvath, T.L.; Kalra, P.S. Interacting appetite-regulating pathways in the hypothalamic regulation of body weight. Endocr. Rev. 1999, 20, 68–100. [Google Scholar] [CrossRef] [PubMed]
- Boeuf, G.; Le Bail, P.Y. Does light have an influence on fish growth? Aquaculture 1999, 177, 129–152. [Google Scholar] [CrossRef]
- Mogi, M.; Yokoi, H.; Suzuki, T. Analyses of the cellular clock gene expression in peripheral tissue, caudal fin, in the Japanese flounder, Paralichthys olivaceus. Gen. Comp. Endocrinol. 2017, 248, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Ramsey, K.M.; Marcheva, B.; Bass, J. Circadian rhythms, sleep, and metabolism. J. Clin. Invest. 2011, 121, 2133–2141. [Google Scholar] [CrossRef]
- McStay, E.; Migaud, H.; Vera, L.M.; Sánchez-Vázquez, F.J.; Davie, A. Comparative study of pineal clock gene and AANAT2 expression in relation to melatonin synthesis in Atlantic salmon (Salmo salar) and European seabass (Dicentrarchus labrax). Comp. Biochem. Physiol. B 2014, 169, 77–89. [Google Scholar] [CrossRef]
- Esteban, M.Á.; Cuesta, A.; Rodríguez, A.; Meseguer, J. Effect of photoperiod on the fish innate immune system: A link between fish pineal gland and the immune system. J. Pineal Res. 2006, 41, 261–266. [Google Scholar] [CrossRef]
- Liebmann, P.M.; Wölfler, A.; Felsner, P.; Hofer, D.; Schauenstein, K. Melatonin and the immune system. Int. Arch. Allergy Immunol. 1997, 112, 203–211. [Google Scholar] [CrossRef]
- Emet, M.; Ozcan, H.; Ozel, L.; Yayla, M.; Halici, Z.; Hacimuftuoglu, A. A review of melatonin, its receptors and drugs. Eurasian J. Med. 2016, 48, 135. [Google Scholar] [CrossRef]
- Carrillo-Vico, A.; Guerrero, J.M.; Lardone, P.J.; Reiter, R.J. A review of the multiple actions of melatonin on the immune system. Endocr. Res. 2005, 27, 189–200. [Google Scholar] [CrossRef]
- Esteban, M.Á.; Cuesta, A.; Chaves-Pozo, E.; Meseguer, J. Influence of melatonin on the immune system of fish: A review. Int. J. Mol. Sci. 2013, 14, 7979–7999. [Google Scholar] [CrossRef] [PubMed]
- FAO. The State of World Fisheries and Aquaculture 2018—Meeting the Sustainable Development Goals; FAO: Rome, Italy, 2018. [Google Scholar]
- Maitra, S.K.; Hasan, K.N. The Role of melatonin as a hormone and an antioxidant in the control of fish reproduction. Front. Endocrinol. 2016, 7, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Cabillon, N.A.R.; Lazado, C.C. Mucosal barrier functions of fish under changing environmental conditions. Fishes 2019, 4, 2. [Google Scholar] [CrossRef]
- Kaplan, J.E.; Chrenek, R.D.; Morash, J.G.; Ruksznis, C.M.; Hannum, L.G. Rhythmic patterns in phagocytosis and the production of reactive oxygen species by zebrafish leukocytes. Comp. Biochem. Physiol. A 2008, 151, 726–730. [Google Scholar] [CrossRef]
- Cordero, H.; Cuesta, A.; Meseguer, J.; Esteban, M.Á. Changes in the levels of humoral immune activities after storage of gilthead seabream (Sparus aurata) skin mucus. Fish shellfish immunol. 2016, 58, 500–507. [Google Scholar] [CrossRef]
- Esteban, M.Á. An Overview of the Immunological Defenses in Fish Skin. ISRN Immunol. 2012, 2012, 1–29. [Google Scholar] [CrossRef]
- Weil, R.J.; Borniger, Z.M.; Cisse, J.C.; Salloum, Y.M.; Nelson, B.A.A. Neuroendocrine control of photoperiodic changes in immune function. Front. Neuroendocr. 2015, 37, 108–118. [Google Scholar] [CrossRef]
- Nakanishi, T. Seasonal changes in the humoral immune response and the lymphoid tissues of the marine teleost, Sebastiscus marmoratus. Vet. Immunol. Immunopathol. 1986, 12, 213–221. [Google Scholar] [CrossRef]
- Bly, L.W.; Quiniou, J.E.; Clem, S.M. Environmental effects on fish immune mechanisms. Dev. Biol. Stand. 1997, 90, 33–43. [Google Scholar]
- Haldar, R.; Ahmad, C. Photoimmunomodulation and melatonin. J. Photochem. Photobiol. B Biol. 2010, 98, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Roberts, J.E. Light and immunomodulation. Ann. N. Y. Acad. Sci. 2000, 917, 435–445. [Google Scholar] [CrossRef] [PubMed]
- Płytycz, S.; Seljelid, R. Rhythms of immunity. Arch. Immunol. Ther. Exp. 1997, 45, 157–162. [Google Scholar]
- Molina-Borja, M.; Falcón, J.; Ravault, J.P. Production of melatonin by the gilthead sea bream pineal: An in vivo and in vitro study. Fish Physiol. Biochem. 1996, 15, 413–419. [Google Scholar] [CrossRef]
- García-Allegue, R.; Madrid, J.A.; Sánchez-Vázquez, F.J. Melatonin rhythms in European sea bass plasma and eye: Influence of seasonal photoperiod and water temperature. J. Pineal Res. 2001, 31, 68–75. [Google Scholar] [CrossRef]
- Tandler, A.; Helps, S. The effects of photoperiod and water exchange rate on growth and survival of gilthead sea bream (Sparus aurata, Linnaeus; Sparidae) from hatching to metamorphosis in mass rearing systems. Aquaculture 1985, 48, 71–82. [Google Scholar] [CrossRef]
- Kissil, G.W.; Lupatsch, I.; Elizur, A.; Zohar, Y. Long photoperiod delayed spawning and increased somatic growth in gilthead seabream (Sparus aurata). Aquaculture 2001, 200, 363–379. [Google Scholar] [CrossRef]
- Bromage, N.; Porter, M.; Randall, C. The environmental regulation of maturation in farmed finfish with special reference to the role of photoperiod and melatonin. Aquaculture 2001, 197, 63–98. [Google Scholar] [CrossRef]
- Chi, L.; Li, X.; Liu, Q.; Liu, Y. Photoperiod regulate gonad development via kisspeptin/kissr in hypothalamus and saccus vasculosus of Atlantic salmon (Salmo salar). PLoS ONE 2017, 12, e0169569. [Google Scholar] [CrossRef]
- Lazado, C.C.; Skov, P.V.; Pedersen, P.B. Innate immune defenses exhibit circadian rhythmicity and differential temporal sensitivity to a bacterial endotoxin in Nile tilapia (Oreochromis niloticus). Fish Shellfish Immunol. 2016, 55, 613–622. [Google Scholar] [CrossRef]
- Guardiola, F.A.; Cuesta, A.; Abellán, E.; Meseguer, J.; Esteban, M.Á. Comparative analysis of the humoral immunity of skin mucus from several marine teleost fish. Fish Shellfish Immunol. 2014, 40, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Itami, I. Defense mechanism of Ayu skin mucus. J. Shimonoseki Univ. Fish. 1993, 42, 71. [Google Scholar]
- Palaksha, K.J.; Shin, G.W.; Kim, Y.R.; Jung, T.S. Evaluation of non-specific immune components from the skin mucus of olive flounder (Paralichthys olivaceus). Fish shellfish immunol. 2008, 24, 479–488. [Google Scholar] [CrossRef] [PubMed]
- Vera, L.M.; Negrini, P.; Zagatti, C.; Frigato, E.; Sanchez-Vazquez, F.J.; Bertolucci, C. Light and feeding entrainment of the molecular circadian clock in a marine teleost (Sparus aurata). Chronobiol. Int. 2013, 30, 649–661. [Google Scholar] [CrossRef]
- Herrero, J.M.; Lepesant, M.J. Daily and seasonal expression of clock genes in the pituitary of the European sea bass (Dicentrarchus labrax). Gen. Comp. Endocrinol. 2014, 208, 30–38. [Google Scholar] [CrossRef]
- Flajnik, M.F. Comparative analyses of immunoglobulin genes: Surprises and portents. Nat. Rev. Immunol. 2002, 2, 688–698. [Google Scholar] [CrossRef]
- Boshra, J.O.; Gelman, H.; Sunyer, A.E. Structural and functional characterization of complement C4 and C1s-like molecules in teleost fish: Insights into the evolution of classical and alternative pathways. J. Immunol. 2004, 173, 349–359. [Google Scholar] [CrossRef]
- Ye, J.; Kaattari, I.M.; Ma, C.; Kaattari, S. The teleost humoral immune response. Fish Shellfish Immunol. 2013, 35, 1719–1728. [Google Scholar] [CrossRef]
- Cain, R.L.; Jones, K.D.; Raison, D.R. Characterisation of mucosal and systemic immune responses in rainbow trout (Oncorhynchus mykiss) using surface plasmon resonance. Fish Shellfish Immunol. 2000, 10, 651–666. [Google Scholar] [CrossRef]
- Subramanian, N.W.; MacKinnon, S.; Ross, S.L. A comparative study on innate immune parameters in the epidermal mucus of various fish species. Comp. Biochem. Physiol. B 2007, 148, 256–263. [Google Scholar] [CrossRef]
- Watts, M.; Munday, B.L.; Burke, C.M. Immune responses of teleost fish. Aust. Vet. J. 2001, 79, 570–574. [Google Scholar] [CrossRef] [PubMed]
- Aranichi, F.; Mano, V.; Nakane, M.; Hirose, H. Effects of thermal stress on skin defence lysins of European eel, Anguilla anguilla L. J. Fish Dis. 1999, 22, 227–229. [Google Scholar] [CrossRef]
- Subramanian, S.; Ross, N.W.; MacKinnon, S.L. Comparison of antimicrobial activity in the epidermal mucus extracts of fish. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2008, 150, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Lazado, C.C.; Gesto, M.; Madsen, L.; Jokumsen, A. Interplay between daily rhythmic serum-mediated bacterial killing activity and immune defence factors in rainbow trout (Oncorhynchus mykiss). Fish Shellfish Immunol. 2018, 72, 418–425. [Google Scholar] [CrossRef]
- Skwarlo-Sonta, K. Melatonin in immunity: Comparative aspects. Neuro Endocrinol. Lett. 2002, 23, 61–66. [Google Scholar]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Cuesta, A.; Meseguer, J.; Esteban, M.A. Total serum immunoglobulin M levels are affected by immunomodulators in seabream (Sparus aurata L.). Vet. Immunol. Immunop. 2004, 101, 203–210. [Google Scholar] [CrossRef]
- Ross, N.W.; Firth, K.J.; Wang, A.; Burka, J.F.; Johnson, S.C. Changes in hydrolytic enzyme activities of naive Atlantic salmon Salmo salar skin mucus due to infection with the salmon louse Lepeophtheirus salmonis and cortisol implantation. Dis. Aquat. Org. 2000, 41, 43–51. [Google Scholar] [CrossRef]
- Hanif, A.; Bakopoulos, V.; Dimitriadis, G.J. Maternal transfer of humoral specific and non-specific immune parameters to sea bream (Sparus aurata) larvae. Fish Shellfish Immunol. 2004, 17, 411–435. [Google Scholar] [CrossRef]
- Quade, M.J.; Roth, J.A. A rapid, direct assay to measure degranulation of bovine neutrophil primary granules. Vet. Immunol. Immunopathol. 1997, 58, 239–248. [Google Scholar] [CrossRef]
- Swain, P.; Dash, S.; Sahoo, P.K.; Routray, P.; Sahoo, S.K.; Gupta, S.D.; Sarangi, N. Non-specific immune parameters of brood Indian major carp Labeo rohita and their seasonal variations. Fish Shellfish Immunol. 2007, 22, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.G.; Jeffries, A.H. A novel assay to detect macrophage bactericidal activity in fish; factors influencing the killing of Aeromonas salmonicida. J. Fish Dis. 1988, 11, 389–396. [Google Scholar]
 and
and  indicate lights on and off, respectively. Results are expressed as mean ± standard error of the mean (SEM) (n = 6). Different letters denote variations in the IgM levels at the sampling times (ANOVA), and the asterisk indicates a significant rhythmic pattern over 24 h (Ritme©). The levels of significance were set at p < 0.05. Letters shared in common among experimental times indicate no significant differences.
indicate lights on and off, respectively. Results are expressed as mean ± standard error of the mean (SEM) (n = 6). Different letters denote variations in the IgM levels at the sampling times (ANOVA), and the asterisk indicates a significant rhythmic pattern over 24 h (Ritme©). The levels of significance were set at p < 0.05. Letters shared in common among experimental times indicate no significant differences.
 and
and  indicate lights on and off, respectively. Results are expressed as mean ± standard error of the mean (SEM) (n = 6). Different letters denote variations in the IgM levels at the sampling times (ANOVA), and the asterisk indicates a significant rhythmic pattern over 24 h (Ritme©). The levels of significance were set at p < 0.05. Letters shared in common among experimental times indicate no significant differences.
indicate lights on and off, respectively. Results are expressed as mean ± standard error of the mean (SEM) (n = 6). Different letters denote variations in the IgM levels at the sampling times (ANOVA), and the asterisk indicates a significant rhythmic pattern over 24 h (Ritme©). The levels of significance were set at p < 0.05. Letters shared in common among experimental times indicate no significant differences.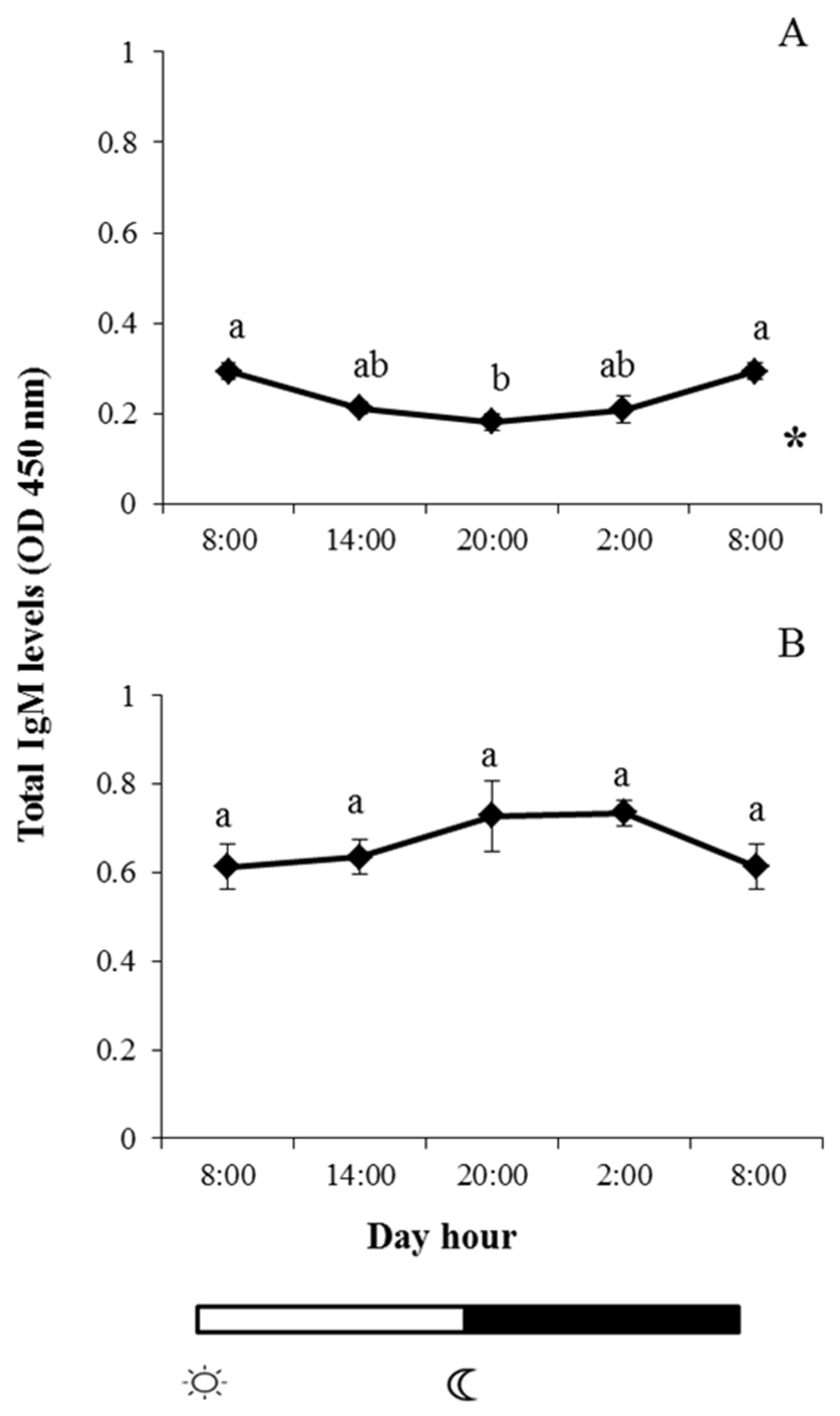
 and
and  indicate lights on and off, respectively. Results are expressed as mean ± SEM (n = 6). Different letters denote variations in the protease activity at the sampling times (ANOVA). The levels of significance were set at p < 0.05. Letters shared in common among experimental times indicate no significant differences.
indicate lights on and off, respectively. Results are expressed as mean ± SEM (n = 6). Different letters denote variations in the protease activity at the sampling times (ANOVA). The levels of significance were set at p < 0.05. Letters shared in common among experimental times indicate no significant differences.
 and
and  indicate lights on and off, respectively. Results are expressed as mean ± SEM (n = 6). Different letters denote variations in the protease activity at the sampling times (ANOVA). The levels of significance were set at p < 0.05. Letters shared in common among experimental times indicate no significant differences.
indicate lights on and off, respectively. Results are expressed as mean ± SEM (n = 6). Different letters denote variations in the protease activity at the sampling times (ANOVA). The levels of significance were set at p < 0.05. Letters shared in common among experimental times indicate no significant differences.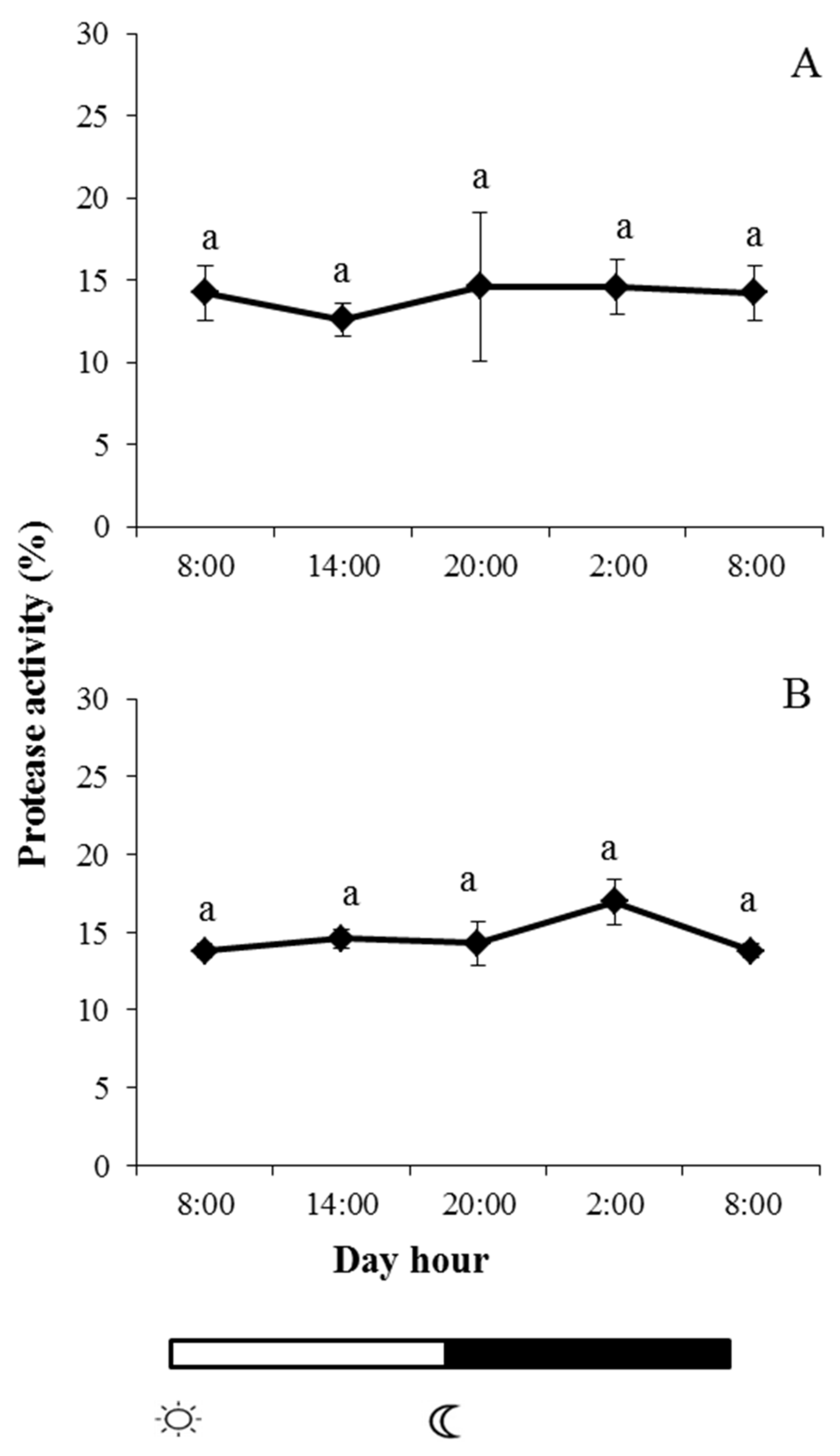
 and
and  indicate lights on and off, respectively. Results are expressed as mean ± SEM (n = 6). Different letters denote variations in the antiprotease activity at the sampling times (ANOVA). The levels of significance were set at p < 0.05. Letters shared in common among experimental times indicate no significant differences.
indicate lights on and off, respectively. Results are expressed as mean ± SEM (n = 6). Different letters denote variations in the antiprotease activity at the sampling times (ANOVA). The levels of significance were set at p < 0.05. Letters shared in common among experimental times indicate no significant differences.
 and
and  indicate lights on and off, respectively. Results are expressed as mean ± SEM (n = 6). Different letters denote variations in the antiprotease activity at the sampling times (ANOVA). The levels of significance were set at p < 0.05. Letters shared in common among experimental times indicate no significant differences.
indicate lights on and off, respectively. Results are expressed as mean ± SEM (n = 6). Different letters denote variations in the antiprotease activity at the sampling times (ANOVA). The levels of significance were set at p < 0.05. Letters shared in common among experimental times indicate no significant differences.
 and
and  indicate lights on and off, respectively. Results are expressed as mean ± SEM (n = 6). Different letters denote variations in the peroxidase activity at the sampling times (ANOVA), and the asterisk indicates a significant rhythmic pattern over 24 h (Ritme©). The levels of significance were set at p < 0.05. Letters shared in common among experimental times indicate no significant differences.
indicate lights on and off, respectively. Results are expressed as mean ± SEM (n = 6). Different letters denote variations in the peroxidase activity at the sampling times (ANOVA), and the asterisk indicates a significant rhythmic pattern over 24 h (Ritme©). The levels of significance were set at p < 0.05. Letters shared in common among experimental times indicate no significant differences.
 and
and  indicate lights on and off, respectively. Results are expressed as mean ± SEM (n = 6). Different letters denote variations in the peroxidase activity at the sampling times (ANOVA), and the asterisk indicates a significant rhythmic pattern over 24 h (Ritme©). The levels of significance were set at p < 0.05. Letters shared in common among experimental times indicate no significant differences.
indicate lights on and off, respectively. Results are expressed as mean ± SEM (n = 6). Different letters denote variations in the peroxidase activity at the sampling times (ANOVA), and the asterisk indicates a significant rhythmic pattern over 24 h (Ritme©). The levels of significance were set at p < 0.05. Letters shared in common among experimental times indicate no significant differences.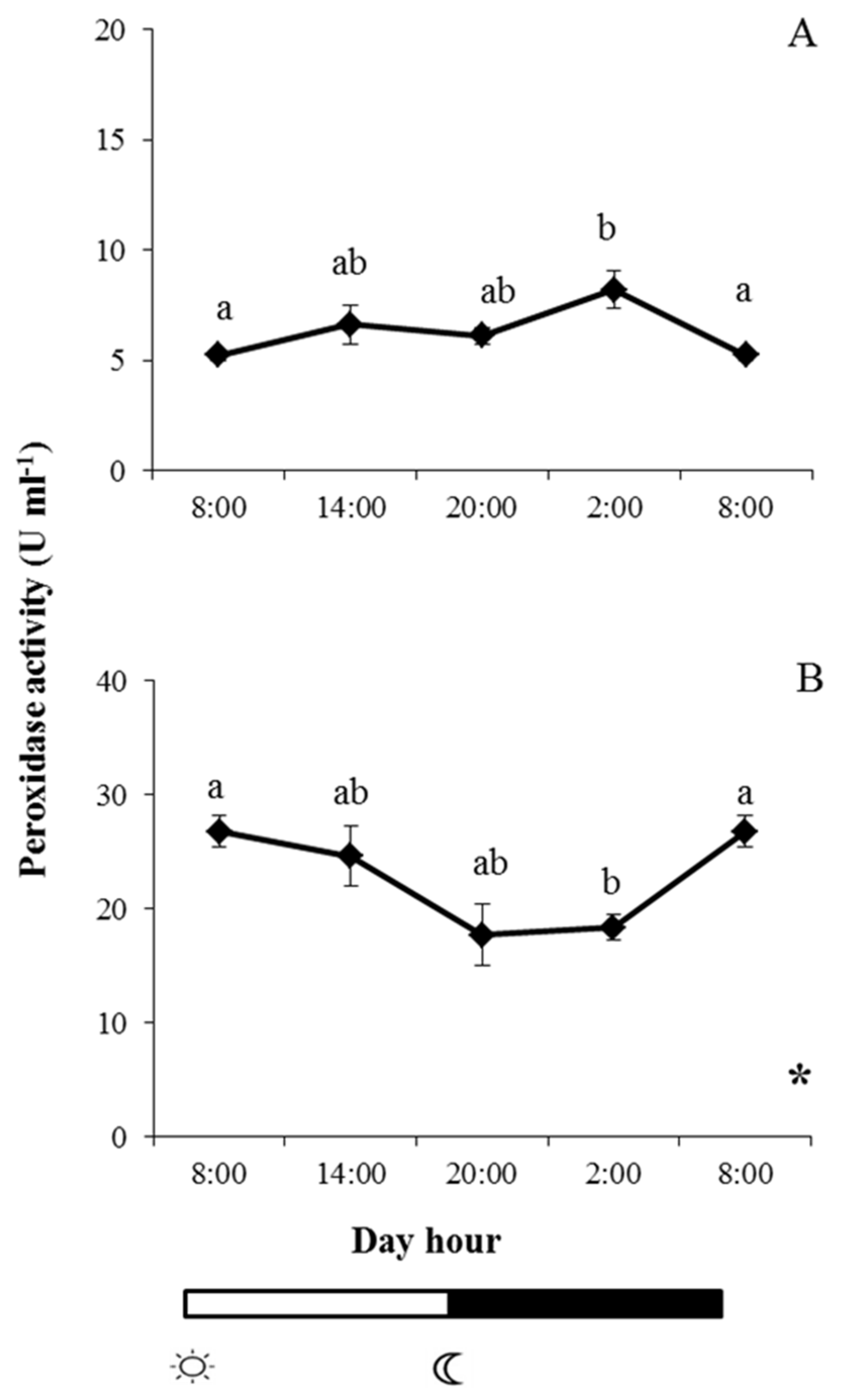
 and
and  indicate lights on and off, respectively. Results are expressed as mean ± SEM (n = 6). Different letters denote variations in the lysozyme activity at the sampling times (ANOVA), and the asterisk indicates a significant rhythmic pattern over 24 h (Ritme©). The levels of significance were set at p < 0.05. Letters shared in common among experimental times indicate no significant differences.
indicate lights on and off, respectively. Results are expressed as mean ± SEM (n = 6). Different letters denote variations in the lysozyme activity at the sampling times (ANOVA), and the asterisk indicates a significant rhythmic pattern over 24 h (Ritme©). The levels of significance were set at p < 0.05. Letters shared in common among experimental times indicate no significant differences.
 and
and  indicate lights on and off, respectively. Results are expressed as mean ± SEM (n = 6). Different letters denote variations in the lysozyme activity at the sampling times (ANOVA), and the asterisk indicates a significant rhythmic pattern over 24 h (Ritme©). The levels of significance were set at p < 0.05. Letters shared in common among experimental times indicate no significant differences.
indicate lights on and off, respectively. Results are expressed as mean ± SEM (n = 6). Different letters denote variations in the lysozyme activity at the sampling times (ANOVA), and the asterisk indicates a significant rhythmic pattern over 24 h (Ritme©). The levels of significance were set at p < 0.05. Letters shared in common among experimental times indicate no significant differences.
 and
and  indicate lights on and off, respectively. Different letters denote variations in the bactericidal activity at the sampling times (ANOVA), and the asterisk indicates a significant rhythmic pattern over 24 h (Ritme©). The levels of significance were set at p < 0.05. Letters shared in common among experimental times indicate no significant differences.
indicate lights on and off, respectively. Different letters denote variations in the bactericidal activity at the sampling times (ANOVA), and the asterisk indicates a significant rhythmic pattern over 24 h (Ritme©). The levels of significance were set at p < 0.05. Letters shared in common among experimental times indicate no significant differences.
 and
and  indicate lights on and off, respectively. Different letters denote variations in the bactericidal activity at the sampling times (ANOVA), and the asterisk indicates a significant rhythmic pattern over 24 h (Ritme©). The levels of significance were set at p < 0.05. Letters shared in common among experimental times indicate no significant differences.
indicate lights on and off, respectively. Different letters denote variations in the bactericidal activity at the sampling times (ANOVA), and the asterisk indicates a significant rhythmic pattern over 24 h (Ritme©). The levels of significance were set at p < 0.05. Letters shared in common among experimental times indicate no significant differences.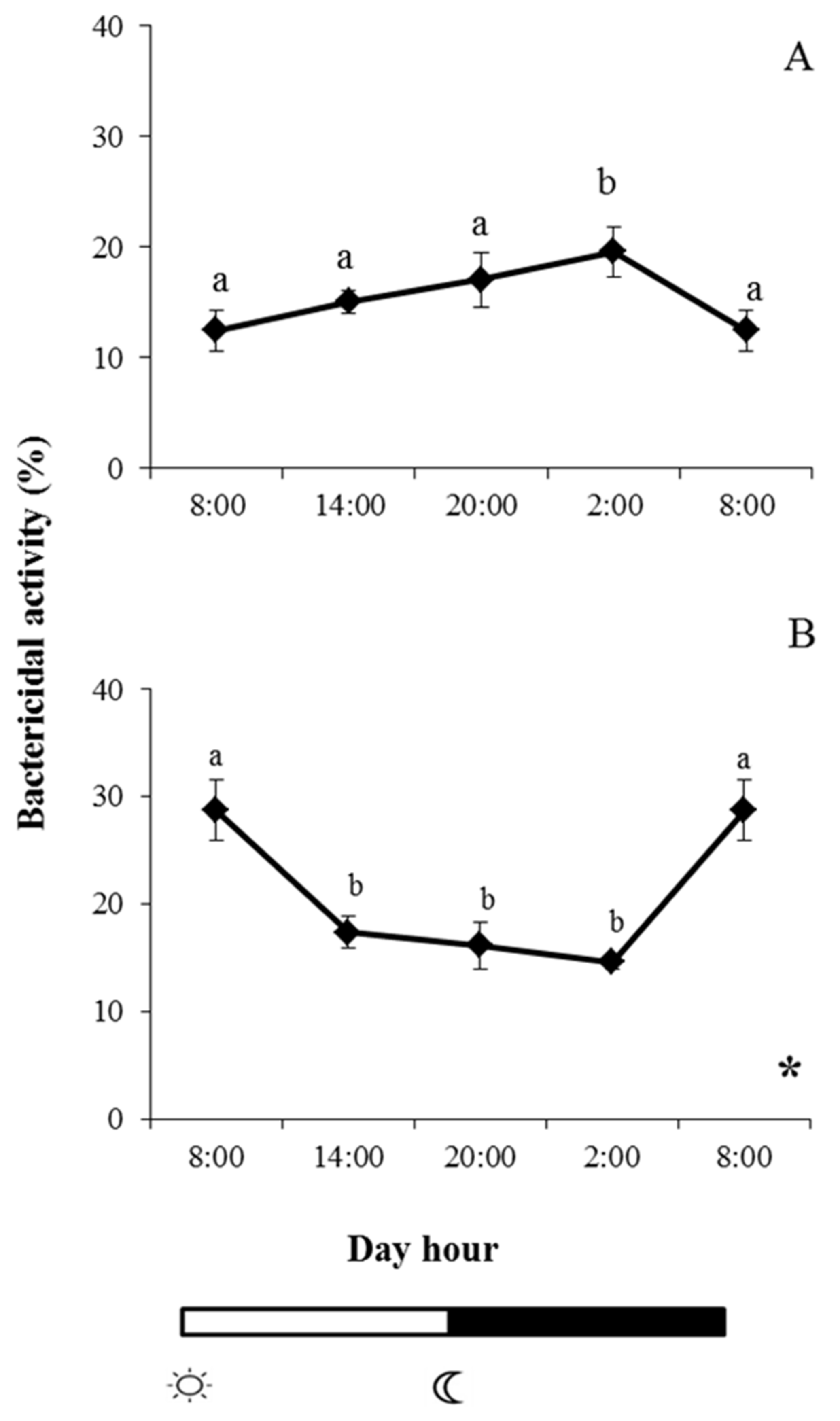
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ceballos-Francisco, D.; Cuesta, A.; Esteban, M.Á. Effect of Light–Dark Cycle on Skin Mucosal Immune Activities of Gilthead Seabream (Sparus aurata) and European Sea Bass (Dicentrarchus labrax). Fishes 2020, 5, 10. https://doi.org/10.3390/fishes5010010
Ceballos-Francisco D, Cuesta A, Esteban MÁ. Effect of Light–Dark Cycle on Skin Mucosal Immune Activities of Gilthead Seabream (Sparus aurata) and European Sea Bass (Dicentrarchus labrax). Fishes. 2020; 5(1):10. https://doi.org/10.3390/fishes5010010
Chicago/Turabian StyleCeballos-Francisco, Diana, Alberto Cuesta, and María Ángeles Esteban. 2020. "Effect of Light–Dark Cycle on Skin Mucosal Immune Activities of Gilthead Seabream (Sparus aurata) and European Sea Bass (Dicentrarchus labrax)" Fishes 5, no. 1: 10. https://doi.org/10.3390/fishes5010010
APA StyleCeballos-Francisco, D., Cuesta, A., & Esteban, M. Á. (2020). Effect of Light–Dark Cycle on Skin Mucosal Immune Activities of Gilthead Seabream (Sparus aurata) and European Sea Bass (Dicentrarchus labrax). Fishes, 5(1), 10. https://doi.org/10.3390/fishes5010010





