Changes in Sex Steroids and Ovarian Steroidogenic Enzyme mRNA Levels in Artificially Maturing Japanese Eel (Anguilla japonica) and Naturally Maturing New Zealand Longfin Eel (Anguilla dieffenbachii) during Vitellogenesis
Abstract
1. Introduction
2. Results
2.1. Gonadosomatic Indices in Japanese and Longfin Eels
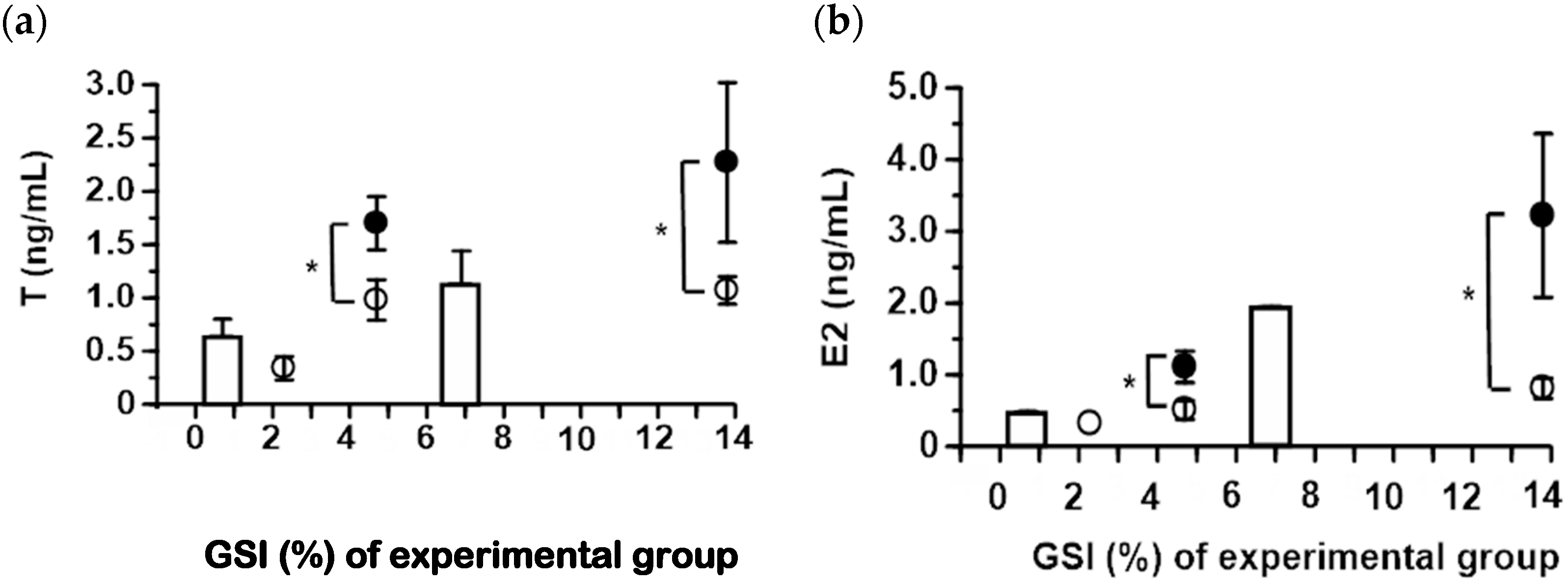
2.2. Serum Steroid Hormone Levels in Japanese and Longfin Eels
2.3. Isolation of Five Steroidogenic Enzyme cDNA Fragments from Longfin Eel Ovary
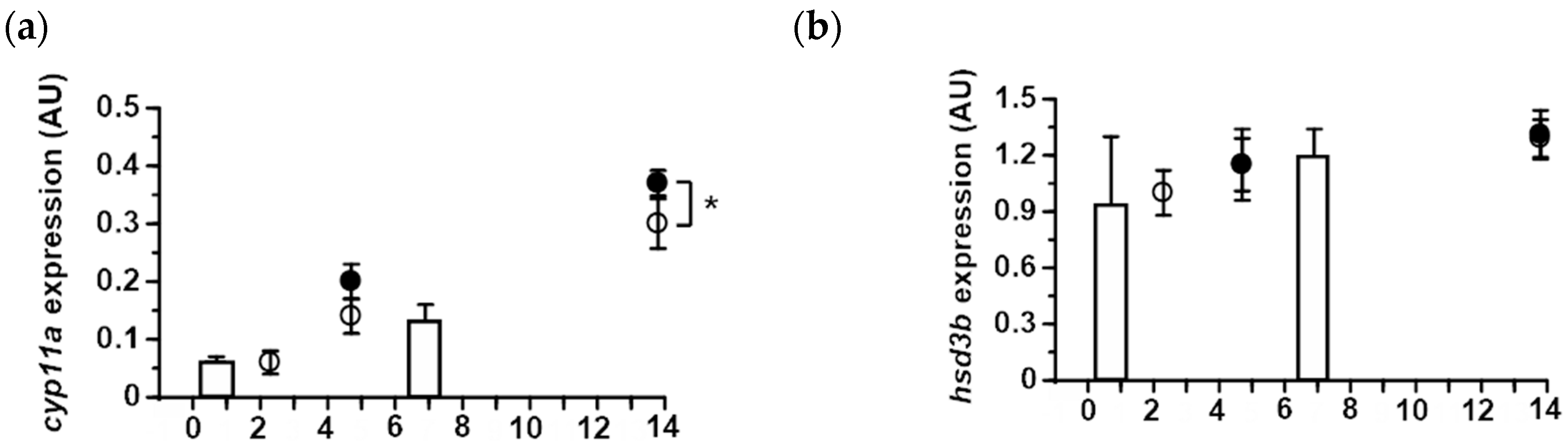
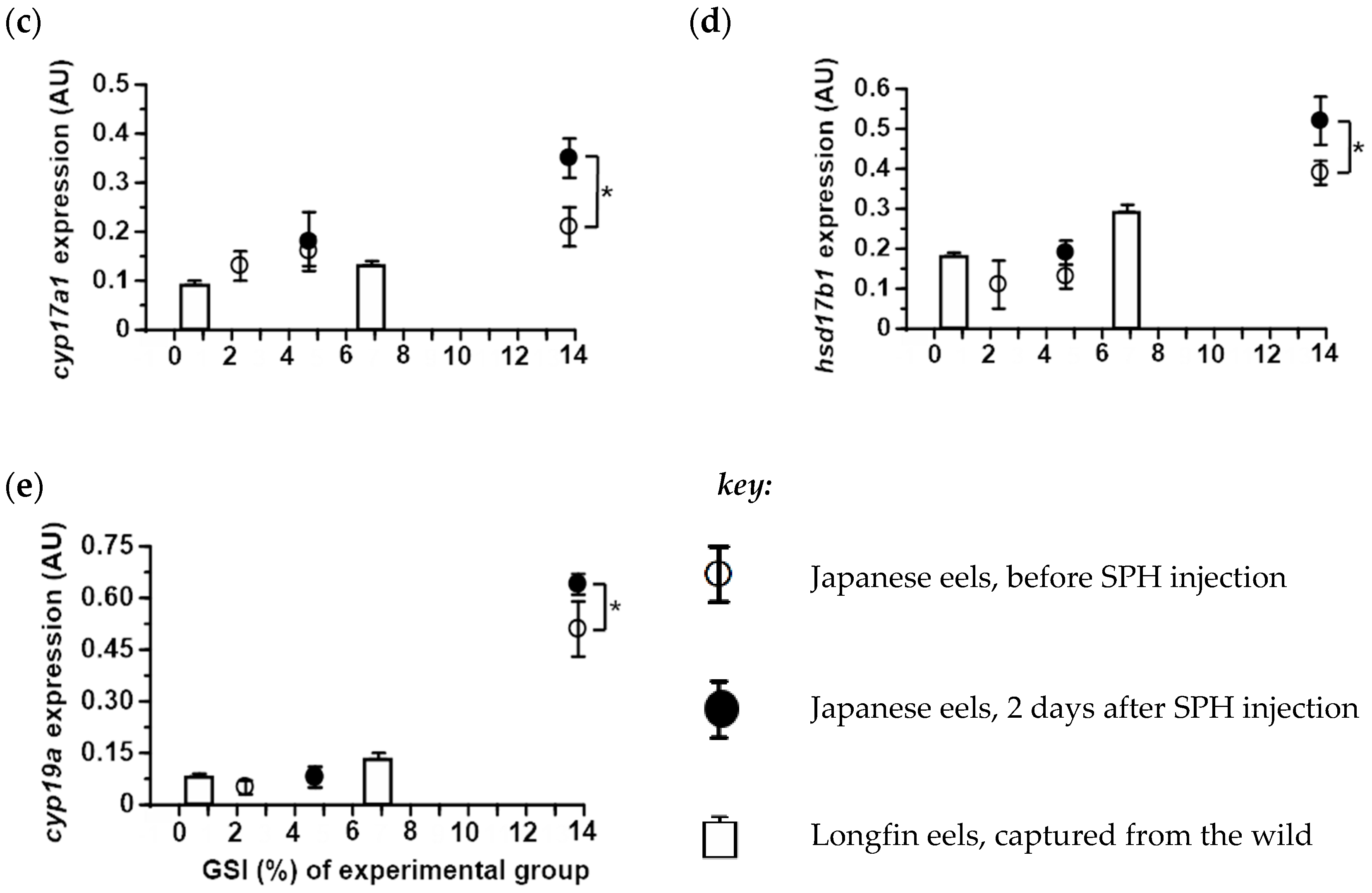
2.4. Expression of Ovarian Steroidogenic Enzymes in Japanese and Longfin Eels
3. Discussion
4. Materials and Methods
4.1. Animals and Sampling Design
4.1.1. Sampling of Japanese Eels
4.1.2. Sampling of Longfin Eels
4.2. Macroscopical Analyses
4.3. Radioimmunoassay
4.4. Isolation of Partial Steroidogenic Enzyme cDNAs from Longfin Eel Ovary
4.5. Real-Time Quantitative PCR (qPCR)
- sense
- 5’-TTAAGCGACAGAGGCAACCT-3’
- antisense
- 5’-CCAACTGTGTACTCCCCGAT-3
- probe
- 5'(FAM)-CCGTATCTTGAGGCCACCATCAGG-3'(TAMURA).
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nagahama, Y.; Yoshikuni, M.; Yamashita, M.; Tokumoto, T.; Katsu, Y. Regulation of Oocyte Growth and Maturation in Fish. Curr. Top. Dev. Biol. 1995, 30, 103–145. [Google Scholar] [PubMed]
- Lubzens, E.; Young, G.; Bobe, J.; Cerdà, J. Oogenesis in teleosts: How fish eggs are formed. Gen. Comp. Endocrinol. 2010, 165, 367–389. [Google Scholar] [CrossRef] [PubMed]
- Kagawa, H. Oogenesis in teleost fish. Aqua-BioSci. Monogr. 2013, 6, 99–127. [Google Scholar] [CrossRef]
- Wallace, R.A.; Jared, D.W.; Kohler, P.O.; Grimley, P.M.; O’Malley, B.W. Estrogen Induces Lipophosphoprotein in Serum of Male Xenopus laevis. Science 1968, 160, 91–92. [Google Scholar] [CrossRef] [PubMed]
- Wallace, R.A. A comprehensive synthesis. In Developmental Biology Volume I; Browder, L.W., Ed.; Plenum Press: New York, NY, USA, 1985; pp. 127–177. [Google Scholar]
- Thomas, P. Rapid steroid hormone actions initiated at the cell surface and the receptors that mediate them with an emphasis on recent progress in fish models. Gen. Com. Endocrinol. 2012, 175, 367–383. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, M.; Tanaka, M.; Sakai, N.; Adachi, S.; Miller, W.L.; Nagahama, Y. Rainbow trout ovarian cholesterol side-chain cleavage cytochrome P450 (P450scc). cDNA cloning and mRNA expression during oogenesis. FEBS Lett. 1993, 319, 45–48. [Google Scholar] [CrossRef]
- Nunez, S.; Trant, J.M. Isolation of the putative cDNA encoding cholesterol side chain cleavage cytochrome P450 (CYP11A) of the southern stingray (Dasyatis americana). Gene 1997, 187, 123–129. [Google Scholar] [CrossRef]
- Sakai, N.; Tanaka, M.; Takahashi, M.; Fukada, S.; I Mason, J.; Nagahama, Y. Ovarian 3 beta-hydroxysteroid dehydrogenase/delta 5-4-isomerase of rainbow trout: Its cDNA cloning and properties of the enzyme expressed in a mammalian cell. FEBS Lett. 1994, 350, 309–313. [Google Scholar] [CrossRef]
- Núñez, S.; Trant, J. Molecular biology and enzymology of elasmobranch 3β-hydroxysteroid dehydrogenase. Fish Physiol. Biochem. 1998, 19, 293–304. [Google Scholar] [CrossRef]
- Sakai, N.; Tanaka, M.; Adachi, S.; Miller, W.L.; Nagahama, Y. Rainbow trout cytochrome P-450c17 (17 alpha-hydroxylase/17,20-lyase). cDNA cloning, enzymatic properties and temporal pattern of ovarian P-450c17 mRNA expression during oogenesis. FEBS Lett. 1992, 301, 60–64. [Google Scholar] [CrossRef]
- Trant, J.M. Isolation and characterization of the cDNA encoding the spiny dogfish shark (Squalus acanthias) form of cytochrome P450c17. J. Exp. Zool. 1995, 272, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Kazeto, Y.; Ijiri, S.; Matsubara, H.; Adachi, S.; Yamauchi, K. Cloning of 17β-Hydroxysteroid Dehydrogenase-I cDNAs from Japanese Eel Ovary. Biochem. Biophys. Res. Commun. 2000, 279, 451–456. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, M.; Telecky, T.M.; Fukada, S.; Adachi, S.; Chen, S.; Nagahama, Y. Cloning and sequence analysis of the cDNA encoding P-450 aromatase (P450arom) from a rainbow trout (Oncorhynchus mykiss) ovary; relationship between the amount of P450arom mRNA and the production of oestradiol-17 beta in the ovary. J. Mol. Endocrinol. 1992, 8, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Trant, J.M. Isolation and Characterization of the cDNA Encoding the Channel Catfish (Ictalurus punctatus) Form of Cytochrome P450arom. Gen. Comp. Endocrinol. 1994, 95, 155–168. [Google Scholar] [CrossRef]
- Zhou, L.-Y.; Wang, D.-S.; Kobayashi, T.; Yano, A.; Paul-Prasanth, B.; Suzuki, A.; Sakai, F.; Nagahama, Y. A Novel Type of P450c17 Lacking the Lyase Activity Is Responsible for C21-Steroid Biosynthesis in the Fish Ovary and Head Kidney. Endocrinology 2007, 148, 4282–4291. [Google Scholar] [CrossRef]
- Su, T.; Ijiri, S.; Kanbara, H.; Hagihara, S.; Wang, D.-S.; Adachi, S. Characterization and expression of cDNAs encoding P450c17-II (cyp17a2) in Japanese eel during induced ovarian development. Gen. Comp. Endocrinol. 2015, 221, 134–143. [Google Scholar] [CrossRef]
- Kumar, R.S.; Trant, J.M.; Ijiri, S. Changes in the Expression of Genes Encoding Steroidogenic Enzymes in the Channel Catfish (Ictalurus punctatus) Ovary Throughout a Reproductive Cycle1. Biol. Reprod. 2000, 63, 1676–1682. [Google Scholar] [CrossRef]
- Ings, J.S.; Van Der Kraak, G.J. Characterization of the mRNA expression of StAR and steroidogenic enzymes in zebrafish ovarian follicles. Mol. Reprod. Dev. 2006, 73, 943–954. [Google Scholar] [CrossRef]
- Nakamura, I.; Evans, J.C.; Kusakabe, M.; Nagahama, Y.; Young, G. Changes in steroidogenic enzyme and steroidogenic acute regulatory protein messenger RNAs in ovarian follicles during ovarian development of rainbow trout (Oncorhynchus mykiss). Gen. Comp. Endocrinol. 2005, 144, 224–231. [Google Scholar] [CrossRef]
- Rocha, A.; Zanuy, S.; Carrillo, M.; Gómez, A. Seasonal changes in gonadal expression of gonadotropin receptors, steroidogenic acute regulatory protein and steroidogenic enzymes in the European sea bass. Gen. Comp. Endocrinol. 2009, 162, 265–275. [Google Scholar] [CrossRef]
- Tsukamoto, K. Oceanic biology: Spawning of eels near a seamount. Nature 2006, 439, 929. [Google Scholar] [CrossRef] [PubMed]
- Chow, S.; Kurogi, H.; Mochioka, N.; Kaji, S.; Okazaki, M.; Tsukamoto, K. Discovery of mature freshwater eels in the open ocean. Fish. Sci. 2009, 75, 257–259. [Google Scholar] [CrossRef]
- Yamamoto, K.; Yamauchi, K. Sexual maturation of Japanese eel and production of eel larvae in the aquarium. Nature 1974, 251, 220–222. [Google Scholar] [CrossRef] [PubMed]
- Adachi, S.; Ijiri, S.; Kazeto, Y.; Yamauchi, K. Oogenesis in the Japanese eel, Anguilla japonica. In Eel Biology; Aida, K., Tsukamoto, K., Yamauchi, K., Eds.; Springer: Tokyo, Japan, 2003; pp. 301–317. [Google Scholar]
- Burgerhout, E.; Minegishi, Y.; Brittijn, S.A.; De Wijze, D.L.; Henkel, C.V.; Jansen, H.J.; Spaink, H.P.; Dirks, R.P.; Thillart, G.E.V.D. Changes in ovarian gene expression profiles and plasma hormone levels in maturing European eel (Anguilla anguilla); Biomarkers for broodstock selection. Gen. Comp. Endocrinol. 2016, 225, 185–196. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, H.; Lokman, P.M.; Kazeto, Y.; Adachi, S.; Yamauchi, K. Serum steroid profiles in artificially maturing female Japanese eel, Anguilla japonica. Aquaculture 2005, 243, 393–402. [Google Scholar] [CrossRef]
- Matsubara, H.; Kazeto, Y.; Ijiri, S.; Hirai, T.; Adachi, S.; Yamauchi, K. Changes in mRNA levels of ovarian steroidogenic enzymes during artificial maturation of Japanese eel Anguilla japonica. Fish. Sci. 2003, 69, 979–988. [Google Scholar] [CrossRef]
- Tanaka, H.; Kagawa, H.; Ohta, H. Production of leptocephali of Japanese eel (Anguilla japonica) in captivity. Aquaculture 2001, 201, 51–60. [Google Scholar] [CrossRef]
- Tanaka, H. Techniques for larval rearing. In Eel Biology; Aida, K., Tsukamoto, K., Yamauchi, K., Eds.; Springer: Tokyo, Japan, 2003; pp. 427–434. [Google Scholar]
- Ohta, H.; Higashimoto, Y.; Koga, S.; Unuma, T.; Nomura, K.; Tanaka, H.; Kagawa, H.; Arai, K. Occurrence of spontaneous polyploids from the eggs obtained by artificial induction of maturation in the Japanese eel (Anguilla japonica). Fish Physiol. Biochem. 2003, 28, 517–518. [Google Scholar] [CrossRef]
- Kurokawa, T.; Okamoto, T.; Gen, K.; Uji, S.; Murashita, K.; Unuma, T.; Nomura, K.; Matsubara, H.; Kim, S.H.; Ohta, H.; et al. Effect of the water temperature on morphological deformities in cultured larvae of Japanese eel Anguilla japonica at competition of yolk absorption. J. World Aquac. Soc. 2008, 39, 726–735. [Google Scholar] [CrossRef]
- Okamura, A.; Yamada, Y.; Mikawa, N.; Horie, N.; Tsukamoto, K. Effect of salinity on occurrence of notochord deformities in Japanese eel Anguilla japonica larvae. Aquac. Int. 2016, 24, 549–555. [Google Scholar] [CrossRef]
- Okamura, A.; Horie, N.; Mikawa, N.; Yamada, Y.; Tsukamoto, K. Recent advances in artificial production of glass eels for conservation of anguillid eel populations. Ecol. Freshwat. Fish 2014, 23, 95–110. [Google Scholar] [CrossRef]
- Todd, P.R. Morphometric changes, gonad histology, and fecundity estimates in maturing New Zealand freshwater eels. N. Z. J. Mar. Freshw. Res. 1981, 15, 155–170. [Google Scholar] [CrossRef]
- Lokman, P.; Young, G. Gonad histology and plasma steroid profiles in wild New Zealand freshwater eels (Anguilla dieffenbachii and A. australis) before and at the onset of the natural spawning migration. II. Males. Fish Physiol. Biochem. 1998, 19, 339–347. [Google Scholar] [CrossRef]
- Reid, P.M.; Divers, S.L.; Zadmajid, V.; Alqaisi, K.M.; Lokman, P.M. Steroidogenic acute regulatory protein transcript abundance in the eel, Anguilla australis: Changes during the induced reproductive cycle and effects of follicle-stimulating hormone during previtellogenesis. J. Steroid Biochem. Mol. Biol. 2013, 138, 464–470. [Google Scholar] [CrossRef] [PubMed]
- Eding, E.H.; van Zanten, G.H.; Bongers, A.B.J.; Richter, C.J.J.; Lambert, J.G.D.; Goos, H.J.T.h. Induction of ovarian development in European eel (Anguilla anguilla) with low dosages of carp pituitary suspension. In Proceedings of the Fourth International Symposium on the Reproductive Physiology of Fish; Scott, A.P., Sumpter, J.P., Kime, D.E., Rolfe, M.S., Eds.; University of East Anglia: Norwich, UK, 1991; p. 321. [Google Scholar]
- Sato, N.; Kawazoe, I.; Suzuki, Y.; Aida, K. Development of an emulsion prepared with lipophilized gelatin and its application for hormone administration in the Japanese eel Anguilla japonica. Fish Physiol. Biochem. 1997, 17, 171–178. [Google Scholar] [CrossRef]
- Ijiri, S.; Kazeto, Y.; Lokman, P.M.; Adachi, S.; Yamauchi, K. Characterization of a cDNA encoding P—450aromatase (P450arom) from Japanese eel ovary and its expression in ovarian follicles during induced ovarian development. Gen. Comp. Endocrinol. 2003, 130, 193–203. [Google Scholar] [CrossRef]
- Ijiri, S.; Kazeto, Y.; Takeda, N.; Chiba, H.; Adachi, S.; Yamauchi, K. Changes in serum steroid hormones and steroidogenic ability of ovarian follicles during artificial maturation of cultivated Japanese eel, Anguilla japonica. Aquaculture 1995, 135, 3–16. [Google Scholar] [CrossRef]
- Lokman, P.M.; Wass, R.T.; Suter, H.C.; Scott, S.G.; Judge, K.F.; Young, G. Changes in steroid hormone profiles and ovarian histology during salmon pituitary-induced vitellogenesis and ovulation in female New Zealand longfinned eels, Anguilla dieffenbachii gray. J. Exp. Zool. 2001, 289, 119–129. [Google Scholar] [CrossRef]
- Pérez, L.M.; Peñaranda, D.S.; Dufour, S.; Baloche, S.; Palstra, A.; Thillart, G.V.D.; Asturiano, J.F. Influence of temperature regime on endocrine parameters and vitellogenesis during experimental maturation of European eel (Anguilla anguilla) females. Gen. Comp. Endocrinol. 2011, 174, 51–59. [Google Scholar] [CrossRef]
- Nguyen, A.T.; Chia, J.H.Z.; Kazeto, Y.; Lokman, P.M. Expression of gonadotropin subunit and gonadotropin receptor genes in wild female New Zealand shortfinned eel (Anguilla australis) during yellow and silver stages. Gen. Comp. Endocrinol. 2019, 272, 83–92. [Google Scholar] [CrossRef]
- Jéhannet, P.; Kruijt, L.; Damsteegt, E.; Swinkels, W.; Heinsbroek, L.; Lokman, P.; Palstra, A. A mechanistic model for studying the initiation of anguillid vitellogenesis by comparing the European eel (Anguilla anguilla) and the shortfinned eel (A. australis). Gen. Comp. Endocrinol. 2019, 279, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Stocco, D.M. Steroidogenic acute regulatory (StAR) protein: what’s new? BioEssays 1999, 21, 768–775. [Google Scholar] [CrossRef]
- Lin, T.-T.; Lan, P.-C.; Hsieh, Y.-J.; Wang, Y.-S. Estrogen suppresses Bcl2 expression through DNMT1 in Japanese eel ovarian follicle. Reproduction 2019, 158, 291–302. [Google Scholar] [CrossRef] [PubMed]
- Kurogi, H.; Okazaki, M.; Mochioka, N.; Jinbo, T.; Hashimoto, H.; Takahashi, M.; Tawa, A.; Aoyama, J.; Shinoda, A.; Tsukamoto, K.; et al. First capture of post-spawning female of the Japanese eel Anguilla japonica at the southern West Mariana Ridge. Fish. Sci. 2011, 77, 199–205. [Google Scholar] [CrossRef]
- Aoyama, J.; Watanabe, S.; Miller, M.J.; Mochioka, N.; Otake, T.; Yoshinaga, T.; Tsukamoto, K. Spawning Sites of the Japanese Eel in Relation to Oceanographic Structure and the West Mariana Ridge. PLoS ONE 2014, 9, e88759. [Google Scholar] [CrossRef]
- Ijiri, S.; Kayaba, T.; Takeda, N.; Tachiki, H.; Adachi, S.; Yamauchi, K. Pretreatment Reproductive Stage and Oocyte Development Induced by Salmon Pituitary Homogenate in the Japanese Eel Anguilla japonica. Fish. Sci. 1998, 64, 531–537. [Google Scholar] [CrossRef]
- Saito, K.; Lokman, P.M.; Young, G.; Ozaki, Y.; Matsubara, H.; Okumura, H.; Kazeto, Y.; Yoshiura, Y.; Aida, K.; Adachi, S.; et al. Follicle-stimulating hormone, luteinizing hormone and glycoprotein hormone subunit mRNA levels in artificially maturing Japanese eel Anguilla japonica and naturally maturing New Zealand longfinned eel Anguilla dieffenbachia. Fish. Sci. 2003, 69, 146–153. [Google Scholar] [CrossRef]
- Kazeto, Y.; Ijiri, S.; Todo, T.; Adachi, S.; Yamauchi, K. Molecular Cloning and Characterization of Japanese Eel Ovarian P450c17 (CYP17) cDNA. Gen. Comp. Endocrinol. 2000, 118, 123–133. [Google Scholar] [CrossRef]

| Gene | Primer | Sequence |
|---|---|---|
| Cyp11a | sense | 5’-GTTCATCGTATCATGCTCAAC-3’ |
| antisense | 5’-AGTCCACCTCTCCTATGTTG-3’ | |
| hsd3b | sense | 5’-TGTGGCTTCCTGGGAGAGA-3’ |
| antisense | 5’-GTACATCTACGGCGATGG-3 | |
| cyp17a1 | sense | 5’-ATAGTGGACACCGTGGCCAAGGA-3 |
| antisense | 5’-CTGCAGGACCACCCCAAATTTGC-3’ | |
| hsd17b1 | sense | 5’-AGAGTGGTGCTGATCACG-3’ |
| antisense | 5’-TGTGAGAGTTTGGCTATTCTC-3’ | |
| cyp19a | sense | 5’-TGGCAGACAGTTCTGATCA-3’ |
| antisense | 5’-AACCGAAGGGCTGGAAGAAA-3’ |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matsubara, H.; Lokman, P.M.; Kazeto, Y.; Okumura, H.; Ijiri, S.; Hirai, T.; Young, G.; Adachi, S.; Yamauchi, K. Changes in Sex Steroids and Ovarian Steroidogenic Enzyme mRNA Levels in Artificially Maturing Japanese Eel (Anguilla japonica) and Naturally Maturing New Zealand Longfin Eel (Anguilla dieffenbachii) during Vitellogenesis. Fishes 2019, 4, 52. https://doi.org/10.3390/fishes4040052
Matsubara H, Lokman PM, Kazeto Y, Okumura H, Ijiri S, Hirai T, Young G, Adachi S, Yamauchi K. Changes in Sex Steroids and Ovarian Steroidogenic Enzyme mRNA Levels in Artificially Maturing Japanese Eel (Anguilla japonica) and Naturally Maturing New Zealand Longfin Eel (Anguilla dieffenbachii) during Vitellogenesis. Fishes. 2019; 4(4):52. https://doi.org/10.3390/fishes4040052
Chicago/Turabian StyleMatsubara, Hajime, P. Mark Lokman, Yukinori Kazeto, Hiromi Okumura, Shigeho Ijiri, Toshiaki Hirai, Graham Young, Shinji Adachi, and Kohei Yamauchi. 2019. "Changes in Sex Steroids and Ovarian Steroidogenic Enzyme mRNA Levels in Artificially Maturing Japanese Eel (Anguilla japonica) and Naturally Maturing New Zealand Longfin Eel (Anguilla dieffenbachii) during Vitellogenesis" Fishes 4, no. 4: 52. https://doi.org/10.3390/fishes4040052
APA StyleMatsubara, H., Lokman, P. M., Kazeto, Y., Okumura, H., Ijiri, S., Hirai, T., Young, G., Adachi, S., & Yamauchi, K. (2019). Changes in Sex Steroids and Ovarian Steroidogenic Enzyme mRNA Levels in Artificially Maturing Japanese Eel (Anguilla japonica) and Naturally Maturing New Zealand Longfin Eel (Anguilla dieffenbachii) during Vitellogenesis. Fishes, 4(4), 52. https://doi.org/10.3390/fishes4040052







