A Global Assessment of Welfare in Farmed Fishes: The FishEthoBase
Abstract
1. Introduction
“1) Welfare is a characteristic of an animal, not something that is given to it; 2) Welfare will vary from very poor to very good, i.e., the individual may be in a poor state at one end of the welfare continuum or in a good state at the other, 3) Welfare can be measured objectively and independently of moral considerations; 4) Measures of failure to cope and measures of how difficult it is for an animal to cope both give information about how poor the welfare is; 5) Knowledge of the preferences of an animal often gives valuable information about what conditions are likely to result in good welfare, but direct measurements of the state of the animal must also be used in attempts to assess welfare and improve it; and 6) Animals may use a variety of methods when trying to cope. There are several consequences of failure to cope, so any one of a variety of measures can indicate that welfare is poor, and the fact that one measure, such as growth, is normal does not mean that welfare is good”.([22], page 4168)
- Describe in detail the FishEthoBase project, an open-access database on fish ethology and welfare.
- Use these data to assess the general welfare state of farmed fishes presently and use the scoring scheme in the database (FishEthoScore) to provide an outlook on the potential of fish species to be farmed in good welfare.
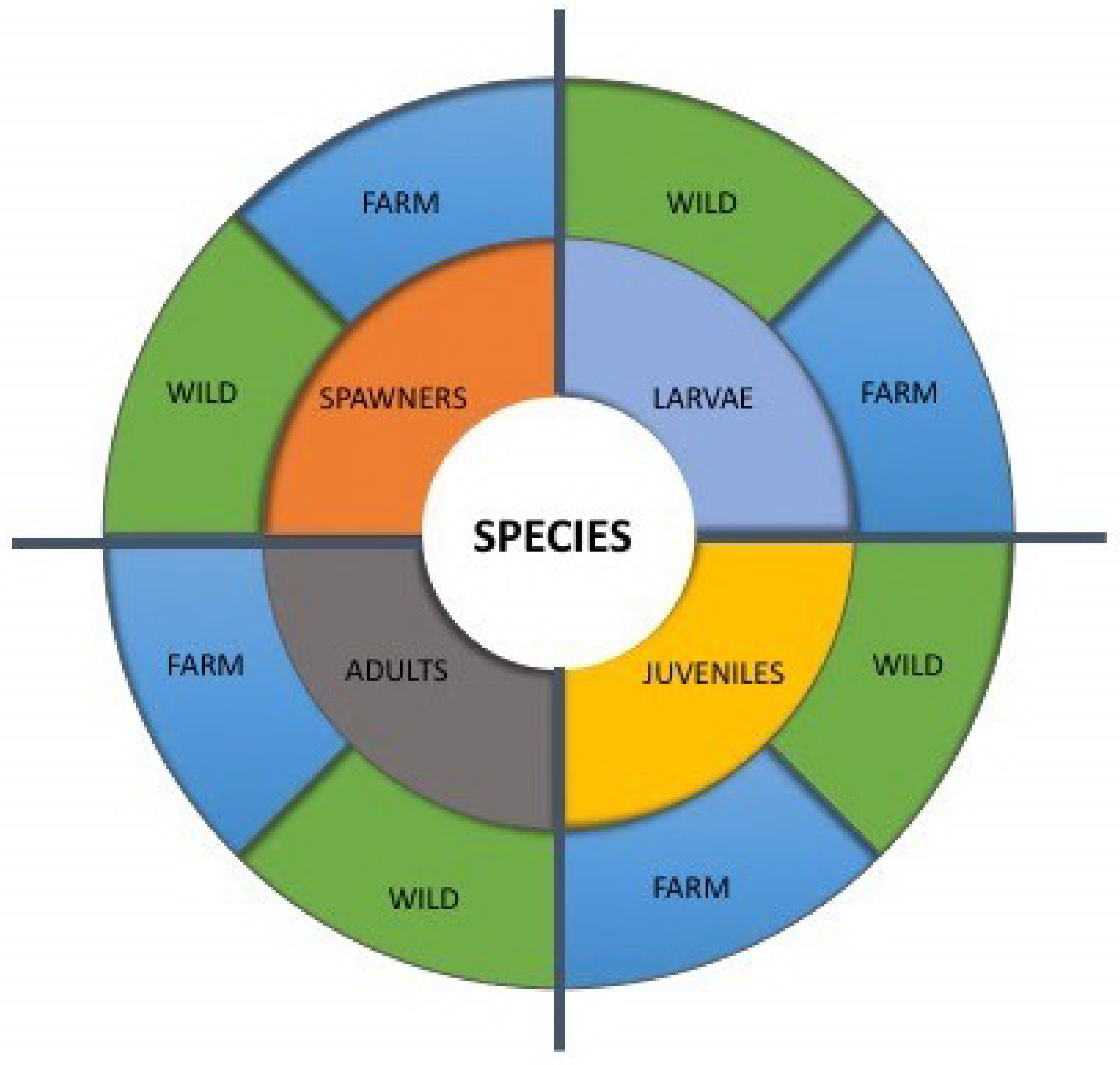
2. The FishEthoBase Project
- (1)
- Full profiles, divided into Findings, where an extensive review on the biology of the species in the wild and in captivity is assessed through bibliographical reviews. Recommendations, where proposals for rearing in captivity under good welfare conditions are made on the basis of the review, and a Summary where this knowledge is condensed.
- (2)
- Short profiles, where a rapid assessment of the welfare state of each species is performed through literature-based answers to 10 core criteria, pointing at main problems and possible solutions, and providing the base for the numerical assessment of welfare—the FishEthoScore.
2.1. Full Profiles—Findings
- Ethograms
- Distribution
- Natural co-existence
- Substrate and/or shelter
- Food, foraging, hunting, feeding
- Photoperiod
- Water parameters
- Swimming
- Growth
- Reproduction
- Senses
- Communication
- Social behaviour
- Cognitive abilities
- Personality, coping styles
- Emotion-like states
- Self-concept, self-recognition
- Reactions to husbandry
2.2. Full Profiles—Recommendations
2.3. Summary
2.4. Short Profiles
2.4.1. Selected Criteria for the Short Profiles
2.4.2. Scoring Framework: The FishEthoScores
- -
- The Likelihood that the fish experience good welfare under the lowest standard farming conditions found in the literature regarding that specific criterion. The possible scores are Low, High, Unclear or No Findings;
- -
- The Potential for the species to experience good welfare in the highest standard rearing conditions regarding that specific criterion (or the expected improvements in the near future). The possible scores are Low, Medium, High, Unclear or No Findings.
- -
- The Certainty of our assessment of the criterion, i.e., a measurement of the general quality, quantity and clarity of the data available for the species. The possible scores are Low, Medium, High or No Findings.
- Likelihood FishEthoScore: The sum of ‘High’ scores throughout the criteria, varies between 0 and 10.
- Potential FishEthoScore: The sum of ‘High’ scores throughout the criteria, varies between 0 and 10.
- Certainty FishEthoScore: The sum of ‘High’ scores throughout the criteria, varies between 0 and 10.
3. Assessment of Welfare in Farmed Fish Species
- -
- Are farmed fish species experiencing good welfare?
- -
- Is there potential for farmed species to experience good welfare?
- -
- Are fish species far from their best possible welfare conditions? Does the existing knowledge influence the current or prospective welfare state of farmed fish?
- -
- Are fish species which currently experience better welfare the ones who show the greatest potential to be farmed in best conditions?
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- FAO. The State of World Fisheries and Aquaculture 2016. Contributing to Food Security and Nutrition for All; FAO: Rome, Italy, 2016; p. 200. [Google Scholar]
- FAO. Domestic Animal Diversity Information System (DAD-IS). Available online: http://www.fao.org/dad-is/data/en/ (accessed on 15 February 2019).
- Saraiva, J.L.; Castanheira, M.F.; Arechavala-Lopez, P.; Volstorf, J.; Studer, B.H. Domestication and Welfare in Farmed Fish. In Animal Domestication; IntechOpen: London, UK, 2018. [Google Scholar]
- Teletchea, F.; Teletchea, F. Domestication and Genetics: What a Comparison between Land and Aquatic Species Can Bring? In Evolutionary Biology: Biodiversification from Genotype to Phenotype; Springer: Cham, Switzerland, 2015; pp. 389–401. [Google Scholar]
- Meeting the Sustainable Development Goals; FAO, Ed.; The State of World Fisheries and Aquaculture: Rome, Italy, 2018; ISBN 978-92-5-130562-1. [Google Scholar]
- Huntingford, F.A. Implications of domestication and rearing conditions for the behaviour of cultivated fishes. J. Fish Biol. 2004, 65, 122–142. [Google Scholar] [CrossRef]
- Brown, C. Fish intelligence, sentience and ethics. Anim. Cogn. 2014, 18, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Sneddon, L.U. The bold and the shy: Individual differences in rainbow trout. J. Fish Biol. 2003, 62, 971–975. [Google Scholar] [CrossRef]
- Yue, S.; Moccia, R.D.; Duncan, I.J.H. Investigating fear in domestic rainbow trout, Oncorhynchus mykiss, using an avoidance learning task. Appl. Anim. Behav. Sci. 2004, 87, 343–354. [Google Scholar] [CrossRef]
- Helfman, G.; Collette, B.B.; Facey, D.E.; Bowen, B.W. The Diversity of Fishes: Biology, Evolution, and Ecology; John Wiley & Sons: Hoboken, NJ, USA, 2009; ISBN 978-1-4443-1190-7. [Google Scholar]
- Ashley, P.J. Fish welfare: Current issues in aquaculture. Appl. Anim. Behav. Sci. 2007, 104, 199–235. [Google Scholar] [CrossRef]
- Cerqueira, M.; Millot, S.; Castanheira, M.F.; Félix, A.S.; Silva, T.; Oliveira, G.A.; Oliveira, C.C.; Martins, C.I.M.; Oliveira, R.F. Cognitive appraisal of environmental stimuli induces emotion-like states in fish. Sci. Rep. 2017, 7, 13181. [Google Scholar] [CrossRef]
- Dawkins, M.S. The Science of Animal Suffering. Ethology 2008, 114, 937–945. [Google Scholar] [CrossRef]
- Martins, C.I.M.; Galhardo, L.; Noble, C.; Damsgård, B.; Spedicato, M.T.; Zupa, W.; Beauchaud, M.; Kulczykowska, E.; Massabuau, J.-C.; Carter, T.; et al. Behavioural indicators of welfare in farmed fish. Fish Physiol. Biochem. 2012, 38, 17–41. [Google Scholar] [CrossRef] [PubMed]
- Segner, H.; Sundh, H.; Buchmann, K.; Douxfils, J.; Sundell, K.S.; Mathieu, C.; Ruane, N.; Jutfelt, F.; Toften, H.; Vaughan, L. Health of farmed fish: Its relation to fish welfare and its utility as welfare indicator. Fish Physiol. Biochem. 2012, 38, 85–105. [Google Scholar] [CrossRef] [PubMed]
- Duncan, I.J. A concept of welfare based on feelings. In The Well-Being of Farm Animals: Challenges and Solutions; Blackwell Publishing: Ames, IA, USA, 2004; pp. 85–101. [Google Scholar]
- Sneddon, L.U. Fish behaviour and welfare. Appl. Anim. Behav. Sci. 2007, 104, 173–175. [Google Scholar] [CrossRef][Green Version]
- Mendl, M.; Burman, O.H.P.; Parker, R.M.A.; Paul, E.S. Cognitive bias as an indicator of animal emotion and welfare: Emerging evidence and underlying mechanisms. Appl. Anim. Behav. Sci. 2009, 118, 161–181. [Google Scholar] [CrossRef]
- Boissy, A.; Manteuffel, G.; Jensen, M.B.; Moe, R.O.; Spruijt, B.; Keeling, L.J.; Winckler, C.; Forkman, B.; Dimitrov, I.; Langbein, J.; et al. Assessment of positive emotions in animals to improve their welfare. Physiol. Behav. 2007, 92, 375–397. [Google Scholar] [CrossRef]
- Noble, E.C.; Gismervik, K.; Iversen, M.H.; Kolarevic, J.; Nilsson, J.; Stien, L.H.; Turnbull, J.F. Welfare Indicators for Farmed Atlantic Salmon—Tools for Assessing Fish Welfare; Nofima: Tromsø, Norway, 2018. [Google Scholar]
- Stien, L.H.; Bracke, M.B.; Folkedal, O.; Nilsson, J.; Oppedal, F.; Torgersen, T.; Kittilsen, S.; Midtlyng, P.J.; Vindas, M.A.; Øverli, Ø.; et al. Salmon Welfare Index Model (SWIM 1.0): A semantic model for overall welfare assessment of caged Atlantic salmon: Review of the selected welfare indicators and model presentation. Rev. Aquac. 2013, 5, 33–57. [Google Scholar] [CrossRef]
- Broom, D.M. Animal welfare: Concepts and measurement. J. Anim. Sci. 1991, 69, 4167–4175. [Google Scholar] [CrossRef] [PubMed]
- Broom, D.M. Indicators of poor welfare. Br. Vet. J. 1986, 142, 524–526. [Google Scholar] [CrossRef]
- Bracke, M.B.M.; Hopster, H. Assessing the Importance of Natural Behavior for Animal Welfare. J. Agric. Environ. Ethics 2006, 19, 77–89. [Google Scholar] [CrossRef]
- Dingemanse, N.J.; Kazem, A.J.N.; Réale, D.; Wright, J. Behavioural reaction norms: Animal personality meets individual plasticity. Trends Ecol. Evol. 2010, 25, 81–89. [Google Scholar] [CrossRef]
- Duponchelle, F.; Legendre, M. Rapid phenotypic changes of reproductive traits in response to experimental modifications of spatial structure in Nile tilapia, Oreochromis niloticus. Aquat. Living Resour. 2001, 14, 145–152. [Google Scholar] [CrossRef]
- Hutchings, J.A. Adaptive phenotypic plasticity in brook trout, Salvelinus fontinalis, life histories. Écoscience 1996, 3, 25–32. [Google Scholar] [CrossRef]
- Lorenzen, K.; Beveridge, M.C.M.; Mangel, M. Cultured fish: Integrative biology and management of domestication and interactions with wild fish. Biol. Rev. 2012, 87, 639–660. [Google Scholar] [CrossRef]
- Brambell, F.W.R. Report of the Technical Committee to Enquire into the Welfare of Animals kept under Intensive Livestock Husbandry Systems; Farm Animal Welfare Council: London, UK, 1965. [Google Scholar]
- Fraser, D. Animal ethics and animal welfare science: Bridging the two cultures. Appl. Anim. Behav. Sci. 1999, 65, 171–189. [Google Scholar] [CrossRef]
- Fraser, D.; Weary, D.; Pajor, E.; Milligan, B. A Scientific Conception of Animal Welfare that Reflects Ethical Concerns. Anim. Welf. 1997, 6, 187–205. [Google Scholar]
- Korte, S.M.; Olivier, B.; Koolhaas, J.M. A new animal welfare concept based on allostasis. Physiol. Behav. 2007, 92, 422–428. [Google Scholar] [CrossRef]
- Sneddon, L.U.; Wolfenden, D.C.C.; Thomson, J.S. 12—Stress Management and Welfare. In Fish Physiology; Schreck, C.B., Tort, L., Farrell, A.P., Brauner, C.J., Eds.; Biology of Stress in Fish; Academic Press: Cambridge, MA, USA, 2016; Volume 35, pp. 463–539. [Google Scholar]
- Bain, M.B.; Webb, D.H.; Tangedal, M.D.; Mangum, L.N. Movements and Habitat Use by Grass Carp in a Large Mainstream Reservoir. Trans. Am. Fish. Soc. 1990, 119, 553–561. [Google Scholar] [CrossRef]
- Kohda, M. Territoriality of male cichlid fishes in Lake Tanganyika. Ecol. Freshw. Fish 1995, 4, 180–184. [Google Scholar] [CrossRef]
- McConnell, R.H.L. Breeding behaviour patterns and ecological differences between tilapia species and their significance for evolution within the genus tilapia (Pisces: Cichlidae). Proceedings of the Zoological Society of London 1959, 132, 1–30. [Google Scholar] [CrossRef]
- Abecasis, D.; Erzini, K. Site fidelity and movements of gilthead sea bream (Sparus aurata) in a coastal lagoon (Ria Formosa, Portugal). Estuar. Coast. Shelf Sci. 2008, 79, 758–763. [Google Scholar] [CrossRef]
- Arechavala-Lopez, P.; Uglem, I.; Fernandez-Jover, D.; Bayle-Sempere, J.T.; Sanchez-Jerez, P. Post-escape dispersion of farmed seabream (Sparus aurata L.) and recaptures by local fisheries in the Western Mediterranean Sea. Fish. Res. 2012, 121–122, 126–135. [Google Scholar] [CrossRef]
- Bruton, M.N. The Habitats and Habitat Preferences of Clarias Gariepinus (pisces: Clariidae) in a Clear Coastal Lake (lake Sibaya, South Africa). J. Limnol. Soc. S. Afr. 1978, 4, 81–88. [Google Scholar]
- Hocutt, C.H. Seasonal and diel behaviour of radio-tagged Clarias gariepinus in Lake Ngezi, Zimbabwe (Pisces: Clariidae). J. Zool. 1989, 219, 181–199. [Google Scholar] [CrossRef]
- Wood-Gush, D.G.M.; Vestergaard, K. Exploratory behavior and the welfare of intensively kept animals. J. Agric. Ethics 1989, 2, 161–169. [Google Scholar] [CrossRef]
- Huntingford, F.A.; Kadri, S. Defining, assessing and promoting the welfare of farmed fish. Rev. Sci. Tech. OIE 2014, 33, 233–244. [Google Scholar] [CrossRef]
- Van de Vis, J.W.; Poelman, M.; Lambooij, E.; Bégout, M.-L.; Pilarczyk, M. Fish welfare assurance system: Initial steps to set up an effective tool to safeguard and monitor farmed fish welfare at a company level. Fish Physiol. Biochem. 2012, 38, 243–257. [Google Scholar] [CrossRef] [PubMed]
- Williot, P.; Chebanov, M.; Nonnotte, G. Welfare in the Cultured Siberian Sturgeon, Acipenser baerii Brandt: State of the Art. In The Siberian Sturgeon (Acipenser baerii, Brandt, 1869) Volume—Farming; Springer: Cham, Switzerland, 2018; pp. 403–450. ISBN 978-3-319-61674-2. [Google Scholar]
- Filiciotto, F.; Giacalone, V.M.; Fazio, F.; Buffa, G.; Piccione, G.; Maccarrone, V.; Di Stefano, V.; Mazzola, S.; Buscaino, G. Effect of acoustic environment on gilthead sea bream (Sparus aurata): Sea and onshore aquaculture background noise. Aquaculture 2013, 414–415, 36–45. [Google Scholar] [CrossRef]
- Bullock, A.M. Solar Ultraviolet Radiation: A Potential Environmental Hazard in the Cultivation of Farmed Finfish. In Recent Advances in Aquaculture: Volume 3; Muir, J.F., Roberts, R.J., Eds.; Springer Netherlands: Dordrecht, The Netherlands, 1988; pp. 139–224. ISBN 978-94-011-9743-4. [Google Scholar]
- Bui, S.; Oppedal, F.; Sievers, M.; Dempster, T. Behaviour in the toolbox to outsmart parasites and improve fish welfare in aquaculture. Rev. Aquac. 2019, 11, 168–186. [Google Scholar] [CrossRef]
- El Naggar, G.O.; John, G.; Rezk, M.A.; Elwan, W.; Yehia, M. Effect of varying density and water level on the spawning response of African catfish Clarias gariepinus: Implications for seed production. Aquaculture 2006, 261, 904–907. [Google Scholar] [CrossRef]
- McCormick, S.D.; Hansen, L.P.; Quinn, T.P.; Saunders, R.L. Movement, migration, and smolting of Atlantic salmon (Salmo salar). Can. J. Fish. Aquat. Sci. 1998, 55, 77–92. [Google Scholar] [CrossRef]
- Huntingford, F.A.; Adams, C.; Braithwaite, V.A.; Kadri, S.; Pottinger, T.G.; Sandøe, P.; Turnbull, J.F. Current issues in fish welfare. J. Fish Biol. 2006, 68, 332–372. [Google Scholar] [CrossRef]
- Methods in Reproductive Aquaculture: Marine and Freshwater Species; Marine Biology Series; Cabrita, E., Robles, V., Herráez, P., Eds.; CRC Press: Boca Raton, FL, USA, 2009; ISBN 978-0-8493-8053-2. [Google Scholar]
- Conte, F.S. Stress and the welfare of cultured fish. Appl. Anim. Behav. Sci. 2004, 86, 205–223. [Google Scholar] [CrossRef]
- Ellis, T.; Scott, S.; Bromage, N.; North, B.; Porter, M. What is stocking density? Trout News 2001, 32, 35–37. [Google Scholar]
- Turnbull, J.F.; North, B.P.; Ellis, T.; Adams, C.E.; Bron, J.; MacIntyre, C.M.; Huntingford, F.A. Stocking density and the welfare of farmed salmonids. Fish Welf. 2008, 111–120. [Google Scholar] [CrossRef]
- Gilmour, K.M.; DiBattista, J.D.; Thomas, J.B. Physiological Causes and Consequences of Social Status in Salmonid Fish. Integr. Comp. Biol. 2005, 45, 263–273. [Google Scholar] [CrossRef] [PubMed]
- Sørensen, C.; Nilsson, G.E.; Summers, C.H.; Øverli, Ø. Social stress reduces forebrain cell proliferation in rainbow trout (Oncorhynchus mykiss). Behav. Brain Res. 2012, 227, 311–318. [Google Scholar] [CrossRef]
- Cammarata, M.; Vazzana, M.; Accardi, D.; Parrinello, N. Seabream (Sparus aurata) long-term dominant-subordinate interplay affects phagocytosis by peritoneal cavity cells. Brain Behav. Immun. 2012, 26, 580–587. [Google Scholar] [CrossRef]
- Sloman, K.A.; Metcalfe, N.B.; Taylor, A.C.; Gilmour, K.M. Plasma cortisol concentrations before and after social stress in rainbow trout and brown trout. Physiol. Biochem. Zool. 2001, 74, 383–389. [Google Scholar] [CrossRef]
- Sloman, K.A.; Montpetit, C.J.; Gilmour, K.M. Modulation of catecholamine release and cortisol secretion by social interactions in the rainbow trout, Oncorhynchus mykiss. Gen. Comp. Endocrinol. 2002, 127, 136–146. [Google Scholar] [CrossRef]
- Olsen, R.E.; Ringø, E. Dominance hierarchy formation in Arctic charr Salvelinus alpinus (L.): Nutrient digestibility of subordinate and dominant fish. Aquac. Res. 1999, 30, 667–671. [Google Scholar] [CrossRef]
- Hatlen, B.; Grisdale-Helland, B.; Helland, S.J. Growth variation and fin damage in Atlantic cod (Gadus morhua L.) fed at graded levels of feed restriction. Aquaculture 2006, 261, 1212–1221. [Google Scholar] [CrossRef]
- Faisal, M.; Chiappelli, F.; Ahmed, I.I.; Cooper, E.L.; Weiner, H. Social confrontation “Stress” in aggressive fish is associated with an endogenous opioid-mediated suppression of proliferative response to mitogens and nonspecific cytotoxicity. Brain Behav. Immun. 1989, 3, 223–233. [Google Scholar] [CrossRef]
- Khater, E.G. Simulation Model for Design and Management of Water Recirculating Systems in Aquaculture; Agricultural Engineering Department, Faculty of Agriculture, Moshtohor, Benha University: Moshtohor, Egypt, 2012. [Google Scholar]
- Malone, R. Recirculating Aquaculture Tank Production Systems; USDA, Southern Regional Aquaculture Center: Stoneville, MS, USA, 2013; p. 12. [Google Scholar]
- Powell, A.; Treasurer, J.W.; Pooley, C.L.; Keay, A.J.; Lloyd, R.; Imsland, A.K.; de Leaniz, C.G. Use of lumpfish for sea-lice control in salmon farming: Challenges and opportunities. Rev. Aquac. 2018, 10, 683–702. [Google Scholar] [CrossRef]
- Daborn, G.R.; Gregory, R.S. Occurrence, distribution, and feeding habits of juvenile lumpfish, Cyclopterus lumpus L. in the Bay of Fundy. Can. J. Zool. 1983, 61, 797–801. [Google Scholar] [CrossRef]
- Lines, J.A.; Spence, J. Safeguarding the welfare of farmed fish at harvest. Fish Physiol. Biochem. 2012, 38, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Boglione, C.; Gisbert, E.; Gavaia, P.; Witten, P.; Moren, M.; Fontagné, S.; Koumoundouros, G. Skeletal anomalies in reared European fish larvae and juveniles. Part 2: Main typologies, occurrences and causative factors. Rev. Aquac. 2013, 5, S121–S167. [Google Scholar] [CrossRef]
- Boglione, C.; Gavaia, P.; Koumoundouros, G.; Gisbert, E.; Moren, M.; Fontagné, S.; Witten, P.E. Skeletal anomalies in reared European fish larvae and juveniles. Part 1: Normal and anomalous skeletogenic processes. Rev. Aquac. 2013, 5, S99–S120. [Google Scholar] [CrossRef]
- Soares, F.; Fernández, I.; Costas, B.; Gavaia, P.J. Non-infectious disorders of warmwater fish. In Diseases and Disorders of Finfish in Cage Culture; CAB International: Wallingford, UK, 2014. [Google Scholar]
- European Food Safety Authority (EFSA). Species-specific welfare aspects of the main systems of stunning and killing of farmed Seabass and Seabream: Species-specific welfare aspects of the main systems of stunning and killing of farmed Seabass and Seabream. EFSA J. 2009, 1010, 1–52. [Google Scholar]
- Digre, H.; Erikson, U.; Misimi, E.; Lambooij, B.; Van De Vis, H. Electrical stunning of farmed Atlantic cod Gadus morhua L.: A comparison of an industrial and experimental method. Aquac. Res. 2010, 41, 1190–1202. [Google Scholar]
- European Food Safety Authority (EFSA). Species-specific welfare aspects of the main systems of stunning and killing of farmed Atlantic Salmon. EFSA J. 2009, 1012, 1–77. [Google Scholar]
- European Food Safety Authority (EFSA). Species-specific welfare aspects of the main systems of stunning and killing of farmed fish: Rainbow Trout. EFSA J. 2009, 7. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA). Species-specific welfare aspects of the main systems of stunning and killing of farmed Carp. EFSA J. 2009, 1013, 1–37. [Google Scholar]
- European Food Safety Authority (EFSA). Species-specific welfare aspects of the main systems of stunning and killing of farmed tuna. EFSA J. 2009, 1072, 1–53. [Google Scholar]
- European Food Safety Authority (EFSA). Scientific opinion of the panel on animal health and welfare on a request from the European commission on animal welfare aspects of husbandry systems for farmed European seabass and gilthead seabream. EFSA J. 2008, 844, 1–21. [Google Scholar]
- Lambooij, E.; Gerritzen, M.A.; Reimert, H.; Burggraaf, D.; van de Vis, J.W. A humane protocol for electro-stunning and killing of Nile tilapia in fresh water. Aquaculture 2008, 275, 88–95. [Google Scholar] [CrossRef]
- Robb, D.H.F.; Kestin, S.C. Methods Used to Kill Fish: Field Observations and Literature Reviewed. Anim. Welf. 2002, 11, 269–282. [Google Scholar]
- Tidwell, J.H.; Allan, G.L. Fish as food: aquaculture’s contribution. Ecological and economic impacts and contributions of fish farming and capture fisheries. EMBO Rep. 2001, 2, 958–963. [Google Scholar] [CrossRef]
- Turchini, G.M.; Torstensen, B.E.; Ng, W.-K. Fish oil replacement in finfish nutrition. Rev. Aquac. 2009, 1, 10–57. [Google Scholar] [CrossRef]
- Jensen, U. Probabilistic Risk Analysis: Foundations and Methods. J. Am. Stat. Assoc. 2002, 97, 925–926. [Google Scholar] [CrossRef]
- Teletchea, F.; Fontaine, P. Levels of domestication in fish: Implications for the sustainable future of aquaculture. Fish Fish. 2012, 15, 181–195. [Google Scholar] [CrossRef]
- Teletchea, F. Domestication of Marine Fish Species: Update and Perspectives. J. Mar. Sci. Eng. 2015, 3, 1227–1243. [Google Scholar] [CrossRef]
- Huntingford, F.A.; Kadri, S. Welfare and fish. Fish Welf. 2008, 1, 19–32. [Google Scholar]
- Poli, B.M.; Parisi, G.; Scappini, F.; Zampacavallo, G. Fish welfare and quality as affected by pre-slaughter and slaughter management. Aquac. Int. 2005, 13, 29–49. [Google Scholar] [CrossRef]
- Morzel, M.; Sohier, D.; Van de Vis, H. Evaluation of slaughtering methods for turbot with respect to animal welfare and flesh quality. J. Sci. Food Agric. 2003, 83, 19–28. [Google Scholar] [CrossRef]
- Napolitano, F.; Girolami, A.; Braghieri, A. Consumer liking and willingness to pay for high welfare animal-based products. Trends Food Sci. Technol. 2010, 21, 537–543. [Google Scholar] [CrossRef]
- Honkanen, P.; Olesen, I.; Mejdell, C.; Nielsen, H.M.; Grimsrud, K.; Gamborg, C.; Navrud, S.; Ellingsen, K.; Sandøe, P. Who cares about fish welfare? A Norwegian study. Br. Food J. 2015, 117, 257–273. [Google Scholar]
- Ottar Olsen, S.; Honkanen, P. Environmental and animal welfare issues in food choice: The case of farmed fish. Br. Food J. 2009, 111, 293–309. [Google Scholar]
- Adams, C.E.; Turnbull, J.F.; Bell, A.; Bron, J.E.; Huntingford, F.A. Multiple determinants of welfare in farmed fish: Stocking density, disturbance, and aggression in Atlantic salmon (Salmo salar). Can. J. Fish. Aquat. Sci. 2007, 64, 336–344. [Google Scholar] [CrossRef]
- Foss, A.; Grimsbø, E.; Vikingstad, E.; Nortvedt, R.; Slinde, E.; Roth, B. Live chilling of Atlantic salmon: Physiological response to handling and temperature decrease on welfare. Fish Physiol. Biochem. 2012, 38, 565–571. [Google Scholar] [CrossRef] [PubMed]
- Cañon Jones, H.A.; Noble, C.; Damsgård, B.; Pearce, G.P. Investigating the influence of predictable and unpredictable feed delivery schedules upon the behaviour and welfare of Atlantic salmon parr (Salmo salar) using social network analysis and fin damage. Appl. Anim. Behav. Sci. 2012, 138, 132–140. [Google Scholar] [CrossRef]
- Noble, C.; Berrill, I.; Waller, B.; Kankainen, M.; Setala, J.; Honkanen, P.; Mejdell, C.M.; Turnbull, J.; Damsgard, B.; Schneider, O.; et al. A multi-disciplinary framework for bio-economic modeling in aquaculture: A welfare case study. Aquac. Econ. Manag. 2012, 16, 297–314. [Google Scholar] [CrossRef]
| Criterion | Type of Constraint | Possible Effects |
|---|---|---|
| 1. Home range | Spatial | Disrupted swimming behaviour, impaired movement. |
| 2. Depth range | Spatial | Disrupted swimming behaviour, impaired movement. |
| 3. Migration and habitat change | Spatial | Disrupted swimming behaviour, migration drivers not met (feeding, reproduction, habitat choice, etc.). |
| 4. Free reproduction | Physiological, Behavioural | Impaired mating, courting and spawning behaviours, inbreeding, disrupted sexual selection. |
| 5. Aggregation | Social | Impaired communication and swimming patterns, disrupted social networks, territoriality and shoaling configurations, increased overall cost of high social stress. |
| 6. Aggression | Behavioural, social | Increased fighting and injuries, increased overall cost of high social stress. |
| 7. Substrate and shelter | Environmental, ecological | Altered swimming and/or feeding patterns, reduced opportunities for escape, disrupted flight response, increased overall cost of high social stress. |
| 8. Handling | Physiological, Mental | Infection, injury, anxiety, chronic stress, pain, death. |
| 9. Malformations | Physiological | Impaired mobility, feeding, breathing or other aspects of biology. |
| 10. Slaughter | Death | Extreme pain and suffering. |
| Criterion | Question(s) | Possible Answers |
|---|---|---|
| 1 | Are minimal farming conditions likely to provide the home range of the species? | Likelihood is High. Likelihood is Low. Data are Unclear. |
| Is there potential for improvement? | Potential is High. Potential is Medium. Potential is Low. Data are Unclear. There are No findings to support scoring. | |
| How certain are these findings? | Certainty is High. Certainty is Medium. Certainty is Low. There are No findings to support scoring. | |
| 2 | Are minimal farming conditions likely to provide the depth range of the species? | Likelihood is High. Likelihood is Low. Data are Unclear. |
| Is there potential for improvement? | Potential is High. Potential is Medium. Potential is Low. Data are Unclear. There are No findings to support scoring. | |
| How certain are these findings? | Certainty is High. Certainty is Medium. Certainty is Low. There are No findings to support scoring. | |
| 3 | Are minimal farming conditions compatible with the migrating or habitat-changing behaviour of the species? | Likelihood is High. Likelihood is Low. Data are Unclear. |
| Is there potential for improvement? | Potential is High. Potential is Medium. Potential is Low. Data are Unclear. There are No findings to support scoring. | |
| How certain are these findings? | Certainty is High. Certainty is Medium. Certainty is Low. There are No findings to support scoring. | |
| 4 | Is the species likely to reproduce in captivity without manipulation? | Likelihood is High. Likelihood is Low. Data are Unclear. |
| Is there potential to allow for it under farming conditions? | Potential is High. Potential is Medium. Potential is Low. Data are Unclear. There are No findings to support scoring. | |
| How certain are these findings? | Certainty is High. Certainty is Medium. Certainty is Low. There are No findings to support scoring. | |
| 5 | Is the aggregation imposed by minimal farming conditions likely to be compatible with the natural behaviour of the species? | Likelihood is High. Likelihood is Low. Data are Unclear. |
| Is there potential to allow for it under farming conditions? | Potential is High. Potential is Medium. Potential is Low. Data are Unclear. There are No findings to support scoring. | |
| How certain are these findings? | Certainty is High. Certainty is Medium. Certainty is Low. There are No findings to support scoring. | |
| 6 | Is the species likely to be non-aggressive and non-territorial? | Likelihood is High. Likelihood is Low. Data are Unclear. |
| Is there potential for improvement? | Potential is High. Potential is Medium. Potential is Low. Data are Unclear. There are No findings to support scoring. | |
| How certain are these findings? | Certainty is High. Certainty is Medium. Certainty is Low. There are No findings to support scoring. | |
| 7 | Are minimal farming conditions likely to match the natural substrate and shelter needs of the species? | Likelihood is High. Likelihood is Low. Data are Unclear. |
| Is there potential for improvement? | Potential is High. Potential is Medium. Potential is Low. Data are Unclear. There are No findings to support scoring. | |
| How certain are these findings? | Certainty is High. Certainty is Medium. Certainty is Low. There are No findings to support scoring. | |
| 8 | Are minimal farming conditions (handling, confinement etc.) likely not to stress the individuals of the species? | Likelihood is High. Likelihood is Low. Data are Unclear. |
| Is there potential for improvement? | Potential is High. Potential is Medium. Potential is Low. Data are Unclear. There are No findings to support scoring. | |
| How certain are these findings? | Certainty is High. Certainty is Medium. Certainty is Low. There are No findings to support scoring. | |
| 9 | Are malformations of this species likely to be rare under farming conditions? | Likelihood is High. Likelihood is Low. Data are Unclear. |
| Is there potential for improvement? | Potential is High. Potential is Medium. Potential is Low. Data are Unclear. There are No findings to support scoring. | |
| How certain are these findings? | Certainty is High. Certainty is Medium. Certainty is Low. There are No findings to support scoring. | |
| 10 | Is a humane slaughter protocol likely to be applied under minimal farming conditions? | Likelihood is High. Likelihood is Low. Data are Unclear. |
| Is there potential for improvement? | Potential is High. Potential is Medium. Potential is Low. Data are Unclear. There are No findings to support scoring. | |
| How certain are these findings? | Certainty is High. Certainty is Medium. Certainty is Low. There are No findings to support scoring. |
| Likelihood | Potential | Certainty | Domestication | Improvement Capacity | |
|---|---|---|---|---|---|
| Likelihood | 1 | ||||
| Potential | 0.60 * | 1 | |||
| Certainty | 0.21 | 0.56 * | 1 | ||
| Domestication | 0.02 | 0.11 | 0.14 | 1 | |
| Improvement Capacity | 0.08 | 0.80 * | 0.49 | 0.18 | 1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saraiva, J.L.; Arechavala-Lopez, P.; Castanheira, M.F.; Volstorf, J.; Heinzpeter Studer, B. A Global Assessment of Welfare in Farmed Fishes: The FishEthoBase. Fishes 2019, 4, 30. https://doi.org/10.3390/fishes4020030
Saraiva JL, Arechavala-Lopez P, Castanheira MF, Volstorf J, Heinzpeter Studer B. A Global Assessment of Welfare in Farmed Fishes: The FishEthoBase. Fishes. 2019; 4(2):30. https://doi.org/10.3390/fishes4020030
Chicago/Turabian StyleSaraiva, João Luis, Pablo Arechavala-Lopez, Maria Filipa Castanheira, Jenny Volstorf, and Billo Heinzpeter Studer. 2019. "A Global Assessment of Welfare in Farmed Fishes: The FishEthoBase" Fishes 4, no. 2: 30. https://doi.org/10.3390/fishes4020030
APA StyleSaraiva, J. L., Arechavala-Lopez, P., Castanheira, M. F., Volstorf, J., & Heinzpeter Studer, B. (2019). A Global Assessment of Welfare in Farmed Fishes: The FishEthoBase. Fishes, 4(2), 30. https://doi.org/10.3390/fishes4020030







