Enrichment Increases Aggression in Zebrafish
Abstract
1. Introduction
2. Results
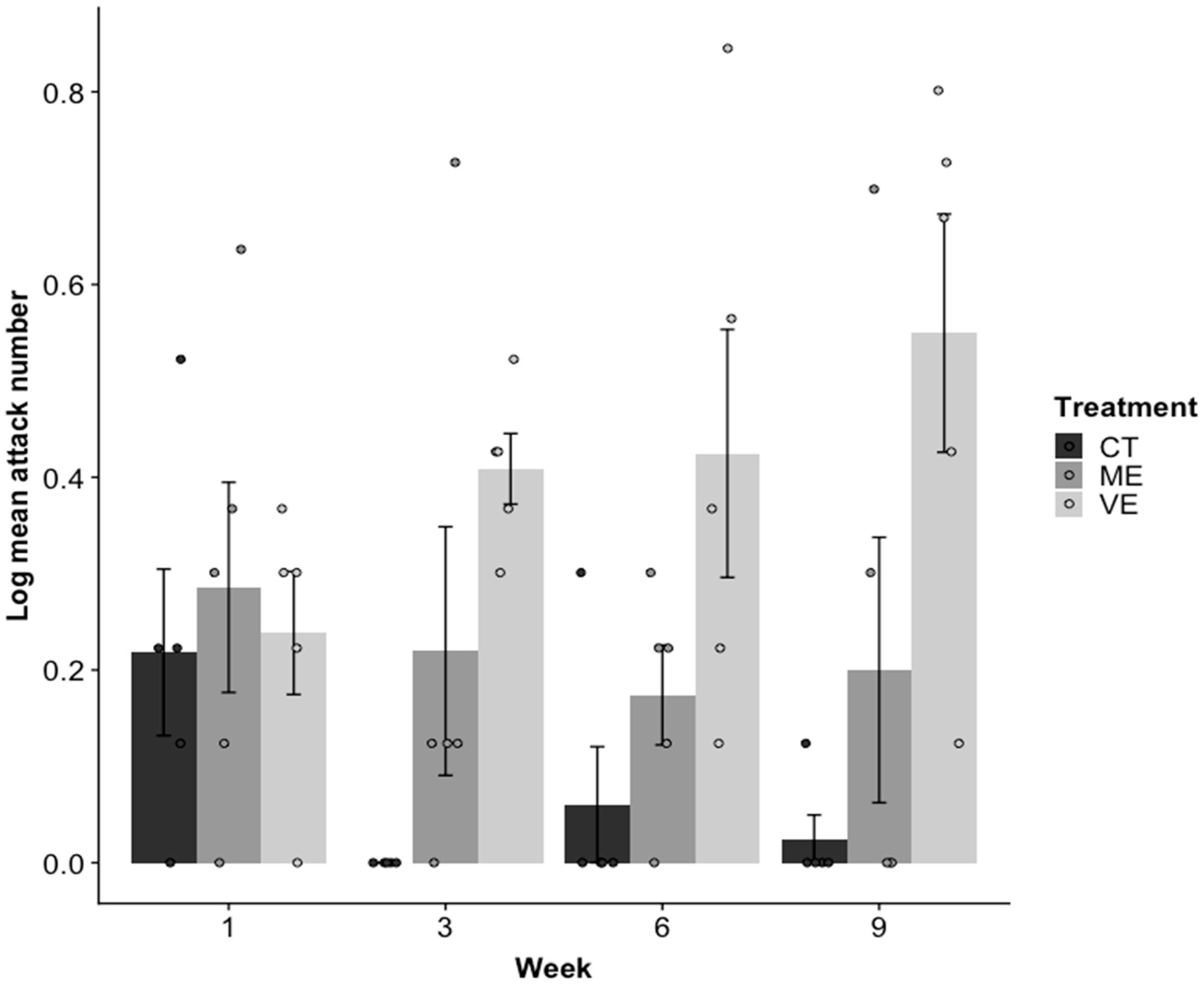
2.1. Aggression
2.2. Boldness
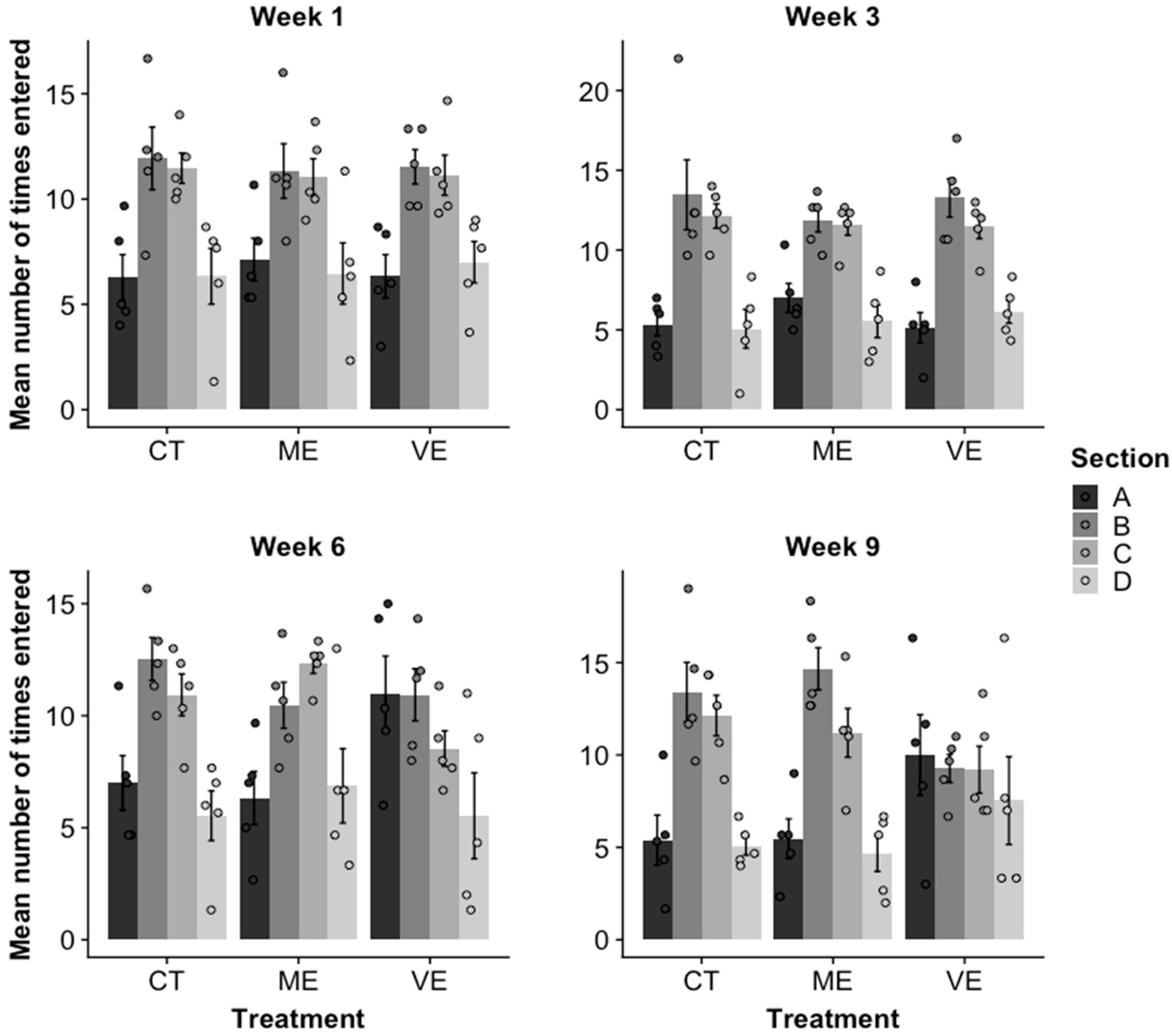
2.3. Activity Level
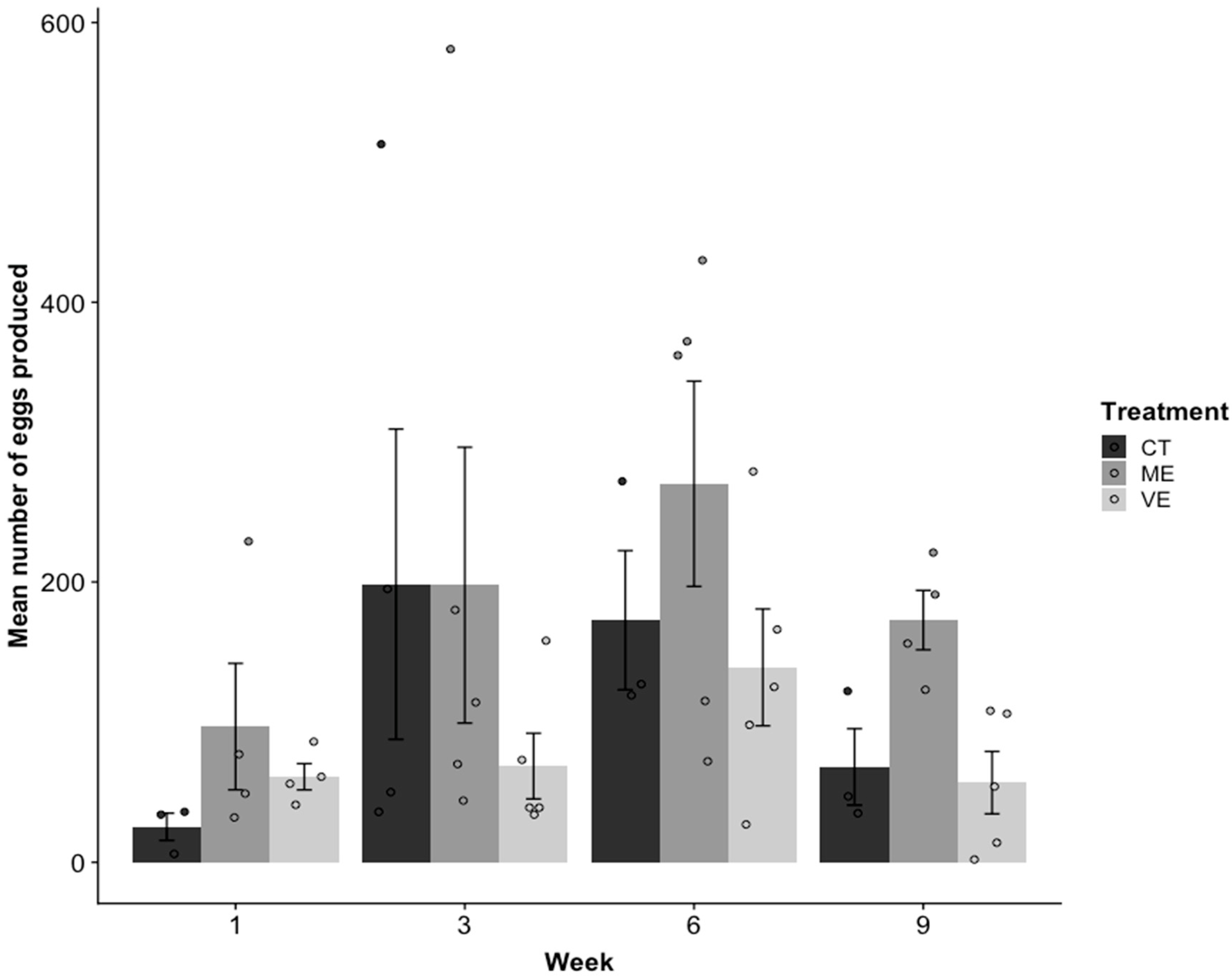
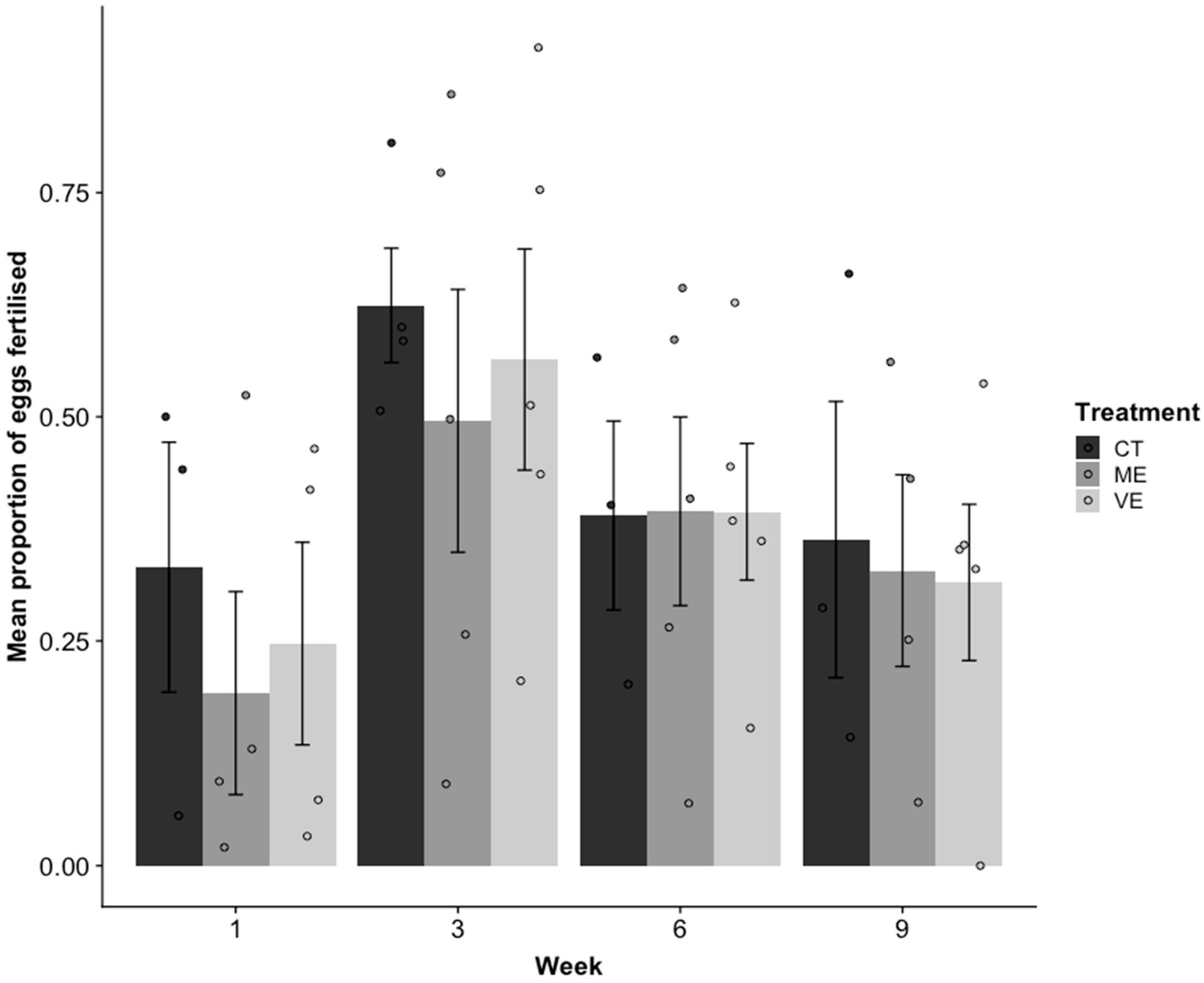
2.4. Reproduction
2.5. Growth
3. Discussion
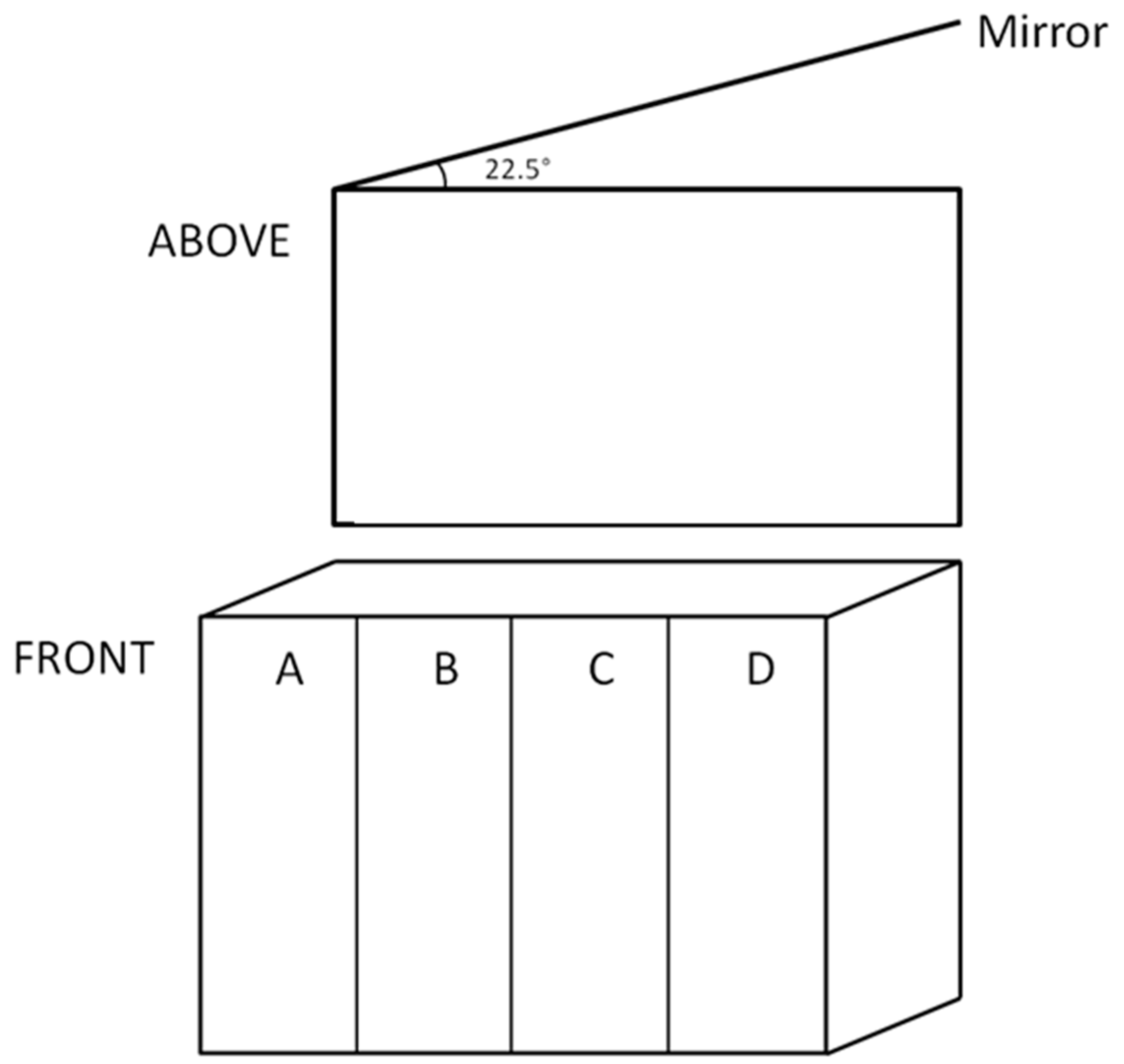
4. Methods
4.1. Fish, Treatments and Growth
4.2. Behavioural Tests
4.2.1. Aggression
4.2.2. Boldness
4.2.3. Activity Level
4.2.4. Reproduction
4.3. Data Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Williams, T.D.; Readman, G.D.; Owen, S.F. Key issues concerning environmental enrichment for laboratory-held fish species. Lab. Anim. 2009, 43, 107–120. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.P.; Taylor, V.P. Environmental enrichment resources for laboratory animals: 1965–1995: Birds, cats, dogs, farm animals, ferrets, rabbits and rodents. AWIC Resour. Ser. 1995, 2, 145–212. [Google Scholar]
- Newberry, R. Environmental enrichment: Increasing the biological relevance of captive environments. Appl. Anim. Behav. Sci. 1995, 44, 229–243. [Google Scholar] [CrossRef]
- Braithwaite, V.A.; Salvanes, A.G.V. Environmental variability in the early rearing environment generates behaviourally flexible cod: Implications for rehabilitating wild populations. Proc. R. Soc. Lond. B 2005, 272, 1107–1113. [Google Scholar] [CrossRef] [PubMed]
- Johnsson, J.I.; Brockmark, S.; Näslund, J. Environmental effects on behavioural development consequences for fitness of captive-reared fishes in the wild. J. Fish Biol. 2014, 85, 1946–1971. [Google Scholar] [CrossRef] [PubMed]
- Rosengren, M.; Kvingedal, E.; Näslund, J.; Johnsson, J.I.; Sundell, K. Born to be wild: Effects of rearing density and environmental enrichment on stress, welfare, and smolt migration in hatchery-reared Atlantic salmon. Can. J. Fish. Aquat. Sci. 2017, 74, 396–405. [Google Scholar] [CrossRef]
- Ellis, T.; North, B.; Scott, A.P.; Bromage, N.R.; Porter, M.; Gadd, D. The relationships between stocking density and welfare in farmed rainbow trout. J. Fish Biol. 2002, 61, 493–531. [Google Scholar] [CrossRef]
- Ashley, P.J. Fish welfare: Current issues in aquaculture. Appl. Anim. Behav. Sci. 2007, 104, 199–235. [Google Scholar] [CrossRef]
- Näslund, J.; Johnsson, J.I. Environmental enrichment for fish in captive environments: Effects of physical structures and substrates. Fish Fish. 2016, 17, 1–30. [Google Scholar] [CrossRef]
- Kemp, P.S.; Armstrong, J.D.; Gilvear, D.J. Behavioural responses of juvenile Atlantic salmon (Salmo salar) to presence of boulders. River Res. Appl. 2005, 21, 1053–1060. [Google Scholar] [CrossRef]
- Kihslinger, R.L.; Lema, S.C.; Nevitt, G.A. Environmental rearing conditions produce forebrain differences in wild Chinook salmon Oncorhynchus tshawytscha. Comp. Biochem. Physiol. A 2006, 145, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Spence, R.; Magurran, A.E.; Smith, C. Spatial cognition in zebrafish: The role of strain and rearing environment. Anim. Cogn. 2011, 14, 607–612. [Google Scholar] [CrossRef] [PubMed]
- Papoutsoglou, S.E.; Mylonakis, G.; Miliou, H.; Karakatsouli, N.P.; Chadio, S. Effects of background colour on growth performances and physiological responses of scaled carp (Cyprinus carpio L.) reared in a closed circulated system. Aquac. Eng. 2000, 22, 309–318. [Google Scholar] [CrossRef]
- Papoutsoglou, S.E.; Karakatsouli, N.; Louizos, E.; Chadio, S.; Kalogiannis, D.; Dalla, C.; Polissidis, A.; Papadopoulou-Daifoti, Z. Effect of Mozart’s music (Romanze-Andante of “Eine Kleine Nacht Musik”, sol major, K525) stimulus on common carp (Cyprinus carpio L.) physiology under different light conditions. Aquac. Eng. 2007, 36, 61–72. [Google Scholar] [CrossRef]
- Martinez-Cardenas, L.; Purser, G.J. Effect of tank colour on Artemia ingestion, growth and survival in cultured early juvenile pot-bellied seahorses (Hippocampus abdominalis). Aquaculture 2007, 264, 92–100. [Google Scholar] [CrossRef]
- Mesquita, F.O.; Godinho, H.P.; Azevedo, P.G.; Young, R.J. A preliminary study into the effectiveness of stroboscopic light as an aversive stimulus for fish. Appl. Anim. Behav. Sci. 2008, 111, 402–407. [Google Scholar] [CrossRef]
- Monk, J.; Puvanendran, V.; Brown, J.A. Does different tank bottom colour affect the growth, survival and foraging behaviour of Atlantic cod (Gadus morhua) larvae? Aquaculture 2008, 277, 197–202. [Google Scholar] [CrossRef]
- Cobcroft, J.M.; Battaglene, S.C. Jaw malformation in striped trumpeter Latris lineate larvae linked to walling behaviour and tank colour. Aquaculture 2009, 289, 274–282. [Google Scholar] [CrossRef]
- El-Sayed, A.F.M.; El-Ghobashy, A.E. Effects of tank colour and feed colour on growth and feed utilization of thinlip mullet (Liza ramada) larvae. Aquac. Res. 2011, 42, 1163–1169. [Google Scholar] [CrossRef]
- Basquill, S.P.; Grant, J.W.A. An increase in habitat complexity reduces aggression and monopolization of food by zebra fish (Danio rerio). Can. J. Zool. 1998, 76, 770–772. [Google Scholar] [CrossRef]
- Carfagnini, A.G.; Rodd, F.H.; Jeffers, K.B.; Bruce, A.E.E. The effects of habitat complexity on aggression and fecundity in zebrafish (Danio rerio). Environ. Biol. Fishes 2009, 86, 403–409. [Google Scholar] [CrossRef]
- Hasegawa, K.; Maekawa, K. Potential of habitat complexity for mitigating interference competition between native and non-native salmonid species. Can. J. Zool. 2008, 86, 386–393. [Google Scholar] [CrossRef]
- Roberts, L.J.; Taylor, J.; Garcia de Leaniz, C. Environmental enrichment reduces maladaptive risk-taking behaviour in salmon reared for conservation. Biol. Conserv. 2011, 144, 1972–1979. [Google Scholar] [CrossRef]
- Finstad, A.G.; Einum, S.; Forseth, T.; Ugedal, O. Shelter availability affects behaviour, size-dependent and mean growth of juvenile Atlantic salmon. Freshwat. Biol. 2007, 52, 1710–1718. [Google Scholar] [CrossRef]
- Reed, B.; Jennings, M. Guidance on the housing and care of zebrafish, Danio rerio; RSPCA: Horsham, UK, 2010. [Google Scholar]
- Kistler, C.; Hegglin, D.; Würbel, H.; König, B. Preference for structured environment on zebrafish (Danio rerio) and checker barbs (Puntius oligolepis). Appl. Anim. Behav. Sci. 2011, 135, 318–327. [Google Scholar] [CrossRef]
- Corkum, L.D.; Cronin, D.J. Habitat complexity reduces aggression and enhances consumption in crayfish. J. Ethol. 2004, 22, 23–27. [Google Scholar] [CrossRef]
- Hamilton, I.M.; Dill, L.M. Monopolization of food by zebrafish (Danio rerio) increases in risky habitats. Can. J. Zool. 2002, 80, 2164–2169. [Google Scholar] [CrossRef]
- Larson, E.T.; O’Malley, D.M.; Melloni, R.H. Aggression and vasotocin are associated with dominant-subordinate relationships in zebrafish. Behav. Brain Res. 2006, 167, 94–102. [Google Scholar] [CrossRef]
- Spence, R.; Gerlach, G.; Lawrence, C.; Smith, C. The behaviour and ecology of the zebrafish, Danio rerio. Biol. Rev. 2008, 83, 13–34. [Google Scholar] [CrossRef]
- Watt, P.J.; Skinner, A.; Hale, M.; Nakagawa, S.; Burke, T. Small subordinate male advantage in the zebrafish. Ethology 2011, 117, 1003–1008. [Google Scholar] [CrossRef]
- Spence, R.; Smith, C. Male territoriality mediates density and sex ratio effects on oviposition in the zebrafish, Danio rerio. Anim. Behav. 2005, 69, 1317–1323. [Google Scholar] [CrossRef]
- Grant, J.W.A.; Kramer, D.L. Temporal clumping of food arrival reduces its monopolization and defense by zebrafish, Brachydanio rerio. Anim. Behav. 1992, 44, 101–110. [Google Scholar] [CrossRef]
- McCarthy, I.D.; Carter, C.G.; Houlihan, D.F. The effect of feeding hierarchy on individual variability in daily feeding of rainbow trout, Oncorhynchus mykiss. J. Fish Biol. 1992, 41, 257–263. [Google Scholar] [CrossRef]
- Johnson, D.W. Combined effects of condition and density on post-settlement survival and growth of a marine fish. Oecologia 2008, 155, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Samhouri, J.F. Food supply influences offspring provisioning but not density-dependent fecundity in a marine fish. Ecology 2009, 90, 3478–3488. [Google Scholar] [CrossRef]
- Pickering, A.D.; Pottinger, T.G. Stress responses and disease resistance in salmonid fish: Effects of chronic elevation of plasma cortisol. Fish Physiol. Biochem. 1989, 7, 253–258. [Google Scholar] [CrossRef]
- Fox, H.E.; White, S.A.; Kao, M.H.F.; Fernald, R.D. Stress and dominance in a social fish. J. Neurosci. 1997, 17, 6463–6469. [Google Scholar] [CrossRef]
- Pottinger, T.G.; Pickering, A.D. The influence of social interaction on the acclimation of rainbow trout, Oncorhynchus mykiss, to chronic stress. J. Fish Biol. 1992, 41, 435–447. [Google Scholar] [CrossRef]
- Wilkes, L.; Owen, S.F.; Readman, G.D.; Sloman, K.A.; Wilson, R.W. Does structural enrichment for toxicology studies improve zebrafish welfare? Appl. Anim. Behav. Sci. 2012, 139, 143–150. [Google Scholar] [CrossRef]
- Keck, V.A.; Edgerton, D.S.; Hajizadeh, S.; Swift, L.L.; Dupont, W.D.; Lawrence, C.; Boyd, K.L. Effects of complexity on pair-housed zebrafish. J. Am. Assoc. Lab. Anim. Sci. 2015, 54, 378–383. [Google Scholar]
- Korte, S.M.; Olivier, B.; Koolhaas, J.M. A new animal welfare concept based on allostasis. Phys. Behav. 2007, 92, 422–428. [Google Scholar] [CrossRef]
- Næsje, T.F.; Thorstad, E.B.; Forseth, T.; Aursand, M.; Saksgård, R.; Finstad, A.G. Lipid class content as an indicator of critical periods for survival in juvenile Atlantic salmon (Salmo salar). Ecol. Freshw. Fish 2006, 15, 572–577. [Google Scholar] [CrossRef]
- Von Krogh, K.; Sørensen, C.; Nilsson, G.E.; Øverli, Ø. Forebrain cell proliferation, behavior, and physiology of zebrafish, Danio rerio, kept in enriched or barren environments. Physiol. Behav. 2010, 101, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Garduño-Paz, M.V.; Couderc, S.; Adams, C.E. Habitat complexity modulates phenotype expression through developmental plasticity in the threespine stickleback. Biol. J. Linn. Soc. 2010, 100, 407–413. [Google Scholar] [CrossRef]
- Brydges, N.M.; Braithwaite, V.A. Does environmental enrichment affect the behaviour of fish commonly used in laboratory work? Appl. Anim. Behav. Sci. 2009, 118, 137–143. [Google Scholar] [CrossRef]
- Burns, J.G.; Saravanan, A.; Rodd, F.H. Rearing environment affects the brain size of guppies: Lab-reared guppies have smaller brains than wild-caught guppies. Ethology 2009, 115, 122–133. [Google Scholar] [CrossRef]
- Réale, D.; Reader, S.M.; Sol, D.; McDougall, P.T.; Dingemanse, N.J. Intergrating animal temperament within ecology and evolution. Biol. Rev. 2007, 82, 291–318. [Google Scholar] [CrossRef]
- Ariyomo, T.O.; Watt, P.J. The effect of variation in boldness and aggressiveness on the reproductive success of zebrafish. Anim. Behav. 2012, 83, 41–46. [Google Scholar] [CrossRef]
- Höglund, E.; Balm, P.H.M.; Winberg, S. Behavioural and neuroendocrine effects of environmental background colour and social interaction in Arctic charr (Salvelinus alpines). J. Exp. Biol. 2002, 205, 2535–2543. [Google Scholar]
- Rosemberg, D.B.; Rico, E.P.; Mussulini, B.H.M.; Paiato, A.L.; Calcagnotto, M.E.; Bonan, C.D.; Dias, R.E.; Blaser, R.E.; Souza, D.O.; de Oliveira, D.L. Differences in spatio-temporal behavior of zebrafish in the open tank paradigm after a short-period confinement into dark and bright environments. PLoS ONE 2011, 6, e19317. [Google Scholar] [CrossRef]
- Gerlai, R.; Lahav, M.; Guo, S.; Rosenthal, A. Drinks like a fish: Zebra fish (Danio rerio) as a behaviour genetic model to study alcohol effects. Pharmacol. Biochem. Behav. 2000, 67, 773–782. [Google Scholar] [CrossRef]
- Moretz, J.A.; Martins, E.P.; Robison, B.D. The effects of early and adult social environment on zebrafish (Danio rerio) behavior. Environ. Biol. Fish. 2007, 80, 91–101. [Google Scholar] [CrossRef]
- Walsh, R.N.; Cummins, R.A. The open-field test: A critical review. Psychol. Bull. 1976, 83, 482–504. [Google Scholar] [CrossRef]
- Moretz, J.A.; Martins, E.P.; Robison, B.D. Behavioral syndromes and the evolution of correlated behaviour in zebrafish. Behav. Ecol. 2007, 18, 556–562. [Google Scholar] [CrossRef]
- Burns, J.G. The validity of three tests of temperament in guppies, Poecilia reticulata. J. Comp. Psychol. 2008, 122, 344–356. [Google Scholar] [CrossRef] [PubMed]
- Westerfield, M. The Zebrafish Book. A Guide for the Laboratory Use of Zebrafish (Danio rerio), 4th ed.; Chapman and Hall: London, UK, 1995. [Google Scholar]
- Brand, M.; Granato, M.; Nüsslein-Volhard, C. Keeping and raising zebrafish. In Zebrafish: A Practical Approach; Nüsslein-Volhard, C., Dahm, R., Eds.; Oxford University Press: Oxford, UK, 2002; pp. 7–37. [Google Scholar]
- Spence, R.; Ashton, R.; Smith, C. Oviposition decisions are mediated by spawning site quality in wild and domesticated zebrafish, Danio rerio. Behaviour 2007, 144, 953–966. [Google Scholar] [CrossRef]
- Mertens, J. Year-round controlled mass reproduction of the zebrafish Brachydanio rerio (Hamilton-Buchanan). Aquaculture 1973, 2, 245–249. [Google Scholar] [CrossRef]
- Gellert, G.; Heinrichsdorff, J. Effect of age on the susceptibility of zebrafish eggs to industrial waste water. Water Res. 2001, 35, 3754–3757. [Google Scholar] [CrossRef]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2011. [Google Scholar]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Woodward, M.A.; Winder, L.A.; Watt, P.J. Enrichment Increases Aggression in Zebrafish. Fishes 2019, 4, 22. https://doi.org/10.3390/fishes4010022
Woodward MA, Winder LA, Watt PJ. Enrichment Increases Aggression in Zebrafish. Fishes. 2019; 4(1):22. https://doi.org/10.3390/fishes4010022
Chicago/Turabian StyleWoodward, Melanie A., Lucy A. Winder, and Penelope J. Watt. 2019. "Enrichment Increases Aggression in Zebrafish" Fishes 4, no. 1: 22. https://doi.org/10.3390/fishes4010022
APA StyleWoodward, M. A., Winder, L. A., & Watt, P. J. (2019). Enrichment Increases Aggression in Zebrafish. Fishes, 4(1), 22. https://doi.org/10.3390/fishes4010022




