Does Addition of Perch Larvae as Prey Affect the Growth, Development and Cannibalism Rate of Pikeperch Larvae?
Abstract
:1. Introduction
2. Results
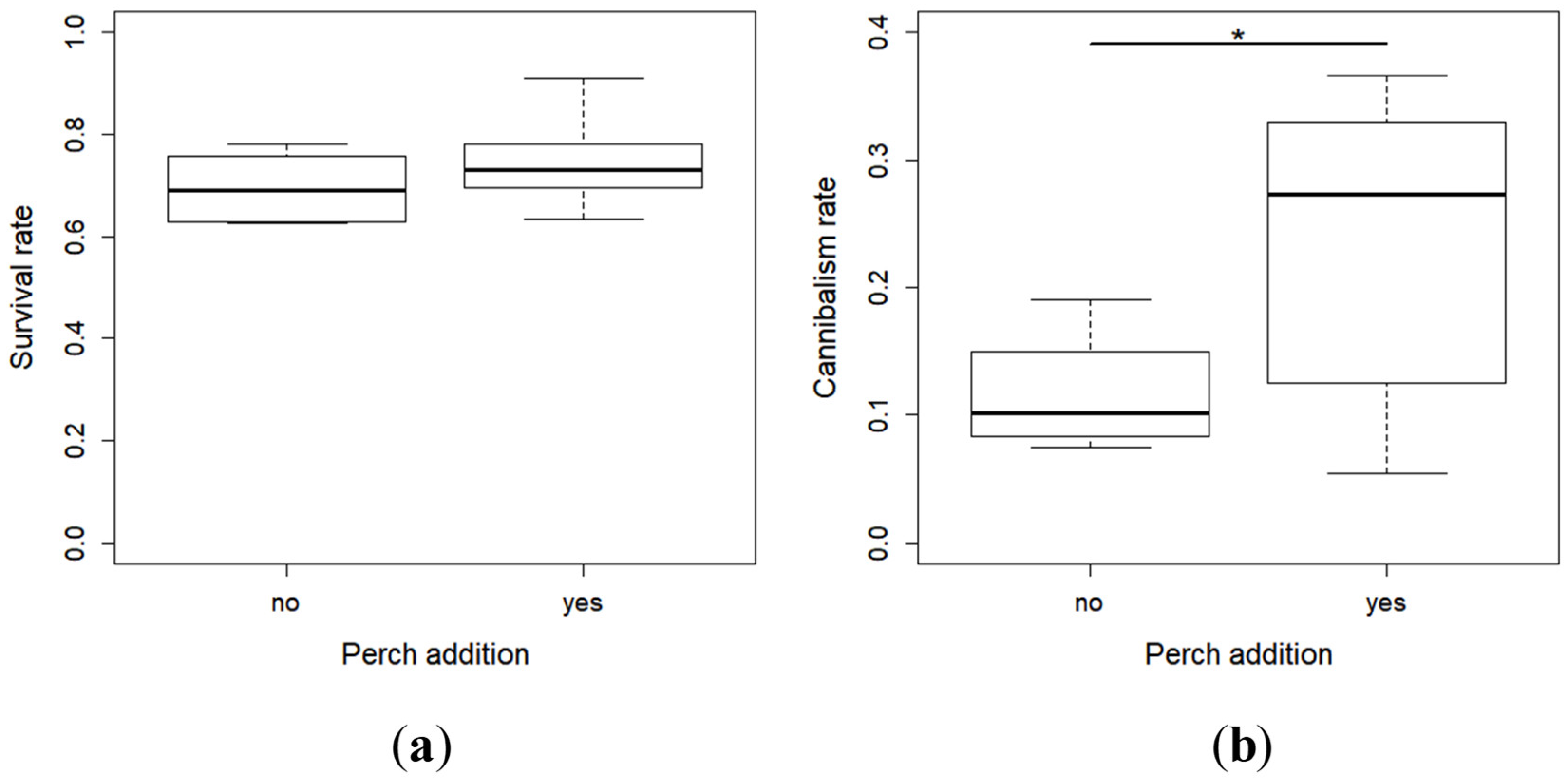
2.1. Survival and Cannibalism Rates
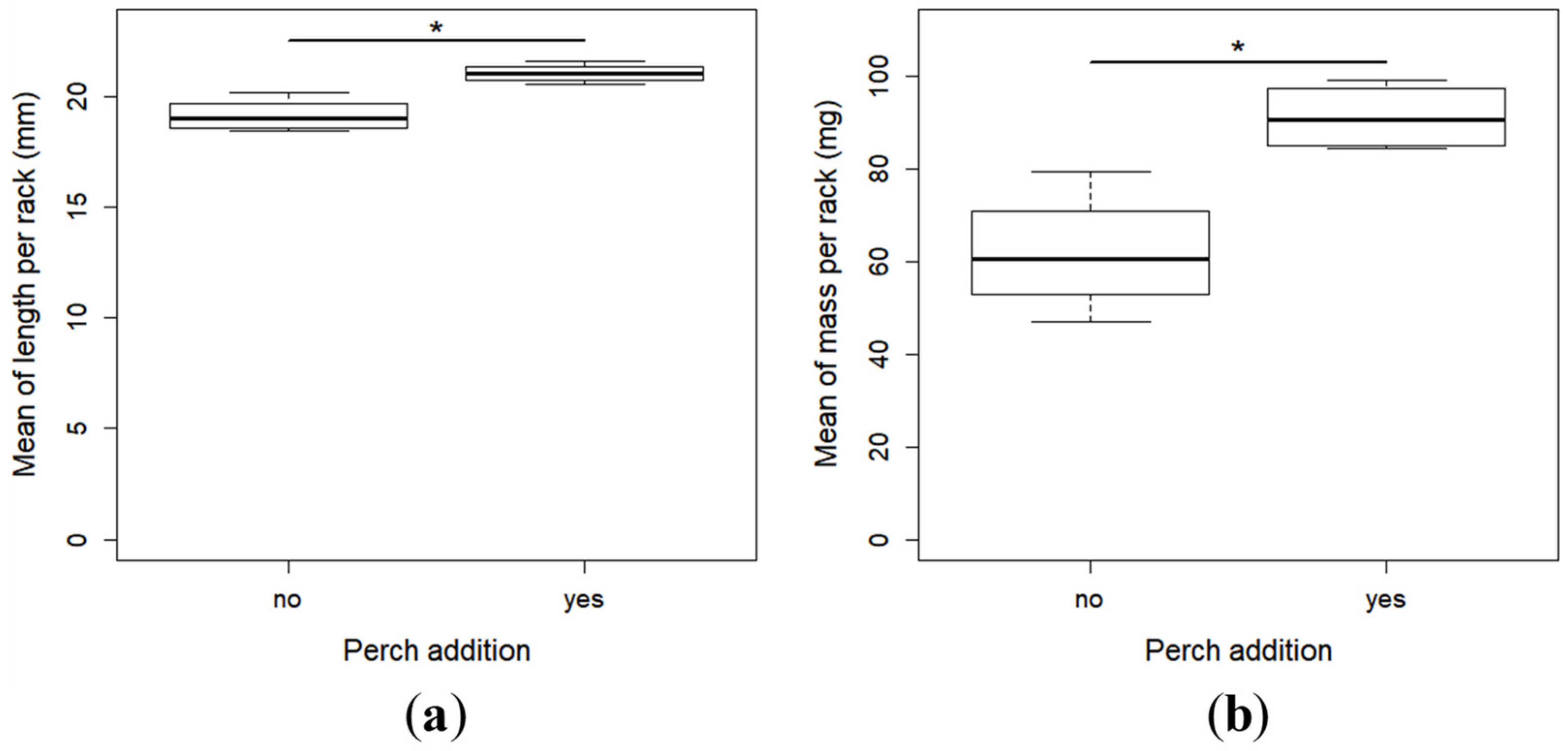
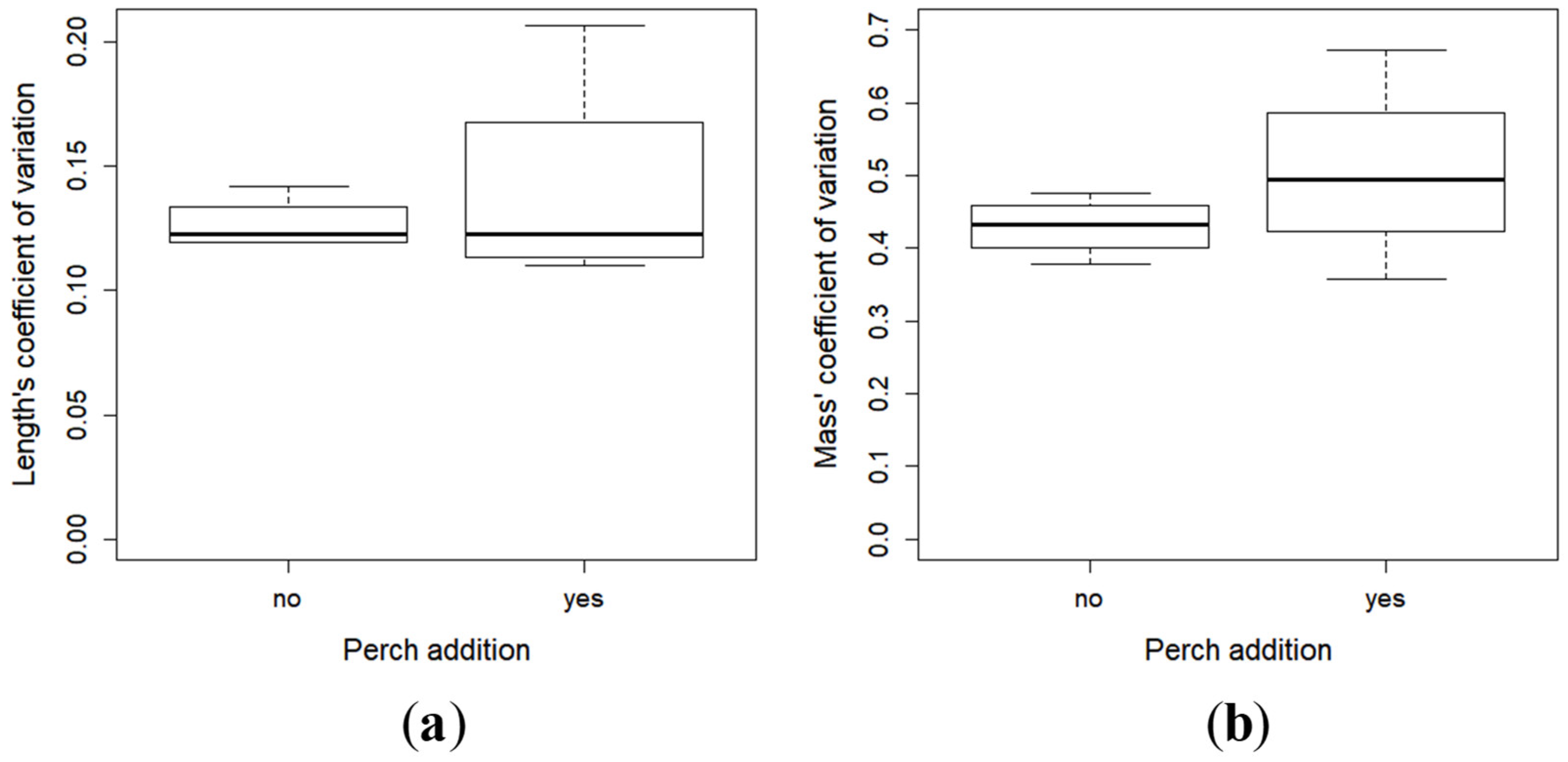
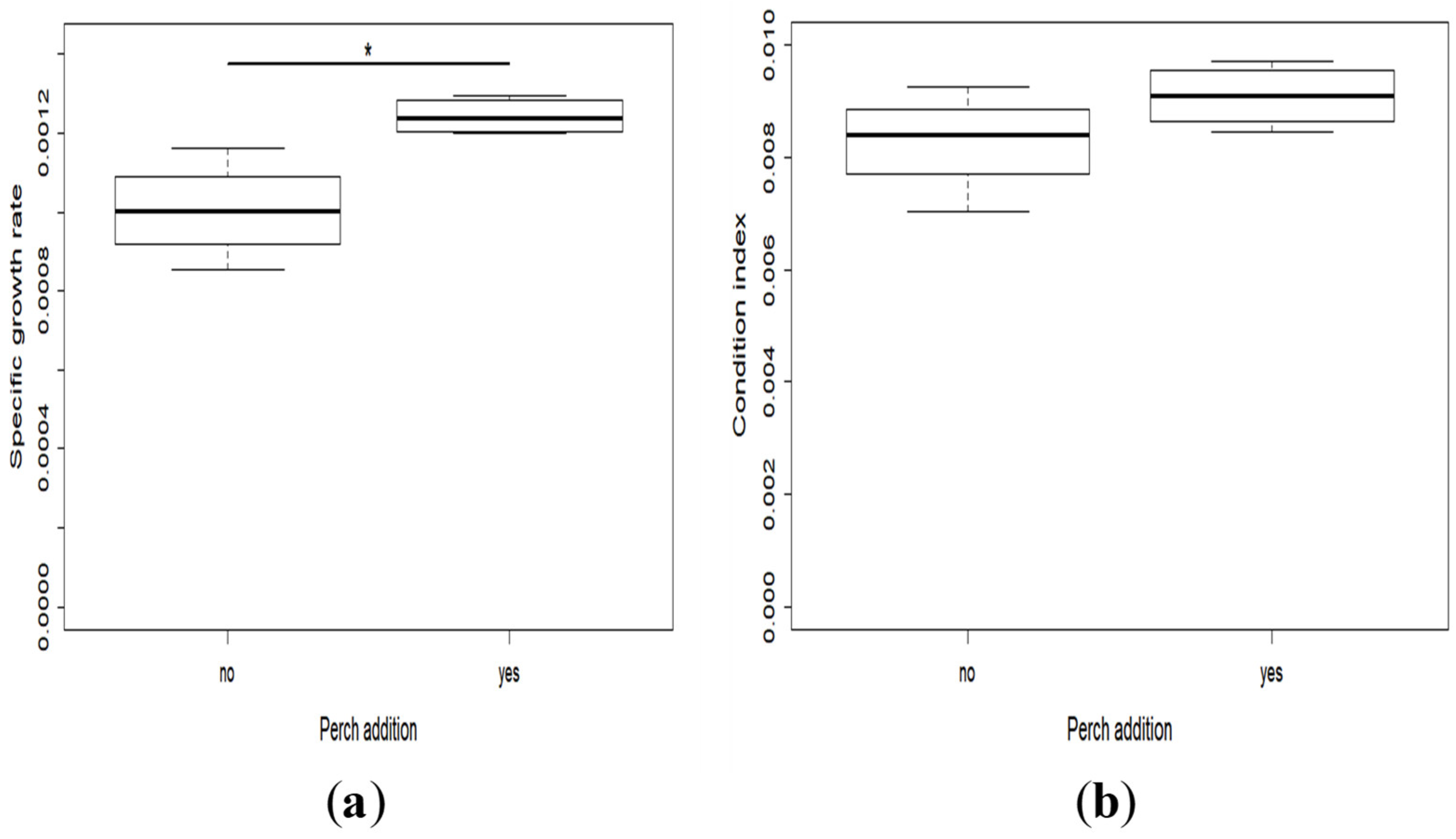
2.2. Growth and Development
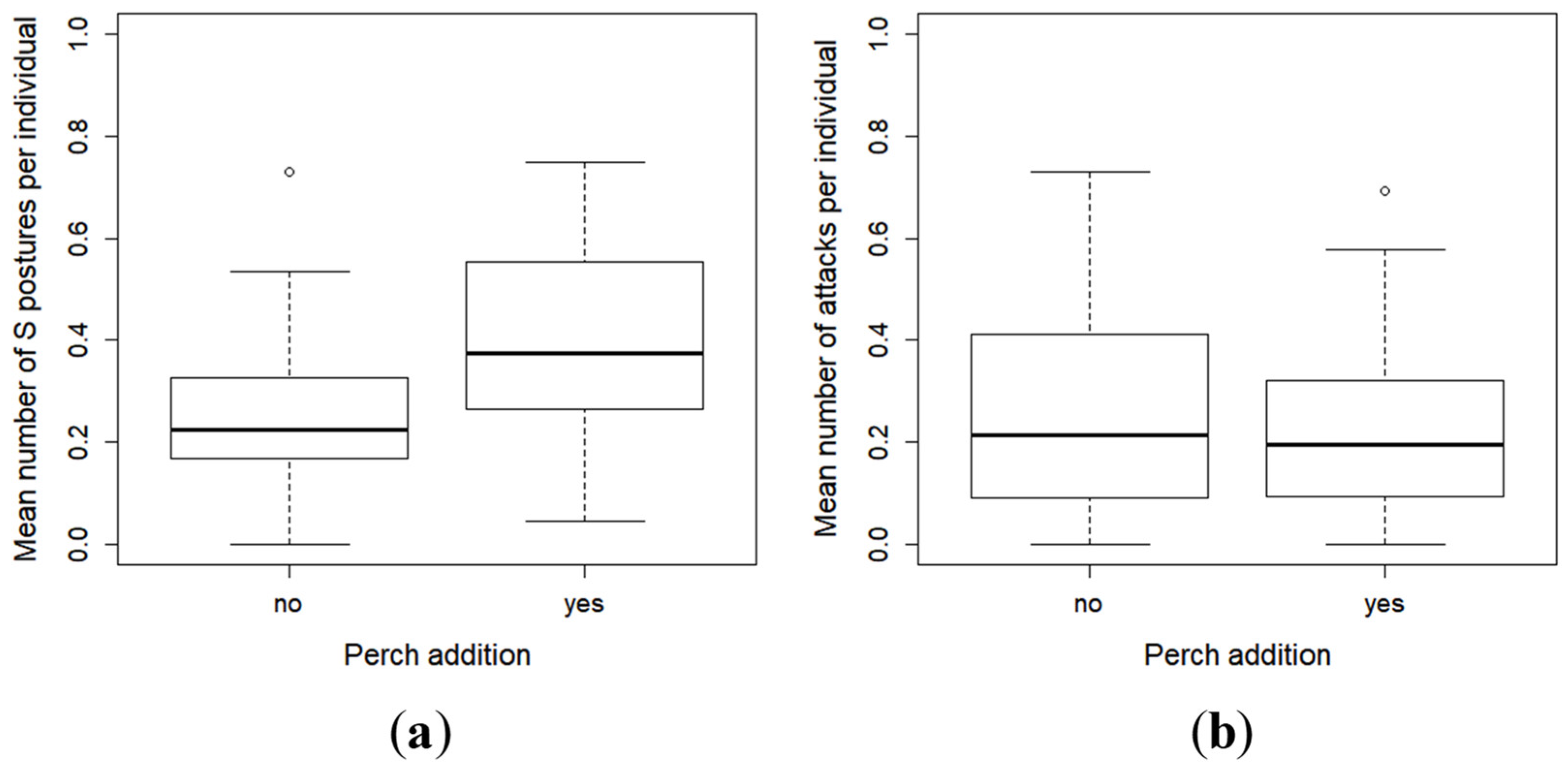
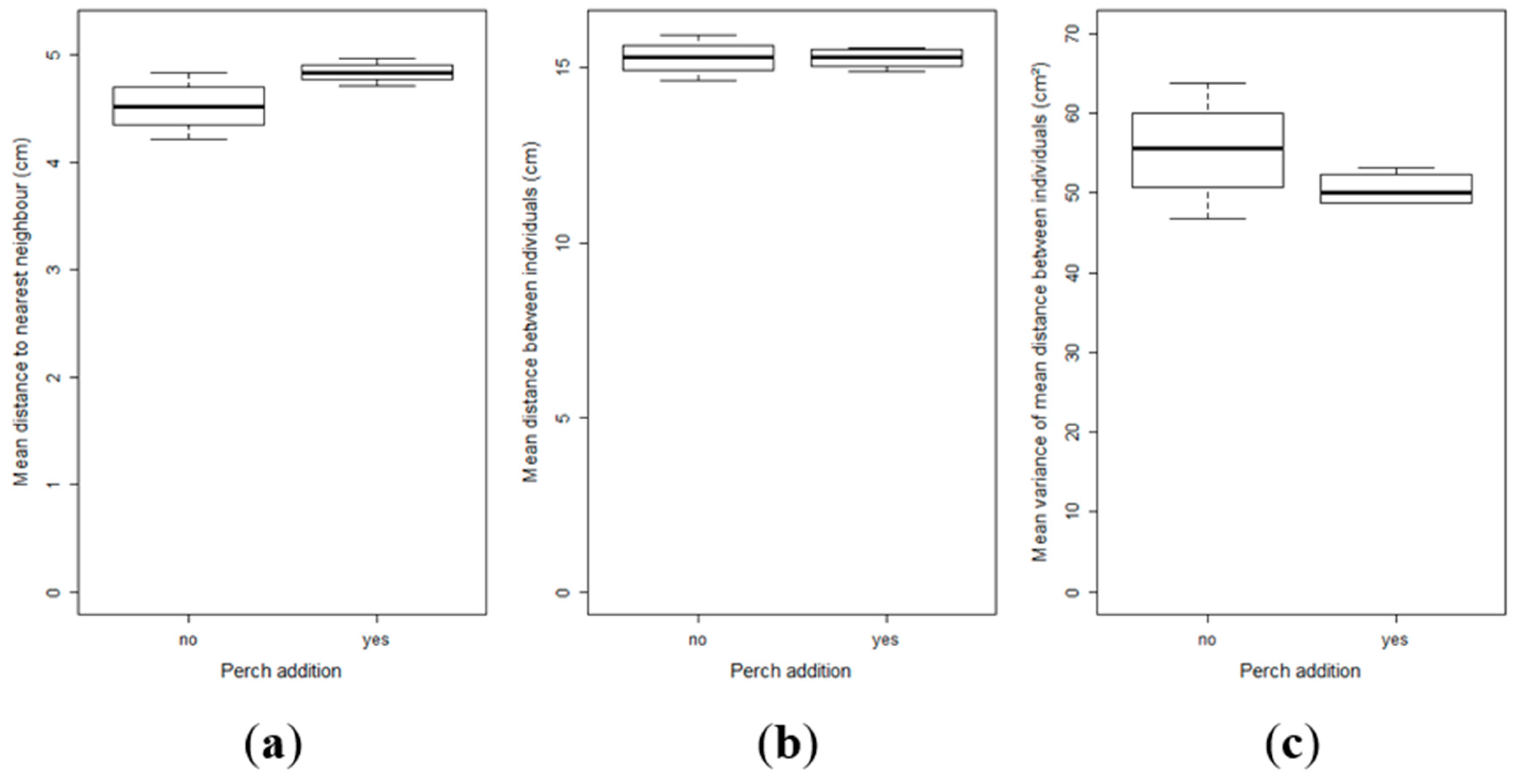
2.3. Effect of Perch as Prey on the Inter-Individual Relationships and Group Structure of Pikeperch Larvae
3. Discussion
4. Material and Methods
4.1. Rearing of the Fish
4.2. Experimental Design
- -
- Survival rate = (Number of living larvae at the end of the period)/(Total number of larvae introduced into the rack (initial larvae [50] + larvae replaced daily)) × 100,
- -
- Cannibalism rate = (Total number of larvae introduced into the rack—Cumulated number of dead larvae during the period—Number of living larvae at the end of the period)/(Total number of larvae introduced into the rack) × 100.
4.3. Influence of Perch as Prey on Zootechnical Parameters
- -
- The specific growth rate, which is the rate of daily mass gain: SGR = (lnWf-lnWi)/Td × 100, where Wf (mg) and Wi (mg) were respectively the mean final and initial body weight, and Td (days) the time of the experiment in days. As it was not possible to weigh individually all the larvae before the experiment, we used the mean mass of 45 larvae collected from the same tank for calculating the initial mean individual mass. It was thus the same for all the racks.
- -
- Fulton’s condition factor: FCF = W/L3, where W (mg) and L (mm) were the weight and total length of each individual at the end of the experiment, respectively. FCF was calculated for each fish of all the racks.
4.4. Influence of Perch as Prey on the Relationships between Pikeperch Larvae
- -
- The S posture: the fish adopts a posture with the shape of S in front of a conspecific and stays motionless.
- -
- The attack: it is characterized by a sudden movement forward in the direction of a conspecific. It can be subsequent to the S posture or not.
- -
- The capture: it occurs when an individual grabs a conspecific with its mouth and attempts to ingest it. The victim can escape or not. In general, it is grabbed by flanks or tail.
- -
- The ingestion: after the capture, the cannibal attempts to ingest its prey. For that, it needs to reorient the prey into a tail-first or head-first position to swallow it.
4.5. Influence of Perch as Prey on the Pikeperch Group Structure
- -
- The distance (cm) to the nearest neighbour for each larva of the group, which is an index of the group aggregation: the shorter the distance is, the greater the aggregation.
- -
- The mean distance (cm) for each larva to all the other members of the group, which is correlated with the group cohesion: the shorter the mean distance is, the greater the group cohesion.
- -
- The variance of the mean distance for each larva, which is a parameter of the group homogeneity: the lower the variance is, the higher the homogeneity of the group.
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Polis, G.A. The evolution and dynamics of intraspecific predation. Ann. Rev. Ecol. Syst. 1981, 12, 225–251. [Google Scholar] [CrossRef]
- Smith, C.; Reay, P. Cannibalism in teleost fish. Rev. Fish Biol. Fish. 1991, 1, 41–64. [Google Scholar] [CrossRef]
- Hecht, T.; Pienaar, A.G. A review of cannibalism and its implications in fish larviculture. J. World Aquac. Soc. 1993, 24, 246–261. [Google Scholar] [CrossRef]
- Baras, E. Cannibalism in fish larvae: What have we learned? In Larval Fish Aquaculture; Qin, J.G., Ed.; ISEM 2012-053; Nova Science Publishers: New York, NY, USA, 2012; pp. 1–37. [Google Scholar]
- Soulsby, D. Animal Cannibalism: The Dark Side of Evolution; 5mEntreprises: Sheffield, UK, 2013. [Google Scholar]
- Van den Bosch, F.; de Roos, A.M.; Gabriel, W. Cannibalism as a lifeboat mechanism. J. Math. Biol. 1988, 26, 619–633. [Google Scholar] [CrossRef]
- Fox, L.R. Cannibalism in natural populations. Ann. Rev. Ecol. Syst. 1975, 6, 87–106. [Google Scholar] [CrossRef]
- Elgar, M.; Crespi, A. Cannibalism: Ecology and Evolution among Diverse Taxa, 1st ed.; Oxford Science Publications: Oxford, UK, 1992. [Google Scholar]
- Pereira, L.S.; Agostinho, A.A.; Winemiller, K.O. Revisiting cannibalism in fishes. Rev. Fish Biol. Fish. 2017, 27, 499–513. [Google Scholar] [CrossRef]
- Babiak, I.; Mandiki, S.N.M.; Ratsinjomanana, M.; Kestemont, P. Initial weight and its variation in post-larval Eurasian perch affect quantitative characteristics of juvenile cohorts under controlled conditions. Aquaculture 2004, 234, 263–276. [Google Scholar] [CrossRef]
- Mandiki, S.N.M.; Babiak, I.; Krol, J.; Rasolo, J.F.R.; Kestemont, P. How initial predator-prey ratio affects intra-cohort cannibalism and growth in Eurasian perch Perca fluviatilis L larvae and juveniles under controlled conditions. Aquaculture 2007, 268, 149–155. [Google Scholar] [CrossRef]
- Naumowicz, K.; Pajdak, J.; Terech-Majewska, E.; Szarek, J. Intracohort cannibalism and methods for its mitigation in cultured freshwater fish. Rev. Fish Biol. Fish. 2017, 27, 193–208. [Google Scholar] [CrossRef]
- Pienaar, A.G. A Study of Coeval Sibling Cannibalism in Larval and Juvenile Fishes and Its Control under Culture Conditions. Ph.D. Thesis, Rhodes University, Grahamstown, South Africa, 1990. [Google Scholar]
- Baras, E.; Jobling, M. Dynamics of intracohort cannibalism in cultured fish. Aquac. Res. 2002, 33, 461–479. [Google Scholar] [CrossRef]
- Buijse, A.D. Dynamics and Exploitation of Unstable Percid Populations; Wageningen Agricultural University: Wageningen, The Netherlands, 1992. [Google Scholar]
- Galarowicz, T.L.; Wahl, D.H. Foraging by a young-of-the-year piscivore: The role of predator size, prey type, and density. Can. J. Fish. Aquat. Sci. 2005, 62, 2330–2342. [Google Scholar] [CrossRef]
- Har, P.J.B.; Ison, S. The influence of prey size and abundance, and individual phenotype on prey choice by the three-spined stickleback, Gasterosteus aculeatm L. J. Fish Biol. 1991, 38, 359–372. [Google Scholar] [CrossRef]
- Pedersen, T.; Falk-Petersen, I.B. Morphological changes during metamorphosis in cod (Gadus morhua L.), with particular reference to the development of the stomach and pyloric caeca. J. Fish Biol. 1992, 41, 449–461. [Google Scholar] [CrossRef]
- Benhaïm, D.; Bégout, M.L.; Lucas, G.; Chatain, B. First Insight into Exploration and Cognition in Wild Caught and Domesticated Sea Bass (Dicentrarchus labrax) in a Maze. PLoS ONE 2013, 8, e65872. [Google Scholar] [CrossRef]
- Heermann, L.; Borcherding, J. Competition, predation, cannibalism: The development of young-of-the-year perch populations in ponds with bream or roach. J. Appl. Ichthy 2013, 29, 549–554. [Google Scholar] [CrossRef]
- García-Berthou, E. Ontogenetic diet shifts and interrupted piscivory in introduced largemouth bass (Micropterus salmoides). Int. Rev. Hydrobiol. 2002, 87, 353–363. [Google Scholar] [CrossRef]
- García-Berthou, E.; Moreno-Amich, R. Food of introduced pumpkinseed sunfish: Ontogenetic diet shift and seasonal variation. J. Fish Biol. 2000, 57, 29–40. [Google Scholar] [CrossRef]
- Persson, A.; Brönmark, C. Foraging capacity and resource synchronization in an ontogenetic diet switcher, pikeperch (Stizostedion lucioperca). Ecology 2002, 83, 3014–3022. [Google Scholar] [CrossRef]
- Mittelbach, G.G.; Persson, L. The ontogeny of piscivory and its ecological consequences. Can. J. Fish. Aquat. Sci. 1998, 55, 1454–1465. [Google Scholar] [CrossRef]
- Diana, M.J.; Diffin, R.J.; Einfalt, L.M.; Walh, D.H. Effect of rearing experience on the survival, growth and behaviour of hatchery-reared large mouth bass. N. Am. J. Fish. Manag. 2018, 38, 794–802. [Google Scholar] [CrossRef]
- Shoup, D.E.; Broderius, C.R. Effects of vegetation density on the ontogeny to piscivory of juvenile largemouth bass. N. Am. J. Fish. Manag. 2018, 38, 630–638. [Google Scholar] [CrossRef]
- Hecht, T.; Appelbaum, S. Observations on intraspecific aggression and coeval sibling cannibalism by larval and juvenile Clarias gariepinus (Clariidae: Pisces) under controlled conditions. J. Zool. 1988, 214, 21–44. [Google Scholar] [CrossRef]
- Otterå, H.; Folkvord, A. Allometric growth in juvenile cod (Gadus morhua) and possible effects on cannibalism. J. Fish Biol. 1993, 43, 643–645. [Google Scholar] [CrossRef]
- Kaji, T.; Kodama, M.; Arai, H.; Tagawa, M.; Tanaka, M. Precocious development of the digestive system in relation to early appearance of piscivory in striped bonito Sarda orientalis larvae. Fish. Sci. 2002, 68, 1212–1218. [Google Scholar] [CrossRef]
- Thorpe, J. Morphology, physiology, behavior, and ecology of Perca fluviatilis L. and P. flavescens Mitchill. J. Fish. Board Can. 1977, 34, 1504–1514. [Google Scholar] [CrossRef]
- Sawada, Y.; Okada, T.; Miyashita, M.; Murata, O.; Kumai, H. Completion of the Pacific Bluefin tuna Thunnus orientalis (Temminck et Schlegel) life cycle. Aquac. Res. 2005, 36, 413–421. [Google Scholar] [CrossRef]
- Tanaka, Y.; Minami, H.; Ishihi, Y.; Kumon, K.; Eba, T.; Nishi, A.; Shiozawa, S. Prey utilization by hatchery reared Pacific Bluefin tuna larvae in mass culture tank estimated using stable isotope analysis, with special reference to their growth variation. Aquac. Sci. 2010, 58, 501–508. [Google Scholar] [CrossRef]
- Tanaka, Y.; Minami, H.; Ishihi, Y.; Kumon, K.; Higuchi, K.; Eba, T.; Nishi, A.; Nikaido, H.; Shiozawa, S. Relationship between prey utilization and growth variation in hatchery reared Pacific Bluefin tuna Thunnus orientalis (Temminck et Schlegel), larvae estimated using nitrogen stable isotope analysis. Aquac. Res. 2014, 45, 537–545. [Google Scholar] [CrossRef]
- Tanaka, Y.; Minami, H.; Ishihi, Y.; Kumon, K.; Higuchi, K.; Eba, T.; Nishi, A.; Nikaido, H.; Shiozawa, S. (Differential growth rates related to initiation to piscivory by hatchery reared larval Pacific Bluefin tuna Thunnus orientalis. Fish. Sci. 2014, 80, 1205–1214. [Google Scholar] [CrossRef]
- Tanaka, Y.; Kumon, K.; Higuchi, K.; Eba, T.; Nishi, A.; Nikaido, H.; Shiozawa, S. Influence of the prey items switched from rotifers to yolk sac larvae on growth of laboratory reared Pacific Bluefin tuna. Aquac. Sci. 2015, 63, 445–457. [Google Scholar] [CrossRef]
- Nyina-wamwiza, L.; Xu, X.L.; Blanchard, G.; Kestemont, P. Effect of dietary protein, lipid and carbohydrate ratio on growth, feed efficiency and body composition of pikeperch Sander lucioperca fingerlings. Aquac. Res. 2005, 36, 486–492. [Google Scholar] [CrossRef]
- Kestemont, P.; Dabrowski, K.; Summerfelt, R.C. Biology and Culture of Percid Fishes, 1st ed.; Springer: Dordrecht, The Netherlands, 2015. [Google Scholar]
- Szczepkowski, M.; Zakęś, Z.; Szczepkowska, B.; Piotrowska, I. Effect of size sorting on the survival, growth and cannibalism in pikeperch (Sander lucioperca L.) larvae during intensive culture in RAS. Czech J. Anim. Sci. 2011, 56, 483–489. [Google Scholar] [CrossRef]
- Mamcarz, A.; Kucharczyk, D.; Kujawa, R.; Skrzypczak, A. Influence of fish density on survival, growth and cannibalism development of pikeperch (Stizostedion lucioperca L.) larvae. In 2nd International Workshop Aquacult Percid Fish; European Aquaculture Society: Oostende, Belgium, 1997; pp. 343–346. [Google Scholar]
- Molnár, T.; Hancz, C.; Bódis, M.; Müller, T.; Bercsényi, M.; Horn, P. The effect of initial stocking density on growth and survival of pike-perch fingerlings reared under intensive conditions. Aquac. Int. 2004, 12, 181–189. [Google Scholar] [CrossRef]
- Lappalainen, J.; Olin, M.; Vinni, M. Pikeperch Cannibalism: Effects of Abundance, Size and Condition. Ann. Zoolo. Fen. 2006, 43, 35–44. [Google Scholar]
- Kestemont, P.; Xu, X.; Hamza, N.; Maboudou, J.; Toko, I.I. Effect of weaning age and diet on pikeperch larviculture. Aquaculture 2007, 264, 197–204. [Google Scholar] [CrossRef]
- Kangur, P.; Kangur, A.; Kangur, K. Dietary importance of various prey fishes for pikeperch Sander lucioperca (L.) in large shallow lake Võrtsjärv (Estonia). Proc. Estonian Acad. Sci. Biol. Ecol. 2007, 56, 154–167. [Google Scholar]
- Ginter, K.; Kangur, K.; Kangur, A.; Kangur, P.; Haldna, M. Diet patterns and ontogenetic diet shift of pikeperch, Sander lucioperca (L.) fry in lakes Peipsi and Vrtsjarv (Estonia). Hydrobiologia 2011, 660, 79–91. [Google Scholar] [CrossRef]
- Colchen, T.; Fontaine, P.; Ledore, Y.; Teletchea, F.; Pasquet, A. Intra cohort cannibalism in early life stages of pikeperch. Aquac. Res. 2019. [Google Scholar] [CrossRef]
- Keskinen, T.; Marjomäki, T.J. Diet and prey size spectrum of pikeperch in lakes in central Finland. J. Fish Biol. 2004, 65, 1147–1153. [Google Scholar] [CrossRef]
- Brownwell, C.L. Laboratory analysis of cannibalism by larvae of the cape anchory Engraulis capensis. Trans. Am. Fishs Soc. 1985, 114, 512–518. [Google Scholar] [CrossRef]
- Bell, A.M.; Stamps, J.A. Development of behavioural differences between individuals and populations of sticklebacks, Gasterosteus aculeatus. Anim. Behav. 2004, 68, 1339–1348. [Google Scholar] [CrossRef]
- Turesson, H.; Persson, A.; Bronmark, C. Prey size selection in piscivorous pikeperch (Stizostedion lucioperca) includes active prey choice. Ecol. Fresh Water Fish 2002, 11, 223–233. [Google Scholar] [CrossRef]
- Macpherson, E.; Gordoa, A. Effect of prey density on cannibalism in cape hake (Merluccius capensis) off Namibia. Mar. Biol. 1994, 119, 145–149. [Google Scholar] [CrossRef]
- Baras, E. Sibling cannibalism in dorada under experimental conditions. I. Ontogeny, dynamics, bioenergetics of cannibalism and prey size selectivity. J. Fish Biol. 2000, 57, 1001–1020. [Google Scholar] [CrossRef]
- Dill, L.M. Adaptative flexibility. Can. J. Fish. Aquat. Sci. 1983, 40, 398–408. [Google Scholar] [CrossRef]
- Iwata, T.; Sakamoto, K.Q.; Edwards, E.W.J.; Staniland, I.J.; Trahtan, P.N.; Goto, Y.; Sato, K.; Naito, Y.; Takahashi, A. The influence of preceding dive cycles on the foraging decisions for antartic fur seals. Biol. Lett. 2015, 11. [Google Scholar] [CrossRef]
- Colchen, T. Etude Multi-Traits du Cannibalisme Intra-Cohorte chez les Premiers Stades de vie du Sandre (Sander lucioperca). Ph.D. Thesis, University of Lorraine, Nancy, France, 2017. [Google Scholar]
- Bumann, D.; Rubenstein, D.; Krause, J. Mortality risk of spatial positions in animal groups: The danger of being in front. Behaviour 1997, 134, 13–14. [Google Scholar] [CrossRef]
- Schram, E.; Phillipsen, E. Production of Market Size Pikeperch (Sander lucioperca) in a Pilot RAS; RIVO (Netherlands Institute for Fisheries Research) Report: Ymuiden, Netherlands, 2003; p. 21. [Google Scholar]
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting linear mixed-effects models using lme4. arXiv, 2014; arXiv:1406.5823. [Google Scholar]
- R Core Team. A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2016; Available online: https://www.R-project.org/ (accessed on 11 March 2019).
- Houde, E.D. Fish larvae. In Encyclopedia of Ocean Sciences; Steele, J.H., Turekian, K.K., Thorpe, S.A., Eds.; Academic Press: London, UK, 2001; pp. 928–938. [Google Scholar]
- Colchen, T.; Faux, E.; Teletchea, F.; Pasquet, A. Is personality of young fish consistent through different behavioural tests? Appl. Anim. Behav. Sci. 2017, 194, 127–134. [Google Scholar] [CrossRef]
- Buske, C.; Gerlai, R. Early embryonic ethanol exposure impairs shoaling and the dopaminergic and serotoninergic system in adult zebrafish. Neurotoxicol. Teratol. 2011, 33, 698–707. [Google Scholar] [CrossRef]

| Periods | Dissolved Oxygen (mg·L−1) | pH | Ammonium Ion Content (mg·L−1) | Nitrous Nitrogen Content (mg·L−1) |
|---|---|---|---|---|
| (a) | 7.8 ± 0.3 | 8.0 ± 0.2 | 0.05 ± 0.04 | 0.01 ± 0.03 |
| (b) | 7.5 ± 0.2 | 6.9 ± 0.7 | 0.07 ± 0,06 | 0.02 ± 0.02 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cortay, A.; Colchen, T.; Fontaine, P.; Pasquet, A. Does Addition of Perch Larvae as Prey Affect the Growth, Development and Cannibalism Rate of Pikeperch Larvae? Fishes 2019, 4, 21. https://doi.org/10.3390/fishes4010021
Cortay A, Colchen T, Fontaine P, Pasquet A. Does Addition of Perch Larvae as Prey Affect the Growth, Development and Cannibalism Rate of Pikeperch Larvae? Fishes. 2019; 4(1):21. https://doi.org/10.3390/fishes4010021
Chicago/Turabian StyleCortay, Aurore, Tatiana Colchen, Pascal Fontaine, and Alain Pasquet. 2019. "Does Addition of Perch Larvae as Prey Affect the Growth, Development and Cannibalism Rate of Pikeperch Larvae?" Fishes 4, no. 1: 21. https://doi.org/10.3390/fishes4010021
APA StyleCortay, A., Colchen, T., Fontaine, P., & Pasquet, A. (2019). Does Addition of Perch Larvae as Prey Affect the Growth, Development and Cannibalism Rate of Pikeperch Larvae? Fishes, 4(1), 21. https://doi.org/10.3390/fishes4010021






