Mucosal Barrier Functions of Fish under Changing Environmental Conditions
Abstract
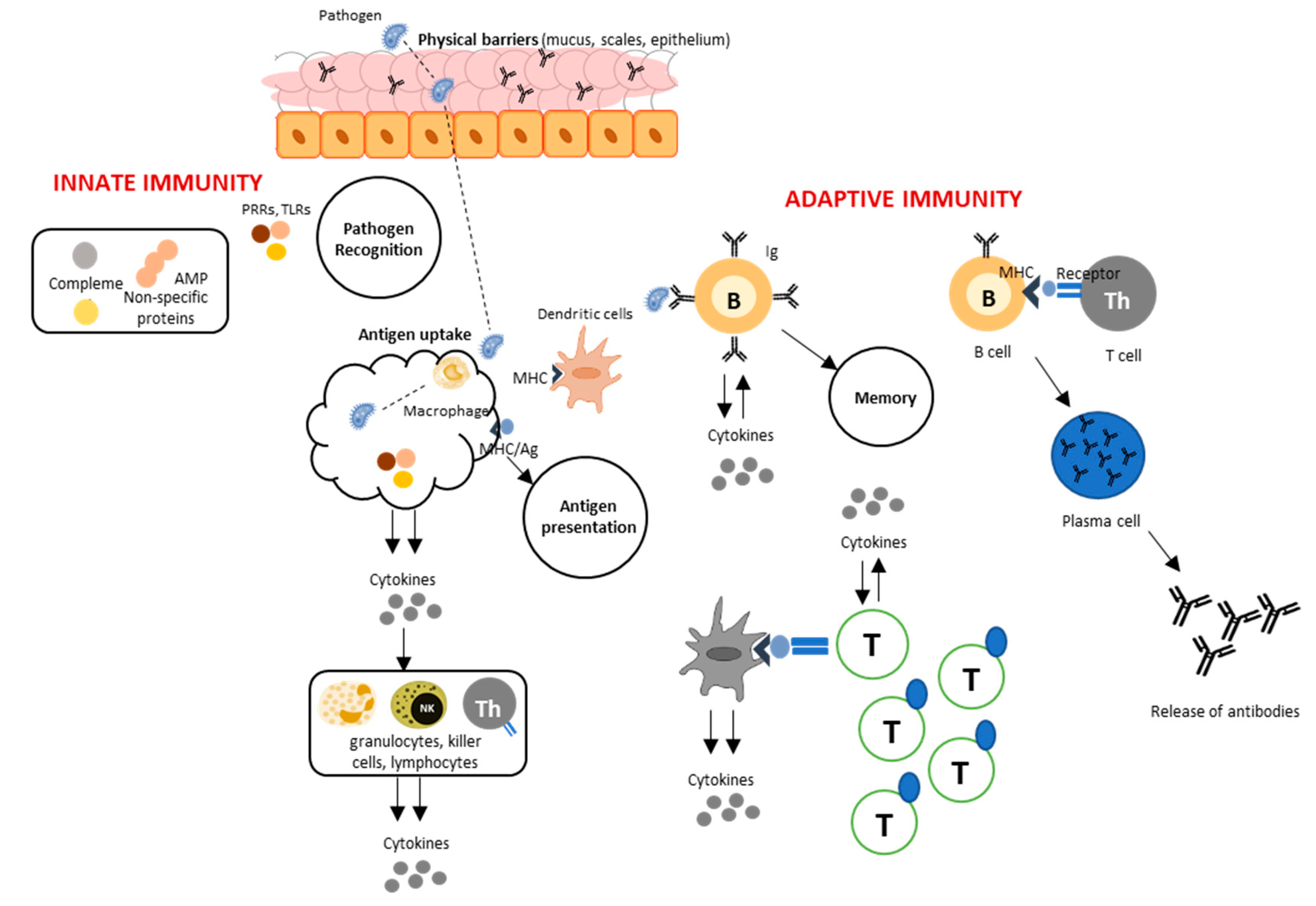
1. Introduction
2. Variability of Environmental Parameters in Aquaculture
3. Dissolved Oxygen
4. Water pH
5. Carbon Dioxide
6. Temperature
7. Salinity
8. Photoperiod
9. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lazado, C.C.; Caipang, C.M.A. Mucosal immunity and probiotics in fish. Fish Shellfish Immunol. 2014, 39, 78–89. [Google Scholar] [CrossRef] [PubMed]
- Salinas, I. The Mucosal Immune System of Teleost Fish. Biology 2015, 4, 525–539. [Google Scholar] [CrossRef] [PubMed]
- Smith, H.W. The absorption and excretion of water and salts by marine teleosts. Am. J. Physiol. Legacy Content 1930, 93, 480–505. [Google Scholar] [CrossRef]
- Evans, D.H.; Piermarini, P.M.; Choe, K.P. The multifunctional fish gill: Dominant site of gas exchange, osmoregulation, acid-base regulation, and excretion of nitrogenous waste. Physiol. Rev. 2005, 85, 97–177. [Google Scholar] [CrossRef] [PubMed]
- Grossel, M.; Farrell, A.; Brauner, C. Fish Physiology: The Multifunctional Gut of Fish, 1st ed.; Academic Press: Cambridge, MA, USA, 2010; Volume 30. [Google Scholar]
- Beck, B.H.; Peatman, E. Mucosal Health in Aquaculture, 1st ed.; Academic Press: Cambridge, MA, USA, 2015. [Google Scholar]
- Lazado, C.C.; Caipang, C.M.A.; Estante, E.G. Prospects of host-associated microorganisms in fish and penaeids as probiotics with immunomodulatory functions. Fish Shellfish Immunol. 2015, 45, 2–12. [Google Scholar] [CrossRef] [PubMed]
- Rombout, J.H.W.M.; Yang, G.; Kiron, V. Adaptive immune responses at mucosal surfaces of teleost fish. Fish Shellfish Immunol. 2014, 40, 634–643. [Google Scholar] [CrossRef]
- Sunyer, J.O. Fishing for mammalian paradigms in the teleost immune system. Nature Immunol. 2013, 14, 320. [Google Scholar] [CrossRef] [PubMed]
- Nigam, A.K.; Kumari, U.; Mittal, A.K. Comparative analysis of innate immune parameters of the skin mucous secretions from certain freshwater teleosts, inhabiting different ecological niches. Fish Physiol. Biochem. 2012, 38, 1245–1256. [Google Scholar] [CrossRef]
- Ellis, A.E. Innate host defense mechanisms of fish against viruses and bacteria. Dev. Comp. Immunol. 2001, 25, 827–839. [Google Scholar] [CrossRef]
- Boutin, S.; Bernatchez, L.; Audet, C.; Derôme, N. Network analysis highlights complex interactions between pathogen, host and commensal microbiota. PLoS ONE 2013, 8, e84772. [Google Scholar] [CrossRef]
- Dudley, S.A.; Schmitt, J. Testing the Adaptive Plasticity Hypothesis: Density-Dependent Selection on Manipulated Stem Length in Impatiens capensis. Am. Nat. 1996, 147, 445–465. [Google Scholar] [CrossRef]
- Meyers, L.A.; Bull, J.J. Fighting change with change: Adaptive variation in an uncertain world. Trends Ecol. Evol. 2002, 17, 551–557. [Google Scholar] [CrossRef]
- Travis, J. Evaluating the adaptive role of morphological plasticity. In Ecological Morphology: Integrative Organismal Biology; University of Chicago Press: Chicago, IL, USA, 1994; pp. 99–122. [Google Scholar]
- West-Eberhard, M.J. Developmental Plasticity and Evolution; Oxford University Press: Cary, NC, USA, 2003; ISBN 978-0-19-512235-0. [Google Scholar]
- Fivelstad, S. Long-term carbon dioxide experiments with salmonids. Aqua. Eng. 2013, 53, 40–48. [Google Scholar] [CrossRef]
- Sollid, J.; De Angelis, P.; Gundersen, K.; Nilsson, G.E. Hypoxia induces adaptive and reversible gross morphological changes in crucian carp gills. J. Exp. Biol. 2003, 206, 3667–3673. [Google Scholar] [CrossRef] [PubMed]
- Sollid, J.; Nilsson, G.E. Plasticity of respiratory structures—Adaptive remodeling of fish gills induced by ambient oxygen and temperature. Respir. Physiol. Neurobiol. 2006, 154, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Matey, V.; Richards, J.G.; Wang, Y.; Wood, C.M.; Rogers, J.; Davies, R.; Murray, B.W.; Chen, X.-Q.; Du, J.; Brauner, C.J. The effect of hypoxia on gill morphology and ionoregulatory status in the Lake Qinghai scaleless carp, Gymnocypris przewalskii. J. Exp. Biol. 2008, 211, 1063–1074. [Google Scholar] [CrossRef]
- Niklasson, L.; Sundh, H.; Fridell, F.; Taranger, G.L.; Sundell, K. Disturbance of the intestinal mucosal immune system of farmed Atlantic salmon (Salmo salar), in response to long-term hypoxic conditions. Fish Shellfish Immunol. 2011, 31, 1072–1080. [Google Scholar] [CrossRef]
- Tzaneva, V.; Bailey, S.; Perry, S.F. The interactive effects of hypoxemia, hyperoxia, and temperature on the gill morphology of goldfish (Carassius auratus). Am. J. Physiol. Regul. Integr. Comp. Physiol. 2011, 300, R1344–R1351. [Google Scholar] [CrossRef] [PubMed]
- Bagherzadeh Lakani, F.; Sattari, M.; Falahatkar, B. Effect of different oxygen levels on growth performance, stress response and oxygen consumption in two weight groups of great sturgeon Huso huso. Iran. J. Fish. Sci. 2013, 12, 533–549. [Google Scholar]
- Machado, M.; Malheiro, D.; Couto, A.; Wilson, J.M.; Guerreiro, M.; Azeredo, R.; Svendsen, J.C.; Afonso, A.; Serradeiro, R.; Costas, B. Acute hyperoxia induces systemic responses with no major changes in peripheral tissues in the Senegalese sole (Solea senegalensis Kaup, 1858). Fish Shellfish Immunol. 2018, 74, 260–267. [Google Scholar] [CrossRef]
- Boyd, C.E.; Tucker, C.S. Pond Aquaculture Water Quality Management; Springer: New York, NY, USA, 1998. [Google Scholar]
- Wilkie, M.P.; Wood, C.M. The adaptations of fish to extremely alkaline environments. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 1996, 113, 665–673. [Google Scholar] [CrossRef]
- Mohammadi, M.; Mahboobi-Soofiani, N.; Farhadian, O.; Malekpouri, P. Metabolic and NH4 excretion rate of fresh water species, Chondrostoma regium in response to environmental stressors, different scenarios for temperature and pH. Sci. Total Environ. 2019, 648, 90–101. [Google Scholar] [CrossRef] [PubMed]
- Chang, W.J.; Horng, J.L.; Yan, J.J.; Hsiao, C.D.; Hwang, P.P. The transcription factor, glial cell missing 2, is involved in differentiation and functional regulation of H+-ATPase-rich cells in zebrafish (Danio rerio). Am. J. Physiol. Regul. Integr. Comp. Physiol. 2009, 296, R1192–R1201. [Google Scholar] [CrossRef] [PubMed]
- Günzel, D.; Fromm, M. Claudins and Other Tight Junction Proteins. In Comprehensive Physiology; American Cancer Society: Atlanta, GA, USA, 2012; pp. 1819–1852. ISBN 978-0-470-65071-4. [Google Scholar]
- Freda, J.; Sanchez, D.A.; Bergman, H.L. Shortening of Branchial Tight Junction Acid-Exposed Rainbow Trout (Oncorhynchus mykiss). Can. J. Fish. Aquat. Sci. 1991, 48, 2028–2033. [Google Scholar] [CrossRef]
- Kwong, R.W.M.; Perry, S.F. Cortisol regulates epithelial permeability and sodium losses in zebrafish exposed to acidic water. J. Endocrinol. 2013, 217, 253–264. [Google Scholar] [CrossRef] [PubMed]
- Kumai, Y.; Bahubeshi, A.; Steele, S.; Perry, S.F. Strategies for maintaining Na+ balance in zebrafish (Danio rerio) during prolonged exposure to acidic water. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2011, 160, 52–62. [Google Scholar] [CrossRef] [PubMed]
- Tiedke, J.; Cubuk, C.; Burmester, T. Environmental acidification triggers oxidative stress and enhances globin expression in zebrafish gills. Biochem. Biophys. Res. Commun. 2013, 441, 624–629. [Google Scholar] [CrossRef]
- Sylvain, F.-É.; Cheaib, B.; Llewellyn, M.; Gabriel Correia, T.; Barros Fagundes, D.; Luis Val, A.; Derome, N. pH drop impacts differentially skin and gut microbiota of the Amazonian fish tambaqui (Colossoma macropomum). Sci. Rep. 2016, 6, 32032. [Google Scholar] [CrossRef]
- Sloman, K.A. Mitochondria-Rich Cell Subtypes in Fish Gill. J. Exp. Biol. 2003, 206, 7–8. [Google Scholar] [CrossRef]
- Cameron, J.N.; Iwama, G.K. Compensation of Progressive Hypercapnia in Channel Catfish and Blue Crabs. J. Exp. Biol. 1987, 133, 183–197. [Google Scholar]
- Hayashi, M.; Kikkawa, T.; Ishimatsu, A. Morphological changes in branchial mitochondria-rich cells of the teleost Paralichthys olivaceus as a potential indicator of CO2 impacts. Mar. Pollut. Bull. 2013, 73, 409–415. [Google Scholar] [CrossRef] [PubMed]
- Goss, G.G.; Laurent, P.; Perry, S.F. Evidence for a morphological component in acid-base regulation during environmental hypercapnia in the brown bullhead (Ictalurus nebulosus). Cell Tissue Res. 1992, 268, 539–552. [Google Scholar] [CrossRef]
- Goss, G.G.; Perry, S.F. Physiological and morphological regulation of acid–base status during hypercapnia in rainbow trout (Oncorhynchus mykiss). Can. J. Zool. 1993, 71, 1673–1680. [Google Scholar] [CrossRef]
- Baker, D.W.; Matey, V.; Huynh, K.T.; Wilson, J.M.; Morgan, J.D.; Brauner, C.J. Complete intracellular pH protection during extracellular pH depression is associated with hypercarbia tolerance in white sturgeon, Acipenser transmontanus. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2009, 296, R1868–R1880. [Google Scholar] [CrossRef] [PubMed]
- Kreiss, C.M.; Michael, K.; Lucassen, M.; Jutfelt, F.; Motyka, R.; Dupont, S.; Pörtner, H.O. Ocean warming and acidification modulate energy budget and gill ion regulatory mechanisms in Atlantic cod (Gadus morhua). J. Comp. Physiol. B 2015, 185, 767–781. [Google Scholar] [CrossRef] [PubMed]
- Kreiss, C.M.; Michael, K.; Bock, C.; Lucassen, M.; Pörtner, H.O. Impact of long-term moderate hypercapnia and elevated temperature on the energy budget of isolated gills of Atlantic cod (Gadus morhua). Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2015, 182, 102–112. [Google Scholar] [CrossRef] [PubMed]
- Sampaio, E.; Lopes, A.R.; Francisco, S.; Paula, J.R.; Pimentel, M.; Maulvault, A.L.; Repolho, T.; Grilo, T.F.; Pousão-Ferreira, P.; Marques, A.; et al. Ocean acidification dampens physiological stress response to warming and contamination in a commercially-important fish (Argyrosomus regius). Sci. Total Environ. 2018, 618, 388–398. [Google Scholar] [CrossRef] [PubMed]
- Mota, V.C.; Nilsen, T.O.; Gerwins, J.; Gallo, M.; Ytteborg, E.; Baeverfjord, G.; Kolarevic, J.; Summerfelt, S.T.; Terjesen, B.F. The effects of carbon dioxide on growth performance, welfare, and health of Atlantic salmon post-smolt (Salmo salar) in recirculating aquaculture systems. Aquaculture 2019, 498, 578–586. [Google Scholar] [CrossRef]
- Bly, J.E.; Clem, L.W. Temperature and teleost immune functions. Fish Shellfish Immunol. 1992, 2, 159–171. [Google Scholar] [CrossRef]
- Bowden, T.J.; Thompson, K.D.; Morgan, A.L.; Gratacap, R.M.L.; Nikoskelainen, S. Seasonal variation and the immune response: A fish perspective. Fish Shellfish Immunol. 2007, 22, 695–706. [Google Scholar] [CrossRef] [PubMed]
- Sollid, J.; Weber, R.E.; Nilsson, G.E. Temperature alters the respiratory surface area of crucian carp Carassius carassius and goldfish Carassius auratus. J. Exp. Biol. 2005, 208, 1109–1116. [Google Scholar] [CrossRef] [PubMed]
- Wentworth, S.A.; Thede, K.; Aravindabose, V.; Monroe, I.; Thompson, A.W.; Molyneaux, N.; Owen, C.L.; Burns, J.R.; Gonzalez-Vicente, A.; Garvin, J.L.; et al. Transcriptomic analysis of changes in gene expression of immune proteins of gill tissue in response to low environmental temperature in fathead minnows (Pimephales promelas). Comp. Biochem. Physiol. Part D Genom. Proteom. 2018, 25, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.-H.; Ma, A.-J.; Wang, X.-A. The immune response of turbot, Scophthalmus maximus (L.), skin to high water temperature. J. Fish Dis. 2011, 34, 619–627. [Google Scholar] [CrossRef] [PubMed]
- Clem, L.W.; Faulmann, E.; Miller, N.W.; Ellsaesser, C.; Lobb, C.J.; Cuchens, M.A. Temperature-mediated processes in teleost immunity: Differential effects of in vitro and in vivo temperatures on mitogenic responses of channel catfish lymphocytes. Dev. Comp. Immunol. 1984, 8, 313–322. [Google Scholar] [CrossRef]
- Parihar, M.S.; Javeri, T.; Hemnani, T.; Dubey, A.K.; Prakash, P. Responses of superoxide dismutase, glutathione peroxidase and reduced glutathione antioxidant defenses in gills of the freshwater catfish (Heteropneustes fossilis) to short-term elevated temperature. J. Therm. Biol. 1997, 22, 151–156. [Google Scholar] [CrossRef]
- Klein, R.D.; Borges, V.D.; Rosa, C.E.; Colares, E.P.; Robaldo, R.B.; Martinez, P.E.; Bianchini, A. Effects of increasing temperature on antioxidant defense system and oxidative stress parameters in the Antarctic fish Notothenia coriiceps and Notothenia rossii. J. Therm. Biol. 2017, 68, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Wen, B.; Jin, S.R.; Chen, Z.Z.; Gao, J.-Z. Physiological responses to cold stress in the gills of discus fish (Symphysodon aequifasciatus) revealed by conventional biochemical assays and GC-TOF-MS metabolomics. Sci. Total Environ. 2018, 640–641, 1372–1381. [Google Scholar] [CrossRef]
- Bleie, H.; Skrudland, A. Tap av laksefisk i sjø. Rapport Fra Mattilsynet; Mattilsynet: Oslo, Norway, 2014.
- Karlsen, C.; Ytteborg, E.; Timmerhaus, G.; Høst, V.; Handeland, S.; Jørgensen, S.M.; Krasnov, A. Atlantic salmon skin barrier functions gradually enhance after seawater transfer. Sci. Rep. 2018, 8, 9510. [Google Scholar] [CrossRef]
- Fast, M.D.; Sims, D.E.; Burka, J.F.; Mustafa, A.; Ross, N.W. Skin morphology and humoral non-specific defence parameters of mucus and plasma in rainbow trout, coho and Atlantic salmon. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2002, 132, 645–657. [Google Scholar] [CrossRef]
- Tipsmark, C.K.; Luckenbach, J.A.; Madsen, S.S.; Kiilerich, P.; Borski, R.J. Osmoregulation and expression of ion transport proteins and putative claudins in the gill of southern flounder (Paralichthys lethostigma). Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2008, 150, 265–273. [Google Scholar] [CrossRef]
- Bui, P.; Kelly, S.P. Claudin-6, -10d and -10e contribute to seawater acclimation in the euryhaline puffer fish Tetraodon nigroviridis. J. Exp. Biol. 2014, 217, 1758–1767. [Google Scholar] [CrossRef] [PubMed]
- Tipsmark, C.K.; Kiilerich, P.; Nilsen, T.O.; Ebbesson, L.O.E.; Stefansson, S.O.; Madsen, S.S. Branchial expression patterns of claudin isoforms in Atlantic salmon during seawater acclimation and smoltification. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2008, 294, R1563–R1574. [Google Scholar] [CrossRef]
- Gu, J.; Dai, S.; Liu, H.; Cao, Q.; Yin, S.; Lai, K.P.; Tse, W.K.F.; Wong, C.K.C.; Shi, H. Identification of immune-related genes in gill cells of Japanese eels (Anguilla japonica) in adaptation to water salinity changes. Fish Shellfish Immunol. 2018, 73, 288–296. [Google Scholar] [CrossRef] [PubMed]
- Lokesh, J.; Kiron, V. Transition from freshwater to seawater reshapes the skin-associated microbiota of Atlantic salmon. Sci. Rep. 2016, 6, 19707. [Google Scholar] [CrossRef]
- Valenzuela, A.; Campos, V.; Yañez, F.; Alveal, K.; Gutiérrez, P.; Rivas, M.; Contreras, N.; Klempau, A.; Fernandez, I.; Oyarzun, C. Application of artificial photoperiod in fish: A factor that increases susceptibility to infectious diseases? Fish Physiol. Biochem. 2012, 38, 943–950. [Google Scholar] [CrossRef] [PubMed]
- Curtis, A.M.; Bellet, M.M.; Sassone-Corsi, P.; O’Neill, L.A.J. Circadian clock proteins and immunity. Immunity 2014, 40, 178–186. [Google Scholar] [CrossRef] [PubMed]
- Lazado, C.C.; Lund, I.; Pedersen, P.B.; Nguyen, H.Q. Humoral and mucosal defense molecules rhythmically oscillate during a light–dark cycle in permit, Trachinotus falcatus. Fish Shellfish Immunol. 2015, 47, 902–912. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cabillon, N.A.R.; Lazado, C.C. Mucosal Barrier Functions of Fish under Changing Environmental Conditions. Fishes 2019, 4, 2. https://doi.org/10.3390/fishes4010002
Cabillon NAR, Lazado CC. Mucosal Barrier Functions of Fish under Changing Environmental Conditions. Fishes. 2019; 4(1):2. https://doi.org/10.3390/fishes4010002
Chicago/Turabian StyleCabillon, Nikko Alvin R., and Carlo C. Lazado. 2019. "Mucosal Barrier Functions of Fish under Changing Environmental Conditions" Fishes 4, no. 1: 2. https://doi.org/10.3390/fishes4010002
APA StyleCabillon, N. A. R., & Lazado, C. C. (2019). Mucosal Barrier Functions of Fish under Changing Environmental Conditions. Fishes, 4(1), 2. https://doi.org/10.3390/fishes4010002




