Wreckfish (Polyprion americanus). New Knowledge About Reproduction, Larval Husbandry, and Nutrition. Promise as a New Species for Aquaculture
Abstract
:1. Introduction
2. Reproduction
2.1. Acquisition of Wild Fish and Establishment of Captive Broodstocks
2.2. Description of the Reproductive Cycle in Captivity
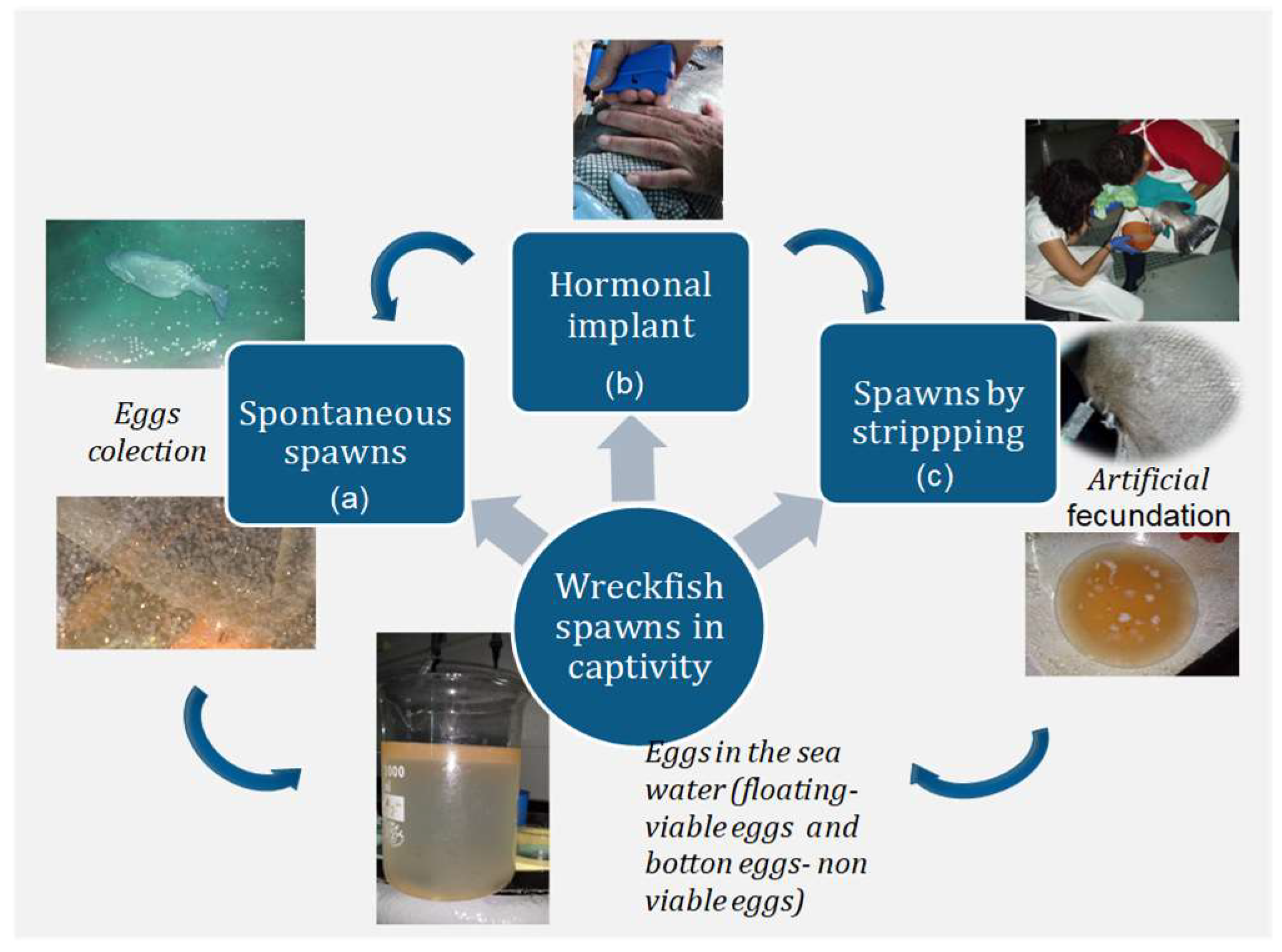
2.3. Spontaneous and Induced Spawning
- Natural and spontaneous spawns in large tanks (>40 m3), collecting the eggs as they exit the outflow of the tank (a).
- Spawning induction with exogenous gonadotropin-releasing hormone agonist (GnRHa) (b) carried out in large tanks (>40 m3) under controlled photothermal conditions, allowing the fish to spawn spontaneously.
- Spawning induction followed by in vitro fertilization by stripping of the mature females and males, maintained in smaller tanks (<40 m3) (c).
2.4. Sperm Characteristics and Cryopreservation
3. Nutrition
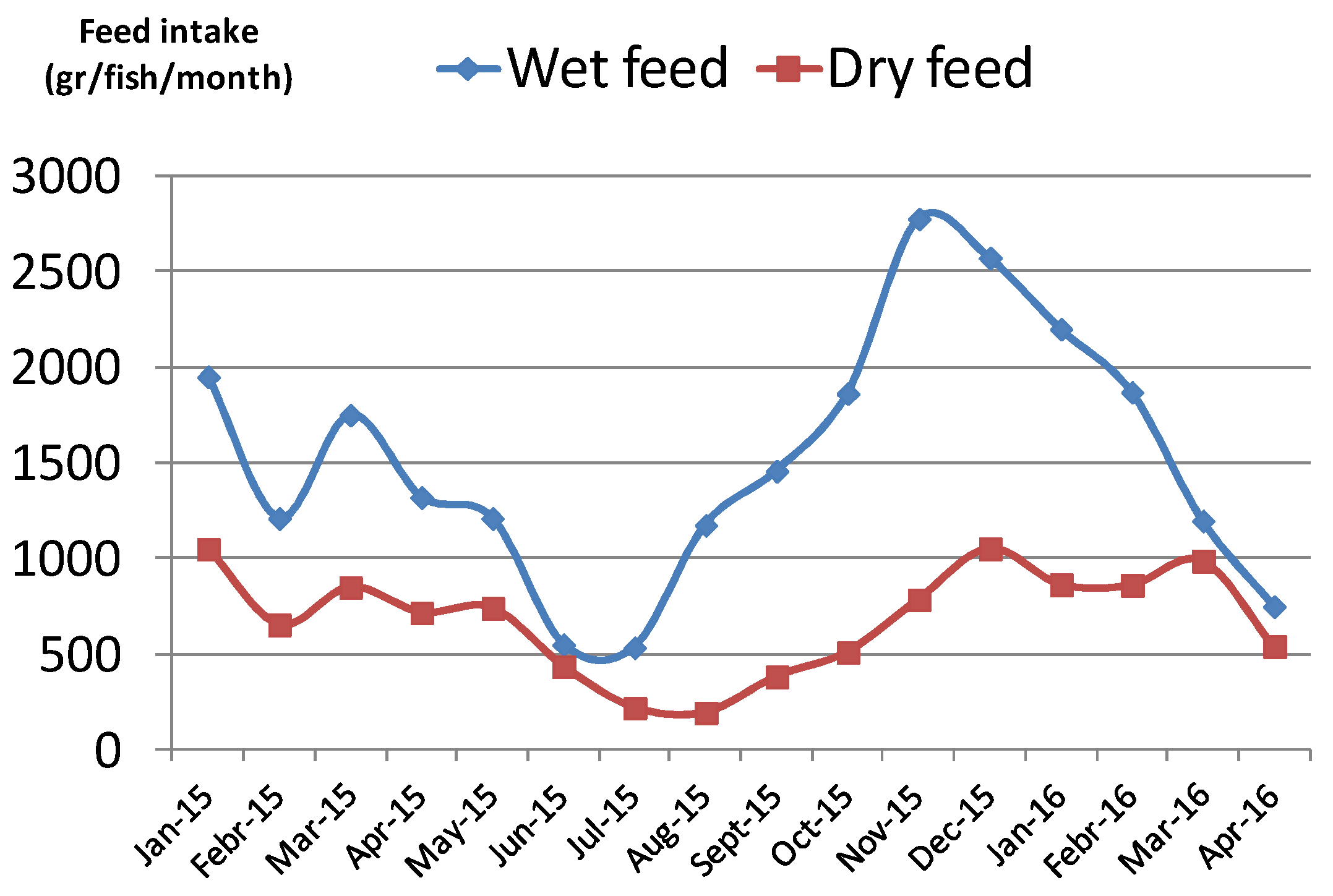
3.1. Effectiveness of Live Prey and the Influence of Enrichments
3.2. Influence of Broodstock Feeds on Fecundity and Spawning Quality
4. Larval Husbandry
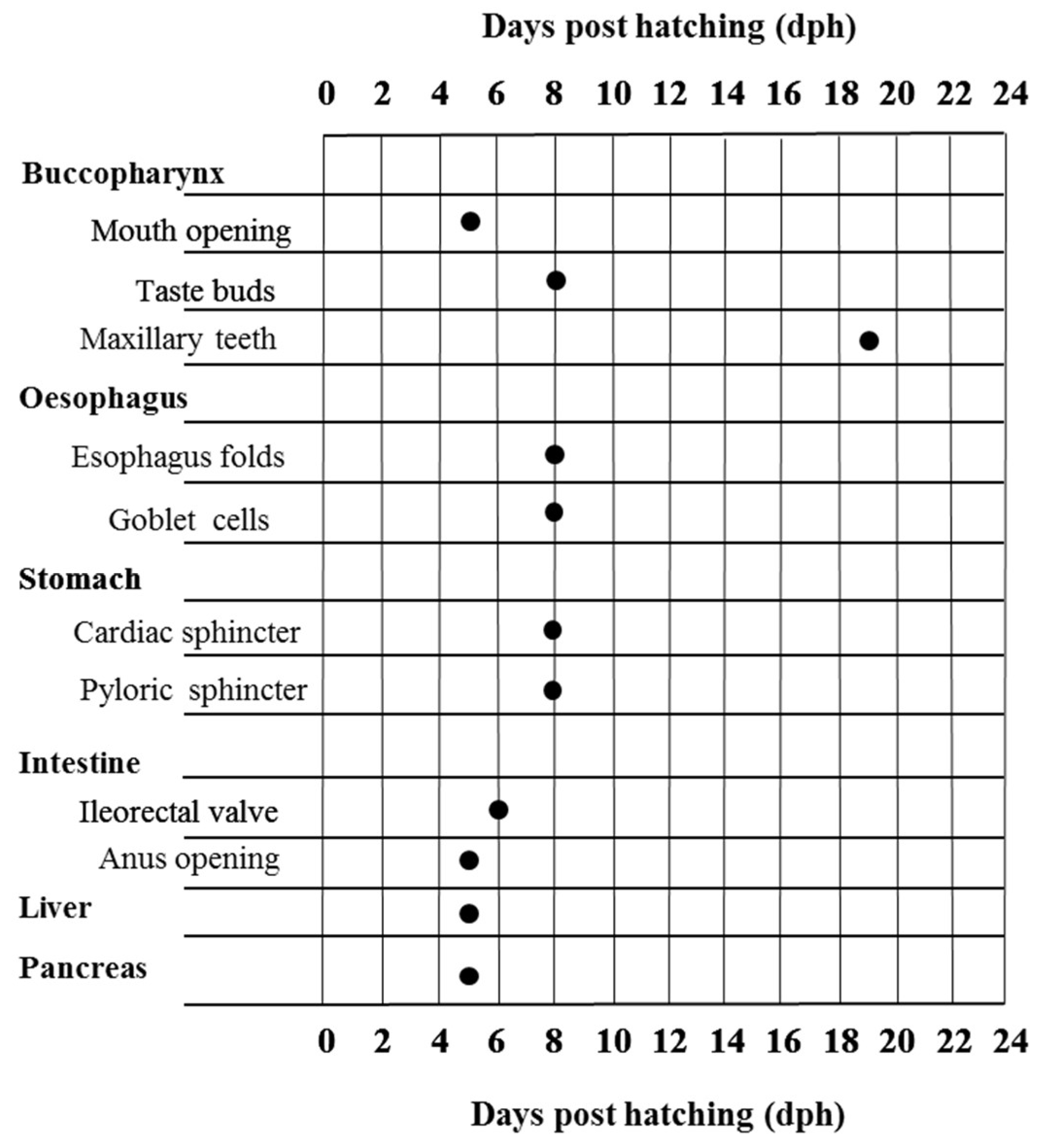
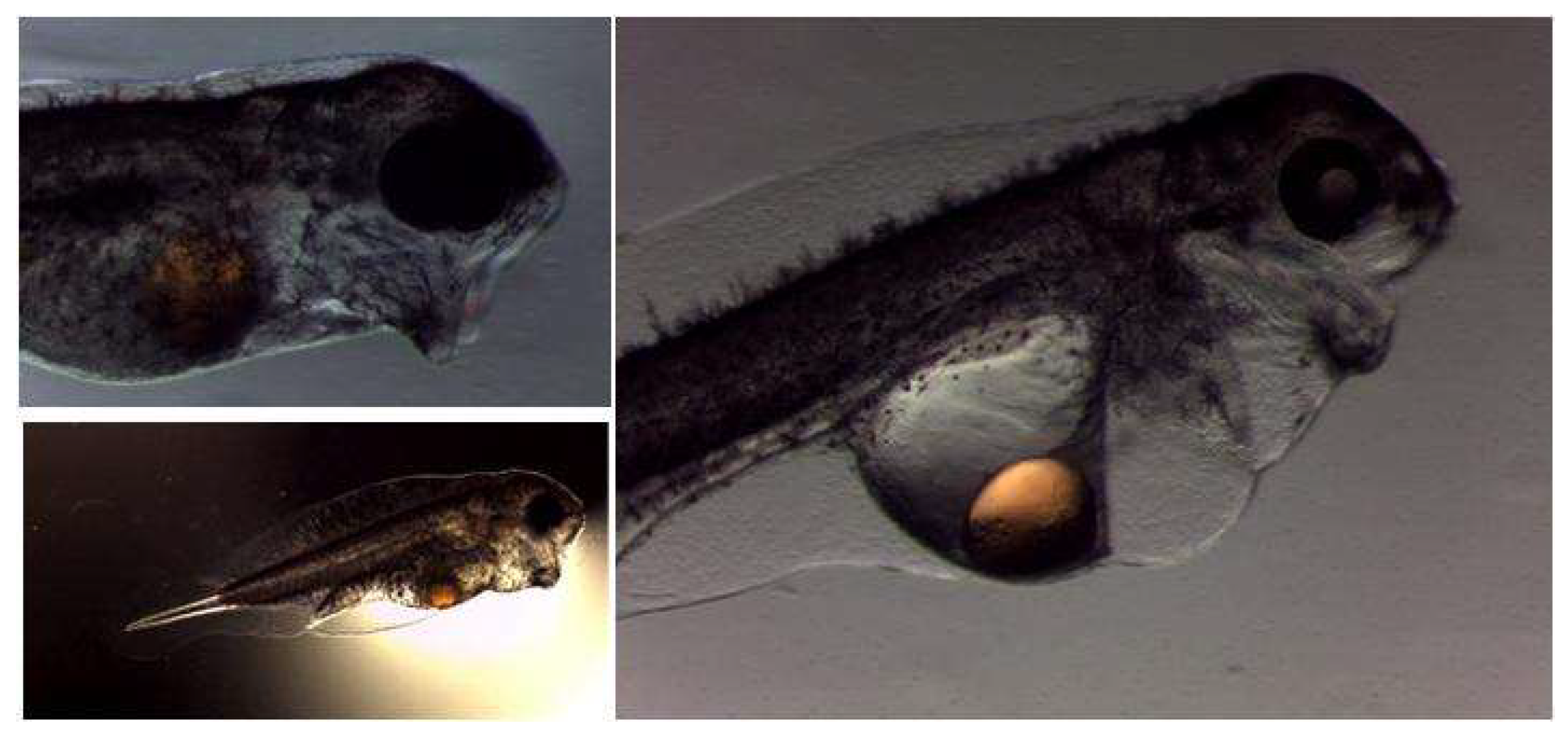
4.1. Development of the Digestive System in Wreckfish
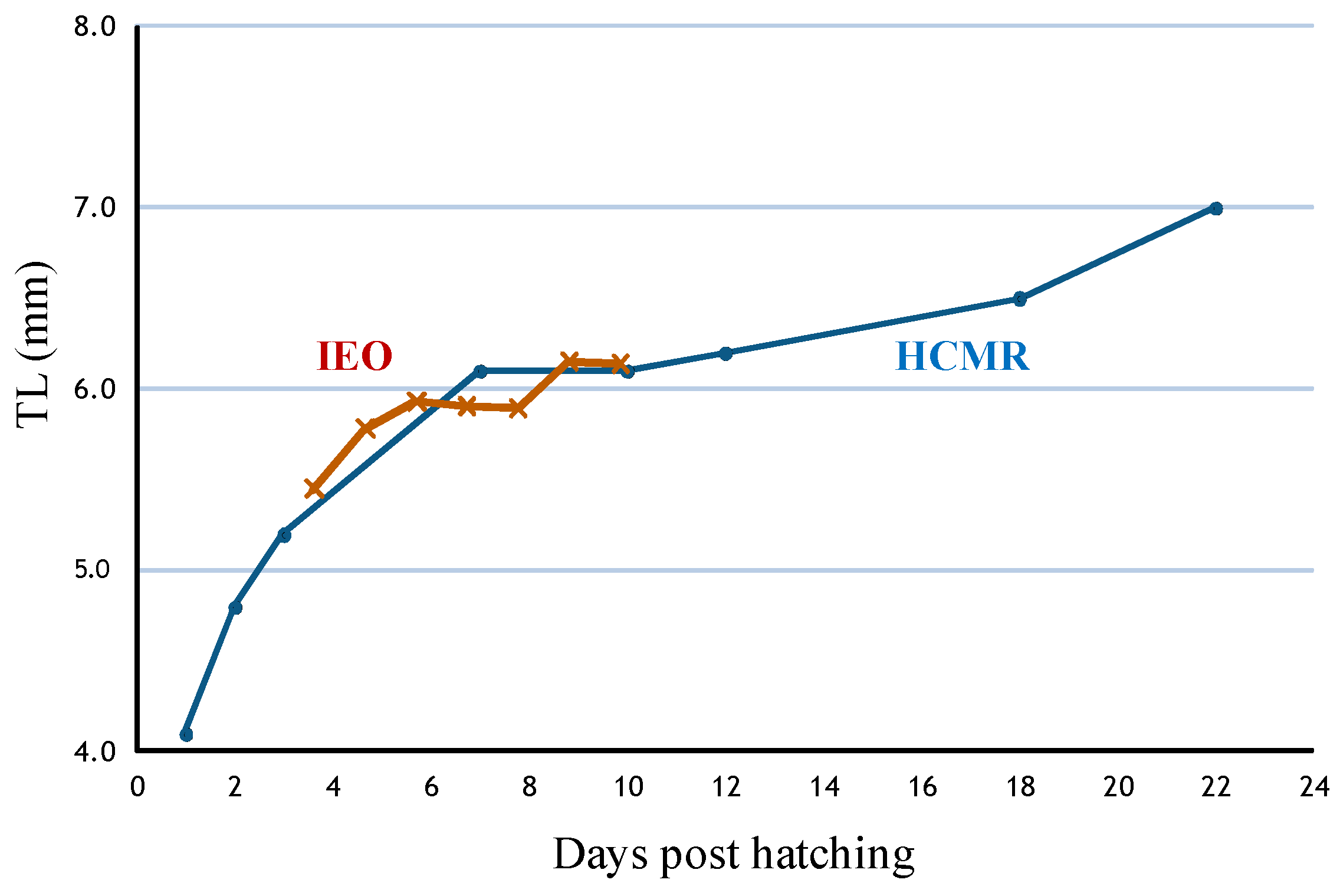
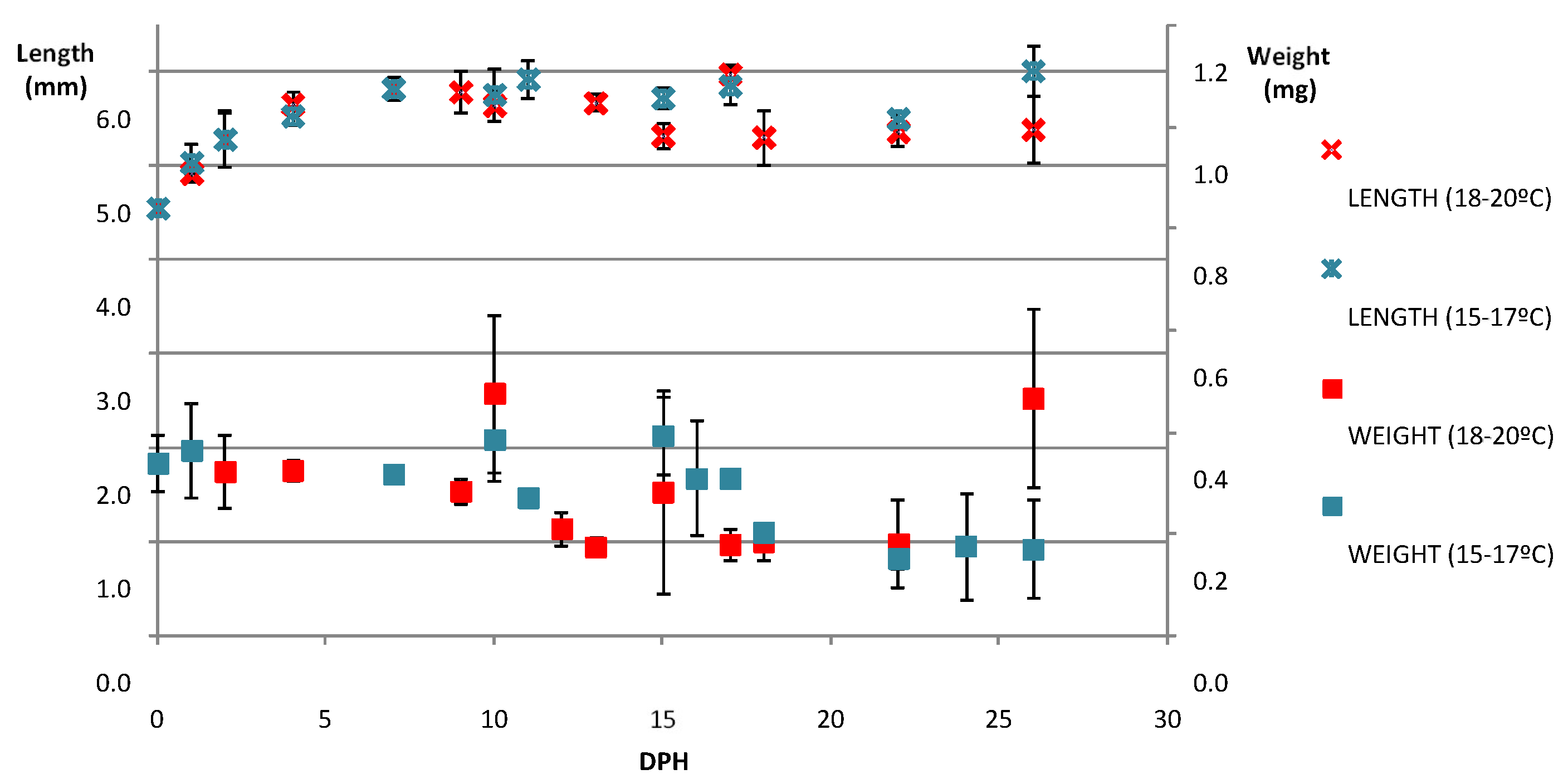
4.2. Optimum Conditions for Larval Rearing
5. Conclusions
Reproduction in captivity:
Sperm characteristics:
Nutrition:
Development of digestive and vision systems:
Larval husbandry:
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Diversify Project. Available online: https://www.diversifyfish.eu/ (accessed on 22 February 2019).
- Sedberry, G.R.; Andrade, C.A.P.; Carlin, J.L.; Chapman, R.W.; Luckhurst, B.E.; Manooch, C.S.; Menezes, G.; Thomsen, B.; Ulrich, G.F. Wreckfish Polyprion americanus in the North Atlantic: Fisheries, biology and management of a widely distributed and long-lived fish. Am. Fish Soc. Symp. 1999, 23, 27–50. [Google Scholar]
- Ball, A.O.; Sedberry, G.R.; Zatcoff, M.S.; Chapman, R.W.; Carlin, J.L. Population structure of wreckfish Polyprion americanus determined with microsatelite genetic markers. Mar. Biol. 2000, 137, 1077–1090. [Google Scholar] [CrossRef]
- Deudero, S.; Morales-Nin, B. Occurrence of Polyprion americanus under floating objects in western Mediterranean oceanic waters, inference from stomach contents analysis. J. Mar. Biol. Assoc. UK 2000, 80, 751–752. [Google Scholar] [CrossRef]
- Suquet, M.; La Pomèlie, C. Le cernier (Polyprion americanus): Biologie, pêche, marché et potentiel aquacole; 279 h. (Ressources de la mer); IFREMER: Plouzané, France, 2002; ISBN 2-84433-075-4. [Google Scholar]
- Rodriguez-Villanueva, J.L.; Peleteiro, J.B.; Pérez-Rial, E.; Soares, E.C.; Álvarez-Bláquez, B.; Mariño, C.; Linares, F.; Mañanós, E. Growth of wreckfish (Polyprion americanus) in Galicia, Spain. In Proceedings of the European Aquaculture 2011, Rhodes, Greece, 18–21 October 2011. [Google Scholar]
- Fishwatch. National Oceanic and Atmospheric Administration (NOAA). Available online: https://www.fishwatch.gov/profiles/wreckfish (accessed on 22 February 2019).
- Pescadegalicia.gal. Xunta de Galicia. Available online: https://www.pescadegalicia.gal/gl/estatisticas (accessed on 22 February 2019).
- Papandroulakis, N.; Mylonas, C.C.; Syggelaki, E.; Katharios, P.; Divakaran, S. First reproduction of captive-reared wreckfish (Polyprion americanus) using GnRH implants. Eur. Aquacult. 2008, 8, 15–18. [Google Scholar]
- Rodriguez, J.L.; Álvarez-Blázquez, B.; Pérez Rial, E.; Martínez, J.M.; Pazos, G.; Linares, F. Evaluation of wreckfish (Polyprion americanus) growth in Galicia (Spain). In Proceedings of the European Aquaculture 2017, Dubrovnik, Croatia, 17–20 October 2017. [Google Scholar]
- Fischer, W.; Bauchot, M.L.; Schneider, M. Fiche FAO d’identification des especes pour les besoins de la peche, Rev 1. Mediterranee et Mer Noire (zone de peche 37); FAO: Rome, Italy, 1987. [Google Scholar]
- Peres, M.B.; Klippel, S. Reproductive biology of southwestern Atlantic wreckfish, Polyprion americanus (Teleostei: Polyprionidae). Environ. Biol. Fishes 2003, 68, 163–173. [Google Scholar] [CrossRef]
- Mylonas, C.C.; Mitrizakis, N.; Papadaki, M.; Sigelaki, I. Reproduction of hatchery-produced meagre Argyrosomus regius in captivity I. Description of the annual reproductive cycle. Aquaculture 2013, 414–415, 309–317. [Google Scholar] [CrossRef]
- Peleteiro, J.B.; Saavedra Penas, C.; Pérez Rial, E.; Álvarez-Blázquez, B.; Soares, E.C.; Vilar Perón, A. Diversificación de especies en acuicultura. Estudio preliminar de la reproducción de la cherna (Polyprion americanus) en cautividad. In Proceedings of the Congreso Nacional de Acuicultura, Castelldefels, Spain, 21–24 November 2011. [Google Scholar]
- Anderson, S.A.; Salinas, I.; Walker, S.P.; Gublin, Y.; Pether, S.; Kohn, Y.Y.; Symonds, J.E. Early development of New Zealand hapuku Polyprion oxygeneios eggs and larvae. J. Fish Biol. 2012, 80, 555–571. [Google Scholar] [CrossRef] [PubMed]
- Symonds, J.E.; Walker, S.P.; Pether, S.; Gublin, Y.; McQueen, D.; King, A.; Irvine, G.W.; Setiawan, A.N.; Forsythe, J.A.; Bruce, M. Developing yellowtail kingfish (Seriola lalandi) and hāpuku (Polyprion oxygeneios) for New Zealand aquaculture. N. Z. J. Mar. Freshw. Res. 2014, 48, 371–384. [Google Scholar] [CrossRef]
- Wylie, M.J.; Setiawan, A.N.; Irvine, G.W.; Symonds, J.E.; Elizur, A.; Dos Santos, M.; Lokman, P.M. Ovarian development of captive F1 wreckfish (hapuku) Polyprion oxygeneios under constant and varying temperature regimes. Implications for broodstock management. Gen. Comp. Endocrinol. 2018, 257, 86–96. [Google Scholar] [CrossRef] [PubMed]
- Wylie, M.J.; Setiawan, A.N.; Irvine, G.W.; Symonds, J.E.; Elizur, A.; Lokman, P.M. Effects of neuropeptides and sex steroids on the pituitary-gonadal axis of pre-pubertal F1 wreckfish (hapuku) Polyprion oxygeneios in vivo: Evidence of inhibitory effects of androgens. Gen. Comp. Endocrinol. 2018, 257, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Mylonas, C.C.; Cardinaletti, G.; Sigelaki, I.; Polzonetti-Magni, A. Comparative efficacy of clove oil and 2-phenoxyethanol as anesthetics in the aquaculture of European sea bass (Dicentrarchus labrax) and gilthead sea bream (Sparus aurata) at different temperatures. Aquaculture 2005, 246, 467–481. [Google Scholar] [CrossRef]
- Papadaki, M.; Peleteiro, J.B.; Álvarez-Blázquez, B.; Rodríguez Villanueva, J.L.; Linares, F.; Vilar, A.; Pérez Rial, E.; Lluch, N.; Fakriadis, I.; Sigelaki, I.; et al. Description of the annual reproductive cycle of wreckfish Polyprion americanus in captivity. Fishes 2018, 3, 43. [Google Scholar] [CrossRef]
- Pérez Rial, E.; Giménez, I.; Rodríguez Villanueva, J.L.; Álvarez-Blázquez, B.; Chaves-Pozo, E.; Lluch, N.; Pazos, G.; Linares, F. Primeras experiencias de inducción a la maduración con gonadotropinas recombinantes en reproductores de cherna (Polyprion americanus). In Proceedings of the Congreso Nacional de Acuicultura, Zaragoza, Spain, 3–5 October 2017. [Google Scholar]
- Siddique, M.A.M.; Butts, I.; Linhart, O.; Macias, D.; Fauvel, C. Fertilization strategies for Sea Bass Dicentrarchus labrax (Linnaeus, 1758): Effects of pre-incubation and duration of egg receptivity in seawater. Aquacult. Res. 2015, 48, 386–394. [Google Scholar] [CrossRef]
- Cabrita, E.; Robles, V.; Cuñado, S.; Wallace, J.C.; Sarasquete, C.; Herráez, M.P. Evaluation of gilt head sea bream, Sparus aurata, sperm quality after cryopreservation in 5 mL macrotubes. Cryobiology 2005, 50, 273–284. [Google Scholar] [CrossRef] [PubMed]
- Mylonas, C.C.; Mitrizakis, N.; Castaldo, C.A.; Cerviño, C.P.; Papadaki, M.; Sigelaki, I. Reproduction of hatchery-produced meagre Argyrosomus regius in captivity II. Hormonal induction of spawning and monitoring of spawning kinetics, egg production and egg quality. Aquaculture 2013, 414–415, 318–327. [Google Scholar] [CrossRef]
- Cabrita, E.; Soares, F.; Dinis, M.T. Characterization of Senegalese sole, Solea senegalensis, male broodstock in terms of sperm production and quality. Aquaculture 2006, 261, 967–975. [Google Scholar] [CrossRef]
- Suquet, M.; Omnes, M.H.; Normant, Y.; Fauvel, C. Assessment of sperm concentration and motility in turbot (Scophthalmus maximus). Aquaculture 1992, 101, 177–185. [Google Scholar] [CrossRef]
- Watanabe, T.; Kitajima, C.; Fujita, S. Nutritional values of live organisms used in Japan for mass propagation of fish: A review. Aquaculture 1983, 34, 115–143. [Google Scholar] [CrossRef]
- Izquierdo, M.S.; Fernandez-Palacios, H.; Tacon, A.G.J. Effect of broodstock nutrition on reproductive performance of fish. Aquaculture 2001, 197, 25–42. [Google Scholar] [CrossRef]
- Sargent, J.R.; McEvoy, L.A.; Bell, J.G. Requirements, presentation and sources of polyunsaturated fatty acids in marine fish larval feeds. Aquaculture 1997, 155, 117–127. [Google Scholar] [CrossRef]
- Tocher, D.R. Metabolism and functions of lipids and fatty acids in teleost fish. Rev. Fish. Sci. 2003, 11, 107–184. [Google Scholar] [CrossRef]
- Izquierdo, M.S.; Koven, W. Lipids. In Larval Fish Nutrition, 1st ed.; Holt, J., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2011; pp. 47–82. ISBN 978-0-8138-1792-7. [Google Scholar]
- Oliva-Teles, A. Nutrition and health of aquaculture fish. J. Fish Dis. 2012, 35, 83–108. [Google Scholar] [CrossRef] [PubMed]
- Roncarati, A.; Cappuccinelli, R.; Stocchi, L.; Melotti, P. Wreckfish, Poliprion americanus (Bloch and Schneider, 1801), a promising species for aquaculture: Proximate composition, fatty acid profile and cholesterol content of wild Mediterranean specimens. J. Food Comp. Anal. 2014, 36, 104–110. [Google Scholar] [CrossRef]
- Saidi, H.; Morales-Medina, R.; Brehouch, A.; Fahd, S.; Guadix Escobar, E.M.; Pérez-Galvez, R. Effect of the supplementation of live preys enriched in cod liver oil on the survival rate, growth and fatty acid profile of meagre (Argyrosomus regius) larvae. Aquacult. Res. 2017, 49, 1133–1141. [Google Scholar] [CrossRef]
- Kamler, E. Ontogeny of yolk-feeding fish: An ecological perspective. Rev. Fish Biol. Fish. 2002, 12, 79–103. [Google Scholar] [CrossRef]
- Pankhurst, P.M.; Eagar, R. Changes in visual morphology through life history stages of the New Zealand snapper, Pagrus auratus. N. Z. J. Mar. Freshwat. Res. 1996, 30, 79–90. [Google Scholar] [CrossRef]
- Pankhurst, P.M.; Hilder, P.E. Effect of light intensity on feeding of striped trumpeter Latris lineata larvae. Mar. Freshwat. Res. 1998, 49, 363–368. [Google Scholar] [CrossRef]
- Pankhurst, P.M.; Pankhurst, N.W.; Montgomery, J.C. Comparison of behavioral and morphological measures of visual acuity during ontogeny in a teleost fish, Fosterygion varium, Tripterygiidae (Foster, 1801). Brain Behav. Evol. 1993, 42, 178–188. [Google Scholar] [CrossRef] [PubMed]
- Pankhurst, P.M.; Pankhurst, N.W.; Parks, M.C. Direct development of the visual system of the coral reef teleost, the spiny damsel, Acanthochromis polyacanthus. Environ. Biol. Fishes. 2002, 65, 431–440. [Google Scholar] [CrossRef]
- Roo, F.J.; Socorro, J.; Izquierdo, M.S.; Caballero, M.J.; Hernández-Cruz, C.M.; Fernández, A.; Fernández-Palacios, H. Development of red porgy Pagrus pagrus visual system in relation to changes in the digestive tract and larval feeding habits. Aquaculture 1999, 179, 499–512. [Google Scholar] [CrossRef]
- Shand, J.; Archer, M.A.; Collin, S.P. Ontogenetic changes in the retinal photoreceptor mosaic in a fish, the black bream, Acanthopagrus butcheri. J. Comp. Neurol. 1999, 412, 203–217. [Google Scholar] [CrossRef]
- Álvarez-Blázquez, B.; Rodríguez, J.L.; Vilar, A.; Mylonas, C.; Papandroulakis, C.; Pérez Rial, E.; Lluch, N.; Pérez, M.; Pazos, G.; Linares, F. Progress in the wreckfish intensive culture. New candidate species for aquaculture. In Proceedings of the European Aquaculture 2017, Dubrovnik, Croatia, 17–20 October 2017. [Google Scholar]
- Gunasekera, R.M.; Gooley, G.J.; De Silva, S.S. Characterization of ‘swollen yolk-sac syndrome’ in the Australian freshwater fish Murray cod, Maccullochella peelii peelii, and associated nutritional implications for large scale aquaculture. Aquaculture 1998, 169, 69–85. [Google Scholar] [CrossRef]
- Brzuzan, P.; Wozny, M.; Dobosz, S.; Kuzminski, H.; Łuczynski, M.K.; Gora, M. Blue sac disease in larval whitefish, Coregonus lavaretus (L.): Pathological changes in mRNA levels of CYP1A, ERa, and p53. J. Fish Dis. 2007, 30, 169–173. [Google Scholar] [CrossRef] [PubMed]
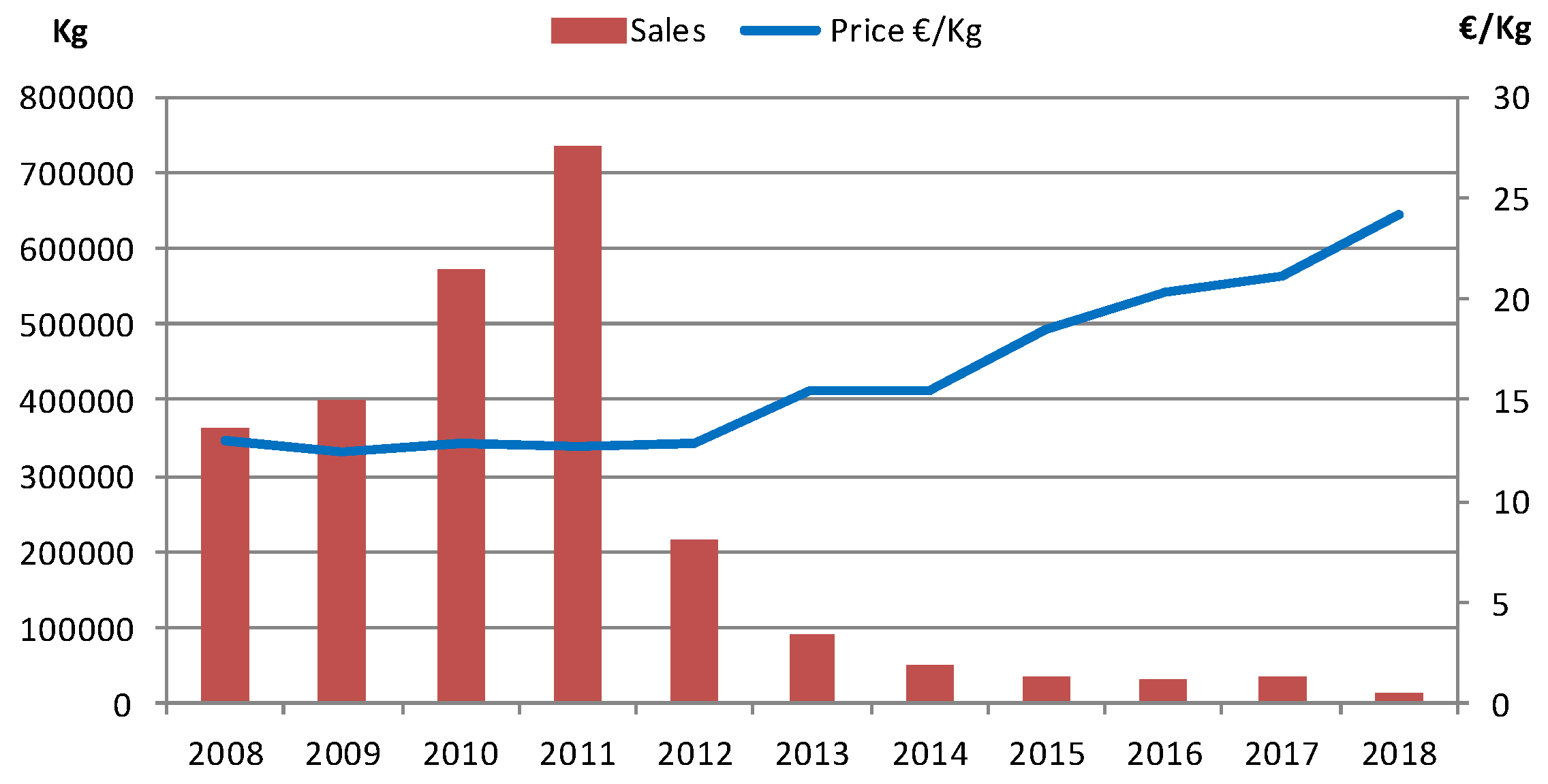
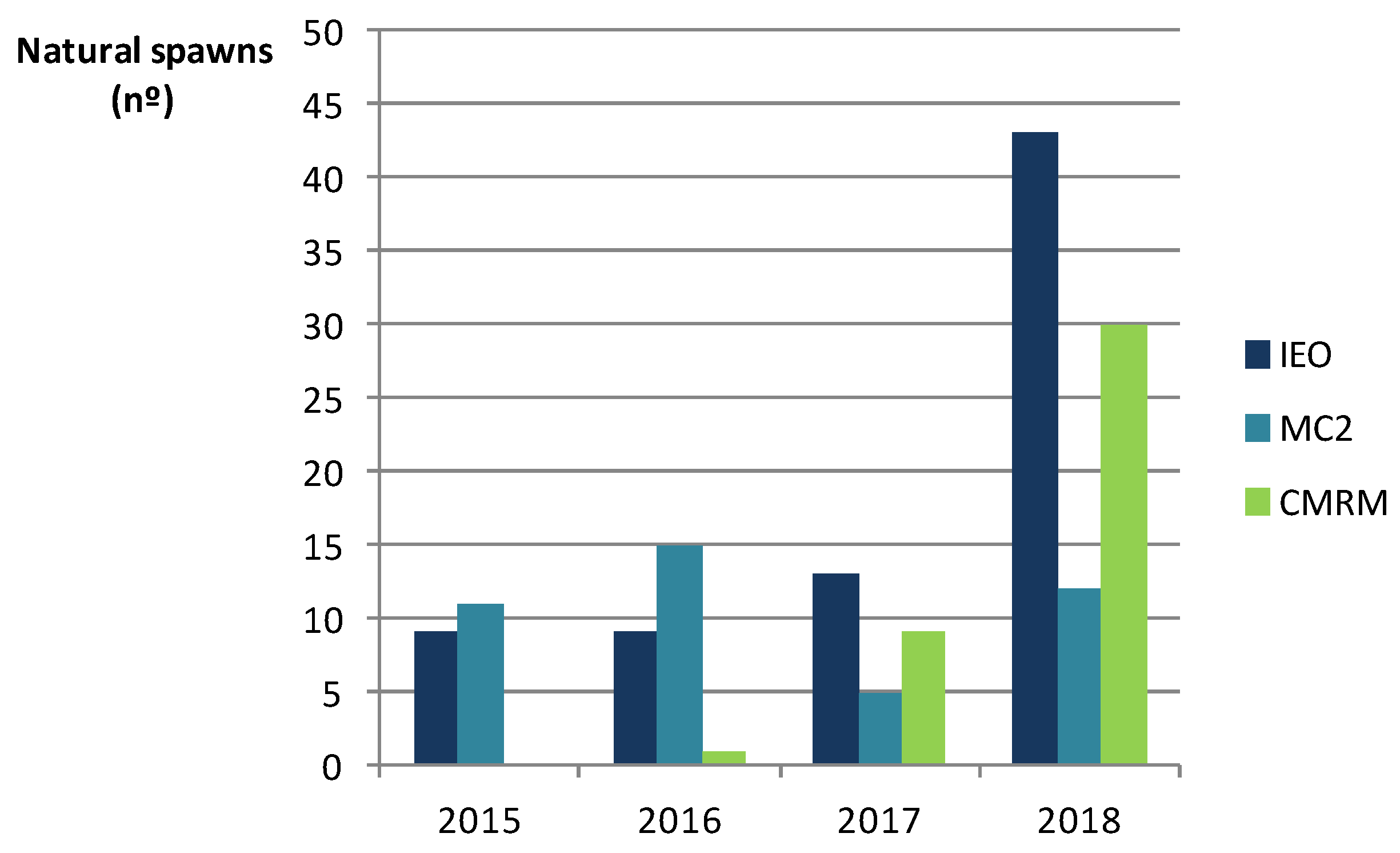
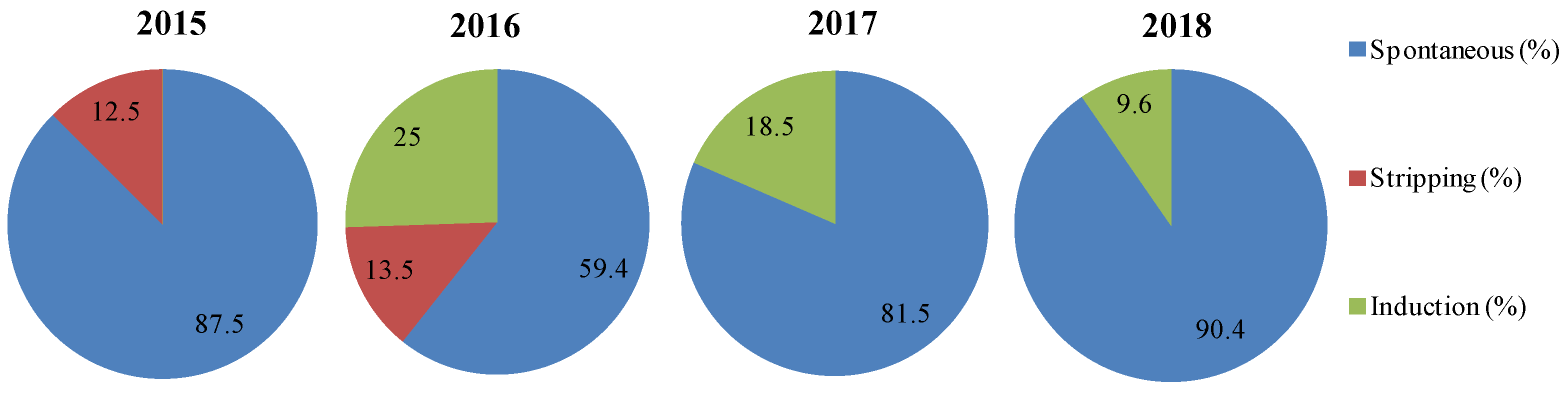

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pérez, E.; Linares, F.; Rodríguez Villanueva, J.L.; Vilar, A.; Mylonas, C.C.; Fakriadis, I.; Papadaki, M.; Papandroulakis, N.; Papadakis, I.; Robles, R.; et al. Wreckfish (Polyprion americanus). New Knowledge About Reproduction, Larval Husbandry, and Nutrition. Promise as a New Species for Aquaculture. Fishes 2019, 4, 14. https://doi.org/10.3390/fishes4010014
Pérez E, Linares F, Rodríguez Villanueva JL, Vilar A, Mylonas CC, Fakriadis I, Papadaki M, Papandroulakis N, Papadakis I, Robles R, et al. Wreckfish (Polyprion americanus). New Knowledge About Reproduction, Larval Husbandry, and Nutrition. Promise as a New Species for Aquaculture. Fishes. 2019; 4(1):14. https://doi.org/10.3390/fishes4010014
Chicago/Turabian StylePérez, Evaristo, Fátima Linares, José Luis Rodríguez Villanueva, Antonio Vilar, Constantinos C. Mylonas, Ioannis Fakriadis, Maria Papadaki, Nikos Papandroulakis, Ioannis Papadakis, Rocío Robles, and et al. 2019. "Wreckfish (Polyprion americanus). New Knowledge About Reproduction, Larval Husbandry, and Nutrition. Promise as a New Species for Aquaculture" Fishes 4, no. 1: 14. https://doi.org/10.3390/fishes4010014
APA StylePérez, E., Linares, F., Rodríguez Villanueva, J. L., Vilar, A., Mylonas, C. C., Fakriadis, I., Papadaki, M., Papandroulakis, N., Papadakis, I., Robles, R., Fauvel, C., Roo, J., Peleteiro, J. B., Lluch, N., Pazos, G., Méndez, B., Sigelaki, I., Gómez, C., Pérez, M., & Álvarez-Blázquez, B. (2019). Wreckfish (Polyprion americanus). New Knowledge About Reproduction, Larval Husbandry, and Nutrition. Promise as a New Species for Aquaculture. Fishes, 4(1), 14. https://doi.org/10.3390/fishes4010014





