Identification of a Novel Nucleobase-Ascorbate Transporter Family Member in Fish and Amphibians
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sequence Mining
2.2. Synteny
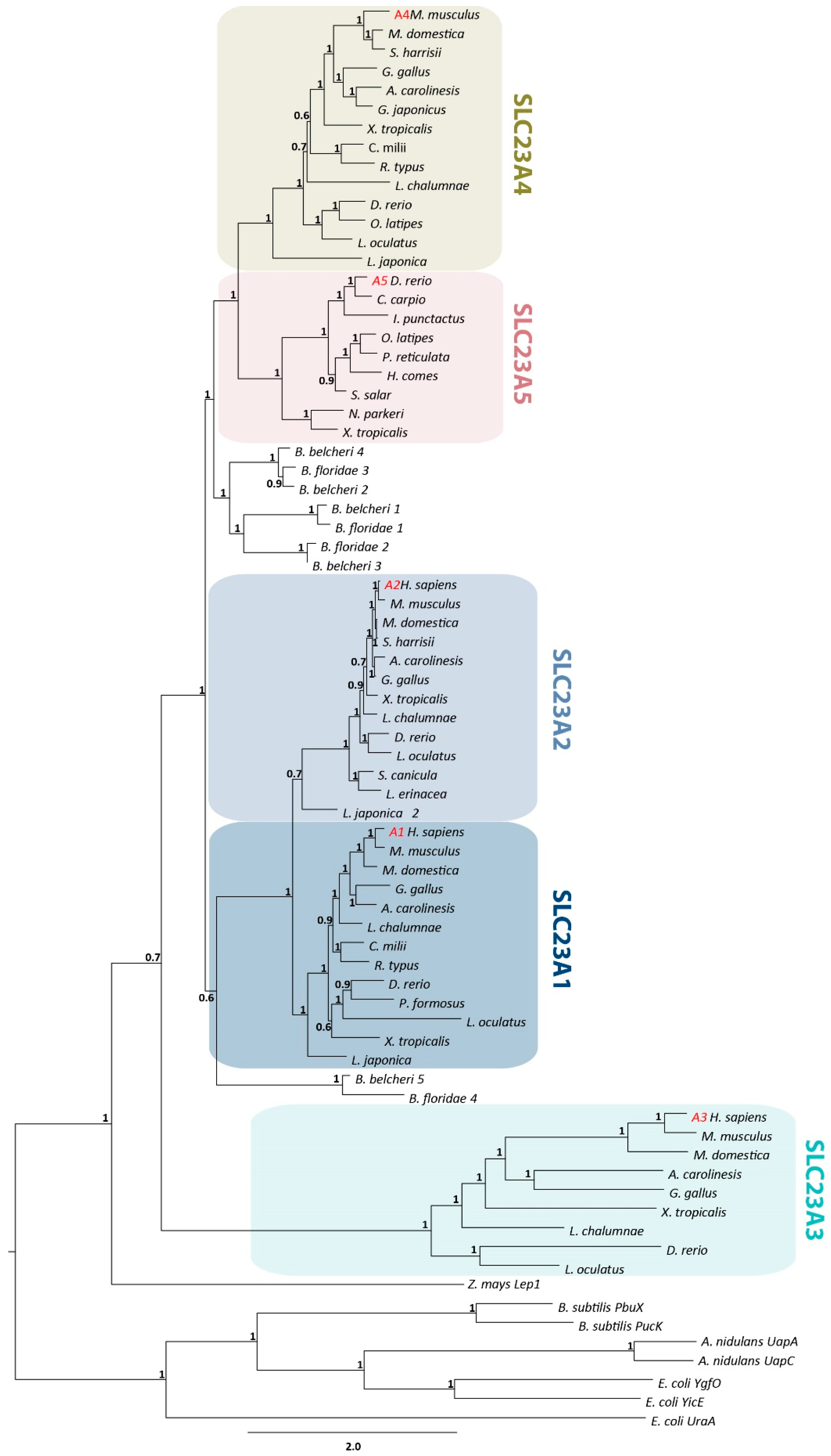
2.3. Phylogenetic Analysis
2.4. Membrane Topology Prediction
2.5. Filtering and Quality Control of Sequence Reading Archive (SRA) Datasets
2.6. Relative Gene Expression Levels
2.7. PCR-Based Validation of Slc23a5 Expression
3. Results
3.1. Identification of a Novel Member of the NAT Family in Neopterygians and Amphibians
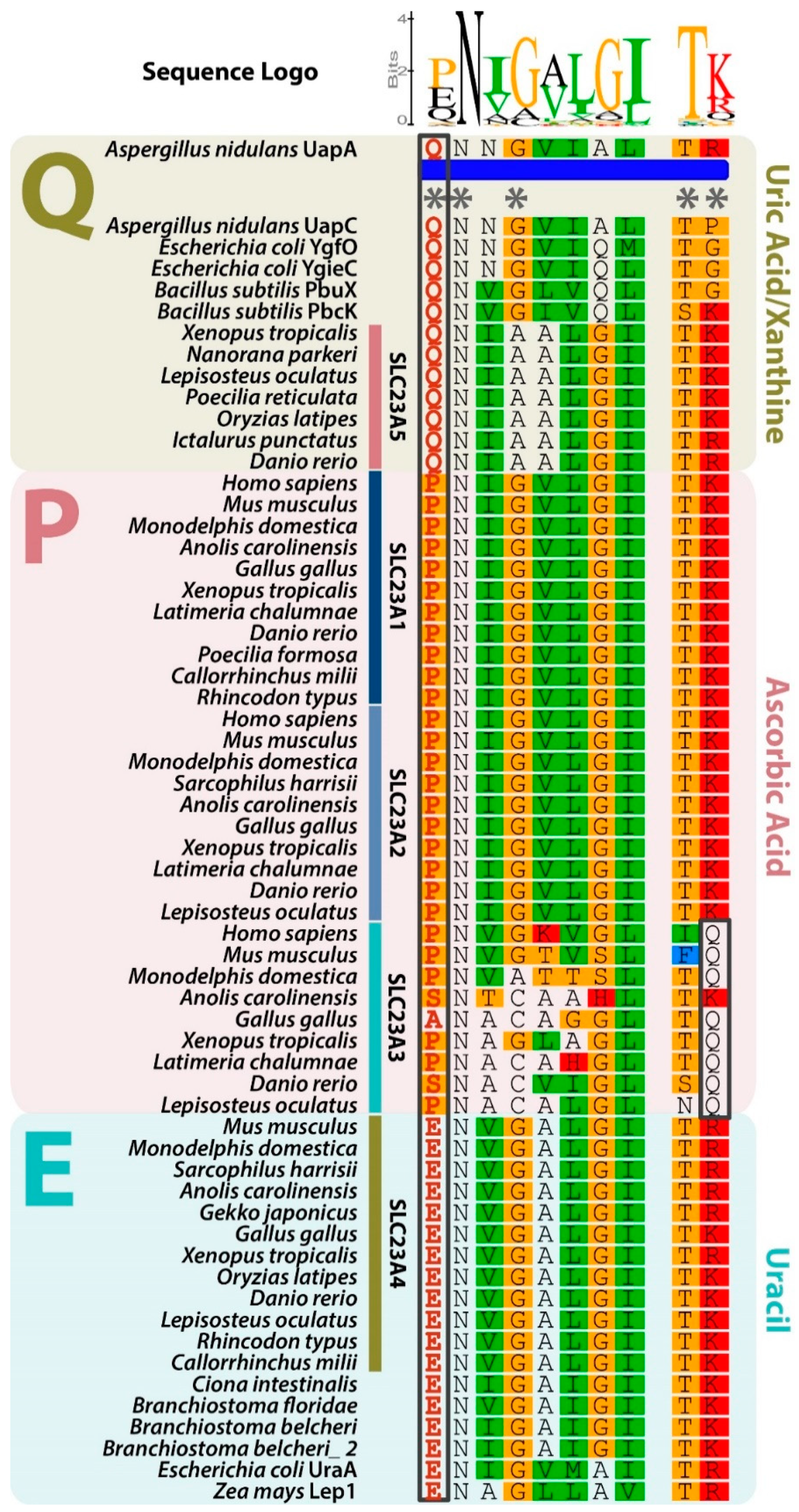
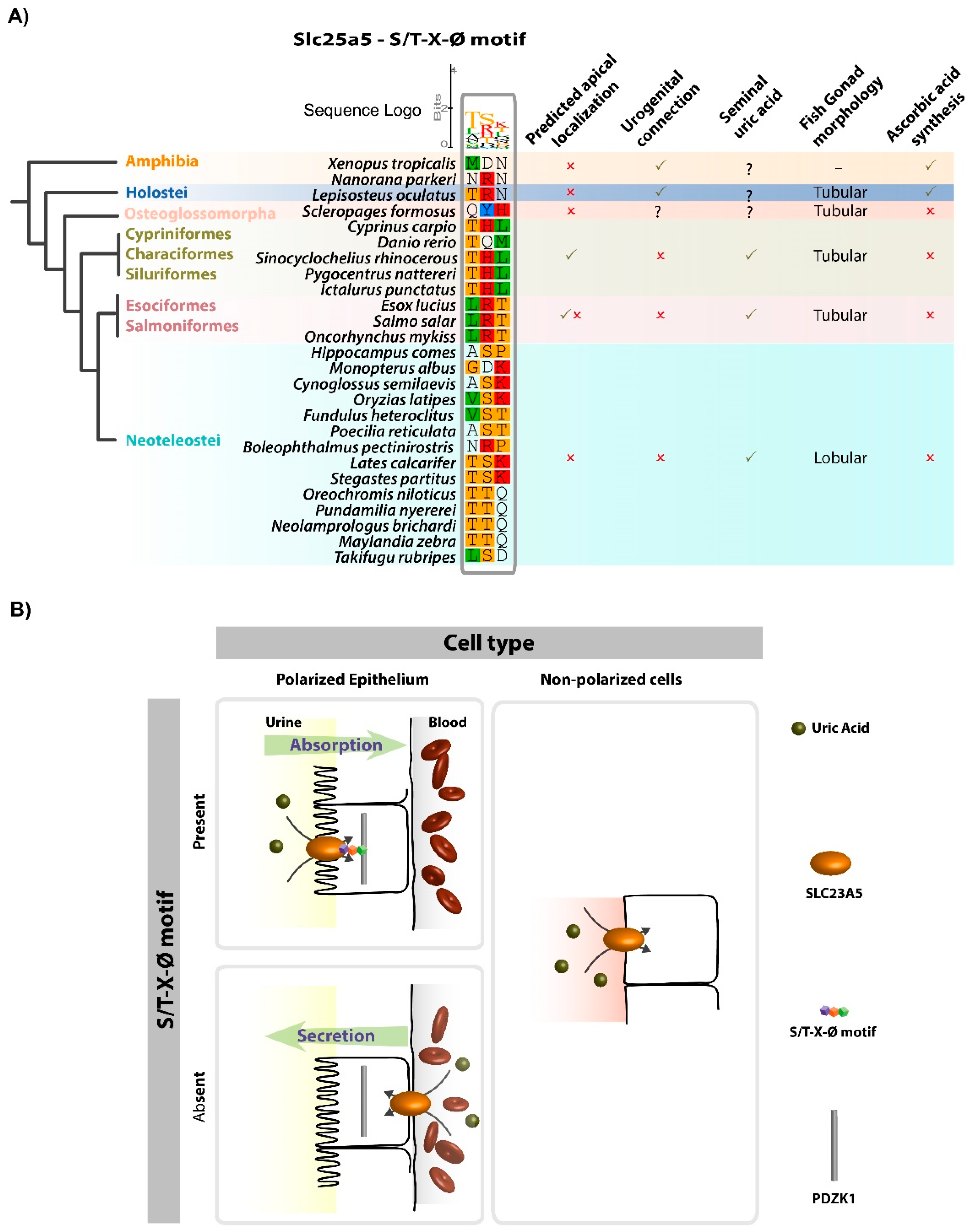
3.2. Slc23a5 Signature Motifs
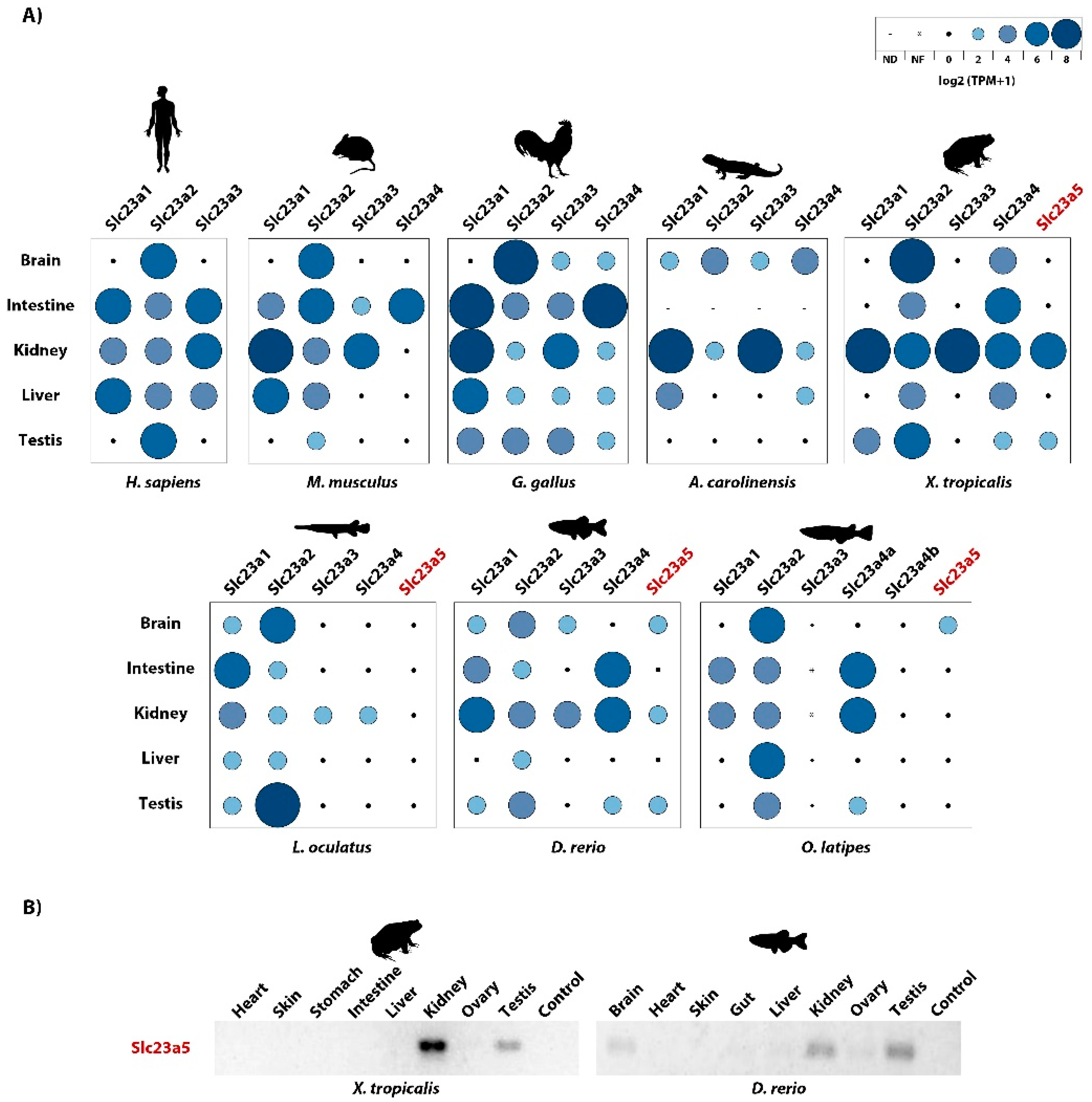
3.3. Slc23a5 Expression Patterns
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Burzle, M.; Suzuki, Y.; Ackermann, D.; Miyazaki, H.; Maeda, N.; Clemencon, B.; Burrier, R.; Hediger, M.A. The sodium-dependent ascorbic acid transporter family SLC23. Mol. Aspects Med. 2013, 34, 436–454. [Google Scholar] [CrossRef]
- Linster, C.L.; Van Schaftingen, E. Vitamin C. FEBS J. 2007, 274, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Drouin, G.; Godin, J.R.; Page, B. The genetics of vitamin C loss in vertebrates. Curr. Genomics 2011, 12, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Dutta Gupta, S.; Choudhury, P.K.; Chatterjee, I.B. Synthesis of l-ascorbic acid from d-glucurono-1,4-lactone conjugates by different species of animals. Int. J. Biochem. 1973, 4, 309–314. [Google Scholar] [CrossRef]
- Wright, P.A. Nitrogen excretion: Three end products, many physiological roles. J. Exp. Biol. 1995, 198, 273–281. [Google Scholar] [PubMed]
- Bobulescu, I.A.; Moe, O.W. Renal transport of uric acid: Evolving concepts and uncertainties. Adv. Chronic Kidney Dis. 2012, 19, 358–371. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Lario, B.; Macarrón-Vicente, J. Evolution of Uric Acid Metabolism in Humans. In eLS; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2001. [Google Scholar] [CrossRef]
- Oda, M.; Satta, Y.; Takenaka, O.; Takahata, N. Loss of urate oxidase activity in hominoids and its evolutionary implications. Mol. Biol. Evol. 2002, 19, 640–653. [Google Scholar] [CrossRef] [PubMed]
- Corti, A.; Casini, A.F.; Pompella, A. Cellular pathways for transport and efflux of ascorbate and dehydroascorbate. Arch. Biochem. Biophys. 2010, 500, 107–115. [Google Scholar] [CrossRef]
- Savini, I.; Rossi, A.; Pierro, C.; Avigliano, L.; Catani, M.V. SVCT1 and SVCT2: Key proteins for vitamin C uptake. Amino Acids 2008, 34, 347–355. [Google Scholar] [CrossRef]
- Yamamoto, S.; Inoue, K.; Murata, T.; Kamigaso, S.; Yasujima, T.; Maeda, J.Y.; Yoshida, Y.; Ohta, K.Y.; Yuasa, H. Identification and functional characterization of the first nucleobase transporter in mammals: Implication in the species difference in the intestinal absorption mechanism of nucleobases and their analogs between higher primates and other mammals. J. Biol. Chem. 2010, 285, 6522–6531. [Google Scholar] [CrossRef]
- Koukaki, M.; Vlanti, A.; Goudela, S.; Pantazopoulou, A.; Gioule, H.; Tournaviti, S.; Diallinas, G. The nucleobase-ascorbate transporter (NAT) signature motif in UapA defines the function of the purine translocation pathway. J. Mol. Biol. 2005, 350, 499–513. [Google Scholar] [CrossRef] [PubMed]
- Papakostas, K.; Frillingos, S. Substrate selectivity of YgfU, a uric acid transporter from Escherichia coli. J. Biol. Chem. 2012, 287, 15684–15695. [Google Scholar] [CrossRef] [PubMed]
- Katoh, K.; Standley, D.M. MAFFT multiple sequence alignment software version 7: Improvements in performance and usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef] [PubMed]
- Guindon, S.; Dufayard, J.F.; Lefort, V.; Anisimova, M.; Hordijk, W.; Gascuel, O. New algorithms and methods to estimate maximum-likelihood phylogenies: Assessing the performance of PhyML 3.0. Syst. Biol. 2010, 59, 307–321. [Google Scholar] [CrossRef]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef] [PubMed]
- Yates, A.; Akanni, W.; Amode, M.R.; Barrell, D.; Billis, K.; Carvalho-Silva, D.; Cummins, C.; Clapham, P.; Fitzgerald, S.; Gil, L.; et al. Ensembl 2016. Nucleic Acids Res. 2016, 44, D710–D716. [Google Scholar] [CrossRef] [PubMed]
- O’Leary, N.A.; Wright, M.W.; Brister, J.R.; Ciufo, S.; Haddad, D.; McVeigh, R.; Rajput, B.; Robbertse, B.; Smith-White, B.; Ako-Adjei, D.; et al. Reference sequence (RefSeq) database at NCBI: Current status, taxonomic expansion, and functional annotation. Nucleic Acids Res. 2016, 44, D733–D745. [Google Scholar] [CrossRef]
- Langmead, B.; Salzberg, S.L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 2012, 9, 357–359. [Google Scholar] [CrossRef] [Green Version]
- Li, B.; Dewey, C.N. RSEM: Accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinformatics 2011, 12, 323. [Google Scholar] [CrossRef]
- Anzai, N.; Jutabha, P.; Amonpatumrat-Takahashi, S.; Sakurai, H. Recent advances in renal urate transport: Characterization of candidate transporters indicated by genome-wide association studies. Clin. Exp. Nephrol. 2012, 16, 89–95. [Google Scholar] [CrossRef]
- Dev, K.K. Making protein interactions druggable: Targeting PDZ domains. Nat. Rev. Drug Discov. 2004, 3, 1047–1056. [Google Scholar] [CrossRef] [PubMed]
- Clapéron, A.; Mergey, M.; Fouassier, L. Roles of the scaffolding proteins NHERF in liver biology. Clin. Res. Hepatol. Gastroenterol. 2011, 35, 176–181. [Google Scholar] [CrossRef] [PubMed]
- Raghuram, V.; Mak, D.-O.D.; Foskett, J.K. Regulation of cystic fibrosis transmembrane conductance regulator single-channel gating by bivalent PDZ-domain-mediated interaction. Proc. Natl. Acad. Sci. USA 2001, 98, 1300–1305. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, S.; Yue, H.; Derin, R.B.; Guggino, W.B.; Li, M. Accessory Protein Facilitated CFTR-CFTR Interaction, a Molecular Mechanism to Potentiate the Chloride Channel Activity. Cell 2000, 103, 169–179. [Google Scholar] [CrossRef] [Green Version]
- Braun, E.J. Comparative renal function in reptiles, birds, and mammals. Semin. Avian Exotic. Pet. Med. 1998, 7, 62–71. [Google Scholar] [CrossRef]
- Singer, M.A. Do mammals, birds, reptiles and fish have similar nitrogen conserving systems? Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2003, 134, 543–558. [Google Scholar] [CrossRef]
- Ciereszko, A.; Dabrowski, K.; Kucharczyk, D.; Dobosz, S.; Goryczko, K.; Glogowski, J. The presence of uric acid, an antioxidantive substance, in fish seminal plasma. Fish Physiol. Biochem. 1999, 21, 313–315. [Google Scholar] [CrossRef]
- Dzyuba, V.; Dzyuba, B.; Cosson, J.; Boryshpolets, S.; Yamaner, G.; Kholodniy, V.; Rodina, M. The antioxidant system of sterlet seminal fluid in testes and Wolffian ducts. Fish Physiol. Biochem. 2014, 40, 1731–1739. [Google Scholar] [CrossRef]
- Lahnsteiner, F.; Mansour, N. A comparative study on antioxidant systems in semen of species of the Percidae, Salmonidae, Cyprinidae, and Lotidae for improving semen storage techniques. Aquaculture 2010, 307, 130–140. [Google Scholar] [CrossRef]
- Lahnsteiner, F.; Mansour, N.; Plaetzer, K. Antioxidant systems of brown trout (Salmo trutta f. fario) semen. Anim. Reprod. Sci. 2010, 119, 314–321. [Google Scholar] [CrossRef]
- Keogh, L.M.; Byrne, P.G.; Silla, A.J. The effect of antioxidants on sperm motility activation in the Booroolong frog. Anim. Reprod. Sci. 2017, 183, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, N.B.; Dulvy, N.K.; Reynolds, J.D. Life-history correlates of the evolution of live bearing in fishes. Philos. Trans. R. Soc. Lond. B. Biol. Sci 2002, 357, 259–267. [Google Scholar] [CrossRef] [Green Version]
- Blün, V. Comparative Anatomy of the Urogenital System. In Vertebrate Reproduction, a Textbook; Springer: Berlin/Heidelberg, Germany; New York, NY, USA; Tokyo, Japan, 1986; pp. 43–74. [Google Scholar] [CrossRef]
- Dzyuba, B.; Cosson, J.; Boryshpolets, S.; Bondarenko, O.; Dzyuba, V.; Prokopchuk, G.; Gazo, I.; Rodina, M.; Linhart, O. In vitro sperm maturation in sterlet, Acipenser ruthenus. Reprod. Biol. 2014, 14, 160–163. [Google Scholar] [CrossRef] [PubMed]
- Ciereszko, A.; Dabrowski, K. Sperm quality and ascorbic acid concentration in rainbow trout semen are affected by dietary vitamin C: An across-season study. Biol. Reprod. 1995, 52, 982–988. [Google Scholar] [CrossRef] [PubMed]
- Ciereszko, A.; Dabrowski, K. Effect of ascorbic acid supplement in vitro onrainbow trout sperm viability. Aquacult. Int. 2000, 8, 1–8. [Google Scholar] [CrossRef]
- Moreau, R.; Dabrowski, K. Biosynthesis of ascorbic acid by extant actinopterygians. J. Fish Biol. 2000, 57, 733–745. [Google Scholar] [CrossRef]
- Forsman, A.; Tibblin, P.; Berggren, H.; Nordahl, O.; Koch-Schmidt, P.; Larsson, P. Pike Esox lucius as an emerging model organism for studies in ecology and evolutionary biology: A review. J. Fish Biol. 2015, 87, 472–479. [Google Scholar] [CrossRef]
- Bone, Q.; Moore, R.H. Biology of Fishes; Taylor & Francis Group: New York, NY, USA, 2008. [Google Scholar]
- Scott, D.B.C.; Fuller, J.D. The reproductive biology of Scleropages formosus (Müller & Schlegel) (Osteoglossomorpha, Osteoglossidae) in Malaya, and the morphology of its pituitary gland. J. Fish Biol. 1976, 8, 45–53. [Google Scholar] [CrossRef]
- Parenti, L.R.; Grier, H.J. Evolution and phylogeny of gonad morphology in bony fishes. Integr. Comp. Biol. 2004, 44, 333–348. [Google Scholar] [CrossRef]
- Uttara, B.; Singh, A.V.; Zamboni, P.; Mahajan, R.T. Oxidative Stress and Neurodegenerative Diseases: A Review of Upstream and Downstream Antioxidant Therapeutic Options. Curr. Neuropharmacol. 2009, 7, 65–74. [Google Scholar] [CrossRef]
- Lopez-Cruz, R.I.; Dafre, A.L.; Filho, D.W. Oxidative Stress in Sharks and Rays. In Oxidative Stress in Aquatic Ecosystems; Abele, D., Vazquez-Medina, J.P., Zenteno-Savin, T., Eds.; Wiley-Blackwell: Hoboken, NJ, USA, 2011; pp. 157–164. [Google Scholar]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oliveira, D.; M. Machado, A.; Cardoso, T.; Lopes-Marques, M.; C. Castro, L.F.; Ruivo, R. Identification of a Novel Nucleobase-Ascorbate Transporter Family Member in Fish and Amphibians. Fishes 2019, 4, 1. https://doi.org/10.3390/fishes4010001
Oliveira D, M. Machado A, Cardoso T, Lopes-Marques M, C. Castro LF, Ruivo R. Identification of a Novel Nucleobase-Ascorbate Transporter Family Member in Fish and Amphibians. Fishes. 2019; 4(1):1. https://doi.org/10.3390/fishes4010001
Chicago/Turabian StyleOliveira, Diogo, André M. Machado, Tiago Cardoso, Mónica Lopes-Marques, L. Filipe C. Castro, and Raquel Ruivo. 2019. "Identification of a Novel Nucleobase-Ascorbate Transporter Family Member in Fish and Amphibians" Fishes 4, no. 1: 1. https://doi.org/10.3390/fishes4010001




