Pancreatitis-Associated Protein in Neonatal Screening for Cystic Fibrosis: Strengths and Weaknesses
Abstract
1. Introduction
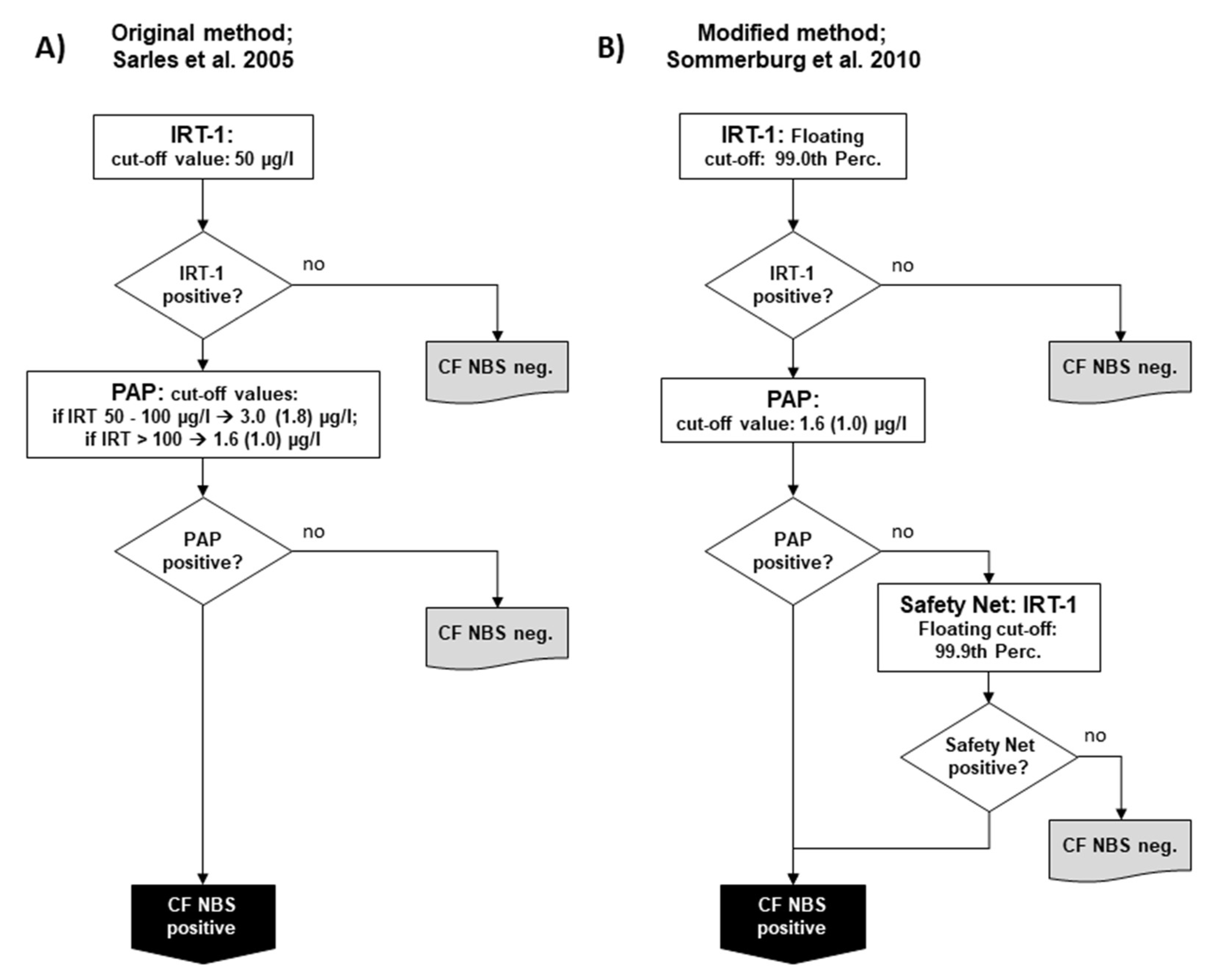
2. The Evolution of the PAP Kit
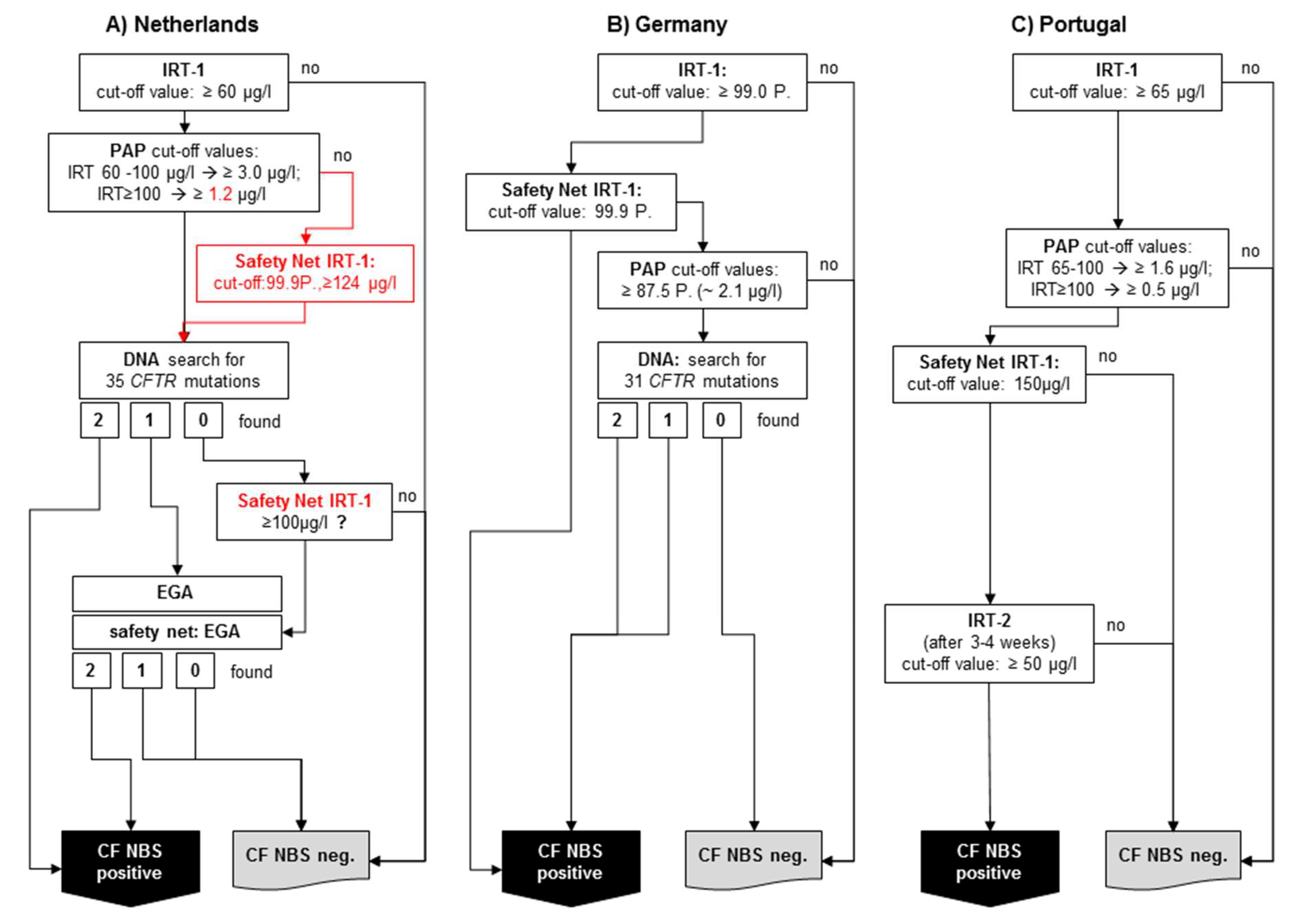
3. Description of Selected European Pilot Studies
4. Findings from the Pilot Studies
4.1. IRT/PAP Protocols Detect Less Healthy Carriers
4.2. IRT/PAP Protocols Detect Less CFSPID
4.3. IRT/PAP Protocols May Show Lower Sensitivity than IRT/DNA Protocols
- The use of an IRT-dependent safety net: When the pilot studies were started in the Germany, the general concern was that the PAP strategy had a worse sensitivity than a well-performing genetic CF NBS. Similar to the IRT/DNA protocols with a restricted mutation panel, an IRT-dependent safety net was added six months after starting the pilot studies. Therefore, CF NBS is considered positive if the initial IRT is above the 99.9th percentile, regardless of the PAP result. When the results of the pilot study conducted in Prague (Czech Republic) were published in 2012, the IRT/PAP strategy showed a very low sensitivity of only 76% [13]. After a re-evaluation for a joint, posthoc analysis of the raw data from Prague, Dresden and Heidelberg, it was found that the sensitivity of the Prague PAP-based CF NBS would have been 89.5% if the colleagues there had used the original IRT/PAP protocol but with the IRT-dependent safety net, as was done in the German centres [18]. Furthermore, a recently published paper on the Dutch CF NBS shows that out of eight CF patients not detected in the IRT/PAP part of the IRT/PAP/DNA(35)/EGA strategy, five would probably have been found if such an IRT-dependent safety net had been used [16].
- Renouncing the two IRT-dependent PAP cut-off values: As mentioned, the reason to use the two IRT-dependent PAP cut-offs was based on the assumption that such a protocol would avoid the detection of CFSPID. In addition, IRT/PAP protocols with two IRT-dependent PAP cut-off values were proposed to detect less healthy newborns as false positives compared to protocols with only one PAP cut-off value. However, the results of the aforementioned joint posthoc analysis of the data from Prague, Dresden, and Heidelberg suggest that IRT/PAP protocols with two IRT-dependent PAP cut-off values may have limited sensitivity compared to those with only one PAP cut-off value. In a joint simulation of raw data from Prague and Heidelberg, it was found that by using two PAP cut-off values, four newborns with two mutations in the CFTR gene would have been missed, but would have been detected by the protocol with one PAP cut-off. Only one out of these four newborns carried a CFTR mutation with varying clinical consequence and had a normal sweat chloride. The other three newborns were diagnosed with classical CF with pancreatic insufficiency. Two out of these three CF patients suffered from MI and would have been diagnosed clinically. However, the third CF patient would have been missed by all IRT/PAP protocols relying on two IRT-dependent PAP cut-offs [18]. It can be argued whether one has to consider three missed patients with CF or only one, since two out of these three presented with MI.
- Anyway, the fact that newborns carrying two CF-causing mutations were not detected due to the IRT/PAP protocol with two PAP cut-offs raises the question of whether such a protocol can achieve sufficient sensitivity. It is interesting to note that if the colleagues in Prague had used the same IRT/PAP protocol as that used in Heidelberg, not only with the IRT dependent safety net, but also with only one PAP cut-off value, the sensitivity would have been 94.7%. Also, in a recently published work on the aforementioned Dutch CF NBS program, it was shown that if only one PAP cut-off value had been used, one CF patient out of the eight CF patients not found would still have been detected. With the five CF patients that would have been found by the safety Net, six of the eight CF patients would have been found [16].
- Lowering of PAP cut-off values: Due to the fact that all the pilot studies mentioned above were started with a MucoPAP kit whose PAP cut-off values had not yet been sufficiently evaluated, the most obvious solution for sensitivity problems would have been to simply adjust the PAP cut-off values downwards. Actually, this was also done later by Sarles et al. and reported in a publication in 2014 [10]. However, significantly lowered PAP cut-off values were not only found there, but were seen in recent years also in other PAP-based protocols (e.g., [17]). Yet, it is precisely this approach that significantly increases the number of false-positive newborns detected.
- Using both biochemical markers, IRT and PAP, at the same time: In all current PAP-based CF-NBS protocols, IRT and PAP are used sequentially. However, the simultaneous use of both biomarkers instead of two steps, e.g., by using the product of IRT and PAP, has the potential to make the screening strategy significantly more sensitive than in the IRT/PAP protocols currently in use. Despite the simultaneous use of both parameters, IRT can still be used as a first-tier-parameter that triggers the PAP measurement if it is above a certain cut-off value. Such an approach was demonstrated by the Dresden group in a posthoc analysis using raw data from the pilot studies of the two German CF NBS centres, i.e., Dresden and Heidelberg [14]. The data from Heidelberg showed the highest sensitivity with the IRTxPAP product (98.3%), in contrast to the revised strategy of Sarles et al. published in 2014 (94.9%), and also in contrast to the Heidelberg IRT/PAP-SN protocol (96.6%).
- Time-dependent sampling of the dried blood for neonatal screening: There is unpublished local experience from Australia, still acknowledged by a number of CF NBS specialists, that the use of PAP is not sensitive enough if the dried blood sample for NBS is taken from the infant before the age of 48 h. As a reason for this, it was assumed that the PAP blood levels in infants with cystic fibrosis increase over time. According to our experience, this could be true, but not only in CF infants. In Germany, the collection of the dried blood sample is usually carried out between the 36th and 72nd hour of life, but for special reasons, we sometimes see early or late sampling. If we group all available PAP values of the infants studied in recent years into 12-h intervals, we see a trend of an increase in the 25th, 50th, and 75th percentiles from 24 h to 72 h (personal communication O. Sommerburg). However, when we focused on CF patients not found in our IRT/PAP protocol, we could not confirm that these CF patients were missed because the time of collection of the dry blood sample was before the 48th hour of life. In this regard, after more than 10 years of PAP-based CF NBS, we consider it to be proven that PAP screening with samples collected between 36 and 48 h of life is feasible. Yet, if the majority of infants in a country are screened for NBS before the 36th hour of life, we might imagine that PAP blood levels might still be too low. In this case, we would recommend a comprehensive pilot study to test the feasibility of a PAP-based CF NBS also under these conditions.
4.4. Pure Biochemical IRT/PAP Protocols Show a Relatively Low Positive Predictive Value
4.5. Current PAP-Based CF Screening Protocols in Use
Funding
Conflicts of Interest
References
- Castellani, C.; Southern, K.W.; Brownlee, K.; Roelse, J.D.; Duff, A.; Farrell, M.; Mehta, A.; Munck, A.; Pollitt, R.; Sermet-Gaudelus, I.; et al. European best practice guidelines for cystic fibrosis neonatal screening. J. Cyst. Fibros. 2009, 8, 153–173. [Google Scholar] [CrossRef] [PubMed]
- Wilcken, B.M.; Wiley, V. Newborn screening methods for cystic fibrosis. Paediatr. Respir. Rev. 2003, 4, 272–277. [Google Scholar] [CrossRef]
- Munck, A.; Delmas, M.; Audrézet, M.-P.; Lemonnier, L.; Cheillan, D.; Roussey, M. Optimization of the French cystic fibrosis newborn screening programme by a centralized tracking process. J. Med Screen. 2017, 25, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Terlizzi, V.; Mergni, G.; Buzzetti, R.; Centrone, C.; Zavataro, L.; Braggion, C. Cystic fibrosis screen positive inconclusive diagnosis (CFSPID): Experience in Tuscany, Italy. J. Cyst. Fibros. 2019, 18, 484–490. [Google Scholar] [CrossRef] [PubMed]
- Iovanna, J.L.; Férec, C.; Sarles, J.; Dagorn, J.C. The pancreatitis-associated protein (PAP). A new candidate for neonatal screening of cystic fibrosis. Comptes Rendus de l’Académie des Sciences Series III Sciences de la Vie 1994, 317, 561–564. [Google Scholar]
- Iovanna, J.L.; Keim, V.; Nordback, I.; Montalto, G.; Camarena, J.; Letoublon, C.; Levy, P.; Berthézène, P.; Dagorn, J.-C. Serum levels of pancreatitis-associated protein as indicators of the course of acute pancreatitis. Gastroenterology 1994, 106, 728–734. [Google Scholar] [CrossRef]
- Sarles, J.; Barthellemy, S.; Férec, C.; Iovanna, J.; Roussey, M.; Farriaux, J.-P.; Toutain, A.; Berthelot, J.; Maurin, N.; Codet, J.-P.; et al. Blood concentrations of pancreatitis associated protein in neonates: Relevance to neonatal screening for cystic fibrosis. Arch. Dis. Child. Fetal Neonatal Ed. 1999, 80, F118–F122. [Google Scholar] [CrossRef] [PubMed]
- Sarles, J.; Berthézène, P.; Le Louarn, C.; Somma, C.; Perini, J.-M.; Catheline, M.; Mirallié, S.; Luzet, K.; Roussey, M.; Farriaux, J.-P.; et al. Combining Immunoreactive Trypsinogen and Pancreatitis-Associated Protein Assays, a Method of Newborn Screening for Cystic Fibrosis that Avoids DNA Analysis. J. Pediatr. 2005, 147, 302–305. [Google Scholar] [CrossRef] [PubMed]
- Barthellemy, S.; Maurin, N.; Roussey, M.; Férec, C.; Murolo, S.; Berthézène, P.; Iovanna, J.L.; Dagorn, J.C.; Sarles, J. Evaluation of 47,213 infants in neonatal screening for cystic fibrosis, using pancreatitis-associated protein and immunoreactive trypsinogen assays. Arch. Pédiatrie 2001, 8, 275–281. [Google Scholar] [CrossRef]
- Sarles, J.; Giorgi, R.; Berthézène, P.; Munck, A.; Cheillan, D.; Dagorn, J.-C.; Roussey, M. Neonatal screening for cystic fibrosis: Comparing the performances of IRT/DNA and IRT/PAP. J. Cyst. Fibros. 2014, 13, 384–390. [Google Scholar] [CrossRef] [PubMed]
- Sommerburg, O.; Lindner, M.; Muckenthaler, M.; Kohlmueller, D.; Leible, S.; Feneberg, R.; Kulozik, A.E.; Mall, M.A.; Hoffmann, G.F. Initial evaluation of a biochemical cystic fibrosis newborn screening by sequential analysis of immunoreactive trypsinogen and pancreatitis-associated protein (IRT/PAP) as a strategy that does not involve DNA testing in a Northern European population. J. Inherit. Metab. Dis. 2010, 33, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Langen, A.M.M.V.-V.; Loeber, J.G.; Elvers, B.; Triepels, R.H.; Gille, J.J.; Van Der Ploeg, C.P.B.; Reijntjens, S.; Dompeling, E.; Dankert-Roelse, J.E. Novel strategies in newborn screening for cystic fibrosis: A prospective controlled study. Thorax 2012, 67, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Krulisova, V.; Balaščaková, M.; Skalická, V.; Piskackova, T.; Holubova, A.; Paděrová, J.; Krenkova, P.; Dvořáková, L.; Zemkova, D.; Kracmar, P.; et al. Prospective and parallel assessments of cystic fibrosis newborn screening protocols in the Czech Republic: IRT/DNA/IRT versus IRT/PAP and IRT/PAP/DNA. Eur. J. Nucl. Med. Mol. Imaging 2012, 171, 1223–1229. [Google Scholar]
- Weidler, S.; Stopsack, K.H.; Hammermann, J.; Sommerburg, O.; Mall, M.A.; Hoffmann, G.F.; Kohlmüller, D.; Okun, J.G.; Macek, M.; Votava, F.; et al. A product of immunoreactive trypsinogen and pancreatitis-associated protein as second-tier strategy in cystic fibrosis newborn screening. J. Cyst. Fibros. 2016, 15, 752–758. [Google Scholar] [CrossRef] [PubMed]
- Sommerburg, O.; Hammermann, J.; Lindner, M.; Stahl, M.; Muckenthaler, M.; Kohlmueller, D.; Happich, M.; Kulozik, A.E.; Stopsack, M.; Gahr, M.; et al. Five years of experience with biochemical cystic fibrosis newborn screening based on IRT/PAP in Germany. Pediatr. Pulmonol. 2015, 50, 655–664. [Google Scholar] [CrossRef] [PubMed]
- Dankert-Roelse, J.E.; Bouva, M.J.; Jakobs, B.S.; Janssens, H.M.; Groot, K.D.W.-D.; Schönbeck, Y.; Gille, J.J.; Gulmans, V.A.; Verschoof-Puite, R.K.; Schielen, P.; et al. Newborn blood spot screening for cystic fibrosis with a four-step screening strategy in the Netherlands. J. Cyst. Fibros. 2019, 18, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Marcão, A.; Barreto, C.; Pereira, J.B.; Vaz, L.; Cavaco, J.; Casimiro, A.; Félix, M.; Silva, T.R.; Barbosa, T.; Freitas, C.; et al. Cystic Fibrosis Newborn Screening in Portugal: PAP Value in Populations with Stringent Rules for Genetic Studies. Int. J. Neonatal Screen. 2018, 4, 22. [Google Scholar] [CrossRef]
- Sommerburg, O.; Krulišová, V.; Hammermann, J.; Lindner, M.; Stahl, M.; Muckenthaler, M.; Kohlmueller, D.; Happich, M.; Kulozik, A.E.; Votava, F.; et al. Comparison of different IRT-PAP protocols to screen newborns for cystic fibrosis in three central European populations. J. Cyst. Fibros. 2014, 13, 15–23. [Google Scholar] [CrossRef]
- Castellani, C.; Duff, A.; Bell, S.C.; Heijerman, H.G.; Munck, A.; Ratjen, F.; Sermet-Gaudelus, I.; Southern, K.W.; Barben, J.; A Flume, P.; et al. ECFS best practice guidelines: The 2018 revision. J. Cyst. Fibros. 2018, 17, 153–178. [Google Scholar] [CrossRef]
- Calvin, J.; Hogg, S.L.; McShane, D.; McAuley, S.A.; Iles, R.; Ross-Russell, R.; MacLean, F.M.; Heeley, M.E.; Heeley, A.F. Thirty-years of screening for cystic fibrosis in East Anglia. Arch. Dis. Child. 2012, 97, 1043–1047. [Google Scholar] [CrossRef]
- Kharrazi, M.; Yang, J.; Bishop, T.; Lessing, S.; Young, S.; Graham, S.; Pearl, M.; Chow, H.; Ho, T.; Currier, R.; et al. Newborn Screening for Cystic Fibrosis in California. Pediatrics 2015, 136, 1062–1072. [Google Scholar] [CrossRef] [PubMed]
- Sontag, M.; Lee, R.; Wright, D.; Freedenberg, D.; Sagel, S.D. Improving the Sensitivity and Positive Predictive Value in a Cystic Fibrosis Newborn Screening Program Using a Repeat Immunoreactive Trypsinogen and Genetic Analysis. J. Pediatr. 2016, 175, 150–158.e1. [Google Scholar] [CrossRef] [PubMed]
- Lundman, E.; Gaup, H.J.; Bakkeheim, E.; Olafsdottir, E.J.; Rootwelt, T.; Storrøsten, O.T.; Pettersen, R.D. Implementation of newborn screening for cystic fibrosis in Norway. Results from the first three years. J. Cyst. Fibros. 2016, 15, 318–324. [Google Scholar] [CrossRef] [PubMed]
- Skov, M.; Baekvad-Hansen, M.; Hougaard, D.M.; Skogstrand, K.; Lund, A.M.; Pressler, T.; Olesen, H.V.; Duno, M. Cystic fibrosis newborn screening in Denmark: Experience from the first 2 years. Pediatr. Pulmonol. 2019, 55, 549–555. [Google Scholar] [CrossRef]
- Gemeinsamer Bundesausschuss. Kinder-Richtlinie: Änderung des Beschlusses zur Neufassung—Screening auf Mukoviszidose (Zystische Fibrose)—Tragende Gründe zum Beschluss. Available online: https://www.g-ba.de/informationen/beschluesse/2316/ (accessed on 3 November 2015).
- Sommerburg, O.; Stahl, M.; Hammermann, J.; Okun, J.G.; Kulozik, A.; Hoffmann, G.; Mall, M. Neugeborenenscreening auf Mukoviszidose in Deutschland: Vergleich des neuen Screening-Protokolls mit einem Alternativprotokoll. Klin. Pädiatrie 2017, 229, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Zeyda, M.; (Medical University of Vienna, Vienna, Austria). Personal communication, 2020.
- Gartner, S.; (Tilburg School of Catholic Theology, Tilburg, The Netherlands). Personal communication, 2020.
| 2nd Tier Test | Reference | Protocol | Region/Country | n Screened | Prevalence of CF | Sensitivity (%) w/o MI | PPV (%) |
|---|---|---|---|---|---|---|---|
| ECFS standard [19] | ≥95 | ≥30 | |||||
| IRT | Calvin et al. 2012 [20] | IRT/IRT | East Anglia (UK) | 582,966 | 1:2286 | 93.8 | 67.3 |
| DNA | Calvin et al. 2012 [20] | IRT/DNA(29)/IRT | East Anglia (UK) | 147,764 | 1:2111 | 90.2 | 85.9 |
| Sommerburg et al. 2015 [15] | IRT/DNA(4)+SN | Southwest Germany | 252,020 | 1:4582 | 95.1 | 15.3 | |
| Kharrazi et al. 2015 [21] | IRT/DNA(28–40)/EGA | California | 2,573,293 | 1:6899 | 92 | 34 | |
| Sontag et al. 2016 [22] | IRT/IRT/DNA(41–48) | Colorado, Wyoming, Texas | 1,520,079 | 1:5548 | 96.2 | 19.7 | |
| Lundman et al. 2016 [23] | IRT/DNA/EGA | Norway | 181,859 | 1:8660 | 95 | 43 | |
| Skov et al. [24] | IRT/DNA(1)/EGA | Denmark | 126,338 | 1:4866 | 91.7 | 84.6 | |
| PAP | Sommerburg et al. 2015 [15] | IRT/PAP+SN | Southwest Germany and East-Saxony (Germany) | 328,176 | 1:4860 | 96.0 | 8.8 |
| Weidler et al. [14] | IRTxPAP | Southwest Germany and East-Saxony (Germany) | 410,111 | 1:5258 | 97.4 | 8.2 | |
| Marcao et al. 2018 [17] | IRT/PAP/IRT | Portugal | 255,000 | 1:7500 | 94.4 | 41.3 | |
| Dankert-Roelse et al. 2019 [16] | IRT/PAP/DNA(35)/EGA | The Netherlands | 819,879 | 1:6029 | 90 | 63 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sommerburg, O.; Hammermann, J. Pancreatitis-Associated Protein in Neonatal Screening for Cystic Fibrosis: Strengths and Weaknesses. Int. J. Neonatal Screen. 2020, 6, 28. https://doi.org/10.3390/ijns6020028
Sommerburg O, Hammermann J. Pancreatitis-Associated Protein in Neonatal Screening for Cystic Fibrosis: Strengths and Weaknesses. International Journal of Neonatal Screening. 2020; 6(2):28. https://doi.org/10.3390/ijns6020028
Chicago/Turabian StyleSommerburg, Olaf, and Jutta Hammermann. 2020. "Pancreatitis-Associated Protein in Neonatal Screening for Cystic Fibrosis: Strengths and Weaknesses" International Journal of Neonatal Screening 6, no. 2: 28. https://doi.org/10.3390/ijns6020028
APA StyleSommerburg, O., & Hammermann, J. (2020). Pancreatitis-Associated Protein in Neonatal Screening for Cystic Fibrosis: Strengths and Weaknesses. International Journal of Neonatal Screening, 6(2), 28. https://doi.org/10.3390/ijns6020028




