Newborn Screening: Current Status in Alberta, Canada
Abstract
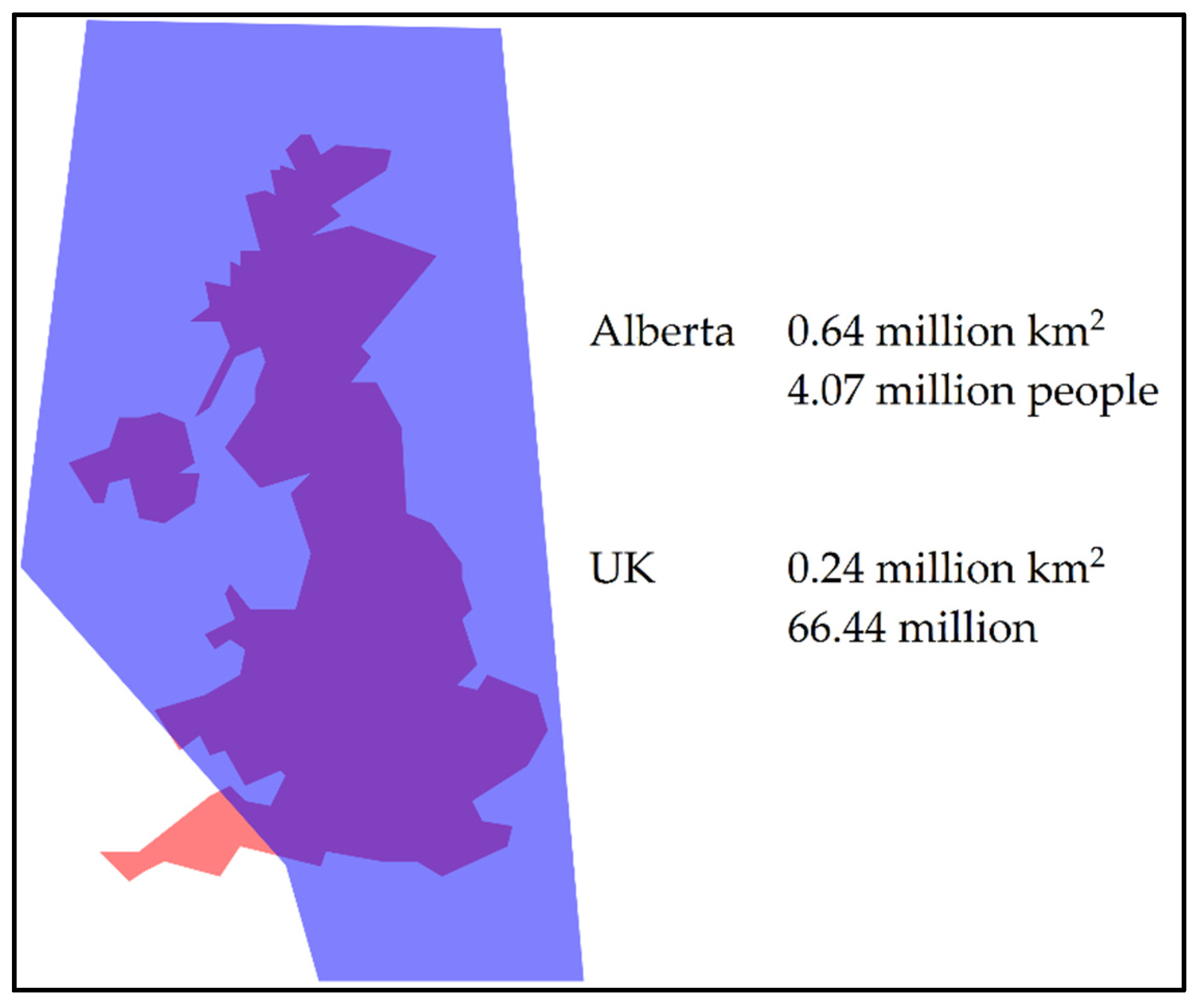
1. Introduction
2. Alberta Newborn Metabolic Screening
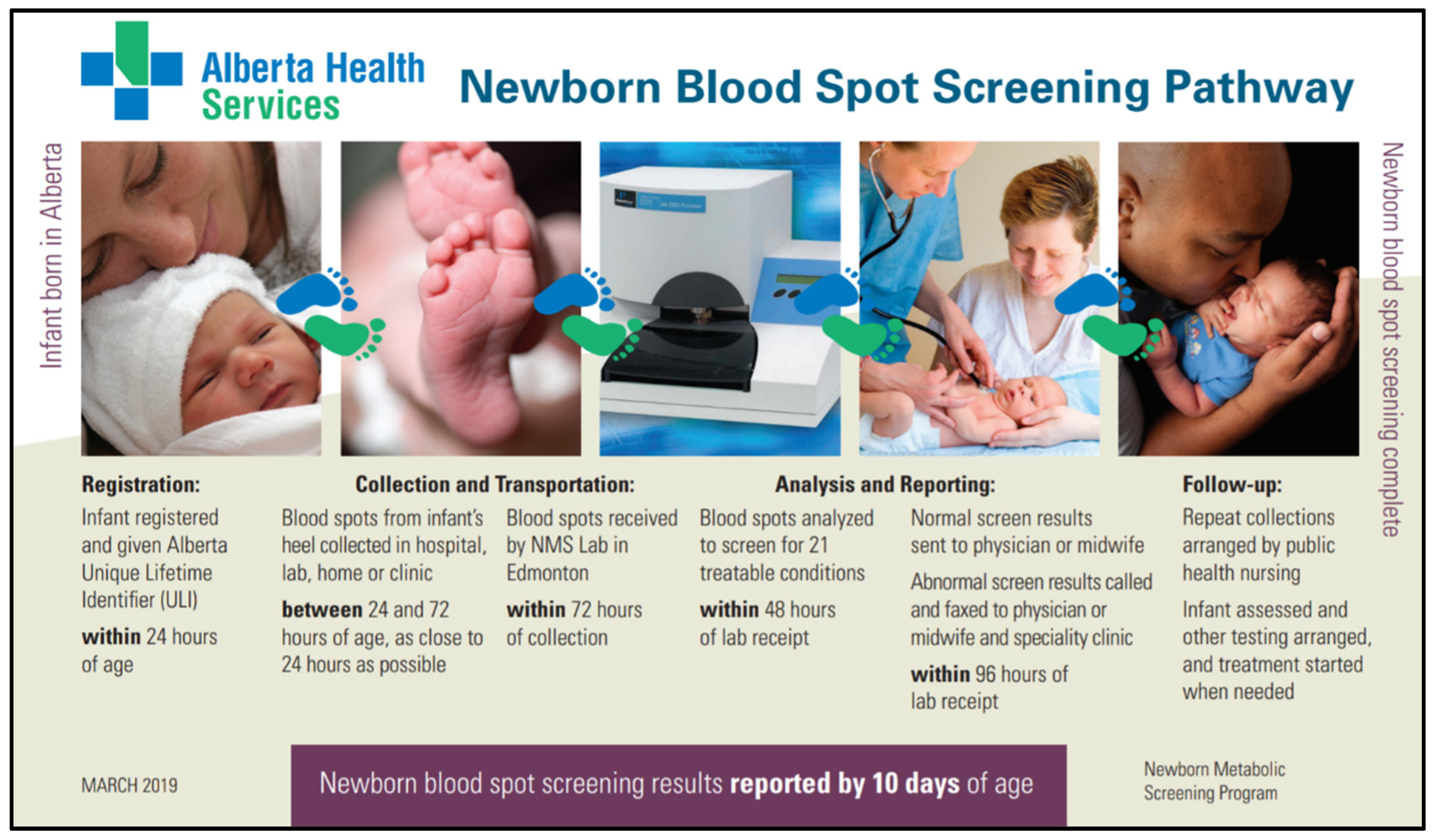
3. Screening Process
4. Alberta NMS 2019 Panel Expansion
5. Key Performance Measures and Outcomes from Alberta Newborn Screening
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Green, N.S.; Dolan, S.M.; Murray, T.H. Newborn screening: Complexities in universal genetic testing. Am. J. Public Health 2006, 96, 1955–1959. [Google Scholar] [CrossRef] [PubMed]
- Guthrie, R.; Susi, A. A simple phenylalanine method for detecting phenylketonuria in large populations of newborn infants. Pediatrics 1963, 32, 338–343. [Google Scholar] [PubMed]
- Chace, D.H.; Millington, D.S.; Terada, N.; Kahler, S.G.; Roe, C.R.; Hofman, L.F. Rapid diagnosis of phenylketonuria by quantitative analysis for phenylalanine and tyrosine in neonatal blood spots by tandem mass spectrometry. Clin. Chem. 1993, 39, 66–71. [Google Scholar] [PubMed]
- Chace, D.H.; Sherwin, J.H.; Hillman, S.L.; Lorey, F.; Cunningham, G.C. Use of phenylalanine-to-tyrosine ratio determined by tandem mass spectrometry to improve newborn screening for phenylketonuria of early discharge specimens collected in the first 24 hours. Clin. Chem. 1998, 44, 2405–2409. [Google Scholar] [PubMed]
- Millington, D.S.; Kodo, N.; Norwood, D.L.; Roe, C.R. Tandem mass spectrometry: A new method for acylcarnitine profiling with potential for neonatal screening for inborn errors of metabolism. J. Inherit. Metab. Dis. 1990, 13, 321–324. [Google Scholar] [CrossRef] [PubMed]
- Millington, D.S.; Kodo, N.; Terada, N.; Roe, D.; Chase, D.H. The analysis of diagnostic markers of genetic disorders in human blood and urine using tandem mass spectrometry with liquid secondary ion mass spectrometry. Int. J. Mass Spectrom. Ion Process. 1991, 111, 211228. [Google Scholar] [CrossRef]
- Chace, D.H.; Hillman, S.L.; Millington, D.S.; Kahler, S.G.; Roe, C.R.; Naylor, E.W. Rapid diagnosis of maple syrup urine disease in blood spots from newborns by tandem mass spectrometry. Clin. Chem. 1995, 41, 62–68. [Google Scholar] [PubMed]
- Chace, D.H.; Hillman, S.L.; Millington, D.S.; Kahler, S.G.; Adam, B.W.; Levy, H.L. Rapid diagnosis of homocystinuria and other hypermethioninemias from newborns’ blood spots by tandem mass spectrometry. Clin. Chem. 1996, 42, 349–355. [Google Scholar] [PubMed]
- Chace, D.H.; Hillman, S.L.; Van Hove, J.L.; Naylor, E.W. Rapid diagnosis of MCAD deficiency: Quantitative analysis of octanoylcarnitine and other acylcarnitines in newborn blood spots by tandem mass spectrometry. Clin. Chem. 1997, 43, 21062113. [Google Scholar]
- Bobadilla, J.L.; Macek, M., Jr.; Fine, J.P.; Farrell, P.M. Cystic fibrosis: A worldwide analysis of CFTR mutations—correlation with incidence data and application to screening. Hum. Mutat. 2002, 19, 575–606. [Google Scholar] [CrossRef] [PubMed]
- Thompson, D.C.; McPhillips, H.; Davis, R.L.; Lieu, T.L.; Homer, C.J.; Helfand, M. Universal newborn hearing screening: Summary of evidence. JAMA 2001, 286, 2000–2010. [Google Scholar] [CrossRef] [PubMed]
- Mahle, W.T.; Newburger, J.W.; Matherne, G.P.; Smith, F.C.; Hoke, T.R.; Koppel, R.; Gidding, S.S.; Beekman, R.H., 3rd; Grosse, S.D.; American Heart Association Congenital Heart Defects Committee of the Council on Cardiovascular Disease in the Young, Council on Cardiovascular Nursing, and Interdisciplinary Council on Quality of Care and Outcomes Research. Role of pulse oximetry in examining newborns for congenital heart disease: A scientific statement from the AHA and AAP. Circulation 2009, 120, 447–458. [Google Scholar] [PubMed]
- Statistics Canada. Available online: https://www.statcan.gc.ca/eng/start (accessed on 10 July 2019).
- Perinatal Services BC. Available online: http://www.perinatalservicesbc.ca/our-services/screening-programs/newborn-screening-program (accessed on 10 July 2019).
- Newborn Metabolic Screening. Available online: https://www.alberta.ca/newborn-metabolic-screening.aspx (accessed on 10 July 2019).
- Winnipeg Regional Health Authority. Available online: http://www.wrha.mb.ca/prog/genetics/newborn-screening.php (accessed on 10 July 2019).
- Dery, M.C. (Shared Health Manitoba, Winnipeg, Manitoba, Canada). Personal communication, 2019.
- Saskatchewan Health Authority. Available online: https://www.saskhealthauthority.ca/Services-Locations/RRPL (accessed on 10 July 2019).
- Newborn Screening Ontario. Available online: https://www.newbornscreening.on.ca/en (accessed on 10 July 2019).
- Neonatal Blood and Urine Screening Program –Services Québec. Available online: http://www4.gouv.qc.ca/EN/Portail/Citoyens/Evenements/DevenirParent/Pages/progr_depst_neont_sangn.aspx (accessed on 10 July 2019).
- Maritime Newborn Screening Program. Available online: http://www.iwk.nshealth.ca/newbornscreening (accessed on 10 July 2019).
- Newfoundland and Labrador Medical Association. Available online: http://www.nlma.nl.ca/News-And-Events/Notices-And-Advisories/Page/0/Article/238 (accessed on 10 July 2019).
- Office for National Statistics. Available online: https://www.ons.gov.uk/ (accessed on 10 July 2019).
- MapFight. Available online: https://mapfight.appspot.com/ (accessed on 18 July 2017).
- Lilley, M.; Christian, S.; Hume, S.; Scott, P.; Montgomery, M.; Semple, L.; Zuberbuhler, P.; Tabak, J.; Bamforth, F.; Somerville, M.J. Newborn screening for cystic fibrosis in Alberta: Two years of experience. Paediatr. Child Health 2010, 15, 590–594. [Google Scholar] [CrossRef] [PubMed]
- Beutler, E.; Baluda, M.C. A simple spot screening test for galactosemia. J. Lab. Clin. Med. 1966, 68, 137–141. [Google Scholar] [PubMed]
- Stuhrman, G.; Perez Juanazo, S.J.; Crivelly, K.; Smith, J.; Anderson, H.; Morava, E. False-positive newborn screen using the Beutler spot assay for galactosemia in glucose-6-phosphate dehydrogenase deficiency. JMID Rep. 2017, 36, 1–5. [Google Scholar]
- Baker, M.W.; Grossman, W.J.; Laessig, R.H.; Hoffman, G.L.; Brokopp, C.D.; Kurtycz, D.F.; Cogley, M.F.; Litsheim, T.J.; Katcher, M.L.; Routes, J.M. Development of a routine newborn screening protocol for severe combined immunodeficiency. J. Allergy Clin. Immunol. 2009, 124, 522–527. [Google Scholar] [CrossRef] [PubMed]
- Therrell, B.L.; Padilla, C.D.; Loeber, J.G.; Kneisser, I.; Saadallah, A.; Borrajo, G.J.C.; Adams, J. Current status of newborn screening worldwide: 2015. Semin. Perinatol. 2015, 39, 171–187. [Google Scholar]

| Classification | Disorders | Screening Initiated | Assay and Instrumentation |
|---|---|---|---|
| Metabolic Disorders—Organic Acid | GA1 | 2007 | PerkinElmer NeoBase™ Non-derivatized MSMS kit PerkinElmer NeoBase™ Succinylacetone Assay Solution Waters™ ACQUITY UPLC™ with XEVO™ TQD |
| HMG deficiency | 2007 | ||
| IVA | 2007 | ||
| MMA | 2007 | ||
| PA | 2007 | ||
| Metabolic Disorders—Fatty Acid Oxidation | CUD | 2007 | |
| LCHAD deficiency | 2007 | ||
| MCAD deficiency | 2007 | ||
| TFP deficiency | 2007 | ||
| VLCAD deficiency | 2007 | ||
| Metabolic Disorders—Amino Acid | CIT | 2007 | |
| MSUD | 2007 | ||
| PKU | 1967 | ||
| TYR1 | 2019 | ||
| Metabolic Disorders—Other | BIOT | 1990 | PerkinElmer Neonatal Biotinidase kit PerkinElmer VICTOR2 ™D Instrument |
| GALT | 2019 | PerkinElmer GSP® Neonatal GALT kit PerkinElmer GSP® Neonatal Total Galactose kit PerkinElmer GSP® Instrument | |
| Endocrine Disorders | CAH | 2007 | PerkinElmer AutoDELFIA® Neonatal 17α-OH-progesterone kit PerkinElmer AutoDELFIA® Instrument |
| CH | 1977 | PerkinElmer AutoDELFIA® Neonatal hTSH kit PerkinElmer AutoDELFIA® Instrument | |
| Other Disorders | CF | 2007 | PerkinElmer AutoDELFIA® Neonatal IRT kit PerkinElmer AutoDELFIA® Instrument Luminex® xTAG® CF39v2 Kit Luminex® 200™ |
| SCID | 2019 | Laboratory developed test Applied Biosystems™ QuantStudio™ 12K Flex Real-Time PCR System | |
| SCD | 2019 | Bio-Rad VARIANT™ Sickle Cell Program kit Bio-Rad VARIANT™ nbs Newborn Screening System |
| Inadequate Sample Alert | Number | |
|---|---|---|
| Less than 24 h | 172 | (16.57%) |
| Blood clotted and layered | 161 | (15.51%) |
| Date or time of birth illegible or not indicated | 2 | (0.19%) |
| Date or time of collection illegible or not indicated | 97 | (9.34%) |
| Pre-expansion requisition | 12 | (1.16%) |
| Query patient identification | 32 | (3.08%) |
| Requisition expired | 15 | (1.45%) |
| Sample contaminated | 186 | (17.92%) |
| Sample insufficient | 206 | (19.85%) |
| Sample stability exceeded | 1 | (0.10%) |
| Specimen abraded | 154 | (14.84%) |
| Total | 1038 | |
| Performance | 2018–2019 | 2017–2018 | 2016–2017 |
|---|---|---|---|
| Registered infants who received an initial blood spot screen 1 | 99.41% (52,005/52,313) | 99.40% (52,898/53,215) | 99.46% (54,891/55,190) |
| Registered infants who did not receive an initial blood spot screen | 0.59% (308/52,313) 57.47% of infants not screened were due to neonatal death (177/308) | 0.60% (317/53,215) 59.62% of infants not screened were due to neonatal death (189/317) | 0.54% (299/55,190) 57.19% of infants not screened were due to neonatal death (171/299) |
| Registered screened infants who had a screen result reported within 10 days of age | 98.74% (51,351/52,005) | 99.15% (52,449/52,898) | 99.22% (52,465/54,891) |
| Screened infants who received normal screen results 2 | 99.50% (51,837/52,099) | 99.42% (52,649/52,958) | 99.36% (54,608/54,960) |
| Screened infants who received abnormal screen results requiring clinical follow-up | 0.36% (188/52,099) (162 critical results and 26 double borderline results) | 0.44% (231/52,958) (200 critical results and 31 double borderline results) | 0.49% (271/54,960) (250 critical results and 21 double borderline results) |
| Screened infants who received unknown screen results 3 | 0.14% (74/52,099) | 0.15% (78/52,958) | 0.15% (81/54,960) |
| Infants with abnormal screen result who received abnormal diagnostic outcomes | 29.26% (55/188) | 32.47% (75/231) | 27.31% (74/271) |
| Infants with abnormal screen result who received likely to be normal diagnostic outcomes | 55.85% (105/188) | 61.47% (142/231) | 68.63% (186/271) |
| Infants with abnormal screen result who received unclear diagnostic outcomes 4 | 1.60% 5 (3/188) | 0.87% (2/231) | 1.85% (5/271) |
| Infants with abnormal screen result who received unknown diagnostic outcomes 6 | 6.91% (13/188) | 5.19% (12/231) | 2.21% (6/271) |
| Infants with abnormal screen result with pending diagnostic outcomes | 6.38% (12/188) | 0.00% (0/231) | 0.00% (0/271) |
| Classification | Abnormal Screen Results | Abnormal Diagnostic Outcomes |
|---|---|---|
| Metabolic Disorders | 44 | BIOT (4), CIT (1), GA1 (1), HMG deficiency (1), LCHAD/TFP deficiency (1), MCAD deficiency (5), MSUD (1), PKU (1) |
| Endocrine Disorders | 42 | CAH (2), CH (28) |
| Other Disorders | 102 | CF (10) |
| Total | 188 | 55 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Souza, A.; Wolan, V.; Battochio, A.; Christian, S.; Hume, S.; Johner, G.; Lilley, M.; Ridsdale, R.; Schnabl, K.; Tran, C.; et al. Newborn Screening: Current Status in Alberta, Canada. Int. J. Neonatal Screen. 2019, 5, 37. https://doi.org/10.3390/ijns5040037
De Souza A, Wolan V, Battochio A, Christian S, Hume S, Johner G, Lilley M, Ridsdale R, Schnabl K, Tran C, et al. Newborn Screening: Current Status in Alberta, Canada. International Journal of Neonatal Screening. 2019; 5(4):37. https://doi.org/10.3390/ijns5040037
Chicago/Turabian StyleDe Souza, Andy, Vanessa Wolan, Angie Battochio, Susan Christian, Stacey Hume, Grace Johner, Margaret Lilley, Ross Ridsdale, Kareena Schnabl, Chi Tran, and et al. 2019. "Newborn Screening: Current Status in Alberta, Canada" International Journal of Neonatal Screening 5, no. 4: 37. https://doi.org/10.3390/ijns5040037
APA StyleDe Souza, A., Wolan, V., Battochio, A., Christian, S., Hume, S., Johner, G., Lilley, M., Ridsdale, R., Schnabl, K., Tran, C., Yuen-Jung, J., & Sosova, I. (2019). Newborn Screening: Current Status in Alberta, Canada. International Journal of Neonatal Screening, 5(4), 37. https://doi.org/10.3390/ijns5040037




