Current Status of Newborn Bloodspot Screening Worldwide 2024: A Comprehensive Review of Recent Activities (2020–2023)
Abstract
1. Introduction
2. Methodology
3. Results
3.1. North America
3.1.1. National NBS Activities Involving Multi-State and Multi-Provincial Collaborations
Multi-Jurisdictional NBS Activities Related to RUSP Expansion (2016–2023)
Multi-Jurisdictional NBS Activities Related to the “Original” RUSP
Multi-Jurisdictional Activities Related to Conditions Not Yet on the RUSP
Other Multi-Jurisdictional NBS Activities
NBS Regional Activities Focused on the Future
3.1.2. NBS Activities within Jurisdictions (State and Provinces)
State Activities (USA)
- Alabama—The Alabama NBS program is one of about 12 states that require a second screen on all babies at 2–6 weeks of age. In March 2023, ALD and adenosine deaminase deficiency (ADA) were added to the screening panel [191].
- Alaska—NBS in Alaska includes laboratory testing by the Iowa NBS laboratory. NBS research sometimes focuses on the CPT-1A Arctic variant, a secondary condition on the RUSP (Table 3) that is highly prevalent among the Indigenous Arctic peoples of Alaska. A recent focus group study in two tribal settings showed opportunities for additional education for healthcare professionals and caregivers, improved educational outreach, and guidance for the care of infants and children who might have the condition [192].
- Arizona—A second NBS is required on all Arizona newborns, although some tests (CF, SCID, SMA, ALD, MPS-I, and PD) may not be run on the second specimen if the first screen is normal. Data from the first two years of NBS for SCID confirmed an incidence 2.5 times above the national average. This likely reflects Arizona’s unique population profile, which includes a higher percentage of Native Americans and an increased percentage of Hispanic/Latinos compared to the general US population [193].
- Arkansas—Consistent with the national push to make state screening mandates responsive to the RUSP and its changes, the Arkansas State Board of Health unanimously approved rules that add conditions to the screening panel as they are approved for the RUSP in July 2023, with the caveat that screening implementation must occur within 36 months following an RUSP addition [194].
- California—The California (CA) NBS program reaches the largest jurisdiction of newborns in the USA. As such, it can provide faster reviews of new conditions under consideration for implementation in state NBS programs. This can assist other programs in their decisions regarding screening methods and related screening issues. An extensive, carefully maintained, temperature-controlled biobank of NBS specimens exists, which is available for research under certain conditions. Several research initiatives have been recently reported that involved improvements in screening laboratory methodology: a comparative effectiveness study of three methods (MS/MS, digital microfluidics, and immunocapture) for detecting four LSDs (Fabry disease, Gaucher disease, PD, MPS-1), which demonstrated that all techniques had high sensitivity and that use of CLIR software tools markedly improved the performance of each [79]; the NBSeq project, using archived residual dried bloodspots (DBSs) and data from 4.5 million California newborns, showed that whole exome sequencing (WES) could be useful as a second-tier test for MS/MS, reducing false-positive results, facilitating timely case resolution, and occasionally suggesting a more appropriate or specific diagnosis [175]; a study showing that a Random Forest machine learning classifier could be trained with screening data to improve prediction of true and false-positive screens for 39 MS/MS metabolic analytes [195]; and a machine learning study that improved screening and demonstrated the utility of supervised machine learning in reducing false-positive screens for an expanded metabolite panel (with second-tier testing of screen-positive cases) of four disorders [GA-1, MMA, ornithine transcarbamylase deficiency (OTCD), and VLCAD] [196].Reports of the results of screening for various conditions have proven useful in decision making in other states. For example, a 2021 review of SCID NBS acknowledged the importance of CA NBS results (3.25 million newborns) in demonstrating the effectiveness of early identification of both SCID NBS and non-SCID T-cell lymphopenia [197]. NBS in CA for other conditions has also proven to be important: the results of screening 1.8 million newborns for ALD in CA during the first four years of NBS (16 February 2016–15 February 2020) [198]; the use of CLIR software tools in affecting the rapid resolution of post-NBS confirmatory testing for ALD [199]; first year results of NBS for PD using a two-tier screening process on 453,152 newborns validated the algorithm and showed that African American and Asian/Pacific Islanders had higher allele frequencies for both pathogenic and pseudodeficiency variants [200]; results of clinical and epidemiological outcomes for the first 18 months of NBS for SMA on 628,791 newborns (including assay specificity and sensitivity) [201]; and a report on the first three years of screening for MPS-I using a two-tiered approach (α-L-iduronidase (IDUA) enzyme activity followed by DNA sequencing for IDUA gene variants) on 1295,515 newborns [202].
- Colorado—The Colorado NBS program requires a second specimen on all newborns between 8–14 days of age and provides laboratory testing for the neighboring state of Wyoming. Recently, the NBS laboratory relocated into a new laboratory space, which includes a larger area for MS/MS and a dedicated molecular suite allowing for easier integration of testing for new disorders.State NBS programs are increasingly responsive to the development of novel therapeutics and political advocacy, and there is increasing oversight and support for harmonization at the federal level. An increased number of conditions screened, increased false-positive screens, and national program improvement initiatives are creating new and increased scrutiny of state NBS systems, potentially posing an existential risk to the public’s acceptance of mandatory NBS. A recent report from Colorado reviews NBS history, current state, and program challenges with suggestions for overcoming some of the challenges [42]. Seeking to continuously evaluate and improve the Colorado NBS program, a review of late-diagnosed CF cases over a five-year period (358,000 newborns—IRT/IRT/DNA protocol) showed a need to change from a fixed IRT screening cutoff (50 ng/mL) to a floating cutoff (96th percentile or 50 ng/mL, whichever is lower) and the importance of continuous quality improvement in preventing late diagnoses [206].
- Connecticut—The Connecticut Newborn Screening Network, established at Connecticut Children’s Medical Center in collaboration with Yale New Haven Hospital in 2018, is a statewide network responding to all reports of out-of-range NBS results. The Network coordinates the diagnostic work-up with the newborn’s family and physician, arranging initial treatment and LTFU care with the appropriate specialty care team if a disorder is identified. The Epic electronic health record (EHR) system (Verona, WI, USA) has been used to establish registries for LTFU tracking, serving as a use case for applying and achieving the adoption of population health tools within the EHR system to track care delivery and quickly fill identified care gaps. Expansion of the LTFU registry to all NBS specialty care teams is underway [207].
- Delaware—Delaware was one of the first states to implement NBS in the early 1960s [3]. In 2018, the NBS program partnered with Nemours Hospital for Children to help manage the screening program. Screening laboratory services were discontinued at the Delaware Department of Health and Social Services and contracted to a private laboratory. In 2022, the Legislature formalized the Newborn Screening Advisory Committee to advise the Director of the Division of Public Health and discontinued the requirement for a second NBS specimen [208].
- District of Columbia (DC)—The DC NBS program uses a private laboratory for NBS and is one of two programs routinely offering NBS for G6PD (Pennsylvania is the other). A recent study of the experiences with MPS-I screening from December 2017–February 2019, reviews the “impact of overrepresentation of screen positives in a minority group and unintentional creation of health disparities and community wariness regarding medical genetics evaluations” and advocates for “MPS I secondary testing … in conjunction with DBS-IDUA activity levels on all infants as part of the initial newborn screen, regardless of race, ethnicity, or ability to pay.” [209].
- Florida—The Florida NBS program began screening for MPS-I and PD in February 2020, SMA in April 2020, MPS-II was approved for addition in 2022 and GAMT in 2023. Florida law requires the addition of conditions added to the RUSP within one year [210]. Genetic counseling via telemedicine is the subject of a 2023 report from one of the Florida follow-up centers. This center was able to demonstrate that genetic counseling for CF via telemedicine was feasible across a variety of distinct geographic locations with diverse socioeconomic status throughout the United States [211].
- Georgia—The Georgia NBS program is a collaboration between the Department of Health for testing and program administration and Emory University for follow-up and treatment/management. A study of 52 SCAD and 9 IBDD cases detected by NBS between 2007–2016 revealed no major health problems overall, no significant difference in cognitive development, and only a slightly higher incidence of reported neonatal hypoglycemia in the SCAD group. As a result, the anxiety reported by parents may be unwarranted and the study authors suggest that these conditions should be considered for removal from NBS panels [212]. As part of NBS follow-up, the Georgia program uses CLIR post-analytical tools to triage abnormal results by assigning each case a risk level, which is used to guide follow-up recommendations. Based on experience, the follow-up program has reportedly moved forward in a more prospective manner with CLIR tools to reduce the number of cases needing follow-up [213]. The efficacy of using CLIR tools were also evaluated for the detection proximal urea cycle disorders (PUCD) in the Georgia NBS program. A high number of false-positives regardless of cutoff adjustments led the authors to conclude that PUCD is not suitable for statewide NBS using their procedure and suggested that a method that separates glutamine from other amino acids may work better [214].Experiences with ALD, PD, MPS I, and SMA have also been reported. The ALD pilot used a two-tiered strategy to quantify very long-chain lysophosphatidylcholines (LPCs) using flow-injection MS/MS (FIA-MS/MS) as an initial screen and LC MS/MS as a second-tier screen [215]. Pilot screening for PD and MPS I involved a two-tier strategy of FIA-MSMS enzyme assays. While false-positive screening results were reduced for PD, the frequency of pseudodeficiency was problematic with MPS-I, suggesting that LC-MS/MS analysis of dermatan and heparan sulfate might be a better alternative [216]. For SMA, a 2022 report detailed findings from a year of pilot screening (February 2019–February 2020) and the first year of standard screening (February 2020–February 2021) using real-time PCR assays. In addition to assay performance data, the report included disease incidence, time to diagnosis and treatment, and early clinical outcomes [217].
- Hawai’i—Since October 2019, NBS specimens on Hawai’i newborns have been tested at the Washington State NBS laboratory, which provides testing services on weekends allowing for more timely testing. On 3 July 2023, both PD and MPS-I were added to the Hawai’i NBS panel and on 1 January 2024, SMA and ALD were added [218].
- Idaho—The Idaho NBS program requires a second screen at 10–15 days of age. Since April 2021, Idaho NBS specimens have been analyzed by the Washington State NBS laboratory. On 1 February 2022, four new conditions were added to the Idaho NBS screening panel—PD, MPS-I, ALD, and SMA [219]. In addition to the NBS success story shared in the Block Grant Executive Summary (formal request for federal funds), Idaho NBS Program staff joined with the Idaho Medicaid staff to successfully remove prior insurance authorization restrictions for diagnostic testing and treatment for NBS conditions that prevented access to early diagnosis and treatment [220]. NBS staff were also successful in meeting with and educating the Director of the Department of Health and Welfare about NBS and getting the Governor to recognize September as “Newborn Screening Awareness Month,” which recognizes the program’s importance and increases awareness among prospective parents [221].
- Illinois—The Illinois Legislature has taken an aggressive approach towards NBS in recent years with Illinois becoming one of the first states to require screening for LSDs. In 2023, Illinois became the first state to require NBS for metachromatic leukodystrophy (MLD) and the Illinois Department of Public Health NBS laboratory is in the early stages of preparation for implementation in 2024 [222]. As a result of the earlier screening in Illinois, publications of Illinois experiences have provided valuable information for other state programs: a report on screening experiences with 684,290 Illinois newborns for PD [223]; a report of the incorporation of psychosine measurements in KD screening to identify newborns with infantile KD and infants with probable late-onset KD [224]; a report on the successful implementation of ALD screening in Illinois reviewing the screening experiences with 276,000 newborns using LC MS/MS [225]; an update on measuring iduronate-2-sulfatase (I2S) activity to detect MPS-II in 339,269 newborns [226]; and a further update on in 586,323 newborns screened for MPS II with the suggestion that, based on Illinois data, MPS-II may be more common than previously recognized with a higher prevalence of attenuated cases [227].
- Indiana—In 2020, the Indiana Legislature passed, and the Governor signed, a law expanding NBS to include PD. This law went into effect on 1 July 2020 and included KD and MPS-I [228]. With an eye towards the future and genomic sequencing, the Rady Children’s Institute for Genomic Medicine is involved in an investigatory project called BeginNGS®, which is an international, pre-competitive, public-private consortium that proposes to implement a self-learning healthcare delivery system for screening all newborns for over four hundred genetic diseases, diagnostic confirmation, implementation of effective treatment, and acceleration of orphan drug development [229,230].
- Iowa—The Iowa NBS laboratory provides laboratory services for Alaska and both North and South Dakota. As with eight other states at the time, the Governor signed legislation in April 2022, that requires the Iowa NBS panel to harmonize with the USA RUSP as new conditions are added/subtracted. Implementation is required within two-and-a-half years of addition with an annual status report of which new conditions were added to the panel and if/why implementation might have been delayed [231]. SMA was added to the screening panel on 13 September 2021 [232], and PD and MPS-I were added on 15 May 2023 [233]. In a letter to the public, the first fee increase for NBS since 2014 was introduced (USD 122 to USD 162) covering increased testing and courier services, among others [234]. A recent review of HGB screening data in Iowa documented the birth prevalence of hemoglobinopathies in Iowa and highlighted the need for periodic outcome evaluation to ensure that the healthcare needs of underserved, minority populations are met [235]. A summary of SCD data for each state from 1 January 1991 through 31 December 2010 has also been published [236].
- Kentucky—A report in 2021 described two late-diagnosed cases of HCY following normal NBS results despite the use of CLIR post-analytical tools as part of the screening algorithm. The performance of methionine and homocysteine analyses as part of the screening process for HCY suggests that a change to NGS might be useful [239].
- Louisiana—In January 2022, the Louisiana Department of Health added NBS for SMA, MPS-I, and PD. Most NBS-related research focuses on clinical applications, which is beyond the scope of this report [240].
- Maine—The Maine NBS program added PD, ALD, MPS-I, and SMA to the screening panel, effective 1 April 2021 [241]. Laboratory testing services are provided by the Massachusetts NBS laboratory as part of the New England NBS collaboration. A 2022 law requires development of a CMV screening program though rulemaking that includes a gradual expansion from a targeted program to universal testing using either urine or saliva and currently does not affect the bloodspot NBS program [242].
- Maryland—The Maryland NBS program strongly recommends a second specimen between 7–14 days of age with a high success rate, and thus it is considered a two-screen state. Consent for NBS, which was required prior to 2008 is no longer required [243]. Maryland became the third state to pass RUSP alignment legislation in 2023 (10th overall) requiring harmonization with the national RUSP. The law requires implementation of a new condition within two years of its addition to the RUSP [244].
- Massachusetts—The New England Newborn Screening Program (NENSP) provides NBS laboratory services for five New England states (Maine, Massachusetts, New Hampshire, Rhode Island, and Vermont) and is part of the University of Massachusetts Chan Medical School. There are two types of NBS in Massachusetts, “required” and “voluntary” (pilot). “Required” NBS is conducted for conditions with known effective treatments and all newborns must be screened for these conditions unless there is a religious objection. “Voluntary” NBS allows for the study of new NBS conditions and is performed at no additional charge [245]. This NBS system has allowed the Massachusetts NBS program to be one of the USA leaders in contributions to NBS research. As an example, SMA analysis originally included a tiered algorithm looking for the absence of SMN1 Exon 7. When results from the first and second tier needed reconciliation, a third tier DNA sequencing assay was developed that confirms SMN2 copy numbers without the need for additional instrumentation [246]. A program report on screening 179,467 newborns confirmed the value of the screening algorithm in detecting SMA-affected infants who show absent SMN1 Exon 7 by Real-Time™ quantitative PCR (qPCR) [247].Results from 10 years of NBS for SCID in Massachusetts support using a single NBS testing-and-referral algorithm for all gestational ages, despite lower median TREC values in premature infants, and that low naïve T-cell percentages are associated with a higher risk of SCID/CID and support the utility of memory/naïve T-cell phenotyping as part of follow-up flow cytometry [248]. As part of NBS research activities during the COVID-19 pandemic, deidentified NBS specimens were used as a source of maternal antibodies to estimate the cumulative incidence of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) to assist in setting public health policies [249]. Additionally, editorials addressing research questions pertinent to NBS have been introduced by Massachusetts researchers: (1) whether NBS for vitamin B12 deficiency be incorporated into all NBS programs via a second-tier homocysteine assay that would increase the vitamin B12 deficiency cases identified and decrease the number of missed (false negative) cases of HCY [250]; and (2) whether more effective NBS for HCY is on the horizon using MS/MS with selective thiol derivatization [251].
- Michigan—The NBS laboratory in Lansing stores a residual NBS specimen on each newborn screened, while five more are sent to the Michigan Neonatal Biobank in Detroit for storage under climate-controlled conditions and possible research use. Since 2010, consent has been required for specimen storage and possible research use. In 2022, the Michigan NBS program agreed to destroy 3.4 million dried bloodspots as part of an ongoing lawsuit on privacy and consent [252]. Currently, legal issues continue with both sides obtaining wins and losses in the courts and continuing appeals.Michigan researchers have recently published reviews discussing both NBS for SCID [58] and NBS generally [253]. A cost-effectiveness and outcomes study for PKU was reported in 2021 based on new treatments and other new data [254]. At least four reports in 2022 addressed various issues pertinent to NBS: a report providing initial evidence of the value of screening for adenosine deaminase deficiency (ADA) using MS/MS [255]; a report showing the benign nature of transcobalamin receptor deficiency and the assertion that its detection should be considered an incidental finding with the caveat that long-term data are needed to ascertain the long-term outcomes of identified children [256]; a report on online NBS education for expectant parents showing that virtual baby fairs are cost effective, convenient, and equitable [257]; and an evaluation of national data currently maintained in a federally-supported database (NewSTEPs) that showed, “… potential usefulness of NewSTEPs for research if investments in higher-quality data are made” [258]. Physician education was the subject of a 2023 report that used case-based scenarios to review SCID NBS results, the principles of TREC-based NBS, the genetics and subtypes of SCID, and patient management for a positive TREC-based screening result [69].
- Minnesota—Press releases in February and August 2023 announced the addition of cCMV and a group of three other conditions (GAMT, MPS-II, and KD) to the Minnesota NBS panel. While the latter group is currently in the implementation phase, the Minnesota NBS program became the first in the nation to implement universal bloodspot screening for cCMV on 6 February 2023 [259]. This builds on earlier target NBS for cCMV in Utah, universal NBS in Ontario, Canada, and a Minnesota study that demonstrated sufficiently high analytical sensitivity and low cost for PCR for DNA from DBSs [260]. A study of urine collected on filter paper showed high sensitivity and specificity, but the collection method requires further study [261]. A 2023 report describing different newborn cCMV NBS approaches makes the case that implementing universal cCMV NBS rather than a targeted screening approach improves cCMV detection, ultimately leading to better health outcomes [262]. A recent report from the Minnesota program considering droplet digital PCR (ddPCR) as a primary DBS screening tool found that ddPCR does not demonstrate any enhancement in sensitivity compared to the use of real-time PCR testing of DNA from DBSs [263].
- Mississippi—The Mississippi Legislature became the first to adopt RUSP alignment legislation in 2022 and the eighth state to implement such legislation overall. In addition to requiring harmonization with the RUSP as new conditions are added/deleted, the legislation requires implementation within 3 years, and a status report every 12 months if the addition of a condition is delayed [264].
- Missouri—The Missouri NBS program was the first USA NBS program to require screening for SMA in 2017 and has been one of the most active programs considering, evaluating, and using the microfluidics assays for various LSDs and one of the earlier state programs implementing LSD NBS (in particular, screening for PD, MPS-I, Fabry disease, Gaucher disease, and KD). As such, its experiences have been helpful for other state programs beginning to implement LSD NBS. In 2020, a report on lessons learned from 6 years of PD NBS noted that the rates of later-onset PD phenotypes and pseudodeficiency alleles were higher than initially anticipated and should be considered during PD NBS implementation [265]. Another 2020 report detailed the analytical and clinical validations of a micro-well fluorometric assay measuring of iduronate-2-sulfatase (IDS) for MPS-II, which was implemented for full population screening after its validation [266]. A report from a Missouri tertiary care center in 2021 described the immunological findings and clinical outcomes of newborns who screened positive for SCID [267].
- Montana—In 2021, the Montana Legislature passed legislation requiring the Department of Public Health and Human Services to set up a Newborn Screening Advisory Committee to impartially hear testimony and approve or deny, by a majority vote, conditions nominated for inclusion on the NBS panel. The Committee was formalized in 2022 and began discussions of adding ALD. At its October 2023 meeting, the addition of ALD was approved [268].
- Nebraska—NBS in Nebraska is compulsory and no exemptions are allowed, including for religious reasons. NBS laboratory testing is through a contract with Revvity (Pittsburgh, PA, USA). A 2023 report outlines the experiences and challenges of implementing NBS for ALD in Nebraska including a lack of genotype-phenotype correlations, absence of predictive biomarkers for childhood cerebral ALD or adrenomyeloneuropathy, and a high proportion of ABCD1 variants of uncertain significance combining to cause unique counseling difficulties [269].
- Nevada—The Nevada NBS program requires a second NBS specimen on all newborns collected at 10–14 days of age. Screening for SMA began 21 December 2023 [270].
- New Hampshire—New Hampshire obtains NBS laboratory services from the NENSP in Massachusetts. Three conditions were added to the New Hampshire NBS panel (MPS-I, ALD, PD) with screening starting on 26 August 2020. The screening fee increased from USD 104 to USD 146 [271]. The NENSP expects to begin screening for MPS-II in August 2024 and GAMT around January 2025, which will impact decisions by the New Hampshire NBS Advisory Committee [272].
- New Jersey—The New Jersey NBS program has found itself involved in lawsuits concerning the use of residual dried bloodspots. The first involved the release of information about police use of bloodspots [273,274] and the second (filed 2 November 2023) concerns the storage of residual bloodspots [275]. Both cases involve complexities outside of the scope of this report. Also in 2023, a report on a series of five cases diagnosed late with negative NBS results led to quality improvement in the New Jersey CF screening laboratory algorithm [276]. A new law requires NBS for cCMV contingent on the development of a suitable bloodspot cCMV test, addition to the RUSP, and acquisition of the necessary equipment to conduct the test, among others [277].
- New Mexico—The New Mexico NBS program requires a second screening specimen for all newborns 10–14 days after birth. Specimen testing is performed at the Oregon NBS laboratory, which began screening for Fabry disease and Gaucher disease in New Mexico on 1 July 2022 [278]. A recent meeting of the Northwest Regional Newborn Bloodspot Screening Program Advisory Board, which advises the Oregon laboratory, considered whether to remove Fabry and Gaucher diseases from the screening panel and a detailed report of current evidence was prepared for its consideration. The decision was not to remove either condition and the Oregon NBS laboratory reported that it would be able to continue testing for New Mexico even if discontinued in Oregon [279].
- New York—New York has the third largest newborn population in the country and significant research efforts exist, often in collaboration with other programs as noted in Section 3.1.1. Currently, screening activities are focused on screening for cCMV which began as a 1-year pilot on 2 October 2023 [280] and Duchenne muscular dystrophy (DMD), which was officially added to the screening panel on 25 October 2023, for implementation in early 2024 [281]. In the latter case, implementation builds on a prospective consented pilot begun in 2019 that was aimed at addition of DMD to the RUSP and was developed in collaboration with expert partners interested in muscular dystrophies and NBS (Duchenne NBS Consortium). The 2-year pilot allowed for validation of the testing protocol and follow-up procedures and demonstrated the feasibility of NBS for DMD through successful case detections [282]. The results demonstrated the value of NBS for DMD [283]. During the validation, several factors affecting variability of creatine kinase-MM (CK-MM) levels were studied. The most prominent was an inverse relationship between the CK-MM concentration and age of collection. This information was used to implement age-related cutoffs for CK-MM [284]. Addressing the need for external quality control materials, in collaboration with the CDC and RTI International, prototype dried bloodspot quality control materials for CK-MM NBS assays were also evaluated [142].A number of other NBS activities have been ongoing since 2020, including experiences during the COVID-19 pandemic, experiences that led to development of new recruitment strategies for pilot studies, which may allow other research studies to adapt novel and more effective recruitment methods [285]. At least four NBS reports were published by New York researchers in 2020: a review of the impact of using CLIR post-analytical tools on NBS for KD (referrals reduced by ~80%) and PD (referrals reduced by ~32%) [286]; a report on the first year of SMA screening in New York (225,093 newborns screened) produced an incidence of SMA 2.6- to 4.7-fold lower than expected [287]; a review describing the current knowledge of the pathophysiology of ALD and updates on NBS, diagnosis, monitoring, and treatment [288]; and a retrospective analysis of data from a SCID Referral Center demonstrating that TREC value can be used to stratify infants for further confirmatory testing [289]. Reports of at least three NBS program activities were reported in 2021: results of a 10-year study on CH explored the effect of manufacturer’s lot changes on the daily mean values of thyroxine (T4) and the influences of seasonal variations on values for both T4 and thyrotropin (TSH), examining the effect of both on screening positive predictive values (PPVs) [290]; details of a modification to the CF NBS algorithm using a new custom NGS platform for more comprehensive CFTR gene analysis improving the PPV of CF NBS from 4% to 25% [291]; and a report on the first two cases of GAMT identified by NBS in New York and Utah [103]. A 2022 report describes a modification to the screening algorithm for GAMT eliminating the need for second-tier screening and reducing referrals for follow-up by 85% [292]. A review of the clinical outcomes from the first three years of SMA NBS was also reported in 2022 [293] along with and a description of potential improvements in the pre-symptomatic detection of MPS-I, PD, and KD using tools based on bivariate normal limits [85]. A 2023 report outlines the basic steps in organizing universal CF NBS with the evolution of CF NBS in New York as a guide, and discusses how to reduce bias, highlights challenges, offers guidance and recommendations for future consideration [294].The New York NBS program is currently actively participating in two consented pilot studies, one exploring the use of first-tier genome sequencing for highly penetrant early onset conditions. This study, Genomic Uniform-screening Against Rare Diseases in All Newborns (GUARDIAN), is screening 100,000 newborns for 250 medical conditions not currently screened by New York state [295]. The second, ScreenPlus, is a consented pilot study screening for 14 diseases (mostly lysosomal disorders), which includes long-term follow-up studies and will evaluate the ethical, legal, and social challenges associated with screening for diseases that include late onset phenotypes [146].
- North Carolina—In 2021, North Carolina became the sixth state nationally to enact RUSP alignment legislation requiring implementation of NBS for a condition within three years of its addition to the RUSP and a report on the status of the addition at 18 months and every 6 months the addition is delayed past 3 years [296]. In February 2023, MPS-I and PD were added to the NBS panel of tests. SMA and ALD were added to the screening panel in 2021 and 2022, respectively [297]. Screening for each built on pilot studies, the results of which have been published: a report on the ALD pilot with particular focus on the single-tier HPLC-MS/MS screening test and its PPV and recall rate [298]; and a report on the SMA pilot that included evaluation of the SMA q-PCR screening method to confirm its robustness [299]. These studies were aided by the existence of the Early Check program, which is a comprehensive opt-in program that supports pilot studies of new NBS conditions. Its overarching objective is to demonstrate the feasibility and acceptability of statewide screening and follow-up for inborn conditions, inform public policy, and support USA RUSP nominations [300]. Other Early Check-related projects have included the use of a patient portal to recruit research participants for Early Check [301], direct mail and email outreach/recruitment of new mothers for NBS research [302], and education and consent in large-scale DNA screening [303]. With the research foundation in place, future studies are beginning including genome sequencing [304]. Other recent NBS activities within the state have included research and development in microfluidic instrumentation directed at mucopolysaccharidoses [81], validation of NBS for Fragile X using a custom FMR1 PCR assay system [305], and evaluation of a commercial assay for DMD NBS (CK-MM) and the DBS stability of CK-MM [306]. A 2024 report chronicles two years of newborn screening for Duchenne muscular dystrophy as a part Early Check [307].
- North Dakota—NBS specimens from North Dakota are analyzed and reported by the Iowa NBS program. A report in 2019 provided preliminary evidence of association between NBS refusal and provider type, home births, and hepatitis-B vaccine refusal while noting that, “Additional studies of obstetric providers, home births and women are needed to improve our understanding of the reasons for NBS refusal to better deliver preventive services to newborns” [308].
- Ohio—In July 2021, the Governor signed RUSP alignment legislation requiring implementation of screening for conditions that are added to the USA RUSP [309]. In July 2023, there was a NBS fee increase, which covered, among other things, testing for SMA and ALD, which were added to the screening panel in October 2022 [310]. Also in July 2023, the Governor signed legislation that requires the Ohio Department of Health Director to specify in rule the addition of DMD as a disorder for NBS making Ohio the first state to have a mandate for DMD NBS [311]. The Ohio NBS program includes KD and a report in 2022 discussed a qualitative assessment of parental experiences with false-positive NBS for KD, which showed concern regarding the lack of knowledge and short-term counseling skills among non-genetics providers and a need to integrate genetic counselors within the NBS result disclosure process [312]. Another 2022 report reviewed the value in additional screening for very low-birthweight newborns with “normal” screening results until thyroid function has been ascertained [313]. A 2024 publication reviewed parental experiences with NBS and gene replacement therapy for SMA [314].
- Oklahoma—The Oklahoma public health laboratory, including NBS, moved from Oklahoma City to Stillwater in 2020. Planning was limited and NBS testing was outsourced to a private laboratory for a brief period. This change added four conditions (PD, ALD, SMA, MPS-I) to the NBS armamentarium along with the addition of screening for SUAC, as a second-tier test for TYR-I. The screening laboratory is now operational in the new facility with these additions, although some challenges remain [315,316,317]. In May 2022, legislation requiring alignment with the USA RUSP was signed into law, which goes into effect in November 2024 [318].
- Oregon—The Oregon NBS program (also known as the Northwest Regional Newborn Bloodspot Screening Program) has been updating its screening algorithms for several screened conditions, including cystic fibrosis. Screening has expanded from 23 conditions to 62, most notably adding SMA in June 2022, and ALD in January 2023. The screening fee has also increased from USD 80 to USD 175 for two screens (August 2022), one near birth and the other at 1–2 weeks of age [319]. The 2022 Advisory Board meeting considered whether to remove Fabry and Gaucher diseases from the NBS panel and decided they did not meet their criteria for removal [279]. Other studies have included a framework to balance sensitivity with the unnecessary treatment of healthy infants due to A143T and other variants of uncertain clinical significance [320], and a review CAH screening accuracy and biochemical and clinical outcomes of CAH cases detected by the Oregon program, which collects specimens at two time periods following birth [321].
- Pennsylvania—The Pennsylvania NBS program is unique in its administration and operation and is governed by the Newborn Child Testing Act, which was amended in 2020. As part of the amendments, the NBS panel and program financing were modified/clarified. The NBS panel was expanded to 34 named conditions, including the addition of KD. Birthing facilities must continue to “… utilize and enter into agreement with the NBS laboratory contracted with the Department [of Health]”. This legislation also required compliance with the USA RUSP. Other program changes have included CF screening algorithm adjustment to include NGS (1 October 2022), addition of MPS II to the screening panel (1 July 2023), and implementation of GAMT screening (1 January 2024) [322]. Other NBS studies ongoing in Pennsylvania include a 2020 report on experiences with PD screening [323] and a 2022 report on data and outcomes for ALD NBS [324].
- Rhode Island—Rhode Island has a small birth population and partners with the Massachusetts NBS laboratory (NENSP) for laboratory services. In 2020, the program expanded by adding screening for SMA [325].
- South Carolina—The South Carolina NBS laboratory implemented Sunday courier service in September 2020, and weekday courier in September 2021. Screening for MPS-I and PD began in February 2021. Screening for SMA, argininemia, and ALD began in 2022 and KD in May 2023. In April 2022, the NBS program began a partnership with the Bureau of Vital Statistics to match newborn vital records with newborn blood-spot screening records to ensure that all newborns receive NBS [326]. Given that the pediatric provider of record has a significant role in NBS and infant health after birth, a recent South Carolina study examined how the pediatric provider of record is selected by parents and factors that might affect NBS education and processes in the perinatal period [327].
- South Dakota—The South Dakota NBS program utilizes the services of the Iowa NBS laboratory. In September 2022, PD was added to the NBS panel, and the fee was increased from USD 91 to USD 98. The South Dakota NBS program began providing long-term follow-up for diagnosed disorders through the Sanford Children’s Specialty Clinic in Sioux Falls in August 2023. The Sanford long-term follow-up team will provide care coordination, insight, primary care and specialist communication, support, and referral services to various state and private programs to NBS patients and parents [328]. A 2022 report summarizes NBS in South Dakota, including changes over time, the groups of conditions included in screening, the process of adding conditions to the screening panel, and the role of the primary care physician when a positive screening finding is reported [329].
- Tennessee—An open-access dashboard was developed to address NBS needs for methods to better visualize performance data, promote data transparency, and drive quality improvement. Eight NBS performance indicators can be visualized across several views, which are designed to provide an overview of NBS performance data at first glance, then allowing a drill-down to specific data. Dashboard development experiences can be applied to future dashboard development in or other public health programs implementing similar measures [330]. Personnel from the Tennessee NBS program have contributed as co-authors to publications in collaboration with other state NBS programs (as examples, see discussions on PD, MPS-I, α-, and ß- thalassemias, CF in Section 3.1.1).
- Texas—The Texas NBS program is one of the largest in the world processing some 800,000 specimens annually for over 30 different conditions (each newborn is required to be tested near birth and at 1–2 weeks of age.) NBS for SMA was begun in June 2021, and in June 2023, Texas became the first state to pass RUSP alignment legislation, and the 11th state overall. The RUSP alignment legislation initiates a three-year timeline in which the screening must begin for new conditions added to the RUSP and requires an annual report to state leadership that outlines the capacity to implement additional RUSP tests [331]. In addition to participating in several projects nationally already noted, a project analyzing the association between NBS analytes measured on a second screen specimen and childhood autism was reported in 2020 [332], a study demonstrating the robustness of the Texas NBS program before and after COVID-19 in South Texas [333], and a 2023 report reviewed cCMV testing in DBSs and found it to be as accurate as traditional urine, saliva, and plasma testing within the first 21 days of life [334]. Texas NBS program personnel have contributed as co-authors to publications in collaboration with other state NBS programs (as examples, see discussions on MPS-II, GAMT, CF in Section 3.1.1).
- Utah—The Utah NBS program is operated by the Utah Department of Health, requires two screening specimens on all newborns, one near birth and one at 7–16 days of age, and provides laboratory testing through the Public Health Laboratory (9 tests) and ARUP Laboratories (30+ tests; Department of Pathology, University of Utah). The NBS fee was increased in July 2003 to USD 140, which covers both screens, and two conditions were added to the NBS panel, MPS-I, and PD [335].In addition to its progressive work with GAMT [96], noted earlier, significant other NBS research is ongoing in Utah. A 2020 report looked at NBS knowledge and attitudes of midwives and out-of-hospital-birth parents to examine factors that drive NBS nonparticipation looking to improve their educational resources [336]. A review of the importance of timing in diagnosing and treating presymptomatic SMA patients identified through NBS was reported in 2021 with a recommendation for treatment by 14 days of life) [337]. Also in 2021, a data model was described that provides a foundation for implementing a standardized electronic data exchange across NBS programs that can accelerate implementation of electronic data exchange between healthcare providers and NBS programs ultimately improving health outcomes [338]. Two other reports in 2021 dealt with secondary screening testing: a report describing a scalable, exome sequencing-based next-generation sequencing (NGS) pipeline with a priori analysis restriction that can be universally applied to any NBS disorder [339]; and a report discussing how NGS can be used as a NBS secondary testing method, the importance of genomic variant repositories for the annotation and interpretation of variants and barriers to incorporation of NGS and bioinformatics into NBS systems [340]. A 2022 report of focus groups of parents who have interacted, or will soon interact with the NBS system, found generally positive reactions with some frustrations with result communications, reliable program/disorder information, and insurance complexities [341]. A recent report reviewed data related to detection of ALD from the perspective of a reference lab concluding that NBS has resulted in a significant increase in follow-up and presymptomatic case detection [342].
- Vermont—Vermont’s NBS specimens are tested as part of the NENSP using the screening laboratory in Massachusetts. The laboratory is currently assessing the capability of testing for MPS-II and GAMT, which were added to the RUSP in 2022 and 2023, respectively [343].
- Virginia—A 2020 report provides an educational review of NBS for both CH and CAH including the published studies of one- and two-screen states, concluding that continued method improvements and cutoff adjustments over time have improved case detection for NBS [344]. A 2022 article reviewed the history of attempts in Virginia to add KD to the screening panel concluding that until and unless KD is added to the USA RUSP, it will continue to face an uphill battle for inclusion in state NBS panels [89]. Recognizing the value of NBS, the Governor’s office announced the addition of SMA and ALD to the screening panel in March 2022 [345]
- Washington—The Washington State NBS laboratory provides testing, including weekends, for Washington, Hawaii (October 2019), and Idaho (April 2021). Washington is considered a “two screen state” since a second NBS at 1–2 weeks is strongly recommended with high compliance. SMA was added to the screening panel in 2020. An expanded CF DNA testing algorithm was recently initiated (1 July 2023). The Washington NBS program has a research collaboration with the Pacific Northwest Research Institute in the five-year CASCADE (Combined Antibody Screening for Celiac and Diabetes Evaluation) study to demonstrate the feasibility of detecting Type 1 diabetes and celiac disease from DBSs through population-based screening. There is also a collaboration with Key Proteo, Inc. to demonstrate that their novel, multiplexed proteomic-based assay can be used for DBS screening to detect Wilson disease, X-linked agammaglobulinemia, Wiskott-Aldrich syndrome, and adenosine deaminase deficiency [346].A 2020 review of the literature involving proximal urea cycle disorders and the possibility of their inclusion in NBS showed that while these conditions meet the medical, diagnostic, treatment, and public health rationales for NBS, screening sensitivity and specificity would need improvement before the screening test would be satisfactory for NBS [347]. A pilot study of 100,000 specimens using a 5-plex LC MS/MS assay for MPS-II, MPS-IIIB, MPS-IVA, MPS-VI, and MPS-VII) demonstrated the feasibility of NBS in a state screening laboratory [348]. A collaborative project involving the Washington NBS program and Seattle Children’s Hospital resulted in the development of a novel proteomics-based assay for rapid and accurate second-tier screening test for PD and MPS I that can possibly reduce the false-positive rate and shorten the clinical follow-up for severe patients resulting in better clinical outcomes [349].
- West Virginia—The West Virginia NBS Program is in the Office of Maternal, Child, and Family Health and the Office of Laboratory Services, in the Bureau for Public Health. On 7 March 2023, the NBS program expanded to include LSDs defined by the Code of State Rules: PD, Fabry disease, MPS-I, MPS-II and Morquio syndrome (MPS-IVa) [350]. A 2020 report from West Virginia researchers reported that parents/guardians of children with a positive NBS result reported less difficulty in receiving needed specialty care than parents/guardians of children with genetic conditions were diagnosed later. This finding implies greater population level benefits realized in jurisdictions with expanded screening versus those conducting minimal testing [351].
- Wisconsin—The Wisconsin NBS laboratory is part of the Wisconsin State Laboratory of Hygiene located within the University of Wisconsin at Madison. The NBS program as a whole is contained within the laboratory and there is an active research component. Given the tendency of NBS programs to move toward molecular testing and based on their own program experience and literature reports, current applications of molecular technologies in routine NBS practice were tabulated in 2020 to begin to catalog NBS molecular uses and assay principles [352]. NBS thyroid research has continued with three recent publications: TSH reference charts from day 1 until day 14 for moderate-to-late preterm infants were constructed using a state-wide cohort of NBS patients with the intent to examine relationship between age-adjusted NBS TSH percentiles and long-term neurodevelopmental outcomes in future studies [353], a study that tested the hypothesis that term-born small for gestational age (SGA) neonates have elevated NBS TSH concentrations and an increased incidence of CH compared with non-SGA term neonates did not support the hypothesis after adjusting for potential confounders, however, NBS TSH concentration were higher in term SGA neonates compared with term non-SGA neonates [354], and a study that looked at a cohort of preterm infants with higher NBS TSH percentiles, suggesting potential subclinical hypothyroidism, found no prediction of any adverse effect on neurodevelopmental or growth outcomes [355]. Two studies relating to improvements in CAH laboratory processes have also recently been reported: a 2020 report described the use of principal component analysis (PCA—a statistical method that reduces high-dimensional data to a small number of components and captures patterns of association relevant to the outcome of interest) to improve the PPV of the current screening algorithm from 20% to 67% [356]; and a 2022 report noted that multiple 17-OHP cutoff co-variates (birthweight, time of collection) failed to improve the accuracy NBS for 21-OH CAH suggesting the need to use alternative approaches not related to 17-OHP for assay improvements [357].The Wisconsin NBS program has continued to assume a leadership research role with CF. In addition to co-authorships in many multi-state and multi-national publications related to CF NBS, Wisconsin researchers have also focused on information and improvements for the local NBS program. In 2020, a comprehensive report outlined the impact of the CFTR gene discovery on CF diagnosis, counseling, and preventive therapy [358]. A 2023 report describes current refinements in NBS algorithms by applying NGS as a screening method to enhance sensitivity and equity while minimizing incidental findings [359]. Even with NGS, the Wisconsin program will require sweat testing on all patients with one identified CFTR variant. In a separate 2023 article, a listing of misperceptions and counter arguments favoring single variant sweat testing are provided [360]. A 2021 report reviewing long-term pulmonary and mortality outcomes of CF patients in the original Wisconsin Cystic Fibrosis Neonatal Screening Project, which ended in 2012, found that, “NBS alone does not improve pulmonary outcomes in CF, particularly when other risk factors supervene. In an era prior to strict infection control and current therapies, NBS for CF may be associated with worse pulmonary outcomes” [361].As one of the first USA NBS programs to implement SCID screening (pilot in 2008), the longer NBS history has allowed for a 10-year review of referrals to a single referral center describing a broad spectrum of medically actionable and idiopathic T-cell lymphopenia [362]. Additionally, the longer SCID testing experience has allowed for modifications in laboratory procedures, including using multiples of the median values for TREC assays, which eliminates the need for standards with known TREC copy numbers and allows for assay normalization and assay comparisons between different laboratories [363], and data suggesting that TRECs increase at a steady rate as gestational age increases, which provides a rationale for Wisconsin’s NBS program’s recommendation for a second NBS test following a screen-positive SCID result in a premature infant (instead of performing flow cytometry) [364].More recently, focus has turned to SMA and ALD. The first year of screening for SMA has been reported in which a multiplex real-time PCR assay was used to detect homozygous SMN1 exon 7 deletion, triggering a droplet digital PCR assay for SMN2 copy number assessment. This method’s positive predictive value was reported to be 100%. and facilitated timely clinical follow-up, family counseling, and treatment planning [365]. For ALD, flow-injection MS/MS in a negative ion mode was reportedly used to shorten ALD analysis run-time to 1.7 min, thus maintaining the advantage of negative mode MS for eliminating isobaric interferences that might lead to false-positive screening results [366].
- Wyoming—The Wyoming NBS program requires a second screening specimen at 7–14 days of age on all newborns. Screening laboratory services are provided in partnership with the neighboring Colorado NBS program and research activities within the state are minimal. Unlike most other states, Wyoming law requires parental consent for NBS [367].
Provincial Activities (Canada)
- Alberta—NBS in Alberta is a collaboration of government and health service organizations providing NBS to newborns in Alberta, the Northwest Territories, and the Kitikmeot region of the Nunavut territory. The Newborn Metabolic Screening Laboratory located at the University of Alberta Hospital (UAH) in Edmonton provides all screening testing except for SCID and second-tier testing for CF, which are performed at the Molecular Diagnostics Laboratory at UAH [368]. NBS for SMA was begun on 28 February 2022 and included a qPCR screening assay to detect the absence of SMN1 exon 7, which was multiplexed with an already established SCID qPCR assay allowing for a smooth cost-effective implementation. The results of screening 47,005 newborns during the first year of the program (prevalence of 1:9401) were recently reported [369]. NBS for classical galactosemia (galactose transferase deficiency—GALT) has been ongoing since April 2019, using a two-tier screening approach, which secondarily identifies infants with glucose-6-phosphate dehydrogenase (G6PD) deficiency. A study of three years of case data shows that reporting G6PD cases “may lead to increased identification of neonatal hyperbilirubinemia and may decrease the incidence of episodes of hemolysis in these patients…[and]… saved a significant number of families from the anxiety associated with the work-up required to rule out a potential GALT diagnosis [370]”.
- British Columbia (BC)—The BC NBS program, administered by the Provincial Health Authority, facilitates screening of approximately 45,000 newborns annually in BC and the Yukon. “Newborn Screening BC” is a collaboration between BC Children’s Hospital, BC Women’s Hospital and Health Centre, Provincial Laboratory Medicine Services and Perinatal Services BC. The NBS program was expanded on 1 October 2022 to include SMA, SCID, and BIO [371]. A 2020 study report on the clinical impact of NBS for CAH and the incremental costs in screened vs. unscreened newborns found that while NBS did not result in cost savings, it was cost effective since screened newborns were less likely to require medical transport and had shorter hospital stays resulting in lower hospitalization costs [372]. Another study reported in 2020 found that an IRT-DNA-IRT algorithm, with a repeat IRT measurement for apparent carriers at 21 days, successfully reduced the number of sweat tests required without significantly impacting CF case detection sensitivity [373]. Report of a program study in 2023 described the validation of a second-tier dual derivatization approach with LC-MS/MS to detect 2-methylcitric acid, methylmalonic acid, and total homocysteine in DBS cards amenable to NBS [374].
- Manitoba—The Cadham Provincial Laboratory (Winnipeg), which provides NBS, announced program expansion to include SMA on 8 June 2022 [375]. A 2020 report details experiences with GA-I cases detected in Manitoba over a 40-year period that included two cohorts of patients (before and after the introduction of NBS in 2000). In addition to reporting on the outcomes observed, suggestions are included for improving the program going forward [376]. A 2021 report noted the high incidence of SCID in Manitoba related to two founder mutations in the Mennonite and First Nations of Northern Cree ancestry populations. The authors report the development, validation, and implementation of a multiplexed qPCR assay to detect both mutations, which should, “…decrease the morbidity and mortality of SCID in Manitoban neonates and establish a precedent for future population-specific screening programs in Canada” [377].
- New Brunswick (NB)—NB, along with Prince Edward Island (PEI), is part of the Maritime NBS Program served by the NBS laboratory at the Izaak Walton Killam (IWK) Health Center in Halifax, Nova Scotia. See Nova Scotia for more information.
- Newfoundland and Labrador—NBS management and laboratory services are provided by the Ontario newborn screening laboratory [378].
- Nova Scotia—The Maritime NBS Program (Nova Scotia, NB and PEI) uses the screening laboratory at IWK Health Center in Halifax, Nova Scotia. There is currently interest in implementing NBS for SMA in the Maritime provinces. A recent report describes the status of SMA NBS in 2022, and illustrates the lack of screening across Canada, including the Maritime provinces [32]. In late 2021 a significant grant (about $322,600) was provided to the Maritime NBS Program by Muscular Dystrophy Canada and Novartis Canada to assist in overcoming the challenges of adding SMA to the NBS panel [379]. As part of the process, a recent epidemiological study looked at SMA prevalence in the region finding an incidence of about 1:11,900 [380]. Another condition, infantile onset CPT-II, has also been suggested as a possibility for screening in a 2021 report [381].
- Ontario—The Ontario NBS began expansion in 2006 guided by the USA RUSP. An updated provincial NBS program was also implemented, which was reported to be different from conventional NBS programs. The updated program is a structured and fully funded partnership between the Ministry of Health (MOH), the NBS laboratory, and the provincial treatment centers, each component with a defined role and accountability. Recalled newborns are immediately referred to a regional treatment center for evaluation and further follow-up service coordination [382]. Considerable research has been ongoing in the Ontario NBS program since 2020 in addition to clinical studies and the international collaborations already noted. A 2020 report on the ability of NBS to detect newborns who will develop inflammatory bowel disease (IBD) in childhood found that an expanded panel of metabolites would be necessary for IBD detection [383]. Reports of two 2021 studies addressed the role of primary care providers (PCPs) in NBS for CF: the role of the PCP in notifying the parents of positive NBS findings [384]; and the PCPs’ preferred roles and confidence in caring for infants with a screen-positive CF NBS result and in assisting with CF family planning issues [385]. The latter showed a lack of confidence in addressing carrier issues demonstrating a need for PCP genetics education. A study evaluating the health outcomes of children with CFSPID (or CFRMS) at school age across Canada and the value of the sweat chloride in predicting CF risk [108]. A commentary on the benefits and harms of NBS identified this study as filling an evidence gap potentially useful as a predictive tool useful in maximizing NBS benefits and minimizing harms [386].Several other publications in 2021 dealt with NBS issues: a study of the predictive value of NBS analytes found a lack of association with infant mortality between 7 days and 6 months of age [387]; a study of sickle cell carrier status, which is not reported in Ontario NBS, identified statistically significant differences in health services use among sickle carriers relative to controls, with small effect sizes and inconsistent association directions across age groups and health service types indicating that carrier status is likely benign in early childhood [119]; a report recommending testing and follow-up protocols for implementation of NBS for SMA in Ontario included a standardized path to early diagnosis and treatment for optimal screening outcome [388]; a report estimating a minimum prevalence for 22q11.2 deletion of 1:2148 making it one of the more common rare genetic conditions and supporting the importance of NBS for early diagnosis and disorder management [389]; and a report of low TREC copy numbers when screening for SCID likely caused by neonatal abstinence syndrome [66].Following the implementation of SMA NBS, a 2022 report of the first year of SMA NBS identified several improvements that could potentially reduce time to treatment: molecular laboratory operation on weekends; reduced specimen transit time; reduced confirmatory testing time; and (4) more efficient approval of treatment medication [390]. Other 2022 reports related to the NBS program concerned prenatal alcohol exposure and transient CH. In the former case, residual DBSs were used as a population resource to estimate the extent of PAE exposure in Ontario by quantifying phosphatidyl ethanol homologues and to use the findings to reassess the effectiveness of current interventions [391]. In the latter case, a possibly more efficient clinical method for differentiating transient from permanent CH was proposed using predictive factors to create a risk score for predicting the likelihood of a successful off-therapy trial at age 1 year [392]. At least three reports in 2023 addressed NBS issues: a report demonstrating how analytical imprecision and biomarker distribution within a population can be used to inform decisions on screening thresholds in general, with NBS for GALT as an example [393]; a report reviewing the pre-analytical factors influencing screen-positive findings for BIO using a time series analysis of print lots of collection cards to demonstrate contributions from both seasonal temperatures and print lots [394]; and a report of a study of parents attitudes about expanded NBS with NGS finding a preference for conditions with supportive interventions in childhood, despite other possible benefits [395].
- Prince Edward Island (PEI)—NBS began in PEI in the mid-1960s and is now a part of the Maritime NBS Program in Halifax, Nova Scotia. See Nova Scotia for more information.
- Quebec—The Quebec NBS program includes two specimens, each tested for its own screening panel. An initial DBS specimen is collected at 24–48 h of age and a urine specimen is collected at 21 days of age. This program has been described as a “unique-in-the-world” program and has functioned within the provincial healthcare system since the early 1970s with over 3.5 million newborns screened. The urine NBS program is complementary to the blood NBS program, allowing detection of disorders not readily identifiable in blood and provides a unique research opportunity, with informed consent of the parents. A 2021 report described the steps being taken to transition the urine screening program from thin layer chromatography to MS/MS [396]. In addition to a probable French Canadian founder effect for hyperornithinemia–hyperammonemia–homocitrullinuria (HHH) syndrome, which is detectable in the urine screening program, there is also an increased prevalence from a founder effect for vitamin D-dependent rickets type 1A in the Saguenay-Lac-Saint-Jean region, and a report on implementation of a local NBS program was reported in 2022 based on the criteria of the Quebec National Screening Committee [397]. Other NBS recent reports include the description of a case of hyperlysinemia with a profile of cystinuria (but with high urinary lysine) identified with urine screening, further establishing this rare condition as a biochemical abnormality, which may not require any intervention [398], and a report on the benefits of early detection and referral of children with SCD detected by NBS to comprehensive care [399].
- Saskatchewan—The Saskatchewan NBS program is administered by the Saskatchewan Health Authority. In February 2022, the program announced its expansion to include screening for SMA, HGB, SCID and cCMV [400].
3.1.3. Tabular Summary of Information (States, Provinces, and Territories)
USA States/Territories
Canadian Provinces/Territories
3.2. Asia–Pacific
3.2.1. NBS Activities Focused on the Asia–Pacific Region
3.2.2. NBS Activities within Countries
- Australia—NBS has been ongoing in Australia since 1964. The Standing Committee on Screening of the Human Genetics Society of Australasia/Royal Australasian College of Physicians provides oversight and advice to the NBS programs in both Australia and New Zealand (six laboratories including five Australian States and New Zealand). The NBS program is delivered at the state and territory level with each acting autonomously with funding from the Australian Government through the National Health Reform Agreement [405]. Shortly after our 2015 report, the NBS policy framework and decision-making process became the subject of studies aimed at developing national NBS policies in Australia [406,407]. Currently, there are five independent Australian NBS laboratories (programs) in New South Wales (NSW), Queensland (QLD), South Australia (SA), Victoria (VIC) and Western Australia (WA). The Australian states/territories that do not have NBS laboratories partner with those that do; therefore, specimens from the Australian Capital Territory are sent to NSW, specimens from Tasmania go to SA, and specimens from the Northern Territory are split between QLD and SA [405]. Further screening details are provided in Section 3.2.3.Currently, all five programs provide similar testing services with the ability to screen for different conditions. Both SCID and SMA are being added to each program on different timeframes. In 2022, an economic evaluation of NBS for SCID was published suggesting the value of SCID screening on both clinical and economic grounds [408] and government cost savings were also illustrated for a combined NBS approach with SCID and SMA [409]. Several reports involving NBS for SMA, including results of pilot studies, have been recently published as Australian NBS programs consider its possible addition: a prospective description of the experiences in implementing NBS for SMA in NSW [410]; a discussion of the first pilot screening project from August 2018 until January 2021 [411]; a thematic analysis of parents’ perceptions and the psychological impact of NBS for SMA [412]; a “value-for-money” assessment of NBS and gene therapy from a clinical and policy point of view [413]; a study demonstrating that NBS and early access to disease-modifying therapies effectively ameliorate the functional burden and associated comorbidities for affected children [414]; and a study demonstrating the technical feasibility of NBS for SMA using NGS in a multiplex platform may provide simultaneous testing for hundreds of inherited conditions [415]. SMA screening has now been recommended by the Australian Government to be rolled out nationally with support from a research team who will help develop standardized guidelines. NBS for SMA is now available in NSW and WA, and NBS for SCID is available only in NSW. Other laboratories have pilot studies either planned or ongoing.Aside from studies of new conditions, studies of long-standing screening conditions have also recently been published, including CH, CAH, and CF: assessing a lower CH (TSH) screening threshold with a concurrent 8-fold increase in the recall rate [416]; harmonizing CH screening protocols across Australasia in response to 50 years of data and variable incidence levels across the six programs [417]; evaluating a two-tier NBS laboratory protocol for CAH in NSW [418]; considering the use of 21-deoxycortisol analysis and LC-MS/MS in the screening/diagnosis protocol for 21-OH CAH [419]; and evaluating increasing median CF survival calculations relative to factors such as NBS, improved care etc. [420]. Looking at other NBS-related research, a 2020 report sought to demonstrate linkages between diabetes, biochemical genetics and NBS suggesting that NBS specimens may be underused in diabetes research [421], and a 2021 publication reported characteristic subtle (nonpathological) changes in neonatal metabolism from fetal metformin exposure evident during NBS [422]. Looking to the future, studies were also reported on the candidate conditions ALD [423] and DMD [424], and the Medical Services Advisory Committee recently announced their recommendations to add ALD and SCD to the Australian NBS panel [425].PD screening was proposed to the Australasian Standing Committee for Screening in 2018 but was not approved prompting publication of a report of PD screening activities in support of NBS [426]. The possibility of NBS for chromosome 15 imprinting disorders using genetic techniques [427] along with the knowledge and attitudes of Australian parents and health professionals towards genomic sequencing in NBS [428] and public perspectives on the risks, benefits, and preferences for implementation of gNBS [429]. A 2023 report notes that the addition of genomic sequencing into population-wide NBS will invariably expand the knowledge of treatable rare diseases, potentially benefiting health over a lifetime. This report discusses the challenges and opportunities ahead, particularly “the need to generate evidence of benefit and to address the ethical, legal and psychosocial issues” raised [180]. An Australian multi-disciplinary expert group has now reviewed 604 of a possible 1279 genes for possible inclusion in gNBS and developed a consensus gene list for possible use in systematic harmonization efforts for gNBS in Australia and internationally [430].
- Bangladesh—The NBS movement for CH was introduced in Bangladesh in 1999 by Bangladesh Atomic Energy Commission (BAEC) and as a regional project of the IAEA. Studies from local pilots have demonstrated an occasionally effective program suitable for government support/management as part of a policy for combatting congenital diseases [431]. The screening laboratory at the Nuclear Medicine Institute at Bangabandhu Sheikh Mujib Medical University, Dhaka is a well-equipped NBS center under BAEC with 3–4 peripheral NBS centers (currently inactive). Currently, screening coverage is <5% and is not considered sustainable and the lack of a national policy that includes funding remains the biggest challenge [432,433]. A recent survey of the NBS knowledge among health professionals found that NBS for CH, CF, PKU, galactosemia (GAL), HGB, and SCID were available in certain locations and suggested an overall lack of awareness of NBS that needed to be addressed nationally [433]. An investigation of PKU in the Rajshahi Division, Bangladesh led to similar conclusions and a national implementation strategy was outlined [434].Other NBS studies on Bangladeshi newborns have been reported with particular emphasis on local incidences of metabolic and other conditions detectable by NBS: a study of LC-MS/MS techniques, establishing cutoff values for various amino acids and acylcarnitines, to assist government authorities in installing and establishing NBS [435]; a Canadian collaboration study of CH and HGB in Matlab, Bangladesh to determine incidences and to validate the use of an offsite remote laboratory [436]; a report on the ease of use of DBSs for remote laboratory testing as part of NBS in LMICs [165]; a study to assess the prevalence of α-thalassemia and provide a model for NBS [437]; and a study to demonstrate the feasibility of NBS for SCD and β-thalassemia [438]. Since NBS for conditions like SCD, thalassemias and other genetic conditions is aided by population education and family counseling when cases are detected, it is important to consider the availability of trained genetic counselors. A 2021 report reviewed the status of genetic counseling in Bangladesh including the current scenario, challenges, and a framework for genetic service implementation [439].Studies have been ongoing in Bangladesh and certain other LMIC countries to determine the feasibility of an algorithm (using NBS specimens) developed in Ontario, Canada to reliably estimate the gestational age of newborns when other methods may not be available: a validation protocol for metabolic gestational age assessment in low-resource settings [164]; a study of the cost-effectiveness of a gestational age metabolic algorithm for preterm and small-for-gestational-age classifications [440]; a study to facilitate the use of machine learning prediction of gestational age using metabolic screening markers resistant to ambient temperature transportation [441]; and a recent report on the development and external validation of machine learning algorithms for postnatal gestational age estimation using clinical data and metabolomic markers [166].
- Cambodia—NBS in Cambodia is in the beginning phase and has been ongoing only in limited hospitals. The Calmette Hospital, Phnom Penh, Cambodia has been providing NBS for CH and G6PDD. In addition, some private clinics are also offering NBS. The major challenges are the lack of trained specialists in endocrinology and difficulties in securing life-long treatment for diagnosed patients [442].
- China—NBS has been ongoing in China since the first CH and PKU pilot in 1981, formalized by law (PKU and CH) in 1994 and management measures in 2009. While somewhat complex, a report of the history of NBS in China until 2012 is available [443]. Today, NBS in China is a complete network system with strict management and quality control throughout. Expert consensus documents have been published under the supervision of the Special Committee of Birth Defects Prevention and Control: organizational management and dry blood sample collection for neonatal inherited metabolic disease screening [444]; diagnosis and treatment of inherited metabolic diseases by NBS [445]; and laboratory testing technical guidelines of neonatal genetic metabolic disease screening [446]. MS/MS testing is now available nationwide, although its implementation may vary by location, and there are over 240 screening centers. NBS is administered at the provincial level, which results in slightly different screening panels as the program expands. There are several recent reports related to expanded NBS with MS/MS: an evaluation of MS/MS implementation in Suqian city (North) [447]; analyses of the cost effectiveness of ENBS in Shenzhen (South) [448] and Zhejiang province (East) [449]; and studies to determine the disease spectrum and genetic characteristics of IEMs detected by ENBS in Xi’an city (Northwest) [450] and Changsha (Central) [451]. A robust system-wide quality management system exists including both laboratories (National Center for Clinical Laboratories) [452] and non-laboratory components [453], sometimes augmented by regional quality improvement activities [454]. A large study was reported in 2021 that determined normal ranges for 35 MS/MS conditions in the Chinese newborn population [455]. Two 2023 reports review IEM findings in both the eastern coastal region [456] and the north [457].Research reports concerning ENBS tend to focus on individual conditions or groups of conditions: investigating the biochemical, clinical, and molecular profiles of IVA patients in Quanzhou, China (incidence 1:84,469) [458]; determining the biochemical and genetic characteristics of patients with primary carnitine deficiency (PCD) and identifying the need for genetic testing to improve screening efficiency [459]; screening for mitochondrial carnitine-acylcarnitine cycle disorders in Zhejiang Province [460]; establishing the incidence and identifying 10 novel variants of PCD in Zhejiang Province [461]; establishing the incidence and identifying 5 novel variants of PCD in Quanzhou [462]; establishing the incidence and identifying 6 novel variants of PCD in Ningbo [463]; establishing the incidence for screened FAODs in Chongqing (PCD is most common FAOD) [464]; using second-tier ultra-performance liquid chromatography (UPLC–MS/MS) to screen for short-chain acyl-CoA dehydrogenase deficiency (SCADD) and isobutyryl-CoA dehydrogenase deficiency (IBDD) to reduce referrals [465]; and assessing the long-term prognosis of 35 patients with methionine adenosyl transferase deficiency (incidence ~1:116,161) based on NBS in Zhejiang [466]. Other recently reported investigations include a method for detecting Duchene muscular dystrophy using creatine kinase isoform MM (CK-MM) [467], estimating the DMD frequency in Henan province (1:4370) [468]; and using MS/MS to define the cutoff values and screen for six newborn LSDs in Shandong province (total incidence ~1:3000) [469].Looking to the future through an ethics-first, forward-looking lens, a group of Chinese NBS experts published a consensus statement in 2021 on NBS for monogenic diseases (genetic NBS) in China to further standardize the genetic screening system for newborns and provide guidance on the application of NGS in genetic NBS [470]. Chinese researchers continue to report results of pilot studies of various gene panels potentially applicable to genetic NBS: a 573-gene panel in combination with biochemical screening [471]; a 134-gene panel for 74 inborn disorders [472]; a 465-gene panel for 596 disorders as part of the Newborn Screening with Targeted Sequencing (NESTS) program in eight hospitals across China and one hospital in Beijing [473]; and a 135-gene panel for 75 disorders [474]. Related reports provide updates on the current status of genetic NBS and a look at the future [475], the use of NGS as a first-tier screening test [476]; the current attitudes and preconceptions on genetic NBS in the Chinese reproductive-aged population [477], the value of combining traditional NBS and genetic screening to reduce false negatives and to improve the early/accurate identification of CH [478], the viability of incorporating genetic screening for neonatal intrahepatic cholestasis caused by citrin deficiency (NICCD) into the current NBS program [479]; and the identification of increased numbers of actionable variants with fewer false-positive NBS results with WGS [480].Research into new procedures and technologies for ENBS not detected through MS/MS has been the subject of at least five studies since 2020: a report on an artificial intelligence (AI) disease model to identify IEMs and decrease the occurrence of false negatives and false positives [481]; a study that develops artificially intelligent disease risk prediction models for quickly, accurately, and with minimal false-positive rates interpreting tandem mass spectrometry data [482]; a report of increased sensitivity and fewer false-positive screens for carnitine-acylcarnitine translocase NBS by using acylcarnitine ratio indices [483]; a pilot study of a commercial assay using matrix-assisted laser desorption/ionization time-of-flight mass spectrometry to screen for SMA [484]; and studies of NBS for ALD using FIA-MS/MS [485,486] and LC MS/MS [487] to detect lysophosphatidylcholines and acylcarnitines. Other published NBS studies include optimizing the PKU cutoff value in Xinjiang Uygur Autonomous Region based on sampling time [488], obtaining the birth prevalence of tetrahydrobiopterin deficiency in China using NBS data [489], improving the sensitivity NBS for CAH by including second-tier steroid profiling and liquid chromatography–tandem mass spectrometry (LC-MS/MS) in Shanghai [490], optimizing delivery and storage conditions to effectively reduce the degradation of certain amino acids and carnitine in DBS to improve assay accuracy and reliability [491], and the clinical features and outcomes of 31 children with CH missed by neonatal screening in Guangzhou [492]. A 2021 review of the national CH data from 245 screening centers in 30 provinces [almost 92 million newborns screened with about 43,000 cases of primary CH detected (1:2145)] between 2013 and 2018 and found a higher incidence of CH in the east than west when all cases were mapped [493]. The cost effectiveness of NBS for congenital cytomegalovirus infection was also studied, but looking at saliva or urine screening instead of blood [494].
- Hong Kong Special Autonomous Region (SAR), China—NBS in Hong Kong has been in place since 1984 with NBS for CH and G6PD provided on cord blood at no cost for babies born in public hospitals with maternity services [495]. A private expanded NBS program at the Chinese University of Hong Kong was begun in 2013 and continues today screening for 31 inherited metabolic disease (IMD) conditions [496]. A 2021 report published in 2023 reviewed the progress with the Chinese University NBS program noting the addition of NBS for CF in 2014, CAH in 2016, ALD, SCID, and SMA in 2021 [497]. Since our 2015 report, there have been at least 2 NBS pilots for multiple IMDs, one led by The University of Hong Kong [498] and one government-led [499] resulting in phased-in ENBS using dried bloodspots. The number of IMD disorders screened in the government-led program increased from 21 to 24 in 2016 and then 24 to 26 in 2019 [495]. A government-led pilot of NBS for SCID was conducted after a follow-up review of NBS for SCID recommended its inclusion on the Hong Kong screening panel before it was formally added to the NBS program in 2023 [500]. Building on a healthcare burden and life cost study of spinal muscular atrophy cases in Hong Kong that supports the idea of NBS [501], SMA is currently in pilot testing in Hong Kong [502].Three reports have been recently published involving parents: one assessing the content of online parental resources, including information on both urine screening and blood screening [503], and two reviewing positive attitudes of parents and healthcare providers on storage and the use of bloodspots after screening [504,505]. An immunoassay for ceruloplasmin concentration on DBS for Wilson’s disease was successfully modified and automated allowing for its inclusion on NBS panels [506]. To summarize, the government led NBS program in Hong Kong covers 26 IMD conditions, SCID and SMA (pilot testing) whereas the private Chinese University of Hong Kong NBS program covers 31 IMD, SCID and SMA.
- India—Despite more than a generation of studies, including government studies, and repeated indications of imminent actions to initiate/recognize a NBS program at the national level, no nationally recognized government-backed NBS program yet exists. While NBS is not widely available in public hospitals where more than 60% of births occur, it is becoming increasingly available in the private sector, including expanded testing. There continue to be data reviews and appeals for government recognition of NBS based on these data: an appeal based on positive experiences from regional (model) programs in Chandigarh, Goa, and Kerala [507]; an appeal for all hospitals in urban areas to initiate NBS for CH, CAH, and G6PD deficiency (the local common disorders) [508,509]; a call for the government to support expanded NBS due to increasing public awareness and large numbers of cases detected annually in regional programs [510]; a review of 1.5 decades of case detection data from three tertiary care centers in Bangalore supporting NBS [511]; a systematic review and meta-analysis of the prevalence, screen positivity rates, and etiology of NBS for CH [512]; an analysis of the biochemical basis, clinical manifestations, treatment advances, and present status of screening [513]; a study of the cost effectiveness and cumulative economic benefits of NBS for CH [514]; and a study to show general acceptance by the public, and the need for increased general awareness of NBS [515].A large multicenter program covering over 230,000 newborns in Delhi, supported by the Science and Engineering Research Board, has recently demonstrated the need and feasibility for NBS for CH, CAH, G6PD, BIO and GAL and has provided information on amino acid, fatty acid and organic acid disorders. These data form the basis of a program in Delhi called Mission NEEV (Neonatal Early Evaluation Vision). It envisages NBS for IMDs, along with POC tests for critical congenital heart disease, hearing, retinopathy of prematurity, and visible birth defects [516].Reports continue to accumulate on screening experiences despite the lack of national coordination. Reviewing reports from 2020 until now, 2 multi-condition studies have been reported: a 3-year study of CH, CAH, G6PDD, GAL, and PKU in Bengaluru, South India [517]; and a Canadian collaboration study of CH, CAH, G6PDD, GAL, PKU and BIO in Udupi district of South India [518]. Reports continue regarding NBS for endocrinopathies: CH screening, prevalence, and etiology in Indian preterm babies [519]; a report of confirmatory testing from the original DBS to speed diagnosis of CAH (assuming that the DBS and patient match) [520]; a review of the challenges and opportunities resulting from NBS for CAH [521]; a case study of a newborn with unnecessary devastating consequences from CAH resulting from no NBS [522]; a standard operating protocol for routine NBS for CAH in Indian settings [523]; and a discussion of genetic confirmatory testing for CAH from the original DBS as a means of speeding the diagnostic process [520].Because the second largest national health burden results from hemoglobinopathies, here is increasing interest in NBS for SCD: a 6-year review of NBS for SCD in the tribal populations of Gujarat and Madhya Pradesh (noting that Indian SCD is not always mild and tertiary care centers are lacking) [524]; a review of the clinical profile of children detected with SCD from NBS in Gujarat and recognizing a high prevalence of α-thalassemia [525]; a description of the development/implementation of a NBS for SCD implemented in 12 SCD-endemic and tribal-dominated primary/community health centers across six districts of India [526]; and an evaluation of a point-of-care test for SCD (HbA, HbS, HbC) suitable for onsite NBS [527]. The Prime Minister’s vision of elimination of SCD by 2047 is an incentive to expand NBS for these diseases by initially targeting pockets in tribal populations as well as areas where SCD is known to be common [528]. The 2020 report on NBS implementation in India, in addition to providing a brief but comprehensive history of NBS in India makes two important points: (1) “the increasing awareness and programs over the past decade have led to more babies being screened every year”; and (2) “the results of the existing programs suggest to the policy makers in India that there is a benefit in implementing a universal NBS program” [507].
- Indonesia—Beginning in 1999, the IAEA aided in starting NBS for CH. A pilot project and health technology assessment was conducted in two hospitals between 2000–2005, which was then followed by the expansion of CH NBS to eight provinces in 2008 (West Sumatra, Jakarta, West Java, Central Java, East Java, Yogyakarta, Bali, and South Sulawesi). Although the MOH released a decree recommending CH NBS in 2014, lack of public awareness and lack of prioritization did not lead to sustainable NBS [529]. An interest in NBS has continued and in 2020 a review of CH and CAH activities in five Indonesian cities from October 2015 to January 2016 reaffirmed the high rates for both conditions, the high false-positive rate when screening for CAH and the overall need for NBS [530]. A 2022 report reviewed the literature over the last decade to identify screening problems and solutions for implementing an improved CH program [531]. In 2022, the coverage of congenital hypothyroidism in Indonesia was 2.3%. The MOH policy reorganized NBS by requiring that all newborns receive screening for CH beginning 1 September 2022 [532]. The year after, the MOH enacted a new decree requiring all healthcare professionals to perform CH NBS as a requirement to claim delivery rates from the national insurance.The challenges and future implications of CH NBS in Indonesia were recently analyzed in view of its importance in improving healthcare. Parent refusal, early hospital discharge, and unavailability of specimencollection cards were identified as the most challenges. [533]. At the 67th U.N. Session of the Commission on the Status of Women in May 2023, the Indonesian Minister of Health, presented a keynote address in which he noted that part of the health system transformation underway in Indonesia would include, “NBS for CH and CCHD for every child born in Indonesia”. With increasing government commitment and NBS being viewed as a national health priority, the country has seen a stark increase in the capacity of NBS for CH nationally with current screening capacity reaching up to 50,000 samples a week at the end of 2023. While there are no laboratories specifically dedicated for NBS, Indonesia maximizes 12 existing national referral laboratories, each responsible for different provinces. In order to increase screening coverage to 80,000 samples a week in 2024, there are plans to add more laboratories to accommodate the geographic extremes that come with 17,000 islands, 38 provinces, 416 counties, and 98 cities. Additional screening for CAH, G6PD, critical congenital heart disease and hearing is currently in the planning stage by the Directorate of Nutrition and Maternal and Child Health in the MOH [533].
- Japan—Japan continues to have a progressive NBS program which began in 1977 with PKU and continues today with over 20 conditions. The screening history has recently been reviewed through a topical collection of articles in this journal, an overview of which is described in the referenced article [534]. Greater details of the history of Japanese NBS are included in the historical review of NBS for CH [535]. As part of this history, Guidelines for Mass Screening of Congenital Hypothyroidism were developed by the Mass Screening Committee of the Japanese Society for Pediatric Endocrinology in 1998, revised in 2014, and slightly revised again in 2021 [536]. Part of the NBS history is also detailed in the 30-year review of lessons learned from CAH screening [537]. And a recent report provides an algorithm for CAH screening using second-tier steroid analysis. The reference ranges for newborns at 4–6 days are also determined [538].The history of certain other NBS conditions and procedures has also been recently reviewed: the discovery of galactose mutarotase (GALM) deficiency in Japan [539]; the measurement of glycosaminoglycans by LC-MS/MS for mucopolysaccharidoses [540]; the development of second-tier LC-MS/MS as part of screening for acylcarnitines, acylglycines, amino acids, and organic acids [541]; the introduction of NBS for ALD and peroxisomal disorders in Japan [542]; and DBS screening for glycogen storage disease type 1a [543,544]. Several studies of MS/MS detectable disorders have also been reported: the clinical and genetic characteristics of patients with mild hyperphenylalaninemia [545]; changes in the frequencies of genetic variants of VLCADD since the implementation of ENBS [546]; the discovery of an unexpectedly high incidence of patients with mild propionic acidemia resulting from a common variant [547]; strict therapeutic observation to prevent sudden death in infants with CPT2 deficiency even if the child appears to have a mild clinical case [548]; CPT2 deficiency does not always have high sensitivity and specificity at the screening level, requiring retesting in a relatively large number of cases. Recently, Japanese researchers have identified markers with high sensitivity and specificity for the detection of CPT2 deficiency by MS/MS [549]; and a pilot study that examines the detection of specific subtypes of MMA and HCY [550]. The long-term outcomes of Japanese adult patients with both HCY [551] and PKU [552] were compared before and after the introduction of NBS, thus demonstrating the importance of lasting strict management in adult life.NBS methodology studies have included the report of a simple screening method to effectively detect isovaleric acidemia by distinguishing isovalerylcarnitine (i-C5) from pivaloylcarnitine (p-C5) by flow-injection MS/MS [553]. There have also been several reports describing NBS for LSDs: the use of gene analysis with NBS for the definitive diagnosis of PD [554]; a study of the frequency of infantile onset PD screening in Kumamoto and Fukuoka prefectures from 2013–2020 preliminary to expansion throughout Japan [555]; a review of the past, present, and future of NBS for mucopolysaccharidoses [556]; a discussion of NBS screening for Fabry disease in western Japan [557]; a study of novel Fabry disease-associated pathogenic variants in Japanese patients detected by newborn and high-risk screening [558]; and a study of DBS detection of bile acid metabolites for bile acid synthesis disorders, Zellweger spectrum disorder, and Niemann–Pick type C1 and the possibility of NBS [559].There is increased interest presently in adding SMA to the Japanese screening panel and several reports informing NBS for SMA have recently been published: a brief review of the current status of SMA treatment and the value of SMA NBS with new drugs [560]; a report on the pilot SMA screening project in Osaka confirming the usefulness of their workflow [561]; a report on pilot NBS using data from Osaka and Hyogo provinces to estimate incidences and to confirm the need for SMA NBS [562]; a review of disease history, treatment options, and NBS algorithm, along with the results of 1 year of SMA screening in Kumamoto Prefecture [563]; a report on 18 months of NBS for SMA in Hyogo Prefecture [564]; validation studies of DBS procedures for detecting SMA carriers suitable for implementation in NBS laboratories [565,566]; and the results of a new simple screening system based on DNA melting peak analysis applicable to a real-world NBS program for SMA with or without PCR equipment [567]. Looking to the future, there are two recent reports on the use of NGS in NBS: the use of 24 causative genes for NGS following a positive screen with TSH [568]; and the use of a 349-gene panel to detect genetic mutations associated with primary immunodeficiencies (PIDs) following a positive screening result using TREC/KREC analysis for SCID [569].Recently, a model for the quantitative assessment of NBS in Japan was developed using the analytic hierarchy process [570]. The emergence of new target NBS conditions such as SMA, LSDs, SCID, or ALD in newly expanded NBS may require a re-evaluation of the hierarchical disorder assignments.
- Korea (South) [Democratic Republic of Korea]—NBS for IEM was adopted by the Ministry of Health and Social Affairs for low-income families in 1991 and expanded to cover all newborns in 1997. There are currently 11 conditions included in routine NBS [571]. Recent publications have dealt with the implementation of NGS in NBS and laboratory issues. A doctoral dissertation in 2019 focused on development of a target NGS panel for NBS [572], which led to a report on the implementation of a 198-gene NGS panel at a hospital in Seoul and its potential to aid in reducing false-positive screening results [573]. Also addressing false-positive screening results was a study of their effect on parents through an analysis of NBS posts regarding NBS from an online parenting community [574]. Other recent publications have focused on laboratory issues. An online report of the annual external quality assurance for the public screening laboratory is published annually [575]. The Korea Research Institute of Standards and Science (KRISS) has developed a limited number of DBS Certified Reference Materials (CRMs) to enhance the reliability of NBS testing, with plans to develop more CRMs for other NBS markers [576]. A report of an issue with a commercial FIA-MS/MS procedure sought to alert product users of the potential problem and its solution [577].
- Laos—Sustainable NBS is under development in Laos. Previously, a 2008–2010 pilot project in the large maternity hospitals in Vientiane utilized laboratory services in Hamburg, Germany to demonstrate the feasibility of NBS for CH and CAH in Laos. The availability of cell phones facilitated retesting follow-up; however, early discharge and physician knowledge were identified as challenges [578]. In 2019, the German group once again assisted with initiation of NBS, but limited to CH. Local laboratory capability was established, workshops were held for health personnel, and screening was again established [579]. Screening with financial support from Else Kröner-Fresenius-Stiftung, a German foundation, is scheduled to continue through 2024 after which time it will require local support [580].
- Macau SAR, China—NBS for G6PD began in 1977 and since then it has expanded slowly in collaboration with other programs, including the Shanghai Xinhua Hospital NBS laboratory. Specimens from Macau babies born in Hong Kong are routinely analyzed in the Xcelom laboratory in Hong Kong [581].
- Malaysia—NBS in Malaysia began with cord-blood screening for glucose-6-phosphate dehydrogenase deficiency (G6PD deficiency) in 1980. In 2003, the Ministry of Health Malaysia implemented a nationwide, stepwise, congenital hypothyroidism (CH) screening program for all babies delivered in government hospitals [582], including a pilot project with MS/MS [583]. While all government maternity facilities offer G6PD and CH screening, ENBS is available only at four main centers on request (i.e., it is not compulsory) and a lack of awareness among clinicians and laboratory diagnostic facilities has likely resulted in under-reported cases [584]. Researchers have taken advantage of MS/MS technology to review and develop reference ranges for Carnitine-Acylcarnitine Translocase (CACT) deficiency and Carnitine Palmitoyl Transferase 2 (CPT II) deficiency [585]. Other research has centered on experiences in applying NGS to screening for thalassemia [586], validating a POC screening test for G6PDD [587], the need for additional training when setting up a new condition based on experiences with CH in 12 government hospitals and 20 health clinics [588], and the need for a mandated IEM-related course for certain healthcare disciplines at the university level [589].
- Mongolia—With some support from the IAEA, pilot NBS for CH began in Mongolia in 2000. The program has grown from three hospitals in 2000 to five city hospitals and five rural hospitals by 2023 primarily in central Mongolia [590,591]. During this period, more than 44,000 infants were examined for CH and CAH, and about 130 children were detected early and monitored. Also, the G6PD pilot study revealed a prevalence of 23.5%. In early 2023, a second laboratory (Center for Newborn Screening and Diagnosis) was established, and its scope of activities expanded. In Hovd (western border) and Dornod (eastern border) provinces, NBS branch centers were established and, in these provinces, in addition to CH and CAH, screening for G6PDD, GAL and CF was started. Beginning in 2024, health insurance will cover NBS for the five diseases. The NBS program continues to grow slowly, and the screening team is working to overcome sustainability challenges [591].
- Myanmar—Despite having participants in some of the IAEA regional NBS activities in the late 1990s, a sustainable public NBS program does not yet exist in Myanmar. There is some NBS availability in the private sector. There is interest in conducting a formal pilot study for at least CH.
- Nepal—There is currently no recognized NBS program in Nepal, although some screening is available in private settings. There have been at least 2 pilot studies in recent years, and while providing incidence information for various conditions and demonstrating that NBS is possible, no program has yet to be sustained: a pilot in collaboration with a NBS laboratory in Switzerland to lay the foundation for NBS and determine reference ranges for various analytes [592]; and a pilot collaboration between Paropakar Maternity and Women’s Hospital and a commercial NBS laboratory in India (NeoGen Labs Pvt. Ltd.; Bangalore, Karnataka, India) that confirmed the feasibility of NBS [593]. The latter pilot suggested that CH, CF, SCD, and LSDs should be considered for screening, although specimen transport issues slowed reporting on occasion. Inclusion of these conditions is supported by case reports and reviews in the literature: a summary of case reports of metabolic conditions, which also suggests inclusion of G6PD deficiency and Wilson’s Disease [594]; and a study of Hb S/S in the Tharu population, an indigenous sub-population living in a malaria endemic area in west Nepal [595].
- New Zealand—NZ was one of the first countries to establish a national NBS program. While it functions independently, NZ enjoys a collaborative NBS relationship with Australia as part of the Human Genetics Society of Australasia. Several significant updates to screening NZ protocols and policies have been published since our 2015 report. In 2015, new evidence was assessed by clinical experts and others, and it was decided that screening for six carboxylase deficiencies (3MCC; HMG; MCD; BKT; 3MGA; 2M3HBA) had no clinical benefit for the child and therefore NBS was discontinued [596]. In June 2017, screening for CUD was also discontinued due to screening sensitivity and treatment/safety concerns [597]. NBS for SCID was initiated in December 2017 [598].In addition to reviewing the factors affecting screening accuracy for CAH in NZ [125], considerable attention also has been given to improving the detection of CAH by including LC-MS/MS second-tier testing [599,600,601,602,603]. Similarly, attention to NBS system improvements in CH screening has been documented: adjustments in the TSH laboratory protocol improving case detection and PPV [604]; examination of the effect that demographic variables (e.g., ethnicity and age at time specimen collection) have on newborn TSH levels [605]; review of the actions to take when obtaining discordant results (case study) [606]; and caution against reducing TSH screening cutoffs to levels at which the diagnosis may not offer long-term benefit for those detected (TSH values less than 15 mIU/L (whole blood) found not to be associated with long-term hypothyroidism or cognitive impairment in NZ) [607].NBS reporting protocols are expected to result in clear and concise information exchange. A recent report in NZ emphasized that, to enable meaningful comparisons, CF screening reports should include the screening algorithm and target along with clarification of the steps in the screening pathway included in the assessment, the screening algorithm, and screening target [608]. To encourage faster recall returns for follow-up testing overall, a structured protocol for follow-up of inadequate and borderline positive NBS (including text messages to specimen submitters) was recently introduced [609].Other reports related to currently screened conditions included a review of the risk of having GALT if NBS results indicate borderline-positive galactose metabolites [610], and an assessment of the genotype–phenotype correlations in CPT1A deficiency detected by NBS, a condition more prevalent in Pacific populations (e.g., NZ and Hawaii) [611]. A study of the potential for NBS for neonatal hypoglycemia also has been recently reported with a call for urgent research to determine the optimal method of screening and which infants would benefit from screening and treatment [612]. The public health agency Te Whatu Ora announced the addition of SMA to the NBS panel in September 2023, with its implementation to take 12 months [613].
- Pakistan—NBS in Pakistan continues in its developing stage. Only one prefecture, Sindh, in the southeast has an NBS law to establish and integrate a sustainable NBS system within the public health delivery system, which became operational in all 29 Sindh districts on 1 January 2020 [614]. Several reviews of NBS operations and their impact on implementation in Pakistan have recently been published: a review of CH and the high burden of disease in Pakistan supporting a national NBS program [615]; a systematic review of CH and a plan for NBS implementation in Pakistan [616]; suggestions as to how a NBS system might best be organized and the conditions that might be a part of a Pakistan screening panel with CH as a prototype and then extending the panel to include CAH, BIO, GAL, G6PDD, SCD, [617]; and a review of why and how screening systems are configured [618]. Encouraging the expansion of NBS efforts around the country, in 2019, Aga Khan University, in collaboration with the Pakistan Society of Chemical Pathology, organized a meeting of genetics researchers, pathologists, and child health specialists from public and private sector organizations to launch an advocacy group, the Pakistan Inherited Metabolic Disease Network (Pak-IMD-Net). This group intends to overcome research challenges, medical education, and clinical practice related to IEMs and to encourage policy reforms to expand access to NBS [619].Recent project reports seeking to determine baseline information on screenable conditions found cases of HCY, MPS, and GAMT through biochemical screening of intellectually disabled patients [620]. Reports of reference range studies have included a multicenter study of TSH [621], a study of BIO [622], and a DBS study of various amino acids, succinylacetone and acylcarnitine [623]. Looking to the future, a study of an international partnership with an overseas facility performing NGS testing for PID patients suggests that resource-limited regions may require immunophenotyping and NGS in rather than TREC analysis, a screening strategy that can be met with through global partnerships [624]. Considering quality improvement of screening where it exists locally, NBS awareness in a hospital performing NBS for CH was found deficient in an audit of short-term follow-up with TSH results [625], a study of pregnant women found a similar lack of understanding about NBS resulting in fewer screens [626], and another study of parents found an increased knowledge from proper counseling led to better uptake NBS [627].
- Philippines—Since beginning as a pilot in 1996 and becoming sustainable through a national law in 2004, the Philippine NBS program has continued to grow in both coverage and disorders included in screening. Reports of expansion include successful implementation of screening for HGB [628] and the addition of MS/MS screening [629] so that the current program includes 29 conditions and newborn coverage exceeds 90%. To improve the quality of the overall program, a Philippine performance evaluation and assessment scheme (PPEAS) was developed as a means of defining responsibilities for various program components (e.g., NBS specimen collection facilities, NBS screening centers, etc.) [630]. Continuity clinics were organized and formalized to provide continuing long-term care assessment [631]. To maximize NBS acceptance, a hospital assessment of maternal utilization of NBS recommended that hospitals assign specific hospital staff to actively offer and be responsible for NBS prior to discharge [632]. A lack of knowledge about the roles of physicians, nurses, and medical students within the NBS were evident in a recent survey suggesting a role for enhanced NBS education among health professionals [633]. Most recently, a review of the challenges in sustaining NBS in the Philippine NBS program has been published [634].
- Singapore—NBS for G6PD deficiency began in 1965 followed by CH screening 25 years later. Universal hearing screening and screening for multiple IEMs were introduced as national programs in 2003 and 2006, respectively, with current NBS coverage at almost 100% without a screening law. A comprehensive review of the history of NBS and other screening activities with newborns was published in 2021 and notes the possibility of Singapore becoming a screening laboratory center for other developing programs in the region (e.g., Brunei, Myanmar, Laos, and Cambodia) [635]. An editorial discussing the history of NBS for CH also was recently published [636] along with an article advising on some of the issues related to cord blood screening for CH (e.g., mode of newborn delivery and TSH measurement method) [637]. In 2018, screening for SCAD and IBDD were removed from the screening panel. In 2019, NBS was expanded to include five additional disorders (CAH, BIO, SCID, GAL, CF). Along with the NBS for SCID came questions about its implementation in settings where BCG (bacille Calmette-Guerin) vaccination was given to newborns. The results of implementing SCID NBS in Singapore were published in 2022 and showed that patients with transient non-SCID T-cell lymphopenia and no underlying primary immunodeficiency can tolerate the BCG vaccination [638].
- Sri Lanka—NBS for CH using DBSs was introduced in 2006 and a regional screening center was established in 2008. In 2010, NBS began for all newborns in the Southern province, which led to financial commitments from the MOH. Program expansion to other provinces continued in 2016 and today the program is sustained and reaching over 95% of newborns in line with the National Strategic for Plan for Maternal and Newborn Health, 2017–2025 which set a goal of >95% of newborns screened annually for CH [639]. A 2018 report outlines the early history of the program and describes an audit process for five provinces (Southern, Uva, Sabaragamuwa, Central and Eastern) describing the need for improved turnaround times and community awareness [640]. A 2021 cost-effectiveness study confirmed the value of NBS for CH [641]. Another 2021 report described the negative health effects of CAH over two decades of unscreened newborns and sought to emphasize the need for adding CAH to the screening panel [642].
- Taiwan—A report on the history of NBS was published in 2019 [643] and updated in 2023 [404]. NBS reaches all newborns, is free to those in need, and subsidized for others. Perhaps the most progressive screening program in the region, the Taiwan NBS program was the first to add PD in 2005, and program researchers have contributed significant knowledge about both infantile onset—and later-onset PD through the years [644,645,646]. Research into various multiplex assays for Pompe and other LSDs recently have included a report of screening 70,000 newborns with an 8-plex assay [PD, Fabry disease, Gaucher disease, MPS-I, MPS-II, MPS-3B, MPS-4A (Morquio syndrome), and MPS-6] [647], and evaluation of initial cutoff values, rates of screen positives, and genotypes when testing over 100,000 Taiwanese newborns for MPS-I, -II, and -6 [648]. A long-term follow-up study of MPS-II from April 2015 until April 2022 provided baseline data on cases for future comparisons regarding treatment and outcome [649]. Research studies on a number of other NBS have also recently been published: a study of ALD showing a high incidence of null variants identified through NBS [650]; a report of the first 50,000 Taiwanese newborns screened for DMD [651]; an assessment of the significance of compound motor action potential (CMAP) amplitude in patients identified through NBS [652]; a report on second-tier molecular testing to improve NBS detection of citrin deficiency [653]; a review of the improved prognosis in early-identified newborns versus later-onset symptomatic infants with neonatal intrahepatic cholestasis caused by citrin deficiency (NICCD), supporting the need for NBS [654]; a report clarifying the etiology of false positives in NBS for citrullinemia. [655]; and a pilot study of a matrix metalloproteinase-7 as a possible screening test for biliary atresia using DBS collected at 3 days of age [656].
- Thailand—Neonatal screening for phenylketonuria (PKU) was introduced as a pilot project in Thailand from 1992–1995, and mass screening was started in 1996 by the Department of Medical Sciences, Ministry of Public Health. [657] NBS is now a mandated program under universal health coverage reaching over 95% of Thai newborns for the past 10 years. There is no parental written consent for NBS, but consent is required to share the leftover bloodspots for future research purposes. The National Health Security Office (NHSO), in October 2022, launched universal healthcare coverage ENBS for 40 treatable inborn metabolic diseases. Currently, there are seven NBS centers covering about one-third of Thai newborns for ENBS using MS/MS. The expansion of new NBS centers and the increase in capability of the existing centers is ongoing and seeks to achieve the government’s goal of 100% coverage by the end of 2024. These NBS centers have become a national network alongside seven rare disease centers, with administrative support from the Thai NHSO and the Ministry of Public Health (Department of Medical Science and Department of Health). The ENBS network meets every 3–4 months to encourage knowledge sharing, mutual assistance, and development of new NBS centers. They also share clinical geneticist consultants and confirmatory tests such as plasma amino acid, urine organic acid and genetic testing, as needed [658,659]. A 2018 report from southern Thailand found a significant increase in CH case detection after the implementation of NBS [660]. Prior to launching the national MS/MS program, a study funded by NHSO in Bangkok showed that Siriraj Hospital could provide ENBS to the 15 public hospitals in Bangkok [661]. A recent assessment of the knowledge and attitudes of parents regarding NBS found a generally favorable outlook but a need for increased awareness and education about the program [662].
- Việt Nam—Since 2006, the government has provided NBS for CH and G6PDD as part of government-paid healthcare. Other tests for a fee may be available, particularly at international and referral hospitals (typical listing shown in reference) [663]. The largest maternity and children hospital in the country offers NBS for CAH. Recently, a MOH spokesperson recently noted that in December 2020, the Prime Minister approved a program expanding the provision of NBS and prenatal screening, diagnosis, and treatment of some diseases and disorders until 2030 with a goal of “…90% of newborn babies for at least 5 of the most common congenital health problems …”. “By 2025, five regional screening and diagnosis centers will be upgraded, and two new similar centers will be built in the northern midland and mountainous region and the Central Highlands”. Facilities supplying NBS and other health services “…will be set up in 90% of the communal-level localities nationwide”. “Prenatal and newborn screening and diagnosis facilities will also be developed at obstetric and pediatric hospitals and general hospitals in 56 provinces and cities” [664]. Looking to the future, a recent study notes the lack of structured economic evaluations of NBS in LMICs to determine the best way forward and uses Việt Nam NBS as the case study [665]. Also looking to the future, studies of at least two screenable “rare” conditions in the country have been reviewed, both with founder effects linked to the Kinh ethnic population, and both noting the need for NBS: a report of MPS-I cases noting five patients in unrelated families from the same small community with the same previously unreported variant [666]; and a review of 10 years’ experience at a medical center in northern Việt Nam with 41 BKT deficiency patients [667].
3.2.3. Tabular NBS Data for Asia–Pacific
3.3. Europe
3.3.1. NBS Activities Focused on the European Region
Review of NBS Harmonization in Europe
NBS in Southeastern Europe
Other European Regional NBS Activities
3.3.2. NBS Activities within Countries
- Armenia—The Republic of Armenia implemented universal NBS for CH in 2012. In 2019, the screening records from 2012 through 2016 were analyzed looking at iodine nutrition status, including time dependence and regional variation and suggesting TSH as a means of monitoring iodine status [711]. In 2018, a Turkmenistan delegation visited to review NBS program operation as a possible model to be copied [712].
- Austria—The National Austrian Newborn Screening Program for inherited metabolic and endocrine disorders was implemented in the late 1960s by the Federal Ministry of Health in collaboration with the Ministry of Finance and the Medical University of Vienna. In addition to a recent review of the 100-year history of the Austrian national birth registry of IMDs [713], a 50-year review of the Austrian NBS program is also available [714]. Recent discussions within the NBS community have focused on whether and how to introduce PAP into the CF screening protocol [715]. Additionally, the NBS program added SMA in 2021 and SCID in 2022 [716].
- Azerbaijan—A study of CH incidence in newborns reported in 2012 [717] and showed an incidence of 1:666, indicating a pressing need for NBS. In 2020, a study of hemoglobinopathies in newborns found the presence of Hb S, Hb D, and pathological genes for α- and β-thalassemia favoring the initiation of NBS for HGB [718], and a 2022 report detailed the data from a NBS study of GALT [719]. Other information on NBS is scarce. A 2022 UNICEF Country Report noted that during the last part of 2022, over 6000 newborns received NBS [720].
- Belgium—Rather than a national NBS program, two regional programs exist in the French-speaking and Dutch-speaking communities, each with slightly differing screening panels. A 2017 report detailed experiences in establishing and evaluating screening methodologies for PD, Fabry disease and MPS I using liquid chromatography–tandem mass spectrometry [721]. A 3-year SMA pilot in Liège begun in 2018 has now expanded to full-scale screening throughout the country [722,723]. A recent publication provides real-world cost-effectiveness information on SMA NBS in Belgium [724]. The experiences of 20 years of limited screening for SCD and the potential impact of adding it to the NBS panel were reported with the hope of adding HGB to the screening panels [725]. A 2020 study in Flanders evaluated the cost effectiveness of four NBS strategies for CF [IRT-DNA (immunoreactive trypsinogen, cystic fibrosis transmembrane conductance regulator (CFTR) gene mutation analysis), IRT-PAP (pancreatitis-associated protein), IRT-PAP-DNA, and IRT-PAP-DNA-EGA (extended CFTR gene analysis)], assessing whether each met the guidelines of the European Cystic Fibrosis Society [726]. A systematic literature review in 2021 identified seven neuromuscular disorders that might be candidates for addition to the Belgium NBS program [SMA, DMD, myotonic dystrophy type 1 (MD 1), PD, ALD, MLD, and KD] [727].
- Bulgaria—While the extent of NBS in Bulgaria remains limited, studies have been initiated looking to future expansion. In 2017, a study was conducted that assessed attitudes and opinions on the potential use of whole-genome sequencing (WGS) with traditional NBS. Both pediatricians and geneticists considered selective WGS as a potential option for improving Bulgarian NBS [728]. The results of a pilot study demonstrating the feasibility of SCID screening were recently reported [729]. Cases of arginase deficiency in the Roma area point to the need for ARG screening in this region [730].
- Croatia—The Croatia genomics activities, including the NBS program, were recently included as part of an online workshop [731]. Building on a NBS program that began screening for PKU in 1978, the centralized testing laboratory at the Zagreb University Hospital Center recently added SMA screening for its expanding panel [732].
- Cyprus—The Cypriot neonatal metabolic screening program began started in 1990 as a civil society effort by a charity, the Centre for Preventive Paediatrics [733]. Ten-year data showed a higher prevalence of CH in Cyprus than in some of the surrounding countries. While a pilot was previously initiated for DMD, the current screening program continues to screen only for CH and PKU, with the Centre responsible for all aspects of the screening system (inviting parents to participate, identifying screen positives, diagnosing conditions, communicating with physicians, assessing long term outcomes, educating the public, and participating in public health policy formation.
- Czechia—Within the country, there are two clusters of laboratories for NBS, one in the Bohemia Region and another in the Moravia/Silesa Region. Each cluster has three laboratories (one for MS/MS, one for immunoanalytical, and one for second-tier genetic testing for CF, and screening has included 18 conditions since 2016 [734]. Pilot testing for SMA and SCID began in January 2022 in Prague and Moravia [735]. Recent research has focused on the epidemiology of diseases detected by NBS [736], with particular emphasis on low birthweight neonates [737] and the growth patterns associated with NBS-detected CAH [738]. A study of the way parents are informed about NBS and the variables associated with their awareness has resulted in several new educational measures including seminars for healthcare providers and the development and distribution of new educational materials [739].
- Denmark—In 2020, researchers described expanded NBS in Denmark, the Faroe Islands and Greenland (the reach of the Danish NBS program), by reviewing a project that began in 2002 and ended in 2019 [740,741]. In the interim CF (in 2016) [742] and SCID (in 2020) [743] became the 17th and 18th conditions added to the Danish NBS panel, and the first case of CF in a native Inuit was detected through NBS in Greenland [744]. In preparation for the addition of other conditions, development of a multiplex assay for SCID, SMA, and X-linked agammaglobulinemia (XLA) was reported [745] along with a MS/MS procedure for galactose-1-phosphate, reportedly superior to the traditional GALT enzyme analysis [746]. The addition of second-tier molecular testing to the Danish NBS program was described in a 2021 report along with the challenges and possible pitfalls of second-tier genetic testing [747]. Also noting the value of second-tier testing was a 10-year review of salt-wasting cases of CAH detected by NBS [748].The secondary use of NBS specimens for other purposes after NBS is a topic of discussion internationally. A 2019 report sought to identify and criticize some of the research uses of specimens contained in the Danish NBS biobank [749], which elicited a response describing the history, organization, and beneficial uses of specimens in this biobank, [750]. A 2022 report discussed the experiences of mothers with both collection and uses of NBS specimens noting their general acceptance of research re-use of the specimens [751]. And a 2023 report sought to provide an understanding of the advantages and possibilities of biobank research in Denmark given the huge number of specimens, the presence of at least a dozen other biobanks, and a unique civil registration number assigned to each Danish citizen [752]. Recent examples of biobank usage include a joint study with the California biobank to develop and validate a new commercial kit for creatine kinase to screen for Duchenne muscular dystrophy (DMD) [753] and LC-MS/MS-based studies of untargeted metabolomics in NBS specimens to define metabolomic profiles for autism [754]. Analyses of specimens in the Danish NBS biobank have been used retrospectively for other epidemiological metabolomic studies [755].
- Estonia—The NBS program in Estonia was re-engineered in 2014 as part of a doctoral project [756]. Vitamin B12 deficiency was included as a NBS pilot and after 2 years, and an incidence of 1:3000, was continued on the screening panel [757]. Several patient reviews have been completed in the past few years looking both to review the NBS program and to consider new conditions for screening. In 2018, a retrospective overview of PKU patients identified a high incidence of PKU estimated at 1:6700 [758]. In addition to a survey of SMA cases to define their extent in the population preparatory to possible inclusion on the NBS panel, a 30-year study of the prevalence of IMDs in Estonia was initiated to define both their prevalence and the effectiveness of new methods for diagnosis [759,760].
- Finland—A regional SCID pilot was initiated in 2019. The Council for Choices in Health Care in Finland (COHERE Finland) recommended NBS for SCID in 2020 and SCID was added to the national NBS panel during 2021–2022 [761]. Similarly, SMA was recommended but not implemented. A proposal for SMA screening has been sent to the Ministry of Social Affairs and Health and is awaiting evaluation [762]. A 2020 report on TYR-I showed improved prognosis from NBS [763]. A 2023 report on the incidence of CH and the proportions of mild, moderate, and severe disease, over the 24-year study period, showed no difference in incidence [764].
- France—NBS began in France in 1967 and its history and status recently have been reviewed [765,766,767]. NBS is mandatory and French overseas territories are included in the screening program. There are 17 regional screening centers (12 mainland and 5 overseas), each associated with a university hospital and a regional health agency. There is a national coordinating center, which is overseen by an epidemiological commission that monitors program effectiveness and a biological commission that defines the screening algorithms, including pertinent biomarkers and cutoffs. A national steering committee in the French MOH provides general NBS policies for NBS and recommended seven new metabolic conditions at the end of 2022 [768]. The history, outcomes, and analytical accuracy of other NBS conditions have been reviewed: PKU [769], CAH [770], TYR-I (with SUAC) [771], CF [772], CH (with an appeal to lower the screening cutoff) [773], and SCD [774]. A recent study of parents’ opinions about the NBS process found that there is a preference for more and better information about NBS during pregnancy and the information should focus on the non-mandatory nature of NBS and the need for informed consent for parents choosing to screen their newborns [775].A number of reports on CF screening recently have been published: a cost-effectiveness analysis that compared four CF neonatal screening strategies with or without DNA testing to assess the value of combining cost effectiveness and ethics evaluation in health policy development (NBS laboratory protocol) [776]; studies analyzing inconclusive diagnoses after a screen-positive test (CFTR issues) including gene-sequencing and genetic counseling [777,778,779,780]; and review of a centralized tracking process to optimize the CF screening program [781]. Similarly, NBS for SCD, presently a targeted condition based on the geographical origins of both parents (malaria endemic areas) and a family history of SCD, has been the subject of several reports: screening and case management [782]; possible harmonization of HPLC result interpretations by applying multiple of median cutoff and ratios [783]; evaluation of POC testing [SickleSCAN™ (BioMedomics, Morrisville, NC USA)] [784]; and a study of targeted screening failures over the years (pushing for universal NBS for SCD—recommended by the Health Technical Agency since 2022) [785].As a prerequisite for addition to the French NBS panel, feasibility and cost-effectiveness studies must be completed along with assay validation. Studies supporting the addition of SCID include performance of the TREC assay for SCID [786], a review of the evidence supporting SCID screening in France [787], and the status of screening implementation, including lessons learned [788]. Recent studies supporting the addition of other conditions also exist: a systematic review of NBS test accuracy for TYR-I using succinylacetone [789]; a world view of practices and pitfalls in defining an algorithm for primary carnitine deficiency screening [790]; and a review of costs and quality of life of patients with SMA [791].Two reports concern conditions already on the screening panel: for CAH, a rise in 17-OHP may be observed in newborns exposed to drugs [792]; and for CH, a change in the screening algorithm has been proposed to allow the detection of hyperthyroidism [131]. While there are a number of studies concerning the addition of NBS for cCMV, each concerned saliva testing instead of blood and will not be discussed here. Finally, a 2021 report provided an overview of the biological techniques currently used for NBS and suggested technological changes were inevitable [793].
- Germany—German NBS is coordinated through a national directive of the Federal Joint Committee (Gemeinsamer Bundesausschuss). Regional pilot studies looking at feasibility, diagnostics, and health benefits are required for new conditions [794]. Recent reports provide an overview of the current NBS program and some of the ongoing issues: a report detailing the legal background for NBS and related issues [795]; the potentials and limitations of second-tier testing in NBS [796]; a report detailing each of the NBS conditions currently included in NBS and the intricacies of their inclusion and follow-up [797]; a review of 20 years of screening for CAH and CH emphasizing the need for sustainable case registries for all screened conditions to better measure screening outcome [798]; and a thought provoking discussion on genomic medicine and the challenges and suggestions for incorporation of genomic NBS into public health programs, including how to balance expected benefits against possible harms to children and their families [799].The addition of SMA and SCD to the screening panel in 2021 has resulted in several reports. For SMA, the focus has been on pilot testing data and the logistics of national expansion, the additional challenges of screening and diagnosis for a new screening condition, and the impact on parents [800,801,802,803,804]. For SCD, a brief narrative reviewing the pathway for adding SCD to the screening panel has been published [805]. Additionally, a report reviews the value of NBS for HGB in recognizing the current immigration patterns across Europe, the needs of the population, and the value of increased case recognition [806]. Recognizing the need to address the transition from pediatric to adult medicine as the screened population ages, a guideline for transition has also been developed [807]. A 2022 report provides pilot testing data for a multi-analyte MS/MS procedure that simultaneously detects SCD, BIO, and TYR-I [808], and a 2023 report provides information on a combination multiplex qPCR and MS/MS procedure for simultaneously detecting SCD, SMA, and SCID [809].Recent reports on other conditions on the screening panel include CF (added 2016) and SCID (added 2019). The SCID report is a review of the first four years of NBS on the largest SCID screening population in Europe noting the apparent success of the TREC assay procedure in case detection [810]. CF studies documented the feasibility of using an IRT/PAP screening protocol [811] and a recent publication reported on the influence of season, storage temperature, and time of sample collection on PAP screening algorithms [812]. A DNA-based third tier for all children with a screen-positive PAP was shown to significantly improve the PPV [813]. The current CF-NBS algorithm combines a three-step protocol (IRT/PAP/DNA) with a “safety-net”, lassifying an ultra-high IRT (>99.9 percentile) as a positive screen directly. The PPV using this algorithm is about 0.2 and adaptation is currently under discussion. Parents’ satisfaction with a more centralized system of confirmatory CF diagnostics has also been reported [814]. Two other reports reviewed the outcomes of screening cases detected over time. One looked at clinical outcome of cases reported between 1999 and 2016 [815], and the other reviewed the annual screening quality reports between 2006 and 2018 [816]. Both found that NBS was successful. As noted in other reports, tracking and case registries were a program deficiency. Other pilot studies have included the rare LSD, metachromatic leukodystrophy [817], and cystinosis, a rare autosomal recessive systemic disease with high morbidity and mortality, both using NGS methodologies as part of the screening algorithm [818].The possibility of NBS for vitamin B12 deficiency has also been reported including strategies, results, and public health implications [819], outcomes of early treatment resulting from NBS [820], and the results of screening 1.2 million newborns for MMA (and vitamin B12 deficiency) using a two-tier screening strategy [821]. A collaborative pilot study conducted at three NBS sites in Germany evaluated 18 candidate disorders for German NBS, many using second-tier strategies, with the majority found suitable for inclusion on the screening panel [822]. Several reports have targeted IVA NBS (added in 2005). Not only have researchers looked at clinical outcome and the issues raised by identification of mild variants [823], but also difficulties with high false-positive rates due to maternal use of pivaloylester-containing antibiotics, which is reduced by the use second-tier testing to differentiate C5 isomers [824].Machine learning (ML) is becoming increasingly popular as a second-tier digital test for testing sensitivity improvement and a literature review was recently published [825]. ML has now been used to improve the reliability of the IVA screening by combining linear discriminant analysis and ridge logistic regression to maintain 100% sensitivity and reduce false-positive screens by almost 70% [826]. Despite the successes of the German NBS program, there are still issues that can be improved (PPV, tracking, education, etc.) as noted in the referenced comments [827]. A recent report reviewed the requirements for NBS infrastructure and screening procedures through a literature search as part of a project “on the quality and shortcomings of the NBS pathway in Germany” [828].
- Greece—NBS dates to the mid-1970s and a detailed look at NBS expansion in Greece exists [829]. A recent publication provides a mini review of the psychological impact on parents of children who have received an inconclusive diagnosis for cystic fibrosis following newborn screening [830]. Looking to the future, a commercial venture to contact at least 1000 families for inclusion in a WGS project looking at over 400 NBS conditions has been announced and is moving forward with plans to reach all Greek newborns by 2027 [831]. The project, BeginNGS, includes the genomic data platform Lifebit (London, U.K.) in collaboration with PlumCare RWE (Delaware, USA), the Rady Children’s Institute for Genomic Medicine (San Diego) and the National Organization of Public Health (Greece).
- Hungary—Expanded NBS was introduced in Hungary in 2007 and experiences in detecting vitamin B12 deficiency and in diagnosing neonatal/infantile vitamin B12 deficiency have been described [832]. A collaboration between universities in Hungary and Germany identified potential interferents in the analysis of methylmalonic acid, which has not previously been reported and represents a pitfall in clinical diagnostics and NBS [833]. Because no commercial quality control material is available for the second-tier NBS CAH assay, a 2021 report compared five different QC preparation approaches used in routine diagnostics for CAH on the concentrations of cortisol, 21-deoxycortisol, 11-deoxycortisol, 4-androstenedione, and 17-hydroxyprogesterone in dried bloodspots [834]. Also in 2021, a report detailed the issues relating to NBS for SMA in Hungary, with particular emphasis on the baby Zente case and the need to have routine NBS for SMA [835].
- Iceland—A 2023 thesis describes a completed CF screening pilot suggesting the addition of CF to the NBS panel in the near future [836]. Two metabolomic studies were reported in 2021. One found that low birth weight and extremely macrosomic newborns showed dissimilar metabolomic profiles when compared to appropriate-for-gestational age neonates [837]. The other estimated potential differences in neonatal metabolomic profiles at birth and at the time of NBS by delivery mode and found small differences, if any, and concluded that the mode of delivery does not affect the results of NBS [838].
- Ireland—The NBS program in Ireland is one of the oldest national programs in the world, having started with PKU in February 1966 [839]. To facilitate NBS expansion systematically and logically, a 2021 guide, “Review of processes in use to inform the expansion of newborn bloodspot screening programmes”, was published [840]. The importance of early diagnosis and skilled multidisciplinary team management is highlighted in another 2021 report showing the value of NBS by reviewing outcomes of MSUD patients detected through NBS since 1972 [841]. In 2022, the Irish NBS program guide featured summary information on adenosine deaminase-deficient SCID (ADA-SCID), which was added in 2022, and updated program consent and feeding requirements [839]. The National Screening Advisory Committee (NSAC) previously recommended inclusion of SCID (not just ADA-SCID) to the NBS panel and in early 2023, this recommendation was accepted by the MOH [842]. The NSAC has also noted the importance of SMA as the next condition and elevated the consideration of SCD, TYR-I, and BIO for the screening panel while suggesting that NBS for PD was not a priority due to its low incidence. Discussions on the prioritization of some 30 other conditions are ongoing.In 2020, a publication on CF NBS, initiated in 2011, brought accolades from the CF community for having “achieved a model CF program” [843,844]. A separate report provided evidence that NBS improved growth, reduced hospitalization for acute episodes, and delayed P. aeruginosa acquisition to age 3 [845]. Beginning first with CF and then expanding to NBS in general, a series of studies from 2016 to 2019 sought to determine the level of knowledge about NBS in the lay community as an aid to developing better educational outreach [846,847,848]. Prior to inclusion of SCID on the panel earlier this year, a study showing its high prevalence in Ireland and the need for NBS was reported in 2021 [849]. Studies supporting the addition of other conditions have also been recently published including one on CAH and another on metachromatic leukodystrophy [850,851].
- Italy—Expanded NBS became required by law in 2016 in all 20 autonomous regions and includes over 40 screened conditions paid by the government. The law not only lists the conditions included in screening, but also other program requirements (consent, specimen collection, result communication, follow-up, etc.) [852]. Further description of the expanded program and the results of the first two years of testing from 15 of 16 screening laboratories are provided in a 2022 report. Also included is a description of the national landscape regarding screening, molecular confirmatory testing, and case management [853]. Additional information on the history and legal aspects of the NBS program is reviewed in a recent editorial [854].Variations in screening and diagnosis protocols between regions have resulted in a number of regional reports on screening and diagnosis outcomes. Since 2020, at least three studies have focused on BIO: a 12-year data review showing a high incidence in the Tuscany and Umbria regions [855]; a study describing experiences in disease management (and unanswered questions) in Verona [856]; and a report documenting a higher rate of partial BIO than predicted in Abruzzo [857]. CF screening experiences also have been reported: improved program efficacy when DNA testing was added to the screening protocol in Tuscany [858]; a six-center study (Ancona, Brescia, Florence, Milan, Naples, Rome) of the psychological impact on parents receiving an inconclusive diagnosis following NBS [859]; and the impact of incorporating PAP into the screening protocol (IRT/PAP/DNA) to decrease screen-positive inconclusive diagnoses [860]. Several reports have focused on screening for various LSDs: 5.5 year review of NBS for Fabry disease in Northeast Italy including clinical, biochemical, and molecular features of detected patients [861]; 7 year review of NBS for PD in Veneto (Northeast Italy) including infantile- and late-onset disease [862]; a review of MPS-I screening in Padua and the inclusion of a second-tier testing to improve specificity [863]; and a comprehensive review of NBS for MPS-I [864]. An earlier report detailed the Tuscany experiences with NBS for PD, Fabry disease, and MPS-I [865]. Other disease reviews have also been recently published: an overview of galactose metabolism, molecular genetics, NBS and novel treatments potentially able to prevent long-term disease complications [866]; and a review of DMD including a proposed NBS pilot project protocol [867].Decreasing unnecessary recall to resolve a positive or inconclusive screening results is an ongoing goal of NBS programs and a discussion of various approaches to its accomplishment has been noted [868]. Several reports of improved screening sensitivity have been published: adding DNA or PAP to the screening protocol in Tuscany (previously mentioned) [858,860]; second-tier testing for CAH in the Northeast region of Italy [869]; and changing the CH cutoff in Western Sicily (to decrease late-diagnosed cases) [870]. Additionally, improvements in CH case detection have been noted in certain cases: use of a lower cutoff on second specimens (when two specimens are required) [871]; and analysis of a second specimen in the case of twins [872]. Preterm newborns are at higher risk for CH and may require a different targeted screening protocol. A study of births in Piedmont indicated that their current two-screen protocol for all newborns was sufficient [873]. In the Apulia region, relocation of the screening laboratory to a more central location has also been shown to improve clinical care for CH through more timely initiation of treatment and improved case management [874].The results of pilot efforts aimed at expanding the NBS panel of conditions continue to be reported: SCD—first report of screening for an entire province [875]; SCD—the feasibility of a multicentric NBS program and epidemiology in two northern areas demonstrating a high percentage of Caucasian carriers (impossible to identify in targeted NBS [876]; ALD—a three-tiered pilot in Lombardy started in 2021 (FIA-MS/MS, ultra-high performance liquid chromatography–tandem mass spectrometry (UHPLC-MS/MS), and focused NGS (genetic confirmation) [877]; and SMA—a pilot in Lazio and Tuscany to determine the proper clinical assessments to systematically evaluate the possible presence of early neurological signs of SMA [878]. Other reports have included the successful inclusion of ADA-SCID [879] and purine nucleoside phosphorylase-deficient SCID (added earlier) [880] on the screening panel, detection of SCAD in family members as a result of NBS [881], mild elevations of citrulline detected through NBS [882] and a possible algorithm to guide the diagnostic process, and a pilot demonstrating the value of dried bloodspot analysis of cCMV in infants with hearing loss [883,884]. A recent report reviews the eight-year experience with screening and follow-up of approximately 250,000 newborns screened for four LSDs (PD, MPS-I, Fabry disease, and Gaucher disease) [885].
- Kazakhstan—NBS for PKU has been ongoing since 2007 and the 10-year history has been reviewed in the Russian language and describes an increasing coverage [886]. Kazakhstan has 16 regions and 3 megalopolises, with screening coverage of approximately 92% [669]. A recent report defines the limits of a study to review various IEMs detectable with LC-MS/MS to determine their frequency in Kazakhstan as part of the effort to expand NBS [887].
- Latvia—A recent report on the 25-year CF diagnostic data in Latvia, including the first report on NBS for CF has been published. Two and one-half years of NBS data were available and indicated a lower disease incidence than expected based on calculations from previous F508del carrier detection studies. There is concern about false negatives and the current IRT-IRT-DNA protocol may soon change to IRT-DNA-IRT [888]. A report on NBS for SMA reviewed implementation issues and improvements in a pilot study that showed the SMA 5q procedure employed could be applied to the whole of Latvia to facilitate early diagnosis and more effective treatment [889].
- Lithuania—NBS has been implemented at the Centre for Medical Genetics of Vilnius University Hospital Santaros Klinikos since 1975, testing for four diseases 3–5 days after birth. Funding is from the Compulsory Health Insurance Fund and NBS is not mandatory. Expanded screening for 30 or more conditions is available on request [890]. A project to compare TSH levels in newborns to evaluate iodine status showed mild iodine deficiency in some areas and a need to reevaluate the proposed satisfactory level of TSH vs. iodine level [891]. A 2021 review of CAH case detection showed 100% sensitivity and specificity of NBS in detecting classical SW CAH, but a poor (4%) positive predictive value [892].
- Luxembourg—NBS in Luxembourg was recently expanded to include screening for CF in addition to PKU (1968), CH (1978), CAH (2001), and MCAD (2008) [893].
- North Macedonia—As part of the MOH’s National program for mothers and children’s care, NBS for PKU and some 30 inborn errors of metabolism by LC/MS/MS has been ongoing in six larger delivery facilities across Macedonia since 2011 [894]. Earlier NBS for both PKU and CH have been documented [895]. Complementing earlier reports [896,897], data from the national CH NBS program over 20 years has recently been analyzed and the prevalence, along with geographic, and ethnic variations, reported [898]. Following a 6-month pilot, NBS for CF by IRT-IRT was introduced in April 2019, and 2-year data show a 4 times higher prevalence of Albanians compared to Macedonians (1:4530 vs. 1:1284) [899].
- Netherlands—NBS was initiated in 1974. The Ministry of Welfare and Sport defines NBS policy and is responsible for funding and facilitation of program coordination, which is a responsibility of the Center for Population Screening (CPS) of the National Institute for Public Health and the Environment (RIVM). An independent national scientific advisory body summarizes scientific evidence and provides policy recommendations. The framework for considering additions to the screening panel has been detailed in a 2021 report [902]. In consideration of professional stakeholder involvement in NBS, a 2017 project evaluated the inter- and intra-personal moral issues that may be encountered by health professionals in NBS, and the support required as they navigate complex NBS system logistics [903]. Multi-disciplinary stakeholder involvement in the NBS system is critical. A 2021 report looked at the perspectives of parents, policy makers and professionals regarding NBS expansion [904]. Studies have recently been reported concerning parents’ views on accepting, declining, and expanding NBS [905] and their perspectives on retention and secondary use of NBS specimens [906]. The Health Council of the Netherlands has specified criteria to assess the long-term harms and benefits of NBS, and in 2021, performed an initial assessment for 11 of the 25 conditions on the NBS panel [907]. In 2022, at the request of the MOH and RIVM CPS, an external consulting service prepared a recently published report on the future of NBS in the Netherlands [908].NBS continues to expand and an evaluation framework for adding new conditions was developed in 2015 when the program was considering expansion from 17 to 31 condition. This framework has been suggested as an ongoing model to assist in future expansion efforts [902]. Recently a report sought to define “treatability” as part of the evaluation framework. This process is envisioned as a starting point for better defining each of the criteria used in selecting screening disorders going forward [909]. Several reports contribute to documenting the process for adding SCID to the NBS panel [910], including preliminary SCID incidence findings [911], screening assay evaluation [912], screening cost effectiveness [913], reporting (or not) incidental findings [914], various screening strategies and their real life economic evaluation [915], and parents’ perspectives and societal acceptance of SCID screening [916,917]. Interestingly, the Health Council has been formally asked for advice on when “non-treatable” disorders might be screened and a report on their response has been published [918]. In addition to SCID, recent reports about other new conditions have included a data report with a recommendation for the addition of GALK [919], a cost-effectiveness study of NBS for SMA [920], two reports concerning the sex-specific NBS pilot for ALD [921,922], a retrospective study of referrals for primary carnitine deficiency in consideration of moving it from an incidental finding to a member of the screening panel [923], and a retrospective evaluation of the Dutch pre-NBS cohort for PA and MMA (what to aim, expect, and evaluate from NBS) [924].PA and MMA were added to the screening panel in 2019, and other recent additions have included GALK (2020), SCID (2021), MPS-I (2021), SMA (2022), and ALD (2023). The program now contains 27 target conditions and two incidental findings (sickle cell trait and primary carnitine deficiency). Evaluation studies ongoing include MPS-I, ALD (5-year study), 3-MCD 3-year study, and the data registry (10-year study) [925]. Recently, there have been several evaluations of different aspects of the NBS program: 11 years of NBS data for MSUD [926]; 11 years of data for CH [927]; 12 years of data for CAH [928]; 15 years of data for SCD [929]; and 10 years for VLCAD [930]. Laboratory-specific studies have also been reported: second-tier 21-deoxycortisol screening for CAH [931]; a 4-step screening protocol for CF (IRT-PAP-DNA-EGA) [932]; a reevaluation of NBS for TYR-I using succinyl acetone in view of a late-diagnosed case (false negative) [933]; and the use of thyroxine-binding globulin concentrations to improve screening for central CH [934]. The stability of analytes in stored specimens has also been reviewed with particular emphasis on instability of certain acylcarnitines [935]. The TSH concentrations in NBS specimens have been studied as a means of monitoring iodine status [936]. Looking to NBS in the future, NBS for CTX has been considered using both flow-injection and UPLC MS/MS [937], machine learning has been studied as a means of improving the PPV for CH NBS [938], and a 3-step study has been outlined as a means of exploring NGS techniques as a first-tier approach in NBS [939].
- Norway—In 2012, the Norwegian NBS program expanded from two conditions to twenty-three and the addition of two more conditions in 2018. A recent report describes the screening results, experiences with second-tier MS/MS methods, DNA testing, use of the Region 4 Stork (R4S)/CLIR post-analytical interpretive tool, and clinical outcomes and follow-up challenges after expansion. To continue program improvements, an appeal is made for coordinated international collaborations [940]. Also in 2018, approval of the use of residual NBS specimens for epidemiological research not related to NBS and the indefinite storage of residual specimens resulted in a critical review of the mechanism for deciding the changes and their extent. A perceived lack of sufficient involvement of ordinary citizens in the decision-making process, the presence of a national personal identity number, and an ambitious national biobanking system (including multiple central health registries) were all identified as contributing to possible “function creep” (serving purposes other than helping newborns) of the NBS program [941].Multiple studies concerning the feasibility and value of NBS for vitamin B12 deficiency were reported. A 2022 study evaluated the predictive value of NBS algorithms from Austria and Germany (Heidelberg) in detecting infants that were clinically diagnosed later with symptomatic B12 deficiency and found that neither could identify most infants diagnosed with symptomatic B12 deficiency after the neonatal period. This study also investigated whether being born in a hospital using nitrous oxide (N2O) as pain relief in labor may have had an impact on total homocysteine at NBS and found that N2O may impact total homocysteine at NBS [942,943]. A more recent study looked at the detection of B12 deficiency when second-tier dried bloodspot (DBS) analyses of total homocysteine and methylmalonic acid are included and concluded that NBS for B12 deficiency is not straightforward. Additionally, there is a need to address whether screening and treatment of maternal B12 deficiency during pregnancy to evaluate its value as a preventive strategy for B12 deficiency in both mother and baby [944].Other reports have included a review of the first 3 years of screening for CF (and the need to revise the IRT/DNA protocol) and the possibility of NBS for branched chain ketoacid dehydrogenase kinase (BCKDK) deficiency, which is linked to a neurodevelopmental disorder characterized by autism, intellectual disability, and microcephaly [945,946]. A report on a pilot using second-tier NGS with an amplicon based targeted gene panel has been shown to provide a rapid molecular diagnosis (or not) of SCID [947]. A recent report notes the value of Sanger sequencing for monogenic disorders caused by variants in one single gene or in a few genes and presents their Sanger methodology including primer sequences and the genetic test algorithms [948].
- Poland—NBS for SMA was approved in 2021 and has undergone staggered implementation across the country with the last provinces completed in 2022 [949]. A discussion of the disease and some of the diagnostic issues was published in 2020 and provides information that is potentially useful as screening policies are developed [950]. The Polish Vaccinology Association published the first recommendations for gene therapy for newborns who received live vaccination against tuberculosis, which might be helpful in other countries where live vaccine against tuberculosis is still used [951]. Looking to the possibility of further program expansion and building on experiences in Germany, where SCID was added in 2019, the first 14 months of a border collaboration to identify newborns with T and/or B immunodeficiency was reported in 2020 [952].Several reports of screening activities for other conditions have been reported since 2020. At least three reports related to CF NBS have been published: a study of the clinical characteristics of Polish patients with rare and novel CFTR mutations to determine their pathogenicity [953]; a 10-year review of the impact of NBS on clinical outcomes of pediatric patients in Lodz Voivodeship [954]; and an investigation into the clinical complications in children with late-diagnosed (false-negative) CF (with a reminder that, “in the presence of clinical symptoms, additional diagnostics must be implemented, in spite of the negative screening results”.) [955]. At least three reports related to screening for endocrinopathies have been published: a case studies of the value of NBS in detecting CAH absent clinical signs in two female newborns [956]; a review of the challenges of CH diagnosis and treatment in preterm newborns and the value of NBS [957]; and a look at the prevalence of hypothyroxinemia in newborns born before the 32nd week of gestation with a birth weight below 1500 g, with a suggestion to measure serum TSH and FT4 between the third and fifth day of life supplementary to NBS [958]. A report on BIO presented the molecular spectrum (profound and partial forms) of the disease in Polish patients and estimated overall disease prevalence to be 1:66,966 (1:178,577 for profound and 1:107,146 for partial) [959]. A study looking at pain management during the heel stick procedure found that over 60% of newborns felt no pain or discomfort and that the non-pharmacological interventions studied (breastfeeding, oral glucose dosing and non-nutritive sucking) were all effective pain management techniques [960].
- Portugal—The NBS program started in 1979 with screening for PKU, and CH was added in 1981. The program expanded to >99% national coverage within 10 years [961]. It is now a voluntary public health program screening for 28 conditions in a single laboratory with specimens collected on the 3rd day of life. A 2024 report reviewed the implementation and results of MS/MS expansion since 2004, including the importance of second tier testing for many of the metabolic conditions [962].Results of a 3-year pilot that included CF resulted in a recommended screening algorithm that included IRT/PAP/DNA [963]. CF NBS was added to the Portuguese NBS program in 2019. A report analyzing data from 1 or 5 CF centers and that the European Cystic Fibrosis Society (ECFS) recommends that NBS programs should aim for a minimum PPV of 30%, a minimum sensitivity of 95% and suggested that these criteria should be applied to a national program evaluation [964].Portugal has one of the highest incidences of Cbl C/D globally (~1:85,000). A 2017 report compared the genotype/phenotype of patients identified with Cbl C/D before and after implementation of expanded NBS with positive findings [965]. A 2022 report noted that, “… early detection through NBS may have prevented hematological abnormalities and irreversible neurological damage in infants with acquired vitamin B12 deficiency due to maternal vitamin B12 deficiency”, and that, “Acquired B12 deficiency should be ruled out before proceeding in a differential diagnosis of cobalamin metabolism deficits, methylmalonic acidemia, and homocystinuria” [966].A recent review on PKU and lipidomics noted that, “MS-based lipidomics is a promising approach to evaluate the effect of the diet restrictions on lipid metabolism in PKU patients, monitor their outcome, …, and find possible prognosis biomarkers” [967]. Another report gave a brief overview of the use of DBSs in lipidomic studies and noted the variables that can affect lipidomic analyses (DBS card matrix, hematocrit, specimen homogeneity, etc.) [968].
- Romania—NBS for phenylketonuria is carried out at the national level in five regional centers, must have the mother’s consent after explanation from her physician, and is funded by the MOH within the framework of the National Women and Child Health Programme [969]. A 2021 report summarizes the importance of NBS for early detection and treatment of both metabolism and endocrine conditions and emphasizes the lack of international standardization in NBS operations and makes the case for a standardization organization in Europe [970]. Several reports have focused on CH NBS in Romania in recent years: a report on 8 years of experiences with the MEDILOG registry for monitoring CH screening steps, CH incidence, diagnosed case management, iodine deficiency (through TSH), and more [971]; a review of CH screening in North-East Romania with suggestions for follow-up improvement—speedier diagnosis communication, tighter follow-up schedule, etc. [972]; and a review of CH data looking at the impact of moving the time of specimen collection and cutoff levels in order to improve patient outcomes [973]. A review of the value of NBS for GAL (all types) was published in 2021 updating the incidence, clinical manifestations, diagnosis, therapy, and prognosis [974].
- Russia—The NBS program expanded to include 36 new conditions, including SCID and SMA, on 1 January 2023 with screening available in 10 centers across the country [975,976]. NBS is carried out at the expense of budgetary allocations of the Regions of the Russian Federation and is free to the public [977]. A 2017 review of the past, present, and future of pediatrics in Russia helps to better understand national pediatric healthcare issues [978]. In preparation for the expansion, three SCID-related studies were published: two reviews of TREC and KREC screening, including the potential role of NGS in increasing the diagnostic accuracy of PIDS in general [979,980]; and determination of the 95% confidence intervals for TREC and KREC levels for different gestational groups with the suggestion that gestational age is an important factor that affects both TREC and KREC levels in newborns [981]. A recent publication reported on a pilot project for SMA NBS in St. Petersburg, which identified cases and supported the idea of earlier detection and diagnosis through NBS [982]. At least two other studies aimed at improving NBS have been reported since 2020: an assessment showing the effectiveness of CF case detection by screening versus clinical symptoms [983]; and a study showing the necessity of introducing differential diagnostics for tetrahydrobiopterin-deficient types of hyperphenylalaninemia to ensure the correct therapy [984].
- San Marino—Newborns in San Marino are screened for the same large number of conditions as newborns in Italy. Their specimens are sent to the Emilia-Romagna regional center laboratory at the Bologna University Hospital [985].
- Serbia—Two studies sought to improve the NBS program by reviewing CH case data over a 30-year time span. The first found a nearly 3-fold increase in detected CH in Central Serbia, which was presumably associated with lowered TSH cutoffs. Other factors affecting the increases were unclear and require additional studies [986]. The second study focused on the 30-year data and 14 late-diagnosed cases, finding 71% due to sample collection errors with the remainder as false-negative screening results with the goal of overall program improvement [987]. Recently, a program expansion pilot for SMA began (April 2022) in Narodni Front maternity hospital in Belgrade and was scheduled for national expansion in April 2023. The State Health Insurance Fund (RFZO) covers the costs of diagnostics, treatment, medical aids, and rehabilitation for all detected patients [988].
- Slovak Republic—The first ethnic results of the National Extended Newborn Screening (ENS) in Slovakia for the majority and the Roma ethnic populations were reported in 2017 for all 23 conditions. Significant differences in the incidence and prevalence of almost all disorders were found, especially in the IEM group, allowing increased ethnic focus on care, especially for children living in socially disadvantaged areas [989].
- Slovenia—A comprehensive review of the earlier years of NBS in Slovenia was published in 2015 [990]. In 2018, building on pilots that experimented with both expanded NBS and NGS confirmatory testing, expanded NBS was introduced in Slovenia, 17 metabolic conditions were added to the Slovenia screening panel, and the NBS program was reorganized and upgraded. Further expansion is expected soon [990,991,992]. A 2021 review gives an overview of the current state of NGS use in NBS, the remaining obstacles to its implementation, and the wider implications of its use in the NBS program [989]. The use of MS/MS has resulted in a study of a higher incidence of VLCAD than expected, which also resulted in one of the first reports of 4 novel variants of the acyl-CoA dehydrogenase very long-chain gene in Central-Southeastern Europe [993]. Two studies of phenylalanine detection also have been recently reported: a parallel study of MS/MS versus fluorometry showed lower analytical results with MS/MS but no missed cases [994]; and a review of fluorometric cutoff values as a protocol review/update showed that a slight upward adjustment (120 µmol/L 160 µmol/L) would make the screen more precise, resulting in fewer patient recalls (false-positive screens) [995].
- Spain—NBS in Spain is complex and is offered as a public health program in 15 screening laboratories within the 17 Autonomous Communities. The 2021 comprehensive review of NBS in Spain provides the history since initial implementation in1968 and details of the current screening situation [996]. A contemporary article reviews the implementation of national legislation specifying conditions that must be a part screening nationally, which begins removing inequalities in access inequalities. Forums coordinated by the MOH with the participation of those responsible for public health from the Autonomous Communities and scientific societies have been fundamental in serving as an example of the convergence of research and science for the benefit of a basic public health program [997]. Two other reports in 2021 draw attention to NBS management issues: one reflects on the revolution envisioned for NBS with the emergence of genomic technology and the continuing need for international harmonization of NBS programs; and the other points to NBS as a healthcare model of precision medicine [998,999]. A three-part series of articles reviewed the history of Spanish NBS focusing on ethical [1000], legal [1001], and social [1002] aspects of the NBS system.As a result of the variations in NBS across the country, several reports since 2020 have reviewed the history and status of various regional screening successes: 50 years of NBS in Catalonia [1003]; 20 years of NBS in Galicia [1004]; 10 years of NBS in Western Andalusia [1005]; 15 years of NBS for SCD in Madrid [1006]; 3 years of NBS for SCD in Western Andalusia [1007]; screening for propionic, methylmalonic acidemia and vitamin B12 deficiency in Madrid [1008]; 3 years’ experience in NBS for SCID in Catalonia [1009,1010]; 8 years’ experience with expanded NBS in Madrid [1011]; evaluation of positive cases from NBS in Madrid [1012]; CAH screening in Madrid [1013]; CH cutoff values in the Balearic Islands [1014]; the role of public health in NBS success in the Basque Country [1015]; and, an improved specimen transport system in Catalonia [1016]. Reports have also addressed issues related to second-tier testing to detect cobalamin (vitamin B12)-related disorders, both genetic and acquired [1017], differences in IRT levels between the different groups of newborns with a positive screen (healthy, healthy carriers, affected by CF or CFSPID) [1018], and the potential usefulness of NBS in detecting cystinuria [1019].Looking to the future, a 2021 review highlights the relevance of NBS for CH reviewing the status nationally and globally and the challenges and future prospects [1020], and a 2023 report notes the usefulness of elevated TSH NBS in detecting other conditions as part of the differential diagnosis of CH [1021]. The economic value of NBS also has been reviewed along with a look at progress in the automated economic assessment of NBS [1022,1023]. In addition to a review of the value of genetic analyses for NBS confirmation [1024], current screening tools, and future screening perspectives [1025], a recent report describes the use of LC-MS/MS as a second-tier panel of 31 metabolites increasing the efficiency of NBS by reducing false-positive and false-negative results, second sample requests, and the time to diagnosis [1026]. A two-part report from the Ethics Commission of the Spanish Society for Human Genetics summarizes and addresses NBS and NGS issues moving forward [1027,1028].
- Sweden—The NBS program began in 1965 and now has one central screening laboratory, two IEM diagnostic centers, three treatment centers (beginning in 2024), two neuromuscular treatment centers, and three SCID diagnostic/treatment centers. CH and CAH are usually handled in pediatric clinics and local hospitals [1029]. Since 1975, all residual bloodspots remaining after NBS have been stored in a biobank for QA and research in accordance with Swedish biobank law. Expansion to include 19 additional disorders by MS/MS occurred in 2010, a national IEM registry was initiated in 2013, and NBS for SCID was added in 2019. Two reports summarize SCID findings: one defines the 2-year pilot study; and one summarizes the first year’s screening results [1030,1031]. In the case of expanded NBS, implementation of second-tier NBS analyses for MMA, PA, IVA, HCY, and the use of collaborative laboratory integrated reports (CLIR) has helped to improve the overall PPV to around 60% [1032]. Additionally, a 2022 report on NBS for VLCAD suggests that a clinical severity score, compiled using treatment interventions and clinical symptoms, may correlate with the NBS CLIR score and residual enzyme activity as an aid in defining treatment [1033].
- Switzerland—NBS for PKU has existed in Switzerland since 1965 and was one of the first programs in Europe to add CH screening (mid 1970s). Specimens are collected on day 4 of life (written informed consent) and submitted to a centralized laboratory, which returns results within 1–3 days [1043]. A NBS TREC/KREC pilot for SCID was introduced in January 2019 to look for both T and B cell deficiencies [1044,1045]. In addition to outlining the NBS screening procedure, a 2020 report gave recommendations for diagnostic evaluations and precautionary measures against infection in children with abnormal screening test results. A recent 2023 Swiss publication reviews Primary Immune Deficiency Treatment Consortium data showing that more than half of NBS identified SCID patients developed at least one infection prior to hematopoietic stem cell transplantation and suggests access to monoclonal antibodies for RSV prevention for all SCID patients in countries with SCID NBS [1046].Other recent NBS activities have focused on laboratory testing: a report reviewing increased laboratory efficiency using a commercial robotic DBS extraction device in combination with LC-MS/MS [1047], a study to define reference intervals for total thyroxine (tT4) in NBS specimens with emphasis on premature and term-born infants [1048], and a clinical comparison and evaluation of two commercial testing platforms for NBS for TSH in DBS [1049]. A recent Swiss study sought to describe the IRT levels in healthy newborns in their first year of life and by gestational age, and to compare IRT values at two time points in healthy newborns and newborns with CF [1050]. While not a part of the Swiss NBS program, 30 years of data from TOXO NBS in Northwestern Switzerland were reported to inform the debate about the need for NBS for TOXO in Europe [1051]. Looking to the future, a commercial laboratory provides their “Baby Health Check” program as a supplemental screen providing testing for 15 additional conditions using “Screening of the Next Generation” [1052].
- Turkey (Türkiye)—Building on the 1983 PKU pilot program that showed a high incidence of PKU in Turkey (recently confirmed in a systematic review and meta-analysis [1053]) NBS was officially initiated by the MOH in 1986 and became the “National Phenylketonuria Screening Program” in 1994. Screening expansion (national) included BIO, CH, CF, and CAH in 2006, 2008, 2015, and 2021, respectively. There are 2 screening laboratories and specimens are collected at 48–72 h with results available in 1–2 days. Pilot testing with MS/MS began in 2002 but it is still not part of the national NBS program [1054]. A 2020 report of MS/MS NBS experiences in a multi-institutional hospital system across the country evaluated the distribution of 26 different acylcarnitine and amino acid disorders, established screening cutoff levels, confirmed the presence of several screened disorders in the population, and in view of the high rate of consanguineous marriages in Turkey, suggested expansion of the national NBS panel [1055].A 2022 mini-review of official NBS policies (or strategies) defining the number of conditions screened in 12 selected countries (including Turkey) confirmed that the Turkish NBS program should be expanded based on epidemiologic information, screening program infrastructure, cost estimates, and pilot screening data [1056]. Early in 2022, the MOH announced the addition of SMA to the Turkish screening panel and it was added in September 2022 [1057,1058]. Also, near the end of 2022, a Turkish molecular diagnostics startup launched an exome-sequencing-based newborn DNA screening test, which has the potential to become a suite of tests for various genetic diseases [1059]. Other recent reports concerning Turkish NBS have been published: a study of lymphopenia screening from cord blood demonstrating a cheaper, less invasive alternative to TREC analyses for SCID [1060]; a report of the status of genetic diagnostic laboratories and the frequency of genetic variants associated with CF detected through the Turkish NBS program [1061]; a 5-year data review of CF screening in a tertiary care center showing low sensitivity and PPV and a need for a national study to establish new cutoff values [1062]; a 6-year data review of the CF NBS protocol also showing low PPV in 3 tertiary care facilities with a similar appeal for re-evaluation of the screening protocol [1063]; and a report showing that mothers in Turkey are not sufficiently knowledgeable about NBS and recommending that education using mass media, educational environments (pregnancy schools), and screening test centers (including increasing awareness of this deficiency among midwives and nurses) [1064]. Most recently, a report on case reviews noted that ENBS can also play a pivotal role in identifying conditions in mothers of newborns identified by NBS [1065].
- Ukraine—Despite the challenges of war currently being faced in Ukraine, efforts to expand the NBS program are underway, beginning with a virtual international conference in 2022. Building on a screening program that includes PKU (2001), CH (2005), and CAH and CF (2012–2014 and 2017), a presidential declaration in 2021 listed 21 conditions for inclusion in the national NBS program as funding allows. There are currently 12 regional/inter-regional screening laboratories with four more for the new conditions requiring MS/MS [1066]. A description of the healthcare system in Ukraine published in 2020 helps to understand responsibilities within the system and how it affects NBS [1067]. Preparing for expanded NBS, a pilot study of the TREC/KREC screening procedure for SCID and other PIDs was recently published [1068].
- United Kingdom (England, Northern Ireland, Scotland, Wales)—The NBS program includes specimen collection at day 5 of life (birth is day 0) at home by a midwife or health visitor with results returned within 6 weeks. Information relating to the nine conditions included in the screening panel (PKU, MCAD, IVA, HCY, MSUD, GA-I, CH, CF and SCD) and other basic program information are included online with similar information available in the four health departments (England, Scotland, Wales, and Northern Ireland) [1069]. Various expert committees and advisory groups make NBS recommendations, including the UK National Screening Committee (UK-NSC), among others, working to ensure that NBS in the UK is evidence based, ethically sound, cost effective, and aligned with national public health goals. A well-defined process for both population and targeted screening exists. Examples of recent costing studies informing the process of the addition of screening conditions include studies on the economic impact of adding ALD [1070], the cost effectiveness of adding SCID [1071], and re-examination of the cost effectiveness adding five conditions (GA-I, HCY, IVA, LCHAD, and MSUD) [1072]. A costing analysis of teleconsultations as a means of communicating positive screening results was recently reported [1073] along with an in-depth description of the communication project and its collaborators (laboratory staff, clinicians communicating positive screening results, and parents) [1074]. Since 2020, several reports on result communications have been published [1075,1076,1077,1078,1079,1080,1081].NBS for CF has been part of the UK screening panel since 2007 and the use of NGS as part of the screening process for this and other conditions has begun. A few CF-related issues have been recently reported: the impact of NBS on outcomes and social inequalities [1082]; the psychological impact of NBS for CF, particularly when awaiting confirmatory results [1083]; parents’ experiences following inconclusive results from CF NBS [1084]; and the quality of the specimen being tested [1085]. A study of parents’ views on the use of NGS for improving the sensitivity and/or specificity of the CF NBS protocol was reported in 2022 [1086]. Discussions on the broader use of NGS as part of NBS, including relevant ethical issues, have also been published: a review of the opportunities and challenges for NBS using genomic sequencing [1087]; a report on the value of the use of ethics, engagement and co-design in developing a national newborn genomes program [1088]; a report of a research project within the National Health Service (NHS) to perform whole-genome sequencing of up to 200,000 newborn babies [189]; a review of the lessons learned from ongoing NBS programs looking at the psychological and ethical challenges of introducing WGS into NBS [1089]; and a 2023 ethics study using newborn genome screening as an example [1090].The UK-NSC has recently reviewed and approved the addition of TYR-I to the NBS screening panel for all UK departments [1091] and is currently considering the addition of SCID. To this end, at least two reports have been published: a review of SCID considering the NBS approach [1092]; and a study design for evaluating the outcomes and care costs for a group of babies identified by NBS compared with a group outside of the screening cohort [1093]. Screening for SMA is being considered by the UK-NSC and the results of a 5000-specimen pilot have been reported [1094], and another independent research study has been launched [1095]. Studies of disease-modifying therapies are underway in the UK [1096]. Already there has been a call to add SMA to NBS in Scotland [1097]. Other published NBS activities include a review of the feasibility and ethics of using data from archived Scottish NBS specimens [1098], a report on the importance NBS and early diagnosis in metachromatic leukodystrophy based on a caregiver survey in the UK and the Republic of Ireland [851], a pre-pilot MLD NBS feasibility study in Manchester UK using a two-tiered screening test algorithm that added to the international evidence recommending NBS new for MLD [1099], and a discussion of whether there is value to assessing maternal alcohol consumption in pregnancy by measuring phosphatidylethanol on day 5 DBS cards [1100]. A recent report on the efficacy of detecting GALT incidentally from the current NBS process notes that cases of GALT are being detected through the process and additional cases would be detected if screening was moved closer to birth [1101].
3.3.3. Tabular NBS Data for Europe
3.4. Latin America (LATAM) and the Caribbean
3.4.1. Regional NBS Activities Involving Multi-National Collaborations
Regional NBS Activities Involving Multi-National Collaborations in LATAM
Regional NBS Activities Involving Multi-National Collaborations in the Caribbean Sea Area
3.4.2. NBS Activities with a Country Focus
NBS Activities with a Country Focus in LATAM
- Argentina—The progression of genetics and NBS in Argentina has recently been reviewed [1132,1133,1134]. NBS was formally established by a law requiring PKU screening in 1986. Laws adding screening for CH (1990), CF (1994), and CAH, BIO, and GAL (2007) have established the framework for NBS implementation. Currently, NBS is conducted through 20 regional programs, one in the Buenos Aires Province (1995), one in the Autonomous City of Buenos Aires (2000), and eighteen other provincial programs organized under the umbrella of the Strengthening National NBS Program of the MOH (2006) with a general coverage of the public sector exceeding 98% [1135,1136,1137]. NBS for MSUD also is included in the provinces of Buenos Aires and Mendoza using a locally developed enzymatic analysis. NBS using MS/MS has been conducted for PKU, MSUD, and MCAD since 2014 in the Autonomous City of Buenos Aires [1134]. Looking to improve the NBS program for CF, researchers have compared the current IRT/IRT method for CF screening to the IRT/PAP approach and found improved sensitivity that should decrease the requisite follow-up actions [1138].
- Bolivia—While there is a 2006 MOH resolution establishing NBS for CH, there is not yet a national program. Instead, screening, including for other conditions such as PKU, CF, and CAH, is progressively occurring in individual hospitals from the nine Bolivian departments presenting diverse percentages of coverage with a national mean for CH around 30% [1139,1140]. In 2016, a new MOH resolution expanded the NBS panel by adding CAH, PKU and CF. A recent report from a regional initiative in La Paz describes the results corresponding to the period 2017–2023, in which 103,220 newborns were tested for CH, PKU, CF, and CAH [1141].
- Brazil—Since 2001, the Brazilian MOH has coordinated the National Neonatal Screening Program, which covers all of the 26 states and the Federal District through 30 reference centers reaching over 82.5% of all newborns [1142]. Information about features of the national NBS program, centers involved in specimen collection, program quality indicators, screened disorders, and the pertinent legislations are available from the MOH [1143]. The 2021 Brazilian legislation is particularly important because it is the first in LATAM to include mandatory NBS by MSMS for aminoacidopathies, urea cycle disorders, FAO disorders, LSDs, and molecular testing for SMA and SCID. A progressive implementation plan was also included [1144]. In recent years, NBS in the Federal District (Brasilia) has included an extensive screening panel including SCID, SMA, and LSDs, NBS in Minas Gerais state has included some MS/MS conditions in addition to the basic six-test panel, and NBS in the São Paulo metropolitan area has included MS/MS and SCID screening.Recent studies have reviewed the progress of screening for various conditions, patient outcomes, and related topics: cost effectiveness, screening equity, and program expansion. Program progress studies have focused on CAH [1143,1145], HGB [1146,1147,1148], and CF [1149,1150,1151]. Outcome studies have targeted patients with CH [1152,1153,1154], PKU [1155], GAL [1156], MSUD [1157], and BIO [1158,1159]. The cost effectiveness of NBS for CAH has been reviewed along with inequities in access to NBS itself [1160,1161]. A need for toxoplasmosis NBS in Brazil and elsewhere has also been questioned [1162]. The opportunities for expanded NBS have included studies of SCID [1163,1164,1165], LSDs [1121,1166,1167,1168], SMA [1169], and Prader–Willi and Angelman syndromes [1170]. NHS also has expanded as equipment and speech therapists have become available so that over 65% of newborns now receive hearing screening. Reports on the extent and outcome of hearing screening have been published, including a look at the possibility of NBS for cCMV in the future [1171,1172,1173,1174].
- Chile—NBS in Chile dates to a pilot program at the National Institute for Nutrition and Food Technology (INTA) in 1989, which was adopted by the MOH in 1992 and expanded step-wise until it covered the full population in 1998 becoming a possible model for other LATAM NBS programs [1123]. Currently, the NBS program is beginning expansion in four phases to include 26 conditions after a pilot implemented at the INTA in 2017 [403,1175]. In addition to research regarding inclusion of other screening conditions like SCID [1118,1176] and cCMV (at least in high risk groups) [1177], others have focused on outcome studies for CH [1178,1179], PKU [1180,1181], and MSUD [1182].
- Colombia—A 2019 law formalized an expansion of the national NBS program from CH to seven conditions and included EHDI and vision screening [1182,1183,1184,1185]. Efforts to continue expanding DBS screening have included developing MS/MS cutoff values and reviewing experiences and policy issues with MPS and other LSDs [1186,1187,1188,1189]. Continued interest in CCHD screening has included cost-effectiveness studies as well as a 14-site collaboration with the Nicklaus Children’s Hospital (Miami, Florida USA) [1190,1191]. Despite work in pursuit of a national NBS program in the past 2 years, NBS is currently only available for CH through various clinical laboratories mostly using cord blood, with newborn coverage exceeding 80%.
- Costa Rica—NBS has continued in Costa Rica since its first pilots in the early 1980s and its history has been detailed elsewhere [1192]. Most recent changes have increased the number of screened disorders to 29 [1193]. A recent evaluation of 3 years of screened newborns, CH referrals, and CH diagnoses confirmed the validity of the CH screening algorithm [1194]. The first mutation study of CAH patients from NBS identified an overabundance of cases with c.292 + 5G > A suggesting a possible founder effect [1195]. NBS expansion to include Wilson’s disease is also of interest because of the highest incidence globally (~1:20,000) [1196]. Wilson’s disease is a “National Interest Disease”, which ensures treatment and management of patients in the Costa Rican Social Security National Health System. Researchers have recently reported genotype and phenotype data on Wilson’s disease in Costa Rican pediatric patients [1197]. Finally, a pilot in 20 collection facilities (of over 1000) has also been implemented to improve and standardize the transport of specimens from collection facilities to the central screening laboratory [1198]. It is important to note that Costa Rica has the largest NBS panel in the region and was the first country to implement expanded NBS by MS/MS at a national level (2004) [1105,1199].
- Cuba—While Cuba had one of the first screening programs in LATAM, program expansion has been relatively slow [1200]. Products and equipment for NBS are generally produced locally and NBS testing is conducted in a decentralized way through more than 175 laboratories. The Ultra Micro Analytical System (SUMA) is an ELISA-based analytical platform, developed and manufactured by the Cuban Immunoassay Center (CIE), and it has previously been used in many LATAM NBS programs. A 2016 progress report detailed the 30-year history of developing and using SUMA® laboratory technology for several different screening conditions [1201]. A report in 2019 described the 20-year SUMA® external QA program, a program limited exclusively to laboratories using the SUMA® technology and confirmed its relevance in supporting quality NBS [1202]. Recently research reports have included CF test development [1203,1204,1205] and test improvement for CAH [1206] and CH [1207,1208]. As part of general health screening for newborns, attention has recently been given to piloting a new POC NHS procedure. A new screening device developed by the Cuban Neurosciences Center detects both auditory and visual disorders in newborns [1209]. The MOH is currently working on a national plan to introduce hearing screening equipment in health institutions with work underway to add a cardiovascular testing module to the equipment [1210].
- Dominican Republic—NBS testing is currently available only on request for private pay patients through hospital and laboratory contracts with a private NBS laboratory in the USA. Efforts to implement a national NBS program have been ongoing. A presidential decree creating the National Newborn Screening and High Risk Program was signed in December 2015, with the goal of screening 95% of all newborns, and a National Newborn Screening Counsel was created for its execution. A national database for metabolic diseases and related conditions was also created [1211]. NBS was to include a screening laboratory in Santo Domingo and evolve in three stages: (1) screening newborns of the three largest maternity facilities in Santo Domingo; (2) screening in centers in the different health regions selected by population density and health risks of the region; and (3) screening nationwide. During its first stage, the NBS program was to include five conditions: HGB, CH, PKU/HPA, GAL, and G6PDD, with specimens collected 48 h after birth. Unfortunately, progress was interrupted due to the pandemic when resources had to be diverted and the NBS decree was repealed due to changes in government authorities. Currently, local professionals interested in NBS are working with lawmakers to enact a comprehensive NBS law. Outside assistance is being provided by the Task Force on Global NBS from the International Federation of Clinical Chemistry and the ISNS [1212]. LATAM members of the Task Force are providing expert advice and assistance in NBS program development and advocacy to local policy makers on NBS cost effectiveness.
- Ecuador—NBS has existed as a program under the MOH since 2011. Specimens are collected in MOH hospitals and health centers around the country and tested for CH, CAH, PKU, and GAL at a private screening laboratory. In 2014, a MOH regulation was issued defining procedures for implementation of metabolic NBS [1213]. A recent review of cases detected through NBS identified different rates across disorders within various provinces, suggesting that in addition to reviewing the quality of testing, further investigations should look at rates versus ethnicities, genetics, consanguinity, and cultural beliefs [1214].
- El Salvador—A regional NBS program for CH was initiated by the MOH in 2008 and Technical Guidelines were published at the end of 2015 [1215]. In 2018, the coverage for CH reached 73% of newborns in the five regions of the country. Besides metabolic screening (CH, CAH, PKU, and CF), hearing (NHS) and cardiac (CCHD) screening are available at the Salvadoran Social Security Institute [1216]. The MOH and Social Security NBS efforts have expanded with two MS/MS systems now installed in each public center.
- Guatemala—Guatemala received technical cooperation support from the IAEA, which included technical visits by one of us (BLT) and contributed to the initiation of CH NBS in Guatemala City as early as 1995 [20,1217]. Expansion since then has been slow. Beginning in 2015, the Institute for Scientific Research and Education on Human Genetic and Metabolic Diseases (INVEGEM) initiated a NBS pilot in national hospitals in Guatemala City and other locations in the country for CH, CAH, PKU, GAL, and CF. The goal of the project is to gather incidence data for use in convincing policy makers to support the program [1218]. A local project in Guatemala City at the two national hospitals is also attempting to better organize NBS and collect similar data to convince policy makers [1219]. Work is also ongoing to determine incidences of other screenable conditions like HGB [1220]. A lack of useful information about NBS has been recognized and is being addressed on an individual hospital basis [1221].
- Haití—In 2010, newborns were screened in St Damien Hospital in Port-au-Prince, and the samples were processed at Pordenone Hospital, Italy as part of a project to determine the incidence of SCD. Despite a reported incidence of SCD of 0.58% (twice the U.S.) demonstrating a need for NBS, no national NBS program was continued [1222]. A 2018 report on capacity building for NBS in Haití noted that NBS for SCD is considered the standard of care in some hospitals and research at others [1223]. This report also noted that equipment for screening was a major need that was being addressed as funds become available, usually from external donations. While administration of the hospital-based programs is similar, the laboratory methods differ, including the use of POC testing in some hospitals. A POC validation study was also conducted in collaboration with the Florida, USA NBS program [1224]. Because of the impact of local advocacy groups in other countries, the Haití Sickle Cell Interest Group has been established [1223]. A recent report reviews the progress of the pilot study in demonstrating the feasibility of NBS while noting the difficulties with follow-up (which are much less with POC testing). The project continues with emphasis on “understanding how to mitigate implementation limitations” [1225].
- Honduras—In September 2016, the National Congress approved the “Mandatory Neonatal Screening Law” to be implemented in Secretariat of Health and Honduran Social Security Institute (IHSS) birthing facilities. The IHSS designed a model for NBS implementation, including a budget forecast and actions to provide personnel, facilities, and screening equipment. A NBS pilot began in 2017, expanding slowly, primarily in IHSS facilities. An operating procedures manual was established by the Secretariat in 2018. Screening includes CH, CAH, PKU, GAL, and CF based on the experiences of others like Cuba and Costa Rica, and the inclusion of HGB has already been suggested. Offering NBS is mandatory, and then parents may then choose whether to accept (informed consent) [1226].
- Mexico—The Mexican health system has recently been detailed including the history/process of overcoming some of the institutional, organizational, and resource barriers to establishing NBS [1227,1228]. A review of NBS also exists [1229], along with a review of over 33 years of outcomes in a group of Mexican patients with inborn errors of intermediary metabolism focusing on disparities and unmet needs [1230]. NBS in Mexico was one of the earlier NBS programs in LATAM, beginning in 1973 but paused from 1977 until 1986. In 1988, NBS for CH was formalized by rule [1231]. Currently, the National Center for Gender Equity and Reproductive Health within the MOH is responsible for NBS policies and the screening panel includes CH, CAH, PKU, GALT, CF, and G6PD [1232]. BIO is included in some parts of the health system, e.g., the Mexican Institute of Social Security (IMSS). There are three different healthcare providers in Mexico that serve the population according to employment-based social insurance, uninsured public assistance, and private employer’s insurance [1227,1233]. Different screening approaches occur in the various hospitals, groups of hospitals, and government departments without any unified central coordination [1231,1234,1235,1236]. The Secretary of Health oversees the highest percentage coverage of Mexican newborns [1237,1238].Interest in G6PD deficiency, and its addition to the official panel of tests in 2017, has resulted in several recent reports [1239,1240,1241]. Some states also offer expanded NBS by MS/MS and HBG screening. Research on CF and LSDs has also been the subject of recent reports along with screening methods for tyrosinemia and the need to screen for toxoplasmosis [1120,1233,1242,1243,1244,1245]. POC reports on NHS and CCHD have focused on hospital experiences for both [1246,1247,1248]. On 1 June 2021, a reform to article 61 of the General Statute of Health institutionalized CCHD screening and a national committee was initiated to assist in its implementation [1249,1250].
- Nicaragua—The NBS program began in 2005 as a collaboration between two local universities, and subsequently, other Spanish institutions. A cord blood NBS program for CH existed until August 2018 when it was discontinued due to the interruption of free testing reagents from the Spanish institutions and the lack of an adequate budget for sustaining the program [1251]. During the first 10 years, nine additional departments joined the screening effort, involving a total of 12 hospitals [1252]. In 2014, a pilot expansion that included CH, CAH, PKU, GAL, and BIO was initiated in the Social Security Institute [1253]. There was interest in HGB screening to the point that a specialty confirmatory laboratory was established through a German academic exchange program [1254]. Aside from blood screening, there has been growing interest in NHS as evidenced by a recent positive cost analysis and an NHS clinic visitation by the U.S. Ambassador [1255,1256].
- Panama—A 2007 law created a national NBS program that mandated testing for CH, PKU, GAL, CAH, G6PD, and HBG. By the end of 2014, screening had extended to 11 health regions of the country [1257]. Published HGB incidence results highlighted the importance of HGB as a public health issue [1258]. A recent comprehensive program overview for 2013–2018 noted that for hospitals throughout the 14 health regions, including MOH, Social Security, and private health facilities, the average NBS coverage was 72.3% [1259]. In 2022, a new law was enacted that established NBS as mandatory and extended the panel of screened diseases to CF, NHS, CCHD, and vision [1260]. Screening expansion has now begun, and new MS/MS equipment has been installed.
- Paraguay—In Paraguay, NBS began in 1999 as a research pilot project, “Program for the Prevention of Mental Retardation,” to detect CH and PKU. In 2003, with the addition of CF screening and a name change to the “Cystic Fibrosis and Mental Retardation Prevention Program”, the NBS program was formalized by law and officially implemented in 2004. It expanded, with assistance from the Japanese International Cooperation Agency (JICA), including technical advice and support by one of us (GJCB) as part of the JICA “Third Country Experts Program” between 2008–2011. In 2016 the national screening program was created by law, changing its name to the “National Neonatal Detection Program”. It now reaches over 90% of newborns in all 18 health regions and has evolved into a formal public health program [1261]. A more detailed history and progress of NBS in Paraguay has been recently reported [1262]. Since 2018, MS/MS testing has been offered to symptomatic newborns or those with a family history of AA, OA, and FAO [1261].
- Peru—Following a 1997 MOH resolution that recognized the need for NBS for CH, a pilot NBS program for CH and CAH was begun by the Social Security Institute of Peru in 2002. The program expanded to include PKU and GAL, and coverage included all Social Security birthing facilities [1263,1264]. In 2012, a law for universal NBS prioritized screening for CH, CAH, PKU, CF, NHS, and congenital cataracts, and in 2019, the MOH issued a Health Technical Rule for NBS for these disorders [1265]. Currently, NBS is available in the main hospitals of the MOH, the Social Security subsystem and the private sector, but it is not completely implemented in some of the public medical centers [1266]. In 2022, a new bill was presented to the Congress contemplating the newborn’s rights to access universal NBS throughout the health system [1267].
- Uruguay—NBS began with CH cord blood screening at the clinical laboratory of the maternity facility of the BPS (Banco de Previsión Social—the state-owned Uruguayan social security institute) in 1990 and became a mandate in 1994. Ancillary assistance from the IAEA and integration with the immunization program for logistics, soon led to over 95% newborn coverage [20,1268,1269]. Various health proclamations related to NBS and a challenging transition to heelstick specimens have led now to one of the more comprehensive, universal, free, and equitable screening programs [1268]. Among other activities, a pilot HGB project [1270] and an evaluation of newborns with abnormal screening results, including NHS, have recently been reported [1271], along with a 25-year history of NBS in Uruguay [1272]. Currently, implementation of a NBS pilot for BIO is planned for 2024 and recently the MOH designated BPS as a national resource center for rare diseases.
- Venezuela—There has been a NBS law in effect since 1999 for PKU and CH but coverage has been slow. NBS was implemented utilizing a network of 19 regional public health laboratories. In 2013, the national NBS program was created and in 2015, the MOH added GAL, CAH, and BIO to the diseases panel. A 2015 report of 2 years of screening for CH and PKU in Zulia state noted difficulties in getting good demographic data. National NBS coverage reported in 2015 was about 65% [1273], decreasing to about 61.5% in 2017 [1274]. In the past few years, the functioning of the national NBS program has been impacted by the economic problems affecting the country, thus reducing NBS program coverage and efficiency.
NBS Activities with a Country Focus in the Caribbean
- Anguilla—A NBS for SCD does not exist in Anguilla. Instead, a program exists to check the sickle status of all pregnant women [1275].
- Antigua and Barbuda—The American University of Antigua College of Medicine (AUA) and the CAREST launched a NBS program for SCD in September 2020 at Mount St. John’s Medical Center (MSJMC.) CAREST paid for specimen transport to the laboratory in Guadeloupe. During and after the project, there is/was no cost to parents or the Antiguan government. Screening continues in Antigua with the intent to extend the program to Barbuda and other private medical facilities soon [1276,1277].
- Aruba—Now a self-governing constituent country of the Kingdom of the Netherlands, its inhabitants were historically not affected by slave trading and SCD as much as other countries in the area. A 2006 pilot showed a lower prevalence of SCD in Aruba than St. Maarten than Curaçao. Study statistics suggested that 2 patients would be detected every 3 years and questioned the true cost effectiveness of a NBS program for SCD. The possibility of a universal NBS program for SCD housed in a single laboratory strategically located in the Caribbean area was noted as a possibility that would also enable screening for other heritable diseases [1278]. In contrast to the BES-islands, Dutch “special municipalities”, there is no national NBS program, however, newborns admitted to the pediatric department of the Dr. Horacio E. Oduber Hospital are offered NBS for CH [1131].
- Bahamas—Expanded NBS is available through a contracted private NBS laboratory in the USA for the birthing hospital and a medical laboratory so that most newborns are screened for the disorders contained in the US RUSP [1279].
- Barbados—A 1999 NBS pilot project with 1000 newborns estimated the SCD incidence to be lower than other nearby islands, but a larger confirmation study was never carried out [1280]. In preparation for another pilot in 2014, a database of persons with sickle cell anemia was developed along with a process for documenting clinical manifestations. A pilot comparing SCD prevalence with Jamaica was planned as a kickoff for NBS; however, the screening pilot was cancelled and replaced by the screening of children and pregnant women thought to be at risk of SCD [1281]. Currently, there is no NBS for SCD despite continued calls for its implementation by NBS advocates and the local HOPE Foundation, which publicly reminds policy makers that Barbados is one of the few Caribbean countries where NBS for SCD is not available publicly [1282]. Specimens from private pay patients are sent for an expanded NBS package at a private NBS laboratory in the USA.
- Belize—Interestingly, while there is currently no formal NBS program in Belize, a pilot study to demonstrate the feasibility of using the Sickle SCAN POC screening test and to gather prevalence data in support of screening was reported in 2017 [1283].
- Cayman Islands—In 1997, routine cord blood NBS for HGB was initiated for babies born at the Cayman Islands Hospital in Grand Cayman and at Faith Hospital in Cayman Brac. In 2002, the Cayman Islands Hospital expanded NBS to include over 50 conditions utilizing the services of Revvity Omics in Pennsylvania, USA. The Doctors Hospital also offers expanded NBS using the same commercial NBS laboratory [1281,1284].
- Curaçao—Curaçao is a constituent country in the Kingdom of the Netherlands. Results of a 2006 pilot have been previously noted under Aruba (above) [1278]. There is no national NBS program but there are hospital-based initiatives that offer limited NBS. Screening for HGB and CH using cord blood is offered to all newborns delivered at the Curaçao Medical Center. NBS for PKU is targeted to newborns that are not, or only partly, of Afro-Caribbean ancestry because of the lower PKU incidence in persons of African descent [1131].
- Dominica—CAREST is working with patient advocates in Dominica to implement NBS for SCD.
- French Guiana—French Guiana is an overseas territory of France and participates in the French NBS program. Specimens are sent to the screening laboratory in Lille for analysis using the French screening panel.
- Grenada—A two-year pilot neonatal screening program to establish the local SCD birth prevalence and to foster NBS in Grenada was undertaken in 2014–2015 at the Grenada General Hospital. Subsidized by CAREST, the pilot was a collaboration between the MOH of Grenada, the Sickle Cell Association of Grenada, and the HGB diagnostic laboratory of the University Hospital of Guadeloupe. This pilot study demonstrated the feasibility of NBS for SCD. The SCD birth prevalence resulted in a commitment from the MOH begin NBS to reduce the burden of disease in Grenada [1285]. NBS was not continued, however, due to funding and a lack of human resources [1107]. NBS HGB/SCD stakeholders continue to explore options for the continuation of the program.
- Guadeloupe—In 1984, a universal NBS program for SCD and other abnormal hemoglobins was initiated. In 1990, NBS became part of a SCD comprehensive management program coordinated by the Comprehensive Sickle Cell Center of Guadeloupe. The NBS program is part of the French national NBS program for SCD supported by the French Association for Screening and Prevention of Child Handicaps and by the national public medical insurance system for salaried persons. Prior to 1995, cord blood specimens collected on the day of birth were used for NBS and afterwards, specimens were dried bloodspots taken two days after birth [1286]. DNA testing is available as a second-tier test in certain instances and the Guadeloupe laboratory serves as a screening laboratory for Grenada and Tobago. Current NBS coverage exceeds 98% [1107].
- Guyana–There is currently no organized NBS program in Guyana. A partnership was established with Newborn Screening Ontario to initiate a pilot in 2016–2017 to report on screen-positive rates, obtain prevalence for CH and SCD, and to assess the feasibility of implementing NBS. This first-time study provided baseline data useful for informing future NBS policies. The small sample size and difficulties in follow-up limit generalizations but suggest that NBS for both conditions could be beneficial, although the data for HGB screening is more compelling. Larger pilots are needed, and a remote screening laboratory may be necessary, at least in the early stages [119].
- Jamaica—The original Jamaican cohort of 100,000 babies has been well studied over time leading to significant knowledge increases in the natural history of SCD [1287,1288,1289]. The current program has been ongoing since 1995, when it was restarted because of advocacy by the Sickle Cell Support Club of Jamaica. Screening coverage reached the full population in 2015. Because there is no legislative mandate, the program previously relied on funding at the discretion of incumbent policy makers [1107]. However, as SCD is now a part of non-communicable disease policy, and it is likely that the program will continue to improve. The collaboration of academics, clinicians, patients, and public health practitioners has accelerated progress [1290].There are two screening laboratories in Jamaica. The lab at The University of the West Indies (UWI) screening laboratory screens the eastern half of the island and the laboratory in the Southern Regional Health Authority screens the western half. Work from Jamaica has highlighted barriers to tracing affected infants and bringing them into care as part of the NBS program [1291]. The UWI screening laboratory has been an essential component of many of the island pilots and pilots in other developing countries internationally. While the typical NBS specimen is whole blood collected from a heel stick onto a special absorptive paper, cord blood is also used in some settings in liquid form. In a 2017 report, the Jamaican program reported their technique for using cord blood to validate its usability in low-resource settings in situations where cord blood is the only available option [1292].
- Martinique—In 1989, NBS for SCD began, mirroring NBS in Guadeloupe. Like Guadeloupe, the NBS program is part of the French national NBS program for SCD supported by the French Association for Screening and Prevention of Child Handicaps and by the national public medical insurance system for salaried persons [1107,1293].
- Sint Maarten—St. Maarten is a “constituent country” in the Kingdom of the Netherlands. Results of a 2006 pilot have been previously noted under Aruba (above) [1278]. There is currently no national NBS program but there are hospital-based initiatives that offer limited NBS. Screening for CH is offered to all newborns delivered at Sint Maarten Medical Center [1133].
- St. Lucia—In the mid-1980s, St. Lucia became the first Caribbean country to attempt NBS for CH. Cord blood samples were collected on filter paper and transported to the Illinois NBS laboratory for testing. While the validity of their blood collection and transport process was demonstrated, no case of CH was detected during the 3-year pilot and the NBS program was not continued [1294]. In 1992, supported by the St. Lucia Sickle Cell Association, cord blood screening for SCD was introduced through the MOH with the goal of working toward universal screening. Because cord blood limited the screening conditions and had unacceptably high false-positive and false-negative rates due to contamination with maternal blood and method-specific errors, heel prick DBS specimens were introduced in 2014 but not widely accepted. In an effort to improve acceptance of DBS specimens, a 2-year iPIER (Improving Program Implementation through Embedded Research) study sought to assess healthcare workers’ knowledge of and attitude toward DBS screening and to determine new mothers’ favorability toward DBS screening [1131,1295]. The use of HP specimens in St. Lucia remains a challenge, and cord blood continues as the specimen of choice [1107].
- St. Vincent and the Grenadines (SVG)—In 2015, a prospective NBS-SCD study of mothers and their newborns was conducted at the maternity hospital that accounts for about 95% of all newborn deliveries in SVG. This study assessed the feasibility of NBS in collaboration with the NBS laboratory at the South Carolina Department of Health and Environmental Control. Results confirmed that partnering with an established NBS laboratory is a feasible way to develop NBS when the infrastructure might not allow an in-country laboratory. It also showed that targeted NBS for SCD using the protocol in place at the time (parents’ sickle history) missed cases and was not acceptable for NBS [1296]. NBS was not continued due to funding and a lack of human resources [1107].
- Trinidad and Tobago—NBS for SCD in Tobago is funded by the Regional Health Authority of Tobago, which made diagnosis and treatment of SCD a priority. It has been ongoing since 2008. It now covers more than 95% of births with family members of identified cases are also screened. Indicating expanding acceptance of the Tobago program, a major obstetric hospital in Trinidad began NBS for HGB in 2018 [1107].
- Turks and Caicos—NBS is available to private pay patients for a limited number of conditions through arrangements with a private NBS laboratory in the USA.
3.4.3. Tabular Display of LATAM NBS Information
3.5. Middle East and North Africa
3.5.1. NBS Activities Focused on the Middle East North Africa Region
3.5.2. NBS Activities within Countries
- Algeria—There continues to be no organized NBS in Algeria despite the recognition that cases of CH are being diagnosed late and that NBS would have a profound impact. A 2016 report showed that, absent NBS, the mean age for referral for 75 suspect cases of CH was 17.6 months with 56 true cases confirmed [1306]. A 2019 review of CH cases detected between 2007 and 2018 showed developmental delay and short stature in a number of cases diagnosed beyond 3 months of age [1307]. Both study groups appealed for CH NBS as a national emergency. While not the subject of our report, we note that NBS for hearing loss has already become available in some settings [1308].
- Bahrain—In 1984, recognizing the large number of genetic blood disorders, the Bahrain MOH organized one of the first genetic clinics in the MENA region and began an educational campaign to reduce the incidence of Hgb. In mid-2007, NBS was begun for HGB and G6PD deficiency in MOH maternity units [1309]. Through the years, retrospective studies have provided incidence estimates for both CH [1310], other IEMs [1311], and NHS [1312], all supporting the need for a national NBS mandate. Absent a government NBS program, private hospitals provide varied screening panels with some NBS specimens reportedly being analyzed for IEMs in Saudi Arabia [30].
- Egypt—Egypt has the largest and one of the fastest growing populations in the MENA Region. The Egyptian NBS program began with CH in 2000 and expanded to include PKU in 2015. The first NBS pilot in 2016 found that Egypt has one of the highest birth prevalence rates for IEMs detectable by MS/MS [1313]. Other pilots have followed, each looking at possible conditions to be added to the NBS panel, including GALT and G6PD deficiency [1314,1315,1316,1317]. In 2021, NBS was expanded nationally to include 19 conditions, with Phase I consisting of screening babies born at public hospitals and Phase II expanding the scope to includes babies born at university hospitals, private hospitals, and healthcare units [1318]. As a part of “Egypt 2030”, many health programs, including NBS, will undergo digital transformations in the coming years. This effort by the Ministry of Communications and Information Technology is intended to make internal communications and data transfer from peripheral sites easier and more efficient [1319]. Building on earlier research from the German NBS program, Egyptian collaborators have also participated in research demonstrating the feasibility of NBS for cystinosis using molecular techniques [818,1320]. Recently, screening for HGB and SMA have been included in the available NBS tests and screening for HGB has been included in premarital screening tests. A 2023 report reviews the impact of rare diseases in Egypt including newborn screening and the Egyptian Genome Project, which began in 2021 [1321].
- Iran—Based on the success of pilot studies in three provinces, and basic support from IAEA, national NBS for CH in Iran was integrated into the national health system in 2005 and newborn coverage now exceeds 95% [20,1322]. To assess the quality and outcomes of the NBS program, at least three audits were completed in recent years, which showed normal growth and development at school age because of early diagnosis and treatment [1323], and good coverage but high recall and losses to follow-up. The latter program issues were addressed through the re-evaluation of cutoff levels and better surveillance of follow-up [1324,1325]. Originally, the national NBS for PKU began because of its relatively high incidence in the Iranian population and corrections were also made through cutoff changes [1326,1327]. A 2014 report noted that when NBS for G6PD deficiency was begun nationally, sensitivity issues with the laboratory procedures were also reported as implementation obstacles [1328].A detailed analysis of the implementation of NBS for PKU and related policies pointed (at least partially) to the continuous persistence of parents and interest at the public health executive level (a top-down approach) as significant factors in program implementation [1329]. Recently, a study on the quality of life of parents of children with PKU showed a need for interventions to help the parents better cope with the problems associated with PKU detected through NBS. In addition to family education to increase awareness and improve attitudes, increased financial support for families, funding assistance for PKU patients from support organizations, and medical cost coverage from insurance companies were identified as probable benefits [1330].Expanded NBS with MS/MS pilots for laboratory and clinical follow-up were begun in six provinces in 2017. Ten laboratories nationally were certified by the MOH, each linked to a referral hospital with a panel of metabolic specialists [1299]. Looking towards future screening expansion, at least one pilot study has evaluated the use of MS/MS and other second-tier tests to improve screening quality and to consider the possibilities of adding up to 38 possible IEMs. This study included the evaluation of reference ranges and screening cutoffs [1331]. To replace the current manual data management system, a framework for implementing electronic data management has been proposed based on literature reviews of the data management systems in the USA, UK, and Australia [1332]. A recent study compared and validated for the first time, two fluorometric methods for measuring α-glucosidase acid activity in dried bloodspot specimens (DBSs), with potential use for NBS and diagnosis of PD. in Iran [1333]
- Iraq—Building on a pilot begun in two provinces in 2013, the U.S. Agency for International Development (USAID) assisted with a 2014 project to screen for PKU, CH, and GAL using the project’s detailed instruction guide [1334,1335]. Reviews of the developing program showed lower coverage than desired but progressing. Recommendations included expansion of the screening panel and increased implementation to more provinces [1336,1337]. A 2020 report noted a continued lack of expansion but plans to cover all 18 governorates and expand the screening panel [1334]. A study of mothers’ knowledge and attitudes towards NBS showed enthusiasm despite a lack of knowledge [1338].A 2019 report of CH birth prevalence in the Kurdish part of Iraq, Duhok Governorate, showed a prevalence of 1:250–1:900 across the governorate, which includes a mountainous iodine-deficient area [1339]. A pilot non-profit, non-governmental program, managed by a private lab, provides seven basic screening tests for a small fee. Expansion to approximately 60 screening tests by this laboratory has been implemented, with the possibility of providing screening on a national level [1340]. The Duhok NBS laboratory is the only facility performing screening for the LSDs. Because NBS is not yet routine across the whole of the country, and there is a high degree of consanguinity lack of metabolic specialists, conditions like PKU (which are likely higher in number than currently reported) are being diagnosed late. There is a growing concern over the lack of full coverage for all newborns through a national NBS program [1341]. There are currently two larger screening laboratories (Baghdad and Karbala) with gradual expansion occurring nationally.
- Israel—Like many Western countries, Israel has maintained a NBS program since the 1960s beginning with PKU and later adding CH [13,1342]. An expanded NBS panel of 11 additional conditions was implemented in 2008 [1343]. Screening is free of charge as part of “The Healthcare Basket” under the National Health Insurance Act [1344]. A website with program information and secure screening results was launched in 2009. A study exploring the views of health professionals regarding parental education and informed consent for screening after expansion indicated a need for in-depth discussions and considerations regarding parental education, result communication, and informed consent processes for screening prior to any future program expansion [1345].Following a successful pilot study and anticipating a high incidence because of consanguinity, NBS was expanded to include SCID. A report on the first year of screening and a 5-year data review have been published [1346,1347] along with an upbeat editorial comment [1348]. Noting an incidence as high as 1:29,000, the authors of the 5-year study called for SCID screening globally, particularly in consanguineous populations. Also, recently there was increased interest in adding NBS for the galactosemias to the screening panel. To develop a NBS pilot algorithm for galactosemia, archived dried bloodspots from newborns with GALT, galactosemia variants, and normal controls were analyzed by MS/MS for galactose-1-phosphate (Gal-1-P) by FIA MS/MS. Based on a successful pilot, a screening algorithm with Gal-1-P as the first-tier screening test, and GALT enzyme activity as the second-tier to identify newborns suspected to be at risk for classical galactosemia has been implemented [1349]. Another FIA MS/MS study demonstrated the feasibility of screening newborns for CTX, a progressive metabolic leukodystrophy, using two-tier screening with FIA MS/MS and LC MS/MS [1350].Also not on the current screening panel, CF case detections identified through the national population carrier screening (PCS) pro/gram have been reviewed and found to support a more balanced case detection approach using both PCS and NBS [1351]. A review of the barriers to adding CF to the NBS screening panel suggests a balance of bioethics and cost effectiveness should be used in considering whether CF should be added to the screening panel [1352]. Other interesting research projects possibly affecting NBS policy include: (1) a study of the effect of NBS on rea productive decision making following the detection of a child with a disorder detected through NBS [1353]; (2) a study of the founder effect of a pathogenic variant among Bukharan Jews associated with severe early-onset Wilson’s disease, for which NBS could lead to early diagnosis, treatment, and improved outcomes [1354]; and (3) decisions of which conditions to screen and exploration of the different ‘gene worlds’ that constitute NBS programs in Israel and the US [1355]. Looking to the future, provocative articles on next generation sequencing in NBS have been published by Israeli researchers [1356,1357].
- Jordan—Following a PKU and CH pilot initiated in 2004, the first national NBS project screening for PKU was begun in 2006 along with a nationwide training program for healthcare workers. The NBS program was expanded to include CH in 2008 and G6PD in 2012. Currently, 29 conditions are included and at least one private laboratory provides a larger number [1358,1359]. A recent report reaffirmed the importance of the NBS program for early identification and treatment of PKU and other genotyped and biochemically characterized Jordanian hyperphenylalaninemia patients for the first time [1360]. A retrospective study of TYR-I found that a lack of clinical familiarity with the disease resulted in delayed diagnosis which might be ameliorated by inclusion of TYR-I on the NBS panel [1361]. A 2022 report on mothers’ NBS knowledge and attitudes confirmed that healthcare providers, particularly nurses, were the main source of mothers’ information and education and their knowledge should be improved [1362]. For health workers and parents, a 2022 comprehensive NBS manual in Arabic exists online [1363].
- Kuwait—The Kuwait NBS program began in 2005 with two conditions, PKU and CH, and was limited to public hospitals. In 2014, the Kuwait Ministry of Health began a publicly funded, expanded NBS program screening for 22 conditions. The aim of the program was to ensure that all infants born in Kuwait were (and are) screened. Before April 2015, NBS was only offered in public hospitals since private hospitals individually offered their own screening testing, often provided by screening laboratories in the U.S. In May 2019, the Kuwait NBS program, located in the Kuwait Medical Genetics Center (KMGC), began covering 100% of newborns born in Kuwait. Noteworthy has been the increased detection of cases of homocystinuria by utilizing the methionine to phenylalanine ratio to improve predictability [1364]. Kuwaiti experiences with NBS for VLCAD deficiency have also been reviewed and reported. This study provides evidence that expanded screening detects cases of VLCAD. Molecular genetic testing for the ACADVL gene was recommended for inclusion in the screening algorithm [1365].
- Lebanon—NBS in Lebanon has been ongoing since 1996 when it began as a fee-for-service program in both Saint Joseph University in Beirut (USJ) and the American University of Beirut Medical Center (AUBMC). Its expansion to include MS/MS technology in 2006, involved collaboration between the NBS laboratory at USJ and the metabolic laboratory at the Hamburg University Medical Center (HUMC) [1366]. Additionally, an independent laboratory began to offer expanded NBS, which in theory made screening available to all. The fact that NBS cost was usually borne by the patient likely decreased the number screened by half. A 2015 cost-effectiveness study provided an evidence-based model to encourage the government to make NBS free and provided a model for other developing countries in the region. It encouraged patient advocacy and other concerned civil activist groups throughout the region, “to revive their demands for mandatory universal publicly funded NBS for IEM [1367].” Also in 2015, a review of genetic disorders highlighted the urgent need for community genetic services nationally. It noted that NBS services were generally localized within the capital and not nationally mandated [1368]. Another 2015 study by AUBMC found a high alpha-globin carrier rate and highlighted the presence of two common alpha gene mutations in the consanguineous Lebanese population [1369]. This supported the conclusions of an earlier study on the presence of sickle cell and other hemoglobin variants and supported the need for national NBS [1370]. In 2018, the government launched its “National Program of Neonatal Screening for Primary Immunodeficiency Diseases” (NaSPID), noting that, “babies born under the coverage of the MOH will have the screening test done with no additional costs” [1371]. Currently, NBS is available to all residents in Lebanon through three community-based universities and screening coverage exceeds 50% despite a severe nationwide economic crisis. The NBS program in Lebanon has assisted several other developing NBS programs [1372].
- Libya—Targeted NBS exists in Libya in certain high-risk situations. For example, babies with diabetic mothers are screened for CH and those with cataracts are screened for GAL [1373]. A 2010 study evaluated the cost- effectiveness of establishing a NBS program for PKU in Libya and showed that implementing a NBS program for PKU would generate a 90% return on investment [1374]. A 2022 report outlined the extent of IEM in Libya and supported implementation of a NBS program [1375]. A survey of Libyan physicians rated lack of technology, infrastructure, and specialized medical professionals as the largest obstacles to developing a NBS program [1373]. A suggested possible solution involved stepwise NBS implementation using a laboratory and/or other support from another country. This type of collaboration was essential to establishing sustainable NBS in Qatar, Lebanon, the Philippines, and others [1366,1376,1377]. In 2019, a new IEM Committee was formed to organize NBS. A central laboratory in Tripoli is planned along with the ambitious goal of screening for a large number of disorders [1378], yet nothing has been implemented due to lack of funding.
- Morocco—The first MENA Regional NBS meeting, Enhancing Neonatal Screening in MENA, was held in Marrakech in 2006 to begin to organize and expand NBS in the Region (see Marrakech Declaration [1379]). During 2010–2011, a NBS steering committee was organized by the MOH and the first phase of a pilot NBS program was launched in the Rabat region. Assistance was provided by the Japanese International Cooperation Agency (JICA) [1380], including training and a program review with recommendations for improvements. In 2013, the MOH created Ministerial Circulars requiring the availability of CH screening at all childbirth facilities and in 2014, designated a specific protocol for diagnosis and therapy of confirmed cases. NBS has continued to expand in university hospitals with continuing interest in adding other conditions such as PKU [1381]. A recent pilot study in 34 centers in Fez determined an incidence of CH of 1:1952 [1382]. While there is not yet a sustainable national NBS program, there is expanding interest and periodically a new facility or laboratory will begin screening, increasing the number of screened babies. A recent report on the use of MS/MS in a targeted study in Rabat showed that IEMs detectable by MS/MS are present in the population and that NBS using MS/MS would be useful for their detection [1383].CCHD screening is also receiving attention in Morocco and a recent collaborative pilot study between Mohammed VI University Hospital of Marrakech and Children’s National Hospital in Washington, DC, USA demonstrated the feasibility of including CCHD screening in the developing NBS program [1384].
- Oman—Building on experiences from a hospital cord blood NBS program initiated in 1991 and a 2004 pilot, the National NBS Program for CH was begun in 2005 using cord blood [1385,1386]. This program was provided to all newborns in all healthcare facilities, including MOH hospitals, extended health centers, sister government and private health institutions. Detailed guidance for CH NBS was created in 2010 by the MOH and updated in 2021 [1385]. Because there is a high incidence of HGBs in Oman and the cord blood used for CH screening can be used for HGB screening, a prospective NBS study provided HGB incidence data and suggested HGB addition to the CH NBS program. [1387]. A later study showed the capabilities of HPLC in detecting HGBs from newborn specimens, but the NBS program was not expanded [1388]. Expansion to include other IEM conditions using MS/MS and other procedures was proposed in detail noting that diagnostic MS/MS screening for multiple IEMs had been available at one Omani university hospital since 2002. The combination of years of diagnostic experience coupled with a recently opened genetic center appeared to provide the necessary expertise for NBS follow-up [1389].While expanded NBS that includes MS/MS technology is not yet available in Oman, this may soon change. In anticipation of the need for reliable references ranges for laboratory testing, a study has already been completed providing reference ranges for most of the conditions that would be included [1390]. Similarly, anticipating the possibility of NBS expansion has resulted in the generation of a study defining how SCID might be added to the program. Finally, NHS was introduced nationally in Oman after limited pilot testing in 2002 [1391]. Experiences with this program are detailed elsewhere along with the challenges of adding NBS to the public health system in a developing economy [1392,1393].
- Qatar—The Qatar NBS program is one of the more advanced in the region. In late 2003, collaboration began between Hamad Medical Corporation and the University Children’s Hospital of Heidelberg in Germany, updating a cord blood CH screening program operational since 1996 [1376]. The NBS collaboration began with 28 screening conditions with an eye towards expansion when and where possible. Recognizing the high numbers of clinical cases and the opportunities for early detection and treatment from NBS, HCY and HGB were added to the program in 2014. Additional MS/MS disorders were also added along with second-tier testing for MMA and methyl citric acids. Methodologies to detect LSDs were reportedly under evaluation [1394,1395]. A two-step NBS approach to diagnosing carnitine deficiency in extremely pre-term newborns has also been reported [1396]. A 2020 report noted that NBS in Qatar included over 80 disorders, which required a wide range of technologies including photometric techniques, MS/MS, and liquid chromatography. Second-tier testing was being used for some disorders (abnormal levels of methionine, homocysteine, etc.) to reduce unnecessary recall. Next generation sequencing (NGS) using an in-house NBS panel of 122 genes associated with 63 metabolic and 18 other NBS disorders was also in use. Screening availability and treatment breakthroughs with SMA make its addition to the Qatari screening panel likely [1299,1397].
- Saudi Arabia—An NBS program for PKU, CH, HGB, and G6PD was initiated by Aramco for its 60,000 employees in 1980 [1398]. Through the combined efforts of the Ministry of Health, King Salman Center for Disability and Research, King Faisal Specialist Hospital, Research Center-Riyadh (KFSHRC), and others, a national NBS program for 16 conditions based on disease frequency and availability/accessibility of confirmatory tests and treatments was begun in August 2005. The national NBS committee that selected the 16 tests was also tasked with overseeing the screening program, future planning, policy development and performance evaluation. The NBS program began with 24 centers and an aim of full population coverage within 10 years (based on funding availability). The panel of conditions was soon expanded to 17, with the addition of VLCAD, and 3 additional conditions were proposed for addition, TYR-I, HCY, and free carnitine [1399].A 2015 report of the knowledge and attitudes of Saudi mothers towards NBS found high acceptance of NBS among Saudi women but a considerable need to increase awareness by improving communications between the medical community and parents [1400]. A later study (2020) concluded similarly that medical healthcare professionals were responsible for interacting with various NBS stakeholders, including families of screened newborns and prenatal educators [1401]. In 2016, keeping with the Kingdom’s “Vision 2030” goals, an Institutional Transformation and Health Care Model was developed that included as one of its foci “Health Promotion Against Health Risks” to increase public awareness of programs such as NBS [1402].In 2016, the MOH announced the inclusion of CCHD and NHS in the national screening effort. Included in the announcement were plans for implementation in 30 MOH referral hospitals [1403]. A 2017 Letter to the Editor questioned national readiness as NBS expanded and noted the need for establishing NBS as a comprehensive system [1404]. A review NBS and the five regional programs in Saudi Arabia the following year noted the success of expanded NBS in the center of the country, and suggested that, “MOH should implement plans to start testing for the maximum number of conditions in all of its five branches at one time and with the same diseases … and there is no single region immune from any disease…” [1405]. Also in 2017, a review and review response provided regional and national incidences for all screened disorders based on data from 2005–2012, with particular emphasis on the apparently low incidence of CH [1406,1407]. In 2018, a unification/harmonization of NBS occurred during the COVID-19 pandemic when MOH laboratories were centralized from three laboratories to one laboratory, National Health Laboratory, in Riyadh. There are several smaller volume laboratories outside of Riyadh and there are now eight NBS laboratories throughout the country working in parallel.Looking to the future, a 2018 report summarized a TREC-based SCID NBS pilot study which confirmed the high incidence of SCID in the Saudi population and demonstrated, “…the feasibility of using T-NGS PID panel from Guthrie card DBSs as a new reliable, rapid, and cost-effective mutation screening method for newborns with low TREC counts” [1408]. A prospective NBS pilot study of TOXO was initiated to serve as a basis for developing neonatal screening policies in the Eastern Province of Saudi Arabia, considering the health plan and the Kingdom’s health system outlook [1409].Since population-based laboratory information on disease ranges, cutoff values, etc. for Saudi Arabia were previously lacking, a 2022 study reported clinical disease ranges for most of the Saudi NBS analytes and several of the ratios used to improve screening specificity and sensitivity [1410]. Also in 2022, a review explored the possible impact of implementing an updated health information exchange (HIE) system into an ongoing LTFU data system and the importance of having a well-established collaboration infrastructure. It also surveyed the current HIE status of the Saudi National NBS program and barriers that might be faced on implementation [1411]. Another 2022 report reviewed readiness for NGS in Saudi Arabia finding that the question of “Are we ready yet?” is still up for debate [1404,1412]. A recent publication reported on the accurate determination of biotinidase activity in serum by HPLC and its utilization as a second-tier test for the confirmation of initial positive newborn screening results [1413].
- Syria—Despite training assistance in NBS for CH from the International Atomic Agency in the 1990s, a national NBS program still does not exist in Syria [20]. There have been several studies relating to NBS in recent years. A 2015 report summarized the results of two studies looking at traditionally screened conditions (CH, GAL, CAH) and conditions identified by MS/MS (FAOs, AAs, OAs). The incidences of these disorders supported “consideration of a comprehensive nationwide neonatal screening program” [1316]. A 2015 report also summarized the results of a 5-year review of CAH cases and supported the idea of a mandatory NBS program, with further efforts needed to confirm its effectiveness in Syrian society [1414]. Another 2015 study looked at OAs and a 2019 study looked at CH with both concluding that NBS was needed [1415,1416]. Other reports supporting NBS are available in non-English journals. In early 2019, the military health authorities began a limited panel of NBS conditions for military newborns.
- Tunisia—Even though in the 1990s Tunisia was featured by the IAEA as a model NBS pilot project for CH with the intent to develop nine screening centers in the country, there is currently no systematic NBS program [1417]. More recently, studies have demonstrated the detection of hemoglobin disorders, a major local health problem, in a more cost-effective way using local materials and methods [1418]. The presence of genetic diseases amenable to NBS, including PKU, have also been reviewed, including a limited number of NBS pilot projects [1419,1420]. These studies not only assess the status of genetic diseases in Tunisia, but also emphasize the importance of early interventions, like NBS, to reduce morbidity and mortality. One report describes many of the reasons to have a national NBS program in Tunisia and the barriers to implementation and suggests a targeted cascading carrier screening alternative to implementing systematic NBS [1421]. A 2018 genetics review identifies some of the major healthcare (and NBS) challenges, noting that, “… there is an urgent need to increase genetic literacy among the health care personnel, in particular primary care practitioners, by providing the appropriate education and training. … The public must be informed and educated to seek services and counseling by creating support groups, organizing workshops, and by creating a network between the different care providers, the school or institution and the parents” [1422].
- United Arab Emirates—A national NBS program for PKU was established in the UAE in 1995. CH was added in 1998 and screening coverage was 50%. Screening conditions gradually expanded over the years to include MS/MS testing in 2011 and screening coverage reached 95%. Expansion to include even more conditions occurred in 2013 [1423]. A 2016 report pointed to possible testing improvements including integration of NBS and the premarital screening program “to better mitigate IEM disorders in the community” [1424]. Interestingly, a recent report from Gulf Cooperation Council Countries that included summaries from 3 studies in UAE noted that in the case of thalassemias, “Despite the premarital screening and genetic counseling (PMSGC) program for thalassemia, the incidence of high-risk couple marriages in GCC countries cannot be effectively diminished”. This study suggested that, at least for thalassemias, an enhanced level of consciousness about the disease and the consequences of consanguinity might be more effective than the PMSGC in reducing the incidence of disease [1425]. A 2021 report of a 5-year project in Dubai found that, among other results, G6PD deficiency was the most prevalent condition found by NBS [1426].Point-of-care screening also occurs in the UAE. A 2013 report on the implementation of CCHD in Abu Dhabi, in collaboration with Children’s National Medical Center (D.C.—USA), suggested the value of a national screening program [1427]. In 2018, the Ministry of Health and Prevention (MOHAP) launched a CCHD screening program for all newborns [1428]. A 2022 report noted that the Emirates Health Services (EHS) had adopted a stringent NBS program for CCHD and showed how an electronic medical record (EMR) could improve the efficiency of the program [1429]. Additionally, a local hearing screening project in 2021 recommended a national NHS program and highlighted “the importance of parental engagement to increase the awareness regarding timely NHS, follow-up NHS, and hearing loss in children in the UAE” [1430]. In addition to CCHD and NHS, 49 conditions from DBSs are included in screening. A national law is under consideration that would “obligate all health sectors, hospitals and families to participate in the test”. Additionally, NBS expansion to include SCID is expected sometime in 2023 [1431].A recent UAE report noted that, “Studies of genomic NBS are highly skewed towards populations in high-income countries. The evidence generated by these studies will be similarly biased and is likely to lead to disparate global implementation. Studies inclusive of historically under-represented populations are needed for equitable global access to genomic newborn screening”. It concluded with a call-to-action, commenting that, “Inclusive studies involving globally diverse populations will be needed to achieve equitable implementation of genomic screening for all newborns irrespective of their ancestries or geographic origins. Failure to achieve this goal will exacerbate the disparate access to genomic medicine for understudied populations” [1432].
- West Bank and Gaza—Approximately 60% of the Palestinian population resides in the West Bank and 40% in Gaza. NBS began in 1994 and currently, while screening laboratory services are provided by two separate laboratories, screening coverage is high and only screening for CH and PKU is universally provided [1433,1434]. Incidence studies of the IEMs in the West Bank (a collaboration between the MOH and Liege University in Belgium) and G6PD studies in Gaza support expanding the NBS program [1435]. A 2020 report defined a PKU cutoff for laboratory use to replace the kit manufacturer’s recommended cutoff, which was in use at the time in the West Bank screening laboratory [1436]. In 2021, a shortage of PKU test materials likely due to supply chain issues from the pandemic caused a backup in testing and threatened the stability of the NBS program in Gaza [1437].
- Yemen—While representatives from Yemen attended the first regional MENA meeting, there has been no information regarding the status of NBS activities in Yemen.
3.5.3. Tabular NBS Data for Middle East North Africa
3.6. Sub-Saharan Africa
3.6.1. NBS Activities with a Regional Focus
3.6.2. NBS Activities with a Country Focus
- Angola—Expanding on the success of a SCD clinic established in 2005 in Luanda, a pilot NBS program focused on sickle cell anemia (SCA) was begun in 2011 in five birthing centers in Luanda. The program was a partnership between the Angolan Ministry of Health, Chevron Corporation, and Baylor College of Medicine and demonstrated that NBS was feasible in a developing country such as Angola. Follow-up was challenging but successful in the majority of cases [1477]. A cost-effectiveness study using data from the pilot program demonstrated that NBS and early treatment would be cost effective across all scenarios by WHO criteria [1478]. The program was continued and expanded to 11 health centers in Luanda and 11 in Cabinda province but was not present in the other 16 provinces [1469]. The funding and project completed in 2020 and in 2022, the project was reported to be on hold while the government considered funding options [1470]. While the pilot utilized IEF as the laboratory screening technique, its use outside of the major population centers continues to be recognized as a challenge due to its relative technical complexity and high cost, among other considerations. As a result, POC tests are still in development as noted previously. One such study demonstrated the feasibility and diagnostic accuracy of a paper-based assay in a resource-limited clinical setting in Cabinda, Angola [1479]. A 2023 report notes that a pilot was begun in June 2023 at the Hospital Materno Infantil Dr. Manuel Pedro Azancot de Menezes in Luanda, a referral of the main maternity facility, to be used as a model for other hospitals. Specimens are sent to Lisbon, Portugal for laboratory testing as part of the ARISE project [1480].
- Benin—Pilot NBS focused on SCA began in 1993 with initial funding from the CAMPUS Program of the French Ministry and the European Union and later as a partnership between the Beninese Ministry of Health, the March of Dimes, and the University of Benin. The program used repeated information and education of pregnant women as a way of getting them to request NBS [1446]. The pilot program was successful and has slowly expanded through the years. A study of hemoglobin abnormalities in Benin highlighted the need for NBS to enhance early disease detection, prevention, and comprehensive care [1481].
- Burkina Faso—The first NBS pilot study was conducted in Ouagadougou in 2004, and the incidence of SCD in Burkina Faso was estimated to be 1 in 57 births [1482]. In 2014, Fondation Pierre Fabre signed a partnership with the Comité d’Initiative contre la Drépanocytose au Burkina (SCD Initiative Committee in Burkina), a non-profit organization, for a 3-year project to raise awareness of SCD, to improve access to care, and to offer systematic neonatal diagnosis. The overall goal was to reduce morbidity and mortality from SCD [1483]. A more recent study between 2015 and 2019 focused on NBS in Ouagadougou and Bobo-Dioulasso, another ethnic area of Burkina Faso, finding a SCD incidence of 1 in 53 births [1484]. Both studies strongly encouraged the development of a national NBS program for SCD.
- Burundi—Aside from a study of a new enzyme-linked immunosorbent assay test conducted in the Great Lakes region in Central Africa from July 2004 until July 2006, there is little information available about NBS in Burundi. In the reported study specimens from Kigali and Butare in Rwanda, Bujumbura in Burundi, and Goma in the East of the Democratic Republic of Congo were analyzed, and the technique found usable for NBS for SCD [1485].
- Cameroon—There are about 20 million inhabitants of Cameroon with a SCD carrier frequency of 8 to 34%, and there are no specialized centers for lifelong medical care of those affected. A 2015 report noted the urgent need to develop and implement policy actions in Cameroon on at least five levels including policies and practices that improve the early detection and care of SCD, e.g., neonatal screening [1486]. A 2018 report documented the use of NBS filter cards in determining the normal range for TSH in newborns at Yaounde Gyneco-Obstetric and Paediatric Hospital. This small pilot (n = 180) did not detect any cases of CH and concluded that a larger study was necessary to determine the potential for NBS in the country [1487]. And a 2020 article on knowledge, attitudes, and practices towards SCD in unmarried youths recommended NBS for SCD as a needed preventive method [1488]. The Fondation Pierre Fabre and partners from Cameroon and three Central African countries (Côte d’Ivoire, Democratic Republic of Congo, Central African Republic and) have designed a project to support the fight against SCD. This multi-country project, headed and coordinated by the Fondation Pierre Fabre and co-financed by the Agence Française de Dévelopement, intends to address the challenge of reducing morbidity and mortality linked to SCD by acting on several levels. One of these is to make NBS available free of charge at more than 50 healthcare structures partnering with the project. Several screening strategies and techniques are being utilized, including POC testing, to maximize the decentralization of access to diagnoses. Other action levels include treating, training, public awareness, and support of public authorities [1489].
- Central African Republic—Since 2019, The Fondation Pierre Fabre has provided support for the Sickle Cell Research and Treatment Centre (CRTD), a national referral center founded in 2018 at the initiative of the Central African Republic Ministry of Health and Population. The CRTD is located on the grounds of Bangui’s National University Hospital Centre and specializes in SCD treatment [1490]. The Fondation Pierre Fabre has also partnered with the Central African Republic and three Central African countries (Côte d’Ivoire, Democratic Republic of Congo, and Cameroon) have designed a project to support the fight against SCD. See a project description under Cameroon [1489]. In September 2022, the “drep.ACCI“ project was launched in Bangui, under the patronage of the First Lady of the Central African Republic. This project is a partnership between the Pasteur Institute of Bangui, the Fondation Pierre Fabre and the AFD—Agence Française de Dévelopement, which is co-financing the project, and is aimed at introducing systematic NBS, using POC tests, in three maternity wards in Bangui (40,000 newborns), as part of the national strategy to combat SCD [1491].
- Comoros—The Comoros islands consist of four volcanic islands to the west of the northern tip of Madagascar and is one of the poorest and least developed countries in the region. Since gaining independence from France in 1975 (except for one island, Mayotte, which remains as a French Department), the country has been characterized by political instability which has adversely affected the health of the population. A comprehensive look at the healthcare issues was published in 2021 [1492]. A relatively high incidence of HGBs and G6PD deficiency has been reported [1493]. To the best of our knowledge, there is no ongoing NBS activity.
- Congo, Democratic Republic—The Democratic Republic of the Congo (DRC), formerly Zaire, is the second-largest country in Africa, after Algeria, with a population of about 100 million. It is a land-locked country bordered by nine other countries and home to over 260 different tribes distinguished into four main groups: Lingala, Luba, Swahili and Kongo. The DRC has made some progress in terms of peace and stability; however, there are still many challenges, including poverty, corruption, and the ongoing conflict in the east of the country. A 2009 report documents the introduction of NBS in the DRC in 2006 and notes the main challenges are tracking new cases for confirmatory testing and early management [1494]. A 2022 study of NBS for SCD in Lubumbashi City found that NBS was widely accepted and the primary challenges to its adoption were likely to be financial and practical rather than social or cultural [1495]. A 2023 report on NBS in Butembo and Beni cities found that while the prevalence of SCD was lower compared to other regions of the country, it was still an alarming public health issue, particularly in view of the lack of SCD policies and priorities. The authors urged decision makers at national and global level to implement systematic NBS, patient/carrier education and early management programs, integrated into the national health system [1496].While some NBS is ongoing in Kinshasa, the capital city, its implementation in the rural and remote areas is extremely difficult for several reasons. One of these centers on the laboratory methodology required for NBS, which is relatively expensive and complex. For this reason, POC tests (both SickleSCAN® and HemoTypeSCTM) have been evaluated in different DRC locations with good results: a 2-month study in 2018 of 310 neonates in 9 maternity homes in the city of Kindu by SickleSCAN® [1497]; a 2-month study of 87 newborns in Kisangani city by HemoTypeSCTM [1498]; testing the parents of 58 newborns screened with HemoTypeSCTM in 8 maternity centers in Kisangani city in 2019 to validate the screening results [1499]; a 10-month study of 448 newborns in Kindu using HemoTypeSCTM during 2019/2020 [1500]; and a 2-year study of 1432 newborns in 12 maternities using HemoTypeSCTM to update the prevalence of SCD (SCA = 2.2%; twice as high as in 2010) as part of a doctoral research study [1501]. A 2022 overview of the current progress and challenges in diagnosis, and management of pediatric SCD in the DRC also noted the use of POC as a possible solution to NBS in outlying areas [1502].The Fondation Pierre Fabre and its partners from DRC and three other Central African countries (Côte d’Ivoire, Central African Republic, and Cameroon) have designed a project to support the fight against SCD. See project description under Cameroon [1489].
- Congo, Republic—The Republic of Congo is made up of 12 departments and lacks a systematic NBS program. A descriptive cross-sectional study was conducted from 1 October 2019 to 31 March 2020 throughout the Congolese national territory, to update Congolese hemoglobinopathy epidemiological data. It used a central NBS laboratory, Centre National de Référence de la drepanocytose (CNRDr), for HPLC screening. In addition to documenting the prevalence of HbS and other hemoglobins, the study concluded that implementation of a national systematic NBS program is needed to identify and provide early care for newborns with SCA, thus providing a better quality of life by limiting infectious and vaso-occlusive complications [1503].
- Cote d’Ivoire—A 2021 report provides evidence to support the use of the HemoTypeSCTM test for the rapid screening of SCD in Côte d’Ivoire. The test was accurate and easy to use, making it a promising option for improving the early diagnosis and treatment of SCD in the country [1504]. The Fondation Pierre Fabre and its partners from Côte d’Ivoire and three Central African countries (Democratic Republic of Congo, Central African Republic, and Cameroon) have designed a project to support the fight against SCD. See a project description under Cameroon [1489].
- Equatorial Guinea—Equatorial Guinea consists of two parts, two small islands and a small mainland region. Bioko Island is the northernmost part and is the site of the country’s capital. A formal NBS program does not yet exist in Equatorial Guinea. An epidemiologic study reported in 2015 confirmed the high prevalence of G6PDD and hemoglobinopathies on Bioko Island and noted that obligatory NBS, prenatal screening, and counseling for these conditions, especially HbS, were needed on the island [1505].
- Eswatini—HIV testing at birth of HIV-exposed infants using polymerase chain reaction (PCR) screening may improve the identification of infants infected with HIV in utero and accelerate antiretroviral treatment (ART) initiation. A pilot program was initiated in two maternity facilities to demonstrate the feasibility of this approach in a low-resource and high-burden setting. While high uptake of testing was documented, few newborns were found to be infected [1506]. A national NBS program is not currently in place. While HIV screening is not generally thought of as NBS, it is still included as one of the screened disorders in the New York NBS program.
- Ethiopia—While there is currently no organized NBS in Ethiopia, there is increasing interest in organizing a pilot and at least one group of university researchers is beginning the process of evaluating condition(s) to be included in a screening program and seeking funding [1507]. Only a single study related to NBS is available assessing the possibility of NBS for CH in Addis Ababa in the 1990s. This study looked at 4206 newborns without identifying a case of CH and concluded that larger studies were needed [1508]. No further studies are known.
- Gabon—There is currently limited NBS for SCD in Gabon. Screening is sometimes carried out only at the request of the mother and a recent study in rural Eastern Gabon found that about 30% of newborns obtained NBS for SCD. The barriers to acceptance of NBS for SCD were more related to habits and customs than to knowledge of the disease. For example, blood sampling at 3–4 days of age was thought to be unrealistic in an environment where traditions confine the newborn during a lunar cycle (1 month) before it is presented to the community. Establishment of rural NBS for SCD will likely require a team composed of a psychosocial and health worker familiar with rural communities, to address inhibitions related to neonatal blood collection [1509]. A 2022 report reviewed data obtained from 6 of the 9 provinces in Gabon as part of a national pilot study of NBS for SCD between January 2007 and September 2010. Results indicated a high prevalence of SCD, likely underreported since study participation required parental consent, and led the authors to conclude that compulsory NBS was needed in countries with a high incidence of malaria, like Gabon [1510].
- Ghana—The NBS program for SCD in Ghana started in 1993 limited to 2 of the 16 health regions—Greater Accra and Ashanti. Significant efforts and sacrifice have contributed to the implementation and expansion of NBS since the 1990s in Ghana and other SSA countries under the leadership of the late Dr. Kwaku Ohene-Frempong [1511]. Recently, at least two POC screening tests for SCD have been evaluated for possible use in Ghana, (see POC discussion in Section 3.6.1): a multicenter (low-, medium- and high-resource environments—Ghana, Martinique, and USA) evaluation of HemoTypeSCTM [1512]; and a prospective diagnostic accuracy study of paper-based microchip electrophoresis using a multispectral platform [1513].Despite the government’s acceptance NBS for SCD as a project for national expansion in 2010, efforts to provide more trained genetic counselors through a SCD genetic counseling training and certification program, and the creation of a mobile application (App) to enhance program efficiency, NBS is still confined to the larger cities of Kumasi and Accra, and a limited number of rural facilities [1457]. The value of the Ghana App has been recognized by ASH, through its CONSA project and, in partnership with Novartis AG (Switzerland), is providing it to the six other CONSA members: Kenya, Liberia, Nigeria, Uganda, Tanzania, and Zambia. The App allows NBS follow-up coordinators to track patients and migrate them to further care. It is being used to collect and store screening results and medical histories for diagnosed newborns (and others) and allows for offline data collection when internet connections are down, and then syncs the data once the connection is restored [1470].The lack of a robust NBS program for SCD in Ghana, and elsewhere in SSA, led to a study of SCD diagnosis patterns at Korle Bu Teaching Hospital (KBTH), the third largest hospital in Africa and a major Ghanaian referral center, between 2009 and 2013, which indicated that a SCD diagnosis was usually made at the first pain crisis. This late diagnosis excludes many patients from life-saving preventive care and argues for a more robust NBS program [1514]. After determining the feasibility and challenges of implementing NBS for SCD at KBTH and relying on a multi-year partnership with SickKids Center for Global Child Health, Toronto, and Pfizer corporation, a comprehensive NBS program was implemented in progressive phases to best overcome the various challenges of sustainability [1472,1515]. This has the potential to provide a model for other hospitals in SSA.Beginning in 2021, the Clinton Health Access Initiative (CHAI), the Gates Foundation, the Ministry of Health, the Ghana Health Service, the Sickle Cell Foundation of Ghana, and the National Newborn Screening Laboratory at Noguchi Memorial Institute for Medical Research have partnered to begin sustainable NBS expansion. To date, NBS is reaching 12 health regions with a focus on closing the gaps in care for SCD, including developing a clear and centrally coordinated pathway to comprehensive NBS, diagnosis, and treatment [1516]. In September of 2023, the American Society of Hematology, in collaboration with Revvity, organized an international, in-person laboratory training summit at the Noguchi Memorial Institute in Accra, Ghana. Representatives from seven SSA CONSA countries came together to learn, share, and develop laboratory skills with Migele® IEF NBS hemoglobinopathy techniques, procedures, and interpretations.
- Guinea—NBS does not yet exist in Guinea despite a 2012 epidemiological study that assessed the frequency of G6PDD and hemoglobinopathies and concluded that the development of NBS was needed and would help patients benefit from early diagnosis and treatment [1517]. A recent Memorandum of Understanding between the Novo Nordisk Haemophilia Foundation (NNHF) and the Fondation Pierre Fabre to jointly improve awareness, diagnosis, care, and advocacy for blood disorders in Guinea is intended to leverage synergies for a joint approach to hemophilia and SCD at the request of Guinea’s MOH. The Programme Manager, Fondation Pierre Fabre, was quoted as saying, “We want to encourage greater screening of newborns…” Implementation of the project is beginning in 2023 [1518].
- Guinea-Bissau—There is currently no organized NBS in Guinea-Bissau. A 2018 report summarized an evaluation study of the feasibility of simultaneously screening for HIV, SCD and TB in a rural area of the country using POC tests for HIV and SCD and a standardized clinical questionnaire with clinical examination for TB. This pilot study demonstrated the feasibility of this approach in rural areas as a model that could be replicated in other rural settings in low-resource countries [1519]. A 2022 article describes an epidemiologic study in two hospitals managed within the National Health System of Guinea Bissau by Italian NGOs. In a collaborative effort, newborn DBS specimens were sent to Padova for Hb quantification and molecular analysis, and Hb haplotypes of the HbSS and HbSA patients were obtained in South Africa. Higher prevalence of the HbS allele in a pediatric population compared to previous studies confirms the need to develop nationwide SCD screening and comprehensive care programs in the country, perhaps building on the POC model referenced above. A high prevalence of G6PDD was also confirmed [1520].
- Kenya—A 2019 report provided data on a NBS pilot in Kisumu Country, Kenya. Specimens were collected by heel prick onto filter paper and analyzed by IEF in the AMPATH (Academic Model for Providing Access to Health Care) reference laboratories at the Moi Teaching and Referral Hospital, Eldoret. High prevalence rates of SCD and SCT were confirmed along with high acceptability of NBS for SCD. These data were important for informing the development and implementation of NBS in Kenya. The study concluded that NBS for SCD was a valuable and needed public health tool for early identification and management of SCD [1521]. A 2020 report also described the critical role played by NBS program for SCD in Western Kenya. This report noted that NBS for SCD was established in 2012 with support from the Indiana Hemophilia and Thrombosis Center (IHTC) who trained laboratory personnel in screening techniques. The use of a POC test was suggested for consideration as screening policies are developed nationally [1522].Through partnerships with ASH, Revvity, and CONSA, and building on the 2019 pilot in Kisumu noted above, a SCD screening center was opened in December 2021. A screening package that included screening equipment to begin NBS was included, and services were intended to serve 17 other counties with high burdens of SCD [1453,1523]. As a CONSA member, Kenya is a recipient of the Ghana App for follow-up and case management (see discussion under Ghana) [1453,1470]. In Kenya, NBS is commercially available and includes 14 other screening conditions in addition to SCD [1524]. Genetic testing for 106 conditions is also commercially available [1525]. Also in Kisumu, researchers at the Kenya Medical Research Institute, Center for Global Health Research, have partnered with researchers in Canada, Bangladesh, Zambia, and Zimbabwe to validate an algorithm that derives gestational age estimates from dried bloodspot samples using metabolic data. Results from this research have the potential to assess gestational age at birth in LMIC countries where reliable estimates may otherwise be unavailable [164].
- Liberia—Updating the epidemiology of SCD in the area is always necessary for a new program such as NBS for SCD. Thus, the NBS pilot program begun in 2013 had two main objectives: to determine the feasibility of NBS in Monrovia, the capital city, and to define the incidence of various abnormal hemoglobins in this post-war, post-migration city [1526]. While the pilot data supported the need for NBS, the Ebola Virus outbreak in 2014 forced the pilot program to pause. In 2021, the Liberian MOH partnered with CONSA to initiate NBS as part of the seven-country CONSA coalition (see Section 3.6.1) and is part of the user community for the Ghana App for NBS follow-up [1453,1470,1527,1528].
- Madagascar—While representatives from Madagascar were present for the signing of the joint statement from attendees at the Paris meeting “Initiative Drépanocytose Afrique” (African Sickle-Cell disease Initiative), hosted by the Fondation Pierre Fabre in 2019, there has been little movement towards a national NBS program (see Section 3.6.1) [1476].
- Malawi—Malawi has a well-established regionalized HIV Early Infant Diagnosis program that uses DBS specimens collected within 6 weeks of birth. The first regional SCD surveillance study in Malawi used leftover HIV screening specimens to estimate the prevalence of SCD and SCT across Central Malawi where 43% of the population resides. This study, reported in 2021, was a collaboration between the Malawi MOH, the University of North Carolina Project-Malawi community tracing team and project laboratories, the Kamuzu Central Hospital, Mzimba District Hospital, Partners in Hope, and UNC Project laboratories, Cincinnati Children’s Hospital Medical Center (CCHMC) and Revvity, and showed that inherited hematological disorders, including G6PDD are common in Central Malawi and that early identification through NBS improves clinical outcomes and should be supported [1529].
- Mali—Traditional NBS methods for SCD have been limited due to cost, lack of laboratory equipment and trained personnel, and an unreliable supply chain. Studies of POC testing have shown promise as a way of initiating a broad NBS program for SCD. The HemoTypeSCTM POC test kit, which only requires capillary blood samples and unfiltered tap water, was tested over a 2-year period in Koutiala hospital in rural Mali and found to be satisfactory for NBS [1530]. Similarly, the acceptability and diagnosis performances of two POC tests (SickleScan® and H HemoTypeSCTM) were evaluated against HPLC methodology in three maternity hospitals in Mali (two urban clinics in Bamako and one rural clinic in Kayes) using cord blood, and both tests were found to be acceptable NBS tests [1531]. A study of the Emmel test routinely used to test adults for SCA was found to be severely lacking when used for screening newborns in Bamako, and its use was NOT recommended despite its low cost and relative simplicity [1532]. A Research Centre to combat SCD (CRLD) was opened in Bamako in 2010, supported by the Fondation Pierre Fabre. The center’s mission was “to screen for the disease, receive and hospitalise patients, conduct training and information campaigns, but also to perform clinical research” [1533]. Despite these efforts, there is not yet an organized national NBS program for SCD in Mali.
- Mauritania—Mauritania is an extremely poor country with a developing healthcare system. Previous studies have confirmed the presence of both G6PD deficiency and HGBs and have supported the concept of NBS as a means of decreasing infant morbidity and mortality [1534,1535,1536]. We are aware of no current efforts to introduce a national NBS program.
- Mauritius—NBS testing has been available in Mauritius since 2019. The test panel includes CF, CH, CAH, PKU and HGB. Screening is performed at C-Lab, Wellkin Hospital, Reduit, Moka. Though screening coverage is still low, 100% of babies born in Wellkin hospital are screened and some samples from other hospitals are sent to Wellkin based on recommendation of the pediatrician. Presently there is no government involvement.
- Namibia—A preliminary report describes pilot testing at Rundu Intermediate hospital in Kavango East Region using the HemoTypeSCTM POC test to determine the prevalence of SCD. Between February and March 2023, slightly more than 200 newborns were screened with the results intended to inform policy on NBS [1537].
- Nigeria—With almost 220 million people, Nigeria is Africa’s most populous country, with one of the highest birth rates in the world, and may become the third most populous country globally by 2050.There is currently no national NBS program in Nigeria; however, there have been several pilot studies in recent years to assess the feasibility and acceptability of NBS for both SCD and CH. A 2018 report on the acceptability of NBS across various attitudes to, and acceptability of, NBS in Nigeria among various socio-demographic groups (including health professionals, undergraduate students, parents of children with SCD, and SCD patients) found good acceptability of NBS with main barriers likely to be financial and practical, rather than social or cultural [1538]. A 2019 report provided a comprehensive assessment of the current burden of SCD in Nigeria with the aim of identifying surveillance and treatment gaps. Surveillance using NBS was noted as a necessity for early detection and management to improve SCD survival [1539]. Also in 2019, NBS for CH was suggested in combination with NBS for SCD, but no serious effort to screen for CH has yet appeared [1451], but no serious effort to screen for CH has yet appeared, although at least one recent report provided information on normative values of cord blood TSH in Nigerian newborns preparatory to developing NBS [1540].A number of recent reports have addressed acceptability and feasibility of NBS for SCD in different areas of the country: validation of the use of IEF as a screening tool in Awka, Southeast Nigeria [1541]; affirmation of the feasibility and acceptability of a SCD screening intervention program in Lagos State using the HemoTypeSCTM POC for ease and reduced cost [1542]; evaluation of HPLC as a suitable NBS method for HGB at the National Hospital Abuja [1543]; documentation of the feasibility and acceptability of integrating HGB screening into an existing community-based maternal–child HIV program to increase awareness and demand for NBS from pregnant women in Benue State, Nigeria [1544]; acceptance of NBS based on awareness, education and cultural beliefs with a preference for screening away from the birthing site in Ibadan, Nigeria [1545]; and improvement of NBS program over time in Kaduna State, Nigeria [1546].The use of POC testing has resulted in studies, including the report from Lagos State mentioned above on HemoTypeSCTM [1542], on their reliability and effectiveness in improving the speed of analysis and impact on acceptability on parents. At least two studies looked at integrating POC NBS into immunization clinic protocols. One addressed the feasibility of eluting blood from DBS for screening using HemoTypeSCTM (to avoid the cumbersome practice of using blood collected in capillary tubes) at immunization clinics in 6 Primary Health Centers in Abuja, Nigeria [1547], and another looked at the feasibility of both HemoTypeSCTM and SickleSCAN® versus HPLC in five immunization clinics in Abuja [1467].Nigeria is part of CONSA, and as such, has formally implemented NBS with the goal of becoming both sustainable and nationally available [1454]. The first babies with SCD identified through the CONSA program were in the Angwan Dodo Primary Health Care Centre in the University of Abuja Teaching Hospital in December 2020, with three more detected by mid-April 2021, via the CONSA Abuja Network. Since then, other babies have also been identified in the CONSA Kaduna location [1548]. Reports from NBS pilots in various locations continue including 2023 reports from Bida, North-Central Nigeria (attendees at routine immunization clinics) [1549], and Abakaliki, South-East Nigeria (post-partum mothers) [1550]. As a member nation of CONSA, the App developed for follow-up should soon be available [1469].
- Rwanda—Despite documenting that SCD should be considered a public health problem in Kigali and Butare in 2006 and that a systematic NBS for those disorders and for G6PDD is deemed reasonable, a systematic national NBS program has not yet been implemented in Rwanda [1551].
- Senegal—A pilot NBS program for SCD was first reported in 2003 [1552], leading Senegal to become one of the first countries in Africa to implement a national NBS program for SCD. More recently, focus has been on development and implementation of a health information system for data management of the blood sampling from newborns at the maternity wards and disease screening at the Center for Research and Ambulatory Care of SCD (CERPAD) [1553,1554,1555]. Significant support has come from the Fondation Pierre Fabre through their “Operational study on early detection and treatment of SCD” [1556].
- Seychelles—The health system in Seychelles was recently reviewed. Significant health system development has occurred over the past three decades, largely because of investments in both the health services and other social sectors that directly impact child survival and the health of individuals. There is an understanding that public services need to use their resources to develop stronger and more sustained initiatives, in partnership with all relevant stakeholders [1557]. As an example, NBS for six conditions (PKU, CH, CAH, CF, HGB, GAL) was implemented in mid-February 2021, made possible by a donation from multiple stakeholders. Additionally, acquisition of MS/MS equipment will allow NBS expansion in the future [1558].
- Sierra Leone—There is currently no NBS in Sierra Leone but at least one stakeholder group, The Africa Sickle Cell Center for Education and Research (ASCC4ER), called on the Sierra Leone President, through the Minister of Health and Sanitation on World Sickle Cell Day (19 June 2023), to develop a policy on NBS for SCD in Sierra Leone. Additionally, ASCC4ER designated the theme for their 2023 Sickle Cell Day commemoration, “Why newborn screening should be a priority in Sierra Leone on sickle cell disorder?” [1559]. Interest in NBS is spreading in Sierra Leone as evidenced by a comment published by collaborators at the University of Sierra Leone, Freetown, concerning the value of carefully considering how NBS results are communicated [1560]. Additional information prior to 2020 is available [1457].
- South Africa—In the 1960s (1964–1967), a urine NBS program was conducted in Johannesburg with no cases detected. A pilot NBS program in Pretoria from 1979–1981 for “amino acidaemias and CH” identified no cases but based on clinical detection of PKU and CH, a second pilot was conducted from 1981–1986 (in mostly White newborns) with sufficient case findings of CH to warrant a recommendation for “…a nationwide unified screening program…based on 5–6 regional screening laboratories” [1561,1562]. Delegates to the first national NBS workshop in Cape Town in 1987 concluded that data were insufficient to justify national NBS. A second workshop (1992) under the joint auspices of the National Pathologists Group of the Medical Association of South Africa and Genetic Services of the Department of National Health and Population Development was held at the Johannesburg airport to review NBS progress and to exchange views and consider recommendations on NBS for CH. Aside from agreeing that there were insufficient screening data on Blacks, there was no consensus on the need for screening and additional workshops were scheduled [1563]. A 2008 report on NBS for CH and GAL in the Nkangala district of the Mpumalanga province noted the continuing lack of national prevalence data. These investigators found that “a screening programme for both disorders (CH and GAL) that integrates disease identification and treatment will be highly cost effective” [1564].Currently, despite the previous studies and continual education of health professionals and the public, there is no national plan for NBS. The HIV and concomitant tuberculosis epidemics that began in the early 2000s resulted in the redirection of both financial and human resources to these infectious diseases to the detriment of genetic services [1565]. NBS is currently only available to less than 5% of births in South Africa, specifically for CH, through a limited number of government-supported facilities in parts of Cape Town, with more comprehensive screening tests offered privately. Testing for CH and MS/MS testing for other IEMs is commercially available with variable pricing “depending on which pathology laboratory the testing is done through” [1566], and are paid out-of-pocket by the patient (through medical savings), even if the patient pays for private medical insurance. The NBS laboratory at North-West University (Centre for Human Metabolomics) has provided testing services for many years with screening services available nationally [1567,1568]. Interestingly, despite the lack of a national NBS program, South Africa is listed as one of the first African countries to explore use of genomics in the diagnosis of singular congenital disorders [1451,1474]. Today, efforts continue to develop an organized, sustainable NBS program in the national public health setting. At a national NBS meeting in February 2023, plans were discussed to potentially expand CH NBS across the country and a NBS demonstration project for CH in two contrasting provinces is under development, at the request of the National Department of Health, to assist in decision making [1569].
- Sudan—A 2012 overview of the diagnosis, management, and outcome of CH in Sudan was intended to “shed a light on this important health problem” and to ask for “help establishing a national screening program”. Moreover, it noted that the cost of long-term treatment of a child with neurodevelopmental delay exceeds the cost of screening and would be cost beneficial in the long run [1570]. A 2016 report on SCD epidemiology in West Kordofan, Sudan provided evidence that the documented HbS allele frequency is one of the highest in the world. Furthermore, the knowledge, attitude and practices towards the disease are unsatisfactory, resulting in the strong recommendation to develop public health programs to control and manage SCD in the western parts of Sudan [1571]. Until now, to our knowledge, a national NBS program does not exist.
- Tanzania—Tanzania in East Africa has a population of over 65 million people with the fifth highest prevalence of SCD in the world estimated at 11,000 new cases (babies) annually. From January 2015 to November 2016, a pilot NBS program for SCD was initiated at Muhimbili National Hospital and Temeke Regional Hospital in Dar-Es-Salaam. The pilot was coordinated by Muhimbili University of Health and Allied Sciences in their hematology research laboratory using IEF. This was the first report on NBS as a health program for SCD in Tanzania and strongly supported the concept of NBS [1572]. A 2020 follow-up report reviewed the past, present, and future of NBS for SCD in Tanzania cautioning that, “The successful introduction and expansion of NBS in Tanzania will require careful planning and advocacy at community to national level” [1573]. To aid in implementation, Tanzania is a member of CONSA (see Section 3.6.1) and is one of the countries receiving the follow-up cell phone App developed in Ghana [1453,1470].Because prevalence data on SCD in Tanzania were sparse and outdated, a surveillance study was performed on residual specimens from the HIV Early Infant Diagnosis program in northwestern Tanzania from February 2017 to May 2018. This was a collaborative effort with Cincinnati Children’s Hospital Medical Center (CCHMC), USA, and found a prevalence rate twice as high as previously reported (SCD ~1.2%), reinforcing the need for NBS and expanded diagnostic services [1574]. High prevalence results (SCD ~4%) were also obtained at a hospital in Shirati (rural northern Tanzania) in a separate study undertaken from February to September 2019. In addition to birth prevalence data, this study also demonstrated the value of POC testing (HemoTypeSCTM) and the need to improve community awareness [1575].A 2021 report reviewed NBS activities related to SCD in Tanzania and analyzed the experiences and perspectives of policy makers, healthcare providers, and families on expanding and sustaining NBS for SCD and related comprehensive care services. The opportunities and areas that need to be addressed for proper implementation and sustainability of NBS for SCD in a low-resource setting were identified. Sustainability efforts at the local level were encouraging and could be a useful model for other low-resource programs [1576]. A 2022 report of a study of POC tests (HemoTypeSCTM and SickleSCAN®) showed reliability of either versus IEF, and both were found to be acceptable for screening [1577]. A 2023 report of a study at three hospitals in Dar-Es-Salaam reported that maternal health education and maternal screening for SCD were feasible and efficacious interventions to increase the knowledge and uptake of NBS, and these interventions were strongly recommended for inclusion in comprehensive care packages for pregnant women attending antenatal clinics [1578].Tanzania NBS projects have also collaborated with North American studies focused on the machine learning prediction of gestational age using metabolic screening markers resistant to ambient temperature during transport. These “Machine learning models applied to metabolomic gestational age dating offer a ladder of opportunity for providing accurate population-level gestational age estimates in LMICs settings” [441,1579,1580].
- Togo—While representatives from Togo were present for the signing of the joint statement from attendees at the Paris meeting “Initiative Drépanocytose Afrique” (African Sickle-Cell disease Initiative) hosted by the Fondation Pierre Fabre in 2019, there has been little movement towards a national NBS program (see Section 3.6.1) [1476].
- Uganda—Design and operation of the Uganda Sickle Surveillance Study (US3) was the result of partnership between the Uganda MOH, Makerere University, and CCHMC, and sought to generate critical data on the prevalence of SCD, SCT, and any comorbidities that might be associated with early mortality in people with SCD. The study period spanned from February 2014 to March 2015 and found evidence for a large sickle burden among HIV-exposed infants and a high prevalence of SCD [1581]. These data resulted in broadening of the NBS program beyond HIV infants. A 3-year (March 2015–April 2018) targeted sickle cell screening project designed by the MOH confirmed a high sickle cell burden and HIV comorbidity, with common genetic modifiers likely influencing laboratory and clinical phenotypes. Further, these data supported the concept of NBS for SCD [1582] and potential improvements by evaluating contributors affecting specimen result turnaround time and determining the associated costs. The NBS program was found to be both efficient and cost-effective with the recommendation that universal NBS using a central laboratory was the best strategy for SCD NBS. A new SCD laboratory outfitted with isoelectric focusing (IEF) equipment was constructed at the Central Public Health Laboratories through a partnership with CCHMC using local personnel recruited and trained on a standardized analytical protocol [1583]. A cost-effectiveness study of POC tests and IEF was also reported in 2019 further informing NBS policy development [1584].To examine epidemiological trends and the degree of screening coverage in high-burden districts, data from the US3 and MOH studies collected on 0–24-month-old children between February 2014 and March 2019 were obtained and analyzed. Pertinent tables and graphs were developed and are included in the referenced report [1585]. More recently, Uganda has become a part of the CONSA group of seven countries working together to improve and sustain NBS in each country (see Section 3.6.1) [1453]. An App for follow-up, developed in Ghana, should help in tracking diagnosed cases and ensuring proper long-term care [1470]. Other NBS research in Uganda has included a study of SCD awareness, and feasibility and acceptability of text messaging about screening follow-up. While over 85% of participants had a cell phone, slightly more than half preferred contact by regular telephone, citing concerns about phone access, privacy, or cost, and readability of messages [1586]. Ugandan researchers have also reported studies with gestational age and metabolic profiles. Their “findings support the notion that NBS metabolic profiles from heel-stick bloodspots can reliably determine gestational age at birth with the additional utility of accurately estimating preterm birth rates”. This has the potential to improve preterm birth surveillance and epidemiology and may, at some point, help to inform treatment and clinical management shortly after delivery [1587].On 24 May 2023, as a side event to the 76th World Health Assembly in Geneva, Switzerland, the World Coalition on Sickle Cell Disease was launched. Dr. Aceng Ocero, Uganda Minister of Health and a pediatrician, embraced the importance of NBS and encouraged other SSA countries to develop sustainable goals.
- Zambia—As one of the seven member countries of CONSA (see Section 3.6.1), the Zambia NBS program was launched on 21 April 2021, at Arthur Davison Children’s Hospital in Ndola, Zambia. NBS is integrated into the Expanded Programme on Immunization and HIV Early Infant Diagnosis (HIV-EID). Screening is initially included in three hospitals with laboratory testing at the Tropical Diseases Research Centre laboratory using IEF. As part of CONSA, a SCD electronic follow-up registry of children diagnosed with SCD will be developed that includes patient-specific demographic, medical, and laboratory information [1548].In Zambia, as in some other African countries, World Sickle Cell Day on June 19 is an opportunity for recognition, education, and renewed emphasis on NBS for SCD. A 2019 report notes the presence of a less than perfect NBS system in Zambia, but a functioning system, nonetheless. A speech by a person living with SCD at Sickle Cell Day 2019 included a passionate appeal to the MOH for comprehensive healthcare services for persons living with SCD at all healthcare levels and was assured that priority was being given. Technical experts were tasked by the MOH to develop a Zambian Strategy for SCD [1588]. At the 2023 annual Sickle Day celebration in Lusaka, the Zambian Health Minister emphasized the ministry’s commitment to expanding screening services across all provinces. “The Zambian health department stated that 8000 babies under the age of three months have been screened for SCD following the introduction of NBS services in selected hospitals in Lusaka and Copperbelt Provinces” [1589].Other NBS research has included collaboration with Canadian researchers in the development and external validation of machine learning algorithms for postnatal gestational age estimation. This allows for a more accurate surveillance of the burden of preterm birth in LMIC jurisdictions where data are currently lacking. The current algorithms developed in Canada provided accurate estimates of gestational age when applied to external cohorts from Zambia and Bangladesh with superior model performance from heel prick data compared to cord blood data [166,1590]. Zambia is also a member nation of CONSA and is a recipient of the Ghana App for follow-up and case management (see discussion under Ghana) [1453,1470].
- Zimbabwe—Despite the absence of a NBS program, researchers at the University of Zimbabwe have participated with researchers in Kenya, Zambia, Bangladesh, and Canada in validating a protocol for determining gestational age from a metabolic profile [1525]. In low-resource settings, reliable estimates of gestational age are necessary to determine the burden of preterm birth, which in turn informs policies for allocating resources and prioritizing interventions. Currently, there is no organized NBS at the national level.
3.6.3. Summary NBS Information for the SSA Region
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| 2M3HBA | 2-Methyl 3-hydroxy butyric aciduria (2-methyl-3-hydroxybutyryl CoA dehydrogenase deficiency) |
| 2MBG | 2-Methylbutyryl-CoA dehydrogenase deficiency |
| 3MCC | 3-Methylcrotonyl-CoA carboxylase deficiency |
| 3MGA | 3-Methylglutaconic aciduria (3-methylglutaconyl-CoA-hydratase deficiency) |
| 22q11.2 | 22q11.2 deletion syndrome (DiGeorge syndrome) |
| 5-OXO | Pyroglutamic acidemia |
| ADA-SCID | Adenosine deaminase-deficient SCID |
| ALD | Adrenoleukodystrophy |
| ARSA | Arylsulfatase AA |
| ASA | Argininosuccinic acidemia |
| ARG | Argininemia |
| BCKDK | Branched chain ketoacid dehydrogenase kinase deficiency |
| BIO | Biotinidase deficiency |
| BIOPT(BS) | Defects of biopterin cofactor biosynthesis |
| BIOPT(REG) | Defects of biopterin cofactor regeneration |
| BKT | β-Ketothiolase deficiency |
| CACT | Carnitine acylcarnitine translocase deficiency |
| CAH | Congenital adrenal hyperplasia |
| CBL A (or B) (or C) (or D) | Cobalamin A or B or C or D (Methylmalonic acidemia) |
| CCHD | Critical congenital heart disease |
| cCMV | Congenital cytomegalovirus |
| CF | Cystic fibrosis |
| CFSPID | Cystic fibrosis screen–positive with inconclusive diagnosis (also CRMS) |
| CRMS | Cystic fibrosis transmembrane conductance regulator related metabolic syndrome (also CFSPID) |
| CH | Congenital hypothyroidism |
| CPS-I | Carbamoyl phosphate synthetase deficiency—Type 1 |
| CTD | Carnitine transport defect |
| CIT I (or II) | Citrullinemia Type 1 (or Type 2) |
| CPT I (or IA) (or II) | Carnitine palmitoyl transferase deficiency Type 1 (or Type 1A) (or Type 2) |
| CTX | Cerebrotendinous xanthomatosis |
| CUD | Carnitine uptake deficiency |
| DE RED | Dienoyl-CoA reductase deficiency |
| DMD | Duchenne muscular dystrophy |
| EHDI | Early hearing detection and intervention (newborn hearing screening) |
| EME | Ethylmalonic encephalopathy |
| FAOD | Fatty oxidation disorder |
| FIGLU | Formiminoglutamic aciduria |
| G6PD | Glucose-6-phosphate dehydrogenase |
| GA-I (or II) | Glutaric acidemia Type 1 (or Type 2) |
| GAL | Galactosemia—non-specific—sometimes used in the enabling legislation for screening. Use of the abbreviation in the text corresponds to the author’s use in the referenced report. |
| GALE | Galactosemia Type 3 (OMIM 230350) epimerase deficiency |
| GALK | Galactosemia Type 2 (OMIM 230200) kinase deficiency |
| GALM | Galactosemia Type 4 (OMIM 618881) mutarotase deficiency |
| GALT | Galactosemia Type 1 (OMIM 230350) transferase deficiency |
| GAMT | Guanidinoacetate methyltransferase deficiency |
| HGB | Hemoglobinopathies—nonspecific, all inclusive—usually used to describe general newborn screening for all hemoglobin disorders |
| HHH | Hyperornithinemia–hyperammonemia–homocitrullinuria syndrome |
| Hb H | Hemoglobin H disease (HbH disease) |
| H-PHE | Benign hyperphenylalaninemia |
| Hb S/β-Th (or Hb S/A) | Hemoglobin S/β-thalassemia (one of the sickle cell diseases) |
| Hb S/S | Hemoglobin S/S disease (sickle cell anemia—one of the sickle cell diseases) |
| Hb S/C | Hemoglobin S/C disease (one of the sickle cell diseases) |
| HCY | Homocystinuria |
| HIV | Human immunodeficiency virus |
| HMG | 3-Hydroxy 3-methyl glutaric aciduria |
| Hyper ORN | Hyperornithinemia with gyrate deficiency |
| IBD | Inflammatory bowel disease |
| IBDD | Isobutyryl-CoA dehydrogenase deficiency |
| IVA | Isovaleric Acidemia |
| KD | Krabbe disease |
| LCHAD | Long-chain L-3-OH acyl-CoA dehydrogenase deficiency |
| LSD | Lysosomal storage disorder |
| MAL | Malonic acidemia |
| MCAD | Medium-chain acyl-CoA dehydrogenase deficiency |
| MCKAT | Medium-chain ketoacyl-CoA thiolase deficiency |
| MCD | Multiple carboxylase deficiency |
| MD 1 | Myotonic dystrophy Type 1 |
| MET | Hypermethioninemia |
| MLD | Metachromatic leukodystrophy |
| MMA | Methylmalonic acidemia (non-specific term describing the disease see CBL, MUT) |
| MPS-I (or II) (etc.) | Mucopolysaccharidosis Type 1 (or Type 2) (or others—3B, 4A, 6, etc.) |
| M/SCHAD | Medium/short-chain L3-OH acyl-CoA-dehydrogenase deficiency |
| MSUD | Maple syrup urine disease |
| MUT | Methylmalonic acidemia (mutase deficiency) |
| NHS | Newborn hearing screening (early hearing detection and intervention) |
| NICCD | Neonatal intrahepatic cholestasis caused by citrin deficiency |
| NKH | Nonketotic hyperglycinemia |
| OA | Organic aciduria |
| OTCD | Ornithine transcarbamylase deficiency |
| PA | Propionic acidemia |
| PCD | Primary carnitine deficiency |
| PD | Pompe disease |
| PID | Primary immunodeficiencies |
| PKU | Phenylketonuria |
| PRO | Prolinemia Type I/ Type II |
| PUCD | Proximal urea cycle disorder |
| SCADD | Short-chain acyl-CoA dehydrogenase deficiency |
| SCD | Sickle cell disease (a group of clinically significant sickle hemoglobin disorders) |
| SCID | Severe combined immunodeficiency |
| SMA | Spinal muscular atrophy |
| SW-CAH | Salt wasting congenital adrenal hyperplasia |
| TCL | T-cell lymphopenia |
| TFP | Trifunctional protein deficiency |
| TOXO | Congenital toxoplasmosis |
| TYR I (or II) (or III) | Tyrosinemia Type 1(or Type 2) (or Type 3) |
| Var Hb | Variant hemoglobinopathies (other than Hb S/S, Hb S/C, Hb S/A) |
| VLCAD | Very long-chain acyl-CoA dehydrogenase deficiency |
| XLA | X-linked agammaglobulinemia |
References
- Guthrie, R. The origin of newborn screening. Screening 1992, 1, 5–15. [Google Scholar] [CrossRef]
- MacCready, R.A.; Hussey, M.G. Newborn phenylketonuria detection program in Massachusetts. Am. J. Public Health Nations Health 1964, 54, 2075–2081. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, D.A., Jr. Delaware’s phenylketonuria early detection and screening program. Del. Med. J. 1963, 35, 232–234. [Google Scholar] [PubMed]
- Guthrie, R. Blood screening for phenylketonuria. JAMA—J. Am. Med. Assoc. 1961, 178, 863. [Google Scholar] [CrossRef]
- Guthrie, R.; Susi, A. A simple phenylalanine method for detecting in large populations of newborns. Pediatrics 1963, 32, 338–343. [Google Scholar] [PubMed]
- Time Inc. New way to detect a dread disease: Phenylketonuria. Life 1962, 52, 45–46. Available online: https://books.google.com/books?id=l00EAAAAMBAJ&q=Phenylketonuria#v=snippet&q=Phenylketonuria&f=false (accessed on 3 February 2024).
- Partington, M.W.; Sinnott, B. Case finding in phenylketonuria. II. The Guthrie test. Can. Med. Assoc. J. 1964, 91, 105–114. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1927280/pdf/canmedaj01057-0012.pdf (accessed on 3 February 2024).
- Cabalska, B.; Nowaczewska, I.; Duczynska, N.; Laskowska-Klita, T. Twenty-five years’ experience with newborn screening for phenylketonuria (PKU) in Poland. Screening 1993, 2, 29–32. Available online: https://www.sciencedirect.com/science/article/abs/pii/092561649390015B (accessed on 3 February 2024).
- Cahalane, S.F. Phenylketonuria. Mass screening of newborns in Ireland. Arch. Dis. Child. 1968, 43, 141–144. [Google Scholar] [CrossRef] [PubMed]
- Alm, J.; Larsson, A. Evaluation of a nation-wide neonatal metabolic screening programme in Sweden 1965–1979. Acta Paediatr. Scand. 1981, 70, 601–607. [Google Scholar] [CrossRef] [PubMed]
- Consultant Paediatricians and Medical Officers of Health of the S.E. Scotland Hospital Region. Population screening by Guthrie test for phenylketonuria in South-east Scotland. Br. Med. J. 1968, 1, 674–676. [Google Scholar] [CrossRef] [PubMed]
- Becroft, D.M.; Horn, C.R. The Guthrie screening test for phenylketonuria: A report on two years participation in the national programme. N. Z. Med. J. 1969, 69, 212–215. [Google Scholar] [PubMed]
- Cohen, B.E.; Szeinberg, A.; Peled, I.; Szeinberg, B.; Bar-Or, R. Screening program for early detection of phenylketonuria in the newborn in Israel. Isr. J. Med. Sci. 1966, 2, 156–164. [Google Scholar] [PubMed]
- Centers for Disease Control and Prevention (CDC). Ten great public health achievements—United States, 2001–2010. Morb. Mortal. Wkly. Rep. 2011, 60, 619–623. Available online: https://www.cdc.gov/mmwr/preview/mmwrhtml/mm6019a5.htm (accessed on 3 February 2024).
- United Nations. Department of Economic and Social Affairs. Transforming Our World: The 2030 Agenda for Sustainable Development. Available online: https://sdgs.un.org/2030agenda (accessed on 1 September 2023).
- Therrell, B.L.; Padilla, C.D.; Loeber, J.G.; Khneisser, I.; Saadallah, A.; Borrajo, G.J.; Adams, J. Current status of newborn screening worldwide: 2015. Semin. Perinatol. 2015, 39, 171–187. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.M.G.; Jungner, G. Principles and Practice of Screening for Disease; Public Health Papers 34; World Health Organization: Geneva, Switzerland, 1968; pp. 26–27. [Google Scholar]
- Therrell, B.L., Jr.; Hannon, W.H. Guidelines on the Prevention and Control of Phenylketonuria (PKU); WHO/HDP/PKU/GL/90.4; World Health Organization, Division of Noncommunicable Diseases and Health Technology, Hereditary Diseases Programme: Geneva, Switzerland, 1990; pp. 1–40. [Google Scholar]
- Hannon, W.H.; Therrell, B.L., Jr. Guidelines on the Prevention and Control of Congenital Hypothyroidism; WHO/HDP/CON.HYPO/GL/90.4; World Health Organization, Division of Noncommunicable Diseases and Health Technology, Hereditary Diseases Programme: Geneva, Switzerland, 1991; pp. 1–39. [Google Scholar]
- Solanki, K.K. Training programmes for developing countries. J. Inherit. Metab. Dis. 2007, 30, 596–599. [Google Scholar] [CrossRef] [PubMed]
- Therrell, B.L., Jr.; David-Padilla, C. Screening of Newborns for Congenital Hypothyroidism; International Atomic Energy Agency: Vienna, Austria, 2005; pp. 1–122. [Google Scholar]
- WHO Fifty-Ninth World Health Assembly. Sickle Cell Anaemia. Report by the Secretariat. 24 April 2006. Available online: https://apps.who.int/gb/archive/pdf_files/WHA59/A59_9-en.pdf (accessed on 1 September 2023).
- WHO Regional Office for Africa. Sickle-Cell Disease: A Strategy for the WHO African Region. AFR/RC60/8. 22 June 2010. Available online: https://apps.who.int/iris/handle/10665/1682 (accessed on 8 September 2023).
- WHO Regional Office for Europe. Screening Programmes: A Short Guide. Increase Effectiveness, Maximize Benefits and Minimize Harm; World Health Organization Regional Office for Europe, UN City: Copenhagen, Denmark, 2020; pp. 14, 19, 28, 30, 32, 40, 53; Available online: https://apps.who.int/iris/handle/10665/330829 (accessed on 3 February 2024).
- March of Dimes. International Conference on Birth Defects and Disabilities in the Developing World. Dr. Anshu Banerjee Keynote Address. 1 March 2023. Available online: https://www.marchofdimes.org/our-work/conference-information (accessed on 10 January 2024).
- Loeber, J.G. Neonatal screening in Europe; the situation in 2004. J. Inherit. Metab. Dis. 2007, 30, 430–438, Erratum in J. Inherit. Metab. Dis. 2008, 31, 469. [Google Scholar] [CrossRef] [PubMed]
- Therrell, B.L.; Adams, J. Newborn screening in North America. J. Inherit. Metab. Dis. 2007, 30, 447–465. [Google Scholar] [CrossRef]
- Bodamer, O.A.; Hoffman, G.F.; Lindner, M. Expanded newborn screening in Europe 2007. J. Inherit. Metab. Dis. 2007, 30, 439–444. [Google Scholar] [CrossRef] [PubMed]
- Padilla, C.D.; Therrell, B.L. Newborn screening in the Asia Pacific region. J. Inherit. Metab. Dis. 2007, 30, 490–506. [Google Scholar] [CrossRef] [PubMed]
- Borrajo, G.J. Newborn screening in Latin America at the beginning of the 21st century. J. Inherit. Metab. Dis. 2007, 30, 466–481. [Google Scholar] [CrossRef] [PubMed]
- Saadallah, A.A.; Rashed, M.S. Newborn screening: Experiences in the Middle East and North Africa. J. Inherit. Metab. Dis. 2007, 30, 482–489. [Google Scholar] [CrossRef] [PubMed]
- Groulx-Boivin, E.; Osman, H.; Chakraborty, P.; Lintern, S.; Oskoui, M.; Selby, K.; Van Caeseele, P.; Wyatt, A.; McMillan, H.J. Variability in newborn screening across Canada: Spinal muscular atrophy and beyond. Can. J. Neurol. Sci. 2024, 51, 203–209. [Google Scholar] [CrossRef]
- U.S. Congress. Public Law 106-310. Title XXVI—Children’s Health Act of 2000. Available online: https://www.govinfo.gov/content/pkg/PLAW-106publ310/html/PLAW-106publ310.htm (accessed on 1 September 2023).
- Health Resources and Services Administration. History of the ACHDNC. Available online: https://www.hrsa.gov/advisory-committees/heritable-disorders/timeline (accessed on 19 September 2023).
- Watson, M.S.; Lloyd-Puryear, M.A.; Howell, R.R. The progress and future of US newborn screening. Int. J. Neonatal Screen. 2022, 8, 41. [Google Scholar] [CrossRef] [PubMed]
- American College of Medical Genetics Newborn Screening Expert Group. Newborn screening: Toward a uniform screening panel and system. Genet. Med. 2006, 8 (Suppl. S1), 1S–252S. [Google Scholar] [CrossRef]
- Canadian Organization for Rare Disorders. Follow Up Letter to CORD from the Ministry of Health about Newborn Screening Recommendations. 29 August 2016. Available online: https://www.raredisorders.ca/content/uploads/NBS-Stakeholder-Letter-2016-CORD.pdf (accessed on 26 December 2023).
- Sweetman, L.; Millington, D.S.; Therrell, B.L.; Hannon, W.H.; Popovich, B.; Watson, M.S.; Mann, M.Y.; Lloyd-Puryear, M.A.; van Dyck, P.C. Naming and counting disorders (conditions) included in newborn screening panels. Pediatrics 2006, 117 (Suppl. 3), S308–S314. [Google Scholar] [CrossRef] [PubMed]
- Kemper, A.R.; Green, N.S.; Calonge, N.; Lam, W.K.; Comeau, A.M.; Goldenberg, A.J.; Ojodu, J.; Prosser, L.A.; Tanksley, S.; Bocchini, J.A., Jr. Decision-making process for conditions nominated to the recommended uniform screening panel: Statement of the US Department of Health and Human Services Secretary’s Advisory Committee on Heritable Disorders in Newborns and Children. Genet. Med. 2014, 16, 183–187. [Google Scholar] [CrossRef] [PubMed]
- Health Resources and Services Administrated. Previously Nominated Conditions. Available online: https://www.hrsa.gov/advisory-committees/heritable-disorders/rusp/previous-nominations.html (accessed on 10 January 2024).
- Beck, M.; Frank, B.; Minaeian, S.; Nicholls, S.G. Decision-making about newborn screening panels in Canada: Risk management and public participation. In Democratizing Risk Governance; Gattinger, M., Ed.; Palgrave Macmillan: Cham, Switzerland, 2023; pp. 217–243. [Google Scholar] [CrossRef]
- McCandless, S.E.; Wright, E.J. Mandatory newborn screening in the United States: History, current status, and existential challenges. Birth Defects Res. 2020, 112, 350–366. [Google Scholar] [CrossRef] [PubMed]
- Sontag, M.K.; Miller, J.I.; McKasson, S.; Sheller, R.; Edelman, S.; Yusuf, C.; Singh, S.; Sarkar, D.; Bocchini, J.; Scott, J.; et al. Newborn screening timeliness quality improvement initiative: Impact of national recommendations and data repository. PLoS ONE 2020, 15, e0231050. [Google Scholar] [CrossRef]
- Simon, N.J.; Atkins, A.; Yusuf, C.; Tarini, B.A. Systems integration: The next frontier in newborn-screening timeliness. J. Public Health Manag. Pract. 2020, 26, E8–E15. [Google Scholar] [CrossRef] [PubMed]
- Kellar-Guenther, Y.; McKasson, S.; Hale, K.; Singh, S.; Sontag, M.K.; Ojodu, J. Implementing statewide newborn screening for new disorders: U.S. program experiences. Int. J. Neonatal Screen. 2020, 6, 35. [Google Scholar] [CrossRef] [PubMed]
- Lloyd-Puryear, M.; Brower, A.; Berry, S.A.; Brosco, J.P.; Bowdish, B.; Watson, M.S. Foundation of the Newborn Screening Translational Research Network and its tools for research. Genet. Med. 2019, 21, 1271–1279. [Google Scholar] [CrossRef]
- Brower, A.; Chan, K.; Williams, M.; Berry, S.; Currier, R.; Rinaldo, P.; Caggana, M.; Gaviglio, A.; Wilcox, W.; Steiner, R.; et al. Population-based screening of newborns: Findings from the NBS expansion study (Part One). Front. Genet. 2022, 13, 867337. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.; Brower, A.; Williams, M.S. Population-based screening of newborns: Findings from the newborn screening expansion study (Part Two). Front. Genet. 2022, 13, 867354. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.; Hu, Z.; Bush, L.W.; Cope, H.; Holm, I.A.; Kingsmore, S.F.; Wilhelm, K.; Scharfe, C.; Brower, A. NBSTRN tools to advance newborn screening research and support newborn screening stakeholders. Int. J. Neonatal Screen. 2023, 9, 63. [Google Scholar] [CrossRef]
- Brower, A.; Chan, K.; Hartnett, M.; Taylor, J. The longitudinal pediatric data resource: Facilitating longitudinal collection of health information to inform clinical care and guide newborn screening efforts. Int. J. Neonatal Screen. 2021, 7, 37. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, K.; Edick, M.J.; Berry, S.A.; Hartnett, M.; Brower, A. Using long-term follow-up data to classify genetic variants in newborn screened conditions. Front. Genet. 2022, 13, 859837. [Google Scholar] [CrossRef] [PubMed]
- Sontag, M.K.; Yusuf, C.; Grosse, S.D.; Edelman, S.; Miller, J.I.; McKasson, S.; Kellar-Guenther, Y.; Gaffney, M.; Hinton, C.F.; Cuthbert, C.; et al. Infants with congenital disorders identified through newborn screening—United States, 2015–2017. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 1265–1268. [Google Scholar] [CrossRef] [PubMed]
- Gaviglio, A.; McKasson, S.; Singh, S.; Ojodu, J. Infants with congenital diseases identified through newborn screening-United States, 2018–2020. Int. J. Neonatal Screen. 2023, 9, 23. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Ojodu, J.; Kemper, A.R.; Lam, W.K.K.; Grosse, S.D. Implementation of newborn screening for conditions in the United States first recommended during 2010–2018. Int. J. Neonatal Screen. 2023, 9, 20. [Google Scholar] [CrossRef] [PubMed]
- Quinn, J.; Orange, J.S.; Modell, V.; Modell, F. The case for severe combined immunodeficiency (SCID) and T cell lymphopenia newborn screening: Saving lives…one at a time. Immunol. Res. 2020, 68, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Sheller, R.; Ojodu, J.; Griffin, E.; Edelman, S.; Yusuf, C.; Pigg, T.; Huston, A.; Fitzek, B.; Boyle, J.G.; Singh, S. The landscape of severe combined immunodeficiency newborn screening in the United States in 2020: A review of screening methodologies and targets, communication pathways, and long-term follow-up practices. Front. Immunol. 2020, 11, 577853. [Google Scholar] [CrossRef] [PubMed]
- Currier, R.; Puck, J.M. SCID newborn screening: What we’ve learned. J. Allergy Clin. Immunol. 2021, 147, 417–426. [Google Scholar] [CrossRef] [PubMed]
- Taki, M.; Miah, T.; Secord, E. Newborn screening for severe combined immunodeficiency. Immunol. Allergy Clin. N. Am. 2021, 41, 543–553. [Google Scholar] [CrossRef] [PubMed]
- Kobrynski, L.J. Newborn screening in the diagnosis of primary immunodeficiency. Clin. Rev. Allergy Immunol. 2022, 63, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Thakar, M.S.; Logan, B.R.; Puck, J.M.; Dunn, E.A.; Buckley, R.H.; Cowan, M.J.; O’Reilly, R.J.; Kapoor, N.; Satter, L.F.; Pai, S.Y.; et al. Measuring the effect of newborn screening on survival after haematopoietic cell transplantation for severe combined immunodeficiency: A 36-year longitudinal study from the Primary Immune Deficiency Treatment Consortium. Lancet 2023, 402, 129–140. [Google Scholar] [CrossRef] [PubMed]
- Barmettler, S.; Coffey, K.; Smith, M.J.; Chong, H.J.; Pozos, T.C.; Seroogy, C.M.; Walter, J.; Abraham, R.S. Functional confirmation of DNA repair defect in ataxia telangiectasia (AT) infants identified by newborn screening for severe combined immunodeficiency (NBS SCID). J. Allergy Clin. Immunol. Pract. 2021, 9, 723–732.e3. [Google Scholar] [CrossRef] [PubMed]
- Carol, H.A.; Ochfeld, E.N.; Ahmed, A. In-utero exposure to immunosuppressive medications resulting in abnormal newborn screening for severe combined immunodeficiency: A case series on natural history and management. Immunol. Res. 2022, 70, 561–565. [Google Scholar] [CrossRef] [PubMed]
- Scott, E.M.; Chandra, S.; Li, J.; Robinette, E.D.; Brown, M.F.; Wenger, O.K. Abnormal newborn screening follow-up for severe combined immunodeficiency in an Amish Cohort with cartilage-hair hypoplasia. J. Clin. Immunol. 2020, 40, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Tallar, M.; Routes, J. Omenn syndrome identified by newborn screening. Clin. Perinatol. 2020, 47, 77–86. [Google Scholar] [CrossRef] [PubMed]
- Buchbinder, D.; Walter, J.E.; Butte, M.J.; Chan, W.Y.; Chitty Lopez, M.; Dimitriades, V.R.; Dorsey, M.J.; Nugent, D.J.; Puck, J.M.; Singh, J.; et al. When screening for severe combined immunodeficiency (SCID) with T cell receptor excision circles is not SCID: A case-based review. J. Clin. Immunol. 2021, 41, 294–302. [Google Scholar] [CrossRef] [PubMed]
- Adatia, A.; Ling, L.; Chakraborty, P.; Brick, L.; Brager, R. Neonatal abstinence syndrome is a potential cause of low TREC copy number. Allergy Asthma Clin. Immunol. 2021, 17, 115. [Google Scholar] [CrossRef] [PubMed]
- Kubala, S.A.; Sandhu, A.; Palacios-Kibler, T.; Ward, B.; Harmon, G.; DeFelice, M.L.; Bundy, V.; Younger, M.E.M.; Lederman, H.; Liang, H.; et al. Natural history of infants with non-SCID T cell lymphopenia identified on newborn screen. Clin. Immunol. 2022, 245, 109182. [Google Scholar] [CrossRef] [PubMed]
- Raspa, M.; Lynch, M.; Squiers, L.; Gwaltney, A.; Porter, K.; Peay, H.; Huston, A.; Fitzek, B.; Boyle, J.G. Information and emotional support needs of families whose infant was diagnosed with SCID through newborn screening. Front. Immunol. 2020, 11, 885. [Google Scholar] [CrossRef] [PubMed]
- Mongkonsritragoon, W.; Huang, J.; Fredrickson, M.; Seth, D.; Poowuttikul, P. Positive newborn screening for severe combined immunodeficiency: What should the pediatrician do? Clin. Med. Insights Pediatr. 2023, 17, 11795565231162839. [Google Scholar] [CrossRef] [PubMed]
- Dvorak, C.C.; Haddad, E.; Heimall, J.; Dunn, E.; Buckley, R.H.; Kohn, D.B.; Cowan, M.J.; Pai, S.Y.; Griffith, L.M.; Cuvelier, G.D.E.; et al. The diagnosis of severe combined immunodeficiency (SCID): The Primary Immune Deficiency Treatment Consortium (PIDTC) 2022 definitions. J. Allergy Clin. Immunol. 2023, 151, 539–546. [Google Scholar] [CrossRef] [PubMed]
- Dvorak, C.C.; Haddad, E.; Heimall, J.; Dunn, E.; Cowan, M.J.; Pai, S.Y.; Kapoor, N.; Satter, L.F.; Buckley, R.H.; O’Reilly, R.J.; et al. The diagnosis of severe combined immunodeficiency: Implementation of the PIDTC 2022 definitions. J. Allergy Clin. Immunol. 2023, 151, 547–555.e5. [Google Scholar] [CrossRef] [PubMed]
- Gaviglio, A.; Lasarev, M.; Sheller, R.; Singh, S.; Baker, M. Newborn Screening for Severe Combined Immunodeficiency: Lessons Learned from Screening and Follow-Up of the Preterm Newborn Population. Int. J. Neonatal Screen. 2023, 9, 68. [Google Scholar] [CrossRef] [PubMed]
- Sakai-Bizmark, R.; Chang, R.R.; Martin, G.R.; Hom, L.A.; Marr, E.H.; Ko, J.; Goff, D.A.; Mena, L.A.; von Kohler, C.; Bedel, L.E.M.; et al. Current postlaunch implementation of state mandates of newborn screening for critical congenital heart disease by pulse oximetry in U.S. states and hospitals. Am. J. Perinatol. 2022. [Google Scholar] [CrossRef]
- Robinson, D.L.; Craig, M.S.; Wells, R.S.; Liesemer, K.N.; Studer, M.A. Newborn screening pulse oximetry to detect critical congenital heart disease: A follow-up survey of current practice at Army, Navy and Air Force Hospitals. Mil. Med. 2019, 184, 826–831. [Google Scholar] [CrossRef] [PubMed]
- Wong, K.K.; Fournier, A.; Fruitman, D.S.; Graves, L.; Human, D.G.; Narvey, M.; Russell, J.L. Canadian Cardiovascular Society/Canadian Pediatric Cardiology Association Position Statement on Pulse Oximetry Screening in Newborns to enhance detection of critical congenital heart disease. Can. J. Cardiol. 2017, 33, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Keutzer, J.M. Establishing Pompe disease newborn screening: The role of industry. Int. J. Neonatal Screen. 2020, 6, 55. [Google Scholar] [CrossRef] [PubMed]
- Ames, E.G.; Fisher, R.; Kleyn, M.; Ahmad, A. Current practices for U.S. newborn screening of Pompe disease and MPSI. Int. J. Neonatal Screen. 2020, 6, 72. [Google Scholar] [CrossRef] [PubMed]
- Hale, K.; Kellar-Guenther, Y.; McKasson, S.; Singh, S.; Ojodu, J. Expanding newborn screening for Pompe Disease in the United States: The NewSTEPs New Disorders Implementation Project, a resource for new disorder implementation. Int. J. Neonatal Screen. 2020, 6, 48. [Google Scholar] [CrossRef] [PubMed]
- Sanders, K.A.; Gavrilov, D.K.; Oglesbee, D.; Raymond, K.M.; Tortorelli, S.; Hopwood, J.J.; Lorey, F.; Majumdar, R.; Kroll, C.A.; McDonald, A.M.; et al. A Comparative effectiveness study of newborn screening methods for four lysosomal storage disorders. Int. J. Neonatal Screen. 2020, 6, 44. [Google Scholar] [CrossRef] [PubMed]
- Smith, L.D.; Bainbridge, M.N.; Parad, R.B.; Bhattacharjee, A. Second tier molecular genetic testing in newborn screening for Pompe disease: Landscape and challenges. Int. J. Neonatal Screen. 2020, 6, 32. [Google Scholar] [CrossRef] [PubMed]
- Washburn, J.; Millington, D.S. Digital microfluidics in newborn screening for mucopolysaccharidoses: A progress report. Int. J. Neonatal Screen. 2020, 6, 78. [Google Scholar] [CrossRef] [PubMed]
- Wasserstein, M.P.; Orsini, J.J.; Goldenberg, A.; Caggana, M.; Levy, P.A.; Breilyn, M.; Gelb, M.H. The future of newborn screening for lysosomal disorders. Neurosci. Lett. 2021, 760, 136080. [Google Scholar] [CrossRef] [PubMed]
- Richardson, J.S.; Kemper, A.R.; Grosse, S.D.; Lam, W.K.K.; Rose, A.M.; Ahmad, A.; Gebremariam, A.; Prosser, L.A. Health and economic outcomes of newborn screening for infantile-onset Pompe disease. Genet. Med. 2021, 23, 758–766. [Google Scholar] [CrossRef] [PubMed]
- Prakash, S.; Penn, J.D.; Jackson, K.E.; Dean, L.W. Newborn screening for Pompe disease: Parental experiences and follow-up care for a late-onset diagnosis. J. Genet. Couns. 2022, 31, 1404–1420. [Google Scholar] [CrossRef] [PubMed]
- Jalal, K.; Carter, R.L.; Barczykowski, A.; Tomatsu, S.; Langan, T.J. A roadmap for potential improvement of newborn screening for inherited metabolic diseases following recent developments and successful applications of bivariate normal limits for pre-symptomatic detection of MPS I, Pompe disease, and Krabbe disease. Int. J. Neonatal Screen. 2022, 8, 61. [Google Scholar] [CrossRef]
- Herbst, Z.M.; Urdaneta, L.; Klein, T.; Burton, B.K.; Basheeruddin, K.; Liao, H.C.; Fuller, M.; Gelb, M.H. Evaluation of two methods for quantification of glycosaminoglycan biomarkers in newborn dried blood spots from patients with severe and attenuated mucopolysaccharidosis type II. Int. J. Neonatal Screen. 2022, 8, 9. [Google Scholar] [CrossRef] [PubMed]
- Millington, D.S.; Ficicioglu, C. Addition of MPS-II to the Recommended Uniform Screening Panel in the United States. Int. J. Neonatal Screen. 2022, 8, 55. [Google Scholar] [CrossRef] [PubMed]
- Ream, M.A.; Lam, W.K.K.; Grosse, S.D.; Ojodu, J.; Jones, E.; Prosser, L.A.; Rosé, A.M.; Comeau, A.M.; Tanksley, S.; Powell, C.M.; et al. Evidence and recommendation for mucopolysaccharidosis type II newborn screening in the United States. Genet. Med. 2023, 25, 100330. [Google Scholar] [CrossRef]
- Dorley, M.C.; Dizikes, G.J.; Pickens, C.A.; Cuthbert, C.; Basheeruddin, K.; Gulamali-Majid, F.; Hetterich, P.; Hietala, A.; Kelsey, A.; Klug, T.; et al. Harmonization of newborn screening results for Pompe disease and mucopolysaccharidosis Type I. Int. J. Neonatal Screen. 2023, 9, 11. [Google Scholar] [CrossRef] [PubMed]
- Turk, B.R.; Theda, C.; Fatemi, A.; Moser, A.B. X-linked adrenoleukodystrophy: Pathology, pathophysiology, diagnostic testing, newborn screening and therapies. Int. J. Dev. Neurosci. 2020, 80, 52–72. [Google Scholar] [CrossRef] [PubMed]
- Mallack, E.J.; Turk, B.R.; Yan, H.; Price, C.; Demetres, M.; Moser, A.B.; Becker, C.; Hollandsworth, K.; Adang, L.; Vanderver, A.; et al. MRI surveillance of boys with X-linked adrenoleukodystrophy identified by newborn screening: Meta-analysis and consensus guidelines. J. Inherit. Metab. Dis. 2021, 44, 728–739. [Google Scholar] [CrossRef] [PubMed]
- Moser, A.B.; Seeger, E.; Raymond, G.V. Newborn screening for X-linked adrenoleukodystrophy: Past, present, and future. Int. J. Neonatal Screen. 2022, 8, 16. [Google Scholar] [CrossRef] [PubMed]
- Pierpont, E.I.; Isaia, A.R.; McCoy, E.; Brown, S.J.; Gupta, A.O.; Eisengart, J.B. Neurocognitive and mental health impact of adrenoleukodystrophy across the lifespan: Insights for the. J. Inherit. Metab. Dis. 2023, 46, 174–193. [Google Scholar] [CrossRef] [PubMed]
- Ramirez Alcantara, J.; Grant, N.R.; Sethuram, S.; Nagy, A.; Becker, C.; Sahai, I.; Stanley, T.; Halper, A.; Eichler, F.S. Early detection of adrenal insufficiency: The impact of newborn screening for adrenoleukodystrophy. J. Clin. Endocrinol. Metab. 2023, 108, e1306–e1315. [Google Scholar] [CrossRef] [PubMed]
- Pitts, L.; White, J.M.; Ladores, S.; Wilson, C.M. The impacts of adrenoleukodystrophy newborn screening on the evaluation of adrenal dysfunction in male children: An integrative literature review. J. Pediatr. Nurs. 2023, 72, e53–e70. [Google Scholar] [CrossRef] [PubMed]
- Jalali, A.; Rothwell, E.; Botkin, J.R.; Anderson, R.A.; Butterfield, R.J.; Nelson, R.E. Cost-Effectiveness of nusinersen and universal newborn screening for spinal muscular atrophy. J. Pediatr. 2020, 227, 274–280.e2. [Google Scholar] [CrossRef]
- Hale, K.; Ojodu, J.; Singh, S. Landscape of spinal muscular atrophy newborn screening in the United States: 2018–2021. Int. J. Neonatal Screen. 2021, 7, 33. [Google Scholar] [CrossRef] [PubMed]
- Ream, M.A.; Lam, W.K.K.; Grosse, S.D.; Ojodu, J.; Jones, E.; Prosser, L.A.; Rose, A.M.; Comeau, A.M.; Tanksley, S.; Powell, C.M.; et al. Evidence and recommendation for guanidinoacetate methyltransferase deficiency newborn screening. Pediatrics 2023, 152, e2023062100. [Google Scholar] [CrossRef] [PubMed]
- Hart, K.; Rohrwasser, A.; Wallis, H.; Golsan, H.; Shao, J.; Anderson, T.; Wang, X.; Szabo-Fresnais, N.; Morrissey, M.; Kay, D.M.; et al. Prospective identification by neonatal screening of patients with guanidinoacetate methyltransferase deficiency. Mol. Genet. Metab. 2021, 134, 60–64. [Google Scholar] [CrossRef] [PubMed]
- Schrier Vergano, S.A.; Kanungo, S.; Arnold, G. Making decisions about Krabbe disease newborn screening. Pediatrics 2022, 149, e2021053175. [Google Scholar] [CrossRef] [PubMed]
- Kurtzberg, J.; Matern, D.; Orsini, J.J.; Gelb, M.; Pike-Langenfeld, S.; Brackbill, L.; Grantham, A.; Steyermark, A.C. Making decisions about Krabbe disease newborn screening. Pediatrics 2022, 150, e2022057888. [Google Scholar] [CrossRef] [PubMed]
- Pike-Langenfeld, S. Why must the debate continue on Krabbe disease newborn screening? Am. J. Med. Genet. C Semin. Med. Genet. 2022, 190, 153–155. [Google Scholar] [CrossRef] [PubMed]
- Bonkowsky, J.L.; Wilkes, J.; Baker, M.; Grantham, A.; Kurtzberg, J.; Orsini, J. Newborn screening for Krabbe Disease and identification of minority patients. Pediatr. Neurol. 2024, 151, 65–67. [Google Scholar] [CrossRef]
- Travert, G.; Heeley, M.; Heeley, A. History of newborn screening for cystic fibrosis-the early years. Int. J. Neonatal Screen. 2020, 6, 8. [Google Scholar] [CrossRef] [PubMed]
- Scotet, V.; Gutierrez, H.; Farrell, P.M. Newborn screening for CF across the globe-where is it worthwhile? Int. J. Neonatal Screen. 2020, 6, 18. [Google Scholar] [CrossRef] [PubMed]
- Farrell, M.H.; Sims, A.M.; La Pean Kirschner, A.; Farrell, P.M.; Tarini, B.A. Vulnerable Child Syndrome and newborn screening carrier results for cystic fibrosis or sickle cell. J. Pediatr. 2020, 224, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Martiniano, S.L.; Elbert, A.A.; Farrell, P.M.; Ren, C.L.; Sontag, M.K.; Wu, R.; McColley, S.A. Outcomes of infants born during the first 9 years of CF newborn screening in the United States: A retrospective Cystic Fibrosis Foundation Patient Registry cohort study. Pediatr. Pulmonol. 2021, 56, 3758–3767. [Google Scholar] [CrossRef] [PubMed]
- Gonska, T.; Keenan, K.; Au, J.; Dupuis, A.; Chilvers, M.A.; Burgess, C.; Bjornson, C.; Fairservice, L.; Brusky, J.; Kherani, T.; et al. Outcomes of cystic fibrosis screening-positive infants with inconclusive diagnosis at school age. Pediatrics 2021, 148, e2021051740. [Google Scholar] [CrossRef] [PubMed]
- Kharrazi, M.; Sacramento, C.; Comeau, A.M.; Hale, J.E.; Caggana, M.; Kay, D.M.; Lee, R.; Reilly, B.; Thompson, J.D.; Nasr, S.Z.; et al. Missed cystic fibrosis newborn screening cases due to immunoreactive trypsinogen levels below program cutoffs: A national survey of risk factors. Int. J. Neonatal Screen. 2022, 8, 58. [Google Scholar] [CrossRef] [PubMed]
- Sontag, M.K.; Miller, J.I.; McKasson, S.; Gaviglio, A.; Martiniano, S.L.; West, R.; Vazquez, M.; Ren, C.L.; Farrell, P.M.; McColley, S.A.; et al. Newborn screening for cystic fibrosis: A qualitative study of successes and challenges from universal screening in the United States. Int. J. Neonatal Screen. 2022, 8, 38. [Google Scholar] [CrossRef] [PubMed]
- Rosenfeld, M.; Ostrenga, J.; Cromwell, E.A.; Magaret, A.; Szczesniak, R.; Fink, A.; Schechter, M.S.; Faro, A.; Ren, C.L.; Morgan, W.; et al. Real-world associations of US cystic fibrosis newborn screening programs with nutritional and pulmonary outcomes. JAMA Pediatr. 2022, 176, 990–999. [Google Scholar] [CrossRef] [PubMed]
- McGarry, M.E.; Ren, C.L.; Wu, R.; Farrell, P.M.; McColley, S.A. Detection of disease-causing CFTR variants in state newborn screening programs. Pediatr. Pulmonol. 2023, 58, 465–474. [Google Scholar] [CrossRef] [PubMed]
- Rehani, M.R.; Marcus, M.S.; Harris, A.B.; Farrell, P.M.; Ren, C.L. Variation in cystic fibrosis newborn screening algorithms in the United States. Pediatr. Pulmonol. 2023, 58, 927–933. [Google Scholar] [CrossRef] [PubMed]
- McColley, S.A.; Martiniano, S.L.; Ren, C.L.; Sontag, M.K.; Rychlik, K.; Balmert, L.; Elbert, A.; Wu, R.; Farrell, P.M. Disparities in first evaluation of infants with cystic fibrosis since implementation of newborn screening. J. Cyst. Fibros. 2023, 22, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Farrell, P.M. Why cystic fibrosis newborn screening programs have failed to meet original expectations… thus far. Mol. Genet. Metab. 2023, 140, 107679. [Google Scholar] [CrossRef] [PubMed]
- van Vliet, K.; Dijkstra, A.M.; Bouva, M.J.; van der Krogt, J.; Bijsterveld, K.; van der Sluijs, F.; de Sain-van der Velden, M.G.; Koop, K.; Rossi, A.; Thomas, J.A.; et al. Maleic acid is a biomarker for maleylacetoacetate isomerase deficiency; implications for newborn screening of tyrosinemia type 1. J. Inherit. Metab. Dis. 2023, 46, 1104–1113. [Google Scholar] [CrossRef] [PubMed]
- Bender, M.A.; Yusuf, C.; Davis, T.; Dorley, M.C.; Del Pilar Aguinaga, M.; Ingram, A.; Chan, M.S.; Ubaike, J.C.; Hassell, K.; Ojodu, J.; et al. Newborn screening practices and alpha-thalassemia detection—United States, 2016. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 1269–1272. [Google Scholar] [CrossRef] [PubMed]
- Bender, M.A.; Hulihan, M.; Dorley, M.C.; Aguinaga, M.D.P.; Ojodu, J.; Yusuf, C. Newborn screening practices for beta-thalassemia in the United States. Int. J. Neonatal Screen. 2021, 7, 83. [Google Scholar] [CrossRef] [PubMed]
- Khangura, S.D.; Potter, B.K.; Davies, C.; Ducharme, R.; Bota, A.B.; Hawken, S.; Wilson, K.; Karaceper, M.D.; Klaassen, R.J.; Little, J.; et al. Health services use by children identified as heterozygous hemoglobinopathy mutation carriers via newborn screening. BMC Pediatr. 2021, 21, 296. [Google Scholar] [CrossRef] [PubMed]
- Sims, A.M.; Cromartie, S.J.; Gessner, L.; Campbell, A.; Coker, T.; Wang, C.J.; Tarini, B.A. Parents’ experiences and needs regarding infant sickle cell trait results. Pediatrics 2022, 149, e2021053454. [Google Scholar] [CrossRef] [PubMed]
- Alladin, B.A.; Mohamed-Rambaran, P.; Grey, V.; Hunter, A.; Chakraborty, P.; Henderson, M.; Milburn, J.; Tessier, L. Cross-sectional prospective feasibility study of newborn screening for sickle cell anaemia and congenital hypothyroidism in Guyana. BMJ Open 2022, 12, e046240. [Google Scholar] [CrossRef] [PubMed]
- Grob, F.; Odame, I.; Van Vliet, G. Worldwide newborn screening and early immunizations: Aligning advances in preventive pediatrics. J. Pediatr. 2024, 264, 113732. [Google Scholar] [CrossRef] [PubMed]
- Edelman, S.; Desai, H.; Pigg, T.; Yusuf, C.; Ojodu, J. Landscape of congenital adrenal hyperplasia newborn screening in the United States. Int. J. Neonatal Screen. 2020, 6, 64. [Google Scholar] [CrossRef] [PubMed]
- Speiser, P.W.; Chawla, R.; Chen, M.; Diaz-Thomas, A.; Finlayson, C.; Rutter, M.M.; Sandberg, D.E.; Shimy, K.; Talib, R.; Cerise, J.; et al. Newborn screening protocols and positive predictive value for congenital adrenal hyperplasia vary across the United States. Int. J. Neonatal Screen. 2020, 6, 37. [Google Scholar] [CrossRef] [PubMed]
- Held, P.K.; Bird, I.M.; Heather, N.L. Newborn screening for congenital adrenal hyperplasia: Review of factors affecting screening accuracy. Int. J. Neonatal Screen. 2020, 6, 67. [Google Scholar] [CrossRef] [PubMed]
- Grosse, S.D.; Van Vliet, G. Challenges in assessing the cost-effectiveness of newborn screening: The example of congenital adrenal hyperplasia. Int. J. Neonatal Screen. 2020, 6, 82. [Google Scholar] [CrossRef] [PubMed]
- Van Vliet, G.; Grosse, S.D. Newborn screening for congenital hypothyroidism and congenital adrenal hyperplasia: Benefits and costs of a successful public health program. Med. Sci. 2021, 37, 528–534. [Google Scholar] [CrossRef] [PubMed]
- Grosse, S.D.; Van Vliet, G. Newborn screening for congenital hypothyroidism and phenylketonuria-Beyond cost savings. J. Pediatr. 2023, 258, 113417. [Google Scholar] [CrossRef] [PubMed]
- Appelberg, K.; Sörensen, L.; Zetterström, R.H.; Henriksson, M.; Wedell, A.; Levin, L.Å. Cost-Effectiveness of newborn screening for phenylketonuria and congenital hypothyroidism. J. Pediatr. 2023, 256, 38–43.e3. [Google Scholar] [CrossRef] [PubMed]
- Rose, S.R.; Wassner, A.J.; Wintergerst, K.A.; Yayah-Jones, N.H.; Hopkin, R.J.; Chuang, J.; Smith, J.R.; Abell, K.; LaFranchi, S.H.; Section on Endocrinology Executive Committee; et al. Congenital hypothyroidism: Screening and management. Pediatrics 2023, 151, e2022060420. [Google Scholar] [CrossRef] [PubMed]
- Banigé, M.; Kariyawasam, D.; Gauthereau, V.; Luton, D.; Polak, M. Neonatal screening for hyperthyroidism proof of concept. J. Clin. Endocrinol. Metab. 2022, 107, e1374–e1381. [Google Scholar] [CrossRef] [PubMed]
- Manousaki, D.; Van Vliet, G. Population-based TSH screening of newborns for hyperthyroidism: It may be feasible, but is it justified? J. Clin. Endocrinol. Metab. 2022, 107, e2630–e2631. [Google Scholar] [CrossRef] [PubMed]
- Banigé, M.; Polak, M. Letter to the editor from Banigé and Polak: “Population-based TSH screening of newborns for hyperthyroidism: It may be feasible, but is it justified? J. Clin. Endocrinol. Metab. 2022, 107, e3536–e3537. [Google Scholar] [CrossRef] [PubMed]
- Peck, D.S.; Lacey, J.M.; White, A.L.; Pino, G.; Studinski, A.L.; Fisher, R.; Ahmad, A.; Spencer, L.; Viall, S.; Shallow, N.; et al. Incorporation of second-tier biomarker testing improves the specificity of newborn screening for mucopolysaccharidosis type I. Int. J. Neonatal Screen. 2020, 6, 10. [Google Scholar] [CrossRef] [PubMed]
- Hong, X.; Sadilek, M.; Gelb, M.H. A highly multiplexed biochemical assay for analytes in dried blood spots: Application to newborn screening and diagnosis of lysosomal storage disorders and other inborn errors of metabolism. Genet. Med. 2020, 22, 1262–1268. [Google Scholar] [CrossRef] [PubMed]
- Khaledi, H.; Gelb, M.H. Tandem mass spectrometry enzyme assays for multiplex detection of 10-mucopolysaccharidoses in dried blood spots and fibroblasts. Anal. Chem. 2020, 92, 11721–11727. [Google Scholar] [CrossRef]
- Herbst, Z.M.; Hong, X.; Sadilek, M.; Fuller, M.; Gelb, M.H. Newborn screening for the full set of mucopolysaccharidoses in dried blood spots based on first-tier enzymatic assay followed by second-tier analysis of glycosaminoglycans. Mol. Genet. Metab. 2023, 140, 107698. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Chopra, S.; Graham, C.; Langer, M.; Ng, R.; Ullal, A.J.; Pamula, V.K. Emerging approaches for fluorescence-based newborn screening of mucopolysaccharidoses. Diagnostics. 2020, 10, 294. [Google Scholar] [CrossRef] [PubMed]
- Parad, R.B.; Sheldon, Y.; Bhattacharjee, A. Implementation of hospital-based supplemental duchenne muscular dystrophy newborn screening (sDMDNBS): A pathway to broadening adoption. Int. J. Neonatal Screen. 2021, 7, 77. [Google Scholar] [CrossRef] [PubMed]
- Gruber, D.; Lloyd-Puryear, M.; Armstrong, N.; Scavina, M.; Tavakoli, N.P.; Brower, A.M.; Caggana, M.; Chung, W.K. Newborn screening for Duchenne muscular dystrophy-early detection and diagnostic algorithm for female carriers of Duchenne muscular dystrophy. Am. J. Med. Genet. C Semin. Med. Genet. 2022, 190, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Maloney, B.; Park, S.; Sowizral, M.; Brackett, I.; Moslehi, R.; Chung, W.K.; Gruber, D.; Brower, A.; Lloyd-Puryear, M.; Caggana, M.; et al. Factors influencing creatine kinase-MM concentrations in newborns and implications for newborn screening for Duchenne muscular dystrophy. Clin. Biochem. 2023, 118, 110614. [Google Scholar] [CrossRef]
- Dantonio, P.; Tavakoli, N.P.; Migliore, B.; McCown, E.; Lim, T.; Park, S.; Caggana, M.; Kucera, K.S.; Phan, H.; Street, N.; et al. Multi-laboratory evaluation of prototype dried blood spot quality control materials for creatine kinase-MM newborn screening assays. Int. J. Neonatal Screen. 2023, 9, 13. [Google Scholar] [CrossRef]
- Gelb, M.H.; Basheeruddin, K.; Burlina, A.; Chen, H.J.; Chien, Y.H.; Dizikes, G.; Dorley, C.; Giugliani, R.; Hietala, A.; Hong, X.; et al. Liquid chromatography-tandem mass spectrometry in newborn screening laboratories. Int. J. Neonatal Screen. 2022, 8, 62. [Google Scholar] [CrossRef]
- Hong, X.; Kumar, A.B.; Daiker, J.; Yi, F.; Sadilek, M.; De Mattia, F.; Fumagalli, F.; Calbi, V.; Damiano, R.; Della Bona, M.; et al. Leukocyte and dried blood spot arylsulfatase A assay by tandem mass spectrometry. Anal. Chem. 2020, 92, 6341–6348. [Google Scholar] [CrossRef] [PubMed]
- Hong, X.; Daiker, J.; Sadilek, M.; Ruiz-Schultz, N.; Kumar, A.B.; Norcross, S.; Dansithong, W.; Suhr, T.; Escolar, M.L.; Ronald Scott, C.; et al. Toward newborn screening of metachromatic leukodystrophy: Results from analysis of over 27,000 newborn dried blood spots. Genet. Med. 2021, 23, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Kelly, N.R.; Orsini, J.J.; Goldenberg, A.J.; Mulrooney, N.S.; Boychuk, N.A.; Clarke, M.J.; Paleologos, K.; Martin, M.M.; McNeight, H.; Caggana, M.; et al. ScreenPlus: A comprehensive, multi-disorder newborn screening program. Mol. Genet. Metab. Rep. 2024, 38, 101037. [Google Scholar] [CrossRef] [PubMed]
- Su, P.; Khaledi, H.; Waggoner, C.; Gelb, M.H. Detection of GM1-gangliosidosis in newborn dried blood spots by enzyme activity and biomarker assays using tandem mass spectrometry. J. Inherit. Metab. Dis. 2021, 44, 264–271. [Google Scholar] [CrossRef] [PubMed]
- Hong, X.; Daiker, J.; Sadilek, M.; DeBarber, A.E.; Chiang, J.; Duan, J.; Bootsma, A.H.; Huidekoper, H.H.; Vaz, F.M.; Gelb, M.H. Toward newborn screening of cerebrotendinous xanthomatosis: Results of a biomarker research study using 32,000 newborn dried blood spots. Genet. Med. 2020, 22, 1606–1612. [Google Scholar] [CrossRef] [PubMed]
- Peretz, R.H.; Ah Mew, N.; Vernon, H.J.; Ganetzky, R.D. Prospective diagnosis of MT-ATP6-related mitochondrial disease by newborn screening. Mol. Genet. Metab. 2021, 134, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Parad, R.B.; Kaler, S.G.; Mauceli, E.; Sokolsky, T.; Yi, L.; Bhattacharjee, A. Targeted next generation sequencing for newborn screening of Menkes disease. Mol. Genet. Metab. Rep. 2020, 24, 100625. [Google Scholar] [CrossRef] [PubMed]
- Marcotte, E.L.; Spector, L.G.; Mendes-de-Almeida, D.P.; Nelson, H.H. The prenatal origin of childhood leukemia: Potential applications for epidemiology and newborn screening. Front. Pediatr. 2021, 9, 639479. [Google Scholar] [CrossRef] [PubMed]
- Vicario-Feliciano, R.; Hernández-Hernández, C.I.; Camacho-Pastor, I.C.; Martínez-Cruzado, J.C. A custom-made newborn screening test for Wilson’s disease in Puerto Rico. Cureus 2022, 14, e24446. [Google Scholar] [CrossRef] [PubMed]
- Gavrilov, D.K.; Piazza, A.L.; Pino, G.; Turgeon, C.; Matern, D.; Oglesbee, D.; Raymond, K.; Tortorelli, S.; Rinaldo, P. The combined impact of CLIR post-analytical tools and second tier testing on the performance of newborn screening for disorders of propionate, methionine, and cobalamin metabolism. Int. J. Neonatal Screen. 2020, 6, 33. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.; Turgeon, C.; Rinaldo, P.; Pop, A.; Salomons, G.S.; Roullet, J.B.; Gibson, K.M. Longitudinal metabolomics in dried bloodspots yields profiles informing newborn screening for succinic semialdehyde dehydrogenase deficiency. JIMD Rep. 2020, 53, 29–38. [Google Scholar] [CrossRef]
- Rowe, A.D.; Stoway, S.D.; Åhlman, H.; Arora, V.; Caggana, M.; Fornari, A.; Hagar, A.; Hall, P.L.; Marquardt, G.C.; Miller, B.J.; et al. A novel approach to improve newborn screening for congenital hypothyroidism by integrating covariate-adjusted results of different tests into CLIR customized interpretive tools. Int. J. Neonatal Screen. 2021, 7, 23. [Google Scholar] [CrossRef] [PubMed]
- Wegwerth, P.J.; White, A.L.; Stoway, S.D.; Loken, P.R.; Oglesbee, D.; Matern, D.; Tortorelli, S.; Raymond, K.M.; Braverman, N.E.; Gavrilov, D.K. A new test method for biochemical analysis of plasmalogens in dried blood spots and erythrocytes from patients with peroxisomal disorders. J. Inherit. Metab. Dis. 2023, 46, 1159–1169. [Google Scholar] [CrossRef]
- Khan, Z.U.N.; Jafri, L.; Hall, P.L.; Schultz, M.J.; Ahmed, S.; Khan, A.H.; Majid, H. Utilizing augmented artificial intelligence for aminoacidopathies using collaborative laboratory integrated reporting—A cross-sectional study. Ann. Med. Surg. 2022, 82, 104651. [Google Scholar] [CrossRef] [PubMed]
- Peng, G.; Zhang, Y.; Zhao, H.; Scharfe, C. dbRUSP: An Interactive database to investigate inborn metabolic differences for improved genetic disease screening. Int. J. Neonatal Screen. 2022, 8, 48. [Google Scholar] [CrossRef] [PubMed]
- Petrick, L.M.; Uppal, K.; Funk, W.E. Metabolomics and adductomics of newborn bloodspots to retrospectively assess the early-life exposome. Curr. Opin. Pediatr. 2020, 32, 300–307. [Google Scholar] [CrossRef] [PubMed]
- Riches, N.O.; Johnson, E.P.; Frost, C.J.; Goldenberg, A.J.; Rothwell, E. The limited use of US residual newborn screening dried bloodspots for health disparity research. Genet. Med. 2020, 22, 1723–1726. [Google Scholar] [CrossRef] [PubMed]
- Peng, G.; Tang, Y.; Gandotra, N.; Enns, G.M.; Cowan, T.M.; Zhao, H.; Scharfe, C. Ethnic variability in newborn metabolic screening markers associated with false-positive outcomes. J. Inherit. Metab. Dis. 2020, 43, 934–943. [Google Scholar] [CrossRef] [PubMed]
- Peng, G.; Tang, Y.; Cowan, T.M.; Zhao, H.; Scharfe, C. Timing of newborn blood collection alters metabolic disease screening performance. Front. Pediatr. 2021, 8, 623184. [Google Scholar] [CrossRef] [PubMed]
- Wilson, K.; Ward, V.; Chakraborty, P.; Darmstadt, G.L. A novel way of determining gestational age upon the birth of a child. J. Glob. Health 2021, 11, 03078. [Google Scholar] [CrossRef] [PubMed]
- Bota, A.B.; Ward, V.; Hawken, S.; Wilson, L.A.; Lamoureux, M.; Ducharme, R.; Murphy, M.S.Q.; Denize, K.M.; Henderson, M.; Saha, S.K.; et al. Metabolic gestational age assessment in low resource settings: A validation protocol. Gates Open Res. 2021, 4, 150. [Google Scholar] [CrossRef] [PubMed]
- Bota, B.; Ward, V.; Lamoureux, M.; Santander, E.; Ducharme, R.; Hawken, S.; Potter, B.K.; Atito, R.; Nyamanda, B.; Munga, S.; et al. Unlocking the global health potential of dried blood spot cards. J. Glob. Health 2022, 12, 03027. [Google Scholar] [CrossRef] [PubMed]
- Hawken, S.; Ducharme, R.; Murphy, M.S.Q.; Olibris, B.; Bota, A.B.; Wilson, L.A.; Cheng, W.; Little, J.; Potter, B.K.; Denize, K.M.; et al. Development and external validation of machine learning algorithms for postnatal gestational age estimation using clinical data and metabolomic markers. PLoS ONE 2023, 18, e0281074. [Google Scholar] [CrossRef] [PubMed]
- Bailey, D.B., Jr.; Porter, K.A.; Andrews, S.M.; Raspa, M.; Gwaltney, A.Y.; Peay, H.L. Expert evaluation of strategies to modernize newborn screening in the United States. JAMA Netw. Open 2021, 4, e2140998. [Google Scholar] [CrossRef] [PubMed]
- Andrews, S.M.; Porter, K.A.; Bailey, D.B., Jr.; Peay, H.L. Preparing newborn screening for the future: A collaborative stakeholder engagement exploring challenges and opportunities to modernizing the newborn screening system. BMC Pediatr. 2022, 22, 90. [Google Scholar] [CrossRef] [PubMed]
- Wojcik, M.H.; Gold, N.B. Implications of genomic newborn screening for infant mortality. Int. J. Neonatal Screen. 2023, 9, 12. [Google Scholar] [CrossRef] [PubMed]
- Goldenberg, A.; Ponsaran, R.; Gaviglio, A.; Simancek, D.; Tarini, B.A. Genomics and newborn screening: Perspectives of public health programs. Int. J. Neonatal Screen. 2022, 8, 11. [Google Scholar] [CrossRef] [PubMed]
- Sobotka, S.A.; Ross, L.F. Newborn screening for neurodevelopmental disorders may exacerbate health disparities. Pediatrics 2023, 152, e2023061727. [Google Scholar] [CrossRef] [PubMed]
- Bailey, D.B., Jr. A window of opportunity for newborn screening. Mol. Diagn. Ther. 2022, 26, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Minear, M.A.; Phillips, M.N.; Kau, A.; Parisi, M.A. Newborn screening research sponsored by the NIH: From diagnostic paradigms to precision therapeutics. Am. J. Med. Genet. C Semin. Med. Genet. 2022, 190, 138–152. [Google Scholar] [CrossRef] [PubMed]
- Woerner, A.C.; Gallagher, R.C.; Vockley, J.; Adhikari, A.N. The use of whole genome and exome sequencing for newborn screening: Challenges and opportunities for population health. Front. Pediatr. 2021, 9, 663752. [Google Scholar] [CrossRef] [PubMed]
- Adhikari, A.N.; Gallagher, R.C.; Wang, Y.; Currier, R.J.; Amatuni, G.; Bassaganyas, L.; Chen, F.; Kundu, K.; Kvale, M.; Mooney, S.D.; et al. The role of exome sequencing in newborn screening for inborn errors of metabolism. Nat. Med. 2020, 26, 1392–1397. [Google Scholar] [CrossRef] [PubMed]
- Lekstrom-Himes, J.; Brooks, P.J.; Koeberl, D.D.; Brower, A.; Goldenberg, A.; Green, R.C.; Morris, J.A.; Orsini, J.J.; Yu, T.W.; Augustine, E.F. Moving away from one disease at a time: Screening, trial design, and regulatory implications of novel platform technologies. Am. J. Med. Genet. C Semin. Med. Genet. 2023, 193, 30–43. [Google Scholar] [CrossRef] [PubMed]
- Parisi, M.A.; Caggana, M.; Cohen, J.L.; Gold, N.B.; Morris, J.A.; Orsini, J.J.; Urv, T.K.; Wasserstein, M.P. When is the best time to screen and evaluate for treatable genetic disorders? A lifespan perspective. Am. J. Med. Genet. C Semin. Med. Genet. 2023, 193, 44–55. [Google Scholar] [CrossRef] [PubMed]
- Ersig, A.L.; Jaja, C.; Tluczek, A. Call to action for advancing equitable genomic newborn screening. Public Health Genom. 2023, 26, 188–193. [Google Scholar] [CrossRef] [PubMed]
- Gold, N.B.; Adelson, S.M.; Shah, N.; Williams, S.; Bick, S.L.; Zoltick, E.S.; Gold, J.I.; Strong, A.; Ganetzky, R.; Roberts, A.E.; et al. Perspectives of rare disease experts on newborn genome sequencing. JAMA Netw. Open 2023, 6, e2312231. [Google Scholar] [CrossRef] [PubMed]
- Stark, Z.; Scott, R.H. Genomic newborn screening for rare diseases. Nat. Rev. Genet. 2023, 24, 755–766. [Google Scholar] [CrossRef] [PubMed]
- Kingsmore, S.F.; Smith, L.D.; Kunard, C.M.; Bainbridge, M.; Batalov, S.; Benson, W.; Blincow, E.; Caylor, S.; Chambers, C.; Del Angel, G.; et al. A genome sequencing system for universal newborn screening, diagnosis, and precision medicine for severe genetic diseases. Am. J. Hum. Genet. 2022, 109, 1605–1619. [Google Scholar] [CrossRef] [PubMed]
- Grosse, S.D.; Cuthbert, C.; Gaffney, M.; Gaviglio, A.; Hinton, C.F.; Kellar-Guenther, Y.; Kemper, A.R.; McKasson, S.; Ojodu, J.; Riley, C.; et al. Progress in expanding newborn screening in the United States. Am. J. Hum. Genet. 2023, 110, 1015–1016. [Google Scholar] [CrossRef] [PubMed]
- Kingsmore, S.F.; Smith, L.D.; Kunard, C.M.; Bainbridge, M.; Batalov, S.; Benson, W.; Blincow, E.; Caylor, S.; Chambers, C.; Del Angel, G.; et al. Response to Grosse et al. Am. J. Hum. Genet. 2023, 110, 1017. [Google Scholar] [CrossRef] [PubMed]
- Hendrix, M.M.; Cuthbert, C.D.; Cordovado, S.K. Assessing the performance of dried-blood-spot DNA extraction methods in next generation sequencing. Int. J. Neonatal Screen. 2020, 6, 36. [Google Scholar] [CrossRef] [PubMed]
- Holm, I.A.; Agrawal, P.B.; Ceyhan-Birsoy, O.; Christensen, K.D.; Fayer, S.; Frankel, L.A.; Genetti, C.A.; Krier, J.B.; LaMay, R.C.; Levy, H.L.; et al. The BabySeq project: Implementing genomic sequencing in newborns. BMC Pediatr. 2018, 18, 225. [Google Scholar] [CrossRef] [PubMed]
- Green, R.C.; Shah, N.; Genetti, C.A.; Yu, T.; Zettler, B.; Uveges, M.K.; Ceyhan-Birsoy, O.; Lebo, M.S.; Pereira, S.; Agrawal, P.B.; et al. Actionability of unanticipated monogenic disease risks in newborn genomic screening: Findings from the BabySeq Project. Am. J. Hum. Genet. 2023, 110, 1034–1045. [Google Scholar] [CrossRef] [PubMed]
- Downie, L.; Halliday, J.; Lewis, S.; Amor, D.J. Principles of genomic newborn screening programs: A systematic review. JAMA Netw. Open 2021, 4, e2114336. [Google Scholar] [CrossRef] [PubMed]
- Currier, R.J. Newborn screening is on a collision course with public health ethics. Int. J. Neonatal Screen. 2022, 8, 51. [Google Scholar] [CrossRef] [PubMed]
- Bick, D.; Ahmed, A.; Deen, D.; Ferlini, A.; Garnier, N.; Kasperaviciute, D.; Leblond, M.; Pichini, A.; Rendon, A.; Satija, A.; et al. Newborn screening by genomic sequencing: Opportunities and challenges. Int. J. Neonatal Screen. 2022, 8, 40. [Google Scholar] [CrossRef] [PubMed]
- Brunelli, L.; Sohn, H.; Brower, A. Newborn sequencing is only part of the solution for better child health. Lancet. Reg. Health Am. 2023, 25, 100581. [Google Scholar] [CrossRef] [PubMed]
- WSFA 12 News. Alabama Begins Screening Newborns or 2 Additional Genetic Disorders. 21 March 2023. Available online: https://www.wsfa.com/2023/03/21/alabama-begins-screening-newborns-2-additional-genetic-disorders/ (accessed on 5 May 2023).
- Beans, J.A.; Trinidad, S.B.; Shane, A.L.; Wark, K.A.; Avey, J.P.; Apok, C.; Guinn, T.; Robler, S.K.; Hirschfeld, M.; Koeller, D.M.; et al. The CPT1A Arctic variant: Perspectives of community members and providers in two Alaska tribal health settings. J. Community Genet. 2023, 14, 613–620. [Google Scholar] [CrossRef] [PubMed]
- Booth, N.A.; Freeman, C.M.; Wright, B.L.; Rukasin, C.; Badia, P.; Daines, M.; Bauer, C.S.; Miller, H. Severe combined immunodeficiency (SCID) screening in Arizona: Lessons learned from the first 2 years. J. Clin. Immunol. 2022, 42, 1321–1329. [Google Scholar] [CrossRef] [PubMed]
- Arkansas Democrat Gazette. Arkansas Adopts Federal List for Newborn Screening; New Screenings Fee Won’t Change. 31 July 2023. Available online: https://www.arkansasonline.com/news/2023/jul/31/arkansas-adopts-federal-list-for-newborn-screening/ (accessed on 19 November 2023).
- Peng, G.; Tang, Y.; Cowan, T.M.; Enns, G.M.; Zhao, H.; Scharfe, C. Reducing false-positive results in newborn screening using machine learning. Int. J. Neonatal Screen. 2020, 6, 16. [Google Scholar] [CrossRef] [PubMed]
- Mak, J.; Peng, G.; Le, A.; Gandotra, N.; Enns, G.M.; Scharfe, C.; Cowan, T.M. Validation of a targeted metabolomics panel for improved second-tier newborn screening. J. Inherit. Metab. Dis. 2023, 46, 194–205. [Google Scholar] [CrossRef] [PubMed]
- Puck, J.M.; Gennery, A.R. Establishing Newborn Screening for SCID in the USA; experience in California. Int. J. Neonatal Screen. 2021, 7, 72. [Google Scholar] [CrossRef] [PubMed]
- Matteson, J.; Sciortino, S.; Feuchtbaum, L.; Bishop, T.; Olney, R.S.; Tang, H. Adrenoleukodystrophy newborn screening in California since 2016: Programmatic outcomes and follow-up. Int. J. Neonatal Screen. 2021, 7, 22. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.; Matteson, J.; Rinaldo, P.; Tortorelli, S.; Currier, R.; Sciortino, S. The clinical impact of CLIR tools toward rapid resolution of post-newborn screening confirmatory testing for X-linked adrenoleukodystrophy in California. Int. J. Neonatal Screen. 2020, 6, 62. [Google Scholar] [CrossRef]
- Tang, H.; Feuchtbaum, L.; Sciortino, S.; Matteson, J.; Mathur, D.; Bishop, T.; Olney, R.S. The first year experience of newborn screening for Pompe Disease in California. Int. J. Neonatal Screen. 2020, 6, 9. [Google Scholar] [CrossRef] [PubMed]
- Matteson, J.; Wu, C.H.; Mathur, D.; Tang, H.; Sciortino, S.; Feuchtbaum, L.; Bishop, T.; Sharma, S.C.; Neogi, P.; Fitzgibbon, I.; et al. California’s experience with SMA newborn screening: A successful path to early intervention. J. Neuromuscul. Dis. 2022, 9, 777–785. [Google Scholar] [CrossRef] [PubMed]
- Fillman, T.; Matteson, J.; Tang, H.; Mathur, D.; Zahedi, R.; Sen, I.; Bishop, T.; Neogi, P.; Feuchtbaum, L.; Olney, R.S.; et al. First three years’ experience of Mucopolysaccharidosis Type-1 newborn screening in California. J. Pediatr. 2023, 263, 113644. [Google Scholar] [CrossRef] [PubMed]
- Tise, C.G.; Morales, J.A.; Lee, A.S.; Velez-Bartolomei, F.; Floyd, B.J.; Levy, R.J.; Cusmano-Ozog, K.P.; Feigenbaum, A.S.; Ruzhnikov, M.R.Z.; Lee, C.U.; et al. Aicardi-Goutières syndrome may present with positive newborn screen for X-linked adrenoleukodystrophy. Am. J. Med. Genet. A 2021, 185, 1848–1853. [Google Scholar] [CrossRef] [PubMed]
- Niehaus, A.D.; Mendelsohn, B.A.; Zimmerman, B.; Lee, C.U.; Manning, M.A.; Cusmano-Ozog, K.P.; Tise, C.G. Neonatal lupus is a novel cause of positive newborn screening for X-linked adrenoleukodystrophy. Am. J. Med. Genet. A 2023, 191, 1412–1417. [Google Scholar] [CrossRef] [PubMed]
- Tise, C.G.; Verscaj, C.P.; Mendelsohn, B.A.; Woods, J.; Lee, C.U.; Enns, G.M.; Stander, Z.; Hall, P.L.; Cowan, T.M.; Cusmano-Ozog, K.P. MT-ATP6 mitochondrial disease identified by newborn screening reveals a distinct biochemical phenotype. Am. J. Med. Genet. A 2023, 191, 1492–1501. [Google Scholar] [CrossRef] [PubMed]
- Martiniano, S.L.; Croak, K.; Bonn, G.; Sontag, M.K.; Sagel, S.D. Improving outcomes for Colorado’s IRT-IRT-DNA cystic fibrosis newborn screening algorithm by implementing floating cutoffs. Mol. Genet. Metab. 2021, 134, 65–67. [Google Scholar] [CrossRef] [PubMed]
- Raboin, K.; Ellis, D.; Nichols, G.; Hughes, M.; Brimacombe, M.; Rubin, K. Advancing newborn screening long-term follow-up: Integration of Epic-based registries, dashboards, and efficient workflows. Int. J. Neonatal Screen. 2024, 10, 27. [Google Scholar] [CrossRef] [PubMed]
- Tullis, K. (Nemours/AI duPont Hospital for Children, Wilmington, DE, USA). Personal communication, 2024.
- Bosfield, K.; Regier, D.S.; Viall, S.; Hicks, R.; Shur, N.; Grant, C.L. Mucopolysaccharidosis type I newborn screening: Importance of second tier testing for ethnically diverse populations. Am. J. Med. Genet. A 2021, 185, 134–140. [Google Scholar] [CrossRef] [PubMed]
- Florida Newborn Screening. Florida Department of Health Newborn Screening Newsletter. April 2023. Available online: https://floridanewbornscreening.com/news-updates/q1-2023/ (accessed on 26 December 2023).
- Stalker, H.J.; Jonasson, A.R.; Hopfer, S.M.; Collins, M.S. Improvement in cystic fibrosis newborn screening program outcomes with genetic counseling via telemedicine. Pediatr. Pulmonol. 2023, 58, 3478–3486. [Google Scholar] [CrossRef] [PubMed]
- Sadat, R.; Hall, P.L.; Wittenauer, A.L.; Vengoechea, E.D.; Park, K.; Hagar, A.F.; Singh, R.; Moore, R.H.; Gambello, M.J. Increased parental anxiety and a benign clinical course: Infants identified with short-chain acyl-CoA dehydrogenase deficiency and isobutyryl-CoA dehydrogenase deficiency through newborn screening in Georgia. Mol. Genet. Metab. 2020, 129, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Hall, P.L.; Wittenauer, A.; Hagar, A. Post-analytical tools for the triage of newborn screening results in follow-up can reduce confirmatory testing and guide performance improvement. Int. J. Neonatal Screen. 2020, 6, 20. [Google Scholar] [CrossRef] [PubMed]
- Hall, P.L.; Wittenauer, A.L.; Wilcox, W.R. Proximal urea cycle defects are challenging to detect with newborn screening: Results of a prospective pilot study using post-analytical tools. Am. J. Med. Genet. C Semin. Med. Genet. 2022, 190, 178–186. [Google Scholar] [CrossRef] [PubMed]
- Hall, P.L.; Li, H.; Hagar, A.F.; Jerris, S.C.; Wittenauer, A.; Wilcox, W. Newborn screening for X-linked adrenoleukodystrophy in Georgia: Experiences from a pilot study screening of 51,081 newborns. Int. J. Neonatal Screen. 2020, 6, 81. [Google Scholar] [CrossRef]
- Hall, P.L.; Sanchez, R.; Hagar, A.F.; Jerris, S.C.; Wittenauer, A.; Wilcox, W.R. Two-tiered newborn screening with post-analytical tools for Pompe disease and mucopolysaccharidosis type I results in performance improvement and future direction. Int. J. Neonatal Screen. 2020, 6, 2. [Google Scholar] [CrossRef] [PubMed]
- Elkins, K.; Wittenauer, A.; Hagar, A.F.; Logan, R.; Sekul, E.; Xiang, Y.; Verma, S.; Wilcox, W.R. Georgia state spinal muscular atrophy newborn screening experience: Screening assay performance and early clinical outcomes. Am. J. Med. Genet. C Semin. Med. Genet. 2022, 190, 187–196. [Google Scholar] [CrossRef] [PubMed]
- State of Hawai’i Department of Health. Ka ‘Oihana Olakino. Newborn Metabolic Screening Program. Available online: https://health.hawaii.gov/genetics/category/announcements/ (accessed on 27 December 2023).
- Idaho Department of Health and Welfare. Idaho Newborns Will Be Screened for Additional Illnesses and Conditions. 26 January 2022. Available online: https://healthandwelfare.idaho.gov/news/idaho-newborns-be-screened-additional-illnesses-and-conditions (accessed on 17 November 2023).
- Idaho Division of Public Health. Idaho Title V Maternal & Child Health Services Block Grant Executive Summary; Idaho Division of Public Health: Boise, ID, USA, 2023; pp. 4–7.
- Idaho Department of Health and Welfare. DHW Communications. Gov. Little declares September Newborn Screening Awareness Month. 26 September 2023. Available online: https://healthandwelfare.idaho.gov/news/gov-little-declares-september-newborn-screening-awareness-month (accessed on 30 November 2023).
- Illinois State Senator Laura Fine. Senator Fine Expands Newborn Screening Test to Include MLD. 28 July 2023. Available online: http://www.senatorfine.com/news/press-releases/240-senator-fine-expands-newborn-screening-test-to-include-mld (accessed on 28 November 2023).
- Burton, B.K.; Charrow, J.; Hoganson, G.E.; Fleischer, J.; Grange, D.K.; Braddock, S.R.; Hitchins, L.; Hickey, R.; Christensen, K.M.; Groepper, D.; et al. Newborn screening for Pompe disease in Illinois: Experience with 684,290 infants. Int. J. Neonatal Screen. 2020, 6, 4. [Google Scholar] [CrossRef]
- Basheeruddin, K.; Shao, R.; Balster, F.; Gardley, P.; Ashbaugh, L. Newborn screening for Krabbe disease-Illinois experience: Role of psychosine in diagnosis of the disease. Int. J. Neonatal Screen. 2021, 7, 24. [Google Scholar] [CrossRef] [PubMed]
- Burton, B.K.; Hickey, R.; Hitchins, L.; Shively, V.; Ehrhardt, J.; Ashbaugh, L.; Peng, Y.; Basheeruddin, K. Newborn screening for X-linked adrenoleukodystrophy: The initial Illinois experience. Int. J. Neonatal Screen. 2022, 8, 6. [Google Scholar] [CrossRef]
- Burton, B.K.; Hickey, R.; Hitchins, L. Newborn screening for mucopolysaccharidosis type II in Illinois: An update. Int. J. Neonatal Screen. 2020, 6, 73. [Google Scholar] [CrossRef] [PubMed]
- Burton, B.K.; Shively, V.; Quadri, A.; Warn, L.; Burton, J.; Grange, D.K.; Christensen, K.; Groepper, D.; Ashbaugh, L.; Ehrhardt, J.; et al. Newborn screening for mucopolysaccharidosis type II: Lessons learned. Mol. Genet. Metab. 2023, 140, 107557. [Google Scholar] [CrossRef] [PubMed]
- McPheron, M.; Sapp, K. Newborn screening for Pompe disease: The Indiana experience. Genet. Med. 2022, 24, S17–S18. [Google Scholar] [CrossRef]
- Rady Children’s Institute for Genomic Medicine. Newborn Genomic Sequencing. To End the Diagnostic Odyssey. Available online: https://radygenomics.org/begin-ngs-newborn-sequencing/ (accessed on 30 November 2023).
- Kingsmore, S.F.; BeginNGS® Consortium. Dispatches from biotech beginning BeginNGS®: Rapid newborn genome sequencing to end the diagnostic and therapeutic odyssey. Am. J. Med. Genet. C Semin. Med. Genet. 2022, 190, 243–256. [Google Scholar] [CrossRef] [PubMed]
- Every Life Foundation for Rare Diseases. Iowa Passes Life-Saving Newborn Screening Legislation. 22 April 2022. Available online: https://everylifefoundation.org/iowa-passes-life-saving-newborn-screening-legislation-2/ (accessed on 27 November 2023).
- State Hygienic Laboratory at the University of Iowa. Technical Updates. Available online: https://www.shl.uiowa.edu/technicalupdates/sma_added_to_newborn_screening_panel.xml (accessed on 30 November 2023).
- State Hygienic Laboratory at the University of Iowa. News—Implementation of MPS-I and Pompe Newborn Screening. 9 May 2023. Available online: https://www.shl.uiowa.edu/news/mps1_and_pompe_testing_xml (accessed on 30 November 2023).
- Iowa.gov. To Whom It May Concern. 15 March 2023. Available online: https://hhs.iowa.gov/media/365/download?inline (accessed on 30 November 2023).
- Jilek, R.; Marcy, J.; Johnson, C.; Younger, G.; Calhoun, A.; Tung, M.L. Iowa newborn screening program experience with hemoglobinopathy screening over the last two decades and its increasing global relevance. Int. J. Neonatal Screen. 2024, 10, 21. [Google Scholar] [CrossRef] [PubMed]
- Therrell, B.L., Jr.; Lloyd-Puryear, M.A.; Eckman, J.R.; Mann, M.Y. Newborn screening for sickle cell diseases in the United States: A review of data spanning 2 decades. Semin. Perinatol. 2015, 39, 238–251. [Google Scholar] [CrossRef] [PubMed]
- Motter, S. Kansas Expands Screening of Newborns. 13WIBW. 6 January 2021. Available online: https://www.wibw.com/2021/01/05/kansas-expands-screening-of-newborns/ (accessed on 27 November 2023).
- Dubay, K.S.; Zach, T.L. Newborn screening. In StatPearls; Updated 1 May 2023; StatPearls Publishing: Treasure Island, FL, USA, 2024. [Google Scholar]
- Asamoah, A.; Wei, S.; Jackson, K.E.; Hersh, J.H.; Levy, H. Diagnosis of classic homocystinuria in two boys presenting with acute cerebral venous thrombosis and neurologic dysfunction after normal newborn screening. Int. J. Neonatal Screen. 2021, 7, 48. [Google Scholar] [CrossRef] [PubMed]
- Louisiana.gov. Louisiana Newborn Screening Panel Update. Available online: https://ldh.la.gov/assets/oph/Center-PHCH/Center-PH/cshs/genetics/NBS_Update_2021_AnnouncementOfNewTests.pdf (accessed on 27 November 2023).
- Maine.gov. Maine Newborn Bloodspot Screening Program. Available online: https://www.maine.gov/dhhs/mecdc/population-health/mch/cshn/bloodspot-screening/ (accessed on 30 November 2023).
- Leigh, V. New Maine Law Could Lead to Early Detection of CMV in Newborns. News Center Maine. 13 May 2022. Available online: https://www.newscentermaine.com/article/news/health/new-maine-law-could-lead-to-early-detection-of-cmv-in-newborns-health-community/97-c0930c45-7d0a-47a8-baa5-36bc9cebcef0 (accessed on 30 November 2023).
- Maryland.gov. Newborn Bloodspot Screening Collection for Provider Offices. Available online: https://health.maryland.gov/phpa/genetics/docs/NBS/specimencollectionforprovideroffices.pdf (accessed on 26 December 2023).
- Every Life Foundation for Rare Diseases. Maryland Passes Life-Saving Newborn Screening Legislation. 12 May 2023. Available online: https://everylifefoundation.org/maryland-passes-life-saving-newborn-screening-legislation/ (accessed on 27 November 2023).
- UMass Chan Medical School: New England Newborn Screening Program. Types of Screening: Routine and Optional. Available online: https://nensp.umassmed.edu/parents/massachusetts-parents/types-screening-routine-and-optional (accessed on 30 November 2023).
- Kumar, B.; Barton, S.; Kordowska, J.; Eaton, R.B.; Counihan, A.M.; Hale, J.E.; Comeau, A.M. Novel modification of a confirmatory SMA sequencing assay that can be used to determine SMN2 copy number. Int. J. Neonatal Screen. 2021, 7, 47. [Google Scholar] [CrossRef] [PubMed]
- Hale, J.E.; Darras, B.T.; Swoboda, K.J.; Estrella, E.; Chen, J.Y.H.; Abbott, M.A.; Hay, B.N.; Kumar, B.; Counihan, A.M.; Gerstel-Thompson, J.; et al. Massachusetts’ findings from statewide newborn screening for spinal muscular atrophy. Int. J. Neonatal Screen. 2021, 7, 26. [Google Scholar] [CrossRef]
- Hale, J.E.; Platt, C.D.; Bonilla, F.A.; Hay, B.N.; Sullivan, J.L.; Johnston, A.M.; Pasternack, M.S.; Hesterberg, P.E.; Meissner, H.C.; Cooper, E.R.; et al. Ten years of newborn screening for severe combined immunodeficiency (SCID) in Massachusetts. J. Allergy Clin. Immunol. Pract. 2021, 9, 2060–2067.e2. [Google Scholar] [CrossRef] [PubMed]
- Ma, K.C.; Hale, J.E.; Grad, Y.H.; Alter, G.; Luzuriaga, K.; Eaton, R.B.; Fischinger, S.; Kaur, D.; Brody, R.; Siddiqui, S.M.; et al. Trends in severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) seroprevalence in Massachusetts estimated from newborn screening specimens. Clin. Infect. Dis. 2022, 75, e105–e113. [Google Scholar] [CrossRef] [PubMed]
- Hawthorne, S.; Levy, H.L. Can newborn screening for vitamin B12 deficiency be incorporated into all newborn screening programs? J. Pediatr. 2020, 216, 9–11.e1. [Google Scholar] [CrossRef] [PubMed]
- Levy, H.L.; Sahai, I. Is more effective newborn screening for homocystinuria on the horizon? Clin. Chem. 2023, 69, 433–434. [Google Scholar] [CrossRef] [PubMed]
- White, E. Michigan to Destroy Millions of Leftover Blood Samples It Took from Newborns. Detroit Free Press. 23 June 2022. Available online: https://www.freep.com/story/news/local/michigan/detroit/2022/06/23/leftover-newborn-blood-spots-consent-fight/7712527001/ (accessed on 30 November 2023).
- Pappas, K.B. Newborn screening. Pediatr. Clin. N. Am. 2023, 70, 1013–1027. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.F.; Rose, A.M.; Waisbren, S.; Ahmad, A.; Prosser, L.A. Newborn screening and treatment of phenylketonuria: Projected health outcomes and cost-effectiveness. Children 2021, 8, 381. [Google Scholar] [CrossRef] [PubMed]
- Holsworth, A.; Hurden, I.; Fredrickson, M.; Kleyn, M.; Walkovich, K.; Secord, E. Newborn tandem mass spectroscopy screening for adenosine deaminase deficiency. Ann. Allergy Asthma Immunol. 2022, 129, 776–783.e2. [Google Scholar] [CrossRef]
- Pappas, K.B.; Younan, M.; Conway, R. Transcobalamin receptor deficiency in seven asymptomatic patients ascertained through newborn screening. Am. J. Med. Genet. A 2022, 188, 1102–1108. [Google Scholar] [CrossRef] [PubMed]
- Thompson, K.; Atkinson, S.; Kleyn, M. Use of online newborn screening educational resources for the education of expectant parents: An improvement in equity. Int. J. Neonatal Screen. 2022, 8, 34. [Google Scholar] [CrossRef] [PubMed]
- Omari, A.; Reeves, S.L.; Prosser, L.A.; Creary, M.S.; Ahmad, A.; Chua, K.P. Usability of NewSTEPs data for assessing the characteristics of infants with newborn screening disorders. Int. J. Neonatal Screen. 2022, 8, 42. [Google Scholar] [CrossRef] [PubMed]
- Minnesota Department of Health. 2023 News Releases. Available online: https://www.health.state.mn.us/news/pressrel/index.html (accessed on 1 December 2023).
- Dollard, S.C.; Dreon, M.; Hernandez-Alvarado, N.; Amin, M.M.; Wong, P.; Lanzieri, T.M.; Osterholm, E.A.; Sidebottom, A.; Rosendahl, S.; McCann, M.T.; et al. Sensitivity of dried blood spot testing for detection of congenital cytomegalovirus infection. JAMA Pediatr. 2021, 175, e205441. [Google Scholar] [CrossRef] [PubMed]
- Amin, M.M.; Wong, P.; McCann, M.; Dollard, S.C. Detection of cytomegalovirus in urine dried on filter paper. J. Pediatr. Infect. Dis. Soc. 2021, 10, 958–961. [Google Scholar] [CrossRef] [PubMed]
- Schleiss, M.R.; Panther, L.; Basnet, S.; Workneh, M.; Diaz-Decaro, J. Comparison of overall sensitivity and specificity across different newborn screening algorithms for congenital cytomegalovirus. Int. J. Neonatal Screen. 2023, 9, 33. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Alvarado, N.; Bierle, C.J.; Schleiss, M.R. Droplet digital PCR (ddPCR) does not enhance the sensitivity of detection of cytomegalovirus (CMV) DNA in newborn dried blood spots evaluated in the context of newborn congenital CMV (cCMV) screening. Int. J. Neonatal Screen. 2024, 10, 1. [Google Scholar] [CrossRef] [PubMed]
- Every Life Foundation for Rare Diseases. Mississippi Passes Life-Saving Newborn Screening Legislation. 16 March 2022. Available online: https://everylifefoundation.org/mississippi-becomes-first-state-in-2022-to-sign-newborn-screening-legislation/ (accessed on 27 November 2023).
- Klug, T.L.; Swartz, L.B.; Washburn, J.; Brannen, C.; Kiesling, J.L. Lessons learned from Pompe disease newborn screening and follow-up. Int. J. Neonatal Screen. 2020, 6, 11. [Google Scholar] [CrossRef] [PubMed]
- Bilyeu, H.; Washburn, J.; Vermette, L.; Klug, T. Validation and implementation of a highly sensitive and efficient newborn screening assay for mucopolysaccharidosis type II. Int. J. Neonatal Screen. 2020, 6, 79. [Google Scholar] [CrossRef] [PubMed]
- Mantravadi, V.; Bednarski, J.J.; Ritter, M.A.; Gu, H.; Kolicheski, A.L.; Horner, C.; Cooper, M.A.; Kitcharoensakkul, M. Immunological findings and clinical outcomes of infants with positive newborn screening for severe combined immunodeficiency from a tertiary care center in the USA. Front. Immunol. 2021, 12, 734096. [Google Scholar] [CrossRef] [PubMed]
- Montana.gov. Newborn Screening Advisory Committee. Available online: https://dphhs.mt.gov/boardscouncils/NBS/index (accessed on 26 December 2023).
- Baker, C.V.; Cady Keller, A.; Lutz, R.; Eveans, K.; Baumert, K.; DiPerna, J.C.; Rizzo, W.B. Newborn screening for X-linked adrenoleukodystrophy in Nebraska: Initial experiences and challenges. Int. J. Neonatal Screen. 2022, 8, 29. [Google Scholar] [CrossRef] [PubMed]
- Cure SMA. Nevada Starts Screening for SMA—Only One State Remains! 21 December 2023. Available online: https://www.curesma.org/nevada-starts-screening-for-sma-dec2023/ (accessed on 26 December 2023).
- New Hampshire DHHS. Newborn Screening Program. 2020 Annual Report on Newborn Screening. Available online: https://www.dhhs.nh.gov/sites/g/files/ehbemt476/files/documents/2021-11/nbs-annual-report-2020.pdf (accessed on 3 December 2023).
- New Hampshire DHHS. Newborn Screening Program. NBS Advisory Committee Minutes. 24 October 2023. Available online: https://www.dhhs.nh.gov/sites/g/files/ehbemt476/files/documents2/nbsac-minutes-10-23.pdf (accessed on 3 December 2023).
- Difilipo, D. Judge Orders State to Release Information about Police Use of Baby Blood Spots. New Jersey Monitor. 4 January 2023. Available online: https://newjerseymonitor.com/2023/01/04/judge-orders-state-to-release-information-about-police-use-of-baby-blood-spots/ (accessed on 30 November 2023).
- Owens, K.; Chapman, C.; Caplan, A. Protect newborn screening programs. Science 2023, 379, 1307–1308. [Google Scholar] [CrossRef] [PubMed]
- Difilipo, D. Civil Rights Group Sues New Jersey to Stop Secret Storage, Use of Baby Blood Spots. New Jersey Monitor. 2 November 2023. Available online: https://newjerseymonitor.com/2023/11/02/civil-rights-group-sues-new-jersey-to-stop-secret-storage-use-of-baby-blood-spots/ (accessed on 30 November 2023).
- Baldwin, K.; Barker, E.M.; Carayannopoulos, M.; Farrell, P.M.; Zanni, R.; Scanlin, T.F. Severe lung disease in children with cystic fibrosis missed in newborn screening. Pediatr. Pulmonol. 2024, 59, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Bill Track 50. NJ S3975. Establishes Requirements to Commence Screening Newborn Infants for Congenital Cytomegalovirus Infection; Establishes Public Awareness Campaign. Available online: https://www.billtrack50.com/billdetail/1381637 (accessed on 4 December 2023).
- New Mexico Department of Health. Newborn Genetic Screening. Available online: https://www.nmhealth.org/about/phd/fhb/cms/nbgs/ (accessed on 26 December 2023).
- Oregon.gov. Northwest Regional Newborn Bloodspot Screening Program Advisory Board. 2022 Advisory Board Report to the Legislature. Available online: https://sharedsystems.dhsoha.state.or.us/DHSForms/Served/le4233.pdf (accessed on 26 December 2023).
- Wadsworth Center, New York State Department of Health. Newborn Screening for Cytomegalovirus (CMV). Available online: https://www.wadsworth.org/sites/default/files/WebDoc/017286_20547_NewbornScreeningCMV_web_final.pdf (accessed on 4 December 2023).
- PPMD30. Duchenne Added to New York State’s Newborn Screening Panel. 25 October 2023. Available online: https://www.parentprojectmd.org/duchenne-added-to-new-york-states-newborn-screening-panel/ (accessed on 30 November 2023).
- Hartnett, M.J.; Lloyd-Puryear, M.A.; Tavakoli, N.P.; Wynn, J.; Koval-Burt, C.L.; Gruber, D.; Trotter, T.; Caggana, M.; Chung, W.K.; Armstrong, N.; et al. Newborn screening for Duchenne muscular dystrophy: First year results of a population-based pilot. Int. J. Neonatal Screen. 2022, 8, 50. [Google Scholar] [CrossRef] [PubMed]
- Tavakoli, N.P.; Gruber, D.; Armstrong, N.; Chung, W.K.; Maloney, B.; Park, S.; Wynn, J.; Koval-Burt, C.; Verdade, L.; Tegay, D.H.; et al. Duchenne Muscular dystrophy pilot study group. Newborn screening for Duchenne muscular dystrophy: A two-year pilot study. Ann. Clin. Transl. Neurol. 2023, 10, 1383–1396. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Maloney, B.; Caggana, M.; Tavakoli, N.P. Creatine kinase-MM concentration in dried blood spots from newborns and implications for newborn screening for Duchenne muscular dystrophy. Muscle Nerve 2022, 65, 652–658. [Google Scholar] [CrossRef] [PubMed]
- Wynn, J.; Tavakoli, N.P.; Armstrong, N.; Gomez, J.; Koval, C.; Lai, C.; Tang, S.; Quevedo Prince, A.; Quevedo, Y.; Rufino, K.; et al. Improving recruitment for a newborn screening pilot study with adaptations in response to the COVID-19 pandemic. Int. J. Neonatal Screen. 2022, 8, 23. [Google Scholar] [CrossRef] [PubMed]
- Martin, M.M.; Wilson, R.; Caggana, M.; Orsini, J.J. The impact of post-analytical tools on New York screening for Krabbe disease and Pompe disease. Int. J. Neonatal Screen. 2020, 6, 65. [Google Scholar] [CrossRef] [PubMed]
- Kay, D.M.; Stevens, C.F.; Parker, A.; Saavedra-Matiz, C.A.; Sack, V.; Chung, W.K.; Chiriboga, C.A.; Engelstad, K.; Laureta, E.; Farooq, O.; et al. Implementation of population-based newborn screening reveals low incidence of spinal muscular atrophy. Genet. Med. 2020, 22, 1296–1302. [Google Scholar] [CrossRef] [PubMed]
- Eng, L.; Regelmann, M.O. Adrenoleukodystrophy in the era of newborn screening. Curr. Opin. Endocrinol. Diabetes Obes. 2020, 27, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Gans, M.D.; Gavrilova, T. Retrospective analysis of a New York newborn screen severe combined immunodeficiency referral center. J. Clin. Immunol. 2020, 40, 456–465. [Google Scholar] [CrossRef] [PubMed]
- McMahon, R.; DeMartino, L.; Sowizral, M.; Powers, D.; Tracy, M.; Caggana, M.; Tavakoli, N.P. The impact of seasonal changes on thyroxine and thyroid-stimulating hormone in newborns. Int. J. Neonatal Screen. 2021, 7, 8. [Google Scholar] [CrossRef] [PubMed]
- Sicko, R.J.; Stevens, C.F.; Hughes, E.E.; Leisner, M.; Ling, H.; Saavedra-Matiz, C.A.; Caggana, M.; Kay, D.M. Validation of a custom next-generation sequencing assay for cystic fibrosis newborn screening. Int. J. Neonatal Screen. 2021, 7, 73. [Google Scholar] [CrossRef] [PubMed]
- Wojcik, M.; Morrissey, M.; Borden, K.; Teta, B.; Sicko, R.; Showers, A.; Sunny, S.; Caggana, M. Method modification to reduce false positives for newborn screening of guanidinoacetate methyltransferase deficiency. Mol. Genet. Metab. 2022, 135, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.H.; Deng, S.; Chiriboga, C.A.; Kay, D.M.; Irumudomon, O.; Laureta, E.; Delfiner, L.; Treidler, S.O.; Anziska, Y.; Sakonju, A.; et al. Newborn screening for spinal muscular atrophy in New York State: Clinical outcomes from the first 3 years. Neurology 2022, 99, e1527–e1537. [Google Scholar] [CrossRef] [PubMed]
- DeCelie-Germana, J.K.; Bonitz, L.; Langfelder-Schwind, E.; Kier, C.; Diener, B.L.; Berdella, M. Diagnostic and communication challenges in cystic fibrosis newborn screening. Life 2023, 13, 1646. [Google Scholar] [CrossRef] [PubMed]
- Columbia-Columbia University Irving Medical Center. Can Genomic Screening of Newborns Help More Children Born with Rare Diseases? 27 February 2023. Available online: https://www.cuimc.columbia.edu/news/can-genomic-screening-newborns-help-more-children-born-rare-diseases (accessed on 2 January 2024).
- Every Life Foundation for Rare Diseases. North Carolina Becomes Fourth State in 2021 to Sign Newborn Screening Legislation. 19 November 2023. Available online: https://everylifefoundation.org/north-carolina-becomes-fourth-state-in-2021-to-sign-newborn-screening-legislation/ (accessed on 27 November 2023).
- NCDHHS. NC Newborns Now Screened for Two Additional Disorders to Help Provide Early Diagnosis and Treatment. 13 February 2023. Available online: https://www.ncdhhs.gov/news/press-releases/2023/02/13/nc-newborns-now-screened-two-additional-disorders-help-provide-early-diagnosis-and-treatment (accessed on 27 November 2023).
- Kucera, K.S.; Taylor, J.L.; Robles, V.R.; Clinard, K.; Migliore, B.; Boyea, B.L.; Okoniewski, K.C.; Duparc, M.; Rehder, C.W.; Shone, S.M.; et al. A voluntary statewide newborn screening pilot for spinal muscular atrophy: Results from Early Check. Int. J. Neonatal Screen. 2021, 7, 20. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Clinard, K.; Young, S.P.; Rehder, C.W.; Fan, Z.; Calikoglu, A.S.; Bali, D.S.; Bailey, D.B., Jr.; Gehtland, L.M.; Millington, D.S.; et al. Evaluation of X-linked adrenoleukodystrophy newborn screening in North Carolina. JAMA Netw. Open 2020, 3, e1920356. [Google Scholar] [CrossRef] [PubMed]
- Bailey, D.B., Jr.; Gehtland, L.M.; Lewis, M.A.; Peay, H.; Raspa, M.; Shone, S.M.; Taylor, J.L.; Wheeler, A.C.; Cotton, M.; King, N.M.P.; et al. Early Check: Translational science at the intersection of public health and newborn screening. BMC Pediatr. 2019, 19, 238. [Google Scholar] [CrossRef] [PubMed]
- Gehtland, L.M.; Paquin, R.S.; Andrews, S.M.; Lee, A.M.; Gwaltney, A.; Duparc, M.; Pfaff, E.R.; Bailey, D.B., Jr. Using a patient portal to increase enrollment in a newborn screening research study: Observational study. JMIR Pediatr. Parent. 2022, 5, e30941. [Google Scholar] [CrossRef] [PubMed]
- Paquin, R.S.; Lewis, M.A.; Harper, B.A.; Moultrie, R.R.; Gwaltney, A.; Gehtland, L.M.; Peay, H.L.; Duparc, M.; Raspa, M.; Wheeler, A.C.; et al. Outreach to new mothers through direct mail and email: Recruitment in the Early Check research study. Clin. Transl. Sci. 2021, 14, 880–889. [Google Scholar] [CrossRef] [PubMed]
- Peay, H.L.; Gwaltney, A.Y.; Moultrie, R.; Cope, H.; Boyea, B.L.; Porter, K.A.; Duparc, M.; Alexander, A.A.; Biesecker, B.B.; Isiaq, A.; et al. Education and consent for population-based DNA Screening: A mixed-methods evaluation of the Early Check newborn screening pilot study. Front. Genet. 2022, 13, 891592. [Google Scholar] [CrossRef] [PubMed]
- UNC School of Medicine Newsroom. Early Check Study to Use Genome Sequencing to Dramatically Expand Voluntary Screening Offered for NC Newborns. 20 September 2023. Available online: https://news.unchealthcare.org/2023/09/early-check-study-to-use-genome-sequencing-to-dramatically-expand-voluntary-screening-offered-for-nc-newborns/ (accessed on 27 November 2023).
- Lee, S.; Taylor, J.L.; Redmond, C.; Hadd, A.G.; Kemppainen, J.A.; Haynes, B.C.; Shone, S.; Bailey, D.B., Jr.; Latham, G.J. Validation of fragile X screening in the newborn population using a fit-for-purpose FMR1 PCR assay system. J. Mol. Diagn. 2020, 22, 346–354. [Google Scholar] [CrossRef] [PubMed]
- Migliore, B.A.; Zhou, L.; Duparc, M.; Robles, V.R.; Rehder, C.W.; Peay, H.L.; Kucera, K.S. Evaluation of the GSP creatine kinase-MM assay and assessment of CK-MM stability in newborn, patient, and contrived dried blood spots for newborn screening for Duchenne muscular dystrophy. Int. J. Neonatal Screen. 2022, 8, 12. [Google Scholar] [CrossRef] [PubMed]
- Kucera, K.S.; Boyea, B.L.; Migliore, B.; Potter, S.N.; Robles, V.R.; Kutsa, O.; Cope, H.; Okoniewski, K.C.; Wheeler, A.; Rehder, C.W.; et al. Two years of newborn screening for Duchenne muscular dystrophy as a part of the statewide Early Check research program in North Carolina. Genet. Med. 2024, 26, 101009. [Google Scholar] [CrossRef] [PubMed]
- Njau, G.; Odoi, A. Investigation of predictors of newborn screening refusal in a large birth cohort in North Dakota, USA. Matern. Child Health J. 2019, 23, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Every Life Foundation for Rare Diseases. Every Life Applauds Newborn Screening Laws in Ohio and Arizona to Save Babies. 1 July 2021. Available online: https://everylifefoundation.org/everylife-applauds-newborn-screening-laws-in-ohio-and-arizona-to-save-babies/ (accessed on 27 November 2023).
- Ohio Hospital Association. Ohio Increases Fees for Newborn Screening Kits, Surprise Billing Arbitration. 30 June 2023. Available online: https://ohiohospitals.org/News-Publications/Subscriptions/Ohio-Hospitals-Newswire/Articles/Ohio-Increases-Fees-for-Newborn-Screening-Kits,-Su (accessed on 30 November 2023).
- Parent Project Muscular Dystrophy. Duchenne Newborn Screening Milestone—First State with Universal Newborn Screening for Duchenne! 5 July 2023. Available online: https://www.parentprojectmd.org/duchenne-newborn-screening-milestone-first-state-with-universal-newborn-screening-for-duchenne/ (accessed on 30 November 2023).
- Peterson, L.; Siemon, A.; Olewiler, L.; McBride, K.L.; Allain, D.C. A qualitative assessment of parental experiences with false-positive newborn screening for Krabbe disease. J. Genet. Couns. 2022, 31, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Rose, S.R.; Blunden, C.E.; Jarrett, O.O.; Kaplan, K.; Caravantes, R.; Akinbi, H.T. Utility of repeat testing for congenital hypothyroidism in infants with very low birth weight. J. Pediatr. 2022, 242, 152–158.e1. [Google Scholar] [CrossRef] [PubMed]
- Meyer, A.P.; Connolly, A.M.; Vannatta, K.; Hacker, N.; Hatfield, A.; Decipeda, A.; Parker, P.; Willoughby, A.; Waldrop, M.A. Parental experiences with newborn screening and gene replacement therapy for spinal muscular atrophy. J. Neuromuscul. Dis. 2024, 11, 129–142. [Google Scholar] [CrossRef] [PubMed]
- Oklahoma State Department of Health. OSDH Temporarily Sends Newborn Screening to Top National Lab. 16 March 2021. Available online: https://oklahoma.gov/health/news---events/newsroom/2021/march/osdh-temporarily-sends-newborn-screening-to-top-national-lab.html (accessed on 3 December 2023).
- Branham, D. Outsourced Newborn Screenings Return to Oklahoma Public Health Lab Amid Move to Stillwater. The Oklahoman. 1 June 2021. Available online: https://www.oklahoman.com/story/news/2021/06/01/stillwater-oklahoma-public-health-lab-newborn-screenings-return/5291738001/ (accessed on 30 November 2023).
- Blevins, J. Newborn Screening Tests Have Improved, but Families Have Suffered from False Positives. The Oklahoman. 17 April 2023. Available online: https://www.oklahoman.com/story/opinion/2023/04/17/positive-action-good-news-for-newborn-testing-but-it-took-two-years/70047467007/ (accessed on 30 November 2023).
- Oklahoma Senate. Bill Updating Newborn Screening Guidelines Signed into Law. 9 May 2022. Available online: https://oksenate.gov/press-releases/bill-updating-newborn-screening-guidelines-signed-law (accessed on 30 November 2023).
- Oregon.gov. Oregon Health Authority, Newborn Screening, Oregon State Public Health Laboratory. Newborn Screening Program Updates. Available online: https://www.oregon.gov/oha/PH/LABORATORYSERVICES/NEWBORNSCREENING/Pages/newborn-screening-news.aspx (accessed on 5 December 2023).
- Viall, S.; Dennis, A.; Yang, A. Newborn screening for Fabry disease in Oregon: Approaching the iceberg of A143T and variants of uncertain significance. Am. J. Med. Genet. C Semin. Med. Genet. 2022, 190, 206–214. [Google Scholar] [CrossRef] [PubMed]
- Eshragh, N.; Doan, L.V.; Connelly, K.J.; Denniston, S.; Willis, S.; LaFranchi, S.H. Outcome of newborn screening for congenital adrenal hyperplasia at two time points. Horm. Res. Paediatr. 2020, 93, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Pennsylvania Bulletin. Newborn Screening Acts and Regulations. Available online: https://www.health.pa.gov/topics/programs/Newborn-Screening/Pages/Acts-Regulations.aspx (accessed on 7 December 2023).
- Ficicioglu, C.; Ahrens-Nicklas, R.C.; Barch, J.; Cuddapah, S.R.; DiBoscio, B.S.; DiPerna, J.C.; Gordon, P.L.; Henderson, N.; Menello, C.; Luongo, N.; et al. Newborn screening for Pompe disease: Pennsylvania experience. Int. J. Neonatal Screen. 2020, 6, 89. [Google Scholar] [CrossRef] [PubMed]
- Priestley, J.R.C.; Adang, L.A.; Drewes Williams, S.; Lichter-Konecki, U.; Menello, C.; Engelhardt, N.M.; DiPerna, J.C.; DiBoscio, B.; Ahrens-Nicklas, R.C.; Edmondson, A.C.; et al. Newborn screening for X-linked adrenoleukodystrophy: Review of data and outcomes in Pennsylvania. Int. J. Neonatal Screen. 2022, 8, 24. [Google Scholar] [CrossRef] [PubMed]
- State of Rhode Island Department of Health. Newborn Blood Screening. Available online: https://health.ri.gov/newbornscreening/blood/ (accessed on 7 December 2023).
- SCDHEC.gov. Newborn Screening. Available online: https://scdhec.gov/health-professionals/health-services-facilities/lab-certification-services/newborn-screening (accessed on 7 December 2023).
- Lowe, T.; Boyd, J.; Shu, L.; DeLuca, J.M. Providers’ perspectives related to parents’ choice of pediatric provider of record and newborn screening: A qualitative study. J. Prim. Care Community Health 2023, 14, 21501319231190274. [Google Scholar] [CrossRef] [PubMed]
- South Dakota Department of Health. Newborn Screening Advisory Committee. Available online: https://doh.sd.gov/topics/maternal-child-health/pregnancy-early-childhood/newborn/newborn-screening/newborn-screening-advisory-committee/ (accessed on 7 December 2023).
- Boyd, J.; Stein, Q.; Davis-Keppen, L. The state of newborn screening in South Dakota. S. D. Med. 2022, 75, 509–512. [Google Scholar] [PubMed]
- Lechner, C.; Rumpler, M.; Dorley, M.C.; Li, Y.; Ingram, A.; Fryman, H. Developing an online dashboard to visualize performance data-Tennessee newborn screening experience. Int. J. Neonatal Screen. 2022, 8, 49. [Google Scholar] [CrossRef]
- Every Life Foundation for Rare Diseases. Texas Passes Life-Saving Newborn Screening Legislation. 13 June 2023. Available online: https://everylifefoundation.org/texas-passes-life-saving-newborn-screening-legislation/ (accessed on 27 November 2023).
- Langlois, P.H.; Canfield, M.A.; Rutenberg, G.W.; Mandell, D.J.; Hua, F.; Reilly, B.; Ruktanonchai, D.J.; Jackson, J.F.; Hunt, P.; Freedenberg, D.; et al. The association between newborn screening analytes as measured on a second screen and childhood autism in a Texas Medicaid population. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2020, 183, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Ward, S.; Mackay, L.P.; Sutton, V.R.; Divin, K. Impact of COVID-19 on newborn screening in South Texas. Mol. Genet. Metab. 2021, 132, S16. [Google Scholar] [CrossRef]
- Del Valle Penella, A.; Miller, J.; Rochat, R.; Demmler-Harrison, G. Utility of dried blood spots for the diagnosis of congenital cytomegaloviruses within the first 21 days of life in a single center. Int. J. Neonatal Screen. 2023, 9, 44. [Google Scholar] [CrossRef] [PubMed]
- Utah Newborn Screening. What Is Newborn Screening? Available online: https://newbornscreening.health.utah.gov/ (accessed on 10 December 2023).
- Coupal, E.; Hart, K.; Wong, B.; Rothwell, E. Newborn screening knowledge and attitudes among midwives and out-of-hospital-birth parents. J. Perinat. Neonatal Nurs. 2020, 34, 357–364. [Google Scholar] [CrossRef] [PubMed]
- Butterfield, R.J. Spinal muscular atrophy treatments, newborn screening, and the creation of a neurogenetics urgency. Semin. Pediatr. Neurol. 2021, 38, 100899. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.; Shao, J.; Wallis, H.; Johansen, C.; Hart, K.; Pasquali, M.; Gouripeddi, R.; Rohrwasser, A. Towards a newborn screening common data model: The Utah newborn screening data model. Int. J. Neonatal Screen. 2021, 7, 70. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Schultz, N.; Sant, D.; Norcross, S.; Dansithong, W.; Hart, K.; Asay, B.; Little, J.; Chung, K.; Oakeson, K.F.; Young, E.L.; et al. Methods and feasibility study for exome sequencing as a universal second-tier test in newborn screening. Genet. Med. 2021, 23, 767–776. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Schultz, N.; Asay, B.; Rohrwasser, A. Scalable newborn screening solutions: Bioinformatics and next-generation sequencing. Int. J. Neonatal Screen. 2021, 7, 63. [Google Scholar] [CrossRef]
- Conway, M.; Vuong, T.T.; Hart, K.; Rohrwasser, A.; Eilbeck, K. Pain points in parents’ interactions with newborn screening systems: A qualitative study. BMC Pediatr. 2022, 22, 167. [Google Scholar] [CrossRef] [PubMed]
- Prinzi, J.; Pasquali, M.; Hobert, J.A.; Palmquist, R.; Wong, K.N.; Francis, S.; De Biase, I. Diagnosing X-linked adrenoleukodystrophy after implementation of newborn screening: A reference laboratory perspective. Int. J. Neonatal Screen. 2023, 9, 64. [Google Scholar] [CrossRef] [PubMed]
- Montgomery, A. Newborn Testing Could See Two More Health Conditions Added to the List. WCAX 3. 29 June 2023. Available online: https://www.wcax.com/2023/06/29/newborn-testing-could-see-two-more-health-conditions-added-list/ (accessed on 10 December 2023).
- Russell, M. Endocrine components of newborn screening. Curr. Probl. Pediatr. Adolesc. Health Care 2020, 50, 100772. [Google Scholar] [CrossRef]
- WAVY.com. 2 Disorders Added to Newborn Screening Program. 16 March 2022. Available online: https://www.wavy.com/news/virginia/2-disorders-added-to-va-newborn-screening-program/ (accessed on 26 December 2023).
- Washington State Department of Health. Newborn Screening. Available online: https://doh.wa.gov/you-and-your-family/infants-and-children/newborn-screening (accessed on 7 December 2023).
- Vasquez-Loarte, T.; Thompson, J.D.; Merritt, J.L., 2nd. Considering proximal urea cycle disorders in expanded newborn screening. Int. J. Neonatal Screen. 2020, 6, 77. [Google Scholar] [CrossRef] [PubMed]
- Scott, C.R.; Elliott, S.; Hong, X.; Huang, J.Y.; Kumar, A.B.; Yi, F.; Pendem, N.; Chennamaneni, N.K.; Gelb, M.H. Newborn screening for mucopolysaccharidoses: Results of a pilot study with 100,000 dried blood spots. J. Pediatr. 2020, 216, 204–207. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Duong, P.; Dayuha, R.; Collins, C.J.; Beckman, E.; Thies, J.; Chang, I.; Lam, C.; Sun, A.; Scott, A.I.; et al. A rapid and non-invasive proteomic analysis using DBS and buccal swab for multiplexed second-tier screening of Pompe disease and Mucopolysaccharidosis type I. Mol. Genet. Metab. 2022, 136, 296–305. [Google Scholar] [CrossRef] [PubMed]
- WV-DHHR. OMCFH/Newborn Screening Home Page. Available online: https://www.wvdhhr.org/nbms/ (accessed on 10 December 2023).
- Wiener, R.C. Newborn genetic testing in the United States and access to needed specialist care, National Survey of Children’s Health, 2020: A cross-sectional study. PLoS ONE 2022, 17, e0279352. [Google Scholar] [CrossRef]
- Furnier, S.M.; Durkin, M.S.; Baker, M.W. Translating molecular technologies into routine newborn screening practice. Int. J. Neonatal Screen. 2020, 6, 80, Erratum in Int. J. Neonatal Screen. 2021, 7. [Google Scholar] [CrossRef]
- Kaluarachchi, D.C.; Nicksic, V.M.; Allen, D.B.; Eickhoff, J.C.; Dawe, S.J.; Baker, M.W. Thyroid-stimulating hormone reference ranges for moderate-to-late preterm infants. J. Perinatol. 2021, 41, 2664–2667. [Google Scholar] [CrossRef] [PubMed]
- Kaluarachchi, D.C.; Nicksic, V.B.; Allen, D.B.; Eickhoff, J.C.; Baker, M.W.; Kling, P.J. Thyroid hormone function in small for gestational age term newborns. J. Pediatr. 2021, 238, 181–186.e3. [Google Scholar] [CrossRef] [PubMed]
- Nicksic, V.B.; Allen, D.B.; Stanley, M.A.; Baker, M.W.; Eickhoff, J.C.; Kaluarachchi, D.C. Lack of association between gestational age adjusted TSH percentiles and neurodevelopmental outcomes among preterm infants. J. Neonatal Perinatal Med. 2021, 15, 243–247. [Google Scholar] [CrossRef] [PubMed]
- Lasarev, M.R.; Bialk, E.R.; Allen, D.B.; Held, P.K. Application of principal component analysis to newborn screening for congenital adrenal hyperplasia. J. Clin. Endocrinol. Metab. 2020, 105, dgaa371. [Google Scholar] [CrossRef] [PubMed]
- Matharu, P.K.; Held, P.K.; Allen, D.B. Multiple 17-OHP cutoff co-variates fail to improve 21-hydroxylase deficiency screening accuracy. Int. J. Neonatal Screen. 2022, 8, 57. [Google Scholar] [CrossRef] [PubMed]
- Farrell, P.M.; Rock, M.J.; Baker, M.W. The impact of the CFTR gene discovery on cystic fibrosis diagnosis, counseling, and preventive therapy. Genes 2020, 11, 401. [Google Scholar] [CrossRef] [PubMed]
- Rock, M.J.; Baker, M.; Antos, N.; Farrell, P.M. Refinement of newborn screening for cystic fibrosis with next generation sequencing. Pediatr. Pulmonol. 2023, 58, 778–787, Erratum in Pediatr. Pulmonol. 2023, 58, 2691. [Google Scholar] [CrossRef] [PubMed]
- Rock, M.J.; Baker, M.; Farrell, P.M. Cystic fibrosis screen-positive neonates with one pathogenic variant still warrant sweat testing. Pediatr. Pulmonol. 2023, 58, 3342–3345. [Google Scholar] [CrossRef] [PubMed]
- Barreda, C.B.; Farrell, P.M.; Laxova, A.; Eickhoff, J.C.; Braun, A.T.; Coller, R.J.; Rock, M.J. Newborn screening alone insufficient to improve pulmonary outcomes for cystic fibrosis. J. Cyst. Fibros. 2021, 20, 492–498. [Google Scholar] [CrossRef] [PubMed]
- Thorsen, J.; Kolbert, K.; Joshi, A.; Baker, M.; Seroogy, C.M. Newborn Screening for Severe Combined Immunodeficiency: 10-Year Experience at a Single Referral Center (2009–2018). J. Clin. Immunol. 2021, 41, 595–602. [Google Scholar] [CrossRef] [PubMed]
- Cogley, M.F.; Wiberley-Bradford, A.E.; Mochal, S.T.; Dawe, S.J.; Piro, Z.D.; Baker, M.W. Newborn screening for severe combined immunodeficiency using the multiple of the median values of T-cell receptor excision circles. Int. J. Neonatal Screen. 2021, 7, 43. [Google Scholar] [CrossRef] [PubMed]
- Atkins, A.E.; Cogley, M.F.; Baker, M.W. Newborn screening for severe combined immunodeficiency: Do preterm infants require special consideration? Int. J. Neonatal Screen. 2021, 7, 40. [Google Scholar] [CrossRef] [PubMed]
- Baker, M.W.; Mochal, S.T.; Dawe, S.J.; Wiberley-Bradford, A.E.; Cogley, M.F.; Zeitler, B.R.; Piro, Z.D.; Harmelink, M.M.; Kwon, J.M. Newborn screening for spinal muscular atrophy: The Wisconsin first year experience. Neuromuscul. Disord. 2022, 32, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Teber, T.A.; Conti, B.J.; Haynes, C.A.; Hietala, A.; Baker, M.W. Newborn screen for X-linked adrenoleukodystrophy using flow injection tandem mass spectrometry in negative ion mode. Int. J. Neonatal Screen. 2022, 8, 27. [Google Scholar] [CrossRef] [PubMed]
- Wyoming Department of Health. Maternal and Child Health Unit. Newborn Screening Program. Available online: https://health.wyo.gov/publichealth/mch/newbornscreening/ (accessed on 26 December 2023).
- De Souza, A.; Wolan, V.; Battochio, A.; Christian, S.; Hume, S.; Johner, G.; Lilley, M.; Ridsdale, R.; Schnabl, K.; Tran, C.; et al. Newborn screening: Current status in Alberta, Canada. Int. J. Neonatal Screen. 2019, 5, 37. [Google Scholar] [CrossRef] [PubMed]
- Niri, F.; Nicholls, J.; Baptista Wyatt, K.; Walker, C.; Price, T.; Kelln, R.; Hume, S.; Parboosingh, J.; Lilley, M.; Kolski, H.; et al. Alberta spinal muscular atrophy newborn screening-results from year 1 pilot project. Int. J. Neonatal Screen. 2023, 9, 42. [Google Scholar] [CrossRef]
- Hoang, S.C.; Blumenschein, P.; Lilley, M.; Olshaski, L.; Bruce, A.; Wright, N.A.M.; Ridsdale, R.; Christian, S. Secondary reporting of G6PD deficiency on newborn screening. Int. J. Neonatal Screen. 2023, 9, 18. [Google Scholar] [CrossRef] [PubMed]
- BC Gov News. B.C. Newborn Screening Expands; Early Detection Improves Quality of Life. 12 October 2022. Available online: https://news.gov.bc.ca/releases/2022HLTH0197-001536 (accessed on 26 December 2023).
- Fox, D.; Ronsley, R.; Khowaja, A.R.; Haim, A.; Vallance, H.; Sinclair, G.; Amed, S. Clinical impact and cost efficacy of newborn screening for congenital adrenal hyperplasia. J. Pediatr. 2020, 220, 101–108.e2, Erratum in J. Pediatr. 2020, 221, 267. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, G.; McMahon, V.; Schellenberg, A.; Nelson, T.N.; Chilvers, M.; Vallance, H. Performance of a three-tier (IRT-DNA-IRT) cystic fibrosis screening algorithm in British Columbia. Int. J. Neonatal Screen. 2020, 6, 46. [Google Scholar] [CrossRef] [PubMed]
- Dubland, J.A.; Rakić, B.; Vallance, H.; Sinclair, G. Analysis of 2-methylcitric acid, methylmalonic acid, and total homocysteine in dried blood spots by LC-MS/MS for application in the newborn screening laboratory: A dual derivatization approach. J. Mass Spectrom. Adv. Clin. Lab. 2021, 20, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Manitoba. Province Announces Expansion of Newborn Screening Program. 8 June 2022. Available online: https://news.gov.mb.ca/news/index.html?item=55077&posted=2022-06-08 (accessed on 26 December 2023).
- Mhanni, A.; Aylward, N.; Boy, N.; Martin, B.; Sharma, A.; Rockman-Greenberg, C. Outcome of the glutaric aciduria type 1 (GA1) newborn screening program in Manitoba: 1980–2020. Mol. Genet. Metab. Rep. 2020, 25, 100666. [Google Scholar] [CrossRef] [PubMed]
- Giffin, C.; Ciapala, A.; Rubin, T.; Greenberg, C.; Cuvelier, G.; Van Caeseele, P. Development of a multiplexed PCR assay to identify pathogenic variants causing severe combined immunodeficiency (SCID) in Manitoba. Univ. Manit. J. Med. 2021, 4, 1–9. Available online: https://umjm.ca/assets/documents/V4I1/V4I1A3.pdf (accessed on 3 February 2024).
- Miele, S. (Provincial Medical Genetics Program, St Johns, NL, Canada). Personal communication, 2022.
- Pinto, V. Funding Bolsters Effort to Bring Newborn Screening to Eastern Canada. SMA News Today. 3 December 2021. Available online: https://smanewstoday.com/news/muscular-dystrophy-canada-novartis-maritime-newborn-sma-screening-program/ (accessed on 26 December 2023).
- McKee-Muir, O.; Dyack, S.; Taillon, M.; Brock, J.A.; Sheriko, J. Epidemiology of spinal muscular atrophy caused by SMN1 deletions in Maritime Canada. Am. J. Med. Genet. A 2023, 191, 2711–2715. [Google Scholar] [CrossRef] [PubMed]
- Mador-House, R.; Liu, Z.; Dyack, S. Detection of early onset carnitine palmitoyltransferase II deficiency by newborn screening: Should CPT II deficiency be a primary disease target? Int. J. Neonatal Screen. 2021, 7, 55. [Google Scholar] [CrossRef] [PubMed]
- Schulze, A.; Chakraborty, P. The Ontario newborn screening program: A novel referral center model. Bundesgesundheitsblatt Gesundheitsforschung Gesundheitsschutz 2023, 66, 1205–1213. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, P.; Sood, M.M.; Benchimol, E.I. Serum newborn screening blood metabolites are not associated with childhood-onset inflammatory bowel disease: A population-based matched case-control study. Inflamm. Bowel Dis. 2020, 26, 1743–1747. [Google Scholar] [CrossRef] [PubMed]
- Hayeems, R.Z.; Miller, F.A.; Barg, C.J.; Bombard, Y.; Chakraborty, P.; Potter, B.K.; Patton, S.; Bytautas, J.P.; Tam, K.; Taylor, L.; et al. Primary care providers’ role in newborn screening result notification for cystic fibrosis. Can. Fam. Physician. 2021, 67, 439–448. [Google Scholar] [CrossRef] [PubMed]
- Carroll, J.C.; Hayeems, R.Z.; Miller, F.A.; Barg, C.J.; Bombard, Y.; Chakraborty, P.; Potter, B.K.; Bytautas, J.P.; Tam, K.; Taylor, L.; et al. Newborn screening for cystic fibrosis: Role of primary care providers in caring for infants with positive screening results. Can. Fam. Physician. 2021, 67, e144–e152. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, P.; Potter, B.K.; Hayeems, R.Z. Maximizing benefits and minimizing harms: Diagnostic uncertainty arising from newborn screening. Pediatrics 2021, 148, e2021052822. [Google Scholar] [CrossRef] [PubMed]
- Potter, B.K.; Little, J.; Chakraborty, P.; Lacaze-Masmonteil, T.; Wilson, K. Association between newborn screening analyte profiles and infant mortality. J. Matern. Fetal Neonatal Med. 2021, 34, 835–838. [Google Scholar] [CrossRef] [PubMed]
- McMillan, H.J.; Kernohan, K.D.; Yeh, E.; Amburgey, K.; Boyd, J.; Campbell, C.; Dowling, J.J.; Gonorazky, H.; Marcadier, J.; Tarnopolsky, M.A.; et al. Newborn screening for spinal muscular atrophy: Ontario testing and follow-up recommendations. Can. J. Neurol. Sci. 2021, 48, 504–511. [Google Scholar] [CrossRef] [PubMed]
- Blagojevic, C.; Heung, T.; Theriault, M.; Tomita-Mitchell, A.; Chakraborty, P.; Kernohan, K.; Bulman, D.E.; Bassett, A.S. Estimate of the contemporary live-birth prevalence of recurrent22q11.2 deletions: A cross-sectional analysis from population-based newborn screening. CMAJ Open 2021, 9, E802–E809. [Google Scholar] [CrossRef] [PubMed]
- Kernohan, K.D.; McMillan, H.J.; Yeh, E.; Lacaria, M.; Kowalski, M.; Campbell, C.; Dowling, J.J.; Gonorazky, H.; Marcadier, J.; Tarnopolsky, M.A.; et al. Ontario newborn screening for spinal muscular atrophy: The first year. Can. J. Neurol. Sci. 2022, 49, 821–823. [Google Scholar] [CrossRef] [PubMed]
- DiBattista, A.; Ogrel, S.; MacKenzie, A.E.; Chakraborty, P. Quantitation of phosphatidylethanols in dried blood spots to determine rates of prenatal alcohol exposure in Ontario. Alcohol Clin. Exp. Res. 2022, 46, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Marr, A.; Yokubynas, N.; Tang, K.; Saleh, D.; Wherrett, D.K.; Stein, R.; Bassilious, E.; Chakraborty, P.; Lawrence, S.E. Transient vs permanent congenital hypothyroidism in Ontario, Canada: Predictive factors and scoring system. J. Clin. Endocrinol. Metab. 2022, 107, 638–648. [Google Scholar] [CrossRef] [PubMed]
- Henderson, M.P.A.; Chakraborty, P. An approach for evaluating potential screening thresholds using biomarker population distribution and analytical imprecision. J. Appl. Lab. Med. 2023, 8, 92–97. [Google Scholar] [CrossRef] [PubMed]
- Henderson, M.P.A.; McIntosh, N.; Chambers, A.; Desormeaux, E.; Kowalski, M.; Milburn, J.; Chakraborty, P. Biotinidase activity is affected by both seasonal temperature and filter collection cards. Clin. Biochem. 2023, 115, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Liang, N.S.Y.; Watts-Dickens, A.; Chitayat, D.; Babul-Hirji, R.; Chakraborty, P.; Hayeems, R.Z. Parental preferences for expanded newborn screening: What are the limits? Children 2023, 10, 1362. [Google Scholar] [CrossRef] [PubMed]
- Auray-Blais, C.; Boutin, M.; Lavoie, P.; Maranda, B. Neonatal urine screening program in the Province of Quebec: Technological upgrade from thin layer chromatography to tandem mass spectrometry. Int. J. Neonatal Screen. 2021, 7, 18. [Google Scholar] [CrossRef] [PubMed]
- Fortin, C.A.; Girard, L.; Bonenfant, C.; Leblanc, J.; Cruz-Marino, T.; Blackburn, M.E.; Desmeules, M.; Bouchard, L. Benefits of newborn screening for vitamin D-dependant Rickets Type 1A in a founder population. Front. Endocrinol. 2022, 13, 887371. [Google Scholar] [CrossRef] [PubMed]
- Yeganeh, M.; Auray-Blais, C.; Maranda, B.; Sabovic, A.; DeVita, R.J.; Lazarus, M.B.; Houten, S.M. A case of hyperlysinemia identified by urine newborn screening. JIMD Rep. 2023, 64, 440–445. [Google Scholar] [CrossRef] [PubMed]
- Kazadi, C.; Robitaille, N.; Forte, S.; Ducruet, T.; Pastore, Y.D. Positive impacts of the universal newborn screening program on the outcome of children with sickle cell disease in the Province of Quebec: A Retrospective Cohort Study. Blood 2023, 142 (Suppl. S1), 370. [Google Scholar] [CrossRef]
- Saskatchewan. Saskatchewan Expanding Newborn Screening Program. 23 February 2022. Available online: https://www.saskatchewan.ca/government/news-and-media/2022/february/23/saskatchewan-expanding-newborn-screening-program (accessed on 26 December 2023).
- Padilla, C.D.; Therrell, B.L., Jr. Working Group of the Asia Pacific Society for Human Genetics on Consolidating Newborn Screening Efforts in the Asia Pacific Region. Consolidating newborn screening efforts in the Asia Pacific region: Networking and shared education. J. Community Genet. 2012, 3, 35–45. [Google Scholar] [CrossRef]
- Shibata, N.; Hasegawa, Y.; Yamada, K.; Kobayashi, H.; Purevsuren, J.; Yang, Y.; Dung, V.C.; Khanh, N.N.; Verma, I.C.; Bijarnia-Mahay, S.; et al. Diversity in the incidence and spectrum of organic acidemias, fatty acid oxidation disorders, and amino acid disorders in Asian countries: Selective screening vs. expanded newborn screening. Mol. Genet. Metab. Rep. 2018, 16, 5–10. [Google Scholar] [CrossRef] [PubMed]
- Wiley, V.; Webster, D.; Loeber, G. Screening pathways through China, the Asia Pacific Region, the World. Int. J. Neonatal Screen. 2019, 5, 26. [Google Scholar] [CrossRef] [PubMed]
- Chien, Y.H.; Hwu, W.L. The modern face of newborn screening. Pediatr. Neonatol. 2023, 64 (Suppl. S1), S22–S29. [Google Scholar] [CrossRef] [PubMed]
- Australian Government. Department of Health and Aging Care. Newborn Bloodspot Screening. Available online: https://www.health.gov.au/our-work/newborn-bloodspot-screening (accessed on 8 May 2023).
- O’Leary, P.; Maxwell, S. Newborn bloodspot screening policy framework for Australia. Australas. Med. J. 2015, 8, 292–298. [Google Scholar] [CrossRef] [PubMed]
- Metternick-Jones, S.C.; Lister, K.J.; Dawkins, H.J.; White, C.A.; Weeramanthri, T.S. Review of current international decision-making processes for newborn screening: Lessons for Australia. Front. Public Health 2015, 3, 214. [Google Scholar] [CrossRef] [PubMed]
- Shih, S.T.F.; Keller, E.; Wiley, V.; Wong, M.; Farrar, M.A.; Chambers, G.M. Economic evaluation of newborn screening for severe combined immunodeficiency. Int. J. Neonatal Screen. 2022, 8, 44. [Google Scholar] [CrossRef] [PubMed]
- Shih, S.T.F.; Keller, E.; Wiley, V.; Farrar, M.A.; Wong, M.; Chambers, G.M. Modelling the cost-effectiveness and budget impact of a newborn screening program for spinal muscular atrophy and severe combined immunodeficiency. Int. J. Neonatal Screen. 2022, 8, 45. [Google Scholar] [CrossRef] [PubMed]
- Kariyawasam, D.S.T.; Russell, J.S.; Wiley, V.; Alexander, I.E.; Farrar, M.A. The implementation of newborn screening for spinal muscular atrophy: The Australian experience. Genet. Med. 2020, 22, 557–565. [Google Scholar] [CrossRef] [PubMed]
- D’Silva, A.M.; Kariyawasam, D.S.T.; Best, S.; Wiley, V.; Farrar, M.A.; NSW SMA NBS Study Group. Integrating newborn screening for spinal muscular atrophy into health care systems: An Australian pilot programme. Dev. Med. Child Neurol. 2022, 64, 625–632. [Google Scholar] [CrossRef] [PubMed]
- Kariyawasam, D.S.T.; D’Silva, A.M.; Vetsch, J.; Wakefield, C.E.; Wiley, V.; Farrar, M.A. “We needed this”: Perspectives of parents and healthcare professionals involved in a pilot newborn screening program for spinal muscular atrophy. eClinicalMedicine 2021, 33, 100742. [Google Scholar] [CrossRef] [PubMed]
- Shih, S.T.; Farrar, M.A.; Wiley, V.; Chambers, G. Newborn screening for spinal muscular atrophy with disease-modifying therapies: A cost-effectiveness analysis. J. Neurol. Neurosurg. Psychiatry 2021, 92, 1296–1304. [Google Scholar] [CrossRef] [PubMed]
- Kariyawasam, D.S.; D’Silva, A.M.; Sampaio, H.; Briggs, N.; Herbert, K.; Wiley, V.; Farrar, M.A. Newborn screening for spinal muscular atrophy in Australia: A non-randomised cohort study. Lancet Child Adolesc. Health 2023, 7, 159–170. [Google Scholar] [CrossRef] [PubMed]
- Shum, B.O.V.; Henner, I.; Cairns, A.; Pretorius, C.; Wilgen, U.; Barahona, P.; Ungerer, J.P.J.; Bennett, G. Technical feasibility of newborn screening for spinal muscular atrophy by next-generation DNA sequencing. Front. Genet. 2023, 14, 1095600. [Google Scholar] [CrossRef] [PubMed]
- Yu, A.; Alder, N.; Lain, S.J.; Wiley, V.; Nassar, N.; Jack, M. Outcomes of lowered newborn screening thresholds for congenital hypothyroidism. J. Paediatr. Child Health 2023, 59, 955–961. [Google Scholar] [CrossRef]
- Huynh, T.; Greaves, R.; Mawad, N.; Greed, L.; Wotton, T.; Wiley, V.; Ranieri, E.; Rankin, W.; Ungerer, J.; Price, R.; et al. Fifty years of newborn screening for congenital hypothyroidism: Current status in Australasia and the case for harmonisation. Clin. Chem. Lab. Med. 2022, 60, 1551–1561. [Google Scholar] [CrossRef] [PubMed]
- Lai, F.; Srinivasan, S.; Wiley, V. Evaluation of a two-tier screening pathway for congenital adrenal hyperplasia in the New South Wales newborn screening programme. Int. J. Neonatal Screen. 2020, 6, 63. [Google Scholar] [CrossRef] [PubMed]
- Greaves, R.F.; Kumar, M.; Mawad, N.; Francescon, A.; Le, C.; O’Connell, M.; Chi, J.; Pitt, J. Best practice for identification of classical 21-hydroxylase deficiency should include 21 deoxycortisol analysis with appropriate isomeric steroid separation. Int. J. Neonatal Screen. 2023, 9, 58. [Google Scholar] [CrossRef] [PubMed]
- Ruseckaite, R.; Salimi, F.; Earnest, A.; Bell, S.C.; Douglas, T.; Frayman, K.; Keatley, L.; King, S.; Kotsimbos, T.; Middleton, P.G.; et al. Survival of people with cystic fibrosis in Australia. Sci. Rep. 2022, 12, 19748. [Google Scholar] [CrossRef] [PubMed]
- Estrella, J.F.G.L.; Immanuel, J.; Wiley, V.; Simmons, D. Newborn screening samples for diabetes research: An underused resource. Cells 2020, 9, 2299. [Google Scholar] [CrossRef] [PubMed]
- Estrella, J.; Wiley, V.; Simmons, D.; Hng, T.M.; McLean, M. Effect of maternal metformin treatment in pregnancy on neonatal metabolism: Evidence from newborn metabolic screening. Diabetes Care 2021, 44, 2536–2541. [Google Scholar] [CrossRef] [PubMed]
- Theda, C.; Gibbons, K.; Defor, T.E.; Donohue, P.K.; Golden, W.C.; Kline, A.D.; Gulamali-Majid, F.; Panny, S.R.; Hubbard, W.C.; Jones, R.O.; et al. Newborn screening for X-linked adrenoleukodystrophy: Further evidence high throughput screening is feasible. Mol. Genet. Metab. 2014, 111, 55–57. [Google Scholar] [CrossRef] [PubMed]
- Farrar, M.A.; Kariyawasam, D.; Grattan, S.; Bayley, K.; Davis, M.; Holland, S.; Waddel, L.B.; Jones, K.; Lorentzos, M.; Ravine, A.; et al. Newborn screening for the diagnosis and treatment of Duchenne muscular dystrophy. J. Neuromuscul. Dis. 2023, 10, 15–28. [Google Scholar] [CrossRef] [PubMed]
- Ministers Department of Health and Aged Care. More Rare Conditions Considered for Newborn Bloodspot Screening and New Treatment Option for Spinal Muscular Atrophy. Available online: https://www.health.gov.au/ministers/the-hon-mark-butler-mp/media/more-rare-conditions-considered-for-newborn-bloodspot-screening-and-new-treatment-option-for-spinal-muscular-atrophy (accessed on 3 February 2024).
- Saich, R.; Brown, R.; Collicoat, M.; Jenner, C.; Primmer, J.; Clancy, B.; Holland, T.; Krinks, S. Is newborn screening the ultimate strategy to reduce diagnostic delays in Pompe disease? The parent and patient perspective. Int. J. Neonatal Screen. 2020, 6, 1. [Google Scholar] [CrossRef] [PubMed]
- Godler, D.E.; Ling, L.; Gamage, D.; Baker, E.K.; Bui, M.; Field, M.J.; Rogers, C.; Butler, M.G.; Murgia, A.; Leonardi, E.; et al. Feasibility of screening for chromosome 15 imprinting disorders in 16 579 newborns by using a novel genomic workflow. JAMA Netw. Open 2022, 5, e2141911. [Google Scholar] [CrossRef] [PubMed]
- White, S.; Mossfield, T.; Fleming, J.; Barlow-Stewart, K.; Ghedia, S.; Dickson, R.; Richards, F.; Bombard, Y.; Wiley, V. Expanding the Australian newborn blood spot screening program using genomic sequencing: Do we want it and are we ready? Eur. J. Hum. Genet. 2023, 31, 703–711. [Google Scholar] [CrossRef] [PubMed]
- Lynch, F.; Best, S.; Gaff, C.; Downie, L.; Archibald, A.D.; Gyngell, C.; Goranitis, I.; Peters, R.; Savulescu, J.; Lunke, S.; et al. Australian public perspectives on genomic newborn screening: Risks, benefits, and preferences for implementation. Int. J. Neonatal Screen. 2024, 10, 6. [Google Scholar] [CrossRef] [PubMed]
- Downie, L.; Bouffler, S.E.; Amor, D.J.; Christodoulou, J.; Yeung, A.; Horton, A.E.; Macciocca, I.; Archibald, A.D.; Wall, M.; Caruana, J.; et al. Gene selection for genomic newborn screening: Moving towards consensus? Genet. Med. 2024, 26, 101077. [Google Scholar] [CrossRef] [PubMed]
- Ul Azim, M.A.; Islam, M.A.; Biswasarma, S.C.; Uddin, M.J.; Jabin, Z.; Begum, F.; Begum, S.M.; Alam, F.; Hasan, M.; Nahar, N.; et al. Current status of newborn screening for the early diagnosis of congenital hypothyroidism in Bangladesh. Bangladesh J. Nucl. Med. 2022, 25, 15–21. [Google Scholar] [CrossRef]
- Hasan, M. (Bangladesh Atomic Energy Commission, Dhaka, Bangladesh). Personal communication, 2023.
- Sumaia, S.N.; Reshad, R.A.I.; Tabassum, F.; Mim, S.K.; Mahmud, M.G.R.; Faruque, C.M.O.; Miah, M.F. Present status of newborn screening in Bangladesh through the participation of health professionals. Clin. Res. Clin. Trials 2023, 7. Available online: https://auctoresonline.org/article/present-status-of-newborn-screening-in-bangladesh-through-the-participation-of-health-professionals (accessed on 5 February 2023).
- Al-Bari, A.A. Current scenario and future direction of newborn screening and management program for phenylketonuria in Bangladesh. J. Inborn Errors Metab. Screen. 2022, 10, e20210024. [Google Scholar] [CrossRef]
- Sarker, S.K.; Islam, M.T.; Biswas, A.; Bhuyan, G.S.; Sultana, R.; Sultana, N.; Rakhshanda, S.; Begum, M.N.; Rahat, A.; Yeasmin, S.; et al. Age-specific cut-off values of amino acids and acylcarnitines for diagnosis of inborn errors of metabolism using liquid chromatography tandem mass spectrometry. BioMed Res. Int. 2019, 2019, 3460902. [Google Scholar] [CrossRef] [PubMed]
- Murphy, M.S.Q.; Chakraborty, P.; Pervin, J.; Rahman, A.; Wilson, L.A.; Lamoureux, M.; Denize, K.; Henderson, M.; Hawken, S.; Potter, B.K.; et al. Incidental screen positive findings in a prospective cohort study in Matlab, Bangladesh: Insights into expanded newborn screening for low-resource settings. Orphanet J. Rare Dis. 2019, 14, 25. [Google Scholar] [CrossRef] [PubMed]
- Anwar, S.; Taslem Mourosi, J.; Hasan, M.K.; Hosen, M.J.; Miah, M.F. Umbilical cord blood screening for the detection of common deletional mutations of α-thalassemia in Bangladesh. Hemoglobin 2020, 44, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Laura, F.K.; Miah, M.F.; Chowdhury, S.F.; Anwar, S.; Reshad, R.A.I.; Mahmud, M.G.R.; Faruque, C.M.O. Inheritance of β hemoglobin gene mutation: Potential method of newborn screening of sickle cell anemia in Bangladesh. J. Clin. Exp. Investig. 2022, 13, em00795. [Google Scholar] [CrossRef]
- Hosen, M.J.; Anwar, S.; Taslem Mourosi, J.; Chakraborty, S.; Miah, M.F.; Vanakker, O.M. Genetic counseling in the context of Bangladesh: Current scenario, challenges, and a framework for genetic service implementation. Orphanet J. Rare Dis. 2021, 16, 168. [Google Scholar] [CrossRef] [PubMed]
- Coyle, K.; Quan, A.M.L.; Wilson, L.A.; Hawken, S.; Bota, A.B.; Coyle, D.; Murray, J.C.; Wilson, K. Cost-effectiveness of a gestational age metabolic algorithm for preterm and small-for-gestational-age classification. Am. J. Obstet. Gynecol. MFM 2021, 3, 100279. [Google Scholar] [CrossRef] [PubMed]
- Sazawal, S.; Das, S.; Ryckman, K.K.; Khanam, R.; Nisar, I.; Deb, S.; Jasper, E.A.; Rahman, S.; Mehmood, U.; Dutta, A.; et al. Machine learning prediction of gestational age from metabolic screening markers resistant to ambient temperature transportation: Facilitating use of this technology in low resource settings of South Asia and East Africa. J. Glob. Health 2022, 12, 04021. [Google Scholar] [CrossRef] [PubMed]
- Bin, S.; Phou, K.; Im, S. Primary congenital hypothyroidism: Challenges in a low-income country without paediatric endocrinologist and universal newborn screening. BMJ Case Rep. 2023, 16, e249997. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.T.; Cai, J.; Wang, Y.Y.; Tu, W.J.; Wang, W.P.; Gong, L.M.; Wang, D.W.; Ye, Y.T.; Fang, S.G.; Jing, P.W. Newborn screening for inborn errors of metabolism in mainland china: 30 years of experience. JIMD Rep. 2012, 6, 79–83. [Google Scholar] [CrossRef] [PubMed]
- Screening Group of Neonatal Genetic Metabolic Disease, Special Committee of Birth Defects Prevention and Control, Chinese Preventive Medical Association; Gu, X.; Zhao, Z. Consensus on the organizational management and dry blood sample collection for neonatal inherited metabolic disease screening. Chin. J. Neonatol. 2023, 38, 321–326. [Google Scholar]
- Screening Group of Neonatal Genetic Metabolic Disease, Special Committee of Birth Defects Prevention and Control, Chinese Preventive Medical Association; Zhao, Z. Consensus on diagnosis and treatment of inherited metabolic diseases by newborn screening. Chin. J. Neonatol. 2023, 38, 385–394. [Google Scholar]
- Screening Group of Neonatal Genetic Metabolic Disease, Special Committee of Birth Defects Prevention and Control, Chinese Preventive Medical Association; Newborn Genetic Metabolic Disease Screening Laboratory Quality Evaluation Professional Committee from Clinical Laboratory Center of National Health Commission; Zhao, Z.; Wang, Z. Consensus on laboratory testing technical guidelines of neonatal genetic metabolic disease screening. Chin. J. Neonatol. 2023, 38, 449–454. [Google Scholar]
- Zhang, H.; Wang, Y.; Qiu, Y.; Zhang, C. Expanded newborn screening for inherited metabolic disorders by tandem mass spectrometry in a northern Chinese population. Front. Genet. 2022, 13, 801447. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Xu, J.; Song, X.; Du, J. Cost-effectiveness analysis of newborn screening by tandem mass spectrometry in Shenzhen, China: Value and affordability of new screening technology. BMC Health Serv. Res. 2022, 22, 1039. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Chen, C.; Sun, X.; Zhou, D.; Huang, X.; Dong, H. Newborn screening for inherited metabolic diseases using tandem mass spectrometry in China: Outcome and cost-utility analysis. J. Med. Screen. 2022, 29, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Qiang, R.; Song, C.; Ma, X.; Zhang, Y.; Li, F.; Wang, R.; Yu, W.; Feng, M.; Yang, L.; et al. Spectrum analysis of inborn errors of metabolism for expanded newborn screening in a northwestern Chinese population. Sci. Rep. 2021, 11, 2699. [Google Scholar] [CrossRef]
- Li, X.; He, J.; He, L.; Zeng, Y.; Huang, X.; Luo, Y.; Li, Y. Spectrum analysis of inherited metabolic disorders for expanded newborn screening in a central Chinese population. Front. Genet. 2022, 12, 763222. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.; Wang, W.; Liu, J.; Zhang, Z.; Zhao, Z.; He, F.; Yuan, S.; Wang, Z. National program for external quality assessment of Chinese newborn screening laboratories. Int. J. Neonatal Screen. 2020, 6, 38. [Google Scholar] [CrossRef] [PubMed]
- Jin, L.; He, F.; Zhang, C. Quality management of the newborn screening network: The Chinese experience. Chin. Med. J. 2023, 136, 2122–2124. [Google Scholar] [CrossRef]
- Yu, C.W.; He, X.Y.; Wan, K.X.; Yuan, Z.J.; Liu, H.; Zhang, J.; Liu, S.; Yang, J.; Zou, L. Improving quality management of newborn screening in southwest China. J. Int. Med. Res. 2021, 49, 3000605211002999. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Yang, R.; Huang, X.; Tian, Y.; Pei, X.; Bohn, M.K.; Zou, L.; Wang, Y.; Li, H.; Wang, T.; et al. Reference standards for newborn screening of metabolic disorders by tandem mass spectrometry: A nationwide study on millions of Chinese neonatal populations. Front. Mol. Biosci. 2021, 8, 719866. [Google Scholar] [CrossRef] [PubMed]
- Men, S.; Liu, S.; Zheng, Q.; Yang, S.; Mao, H.; Wang, Z.; Gu, Y.; Tang, X.; Wang, L. Incidence and genetic variants of inborn errors of metabolism identified through newborn screening: A 7-year study in eastern coastal areas of China. Mol. Genet. Genomic Med. 2023, 11, e2152. [Google Scholar] [CrossRef]
- Liu, G.; Liu, X.; Lin, Y. Newborn screening for inborn errors of metabolism in a northern Chinese population. J. Pediatr. Endocrinol. Metab. 2023, 36, 278–282. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Chen, D.; Peng, W.; Wang, K.; Lin, W.; Zhuang, J.; Zheng, Z.; Li, M.; Fu, Q. Newborn screening for isovaleric acidemia in Quanzhou, China. Clin. Chim. Acta 2020, 509, 25–29. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Lin, B.; Chen, Y.; Zheng, Z.; Fu, Q.; Lin, W.; Zhang, W. Biochemical and genetic characteristics of patients with primary carnitine deficiency identified through newborn screening. Orphanet J. Rare Dis. 2021, 16, 503, Erratum in Orphanet J. Rare Dis. 2022, 17, 21. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.; Cheng, Y.; Yin, X.; Miao, H.; Hu, Z.; Yang, J.; Zhang, Y.; Wu, B.; Huang, X. Newborn screening for mitochondrial carnitine-acylcarnitine cycle disorders in Zhejiang Province, China. Front. Genet. 2022, 13, 823687. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Xu, H.; Zhou, D.; Hu, Z.; Zhang, C.; Hu, L.; Zhang, Y.; Zhu, L.; Lu, B.; Zhang, T.; et al. Screening 3.4 million newborns for primary carnitine deficiency in Zhejiang Province, China. Clin. Chim. Acta 2020, 507, 199–204. [Google Scholar] [CrossRef]
- Lin, W.; Wang, K.; Zheng, Z.; Chen, Y.; Fu, C.; Lin, Y.; Chen, D. Newborn screening for primary carnitine deficiency in Quanzhou, China. Clin. Chim. Acta 2021, 512, 166–171. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Li, Q.; Wang, F.; Yan, L.; Zhuang, D.; Qiu, H.; Li, H.; Chen, L. Newborn screening and genetic analysis identify six novel genetic variants for primary carnitine deficiency in Ningbo Area, China. Front. Genet. 2021, 12, 686137. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Yin, Y.; Liu, H.; Peng, Y.; Ye, L.; Luo, Q.; Miao, J. Screening for newborn fatty acid oxidation disorders in Chongqing and the follow-up of confirmed children. J. Zhejiang Univ. 2022, 51, 290–297. [Google Scholar] [CrossRef] [PubMed]
- Tong, F.; Zhang, Y.; Chen, C.; Zhu, L.; Lu, Y.; Zhang, Z.; Chen, T.; Yan, J.; Zheng, J.; Zhao, X.; et al. Long-term prognosis of 35 patients with methionine adenosyltransferase deficiency based on newborn screening in China. Front. Cell Dev. Biol. 2023, 10, 1059680. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Cai, H.; Li, H.; Ji, Z.; Gu, M. Quantification of differential metabolites in dried blood spots using second-tier testing for SCADD/IBDD disorders based on large-scale newborn screening in a Chinese population. Front. Pediatr. 2021, 9, 757424. [Google Scholar] [CrossRef] [PubMed]
- Zhonghua, Y.; Xue, Y.; Chuan, X.; Za, Z. Study on newborn screening for Duchenne muscular dystrophy and diagnostic strategy. Chin. J. Neonatol. 2021, 38, 430–434. [Google Scholar] [CrossRef] [PubMed]
- Jia, C.; Zhao, D.; Li, Y.; Gao, Y.; Zhang, X.; Li, X.; Lv, S.; Li, R.; Zhu, X.; Liu, S. Newborn screening and genomic analysis of Duchenne muscular dystrophy in Henan, China. Clin. Chim. Acta 2023, 539, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Tian, L.; Gao, Q.; Guo, Y.; Li, G.; Li, Y.; Sun, M.; Yan, Y.; Li, Q.; Nie, W.; et al. Establishment of cutoff values for newborn screening of six lysosomal storage disorders by tandem mass spectrometry. Front. Pediatr. 2022, 10, 814461. [Google Scholar] [CrossRef] [PubMed]
- Tong, F.; Wang, J.; Xiao, R.; Wu, B.B.; Zou, C.C.; Wu, D.W.; Wang, H.; Zou, H.; Han, L.S.; Yang, L.; et al. Application of next generation sequencing in the screening of monogenic diseases in China, 2021: A consensus among Chinese newborn screening experts. World J. Pediatr. 2022, 18, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Sun, Y.; Xu, F.; Guo, J.; Li, L.; Lin, Z.; Ye, J.; Gu, X.; Yu, Y. A pilot study of expanded newborn screening for 573 genes related to severe inherited disorders in China: Results from 1,127 newborns. Ann. Transl. Med. 2020, 8, 1058. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Wu, D.; Zhu, L.; Wang, W.; Yang, R.; Yang, J.; He, Q.; Zhu, B.; You, Y.; Xiao, R.; et al. Application of a next-generation sequencing (NGS) panel in newborn screening efficiently identifies inborn disorders of neonates. Orphanet J. Rare Dis. 2022, 17, 66. [Google Scholar] [CrossRef] [PubMed]
- Hao, C.; Guo, R.; Hu, X.; Qi, Z.; Guo, Q.; Liu, X.; Liu, Y.; Sun, Y.; Zhang, X.; Jin, F.; et al. Newborn screening with targeted sequencing: A multicenter investigation and a pilot clinical study in China. J. Genet. Genom. 2022, 49, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.L.; Qian, G.L.; Wu, D.W.; Miao, J.K.; Yang, X.; Wu, B.Q.; Yan, Y.Q.; Li, H.B.; Mao, X.M.; He, J.; et al. A multicenter prospective study of next-generation sequencing-based newborn screening for monogenic genetic diseases in China. World J. Pediatr. 2023, 19, 663–673. [Google Scholar] [CrossRef] [PubMed]
- Ding, S.; Han, L. Newborn screening for genetic disorders: Current status and prospects for the future. Pediatr. Investig. 2022, 6, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Fan, C.; Huang, Y.; Feng, J.; Zhang, Y.; Miao, J.; Wang, X.; Li, Y.; Huang, C.; Jin, W.; et al. Genomic sequencing as a first-tier screening test and outcomes of newborn screening. JAMA Netw. Open 2023, 6, e2331162. [Google Scholar] [PubMed]
- Wang, X.; Guan, X.W.; Wang, Y.Y.; Zhang, Z.L.; Li, Y.H.; Yang, P.Y.; Sun, Y.; Jiang, T. Current attitudes and preconceptions on newborn genetic screening in the Chinese reproductive-aged population. Orphanet J. Rare Dis. 2022, 17, 322. [Google Scholar] [CrossRef] [PubMed]
- Ye, L.; Yin, Y.; Chen, M.; Gong, N.; Peng, Y.; Liu, H.; Miao, J. Combined genetic screening and traditional newborn screening to improve the screening efficiency of congenital hypothyroidism. Front. Pediatr. 2023, 11, 1185802. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Liu, Y.; Zhu, L.; Le, K.; Shen, Y.; Yang, C.; Chen, X.; Hu, H.; Ma, Q.; Shi, X.; et al. Combining newborn metabolic and genetic screening for neonatal intrahepatic cholestasis caused by citrin deficiency. J. Inherit. Metab. Dis. 2020, 43, 467–477. [Google Scholar] [CrossRef] [PubMed]
- Jian, M.; Wang, X.; Sui, Y.; Fang, M.; Feng, C.; Huang, Y.; Liu, C.; Guo, R.; Guan, Y.; Gao, Y.; et al. A pilot study of assessing whole genome sequencing in newborn screening in unselected children in China. Clin. Transl. Med. 2022, 12, e843. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Deng, L.; Huang, Y.; Xiao, Y.; Wen, J.; Liu, N.; Zeng, Y.; Zhang, H. Application of the artificial intelligence algorithm model for screening of inborn errors of metabolism. Front. Pediatr. 2022, 10, 855943. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.L.; Yang, Y.L.; Wang, T.; Xu, W.Z.; Yu, G.; Yang, J.B.; Sun, Q.L.; Gu, M.S.; Li, H.B.; Zhao, D.H.; et al. Establishment of an auxiliary diagnosis system of newborn screening for inherited metabolic diseases based on artificial intelligence technology and a clinical trial. Zhonghua Er Ke Za Zhi 2021, 59, 286–293. (In Chinese) [Google Scholar] [CrossRef] [PubMed]
- Shi, C.; Ao, Z.; Liu, B.; Xiao, X.; Gu, X.; Yang, Q.; Hao, H.; Cai, Y.; Li, S. Increased acylcarnitine ratio indices in newborn screening for carnitine-acylcarnitine translocase deficiency shows increased sensitivity and reduced false-positivity. Transl. Pediatr. 2023, 12, 871–881. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Lin, C.H.; Yin, X.; Zhu, L.; Yang, J.; Shen, Y.; Yang, C.; Chen, X.; Hu, H.; Ma, Q.; et al. Newborn screening for spinal muscular atrophy in China using DNA mass spectrometry. Front. Genet. 2019, 10, 1255. [Google Scholar] [CrossRef] [PubMed]
- Tian, G.L.; Xu, F.; Jiang, K.; Wang, Y.M.; Ji, W.; Zhuang, Y.P. Evaluation of a panel of very long-chain lysophosphatidylcholines and acylcarnitines for screening of X-linked adrenoleukodystrophy in China. Clin. Chim. Acta 2020, 503, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Yue, X.; Liu, W.; Liu, Y.; Shen, M.; Zhai, Y.; Ma, Z.; Cao, Z. Development, validation, and clinical application of an FIA-MS/MS method for the quantification of lysophosphatidylcholines in dried blood spots. J. Clin. Lab. Anal. 2022, 36, e24099. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.; Tang, F.; Cai, Y.; Tan, M.; Liu, S.; Xie, T.; Jiang, X.; Huang, Y. A pilot study of newborn screening for X-linked adrenoleukodystrophy based on liquid chromatography-tandem mass spectrometry method for detection of C26:0-lysophosphatidylcholine in dried blood spots: Results from 43,653 newborns in a southern Chinese population. Clin. Chim. Acta 2024, 552, 117653. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Jin, H.; Ye, M.; Han, R.; Han, N.; Zhao, J. Optimization of phenylalanine cut-off value in newborn screening based on blood sampling time. Am. J. Perinatol. 2022. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.; Zhu, J.; Liu, H.; Xiang, L.; Yao, Y.; Li, Q.; Deng, K.; Li, X. Birth prevalence of tetrahydrobiopterin deficiency in China: Data from the national newborn screening program, 2013–2019. J. Pediatr. Endocrinol. Metab. 2021, 34, 835–841. [Google Scholar] [CrossRef]
- Zhan, X.; Han, L.; Qiu, W.; Gu, X.; Guo, J.; Chang, S.; Wang, Y.; Zhang, H. Steroid profile in dried blood spots by liquid chromatography tandem mass spectrometry: Application to newborn screening for congenital adrenal hyperplasia in China. Steroids 2022, 185, 109056. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.W.; Hu, Z.Z.; Yang, J.B.; Zhang, Y.; Shi, Y.Z.; Zhu, S.S.; Yang, R.L.; Huang, X.W. Effects of delivery and storage conditions on concentrations of amino acids and carnitines in neonatal dried blood spots. J. Zhejiang Univ. (Med. Sci.) 2020, 49, 565–573. [Google Scholar]
- Xie, T.; Tan, M.; Jiang, X.; Feng, Y.; Chen, Q.; Mei, H.; Cai, Y.; Zou, H.; Huang, Y. Clinical features and outcomes of 31 children with congenital hypothyroidism missed by neonatal screening. J. Zhejiang Univ. 2022, 51, 314–320. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.N.; Yuan, X.L.; Zhu, J.; Xiang, L.C.; Li, Q.; Deng, K.; Li, X.H.; Liu, H.M. Geographic variations in the incidence of congenital hypothyroidism in China: A retrospective study based on 92 million newborns screened in 2013–2018. Chin. Med. J. 2021, 134, 2223–2230. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Zhong, Y.; Gu, Y.; Sharma, R.; Li, M.; Zhou, J.; Wu, Y.; Gao, Y.; Qin, G. Estimated cost-effectiveness of newborn screening for congenital cytomegalovirus infection in China using a Markov model. JAMA Netw. Open 2020, 3, e2023949. [Google Scholar] [CrossRef] [PubMed]
- Department of Health, Government of Hong Kong. Document Number: NBSIEM/1-60-2/04. Newborn Screening Program for Inborn Errors of Metabolism. April 2023. Available online: https://www21.ha.org.hk/smartpatient/SPW/MediaLibraries/SPW/SPWMedia/Education-pamphlet_NBS-IEM_Eng-(for-Apr-Sep-2023).pdf?ext=.pdf (accessed on 31 December 2023).
- Belaramani, K.M.; Chan, T.C.H.; Hau, E.W.L.; Yeung, M.C.W.; Kwok, A.M.K.; Lo, I.F.M.; Law, T.H.F.; Wu, H.; Wong, S.S.N.; Lam, S.W.; et al. Expanded Newborn Screening for Inborn Errors of Metabolism in Hong Kong: Results and Outcome of a 7 Year Journey. Int. J. Neonatal Screen. 2024, 10, 23. [Google Scholar] [CrossRef] [PubMed]
- The Chinese University of Hong Kong & Prince of Wales Hospital. JHF Newborn Metabolic Screening Program. 23 September 2023. Available online: https://www.obg.cuhk.edu.hk/services/clinical-services/obstetrics/fetal-medicine/jhf-newborn-metabolic-screening-program/ (accessed on 6 November 2023).
- Mak, C.M.; Law, E.C.; Lee, H.H.; Siu, W.K.; Chow, K.M.; Au Yeung, S.K.; Ngan, H.Y.; Tse, N.K.; Kwong, N.S.; Chan, G.C.; et al. The first pilot study of expanded newborn screening for inborn errors of metabolism and survey of related knowledge and opinions of health care professionals in Hong Kong. Hong Kong Med. J. 2018, 24, 226–237. [Google Scholar] [CrossRef] [PubMed]
- Lam, R.K.Y.; Lo, I.F.M.; Leung, C.M.; Tong, A.Y.H.; Yeung, S.W.C.; Leung, K.Y.; Hui, P.W.; Tsang, S.L.; Ma, G.G.Y.; But, B.W.M.; et al. Evaluation of the 18-month “Pilot Study of Newborn Screening for Inborn Errors of Metabolism” in Hong Kong. Hong Kong J. Pediatr. (New Ser.) 2020, 25, 16–22. Available online: https://www.hkjpaed.org/details.asp?id=1266&show=1234 (accessed on 3 February 2024).
- Leung, D.; Lee, P.P.W.; Lau, Y.L. Review of a decade of international experiences in severe combined immunodeficiency newborn screening using T-cell receptor excision Circle. Hong Kong J. Pediatr. (New Ser.) 2020, 25, 30–41. Available online: https://www.hkjpaed.org/details.asp?id=1268&show=1234 (accessed on 3 February 2024).
- Chan, S.H.S.; Wong, C.K.H.; Wu, T.; Wong, W.; Yu, M.K.L.; Au, I.C.H.; Chan, G.C.F. Significant healthcare burden and life cost of spinal muscular atrophy: Real-world data. Eur. J. Health Econ. 2023, 24, 1373–1382. [Google Scholar] [CrossRef] [PubMed]
- Hong Kong Hospital Authority Annual Plan 2023–3024; Hong Kong Hospital Authority: Hong Kong, China, 2023; pp. 9, 46, 48.
- Ngan, O.M.Y.; Wong, W.K.; Tam, J.C.; Li, C.K. Assessing the content quality of online parental resources about newborn metabolic disease screening: A content analysis. Int. J. Neonatal Screen. 2022, 8, 63. [Google Scholar] [CrossRef]
- Hui, L.L.; Nelson, E.A.S.; Deng, H.B.; Leung, T.Y.; Ho, C.H.; Chong, J.S.C.; Fung, G.P.G.; Hui, J.; Lam, H.S. The view of Hong Kong parents on secondary use of dried blood spots in newborn screening program. BMC Med. Ethics 2022, 23, 105. [Google Scholar] [CrossRef]
- Belaramani, K.M.; Fung, C.W.; Kwok, A.M.K.; Lee, S.Y.R.; Yau, E.K.C.; Luk, H.M.; Mak, C.M.; Yeung, M.C.W.; Ngan, O.M.Y. Public and healthcare provider receptivity toward the retention of dried blood spot cards and their usage for extended genetic testing in Hong Kong. Int. J. Neonatal Screen. 2023, 9, 45. [Google Scholar] [CrossRef] [PubMed]
- Mak, C.M.; Choi, C.T.; Wong, T.K.; Chin, H.H.; Lai, H.K.Y.; Yuet, K.Y. Technical study of automated high-throughput high-sensitive ceruloplasmin assay on dried blood spots-reinstate the potential use for newborn screening of Wilson Disease. Int. J. Neonatal Screen. 2022, 8, 64. [Google Scholar] [CrossRef] [PubMed]
- Mookken, T. Universal implementation of newborn screening in India. Int. J. Neonatal Screen. 2020, 6, 24. [Google Scholar] [CrossRef] [PubMed]
- Verma, J.; Roy, P.; Thomas, D.C.; Jhingan, G.; Singh, A.; Bijarnia-Mahay, S.; Verma, I.C. Newborn screening for congenital hypothyroidism, congenital adrenal hyperplasia, and glucose-6-phosphate dehydrogenase deficiency for improving health care in India. J. Pediatr. Intensive Care 2020, 9, 40–44. [Google Scholar] [CrossRef] [PubMed]
- Jalan, A.B.; Kudalkar, K.V. Newborn screening: Need of the hour. Karnataka Paediatr. J. 2021, 36, 35–41. Available online: https://iap-kpj.org/newborn-screening-need-of-the-hour/ (accessed on 3 February 2024). [CrossRef]
- Narayanan, M.P. Inherited metabolic disorders—Relevance of newborn screening in India. Acta Scientific Paediatr. 2022, 5, 25–26. Available online: https://actascientific.com/ASPE/ASPE-05-0507.php (accessed on 3 February 2024).
- Kumar, R.K.; Nevilebasappa, A.; Shenoi, A.; Nagar, N.; Das, H. Experience of one and half decades of newborn screening in India. EC Pediatr. 2022, 11, 17–26. Available online: https://ecronicon.org/ecpe/Experience-of-One-and-Half-Decades-of-Newborn-Screening-in-India (accessed on 3 February 2024).
- Anne, R.P.; Rahiman, E.A. Congenital hypothyroidism in India: A systematic review and meta-analysis of prevalence, screen positivity rates, and etiology. Lancet Reg. Health Southeast Asia 2022, 5, 100040. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.; Ray, S.K. Inborn errors of metabolism screening in neonates: Current perspective with diagnosis and therapy. Curr. Pediatr. Rev. 2022, 18, 274–285. [Google Scholar] [CrossRef] [PubMed]
- Vidavalur, R. Human and economic cost of disease burden due to congenital hypothyroidism in India: Too little, but not too late. Front. Pediatr. 2022, 10, 788589. [Google Scholar] [PubMed]
- Amalakani, S.; Raman Arepalli, K.V.; Suvvari, T.K.; Jillella, J.P. Perception towards newborn screening. J. Med. Evid. 2023, 4, 34–36. Available online: http://www.journaljme.org/text.asp?2023/4/1/34/374723 (accessed on 3 February 2024).
- DNB Pediatrics. Mission NEEV—Objectives and Workflow|DNB Solved Questions. Available online: https://www.dnbpediatrics.com/2022/11/mission-neev.html (accessed on 8 November 2023).
- Kommalur, A.; Devadas, S.; Kariyappa, M.; Sabapathy, S.; Benakappa, A.; Gagandeep, V.; Veranna Sajjan, S.; Krishnapura Lakshminarayana, S.; Dakshayani, B.; Devi Chinnappa, G. Newborn screening for five conditions in a tertiary care government hospital in Bengaluru, South India-three years’ experience. J. Trop. Pediatr. 2020, 66, 284–289. [Google Scholar] [CrossRef] [PubMed]
- Raveendran, A.; Chacko, T.J.; Prabhu, P.; Varma, R.; Lewis, L.E.; Rao, P.; Shetty, P.P.; Mallimoggala, Y.S.P.; Hedge, A.; Nayak, D.M.; et al. Need and viability of newborn screening programme in India: Report from a pilot study. Int. J. Neonatal Screen. 2022, 8, 26. [Google Scholar] [CrossRef]
- Krishna Prasad, H.; Pulluru, P.; Venugopalan, L.; Murugesan, G.; Ramanathan, S. Congenital hypothyroidism in Indian preterm babies—Screening, prevalence, and etiology. Pediatr. Endocrinol. Diabetes Metab. 2021, 27, 82–86. [Google Scholar] [CrossRef] [PubMed]
- Tippabathani, J.; Seenappa, V.; Murugan, A.; Phani, N.M.; Hampe, M.H.; Appaswamy, G.; Sadashiv Gambhir, P. Neonatal screening for congenital adrenal hyperplasia in Indian newborns with reflex genetic analysis of 21-hydroxylase deficiency. Int. J. Neonatal Screen. 2023, 9, 9. [Google Scholar] [CrossRef] [PubMed]
- Dabas, A.; Bothra, M.; Kapoor, S. CAH newborn screening in India: Challenges and opportunities. Int. J. Neonatal Screen. 2020, 6, 70. [Google Scholar] [CrossRef] [PubMed]
- Reddy, N.A.; Sharma, S.; Das, M.; Kapoor, A.; Maskey, U. Devastating salt-wasting crisis in a four-month-old male child with congenital adrenal hyperplasia, highlighting the essence of neonatal screening. Clin. Case Rep. 2022, 10, e6010. [Google Scholar] [CrossRef] [PubMed]
- Vats, P.; Dabas, A.; Jain, V.; Seth, A.; Yadav, S.; Kabra, M.; Gupta, N.; Singh, P.; Sharma, R.; Kumar, R.; et al. Newborn screening and diagnosis of infants with congenital adrenal hyperplasia. Indian Pediatr. 2020, 57, 49–55. Available online: https://www.indianpediatrics.net/jan2020/49.pdf (accessed on 3 February 2024). [CrossRef] [PubMed]
- Thaker, P.; Colah, R.B.; Patel, J.; Raicha, B.; Mistry, A.; Mehta, V.; Italia, Y.; Desai, S.; Dave, K.; Shanmugam, R.; et al. Newborn screening for sickle cell disease among tribal populations in the States of Gujarat and Madhya Pradesh in India: Evaluation and outcome over 6 years. Front. Med. 2022, 8, 731884. [Google Scholar] [CrossRef] [PubMed]
- Dave, K.; Desai, S.; Italia, Y.; Mukherjee, M.B.; Mehta, P.; Desai, G. Newborn screening and clinical profile of children with sickle cell disease in a tribal area of Gujarat. Indian Pediatr. 2022, 59, 230–233. [Google Scholar] [CrossRef] [PubMed]
- Babu, B.V.; Sharma, Y.; Sridevi, P.; Surti, S.B.; Ranjit, M.; Bhat, D.; Sarmah, J.; Sudhakar, G. Feasibility of population-based screening of sickle cell disease through the primary health care system in tribal areas of India. J. Med. Screen. 2023, 30, 28–35. [Google Scholar] [CrossRef]
- Mukherjee, M.B.; Colah, R.B.; Mehta, P.R.; Shinde, N.; Jain, D.; Desai, S.; Dave, K.; Italia, Y.; Raicha, B.; Serrao, E. Multicenter evaluation of HemoTypeSC ™as a point-of-care sickle cell disease rapid diagnostic test for newborns and adults across India. Am. J. Clin. Pathol. 2020, 153, 82–87. [Google Scholar] [CrossRef]
- Ministry of Health and Welfare. Prime Minister Launches National Sickle Cell Anaemia Elimination Mission from Shahdol, Madhya Pradesh. 1 July 2023. Available online: https://pib.gov.in/PressReleaseIframePage.aspx?PRID=1936735 (accessed on 8 November 2023).
- Octavius, G.S.; Daleni, V.A.; Sagala, Y.D.S. An insight into Indonesia’s challenges in implementing newborn screening programs and their future implications. Children 2023, 10, 1216. [Google Scholar] [CrossRef]
- Pulungan, A.B.; Soesanti, F.; Utari, A.; Pritayati, N.; Julia, M.; Annisa, D.; Andarie, A.A.; Bikin, I.W. Preliminary study of newborn screening for congenital hypothyroidism and congenital adrenal hyperplasia in Indonesia. eJournal Kedokt. Indones. 2020, 8, 98–103. [Google Scholar]
- Setyaningsih, W.; Wulandari, R.D. The evaluation of congenital hypothyroidism screening program in Indonesia: Literature review. J. Aisyah J. Ilmu Kesehat 2022, 7, 495–502. Available online: https://aisyah.journalpress.id/index.php/jika/article/view/1161 (accessed on 3 February 2024).
- VOI. Newborns in Indonesia Now Mandatory to Screen the Hypothyroid Concentric, This Is an Explanation from the Ministry of Health. 1 September 2022. Available online: https://voi.id/en/news/206259 (accessed on 15 November 2023).
- Pulungan, A.B.; Puteri, H.A.; Faizi, M.; Hofman, P.L.; Utari, A.; Chanoine, J.-P. Experiences and challenges with congenital hypothyroidism newborn screening in Indonesia: A national cross-sectional survey. Int. J. Neonatal Screen. 2024, 10, 8. [Google Scholar] [CrossRef] [PubMed]
- Tajima, T. Newborn screening in Japan—2021. Int. J. Neonatal Screen. 2022, 8, 3. [Google Scholar] [CrossRef] [PubMed]
- Minamitani, K. Newborn screening for congenital hypothyroidism in Japan. Int. J. Neonatal Screen. 2021, 7, 34. [Google Scholar] [CrossRef] [PubMed]
- Nagasaki, K.; Minamitani, K.; Nakamura, A.; Kobayashi, H.; Numakura, C.; Itoh, M.; Mushimoto, Y.; Fujikura, K.; Fukushi, M.; Tajima, T. Guidelines for newborn screening of congenital hypothyroidism (2021 Revision). Clin. Pediatr. Endocrinol. 2023, 32, 26–51. [Google Scholar] [CrossRef] [PubMed]
- Tsuji-Hosokawa, A.; Kashimada, K. Thirty-year lessons from the newborn screening for congenital adrenal hyperplasia (CAH) in Japan. Int. J. Neonatal Screen. 2021, 7, 36. [Google Scholar] [CrossRef]
- Watanabe, K.; Tsuji-Hosokawa, A.; Hashimoto, A.; Konishi, K.; Ishige, N.; Yajima, H.; Sutani, A.; Nakatani, H.; Gau, M.; Takasawa, K.; et al. The high relevance of 21-deoxycortisol, (androstenedione + 17α-hydroxyprogesterone)/cortisol, and 11-deoxycortisol/17α-hydroxyprogesterone for newborn screening of 21-hydroxylase deficiency. J. Clin. Endocrinol. Metab. 2022, 107, 3341–3352. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, A.; Wada, Y.; Ohura, T.; Kure, S. The discovery of GALM deficiency (type IV galactosemia) and newborn screening system for galactosemia in Japan. Int. J. Neonatal Screen. 2021, 7, 68. [Google Scholar] [CrossRef] [PubMed]
- Stapleton, M.; Kubaski, F.; Mason, R.W.; Shintaku, H.; Kobayashi, H.; Yamaguchi, S.; Taketani, T.; Suzuki, Y.; Orii, K.; Orii, T.; et al. Newborn screening for mucopolysaccharidoses: Measurement of glycosaminoglycans by LC-MS/MS. Mol. Genet. Metab. Rep. 2020, 22, 100563. [Google Scholar] [CrossRef] [PubMed]
- Shigematsu, Y.; Yuasa, M.; Ishige, N.; Nakajima, H.; Tajima, G. Development of second-tier liquid chromatography-tandem mass spectrometry analysis for expanded newborn screening in Japan. Int. J. Neonatal Screen. 2021, 7, 44. [Google Scholar] [CrossRef] [PubMed]
- Shimozawa, N.; Takashima, S.; Kawai, H.; Kubota, K.; Sasai, H.; Orii, K.; Ogawa, M.; Ohnishi, H. Advanced diagnostic system and introduction of newborn screening of adrenoleukodystrophy and peroxisomal disorders in Japan. Int. J. Neonatal Screen. 2021, 7, 58. [Google Scholar] [CrossRef] [PubMed]
- Niba, E.T.E.; Wijaya, Y.O.S.; Awano, H.; Taniguchi, N.; Takeshima, Y.; Nishio, H.; Shinohara, M. DBS screening for glycogen storage disease type 1a: Detection of c.648G>T mutation in G6PC by combination of modified competitive oligonucleotide priming-PCR and melting curve analysis. Int. J. Neonatal Screen. 2021, 7, 79. [Google Scholar] [CrossRef] [PubMed]
- Wijaya, Y.O.S.; Niba, E.T.E.; Yabushita, R.; Bouike, Y.; Nishio, H.; Awano, H. Glycogen storage disease type Ia screening using dried blood spots on filter paper: Application of COP-PCR for detection of the c.648G>T G6PC gene mutation. Kobe J. Med. Sci. 2021, 67, E71–E78. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8622217/ (accessed on 3 February 2024). [PubMed]
- Odagiri, S.; Kabata, D.; Tomita, S.; Kudo, S.; Sakaguchi, T.; Nakano, N.; Yamamoto, K.; Shintaku, H.; Hamazaki, T. Clinical and genetic characteristics of patients with mild hyperphenylalaninemia identified by newborn screening program in Japan. Int. J. Neonatal Screen. 2021, 7, 17. [Google Scholar] [CrossRef] [PubMed]
- Osawa, Y.; Kobayashi, H.; Tajima, G.; Hara, K.; Yamada, K.; Fukuda, S.; Hasegawa, Y.; Aisaki, J.; Yuasa, M.; Hata, I.; et al. The frequencies of very long-chain acyl-CoA dehydrogenase deficiency genetic variants in Japan have changed since the implementation of expanded newborn screening. Mol. Genet. Metab. 2022, 136, 74–79. [Google Scholar] [CrossRef] [PubMed]
- Tajima, G.; Kagawa, R.; Sakura, F.; Nakamura-Utsunomiya, A.; Hara, K.; Yuasa, M.; Hasegawa, Y.; Sasai, H.; Okada, S. Current perspectives on neonatal screening for propionic acidemia in Japan: An unexpectedly high incidence of patients with mild disease caused by a common PCCB variant. Int. J. Neonatal Screen. 2021, 7, 35. [Google Scholar] [CrossRef] [PubMed]
- Bo, R.; Musha, I.; Yamada, K.; Kobayashi, H.; Hasegawa, Y.; Awano, H.; Arao, M.; Kikuchi, T.; Taketani, T.; Ohtake, A.; et al. Need for strict clinical management of patients with carnitine palmitoyltransferase II deficiency: Experience with two cases detected by expanded newborn screening. Mol. Genet. Metab. Rep. 2020, 24, 100611. [Google Scholar] [CrossRef] [PubMed]
- Kagawa, R.; Tajima, G.; Maeda, T.; Sakura, F.; Nakamura-Utsunomiya, A.; Hara, K.; Nishimura, Y.; Yuasa, M.; Shigematsu, Y.; Tanaka, H.; et al. Pilot study on neonatal screening for methylmalonic acidemia caused by defects in the adenosylcobalamin synthesis pathway and homocystinuria caused by defects in homocysteine remethylation. Int. J. Neonatal Screen. 2021, 7, 39. [Google Scholar] [CrossRef] [PubMed]
- Tajima, G.; Hara, K.; Tsumura, M.; Kagawa, R.; Sakura, F.; Sasai, H.; Yuasa, M.; Shigematsu, Y.; Okada, S. Newborn screening with (C16 + C18:1)/C2 and C14/C3 for carnitine palmitoyltransferase II deficiency throughout Japan has revealed C12/C0 as an index of higher sensitivity and specificity. Int. J. Neonatal Screen. 2023, 9, 62. [Google Scholar] [CrossRef] [PubMed]
- Yamada, K.; Yokoyama, K.; Aoki, K.; Taketani, T.; Yamaguchi, S. Long-term outcomes of adult patients with homocystinuria before and after newborn screening in Japan. Int. J. Neonatal Screen. 2020, 6, 60. [Google Scholar] [CrossRef] [PubMed]
- Yamada, K.; Yamaguchi, S.; Yokoyama, K.; Aoki, K.; Taketani, T. Long-term neurological outcomes of adult patients with phenylketonuria before and after newborn screening in Japan. Int. J. Neonatal Screen. 2021, 7, 21. [Google Scholar] [CrossRef] [PubMed]
- Hattori, T.; Notsu, Y.; Tanaka, M.; Matsui, M.; Tetsuo Iida, T.; Watanabe, J.; Osawa, Y.; Yamaguchi, S.; Yano, S.; Taketani, T.; et al. A simple flow injection analysis–tandem mass spectrometry method to reduce false positives of C5-acylcarnitines due to pivaloylcarnitine using reference ions. Children 2022, 9, 694. [Google Scholar] [CrossRef] [PubMed]
- Sawada, T.; Kido, J.; Nakamura, K. Newborn screening for Pompe Disease. Int. J. Neonatal Screen. 2020, 6, 31. [Google Scholar] [CrossRef] [PubMed]
- Sawada, T.; Kido, J.; Sugawara, K.; Momosaki, K.; Yoshida, S.; Kojima-Ishii, K.; Inoue, T.; Matsumoto, S.; Endo, F.; Ohga, S.; et al. Current status of newborn screening for Pompe disease in Japan. Orphanet J. Rare Dis. 2021, 16, 516. [Google Scholar] [CrossRef] [PubMed]
- Arunkumar, N.; Langan, T.J.; Stapleton, M.; Kubaski, F.; Mason, R.W.; Singh, R.; Kobayashi, H.; Yamaguchi, S.; Suzuki, Y.; Orii, K.; et al. Newborn screening of mucopolysaccharidoses: Past, present, and future. J. Hum. Genet. 2020, 65, 557–567. [Google Scholar] [CrossRef] [PubMed]
- Sawada, T.; Kido, J.; Yoshida, S.; Sugawara, K.; Momosaki, K.; Inoue, T.; Tajima, G.; Sawada, H.; Mastumoto, S.; Endo, F.; et al. Newborn screening for Fabry disease in the western region of Japan. Mol. Genet. Metab. Rep. 2020, 22, 100562. [Google Scholar] [CrossRef] [PubMed]
- Sawada, T.; Kido, J.; Sugawara, K.; Matsumoto, S.; Takada, F.; Tsuboi, K.; Ohtake, A.; Endo, F.; Nakamura, K. Detection of novel Fabry disease-associated pathogenic variants in Japanese patients by newborn and high-risk screening. Mol. Genet. Genomic Med. 2020, 8, e1502. [Google Scholar] [CrossRef] [PubMed]
- Muto, Y.; Suzuki, M.; Takei, H.; Saito, N.; Mori, J.; Sugimoto, S.; Imagawa, K.; Nambu, R.; Oguri, S.; Itonaga, T.; et al. Dried blood spot-based newborn screening for bile acid synthesis disorders, Zellweger spectrum disorder, and Niemann-Pick type C1 by detection of bile acid metabolites. Mol. Genet. Metab. 2023, 140, 107703. [Google Scholar] [CrossRef]
- Nishio, H. Newborn screening for spinal muscular atrophy. Lancet Child Adolesc. Health 2023, 7, 146–147. [Google Scholar] [CrossRef] [PubMed]
- Kimizu, T.; Ida, S.; Oki, K.; Shima, M.; Nishimoto, S.; Nakajima, K.; Ikeda, T.; Mogami, Y.; Yanagihara, K.; Matsuda, K.; et al. Newborn screening for spinal muscular atrophy in Osaka—Challenges in a Japanese pilot study. Brain Dev. 2023, 45, 363–371. [Google Scholar] [CrossRef]
- Kimizu, T.; Ida, S.; Okamoto, K.; Awano, H.; Niba, E.T.E.; Wijaya, Y.O.S.; Okazaki, S.; Shimomura, H.; Lee, T.; Tominaga, K.; et al. Spinal muscular atrophy: Diagnosis, incidence, and newborn screening in Japan. Int. J. Neonatal Screen. 2021, 7, 45. [Google Scholar] [CrossRef]
- Sawada, T.; Kido, J.; Sugawara, K.; Yoshida, S.; Ozasa, S.; Nomura, K.; Okada, K.; Fujiyama, N.; Nakamura, K. Newborn screening for spinal muscular atrophy in Japan: One year of experience. Mol. Genet. Metab. Rep. 2022, 32, 100908. [Google Scholar] [CrossRef] [PubMed]
- Noguchi, Y.; Bo, R.; Nishio, H.; Matsumoto, H.; Matsui, K.; Yano, Y.; Sugawara, M.; Ueda, G.; Wijaya, Y.O.S.; Niba, E.T.E.; et al. PCR-based screening of spinal muscular atrophy for newborn infants in Hyogo Prefecture, Japan. Genes 2022, 13, 2110, Erratum in Genes 2023, 14, 759. [Google Scholar] [CrossRef] [PubMed]
- Wijaya, Y.O.S.; Purevsuren, J.; Harahap, N.I.F.; Niba, E.T.E.; Bouike, Y.; Nurputra, D.K.; Rochmah, M.A.; Thursina, C.; Hapsara, S.; Yamaguchi, S.; et al. Assessment of spinal muscular atrophy carrier status by determining SMN1 copy number using dried blood spots. Int. J. Neonatal Screen. 2020, 6, 43. [Google Scholar] [CrossRef] [PubMed]
- Harahap, N.I.; Harahap, I.S.; Kaszynski, R.H.; Nurputra, D.K.; Hartomo, T.B.; Pham, H.T.; Yamamoto, T.; Morikawa, S.; Nishimura, N.; Rusdi, I.; et al. Spinal muscular atrophy patient detection and carrier screening using dried blood spots on filter paper. Genet. Test Mol. Biomark. 2012, 16, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Harahap, N.I.F.; Nurputra, D.K.; Okamoto, K.; Saito, T.; Takeuchi, A.; Lai, P.S.; Yamaguchi, S.; Shinohara, M. Dried blood spot screening system for spinal muscular atrophy with allele-specific polymerase chain reaction and melting peak analysis. Genet. Test. Mol. Biomark. 2021, 25, 293–301. [Google Scholar] [CrossRef]
- Yamaguchi, T.; Nakamura, A.; Nakayama, K.; Hishimura, N.; Morikawa, S.; Ishizu, K.; Tajima, T. Targeted next-generation sequencing for congenital hypothyroidism with positive neonatal TSH screening. J. Clin. Endocrinol. Metab. 2020, 105, dgaa308. [Google Scholar] [CrossRef] [PubMed]
- Wakamatsu, M.; Kojima, D.; Muramatsu, H.; Okuno, Y.; Kataoka, S.; Nakamura, F.; Sakai, Y.; Tsuge, I.; Ito, T.; Ueda, K.; et al. TREC/KREC newborn screening followed by next-generation sequencing for severe combined immunodeficiency in Japan. J. Clin. Immunol. 2022, 42, 1696–1707. [Google Scholar] [CrossRef] [PubMed]
- Konomura, K.; Hoshino, E.; Sakai, K.; Fukuda, T.; Tajima, G. Development of a model for quantitative assessment of newborn screening in Japan using the analytic hierarchy process. Int. J. Neonatal Screen. 2023, 9, 39. [Google Scholar] [CrossRef] [PubMed]
- Morning Calm Birthing Services. Newborn Screening Tests in Korea. Available online: https://www.morningcalmbirthing.com/resources/newborn-screening-tests-korea/ (accessed on 15 June 2023).
- Lee, H.-J. Application of a Targeted Next Generation Sequencing Panel for Newborn Screening. Ph.D. Dissertation, Yonsei University, Seoul, Republic of Korea, 2019. [Google Scholar]
- Lee, H.; Lim, J.; Shin, J.E.; Eun, H.S.; Park, M.S.; Park, K.I.; Namgung, R.; Lee, J.S. Implementation of a targeted next-generation sequencing panel for constitutional newborn screening in high-risk neonates. Yonsei Med. J. 2019, 60, 1061–1066. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.S.; Chung, H.S.; Kim, J.S. Analysis of online parenting community posts on expanded newborn screening for metabolic disorders using topic modeling: A quantitative content analysis. Korean J. Women Health Nurs. 2023, 29, 20–31. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-Y.; Ahn, S. Report of the Korean Association of External Quality assessment service on metabolite testing in Korea (2020–2021). Lab. Med. Qual. Assur. 2022, 44, 143–158. [Google Scholar] [CrossRef]
- Michael, I. CRMs for Dried Blood Spot Testing for Newborn Screening. Spectroscopy—Europe World. 25 January 2023. Available online: https://www.spectroscopyeurope.com/product/crms-dried-blood-spot-testing-newborn-screening (accessed on 13 June 2023).
- Kim, K.; Lee, H.; Lee, J.J.; Cha, K.; Hyun Park, N.; Shin, Y.K.; Chae, H.; Oh, E.J. Identification of a frit-related sample carryover in newborn screening by tandem mass spectrometry. J. Mass Spectrom. Adv. Clin. Lab. 2023, 27, 56–60. [Google Scholar] [CrossRef]
- Hoehn, T.; Lukacs, Z.; Stehn, M.; Mayatepek, E.; Philavanh, K.; Bounnack, S. Establishment of the first newborn screening program in the People’s Democratic Republic of Laos. J. Trop. Pediatr. 2013, 59, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Vesaphong, P.; Lindemuth, L.; Istaz, B.; Kedsatha, P.; Saysanasongkham, B.; Hoehn, T. Establishment of a sustainable newborn TSH-screening program in the People’s Democratic Republic of Laos. Early Hum. Dev. 2021, 154, 105306. [Google Scholar] [CrossRef] [PubMed]
- Höhn, T. (Heinrich-Heine-Universität, Düsseldorf, Germany). Personal communication, 2023.
- Kam, K. (Marketing Department, Xcelom, Hong Kong, China). Personal communication, 2024.
- Leong, Y.H.; Gan, C.Y.; Tan, M.A.; Majid, M.I. Present status and future concerns of expanded newborn screening in Malaysia: Sustainability, challenges and perspectives. Malays. J. Med. Sci. 2014, 21, 63–67. [Google Scholar] [PubMed]
- Yunus, Z.M.; Rahman, S.A.; Choy, Y.S.; Keng, W.T.; Ngu, L.H. Pilot study of newborn screening of inborn error of metabolism using tandem mass spectrometry in Malaysia: Outcome and challenges. J. Pediatr. Endocrinol. Metab. 2016, 29, 1031–1039. [Google Scholar] [CrossRef] [PubMed]
- Shafie, A.A.; Supian, A.; Ahmad Hassali, M.A.; Ngu, L.H.; Thong, M.K.; Ayob, H.; Chaiyakunapruk, N. Rare disease in Malaysia: Challenges and solutions. PLoS ONE 2020, 15, e0230850, Erratum in PLoS ONE 2022, 17, e0273939. [Google Scholar] [CrossRef] [PubMed]
- Habib, A.; Nazri, M.I.B.A.; Rahman, S.A. Dataset from dried blood spot acylcarnitine for detection of carnitine translocase (CACT) deficiency and carnitine palmitoyl transferase 2 (CPT2) deficiency. Data Brief. 2023, 48, 109090. [Google Scholar] [CrossRef] [PubMed]
- Zulkeflee, R.H.; Bahar, R.; Abdullah, M.; Mohd Radzi, M.A.R.; Md Fauzi, A.; Hassan, R. Application of targeted next-generation sequencing for the investigation of thalassemia in a developing country: A single center experience. Diagnostics 2023, 13, 1379. [Google Scholar] [CrossRef]
- Zailani, M.A.H.; Raja Sabudin, R.Z.A.; Abdul Jalil, D.; Ithnin, A.; Ayub, N.A.; Alauddin, H.; Jalil, N.; Md Fauzi, A.; Cheah, F.C.; Lim, L.S.; et al. Evaluation of quantitative point-of-care test for measurement of glucose-6-phosphate dehydrogenase enzyme activity in Malaysia. Malays. J. Pathol. 2023, 45, 31–41. [Google Scholar] [PubMed]
- Hamzah, H.; Sutan, R.; Mohd Tamil, A.; Mohd Kassim, A.B.; Mohamed Soid, A.; Singh, A. Assessment of healthcare workers’ behavior in implementing a newborn screening program for congenital hypothyroidism in Perak, Malaysia. J. Health Res. 2021, 35, 172–185. [Google Scholar] [CrossRef]
- Liew, S.H.; Lim, J.Y.; Yahya, H.M.; Rajikan, R. Knowledge and perception of inborn errors of metabolism (IEMs) among healthcare students at a selected public university in Klang Valley, Malaysia. Intractable Rare Dis. Res. 2022, 11, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Tsevgee, A.; Batjargal, K.; Munkhchuluun, T.; Khurelbaatar, N.; Nansal, G.; Bulgan, O.E.; Nyamjav, S.; Zagd, G.; Ganbaatar, E. First experiences with newborn screening for congenital hypothyroidism in Ulaanbaatar, Mongolia. Int. J. Neonatal Screen. 2021, 7, 29. [Google Scholar] [CrossRef]
- Ganbaatar, E. (Kazakh National Medical University, Ulanbaatar, Mongolia). Personal communication, 2023.
- Sharma Pandey, A.; Joshi, S.; Rajbhandari, R.; Kansakar, P.; Dhakal, S.; Fingerhut, R. Newborn screening for selected disorders in Nepal: A pilot study. Int. J. Neonatal Screen. 2019, 5, 18. [Google Scholar] [CrossRef] [PubMed]
- Khan, J.K.; Manandhar, S.; Subedi, K.U.; Paudel, P.; Bir Karmancharya, S.; Vaidya, K.M.; Manandhar, D.S.; Chhantel, R.; Garza, R.; Dookeran, K.A. Determination of feasibility and acceptability of newborn screening in Nepal—A pilot study. Poster PC2106. In Proceedings of the American Academy of Pediatrics National Conference & Exhibition 2022, Anaheim, CA, USA, 7–11 October 2022. Available online: https://www.eventscribe.net/2022/AAPexperience/fsPopup.asp?PosterID=524433&mode=posterinfo (accessed on 26 December 2023).
- Sharma Pandey, A. Case reports of metabolic disorders from Nepal. Mol. Genet. Metab. Rep. 2019, 21, 100542. [Google Scholar] [CrossRef] [PubMed]
- Pandey, S.; Shrestha, N. Sickle cell anaemia among Tharu population visiting the outpatient department of general medicine in a secondary care centre: A descriptive cross-sectional study. J. Nepal Med. Assoc. 2022, 60, 774–776. [Google Scholar] [CrossRef] [PubMed]
- National Screening Unit. Changes to the Disorders Screened for: Conditions No Longer Being Screened for. Available online: https://www.nsu.govt.nz/pregnancy-newborn-screening/newborn-metabolic-screening-programme-heel-prick-test/about-test (accessed on 8 November 2023).
- Wilson, C.; Knoll, D.; de Hora, M.; Kyle, C.; Glamuzina, E.; Webster, D. The decision to discontinue screening for carnitine uptake disorder in New Zealand. J. Inherit. Metab. Dis. 2019, 42, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Heather, N.; de Hora, M.; Brothers, S.; Grainger, P.; Knoll, D.; Webster, D. Introducing newborn screening for severe combined immunodeficiency-the New Zealand experience. Int. J. Neonatal Screen. 2022, 8, 33. [Google Scholar] [CrossRef] [PubMed]
- de Hora, M.R.; Heather, N.L.; Patel, T.; Bresnahan, L.G.; Webster, D.; Hofman, P.L. Measurement of 17-hydroxyprogesterone by LC MSMS improves newborn screening for CAH due to 21-Hydroxylase deficiency in New Zealand. Int. J. Neonatal Screen. 2020, 6, 6. [Google Scholar] [CrossRef] [PubMed]
- de Hora, M.; Heather, N.; Webster, D.; Albert, B.; Hofman, P. The use of liquid chromatography-tandem mass spectrometry in newborn screening for congenital adrenal hyperplasia: Improvements and future perspectives. Front. Endocrinol. 2023, 14, 1226284. [Google Scholar] [CrossRef] [PubMed]
- de Hora, M.R.; Heather, N.L.; Patel, T.; Bresnahan, L.G.; Webster, D.; Hofman, P.L. Implementing steroid profiling by liquid chromatography-tandem mass spectrometry improves newborn screening for congenital adrenal hyperplasia in New Zealand. Clin. Endocrinol. 2021, 94, 904–912. [Google Scholar] [CrossRef]
- de Hora, M.R.; Heather, N.L.; Webster, D.; Albert, B.B.; Hofman, P.L. Birth weight- or gestational age-adjusted second-tier LCMSMS cutoffs improve newborn screening for CAH in New Zealand. J. Clin. Endocrinol. Metab. 2021, 106, e3390–e3399. [Google Scholar] [CrossRef] [PubMed]
- de Hora, M.R.; Heather, N.L.; Webster, D.; Albert, B.B.; Hofman, P.L. Evaluation of a new laboratory protocol for newborn screening for congenital adrenal hyperplasia in New Zealand. Int. J. Neonatal Screen. 2022, 8, 56. [Google Scholar] [CrossRef] [PubMed]
- Heather, N.L.; Hofman, P.L.; de Hora, M.; Carll, J.; Derraik, J.G.; Webster, D. Evaluation of the revised New Zealand national newborn screening protocol for congenital hypothyroidism. Clin. Endocrinol. 2017, 86, 431–437. [Google Scholar] [CrossRef] [PubMed]
- Heather, N.L.; Derraik, J.G.B.; Webster, D.; Hofman, P.L. The impact of demographic factors on newborn TSH levels and congenital hypothyroidism screening. Clin. Endocrinol. 2019, 91, 456–463. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.; Hofman, P.; Li, B.; Heron, C.; Heather, N. A positive newborn screen for congenital hypothyroidism in a clinically euthyroid neonate-avoiding unnecessary treatment. Int. J. Neonatal Screen. 2023, 9, 16. [Google Scholar] [CrossRef] [PubMed]
- West, R.; Hong, J.; Derraik, J.G.B.; Webster, D.; Heather, N.L.; Hofman, P.L. Newborn screening TSH values less than 15 mIU/L are not associated with long-term hypothyroidism or cognitive impairment. J. Clin. Endocrinol. Metab. 2020, 105, dgaa415. [Google Scholar] [CrossRef] [PubMed]
- Heather, N.; Webster, D. It all depends on what you count-the importance of definitions in evaluation of CF screening performance. Int. J. Neonatal Screen. 2020, 6, 47. [Google Scholar] [CrossRef]
- Heather, N.; Morgan, L.; Knoll, D.; Shore, K.; de Hora, M.; Webster, D. Introduction of a protocol for structured follow-up and texting of inadequate and borderline-positive newborn metabolic screening results. Int. J. Neonatal Screen. 2022, 8, 30. [Google Scholar] [CrossRef] [PubMed]
- Bernhardt, I.; Glamuzina, E.; Ryder, B.; Knoll, D.; Heather, N.; De Hora, M.; Webster, D.; Wilson, C. The risk of classical galactosaemia in newborns with borderline galactose metabolites on newborn screening. JIMD Rep. 2022, 64, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Bernhardt, I.; Glamuzina, E.; Dowsett, L.K.; Webster, D.; Knoll, D.; Carpenter, K.; Bennett, M.J.; Maeda, M.; Wilson, C. Genotype-phenotype correlations in CPT1A deficiency detected by newborn screening in Pacific populations. JIMD Rep. 2022, 63, 322–329. [Google Scholar] [CrossRef] [PubMed]
- Alsweiler, J.M.; Heather, N.; Harris, D.L.; McKinlay, C.J.D. Application of the screening test principles to screening for neonatal hypoglycemia. Front. Pediatr. 2022, 10, 1048897. [Google Scholar] [CrossRef] [PubMed]
- Te Whatu Ora, Health New Zealand. Newborn Screening for Spinal Muscular Atrophy (SMA) to Help Babies Access Life-Saving Treatments Earlier. Available online: https://www.tewhatuora.govt.nz/whats-happening/news-and-updates/older-news-items/new-screening-for-spinal-muscular-atrophy-sma-to-help-babies-access-life-saving-treatments-earlier/ (accessed on 17 November 2023).
- Provincial Assembly of Sindh. The Sindh Newborn Screening Act of 2013. 23 September 2013. Available online: http://www.pas.gov.pk/index.php/acts/details/en/31/255 (accessed on 17 June 2023).
- Mansoor, S. Trends of congenital hypothyroidism and inborn errors of metabolism in Pakistan. Orphanet J. Rare Dis. 2020, 15, 321. [Google Scholar] [CrossRef] [PubMed]
- Majid, H.; Jafri, L.; Khan, N.; Khan, A.H. Establishing a nationwide newborn screening program for congenital hypothyroidism: A systematic review. Pak. J. Pathol. 2022, 33, 104–108. [Google Scholar] [CrossRef]
- Majid, H.; Jafri, L.; Ahmed, S.; Humayun, K.; Kirmani, S.; Ali, N.; Moiz, B.; Khan, A.H.; Afroze, B. Perspective on newborn screening (NBS): Evidence sharing on conditions to be included in NBS in Pakistan. J. Pak. Med. Assoc. 2022, 72, 526–531. [Google Scholar] [CrossRef] [PubMed]
- Wasim, M.; Khan, H.N.; Ayesha, H.; Awan, F.R. Need and challenges in establishing newborn screening programs for inherited metabolic disorders in developing countries. Adv. Biol. 2023, 7, e2200318. [Google Scholar] [CrossRef]
- The Aga Khan University. Nationwide Screening Needed to Protect Newborns. 28 February 2019. Available online: https://www.aku.edu/news/Pages/News_Details.aspx?nid=NEWS-001762 (accessed on 17 June 2023).
- Wasim, M.; Khan, H.N.; Ayesha, H.; Goorden, S.M.I.; Vaz, F.M.; van Karnebeek, C.D.M.; Awan, F.R. Biochemical screening of intellectually disabled patients: A stepping stone to initiate a newborn screening program in Pakistan. Front. Neurol. 2019, 10, 762. [Google Scholar] [CrossRef] [PubMed]
- Bibi, A.; Haroon, Z.H.; Shujaat, A.; Aamir, M.; Irum, S.; Jaffar, S.R. Establishing biotinidase reference interval: A foundation stone for newborn screening of biotinidase deficiency in Pakistan. J. Pak. Med. Assoc. 2022, 72, 97–100. [Google Scholar] [CrossRef] [PubMed]
- Chaudhri, N.; Haider, S. Diagnostic evaluation of heel prick newborn screening of thyroid stimulating hormone on dissociation-enhanced lanthanide fluorescence immunoassay with the establishment of reference value in Pakistani neonates. J. Pak. Med. Assoc. 2021, 71, 191–194. [Google Scholar] [CrossRef] [PubMed]
- Jafri, L.; Khan, A.H.; Ilyas, M.; Nisar, I.; Khalid, J.; Majid, H.; Hotwani, A.; Jehan, F. Metabolomics of a neonatal cohort from the Alliance for Maternal and Newborn Health Improvement biorepository: Effect of preanalytical variables on reference intervals. PLoS ONE 2023, 18, e0279931. [Google Scholar] [CrossRef] [PubMed]
- Wallace, J.G.; Tipu, H.N.; Stafstrom, K.; Alosaimi, M.F.; Massaad, M.J.; Bainter, W.; Geha, R.S.; Chou, J. Rethinking newborn screening for severe combined immunodeficiency: Lessons from an international partnership for patients with primary immunodeficiencies in Pakistan. Clin. Immunol. 2019, 202, 29–32. [Google Scholar] [CrossRef] [PubMed]
- Majid, H.; Ahmed, S.; Siddiqui, I.; Humayun, K.; Karimi, H.; Khan, A.H. Newborn screening for congenital hypothyroidism: Improvement in short-term follow-up by audit and monitoring. BMC Res. Notes 2020, 13, 563. [Google Scholar] [CrossRef] [PubMed]
- Tariq, B.; Ahmed, A.; Habib, A.; Turab, A.; Ali, N.; Soofi, S.B.; Nooruddin, S.; Kumar, R.J.; Tariq, A.; Shaheen, F.; et al. Assessment of knowledge, attitudes and practices towards newborn screening for congenital hypothyroidism before and after a health education intervention in pregnant women in a hospital setting in Pakistan. Int. Health 2018, 10, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Mohsin, S.N.; Zulfiqar, S.; Javed, R.; Razi, A. Newborn screening for congenital hypothyroidism: Impact of parents counseling on the uptake of program. Prof. Med. J. 2020, 27, 2350–2356. [Google Scholar] [CrossRef]
- Padilla, C.D.; Therrell, B.L., Jr.; Alcausin, M.M.L.B.; de Castro, R.C., Jr.; Gepte, M.B.P.; Reyes, M.E.L.; Jomento, C.M.; Suarez, R.C.N.; Maceda, E.B.G.; Abarquez, C.G.; et al. Successful implementation of newborn screening for hemoglobin disorders in the Philippines. Int. J. Neonatal Screen. 2021, 7, 30. [Google Scholar] [CrossRef] [PubMed]
- Padilla, C.D.; Therrell, B.L., Jr.; Alcausin, M.M.L.B.; Chiong, M.A.D.; Abacan, M.A.R.; Reyes, M.E.L.; Jomento, C.M.; Dizon-Escoreal, M.T.T.; Canlas, M.A.E.; Abadingo, M.E.; et al. Successful implementation of expanded newborn screening in the Philippines using tandem mass spectrometry. Int. J. Neonatal Screen. 2022, 8, 8. [Google Scholar] [CrossRef] [PubMed]
- Padilla, C.D.; Therrell, B.L.; Panol, K.A.R.; Suarez, R.C.N.; Reyes, M.E.L.; Jomento, C.M.; Maceda, E.B.G.; Lising, J.A.C.; Beltran, F.D.E.; Orbillo, L.L. Philippine performance evaluation and assessment scheme (PPEAS): Experiences in newborn screening system quality improvement. Int. J. Neonatal Screen. 2020, 6, 95. [Google Scholar] [CrossRef] [PubMed]
- Maceda, E.B.G.; Abadingo, M.E.; Panol, K.A.R.; Beltran, F.D.E.; Valdez-Acosta, I.R.C.; Taquiqui, G.D.; Gawigawen, S.B.; Macalino, M.V.L.; Aguirre-Aguinaldo, L.M.S.M.; Flores-Declaro, M.A.; et al. Newborn screening long-term follow-up clinics (continuity clinics) in the Philippines during the COVID-19 pandemic: Continuing quality patient care. Int. J. Neonatal Screen. 2022, 9, 2. [Google Scholar] [CrossRef] [PubMed]
- Del Mundo, M.P.F.A.; Feliciano, A.I.C.; Habaluyas, K.E.R.; Lirio, A.E.; Santiago, D.K.A.; Young, A.T. Predictors of newborn screening utilization in Quezon City, Philippines. Int. J. Health Wellness Soc. 2020, 10, 61–76. [Google Scholar] [CrossRef]
- Padilla, P.J.D.; Manalo, E.M. Newborn screening knowledge, attitudes and practices among obstetrics-gynecology residents, pediatric residents, and newborn screening nurses in a tertiary government hospital in the Philippines during the COVID-19 pandemic. Int. J. Neonatal Screen. 2023, 9, 19. [Google Scholar] [CrossRef] [PubMed]
- Padilla, C.D.; Abadingo, M.E.; Munda, K.V.; Therrell, B.L. Overcoming challenges in sustaining newborn screening: The Philippine Newborn Screening System. Rare Dis. Orphan Drugs J. 2023, 2, 27. [Google Scholar] [CrossRef]
- Rajadurai, V.S.; Yip, W.Y.; Lim, J.S.C.; Khoo, P.C.; Tan, E.S.; Mahadev, A.; Joseph, R. Evolution and expansion of newborn screening programmes in Singapore. Singap. Med. J. 2021, 62 (Suppl. S1), S26–S35. [Google Scholar] [CrossRef]
- Lau, C.S.; Joseph, R.; Aw, T.C. Screening for congenital hypothyroidism. Ann. Acad. Med. Singap. 2020, 49, 934–936. [Google Scholar] [CrossRef] [PubMed]
- Tan, K.M.L.; Chu, A.H.Y.; Loy, S.L.; Rajadurai, V.S.; Ho, C.K.M.; Chong, Y.S.; Karnani, N.; Lee, Y.S.; Yap, F.K.P.; Chan, S.Y. Association of cord blood thyroid-stimulating hormone levels with maternal, delivery and infant factors. Ann. Acad. Med. Singap. 2020, 49, 937–947. [Google Scholar] [CrossRef] [PubMed]
- Chan, S.B.; Zhong, Y.; Lim, S.C.J.; Poh, S.; The, K.L.; Soh, J.Y.; Chong, C.Y.; Thoon, K.C.; Seng, M.; Tan, E.S.; et al. Implementation of universal newborn screening for severe combined immunodeficiency in Singapore while continuing routine Bacille-Calmette-Guerin vaccination given at birth. Front. Immunol. 2022, 12, 794221. [Google Scholar] [CrossRef] [PubMed]
- Family Health Bureau. National Strategic Plan, Maternal and Newborn Health, 2017–2025; Family Health Bureau, Ministry of Health: Colombo, Sri Lanka, 2017; pp. 26, 37, 62.
- Amarasena, S.; Hettiarachchi, M. Establishment of National programme on newborn screening for congenital hypothyroidism. Sri Lanka J. Diab. Endocrinol. Metabol. 2018, 8, 13–18. [Google Scholar] [CrossRef]
- Karunarathna, N.; Hettiarachchi, M. Cost-effective analysis of the congenital hypothyroidism screening program in Sri Lanka. Value Health Reg. Issues 2021, 24, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Seneviratne, S.N.; Sandakelum, U.; Jayawardena, C.H.; Weerasinghe, A.M.; Wickramarachchi, P.S.; de Silva, S. Presenting status of children with classical congenital adrenal hyperplasia over two decades (1999–2018) in the absence of newborn screening in Sri Lanka. J. Pediatr. Endocrinol. Metab. 2021, 34, 1131–1137. [Google Scholar] [CrossRef] [PubMed]
- Chien, Y.H.; Hwu, W.L.; Lee, N.C. Newborn screening: Taiwanese experience. Ann. Transl. Med. 2019, 7, 281. [Google Scholar] [CrossRef] [PubMed]
- Hwu, W.L.; Chien, Y.H. Development of newborn screening for Pompe Disease. Int. J. Neonatal Screen. 2020, 6, 5. [Google Scholar] [CrossRef] [PubMed]
- Chiang, S.C.; Chien, Y.H.; Chang, K.L.; Lee, C.; Hwu, W.L. The timely needs for infantile onset Pompe disease newborn screening-practice in Taiwan. Int. J. Neonatal Screen. 2020, 6, 30. [Google Scholar] [CrossRef] [PubMed]
- Lee, N.C.; Chang, K.L.; In’t Groen, S.L.M.; de Faria, D.O.S.; Huang, H.J.; Pijnappel, W.W.M.P.; Hwu, W.L.; Chien, Y.H. Outcome of later-onset Pompe disease identified through newborn screening. J. Pediatr. 2022, 244, 139–147.e2. [Google Scholar] [CrossRef] [PubMed]
- Chien, Y.H.; Lee, N.C.; Chen, P.W.; Yeh, H.Y.; Gelb, M.H.; Chiu, P.C.; Chu, S.Y.; Lee, C.H.; Lee, A.R.; Hwu, W.L. Newborn screening for Morquio disease and other lysosomal storage diseases: Results from the 8-plex assay for 70,000 newborns. Orphanet J. Rare Dis. 2020, 15, 38. [Google Scholar] [CrossRef] [PubMed]
- Chan, M.J.; Liao, H.C.; Gelb, M.H.; Chuang, C.K.; Liu, M.Y.; Chen, H.J.; Kao, S.M.; Lin, H.Y.; Huang, Y.H.; Kumar, A.B.; et al. Taiwan national newborn screening program by tandem mass spectrometry for mucopolysaccharidoses types I, II, and VI. J. Pediatr. 2019, 205, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.Y.; Chang, Y.H.; Lee, C.L.; Tu, Y.R.; Lo, Y.T.; Hung, P.W.; Niu, D.M.; Liu, M.Y.; Liu, H.Y.; Chen, H.J.; et al. Newborn screening program for mucopolysaccharidosis type II and long-term follow-up of the screen-positive subjects in Taiwan. J. Pers. Med. 2022, 12, 1023. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.A.; Hsu, R.H.; Chen, P.W.; Lee, N.C.; Chiu, P.C.; Hwu, W.L.; Chien, Y.H. High incidence of null variants identified from newborn screening of X-linked adrenoleukodystrophy in Taiwan. Mol. Genet. Metab. Rep. 2022, 32, 100902. [Google Scholar] [CrossRef] [PubMed]
- Chien, Y.H.; Lee, N.C.; Weng, W.C.; Chen, L.C.; Huang, Y.H.; Wu, C.S.; Hwu, W.L. Duchenne muscular dystrophy newborn screening: The first 50,000 newborns screened in Taiwan. Neurol. Sci. 2022, 43, 4563–4566. [Google Scholar] [CrossRef] [PubMed]
- Weng, W.C.; Hsu, Y.K.; Chang, F.M.; Lin, C.Y.; Hwu, W.L.; Lee, W.T.; Lee, N.C.; Chien, Y.H. CMAP changes upon symptom onset and during treatment in spinal muscular atrophy patients: Lessons learned from newborn screening. Genet. Med. 2021, 23, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.A.; Hsu, R.H.; Chen, Y.H.; Hsu, L.W.; Chiang, S.C.; Lee, N.C.; Hwu, W.L.; Chiu, P.C.; Chien, Y.H. Improved diagnosis of citrin deficiency by newborn screening using a molecular second-tier test. Mol. Genet. Metab. 2022, 136, 330–336. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-Y.; Chang, M.-H.; Chen, H.-L.; Chien, Y.-H.; Wu, J.-F. The prognosis of citrin deficiency differs between early-identified newborn and later-onset symptomatic infants. Pediatr. Res. 2023, 94, 1151–1157. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.A.; Hsu, R.H.; Chang, K.L.; Huang, Y.C.; Chiang, Y.C.; Lee, N.C.; Hwu, W.L.; Chiu, P.C.; Chien, Y.H. Asymptomatic ASS1 carriers with high blood citrulline levels. Mol. Genet. Genomic Med. 2022, 10, e2007. [Google Scholar] [CrossRef] [PubMed]
- Chien, Y.H.; Lee, N.C.; Hwu, W.L.; Yen, T.A.; Chua, H.H.; Chen, Y.J.; Wang, Y.L.; Chang, M.H. A pilot study of biliary atresia newborn screening using dried blood spot matrix metalloproteinase-7. J. Pediatr. Gastroenterol. Nutr. 2023, 76, 418–423. [Google Scholar] [CrossRef] [PubMed]
- Pangkanon, S.; Ratrisawadi, V.; Charoensiriwatana, W.; Techasena, W.; Boonpuan, K.; Srisomsap, C.; Svasti, J. Phenylketonuria detected by the neonatal screening program in Thailand. Southeast Asian J. Trop. Med. Public Health 2003, 34 (Suppl. S3), 179–181. [Google Scholar] [PubMed]
- CTN News. Thailand’s Free Newborn Screening for 24 Rare Conditions. 25 August 2023. Available online: https://www.chiangraitimes.com/news/thailands-free-newborn-screening-for-24-rare-conditions/?amp=1 (accessed on 8 November 2023).
- Wattanasirichaigoon, D. (Mahidol University, Bangkok, Thailand). Personal communication, 2023.
- Jaruratanasirikul, S.; Piriyaphan, J.; Saengkaew, T.; Janjindamai, W.; Sriplung, H. The etiologies and incidences of congenital hypothyroidism before and after neonatal TSH screening program implementation: A study in southern Thailand. J. Pediatr. Endocrinol. Metab. 2018, 31, 609–617. [Google Scholar] [CrossRef] [PubMed]
- Liammongkolkul, S.; Sanomcham, K.; Vatanavicharn, N.; Sathienkijkanchai, A.; Ranieri, E.; Wasant, P. Expanded newborn screening program in Thailand. Ann. Transl. Med. 2017, 5 (Suppl. S2), AB133. [Google Scholar] [CrossRef]
- Wilaiwongsathien, K.; Wattanasirichaigoon, D.; Rattanasiri, S.; Aonnuam, C.; Tangshewinsirikul, C.; Tim-Aroon, T. Parental awareness, knowledge, and attitudes regarding current and future newborn bloodspot screening: The first report from Thailand. Int. J. Neonatal Screen. 2023, 9, 25. [Google Scholar] [CrossRef] [PubMed]
- Family Medical Practice Vietnam. Baby’s First Test. Available online: https://fmp.bliss.build/hcmc/en/news/babys-first-test (accessed on 15 November 2023).
- Vietnam Plus. Prenatal, Newborn Screening Programme Helps Improve Population Quality. 26 March 2023. Available online: https://en.vietnamplus.vn/prenatal-newborn-screening-programme-helps-improve-population-quality/250241.vnp (accessed on 20 June 2023).
- Ho, V.H.; Reinharz, D. Expected benefits and challenges of using economic evaluations to make decisions about the content of newborn screening programs in Vietnam: A scoping review of the literature. J. Inborn Errors Metab. Screen. 2023, 11, e20220011. [Google Scholar] [CrossRef]
- Can, N.T.B.; Tran, D.M.; Bui, T.P.; Nguyen, K.N.; Nguyen, H.H.; Nguyen, T.V.; Hwu, W.L.; Tomatsu, S.; Vu, D.C. Molecular analysis of Vietnamese patients with mucopolysaccharidosis type I. Life 2021, 11, 1162. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, S.; Fukao, T.; Vu, D.C. Characterization and outcome of 41 patients with beta-ketothiolase deficiency: 10 years’ experience of a medical center in northern Vietnam. J. Inherit. Metab. Dis. 2017, 40, 395–401. [Google Scholar] [CrossRef]
- Loeber, J.G.; Platis, D.; Zetterström, R.H.; Almashanu, S.; Boemer, F.; Bonham, J.R.; Borde, P.; Brincat, I.; Cheillan, D.; Dekkers, E.; et al. Neonatal screening in Europe revisited: An ISNS perspective on the current state and developments since 2010. Int. J. Neonatal Screen. 2021, 7, 15. [Google Scholar] [CrossRef] [PubMed]
- Koracin, V.; Mlinaric, M.; Baric, I.; Brincat, I.; Djordjevic, M.; Drole Torkar, A.; Fumic, K.; Kocova, M.; Milenkovic, T.; Moldovanu, F.; et al. Current status of newborn screening in Southeastern Europe. Front. Pediatr. 2021, 9, 648939. [Google Scholar] [CrossRef] [PubMed]
- Sikonja, J.; Groselj, U.; Scarpa, M.; la Marca, G.; Cheillan, D.; Kölker, S.; Zetterström, R.H.; Kožich, V.; Le Cam, Y.; Gumus, G.; et al. Towards achieving equity and innovation in newborn screening across Europe. Int. J. Neonatal Screen. 2022, 8, 31. [Google Scholar] [CrossRef] [PubMed]
- Bussé, A.M.L.; Mackey, A.R.; Hoeve, H.L.J.; Goedegebure, A.; Carr, G.; Uhlén, I.M.; Simonsz, H.J.; EUS€REEN Foundation. Assessment of hearing screening programmes across 47 countries or regions I: Provision of newborn hearing screening. Int. J. Audiol. 2021, 60, 821–830. [Google Scholar] [CrossRef]
- Mackey, A.R.; Bussé, A.M.L.; Hoeve, H.L.J.; Goedegebure, A.; Carr, G.; Simonsz, H.J.; Uhlén, I.M.; EUS€REEN Foundation. Assessment of hearing screening programmes across 47 countries or regions II: Coverage, referral, follow-up and detection rates from newborn hearing screening. Int. J. Audiol. 2021, 60, 831–840. [Google Scholar] [CrossRef] [PubMed]
- Bussé, A.M.L.; Mackey, A.R.; Carr, G.; Hoeve, H.L.J.; Uhlén, I.M.; Goedegebure, A.; Simonsz, H.J.; EUS€REEN Foundation. Assessment of hearing screening programmes across 47 countries or regions III: Provision of childhood hearing screening after the newborn period. Int. J. Audiol. 2021, 60, 841–848. [Google Scholar] [CrossRef] [PubMed]
- Cornel, M.C.; Rigter, T.; Weinreich, S.S.; Burgard, P.; Hoffmann, G.F.; Lindner, M.; Loeber, J.G.; Rupp, K.; Taruscio, D.; Vittozzi, L. A framework to start the debate on neonatal screening policies in the EU: An Expert Opinion Document. Eur. J. Hum. Genet. 2014, 22, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Loeber, J.G.; Burgard, P.; Cornel, M.C.; Rigter, T.; Weinreich, S.S.; Rupp, K.; Hoffmann, G.F.; Vittozzi, L. Newborn screening programmes in Europe; arguments and efforts regarding harmonization. Part 1. From blood spot to screening result. J. Inherit. Metab. Dis. 2012, 35, 603–611. [Google Scholar] [CrossRef] [PubMed]
- Burgard, P.; Rupp, K.; Lindner, M.; Haege, G.; Rigter, T.; Weinreich, S.S.; Loeber, J.G.; Taruscio, D.; Vittozzi, L.; Cornel, M.C.; et al. Newborn screening programmes in Europe; arguments and efforts regarding harmonization. Part 2. From screening laboratory results to treatment, follow-up and quality assurance. J. Inherit. Metab. Dis. 2012, 35, 613–625. [Google Scholar] [CrossRef] [PubMed]
- Aymé, S.; Rodwell, C. The European Union Committee of Experts on Rare Diseases: Three productive years at the service of the rare disease community. Orphanet J. Rare Dis. 2014, 9, 30. [Google Scholar] [CrossRef] [PubMed]
- EUCERD—European Union Committee of Experts on Rare Diseases. EUCERD Opinion on Potential Areas of European Collaboration in the Field of New Born Screening—July 2013. Available online: http://www.rd-action.eu/eucerd/RDACTION_EUCERDARCHIVE.html (accessed on 26 March 2023).
- Loeber, J.G. European Union should actively stimulate and harmonise neonatal screening initiatives. Int. J. Neonatal Screen. 2018, 4, 32. [Google Scholar] [CrossRef] [PubMed]
- EURORDIS Rare Diseases Newborn Screening. Key Principles for Newborn Screening. A EURORDIS Position Paper. January 2021. Available online: http://eurordis.org/newbornscreening (accessed on 26 March 2023).
- Screen4Rare. Call to Action: Newborn Screening for Rare Diseases. Available online: https://screen4rare.org/calltoaction/ (accessed on 26 March 2023).
- Scarpa, M.; Bonham, J.R.; Dionisi-Vici, C.; Prevot, J.; Pergent, M.; Meyts, I.; Mahlaoui, N.; Schielen, P.C. Newborn screening as a fully integrated system to stimulate equity in neonatal screening in Europe. Lancet Reg. Health Eur. 2022, 13, 100311. [Google Scholar] [CrossRef]
- Screen4Rare. Adherence Principles. Available online: https://screen4rare.org/wp-content/uploads/2023/10/Adherence-Principles-Screen4Rare-2023.pdf) (accessed on 27 December 2023).
- MetabERN European Reference Network for Hereditary Metabolic Disorders. Rare Diseases Events under the Czech EU Council Presidency. 23 July 2022. Available online: https://metab.ern-net.eu/rare-diseases-events-under-the-czech-eu-council-presidency/ (accessed on 26 March 2023).
- Zerjav Tansek, M.; Groselj, U.; Angelkova, N.; Anton, D.; Baric, I.; Djordjevic, M.; Grimci, L.; Ivanova, M.; Kadam, A.; Kotori, V.; et al. Phenylketonuria screening and management in southeastern Europe—Survey results from 11 countries. Orphanet J. Rare Dis. 2015, 10, 68. [Google Scholar] [CrossRef] [PubMed]
- IJzebrink, A.; van Dijk, T.; Franková, V.; Loeber, G.; Kožich, V.; Henneman, L.; Jansen, M. Informing Parents about newborn screening: A European comparison study. Int. J. Neonatal Screen. 2021, 7, 13. [Google Scholar] [CrossRef]
- Franková, V.; Driscoll, R.O.; Jansen, M.E.; Loeber, J.G.; Kožich, V.; Bonham, J.; Borde, P.; Brincat, I.; Cheillan, D.; Dekkers, E.; et al. Regulatory landscape of providing information on newborn screening to parents across Europe. Eur. J. Hum. Genet. 2021, 29, 67–78. [Google Scholar] [CrossRef] [PubMed]
- Burlina, A.; Jones, S.A.; Chakrapani, A.; Church, H.J.; Heales, S.; Wu, T.H.Y.; Morton, G.; Roberts, P.; Sluys, E.F.; Cheillan, D. A new approach to objectively evaluate inherited metabolic diseases for inclusion on newborn screening programmes. Int. J. Neonatal Screen. 2022, 8, 25. [Google Scholar] [CrossRef] [PubMed]
- Jones, S.A.; Cheillan, D.; Chakrapani, A.; Church, H.J.; Heales, S.; Wu, T.H.Y.; Morton, G.; Roberts, P.; Sluys, E.F.; Burlina, A. Application of a novel algorithm for expanding newborn screening for inherited metabolic disorders across Europe. Int. J. Neonatal Screen. 2022, 8, 20. [Google Scholar] [CrossRef] [PubMed]
- Maase, R.; Jansen, M.E.; Heijnen, M.-L.; Dekkers, E. Comment on Jones et al. Application of a novel algorithm for expanding newborn screening for inherited metabolic disorders across Europe. Int. J. Neonatal Screen. 2022, 8, 20, Comment in Int. J. Neonatal Screen. 2023, 9, 7. [Google Scholar] [CrossRef] [PubMed]
- Jones, S.A.; Cheillan, D.; Chakrapani, A.; Church, H.J.; Heales, S.; Wu, T.H.Y.; Morton, G.; Roberts, P.; Sluys, E.F.; Burlina, A. Reply to Maase et al. Comment on “Jones et al. Application of a novel algorithm for expanding newborn screening for inherited metabolic disorders across Europe. Int. J. Neonatal Screen. 2022, 8, 20, Erratum in Int. J. Neonatal Screen. 2023, 9, 8. [Google Scholar] [CrossRef] [PubMed]
- Dangouloff, T.; Burghes, A.; Tizzano, E.F.; Servais, L.; NBS SMA Study Group. 244th ENMC international workshop: Newborn screening in spinal muscular atrophy May 10-12, 2019, Hoofdorp, The Netherlands. Neuromuscul Disord. 2020, 30, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Dangouloff, T.; Vrščaj, E.; Servais, L.; Osredkar, D.; SMA NBS World Study Group. Newborn screening programs for spinal muscular atrophy worldwide: Where we stand and where to go. Neuromuscul. Disord. 2021, 31, 574–582. [Google Scholar] [CrossRef]
- SMA NBS Alliance. European Alliance for Newborn Screening in Spinal Muscular Atrophy. Available online: https://www.sma-screening-alliance.org/ (accessed on 26 March 2023).
- Hörster, F.; Kölker, S.; Loeber, J.G.; Cornel, M.C.; Hoffmann, G.F.; Burgard, P. Newborn screening programmes in Europe, arguments and efforts regarding harmonisation: Focus on organic acidurias. JIMD Rep. 2017, 32, 105–115. [Google Scholar] [CrossRef] [PubMed]
- Lobitz, S.; Telfer, P.; Cela, E.; Allaf, B.; Angastiniotis, M.; Backman Johansson, C.; Badens, C.; Bento, C.; Bouva, M.J.; Canatan, D.; et al. Newborn screening for sickle cell disease in Europe: Recommendations from a Pan-European Consensus Conference. Br. J. Haematol. 2018, 183, 648–660. [Google Scholar] [CrossRef] [PubMed]
- Daniel, Y.; Elion, J.; Allaf, B.; Badens, C.; Bouva, M.J.; Brincat, I.; Cela, E.; Coppinger, C.; de Montalembert, M.; Gulbis, B.; et al. Newborn screening for sickle cell disease in Europe. Int. J. Neonatal Screen. 2019, 5, 15. [Google Scholar] [CrossRef]
- Barben, J.; Castellani, C.; Dankert-Roelse, J.; Gartner, S.; Kashirskaya, N.; Linnane, B.; Mayell, S.; Munck, A.; Sands, D.; Sommerburg, O.; et al. The expansion and performance of national newborn screening programmes for cystic fibrosis in Europe. J. Cyst. Fibros. 2017, 16, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Sommerburg, O.; Hammermann, J. Pancreatitis-associated protein in neonatal screening for cystic fibrosis: Strengths and weaknesses. Int. J. Neonatal Screen. 2020, 6, 28. [Google Scholar] [CrossRef] [PubMed]
- Munck, A.; Berger, D.O.; Southern, K.W.; Carducci, C.; de Winter-de Groot, K.M.; Gartner, S.; Kashirskaya, N.; Linnane, B.; Proesmans, M.; Sands, D.; et al. European survey of newborn bloodspot screening for CF: Opportunity to address challenges and improve performance. J. Cyst. Fibros. 2023, 22, 484–495. [Google Scholar] [CrossRef] [PubMed]
- Naehrlich, L. The changing face of cystic fibrosis and its implications for screening. Int. J. Neonatal Screen. 2020, 6, 54. [Google Scholar] [CrossRef]
- Chudleigh, J.; Ren, C.L.; Barben, J.; Southern, K.W. International approaches for delivery of positive newborn bloodspot screening results for CF. J. Cyst. Fibros. 2019, 18, 614–621. [Google Scholar] [CrossRef]
- Munck, A.; Southern, K.W.; Murphy, J.; de Winter-de Groot, K.M.; Gartner, S.; Karadag, B.; Kashirskaya, N.; Linnane, B.; Proesmans, M.; Sands, D.; et al. Cystic Fibrosis Cases Missed by Newborn Bloodspot Screening—Towards a Consistent Definition and Data Acquisition. Int. J. Neonatal Screen. 2023, 9, 65. [Google Scholar] [CrossRef] [PubMed]
- Munck, A.; Southern, K.W.; Castellani, C.; de Winter-de Groot, K.M.; Gartner, S.; Kashirskaya, N.; Linnane, B.; Mayell, S.J.; Proesmans, M.; Sands, D.; et al. Defining key outcomes to evaluate performance of newborn screening programmes for cystic fibrosis. J. Cyst. Fibros. 2021, 20, 820–823. [Google Scholar] [CrossRef] [PubMed]
- Cornel, M.C.; Rigter, T.; Jansen, M.E.; Henneman, L. Neonatal and carrier screening for rare diseases: How innovation challenges screening criteria worldwide. J. Community Genet. 2021, 12, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Kölker, S.; Gleich, F.; Mütze, U.; Opladen, T. Rare disease registries are key to evidence-based personalized medicine: Highlighting the European experience. Front. Endocrinol. 2022, 13, 832063. [Google Scholar] [CrossRef] [PubMed]
- Tsagkaris, C.; Eleftheriades, A.; Moysidis, D.V.; Papazoglou, A.S.; Loudovikou, A.; Panagiotopoulos, D.; Christodoulaki, C.; Panagopoulos, P. Migration and newborn screening: Time to build on the European Asylum, Integration and Migration Fund? Eur. J. Contracept. Reprod. Health Care 2022, 27, 431–435. [Google Scholar] [CrossRef] [PubMed]
- Kouřil, Š.; de Sousa, J.; Fačevicová, K.; Gardlo, A.; Muehlmann, C.; Nordhausen, K.; Friedecký, D.; Adam, T. Multivariate independent component analysis identifies patients in newborn screening equally to adjusted reference ranges. Int. J. Neonatal Screen. 2023, 9, 60. [Google Scholar] [CrossRef] [PubMed]
- Bussé, A.M.; Qirjazi, B.; Goedegebure, A.; Toll, M.; Hoeve, H.L.; Toçi, E.; Roshi, E.; Carr, G.; Simonsz, H.J. Implementation of a neonatal hearing screening programme in three provinces in Albania. Int. J. Pediatr. Otorhinolaryngol. 2020, 134, 110039. [Google Scholar] [CrossRef]
- Bartling, T. Infant Mortality and Nationwide Newborn Bloodspot Screening Programs in the European Union. Instituti I Shëndetit Publik. Available online: https://www.ishp.gov.al/infant-mortality-and-nationwide-newborn-bloodspot-screening-programs-in-the-european-union/ (accessed on 18 April 2023).
- Hutchings, N.; Tovmasyan, I.; Hovsepyan, M.; Qefoyan, M.; Baghdasaryan, S.; Bilezikian, J.P. Neonatal thyrotropin (TSH) screening as a tool for monitoring iodine nutrition in Armenia. Eur. J. Clin. Nutr. 2019, 73, 905–909. [Google Scholar] [CrossRef] [PubMed]
- Armenia News—NEWS.am. Parliament Speaker Briefs Turkmenistan Delegation on Newborn Screening in Armenia. 28 November 2018. Available online: https://news.am/eng/news/483393.html (accessed on 18 April 2023).
- Ramoser, G.; Caferri, F.; Radlinger, B.; Brunner-Krainz, M.; Herbst, S.; Huemer, M.; Hufgard-Leitner, M.; Kircher, S.G.; Konstantopoulou, V.; Löscher, W.; et al. 100 years of inherited metabolic disorders in Austria—A national registry of minimal birth prevalence, diagnosis, and clinical outcome of inborn errors of metabolism in Austria between 1921 and 2021. J. Inherit. Metab. Dis. 2022, 45, 144–156. [Google Scholar] [CrossRef] [PubMed]
- Pollak, A.; Kasper, D.C. Austrian newborn screening program: A perspective of five decades. J. Perinat. Med. 2014, 42, 151–158. [Google Scholar] [CrossRef]
- Zeyda, M.; Schanzer, A.; Basek, P.; Bauer, V.; Eber, E.; Ellemunter, H.; Kallinger, M.; Riedler, J.; Thir, C.; Wadlegger, F.; et al. Cystic fibrosis newborn screening in Austria using PAP and the numeric product of PAP and IRT concentrations as second-tier parameters. Diagnostics 2021, 11, 299. [Google Scholar] [CrossRef] [PubMed]
- Medical University of Vienna. Neonatal Screening: Programme Successfully Expanded—Screening for Spinal Muscular Atrophy (SMA) and Congenital Immunodeficiencies (SCID). 27 June 2022. Available online: https://www.meduniwien.ac.at/web/en/about-us/news/2022/news-in-june-2022/neugeborenen-screening-erweiterung-erfolgreich-umgesetzt/ (accessed on 6 April 2023).
- Zeinalzadeh, A.H.; Talebi, M. Neonatal screening for congenital hypothyroidism in East Azerbaijan, Iran: The first report. J. Med. Screen. 2012, 19, 123–126. [Google Scholar] [CrossRef] [PubMed]
- Babaev, M.S.; Aliyeva, K.A.; Mammadova, R.F. Genetic screening of newborns in Baku for hereditary hemoglobinopathies. Med. Genet. 2020, 19, 56–60. [Google Scholar]
- Hajiyeva, N. GALT enzyme deficiency identification in newborns in Azerbaijan. Bull. Sci. Pract. 2022, 8, 284–291. Available online: https://cyberleninka.ru/article/n/galt-enzyme-deficiency-identification-in-newborns-in-azerbaijan/viewer (accessed on 3 February 2024).
- UNICEF. Country Office Annual Report 2022—Azerbaijan. Available online: https://www.unicef.org/media/135431/file/Aberbaijan-2022-COAR.pdf (accessed on 2 January 2024).
- Eyskens, F.; Sylvie Devos, S. Newborn screening for lysosomal storage disorders in Belgium. The importance of sex- and age-dependent reference ranges. J. Inborn Errors Metab. Screen. 2017, 5, 2326409817744231. [Google Scholar] [CrossRef]
- Boemer, F.; Caberg, J.-H.; Dideberg, V.; Dardenne, D.; Bours, V.; Hiligsmann, M.; Dangouloff, T.; Servais, L. Newborn screening for SMA in Southern Belgium. Neuromuscul. Disord. 2019, 29, 343–349. [Google Scholar] [CrossRef] [PubMed]
- Boemer, F.; Caberg, J.H.; Beckers, P.; Dideberg, V.; di Fiore, S.; Bours, V.; Marie, S.; Dewulf, J.; Marcelis, L.; Deconinck, N.; et al. Three years pilot of spinal muscular atrophy newborn screening turned into official program in Southern Belgium. Sci. Rep. 2021, 11, 19922. [Google Scholar] [CrossRef]
- Dangouloff, T.; Thokala, P.; Stevenson, M.D.; Deconinck, N.; D’Amico, A.; Daron, A.; Delstanche, S.; Servais, L.; Hiligsmann, M. Cost-effectiveness of spinal muscular atrophy newborn screening based on real-world data in Belgium. Neuromuscul. Disord. 2024, 34, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Gulbis, B.; Lê, P.-Q.; Ketelslegers, O.; Dresse, M.-F.; Adam, A.-S.; Cotton, F.; Boemer, F.; Bours, V.; Minon, J.-M.; Ferster, A. Neonatal screening for sickle cell disease in Belgium for more than 20 Years: An experience for comprehensive care improvement. Int. J. Neonatal Screen. 2018, 4, 37. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.; Werbrouck, A.; Verhaeghe, N.; De Wachter, E.; Simoens, S.; Annemans, L.; Putman, K. A model-based economic evaluation of four newborn screening strategies for cystic fibrosis in Flanders, Belgium. Acta Clin. Belg. 2020, 75, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Dangouloff, T.; Boemer, F.; Servais, L. Newborn screening of neuromuscular diseases. Neuromuscul. Disord. 2021, 31, 1070–1080. [Google Scholar] [CrossRef] [PubMed]
- Iskrov, G.; Ivanov, S.; Wrenn, S.; Stefanov, R. Whole-genome sequencing in newborn screening-attitudes and opinions of Bulgarian pediatricians and geneticists. Front. Public Health 2017, 5, 308. [Google Scholar] [CrossRef] [PubMed]
- Marinova, M.; Georgyeva, A.; Yordanova, V.; Ivanov, N.; Atanasova, V.; Naumova, E.; Kandilarova, S.M. Implementation of TREC/KREC detection protocol for newborn SCID screening in Bulgaria: A pilot study. Cent. Eur. J. Immunol. 2022, 47, 339–349. [Google Scholar] [CrossRef] [PubMed]
- Atemin, S.; Todorov, T.; Tourtourikov, I.; Ivanova, M.B.; Chamova, T.; Avdjieva-Tzavella, D.; Kathom, H.; Georgieva, B.; Guergueltcheva, V.; Savov, I.B.A.; et al. Arginase deficiency in Bulgaria: First cases and potential endemic region for the disorder. J. Genet. 2023, 102, 2. Available online: http://www.ias.ac.in/article/fulltext/jgen/102/0002 (accessed on 3 February 2024).
- Roque, B. Front Line Genomics. World of Genomics: Croatia. 1 February 2023. Available online: https://frontlinegenomics.com/world-of-genomics-croatia/ (accessed on 19 April 2023).
- N1 Newsnight. Croatia Introduces SMA Testing for Every Newborn. Available online: https://n1info.hr/english/news/croatia-introduces-sma-testing-for-every-newborn/ (accessed on 2 January 2024).
- Skordis, N.; Toumba, M.; Savva, S.C.; Erakleous, E.; Topouzi, M.; Vogazianos, M.; Argyriou, A. High prevalence of congenital hypothyroidism in the Greek Cypriot population: Results of the neonatal screening program 1990–2000. J. Pediatr. Endocrinol. Metab. 2005, 18, 453–461. [Google Scholar] [CrossRef] [PubMed]
- Kožich, V. Focus on National Experiences and Best Practices Czech Republic, Viktor Kožich. YouTube. 11 October 2021. Available online: https://www.youtube.com/watch?v=9MDNHzmcqzs (accessed on 19 April 2023).
- General University Hospital in Prague. Pilot Screening Reveals New Disease. 19 January 2022. Available online: https://www.vfn.cz/en/aktuality/pilotni-screening-odhaluje-nove-nemoci/ (accessed on 19 April 2023).
- David, J.; Chrastina, P.; Pešková, K.; Kožich, V.; Friedecký, D.; Adam, T.; Hlídková, E.; Vinohradská, H.; Novotná, D.; Hedelová, M.; et al. Epidemiology of rare diseases detected by newborn screening in the Czech Republic. Cent. Eur. J. Public Health 2019, 27, 153–159. [Google Scholar] [CrossRef] [PubMed]
- David, J.; Chrastina, P.; Vinohradska, H.; Al Taji, E.; Holubova, A.; Hlidkova, E.; Kožich, V.; Votava, F. Neonatal screening in the Czech Republic: Increased prevalence of selected diseases in low birthweight neonates. Eur. J. Pediatr. 2018, 177, 1697–1704. [Google Scholar] [CrossRef] [PubMed]
- David, J.; Hruba, Z.; Kolouskova, S.; Votava, F. Newborn screening-detected 21-hydroxylase deficiency: Growth pattern is not associated with the genotype. Minerva Pediatr. 2024, 76, 24–29. [Google Scholar] [CrossRef] [PubMed]
- Franková, V.; Dohnalová, A.; Pešková, K.; Hermánková, R.; O’Driscoll, R.; Ješina, P.; Kožich, V. Factors influencing parental awareness about newborn screening. Int. J. Neonatal Screen. 2019, 5, 35. [Google Scholar] [CrossRef] [PubMed]
- Lund, A.M.; Hougaard, D.M.; Simonsen, H.; Andresen, B.S.; Christensen, M.; Dunø, M.; Skogstrand, K.; Olsen, R.K.; Jensen, U.G.; Cohen, A.; et al. Biochemical screening of 504,049 newborns in Denmark, the Faroe Islands and Greenland--experience and development of a routine program for expanded newborn screening. Mol. Genet. Metab. 2012, 107, 281–293. [Google Scholar] [CrossRef] [PubMed]
- Lund, A.; Wibrand, F.; Skogstrand, K.; Cohen, A.; Christensen, M.; Jäpelt, R.B.; Dunø, M.; Skovby, F.; Nørgaard-Pedersen, B.; Gregersen, N.; et al. Danish expanded newborn screening is a successful preventive public health programme. Dan. Med. J. 2020, 67, A06190341. [Google Scholar] [PubMed]
- Skov, M.; Baekvad-Hansen, M.; Hougaard, D.M.; Skogstrand, K.; Lund, A.M.; Pressler, T.; Olesen, H.V.; Duno, M. Cystic fibrosis newborn screening in Denmark: Experience from the first 2 years. Pediatr. Pulmonol. 2020, 55, 549–555. [Google Scholar] [CrossRef] [PubMed]
- Bækvad-Hansen, M.; Adamsen, D.; Bybjerg-Grauholm, J.; Hougaard, D.M. Implementation of SCID screening in Denmark. Int. J. Neonatal Screen. 2021, 7, 54. [Google Scholar] [CrossRef] [PubMed]
- Skov, M.; Koch, A.; Duno, M. First case of cystic fibrosis in Greenland—Diagnosed by neonatal screening. J. Cyst. Fibros. 2020, 19, e14–e15. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez-Mateo, C.; Timonen, A.; Vaahtera, K.; Jaakkola, M.; Hougaard, D.M.; Bybjerg-Grauholm, J.; Baekvad-Hansen, M.; Adamsen, D.; Filippov, G.; Dallaire, S.; et al. Development of a multiplex real-time PCR assay for the newborn screening of SCID, SMA, and XLA. Int. J. Neonatal Screen. 2019, 5, 39. [Google Scholar] [CrossRef] [PubMed]
- Cohen, A.S.; Baurek, M.; Lund, A.M.; Dunø, M.; Hougaard, D.M. Including classical galactosaemia in the expanded newborn screening panel using tandem mass spectrometry for galactose-1-phosphate. Int. J. Neonatal Screen. 2019, 5, 19. [Google Scholar] [CrossRef] [PubMed]
- Lund, A.M.; Wibrand, F.; Skogstrand, K.; Bækvad-Hansen, M.; Gregersen, N.; Andresen, B.S.; Hougaard, D.M.; Dunø, M.; Olsen, R.K.J. Use of molecular genetic analyses in Danish routine newborn screening. Int. J. Neonatal Screen. 2021, 7, 50. [Google Scholar] [CrossRef] [PubMed]
- Lind-Holst, M.; Bækvad-Hansen, M.; Berglund, A.; Cohen, A.S.; Melgaard, L.; Skogstrand, K.; Dunø, M.; Main, K.M.; Hougaard, D.M.; Gravholt, C.H.; et al. Neonatal screening for congenital adrenal hyperplasia in Denmark: 10 years of experience. Horm. Res. Paediatr. 2022, 95, 35–42. [Google Scholar] [CrossRef]
- Nordfalk, F.; Ekstrøm, C.T. Newborn dried blood spot samples in Denmark: The hidden figures of secondary use and research participation. Eur. J. Hum. Genet. 2019, 27, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Hougaard, D.M.; Bybjerg-Grauholm, J.; Christiansen, M.; Nørgaard-Pedersen, B. Response to “Newborn dried blood spot samples in Denmark: The hidden figures of secondary use and research participation”. Eur. J. Hum. Genet. 2019, 27, 1625–1627. [Google Scholar] [CrossRef] [PubMed]
- Nordfalk, F.; Jensen, A.M.B. Mothering a population: How Danish mothers experience newborn dried blood spot samples and their considerations about re-use of samples for research purposes. Eur. J. Midwifery 2022, 6, 58. [Google Scholar] [CrossRef] [PubMed]
- Kjaergaard, A.D.; Sørensen, E.; Brügmann, A.; Petersen, E.R.B.; Brandslund, I.; Nordestgaard, B.G.; Jensen, G.B.; Skajaa, N.; Troelsen, F.S.; Fuglsang, C.H.; et al. A review of major Danish biobanks: Advantages and possibilities of health research in Denmark. Clin. Epidemiol. 2023, 15, 213–239. [Google Scholar] [CrossRef] [PubMed]
- Timonen, A.; Lloyd-Puryear, M.; Hougaard, D.M.; Meriö, L.; Mäkinen, P.; Laitala, V.; Pölönen, T.; Skogstrand, K.; Kennedy, A.; Airenne, S.; et al. Duchenne muscular dystrophy newborn screening: Evaluation of a new GSP® Neonatal Creatine Kinase-MM Kit in a US and Danish population. Int. J. Neonatal Screen. 2019, 5, 27. [Google Scholar] [CrossRef] [PubMed]
- Courraud, J.; Ernst, M.; Svane Laursen, S.; Hougaard, D.M.; Cohen, A.S. Studying autism using untargeted metabolomics in newborn screening samples. J. Mol. Neurosci. 2021, 71, 1378–1393. [Google Scholar] [CrossRef] [PubMed]
- MacSween, N.; Courraud, J.; Nielsen, Z.K.; Hougaard, D.M.; Cohen, A.S.; Ernst, M. Effects of long-term storage on the biobanked neonatal dried blood spot Mmetabolome. J. Am. Soc. Mass Spectrom. 2023, 34, 685–694. [Google Scholar] [CrossRef] [PubMed]
- Reinson, K.; Universitas Tartuensis DSpace. New Diagnostic Methods for Early Detection of Inborn Errors of Metabolism in Estonia. 16 November 2018. Available online: https://dspace.ut.ee/handle/10062/62653 (accessed on 22 April 2023).
- Reinson, K.; Künnapas, K.; Kriisa, A.; Vals, A.; Muru, K.; Õunap, K. High incidence of low vitamin B12 levels in Estonian newborns. Mol. Genet. Metab. Rep. 2018, 15, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Lilleväli, H.; Reinson, K.; Muru, K.; Simenson, K.; Murumets, Ü.; Möls, T.; Õunap, K. Hyperphenylalaninaemias in Estonia: Genotype-phenotype correlation and comparative overview of the patient cohort before and after nation-wide neonatal screening. JIMD Rep. 2018, 40, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Sarv, S.; Kahre, T.; Vaidla, E.; Pajusalu, S.; Muru, K.; Põder, H.; Gross-Paju, K.; Ütt, S.; Žordania, R.; Talvik, I.; et al. The birth prevalence of spinal muscular atrophy: A population specific approach in Estonia. Front. Genet. 2021, 12, 796862. [Google Scholar] [CrossRef] [PubMed]
- Tiivoja, E.; Reinson, K.; Muru, K.; Rähn, K.; Muhu, K.; Mauring, L.; Kahre, T.; Pajusalu, S.; Õunap, K. The prevalence of inherited metabolic disorders in Estonian population over 30 years: A significant increase during study period. JIMD Rep. 2022, 63, 604–613. [Google Scholar] [CrossRef] [PubMed]
- Palveluvalikoima. Summary of a Recommendation by Council for Choices in Health Care in Finland (COHERE Finland)—Screening for Severe Combined Immunodeficiency (SCID) Using a Heel Prick Test for Newborn Babies. Available online: https://palveluvalikoima.fi/documents/1237350/38358696/Tiivistelm%C3%A4_SCID+seulonta_en.pdf/a14177c3-2d0c-dcdd-cbca-2d1c202e4b65/Tiivistelm%C3%A4_SCID+seulonta_en.pdf?t=1602084321143 (accessed on 23 April 2023).
- Roche. SMA Screening for Newborns in Europe. 8 February 2022. Available online: https://www.roche.com/stories/sma-screening-for-newborns-in-europe/ (accessed on 23 April 2023).
- Äärelä, L.; Hiltunen, P.; Soini, T.; Vuorela, N.; Huhtala, H.; Nevalainen, P.I.; Heikinheimo, M.; Kivelä, L.; Kurppa, K. Type 1 tyrosinemia in Finland: A nationwide study. Orphanet J. Rare Dis. 2020, 15, 281. [Google Scholar] [CrossRef] [PubMed]
- Danner, E.; Niuro, L.; Huopio, H.; Niinikoski, H.; Viikari, L.; Kero, J.; Jääskeläinen, J. Incidence of primary congenital hypothyroidism over 24 years in Finland. Pediatr. Res. 2023, 93, 649–653. [Google Scholar] [CrossRef] [PubMed]
- Farriaux, J.P. Histoire et organisation du programme Français de dépistage néonatal systématiq (1967–2020) [History and organization of the French systematic neonatal screening program (1967–2020)]. Med. Sci. 2021, 37, 433–440. (In French) [Google Scholar] [CrossRef] [PubMed]
- Munck, A.; Gauthereau, V.; Czernichow, P. Organisation du dépistage néonatal en France [Organization of newborn screening in France]. Med. Sci. 2021, 37, 457–460. (In French) [Google Scholar] [CrossRef] [PubMed]
- Coutant, R.; Feillet, F. Présentation de l’état des lieux du dépistage néonatal en France [State of play of neonatal screening in France]. Med. Sci. 2018, 34, 19–21. (In French) [Google Scholar] [CrossRef] [PubMed]
- Haute Autorité de Santé. Évaluation a priori de l’extension du dépistage néonatal à une ou plusieurs erreurs innées du métabolisme par spectrométrie de masse en tandem. Volet 2. 3 February 2020. Available online: https://www.has-sante.fr/jcms/c_2866458/fr/evaluation-a-priori-de-l-extension-du-depistage-neonatal-a-une-ou-plusieurs-erreurs-innees-du-metabolisme-par-spectrometrie-de-masse-en-tandem-volet-2 (accessed on 23 April 2023).
- Wiedemann, A.; Jeannesson, É.; Oussalah, A.; Guéant, J.L.; Guéant-Rodriguez, R.M.; Feillet, F. Le dépistage de la phénylcétonurie en France [Newborn screening of phenylketonuria in France]. Med. Sci. 2021, 37, 468–473. (In French) [Google Scholar] [CrossRef] [PubMed]
- Kariyawasam, D.; Nguyen-Khoa, T.; Gonzalez Briceño, L.; Polak, M. Le dépistage néonatal de l’hyperplasie congénitale des glandes surrénales [Newborn screening for congenital adrenal hyperplasia in France]. Med. Sci. 2021, 37, 500–506. (In French) [Google Scholar] [CrossRef] [PubMed]
- Munck, A.; Cheillan, D.; Audrezet, M.P.; Guenet, D.; Huet, F. Dépistage néonatal de la mucoviscidose [Newborn screening for cystic fibrosis in France]. Med. Sci. 2021, 37, 491–499. (In French) [Google Scholar] [CrossRef] [PubMed]
- Audrézet, M.P.; Munck, A. Newborn screening for CF in France: An exemplary national experience. Arch. Pediatr. 2020, 27 (Suppl. S1), eS35–eS40. [Google Scholar] [CrossRef] [PubMed]
- Levaillant, L.; Huet, F.; Bretones, P.; Corne, C.; Dupuis, C.; Reynaud, R.; Somma, C.; Barat, P.; Corcuff, J.B.; Bouhours-Nouet, N.; et al. Neonatal screening for congenital hypothyroidism: Time to lower the TSH threshold in France. Arch. Pediatr. 2022, 29, 253–257. [Google Scholar] [CrossRef]
- Brousse, V.; Allaf, B.; Benkerrou, M. Dépistage néonatal de la drépanocytose en France [Newborn screening for sickle cell disease in France]. Med. Sci. 2021, 37, 482–490. (In French) [Google Scholar] [CrossRef] [PubMed]
- Pinel, J.; Bellanger, A.; Jamet, C.; Moreau, C. Information and Parental Consent for French Neonatal Screening: A Qualitative Study on Parental Opinion. Int. J. Neonatal Screen. 2023, 9, 26. [Google Scholar] [CrossRef] [PubMed]
- Raimond, V.; Sambuc, C.; Pibouleau, L. Ethics evaluation revealing decision-maker motives: A case of neonatal screening. Int. J. Technol. Assess. Health Care 2018, 34, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Munck, A.; Bourmaud, A.; Bellon, G.; Picq, P.; Farrell, P.M.; DPAM Study Group. Phenotype of children with inconclusive cystic fibrosis diagnosis after newborn screening. Pediatr. Pulmonol. 2020, 55, 918–928. [Google Scholar] [CrossRef]
- Bergougnoux, A.; Lopez, M.; Girodon, E. The role of extended CFTR gene sequencing in newborn screening for cystic fibrosis. Int. J. Neonatal Screen. 2020, 6, 23. [Google Scholar] [CrossRef] [PubMed]
- Hatton, A.; Bergougnoux, A.; Zybert, K.; Chevalier, B.; Mesbahi, M.; Altéri, J.P.; Walicka-Serzysko, K.; Postek, M.; Taulan-Cadars, M.; Edelman, A.; et al. Reclassifying inconclusive diagnosis after newborn screening for cystic fibrosis. Moving forward. J. Cyst. Fibros. 2022, 21, 448–455. [Google Scholar] [CrossRef] [PubMed]
- Bienvenu, T.; Lopez, M.; Girodon, E. Molecular diagosis and genetic counseling of cystic fibrosis and related disorders: New challenges. Genes 2020, 11, 619. [Google Scholar] [CrossRef] [PubMed]
- Munck, A.; Delmas, D.; Audrézet, M.P.; Lemonnier, L.; Cheillan, D.; Roussey, M. Optimization of the French cystic fibrosis newborn screening programme by a centralized tracking process. J. Med. Screen. 2018, 25, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Allaf, B.; Couque, N.; de Montalembert, M. Dépistage néonatal de la drépanocytose et filières d’organisation des soins [Newborn screening of sickle cell disease and management of care]. Rev. Prat. 2019, 69, 411–416. (In French) [Google Scholar]
- Allaf, B.; Patin, F.; Elion, J.; Couque, N. New approach to accurate interpretation of sickle cell disease newborn screening by applying multiple of median cutoffs and ratios. Pediatr. Blood Cancer 2018, 65, e27230. [Google Scholar] [CrossRef] [PubMed]
- Nguyen-Khoa, T.; Mine, L.; Allaf, B.; Ribeil, J.A.; Remus, C.; Stanislas, A.; Gauthereau, V.; Enouz, S.; Kim, J.S.; Yang, X.; et al. Sickle SCAN™ (BioMedomics) fulfills analytical conditions for neonatal screening of sickle cell disease. Ann. Biol. Clin. 2018, 76, 416–420. [Google Scholar] [CrossRef] [PubMed]
- Remion, J.; Pluchart, C. Targeted screening failures in neonates with sickle cell disease in metropolitan France from 2005 to 2017: Number of cases, failure circumstances and health at diagnosis. Arch. Pediatr. 2019, 26, 451–452. [Google Scholar] [CrossRef] [PubMed]
- Audrain, M.A.P.; Léger, A.J.C.; Hémont, C.A.F.; Mirallié, S.M.; Cheillan, D.; Rimbert, M.G.M.; Le Thuaut, A.M.; Sébille-Rivain, V.A.; Prat, A.; Pinel, E.M.Q.; et al. Newborn screening for severe combined immunodeficiency: Analytic and clinical performance of the T cell receptor excision circle assay in France (DEPISTREC Study). J. Clin. Immunol. 2018, 38, 778–786. [Google Scholar] [CrossRef] [PubMed]
- Thomas, C.; Hubert, G.; Catteau, A.; Danielo, M.; Riche, V.P.; Mahlaoui, N.; Moshous, D.; Audrain, M. Review: Why screen for severe combined immunodeficiency disease? Arch. Pediatr. 2020, 27, 485–489. [Google Scholar] [CrossRef]
- Audrain, M.; Thomas, C. Neonatal screening for SCID: The French experience. Int. J. Neonatal Screen. 2021, 7, 42. [Google Scholar] [CrossRef]
- Stinton, C.; Geppert, J.; Freeman, K.; Clarke, A.; Johnson, S.; Fraser, H.; Sutcliffe, P.; Taylor-Phillips, S. Newborn screening for tyrosinemia type 1 using succinylacetone—A systematic review of test accuracy. Orphanet J. Rare Dis. 2017, 12, 48. [Google Scholar] [CrossRef] [PubMed]
- Lefèvre, C.R.; Labarthe, F.; Dufour, D.; Moreau, C.; Faoucher, M.; Rollier, P.; Arnoux, J.B.; Tardieu, M.; Damaj, L.; Bendavid, C.; et al. Newborn screening of primary carnitine deficiency: An overview of worldwide practices and pitfalls to define an algorithm before expansion of newborn screening in France. Int. J. Neonatal Screen. 2023, 9, 6. [Google Scholar] [CrossRef] [PubMed]
- Dangouloff, T.; Hiligsmann, M.; Deconinck, N.; D’Amico, A.; Seferian, A.M.; Boemer, F.; Servais, L. Financial cost and quality of life of patients with spinal muscular atrophy identified by symptoms or newborn screening. Dev. Med. Child Neurol. 2023, 65, 67–77. [Google Scholar] [CrossRef] [PubMed]
- Francois-Bic, S.; Legagneur, C.; Hubert, C.; Guéant-Rodriguez, R.M.; Leheup, B.; Albuisson, E.; Renard, E. Maternal opioid addiction: A potential cause of elevated 17-OH progesterone in neonatal screening. Arch. Pediatr. 2023, 30, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Cheillan, D. Les principaux outils biologiques appliqués au dépistage néonatal—État des lieux et perspectives d’avenir [Main biological tools applied to newborn screening: Landscape and future perspectives]. Med. Sci. 2021, 37, 461–467. (In French) [Google Scholar] [CrossRef] [PubMed]
- Tönnies, H.; Nennstiel, U. Newborn screening in Germany. Med. Genet. 2022, 34, 1–2. [Google Scholar] [CrossRef]
- Rosenau, H.; Steffen, F. Legal aspects of newborn screening. Med. Genet. 2022, 34, 3–11. [Google Scholar] [CrossRef]
- Gramer, G.; Hoffmann, G.F. Second-tier strategies in newborn screening—Potential and limitations. Med. Genet. 2022, 34, 21–28. [Google Scholar] [CrossRef]
- Spiekerkoetter, U.; Krude, H. Target diseases for neonatal screening in Germany. Challenges for treatment and long-term care. Dtsch. Arztebl. Int. 2022, 119, 306–316. [Google Scholar] [CrossRef] [PubMed]
- Hammersen, J.; Bettendorf, M.; Bonfig, W.; Schönau, E.; Warncke, K.; Eckert, A.J.; Fricke-Otto, S.; Palm, K.; Holl, R.W.; Woelfle, J. Twenty years of newborn screening for congenital adrenal hyperplasia and congenital primary hypothyroidism—Experiences from the DGKED/AQUAPE study group for quality improvement in Germany. Med. Genet. 2022, 34, 29–40. [Google Scholar] [CrossRef]
- Dikow, N.; Ditzen, B.; Kölker, S.; Hoffmann, G.F.; Schaaf, C.P. From newborn screening to genomic medicine: Challenges and suggestions on how to incorporate genomic newborn screening in public health programs. Med. Genet. 2022, 34, 13–20. [Google Scholar] [CrossRef]
- Vill, K.; Schwartz, O.; Blaschek, A.; Gläser, D.; Nennstiel, U.; Wirth, B.; Burggraf, S.; Röschinger, W.; Becker, M.; Czibere, L.; et al. Newborn screening for spinal muscular atrophy in Germany: Clinical results after 2 years. Orphanet J. Rare Dis. 2021, 16, 153. [Google Scholar] [CrossRef] [PubMed]
- Müller-Felber, W.; Blaschek, A.; Schwartz, O.; Gläser, D.; Nennstiel, U.; Brockow, I.; Wirth, B.; Burggraf, S.; Röschinger, W.; Becker, M.; et al. Newborn screening SMA—From pilot project to nationwide screening in Germany. J. Neuromuscul. Dis. 2023, 10, 55–65. [Google Scholar] [CrossRef]
- Mueller-Felber, W. Newborn infant screening for spinal muscular atrophy: Chances and challenges. Dev. Med. Child Neurol. 2022, 64, 535. [Google Scholar] [CrossRef] [PubMed]
- Müller-Felber, W.; Vill, K.; Schwartz, O.; Gläser, D.; Nennstiel, U.; Wirth, B.; Burggraf, S.; Röschinger, W.; Becker, M.; Durner, J.; et al. Infants diagnosed with spinal muscular atrophy and 4 SMN2 copies through newborn screening—Opportunity or burden? J. Neuromuscul. Dis. 2020, 7, 109–117, Erratum in J. Neuromuscul. Dis. 2021, 8, 335–336. [Google Scholar] [CrossRef] [PubMed]
- Kölbel, H.; Modler, L.; Blaschek, A.; Schara-Schmidt, U.; Vill, K.; Schwartz, O.; Müller-Felber, W. Parental burden and quality of life in 5q-SMA diagnosed by newborn screening. Children 2022, 9, 1829. [Google Scholar] [CrossRef] [PubMed]
- Lobitz, S.; Kunz, J.B.; Cario, H.; Hakimeh, D.; Jarisch, A.; Kulozik, A.E.; Oevermann, L.; Grosse, R. Introduction of universal newborn screening for sickle cell disease in Germany—A brief narrative review. Int. J. Neonat. Screen. 2021, 7, 7. [Google Scholar] [CrossRef] [PubMed]
- Aramayo-Singelmann, C.; Halimeh, S.; Proske, P.; Vignalingarajah, A.; Cario, H.; Christensen, M.O.; Yamamoto, R.; Röth, A.; Reinhardt, D.; Reinhardt, H.C.; et al. Screening and diagnosis of hemoglobinopathies in Germany: Current state and future perspectives. Sci. Rep. 2022, 12, 9762. [Google Scholar] [CrossRef] [PubMed]
- Alashkar, F.; Aramayo-Singelmann, C.; Böll, J.; Hoferer, A.; Jarisch, A.; Kamal, H.; Oevermann, L.; Schwarz, M.; Cario, H. Transition in sickle cell disease (SCD): A German consensus recommendation. J. Pers. Med. 2022, 12, 1156. [Google Scholar] [CrossRef] [PubMed]
- Lobitz, S.; Frömmel, C.; Brose, A.; Blankenstein, O.; Turner, C.; Dalton, R.N.; Daniel, Y.; Klein, J. Simultaneous newborn screening for sickle cell disease, biotinidase deficiency, and hereditary tyrosinemia type 1 with an optimized tandem mass spectrometry protocol. Ann. Hematol. 2022, 101, 1859–1860. [Google Scholar] [CrossRef] [PubMed]
- Tesorero, R.; Janda, J.; Hörster, F.; Feyh, P.; Mütze, U.; Hauke, J.; Schwarz, K.; Kunz, J.B.; Hoffmann, G.F.; Okun, J.G. A high-throughput newborn screening approach for SCID, SMA, and SCD combining multiplex qPCR and tandem mass spectrometry. PLoS ONE 2023, 18, e0283024. [Google Scholar] [CrossRef] [PubMed]
- Speckmann, C.; Nennstiel, U.; Hönig, M.; Albert, M.H.; Ghosh, S.; Schuetz, C.; Brockow, I.; Hörster, F.; Niehues, T.; Ehl, S.F.; et al. Prospective newborn screening for SCID in Germany: A first analysis by the Pediatric Immunology Working Group (API). J. Clin. Immunol. 2023, 43, 965–978. [Google Scholar] [CrossRef] [PubMed]
- Sommerburg, O.; Stahl, M.; Hämmerling, S.; Gramer, G.; Muckenthaler, M.U.; Okun, J.; Kohlmüller, D.; Happich, M.; Kulozik, A.E.; Mall, M.A.; et al. Final results of the southwest German pilot study on cystic fibrosis newborn screening—Evaluation of an IRT/PAP protocol with IRT-dependent safety net. J. Cyst. Fibros. 2022, 21, 422–433. [Google Scholar] [CrossRef] [PubMed]
- Maier, P.; Jeyaweerasinkam, S.; Eberhard, J.; Soueidan, L.; Hämmerling, S.; Kohlmüller, D.; Feyh, P.; Gramer, G.; Garbade, S.F.; Hoffmann, G.F.; et al. Influence of season, storage temperature and time of sample collection in pancreatitis-associated protein-based algorithms for newborn screening for cystic fibrosis. Int. J. Neonatal Screen. 2024, 10, 5. [Google Scholar] [CrossRef] [PubMed]
- Schütz, K.; Kontsendorn, J.; Janzen, N.; Fuge, J.; Grewendorf, S.; Klemann, C.; Happle, C.; Junge, S.; Rudolf, I.; Dopfer, C.; et al. The first 4 years—Outcome of children identified by newborn screening for CF in Germany. Klin. Padiatr. 2022, 234, 284–292. [Google Scholar] [CrossRef] [PubMed]
- Gapp, S.; Garbade, S.F.; Feyh, P.; Brockow, I.; Nennstiel, U.; Hoffmann, G.F.; Sommerburg, O.; Gramer, G. German newborn screening for Cystic fibrosis: Parental perspectives and suggestions for improvements. Pediatr. Pulmonol. 2023, 58, 844–852. [Google Scholar] [CrossRef] [PubMed]
- Mütze, U.; Garbade, S.F.; Gramer, G.; Lindner, M.; Freisinger, P.; Grünert, S.C.; Hennermann, J.; Ensenauer, R.; Thimm, E.; Zirnbauer, J.; et al. Long-term outcomes of individuals with metabolic diseases identified through newborn screening. Pediatrics 2020, 146, e20200444. [Google Scholar] [CrossRef] [PubMed]
- Lüders, A.; Blankenstein, O.; Brockow, I.; Ensenauer, R.; Lindner, M.; Schulze, A.; Nennstiel, U. Screening Laboratories in Germany. Neonatal screening for congenital metabolic and endocrine disorders—Results from Germany for the years 2006–2018. Dtsch. Arztebl. Int. 2021, 118, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Oliva, P.; Mechtler, T.P.; Schwarz, M.; Streubel, B.; Chanson, C.; Essing, M.M.; Janzen, N.; Kasper, D.C. Newborn screening for metachromatic leukodystrophy in Northern Germany-a prospective study. Molec. Genet. Metab. 2022, 135, S91–S92. [Google Scholar]
- Hohenfellner, K.; Elenberg, E.; Ariceta, G.; Nesterova, G.; Soliman, N.A.; Topaloglu, R. Newborn screening: Review of its impact for cystinosis. Cells 2022, 11, 1109. [Google Scholar] [CrossRef] [PubMed]
- Gramer, G.; Fang-Hoffmann, J.; Feyh, P.; Klinke, G.; Monostori, P.; Mütze, U.; Posset, R.; Weiss, K.H.; Hoffmann, G.F.; Okun, J.G. Newborn screening for vitamin B12 deficiency in Germany—Strategies, results, and public health implications. J. Pediatr. 2020, 216, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Mütze, U.; Walter, M.; Keller, M.; Gramer, G.; Garbade, S.F.; Gleich, F.; Haas, D.; Posset, R.; Grünert, S.C.; Hennermann, J.B.; et al. Health outcomes of infants with vitamin B12 deficiency identified by newborn screening and early treated. J. Pediatr. 2021, 235, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Weiss, K.J.; Röschinger, W.; Blessing, H.; Lotz-Havla, A.S.; Schiergens, K.A.; Maier, E.M. Diagnostic challenges using a 2-tier strategy for methylmalonic acidurias: Data from 1.2 million dried blood spots. Ann. Nutr. Metab. 2020, 76, 268–276. [Google Scholar] [CrossRef] [PubMed]
- Maier, E.M.; Mütze, U.; Janzen, N.; Steuerwald, U.; Nennstiel, U.; Odenwald, B.; Schuhmann, E.; Lotz-Havla, A.S.; Weiss, K.J.; Hammersen, J.; et al. Collaborative evaluation study on 18 candidate diseases for newborn screening in 1.77 million samples. J. Inherit. Metab. Dis. 2023, 46, 043–1062. [Google Scholar] [CrossRef] [PubMed]
- Mütze, U.; Henze, L.; Gleich, F.; Lindner, M.; Grünert, S.C.; Spiekerkoetter, U.; Santer, R.; Blessing, H.; Thimm, E.; Ensenauer, R.; et al. Newborn screening and disease variants predict neurological outcome in isovaleric aciduria. J. Inherit. Metab. Dis. 2021, 44, 857–870. [Google Scholar] [CrossRef] [PubMed]
- Murko, S.; Aseman, A.D.; Reinhardt, F.; Gramer, G.; Okun, J.G.; Mütze, U.; Santer, R. Neonatal screening for isovaleric aciduria: Reducing the increasingly high false-positive rate in Germany. JIMD Rep. 2022, 64, 114–120. [Google Scholar] [CrossRef] [PubMed]
- Zaunseder, E.; Haupt, S.; Mütze, U.; Garbade, S.F.; Kölker, S.; Heuveline, V. Opportunities and challenges in machine learning-based newborn screening—A systematic literature review. JIMD Rep. 2022, 63, 250–261. [Google Scholar] [CrossRef] [PubMed]
- Zaunseder, E.; Mütze, U.; Garbade, S.F.; Haupt, S.; Feyh, P.; Hoffmann, G.F.; Heuveline, V.; Kölker, S. Machine learning methods improve specificity in newborn screening for isovaleric aciduria. Metabolites 2023, 13, 304. [Google Scholar] [CrossRef]
- Zimmer, K.P. Newborn screening: Still room for improvement. Dtsch. Arztebl. Int. 2021, 118, 99–100. [Google Scholar] [CrossRef] [PubMed]
- Odenwald, B.; Brockow, I.; Hanauer, M.; Lüders, A.; Nennstiel, U. Is our newborn screening working well? A literature review of quality requirements for newborn blood spot screening (NBS) infrastructure and procedures. Int. J. Neonatal Screen. 2023, 9, 35. [Google Scholar] [CrossRef]
- Loukas, Y.L.; Soumelas, G.S.; Dotsikas, Y.; Georgiou, V.; Molou, E.; Thodi, G.; Boutsini, M.; Biti, S.; Papadopoulos, K. Expanded newborn screening in Greece: 30 months of experience. J. Inherit. Metab. Dis. 2010, 33 (Suppl. S3), S341–S348. [Google Scholar] [CrossRef]
- Loukou, I.; Moustaki, M.; Douros, K. The psychological impact on parents of children who receive an inconclusive diagnosis for cystic fibrosis following newborn screening: A systematic mini-review. Children 2024, 11, 93. [Google Scholar] [CrossRef] [PubMed]
- Inside Precision Medicine. Genomics Data Players Unite to Power Greek Newborn Screening Project. 25 January 2023. Available online: https://www.insideprecisionmedicine.com/topics/molecular-dx-topic/genetic-disease-testing/genomics-data-players-unite-to-power-greek-newborn-screening-project/ (accessed on 28 April 2023).
- Kósa, M.; Galla, Z.; Lénárt, I.; Baráth, Á.; Grecsó, N.; Rácz, G.; Bereczki, C.; Monostori, P. Vitamin B12 (Cobalamin): Its fate from ingestion to metabolism with particular emphasis on diagnostic approaches of acquired neonatal/infantile deficiency detected by newborn screening. Metabolites 2022, 12, 1104. [Google Scholar] [CrossRef] [PubMed]
- Monostori, P.; Godejohann, M.; Janda, J.; Galla, Z.; Rácz, G.; Klinke, G.; Szatmári, I.; Zsidegh, P.; Kohlmüller, D.; Kölker, S.; et al. Identification of potential interferents of methylmalonic acid: A previously unrecognized pitfall in clinical diagnostics and newborn screening. Clin. Biochem. 2023, 111, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Grecsó, N.; Zádori, A.; Baráth, Á.; Galla, Z.; Rácz, G.; Bereczki, C.; Monostori, P. Comparison of different preparation techniques of dried blood spot quality controls in newborn screening for congenital adrenal hyperplasia. PLoS ONE 2021, 16, e0252091. [Google Scholar] [CrossRef] [PubMed]
- Bartos, B. The reform of the newborn screening policy: Spinal muscular atrophy. Issues Law Med. 2021, 36, 211–220. [Google Scholar] [PubMed]
- Marchadesch, L. Newborn Screening for Cystic Fibrosis in Iceland: A Pilot Study. Master’s Thesis, University of Iceland School of Health Sciences, Reykjavík, Iceland, 2023. [Google Scholar]
- Vidarsdottir, H.; Thorkelsson, T.; Halldorsson, T.I.; Bjarnason, R.; Geirsson, R.T.; Rinaldo, P.; Franzson, L. Does metabolomic profile differ with regard to birth weight? Pediatr. Res. 2021, 89, 1144–1151. [Google Scholar] [CrossRef] [PubMed]
- Vidarsdottir, H.; Halldorsson, T.I.; Geirsson, R.T.; Bjarnason, R.; Franzson, L.; Valdimarsdottir, U.A.; Thorkelsson, T. Mode of delivery was associated with transient changes in the metabolomic profile of neonates. Acta Paediatr. 2021, 110, 2110–2118. [Google Scholar] [CrossRef] [PubMed]
- National Newborn Bloodspot Screening Laboratory, Children’s Health Ireland at Temple Street. A Practical Guide to Newborn Bloodspot Screening in Ireland, 9th ed.; National Newborn Bloodspot Screening Laboratory, Children’s Health Ireland at Temple Street: Dublin, Ireland, 2022; pp. 1–73. [Google Scholar]
- Health Information and Quality Authority. Review of Processes in Use to Inform the Expansion of Newborn Bloodspot Screening Programmes; Health Information and Quality Authority: Dublin, Ireland, 2021; pp. 1–164. [Google Scholar]
- O’Reilly, D.; Crushell, E.; Hughes, J.; Ryan, S.; Rogers, Y.; Borovickova, I.; Mayne, P.; Riordan, M.; Awan, A.; Carson, K.; et al. Maple syrup urine disease: Clinical outcomes, metabolic control, and genotypes in a screened population after four decades of newborn bloodspot screening in the Republic of Ireland. J. Inherit. Metab. Dis. 2021, 44, 639–655. [Google Scholar] [CrossRef] [PubMed]
- Mulholland, P. The NSAC and the Work of Expanding the Newborn Screening Programme. The Medical Independent. 19 March 2023. Available online: https://www.medicalindependent.ie/in-the-news/news-features/the-nsac-and-the-work-of-expanding-the-newborn-screening-programme/ (accessed on 29 April 2023).
- Sasaki, E.; Kostocenko, M.; Lang, N.; Clark, T.; Rogers, M.; Muldowney, R.; Walsh, O.; O’Grady, L.; Edge, G.; Ward, A.; et al. National newborn screening for cystic fibrosis in the Republic of Ireland: Genetic data from the first 6.5 years. Eur. J. Hum. Genet. 2020, 28, 1669–1674. [Google Scholar] [CrossRef] [PubMed]
- Farrell, P.M. Setting a new standard in cystic fibrosis newborn screening illustrates controversial issues as new data emerge. Eur. J. Hum. Genet. 2020, 28, 1305–1306. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, C.; Linnane, B.; George, S.; Ni Chroinin, M.; Mullane, D.; Herzig, M.; Greally, P.; Elnazir, B.; Healy, F.; McNally, P.; et al. Neonatal screening programme for CF: Results from the Irish Comparative Outcomes Study (ICOS). Pediatr. Pulmonol. 2020, 55, 2323–2329. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, C.; Linnane, B.; Heery, E.; Conneally, N.; George, S.; Fitzpatrick, P. Newborn bloodspot screening for cystic fibrosis: What do antenatal and postnatal women know about cystic fibrosis? J. Cyst. Fibros. 2016, 15, 436–442. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, C.; Heery, E.; Conneally, N.; Linnane, B.; George, S.; Fitzpatrick, P. An evaluation of pregnant women’s knowledge and attitudes about newborn bloodspot screening. Midwifery 2017, 45, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Fitzpatrick, P.; Fitzgerald, C.; Somerville, R.; Linnane, B. Parental awareness of newborn bloodspot screening in Ireland. Ir. J. Med. Sci. 2019, 188, 921–923. [Google Scholar] [CrossRef] [PubMed]
- Burns, H.; Collins, A.; Marsden, P.; Flood, T.J.; Slatter, M.A.; Booth, C.; Xu-Bayford, J.; Leahy, T.R. Severe combined immunodeficiency (SCID)-the Irish experience. J. Clin. Immunol. 2021, 41, 1950–1953. [Google Scholar] [CrossRef] [PubMed]
- Conlon, T.A.; Hawkes, C.P.; Brady, J.J.; Murphy, N.P. The presentation of congenital adrenal hyperplasia in an unscreened population. J. Pediatr. Endocrinol. Metab. 2021, 34, 1123–1129. [Google Scholar] [CrossRef] [PubMed]
- Morton, G.; Thomas, S.; Roberts, P.; Clark, V.; Imrie, J.; Morrison, A. The importance of early diagnosis and views on newborn screening in metachromatic leukodystrophy: Results of a caregiver survey in the UK and Republic of Ireland. Orphanet J. Rare Dis. 2022, 17, 403. [Google Scholar] [CrossRef] [PubMed]
- la Marca, G. La organización del cribado neonatal en Italia: Comparación con Europa y el resto del mundo [The newborn screening program in Italy: Comparison with Europe and other countries]. Rev. Esp. Salud Publica 2021, 95, e202101007. (In Spanish) [Google Scholar] [PubMed]
- Ruoppolo, M.; Malvagia, S.; Boenzi, S.; Carducci, C.; Dionisi-Vici, C.; Teofoli, F.; Burlina, A.; Angeloni, A.; Aronica, T.; Bordugo, A.; et al. Expanded newborn screening in Italy using tandem mass spectrometry: Two years of national experience. Int. J. Neonatal Screen. 2022, 8, 47. [Google Scholar] [CrossRef] [PubMed]
- Taruscio, D.; Piccioli, A. Newborn screening in Italy: A unique program of public health in Europe. Editorial. Ann. Ist. Super. Sanita 2023, 59, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Funghini, S.; Tonin, R.; Malvagia, S.; Caciotti, A.; Donati, M.A.; Morrone, A.; la Marca, G. High frequency of biotinidase deficiency in Italian population identified by newborn screening. Mol. Genet. Metab. Rep. 2020, 25, 100689. [Google Scholar] [CrossRef]
- Maguolo, A.; Rodella, G.; Dianin, A.; Monge, I.; Messina, M.; Rigotti, E.; Pellegrini, F.; Molinaro, G.; Lupi, F.; Pasini, A.; et al. Newborn Screening for biotinidase deficiency. The experience of a regional center in Italy. Front. Pediatrics 2021, 9, 661416. [Google Scholar] [CrossRef]
- Semeraro, D.; Verrocchio, S.; Di Dalmazi, G.; Rossi, C.; Pieragostino, D.; Cicalini, I.; Ferrante, R.; Di Michele, S.; Stuppia, L.; Rizzo, C.; et al. High incidence of partial biotinidase deficiency in the first 3 years of a regional newborn screening program in Italy. Int. J. Environ. Res. Public Health 2022, 19, 8141. [Google Scholar] [CrossRef] [PubMed]
- Botti, M.; Terlizzi, V.; Francalanci, M.; Dolce, D.; Cavicchi, M.C.; Neri, A.S.; Galici, V.; Mergni, G.; Zavataro, L.; Centrone, C.; et al. Cystic fibrosis in Tuscany: Evolution of newborn screening strategies over time to the present. Ital. J. Pediatr. 2021, 47, 2. [Google Scholar] [CrossRef] [PubMed]
- Terlizzi, V.; Claut, L.; Tosco, A.; Colombo, C.; Raia, V.; Fabrizzi, B.; Lucarelli, M.; Angeloni, A.; Cimino, G.; Castaldo, A.; et al. A survey of the prevalence, management and outcome of infants with an inconclusive diagnosis following newborn bloodspot screening for cystic fibrosis (CRMS/CFSPID) in six Italian centres. J. Cyst. Fibros. 2021, 20, 828–834. [Google Scholar] [CrossRef] [PubMed]
- Bianchimani, C.; Dolce, D.; Centrone, C.; Campana, S.; Ravenni, N.; Orioli, T.; Camera, E.; Mergni, G.; Fevola, C.; Bonomi, P.; et al. Impact of pancreatitis-associated protein on newborn screening outcomes and detection of CFTR-related metabolic syndrome (CRMS)/cystic fibrosis screen positive, inconclusive diagnosis (CFSPID): A monocentric prospective pilot experience. Int. J. Neonatal Screen. 2022, 8, 46. [Google Scholar] [CrossRef] [PubMed]
- Gragnaniello, V.; Burlina, A.P.; Polo, G.; Giuliani, A.; Salviati, L.; Duro, G.; Cazzorla, C.; Rubert, L.; Maines, E.; Germain, D.P.; et al. Newborn Screening for Fabry disease in Northeastern Italy: Results of five years of experience. Biomolecules 2021, 11, 951. [Google Scholar] [CrossRef] [PubMed]
- Gragnaniello, V.; Pijnappel, P.W.W.M.; Burlina, A.P.; In’t Groen, S.L.M.; Gueraldi, D.; Cazzorla, C.; Maines, E.; Polo, G.; Salviati, L.; Di Salvo, G.; et al. Newborn screening for Pompe disease in Italy: Long-term results and future challenges. Mol. Genet. Metab. Rep. 2022, 33, 100929. [Google Scholar] [CrossRef] [PubMed]
- Polo, G.; Gueraldi, D.; Giuliani, A.; Rubert, L.; Cazzorla, C.; Salviati, L.; Marzollo, A.; Biffi, A.; Burlina, A.P.; Burlina, A.B. The combined use of enzyme activity and metabolite assays as a strategy for newborn screening of mucopolysaccharidosis type I. Clin. Chem. Lab. Med. 2020, 58, 2063–2072. [Google Scholar] [CrossRef] [PubMed]
- Burlina, A.B.; Gragnaniello, V. Newborn screening of mucopolysaccharidosis type I. Crit. Rev. Clin. Lab. Sci. 2022, 59, 257–277. [Google Scholar] [CrossRef] [PubMed]
- Donati, M.A.; Pasquini, E.; Spada, M.; Polo, G.; Burlina, A. Newborn screening in mucopolysaccharidoses. Ital. J. Pediatr. 2018, 44 (Suppl. S2), 126, Erratum in Ital. J. Pediatr. 2019, 45, 71. [Google Scholar] [CrossRef] [PubMed]
- Succoio, M.; Sacchettini, R.; Rossi, A.; Parenti, G.; Ruoppolo, M. Galactosemia: Biochemistry, molecular genetics, newborn screening, and treatment. Biomolecules 2022, 12, 968. [Google Scholar] [CrossRef]
- Vita, G.L.; Vita, G. Is it the right time for an infant screening for Duchenne muscular dystrophy? Neurol. Sci. 2020, 41, 1677–1683. [Google Scholar] [CrossRef] [PubMed]
- Malvagia, S.; Forni, G.; Ombrone, D.; la Marca, G. Development of strategies to decrease false positive results in newborn screening. Int. J. Neonatal Screen. 2020, 6, 84. [Google Scholar] [CrossRef] [PubMed]
- Cavarzere, P.; Camilot, M.; Palma, L.; Lauriola, S.; Gaudino, R.; Vincenzi, M.; Antoniazzi, F.; Teofoli, F.; Piacentini, G. Twenty years of neonatal screening for congenital adrenal hyperplasia in North-Eastern Italy: Role of liquid chromatography-tandem mass spectrometry as a second-tier test. Horm. Res. Paediatr. 2022, 95, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Maggio, M.C.; Ragusa, S.S.; Aronica, T.S.; Granata, O.M.; Gucciardino, E.; Corsello, G. Neonatal screening for congenital hypothyroidism in an Italian Centre: A 5-years real-life retrospective study. Ital. J. Pediatr. 2021, 4, 108. [Google Scholar] [CrossRef] [PubMed]
- Caiulo, S.; Corbetta, C.; Di Frenna, M.; Medda, E.; De Angelis, S.; Rotondi, D.; Vincenzi, G.; de Filippis, T.; Patricelli, M.G.; Persani, L.; et al. Newborn screening for congenital hypothyroidism: The benefit of using differential tsh cutoffs in a 2-screen program. J. Clin. Endocrinol. Metab. 2021, 106, e338–e349. [Google Scholar] [CrossRef] [PubMed]
- Medda, E.; Vigone, M.C.; Cassio, A.; Calaciura, F.; Costa, P.; Weber, G.; de Filippis, T.; Gelmini, G.; Di Frenna, M.; Caiulo, S.; et al. Neonatal screening for congenital hypothyroidism: What can we learn from discordant twins? J. Clin. Endocrinol. Metab. 2019, 104, 5765–5779. [Google Scholar] [CrossRef]
- Tuli, G.; Munarin, J.; Topalli, K.; Pavanello, E.; de Sanctis, L. Neonatal screening for congenital hypothyroidism in preterm infants: Is a targeted strategy required? Thyroid 2023, 33, 440–448. [Google Scholar] [CrossRef] [PubMed]
- Simonetti, S.; D’Amato, G.; Esposito, B.; Chiarito, M.; Dentico, D.; Lorè, T.; Cardinali, R.; Russo, S.; Laforgia, N.; Faienza, M.F. Congenital hypothyroidism after newborn screening program reorganization in the Apulia region. Ital. J. Pediatr. 2022, 48, 131. [Google Scholar] [CrossRef] [PubMed]
- Venturelli, D.; Rolli, M.; Vecchi, L.; Bensi, L.; Savarino, M.; Palazzi, G.; Bergonzini, G.; Ceccherelli, G.B. Newborn Screening for sickle cell disease in Italy: Report of 7 years of a low cost and reproducible program in an area of immigration of high risk population. Blood 2022, 140 (Suppl. S1), 2549–2550. [Google Scholar] [CrossRef]
- Colombatti, R.; Martella, M.; Cattaneo, L.; Viola, G.; Cappellari, A.; Bergamo, C.; Azzena, S.; Schiavon, S.; Baraldi, E.; Dalla Barba, B.; et al. Results of a multicenter universal newborn screening program for sickle cell disease in Italy: A call to action. Pediatr. Blood Cancer 2019, 66, e27657. [Google Scholar] [CrossRef] [PubMed]
- Bonaventura, E.; Alberti, L.; Lucchi, S.; Cappelletti, L.; Fazzone, S.; Cattaneo, E.; Bellini, M.; Izzo, G.; Parazzini, C.; Bosetti, A.; et al. Newborn screening for X-linked adrenoleukodystrophy in Italy: Diagnostic algorithm and disease monitoring. Front. Neurol. 2023, 13, 1072256. [Google Scholar] [CrossRef]
- Pane, M.; Donati, M.A.; Cutrona, C.; De Sanctis, R.; Pirinu, M.; Coratti, G.; Ricci, M.; Palermo, C.; Berti, B.; Leone, D.; et al. Neurological assessment of newborns with spinal muscular atrophy identified through neonatal screening. Eur. J. Pediatr. 2022, 181, 2821–2829. [Google Scholar] [CrossRef] [PubMed]
- Malvagia, S.; Funghini, S.; Della Bona, M.; Ombrone, D.; Mura, M.; Damiano, R.; Ricci, S.; Cortimiglia, M.; Azzari, C.; la Marca, G. The successful inclusion of ADA SCID in Tuscany expanded newborn screening program. Clin. Chem. Lab. Med. 2021, 59, e401–e404. [Google Scholar] [CrossRef] [PubMed]
- la Marca, G.; Canessa, C.; Giocaliere, E.; Romano, F.; Malvagia, S.; Funghini, S.; Moriondo, M.; Valleriani, C.; Lippi, F.; Ombrone, D.; et al. Diagnosis of immunodeficiency caused by a purine nucleoside phosphorylase defect by using tandem mass spectrometry on dried blood spots. J. Allergy Clin. Immunol. 2014, 134, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Messina, M.; Arena, A.; Fiumara, A.; Iacobacci, R.; Meli, C.; Raudino, F. Neonatal screening on tandem mass spectrometry as a powerful tool for the reassessment of the prevalence of underestimated diseases in newborns and their family members: A focus on short chain acyl-coa dehydrogenase deficiency. Int. J. Neonatal Screen. 2020, 6, 58. [Google Scholar] [CrossRef] [PubMed]
- Carducci, C.; Cotugno, G.; Di Michele, S.; Giovanniello, T.; La Marca, G.; Lepri, F.R.; Novelli, A.; Rossi, C.; Semeraro, M.; Dionisi-Vici, C. The diagnostic challenge of mild citrulline elevation at newborn screening. Mol. Genet. Metab. 2022, 135, 327–332. [Google Scholar] [CrossRef] [PubMed]
- Pellegrinelli, L.; Galli, C.; Primache, V.; Alde’, M.; Fagnani, E.; Di Berardino, F.; Zanetti, D.; Pariani, E.; Ambrosetti, U.; Binda, S. Diagnosis of congenital CMV infection via DBS samples testing and neonatal hearing screening: An observational study in Italy. BMC Infect. Dis. 2019, 19, 652. [Google Scholar] [CrossRef] [PubMed]
- Pellegrinelli, L.; Alberti, L.; Pariani, E.; Barbi, M.; Binda, S. Diagnosing congenital cytomegalovirus infection: Don’t get rid of dried blood spots. BMC Infect. Dis. 2020, 20, 217. [Google Scholar] [CrossRef] [PubMed]
- Gragnaniello, V.; Cazzorla, C.; Gueraldi, D.; Puma, A.; Loro, C.; Porcù, E.; Stornaiuolo, M.; Miglioranza, P.; Salviati, L.; Burlina, A.P.; et al. Light and shadows in newborn screening for lysosomal storage disorders: Eight years of experience in northeast Italy. Int. J. Neonatal Screen. 2024, 10, 3. [Google Scholar] [CrossRef] [PubMed]
- Svyatova, G.S.; Salimbaeva, D.N.; Kirikbaeva, M.S.; Berezina, G.M. 10 years of neonatal screening in the Republican of Kazakhstan: Results and prospects. Med. Genet. 2017, 16, 14–17. (In Russian) [Google Scholar]
- Zharmakhanova, G.; Kononets, V.; Balmagambetova, S.; Syrlybayeva, L.; Nurbaulina, E.; Zhusupova, Z.; Sakhanova, S.; Ayaganov, D.; Kim, S.; Zhumalina, A. Selective screening for inborn errors of metabolism using tandem mass spectrometry in west Kazakhstan children: Study protocol. Front. Genet. 2024, 14, 1278750. [Google Scholar] [CrossRef] [PubMed]
- Auzenbaha, M.; Aleksejeva, E.; Taurina, G.; Kornejeva, L.; Kesmpa, I.; Svabe, V.; Gailite, L. Clinical and genetic characterisation of cystic fibrosis patients in Latvia: A twenty-five-year experience. Diagnostics 2022, 12, 2893. [Google Scholar] [CrossRef] [PubMed]
- Gailite, L.; Sterna, O.; Konika, M.; Isakovs, A.; Isakova, J.; Micule, I.; Setlere, S.; Diriks, M.; Auzenbaha, M. New-born screening for spinal muscular atrophy: Results of a Latvian pilot study. Int. J. Neonatal Screen. 2022, 8, 15. [Google Scholar] [CrossRef] [PubMed]
- Visuotinis Naujagimiu Tikrinimas. Newborn Screening. Available online: http://www.patikrinkmane.lt/newborn-screening/ (accessed on 30 April 2023).
- Zabuliene, L.; Miglinas, M.; Bradziunaite, D.; Smirnova, M.; Songailiene, J.; Bratcikoviene, N.; Banys, V.; Macioniene, E.; Utkus, A. Neonatal thyroid stimulating hormone as indicator of iodine status in Lithuania. Endocr. Abstr. 2022, 81, P649. [Google Scholar] [CrossRef]
- Navardauskaitė, R.; Banevičiūtė, K.; Songailienė, J.; Grigalionienė, K.; Čereškevičius, D.; Šukys, M.; Mockevicienė, G.; Smirnova, M.; Utkus, A.; Verkauskienė, R. Impact of newborn screening on clinical presentation of congenital adrenal hyperplasia. Medicina 2021, 57, 1035. [Google Scholar] [CrossRef] [PubMed]
- LNS Luxembourg. Day of the Unborn Child, March 25: “Preventive Health Care from the Very First Hour”. Available online: https://lns.lu/en/day-of-the-unborn-child-march-25-preventive-health-care-from-the-very-first-hour/ (accessed on 2 January 2024).
- Kocova, M.; Anastasovska, V. Phenylketonuria screening in the Republic of Macedonia. Orphanet J. Rare Dis. 2016, 11, 112. [Google Scholar] [CrossRef] [PubMed]
- Gucev, Z. Comments on: Kocova, M.; Anastasovska, V. Comments on ‘Newborn screening in southeastern Europe’ published in Mol. Genet. Metab. 2014, 113, 42–45 by Groselj, U.; ZerjavTansek, M.; Smon, A.; Angelkova, N.; Anton, D.; Baric, I.; Djordjevic, M.; et al. in Mol. Genet. Metab. Rep. 2015, 5, 110. Mol. Genet. Metab. Rep. 2017, 13, 13. [Google Scholar] [CrossRef] [PubMed]
- Anastasovska, V.; Kocova, M. Ethnicity and incidence of congenital hypothyroidism in the capital of Macedonia. J. Pediatr. Endocrinol. Metab. 2017, 30, 405–409. [Google Scholar] [CrossRef] [PubMed]
- Anastasovska, V.; Sukarova-Angelovska, E.; Pesevska, M.; Taseva, E.; Kocova, M. Regional variation in the incidence of congenital hypothyroidism in Macedonia. Int. J. Neonatal Screen. 2017, 3, 22. [Google Scholar] [CrossRef]
- Anastasovska, V.; Pesevska, M.; Zdraveska, N.; Zafirova, B.; Jovcevska, J.M.; Kocova, M. Prevalence of congenital hypothyroidism in North Macedonia: Data from a newborn screening program conducted for twenty years. Turk. J. Pediatr. 2023, 65, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Fustik, S.; Anastasovska, V.; Plaseska-Karanfilska, D.; Stamatova, A.; Spirevska, L.; Pesevska, M.; Terzikj, M.; Vujovic, M. Two years of newborn screening for cystic fibrosis in North Macedonia: First experience. Balkan J. Med. Genet. 2021, 24, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Malta Independent. New Form of Screening for Medical Condition in Newborns. 13 July 2017. Available online: https://www.independent.com.mt/articles/2017-07-13/local-news/New-form-of-screening-for-medical-condition-in-newborns-6736176539 (accessed on 26 May 2023).
- TVM News. Funds from Norway to Be Used in Malta to Detect Rare Disease Found in Newborns. Available online: https://tvmnews.mt/en/news/funds-from-norway-to-be-used-in-malta-to-detect-rare-disease-found-in-newborns/ (accessed on 26 May 2023).
- Jansen, M.E.; Klein, A.W.; Buitenhuis, E.C.; Rodenburg, W.; Cornel, M.C. Expanded neonatal bloodspot screening programmes: An evaluation framework to discuss new conditions with stakeholders. Front. Pediatr. 2021, 9, 635353. [Google Scholar] [CrossRef] [PubMed]
- Oerlemans, A.J.M.; Kluijtmans, L.A.J.; van der Burg, S. The moral life of professionals in newborn screening in the Netherlands: A qualitative study. Public Health Ethics 2017, 10, 19–34. [Google Scholar] [CrossRef][Green Version]
- van Dijk, T.; Kater, A.; Jansen, M.; Dondorp, W.J.; Blom, M.; Kemp, S.; Langeveld, M.; Cornel, M.C.; van der Pal, S.M.; Henneman, L. Expanding neonatal bloodspot screening: A multi-stakeholder perspective. Front. Pediatr. 2021, 9, 706394. [Google Scholar] [CrossRef] [PubMed]
- van der Pal, S.M.; Wins, S.; Klapwijk, J.E.; van Dijk, T.; Kater-Kuipers, A.; van der Ploeg, C.P.B.; Jans, S.M.P.J.; Kemp, S.; Verschoof-Puite, R.K.; van den Bosch, L.J.M.; et al. Parents’ views on accepting, declining, and expanding newborn bloodspot screening. PLoS ONE 2022, 17, e0272585. [Google Scholar] [CrossRef] [PubMed]
- Jansen, M.E.; van den Bosch, L.J.M.; Hendriks, M.J.; Scheffer, M.M.J.; Heijnen, M.L.; Douglas, C.M.W.; van El, C.G. Parental perspectives on retention and secondary use of neonatal dried bloodspots: A Dutch mixed methods study. BMC Pediatr. 2019, 19, 230. [Google Scholar] [CrossRef] [PubMed]
- Health Council of the Netherlands. Evaluating Newborn Blood Spot Screening: A Framework and Initial Assessment. 14 December 2021. Available online: https://www.healthcouncil.nl/documents/advisory-reports/2021/12/14/evaluating-newborn-blood-spot-screening-a-framework-and-initial-assessment (accessed on 20 January 2024).
- National Government of the Netherlands (Rijksoverheid). Toekomstverkenning Neonatale Hielprikscreening (Exploring the Future of Neonatal Heel Prick Screening—In Dutch). 31 March 2023. Available online: https://www.rijksoverheid.nl/documenten/rapporten/2023/03/31/toekomstverkenning-neonatale-hielprikscreening (accessed on 20 January 2024).
- Veldman, A.; Kiewiet, M.B.G.; Westra, D.; Bosch, A.M.; Brands, M.M.G.; de Coo, R.I.F.M.; Derks, T.G.J.; Fuchs, S.A.; van den Hout, J.M.P.; Huidekoper, H.H.; et al. A Delphi survey study to formulate statements on the treatability of inherited metabolic disorders to decide on eligibility for newborn screening. Int. J. Neonatal Screen. 2023, 9, 56. [Google Scholar] [CrossRef]
- Blom, M.; Bredius, R.G.M.; Weijman, G.; Dekkers, E.H.B.M.; Kemper, E.A.; van den Akker-van Marle, M.E.; van der Ploeg, C.P.B.; van der Burg, M.; Schielen, P.C.J.I. Introducing newborn screening for severe combined immunodeficiency (SCID) in the Dutch neonatal screening program. Int. J. Neonatal Screen. 2018, 4, 40. [Google Scholar] [CrossRef] [PubMed]
- de Pagter, A.P.; Bredius, R.G.; Kuijpers, T.W.; Tramper, J.; van der Burg, M.; van Montfrans, J.; Driessen, G.J.; Dutch Working Party for Immunodeficiencies. Overview of 15-year severe combined immunodeficiency in the Netherlands: Towards newborn blood spot screening. Eur. J. Pediatr. 2015, 174, 1183–1188. [Google Scholar] [CrossRef] [PubMed]
- Blom, M.; Pico-Knijnenburg, I.; Sijne-van Veen, M.; Boelen, A.; Bredius, R.G.M.; van der Burg, M.; Schielen, P.C.J.I. An evaluation of the TREC assay with regard to the integration of SCID screening into the Dutch newborn screening program. Clin. Immunol. 2017, 180, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Van der Ploeg, C.P.B.; Blom, M.; Bredius, R.G.M.; van der Burg, M.; Schielen, P.C.J.I.; Verkerk, P.H.; Van den Akker-van Marle, M.E. Cost-effectiveness of newborn screening for severe combined immunodeficiency. Eur. J. Pediatr. 2019, 178, 721–729. [Google Scholar] [CrossRef] [PubMed]
- Blom, M.; Schoenaker, M.H.D.; Hulst, M.; de Vries, M.C.; Weemaes, C.M.R.; Willemsen, M.A.A.P.; Henneman, L.; van der Burg, M. Dilemma of reporting incidental findings in newborn screening programs for SCID: Parents’ perspective on ataxia telangiectasia. Front. Immunol. 2019, 10, 2438. [Google Scholar] [CrossRef] [PubMed]
- van den Akker-van Marle, M.E.; Blom, M.; van der Burg, M.; Bredius, R.G.M.; Van der Ploeg, C.P.B. Economic evaluation of different screening strategies for severe combined immunodeficiency based on real-life data. Int. J. Neonatal Screen. 2021, 7, 60. [Google Scholar] [CrossRef] [PubMed]
- Blom, M.; Bredius, R.G.M.; Jansen, M.E.; Weijman, G.; Kemper, E.A.; Vermont, C.L.; Hollink, I.H.I.M.; Dik, W.A.; van Montfrans, J.M.; van Gijn, M.E.; et al. Parents’ perspectives and societal acceptance of implementation of newborn screening for SCID in the Netherlands. J. Clin. Immunol. 2021, 41, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Stroek, K. Parents’ Perspectives and Societal Acceptance of Implementation of Newborn Screening for SCID in The Netherlands. Ph.D. Thesis, University van Amsterdam Faculty of Medicine, Amsterdam, The Netherlands, 2021. [Google Scholar]
- Kalkman, S.; Dondorp, W. The case for screening in early life for ‘non-treatable’ disorders: Ethics, evidence and proportionality. A report from the Health Council of the Netherlands. Eur. J. Hum. Genet. 2022, 30, 1155–1158. [Google Scholar] [CrossRef] [PubMed]
- Stroek, K.; Bouva, M.J.; Schielen, P.C.J.I.; Vaz, F.M.; Heijboer, A.C.; de Jonge, R.; Boelen, A.; Bosch, A.M. Recommendations for newborn screening for galactokinase deficiency: A systematic review and evaluation of Dutch newborn screening data. Mol. Genet. Metab. 2018, 124, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Velikanova, R.; van der Schans, S.; Bischof, M.; van Olden, R.W.; Postma, M.; Boersma, C. Cost-effectiveness of newborn screening for spinal muscular atrophy in the Netherlands. Value Health 2022, 25, 1696–1704. [Google Scholar] [CrossRef] [PubMed]
- Barendsen, R.W.; Dijkstra, I.M.E.; Visser, W.F.; Alders, M.; Bliek, J.; Boelen, A.; Bouva, M.J.; van der Crabben, S.N.; Elsinghorst, E.; van Gorp, A.G.M.; et al. Adrenoleukodystrophy newborn screening in the Netherlands (SCAN Study): The X-factor. Front. Cell Dev. Biol. 2020, 8, 499. [Google Scholar] [CrossRef] [PubMed]
- Albersen, M.; van der Beek, S.L.; Dijkstra, I.M.E.; Alders, M.; Barendsen, R.W.; Bliek, J.; Boelen, A.; Ebberink, M.S.; Ferdinandusse, S.; Goorden, S.M.I.; et al. Sex-specific newborn screening for X-linked adrenoleukodystrophy. J. Inherit. Metab. Dis. 2023, 46, 116–128. [Google Scholar] [CrossRef]
- Crefcoeur, L.; Ferdinandusse, S.; van der Crabben, S.N.; Dekkers, E.; Fuchs, S.A.; Huidekoper, H.; Janssen, M.; Langendonk, J.; Maase, R.; de Sain, M.; et al. Newborn screening for primary carnitine deficiency: Who will benefit?—A retrospective cohort study. J. Med. Genet. 2023, 60, 1177–1185. [Google Scholar] [CrossRef] [PubMed]
- Haijes, H.A.; Molema, F.; Langeveld, M.; Janssen, M.C.; Bosch, A.M.; van Spronsen, F.; Mulder, M.F.; Verhoeven-Duif, N.M.; Jans, J.J.M.; van der Ploeg, A.T.; et al. Retrospective evaluation of the Dutch pre-newborn screening cohort for propionic acidemia and isolated methylmalonic acidemia: What to aim, expect, and evaluate from newborn screening? J. Inherit. Metab. Dis. 2020, 43, 424–437. [Google Scholar] [CrossRef] [PubMed]
- Masse, R.; Deckers, E.; Rijksinstituut voor Volksgezondheid en Milieu (RIVM), Bilthoven, The Netherlands. Personal communication, 2024.
- Stroek, K.; Boelen, A.; Bouva, M.J.; De Sain-van der Velden, M.; Schielen, P.C.J.I.; Maase, R.; Engel, H.; Jakobs, B.; Kluijtmans, L.A.J.; Mulder, M.F.; et al. Evaluation of 11 years of newborn screening for maple syrup urine disease in the Netherlands and a systematic review of the literature: Strategies for optimization. JIMD Rep. 2020, 54, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Stroek, K.; Heijboer, A.C.; Bouva, M.J.; van der Ploeg, C.P.B.; Heijnen, M.A.; Weijman, G.; Bosch, A.M.; de Jonge, R.; Schielen, P.C.J.I.; van Trotsenburg, A.S.P.; et al. Critical evaluation of the newborn screening for congenital hypothyroidism in the Netherlands. Eur. J. Endocrinol. 2020, 183, 265–273. [Google Scholar] [CrossRef] [PubMed]
- van der Linde, A.A.A.; Schönbeck, Y.; van der Kamp, H.J.; van den Akker, E.L.T.; van Albada, M.E.; Boelen, A.; Finken, M.J.J.; Hannema, S.E.; Hoorweg-Nijman, G.; Odink, R.J.; et al. Evaluation of the Dutch neonatal screening for congenital adrenal hyperplasia. Arch. Dis. Child. 2019, 104, 653–657. [Google Scholar] [CrossRef] [PubMed]
- Vuong, C.; Eckhardt, C.L.; de Ligt, L.; Heijboer, H.; Rettenbacher, E.; van der Veer, A.; Stein-Wit, M.; Brons, P.; Smiers, F.J.; Suijker, M.; et al. Long-term follow up of Dutch patients with SCD diagnosed by neonatal screening—Effect on the morbidity and mortality in The Netherlands. Blood 2022, 140 (Suppl. S1), 2562–2563. [Google Scholar] [CrossRef]
- Bleeker, J.C.; Kok, I.L.; Ferdinandusse, S.; van der Pol, W.L.; Cuppen, I.; Bosch, A.M.; Langeveld, M.; Derks, T.G.J.; Williams, M.; de Vries, M.; et al. Impact of newborn screening for very-long-chain acyl-CoA dehydrogenase deficiency on genetic, enzymatic, and clinical outcomes. J. Inherit. Metab. Dis. 2019, 42, 414–423. [Google Scholar] [CrossRef] [PubMed]
- Stroek, K.; Ruiter, A.; van der Linde, A.; Ackermans, M.; Bouva, M.J.; Engel, H.; Jakobs, B.; Kemper, E.A.; van den Akker, E.L.T.; van Albada, M.E.; et al. Second-tier testing for 21-hydroxylase deficiency in the Netherlands: A newborn screening pilot study. J. Clin. Endocrinol. Metab. 2021, 106, e4487–e4496. [Google Scholar] [CrossRef]
- Dankert-Roelse, J.E.; Bouva, M.J.; Jakobs, B.S.; Janssens, H.M.; Groot, K.D.W.-D.; Schönbeck, Y.; Gille, J.J.; Gulmans, V.A.; Verschoof-Puite, R.K.; Schielen, P.; et al. Newborn blood spot screening for cystic fibrosis with a four-step screening strategy in the Netherlands. J. Cyst. Fibros. 2019, 18, 54–63. [Google Scholar] [CrossRef]
- Dijkstra, A.M.; Evers-van Vliet, K.; Heiner-Fokkema, M.R.; Bodewes, F.A.J.A.; Bos, D.K.; Zsiros, J.; van Aerde, K.J.; Koop, K.; van Spronsen, F.J.; Lubout, C.M.A. A false-negative newborn screen for tyrosinemia type 1—Need for re-evaluation of newborn screening with succinylacetone. Int. J. Neonatal Screen. 2023, 9, 66. [Google Scholar] [CrossRef] [PubMed]
- Stroek, K.; Heijboer, A.C.; van Veen-Sijne, M.; Bosch, A.M.; van der Ploeg, C.P.B.; Zwaveling-Soonawala, N.; de Jonge, R.; van Trotsenburg, A.S.P.; Boelen, A. Improving the Dutch newborn screening for central congenital hypothyroidism by using 95% reference intervals for thyroxine-binding globulin. Eur. Thyroid J. 2021, 10, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Van Rijt, W.J.; Schielen, P.C.; Özer, Y.; Bijsterveld, K.; Van der Sluijs, F.H.; Derks, T.G.; Heiner-Fokkema, M.R. Instability of acylcarnitines in stored dried blood spots: The impact on retrospective analysis of biomarkers for inborn errors of metabolism. Int. J. Neonatal Screen. 2020, 6, 83. [Google Scholar] [CrossRef] [PubMed]
- Verkaik-Kloosterman, J. Neonatal heel prick screening TSH concentration in the Netherlands as indicator of iodine status. Nutr. J. 2021, 20, 63. [Google Scholar] [CrossRef] [PubMed]
- Vaz, F.M.; Jamal, Y.; Barto, R.; Gelb, M.H.; DeBarber, A.E.; Wevers, R.A.; Nelen, M.R.; Verrips, A.; Bootsma, A.H.; Bouva, M.J.; et al. Newborn screening for cerebrotendinous xanthomatosis: A retrospective biomarker study using both flow-injection and UPLC-MS/MS analysis in 20,000 newborns. Clin. Chim. Acta 2023, 539, 170–174. [Google Scholar] [CrossRef] [PubMed]
- Stroek, K.; Visser, A.; van der Ploeg, C.P.B.; Zwaveling-Soonawala, N.; Heijboer, A.C.; Bosch, A.M.; van Trotsenburg, A.S.P.; Boelen, A.; Hoogendoorn, M.; de Jonge, R. Machine learning to improve false-positive results in the Dutch newborn screening for congenital hypothyroidism. Clin. Biochem. 2023, 116, 7–10. [Google Scholar] [CrossRef]
- Veldman, A.; Kiewiet, M.B.G.; Heiner-Fokkema, M.R.; Nelen, M.R.; Sinke, R.J.; Sikkema-Raddatz, B.; Voorhoeve, E.; Westra, D.; Dollé, M.E.T.; Schielen, P.C.J.I.; et al. Towards next-generation sequencing (NGS)-based newborn screening: A technical study to prepare for the challenges ahead. Int. J. Neonatal Screen. 2022, 8, 17. [Google Scholar] [CrossRef] [PubMed]
- Tangeraas, T.; Sæves, I.; Klingenberg, C.; Jørgensen, J.; Kristensen, E.; Gunnarsdottir, G.; Hansen, E.; Strand, J.; Lundman, E.; Ferdinandusse, S.; et al. Performance of expanded newborn screening in Norway supported by post-analytical bioinformatics tools and rapid second-tier DNA analyses. Int. J. Neonatal Screen. 2020, 6, 51. [Google Scholar] [CrossRef] [PubMed]
- Evans-Jordan, S.B.; Skolbekken, J.-A. Scientific citizenship’s youngest domain: Function creep in Norway’s newborn screening programme. Sci. Technol. Soc. 2021, 26, 98–115. [Google Scholar] [CrossRef]
- Ljungblad, U.W.; Lindberg, M.; Eklund, E.A.; Sæves, I.; Sagredo, C.; Bjørke-Monsen, A.L.; Tangeraas, T. A retrospective evaluation of the predictive value of newborn screening for vitamin B12 deficiency in symptomatic infants below 1 year of age. Int. J. Neonatal Screen. 2022, 8, 66. [Google Scholar] [CrossRef] [PubMed]
- Ljungblad, U.W.; Lindberg, M.; Eklund, E.A.; Saeves, I.; Bjørke-Monsen, A.L.; Tangeraas, T. Nitrous oxide in labour predicted newborn screening total homocysteine and is a potential risk factor for infant vitamin B12 deficiency. Acta Paediatr. 2022, 111, 2315–2321. [Google Scholar] [CrossRef] [PubMed]
- Tangeraas, T.; Ljungblad, U.W.; Lutvica, E.; Kristensen, E.; Rowe, A.D.; Bjørke-Monsen, A.L.; Rootwelt-Revheim, T.; Sæves, I.; Pettersen, R.D. Vitamin B12 deficiency (un-)detected using newborn screening in Norway. Int. J. Neonatal Screen. 2023, 9, 3. [Google Scholar] [CrossRef] [PubMed]
- Lundman, E.; Gaup, H.J.; Bakkeheim, E.; Olafsdottir, E.J.; Rootwelt, T.; Storrøsten, O.T.; Pettersen, R.D. Implementation of newborn screening for cystic fibrosis in Norway. Results from the first three years. J. Cyst. Fibros. 2016, 15, 318–324. [Google Scholar] [CrossRef] [PubMed]
- Tangeraas, T.; Constante, J.R.; Backe, P.H.; Oyarzábal, A.; Neugebauer, J.; Weinhold, N.; Boemer, F.; Debray, F.G.; Ozturk-Hism, B.; Evren, G.; et al. BCKDK deficiency: A treatable neurodevelopmental disease amenable to newborn screening. Brain 2023, 146, 3003–3013. [Google Scholar] [CrossRef] [PubMed]
- Strand, J.; Gul, K.A.; Erichsen, H.C.; Lundman, E.; Berge, M.C.; Trømborg, A.K.; Sørgjerd, L.K.; Ytre-Arne, M.; Hogner, S.; Halsne, R.; et al. Second-tier next generation sequencing integrated in nationwide newborn screening provides rapid molecular diagnostics of severe combined immunodeficiency. Front. Immunol. 2020, 11, 1417. [Google Scholar] [CrossRef]
- Hogner, S.; Lundman, E.; Strand, J.; Ytre-Arne, M.E.; Tangeraas, T.; Stray-Pedersen, A. Newborn genetic screening—Still a role for Sanger sequencing in the era of NGS. Int. J. Neonatal Screen. 2023, 9, 67. [Google Scholar] [CrossRef] [PubMed]
- SMA NBS Alliance. Newborn Screening for SMA in Poland. Available online: https://www.sma-screening-alliance.org/newborn-screening-for-sma-in-poland/ (accessed on 6 May 2023).
- Jędrzejowska, M. Advances in newborn screening and presymptomatic diagnosis of spinal muscular atrophy. Degener. Neurol. Neuromuscul. Dis. 2020, 10, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Kotulska, K.; Jozwiak, S.; Jedrzejowska, M.; Gos, M.; Ogrodnik, M.; Wysocki, J.; Czajka, H.; Kuchar, E. Newborn screening and gene therapy in SMA: Challenges related to vaccinations. Front. Neurol. 2022, 13, 890860. [Google Scholar] [CrossRef]
- Giżewska, M.; Durda, K.; Winter, T.; Ostrowska, I.; Ołtarzewski, M.; Klein, J.; Blankenstein, O.; Romanowska, H.; Krzywińska-Zdeb, E.; Patalan, M.F.; et al. Newborn screening for SCID and other severe primary immunodeficiency in the Polish-German transborder area: Experience from the first 14 months of collaboration. Front. Immunol. 2020, 11, 1948. [Google Scholar] [CrossRef]
- Zybert, K.; Wozniacki, L.; Tomaszewska-Sobczyńska, A.; Wertheim-Tysarowska, K.; Czerska, K.; Ołtarzewski, M.; Sands, D. Clinical characteristics of rare CFTR mutations causing cystic fibrosis in Polish population. Pediatr. Pulmonol. 2020, 55, 2097–2107. [Google Scholar] [CrossRef] [PubMed]
- Olszowiec-Chlebna, M.; Mospinek, E.; Jerzynska, J. Impact of newborn screening for cystic fibrosis on clinical outcomes of pediatric patients: 10 years’ experience in Lodz Voivodship. Ital. J. Pediatr. 2021, 47, 87. [Google Scholar] [CrossRef] [PubMed]
- Zybert, K.; Borawska-Kowalczyk, U.; Wozniacki, L.; Dawidziuk, M.; Ołtarzewski, M.; Sands, D. Clinical complications in children with false-negative results in cystic fibrosis newborn screening. J. Pediatr. 2022, 98, 419–424. [Google Scholar] [CrossRef] [PubMed]
- Banaszak-Ziemska, M.; Małecka, E.; Łacna, K.; Ginalska-Malinowska, M.; Niedziela, M. Moderate congenital adrenal hyperplasia in two girls diagnosed by newborn screening. Pediatr. Endocrinol. Diabetes Metab. 2021, 27, 291–297. [Google Scholar] [CrossRef]
- Klosinska, M.; Kaczynska, A.; Ben-Skowronek, I. Congenital hypothyroidism in preterm newborns—The challenges of diagnostics and treatment: A review. Front. Endocrinol. 2022, 13, 860862. [Google Scholar] [CrossRef] [PubMed]
- Stawerska, R.; Nowak-Bednarek, M.; Talar, T.; Kolasa-Kicińska, M.; Łupińska, A.; Hilczer, M.; Gulczyńska, E.; Lewiński, A. The prevalence of hypothyroxinemia in premature newborns. Front. Endocrinol. 2022, 13, 940152. [Google Scholar] [CrossRef] [PubMed]
- Taybert, J.; Ołtarzewski, M.; Sykut-Cegielska, J. Molecular background and disease prevalence of biotinidase deficiency in a Polish population-data based on the national newborn screening programme. Genes 2022, 13, 802. [Google Scholar] [CrossRef] [PubMed]
- Napiórkowska-Orkisz, M.; Gutysz-Wojnicka, A.; Tanajewska, M.; Sadowska-Krawczenko, I. Evaluation of methods to minimize pain in newborns during capillary blood sampling for screening: A randomized clinical trial. Int. J. Environ. Res. Public Health 2022, 19, 870. [Google Scholar] [CrossRef]
- Costeira, M.J.; Costa, P.; Roque, S.; Carvalho, I.; Vilarinho, L.; Palha, J.A. History of neonatal screening of congenital hypothyroidism in Portugal. Int. J. Neonatal Screen. 2024, 10, 16. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, M.M.; Marcão, A.; Sousa, C.; Nogueira, C.; Fonseca, H.; Rocha, H.; Vilarinho, L. Portuguese neonatal screening program: A cohort study of 18 years using MS/MS. Int. J. Neonatal Screen. 2024, 10, 25. [Google Scholar] [CrossRef] [PubMed]
- Marcão, A.; Barreto, C.; Pereira, L.; Vaz, L.G.; Cavaco, J.; Casimiro, A.; Félix, M.; Silva, T.R.; Barbosa, T.; Freitas, C.; et al. Cystic fibrosis newborn screening in Portugal: PAP value in populations with stringent rules for genetic studies. Int. J. Neonatal Screen. 2018, 4, 22. [Google Scholar] [CrossRef] [PubMed]
- Camacho, B.; Constant, C.; Pereira, L.; Barreto, C. Cystic fibrosis newborn screening in a Portuguese centre: Are we complying with European recommendations? Lusíadas Sci. J. 2022, 3. [Google Scholar] [CrossRef]
- Nogueira, C.; Marcão, A.; Rocha, H.; Sousa, C.; Fonseca, H.; Valongo, C.; Vilarinho, L. Molecular picture of cobalamin C/D defects before and after newborn screening era. J. Med. Screen. 2017, 24, 6–11. [Google Scholar] [CrossRef] [PubMed]
- Lipari Pinto, P.; Florindo, C.; Janeiro, P.; Santos, R.L.; Mexia, S.; Rocha, H.; Tavares de Almeida, I.; Vilarinho, L.; Gaspar, A. Acquired vitamin B12 deficiency in newborns: Positive impact on newborn health through early detection. Nutrients 2022, 14, 4397. [Google Scholar] [CrossRef] [PubMed]
- Guerra, I.M.S.; Ferreira, H.B.; Neves, B.; Melo, T.; Diogo, L.M.; Domingues, M.R.; Moreira, A.S.P. Lipids and phenylketonuria: Current evidence points to the need for lipidomics studies. Arch. Biochem. Biophys. 2020, 688, 108431. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, H.B.; Guerra, I.M.S.; Melo, T.; Rocha, H.; Moreira, A.S.P.; Paiva, A.; Domingues, M.R. Dried blood spots in clinical lipidomics: Optimization and recent findings. Anal. Bioanal. Chem. 2022, 414, 7085–7101. [Google Scholar] [CrossRef] [PubMed]
- PKU Romania. Neonatal Screening for Phenylketonuria. 2020. Available online: https://www.pku-romania.ro/en/about-pku/pku-diagnostic (accessed on 6 May 2023).
- Dima, V. Actualities in neonatal endocrine and metabolic screening. Acta Endocrinol. 2021, 17, 416–421. [Google Scholar] [PubMed]
- Nanu, M.; Ardeleanu, I.S.; Brezan, F.; Nanu, I.; Apostol, A.; Moldovanu, F.; Lazarescu, H.; Gheorghiu, M.L.; Kozma, A. Neonatal screening for congenital hypothyroidism in Romania: Data from MEDILOG medical information registry. Acta Endocrinol. 2019, 15, 209–214. [Google Scholar] [CrossRef] [PubMed]
- Anton-Paduraru, D.T.; Bilha, S.; Miftode, E.G.; Iliescu, M.L.; Leustean, L.; Ungureanu, M.C. Screening of congenital hypothyroidism in North-East Romania. Benefits and messages for further improvement. Acta Endocrinol. 2020, 16, 437–442. [Google Scholar] [CrossRef] [PubMed]
- Oprea, O.R.; Barbu, S.V.; Kodori, D.R.; Dobreanu, M. Recall rate in congenital hypothyroidism screening: Influence of the day of sample collection and lower cutoff. Acta Endocrinol. 2021, 17, 22–25. [Google Scholar] [CrossRef] [PubMed]
- Badiu Tișa, I.; Achim, A.C.; Cozma-Petruț, A. The importance of neonatal screening for galactosemia. Nutrients 2022, 15, 10. [Google Scholar] [CrossRef] [PubMed]
- Nova Medica. More than 95,000 Newborns Have Undergone the Neonatal Screening Program in the Russian Federation. 20 February 2023. Available online: https://novamedica.com/media/theme_news/p/13731-more-than-95000-newborns-have-undergone-the-neonatal-screening-program-in-the-russian-federation (accessed on 8 May 2023).
- GxP News. Expanded Newborn Screening in Russia Will Be Provided in 10 Clinics. 20 December 2022. Available online: https://gxpnews.net/en/2022/12/expanded-newborn-screening-in-russia-will-be-provided-in-10-clinics/ (accessed on 6 May 2023).
- Volgina, S.Y.; Sokolov, A.A. An analysis of medical care services for children with rare diseases in the Russian Federation. Front. Pharmacol. 2021, 12, 754073. [Google Scholar] [CrossRef]
- Baranov, A.; Namazova-Baranova, L.; Albitsky, V.; Ustinova, N.; Terletskaya, R.; Komarova, O. Paediatrics in Russia: Past, present and future. Arch Dis. Child. 2017, 102, 774–778. [Google Scholar] [CrossRef] [PubMed]
- Shinwari, K.; Bolkov, M.; Tuzankina, I.A.; Chereshnev, V.A. Newborn screening through TREC, TREC/KREC system for primary immunodeficiency with limitation of TREC/KREC. Comprehensive review. Antiinflamm. Antiallergy Agents Med. Chem. 2021, 20, 132–149. [Google Scholar] [CrossRef] [PubMed]
- Khalturina, E.O.; Degtyareva, N.D.; Bairashevskaia, A.V.; Mulenkova, A.V.; Degtyareva, A.V. Modern diagnostic capabilities of neonatal screening for primary immunodeficiencies in newborns. Clin. Exp. Pediatr. 2021, 64, 504–510. [Google Scholar] [CrossRef] [PubMed]
- Cheremokhin, D.A.; Shinwari, K.; Deryabina, S.S.; Bolkov, M.A.; Tuzankina, I.A.; Kudlay, D.A. Analysis of the TREC and KREC levels in the dried blood spots of healthy newborns with different gestational ages and weights. Acta Naturae 2022, 14, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Kiselev, A.; Maretina, M.; Shtykalova, S.; Al-Hilal, H.; Maslyanyuk, N.; Plokhih, M.; Serebryakova, E.; Frolova, M.; Shved, N.; Krylova, N.; et al. Establishment of a Pilot Newborn Screening Program for Spinal Muscular Atrophy in Saint Petersburg. Int. J. Neonatal Screen. 2024, 10, 9. [Google Scholar] [CrossRef]
- Sherman, V.; Kondratyeva, E.; Kashirskaya, N.; Voronkova, A.; Nikonova, V.; Zhekaite, E.; Kutsev, S. Newborn screening for cystic fibrosis in Russia: A catalyst for improved care. Int. J. Neonatal Screen. 2020, 6, 34. [Google Scholar] [CrossRef] [PubMed]
- Gundorova, P.; Kuznetcova, I.A.; Baydakova, G.V.; Stepanova, A.A.; Itkis, Y.S.; Kakaulina, V.S.; Alferova, I.P.; Lyazina, L.V.; Andreeva, L.P.; Kanivets, I.; et al. BH4-deficient hyperphenylalaninemia in Russia. PLoS ONE 2021, 16, e0249608. [Google Scholar] [CrossRef] [PubMed]
- Regione Emilia-Romagna. Newborn Screening, a Simple Test to Protect the Health of Babies. Information for Parents. 8 May 2020. Available online: https://salute.regione.emilia-romagna.it/normativa-e-documentazione/informative-material/opuscolo-screening-neonatale (accessed on 6 May 2023).
- Mitrovic, K.; Vukovic, R.; Milenkovic, T.; Todorovic, S.; Radivojcevic, J.; Zdravkovic, D. Changes in the incidence and etiology of congenital hypothyroidism detected during 30 years of a screening program in central Serbia. Eur. J. Pediatr. 2016, 175, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Milenkovic, T.; Vukovic, R.; Radojicic, B.; Mitrovic, K.; Todorovic, S.; Zatezalo, L. Thirty years of the newborn screening program in Central Serbia: The missed cases of congenital hypothyroidism. Turk. J. Pediatr. 2019, 61, 319–324. [Google Scholar] [CrossRef] [PubMed]
- The Serbian Monitor.com. Spinal Muscular Atrophy Screenings in All Maternity Hospitals as of April. 23 March 2023. Available online: https://www.serbianmonitor.com/en/spinal-muscular-atrophy-screenings-in-all-maternity-hospitals-as-of-april/ (accessed on 6 May 2023).
- Dluholucký, S.; Knapková, M. The first results of extended newborn screening in Slovakia—Differences between the Majority and the Roma Ethnic Group. Int. J. Neonatal Screen. 2017, 3, 25. [Google Scholar] [CrossRef]
- Šmon, A.; Grošelj, U.; Žerjav Tanšek, M.; Biček, A.; Oblak, A.; Zupančič, M.; Kržišnik, C.; Repič Lampret, B.; Murko, S.; Hojker, S.; et al. Newborn screening in Slovenia. Zdr. Varst. 2015, 54, 86–90. [Google Scholar] [CrossRef] [PubMed]
- Šmon, A.; Repič Lampret, B.; Grošelj, U.; Žerjav Tanšek, M.; Kovac, J.; Perko, D.; Bertok, S.; Battelino, T.; Trebusak Podkrajsek, K. Next generation sequencing as a follow-up test in an expanded newborn screening programme. Clin. Biochem. 2018, 52, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Repič Lampret, B.; Remec, Ž.I.; Drole Torkar, A.; Žerjav Tanšek, M.; Šmon, A.; Koračin, V.; Čuk, V.; Perko, D.; Ulaga, B.; Jelovšek, A.M.; et al. Expanded newborn screening program in Slovenia using tandem mass spectrometry and confirmatory next generation sequencing genetic testing. Zdr. Varst. 2020, 59, 256–263. [Google Scholar] [CrossRef] [PubMed]
- Remec, Ž.I.; Grošelj, U.; Drole Torkar, A.; Žerjav Tanšek, M.; Čuk, V.; Perko, D.; Ulaga, B.; Lipovec, N.; Debeljak, M.; Kovac, J.; et al. Very long-chain acyl-coA dehydrogenase deficiency: High incidence of detected patients with expanded newborn screening program. Front. Genet. 2021, 12, 648493. [Google Scholar] [CrossRef]
- Perko, D.; Grošelj, U.; Čuk, V.; Remec, Ž.I.; Žerjav Tanšek, M.; Drole Torkar, A.; Krhin, B.; Bicek, A.; Oblak, A.; Battelino, T.; et al. Comparison of tandem mass spectrometry and the fluorometric method—Parallel phenylalanine measurement on a large fresh sample series and implications for newborn screening for phenylketonuria. Int. J. Mol. Sci. 2023, 24, 2487. [Google Scholar] [CrossRef] [PubMed]
- Perko, D.; Repič Lampret, B.; Remec, Ž.I.; Žerjav Tanšek, M.; Drole Torkar, A.; Krhin, B.; Bicek, A.; Oblak, A.; Battelino, T.; Grošelj, U. Optimizing the phenylalanine cut-off value in a newborn screening program. Genes 2022, 13, 517. [Google Scholar] [CrossRef] [PubMed]
- Marín Soria, J.L.; González de Aledo Castillo, J.M.; Argudo Ramírez, A.; López Galera, R.M.; Pajares García, S.; Ribes Rubió, A.; García Villoria, J.; Yahyaoui Macías, R.; Álvarez Ríos, A.I.; Melguizo Madrid, E.; et al. Inicio, evolución y situación actual de los programas de cribado neonatal en España [Beginnings, evolution and current situation of the newborn screening programs in Spain]. Rev. Esp. Salud Publica 2021, 95, e202102041. (English abstract). (In Spanish) [Google Scholar] [PubMed]
- Dulín Iñiguez, E.; Eguileor Gurtubai, I.; Espada Sáenz-Torre, M. Los programas de cribado neonatal en España. Ciencia, investigación y salud pública, claves para su calidad y eficacia [The neonatal screening programs in Spain. Science, research and public health, keys to their quality and effectiveness]. Rev. Esp. Salud Publica 2021, 95, e202101024. (English abstract). (In Spanish) [Google Scholar]
- González-Irazabal, Y.; Hernandez de Abajo, G.; Martínez-Morillo, E. Identifying and overcoming barriers to harmonize newborn screening programs through consensus strategies. Crit. Rev. Clin. Lab. Sci. 2021, 58, 29–48. [Google Scholar] [CrossRef] [PubMed]
- González-Lamuño Leguina, D.; Bóveda Fontán, M.D.; Bueno Delgado, M.; Gort Mas, L.; Unceta Suárez, M.; Morales Conejo, M. El cribado metabólico del recién nacido como modelo asistencial de la medicina de precisión. Perspectiva desde la Asociación Española para el Estudio de los Errores Congénitos del Metabolismo (AECOM) [The metabolic newborn screening as a healthcare model of the precision medicine. Perspective from the Spanish Association for the Study of Congenital Errors of Metabolism (AECOM)]. Rev. Esp. Salud Publica 2021, 95, e202101021. (In Spanish) [Google Scholar] [PubMed]
- Pàmpols, R.T.; Pérez Aytés, A.; García Sagredo, J.M.; Díaz de Bustamante, A.; Martín Arribas, C.; García López, F.J.; Nicolás Jiménez, P.; Dulín Íñiguez, E.; Labrador Cañadas, V. Medio siglo de cribado neonatal en España: Evolución de los aspectos éticos, legales y sociales (AELS). Parte I, aspectos éticos. Rev. Esp. Salud Publica 2021, 95, e1–e22. (In Spanish) [Google Scholar]
- Nicolás Jiménez, P.; Pàmpols, R.T.; García López, F.J.; Martín Arribas, C.; Pérez Aytés, A.; García Sagredo, J.M.; Díaz de Bustamante, A.; Dulín Íñiguez, E.; Labrador Cañadas, V. Medio siglo de cribado neonatal en España: Evolución de los aspectos éticos, legales y sociales (aels). Parte II, marco legal. Rev. Esp. Salud Publica 2021, 95, e1–e14. (In Spanish) [Google Scholar]
- Labrador Cañadas, M.V.; Pàmpols Ros, T.; Dulín Íñiguez, E.; Pérez Aytés, A.; García Sagredo, J.M.; Díaz de Bustamante, A.; Martín Arribas, C.; García López, F.J.; Nicolás Jiménez, P. Medio siglo de cribado neonatal en España: Evolución de los aspectos éticos, legales y sociales (AELS). Parte III, aspectos sociales [Half a century of newborn screening in Spain: Evolution of ethical, legal and social issues (ELSIs). Part III, social issues]. Rev. Esp. Salud Publica 2021, 95, e2 02101016. (In Spanish) [Google Scholar]
- Marín Soria, J.L.; López Galera, R.M.; Argudo Ramírez, A.; González de Aledo, J.M.; Pajares García, S.; Navarro Sastre, A.; Hernandez Pérez, J.M.; Ribes Rubio, A.; Gort Mas, L.; García Villoria, J.; et al. 50 años del programa de cribado neonatal en Cataluña [50 years of the neonatal screening program in Catalonia]. Rev. Esp. Salud Publica 2020, 94, e202012177. (In Spanish) [Google Scholar] [PubMed]
- Sánchez Pintos, P.; Cocho de Juan, J.Á.; Bóveda Fontán, M.D.; Castiñeiras Ramos, D.E.; Colón Mejeras, C.; Iglesias Rodríguez, A.J.; de Castro López, M.J.; Alonso Fernández, J.R.; Fraga Bermúdez, J.M.; Couce Pico, M.L. Evaluación y perspectiva de 20 años de cribado neonatal en Galicia. Resultados del programa [Evaluation and perspective of 20 years of neonatal screening in Galicia. Program results]. Rev. Esp. Salud Publica 2020, 94, e202012161. (In Spanish) [Google Scholar] [PubMed]
- Delgado-Pecellín, C.; Álvarez Ríos, I.; Bueno Delgado, M.D.A.; Jiménez Jambrina, M.M.; Quintana Gallego, M.E.; Ruiz Salas, P.; Marcos Luque, I.; Melguizo Madrid, E. Resultados del cribado neonatal de Andalucía Occidental tras una década de experiencia [Results of the neonatal screening on Western Andalusia after a decade of experience]. Rev. Esp. Salud Publica 2020, 94, e202012174. (In Spanish) [Google Scholar] [PubMed]
- García-Morín, M.; Bardón-Cancho, E.J.; Beléndez, C.; Zamarro, R.; Béliz-Mendiola, C.; González-Rivera, M.; Vecilla, C.; Llorente-Otones, L.; Pérez-Alonso, V.; Román, S.S.; et al. Fifteen years of newborn sickle cell disease screening in Madrid, Spain: An emerging disease in a European country. Ann. Hematol. 2020, 99, 1465–1474. [Google Scholar] [CrossRef] [PubMed]
- Núñez-Jurado, D.; Payán-Pernía, S.; Álvarez-Ríos, A.I.; Jiménez-Jambrina, M.; Concepcion Pérez-De-Soto, I.; José Palma-Vallellano, A.; Zapata-Bautista, R.; Carlos Hernández-Castellet, J.; Paz Garrastazul-Sánchez, M.; Arqueros-Martínez, V.; et al. Neonatal screening for sickle cell disease in Western Andalusia: Results and lessons learnt after 3 years of implementation. Am. J. Perinatol. 2022. [Google Scholar] [CrossRef] [PubMed]
- Martín-Rivada, Á.; Cambra Conejero, A.; Martín-Hernández, E.; Moráis López, A.; Bélanger-Quintana, A.; Cañedo Villarroya, E.; Quijada-Fraile, P.; Bellusci, M.; Chumillas Calzada, S.; Bergua Martínez, A.; et al. Newborn screening for propionic, methylmalonic acidemia and vitamin B12 deficiency. Analysis of 588,793 newborns. J. Pediatr. Endocrinol. Metab. 2022, 35, 1223–1231. [Google Scholar] [CrossRef] [PubMed]
- Argudo Ramírez, A.; Martín Nalda, A.; Marín Soria, J.L.; López Galera, R.M.; González de Aledo Castillo, J.M.; Pajares García, S.; Rivière, J.G.; Martínez Gallo, M.; Colobran, R.; Parra Martínez, A.; et al. Primer programa europeo de cribado neonatal para la inmunodeficiencia combinada grave: Experiencia de tres años en Cataluña [First universal newborn screening program for severe combined immunodeficiency in Europe. Three-years’ experience in Catalonia]. Rev. Esp. Salud Publica 2020, 94, e202012153. (In Spanish) [Google Scholar] [PubMed]
- Argudo-Ramírez, A.; Martín-Nalda, A.; González de Aledo-Castillo, J.M.; López-Galera, R.; Marín-Soria, J.L.; Pajares-García, S.; Martínez-Gallo, M.; García-Prat, M.; Colobran, R.; Riviere, J.G.; et al. Newborn screening for SCID. Experience in Spain (Catalonia). Int. J. Neonatal Screen. 2021, 7, 46. [Google Scholar] [CrossRef] [PubMed]
- Martín-Rivada, Á.; Palomino Pérez, L.; Ruiz-Sala, P.; Navarrete, R.; Cambra Conejero, A.; Quijada Fraile, P.; Moráis López, A.; Belanger-Quintana, A.; Martín-Hernández, E.; Bellusci, M.; et al. Diagnosis of inborn errors of metabolism within the expanded newborn screening in the Madrid region. JIMD Rep. 2022, 63, 146–161. [Google Scholar] [CrossRef] [PubMed]
- Cambra Conejero, A.; Martínez Figueras, L.; Ortiz Temprado, A.; Blanco Soto, P.; Martín Rivada, Á.; Palomino Pérez, L.; Cañedo Villarroya, E.; Pedrón Giner, C.; Quijada Fraile, P.; Martín-Hernández, E.; et al. Análisis de casos positivos de cribado neonatal de errores congénitos del metabolismo en la Comunidad de Madrid [Newborn Screening Program in the Community of Madrid: Evaluation of positive cases]. Rev. Esp. Salud Publica 2020, 94, e202012185. (In Spanish) [Google Scholar] [PubMed]
- Sanz Fernández, M.; Mora Sitja, M.; Carrascón González Pinto, L.; Rodríguez Sánchez, A. Análisis del grado de sospecha clínica en pacientes con hiperplasia suprarrenal congénita por déficit de 21 hidroxilasa antes de la obtención del resultado del cribado neonatal en la Comunidad de Madrid [Analysis of the degree of clinical suspect in patients with congenital adrenal hyperplasia by 21-hydroxylase deficiency before obtaining the result of the newborn screening program of the autonomous Community of Madrid]. Rev. Esp. Salud Publica 2020, 94, e202012186. (In Spanish) [Google Scholar] [PubMed]
- Delgado, J.A.; Bauça, J.M.; Pérez Esteban, G.; Caimari Jaume, M.; Robles Bauza, J. Challenges in screening for congenital hypothyroidism: Optimization of thyrotropin cut-off values. Clin. Chim. Acta 2021, 512, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Espada Sáenz-Torre, M.; Peiró Callizo, E.; Eguileor Gurtubai, I. El papel de la salud publica como clave del éxito del programa de cribado neonatal en el País Vasco [The role of public health as a key to the success of the neonatal screening program in the Basque Country]. Rev. Esp. Salud Publica 2020, 94, e202012175. (In Spanish) [Google Scholar] [PubMed]
- Asso Ministral, L.; Marín Soria, J.L.; López Galera, R.M.; Argudo Ramírez, A.; González de Aledo Castillo, J.M.; Prats Viedma, B.; Casas Puig, C.; García Villoria, J.; Cabezas Peña, C. Análisis de la implementación de un sistema de transporte unificado de las muestras de cribado neonatal en Cataluña [Analysis of the implementation of a unified transportation system of the neonatal screening samples in Catalonia]. Rev. Esp. Salud Publica 2020, 94, e202012163. (In Spanish) [Google Scholar] [PubMed]
- Pajares, S.; Arranz, J.A.; Ormazabal, A.; Del Toro, M.; García-Cazorla, Á.; Navarro-Sastre, A.; López, R.M.; Meavilla, S.M.; de Los Santos, M.M.; García-Volpe, C.; et al. Implementation of second-tier tests in newborn screening for the detection of vitamin B12 related acquired and genetic disorders: Results on 258,637 newborns. Orphanet. J. Rare Dis. 2021, 16, 195. [Google Scholar] [CrossRef]
- Arrudi-Moreno, M.; García-Romero, R.; Samper-Villagrasa, P.; Sánchez-Malo, M.J.; Martin-de-Vicente, C. Neonatal cystic fibrosis screening: Analysis and differences in immunoreactive trypsin levels in newborns with a positive screen. An. Pediatr. (Engl. Ed.) 2021, 95, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Piñero-Fernández, J.A.; Vicente-Calderón, C.; Lorente-Sánchez, M.J.; Juan-Fita, M.J.; Egea-Mellado, J.M.; González-Gallego, I.C. Phenotypic characterization of a pediatric cohort with cystinuria and usefulness of newborn screening. Pediatr. Nephrol. 2023, 38, 1513–1521. [Google Scholar] [CrossRef] [PubMed]
- López Galera, R.M.; Castiñeiras Ramos, D.; Rocha, H. Cribado neonatal del hipotiroidismo congénito [Neonatal screening for congenital hypothyroidism]. Rev. Esp. Salud Publica 2021, 95, e202101010. (In Spanish) [Google Scholar] [PubMed]
- Rodríguez Del Rosario, S.; Gutiérrez Zamorano, M.; Pérez de Nanclares Leal, G.; Rellán Rodríguez, S.; Bahíllo-Curieses, M.P. TSH elevation in neonatal screening as the first manifestation of other associated diseases. Endocrinol. Diabetes Nutr. (Engl. Ed.) 2023, 70, 297–299. [Google Scholar] [CrossRef] [PubMed]
- Valcárcel Nazco, C.; García Pérez, L.; Linertová, R.; Castilla, I.; Vallejo Torres, L.; Ramos Goñi, J.M.; Labrador Cañadas, V.; Couce, M.L.; Espada Sáenz-Torres, M.; Dulín Íñiguez, E.; et al. Métodos para la evaluación económica de programas de cribado neonatal [Cost-effectiveness methods of newborn screening assessment]. Rev. Esp. Salud Publica 2021, 95, e202101009. [Google Scholar] [PubMed]
- Prieto-González, D.; Castilla-Rodríguez, I.; González, E.; Couce, M.L. Towards the automated economic assessment of newborn screening for rare diseases. J. Biomed. Inform. 2019, 95, 103216. [Google Scholar] [CrossRef] [PubMed]
- Navarrete, R.; Leal, F.; Vega, A.I.; Morais-López, A.; Garcia-Silva, M.T.; Martín-Hernández, E.; Quijada-Fraile, P.; Bergua, A.; Vives, I.; García-Jiménez, I.; et al. Value of genetic analysis for confirming inborn errors of metabolism detected through the Spanish neonatal screening program. Eur. J. Hum. Genet. 2019, 27, 556–562. [Google Scholar] [CrossRef]
- Macias, R.Y. History, current situation and future perspectives of neonatal screening for rare diseases in Spain. Asociacion Española de Laboratorios de Medicamentos Huérfanos y Ultrahuérfanos (ELMHU). 3 August 2022. Available online: https://aelmhu.es/en/articulos/cribado-neonatal-de-enfermedades-raras/ (accessed on 3 February 2024).
- Pajares-García, S.; González de Aledo-Castillo, J.M.; Flores-Jiménez, J.E.; Collado, T.; Pérez, J.; Paredes-Fuentes, A.J.; Argudo-Ramírez, A.; López-Galera, R.M.; Prats, B.; García-Villoria, J. Analysis of a second-tier test panel in dried blood spot samples using liquid chromatography-tandem mass spectrometry in Catalonia’s newborn screening programme. Clin. Chem. Lab. Med. 2024, 62, 493–505. [Google Scholar] [CrossRef] [PubMed]
- Pàmpols Ros, T.; Pérez Aytés, A.; García Sagredo, J.M.; Díaz de Bustamante, A.; Blanco Guillermo, I. Cribado neonatal genómico. Perspectiva de la Comisión de ética de la Asociación Española de Genética Humana. Parte I. Las tecnologías de secuenciación masiva (NGS) y su aplicación al cribado neonatal. Desafíos y oportunidades [Genomic newborn screening. Perspective from the Ethics commission of the Spanish Society for Human Genetics. Part I. Next generation sequencing technologies applied to newborn screening. Challenges and opportunities]. Rev. Esp. Salud Publica 2022, 96, e202202012. (In Spanish) [Google Scholar] [PubMed]
- Pàmpols Ros, T.; Pérez Aytés, A.; García Sagredo, J.M.; Díaz de Bustamante, A.; Blanco Guillermo, I. Cribado neonatal genómico. Perspectiva de la Comisión de ética de la Asociación Española de Genética Humana. Parte II: Aspectos Éticos, Legales y Sociales (AELS) de la introducción de las tecnologías de secuenciación masiva (NGS) en un programa de cribado neonatal de salud pública [Genomic newborn screening. Perspective from the Ethics Commission of the Spanish Society for Human Genetics. Part II: Ethical, legal and social issues (ELSIs) of the introduction of next generation sequencing technologies in a public health newborn screening program]. Rev. Esp. Salud Publica 2022, 96, e202203030. (In Spanish) [Google Scholar] [PubMed]
- Zetterström, R.H. (Karolinska University Hospital, Stockholm, Sweden). Personal communication. 2023. [Google Scholar]
- Barbaro, M.; Ohlsson, A.; Borte, S.; Jonsson, S.; Zetterström, R.H.; King, J.; Winiarski, J.; von Döbeln, U.; Hammarström, L. Newborn Screening for severe primary immunodeficiency diseases in Sweden-a 2-year pilot TREC and KREC screening study. J. Clin. Immunol. 2017, 37, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Göngrich, C.; Ekwall, O.; Sundin, M.; Brodszki, N.; Fasth, A.; Marits, P.; Dysting, S.; Jonsson, S.; Barbaro, M.; Wedell, A.; et al. First year of TREC-based national SCID screening in Sweden. Int. J. Neonatal Screen. 2021, 7, 59. [Google Scholar] [CrossRef] [PubMed]
- Sörensen, L.; Von Döbeln, U.; Åhlman, H.; Ohlsson, A.; Engvall, M.; Naess, K.; Backman-Johansson, C.; Nordqvist, Y.; Wedell, A.; Zetterström, R.H. Expanded screening of one million Swedish babies with R4S and CLIR for post-analytical evaluation of data. Int. J. Neonatal Screen. 2020, 6, 42. [Google Scholar] [CrossRef] [PubMed]
- Olsson, D.; Barbaro, M.; Haglind, C.; Halldin, M.; Lajic, S.; Tucci, S.; Zetterström, R.H.; Nordenström, A. Very long-chain acyl-CoA dehydrogenase deficiency in a Swedish cohort: Clinical symptoms, newborn screening, enzyme activity, and genetics. JIMD Rep. 2022, 63, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Heather, N.L.; Nordenström, A. Newborn screening for CAH—challenges and opportunities. Int. J. Neonatal Screen. 2021, 7, 11. [Google Scholar] [CrossRef] [PubMed]
- Lajic, S.; Karlsson, L.; Zetterström, R.H.; Falhammar, H.; Nordenström, A. The success of a screening program is largely dependent on close collaboration between the laboratory and the clinical follow-up of the patients. Int. J. Neonatal Screen. 2020, 6, 68. [Google Scholar] [CrossRef] [PubMed]
- Zetterström, R.H.; Karlsson, L.; Falhammar, H.; Lajic, S.; Nordenström, A. Update on the Swedish newborn screening for congenital adrenal hyperplasia due to 21-hydroxylase deficiency. Int. J. Neonatal Screen. 2020, 6, 71. [Google Scholar] [CrossRef] [PubMed]
- Gunnerbeck, A.; Lundholm, C.; von Döbeln, U.; Zetterström, R.H.; Almqvist, C.; Nordenström, A. Neonatal screening for congenital hypothyroidism in Sweden 1980–2013: Effects of lowering the thyroid-stimulating hormone threshold. Eur. J. Endocrinol. 2023, 188, 536–546. [Google Scholar] [CrossRef] [PubMed]
- Beck, O.; Kenan Modén, N.; Seferaj, S.; Lenk, G.; Helander, A. Study of measurement of the alcohol biomarker phosphatidylethanol (PEth) in dried blood spot (DBS) samples and application of a volumetric DBS device. Clin. Chim. Acta 2018, 479, 38–42. [Google Scholar] [CrossRef] [PubMed]
- Ohlsson, A.; Rehnholm, K.; Shubham, K.; von Döbeln, U. Incidence of glucose-6-phosphate dehydrogenase deficiency among Swedish newborn infants. Int. J. Neonatal Screen. 2019, 5, 38. [Google Scholar] [CrossRef]
- Beck, O.; Mellring, M.; Löwbeer, C.; Seferaj, S.; Helander, A. Measurement of the alcohol biomarker phosphatidylethanol (PEth) in dried blood spots and venous blood-importance of inhibition of post-sampling formation from ethanol. Anal. Bioanal. Chem. 2021, 413, 5601–5606. [Google Scholar] [CrossRef] [PubMed]
- Ahrens-Nicklas, R.C.; Ganetzky, R.D.; Rush, P.W.; Conway, R.L.; Ficicioglu, C. Characteristics and outcomes of patients with formiminoglutamic aciduria detected through newborn screening. J. Inherit. Metab. Dis. 2019, 42, 140–146, Erratum in J. Inherit. Metab. Dis. 2019, 42, 1040. [Google Scholar] [CrossRef] [PubMed]
- Uppsala Univesitat. Screen4Care: Accelerating Diagnosis for Rare Disease Patients through Genetic Newborn Screening & AI. Center for Research Ethics & Bioethics (CRB). 19 October 2021. Available online: https://www.crb.uu.se/news/item/?tarContentId=975702 (accessed on 12 May 2023).
- Newborn Screening. Newborn Screening Switzerland. Interesting Facts for Parents and Professionals. 2023. Available online: https://www.neoscreening.ch/en/ (accessed on 20 May 2023).
- Fouriki, A.; Schnider, C.; Theodoropoulou, K.; Pachlopnik, J.; Hofer, M.; Candotti, F. Pédiatrie—Dépistage des nouveau-nés en Suisse pour les déficiences sévères à lymphocytes T et B [Newborn screening for severe T and B lymphocyte deficiencies in Switzerland]. Rev. Med. Suisse 2021, 17, 68–76. (In French) [Google Scholar] [PubMed]
- Trück, J.; Prader, S.; Natalucci, G.; Hagmann, C.; Brotschi, B.; Kelly, J.; Bassler, D.; Steindl, K.; Rauch, A.; Baumgartner, M.; et al. Swiss newborn screening for severe T and B cell deficiency with a combined TREC/KREC assay—Management recommendations. Swiss Med. Wkly. 2020, 150, w20254. [Google Scholar] [CrossRef] [PubMed]
- Soomann, M.; Prader, S.; Pachlopnik Schmid, J.; Güngör, T.; Trück, J. Fatal RSV in SCID: The importance of infection prevention despite newborn screening. J. Clin. Immunol. 2023, 43, 554–556. [Google Scholar] [CrossRef] [PubMed]
- Luginbühl, M.; Gaugler, S. The application of fully automated dried blood spot analysis for liquid chromatography-tandem mass spectrometry using the CAMAG DBS-MS 500 autosampler. Clin. Biochem. 2020, 82, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Hijman, A.I.; Konrad, D.; Fingerhut, R. Determining reference ranges for total t4 in dried blood samples for newborn screening. Int. J. Neonatal Screen. 2020, 6, 17. [Google Scholar] [CrossRef] [PubMed]
- Fingerhut, R. Newborn screening for congenital hypothyroidism—Clinical evaluation and comparison of different test kits for the determination of TSH in dried blood samples on two different platforms. Int. J. Neonatal Screen. 2021, 7, 51. [Google Scholar] [CrossRef] [PubMed]
- Fingerhut, R.; Rueegg, C.S.; Imahorn, O.; Pedersen, E.S.L.; Kuehni, C.E.; Gallati, S.; Regamey, N.; Barben, J. Immunoreactive trypsinogen in healthy newborns and infants with cystic fibrosis. Arch. Dis. Child. Fetal Neonatal Ed. 2023, 108, 176–181. [Google Scholar] [CrossRef] [PubMed]
- Rudin, C.; Hirsch, H.H.; Spaelti, R.; Schaedelin, S.; Klimkait, T. Decline of seroprevalence and incidence of congenital toxoplasmosis despite changing prevention policy—Three decades of cord-blood screening in Northwestern Switzerland. Pediatr. Infect. Dis. J. 2018, 37, 1087–1092. [Google Scholar] [CrossRef] [PubMed]
- Baby Health Check. Newborn Screening of the Next Generation. Available online: https://www.babyhealthcheck.com/ch/?lang=en (accessed on 14 May 2023).
- Shoraka, H.R.; Haghdoost, A.A.; Baneshi, M.R.; Bagherinezhad, Z.; Zolala, F. Global prevalence of classic phenylketonuria based on neonatal screening program data: Systematic review and meta-analysis. Clin. Exp. Pediatr. 2020, 63, 34–43. [Google Scholar] [CrossRef] [PubMed]
- Aktuğlu Zeybek, A.Ç. Newborn screening: From the past to the future. Turk. Arch. Pediatr. 2022, 57, 473–475. [Google Scholar] [CrossRef] [PubMed]
- Demirelce, Ö.; Aksungar, F.B.; Saral, N.Y.; Kilercik, M.; Serteser, M.; Unsal, I. Institutional experience of newborn screening for inborn metabolism disorders by tandem MS in the Turkish population. J. Pediatr. Endocrinol. Metab. 2020, 33, 703–711. [Google Scholar] [CrossRef] [PubMed]
- Fidan, Ç.; Örün, H.; Alper, A.B.; Ünver, Ç.N.; Şahin, Ö.C.; Uğurlu, Z.; Akdur, R.; Taruscio, D. Expanded newborn bloodspot screening: Developed country examples and what can be done in Turkey. Intractable Rare Dis. Res. 2022, 11, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Daily Sabah. Turkey Will Soon Screen Newborns for Rare Disease SMA. 28 February 2022. Available online: https://www.dailysabah.com/turkey/turkey-will-soon-screen-newborns-for-rare-disease-sma/news (accessed on 17 May 2023).
- Türkiye SMA Vakfi. Newborn Screening. Available online: https://www.sma.org.tr/en/newborn-screening (accessed on 17 May 2023).
- Genome Web. Turkish MDx Startup Elm Genomics Rolls Out Newborn Sequencing Test. 17 November 2022. Available online: https://www.genomeweb.com/molecular-diagnostics/turkish-mdx-startup-elm-genomics-rolls-out-newborn-sequencing-test#.ZGVwptbMI1g (accessed on 17 May 2023).
- Poyraz, A.; Cansever, M.; Muderris, I.; Patiroglu, T. Neonatal lymphopenia screening is important for early diagnosis of severe combined immunodeficiency. Am. J. Perinatol. 2023, 40, 748–752. [Google Scholar] [CrossRef] [PubMed]
- Bozdogan, S.T.; Mujde, C.; Boga, I.; Sonmezler, O.; Hanta, A.; Rencuzogullari, C.; Ozcan, D.; Altintas, D.U.; Bisgin, A. Current status of genetic diagnosis laboratories and frequency of genetic variants associated with cystic fibrosis through a newborn-screening program in Turkey. Genes 2021, 12, 206. [Google Scholar] [CrossRef] [PubMed]
- Nayir Buyuksahin, H.; Emiralioglu, N.; Ozsezen, B.; Ademhan Tural, D.; Sunman, B.; Guzelkas, I.; Tezel, B.; Dayangaç Erden, D.; Yalçın, E.; Dogru, D.; et al. Cystic fibrosis newborn screening: Five-year experience from a tertiary care center. Pediatr. Pulmonol. 2022, 57, 403–410. [Google Scholar] [CrossRef] [PubMed]
- Ramasli Gursoy, T.; Asfuroglu, P.; Sismanlar Eyuboglu, T.; Aslan, A.T.; Yilmaz, A.I.; Unal, G.; Kibar, B.S.; Pekcan, S.; Hangul, M.; Kose, M.; et al. Evaluation of specificity and sensitivity of IRT/IRT protocol in the cystic fibrosis newborn screening program: 6-year experience of three tertiary centers. Eur. J. Pediatr. 2023, 182, 1067–1076. [Google Scholar] [CrossRef] [PubMed]
- Topuz, Ş.; Taşgıt, A.; Çilesiz, E.; Gökçe, R.; Ülger, D.A.; Oğuz, Ş.S. Views of mothers about national newborn screening tests performed in Turkey. Iran. Red Crescent Med. J. 2021, 23. [Google Scholar] [CrossRef]
- Tin, O.; Zübarioğlu, T.; Cansever, M.Ş.; Kıykım, E.; Aktuğlu-Zeybek, Ç. Maternal Inborn Errors of Metabolism Detected in Expanded Newborn Metabolic Screening. Turk. Arch. Pediatr. 2023, 58, 382–387. [Google Scholar] [CrossRef] [PubMed]
- YouTube. Expanded Neonatal Screening in Ukraine: Implementation of European and World Experience. 28 June 2022. Available online: https://www.youtube.com/watch?v=nJ9MAKiRUcc (accessed on 7 May 2023).
- Loboda, A.; Smiyan, O.; Popov, S.; Petrashenko, V.; Zaitsev, I.; Redko, O.; Zahorodnii, M.; Kasyan, S. Child health care system in Ukraine. Turk. Arch. Pediatr. 2020, 55 (Suppl. S1), 98–104. [Google Scholar] [CrossRef] [PubMed]
- Boyarchuk, O.; Yarema, N.; Kravets, V.; Shulhai, O.; Shymanska, I.; Chornomydz, I.; Hariyan, T.; Volianska, L.; Kinash, M.; Makukh, H. Newborn screening for severe combined immunodeficiency: The results of the first pilot TREC and KREC study in Ukraine with involving of 10,350 neonates. Front. Immunol. 2022, 13, 999664. [Google Scholar] [CrossRef] [PubMed]
- NHS Inform. Blood Spot Test. 7 March 2023. Available online: https://www.nhsinform.scot/healthy-living/screening/newborn-screening/blood-spot-test (accessed on 23 May 2023).
- Bessey, A.; Chilcott, J.B.; Leaviss, J.; Sutton, A. Economic impact of screening for X-linked Adrenoleukodystrophy within a newborn blood spot screening program. Orphanet J. Rare Dis. 2018, 13, 179. [Google Scholar] [CrossRef] [PubMed]
- Bessey, A.; Chilcott, J.; Pandor, A.; Paisley, S. The cost-effectiveness of expanding the UK newborn bloodspot screening programme to include five additional inborn errors of metabolism. Int. J. Neonatal Screen. 2020, 6, 93. [Google Scholar] [CrossRef] [PubMed]
- Bessey, A.; Chilcott, J.; Leaviss, J.; de la Cruz, C.; Wong, R. A cost-effectiveness analysis of newborn screening for severe combined immunodeficiency in the UK. Int. J. Neonatal Screen. 2019, 5, 28. [Google Scholar] [CrossRef]
- Fusco, F.; Chudleigh, J.; Holder, P.; Bonham, J.R.; Southern, K.W.; Simpson, A.; Moody, L.; Olander, E.K.; Chinnery, H.; Morris, S. Delivering positive newborn screening results: Cost analysis of existing practice versus innovative, co-designed strategies from the ReSPoND study. Int. J. Neonatal Screen. 2022, 8, 19. [Google Scholar] [CrossRef] [PubMed]
- Chudleigh, J.; Holder, P.; Fusco, F.; Bonham, J.R.; Bryon, M.; Moody, L.; Morris, S.; Olander, E.K.; Simpson, A.; Chinnery, H.; et al. Co-Designed Strategies for Delivery of Positive Newborn Bloodspot Screening Results to Parents: The ReSPoND Mixed-Methods Study; National Institute for Health and Care Research: Southampton, UK, 2022. [Google Scholar]
- Chudleigh, J.; Chinnery, H.; Bonham, J.R.; Olander, E.; Moody, L.; Simpson, A.; Morris, S.; Ulph, F.; Bryon, M.; Southern, K. Qualitative exploration of health professionals’ experiences of communicating positive newborn bloodspot screening results for nine conditions in England. BMJ Open 2020, 10, e037081. [Google Scholar] [CrossRef]
- Barben, J.; Chudleigh, J. Processing newborn bloodspot screening results for CF. Int. J. Neonatal Screen. 2020, 6, 25. [Google Scholar] [CrossRef] [PubMed]
- Chudleigh, J.; Chinnery, H.; Holder, P.; Carling, R.S.; Southern, K.; Olander, E.; Moody, L.; Morris, S.; Ulph, F.; Bryon, M.; et al. Processing of positive newborn screening results: A qualitative exploration of current practice in England. BMJ Open 2020, 10, e044755. [Google Scholar] [CrossRef] [PubMed]
- Holder, P.; Cheetham, T.; Cocca, A.; Chinnery, H.; Chudleigh, J. Processing of positive newborn screening results for congenital hypothyroidism: A qualitative exploration of current practice in England. Int. J. Neonatal Screen. 2021, 7, 64. [Google Scholar] [CrossRef] [PubMed]
- Chudleigh, J.; Holder, P.; Moody, L.; Simpson, A.; Southern, K.; Morris, S.; Fusco, F.; Ulph, F.; Bryon, M.; Bonham, J.R.; et al. Process evaluation of co-designed interventions to improve communication of positive newborn bloodspot screening results. BMJ Open 2021, 11, e050773. [Google Scholar] [CrossRef] [PubMed]
- Seddon, L.; Dick, K.; Carr, S.B.; Balfour-Lynn, I.M. Communicating cystic fibrosis newborn screening results to parents. Eur. J. Pediatr. 2021, 180, 1313–1316. [Google Scholar] [CrossRef] [PubMed]
- White, A.L.; Boardman, F.; McNiven, A.; Locock, L.; Hinton, L. Absorbing it all: A meta-ethnography of parents’ unfolding experiences of newborn screening. Soc. Sci. Med. 2021, 287, 114367. [Google Scholar] [CrossRef] [PubMed]
- Schlüter, D.K.; Southern, K.W.; Dryden, C.; Diggle, P.; Taylor-Robinson, D. Impact of newborn screening on outcomes and social inequalities in cystic fibrosis: A UK CF registry-based study. Thorax 2020, 75, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Chudleigh, J.; Chinnery, H. Psychological Impact of NBS for CF. Int. J. Neonatal Screen. 2020, 6, 27. [Google Scholar] [CrossRef] [PubMed]
- Boardman, F.; Clark, C. ‘We’re kind of like genetic nomads’: Parents’ experiences of biographical disruption and uncertainty following in/conclusive results from newborn cystic fibrosis screening. Soc. Sci. Med. 2022, 301, 114972. [Google Scholar] [CrossRef] [PubMed]
- Doull, I.; Course, C.W.; Hanks, R.E.; Southern, K.W.; Forton, J.T.; Thia, L.P.; Moat, S.J. Cystic fibrosis newborn screening: The importance of bloodspot sample quality. Arch. Dis. Child. 2021, 106, 253–257. [Google Scholar] [CrossRef] [PubMed]
- Kinsella, S.; Hopkins, H.; Cooper, L.; Bonham, J.R. A public dialogue to inform the use of wider genomic testing when used as part of newborn screening to identify cystic fibrosis. Int. J. Neonatal Screen. 2022, 8, 32. [Google Scholar] [CrossRef] [PubMed]
- Burki, T. UK explores whole-genome sequencing for newborn babies. Lancet 2022, 400, 260–261. [Google Scholar] [CrossRef] [PubMed]
- Pichini, A.; Ahmed, A.; Patch, C.; Bick, D.; Leblond, M.; Kasperaviciute, D.; Deen, D.; Wilde, S.; Garcia Noriega, S.; Matoko, C.; et al. Developing a national newborn genomes program: An approach driven by ethics, engagement and co-design. Front. Genet. 2022, 13, 866168. [Google Scholar] [CrossRef]
- Ulph, F.; Bennett, R. Psychological and ethical challenges of introducing whole genome sequencing into routine newborn screening: Lessons learned from existing newborn screening. New Bioeth. 2023, 29, 52–74. [Google Scholar] [CrossRef] [PubMed]
- Horton, R.; Lucassen, A. Ethical issues raised by new genomic technologies: The case study of newborn genome screening. Camb. Prism. Precis. Med. 2023, 1, e2. [Google Scholar] [CrossRef] [PubMed]
- Harris, M. UK NSC Recommends Introduction of Screening for Tyrosinaemia in Newborns. GOV.UK. Blog UK National Screening Committee. 2 February 2023. Available online: https://nationalscreening.blog.gov.uk/2023/02/02/uk-nsc-recommends-introduction-of-screening-for-tyrosinaemia-in-newborns/ (accessed on 27 May 2023).
- Elliman, D.A.C.; Gennery, A.R. Newborn screening for severe combined immunodeficiency—Coming to a region near you soon. Clin. Exp. Immunol. 2021, 205, 343–345. [Google Scholar] [CrossRef] [PubMed]
- Elliman, D. Study design for an evaluation of newborn screening for SCID in the UK. Int. J. Neonatal Screen. 2022, 8, 4. [Google Scholar] [CrossRef] [PubMed]
- Adams, S.P.; Gravett, E.; Kent, N.; Kricke, S.; Ifederu, A.; Scoto, M.; Samsuddin, S.; Muntoni, F. Screening of neonatal UK dried blood spots using a duplex SMN1 screening assay. Int. J. Neonatal Screen. 2021, 7, 69. [Google Scholar] [CrossRef]
- University of Oxford—News and Events. First UK Pilot Study of Newborn Screening for Spinal Muscular Atrophy Launched in Oxford. 11 March 2022. Available online: https://www.ox.ac.uk/news/2022-03-11-first-uk-pilot-study-newborn-screening-spinal-muscular-atrophy-launched-oxford (accessed on 23 March 2021).
- Gillingwater, T.H. Maximising returns: Combining newborn screening with gene therapy for spinal muscular atrophy. J. Neurol. Neurosurg. Psychiatry 2021, 92, 1252. [Google Scholar] [CrossRef] [PubMed]
- Gillingwater, T.H.; McWilliam, C.; Horrocks, I.; McWilliam, K.; Hamilton, M.; Fletcher, E.; Williams, N.; Smith, S.; Parson, S.H. A call to introduce newborn screening for spinal muscular atrophy (SMA) in Scotland. Scott. Med. J. 2022, 67, 46–47. [Google Scholar] [CrossRef] [PubMed]
- Cunningham-Burley, S.; McCartney, D.L.; Campbell, A.; Flaig, R.; Orange, C.E.L.; Porteous, C.; Aitken, M.; Mulholland, C.; Davidson, S.; McCafferty, S.M.; et al. Feasibility and ethics of using data from the Scottish newborn blood spot archive for research. Commun. Med. 2022, 2, 126. [Google Scholar] [CrossRef]
- Wu, T.H.Y.; Brown, H.A.; Church, H.J.; Kershaw, C.J.; Hutton, R.; Egerton, C.; Cooper, J.; Tylee, K.; Cohen, R.N.; Gokhale, D.; et al. Improving newborn screening test performance for metachromatic leukodystrophy: Recommendation from a pre-pilot study that identified a late-infantile case for treatment. Mol. Genet. Metab. 2024, 142, 108349. [Google Scholar] [CrossRef] [PubMed]
- Henderson, E.M.A.; Tappin, D.; Young, D.; Favretto, D.; Mactier, H. Assessing maternal alcohol consumption in pregnancy: Does phosphatidylethanol measured from day 5 newborn blood spot cards have any value? An observational, population-based study. Arch. Dis. Child. 2023, 108, 36–41. [Google Scholar] [CrossRef]
- Cantley, N.W.P.; Barski, R.; Kemp, H.; Hogg, S.L.; Wu, H.Y.T.; Bowron, A.; Collingwood, C.; Cundick, J.; Hart, C.; Shakespeare, L.; et al. Incidental detection of classical galactosemia through newborn screening for phenylketonuria: A 10-year retrospective audit to determine the efficacy of this approach. Int. J. Neonatal Screen. 2024, 10, 2. [Google Scholar] [CrossRef] [PubMed]
- Wilsdon, T.; Saada, R.; Ferguson, M. A Landscape Assessment of Newborn Screening in Europe. Technology Network Diagnostics. 4 April 2022. Available online: https://www.technologynetworks.com/diagnostics/blog/a-landscape-assessment-of-newborn-screening-in-europe-359964 (accessed on 20 January 2024).
- United Nations—ECLAC. Member States and Associate Members. Available online: https://www.cepal.org/en/about/member-states (accessed on 11 March 2023).
- United Nations. About Us [Economic Commission for Latin America and the Caribbean—ECLAC]. Available online: https://www.cepal.org/en/headquarters-and-offices/eclac-caribbean/about-us (accessed on 23 February 2023).
- Borrajo, G.J.C. Newborn screening in Latin America: A brief overview of the state of the art. Am. J. Med. Genet. C Semin. Med. Genet. 2021, 187, 322–328. [Google Scholar] [CrossRef]
- Giugliani, R.; Castillo Taucher, S.; Hafez, S.; Oliveira, J.B.; Rico-Restrepo, M.; Rozenfeld, P.; Zarante, I.; Gonzaga-Jauregui, C. Opportunities and challenges for newborn screening and early diagnosis of rare diseases in Latin America. Front. Genet. 2022, 13, 1053559. [Google Scholar] [CrossRef] [PubMed]
- Knight-Madden, J.; Lee, K.; Elana, G.; Elenga, N.; Marcheco-Teruel, B.; Keshi, N.; Etien ne-Julan, M.; King, L.; Asnani, M.; Romana, M.; et al. Newborn screening for sickle cell disease in the Caribbean: An update of the present situation and of the disease prevalence. Int. J. Neonatal Screen. 2019, 5, 5. [Google Scholar] [CrossRef] [PubMed]
- Borrajo, G.; Pistaccio, L.; Parente, M. External quality assurance program for neonatal screening (PEEC-PN): A 15-year trajectory. J. Inborn Errors Metab. Screen. 2015, 3, 56. Available online: http://www.jiems-journal.org/storage/files/20190513155803_2015.pdf (accessed on 3 February 2024).
- Borrajo, G.J.C. Programa de evaluación externa de calidad—Pesquisa neonatal. Fundación Bioquímica Argentina, La Plata, Argentina. Encuesta # 136—Junio 2023. Personal communication, 2023. [Google Scholar]
- Borrajo, G.J.C.; Pistaccio, L.G.; Parente, M.J. External quality assurance program for neonatal screening in Latin America: Pitfalls and errors in the measurements and reporting results. J. Inborn Errors Metab. Screen. 2019. Available online: https://jiems-journal.org/storagefiles/2019.pdf (accessed on 3 February 2024).
- Borrajo, G.J.C.; Doña, V.V. How long are residual newborn screening specimens useful for retesting when stored in suboptimal and uncontrolled conditions of temperature and humidity? J. Inborn Errors Metab. Screen. 2021, 9, e20210010. [Google Scholar] [CrossRef]
- Borrajo, G.J.C. Newborn screening for phenylketonuria: Latin American consensus guidelines. J. Inborn Errors Metab. Screen. 2016, 4. [Google Scholar] [CrossRef]
- Chiesa, A.; Spécola, N.; Poubel, M.; Vela-Amieva, M.; Jurecki, E.; Vilela, D.R.; Mesojedovas, D.; Carneiro, G.C.; Eiroa, H.; Nakamura, K.H.; et al. Adherence to PKU guidelines among patients with phenylketonuria: A cross-sectional national multicenter survey-based study in Argentina, Brazil, and Mexico. Mol. Genet. Metab. Rep. 2023, 38, 101026. [Google Scholar] [CrossRef] [PubMed]
- Poloni, S.; Dos Santos, B.B.; Chiesa, A.; Specola, N.; Pereyra, M.; Saborío-Rocafort, M.; Salazar, M.F.; Leal-Witt, M.J.; Castro, G.; Peñaloza, F.; et al. Current practices and challenges in the diagnosis and management of PKU in Latin America: A multicenter survey. Nutrients 2021, 13, 2566. [Google Scholar] [CrossRef] [PubMed]
- Pessoa, A.L.S.; Martins, A.M.; Ribeiro, E.M.; Specola, N.; Chiesa, A.; Vilela, D.; Jurecki, E.; Mesojedovas, D.; Schwartz, I.V.D. Burden of phenylketonuria in Latin American patients: A systematic review and meta-analysis of observational studies. Orphanet J. Rare Dis. 2022, 17, 302. [Google Scholar] [CrossRef] [PubMed]
- Huttle, A.; Maestre, G.E.; Lantigua, R.; Green, N.S. Sickle cell in Latin America and the United States. Pediatr. Blood Cancer 2015, 62, 1131–1136, Erratum in Pediatr. Blood Cancer 2015, 62, 1493. [Google Scholar] [CrossRef] [PubMed]
- Silva Filho, L.V.; Castaños, C.; Ruíz, H.H. Cystic fibrosis in Latin America-improving the awareness. J. Cyst. Fibros. 2016, 15, 791–793. [Google Scholar] [CrossRef] [PubMed]
- Hoyos Bachiloglu, R.; Sotomayor, F.C.; Poli, H.C. Inmunodeficiencia combinada severa: Es tiempo de su detección precoz [Severe combined immunodeficiency: The time for newborn screening has come]. Rev. Chil. Pediatr. 2019, 90, 581–588. (In Spanish) [Google Scholar] [PubMed]
- Bustamante Ogando, J.C.; Partida Gaytán, A.; Aldave Becerra, J.C.; Álvarez Cardona, A.; Bezrodnik, L.; Borzutzky, A.; Blancas Galicia, L.; Cabanillas, D.; Condino-Neto, A.; De Colsa Ranero, A.; et al. Latin American consensus on the supportive management of patients with severe combined immunodeficiency. J. Allergy Clin. Immunol. 2019, 144, 897–905. [Google Scholar] [CrossRef]
- Kubaski, F.; Sousa, I.; Amorim, T.; Pereira, D.; Trometer, J.; Souza, A.; Ranieri, E.; Polo, G.; Burlina, A.; Brusius-Facchin, A.C.; et al. Neonatal screening for MPS disorders in Latin America: A survey of pilot initiatives. Int. J. Neonatal Screen. 2020, 6, 90. [Google Scholar] [CrossRef] [PubMed]
- Kubaski, F.; Sousa, I.; Amorim, T.; Pereira, D.; Silva, C.; Chaves, V.; Brusius-Facchin, A.C.; Netto, A.B.O.; Soares, J.; Vairo, F.; et al. Pilot study of newborn screening for six lysosomal diseases in Brazil. Mol. Genet. Metab. 2023, 140, 107654. [Google Scholar] [CrossRef] [PubMed]
- Colomé-Hidalgo, M.; Campos, J.D.; de Miguel, Á.G. Exploring wealth-related inequalities in maternal and child health coverage in Latin America and the Caribbean. BMC Public Health 2021, 21, 115. [Google Scholar] [CrossRef] [PubMed]
- Cabello, J.F.; Novoa, F.; Huff, H.V.; Colombo, M. Expanded newborn screening and genomic sequencing in Latin America and the resulting social justice and ethical considerations. Int. J. Neonatal Screen. 2021, 7, 6. [Google Scholar] [CrossRef]
- Brosco, J.P.; Castellands, T.H.; Gelpi, A. Newborn screening in Latin America: A window on the evolution of health policy. OBM Genet. 2022, 6, 146. [Google Scholar] [CrossRef]
- Sola, A.; Rodríguez, S.; Young, A.; Lemus Varela, L.; Villamayor, R.M.; Cardetti, M.; Pleitez Navarrete, J.; Favareto, M.V.; Lima, V.; Baquero, H.; et al. CCHD screening implementation efforts in Latin American countries by the Ibero American Society of Neonatology (SIBEN). Int. J. Neonatal Screen. 2020, 6, 21. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.C.; Zin, A.; Gilbert, C. Retinopathy of prematurity prevention, screening and treatment programmes: Progress in South America. Semin Perinatol. 2019, 43, 348–351. [Google Scholar] [CrossRef]
- Gerner de Garcia, B.; Gaffney, C.; Chacon, S.; Gaffney, M. Overview of newborn hearing screening activities in Latin America. Rev. Panam. Salud Publica 2011, 29, 145–152. [Google Scholar] [PubMed]
- Neumann, K.; Mathmann, P.; Chadha, S.; Euler, H.A.; White, K.R. Newborn hearing screening benefits children, but global disparities persist. J. Clin. Med. 2022, 11, 271. [Google Scholar] [CrossRef]
- Knight-Madden, J.; Romana, M.; Villaescusa, R.; Reid, M.; Etienne-Julan, M.; Boutin, L.; Elana, G.; Elenga, N.; Wheeler, G.; Lee, K.; et al. CAREST--Multilingual regional integration for health promotion and research on sickle cell disease and thalassemia. Am. J. Public Health 2016, 106, 851–853. [Google Scholar] [CrossRef] [PubMed]
- Langlois, E.V.; Mancuso, A.; Elias, V.; Reveiz, L. Embedding implementation research to enhance health policy and systems: A multi-country analysis from ten settings in Latin America and the Caribbean. Health Res. Policy Syst. 2019, 17, 85. [Google Scholar] [CrossRef] [PubMed]
- Verberne, E.A.; Ecury-Goossen, G.M.; Manshande, M.E.; Ponson-Wever, M.; de Vroomen, M.; Tilanus, M.; Mannens, M.M.A.M.; Henneman, L.; van Haelst, M.M. Clinical and community genetics services in the Dutch Caribbean. J. Community Genet. 2021, 12, 497–501. [Google Scholar] [CrossRef] [PubMed]
- Bidondo, M.P.; Groisman, B.; Barbero, P.; Liascovich, R. Public health approach to birth defects: The Argentine experience. J. Community Genet. 2015, 6, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Vishnopolska, S.A.; Turjanski, A.G.; Herrera Piñero, M.; Groisman, B.; Liascovich, R.; Chiesa, A.; Marti, M.A. Genetics and genomic medicine in Argentina. Mol. Genet. Genomic Med. 2018, 6, 481–491. [Google Scholar] [CrossRef] [PubMed]
- Cotignola, J.; Rozental, S.; Buzzalino, N.; Dain, L. Genetics and genomic medicine in Argentina. Mol. Genet. Genomic Med. 2019, 7, e00571. [Google Scholar] [CrossRef]
- Dratler, G.A. Pesquisa neonatal: Antecedentes, presente y perspectivas. Med. Infant. 2022, XXIX, pp. 48–60. Available online: https://medicinainfantil.org.ar/images/stories/volumen/2022/xxix_1_048.pdf (accessed on 3 February 2024).
- Borrajo, G.J.C. Estado actual de la pesquisa neonatal en Argentina. Acta Bioquím Clín Latinoam. Acta de Congreso 2. 2022, p. 44. Available online: https://calilab.fba.org.ar/wp-content/uploads/2022/11/Acta-Bioq-Clin-Lat-Actas-de-Congreso-2-Calilab-2022.pdf (accessed on 3 February 2024).
- Boletin Integrado de Vigilancia BIV 545—SE 15. Informe Especial: Pesquisa Neonatal de Enfermedades Congénitas (Ley Nacional 26279). Dirección de Epidemiología. Ministerio de Salud Nacional. 2021, pp. 49–59. Available online: https://bancos.salud.gob.ar/recurso/boletin-integrado-de-vigilancia-n545-se15-2021 (accessed on 3 February 2024).
- Teper, A.; Smithuis, F.; Rodríguez, V.; Salvaggio, O.; Maccallini, G.; Aranda, C.; Lubovich, S.; Zaragoza, S.; García-Bournissen, F. Comparison between two newborn screening strategies for cystic fibrosis in Argentina: IRT/IRT versus IRT/PAP. Pediatr. Pulmonol. 2021, 56, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Seoane, B.; Suárez, S. Congenital adrenal hyperplasia, before and after neonatal screening. 13 years of experience in Santa Cruz, Bolivia. J. Inborn Errors Metab. Screen. 2022. Available online: https://www.jiems-journal.org/storage/files/20220505102450_special-supplement-2022_compressed.pdf (accessed on 1 April 2024).
- Jove, A.; Salvatierra, I.; Siacar, S. Congenital hypothyroidism. Progression of the neonatal screening program in the Department of La Paz, Bolivia. J. Inborn Errors Metab. Screen. 2015, 3, 14. Available online: http://www.jiems-journal.org/storage/files/20190513155803_2015.pdf (accessed on 3 February 2024).
- Cortez Copa, P.; Andrade Cruz, N.; Limachi Vazquez, R.; Pardo Boyan, J.; Patiño Azuga, K.; Bravo Villalta, H.; Larrandaburu, M. Newborn screening in Bolivia: A regional initiative 2017 to 2022. Program & Abstract Book. In Proceedings of the International Conference on Birth Defects and Disabilities in the Developing World—ICBD 2023, Santiago, Chile, 1–4 March 2023; p. 121. [Google Scholar]
- Gov.Br—Ministério da Saúde. Programa Nacional da Triagem Neonatal. Available online: https://www.gov.br/saude/pt-br/composicao/saes/sangue/pntn (accessed on 30 August 2023).
- Kopacek, C.; de Castro, S.M.; Prado, M.J.; da Silva, C.M.; Beltrão, L.A.; Spritzer, P.M. Neonatal screening for congenital adrenal hyperplasia in Southern Brazil: A population based study with 108,409 infants. BMC Pediatr. 2017, 17, 22. [Google Scholar] [CrossRef] [PubMed]
- Jusbrasil. Legislação—Lei Nº 14.154, de 26 de Maio de 2021. Law Creating a National Newborn Screening Program. Available online: https://www.jusbrasil.com.br/legislacao/1218254408/lei-14154-21 (accessed on 1 September 2023).
- Castro, S.M.; Wiest, P.; Spritzer, P.M.; Kopacek, C. The impact of neonatal 17-hydroxyprogesterone cutoff determination in a public newborn screening program for congenital adrenal hyperplasia in Southern Brazil: 3 years’ experience. Endocr. Connect. 2023, 12, e230162. [Google Scholar] [CrossRef] [PubMed]
- Pimentel dos Santos, H.; Bonini Domingos, C.R.; Simone Martins de Castro, S. Twenty years of neonatal screening for sickle cell disease in Brazil: The challenges of a continental country with high genetic heterogeneity. J. Inborn Errors Metab. Screen. 2021, 9, e20210002. [Google Scholar] [CrossRef]
- Silva-Pinto, A.C.; Alencar de Queiroz, M.C.; Antoniazzo Zamaro, P.J.; Arruda, M.; Pimentel Dos Santos, H. The neonatal screening program in Brazil, focus on sickle cell disease (SCD). Int. J. Neonatal Screen. 2019, 5, 11. [Google Scholar] [CrossRef] [PubMed]
- Cândido-Bacani, P.M.; Grilo, P.M.S.; Ramos, V.D.S.; Zanchin, M.; Pereira, I.C.; Oliveira, J.S.P.; Bacani, V.M.; Belini Junior, E. Incidence of hemoglobinopathies and spatialization of newborns with sickle cell trait in Mato Grosso do Sul, Brazil. Einstein 2022, 20, eAO6535. [Google Scholar] [CrossRef] [PubMed]
- Maciel, L.M.Z.; Magalhães, P.K.R.; Ciampo, I.R.L.D.; Sousa, M.L.B.; Fernandes, M.I.M.; Sawamura, R.; Bittar, R.R.; Molfetta, G.A.; Silva Júnior, W.A.D. The first five-year evaluation of cystic fibrosis neonatal screening program in São Paulo State, Brazil. Cad. Saude Publica 2020, 36, e00049719. [Google Scholar] [CrossRef] [PubMed]
- Sadigurschi, G.; Vaz Micherino, B.; Assunção Mendes da Cunha, M.B.; Antão Paiva, C.L.; da Silva, E.; Sá, G.R. Analysis of national coverage of neonatal cystic fibrosis screening in Brazil from 2008 to 2017. J. Matern. Fetal Neonatal Med. 2022, 35, 5204–5209. [Google Scholar] [CrossRef] [PubMed]
- Godoy, C.; Paixão, D.C.; Boa-Sorte, N.C.A.; Amorim, T.; da Silva Filho, L.V.R.F.; Souza, E.L. Five-year performance analysis of a cystic fibrosis newborn screening program in northeastern Brazil. J. Pediatr. 2023, 99, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Matos, D.M.; Ramalho, R.J.; Carvalho, B.M.; Almeida, M.A.; Passos, L.F.; Vasconcelos, T.T.; Melo, E.V.; Oliveira, C.R.; Santos, E.G.; Resende, K.F.; et al. Evolution to permanent or transient conditions in children with positive neonatal TSH screening tests in Sergipe, Brazil. Arch. Endocrinol. Metab. 2016, 60, 450–456. [Google Scholar] [CrossRef]
- Gumes-Felix, H.M.; Ramalho, R.J.R.; Melo, E.V.; Matos, D.M.; Menezes, N.V.; Oliveira, C.R.P.; Campos, V.C.; Santos, E.G.; da S Marques, D.; Vaz Dos Santos, B.; et al. Predictive factors for the diagnosis of permanent congenital hypothyroidism and its temporal changes in Sergipe, Brazil—A real-life retrospective study. Arch. Endocrinol. Metab. 2023, 67, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, J.A.R.; Pereira, R.M.; De Lacerda, L. Pubertal development and adult height in patients with congenital hypothyroidism detected by neonatal screening in southern Brazil. J. Pediatr. Endocrinol. Metab. 2020, 33, 1449–1455. [Google Scholar] [CrossRef] [PubMed]
- Martins, A.M.; Pessoa, A.L.S.; Quesada, A.A.; Ribeiro, E.M. Unmet needs in PKU and the disease impact on the day-to-day lives in Brazil: Results from a survey with 228 patients and their caregivers. Mol. Genet. Metab. Rep. 2020, 24, 100624. [Google Scholar] [CrossRef] [PubMed]
- Garcia, D.F.; Camelo, J.S., Jr.; Molfetta, G.A.; Turcato, M.; Souza, C.F.; Porta, G.; Steiner, C.E.; Silva, W.A., Jr. Clinical profile and molecular characterization of galactosemia in Brazil: Identification of seven novel mutations. BMC Med. Genet. 2016, 17, 39. [Google Scholar] [CrossRef] [PubMed]
- Herber, S.; Schwartz, I.V.; Nalin, T.; Oliveira Netto, C.B.; Camelo Junior, J.S.; Santos, M.L.; Ribeiro, E.M.; Schüler-Faccini, L.; Souza, C.F. Maple syrup urine disease in Brazil: A panorama of the last two decades. J. Pediatr. 2015, 91, 292–298. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, N.O.; Januário, J.N.; Felix, G.L.P.; Nolasco, D.M.; Ladeira, R.V.P.; Del Castillo, D.M.; Starling, A.L.P.; Norton, R.C.; Viana, M.B. Frequency of biotinidase gene variants and incidence of biotinidase deficiency in the newborn screening program in Minas Gerais, Brazil. J. Med. Screen. 2020, 27, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, N.O.; Del Castillo, D.M.; Januário, J.N.; Starling, A.L.P.; Arantes, R.R.; Norton, R.C.; Viana, M.B. Novel mutations causing biotinidase deficiency in individuals identified by the newborn screening program in Minas Gerais, Brazil. Am. J. Med. Genet. A 2019, 179, 978–982. [Google Scholar] [CrossRef] [PubMed]
- de Miranda, M.C.; Haddad, L.B.P.; Trindade, E.; Cassenote, A.; Hayashi, G.Y.; Damiani, D.; Costa, F.C.; Madureira, G.; de Mendonca, B.B.; Bachega, T.A.S.S. The cost-effectiveness of congenital adrenal hyperplasia newborn screening in Brazil: A comparison between screened and unscreened cohorts. Front. Pediatr. 2021, 9, 659492. [Google Scholar] [CrossRef] [PubMed]
- Mallmann, M.B.; Tomasi, Y.T.; Boing, A.F. Neonatal screening tests in Brazil: Prevalence rates and regional and socioeconomic inequalities. J. Pediatr. 2020, 96, 487–494. [Google Scholar] [CrossRef] [PubMed]
- Paraguassú-Chaves, C.A.; Moreira Dantas, L.R.; de Almeida, F.M.; Serruta, A.J.; Soares Leite, J.M.; da Luz Neto, L.S.; Neves Calderaro, I.F.; Barcellos Xavier, D.F. Incidence of congenital toxoplasmosis in newborn infant in the Western Amazon, Brazil. Int. J. Adv. Eng. Res. Sci. 2019, 6, 659–670. [Google Scholar] [CrossRef]
- Kanegae, M.P.P.; Barreiros, L.A.; Mazzucchelli, J.T.L.; Hadachi, S.M.; Guilhoto, L.M.F.F.; Acquesta, A.L.; Genov, I.R.; Holanda, S.M.; Guilhoto, L.M.F.F.; Acquesta, A.L.; et al. Neonatal screening for severe combined immunodeficiency in Brazil. J. Pediatr. 2016, 92, 374–380. [Google Scholar] [CrossRef]
- Meehan, C.; Bonfim, C.; Dasso, J.F.; Costa-Carvalho, B.T.; Condino-Neto, A.; Walter, J. In time: The value and global implications of newborn screening for severe combined immunodeficiency. Rev. Paul. Pediatr. 2018, 36, 388–397, Erratum in Rev. Paul. Pediatr. 2020, 38, e00020. [Google Scholar] [CrossRef]
- Barreiros, L.A.; Sousa, J.L.; Geier, C.; Leiss-Piller, A.; Kanegae, M.P.P.; França, T.T.; Boisson, B.; Lima, A.M.; Costa-Carvalho, B.T.; Aranda, C.S.; et al. SCID and other inborn errors of immunity with low TRECs—The Brazilian Experience. J. Clin. Immunol. 2022, 42, 1171–1192. [Google Scholar] [CrossRef] [PubMed]
- Bravo, H.; Neto, E.C.; Schulte, J.; Pereira, J.; Filho, C.S.; Bittencourt, F.; Sebastião, F.; Bender, F.; de Magalhães, A.P.S.; Guidobono, R.; et al. Investigation of newborns with abnormal results in a newborn screening program for four lysosomal storage diseases in Brazil. Mol. Genet. Metab. Rep. 2017, 12, 92–97. [Google Scholar] [CrossRef]
- Camargo Neto, E.; Schulte, J.; Pereira, J.; Bravo, H.; Sampaio-Filho, C.; Giugliani, R. Neonatal screening for four lysosomal storage diseases with a digital microfluidics platform: Initial results in Brazil. Genet. Mol. Biol. 2018, 41, 414–416. [Google Scholar] [CrossRef] [PubMed]
- Bender, F.; Burin, M.G.; Tirelli, K.M.; Medeiros, F.; Bitencourt, F.H.; Civallero, G.; Kubaski, F.; Bravo, H.; Daher, A.; Carnier, V.; et al. Newborn screening for lysosomal disorders in Brazil: A pilot study using customized fluorimetric assays. Genet. Mol. Biol. 2020, 43, e20180334. [Google Scholar] [CrossRef]
- Romanelli Tavares, V.L.; Monfardini, F.; Lourenço, N.C.V.; da Rocha, K.M.; Weinmann, K.; Pavanello, R.; Zatz, M. Newborn Screening for 5q spinal muscular atrophy: Comparisons between real-time PCR methodologies and cost estimations for future implementation programs. Int. J. Neonatal Screen. 2021, 7, 53. [Google Scholar] [CrossRef]
- Ferreira, I.R.; Costa, R.A.; Gomes, L.H.F.; Dos Santos Cunha, W.D.; Tyszler, L.S.; Freitas, S.; Llerena Junior, J.C.; de Vasconcelos, Z.F.M.; Nicholls, R.D.; Guida, L.D.C. A newborn screening pilot study using methylation-sensitive high resolution melting on dried blood spots to detect Prader-Willi and Angelman syndromes. Sci. Rep. 2020, 10, 3026. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, T.D.S.; Dutra, M.R.P.; Cavalcanti, H.G. Newborn hearing screening: Association between coverage, and the availability of speech therapists and equipment in Brazil. Codas 2021, 33, e20190259. (In Portuguese) [Google Scholar] [CrossRef] [PubMed]
- Vernier, L.S.; Cazella, S.C.; Levandowski, D.C. Neonatal hearing screening: Protocols, obstacles and perspectives of speech therapists in Brazil—10 years of Brazilian Federal Law 12,303/2010. Codas 2022, 34, e20200331. (In Portuguese) [Google Scholar] [CrossRef] [PubMed]
- Marinho, A.C.A.; Pereira, E.C.S.; Torres, K.K.C.; Miranda, A.M.; Ledesma, A.L.L. Evaluation of newborn hearing screening program. Rev. Saúde Pública 2020, 54, 44. [Google Scholar] [CrossRef] [PubMed]
- Isaac, M.L.; Manfredi, A.K.S.; Cavalcante, J.M.S.; Carnevale-Silva, A.; Fowler, K.B.; Boppana, S.B.; Britt, W.J.; Mussi-Pinhata, M.M. Contribution of congenital cytomegalovirus infection to permanent hearing loss in a highly seropositive population: The Brazilian cytomegalovirus hearing and maternal secondary infection study. Clin. Infect. Dis. 2020, 70, 1379–1384. [Google Scholar] [CrossRef] [PubMed]
- Valiente, A.G.; Faundez, A.; Cabello, J.F.; Lobo, G.; Berrios, C.G.; Valdebenito, S.; Cornejo, V. Expanded newborn screening in Chile (26 conditions). Results of a pilot program. J. Inborn Errors Metab. Screen. 2019, Special Suppl. SLEIMPN 2019, 17–18. [Google Scholar]
- Hoyos-Bachiloglu, R.; Rojas, J.; Borzutzky, A.; Hernández, P.; Vinet, A.M.; Bustos, P.; Fernández, F.; Lagos, M.; Strickler, A.; Marinovic, M.A.; et al. Severe combined immunodeficiency, report of Chilean patients diagnosed during the 1999–2020 period. Rev. Chil. Pediatr. 2020, 91, 908–916. [Google Scholar] [PubMed]
- Chuang Chuang, Á.; Ramos Hernández, H.; Zelada Bacigualupo, Ú.; López Castillo, M.T.; Villavicencio Landeros, L.; Peret, L.M.; González Muñoz, C.; Barría Espinoza, T.; Izquierdo Copiz, G. Congenital cytomegalovirus infection screening in high risk newborn. Rev. Chilena Infectol. 2021, 38, 45–53. (In Spanish) [Google Scholar] [CrossRef] [PubMed]
- Núñez, A.; Bedregal, P.; Becerra, C.; Grob, L.F. Neurodevelopmental assessment of patients with congenital hypothyroidism. Rev. Med. Chil. 2017, 145, 1579–1587. (In Spanish) [Google Scholar] [CrossRef][Green Version]
- Grob, F.; Bedregal, P.; Viviani, P.; Torres, C.; Becerra, C.; Leyton, B. Cognitive outcomes in congenital hypothyroidism: Impact of socioeconomic status—A case-control Study. Horm. Res. Paediatr. 2022, 95, 224–232. [Google Scholar] [CrossRef] [PubMed]
- Leal-Witt, M.J.; Salazar, M.F.; Peñaloza, F.; Castro, G.; Hamilton, V.; Arias, C.; Peredo, P.; Valiente, A.; De la Parra, A.; Cabello, J.F.; et al. Update on dietary compliance, nutritional status, and neuropsychological functioning in the Chilean phenylketonuria cohort. J. Inborn Errors Metab. Screen. 2021, 9, e20210003. [Google Scholar] [CrossRef]
- Hamilton, V.; Santa María, L.; Fuenzalida, K.; Morales, P.; Desviat, L.R.; Ugarte, M.; Pérez, B.; Cabello, J.F.; Cornejo, V. Characterization of phenyalanine hydroxylase gene mutations in Chilean PKU patients. JIMD Rep. 2018, 42, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, I.V.D.; Cornejo, V.; Hamilton, V.; Castro, G.; Sperb-Ludwig, F.; Borges, E.S.; Camelo, J.S., Jr. Molecular basis of various forms of maple syrup urine disease in Chilean patients. Mol. Genet. Genom. Med. 2021, 9, e1616. [Google Scholar] [CrossRef]
- Valera Antequera, D.; Montealegre Páez, A.L.; Bermúdez, A.; García Robles, R. Importancia de una propuesta para la implementación de un programa de tamizaje neonatal expandido en Colombia. Rev. Med. 2019, 27, 21–33. [Google Scholar] [CrossRef]
- Ministerio de Salud y Protección Social. Neonatal Screening Is a Transcendental Measure of Equity in Colombia: President Duque. Press Release, Bogotá DC. 14 August 2019. Available online: https://www.minsalud.gov.co/Paginas/Neonatal-Screening-is-a-Transcendental-Measure-of-Equity-in-Colombia-President-Duque.aspx (accessed on 21 January 2023).
- Peñaloza, M. What Is Screening and Why Does It Benefit Babies? LatinAmerican Post. 14 May 2019. Available online: https://latinamericanpost.com/28050-what-is-screening-and-why-does-it-benefit-babies (accessed on 3 February 2024).
- Céspedes, N.; Valencia, A.; Echeverry, C.A.; Arce-Plata, M.I.; Colón, C.; Castiñeiras, D.E.; Hurtado, P.M.; Cocho, J.A.; Herrera, S.; Arévalo-Herrera, M. Reference values of amino acids, acylcarnitines and succinylacetone by tandem mass spectrometry for use in newborn screening in southwest Colombia. Colomb. Medica 2017, 48, 113–119. [Google Scholar] [CrossRef]
- Bermúdez, A.; Robayo, D.; Porras, G.; Acosta, A.M. Amino acids and acylcarnitines reference values for neonatal screening of inborn errors of metabolism in Colombia by tandem mass spectrometry. J. Inborn Errors Metab. Screen. 2021, 9, e20210012. [Google Scholar] [CrossRef]
- Uribe-Ardila, A.; Ramirez-Borda, J.; Ayala, A. Twenty years of Colombian experience with enzymatic screening in patients with features of mucopolysaccharidosis. JIMD Rep. 2022, 63, 475–483. [Google Scholar] [CrossRef] [PubMed]
- Puentes-Tellez, M.A.; Lerma-Barbosa, P.A.; Garzón-Jaramillo, R.G.; Suarez, D.A.; Espejo-Mojica, A.J.; Guevara, J.M.; Echeverri, O.Y.; Solano-Galarza, D.; Uribe-Ardila, A.; Alméciga-Díaz, C.J. A perspective on research, diagnosis, and management of lysosomal storage disorders in Colombia. Heliyon 2020, 6, e03635. [Google Scholar] [CrossRef] [PubMed]
- Londoño Trujillo, D.; Sandoval Reyes, N.F.; Taborda Restrepo, A.; Chamorro Velasquez, C.L.; Dominguez Torres, M.T.; Romero Ducuara, S.V.; Troncoso Moreno, G.A.; Aranguren Bello, H.C.; Fonseca Cuevas, A.; Bermudez Hernandez, P.A.; et al. Cost-effectiveness analysis of newborn pulse oximetry screening to detect critical congenital heart disease in Colombia. Cost Eff. Resour. Alloc. 2019, 17, 11. [Google Scholar] [CrossRef] [PubMed]
- Nicklaus Children’s Hospital. Global Health Initiative for the Early Detection and Treatment of Congenital Heart Disease in Newborns. Available online: https://www.nicklauschildrens.org/programs/global-health-initiative (accessed on 21 January 2023).
- De Céspedes, C.; Saborío, M.; Trejos, R.; Abarca, G.; Sánchez, A.; Rojas, L. Evolution and innovations of the national neonatal and high risk screening program in Costa Rica. Rev. Biol. Trop. 2004, 52, pp. 451–466. Available online: https://www.readcube.com/articles/10.15517%2Frbt.v1i2.15278 (accessed on 3 February 2024).
- The Costa Rica News. Free Costa Rican Neonatal ‘Heel Test’ Diagnoses and Allows Treatment for 29 Diseases. 15 October 2014. Available online: https://thecostaricanews.com/free-costa-rican-neonatal-heel-test-diagnoses-and-allows-treatment-for-29-diseases/ (accessed on 22 January 2023).
- Cob, A.; Aita, F.C.; Rodríguez, A. Costa Rican national primary congenital hypothyroidism screening program evaluation. Retrospective cohort trial between 2015 and 2017. J. Endocr. Soc. 2020, 4 (Suppl. S1), A69. [Google Scholar] [CrossRef]
- Umaña-Calderón, A.; Acuña-Navas, M.J.; Alvarado, D.; Jiménez, M.; Cavallo-Aita, F. CYP21A2 mutations in pediatric patients with congenital adrenal hyperplasia in Costa Rica. Mol. Genet. Metab. Rep. 2021, 27, 100728. [Google Scholar] [CrossRef] [PubMed]
- Hevia-Urrutia, F.; Alvarado-Echeverría, I.; Sanabria-Castro, A.; Sánchez-Molina, M.; Meza-Sierra, L.; Parajeles-Vindas, A.; Méndez-Blanca, O.; Sánchez-Siles, Á.; Saborío-Rocafort, M.; Barguil-Gallardo, M.; et al. National alliance for Wilson’s disease: Health policy in Costa Rica. Hepatol. Med. Policy 2012, 2, 5. [Google Scholar] [CrossRef][Green Version]
- Penon-Portmann, M.; Lotz-Esquivel, S.; Chavez Carrera, A.; Jiménez-Hernández, M.; Alvarado-Romero, D.; Segura-Cordero, S.; Rimolo-Donadio, F.; Hevia-Urrutia, F.; Mora-Guevara, A.; Saborío-Rocafort, M.; et al. Wilson disease in Costa Rica: Pediatric phenotype and genotype characterization. JIMD Rep. 2020, 52, 55–62. [Google Scholar] [CrossRef]
- Muñoz, I.; Obando, S.O.; Jiménez, M.; Saborío, M. Pilot plan for improvement of newborn screening samples transportation in Costa Rica. J. Inborn Errors Metab. Screen. 2022. Available online: https://www.jiems-journal.org/storage/files/20220505102450_special-supplement-2022_compressed.pdf (accessed on 1 April 2024).
- Alvarado, D.; Gutiérrez, J.D.; Solano, M.; Reuben, A.; Camacho, N.; Jiménez, M.; Saborío, M. Medium-chain acyl-CoA dehydrogenase deficiency (MCADD) in Costa Rica: Molecular characterization of 38 patients identified through the national newborn screening program. J. Inborn Errors Metab. Screen. 2022. Available online: https://www.jiems-journal.org/storage/files/20220505102450_special-supplement-2022_compressed.pdf (accessed on 1 April 2024).
- Sánchez-Gutiérrez, A.; Arteaga-Year, A.L.; Ozunas-Fernández, N.; Frómeta-Suárez, A.; Castells-Martínez, E.M.; Del Río-Fabré, L.; Espinosa-Morales, M.; Martín-González, O.; Pérez-Molina, H.L.; Rego-Diaz, A.; et al. Results of the neonatal screening program in Cuba: 33 years of experience. J. Inborn Errors Metab. Screen. 2019. Available online: https://jiems-journal.org/storagefiles/2019.pdf (accessed on 3 February 2024).
- González Reyes, E.C.; Castells, E.M.; Frómeta, A.; Arteaga, A.L.; Del Rio, L.; Tejeda, Y.; Perez, L.T.; Segura, M.T.; Almenares, P.; Perea, Y.; et al. SUMA technology and newborn screening tests for inherited metabolic diseases in Cuba: An overview of the first 30 years. J. Inborn Errors Metabol. Screen. 2016, 4. [Google Scholar] [CrossRef]
- Pérez-Molina, H.L.; Palenzuela, A.; Carlos, N.M.; Rego-Diaz, A.; Turro, G.A.; Baró, G.; Rodríguez, R.; Rojas, M.M. External quality assessment of neonatal screening with ultramicroanalytic system in Latin America. General evaluation of more than two decades of work. J. Inborn Errors Metab. Screen. 2019. Available online: https://jiems-journal.org/storagefiles/2019.pdf (accessed on 3 February 2024).
- Morejón García, G.; García de la Rosa, I.; Feal Carballo, S.; Castells Martínez, E.M.; Stable Vernier, I.C.; Quintana Guerra, J.M.; Hernández Pérez, L.; Lafita Delfino, Y.; Pérez Morás, P.L.; Pupo Infante, M.; et al. Generation and characterization of murine monoclonal antibodies against immunoreactive trypsinogen for newborn screening of cystic fibrosis. Anal. Biochem. 2020, 591, 113569. [Google Scholar] [CrossRef] [PubMed]
- Castells, E.M.; Sánchez, A.; Frómeta, A.; Mokdse, Y.; Ozunas, N.; Licourt, T.; Arteaga, A.L.; Silva, E.; Collazo, T.; Rodríguez, F.; et al. Pilot study for cystic fibrosis neonatal screening: The Cuban experience. Clin. Chem Lab Med. 2020, 58, 1857–1864. [Google Scholar] [CrossRef]
- Castells Martínez, E.M.; González, E.C.; Tejeda, Y.; Frómeta, A.; Martín, O.; Espinosa, M.; Del Río, L.; Pérez, P.L.; Morejón, G.; Perea, Y.; et al. An enzyme immunoassay for determining immunoreactive trypsinogen (IRT) in dried blood spots on filter paper using an ultra-microanalytical system. Appl. Biochem. Biotechnol. 2018, 186, 1034–1046. [Google Scholar] [CrossRef] [PubMed]
- Morejón García, G.; García de la Rosa, I.; González Reyes, E.C.; Rubio Torres, A.; Quintana Guerra, J.M.; Hernández Marín, M.; Pérez Mora, P.L.; Feal Carballo, S.; Lafita Delfino, Y.; Pupo Infante, M.; et al. Generation of monoclonal antibodies against 17α-hydroxyprogesterone for newborn screening of congenital adrenal hyperplasia. Clin. Chim Acta 2018, 485, 311–315. [Google Scholar] [CrossRef] [PubMed]
- Castells, E.M.; González, E.C.; Pérez, P.L.; Del Río, L.; Tejeda, Y.; Perea, Y.; Martín, O.; Espinosa, M.; Rivero, J.A.; Frómeta, A. Evaluation of UMELISA® T4 NEONATAL and UMELISA® T4 using polystyrene plates coated with anti-thyroxine (T4) monoclonal antibodies. J. Immunoass. Immunochem. 2018, 39, 30–42. [Google Scholar] [CrossRef] [PubMed]
- García de la Rosa, I.; Feal Carballo, S.; Almenares Guasch, P.R.; Segura González, M.T.; Pupo Infante, M.; Frómeta Suárez, A.; Martínez Beltrán, L.; Quintana Guerra, J.M.; Lafita Delfino, Y.; Morejón García, G.; et al. Generation and characterization of monoclonal antibodies against thyroid-stimulating hormone for newborn screening of congenital hypothyroidism. J. Immunoass. Immunochem. 2019, 40, 350–366. [Google Scholar] [CrossRef] [PubMed]
- Delgado Sanchez, O. Universal newborn hearing screening in the neonatal population. Cuba, 2018. Horiz. Sanit. 2020, 19, 137–151. [Google Scholar] [CrossRef]
- Del Sol Gonzalez, Y.; Rodriguez, M.M. Infantix: A Cuban Screening System, for Our Children. Granma, Havana. 25 June 2021. Available online: https://en.granma.cu/cuba/2021-06-25/infantix-a-cuban-screening-system-for-our-children (accessed on 23 January 2023).
- Estrada-Veras, J.I.; Cabrera-Peña, G.A.; Pérez-Estrella de Ferrán, C. Medical genetics and genomic medicine in the Dominican Republic: Challenges and opportunities. Mol. Genet. Genomic Med. 2016, 4, 243–256. [Google Scholar] [CrossRef] [PubMed]
- Olney, R.S.; Bonham, J.R.; Schielen, P.C.J.I.; Slavin, D.; Ojodu, J. 2023 APHL/ISNS Newborn Screening Symposium. Int. J. Neonatal Screen. 2023, 9, 54. [Google Scholar] [CrossRef] [PubMed]
- Ministerio de Salud Pública de la República del Ecuador. Reglamento para Establecer el Procedimiento para el Desarrollo de las Pruebas de Tamizaje Metabólico Neonatal en el Ecuador. 2014. Available online: https://www.salud.gob.ec/wp-content/uploads/2016/09/AM-5104-REGLAMENTO-TAMIZAJE.pdf (accessed on 1 September 2023).
- Pozo-Palacios, J.; Garcia-Díaz, G.; Cruz, F.; Porras, F.; Heras, J.; Cano-Pérez, E. Spatial Distribution of congenital disorders diagnosed by the newborn screening program in Ecuador. J. Inborn Errors Metab. Screen. 2021, 9, e20200016. [Google Scholar] [CrossRef]
- Ministerio de Salud de El Salvador. Lineamientos Técnicos de Tamizaje Neonatal para Hipotiroidismo Congénito. 2015. Available online: http://asp.salud.gob.sv/regulacion/pdf/lineamientos/lineamientos_tamizaje_neonatal.pdf (accessed on 1 September 2023).
- Gobierno de El Salvador—Instituto Salvadoreño del Seguro Social. ISSS Realiza Tamizajes Neonatales para Detectar Enfermedades Congénitas en los Recién Nacidos. 2021. Available online: https://www.isss.gob.sv/isss-capacita-a-personal-administrativo-en-gestion-de-cobros/#:~:text=El%20Instituto%20Salvadore%C3%B1o%20del%20Seguro,el%20auditivo%20y%20el%20card%C3%ADaco (accessed on 1 September 2023).
- Salerno, R.; Schmidt, J. Lessons Learned from IAEA Technical Cooperation Programme: Successes and Challenge. 6 October 2005. Available online: https://www.academia.edu/download/33586729/PublicationRESEARCH_POSITIVO.pdf (accessed on 3 February 2024).
- Escobar Mena, N.G.; Bremily Chinchilla, B.; Soto, M.A.; Raul Velasco, R.; Marroquín, N.; Herrera, M.; Claudia Osorio, C.; Zamora, N.; Rosales, L.; Mariela Guerra, M.; et al. Pilot program of the neonatal screening program in Guatemala. J. Inborn Errors Metab. Screen. 2017, 5, 12. [Google Scholar] [CrossRef]
- Lemus, G.A.; Albanés, E.J.; Tzorín, P.A. Newborn screening in Guatemala: New strategies in public health. J. Inborn Errors Metab. Screen. 2019. Available online: https://jiems-journal.org/storagefiles/2019.pdf (accessed on 3 February 2024).
- Albanés, E.J.; Jacinto, A.C.; Chiroy, A.S.; Gaitán, I.C.; Tzorín, P.A. Preliminary incidence of structural hemoglobinopathies in Guatemala. J. Inborn Errors Metab. Screen. 2022. Available online: https://jiems-journal.org/storage/files/20220505102450_special-supplement-2022_compressed.pdf (accessed on 3 February 2024).
- Tzorín, P.A.; Morales, L.A.; Lemus, G.A.; Albanés, E.J. Proposal of informative material for the newborn screening program in Guatemala. J. Inborn Errors Metab. Screen. 2022. Available online: https://jiems-journal.org/storage/files/20220505102450_special-supplement-2022_compressed.pdf (accessed on 3 February 2024).
- Rotz, S.; Arty, G.; Dall’Amico, R.; De Zen, L.; Zanolli, F.; Bodas, P. Prevalence of sickle cell disease, hemoglobin S, and hemoglobin C among Haitian newborns. Am. J. Hematol. 2013, 88, 827–828. [Google Scholar] [CrossRef] [PubMed]
- Saint Fleur, R.; Archer, N.; Hustace, T.; Louis, R.J.; Bellevue, R.; Gautier, J.; Lerebours, E.; Voltaire, M.; Alvarez, O. Capacity building and networking to make newborn screening for sickle cell disease a reality in Haití. Blood Adv. 2018, 2 (Suppl. S1), 54–55. [Google Scholar] [CrossRef]
- Alvarez, O.A.; Hustace, T.; Voltaire, M.; Mantero, A.; Liberus, U.; Saint Fleur, R. Newborn screening for sickle cell disease using point-of-care testing in low-income setting. Pediatrics 2019, 144, e20184105. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, O.A.; St. Victor Dély, N.; Paul Hanna, M.; Saint Fleur, R.; Cetoute, M.; Metalonis, S.; Hustace, T.; Brown, E.C.; Marcelin, L.H.; Muscadin, E.; et al. Implementation of hospital-based sickle cell newborn screening and follow-up programs in Haití. Blood Adv. 2024, 8, 14–22. [Google Scholar] [CrossRef] [PubMed]
- Buckley, M.M.M.; Aguilar, L.B.; Lainez, R.C.; Valenzuela, H.J.A.; Ponce, F.; Melo, D.G. Neonatal screening program for five conditions in Honduras. J. Community Genet. 2021, 12, 389–395. [Google Scholar] [CrossRef] [PubMed]
- González Block, M.Á.; Reyes Morales, H.; Hurtado, L.C.; Balandrán, A.; Méndez, E. Mexico: Health system review. Health Syst Transit. 2020, 22, 1–222. [Google Scholar]
- Vargas, A.T.; Betsabé Castellanos Gómez, B.C. Barriers and facilitators of knowledge use in the health care system in Mexico: The newborn screening programme. Innov. Dev. 2019, 9, 305–321. [Google Scholar] [CrossRef]
- Vela-Amieva, M.; Belmont-Martínez, L.; Ibarra-González, I.; Fernández-Lainez, C. Institutional variability of neonatal screening in Mexico. Bol. Med. Hosp. Infant. Mex. 2009, 66, 431–439. [Google Scholar]
- Ibarra-González, I.; Fernández-Lainez, C.; Vela-Amieva, M.; Guillén-López, S.; Belmont-Martínez, L.; López-Mejía, L.; Carrillo-Nieto, R.I.; Guillén-Zaragoza, N.A. A Review of Disparities and Unmet Newborn Screening Needs over 33 Years in a Cohort of Mexican Patients with Inborn Errors of Intermediary Metabolism. Int. J. Neonatal Screen. 2023, 9, 59. [Google Scholar] [CrossRef] [PubMed]
- Cantú-Reyna, C.L.; Zepeda, L.M.; Montemayor, R.; Benavides, S.; González, H.J.; Vázquez-Cantú, M.; Cruz-Camino, H. Incidence of inborn errors of metabolism by expanded newborn screening in a Mexican hospital. J. Inborn Errors Metab. Screen. 2016, 4, 1–8. [Google Scholar] [CrossRef][Green Version]
- Vela-Amieva, M. (Laboratorio de Errores Innatos del Metabolismo y Tamiz, Instituto Nacional de Pediatría, CDMX, Mexico). Personal communication, 2023.
- Navarrete-Martínez, J.I.; Limón-Rojas, A.E.; Gaytán-García, M.J.; Reyna-Figueroa, J.; Wakida-Kusunoki, G.; Delgado-Calvillo, M.D.R.; Cantú-Reyna, C.; Cruz-Camino, H.; Cervantes-Barragán, D.E. Newborn screening for six lysosomal storage disorders in a cohort of Mexican patients: Three-year findings from a screening program in a closed Mexican health system. Mol. Genet. Metab. 2017, 121, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Camino, H.; Martínez Cervantes, E.A.; Cantú-Reyna, C.; Vázquez-Cantu, D.L.; Zea-Rey, A.; Gómez Gutiérrez, R.; Vera Delgado, J.A. Incidence of inborn errors of metabolism and other diseases detected in a newborn screening program within a group of Mexican private hospitals. Acta Pediatr. Esp. 2020, 78, e25–e32. [Google Scholar]
- De-la-Torre-García, O.; Mar-Aldana, R.; Santiago-Pérez, A.L.; Salgado-Sangri, R.E.; Fernández-Luna, C.P.; Herrera-Pérez, L.A.; Moreno-Graciano, C.M.; Quintero-Rodríguez, G.I.; Burgos-Lizama, J.Y.; Cardeña-Carballo, Z.M. Detection of newborns in the neonatal screening program of the health services of the Mexican Navy: An experience of 9 years. J. Inborn Errors Metab. Screen. 2022. Available online: https://jiems-journal.org/storage/files/20220505102450_special-supplement-2022_compressed.pdf (accessed on 3 February 2024).
- García-Martínez, E.; Terán-Montiel, L.; Rodríguez-Bazaldua, J.A.; Maldonado-Solís, F.A.; Moreno-Graciano, C.M.; Herrera-Pérez, L.A.; Quintero-Rodríguez, G.I.; Arias-Vidal, C.E.; Hernández-Pérez, H.; Rosique-Hipólito, N.T. Expanded neonatal screening program in the Mexican Army health services. An experience of 8 years. J. Inborn Errors Metab. Screen. 2022. Available online: https://jiems-journal.org/storage/files/20220505102450_special-supplement-2022_compressed.pdf (accessed on 3 February 2024).
- García-Flores, E.P.; Herrera-Maldonado, N.; Hinojosa-Trejo, M.A.; Vergara-Vázquez, M.; Halley-Castillo, M.E. Advances and achievements of the newborn screening program (2012–2018). Acta Pediatr. Mex. 2018, 39 (Suppl. S1), S57–S65. [Google Scholar]
- Herrera-Maldonado, N.; Vergara-Vázquez, M.; Hinojosa-Trejo, M.A.; García-Flores, E.P. Challenges of the newborn screening program at the Ministry of Health in Mexico. Acta Pediatr. Mex. 2018, 39 (Suppl. S1), S1–S4. [Google Scholar]
- Cantú-Reyna, C.; Santos-Guzmán, J.; Cruz-Camino, H.; Vazquez Cantu, D.L.; Góngora-Cortéz, J.J.; Gutiérrez-Castillo, A. Glucose-6-phosphate dehydrogenase deficiency incidence in a Hispanic population. J. Neonatal Perinat. Med. 2019, 12, 203–207. [Google Scholar] [CrossRef] [PubMed]
- Vela-Amieva, M.; Alcántara-Ortigoza, M.A.; González-Del Angel, A.; Belmont-Martínez, L.; López-Candiani, C.; Ibarra-González, I. Genetic spectrum and clinical early natural history of glucose-6-phosphate dehydrogenase deficiency in Mexican children detected through newborn screening. Orphanet J. Rare Dis. 2021, 16, 103. [Google Scholar] [CrossRef] [PubMed]
- Alcántara-Ortigoza, M.A.; Hernández-Ochoa, B.; González-Del Angel, A.; Ibarra-González, I.; Belmont-Martínez, L.; Gómez-Manzo, S.; Vela-Amieva, M. Functional characterization of the p.(Gln195His) or Tainan and novel p.(Ser184Cys) or Toluca glucose-6-phosphate dehydrogenase (G6PD) gene natural variants identified through Mexican newborn screening for glucose-6-phosphate dehydrogenase deficiency. Clin. Biochem. 2022, 109–110, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Ibarra-González, I.; Campos-Garcia, F.J.; Herrera-Pérez, L.D.A.; Martínez-Cruz, P.; Moreno-Graciano, C.M.; Contreras-Capetillo, S.N.; León-Burgos, V.; Maldonado-Solis, F.A.; Alcántara-Ortigoza, M.A.; González Del Angel, A.; et al. Newborn cystic fibrosis screening in southeastern Mexico: Birth prevalence and novel CFTR gene variants. J. Med. Screen. 2018, 25, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Ibarra-González, I.; Ridaura-Sanz, C.; Fernández-Lainez, C.; Guillén-López, S.; Belmont-Martínez, L.; Vela-Amieva, M. Hepatorenal tyrosinemia in Mexico: A call to action. Adv. Exp. Med. Biol. 2017, 959, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Zea-Rey, A.V.; Cruz-Camino, H.; Vazquez-Cantu, D.L.; Gutiérrez-García, V.M.; Santos-Guzmán, J.; Cantú-Reyna, C. The incidence of transient neonatal tyrosinemia within a Mexican population. J. Inborn Errors Metab. Screen. 2017, 5, 1–4. [Google Scholar] [CrossRef]
- Galvan-Ramírez Mde, L.; Troyo-Sanroman, R.; Roman, S.; Bernal-Redondo, R.; Vázquez Castellanos, J.L. Prevalence of toxoplasma infection in Mexican newborns and children: A systematic review from 1954 to 2009. ISRN Pediatr. 2012, 2012, 501216. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Martínez-Cruz, C.F.; Ramírez-Vargas, M.N.; García-Alonso Themann, P. Results of the universal neonatal hearing screening in a tertiary care hospital in Mexico City. Int. J. Pediatr. Otorhinolaryngol. 2020, 139, 110412. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Gutiérrez, R.; Galindo-Hayashi, J.M.; Cantú-Reyna, C.; Vazquez-Cantu, D.L.; Britton-Robles, C.; Cruz-Camino, H. Critical CHD screening programme: A 3-year multicentre experience in Mexico. Cardiol. Young. 2023, 33, 952–958. [Google Scholar] [CrossRef]
- Orozco, O.S.R.; Nkemdirim, W.; Gutierrez Alvarez, I.A.; Saarinen, A.; Zanella, E.; Muñoz, J.E.; Ledesma, P.; Alejandra Guzman, L.; Perez, B.M. The predictive power of concurrent pulse oximetry readings in the detection of congenital heart defects in newborns of The Hospital of Specialties of Children and Women in Queretaro, Mexico. Neonatol. Today 2020, 15, 3–10. [Google Scholar] [CrossRef]
- Rubens-Figueroa, J.; Mier-Martínez, M.; Jiménez-Carbajal, M.G.; García-Aguilar, H. Neonatal cardiac screening in Mexico, a tool for critical heart disease early diagnosis. Gac. Med. Mex. 2022, 158, 65–68. [Google Scholar] [CrossRef] [PubMed]
- Comité MTNC. Introduction, creation and development of the Mexican Committee for Neonatal Cardiac Screening. Rev. Mex. Pediatr 2022, 89 (Suppl. S1), s3–s4. [Google Scholar] [CrossRef] [PubMed]
- Lara Toruño, L. (Departamento de Ciencias Morfológicas, Facultad de Ciencias Médicas, Universidad Nacional Autónoma de Nicaragua, León, Nicaragua). Personal communication, 2022.
- Fúnez, A.; Lara, M.E.; Chévez, A.C.; Castellón, E.A.; Perán, S.; Toro, M.J.; Montoya, E.; Moreno, J.C. Neonatal Screening for Congenital Hypothyroidism in Nicaragua: Audit of a Cord-blood Thyrotropin-based Program (2005–2015). J. Inborn Errors Metab. Screen. 2019, 7, e20190003. [Google Scholar] [CrossRef]
- Del Río Fabre, L.; Alvarez Abreu, M.; Sequeira Gudiel, B.; Delgado Pérez, R.; Barrios Rocha, L.; Salazar Ortiz, Y. Implementation of the Newborn Screening Program at the Nicaraguan Social Security Institute: First Year Results. J. Inborn Errors Metab. Screen. 2015, 3, 91–92. [Google Scholar]
- Pernudy-Ubau, A.; Puller, A.-C.; Mejía-Baltodano, G.; Rodríguez-Romer, W. Establishment of a specialized laboratory for hemoglobin analysis in Nicaragua. Blood Adv. 2018, 2 (Suppl. S1), 58–59. [Google Scholar] [CrossRef] [PubMed]
- Wong, L.Y.; Espinoza, F.; Alvarez, K.M.; Molter, D.; Saunders, J.E. Otoacoustic emissions in rural Nicaragua: Cost analysis and implications for newborn hearing screening. Otolaryngol. Head Neck Surg. 2017, 156, 877–885. [Google Scholar] [CrossRef]
- Stein-Meyers, A. Bringing Newborn Hearing Screening to Nicaragua. Center for Hearing and Communication. 14 August 2017. Available online: https://www.chchearing.org/post/bringing-newborn-hearing-screening-to-nicaragua (accessed on 31 January 2023).
- Cossio, G. Informe Programa Nacional de Tamizaje Neonatal 2014. (Centro de Referencia). Available online: https://www.minsa.gob.pa/sites/default/files/programas/informe_programa_nacional_de_tamizaje_neonatal_ano_2014.pdf (accessed on 1 September 2023).
- Cossio, G.; Chávez, T.; Gónzalez, D.; Delgado, M. Estudio preliminar de incidencia de hemoglobinopatías, en neonatos tamizados en el Hospital del Niño de Panamá. Agosto-diciembre 2009. Pediátr. Panamá 2011, 40, 15–18. [Google Scholar]
- Chung Chong, M.; Herrera Rodriguez, I.; Chávez Peña, T.; Saldaña Bethancourth, O.; Rodríguez, M.; Cepeda, C. Panamá en cifras: Tamizaje neonatal metabólico 2013 al 2018. Pediátr. Panamá 2022, 51, 94–99. [Google Scholar] [CrossRef]
- Gaceta Oficial Digital. Gaceta Oficial Digital. República de Panamá—Gobierno Nacional. Ley 329/2022 Tamizaje Neonatal. Año CXXI. N° 29643. 11 October 2022. Available online: https://www.gacetaoficial.gob.pa/pdfTemp/29643/GacetaNo_29643_20221013.pdf (accessed on 1 September 2023).
- Ascurra, M. Challenges in the design and implementation of the neonatal screening program—Paraguay. Program & Abstract Book. In Proceedings of the International Conference on Birth Defects and Disabilities in the Developing World—ICBD 2023, Santiago, Chile, 1–4 March 2023; p. 43. [Google Scholar]
- Ascurra, M.; Alvarez, P.; Ortíz-Paranza, L.; Blanco, F.; Valenzuela, A.; Insaurralde, A.; Rodríguez, S.; Salinas, M.; Porzio, G.; Núñez, A. Consolidation of the neonatal screening program as a public health program in Paraguay. J. Inborn Errors Metab. Screen. 2021, 9, e20210006. [Google Scholar] [CrossRef]
- Galán-Rodas, E.; Dueñas, M.; Obando, S.; Saborio, M. Newborn screening in Peru: Where are we going? Rev. Peru Med. Exp. Salud Publica 2013, 30, 724–725. [Google Scholar]
- Huerta-Sáenz, L.; Del Águila, C.; Espinoza, O.; Falen-Boggio, J.; Mitre, N. Tamizaje nacional unificado de hipotiroidismo congénito en el Perú: Un programa inexistente [National congenital hypothyroidism screening in Peru: A broken program]. Rev. Peru Med. Exp. Salud Publica 2015, 32, 579–585. [Google Scholar] [CrossRef] [PubMed]
- Dirección General de Intervenciones Estratégicas en Salud Pública. Ministerio de Salud. Lima—Perú. Norma técnica de salud para el tamizaje neonatal de hipotiroidismo congénito, hiperplasia suprarrenal congénita, fenilcetonuria, fibrosis quística, hipoacusia congénita y catarata congénita. R.M. N° 558-2019/MINSA. 2020. Available online: http://bvs.minsa.gob.pe/local/MINSA/5045.pdf (accessed on 3 February 2024).
- Guio, H.; Poterico, J.A.; Levano, K.S.; Cornejo-Olivas, M.; Mazzetti, P.; Manassero-Morales, G.; Ugarte-Gil, M.F.; Acevedo-Vásquez, E.; Dueñas-Roque, M.; Piscoya, A.; et al. Genetics and genomics in Peru: Clinical and research perspective. Mol. Genet. Genomic Med. 2018, 6, 873–886. [Google Scholar] [CrossRef] [PubMed]
- Congreso de la República del Perú. Proyecto de Ley 4210. Ley que Implementa el Derecho del Nacido vivo al Tamizaje Neonatal Universal en todo el Sistema de Salud. 2022. Available online: https://www.congreso.gob.pe/Docs/comisiones2022/Salud/files/proyectos_de_ley/proyecto_de_ley_4210.pdf (accessed on 1 September 2023).
- Larrandaburu, M.; Matte, U.; Noble, A.; Olivera, Z.; Sanseverino, M.T.; Nacul, L.; Schuler-Faccini, L. Ethics, genetics and public policies in Uruguay: Newborn and infant screening as a paradigm. J. Community Genet. 2015, 6, 241–249. [Google Scholar] [CrossRef] [PubMed]
- International Atomic Energy Agency. Screening Newborns for Health: Simple, Cost-Effective Nuclear Technique Prevents Mental Retardation in Developing Countries; International Atomic Energy Agency: Vienna, Austria, 2018; Available online: https://www.iaea.org/newscenter/pressreleases/screening-newborns-health-simple-cost-effective-nuclear-technique-prevents-mental-retardation-developing-countries (accessed on 7 February 2023).
- Audicio, P.; Segobia, B.; Queijo, C.; Queiruga, G. Newborn screening pilot program of hemoglobinopathies: First results in Uruguay. Acta Bioquím. Clín. Latinoam. 2017, 51, 243–248. [Google Scholar]
- Larrandaburu, M.; Vianna, F.L.S.; Griot, K.; Queijo, C.; Monzón, G.; Ugarte, C.; Nacul, L.; Schuler-Faccini, L.; Sanseverino, M.T.V. Rare diseases in Uruguay: Focus on infants with abnormal newborn screening. J. Inborn Errors Metab. Screen. 2019, 7, e20190002. [Google Scholar] [CrossRef]
- Queijo, C.; Lemes, A.; Queiruga, G. 25 Years of Newborn Screening in Uruguay. J. Inborn Errors Metab. Screen. 2021, 9, e20210008. [Google Scholar] [CrossRef]
- Costagliola Martorona, A.; Chacin Hernandez, J.; Ocando Pino, L.; Mejìa Villan, I.; Torres Quevedo, E.; Torres Chirinos, Y.; Picado Gutierrez, J. Neonatal Screening Program (NSP) in Zulia—Venezuela: Evaluation of Twelve Years of Experience. J. Inborn Errors Metab. Screen. 2015, 3, 18–19. [Google Scholar]
- Roz, G. (Programa Nacional de Pesquisa Neonatal, División de Enfermedades Heredometabólicas, Dirección de Salud de la Familia, Ministerio del Poder Popular para la Salud, Gobierno Bolivariano de Venezuela). Personal communication, 2017.
- The Anguillan. Ask Your Doctor: Sickle Cell Disease. 28 June 2021. Available online: https://theanguillian.com/2021/06/ask-your-doctor-sickle-cell-disease-3/ (accessed on 28 February 2023).
- American University of Antigua College of Medicine. AUA Sponsors Free Sickle Cell Screening Program for Newborns in Antigua. 2 October 2020. Available online: https://www.auamed.org/news/aua-sponsors-free-sickle-cell-screening-program-for-newborns-in-antigua/ (accessed on 26 February 2023).
- Belle Jarvis, S.; Hadeed, E.; Lee, K.; Hardy-Dessources, M.D.; Knight-Madden, J.M.; Richardson, C. Sickle cell disease newborn screening—An audit of a twin island state pilot program. Int. J. Neonatal Screen. 2023, 9, 14. [Google Scholar] [CrossRef] [PubMed]
- van Heyningen, A.M.; Levenston, M.J.; Tamminga, N.; Scoop-Martijn, E.G.; Wever, R.M.F.; Verhagen, A.A.E.; van der Dijs, F.P.L.; Dijck-Brouwer, D.A.J.; Offringa, P.J.; Muskiet, F.A.J. Estimated incidence of sickle-cell disease in Aruba and St Maarten suggests cost-effectiveness of a universal screening programme for St. Maarten. West Indian Med. J. 2009, 58, 301–304. [Google Scholar] [PubMed]
- Quashnock, J. (Revvity, Pittsburg, PA, USA). Personal communication, 2023.
- St. John, M.A.; Lungu, F.N. Haemoglobin electrophoresis patterns in Barbados. West Indian Med. J. 1999, 48, 221–222. [Google Scholar] [PubMed]
- Quimby, K.R.; Moe, S.; Sealy, I.; Nicholls, C.; Hambleton, I.R.; Landis, R.C. Clinical findings associated with homozygous sickle cell disease in the Barbadian population—Do we need a national SCD registry? BMC Res. Notes 2014, 7, 102. [Google Scholar] [CrossRef] [PubMed]
- Barbados Today. HOPE Urges More Screening for Sickle Cell. 21 June 2022. Available online: https://barbadostoday.bb/2022/06/21/hope-urges-more-screening-for-sickle-cell/ (accessed on 20 February 2023).
- Jaffar, S.; Ellsworth, G. A pilot study of sickle cell anemia in the Belizean Population. Blood 2017, 130 (Suppl. S1), 5584. [Google Scholar]
- Ferreira, C. Genetic Disorders in the Cayman Islands. Public Health Spotlight. 17 February 2023. Available online: https://www.gov.ky/health-wellness/Public-Health.html (accessed on 1 March 2023).
- Antoine, M.; Lee, K.; Donald, T.; Belfon, Y.; Drigo, A.; Polson, S.; Martin, F.; Mitchell, G.; Etienne-Julan, M. Marie-Dominique Hardy-Dessources. Prevalence of sickle cell disease among Grenadian newborns. J. Med. Screen. 2018, 25, 49–50. [Google Scholar] [CrossRef] [PubMed]
- Saint-Martin, C.; Romana, M.; Bibrac, A.; Brudey, K.; Tarer, V.; Divialle-Doumdo, L.; Petras, M.; Keclard-Christophe, L.; Lamothe, S.; Broquere, C.; et al. Universal newborn screening for haemoglobinopathies in Guadeloupe (French West Indies): A 27-year experience. J. Med. Screen. 2013, 20, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Serjeant, G. Sickle Cell, Jamaica and Beyond: A Life; Ian Randle: Kingston, Jamaica, 2021; pp. 40–41. [Google Scholar]
- Serjeant, G.; Serjeant, B.E.; Forbes, M.; Hayes, R.J.; Higgs, D.R.; Lehmann, H. Haemoglobin gene frequencies in the Jamaican population: A study in 100,000 newborns. Br. J. Haematol. 1986, 64, 253–262. [Google Scholar] [CrossRef]
- King, L.; Knight-Madden, J.; Reid, M. Newborn screening for sickle cell disease in Jamaica: A review—Past, present and future. West Indian Med. J. 2014, 63, 147–150. [Google Scholar] [CrossRef] [PubMed]
- Knight-Madden, J.; Asnani, M.; King, L.; Hardy-Dessources, M.D. Sickle cell disease in the Caribbean: Progress in newborn screening, clinical care, and research through collaboration. Lancet Haematol. 2023, 10, e581–e582. [Google Scholar] [CrossRef] [PubMed]
- Knight-Madden, J.; Abar, A.; Smith, M.; Asnani, M. Factors affecting age of initial specialty clinic evaluation among infants with sickle cell disease in Kingston, Jamaica. J. Pediatr. 2023, 257, 113384. [Google Scholar] [CrossRef] [PubMed]
- Serjeant, G.R.; Serjeant, B.E.; Mason, K.P.; Gardner, R.; Warren, L.; Gibson, F.; Coombs, M. Newborn screening for sickle cell disease in Jamaica: Logistics and experience with umbilical cord samples. J. Community Genet. 2017, 8, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Etienne-Julan, M.; Christian Saint-Martin, C. Sickle cell disease in the French West Indies. Rev. Francoph. Lab. 2005, 2005, 61–66. [Google Scholar] [CrossRef]
- Sajous, C.; Goto, M.; Fitzgerald, M.; Anderson, C.L.; Craft, W.; Hurley, R.M.; Zeller, W.P. Cord blood thyrotropin screening for congenital hypothyroidism. Three years’ experience on the Island of Saint Lucia. Ann. Clin. Lab. Sci. 1991, 21, 426–429. [Google Scholar] [PubMed]
- Alexander, S.; Belmar-George, S.; Eugene, A.; Elias, V. Knowledge of and attitudes toward heel prick screening for sickle cell disease in Saint Lucia. Rev. Panam. Salud Publica 2017, 41, e70. [Google Scholar] [CrossRef] [PubMed]
- Williams, S.A.; Browne-Ferdinand, B.; Smart, Y.; Morella, K.; Reed, S.G.; Kanter, J. Newborn screening for sickle cell disease in St. Vincent and the Grenadines: Results of a pilot newborn screening program. Glob. Pediatr. Health 2017, 4, 2333794X17739191. [Google Scholar] [CrossRef] [PubMed]
- Marrakech Declaration. Available online: https://cdn-links.lww.com/permalink/gim/a/gim_11_9_2009_06_08_krotoski_200542_sdc1.pdf (accessed on 13 July 2023).
- Shawky, R.M. Newborn screening in the Middle East and North Africa—Challenges and recommendations. Hamdan Med. J. 2012, 5, 191. [Google Scholar] [CrossRef]
- Skrinska, V.; Khneisser, I.; Schielen, P.; Loeber, G. Introducing and expanding newborn screening in the MENA Region. Int. J. Neonatal Screen. 2020, 6, 12. [Google Scholar] [CrossRef] [PubMed]
- Krotoski, D.; Namaste, S.; Raouf, R.K.; El Nekhely, I.; Hindi-Alexander, M.; Engelson, G.; Hanson, J.W.; Howell, R.R.; MENA NBS Steering Committee. Conference report: Second conference of the Middle East and North Africa newborn screening initiative: Partnerships for sustainable newborn screening infrastructure and research opportunities. Genet. Med. 2009, 11, 663–668. [Google Scholar] [CrossRef]
- Al-Sadeq, D.; Abunada, T.; Dalloul, R.; Fahad, S.; Taleb, S.; Aljassim, K.; Al Hamed, F.A.; Zayed, H. Spectrum of mutations of cystic fibrosis in the 22 Arab countries: A systematic review. Respirology 2019, 24, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Al-Sadeq, D.W.; Nasrallah, G.K. The spectrum of mutations of homocystinuria in the MENA Region. Genes 2020, 11, 330. [Google Scholar] [CrossRef] [PubMed]
- Al-Mousa, H.; Al-Saud, B. Primary immunodeficiency diseases in highly consanguineous populations from Middle East and North Africa: Epidemiology, diagnosis, and care. Front. Immunol. 2017, 8, 678. [Google Scholar] [CrossRef] [PubMed]
- El-Sayed, Z.A.; Radwan, N. Newborn screening for primary immunodeficiencies: The gaps, challenges, and outlook for developing countries. Front. Immunol. 2020, 10, 2987. [Google Scholar] [CrossRef] [PubMed]
- Hom, L.A.; Martin, G.R. Newborn critical congenital heart disease screening using pulse oximetry: Value and unique challenges in developing regions. Int. J. Neonatal Screen. 2020, 6, 74. [Google Scholar] [CrossRef] [PubMed]
- Ouarezki, Y.; Ladjouze, A.; Kherra, S.; Djermane, A.; Laraba, A. Age at diagnosis and mental development in children with congenital hypothyroidism in the absence of newborn screening programme. Horm. Res. Paediatr. 2016, 86 (Suppl. 1), 506–507. [Google Scholar] [CrossRef] [PubMed]
- Adel, D.; Asmahane, L.; Yasmine, O.; Ourmiida, T.N.; Kafia, Z.; Samira, A.; Bouzerar, Z.; Hachemi, M. Outcome of congenital hypothyroidism in Algeria: The urgent need to implement a national newborn screening program. Horm. Res. Paediatr. 2019, 89 (Suppl. 1), 275. [Google Scholar] [CrossRef]
- Boudjenah, F.; Hasbellaoui, M.; Zemirli, O. Results of the first universal newborn hearing screening in Algeria. Adv. Arab Acad. Audio-Vestibul J. 2015, 2, 54–58. [Google Scholar] [CrossRef]
- Al Arrayed, S.; Al Hajeri, A. Newborn screening services in Bahrain between 1985 and 2010. Adv. Hematol. 2012, 2012, 903219. [Google Scholar] [CrossRef] [PubMed]
- Golbahar, J.; Al-Khayyat, H.; Hassan, B.; Agab, W.; Hassan, E.; Darwish, A. Neonatal screening for congenital hypothyroidism: A retrospective hospital based study from Bahrain. J. Pediatr. Endocrinol. Metab. 2010, 23, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Golbahar, J.; Al-Jishi, E.A.; Altayab, D.D.; Carreon, E.; Bakhiet, M.; Alkhayyat, H. Selective newborn screening of inborn errors of amino acids, organic acids and fatty acids metabolism in the Kingdom of Bahrain. Mol. Genet. Metab. 2013, 110, 98–101. [Google Scholar] [CrossRef] [PubMed]
- George, S.A.; Alawadhi, A.; Al Reefy, H.; Riskalla, A. Newborn hearing screening. Bahrain Med. Bull. 2016, 38, 148–150. Available online: https://www.bahrainmedicalbulletin.com/september_2016/Newborn.pdf (accessed on 3 November 2022).
- Hassan, F.A.; El-Mougy, F.; Sharaf, S.A.; Mandour, I.; Morgan, M.F.; Selim, L.A.; Hassan, S.A.; Salem, F.; Oraby, A.; Girgis, M.Y.; et al. Inborn errors of metabolism detectable by tandem mass spectrometry in Egypt: The first newborn screening pilot study. J. Med. Screen. 2016, 23, 124–129. [Google Scholar] [CrossRef] [PubMed]
- Kotb, M.A.; Mansour, L.; William Shaker Basanti, C.; El Garf, W.; Ali, G.I.Z.; Mostafa El Sorogy, S.T.; Kamel, I.E.M.; Kamal, N.M. Pilot study of classic galactosemia: Neurodevelopmental impact and other complications urge neonatal screening in Egypt. J. Adv. Res. 2018, 12, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Hagag, A.A.; Badraia, I.M.; Elfarargy, M.S.; Abd Elmageed, M.M.; Abo-Ali, E.A. Study of glucose-6.-phosphate dehydrogenase deficiency: 5 years retrospective Egyptian study. Endocr. Metab Immune Disord. Drug Targets 2018, 18, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Kotb, M.A.; Mansour, L.; Shamma, R.A. Screening for galactosemia: Is there a place for it? Int. J. Gen. Med. 2019, 12, 193–205. [Google Scholar] [CrossRef] [PubMed]
- Magdy, R.M.; Abd-Elkhalek, H.S.; Bakheet, M.A.; Mohamed, M.M. Selective screening for inborn errors of metabolism by tandem mass spectrometry at Sohag University Hospital, Egypt. Arch. Pediatr. 2022, 29, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Egypt Today. Egypt Screens 107,000 Neonates under Initiative to Detect Genetic Disease. Available online: https://www.egypttoday.com/Article/1/115524/Egypt-screens-107-000-neonates-under-initiative-to-detect-genetic (accessed on 7 November 2022).
- Khafagy, H. Future view of Egyptian neonatal screening program. In Proceedings of the 4th Meeting of the Middle East North Africa (MENA) Region of the International Society for Neonatal Screening (ISNS), Limassol, Cyprus, 8–11 March 2020. [Google Scholar]
- Hohenfellner, K.; Bergmann, C.; Fleige, T.; Janzen, N.; Burggraf, S.; Olgemöller, B.; Gahl, W.A.; Czibere, L.; Froschauer, S.; Röschinger, W.; et al. Molecular based newborn screening in Germany: Follow-up for cystinosis. Mol. Genet. Metab. Rep. 2019, 21, 100514. [Google Scholar] [CrossRef] [PubMed]
- Taha, T.; Ahmed, D.; El-Gammal, Z.; Oura, G.A.; Elshenawy, S.E.; Gaber, Y.; Elnagdy, T.; Amer, K. A brief insight into the rare diseases in Egypt. J. Rare Dis. 2023, 2, 6. [Google Scholar] [CrossRef]
- Yarahmadi, S.; Azhang, N.; Nikkhoo, B.; Rahmani, K. A success story: Review of the implementation and achievements of the national newborn screening program for congenital hypothyroidism in Iran. Int. J. Endocrinol. Metab. 2020, 18, e99099. [Google Scholar] [CrossRef] [PubMed]
- Yarahmadi, S.; Nikkhoo, B.; Parvizi, H.; Motaghi, R.; Rahmani, K. Physical development at school entry in children with congenital hypothyroidism diagnosed by the national program of newborn screening in Iran. Int. J. Endocrinol. Metab. 2022, 21, e131081. [Google Scholar] [CrossRef] [PubMed]
- Mehran, L.; Khalili, D.; Yarahmadi, S.; Delshad, H.; Mehrabi, Y.; Amouzegar, A.; Ajang, N.; Azizi, F. Evaluation of the congenital hypothyroidism screening programme in Iran: A 3-year retrospective cohort study. Arch. Dis. Child. Fetal Neonatal Ed. 2019, 104, F176–F181. [Google Scholar] [CrossRef] [PubMed]
- Mehran, L.; Yarahmadi, S.; Khalili, D.; Hedayati, M.; Amouzegar, A.; Mousapour, P.; Ajang, N.; Azizi, F. Audit of the congenital hypothyroidism screening program in 15 provinces of Iran. Arch. Iran. Med. 2019, 22, 310–317. [Google Scholar] [PubMed]
- Heidari, A.; Arab, M.; Etemad, K.; Damari, B.; Kabir, M.J. Challenges of implementation of the national phenylketonuria screening program in Iran: A qualitative study. Electron. Physician 2016, 8, 3048–3056. [Google Scholar] [CrossRef] [PubMed]
- Shokri, M.; Karimi, P.; Zamanifar, H.; Kazemi, F.; Badfar, G.; Azami, M. Phenylketonuria screening in Iranian newborns: A systematic review and meta-analysis. BMC Pediatr. 2020, 20, 352. [Google Scholar] [CrossRef] [PubMed]
- Kosaryan, M.; Mahdavi, M.R.; Jalali, H.; Roshan, P. Why does the Iranian national program of screening newborns for G6PD enzyme deficiency miss a large number of affected infants? Pediatr. Hematol. Oncol. 2014, 31, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Heidari, A.; Arab, M.; Damari, B. A policy analysis of the national phenylketonuria screening program in Iran. BMC Health Serv. Res. 2021, 21, 120. [Google Scholar] [CrossRef] [PubMed]
- Etemad, K.; Heidari, A.; Setoodeh, A.; Shayeganrad, A.; Akhlaghi, A.; Azizi, M.; Nejatbakhsh, R.; Beglar, M.B.; Lotfi, M. Health-related quality of life of parents of children with phenylketonuria in Tehran Province, Islamic Republic of Iran. East. Mediterr. Health J. 2020, 26, 331–339. [Google Scholar] [CrossRef] [PubMed]
- Younesi, S.; Yazdani, B.; Taheri Amin, M.M.; Saadati, P.; Jamali, S.; Modarresi, M.H.; Savad, S.; Amidi, S.; Razavi, H.; Ghafouri-Fard, S. Incorporation of second-tier tests and secondary biomarkers to improve positive predictive value (PPV) rate in newborn metabolic screening program. J. Clin. Lab. Anal. 2022, 36, e24471. [Google Scholar] [CrossRef] [PubMed]
- Ayatollahi, H.; Karimi, S.; Ahmadi, M. Newborn screening data management: Proposing a framework for Iran. J. Innov. Health Inform. 2018, 25, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Tajmir-Riahi, A.; Khatami, S.; Shemirani, F.; Mirzazadeh, R. Two fluorimetric determinations of acid α-glucosidase activity in dried blood spot: Pompe disease in Iranian population. Anal. Biochem. 2023, 682, 115346. [Google Scholar] [CrossRef]
- Mahmood, R.H. Newborn screening in Iraq. In Proceedings of the 4th Meeting of the Middle East North Africa (MENA) Region of the International Society for Neonatal Screening (ISNS), Limassol, Cyprus, 8–11 March 2020. [Google Scholar]
- U.S. Agency for International Development (USAID) Primary Health Care Project in Iraq. National Guideline of Newborn Screening for Care Providers in PHC Centers Iraq 2014. University Research Co., LLC. 2014. Available online: https://studylib.net/doc/6617400/newborn-screening---primary-health-care-iraq (accessed on 8 December 2022).
- Alkhazrajy, L.A.; Hassan, A.A. Overview of neonatal screening program applied at primary health care centers in Baghdad/Iraq. Int. J. Community Coop. Stud. 2016, 4, 40–56. [Google Scholar]
- Atshan, F.H.; Hassan, I.F.; Hommadi, S.A.W. Evaluation of newborn screening program in Baghdad Al-Karkh health directorate in 2018. Pharma Innovat. J. 2019, 8, 6–11. [Google Scholar]
- Twfeeq, W.F.; Abed, L.M. Knowledge and attitudes of mothers toward newborn screening test in Baghdad City. Diyala J. Med. 2016, 10, 66–74. [Google Scholar]
- Al-Timimi, D.J.; Mustafa, A.H. Neonatal screening for congenital hypothyroidism in Duhok Governorate (Iraq): A preliminary study. Duhok Med. J. 2019, 13, 20–28. [Google Scholar] [CrossRef]
- Khneisser, I. Targeted NBS Program in Kurdistan Region—Iraq. Paper presented at the 4th Meeting of the Middle East North Africa (MENA) Region of the International Society for Neonatal Screening (ISNS), Limassol, Cyprus, 8–11 March 2020. [Google Scholar]
- Persike de Oliveira, D.S.; Bjoraker, K.J. Serendipitous discovery of phenylketonuria in Iraq—How to identify and treat? Mol. Genet. Metab. Rep. 2021, 27, 100737. [Google Scholar] [CrossRef] [PubMed]
- Abbasi, R.; Harari-Kremer, R.; Haim, A.; Raz, R. Risk factors of congenital hypothyroidism in Israel. Isr. Med. Assoc. J. 2022, 25, 808–814. [Google Scholar]
- Israel Ministry of Health. The Ministry of Health National Program of Infant Screening Tests. Available online: https://www.health.gov.r/English/Topics/Genetics/InfantScreening/Pages/default.aspx (accessed on 13 December 2022).
- Zchut, K. Screening Newborns for Serious Diseases. Israel Ministry of Health. The Ministry of Health National Screening Newborns for Serious Disease. Available online: https://wchutww.kolzchut.org.il/en/Screening_Newborns_for_Serious_Diseases (accessed on 13 December 2022).
- Zuckerman, S. Indifferent or uninformed? Reflections of health professionals on parental education and consent for expanded newborn screening in Israel, 2008–2016. Int. J. Neonatal Screen. 2017, 3, 12. [Google Scholar] [CrossRef]
- Rechavi, E.; Lev, A.; Simon, A.J.; Stauber, T.; Daas, S.; Saraf-Levy, T.; Broides, A.; Nahum, A.; Marcus, N.; Hanna, S.; et al. First year of Israeli newborn screening for severe combined immunodeficiency—Clinical achievements and insights. Front. Immunol. 2017, 8, 1448. [Google Scholar] [CrossRef] [PubMed]
- Lev, A.; Sharir, I.; Simon, A.J.; Levy, S.; Lee, Y.N.; Frizinsky, S.; Daas, S.; Saraf-Levy, T.; Broides, A.; Nahum, A.; et al. Lessons learned from five years of newborn screening for severe combined immunodeficiency in Israel. J. Allergy Clin. Immunol. Pract. 2022, 10, 2722–2731.e9. [Google Scholar] [CrossRef] [PubMed]
- Puck, J.M. A spot of good news: Israeli experience with SCID newborn screening. J. Allergy Clin. Immunol. Pract. 2022, 10, 2732–2733. [Google Scholar] [CrossRef] [PubMed]
- Daas, S.; Abu Salah, N.; Anikster, Y.; Barel, O.; Damseh, N.S.; Dumin, E.; Fattal-Valevski, A.; Falik-Zaccai, T.C.; Habib, C.; Josefsberg, S.; et al. Addition of galactose-1-phosphate measurement enhances newborn screening for classical galactosemia. J. Inherit. Metab. Dis. 2023, 46, 232–242. [Google Scholar] [CrossRef] [PubMed]
- DeBarber, A.E.; Kalfon, L.; Fedida, A.; Fleisher Sheffer, V.; Ben Haroush, S.; Chasnyk, N.; Shuster Biton, E.; Mandel, H.; Jeffries, K.; Shinwell, E.S.; et al. Newborn screening for cerebrotendinous xanthomatosis is the solution for early identification and treatment. J. Lipid Res. 2018, 59, 2214–2222. [Google Scholar] [CrossRef] [PubMed]
- Prais, D.; Steuer, G.; Inbar, O.; Kerem, E.; Blau, H. The impact of a national population carrier screening program on cystic fibrosis birth rate and age at diagnosis: Implications for newborn screening. J. Cyst Fibros. 2016, 15, 460–466. [Google Scholar] [CrossRef] [PubMed]
- Blau, H. Barrers to newborn screening for CF: Israel. Paper Presented at the 40th European Cystic Fibrosis Meeting, Seville, Spain, 7–10 June 2017. [Google Scholar]
- Raz, A.E.; Amano, Y.; Timmermans, S. Coming to terms with the imperfectly normal child: Attitudes of Israeli parents of screen-positive infants regarding subsequent prenatal diagnosis. J. Community Genet. 2019, 10, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Orenstein, N.; Glassberg, Y.M.; Shkalim-Zemer, V.; Basel-Salmon, L.; Averbuch, N.S.; Lagovsky, I.; Mark, A.G.; Amir, A.Z.; Bazak, L.; Cooper, S.; et al. Severe early-onset Wilson disease caused by a common pathogenic variant in the Bukharan Jewish population in Israel. Gene 2023, 887, 147728. [Google Scholar] [CrossRef] [PubMed]
- Raz, A.E.; Timmermans, S. Divergent evolution of newborn screening: Israel and the US as gene worlds. BioSocieties 2018, 13, 580–600. [Google Scholar] [CrossRef]
- Friedman, E. Next generation sequencing for newborn screening: Are we there yet? Genet. Res. 2015, 97, e17. [Google Scholar] [CrossRef] [PubMed]
- Reinstein, E. Challenges of using next generation sequencing in newborn screening. Genet. Res. 2015, 97, e21. [Google Scholar] [CrossRef]
- Jaradat, N. Establishing a National Newborn Screening Program in Jordan and Review of the Literature. Master’s Thesis, Sarah Lawrence College, Yonkers, NY, USA. May 2009. Available online: https://www.proquest.com/docview/305076548?pq-origsite=gscholar&fromopenview=true (accessed on 8 December 2022).
- Dababneh, S.; Alsbou, M.; Taani, N.; Sharkas, G.; Ismael, R.; Maraqa, L.; Nemri, O.; Al-Jawaldeh, H.; Kopti, N.; Atieh, E.; et al. Epidemiology of phenylketonuria disease in Jordan: Medical and nutritional challenges. Children 2022, 9, 402. [Google Scholar] [CrossRef]
- Carducci, C.; Amayreh, W.; Ababneh, H.; Mahasneh, A.; Al Rababah, B.; Al Qaqa, K.; Al Aqeel, M.; Artiola, C.; Tolve, M.; D’Amici, S.; et al. Molecular genetics of phenylketonuria and tetrahydrobiopterin deficiency in Jordan. JIMD Rep. 2020, 55, 59–67. [Google Scholar] [CrossRef]
- Megdadi, N.A.; Almigdad, A.; Alakil, M.O.; Alqiam, S.M.; Rababah, S.G.; Dwiari, M.A. Hereditary tyrosinemia type 1 in Jordan: A retrospective study. Int. J. Pediatr. 2021, 2021, 3327277. [Google Scholar] [CrossRef] [PubMed]
- Kasem, A.; Razeq, N.M.A.; Abuhammad, S.; Alkhazali, H. Mothers’ knowledge and attitudes about newborn screening in Jordan. J. Community Genet. 2022, 13, 215–225. [Google Scholar] [CrossRef] [PubMed]
- UNICEF and Ministry of Health. Newborn Screening Program Manual. January 2022. Available online: https://www.unicef.org/jordan/reports/newborns-screening-program-manual (accessed on 8 December 2022).
- Alsharhan, H.; Ahmed, A.A.; Ali, N.M.; Alahmad, A.; Albash, B.; Elshafie, R.M.; Alkanderi, S.; Elkazzaz, U.M.; Cyril, P.X.; Abdelrahman, R.M.; et al. Early Diagnosis of Classic homocystinuria in Kuwait through newborn screening: A 6-year experience. Int. J. Neonatal Screen. 2021, 7, 56. [Google Scholar] [CrossRef] [PubMed]
- Alabdulrazzaq, F.; AlSharhan, H.; Ahmed, A.; Marafie, M.; Sulaiman, I.; Alshafie, R.; AlAhmad, A.; AlBash, B.; Ali, N.; Cyril, P.; et al. Newborn screening experience for very long chain Acyl-CoA Dehydrogenase (VLCAD) deficiency in Kuwait. Genet. Med. 2022, 24 (Suppl. S1), S1. [Google Scholar] [CrossRef]
- Khneisser, I.; Adib, S.M.; Megarbane, A.; Lukacs, Z. International cooperation in the expansion of a newborn screening programme in Lebanon: A possible model for other programmes. J. Inherit. Metab. Dis. 2008, 31 (Suppl. S2), S441–S446. [Google Scholar] [CrossRef] [PubMed]
- Khneisser, I.; Adib, S.; Assaad, S.; Megarbane, A.; Karam, P. Cost-benefit analysis: Newborn screening for inborn errors of metabolism in Lebanon. J. Med. Screen. 2015, 22, 182–186. [Google Scholar] [CrossRef]
- Nakouzi, G.; Kreidieh, K.; Yazbek, S. A review of the diverse genetic disorders in the Lebanese population: Highlighting the urgency for community genetic services. J. Community Genet. 2015, 6, 83–105. [Google Scholar] [CrossRef] [PubMed]
- Farra, C.; Daher, R.; Badra, R.; el Rafei, R.; Bejjany, R.; Charafeddine, L.; Yunis, K. Incidence of alpha-globin gene defect in the Lebanese population: A pilot study. BioMed Res. Int. 2015, 2015, 517679. [Google Scholar] [CrossRef] [PubMed]
- Khoriaty, E.; Halaby, R.; Berro, M.; Sweid, A.; Abbas, H.A.; Inati, A. Incidence of sickle cell disease and other hemoglobin variants in 10,095 Lebanese neonates. PLoS ONE 2014, 9, e105109. [Google Scholar] [CrossRef] [PubMed]
- American University of Beirut Medical Center (AUBMC). Launching of the National Program of Neonatal Screening for Primary Immunodeficiency Diseases (NaSPID). 15 February 2018. Available online: https://aubmc.org.lb/Pages/National-Program-of-Neonatal-Screening-for-Primary-Immunodeficiency-Diseases.aspx (accessed on 15 November 2022).
- Khneisser, I. Newborn screening in Lebanon: 25 years’ experience. Paper Presented at the 4th Meeting of the Middle East North Africa (MENA) Region of the International Society for Neonatal Screening (ISNS), Limassol, Cyprus, 8–11 March 2020. [Google Scholar]
- Schickh, S.A. The Status of Newborn Screening in Libya: The Experiences of Physicians in Libya with Regards to Congenital Disorders and Their Attitudes toward Newborn Screening. Master’s Thesis, Sarah Lawrence College, Bronxville, NY, USA, 2014. [Google Scholar]
- Sladkevicius, E.; Pollitt, R.J.; Mgadmi, A.; Guest, J.F. Cost effectiveness of establishing a neonatal screening programme for phenylketonuria in Libya. Appl. Health Econ. Health Policy 2010, 8, 407–420. [Google Scholar] [CrossRef]
- Ismail, M.E.; Ben-Omran, T.; Mustafa, A.B.; Abdel-Azeem, A.M. Prevalence and profile of inherited metabolic disorders in Libya: Single center experience. World J. Biol. Pharm. Health Sci. 2022, 10, 60–64. [Google Scholar] [CrossRef]
- Lindner, M.; Abdoh, G.; Fang-Hoffmann, J.; Shabeck, N.; Al-Sayrafi, M.; Al-Janahi, M.; Ho, S.; Abdelrahman, M.O.; Ben-Omran, T.; Bener, A.; et al. Implementation of extended neonatal screening and a metabolic unit in the State of Qatar: Developing and optimizing strategies in cooperation with the Neonatal Screening Center in Heidelberg. J. Inherit. Metab. Dis. 2007, 30, 522–529. [Google Scholar] [CrossRef]
- David-Padilla, C.D.; Basilio, J.A.; Oliveros, Y.E. Newborn screening: Research to policy. Acta Med. Philipp. 2009, 43, 6–14. Available online: https://actamedicaphilippina.upm.edu.ph/index.php/acta/article/view/2554 (accessed on 3 February 2024).
- Rfifi, K.E. IEM and NSP Libya. Paper Presented at the 4th Meeting of the Middle East North Africa (MENA) Region of the International Society for Neonatal Screening (ISNS), Limassol, Cyprus, 8–11 March 2020. [Google Scholar]
- U.S. National Institutes of Health. Marrakech Declaration. Available online: http://links.lww.com/GIM/A81 (accessed on 2 November 2022).
- Fukushi, M. An international training and support programme for the establishment of neonatal screening in developing countries. J. Inherit. Metab. Dis. 2007, 30, 593–595. [Google Scholar] [CrossRef] [PubMed]
- Chabraoui, L. The Status of Newborn Screening Program in Morocco. Paper Presented at the 4th Meeting of the Middle East North Africa (MENA) Region of the International Society for Neonatal Screening (ISNS), Limassol, Cyprus, 8–11 March 2020. [Google Scholar]
- Maniar, S.; Amor, C.; Bijjou, A. Depistage de l’hypothyroidie congenitale au Maroc: Etude pilote. East. Mediterr. Health J. 2019, 24, 1066–1073. [Google Scholar] [CrossRef] [PubMed]
- Meiouet, F.; El Kabbaj, S.; Abilkassem, R.; Boemer, F. Moroccan experience of targeted screening for inborn errors of metabolism by tandem mass spectrometry. Pediatr. Rep. 2023, 15, 227–236. [Google Scholar] [CrossRef] [PubMed]
- El Idrissi Slitine, N.; Bennaoui, F.; Sable, C.A.; Martin, G.R.; Hom, L.A.; Fadel, A.; Moussaoui, S.; Inajjarne, N.; Boumzebra, D.; Mouaffak, Y.; et al. Pulse oximetry and congenital heart disease screening: Results of the first pilot study in Morocco. Int. J. Neonatal Screen. 2020, 6, 53. [Google Scholar] [CrossRef] [PubMed]
- Elbualy, M.; Bold, A.; De Silva, V.; Gibbons, U. Congenital hypothyroid screening: The Oman experience. J. Trop. Pediatr. 1998, 44, 81–83. [Google Scholar] [CrossRef] [PubMed]
- Oman Ministry of Health. Congenital Hypothyroidism Guideline for Neonatal Screening and Management. May 2021. Available online: https://www.moh.gov.om/documents/272928/4017900/Updated+Guidelines+for+the+Management+of+Congenital+Hypothyroidism+-+2021+%28Final%29.pdf/76648589-7fac-c2e9-b1ff-935086b0372e (accessed on 7 November 2022).
- Alkindi, S.; Al Zadjali, S.; Al Madhani, A.; Daar, S.; Al Haddabi, H.; Al Abri, Q.; Gravell, D.; Berbar, T.; Pravin, S.; Pathare, A.; et al. Forecasting hemoglobinopathy burden through neonatal screening in Omani neonates. Hemoglobin 2010, 34, 135–144. [Google Scholar] [CrossRef]
- Al-Madhani, A.; Pathare, A.; Al Zadjali, S.; Al Rawahi, M.; Al-Nabhani, I.; Alkindi, S. The use of HPLC as a tool for neonatal cord blood screening of haemoglobinopathy: A validation study. Mediterr. J. Hematol. Infect. Dis. 2019, 11, e2019005. [Google Scholar] [CrossRef] [PubMed]
- Joshi, S.N.; Bayoumi, R. Newborn screening program for Oman: The time is here and now. Oman Med. J. 2012, 27, 346–347. [Google Scholar] [CrossRef]
- Al-Riyami, S.; Al-Manei, M.; Al-Fahdi, A.; Al-Thihli, K. Establishment and validation of reference values for amino acids and acylcarnitines in dried blood spots for Omani newborns using tandem mass spectrometry. Oman Med. J. 2022, 37, e426. [Google Scholar] [CrossRef] [PubMed]
- Cook, M.C.; Farsi, T.A. A decade experience on severe combined immunodeficiency phenotype in Oman, bridging to newborn screening. Front. Immunol. 2021, 11, 623199. [Google Scholar] [CrossRef] [PubMed]
- Khandekar, R.; Khabori, M.; Jaffer Mohammed, A.; Gupta, R. Neonatal screening for hearing impairment—The Oman experience. Int. J. Pediatr. Otorhinolaryngol. 2006, 70, 663–670. [Google Scholar] [CrossRef] [PubMed]
- Mullaicharam, A.R. Challenges in neonatal screening in health care system. Int. J. Clin. Pharmacol. Pharmacother. 2018, 3, 139. [Google Scholar] [CrossRef] [PubMed]
- Newborn Screening in Qatar: An Interview with Dr. Skrinska. 13 September 2019. Available online: https://baebies.com/newborn-screening-qatar-skrinska/ (accessed on 2 December 2022).
- Gramer, G.; Abdoh, G.; Ben-Omran, T.; Shahbeck, N.; Ali, R.; Mahmoud, L.; Fang-Hoffmann, J.; Hoffmann, G.F.; Al Rifai, H.; Okun, J.G. Newborn screening for remethylation disorders and vitamin B12 deficiency-evaluation of new strategies in cohorts from Qatar and Germany. World J. Pediatr. 2017, 13, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Ramaswamy, M.; Skrinska, V.A.; Mitri, R.F.; Abdoh, G. Diagnosis of carnitine deficiency in extremely preterm neonates related to parenteral nutrition: Two step newborn screening approach. Int. J. Neonatal Screen. 2019, 5, 29. [Google Scholar] [CrossRef]
- Hamad Medical Corporation. For a Better Health of Qatar’s Newborns, HMC Rare Disease Center launches the First Q-Chip Based Expanded Genomic Newborn Screening Pilot Study. 5 March 2024. Available online: https://www.hamad.qa/EN/news/2024/March/Pages/For-a-better-health-of-Qatar-newborns-HMC-Rare-Disease-Center-launches.aspx (accessed on 27 March 2024).
- Abu-Osba, Y.K.; Mallouh, A.; Salamah, M.; Hann, R.; Thalji, A.; Hamdan, J.; Sa’di, A.A. Comprehensive newborn screening program: Aramco experience, the national need and recommendations. Ann. Saudi Med. 1992, 12, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, S.; Elsheikh, W.; Al-Aqeel, A.I.; Alhashem, A.M.; Alodaib, A.; Alahaideb, L.; Almashary, M.; Alharbi, F.; AlMalawi, H.; Ammari, A.; et al. Incidence of newborn screening disorders among 56,632 infants in Central Saudi Arabia. A 6-year study. Saudi Med. J. 2020, 41, 703–708. [Google Scholar] [CrossRef] [PubMed]
- Al-Sulaiman, A.; Kondkar, A.A.; Saeedi, M.Y.; Saadallah, A.; Al-Odaib, A.; Abu-Amero, K.K. Assessment of the knowledge and attitudes of Saudi mothers towards newborn screening. BioMed Res. Int. 2015, 2015, 718674. [Google Scholar] [CrossRef] [PubMed]
- Raja, E.H.-S.; Najwa, A.R.; Cordero, M.A.; Ganesh, K. Expanded newborn screening programs in Saudi Arabia; A questionnaire-based study. Biomed. J. Sci. Tech. Res. 2020, 32, 24632–24638. [Google Scholar] [CrossRef]
- Gosadi, I.M. National screening programs in Saudi Arabia: Overview, outcomes, and effectiveness. J. Infect. Public Health 2019, 12, 608–614. [Google Scholar] [CrossRef] [PubMed]
- Saudi Arabia Ministry of Health. MOH News: MOH Launches the 1st Phase of Newborn Screening for Hearing-Loss and CCHD Program. 9 October 2016. Available online: https://www.moh.gov.sa/en/Ministry/MediaCenter/News/Pages/News-2016-10-09-001.aspx (accessed on 11 November 2022).
- Aqeel, A.I. Expanded newborn screening programme in Saudi Arabia: Are We Ready? J. Paediatr. Child Health 2017, 53, 1242. [Google Scholar] [CrossRef] [PubMed]
- Bahshir, A.; Weissbecker, K.; Abdul-Mageed, A.; Andersson, H. Towards a uniform newborn screening panel in the Kingdom of Saudi Arabia. Acad. J. Ped. Neonatol. 2018, 6, 555753. [Google Scholar] [CrossRef]
- Alfadhel, M.; Al Othaim, A.; Al Saif, S.; Al Mutairi, F.; Alsayed, M.; Rahbeeni, Z.; Alzaidan, H.; Alowain, M.; Al-Hassnan, Z.; Saeedi, M.; et al. Expanded newborn screening program in Saudi Arabia: Incidence of screened disorders. J. Paediatr. Child Health 2017, 53, 585–591. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, S. Reflection on the expanded newborn screening programme in Saudi Arabia: Incidence of screened disorders. J. Paediatr. Child Health 2017, 53, 1034–1035. [Google Scholar] [CrossRef] [PubMed]
- Al-Mousa, H.; Al-Dakheel, G.; Jabr, A.; Elbadaoui, F.; Abouelhoda, M.; Baig, M.; Monies, D.; Meyer, B.; Hawwari, A.; Dasouki, M. High incidence of severe combined immunodeficiency disease in Saudi Arabia detected through combined T cell receptor excision circle and next generation sequencing of newborn dried blood spots. Front. Immunol. 2018, 9, 782. [Google Scholar] [CrossRef] [PubMed]
- Al-Yami, F.S.; Dar, F.K.; Yousef, A.I.; Al-Qurouni, B.H.; Al-Jamea, L.H.; Rabaan, A.A.; Quiambao, J.V.; Arulanantham, Z.J.; Woodman, A. A pilot study on screening for gestational/congenital toxoplasmosis of pregnant women at delivery in the Eastern Province of Saudi Arabia. Saudi Pharm. J. 2021, 29, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.R.; Alothaim, A.; Alfares, A.; Jowed, A.; Enazi, S.M.A.; Ghamdi, S.M.A.; Seneid, A.A.; Algahtani, A.; Zahrani, S.A.; AlFadhel, M.; et al. Cut-off values in newborn screening for inborn errors of metabolism in Saudi Arabia. Ann. Saudi Med. 2022, 42, 107–118. [Google Scholar] [CrossRef] [PubMed]
- AlAhaidib, L.Y.; AlOdaib, A.N. Health information exchange in relation to long-term follow-up data system in newborn screening program: General overview and the Saudi status. Saudi J. Health Syst. Res. 2022, 2, 45–53. [Google Scholar] [CrossRef]
- Mujamammi, A.H. Insights into national laboratory newborn screening and future prospects. Medicina 2022, 58, 272. [Google Scholar] [CrossRef] [PubMed]
- Saad, S.M.; Khan, K.M.; Alothaim, A. Accurate determination of biotinidase activity in serum by HPLC and its utilization as second tier test for the confirmation of initial positive newborn screening results. Mol. Genet. Metab. Rep. 2023, 38, 101045. [Google Scholar] [CrossRef] [PubMed]
- Sheikh Alshabab, L.I.; AlebrahIm, A.; Kaddoura, A.; Al-Fahoum, S. Congenital adrenal hyperplasia due to 21-hydroxylase deficiency: A five-year retrospective study in the Children’s Hospital of Damascus, Syria. Qatar Med. J. 2015, 2015, 11. [Google Scholar] [CrossRef]
- Saoud, M.; Al-Fahoum, S.; Kabalan, Y. Congenital hypothyroidism: A five-year retrospective study at Children’s University Hospital, Damascus, Syria. Qatar Med. J. 2019, 2019, 7. [Google Scholar] [CrossRef] [PubMed]
- Shennar, H.K.; Al-Asmar, D.; Kaddoura, A.; Al-Fahoum, S. Diagnosis and clinical features of organic acidemias: A hospital-based study in a single center in Damascus, Syria. Qatar Med. J. 2015, 2015, 9. [Google Scholar] [CrossRef] [PubMed]
- Screening for Health: Tunisia; International Atomic Energy Agency (IAEA), Department of Technical Cooperation: Vienna, Austria. 2003. Available online: https://inis.iaea.org/collection/NCLCollectionStore/_public/34/028/34028287.pdf (accessed on 2 November 2022).
- Hajer, S.; Neila, T.; Sondess, H.F.; Fekria, O.; Nabila, A.; Mahbouba, K.; Melika, D.; Faida, O.; Amina, B.; Raja, B.; et al. A lower-cost protocol for sickle cell disease neonatal screening in Tunisia. Ann. Saudi Med. 2012, 32, 49–52. [Google Scholar] [CrossRef] [PubMed]
- Khemir, S.; El Asmi, M.; Sanhaji, H.; Feki, M.; Jemaa, R.; Tebib, N.; Dhondt, J.L.; Ben Dridi, M.F.; Mebazaa, A.; Kaabachi, N. Phenylketonuria is still a major cause of mental retardation in Tunisia despite the possibility of treatment. Clin. Neurol. Neurosurg. 2011, 113, 727–730. [Google Scholar] [CrossRef] [PubMed]
- Mezzi, N.; Messaoud, O.; Mkaouar, R.; Zitouna, N.; Romdhane, S.; Abdessalem, G.; Charfeddine, C.; Maazoul, F.; Ouerteni, I.; Hamdi, Y.; et al. Spectrum of genetic diseases in Tunisia: Current situation and main milestones achieved. Genes 2021, 12, 1820. [Google Scholar] [CrossRef]
- Elloumi-Zghal, H.; Chaabouni, B.H. Genetics and genomic medicine in Tunisia. Mol. Genet. Genomic Med. 2018, 6, 134–159. [Google Scholar] [CrossRef] [PubMed]
- Al Hosani, H.; Salah, M.; Osman, H.M.; Farag, H.M.; El-Assiouty, L.; Saade, D.; Hertecant, J. Expanding the comprehensive national neonatal screening programme in the United Arab Emirates from 1995 to 2011. East. Mediterr. Health J. 2014, 20, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Al-Jasmi, F.A.; Al-Shamsi, A.; Hertecant, J.L.; Al-Hamad, S.M.; Souid, A.K. Inborn errors of metabolism in the United Arab Emirates: Disorders detected by newborn screening (2011–2014). JIMD Rep. 2016, 28, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Abdelmoety, D.A.; Almatary, A.; Alsheef, M.; AlFayyad, I. Epidemiology of thalassemia in Gulf Cooperation Council Countries: A systematic review. BioMed Res. Int. 2020, 2020, 1509501. [Google Scholar] [CrossRef] [PubMed]
- Elsaban, M.; Ahmad, D.; Toba, N.J.A.; Alsaeed, T.; Abusalah, Z.G. Five year evaluation of the newborn screening programme in Dubai, United Arab Emirates: A cross sectional study. In Proceedings of the Royal College of Pediatrics and Child Health Conference, Singapore, 6–7 March 2021. [Google Scholar]
- Al Mazrouei, S.K.; Moore, J.; Ahmed, F.; Mikula, E.B.; Martin, G.R. Regional implementation of newborn screening for critical congenital heart disease screening in Abu Dhabi. Pediatr. Cardiol. 2013, 34, 1299–1306. [Google Scholar] [CrossRef] [PubMed]
- Emirates News Agency—WAM. Ministry of Health Launches Critical Congenital Heart Disease Screening Programme for newborns. 27 June 2018. Available online: http://wam.ae/en/details/1395302696427 (accessed on 4 December 2022).
- Zarouni, S.A.; Mheiri, N.M.A.; Blooshi, K.A.; Serkal, Y.A.; Preman, N.; Naqvi, S.A.; Khan, Y. Impact of an electronic medical record-based automated screening program for critical congenital heart disease: Emirates Health Services, United Arab Emirates. BMC Med. Inform. Decis. Mak. 2022, 22, 165. [Google Scholar] [CrossRef] [PubMed]
- Ayas, M.; Yaseen, H. Emerging data from a newborn hearing screening program in Sharjah, United Arab Emirates. Int. J. Pediatr. 2021, 1, 2616890. [Google Scholar] [CrossRef] [PubMed]
- El-Din Riad, M. Newborn screening in United Arab Emirates. In Proceedings of the 4th Meeting of the Middle East North Africa (MENA) Region of the International Society for Neonatal Screening (ISNS), Limassol, Cyprus, 8–11 March 2020. [Google Scholar]
- Abou Tayoun, A.N. Unequal global implementation of genomic newborn screening. Nat. Rev. Genet. 2023, 24, 801–802. [Google Scholar] [CrossRef] [PubMed]
- Sirdah, M. (Biology Department, Al Azhar University-Gaza, Gaza, Palestine and Division of Hematology, Department of Internal Medicine, School of Medicine, University of Utah, Salt Lake City, UT, USA). Personal communication, 8 December 2022.
- Khatib, S.; Ayyad, A. A pilot study on an expanded newborn screening program in Palestine—Phase II. In Proceedings of the U.S. Newborn Screening and Genetic Testing Symposium, Anaheim, CA, USA, 27–30 October 2014; Available online: https://www.aphl.org/conferences/proceedings/documents/2014/nbs/59katib.pdf (accessed on 5 December 2022).
- Sirdah, M.M.; Al-Kahlout, M.S.; Reading, N.S. National G6PD neonatal screening program in Gaza Strip of Palestine: Rationale, challenges and recommendations. Clin. Genet. 2016, 90, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Ghannam, I.A.; Kharroubi, A.T.; Mustafa, R.M.; Jamous, S.M.; Almufreh, S.M.; Othman, N.M. Establishment of a cutoff value of phenylalanine for phenylketonuria among West Bank-Palestinian neonates. Jordan J. Biol. Sci. 2020, 13, 177–181. [Google Scholar]
- Barakat, M. Shortage of PKU Test Materials Threatens Gaza Newborns. Nour Abu Aisha. 26 January 2021. Available online: https://www.aa.com.tr/en/world/shortage-of-pku-test-materials-threatens-gaza-newborns/212333 (accessed on 3 February 2024).
- Grant, M.; Kabakian-Khasholian, T.; Yazbek, S. Interventions addressing genetic disease burdens within selected countries in the MENA region: A scoping review. J. Community Genet. 2023, 14, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Saffi, M.; Howard, N. Exploring the effectiveness of mandatory premarital screening and genetic counselling programmes for β-thalassaemia in the Middle East: A scoping review. Public Health Genom. 2015, 18, 193–203. [Google Scholar] [CrossRef] [PubMed]
- Inati, A.; Zeineh, N.; Isma’eel, H.I.; Koussa, S. β-Thalassaemia: The Lebanese experience. Clin. Lab. Haem. 2006, 28, 217–227. [Google Scholar] [CrossRef] [PubMed]
- Rouh AlDeen, N.; Osman, A.A.; Alhabashi, M.J.; Al Khaldi, R.; Alawadi, H.; Alromh, M.K.; Alyafai, E.G.; Akbulut-Jeradi, N. The prevalence of β-thalassemia and other hemoglobinopathies in Kuwaiti premarital screening program: An 11-year experience. J. Pers. Med. 2021, 11, 980. [Google Scholar] [CrossRef] [PubMed]
- Al-Shafai, M.; Al-Romaihi, A.; Al-Hajri, N.; Islam, N.; Adawi, K. Knowledge and Perception of and Attitude toward a Premarital Screening Program in Qatar: A Cross-Sectional Study. Int. J. Environ. Res. Public Health 2022, 19, 4418. [Google Scholar] [CrossRef] [PubMed]
- Ahramonline. Explainer: Egypt’s Pre-Marriage Medical Tests Go into Effect. 26 February 2023. Available online: https://english.ahram.org.eg/News/489781.aspx (accessed on 21 August 2023).
- United Nations—Population Division. World Population Prospects 2022: Summary of Results. Available online: https://www.un.org/development/desa/pd/content/World-Population-Prospects-2022 (accessed on 6 July 2023).
- GBD 2021 Sickle Cell Disease Collaborators. Global, Regional, and National Prevalence and Mortality Burden of Sickle Cell Disease, 2000–2021: A Systematic Analysis from the Global Burden of Disease Study 2021. Lancet Haematol. 2023, 10, e585–e599. [Google Scholar] [CrossRef] [PubMed]
- Piel, F.B.; Rees, D.C.; DeBaun, M.R.; Nnodu, O.; Ranque, B.; Thompson, A.A.; Ware, R.E.; Abboud, M.R.; Abraham, A.; Ambrose, E.E.; et al. Defining global strategies to improve outcomes in sickle cell disease: A Lancet Haematology Commission. Lancet Haematol. 2023, 10, e633–e686. [Google Scholar] [CrossRef] [PubMed]
- Rahimy, M.C.; Gangbo, A.; Ahouignan, G.; Alihonou, E. Newborn screening for sickle cell disease in the Republic of Benin. J. Clin. Pathol. 2009, 62, 46–48. [Google Scholar] [CrossRef] [PubMed]
- Ohene-Frempong, K.; Oduro, J.; Tetteh, H.; Nkrumah, F. Screening newborns for sickle cell disease in Ghana. Pediatrics 2008, 121, S120–S121. [Google Scholar] [CrossRef]
- Tshilolo, L.; Kafando, E.; Sawadogo, M.; Cotton, F.; Vertongen, F.; Ferster, A.; Gulbis, B. Neonatal screening and clinical care programmes for sickle cell disorders in sub-Saharan Africa: Lessons from pilot studies. Public Health 2008, 122, 933–941. [Google Scholar] [CrossRef] [PubMed]
- Kuznik, A.; Habib, A.G.; Munube, D.; Lamorde, M. Newborn screening and prophylactic interventions for sickle cell disease in 47 countries in sub-Saharan Africa: A cost-effectiveness analysis. BMC Health Serv. Res. 2016, 16, 304. [Google Scholar] [CrossRef] [PubMed]
- Adeniran, K.; Limbe, M. Review article on congenital hypothyroidism and newborn screening program in Africa; the present situation and the way forward. J. Thyroid Disord. Ther. 2012, 1, 102. Available online: https://www.longdom.org/open-access/review-article-on-congenital-hypothyroidism-and-newborn-screening-program-in-africa-the-present-situation-and-the-way-fo-6208.html (accessed on 3 February 2024).
- Yarhere, I.E.; Jaja, T.; Briggs, D.; Iughetti, L. Newborn screening in Nigeria: Associating the screening of congenital hypothyroidism and sickle cell disease can be a winning choice? Acta Biomed. 2019, 90, 316–320. [Google Scholar] [CrossRef] [PubMed]
- Twum, S.; Fosu, K.; Felder, R.A.; Sarpong, K.A.N. Bridging the gaps in newborn screening programmes: Challenges and opportunities to detect haemoglobinopathies in Africa. Afr. J. Lab. Med. 2023, 12, 2225. [Google Scholar] [CrossRef] [PubMed]
- American Society of Hematology. Consortium on Newborn Screening in Africa. Available online: https://www.hematology.org/global-initiatives/consortium-on-newborn-screening-in-africa (accessed on 6 July 2023).
- Green, N.S.; Zapfel, A.; Nnodu, O.E.; Franklin, P.; Tubman, V.N.; Chirande, L.; Kiyaga, C.; Chunda-Liyoka, C.; Awuonda, B.; Ohene-Frempong, K.; et al. The Consortium on Newborn Screening in Africa for sickle cell disease: Study rationale and methodology. Blood Adv. 2022, 6, 6187–6197. [Google Scholar] [CrossRef] [PubMed]
- Paintsil, V.; Amuzu, E.X.; Nyanor, I.; Asafo-Adjei, E.; Mohammed, A.R.; Yawnumah, S.A.; Oppong-Mensah, Y.G.; Nguah, S.B.; Obeng, P.; Dogbe, E.E.; et al. Establishing a sickle cell disease registry in Africa: Experience from the Sickle Pan-African Research Consortium, Kumasi-Ghana. Front. Genet. 2022, 13, 802355. [Google Scholar] [CrossRef] [PubMed]
- Makani, J.; Sangeda, R.Z.; Nnodu, O.; Nembaware, V.; Osei-Akoto, A.; Paintsil, V.; Balandya, E.; Kent, J.; Luzzatto, L.; Ofori-Acquah, S.; et al. SickleInAfrica. Lancet Haematol. 2020, 7, e98–e99. [Google Scholar] [CrossRef] [PubMed]
- Therrell, B.L., Jr.; Lloyd-Puryear, M.A.; Ohene-Frempong, K.; Ware, R.E.; Padilla, C.D.; Ambrose, E.E.; Barkat, A.; Ghazal, H.; Kiyaga, C.; Mvalo, T.; et al. Empowering newborn screening programs in African countries through establishment of an international collaborative effort. J. Community Genet. 2020, 11, 253–268. [Google Scholar] [CrossRef] [PubMed]
- Nnodu, O.; Isa, H.; Nwegbu, M.; Ohiaeri, C.; Adegoke, S.; Chianumba, R.; Ugwu, N.; Brown, B.; Olaniyi, J.; Okocha, E.; et al. HemoTypeSC, a low-cost point-of-care testing device for sickle cell disease: Promises and challenges. Blood Cells Mol. Dis. 2019, 78, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Ilyas, S.; Simonson, A.E.; Asghar, W. Emerging point-of-care technologies for sickle cell disease diagnostics. Clin. Chim. Acta 2020, 501, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Dexter, D.; McGann, P.T. Saving lives through early diagnosis: The promise and role of point of care testing for sickle cell disease. Br. J. Haematol. 2022, 196, 63–69. [Google Scholar] [CrossRef] [PubMed]
- McGann, P.T.; Hoppe, C. The pressing need for point-of-care diagnostics for sickle cell disease: A review of current and future technologies. Blood Cells Mol. Dis. 2017, 67, 104–113. [Google Scholar] [CrossRef] [PubMed]
- Hsu, L.; Nnodu, O.E.; Brown, B.J.; Tluway, F.; King, S.; Dogara, L.G.; Patil, C.; Shevkoplyas, S.S.; Lettre, G.; Cooper, R.S.; et al. White paper: Pathways to progress in newborn screening for sickle cell disease in sub-Saharan Africa. J. Trop. Dis. Public Health 2018, 6, 260. [Google Scholar] [CrossRef] [PubMed]
- Oron, A.P.; Chao, D.L.; Ezeanolue, E.E.; Ezenwa, L.N.; Piel, F.B.; Ojogun, O.T.; Uyoga, S.; Williams, T.N.; Nnodu, O.E. Caring for Africa’s sickle cell children: Will we rise to the challenge? BMC Med. 2020, 18, 92. [Google Scholar] [CrossRef] [PubMed]
- Esoh, K.; Wonkam-Tingang, E.; Wonkam, A. Sickle cell disease in sub-Saharan Africa: Transferable strategies for prevention and care. Lancet Haematol. 2021, 8, e744–e755. [Google Scholar] [CrossRef] [PubMed]
- Egesa, W.I.; Nakalema, G.; Waibi, W.M.; Turyasiima, M.; Amuje, E.; Kiconco, G.; Odoch, S.; Kumbakulu, P.K.; Abdirashid, S.; Asiimwe, D. Sickle cell disease in children and adolescents: A review of the historical, clinical, and public health perspective of sub-Saharan Africa and beyond. Int. J. Pediatr. 2022, 2022, 3885979. [Google Scholar] [CrossRef] [PubMed]
- Nnodu, O.E.; Sopekan, A.; Nnebe-Agumadu, U.; Ohiaeri, C.; Adeniran, A.; Shedul, G.; Isa, H.A.; Owolabi, O.; Chianumba, R.I.; Tanko, Y.; et al. Implementing newborn screening for sickle cell disease as part of immunisation programmes in Nigeria: A feasibility study. Lancet Haematol. 2020, 7, e534–e540, Erratum in Lancet Haematol 2020, 7, e560. [Google Scholar] [CrossRef] [PubMed]
- Streetly, A. Screening infants for sickle cell disease in sub-Saharan Africa: Starting the journey to a sustainable model in primary care. Lancet Haematol. 2020, 7, e503–e504. [Google Scholar] [CrossRef] [PubMed]
- Smart, L.R.; Ware, R.E. Newborn screening for sickle cell disease in sub-Saharan Africa: Is the glass half-full yet? Pediatr. Blood Cancer 2021, 68, e29137. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization, Regional Committee for Africa. Progress in the Implementation of the African Region Sickle-Cell Strategy 2010–2020. Available online: https://apps.who.int/iris/handle/10665/92663 (accessed on 8 July 2023).
- Ezenwa, M.O.; Dennis-Antwi, J.A.; Dallas, C.M.; Amarachukwu, C.; Ezema, C.I.; Yao, Y.; Gallo, A.M.; Wilkie, D.J. The crisis of sickle cell disease in Africa from insights into primary prevention in Ghana and Nigeria: Notes from the field. J. Immigr. Minor. Health 2021, 23, 871–878. [Google Scholar] [CrossRef] [PubMed]
- Maia, M. App Will Track Newborns Diagnosed with SCD in 6 African Countries. Sickle Cell Disease News. 14 June 2022. Available online: https://sicklecellanemianews.com/news/tracking-app-newborns-with-scd-be-introduced-african-countries/ (accessed on 7 July 2023).
- Archer, N.M.; Inusa, B.; Makani, J.; Nkya, S.; Tshilolo, L.; Tubman, V.N.; McGann, P.T.; Ambrose, E.E.; Henrich, N.; Spector, J.; et al. Enablers and barriers to newborn screening for sickle cell disease in Africa: Results from a qualitative study involving programmes in six countries. BMJ Open 2022, 12, e057623. [Google Scholar] [CrossRef] [PubMed]
- Segbefia, C.I.; Goka, B.; Welbeck, J.; Amegan-Aho, K.; Dwuma-Badu, D.; Rao, S.; Salifu, N.; Oppong, S.A.; Odei, E.; Ohene-Frempong, K.; et al. Implementing newborn screening for sickle cell disease in Korle Bu Teaching Hospital, Accra: Results and lessons learned. Pediatr. Blood Cancer 2021, 68, e29068. [Google Scholar] [CrossRef] [PubMed]
- Wesonga, R.M.; Awe, O.I. An assessment of traditional and genomic screening in newborns and their applicability for Africa. Inform. Med. Unlocked. 2022, 32, 101050. [Google Scholar] [CrossRef]
- Thalassaemia International Federation. Sickle Cell Disease—African Health Ministers Launch Drive to Curb SCD’s Toll. Available online: https://thalassaemia.org.cy/news/african-health-ministers-launch-drive-to-curb-scd-toll/ (accessed on 6 July 2023).
- Fondation Pierre Fabre. African Sickle-Cell Initiative: Advocating for Sickle-Cell Disease to Be Included in the Global Health Agenda. Available online: https://www.fondationpierrefabre.org/en/current-initiatives/african-sickle-cell-disease-initiative-advocating-for-sickle-cell-disease-to-be-included-in-the-global-health-agenda/ (accessed on 7 August 2023).
- McGann, P.T.; Ferris, M.G.; Ramamurthy, U.; Santos, B.; de Oliveira, V.; Bernardino, L.; Ware, R.E. A prospective newborn screening and treatment program for sickle cell anemia in Luanda, Angola. Am. J. Hematol. 2013, 88, 984–989. [Google Scholar] [CrossRef] [PubMed]
- McGann, P.T.; Grosse, S.D.; Santos, B.; de Oliveira, V.; Bernardino, L.; Kassebaum, N.J.; Ware, R.E.; Airewele, G.E. A cost-effectiveness analysis of a pilot neonatal screening program for sickle cell anemia in the Republic of Angola. J. Pediatr. 2015, 167, 1314–1319. [Google Scholar] [CrossRef] [PubMed]
- Piety, N.; George, A.; Serrano, S.; Lanzi, M.R.; Patel, P.R.; Noli, M.P.; Kahan, S.; Nirenberg, D.; Camanda, J.F.; Airewele, G.; et al. A Paper-based test for screening newborns for sickle cell disease. Sci. Rep. 2017, 7, 45488. [Google Scholar] [CrossRef]
- Brito, M.; Inusa, B.P.D.; Ginete, C.; Mendes, M.; Afonso, R.; Valentim, I.; Siatembo, A.; Vaconcelos, J.N. Implementation of a newborn screening for sickle cell diseae, at the Hospital Materno Infantil Dr. Manuel Pedro Azancot de Menezes, Angola. Blood 2023, 142 (Suppl. S1), 5300. [Google Scholar] [CrossRef]
- Zohoun, A.; Baglo Agbodande, T.; Zohoun, L.; Anani, L. Prevalence of hemoglobin abnormalities in an apparently healthy population in Benin. Hematol. Transfus. Cell Ther. 2020, 42, 145–149. [Google Scholar] [CrossRef] [PubMed]
- Kafando, E.; Nacoulma, E.; Ouattara, Y.; Ayéroué, J.; Cotton, F.; Sawadogo, M.; Gulbis, B. Neonatal haemoglobinopathy screening in Burkina Faso. J. Clin. Pathol. 2009, 62, 39–41. [Google Scholar] [CrossRef] [PubMed]
- Fondation Pierre Fabre. Prevention, Diagnosis and Treatment of Sickle Cell Disease—Burkina Faso. Available online: https://www.fondationpierrefabre.org/en/our-programmes/combatting-sickle-cell-disease/prevention-diagnosis-treatment-sickle-cell-disease/ (accessed on 7 August 2023).
- Sawadogo, S.; Nebie, K.; Kima, D.; Savadogo, H.; Sanou, J.; Ouedraogo, D.; Ouedraogo, P.; Zamane, H.; Ndiaye, A.; Kafando, E. Incidence of sickle cell disease and other hemoglobinopathies in Burkina Faso: Results of a five-year systematic neonatal screening (2015–2019) in four urban hospitals. Open J. Blood Dis. 2022, 12, 87–97. [Google Scholar] [CrossRef]
- Mutesa, L.; Boemer, F.; Ngendahayo, L.; Rulisa, S.; Rusingiza, E.K.; Cwinya-Ay, N.; Mazina, D.; Kariyo, P.C.; Bours, V.; Schoos, R. Neonatal screening for sickle cell disease in Central Africa: A study of 1825 newborns with a new enzyme-linked immunosorbent assay test. J. Med. Screen. 2007, 14, 113–116. [Google Scholar] [CrossRef] [PubMed]
- Wonkam, A.; Ngo Bitoungui, V.J.; Ngogang, J. Perspectives in genetics and sickle cell disease prevention in Africa: Beyond the preliminary data from Cameroon. Public Health Genom. 2015, 18, 237–241. [Google Scholar] [CrossRef] [PubMed]
- Adele, B.C.; Mungyeh, M.E.; Suzanne, N.U.S.; Alkali, Y.S.; Agwu, C.; Bernard, C.C.; Ekoe, T.; Oduwole, A.; Andreas, C. Newborn thyroid stimulating hormone levels in heel prick blood at the Yaounde Gyneco-Obstetric and Paediatric Hospital Cameroon. Endocrinol. Metab. Syndr. 2018, 7, 287. [Google Scholar] [CrossRef]
- Ngwengi, N.Y.; Fon, P.N.; Mbanya, D. Distribution of haemoglobin genotypes, knowledge, attitude and practices towards sickle cell disease among unmarried youths in the Buea Health District, Cameroon. Pan Afr. Med. J. 2020, 37, 109. [Google Scholar] [CrossRef] [PubMed]
- Fondation Pierre Fabre. Supporting the Fight against Sickle Cell Disease in Sub-Saharan Africa. Available online: https://www.fondationpierrefabre.org/en/our-programmes/combatting-sickle-cell-disease/supporting-the-fight-against-sickle-cell-disease-in-sub-saharan-africa/ (accessed on 7 August 2023).
- Fondation Pierre Fabre. Support for the Sickle Cell Research and Treatment Centre (CRTD). Available online: https://www.fondationpierrefabre.org/en/our-programmes/combatting-sickle-cell-disease/support-for-the-sickle-cell-research-and-treatment-centre-crtd/ (accessed on 7 August 2023).
- Fondation Pierre Fabre. The Fight against Sickle Cell Disease in Central African Republic Is Moving Forward! Available online: https://www.fondationpierrefabre.org/en/current-initiatives/the-fight-against-sickle-cell-disease-in-the-central-african-republic-is-moving-forward/ (accessed on 7 August 2023).
- Mohamed, K.S.; Abasse, K.S.; Abbas, M.; Sintali, D.N.; Baig, M.M.F.A.; Cote, A. An overview of the healthcare systems in Comoros: The effects of two decades of political instability. Ann. Glob. Health 2021, 87, 84. [Google Scholar] [CrossRef] [PubMed]
- Badens, C.; Martinez di Montemuros, F.; Thuret, I.; Michel, G.; Mattei, J.F.; Cappellini, M.D.; Lena-Russo, D. Molecular basis of haemoglobinopathies and G6PD deficiency in the Comorian population. Hematol. J. 2000, 1, 264–268. [Google Scholar] [CrossRef] [PubMed]
- Tshilolo, L.; Aissi, L.M.; Lukusa, D.; Kinsiama, C.; Wembonyama, S.; Gulbis, B.; Vertongen, F. Neonatal screening for sickle cell anaemia in the Democratic Republic of the Congo: Experience from a pioneer project on 31 204 newborns. J. Clin. Pathol. 2009, 62, 35–38. [Google Scholar] [CrossRef] [PubMed]
- Katamea, T.; Mukuku, O.; Mpoy, C.W.; Mutombo, A.K.; Luboya, O.N.; Wembonyama, S.O. Factors associated with acceptability of newborn screening for sickle cell disease in Lubumbashi City, Democratic Republic of the Congo. Glob. J. Med. Pharm. Biomed. Update 2022, 17, 5. [Google Scholar] [CrossRef]
- Mumbere, M.; Batina-Agasa, S.; Uvoya, N.A.; Kasai, E.T.; Kombi, P.K.; Djang’eing’a, R.M.; Alworong’a Opara, J.-P. Newborn screening for sickle cell disease in Butembo and Beni: A pilot experience in a highland region of the Democratic Republic of Congo. Pan. Afr. Med. J. 2023, 45, 56. Available online: https://www.panafrican-med-journal.com//content/article/45/56/full (accessed on 3 February 2024).
- Katawandja, A.L.; Nzengu-Lukusa, F.; Nzongola-Nkasu, D.K.; Muepu, L.T. Neonatal screening for sickle cell disease in urban areas with limited resources: Case of Kindu City, East of the Democratic Republic of Congo. Open Access Libr. J. 2021, 8, e7097. [Google Scholar] [CrossRef]
- Kasai, E.T.; Boemer, F.; Djang’eing’a, R.M.; Ntokamunda, J.K.; Batina Agasa, S.; Dauly, N.N.; Alworong’a Opara, J.-P. Systematic screening of neonatal sickle cell disease with HemoTypeSCTM Kit-test: Case study and literature review. Open J. Blood Dis. 2020, 10, 12–21. [Google Scholar] [CrossRef]
- Kasai, E.T.; Kadima, J.N.; Alworong’a Opara, J.-P.; Boemer, F.; Dresse, M.F.; Makani, J.; Bours, V.; Marini Djang’eing’a, R.; Paul, K.K.; Batina Agasa, S. Pairing parents and offspring’s HemoTypeSCTM test to validate results and confirm sickle cell pedigree: A case study in Kisangani, the Democratic Republic of the Congo. Hematology 2022, 27, 853–859. [Google Scholar] [CrossRef]
- Aimé, A.K.; Etienne, S.M.; Mbongi, D.; Nsonso, D.; Serrao, E.; Léon, T.M.M.; Oscar, L.N.; Stanis, W.O. Dépistage hospitalier de la drépanocytose en République Démocratique du Congo (RDC) par HemoTypeSC: Cas de la ville de Kindu [HemoTypeSC screening for sickle cell disease in the Democratic Republic of Congo (DRC): A case from the city of Kindu]. Pan. Afr. Med. J. 2022, 41, 134. (In French) [Google Scholar] [CrossRef] [PubMed]
- Kasai, E.T.; Gulbis, B.; Ntukamunda, J.K.; Bours, V.; Batina Agasa, S.; Marini Djang’eing’a, R.; Boemer, F.; Katenga Bosunga, G.; Ngbonda Dauly, N.; Sokoni Vutseme, J.; et al. Newborn screening for sickle cell disease in Kisangani, Democratic Republic of the Congo: An update. Hematology 2023, 28, 2213043. [Google Scholar] [CrossRef] [PubMed]
- Kasai, E.T.; Alworong’a Opara, J.P.; Ntokamunda, J.K.; Kalenga, M.; Batina Agasa, S.; Marini Djang’eing’a, R.; Boemer, F. Overview of current progress and challenges in diagnosis, and management of pediatric sickle cell disease in Democratic Republic of the Congo. Hematology 2022, 27, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Dokekias, A.E.; Ocko Gokaba, L.T.; Louokdom, J.S.; Ocini, L.N.; Galiba Atipo Tsiba, F.O.; Ondzotto Ibatta, C.I.; Kouandzi, Q.N.; Tamekue, S.T.; Bango, J.C.; Nziengui Mboumba, J.V.; et al. Neonatal screening for sickle cell disease in Congo. Anemia 2022, 2022, 9970315. [Google Scholar] [CrossRef] [PubMed]
- Kakou Danho, J.B.; Atiméré, Y.N.; Koné, D.; Yéo, D.D.; Couitchéré, L. Feasibility study of the “HemoTypeSC” Test for the rapid screening of sickle cell disease in Côte D’Ivoire. Adv. Hematol. 2021, 2021, 8862039. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.; Yang, L.Y.; Xie, D.D.; Chen, J.T.; Nguba, S.M.; Ehapo, C.S.; Zhan, X.F.; Eyi, J.U.; Matesa, R.A.; Obono, M.M.; et al. G6PD deficiency and hemoglobinopathies: Molecular epidemiological characteristics and healthy effects on malaria endemic Bioko Island, Equatorial Guinea. PLoS ONE 2015, 10, e0123991. [Google Scholar] [CrossRef] [PubMed]
- Teasdale, C.A.; Tsiouries, F.; Mafukidze, A.; Shongwe, S.; Choy, M.; Nhlengetfwa, H.; Simelane, S.; Mthethwa, S.; Ao, T.; Ryan, C.; et al. Birth testing for infant HIV diagnosis in Eswatini: Implementation experience and uptake among women living with HIV in Manzini Region. Pediatr. Infect. Dis. J. 2020, 39, e235–e241. [Google Scholar] [CrossRef] [PubMed]
- Tarekegn, B.G. (University of Gondor, Gondor, Ethiopia). Personal communication, 2022.
- Feleke, Y.; Enquoselassie, F.; Deneke, F.; Abdulkadir, J.; Hawariat, G.W.; Tilahun, M.; Mekbib, T. Neonatal congenital hypothyroidism screening in Addis Ababa, Ethiopia. East Afr. Med. J. 2000, 77, 377–381. [Google Scholar] [PubMed]
- Mombo, L.E.; Makosso, L.K.; Bisseye, C.; Mbacky, L.; Setchell, J.M.; Edou, A. Acceptability of neonatal sickle cell disease screening among parturient women at the Paul Moukambi Regional Hospital in rural Eastern Gabon, Central Africa. Af. J. Reprod. Health 2021, 25, 72–77. [Google Scholar] [PubMed]
- Loembet, L.M.D.; Mezui-me-ndong, J.; Mboro, T.M.; Sicas, L.; Feudjo, M.; Bisvigou, U.; Koko, J.; Ducrocq, R.; Gonzalez, J.-P. Neonatal screening of sickle cell disease in Gabon: A nationwide study. J. Neonatal Biol. 2022, 11, 376. [Google Scholar]
- Wikipedia. Kwaku Ohene-Frempong. Available online: https://en.wikipedia.org/wiki/Kwaku_Ohene-Frempong (accessed on 26 July 2023).
- Steele, C.; Sinski, A.; Asibey, J.; Hardy-Dessources, M.D.; Elana, G.; Brennan, C.; Odame, I.; Hoppe, C.; Geisberg, M.; Serrao, E.; et al. Point-of-care screening for sickle cell disease in low-resource settings: A multi-center evaluation of HekimoTypeSC, a novel rapid test. Am. J. Hematol. 2019, 94, 39–45. [Google Scholar] [CrossRef] [PubMed]
- An, R.; Huang, Y.; Rocheleau, A.; Avanaki, A.; Thota, P.; Zhang, Q.; Man, Y.; Sekyonda, Z.; Segbefia, C.I.; Dei-Adomakoh, Y.; et al. Multispectral imaging for microchip electrophoresis enables point-of-care newborn hemoglobin variant screening. Heliyon 2022, 8, e11778. [Google Scholar] [CrossRef] [PubMed]
- Sims, A.M.; Bonsu, K.O.; Urbonya, R.; Farooq, F.; Tavernier, F.; Yamamoto, M.; VanOmen, S.; Halford, B.; Gorodinsky, P.; Issaka, R.; et al. Diagnosis patterns of sickle cell disease in Ghana: A secondary analysis. BMC Public Health 2021, 21, 1719. [Google Scholar] [CrossRef] [PubMed]
- Pfizer. Pfizer 2019 Annual Review: Partnering to Uncover Sickle Cell Disease in Sub-Saharan Africa. Available online: https://www.pfizer.com/sites/default/files/investors/financial_reports/annual_reports/2019/our-bold-moves/lead-the-conversation/partnering-to-uncover-sickle-cell-disease-in-sub-saharan-africa/index.html (accessed on 27 July 2023).
- Clinton Health Access Initiative. Ghana Sickle Cell Disease Program Screens Over 24,000 Infants. 19 June 2023. Available online: https://www.clintonhealthaccess.org/blog/sickle-cell-disease-program-in-ghana-screens-over-24000-infants/ (accessed on 26 July 2023).
- Millimono, T.S.; Loua, K.M.; Rath, S.L.; Relvas, L.; Bento, C.; Diakite, M.; Jarvis, M.; Daries, N.; Ribeiro, L.M.; Manco, L.; et al. High prevalence of hemoglobin disorders and glucose-6-phosphate dehydrogenase (G6PD) deficiency in the Republic of Guinea (West Africa). Hemoglobin 2012, 36, 25–37. [Google Scholar] [PubMed]
- Fondation Pierre Fabre. Haemophilia and Sickle Cell Disease: Increasing Our Impact Together in Guinea. Available online: https://www.fondationpierrefabre.org/en/combating-sickle-cell-disease/haemophilia-and-sickle-cell-disease-increasing-our-impact-together-in-guinea/ (accessed on 7 August 2023).
- Menzato, F.; Bosa, L.; Sifna, A.; Da Silva, L.; Gasperoni, E.; Martella, M.; Sanha, M.S.; Mustik, A.; Da Dalt, L.; Riccarddi, F.; et al. Successful simultaneous screening of sickle cell disease, HIV and tuberculosis in rural Guinea Bissau, West Africa through rapid tests and a standardized clinical questionnaire: An outreach program due to a public-private partnership. Blood 2018, 132, 4715. [Google Scholar] [CrossRef]
- Martella, M.; Campeggio, M.; Pulè, G.; Wonkam, A.; Menzato, F.; Munaretto, V.; Viola, G.; Da Costa, S.P.; Reggiani, G.; Araujo, A.; et al. Distribution of HbS allele and haplotypes in a multiethnic population of Guinea Bissau, West Africa: Implications for public health screening. Front. Pediatr. 2022, 10, 826262. [Google Scholar] [CrossRef]
- Kuta, E.S.M.; Tenge, C.N.; Ganda, B.K.O.; Njuguna, F.M. Newborn screening for sickle cell disease at Kisumu County Hospital, Kisumu—Kenya. East Afr. Med. J. 2019, 96, 2419–2429. Available online: https://www.ajol.info/index.php/eamj/article/view/198133 (accessed on 3 February 2024).
- Korir, R.K. Newborn Screening Program for Sickle Cell Disease in Western Kenya. In Proceedings of the 18th World Hematology Congress, London, UK, 16–17 March 2020; Available online: https://hematology.insightconferences.com/abstract/2020/newborn-screening-program-for-sickle-cell-disease-in-western-kenya (accessed on 1 August 2023).
- Science Africa. Kenya: Kisumu Launches Newborn Sickle Cell Screening Centre. 16 December 2021. Available online: https://news.scienceafrica.co.ke/kenya-kisumu-launches-newborn-sickle-cell-screening-centre/ (accessed on 1 August 2023).
- Quest Laboratories. Newborn Screening. Available online: https://questlabs-kenya.com/newborn-screening/ (accessed on 27 July 2023).
- ibs (Kenya Institute of Biological Sciences). Newborn Screening. Available online: https://www.kibs.co.ke/services/postnatal-genetic-testing-Kenya.php (accessed on 27 July 2023).
- Tubman, V.N.; Marshall, R.; Jallah, W.; Guo, D.; Ma, C.; Ohene-Frempong, K.; London, W.B.; Heeney, M.M. Newborn screening for sickle cell disease in Liberia: A pilot study. Pediatr. Blood Cancer 2016, 63, 671–676. [Google Scholar] [CrossRef] [PubMed]
- Udhayashankar, K.; Franklin, P.D.; Nuta, C.J.; Cherue, A.K.; Haq, H.; Thompson, D.; Tubman, V.N. Determinants of retention in care of newborns diagnosed with sickle cell disease in Liberia: Results from a mixed-methods study of caregivers. PLoS Glob. Public Health 2023, 3, e0001705. [Google Scholar] [CrossRef]
- Business Insider Africa. Liberia: Ministry of Health, Consortium on Newborn Screening in Africa (CONSA) Launch Newborn Screening for Sickle Cell Disease in Liberia. 20 September 2021. Available online: https://africa.businessinsider.com/apo/liberia-ministry-of-health-consortium-on-newborn-screening-in-africa-consa-launch/c3w7gy7 (accessed on 3 August 2023).
- Tegha, G.; Topazian, H.M.; Kamthunzi, P.; Howard, T.; Tembo, Z.; Mvalo, T.; Chome, N.; Kumwenda, W.; Mkochi, T.; Hernandez, A.; et al. Prospective newborn screening for sickle cell disease and other inherited blood disorders in Central Malawi. Int. J. Public Health 2021, 66, 629338. [Google Scholar] [CrossRef] [PubMed]
- Anderson, A.R.; Guindo, A.; Cablay, K.; MacLean, B.; Kamate, J. Systematic, point-of-care newborn screening and treatment for sickle cell disease in rural Mali, West Africa: Results from an ongoing implementation project. Blood 2023, 142 (Suppl. S1), 796. [Google Scholar] [CrossRef]
- Guindo, A.; Cisse, Z.; Keita, I.; Desmonde, S.; Sarro, Y.D.S.; Touré, B.A.; Baraika, M.A.; Tessougué, O.; Guindo, P.; Coulibaly, M.; et al. Potential for a large-scale newborn screening strategy for sickle cell disease in Mali: A comparative diagnostic performance study of two rapid diagnostic tests (SickleScan® and HemotypeSC®) on cord blood. Br. J. Haematol. 2024, 204, 337–345. [Google Scholar] [CrossRef]
- Diallo, D.A.; Guindo, A.; Touré, B.A.; Sarro, Y.S.; Sima, M.; Tessougué, O.; Baraika, M.A.; Guindo, P.; Traoré, M.; Diallo, M.; et al. Dépistage néonatal ciblé de la drépanocytose: Limites du test de falciformation (test d’Emmel) dans le bilan prénatal en zone ouest africaine [Targeted newborn screening for sickle-cell anemia: Sickling test (Emmel test) boundaries in the prenatal assessment in West African area]. Rev. Epidemiol. Sante Publique 2018, 66, 181–185. (In French) [Google Scholar] [CrossRef] [PubMed]
- Fondation Pierre Fabre. Creating and Developing the Research Centre to Combat Sickle Cell Disease. 30 July 2018. Available online: https://www.fondationpierrefabre.org/en/our-programmes/combatting-sickle-cell-disease/creating-developing-research-centre-sickle-cell-disease/ (accessed on 7 August 2023).
- Mohamed, G.S.; Lemine, S.M.; Cheibetta, S.; Mohamed, A. Dépistage néonatal du déficit en glucose 6 phosphate déshydrogénase (G6PD) en Mauritanie [Neonatal screening for glucose-6-phosphate dehydrogenase (G6PD) deficiency in Mauritania]. Pan Afr. Med. J. 2018, 30, 224. (In French) [Google Scholar] [CrossRef] [PubMed]
- Djigo, O.K.M.; Ould Ahmedou Salem, M.S.; Diallo, S.M.; Bollahi, M.A.; Boushab, B.M.; Garre, A.; Papa Mze, N.; Basco, L.; Briolant, S.; Ould Mohamed Salem Boukhary, A. Molecular epidemiology of G6PD genotypes in different ethnic groups residing in Saharan and Sahelian Zones of Mauritania. Pathogens 2021, 10, 931. [Google Scholar] [CrossRef] [PubMed]
- Laghdaf Sidi, M.; N’Diaye, A.M.; Cheikh, M.; Heinhane, M.M. Sickle cell disease in Mauritania: Epidemiological, clinical and therapeutic aspects about 135 cases. Tunis. Med. 2022, 100, 313–322. [Google Scholar] [PubMed]
- Mano, R.M.; Kuona, P.; Misihairabgwi, J.M. Determination of Birth Prevalence of Sickle Cell Disease Using Point of Care Test HemoTypeSCTM at Rundu Hospital, Namibia. Research Square. Available online: https://www.researchsquare.com/article/rs-3753291/v1 (accessed on 10 January 2024).
- Nnodu, O.E.; Adegoke, S.A.; Ezenwosu, O.U.; Emodi, I.I.; Ugwu, N.I.; Ohiaeri, C.N.; Brown, B.J.; Olaniyi, J.A.; Isa, H.; Okeke, C.C.; et al. A multi-centre survey of acceptability of newborn screening for sickle cell disease in Nigeria. Cureus 2018, 10, e2354. [Google Scholar] [CrossRef] [PubMed]
- Ojewunmi, O.O.; Adeyemo, T.A.; Ayinde, O.C.; Iwalokun, B.; Adekile, A. Current perspectives of sickle cell disease in Nigeria: Changing the narratives. Expert Rev. Hematol. 2019, 12, 609–620. [Google Scholar] [CrossRef]
- Bashir, M.F.; Elechi, H.A.; Jarrett, O.O.; Oyenusi, E.E.; Oduwole, A.; Ibrahim, N.; Ohuche, I.; Adedeji, I.A.; Adamu, S.; Jingina, J.; et al. Cord blood thyroid stimulating hormone values in healthy term babies delivered at Abubakar Tafawa Balewa University Teaching Hospital Bauchi, Northeastern Nigeria. West Afr. J. Med. 2022, 39, 603–608. [Google Scholar] [PubMed]
- Ejiofor, O.S.; Efobi, C.; Emechebe, G.O.; Ifezulike, C.C.; Okeke, K.N.; Mokebe, T.C.; Obijiofor, K.; Ideh, C.C.; Onuh, U. Newborn screening for sickle cell disease (SCD) in Awka South-East Nigeria. J. Blood Disord. Transfus. 2018, 9, 398. [Google Scholar] [CrossRef]
- Oluwole, E.O.; Adeyemo, T.A.; Osanyin, G.E.; Odukoya, O.O.; Kanki, P.J.; Afolabi, B.B. Feasibility and acceptability of early infant screening for sickle cell disease in Lagos, Nigeria-A pilot study. PLoS ONE 2020, 15, e0242861. [Google Scholar] [CrossRef]
- Mohammed-Nafi’u, R.; Audu, L.I.; Ibrahim, M.; Wakama, T.T.; Okon, E.J. Pattern of haemoglobin phenotypes in newborn infants at the National Hospital Abuja using high performance liquid chromatography. Niger Postgrad. Med. J. 2020, 27, 190–195. [Google Scholar] [CrossRef]
- Ezenwosu, O.U.; Itanyi, I.U.; Nnodu, O.E.; Ogidi, A.G.; Mgbeahurike, F.; Ezeanolue, E.E. Community based screening for sickle haemoglobin among pregnant women in Benue State, Nigeria: I-Care-to-Know, A Healthy Beginning initiative. BMC Pregnancy Childbirth 2021, 21, 498. [Google Scholar] [CrossRef] [PubMed]
- Babalola, O.A.; Chen, C.S.; Brown, B.J.; Cursio, J.F.; Falusi, A.G.; Olopade, O.I. Knowledge and health beliefs assessment of Sickle cell disease as a prelude to neonatal screening in Ibadan, Nigeria. J. Glob. Health Rep. 2019, 3, e2019062. [Google Scholar] [CrossRef]
- Ijei-Enesi, I.; Dogara, L.; Inusa, P.B. Implementing a Newborn Screening Programme: Our Experience in Kaduna State, Nigeria. Annual Report 2021–2022. The Royal College of Pathologists. Available online: https://www.rcpath.org/profession/publications/annual-reports/annual-report-2021-2022/implementing-a-newborn-screening-programme-our-experience-in-kaduna-state-nigeria.html (accessed on 7 July 2023).
- Okeke, C.O.; Chianumba, R.I.; Isa, H.; Asala, S.; Nnodu, O.E. Using dried blood spot on HemoTypeSC™, a new frontier for newborn screening for sickle cell disease in Nigeria. Front. Genet. 2022, 13, 1013858. [Google Scholar] [CrossRef] [PubMed]
- Makoni, M. Newborn screening for sickle cell disease in Africa. Lancet Haematol. 2021, 8, e476. [Google Scholar] [CrossRef]
- Folayan, O.S.; Bello, A.O.; Ernest, S.K. Newborn screening for haemoglobinopathies in Bida, North-Central Nigeria. J. Hematol. Thrombo. Dis. 2023, 11, 537. [Google Scholar]
- Nnachi, O.C.; Umeokonkwo, A.A.; Okoye, H.C.; Ekwe, A.N.; Akpa, C.O.; Okoye, A.E. Acceptability of newborn screening for sickle cell disease among post-partum mothers in Abakaliki, South-East Nigeria. West Afr. J. Med. 2023, 40, 298–304. [Google Scholar] [PubMed]
- Munyanganizi, R.; Cotton, F.; Vertongen, F.; Gulbis, B. Red blood cell disorders in Rwandese neonates: Screening for sickle cell disease and glucose-6-phosphate dehydrogenase deficiency. J. Med. Screen. 2006, 13, 129–131. [Google Scholar] [CrossRef] [PubMed]
- Mbodj, M.; Ndoye, O.; Diarra, M.; Mbaye, B.N.; Sow Toure, H.; Diouf, L.; Gassama Seck, S.; Dhondt, J.L.; Farriaux, J.P. Dépistage néonatal de la drépanocytose au CHU de Dakar: Premier bilan [Sickle cell disease neonatal screening. First evaluation]. Dakar Med. 2003, 48, 202–205. (In French) [Google Scholar] [PubMed]
- Diallo, A.H.; Camara, G.; Lamy, J.B.; Lo, M.; Diagne, I.; Makalou, D.; Diop, M.; Doupa, D. Towards an Information System for Sickle Cell Neonatal Screening in Senegal. Stud. Health Technol. Inform. 2019, 258, 95–99. [Google Scholar] [PubMed]
- Diallo, A.H.; Camara, G.; Lamy, J.B.; Lo, M.; Diagne, I.; Makalou, D.; Diop, M.; Doupa, D. SIMENS-LIS4SC, a laboratory information system for biological tests of sickle cell screening and healthcare. Stud. Health Technol. Inform. 2019, 264, 611–615. [Google Scholar] [CrossRef] [PubMed]
- Diallo, A.H.; Camara, G.; Lo, M.; Diagne, I.; Lamy, J.B. Iconic visualization of sickle cell patients current and past health status. Stud. Health Technol. Inform. 2020, 272, 71–74. [Google Scholar] [CrossRef] [PubMed]
- Fondation Pierre Fabre. Operational Study on Early Detection and Treatment of Sickle Cell Disease. Available online: https://www.fondationpierrefabre.org/en/our-programmes/combatting-sickle-cell-disease/operational-study-early-detection-treatment-sickle-cell-disease/ (accessed on 7 August 2023).
- Shamlaye, C.; Shamlaye, H.; Brewer, R. Health in Seychelles—An overview. Neurotoxicology 2020, 81, 230–237. [Google Scholar] [CrossRef] [PubMed]
- Seychelles News Agency. Karapetyan, S. Babies Born in Seychelles to Undergo Screenings for More Health Conditions. 10 February 2021. Available online: http://www.seychellesnewsagency.com/articles/14322/Babies+born+in+Seychelles+to+undergo+screenings+for+more+health+conditions (accessed on 3 August 2023).
- Gibson, R.W.; Edem-Hotah, J. Communicating sickle cell disease point-of-care testing results. Lancet Haematol. 2020, 7, e708–e709. [Google Scholar] [CrossRef] [PubMed]
- allAfrica. Sierra Leone: Today Is World Sickle Cell Day—Call to Implement Screening of Newborn. Concord Times (Freetown). 22 June 2023. Available online: https://allafrica.com/stories/202306230113.html (accessed on 3 August 2023).
- Bernstein, R.E.; Op’t Hof, J.; Hitzeroth, H.W. Neonatal screening for congenital hypothyroidism. A decade’s review, including South Africa. S. Afr. Med. J. 1988, 73, 339–343. [Google Scholar] [PubMed]
- Hitzeroth, H.W.; Niehaus, C.E.; Brill, D.C. Phenylketonuria in South Africa. A report on the status quo. S. Afr. Med. J. 1995, 85, 33–36. [Google Scholar] [PubMed]
- Hitzeroth, H.W. Screening for congenital hypothyroidism in South Africa. Report on a national workshop (1992). S. Afr. Med. J. 1994, 84, 106–108. [Google Scholar]
- Lebea, P.J.; Pretorius, P.J. Newborn screening for classic galactosemia and primary congenital hypothyroidism in the Nkangala district of the Mpumalanga province of South Africa. S. Afr. J. Child Health 2008, 2, 19–22. [Google Scholar]
- Malherbe, H.L.; Christianson, A.L.; Aldous, C. Need for services for the care and prevention of congenital disorders in South Africa as the country’s epidemiological transition evolves. S. Afr. Med. J. 2015, 105, 186–188. [Google Scholar] [CrossRef] [PubMed]
- Next Biosciences. FirstScreen—Newborn Screening [NBS] of Babies 1–3 Days after Birth for Early Detection of Metabolic Disorders to Allow for Prompt Intervention and Treatment. Available online: https://nextbio.co.za/divisions/patients/genetics/firstscreen (accessed on 3 August 2023).
- NWU. Centre for Human Metabolomics: NBS (Newborn Screening). Available online: https://natural-sciences.nwu.ac.za/centre-human-metabolomics/nbs-newborn-screening (accessed on 3 August 2023).
- NWU. News@NWU: Screening at Birth Can Spare Babies and Parents Much Pain. Available online: https://news.nwu.ac.za/screening-birth-can-spare-babies-and-parents-much-pain (accessed on 3 August 2023).
- Malherbe, H.L.; Bonham, J.; Carrihill, M.; Chetty, K.; Conradie, E.H.; Dercksen, M.; Goeiman, H.; Gomes, M.C.M.; Klopper, B.; McKerrow, N.; et al. Newborn screening in South Africa: The past, present and plans for the future. Rare Dis. Orphan Drugs J. 2024, 3, 7. [Google Scholar] [CrossRef]
- Babiker, A.M.; Jurayyan, N.A.; Mohamed, S.H.; Abdullah, M.A. Overview of diagnosis, management and outcome of congenital hypothyroidism: A call for a national screening programme in Sudan. Sudan J. Paediatr. 2012, 12, 7–16. [Google Scholar]
- Daak, A.A.; Elsamani, E.; Ali, E.H.; Mohamed, F.A.; Abdel-Rahman, M.E.; Elderdery, A.Y.; Talbot, O.; Kraft, P.; Ghebremeskel, K.; Elbashir, M.I.; et al. Sickle cell disease in western Sudan: Genetic epidemiology and predictors of knowledge attitude and practices. Trop. Med. Int. Health 2016, 21, 642–653. [Google Scholar] [CrossRef] [PubMed]
- Nkya, S.; Mtei, L.; Soka, D.; Mdai, V.; Mwakale, P.B.; Mrosso, P.; Mchoropa, I.; Rwezaula, S.; Azayo, M.; Ulenga, N.; et al. Newborn screening for sickle cell disease: An innovative pilot program to improve child survival in Dar es Salaam, Tanzania. Int. Health 2019, 11, 589–595. [Google Scholar] [CrossRef] [PubMed]
- Luoga, F.; Jonathan, A.; Chirande, L.; Balandya, E. Newborn Screening for Sickle Cell Disease in Tanzania: The Past, Present and Future. Tanzania Med. J. 2020, 31, 79–82. [Google Scholar] [CrossRef]
- Ambrose, E.E.; Smart, L.R.; Charles, M.; Hernandez, A.G.; Latham, T.; Hokororo, A.; Beyanga, M.; Howard, T.A.; Kamugisha, E.; McElhinney, K.E.; et al. Surveillance for sickle cell disease, United Republic of Tanzania. Bull. World Health Organ. 2020, 98, 859–868. [Google Scholar] [CrossRef] [PubMed]
- Eastburg, L.; Peckham, A.; Kawira, E.; Chirangi, B.; Adler, D.; Akungo, B.D.; Smart, L.R.; Ambrose, E.E. Extremely high birth prevalence of sickle cell disease in rural Tanzania. Pediatr. Blood Cancer 2020, 67, e28620. [Google Scholar] [CrossRef] [PubMed]
- Bukini, D.; Nkya, S.; McCurdy, S.; Mbekenga, C.; Manji, K.; Parker, M.; Makani, J. Perspectives on building sustainable newborn screening programs for sickle cell disease: Experience from Tanzania. Int. J. Neonatal Screen. 2021, 7, 12. [Google Scholar] [CrossRef] [PubMed]
- Christopher, H.; Josephat, E.; Kaywanga, F.; Saul, S.; Mshana, I.; Kunambi, P.; Nasser, A.; Chamba, C.; Makani, J.; Nkya, S. Potential of point of care tests for newborn screening for sickle cell disease: Evaluation of HemotypeSC™ and sickle SCAN® in Tanzania. Int. J. Lab. Hematol. 2022, 44, 959–965. [Google Scholar] [CrossRef] [PubMed]
- Marco, E.; Makani, J.; Ruggajo, P.; Kidenya, B.R.; Minja, I.K.; Balandya, E. The efficacy of maternal health education and maternal screening on knowledge and the uptake of infant screening for sickle cell disease in Dar-Es-Salaam, Tanzania; a quasi-experimental study. BMC Public Health 2023, 23, 70. [Google Scholar] [CrossRef] [PubMed]
- Sazawal, S.; Ryckman, K.K.; Das, S.; Khanam, R.; Nisar, I.; Jasper, E.; Dutta, A.; Rahman, S.; Mehmood, U.; Bedell, B.; et al. Machine learning guided postnatal gestational age assessment using new-born screening metabolomic data in South Asia and sub-Saharan Africa. BMC Pregnancy Childbirth 2021, 21, 609. [Google Scholar] [CrossRef] [PubMed]
- Jehan, F.; Sazawal, S.; Baqui, A.H.; Nisar, M.I.; Dhingra, U.; Khanam, R.; Ilyas, M.; Dutta, A.; Mitra, D.K.; Mehmood, U.; et al. Multiomics characterization of preterm birth in low- and middle-income countries. JAMA Netw. Open 2020, 3, e2029655, Erratum in: JAMA Netw. Open. 2021, 4, e210399. [Google Scholar] [CrossRef]
- Ndeezi, G.; Kiyaga, C.; Hernandez, A.G.; Munube, D.; Howard, T.A.; Ssewanyana, I.; Nsungwa, J.; Kiguli, S.; Ndugwa, C.M.; Ware, R.E.; et al. Burden of sickle cell trait and disease in the Uganda Sickle Surveillance Study (US3): A cross-sectional study. Lancet Glob. Health 2016, 4, e195–e200. [Google Scholar] [CrossRef]
- Kiyaga, C.; Hernandez, A.G.; Ssewanyana, I.; Schaefer, B.A.; McElhinney, K.E.; Ndeezi, G.; Howard, T.A.; Ndugwa, C.M.; Ware, R.E.; Aceng, J.R. Sickle cell screening in Uganda: High burden, human immunodeficiency virus comorbidity, and genetic modifiers. Pediatr. Blood Cancer 2019, 66, e27807. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, A.G.; Kiyaga, C.; Howard, T.A.; Ssewanyana, I.; Ndeezi, G.; Aceng, J.R.; Ware, R.E. Operational analysis of the national sickle cell screening programme in the Republic of Uganda. Afr. J. Lab. Med. 2021, 10, 1303. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.M.; Maiteki-Sebuguzi, C.; Coffey, P.S. Cost for sickle cell disease screening using isoelectric focusing with dried blood spot samples and estimation of price thresholds for a point-of-care test in Uganda. J. Blood Med. 2019, 10, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, A.G.; Kiyaga, C.; Howard, T.A.; Ssewanyana, I.; Ndeezi, G.; Aceng, J.R.; Ware, R.E. Trends in sickle cell trait and disease screening in the Republic of Uganda, 2014–2019. Trop. Med. Int. Health 2021, 26, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Oltman, S.P.; Jasper, E.A.; Kajubi, R.; Ochieng, T.; Kakuru, A.; Adrama, H.; Okitwi, M.; Olwoch, P.; Kamya, M.; Bedell, B.; et al. Gestational age dating using newborn metabolic screening: A validation study in Busia, Uganda. J. Glob. Health 2021, 11, 04012. [Google Scholar] [CrossRef]
- Fashakin, V.; Mathur, S.; Kwagala, P.; Mupere, E.; Kiguli, S.; Kitaka, S.B.; Henry, M.B.; Van Ells, L.; Tusuubira, S.; Mulumba, L.; et al. Parental notification via text messaging for infant sickle screening programs: Exploration of feasibility and acceptability in Uganda. J. Pediatr. Hematol. Oncol. 2020, 42, e593–e600. [Google Scholar] [CrossRef] [PubMed]
- Mantina, H.M. Sickle cell disease—Getting the act right. Health Press Zambia Bull. 2019, 3, 2–4. [Google Scholar]
- TrendsNAfrica. News Desk. Zambian Government Intensifies Screening for Sickle Cell Disease. 19 June 2023. Available online: https://trendsnafrica.com/zambian-government-intensifies-screening-for-sickle-cell-disease/ (accessed on 5 August 2023).
- Murphy, M.S.Q.; Hawken, S.; Atkinson, K.M.; Milburn, J.; Pervin, J.; Gravett, C.; Stringer, J.S.A.; Rahman, A.; Lackritz, E.; Chakraborty, P.; et al. Postnatal gestational age estimation using newborn screening blood spots: A proposed validation protocol. BMJ Glob. Health 2017, 2, e000365. [Google Scholar] [CrossRef] [PubMed]

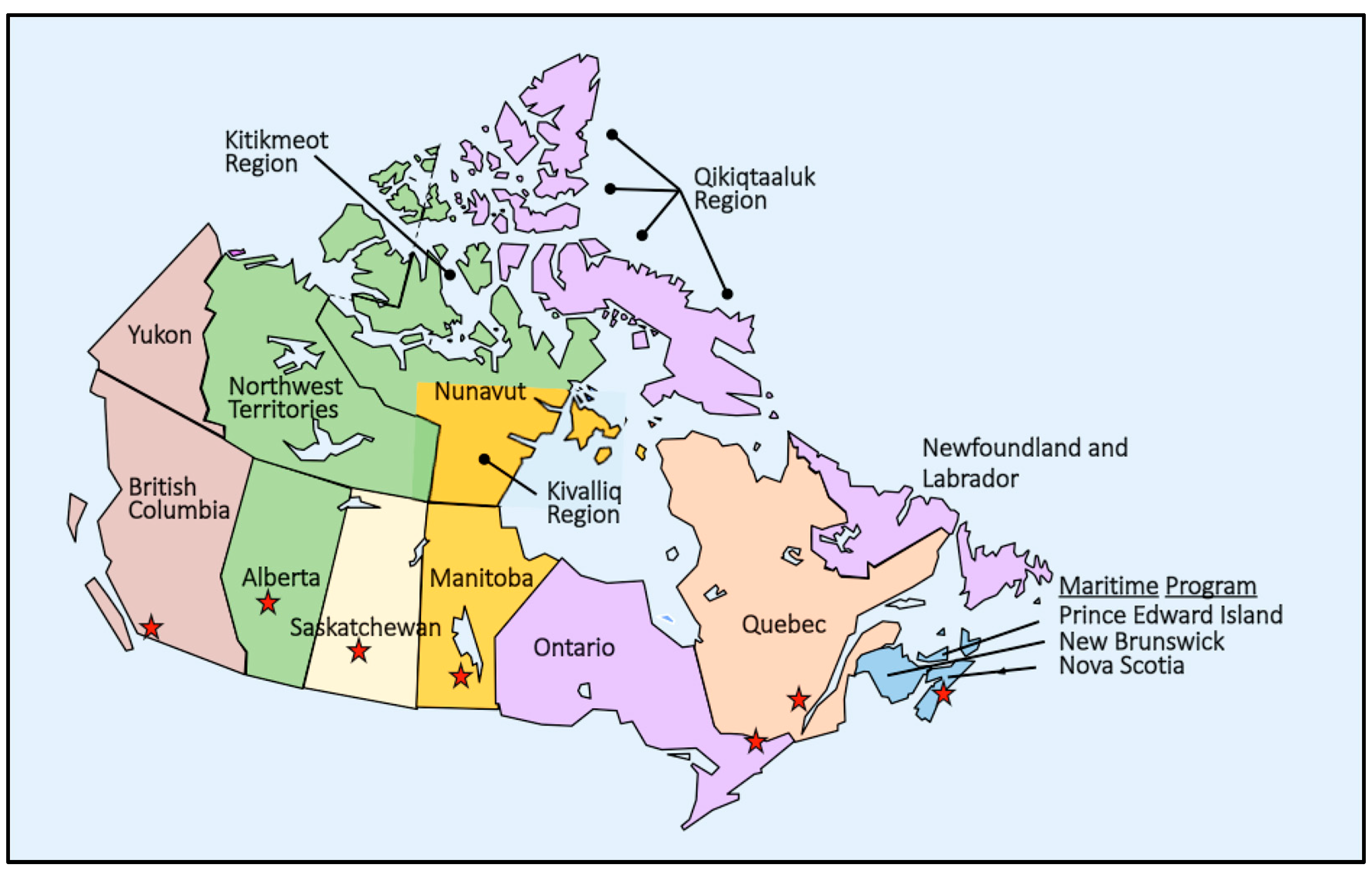
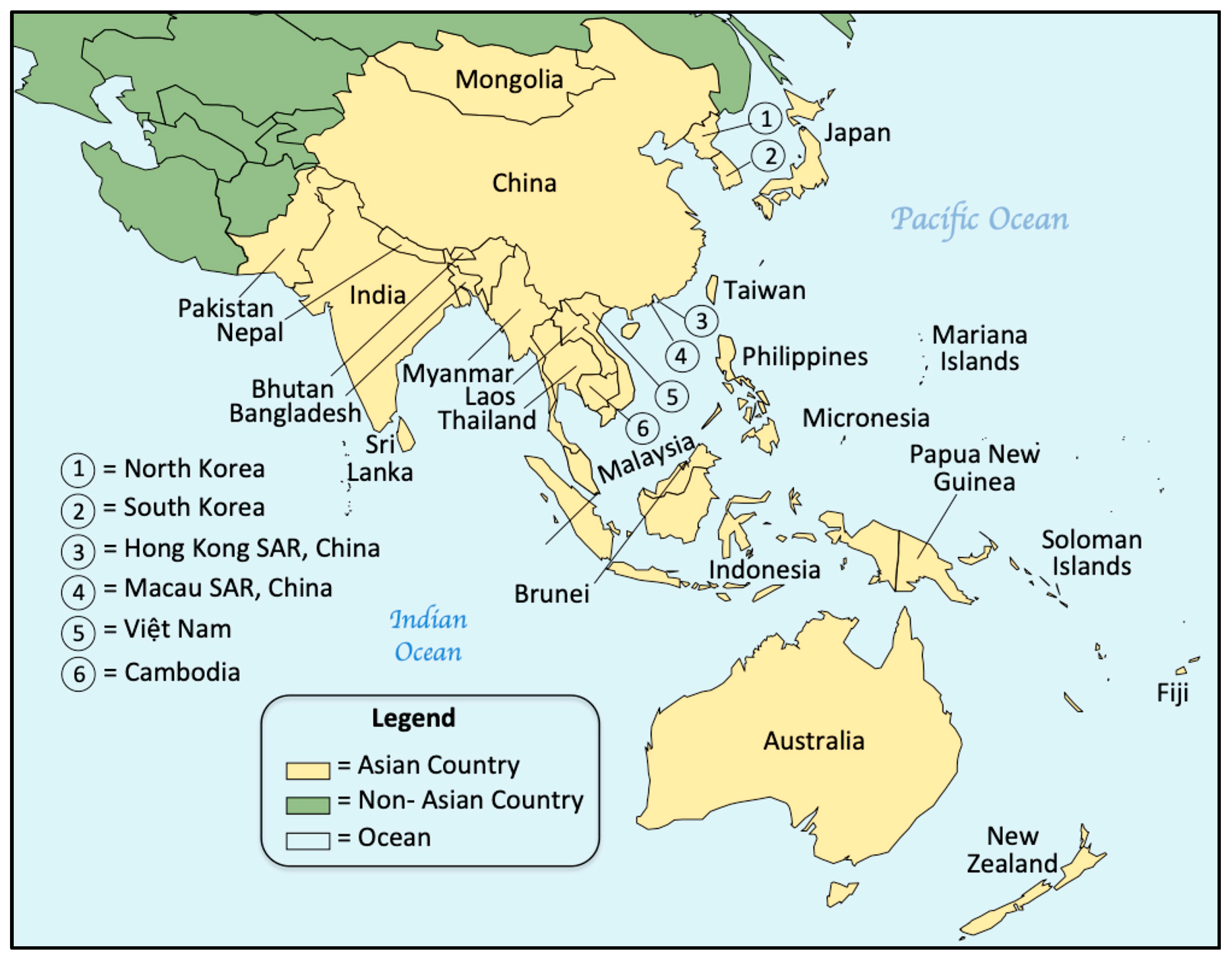
| Organic Acid Disorders (OA) | Abbreviation | Fatty Acid Oxidation Disorders (FAO) | Abbreviation | ||
|---|---|---|---|---|---|
 | Isovaleric acidemia | IVA |  | Medium-chain acyl-CoA dehydrogenase deficiency | MCAD |
 | Glutaric acidemia-Type I | GA-I |  | Very long-chain acyl-CoA dehydrogenase deficiency | VLCAD |
| 3-Hydroxy 3-methyl glutaric aciduria | HMG |  | Long-chain L-3-OH acyl-CoA dehydrogenase deficiency | LCHAD | |
| Multiple carboxylase deficiency | MCD |  | Trifunctional protein deficiency | TFP | |
 | Methylmalonic acidemia (mutase deficiency) | MUT |  | Carnitine uptake defect | CUD |
| 3-Methylcrotonyl-CoA carboxylase deficiency | 3MCC | Hemoglobinopathies (HGB) | Abbreviation | ||
 | Methylmalonic acidemia (Cobalamin A and B) | CBL A, B |  | Hemoglobin S/S disease (Sickle cell anemia) | Hb S/S |
 | Propionic acidemia | PA |  | Hemoglobin S/β-thalassemia | Hb S/β-Th |
| β-Ketothiolase deficiency | BKT |  | Hemoglobin S/C disease | Hb S/C | |
| Amino Acid Disorders (AA) | Abbreviation | Other Clinically Significant Disorders | Abbreviation | ||
 | Phenylketonuria | PKU |  | Congenital Hypothyroidism | CH |
 | Maple syrup urine disease | MSUD |  | Biotinidase Deficiency | BIO |
| Homocystinuria | HCY |  | Congenital Adrenal Hyperplasia | CAH | |
 | Citrullinemia | CIT |  | Galactosemia (classical transferase deficiency) | GALT |
 | Argininosuccinic acidemia | ASA | Early hearing detection and intervention | EHDI | |
 | Tyrosinemia type I | TYR I |  | Cystic fibrosis | CF |
 Indicates conditions included in the 2016 recommendations to the Canadian health ministry—See reference [36]. Indicates conditions included in the 2016 recommendations to the Canadian health ministry—See reference [36]. | |||||
| Organic Acid Disorders (OA) | Abbreviation | Fatty Acid Oxidation Disorders (FAO) | Abbreviation |
|---|---|---|---|
| Methylmalonic acidemia (Cobalamin C and D) | CBL C, D | Short-chain acyl-CoA dehydrogenase deficiency | SCAD |
| Malonic acidemia | MAL | Glutaric acidemia type II | GA-II |
| Isobutyryl-CoA dehydrogenase deficiency | IBDD | Medium/short-chain L3-OH acyl-CoA-dehydrogenase deficiency | M/SCHAD |
| 2-Methyl 3-hydroxy butyric aciduria | 2M3HBA | Medium-chain ketoacyl-CoA thiolase deficiency | MCKAT |
| 2-methylbutyryl-CoA dehydrogenase deficiency | 2MBG | Carnitine palmitoyl transferase II deficiency | CPT II |
| 3-Methylglutaconic aciduria | 3MGA | Carnitine acylcarnitine translocase deficiency | CACT |
| Amino Acid Disorders (AA) | Abbreviation | Carnitine palmitoyl transferase IA deficiency (liver) | CPT IA |
| Benign hyperphenylalaninemia | H-PHE | Dienoyl-CoA reductase deficiency | DE RED |
| Tyrosinemia type II | TYR II | Hemoglobinopathies (HGB) | Abbreviation |
| Defects of biopterin cofactor biosynthesis | BIOPT(BS) | Variant hemoglobinopathies (including Hb E) | Var HGB |
| Argininemia | ARG | Other Clinically Significant Disorders | Abbreviation |
| Tyrosinemia type II | TYR III | Galactokinase deficiency | GALK |
| Defects of biopterin cofactor regeneration | BIOPT(REG) | Galactose epimerase deficiency | GALE |
| Hypermethioninemia | MET | ||
| Citrullinemia type II | CIT II |
| Approved | Disapproved | ||
|---|---|---|---|
| Condition | Date Added to RUSP | Condition | Date Disapproved |
| a Infantile Krabbe disease (KD) | Sent to SHHS * | Duchenne muscular dystrophy (DMD) | February 2023 |
| b Guanidinoacetate methyltransferase deficiency (GAMT) | January 2023 | Congenital cytomegalovirus (cCMV) | August 2022 |
| Mucopolysaccharidosis II (MPS-II) | August 2022 | Cerebrotendinous xanthomatosis (CTX) | November 2018 |
| c Spinal muscular atrophy (SMA) | July 2018 | 22q11.2 deletion syndrome | January 2012 |
| c Adrenoleukodystrophy (ALD) | February 2016 | Neonatal hyperbilirubinemia/kernicterus | January 2012 |
| Mucopolysaccharidosis I (MPS-I) | February 2016 | Hemoglobin H disease (HbH disease) | May 2010 |
| Pompe disease (PD) | March 2015 | Niemann–Pick disease | October 2008 |
| Critical congenital heart disease (CCHD) | September 2011 | Fabry disease | August 2008 |
| d Severe combined immunodeficiency (SCID) | February2010 | ||
| * Secretary of the USA Department of Health and Human Services. a Approved on 3rd nomination October 2023. Disapproved on 2 previous nominations: September 2009 and February 2023. Recommendation for infantile KD, as defined by low GALC enzyme and psychosine ≥ 10 nM, forwarded to SHHS on 1 March 2024. b Approved on 3rd nomination. Not approved June 2016 and November 2016. c Approved on 2nd nomination. SMA not approved November 20008; ALD not approved September 2012. d T-cell lymphocyte deficiencies were included as a secondary condition. | |||
| Jurisdiction | Demographic Information | Disorder | |||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Core RUSP | Secondary RUSP | Miscellaneous | |||||||||||||||||||||||||||||||
Shading Indicates a Shared Laboratory: Iowa Iowa Colorado Colorado Revvity Revvity Washington Washington Massachusetts Massachusetts Oregon Oregon | a Population (× 1000) Latest Data—2023 | b Births (Thousands) 2022, Provisional | Date Universal Screening Required | c Newborn Screening Fee (US$) | d 28 Core RUSP—DBS | EHDI | SCID | CCHD | Pompe | MPS-I | ALD | SMA | MPS-II | GAMT Deficiency | Other Hemoglobins | Galactokinase | Galactoepimerase | d 8 Secondary AAs | d 8 Secondary FAOs | d 6 Secondary OAs | T-cell Defect | Krabbe | Fabry | Gaucher | Niemann Pick | FIGLU | HHH | G6PD Deficiency | HIV | TOXO | 5-OXO | cCMV | Other |
| Alabama | 5108 | 58.1 | 1965 | 150.00 e | ● | ● | ● | ● | 🞇 | 🞇 | 🞇 | ● | ● | ● | ● | ● | ● | ||||||||||||||||
| Alaska | 733 | 9.3 | 1965 | 190.50 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||||||||||
| Arizona | 7431 | 78.5 | 1979 | 171.00 e | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||||||||
| Arkansas | 3068 | 35.3 | 1967 | 131.00 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||||||||||
| California | 38,965 | 18.5 | 1965 | 210.00 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||
| Colorado | 5878 | 62.33 | 1965 | 111.00 e | ● | ● | ● | ● | 🞇 | 🞇 | 🞇 | ● | ● | ● | ● | ● | ● | ||||||||||||||||
| Connecticut | 3617 | 35.3 | 1965 | 113.00 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | 🞇 | ||||||||||||
| Delaware | 1032 | 10.7 | 1962 | 135.00 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||||
| District of Columbia | 679 | 8.0 | 1980 | 0.00 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||||
| Florida | 22,611 | 224.6 | 1965 | 15.00 f | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||||||
| Georgia | 11,029 | 125.8 | 1966 | 80.40 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | 🞇 | |||||||||||||||
| Hawaii | 1435 | 15.2 | 1965 | 99.00 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||||||||||
| Idaho | 1965 | 22.3 | 1965 | 120.70 e | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||||||
| Illinois | 12,550 | 128.3 | 1965 | 128.00 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||
| Indiana | 6862 | 79.6 | 1965 | 120.00 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||||
| Iowa | 3207 | 36.4 | 1965 | 162.00 | ● | ● | ● | ● | 🞇 | 🞇 | 🞇 | ● | ● | ● | ● | ● | ● | ||||||||||||||||
| Kansas | 2941 | 34.4 | 1965 | 0.00 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||||||||
| Kentucky | 4526 | 52.2 | 1966 | 150.00 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||||||
| Louisiana | 4474 | 56.1 | 1964 | 30.00 | ● | ● | ● | ● | ● | ● | 🞇 | ● | ● | ● | ● | ● | ● | ||||||||||||||||
| Maine | 1396 | 12.1 | 1965 | 220.00 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | 🞇 | ● | ||||||||||||
| Maryland | 6180 | 68.7 | 1965 | 106.00 e | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||||
| Massachusetts | 7001 | 68.6 | 1963 | 170.21 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | 🞇 | ● | ● | |||||||||||
| Michigan | 10,037 | 102.2 | 1965 | 166.38 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||||
| Minnesota | 5738 | 63.9 | 1965 | 235.00 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||||
| Mississippi | 2940 | 34.6 | 1985 | 110.00 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||||
| Missouri | 6196 | 69.0 | 1965 | 102.00 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||
| Montana | 1133 | 11.2 | 1965 | 134.00 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||||||||||
| Nebraska | 1978 | 24.3 | 1967 | 86.00 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||||
| Nevada | 3194 | 33.2 | 1967 | 81.00 e | ● | ● | ● | ● | 🞇 | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||||||
| New Hampshire | 1402 | 12.1 | 1965 | 146.00 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | 🞇 | ● | ● | ● | ||||||||||||
| New Jersey | 9291 | 102.9 | 1964 | 150.00 | ● | ● | ● | ● | ● | ● | 🞇 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||
| New Mexico | 2114 | 19.5 | 1966 | 240.00 e | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||||
| New York | 19,571 | 207.5 | 1964 | 0.00 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||||
| North Carolina | 10,835 | 121.4 | 1983 | 128.00 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||||
| North Dakota | 784 | 9.6 | 1967 | 109.00 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||||||||||
| Ohio | 11,786 | 128.2 | 1965 | 98.63 | ● | ● | ● | ● | ● | ● | 🞇 | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||||
| Oklahoma | 4054 | 48.3 | 1965 | 160.42 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||||
| Oregon | 4233 | 39.5 | 1963 | 175.00 e | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||||
| Pennsylvania | 12,962 | 130.0 | 1965 | 41.72 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | 🞇 | ||||||||||
| Rhode Island | 1096 | 10.2 | 1965 | 162.98 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | 🞇 | 🞇 | ||||||||||||||
| South Carolina | 5374 | 57.8 | 1965 | 127.00 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||||||
| South Dakota | 919 | 11.2 | 1973 | 98.00 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||||||||||
| Tennessee | 7126 | 82.3 | 1968 | 185.00 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||
| Texas | 30,503 | 389.5 | 1965 | 127.10 e | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||||||||
| Utah | 3418 | 45.8 | 1965 | 140.00 e | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||||||||
| Vermont | 647 | 5.3 | 1962 | 203.00 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | 🞇 | ● | ||||||||||||
| Virginia | 8716 | 95.4 | 1966 | 138.00 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | 🞇 | |||||||||||||||
| Washington | 7813 | 83.2 | 1967 | 135.10 e | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||||||
| West Virginia | 1770 | 16.9 | 1965 | 241.35 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||||||
| Wisconsin | 5911 | 59.9 | 1965 | 195.00 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||||||||
| Wyoming | 584 | 6.0 | 1983 | 97.32 e | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||||||||
| Total | 334,813 | ||||||||||||||||||||||||||||||||
| Guam | 153 | 2.6 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||||||||
| Northern Mariana Islands | 47 | 0.6 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||||||||||
| Puerto Rico | 3206 | 19.3 | 130.00 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||||||||||
| American Samoa | 50 | - | |||||||||||||||||||||||||||||||
| Virgin Islands | 87 | 0.9 | ● | ● | ● | ||||||||||||||||||||||||||||
| Abbreviations not in the text: FIGLU = Formiminoglutamic Acid; HHH = Hyperornithinemia-Hyperammonemia-Homocitrullinuria; G6PD = Glucose-6-Phosphate Dehydrogenase; HIV = Human Immunodeficiency Virus; 5-OXO = 5-Oxoproline; cCMV = Congenital Cytomegalovirus; DBS = dried blood spot ● Indicates full population mandate. 🞇 Indicates less than full population mandate—includes pilot testing or testing on a part of the population based on another test (e.g., cCMV after positive EHDI) a Available at: https://worldpopulationreview.com/states (accessed 10 January 2024) b https://www.cdc.gov/nchs/data/vsrr/vsrr028.pdf (provisional data for 2022—accessed 10 January 2024) c Available: https://www.newsteps.org/resources/data-visualizations/newborn-screening-fees-and-fee-details (accessed 10 January 2024) d See Table 1 e Indicates a program requiring 2 screens—fee indicated covers the 2 required screens (fee may be charged in 2 parts—Texas charges $63.55 twice) f An additional fee is charged to insurance by the NBS laboratory | |||||||||||||||||||||||||||||||||
| Demographics | Disorders | |||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Jurisdiction | Details | Miscellaneous | Amino Acid | Fatty Acid Oxidation | Organic Acid | |||||||||||||||||||||||||||||||
| (See Footnotes in Table regarding Collaborations.) | a Population (x1000) Latest Data—2023 | b Births 2022/2023 Latest Official Data. | c Date Screening Began | d CH | d CAH | d GAL | d BIO | d HGB | d CF | d SCID | SMA | cCMV | GAMT | Other (See below) | d PKU | d MSUD | d TYR-I | d ASA | d CIT | HCY | d CUD | d TFP | d MCAD | d LCHAD/TFP | d VLCAD | GA-II | CPT- I/CPT -II | CACT | d IVA | d MMA (MUT)/(CBL) | d PA | d GA-I | MCD | BKT | HMG | 3-MCC |
| Provinces | ||||||||||||||||||||||||||||||||||||
| eAlberta | 4756.4 | 48,523 | 1967 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||
| f British Columbia | 581.15 | 4103 | 1964 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||
| g Manitoba | 1465.4 | 17,146 | 1965 | ● | ● | ● | ● | ● | ● | ● | ● | 1 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||
| h New Brunswick | 842.7 | 6388 | 1966 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||
| New Foundland and Labrador | 540.4 | 3566 | 1978 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||
| h Nova Scotia (Maritime) | 1066.4 | 8122 | 1966 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||
| i Ontario | 15,801.8 | 137,748 | 1965 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | 2 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||
| h Prince Edward Island | 175.9 | 1373 | 1963 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||
| Quebec | 8948.5 | 79,050 | 1971 | ● | ● | ● | ● | ● | ● | 3 | ● | ● | ● | ● | † | ● | ● | ● | ● | † | † | ● | ||||||||||||||
| Saskatchewan | 1219.0 | 13,343 | 1965 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | 4 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |
| Territories | ||||||||||||||||||||||||||||||||||||
| e Northwest Territories | 44.8 | 502 | 1965 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||
| e Nunavut-Kitikmeot | 40.8 | 708 | 1965 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||
| g - Kivalliq | 1965 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||
| i - Kivalliq Qikiqtaaluk | 1965 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||
| f Yukon | 45.1 | 399 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||
| Totals | 40,528.4 | 357,903 | ||||||||||||||||||||||||||||||||||
| ● Indicates availability to full population † Screening performed using urine specimens (versus dried bloodspots). a Statistics Canada. 2023 Quarter 4 population estimates. Available: https://doi.org/10.25318/1710000901-eng (accessed 26 December 2023). b Statistics Canada. Table: 17-10-0008-01 Release date: 2023-09-27. Available: https://doi.org/10.25318/1710000801-eng (accessed 26 December 2023). c Dates previously published—see Reference [27] d Twenty-two conditions have been suggested for NBS in Canada, including mutase and cobalamin disorders (counted as one condition in the table). See Reference [37]. e University of Alberta Hospital (UAH); Edmonton, Alberta, performs screening testing for Alberta, the Northwest Territories and Kitikmeot, Nunavut. f British Columbia Children’s Hospital; Vancouver, British Columbia, performs screening testing for British Columbia and for the Yukon. g The Cadham Provincial Laboratory; Winnipeg, Manitoba, performs screening testing for Manitoba and for a limited number of newborns in Kivalliq, Nunavut. h (Maritime Program) Izaak Walton Killam (IWK) Children’s Health Centre; Halifax, Nova Scotia, performs testing for Nova Scotia, New Brunswick, and Prince Edward Island. i The Ontario laboratory at Children’s Hospital of Eastern Ontario (CHEO); Ottawa, Ontario, performs screening testing for Ontario, Qikiqtaaluk (Baffin), Kitikmeot, Nunavut. 1 Also screens for congenital cytomegalovirus (cCMV) 2 Also screens for mucopolysaccharidosis type 1 (MPS-I) 3 Also performs urine screening for hyperornithinemia-hyperammonemia-homocitrullinuria (HHH) syndrome 4 Babies automatically receive testing in the Keewatin Yatthé Health Region, all Meadow Lake Tribal Council communities, and all babies born at the Meadow Lake Hospital due to overrepresentation in northwest Saskatchewan. Performed by special request in other areas of the province. | ||||||||||||||||||||||||||||||||||||
| Demographic Information | Disorder | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Miscellaneous | Amino Acids | Fatty Acids | Organic Acids | ||||||||||||||||||||||||||||||||||
| Country | a Population (Millions) 2023 UNICEF Data | b Births (Thousands) 2023 UNICEF Data | c Infant (<1 Year) Mortality Rate (per 1000) 2022 | Date Screening Began | Screening Laboratories | Specimen Collection Time (Hrs.) - | d CH | d CAH | d GAL | d BIO | d HGB | G6PDD | d SCID | SMA | d CF | Other (see key below) | d PKU | d MSUD | d TYR-I | d ASA | d CIT | HCY | d CUD | CACT | d MCAD | d LCHAD/TFP | d VLCAD | SCAD | CPT- I/CPT -II | d IVA | d MMA (MUT)/(CBL) | d PA | d GA-I | MCD | BKT | HMG | 3-MCC |
| Australia | 26.4 | 300.2 | 3.3 | 1964 | 5 | 48–72 | ● | ● | ● | 🞇 | 🞇 | ● | 1 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||
| Bangladesh | 173.0 | 2995 | 22.9 | 1999 | 1 | 168–240 | § | ||||||||||||||||||||||||||||||
| Bhutan | 0.8 | 9.6 | 22.5 | n/a | No information | ||||||||||||||||||||||||||||||||
| Brunei Darussalam | 0.5 | 6.1 | 9.6 | n/a | No information | ||||||||||||||||||||||||||||||||
| Cambodia | 16.9 | 318.3 | 21.3 | 2013 | 1 | 48 | § | § | |||||||||||||||||||||||||||||
| China | 1425.7 | 10,758 | 5.1 | 1981 | 269 | 48 | ● | ● | § | 🞇 | ● | § | § | ● | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | |||
| Hong Kong SAR, China | 41.9 | 1.2 | 1984 | 1 # | 24–72 | ● | ● | ● | ● | ● | ● | § | 2 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||
| India | 1428.6 | 23,056 | 25.5 | 1980 | 49 † | 24–72 | § | § | § | § | § | § | § | § | § | § | § | § | § | § | § | § | § | § | § | § | § | § | § | § | § | § | § | ||||
| Indonesia | 277.5 | 4462 | 18.9 | 1999 | 12 * | 24–72 | ● | ||||||||||||||||||||||||||||||
| Japan | 123.3 | 814.7 | 1.7 | 1977 | 35 | 120–168 | ● | ● | ● | § | § | 3 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||
| Korea (North) | 340.4 | 10.1 | n/a | No Information | |||||||||||||||||||||||||||||||||
| Korea (South) | 285.7 | 2.5 | 1991 | 13 | 24–72 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||
| Lao, PDR | 7.6 | 161.7 | 34.2 | 2008 | 1 | - | † | ||||||||||||||||||||||||||||||
| Macau SAR, China | 7.0 | 4.7 | 1977 | 1 | 48 | ● | ● | § | 🞇 | ● | § | § | ● | § | § | § | § | § | § | § | § | § | § | § | § | § | § | § | § | § | § | § | § | ||||
| Malaysia | 34.3 | 509.2 | 6.5 | 1980 | 4# | 24–168 | ● | ● | § | § | § | § | § | § | § | § | § | § | § | § | § | § | § | § | § | § | § | § | § | ||||||||
| Mongolia | 3.4 | 69.5 | 12.7 | 2000 | 2 | 24–72 | § | § | § | § | § | ||||||||||||||||||||||||||
| Myanmar | 54.6 | 912.3 | 33.7 | n/a | - | § | |||||||||||||||||||||||||||||||
| Nepal | 30.9 | 616.7 | 22.8 | n/a | 1 | 24–48 | § | § | § | § | § | ||||||||||||||||||||||||||
| New Zealand | 5.2 | 64.1 | 3.9 | 1964 | 1 | 24–72 | ● | ● | ● | ● | ● | § | ● | 4 | ● | ● | ● | ● | ● | ● | × | ● | ● | ● | ● | ● | ● | ● | ● | ● | × | × | × | × | |||
| Pakistan | 240.5 | 6425 | 52.8 | 2000 | 1 | 48–240 | § | § | § | § | § | ||||||||||||||||||||||||||
| Papua New Guinea | 10.3 | 254.7 | 34.4 | n/a | No Information | ||||||||||||||||||||||||||||||||
| Philippines | 117.3 | 2499 | 20.5 | 1996 | 7 | 24–48 | ● | ● | ● | ● | ● | ● | ● | 5 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||
| Singapore | 6.0 | 42.3 | 1.7 | 1965 | 1 # | 24–72 | ● | ● | ● | ● | ● | ● | ● | 6 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||
| Sri Lanka | 21.9 | 301.4 | 5.8 | 2006 | 2 | 6–48 | ● | ||||||||||||||||||||||||||||||
| Taiwan | 186.2 | 4.1 | 1981 | 3 | 48–72 | ● | ● | ● | ● | ● | ● | ● | 7 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||
| Thailand | 71.8 | 631.2 | 7.1 | 1992 | 2 | 48–72 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||
| Việt Nam | 98.9 | 1443 | 16.4 | 2000 | 8 | 24–48 | ● | 🞇 | 🞇 | 🞇 | 🞇 | ● | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | |
| Totals | 57,511 | 15.0 | - | - | - | ||||||||||||||||||||||||||||||||
| Abbreviations: SAR = Special Administrative Region; PDR—Peoples’ Democratic Republic ● More than 50% coverage; part of national or administrative region implementation/law 🞇 Less than 50% coverage; part of national or administrative region implementation/law × Discontinued testing—see appropriate note under ‘Other’ § Only available on a limited basis—screening may part of a pilot or limited to a specific birthing facility by contract and not generally available to the full public † Pilot has been completed but program is not yet stable, and the number of screening laboratories is not yet fixed # NBS using cord blood is performed in all hospitals for CH and G6PDD * NBS samples are analyzed within the existing laboratories of the hospitals a https://data.unicef.org/resources/data_explorer/unicef_f/?ag=UNICEF&df=GLOBAL_DATAFLOW&ver=1.0&dq=.DM_POP_TOT..&startPeriod=2023&endPeriod=2023 (accessed 27 March 2024) bhttps://data.unicef.org/resources/data_explorer/unicef_f/?ag=UNICEF&df=GLOBAL_DATAFLOW&ver=1.0&dq=.DM_BRTS.&startPeriod=2023&endPeriod=2023 (accessed 27 March 2024) c https://data.unicef.org/resources/data_explorer/unicef_f/?ag=UNICEF&df=GLOBAL_DATAFLOW&ver=1.0&dq=.CME_MRY0._T.&startPeriod=2021&endPeriod=2023 (accessed 27 March 2024) d Indicates a condition chosen to be on the Canadian newborn screening panel and which is being used to harmonize the tables in this report. 1 =Multiple acyl-CoA dehydrogenase deficiency (MADD);GA-2, Holocarboxylase synthetase deficiency, Tyr II, III; For an up-to date listing, see: https://www.health.gov.au/our-work/newborn-bloodspot-screening/what-is-screened (accessed 27 March 2024) 2 = Argininemia,6-pyruvoyl-tetrahydropterin synthase deficiency, Glutaric Acidemia Type II. Citrullinemia type II. Information shown is for government program. Additional testing may be available in the private sector. 3 = GA-2, citrin deficiency, methylglutaconic aciduria—pilot 4 = TFP, MADD—Discontinued August 2015: 3MCC; HMG; MCD; BKT; 3-methylglutaconyl-CoA-hydratase deficiency; 2-methyl-3-hydroxybutyryl CoA dehydrogenase deficiency—Discontinued April 2017: CUD (See: https://www.nsu.govt.nz/pregnancy-newborn-screening/newborn-metabolic-screening-programme-heel-prick-test/about-test) (accessed 27 March 2024) 5 = MAT, GA-2, TFP (See: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8883932/)(accessed 27 March 2024) 6 = GA-2, Medium-chain ketoacyl-CoA thiolase deficiency (MCAT), Ethylmalonic encephalopathy(EE), 2-methyl-3-hydroxybutyric aciduria (HSD10),2-Methyl-3-hydroxybutyryl-CoA dehydrogenase deficiency (2M3HBD), 2-methylbutyryl-CoA dehydrogenase deficiency, 6-pyruvoyl-tetrahydropterin synthase deficiency and 3-methylglutaconic aciduria 7 = Pompe, Gaucher, citrin, MPS-1,2,3b, ALD—Not fully implemented at the time of writing: Fabry, DMD, MPS 4a, 6,aromatic 1-amino acid decarboxylase deficiency (AADC) | |||||||||||||||||||||||||||||||||||||
| Demographic Information | Disorder | |||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Miscellaneous | Amino Acid | Fatty Acid Oxidation | Organic Acid | |||||||||||||||||||||||||||||||||
| Country [Screening Covered By]  Member of European Union Member of European Union | a Population (Millions) 2023 UNICEF Data | b Births (Thousands) 2023 UNICEF Data | Date Screening Began | c Screening Laboratories | c Specimen Collection Time (Hrs.) | CH | CAH | GAL | BIO | HGB | G6PD | SCID | CF | SMA | Other (see key below) | PKU | MSUD | TYR-I | ASA | CIT | HCY | CUD | CACT | MCAD | LCHAD/TFP | VLCAD | SCAD | CPT- I/CPT -II | IVA | MMA (MUT)/(CBL) | PA | GA-I | MCD | BKT | HMG | 3-MCC |
| Albania | 2.8 | 28.5 | ||||||||||||||||||||||||||||||||||
| Andorra [France] | 0.1 | 0.6 | 48–72 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||||||||||
| Armenia | 2.8 | 31.6 | 2 | 48–96 | ● | ● | ||||||||||||||||||||||||||||||
| Austria | 9.0 | 84.1 | 1967 | 1 | 36–72 | ● | ● | ● | ● | ● | ● | 1, 4 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||
| Azerbaijan | 10.4 | 120.5 | 1 | 48–72 | ● | ● | ● | † | ● | ● | ||||||||||||||||||||||||||
| Belarus | 9.5 | 86.2 | 1978 | 1 | 72–120 | ● | ● | |||||||||||||||||||||||||||||
| Belgium | 11.7 | 116.8 | 1964 | 4 | 48–120 | ● | ● | ● | ● | 🞇 | 🞇 | ● | ● | 1 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||
| Bosnia-Herzegovina | 3.2 | 26.3 | 2000 | 3 | 48–96 | ● | ● | ● | ||||||||||||||||||||||||||||
| Bulgaria | 6.7 | 55.5 | 1978 | 2 | 72–120 | ● | ● | ● | ||||||||||||||||||||||||||||
| Croatia | 4.0 | 33.5 | 1978 | 1 | 48–72 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||||||||||||
| Cyprus | 1.3 | 12.3 | 1988 | 1 | 48–168 | ● | ● | |||||||||||||||||||||||||||||
| Czechia | 10.5 | 99.7 | 1975 | 4 | 48–72 | ● | ● | ● | ● | 2 | 4 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||||
| Denmark | 5.9 | 65.0 | 1978 | 1 | 48–72 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||
| Estonia | 1.3 | 13.1 | 1993 | 1 | 48–72 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||||
| Finland | 5.5 | 47.1 | 1980 | 1 | 36–120 | ● | ● | ● | 1 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||
| France | 64.8 | 671.4 | 1967 | 16 | 48–72 | ● | ● | ● | ● | 2 | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||||||||
| Georgia | 3.7 | 47.8 | 2007 | 1 | 48–72 | ● | ● | ● | ||||||||||||||||||||||||||||
| Germany | 83.3 | 755.1 | 1969 | 11 | 36–72 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||
| Greece | 10.3 | 76.1 | 1974 | 1 | 48–72 | ● | ● | ● | ● | ● | ||||||||||||||||||||||||||
| Hungary | 10.2 | 104.8 | 1975 | 2 | 48–72 | ● | ● | ● | 🞇 | 2 | 1 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||
| Iceland | 0.4 | 4.6 | 1972 | 1 | 48–72 | ● | ● | ● | 1, 3 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||
| Ireland | 5.1 | 56.7 | 1966 | 1 | 72–120 | ● | ● | 🞇 | ● | ● | † | ● | ● | ● | ● | ● | ||||||||||||||||||||
| Italy | 58.9 | 406.2 | 1983 | 15 | 48–72 | ● | 🞇 | ● | ● | 🞇 | 🞇 | 🞇 | ● | † | 1, 4 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● |
| Kazakhstan | 19.6 | 395.3 | 2007 | 21 | 24–72 | ● | 🞇 | ● | ||||||||||||||||||||||||||||
| Kosovo | 1.7 | 18.5 | ||||||||||||||||||||||||||||||||||
| Kyrgyz Republic | 6.7 | 152.4 | 1 | 48–72 | ● | |||||||||||||||||||||||||||||||
| Latvia | 1.8 | 16.0 | 1980 | 1 | 48–72 | ● | ● | ● | ● | ● | ● | ● | ||||||||||||||||||||||||
| Liechtenstein [Switzerland] | 0.04 | 0.4 | 72–96 | ● | ● | ● | ● | ● | ● | 2 | ● | ● | ● | ● | ||||||||||||||||||||||
| Lithuania | 2.7 | 25.6 | 1975 | 1 | 48–96 | ● | ● | ● | ● | |||||||||||||||||||||||||||
| Luxembourg | 0.7 | 6.7 | 1968 | 1 | 48–72 | ● | ● | ● | ● | ● | ||||||||||||||||||||||||||
| North Macedonia | 2.1 | 19.8 | 1976 | 1 | 32–72 | ● | 🞇 | ● | 1 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||
| Malta | 0.5 | 4.9 | 2017 | 2 | 72–120 | ● | ● | ● | ||||||||||||||||||||||||||||
| Moldova | 3.4 | 49.7 | 1989 | 1 | >48 | ● | ||||||||||||||||||||||||||||||
| Monaco [France] | 0.04 | 0.3 | 48–72 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||||||||||
| Montenegro | 0.6 | 6.8 | 2007 | 1 | 24–72 | ● | ||||||||||||||||||||||||||||||
| Netherlands | 17.6 | 182.4 | 1974 | 5 | 72–96 | ● | ● | ● | ● | ● | ● | ● | ● | 3 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||
| Norway | 5.5 | 54.5 | 1965 | 1 | 48–72 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||
| Poland | 41.0 | 423.7 | 1964 | 6 | 48–96 | ● | ● | 🞇 | ● | 🞇 | ● | ● | 1 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||
| Portugal | 10.2 | 79.5 | 1979 | 1 | 48–72 | ● | ● | ● | 1 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||
| Romania | 19.9 | 214.2 | 1999 | 5 | 48–72 | ● | 2 | ● | ● | |||||||||||||||||||||||||||
| Russia | 144.4 | 1376 | 1993 | 78 | 48–72 | ● | ● | ● | ● | ● | ● | |||||||||||||||||||||||||
| San Marino [Italy] | 0.03 | 0.2 | 1983 | 48–72 | ● | 🞇 | ● | ● | 🞇 | 🞇 | 🞇 | ● | 1, 4 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||
| Serbia | 7.1 | 65.2 | 1982 | 2 | 48–72 | ● | 🞇 | ● | ● | ● | ● | |||||||||||||||||||||||||
| Slovak Republic | 5.8 | 62.5 | 1985 | 1 | 72–96 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||
| Slovenia | 2.1 | 18.5 | 1979 | 1 | 48–72 | ● | 3 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||||
| Spain | 47.5 | 351.9 | 1968 | 15 | 24–72 | ● | 🞇 | 🞇 | 🞇 | ● | 🞇 | ● | † | 1, 4 | ● | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | ● | ● | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | ● | 🞇 | 🞇 | 🞇 | 🞇 | ||
| Sweden | 10.6 | 113.7 | 1965 | 1 | 48–72 | ● | ● | ● | ● | ● | ● | 1 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||
| Switzerland | 8.8 | 86.1 | 1965 | 1 | 72–96 | ● | ● | ● | ● | ● | ● | 2 | ● | ● | ● | ● | ||||||||||||||||||||
| Tajikistan | 10.1 | 256.6 | ||||||||||||||||||||||||||||||||||
| Turkey (Türkiye) | 85.8 | 1214 | 1983 | 2 | 48–72 | ● | ● | ● | ● | ● | ● | |||||||||||||||||||||||||
| Turkmenistan | 6.5 | 130.7 | 1 | 48–72 | ● | ● | ● | ● | ● | |||||||||||||||||||||||||||
| Ukraine | 36.7 | 184.5 | 2001 | 7 | 48–72 | ● | ● | 🞇 | 🞇 | ● | ● | 1 | ● | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | |||||||
| United Kingdom | 67.7 | 677.7 | 1969 | 16 | 120 | ● | ● | ● | † | ● | ● | ● | ● | ● | ● | |||||||||||||||||||||
| Uzbekistan | 35.2 | 755.1 | 14 | 72–96 | ● | 🞇 | 🞇 | ● | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | |||||||||||||
| Total | 923.8 | 9886.9 | ||||||||||||||||||||||||||||||||||
| ● More than 50% coverage; part of national plan/law 🞇 Less than 50% coverage; part of national plan/law † Only available on limited basis—pilot, contracted service of specific birthing facility, etc. not generally available to all public a https://data.unicef.org/resources/data_explorer/unicef_f/?ag=UNICEF&df=GLOBAL_DATAFLOW&ver=1.0&dq=.DM_POP_TOT..&startPeriod=2023&endPeriod=2023 (accessed 27 March 2024) b https://data.unicef.org/resources/data_explorer/unicef_f/?ag=UNICEF&df=GLOBAL_DATAFLOW&ver=1.0&dq=.DM_BRTS.&startPeriod=2023&endPeriod=2023 (accessed 27 March 2024) c See Reference [666]. Available online: https://www.mdpi.com/2409-515X/7/1/15 (accessed 27 May 2023) 1 = GA-2 (pilot in Spain and Ukraine) 2 = SMA pilot completed; awaiting decision on implementation: see details at https://old.sma-screening-alliance.org/map/ (accessed 21 January 2024) 3 = ALD (pilot in Slovenia and Iceland) 4 = RMD (pilot in Spain) | ||||||||||||||||||||||||||||||||||||
| Demographic Information | Disorder | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Miscellaneous | Amino Acid | Fatty Acid Oxidation | Organic Acid | ||||||||||||||||||||||||||||||||||
| Country | a Population (Millions) 2023 UNICEF Data 0 | b Births (Thousands) 2023 UNICEF Data | c Infant (<1 Year) Mortality Rate (per 1000) 2022 | Date Screening Began (P) = Public pilot (N) = National (L) = Limited (X) = Private | Screening Laboratories | Specimen Collection Time (Hrs.) | CH | CAH | GAL | BIO | HGB | G6PD | SCID | CF | Other (see key below) | PKU | MSUD | TYR-I | ASA | CIT | HCY | CUD | CACT | MCAD | LCHAD/TFP | VLCAD | SCAD | CPT- I/CPT -II | IVA | MMA (MUT)/(CBL) | PA | GA-I | MCD | BKT | HMG | 3-MCC | |
| Latin America | |||||||||||||||||||||||||||||||||||||
| Argentina | 45.8 | 624.9 | 8.4 | 1983 (P) 1995 (R) | 2006 (N) | 23 | Var. | ● | ● | ● | ● | ● | ● | ● | 🞇 | ||||||||||||||||||||||
| Bolivia | 12.4 | 263.9 | 19.6 | 1993 (P) | 2008 (R) | 9+ | 🞇 | † | † | † | |||||||||||||||||||||||||||
| Brazil | 216.4 | 2700.0 | 12.5 | 1976 (P) | 2001(N) | 30 | 48–72 | ● | ● | † | ● | ● | † | ● | 1 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | |
| Chile | 19.6 | 226.3 | 5.4 | 1989 (P) | 1992 (N) | 2 | 40–48 | ● | ● | ||||||||||||||||||||||||||||
| Colombia | 52.1 | 714.4 | 10.6 | 1986 (P) | 2000 (N) | § | >24 | ● | † | † | † | † | † | † | |||||||||||||||||||||||
| Costa Rica | 5.2 | 60.1 | 6.9 | 1982 (L) 1988 (P) | 1990 (N) | 1 | ● | ● | ● | ● | ● | 2 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||
| Cuba | 11.2 | 98.9 | 6.2 | 1986 (N) | 175+ | ● | ● | ● | ● | ● | ● | ||||||||||||||||||||||||||
| Dominican Republic | 11.3 | 200.6 | 26.8 | 1989 (L) | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | ||||||
| Ecuador | 18.2 | 298.5 | 10.5 | 1987 (X) | 2011 (N) | 1? | ● | ● | ● | ● | |||||||||||||||||||||||||||
| El Salvador | 6.4 | 99.7 | 10.2 | 2007 (P) | 2008 (R) | >24 | ● | † | † | † | † | ||||||||||||||||||||||||||
| Guatemala | 18.1 | 373.4 | 18.8 | 1995 (L) | 2003 (L) | 2 | 🞇 | † | † | † | † | ||||||||||||||||||||||||||
| Haiti | 11.7 | 267.6 | 44.0 | 2010 (P) | † | ||||||||||||||||||||||||||||||||
| Honduras | 10.6 | 217.2 | 13.8 | 2017 (R) | 1 | 🞇 | 🞇 | 🞇 | † | 🞇 | 🞇 | ||||||||||||||||||||||||||
| Mexico | 128.5 | 1857.0 | 11.0 | 1973 (P) | 1988 (L) 1995 (R) | § | 72–120 | ● | ● | ● | ● | ● | ● | 3 | ● | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | ||
| Nicaragua | 7.0 | 138.1 | 13.1 | 2005 (R) | 2014 (N) | ||||||||||||||||||||||||||||||||
| Panama | 4.5 | 76.6 | 11.4 | 2007 (P) | 2023 (N) | 2 | ● | ● | ● | ● | ● | ● | ● | ||||||||||||||||||||||||
| Paraguay | 6.9 | 137.4 | 15.0 | 1999 (L) | 2003 (N) | 1 | 24–48 | ● | ● | ● | |||||||||||||||||||||||||||
| Peru | 34.4 | 588.9 | 11.7 | 2002 (X) | 2012 (N) | 3+ | 48–72 | 🞇 | 🞇 | 🞇 | 🞇 | ||||||||||||||||||||||||||
| Uruguay | 3.4 | 35.6 | 5.6 | 1990 (L) | 1994 (N) | 1 | 40 | ● | ● | ● | ● | 4 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||
| Venezuela | 28.8 | 454.9 | 21.2 | 1985 (P) 1999 (R) | 2013 (N) | 2+ | ● | ● | ● | ● | ● | ||||||||||||||||||||||||||
| Total | 652.5 | 9434.0 | Var. | ||||||||||||||||||||||||||||||||||
| Caribbean | |||||||||||||||||||||||||||||||||||||
| Anguilla | 0.02 | 0.15 | 5.4 | ||||||||||||||||||||||||||||||||||
| Antigua and Barbuda | 0.1 | 1.12 | 8.1 | 2020 (P) | † | ||||||||||||||||||||||||||||||||
| Aruba | 0.1 | 0.77 | n/a | 2006(L) | † | ||||||||||||||||||||||||||||||||
| Bahamas | 0.4 | 4.67 | 11.2 | † | † | † | † | † | † | 5 | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | ||||||
| Barbados | 0.3 | 3.01 | 10.1 | 1999 (L) | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | ||||||
| Belize | 0.4 | 7.24 | 9.4 | ||||||||||||||||||||||||||||||||||
| British Virgin Islands | 0.03 | 0.24 | 11.6 | ||||||||||||||||||||||||||||||||||
| Cayman Islands | 0.07 | 0.65 | n/a | 1997 (L) | 2002 (N) | † | † | † | † | † | † | 5 | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | ||||
| Curaçao | 0.2 | 2.20 | n/a | 2006 (L) | 🞇 | 🞇 | † | ||||||||||||||||||||||||||||||
| Dominica | 0.1 | 0.96 | 30.8 | ||||||||||||||||||||||||||||||||||
| French Guiana | 0.3 | 7.66 | n/a | ● | ● | ● | ● | ● | ● | 6 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||||||||
| Grenada | 0.1 | 1.92 | 14.4 | 2014 (P) | † | ||||||||||||||||||||||||||||||||
| Guadeloupe | 0.4 | 4.59 | n/a | 1984 (P) | 1992 (N) | ● | |||||||||||||||||||||||||||||||
| Guyana | 0.8 | 15.94 | 22.3 | 2016 (L) | † | ||||||||||||||||||||||||||||||||
| Jamaica | 2.8 | 32.25 | 16.1 | 1995 (N) | ● | ||||||||||||||||||||||||||||||||
| Martinique | 0.4 | 3.53 | n/a | 1989 (N) | ● | ||||||||||||||||||||||||||||||||
| Montserrat | 0.01 | 0.04 | 5.7 | ||||||||||||||||||||||||||||||||||
| Sint Maarten | 0.04 | 0.40 | n/a | 2006 (L) | † | ||||||||||||||||||||||||||||||||
| St. Kitts and Nevis | 0.05 | 0.56 | 13.3 | ||||||||||||||||||||||||||||||||||
| St. Lucia | 0.2 | 2.01 | 16.0 | 1986 (L) | 1992 (N) | 🞇 | |||||||||||||||||||||||||||||||
| St. Vincent and the Grenadines | 0.05 | 1.31 | 10.1 | 2015 (L) | † | ||||||||||||||||||||||||||||||||
| Suriname | 0.6 | 11.09 | 14.8 | ||||||||||||||||||||||||||||||||||
| Trinidad & Tobago | 1.5 | 17.01 | 13.8 | 2008 (N) | 🞇 | ||||||||||||||||||||||||||||||||
| Turks and Caicos | 0.05 | 0.55 | 4.1 | † | † | † | † | † | |||||||||||||||||||||||||||||
| Total | 9.02 | 119.87 | |||||||||||||||||||||||||||||||||||
| ● More than 50% coverage; part of national implementation/law 🞇 Less than 50% coverage; part of national implementation/law † Only available on limited basis—pilot, contracted service of specific birthing facility, etc.; not generally available to all public § Complex laboratory services. Colombia decentralized through private clinical providers; Mexico has a variety of NBS providers a https://data.unicef.org/resources/data_explorer/unicef_f/?ag=UNICEF&df=GLOBAL_DATAFLOW&ver=1.0&dq=.DM_POP_TOT..&startPeriod=2023&endPeriod=2023 (accessed 27 March 2024) b https://data.unicef.org/resources/data_explorer/unicef_f/?ag=UNICEF&df=GLOBAL_DATAFLOW&ver=1.0&dq=.DM_BRTS.&startPeriod=2023&endPeriod=2023 (accessed 27 March 2024) c https://data.unicef.org/resources/data_explorer/unicef_f/?ag=UNICEF&df=GLOBAL_DATAFLOW&ver=1.0&dq=.CME_MRY0._T.&startPeriod=2021&endPeriod=2023 (accessed 27 March 2024) Var. = Variable recommendations: National program (48 h to 120 h); Buenos Aires Province (24 h after first milk 0 120 h); Buenos Aires Autonomous (48 h to 72 h) 1 = Limited screening for SMA, some LSDs, Prader-Willi, Angelman syndrome 2 = Also includes GA-2 3 = Complex system—policies set by National Center for Gender Equity and Reproductive Health of the Ministry of Health (Centro Nacional de Equidad de Género y Salud Reproductiva). 4 = Also includes HHH, argininemia 5 = expanded newborn screening available from overseas private laboratory 6 = specimens sent to France and get French screening panel, including hemoglobinopathies | |||||||||||||||||||||||||||||||||||||
| Demographic Information | Disorder | |||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Miscellaneous | Amino Acid | Fatty Acid Oxidation | Organic Acid | |||||||||||||||||||||||||||||||||||
| Country | a Population (Millions) 2023 UNICEF Data | b Births (Thousands) 2023 UNICEF Data | c Est. Birth Rate (Births/1000 Pop.) 2024 | d Infant (<1 Year) Mortality Rate (per 1000) 2022 | e Date Screening Began | Screening Laboratories | Specimen Collection Time (Hrs.)—2021 | CH | HGB | GAL | BIO | CAH | G6PD | SCID | CF | Other (see key below) | PKU | MSUD | TYR-I | ASA | CIT | HCY | CUD | CACT | MCAD | LCHAD/TFP | VLCAD | SCAD | CPT- I/CPT -II | IVA | MMA (MUT)/(CBL) | PA | GA-I | MCD | BKT | HMG | 3-MCC | |
| Algeria | 45.6 | 895.4 | 20.2 | 18.7 | -- | † | ||||||||||||||||||||||||||||||||
| Bahrain | 1.5 | 16.6 | 12.2 | 5.6 | 2007 | 1 | §† | † | ● | ● | † | 1 | † | † | † | † | ||||||||||||||||||||||
| Egypt, Arab Republic | 112.7 | 2,449.0 | 19.5 | 15.5 | 2000 | 1+1 | 24–48 | ● | § | 🞇 | 🞇 | 🞇 | 🞇 | † | 2 | ● | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | ||||||||
| Iran, Islamic Republic | 89.2 | 1,145.0 | 14.3 | 10.3 | 2005 | 10 | 72–120 | ● | § | 🞇 | 🞇 | 3 | ● | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | ||||||||||
| Iraq | 45.5 | 1,216.0 | 23.7 | 20.0 | 2013 | 2+1 | 72–120 | 🞇 | §† | 🞇 | † | † | † | † | † | 4 | 🞇 | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | † | |
| Israel | 9.2 | 174.6 | 19.1 | 2.7 | 1964 | >36 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||||||||||||
| Jordan | 11.3 | 243.2 | 22.2 | 12.2 | 2004 | 1 | 72–96 | ● | § | † | † | ● | ● | |||||||||||||||||||||||||
| Kuwait | 4.3 | 39.9 | 17.5 | 7.4 | 2005 | 1 | 24–48 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||||||
| Lebanon | 5.4 | 76.1 | 12.6 | 14.9 | 1996 | 3 | 24–48 | ● | § | 🞇 | ● | 🞇 | ● | ● | 🞇 | ● | ● | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | 🞇 | |||||||||||
| Libya | 6.9 | 116.4 | 20.3 | 8.9 | -- | |||||||||||||||||||||||||||||||||
| Morocco | 37.8 | 637.5 | 16.8 | 14.8 | 2010 | 6 | 24–48 | 🞇 | ||||||||||||||||||||||||||||||
| Oman | 4.6 | 79.5 | 21.1 | 9.0 | 2005 | 1 | 24–48 | ● | ||||||||||||||||||||||||||||||
| Palestine | Gaza | 5.4 | 146.6 | 26.8 | 12.3 | 1994 | ● | § | ● | |||||||||||||||||||||||||||||
| West Bank | 27.8 | 1994 | ● | § | ● | |||||||||||||||||||||||||||||||||
| Qatar | 2.7 | 23.9 | 9.2 | 4.5 | 1996 | 1 | 24–48 | ● | ● | ● | ● | ● | ● | 5 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | |||||
| Saudi Arabia | 36.9 | 606.7 | 13.6 | 5.5 | 1980 | 1+7 | 24–48 | ● | §● | ● | ● | ● | 6 | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||||||||||||
| Syrian Arab Republic | 23.2 | 490.8 | 21.7 | 17.8 | -- | 1 | 🞇 | 🞇 | ||||||||||||||||||||||||||||||
| Tunisia | 12.5 | 188.3 | 13.5 | 9.9 | -- | † | ||||||||||||||||||||||||||||||||
| United Arab Emirates | 9.5 | 91.8 | 10.7 | 4.3 | 1995 | 5 | 24–48 | ● | §● | ● | ● | ● | ● | § | † | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ● | ||
| Yemen | 34.4 | 1012.0 | 23.4 | 33.1 | -- | |||||||||||||||||||||||||||||||||
| Totals | 499.4 | 9665.2 | ||||||||||||||||||||||||||||||||||||
| Abbreviations: n/a = not available ● More than 50% coverage; part of national implementation/law 🞇 Less than 50% coverage; part of national implementation/law † Only available on limited basis—pilot, contracted service of specific birthing facility, etc.; not generally available to all public § an extensive national prenatal screening program exists a https://data.unicef.org/resources/data_explorer/unicef_f/?ag=UNICEF&df=GLOBAL_DATAFLOW&ver=1.0&dq=.DM_POP_TOT..&startPeriod=2023&endPeriod=2023 (accessed 27 March 2024) b https://data.unicef.org/resources/data_explorer/unicef_f/?ag=UNICEF&df=GLOBAL_DATAFLOW&ver=1.0&dq=.DM_BRTS.&startPeriod=2023&endPeriod=2023 (accessed 27 March 2024) c https://www.cia.gov/the-world-factbook/field/birth-rate/country-comparison/ (accessed 27 March 2024) d https://data.unicef.org/resources/data_explorer/unicef_f/?ag=UNICEF&df=GLOBAL_DATAFLOW&ver=1.0&dq=.CME_MRY0._T.&startPeriod=2021&endPeriod=2023 (accessed 27 March 2024) e In some cases there may be a single “main” screening laboratory(ies) with smaller laboratories operating in some regions of the country—first number indicates number of “main” laboratory(ies) 1 = Tests marked with † are available from Saudi Arabia laboratories 2 = SMA 3 = GA II, NKH, UCD 4 = SMA, Pompe, Gaucher, Krabbe, MPS I, Fabry and Neiman Pick A&B 5 = IBG, CTD, MADD 6 = ASL | ||||||||||||||||||||||||||||||||||||||
| Demographic Information | Disorders Screened | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Country | a Population (Millions) 2023 UNICEF Data | b Births (Thousands) 2023 UNICEF Data | c Est. Birth Rate 2024 (Births/1000 Pop.) | c Global Rank Birth Rate 2024 | d Global Rank Poorest Countries 2024 | e Child (<5 Year) Mortality Rate (per 1000) 2022 | f Infant (<1 Year) Mortality Rate (per 1000) 2022 | g Sickle Cell Disease Births—2000 | g Sickle Cell Disease Births—2021 | HGB | CH | CAH | G6PD | CF | Other (See Footnotes) |
| Angola | 36.7 | 1382.0 | 41.1 | 2 | 52 | 66.8 | 45.7 | 8930 | 13,700 | † | |||||
| Benin | 13.7 | 487.5 | 40.3 | 3 | 36 | 80.8 | 53.6 | 10,600 | 13,400 | † | |||||
| Botswana | 2.7 | 60.2 | 19.6 | 72 | 104 | 38.7 | 31.2 | 20 | 21 | No Information | |||||
| Burkina Faso | 23.3 | 801.0 | 31.9 | 22 | 16 | 78.8 | 50.1 | 18,100 | 22,000 | † | |||||
| Burundi | 13.2 | 443.1 | 34.6 | 14 | 2 | 50.5 | 36.4 | 188 | 316 | † | |||||
| Cabo Verde | 0.6 | 9.8 | 17.9 | 79 | 64 | 12.3 | 10.6 | 15 | 12 | No Information | |||||
| Cameroon | 28.6 | 967.7 | 34.7 | 13 | 40 | 69.8 | 47.0 | 5050 | 7310 | † | |||||
| Central African Republic | 5.7 | 243.5 | 31.9 | 21 | 3 | 96.8 | 73.5 | 1420 | 1790 | † | |||||
| Chad | 18.3 | 779.3 | 39.2 | 7 | 13 | 102.8 | 64.1 | 940 | 1790 | No Information | |||||
| Comoros | 0.9 | 24.2 | 21.6 | 58 | 28 | 48.2 | 38.2 | 1 | 1 | Interest but No Program | |||||
| Congo, Democratic Republic | 102.3 | 4237.0 | 39.2 | 6 | 4 | 75.6 | 60.1 | 36,900 | 47,400 | † | |||||
| Congo, Republic | 6.1 | 182.2 | 28.7 | 30 | 39 | 41.6 | 31.2 | 782 | 851 | † | |||||
| Cote d’Ivoire | 28.9 | 960.0 | 27.5 | 35 | 49 | 69.4 | 52.4 | 5080 | 6050 | † | |||||
| Djibouti | 1.1 | 24.6 | 21.8 | 56 | 57 | 51.9 | 44.1 | 6 | 8 | No Information | |||||
| Equatorial Guinea | 1.7 | 50.3 | 29.0 | 28 | 95 | 73.4 | 55.1 | 663 | 801 | † | † | ||||
| Eritrea | 3.7 | 106.3 | 26.3 | 41 | n/a | 36.6 | 28.0 | 42 | 49 | No Information | |||||
| Eswatini (Swaziland) | 1.2 | 28.4 | 22.3 | 51 | 76 | 50.0 | 39.7 | 1 | 1 | 🞇 | |||||
| Ethiopia | 126.5 | 3965.0 | 29.6 | 27 | 33 | 46.2 | 33.9 | 147 | 184 | Interest but No Program | |||||
| Gabon | 2.4 | 63.8 | 25.7 | 43 | 99 | 38.8 | 28.8 | 439 | 412 | 🞇 | |||||
| Gambia | 2.8 | 89.5 | 27.3 | 36 | 20 | 45.6 | 32.8 | 414 | 443 | No Information | |||||
| Ghana | 34.1 | 909.9 | 27.6 | 33 | 53 | 42.3 | 31.6 | 11,900 | 13,700 | 🞇 | |||||
| Guinea | 14.2 | 473.4 | 35.3 | 12 | 25 | 96.0 | 62.2 | 6560 | 9200 | † | |||||
| Guinea-Bissau | 2.2 | 64.8 | 36.0 | 11 | 23 | 71.9 | 48.6 | 37 | 41 | † | † | ||||
| Kenya | 55.1 | 1503.0 | 25.6 | 44 | 51 | 41.1 | 30.5 | 15,600 | 18,100 | 🞇 | † | † | † | 1 | |
| Lesotho | 2.3 | 59.4 | 22.9 | 50 | 22 | 72.2 | 56.4 | 2 | 1 | No Information | |||||
| Liberia | 5.4 | 167.0 | 32.4 | 20 | 8 | 73.2 | 54.9 | 669 | 682 | 🞇 | |||||
| Madagascar | 30.3 | 918.2 | 27.6 | 34 | 9 | 65.8 | 45.1 | 2530 | 3240 | † | |||||
| Malawi | 20.9 | 679.0 | 26.6 | 40 | 7 | 40.1 | 30.1 | 404 | 422 | † | † | ||||
| Mali | 23.3 | 952.5 | 40.0 | 4 | 15 | 93.8 | 60.1 | 3620 | 6720 | † | |||||
| Mauritania | 4.9 | 158.7 | 27.2 | 37 | 56 | 39.2 | 31.4 | 208 | 221 | † | † | ||||
| Mauritius | 1.3 | 13.2 | 9.8 | 191 | 126 | 15.0 | 13.4 | 0 | 0 | † | † | † | † | 2 | |
| Mozambique | 33.9 | 1208.0 | 36.5 | 9 | 5 | 66.2 | 48.7 | 155 | 220 | No Information | |||||
| Namibia | 2.6 | 69.2 | 24.3 | 47 | 72 | 37.9 | 28.6 | 2 | 2 | † | |||||
| Niger | 27.2 | 1217.0 | 46.6 | 1 | 6 | 117.3 | 60.3 | 9640 | 14,500 | No Information | |||||
| Nigeria | 223.8 | 8109.0 | 33.8 | 17 | 46 | 107.2 | 68.5 | 145,000 | 179,000 | 🞇 | † | ||||
| Rwanda | 14.1 | 409.8 | 25.0 | 46 | 26 | 38.0 | 28.8 | 76 | 84 | † | |||||
| Sao Tome and Principe | 0.2 | 6.4 | 26.7 | 39 | 34 | 14.5 | 11.3 | 54 | 46 | No Information | |||||
| Senegal | 17.8 | 562.5 | 30.2 | 25 | 37 | 37.0 | 28.2 | 2160 | 2620 | 🞇 | |||||
| Seychelles | 0.1 | 1.6 | 11.8 | 152 | 137 | 14.5 | 12.5 | 0 | 0 | 🞇 | 🞇 | 🞇 | 🞇 | 3 | |
| Sierra Leone | 8.8 | 266.8 | 30.8 | 24 | 12 | 100.8 | 76.0 | 5040 | 7250 | Pilot Data but No Program | |||||
| Somalia | 18.1 | 774.6 | 37.4 | 8 | 11 | 106.1 | 68.0 | 27 | 48 | No Information | |||||
| South Africa | 60.4 | 1141.0 | 17.7 | 72 | 85 | 34.5 | 27.7 | 19 | 18 | † | 4 | ||||
| South Sudan | 11.1 | 321.4 | 36.4 | 10 | 1 | 98.8 | 63.8 | 97 | 105 | No Information | |||||
| Sudan | 48.1 | 1567.0 | 33.1 | 18 | 27 | 51.6 | 37.0 | 9070 | 5870 | Studies but No Program | |||||
| Tanzania, United Republic of | 67.4 | 2385.0 | 32.5 | 19 | 31 | 40.5 | 30.0 | 9600 | 13,100 | 🞇 | |||||
| Togo | 9.1 | 282.0 | 30.9 | 23 | 17 | 60.4 | 42.1 | 4200 | 4870 | † | |||||
| Uganda | 48.6 | 1729.0 | 39.6 | 5 | 24 | 40.5 | 30.2 | 7250 | 9410 | 🞇 | |||||
| Zambia | 20.6 | 694.8 | 34.1 | 16 | 35 | 55.6 | 39.0 | 3420 | 4700 | 🞇 | |||||
| Zimbabwe | 16.7 | 493.8 | 28.8 | 29 | 19 | 47.7 | 34.6 | 71 | 68 | Interest but No Program | |||||
| SSA Total | ~1212 | 42,043.4 | 327,149 | 410,777 | |||||||||||
| Global Total | ~8000 | 134,280.0 | 453,000 | 515,000 | |||||||||||
| 🞇 Less than 50% coverage; part of national implementation/law † Only available on limited basis—pilot, contracted service of specific birthing facility, etc.; not generally available to all public a https://data.unicef.org/resources/data_explorer/unicef_f/?ag=UNICEF&df=GLOBAL_DATAFLOW&ver=1.0&dq=.DM_POP_TOT..&startPeriod=2023&endPeriod=2023 (accessed 27 March 2024) b https://data.unicef.org/resources/data_explorer/unicef_f/?ag=UNICEF&df=GLOBAL_DATAFLOW&ver=1.0&dq=.DM_BRTS.&startPeriod=2023&endPeriod=2023 (accessed 27 March 2024) c https://www.cia.gov/the-world-factbook/field/birth-rate/country-comparison/ (accessed 27 March 2024) d https://www.gfmag.com/global-data/economic-data/the-poorest-countries-in-the-world According to gross domestic product per capita – purchasing power parity) (accessed 27 March 2024) e https://data.unicef.org/resources/data_explorer/unicef_f/?ag=UNICEF&df=GLOBAL_DATAFLOW&ver=1.0&dq=.CME_MRY0T4._T&startPeriod=2021&endPeriod=2023 (accessed 27 March 2024) f https://data.unicef.org/resources/data_explorer/unicef_f/?ag=UNICEF&df=GLOBAL_DATAFLOW&ver=1.0&dq=.CME_MRY0._T.&startPeriod=2021&endPeriod=2023 (accessed 27 March 2024) g See reference [1444]. Supplemental Table S9, pp. 69–95. https://www.thelancet.com/journals/lanhae/article/PIIS2352-3026(23)00118-7/fulltext#sec1 (accessed 3 February 2024) 1 = Commercial laboratory services available (FirstScreen). Other advertised conditions: Multiple Acyl-CoA Dehydrogenase Deficiency (MADD or Glutaric Acidemia II) 2 = PKU 3 = Pilot also includes PKU and GAL, 6 conditions total 4 = Various Inborn Errors of Metabolism (IEM) conditions available through MS/MS testing at North-West University Potchestroom IEM Laboratory | |||||||||||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Therrell, B.L.; Padilla, C.D.; Borrajo, G.J.C.; Khneisser, I.; Schielen, P.C.J.I.; Knight-Madden, J.; Malherbe, H.L.; Kase, M. Current Status of Newborn Bloodspot Screening Worldwide 2024: A Comprehensive Review of Recent Activities (2020–2023). Int. J. Neonatal Screen. 2024, 10, 38. https://doi.org/10.3390/ijns10020038
Therrell BL, Padilla CD, Borrajo GJC, Khneisser I, Schielen PCJI, Knight-Madden J, Malherbe HL, Kase M. Current Status of Newborn Bloodspot Screening Worldwide 2024: A Comprehensive Review of Recent Activities (2020–2023). International Journal of Neonatal Screening. 2024; 10(2):38. https://doi.org/10.3390/ijns10020038
Chicago/Turabian StyleTherrell, Bradford L., Carmencita D. Padilla, Gustavo J. C. Borrajo, Issam Khneisser, Peter C. J. I. Schielen, Jennifer Knight-Madden, Helen L. Malherbe, and Marika Kase. 2024. "Current Status of Newborn Bloodspot Screening Worldwide 2024: A Comprehensive Review of Recent Activities (2020–2023)" International Journal of Neonatal Screening 10, no. 2: 38. https://doi.org/10.3390/ijns10020038
APA StyleTherrell, B. L., Padilla, C. D., Borrajo, G. J. C., Khneisser, I., Schielen, P. C. J. I., Knight-Madden, J., Malherbe, H. L., & Kase, M. (2024). Current Status of Newborn Bloodspot Screening Worldwide 2024: A Comprehensive Review of Recent Activities (2020–2023). International Journal of Neonatal Screening, 10(2), 38. https://doi.org/10.3390/ijns10020038








