3D Single-Breath Chemical Shift Imaging Hyperpolarized Xe-129 MRI of Healthy, CF, IPF, and COPD Subjects
Abstract
1. Introduction
2. Materials and Methods
3. Results
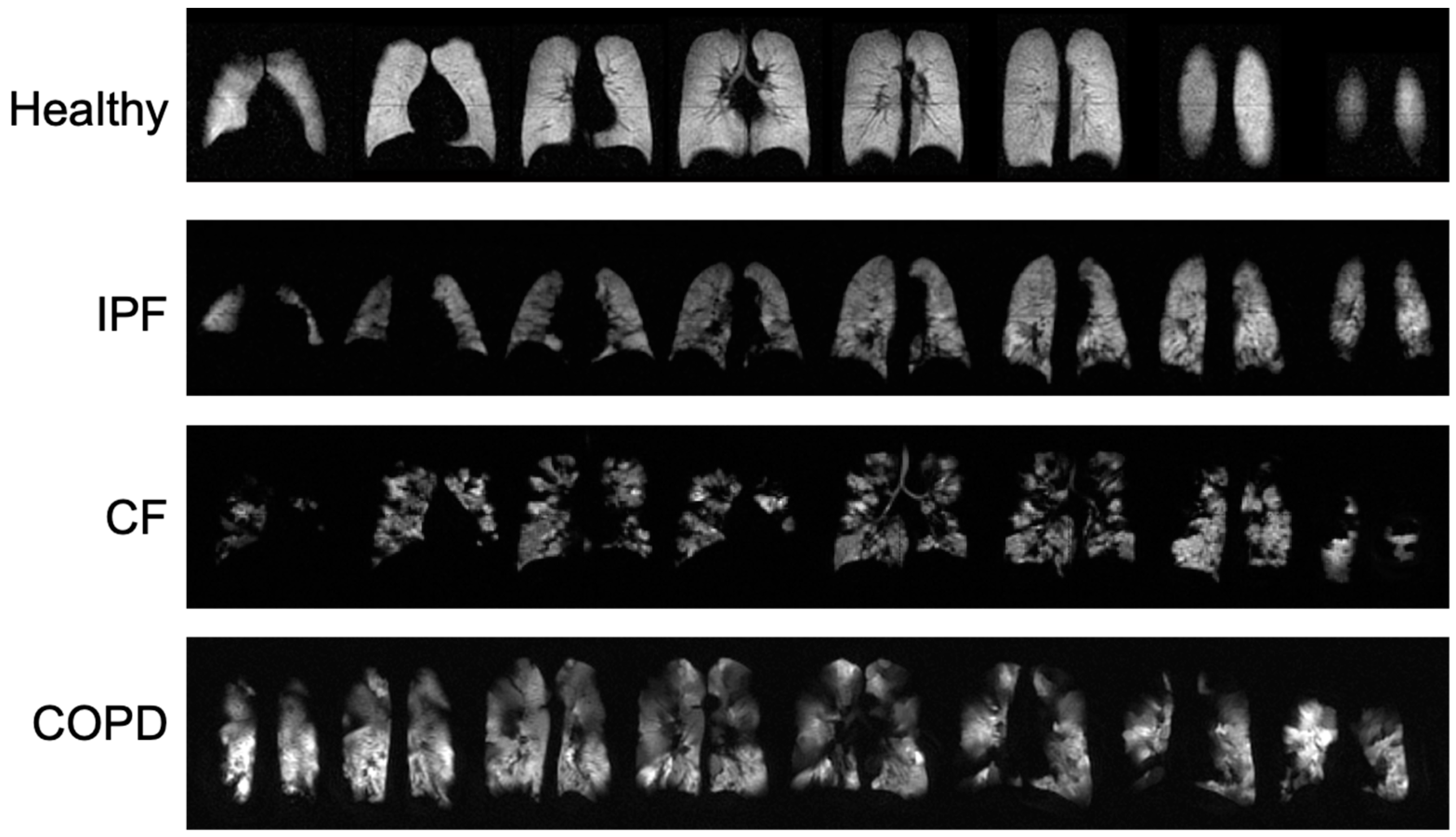
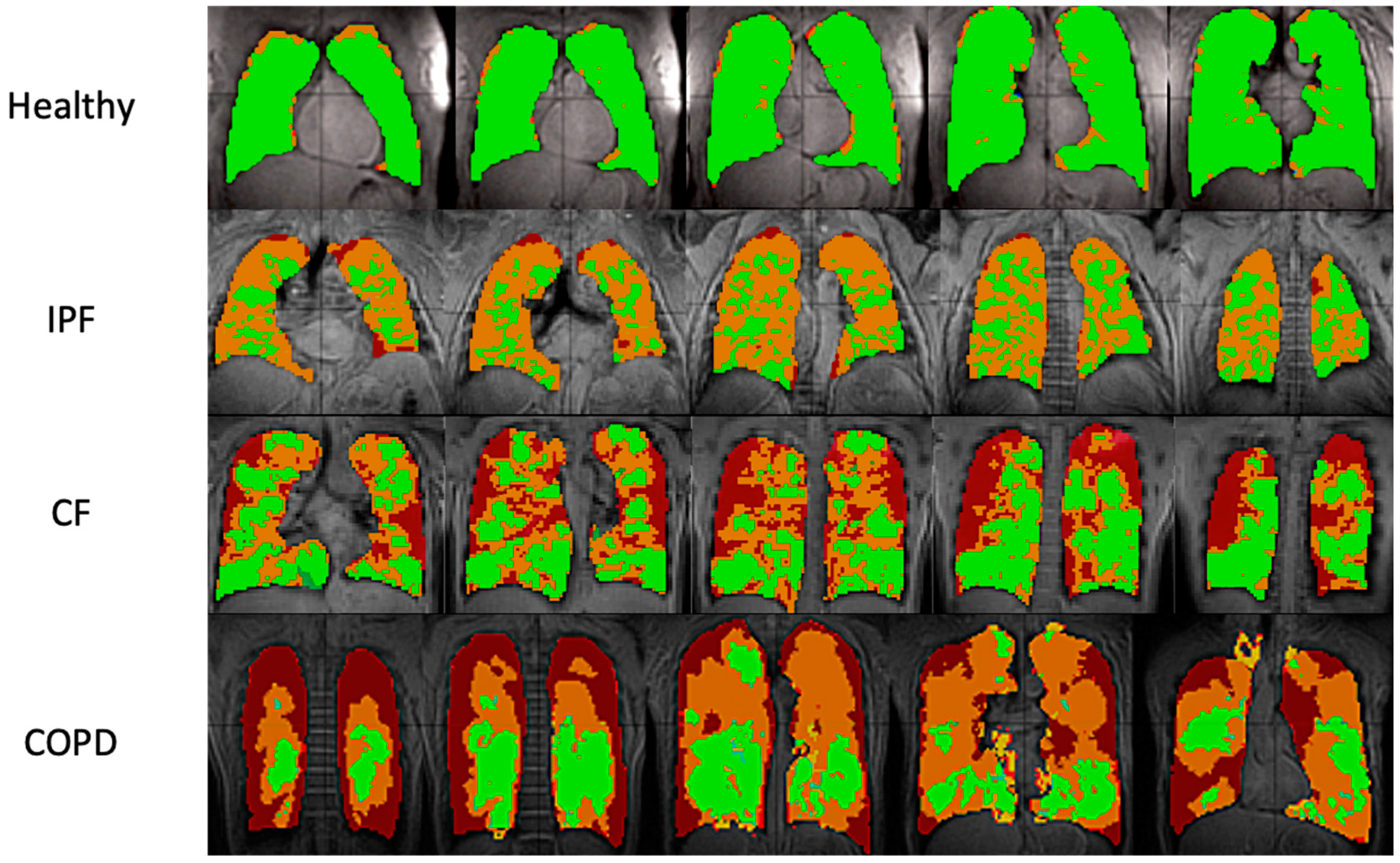
3.1. Comparison of Ventilation Images
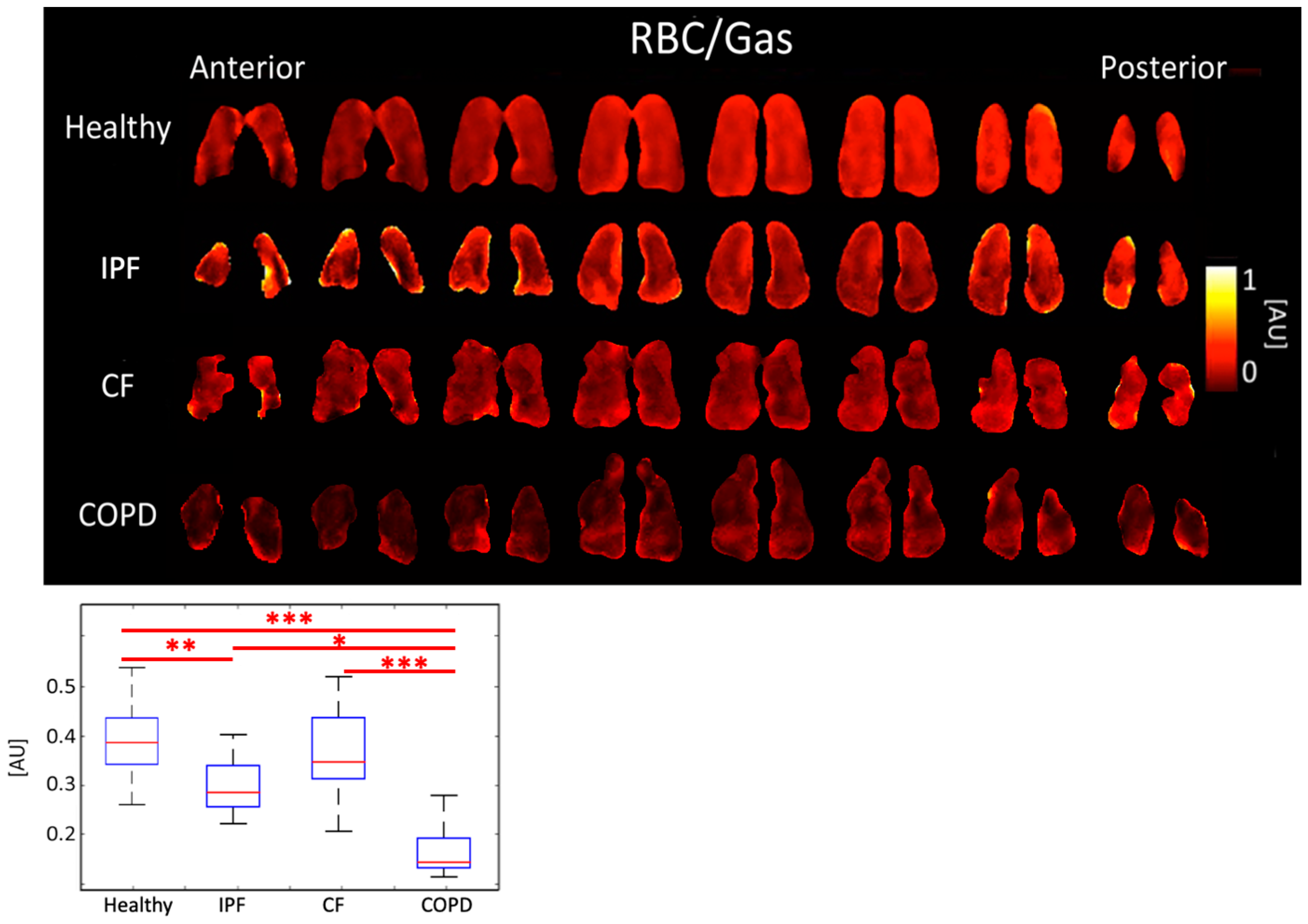
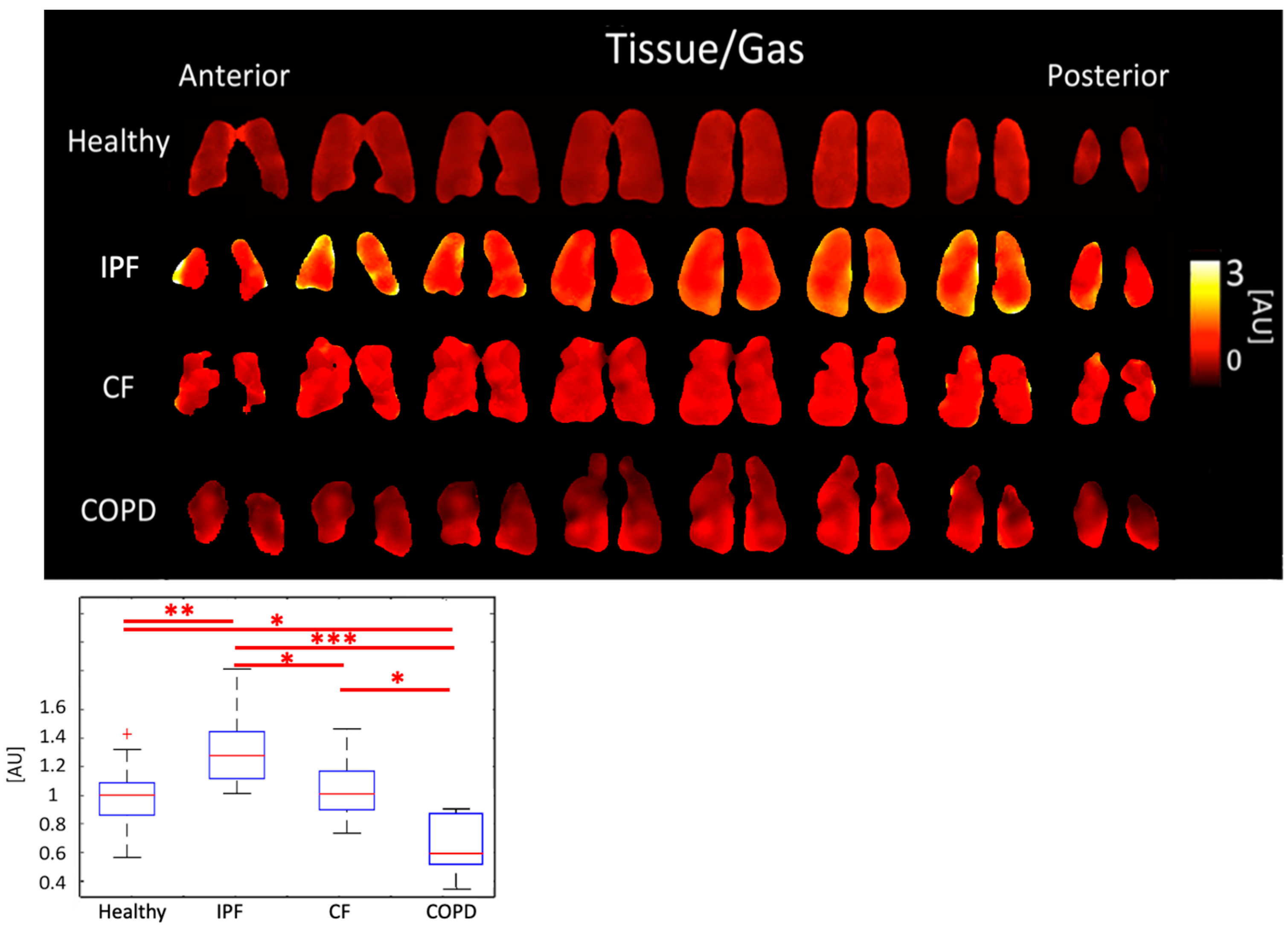
3.2. Comparison of Peak Ratios
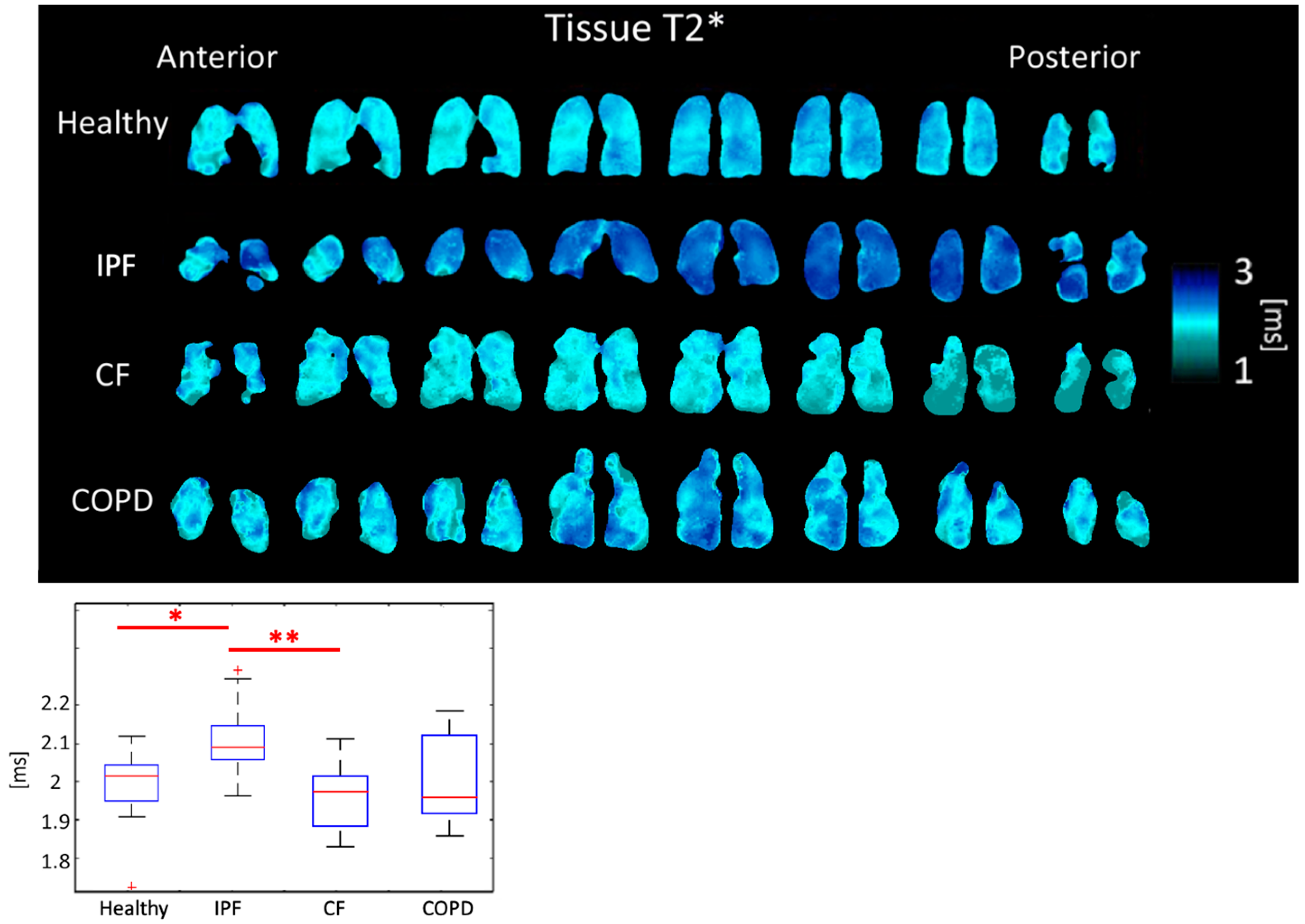
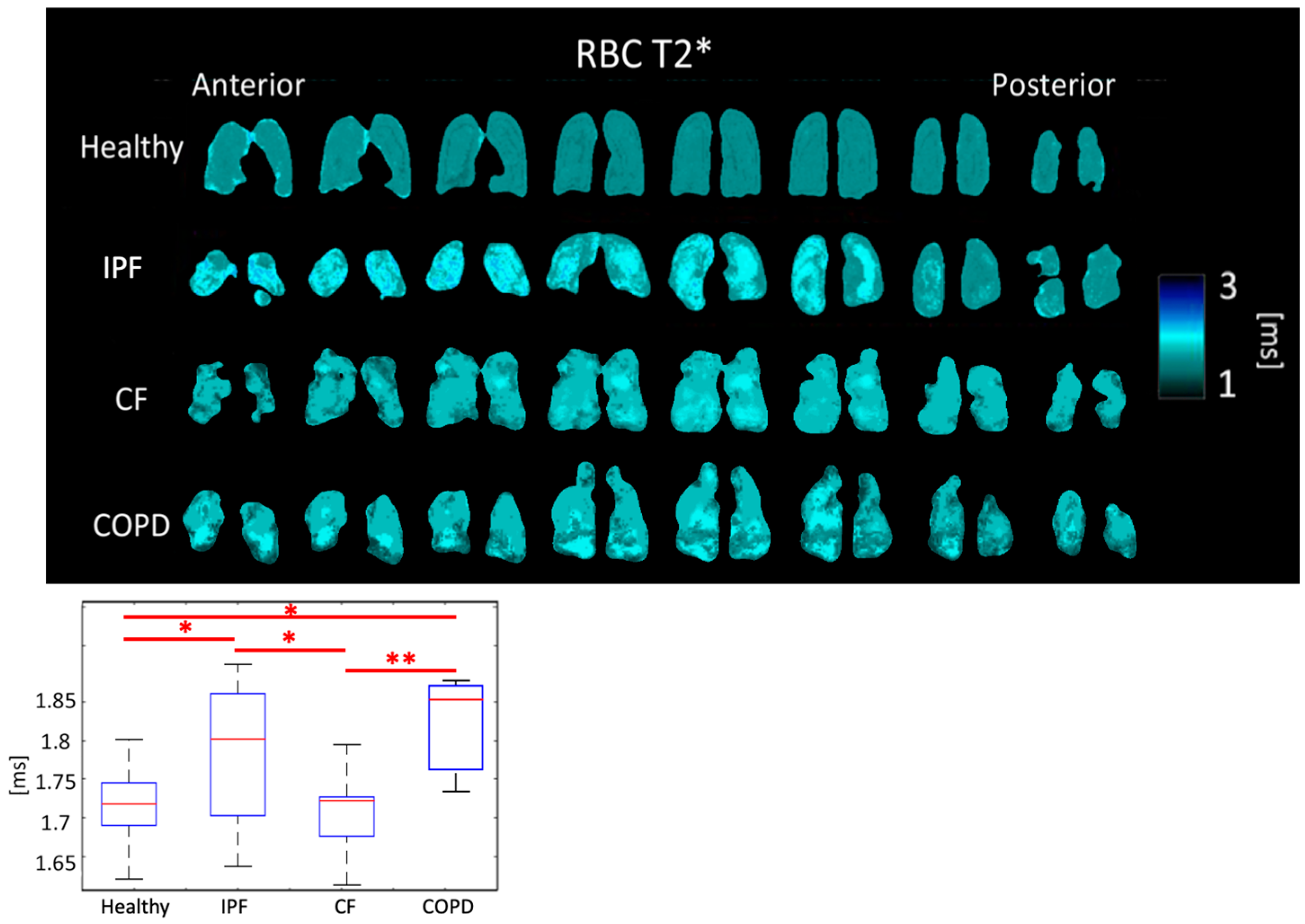
3.3. Comparison of T2*
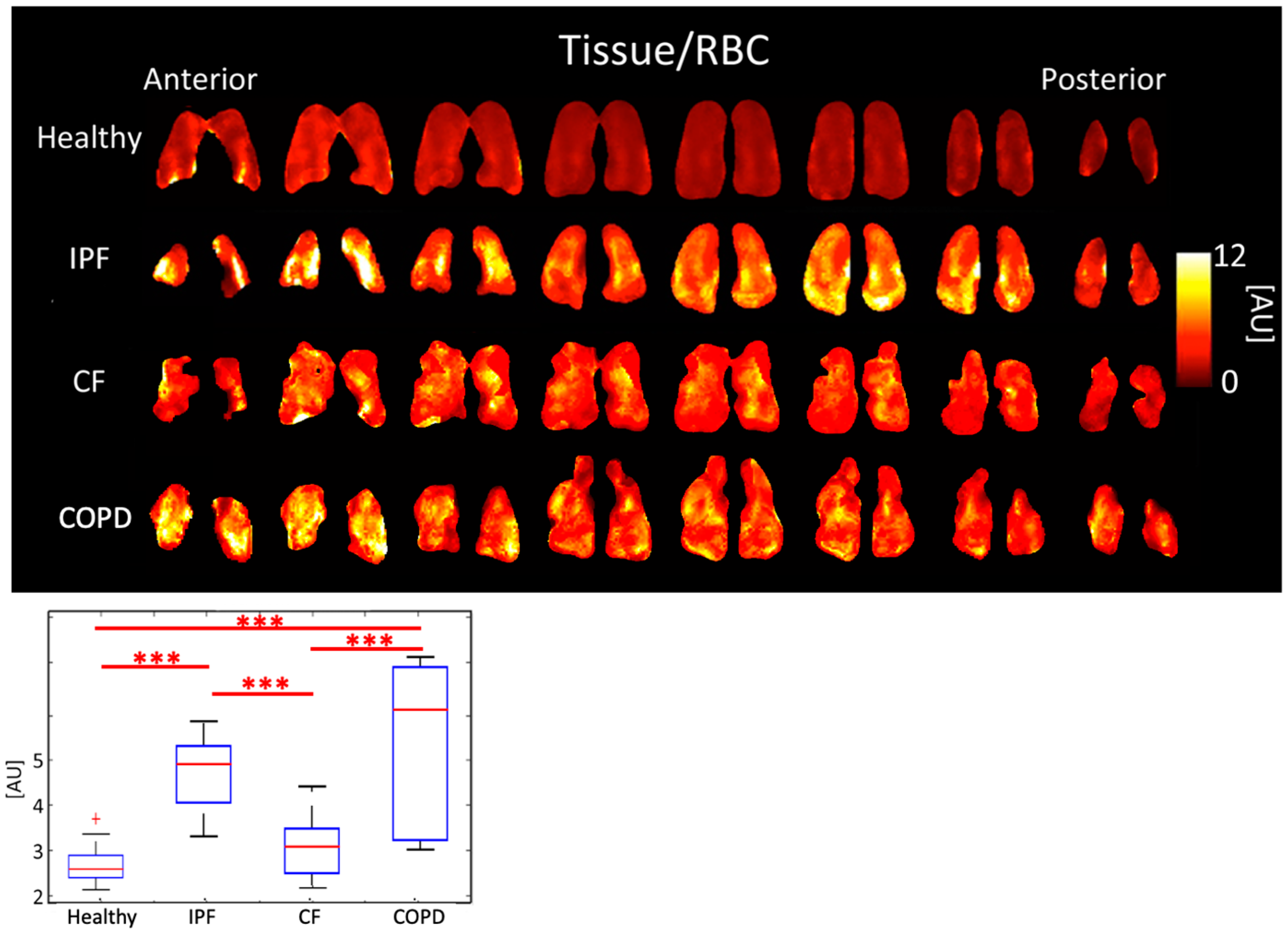
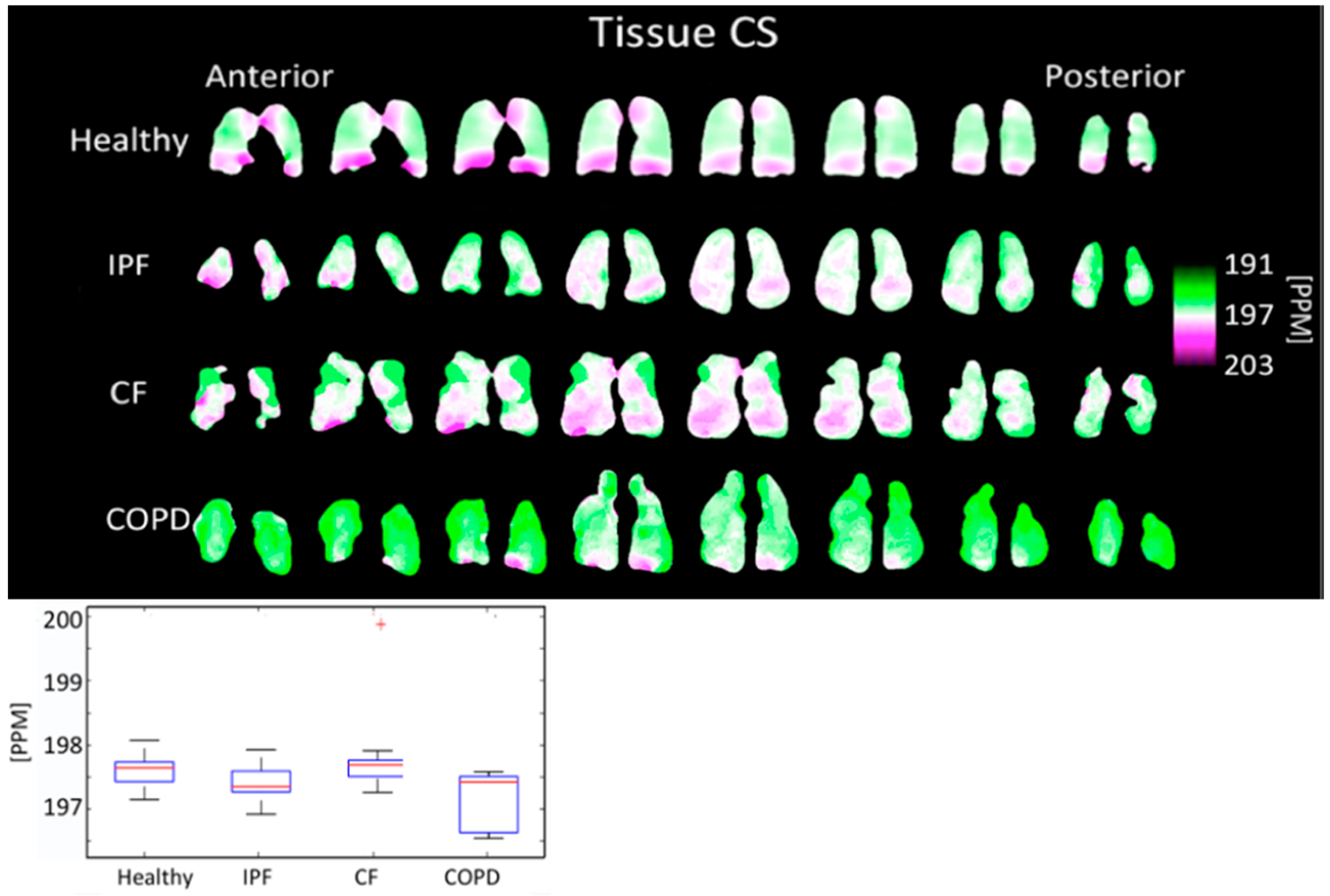
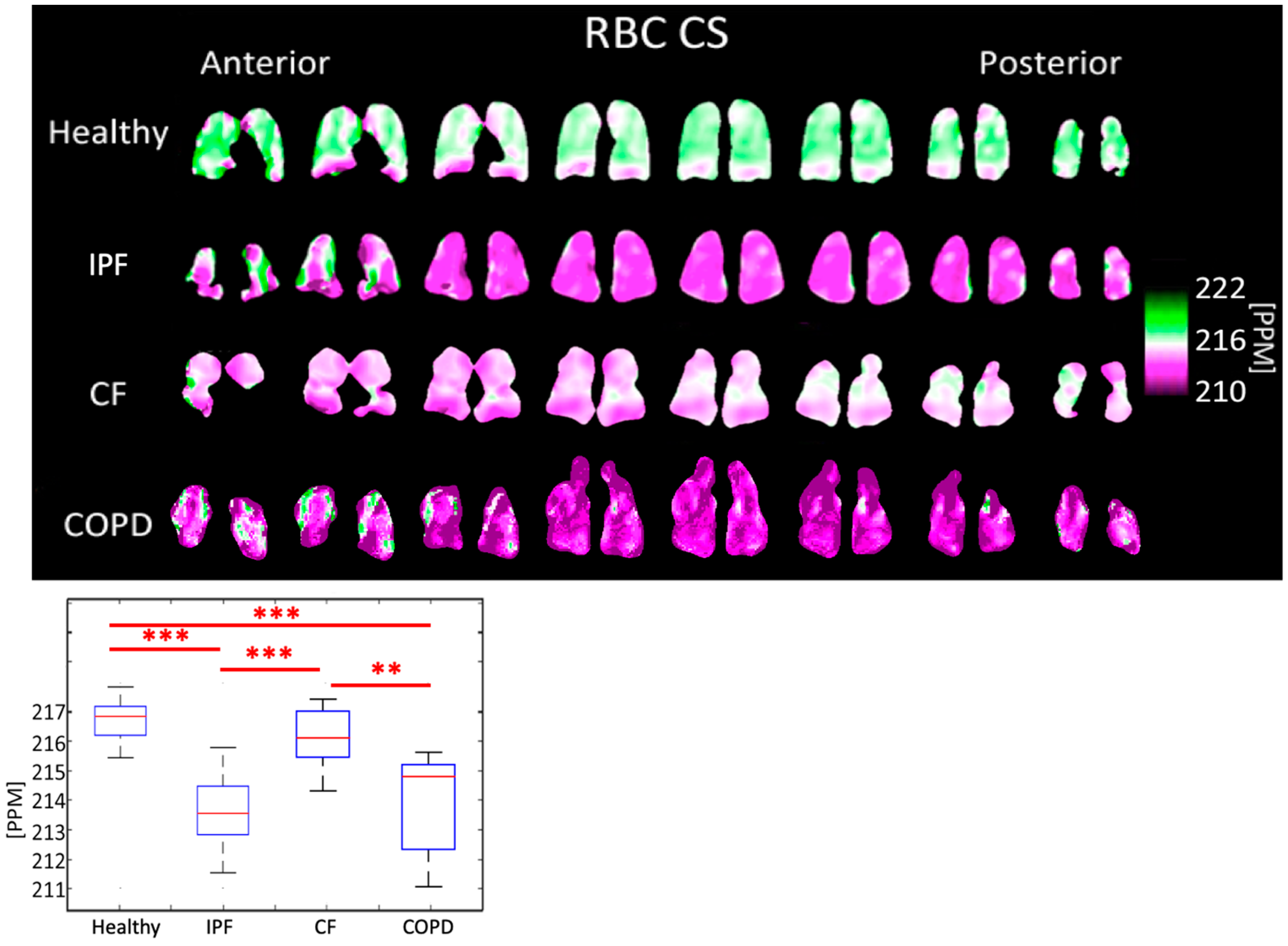
3.4. Comparison of Chemical Shifts
3.5. Correlation with Spirometry
3.6. Repeatability
4. Discussion
4.1. Comparison of Ventilation Images
4.2. Comparison of Peak Ratios
4.3. Comparison of T2*
4.4. Comparison of Chemical Shifts
4.5. Correlation with Spirometry
4.6. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Di Nardo, F.; Laurenti, P. Respiratory Diseases and Health Disorders Related to Indoor and Outdoor Air Pollution. In A Systematic Review of Key Issues in Public Health; Boccia, S., Villari, P., Ricciardi, W., Eds.; Springer International Publishing: Berlin/Heidelberg, Germany, 2015; pp. 109–127. [Google Scholar] [CrossRef]
- Vijayan, V.K. Chronic obstructive pulmonary disease. Indian J. Med. Res. 2013, 137, 251–269. [Google Scholar] [PubMed]
- O’Sullivan, B.P.; Freedman, S.D. Cystic fibrosis. Lancet Lond. Engl. 2009, 373, 1891–1904. [Google Scholar] [CrossRef]
- Heukels, P.; Moor, C.C.; von der Thüsen, J.H.; Wijsenbeek, M.S.; Kool, M. Inflammation and immunity in IPF pathogenesis and treatment. Respir. Med. 2019, 147, 79–91. [Google Scholar] [CrossRef] [PubMed]
- Ley, B.; Collard, H.R.; King, T.E. Clinical Course and Prediction of Survival in Idiopathic Pulmonary Fibrosis. Am. J. Respir. Crit. Care Med. 2011, 183, 431–440. [Google Scholar] [CrossRef]
- Brown, J.P.; Martinez, C.H. Chronic obstructive pulmonary disease comorbidities. Curr. Opin. Pulm. Med. 2016, 22, 113–118. [Google Scholar] [CrossRef]
- Margaritopoulos, G.A.; Antoniou, K.M.; Wells, A.U. Comorbidities in interstitial lung diseases. Eur. Respir. Rev. 2017, 26, 160027. [Google Scholar] [CrossRef]
- Ronan, N.J.; Elborn, J.S.; Plant, B.J. Current and emerging comorbidities in cystic fibrosis. Presse Médicale 2017, 46, e125–e138. [Google Scholar] [CrossRef]
- Kalra, M.K.; Maher, M.M.; Rizzo, S.; Kanarek, D.; Shephard, J.A.O. Radiation exposure from Chest CT: Issues and Strategies. J. Korean Med. Sci. 2004, 19, 159. [Google Scholar] [CrossRef]
- Giuranno, L.; Ient, J.; De Ruysscher, D.; Vooijs, M.A. Radiation-Induced Lung Injury (RILI). Front. Oncol. 2019, 9, 877. [Google Scholar] [CrossRef]
- Liu, Z.; Araki, T.; Okajima, Y.; Albert, M.; Hatabu, H. Pulmonary hyperpolarized noble gas MRI: Recent advances and perspectives in clinical application. Eur. J. Radiol. 2014, 83, 1282–1291. [Google Scholar] [CrossRef]
- Mugler, J.P.; Altes, T.A.; Ruset, I.C.; Dregely, I.M.; Mata, J.F.; Miller, G.W.; Ketel, S.; Ketel, J.; Hersman, F.W.; Ruppert, K. Simultaneous magnetic resonance imaging of ventilation distribution and gas uptake in the human lung using hyperpolarized xenon-129. Proc. Natl. Acad. Sci. USA 2010, 107, 21707–21712. [Google Scholar] [CrossRef] [PubMed]
- Mugler, J.P.; Altes, T.A. Hyperpolarized 129Xe MRI of the human lung. J. Magn. Reson. Imaging JMRI 2013, 37, 313–331. [Google Scholar] [CrossRef] [PubMed]
- Ruppert, K.; Brookeman, J.R.; Hagspiel, K.D.; Mugler, J.P. Probing lung physiology with xenon polarization transfer contrast (XTC). Magn. Reson. Med. 2000, 44, 349–357. [Google Scholar] [CrossRef]
- Wagshul, M.E.; Button, T.M.; Li, H.F.; Liang, Z.; Springer, C.S.; Zhong, K.; Wishnia, A. In vivo MR imaging and spectroscopy using hyperpolarized 129Xe. Magn. Reson. Med. 1996, 36, 183–191. [Google Scholar] [CrossRef]
- Sakaia, K.; Bilek, A.M.; Oteiza, E.R.; Walsworth, R.L.; Balamorec, D.; Jolesz, F.A.; Albert, M.S. Temporal Dynamics of Hyperpolarized129Xe Resonances in Living Rats. J. Magn. Reson. B 1996, 111, 300–304. [Google Scholar] [CrossRef] [PubMed]
- Miller, K.W.; Reo, N.V.; Schoot Uiterkamp, A.J.; Stengle, D.P.; Stengle, T.R.; Williamson, K.L. Xenon NMR: Chemical shifts of a general anesthetic in common solvents, proteins, and membranes. Proc. Natl. Acad. Sci. USA 1981, 78, 4946–4949. [Google Scholar] [CrossRef]
- Cleveland, Z.I.; Virgincar, R.S.; Qi, Y.; Robertson, S.H.; Degan, S.; Driehuys, B. 3D MRI of impaired hyperpolarized 129Xe uptake in a rat model of pulmonary fibrosis. NMR Biomed. 2014, 27, 1502–1514. [Google Scholar] [CrossRef]
- Fernandes, C.; Ruppert, K.; Altes, T.; Mugler, J.; Ruset, I.; Miller, W.; Hersman, W.; Mata, J. Hyperpolarized xenon-129 3D-Chemical Shift Imaging of the lung in subjects with a history of smoke exposure. In Proceedings of the International Society for Magnetic Resonance in Medicine, Salt Lake City, UT, USA, 20–26 April 2013; p. 1450. [Google Scholar]
- Guan, S.; Qing, K.; Altes, T.J.M., III; Fernandes, C.; Ruppert, K.; Ruset, I.; Hersman, F.W.; Froh, D.; Teague, W.; Miller, G. Regional Quantification of Lung Function in Cystic Fibrosis using 3D Single-Breath CSI. In Proceedings of the International Society for Magnetic Resonance in Medicine, Milan, Italy, 10–16 May 2014; Volume 22. [Google Scholar]
- Guan, S.; Qing, K.; Altes, T.; Mugler, J.P., 3rd; Ruset, I.; Froh, D.; Grady, M.; Brookeman, J.; Mata, J.F. T2* and Frequency Shift Maps of Healthy and CF Subjects. In Proceedings of the 23rd Annual Meeting of the ISMRM, Toronto, ON, Canada, 30 May–5 June 2015. [Google Scholar]
- Eddy, R.L.; Parraga, G. Pulmonary xenon-129 MRI: New opportunities to unravel enigmas in respiratory medicine. Eur. Respir. J. 2020, 55, 1901987. [Google Scholar] [CrossRef] [PubMed]
- Svenningsen, S.; Kirby, M.; Starr, D.; Leary, D.; Wheatley, A.; Maksym, G.N.; McCormack, D.G.; Parraga, G. Hyperpolarized (3) He and (129) Xe MRI: Differences in asthma before bronchodilation: MRI of Asthma. J. Magn. Reson. Imaging 2013, 38, 1521–1530. [Google Scholar] [CrossRef] [PubMed]
- Kirby, M.; Svenningsen, S.; Owrangi, A.; Wheatley, A.; Farag, A.; Ouriadov, A.; Santyr, G.E.; Etemad-Rezai, R.; Coxson, H.O.; McCormack, D.G.; et al. Hyperpolarized (3) He and (129) Xe MR Imaging in Healthy Volunteers and Patients with Chronic Obstructive Pulmonary Disease. Radiology 2012, 265, 600–610. [Google Scholar] [CrossRef] [PubMed]
- Kaushik, S.S.; Robertson, S.H.; Freeman, M.S.; He, M.; Kelly, K.T.; Roos, J.E.; Rackley, C.R.; Foster, W.M.; McAdams, H.P.; Driehuys, B. Single-breath clinical imaging of hyperpolarized (129)Xe in the airspaces, barrier, and red blood cells using an interleaved 3D radial 1-point Dixon acquisition. Magn. Reson. Med. 2016, 75, 1434–1443. [Google Scholar] [CrossRef] [PubMed]
- Mata, J.; Guan, S.; Qing, K.; Tustison, N.; Shim, Y.; Mugler, J.P.; Altes, T.; Huaromo, J.; Mehrad, B. Evaluation of Regional Lung Function in Pulmonary Fibrosis with Xenon-129 MRI. Tomogr. Ann. Arbor. Mich. 2021, 7, 452–465. [Google Scholar] [CrossRef] [PubMed]
- Avants, B.B.; Tustison, N.J.; Wu, J.; Cook, P.A.; Gee, J.C. An Open Source Multivariate Framework for n-Tissue Segmentation with Evaluation on Public Data. Neuroinformatics 2011, 9, 381–400. [Google Scholar] [CrossRef]
- Tustison, N.J.; Qing, K.; Wang, C.; Altes, T.A.; Mugler, J.P. Atlas-based estimation of lung and lobar anatomy in proton MRI: Atlas-Based Estimation of Lung and Lobar Anatomy in Proton MRI. Magn. Reson. Med. 2016, 76, 315–320. [Google Scholar] [CrossRef]
- Yushkevich, P.A.; Piven, J.; Hazlett, H.C.; Smith, R.G.; Ho, S.; Gee, J.C.; Gerig, G. User-guided 3D active contour segmentation of anatomical structures: Significantly improved efficiency and reliability. NeuroImage 2006, 31, 1116–1128. [Google Scholar] [CrossRef]
- Tustison, N.J.; Altes, T.A.; Qing, K.; He, M.; Miller, G.W.; Avants, B.B.; Shim, Y.M.; Gee, J.C.; Mugler, J.P., 3rd; Mata, J.F. Image- versus histogram-based considerations in semantic segmentation of pulmonary hyperpolarized gas images. Magn. Reson. Med. 2021, 86, 2822–2836. [Google Scholar] [CrossRef]
- Smith, L.J.; Horsley, A.; Bray, J.; Hughes, P.J.; Biancardi, A.; Norquay, G.; Wildman, M.; West, N.; Marshall, H.; Wild, J.M. The assessment of short- and long-term changes in lung function in cystic fibrosis using 129Xe MRI. Eur. Respir. J. 2020, 56, 2000441. [Google Scholar] [CrossRef]
- Willmering, M.M.; Roach, D.J.; Kramer, E.L.; Walkup, L.L.; Cleveland, Z.I.; Woods, J.C. Sensitive structural and functional measurements and 1-year pulmonary outcomes in pediatric cystic fibrosis. J. Cyst. Fibros. Off. J. Eur. Cyst. Fibros. Soc. 2021, 20, 533–539. [Google Scholar] [CrossRef] [PubMed]
- Crews, M.S.; Bartholmai, B.J.; Adegunsoye, A.; Oldham, J.M.; Montner, S.M.; Karwoski, R.A.; Husain, A.N.; Vij, R.; Noth, I.; Strek, M.E.; et al. Automated CT Analysis of Major Forms of Interstitial Lung Disease. J. Clin. Med. 2020, 9, 3776. [Google Scholar] [CrossRef] [PubMed]
- Wallis, A.; Spinks, K. The diagnosis and management of interstitial lung diseases. BMJ 2015, 350, h2072. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.M.; Robertson, S.H.; Wang, Z.; He, M.; Virgincar, R.S.; Schrank, G.M.; Smigla, R.M.; O’Riordan, T.G.; Sundy, J.; Ebner, L.; et al. Using hyperpolarized (129) Xe MRI to quantify regional gas transfer in idiopathic pulmonary fibrosis. Thorax 2018, 73, 21–28. [Google Scholar] [CrossRef]
- Wolber, J.; Cherubini, A.; Santoro, D.; Payne, G.S.; Leach, M.O.; Bifone, A. Linewidths of Hyperpolarized 129Xe NMR Spectra in Human Blood at 1.5T. Proc. Int. Soc. Magn. Reson. Med. 2000, 8, 970. [Google Scholar]
- Bauman, J.H.; Harris, J.W. Estimation of hepatic iron stores by vivo measurement of magnetic susceptibility. J. Lab. Clin. Med. 1967, 70, 246–257. [Google Scholar]
- Kaushik, S.S.; Freeman, M.S.; Yoon, S.W.; Liljeroth, M.G.; Stiles, J.V.; Roos, J.E.; Michael Foster, W.S.; Rackley, C.R.; McAdams, H.P.; Driehuys, B. Measuring diffusion limitation with a perfusion-limited gas—Hyperpolarized 129Xe gas-transfer spectroscopy in patients with idiopathic pulmonary fibrosis. J. Appl. Physiol. 2014, 117, 577–585. [Google Scholar] [CrossRef]
- Norquay, G.; Leung, G.; Stewart, N.J.; Wolber, J.; Wild, J.M. 129Xe chemical shift in human blood and pulmonary blood oxygenation measurement in humans using hyperpolarized 129Xe NMR. Magn. Reson. Med. 2017, 77, 1399–1408. [Google Scholar] [CrossRef]
- Wolber, J.; Cherubini, A.; Leach, M.O.; Bifone, A. Hyperpolarized 129Xe NMR as a probe for blood oxygenation. Magn. Reson. Med. 2000, 43, 491–496. [Google Scholar] [CrossRef]
- MacIntyre, N. Standardisation of the single-breath determination of carbon monoxide uptake in the lung. Eur. Respir. J. 2005, 26, 720–735. [Google Scholar] [CrossRef]
| Healthy | IPF | CF | COPD | ||
|---|---|---|---|---|---|
| Age | 28 ± 9.8 | 66 ± 11.6 | 24 ± 8.7 | 64 ± 11.8 | |
| M/F | 5/11 | 8/3 | 3/10 | 4/1 | |
| FVC Predicted [%] | 103 ± 8.8 | 66 ± 15.0 | 92 ± 19.6 | 91 ± 6.3 | |
| FEV1 Predicted [%] | 99 ± 7.6 | 67 ± 14.7 | 76 ± 23.6 | 55 ± 20.9 | |
| Ventilation | No Ventilation [%] | 0.4 ± 0.50 | 2.2 ± 0.91 | 14.1 ± 8.37 | 20.3 ± 11.25 |
| Hypoventilation [%] | 9.2 ± 6.96 | 25.8 ± 6.31 | 25.0 ± 6.41 | 39.1 ± 2.23 | |
| Normal Ventilation [%] | 90.4 ± 7.37 | 72.0 ± 6.44 | 60.9 ± 13.86 | 40.6 ± 9.14 | |
| Whole-Lung CSI Averages | Tissue/RBC [AU] | 2.66 ± 0.448 | 4.71 ± 0.807 | 3.06 ± 0.640 | 5.30 ± 2.040 |
| RBC/Gas [AU] | 0.39 ± 0.079 | 0.28 ± 0.061 | 0.35 ± 0.094 | 0.15 ± 0.068 | |
| Tissue/Gas [AU] | 0.99 ± 0.196 | 1.31 ± 0.259 | 1.02 ± 0.196 | 0.66 ± 0.220 | |
| Tissue T2* [ms] | 2.00 ± 0.089 | 2.12 ± 0.093 | 1.97 ± 0.084 | 2.02 ± 0.131 | |
| RBC T2* [ms] | 1.72 ± 0.040 | 1.79 ± 0.086 | 1.71 ± 0.053 | 1.82 ± 0.061 | |
| Tissue CS [PPM] | 197.69 ± 0.227 | 197.48 ± 0.292 | 197.87 ± 0.655 | 197.28 ± 0.434 | |
| RBC CS [PPM] | 216.60 ± 0.645 | 213.49 ± 1.254 | 215.99 ± 0.910 | 213.74 ± 1.872 | |
| RBC-Tissue CS [PPM] | 18.90 ± 0.627 | 16.13 ± 1.21 | 18.34 ± 0.859 | 16.55 ± 1.698 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guan, S.; Tustison, N.; Qing, K.; Shim, Y.M.; Mugler, J., III; Altes, T.; Albon, D.; Froh, D.; Mehrad, B.; Patrie, J.; et al. 3D Single-Breath Chemical Shift Imaging Hyperpolarized Xe-129 MRI of Healthy, CF, IPF, and COPD Subjects. Tomography 2022, 8, 2574-2587. https://doi.org/10.3390/tomography8050215
Guan S, Tustison N, Qing K, Shim YM, Mugler J III, Altes T, Albon D, Froh D, Mehrad B, Patrie J, et al. 3D Single-Breath Chemical Shift Imaging Hyperpolarized Xe-129 MRI of Healthy, CF, IPF, and COPD Subjects. Tomography. 2022; 8(5):2574-2587. https://doi.org/10.3390/tomography8050215
Chicago/Turabian StyleGuan, Steven, Nick Tustison, Kun Qing, Yun Michael Shim, John Mugler, III, Talissa Altes, Dana Albon, Deborah Froh, Borna Mehrad, James Patrie, and et al. 2022. "3D Single-Breath Chemical Shift Imaging Hyperpolarized Xe-129 MRI of Healthy, CF, IPF, and COPD Subjects" Tomography 8, no. 5: 2574-2587. https://doi.org/10.3390/tomography8050215
APA StyleGuan, S., Tustison, N., Qing, K., Shim, Y. M., Mugler, J., III, Altes, T., Albon, D., Froh, D., Mehrad, B., Patrie, J., Ropp, A., Miller, B., Nehrbas, J., & Mata, J. (2022). 3D Single-Breath Chemical Shift Imaging Hyperpolarized Xe-129 MRI of Healthy, CF, IPF, and COPD Subjects. Tomography, 8(5), 2574-2587. https://doi.org/10.3390/tomography8050215







