A New Classification of the Anatomical Variations of Labbé’s Inferior Anastomotic Vein
Abstract
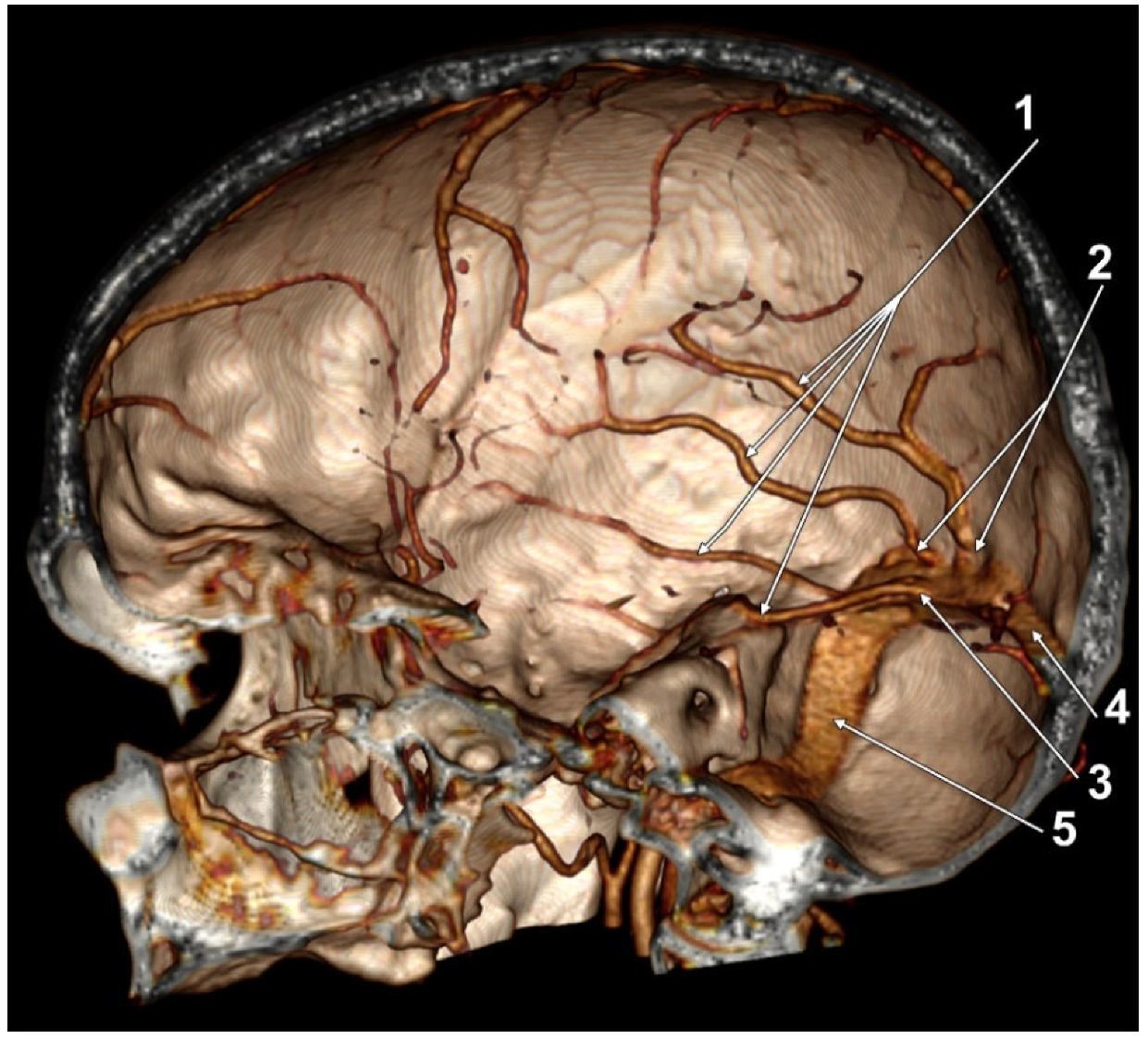
:1. Introduction
2. Materials and Methods
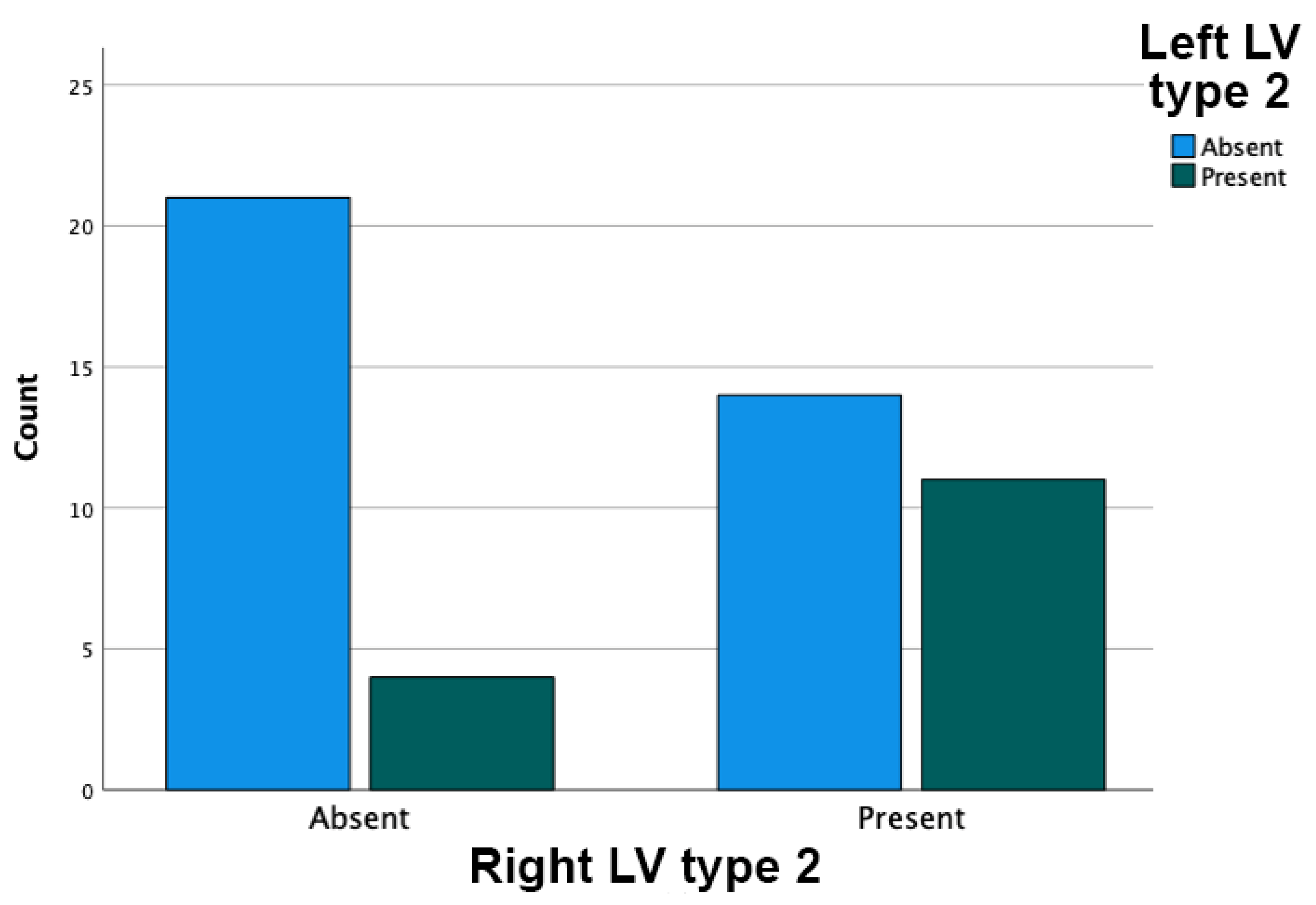
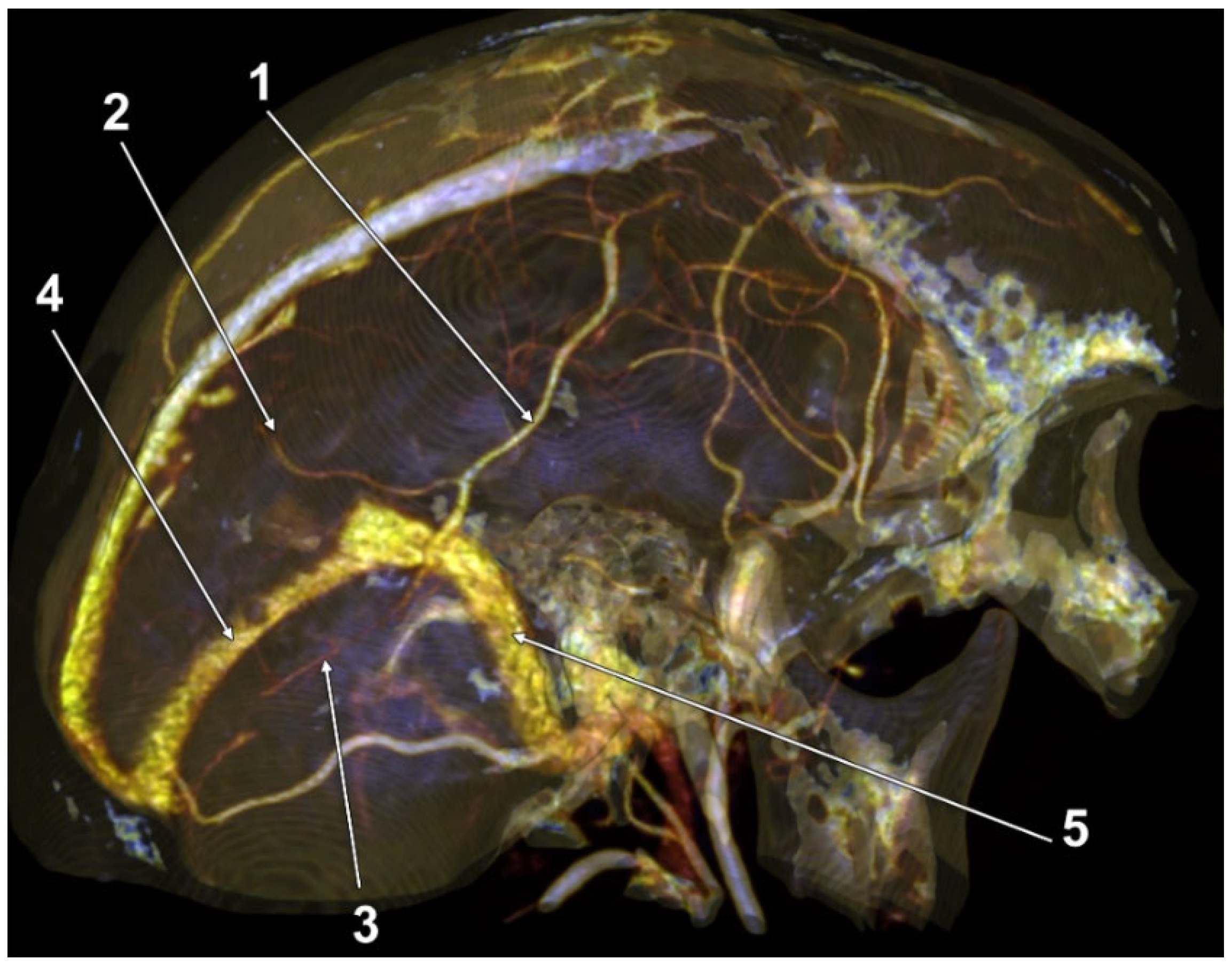
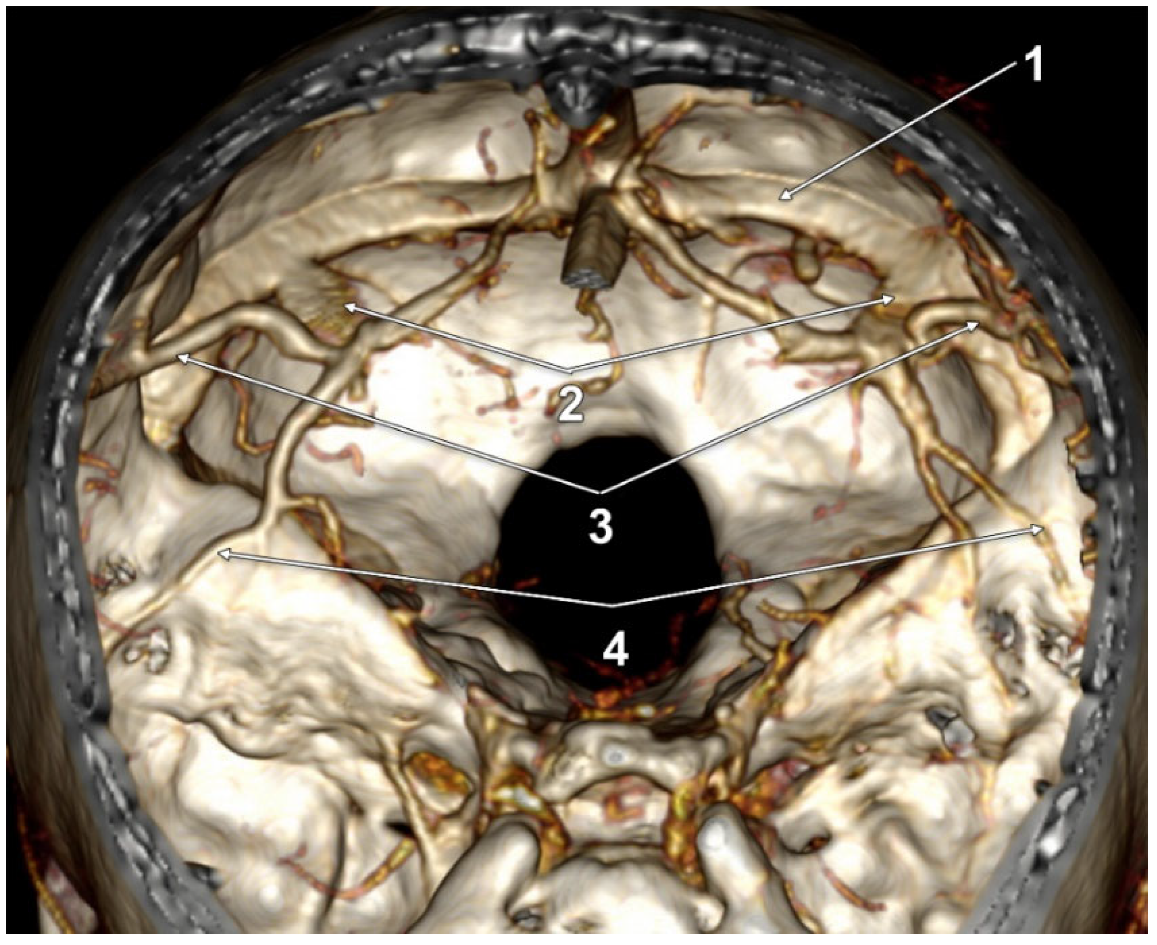
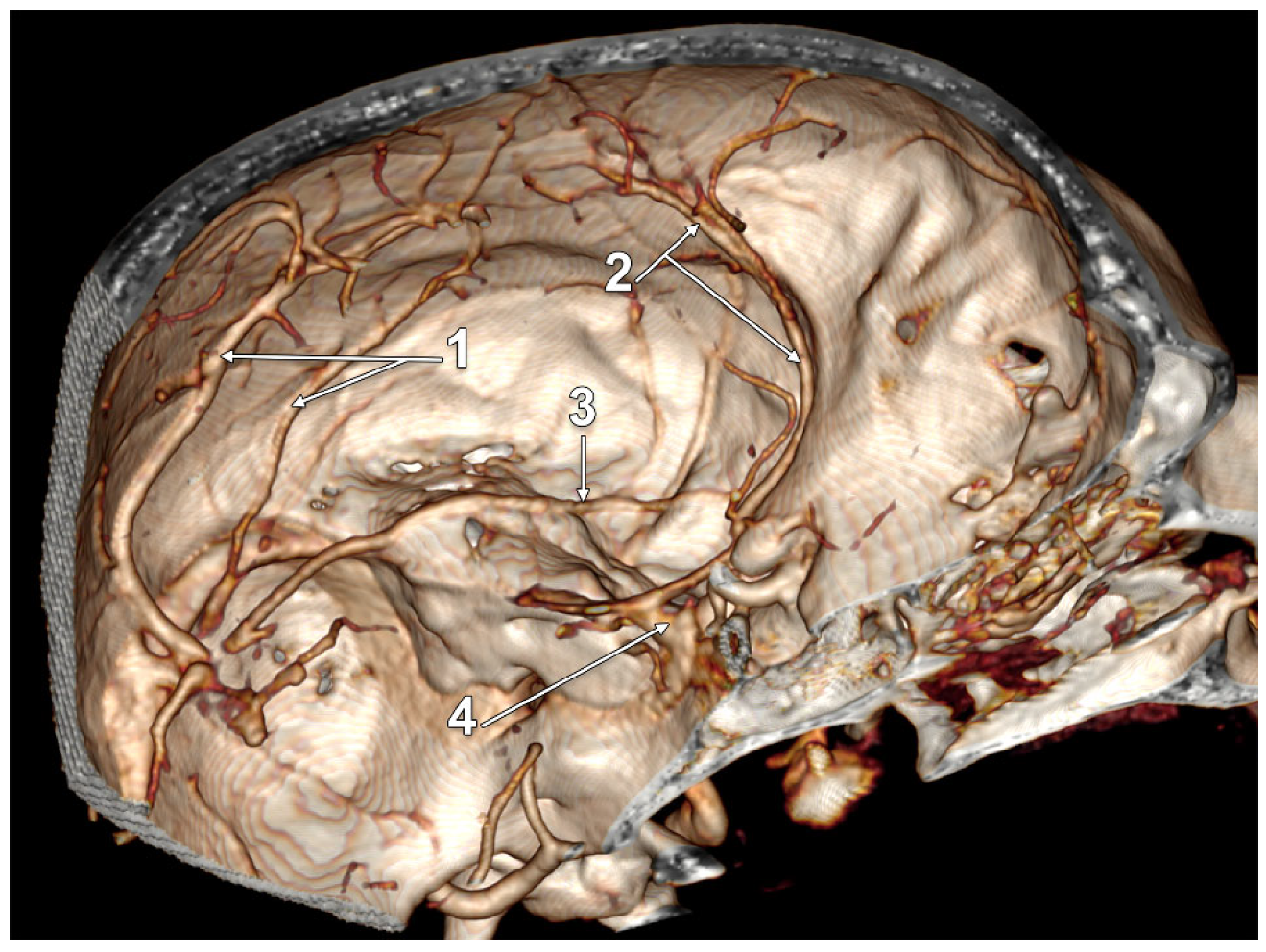
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Avci, E.; Dagtekin, A.; Akture, E.; Uluc, K.; Baskaya, M.K. Microsurgical anatomy of the vein of Labbe. Surg. Radiol. Anat. 2011, 33, 569–573. [Google Scholar] [CrossRef] [PubMed]
- Gray, H.; Standring, S.; Anand, N.; Birch, R.; Collins, P.; Crossman, A.; Gleeson, M.; Jawaheer, G.; Smith, A.L.; Spratt, J.D.; et al. Gray’s Anatomy: The Anatomical Basis of Clinical Practice, 41st ed.; Elsevier: Amsterdam, The Netherlands, 2016. [Google Scholar]
- Shoman, N.M.; Patel, B.; Cornelius, R.S.; Samy, R.N.; Pensak, M.L. Contemporary angiographic assessment and clinical implications of the vein of labbe in neurotologic surgery. Otol. Neurotol. 2011, 32, 1012–1016. [Google Scholar] [CrossRef] [PubMed]
- Boukobza, M.; Crassard, I.; Bousser, M.G.; Chabriat, H. Labbe vein thrombosis. Neuroradiology 2020, 62, 935–945. [Google Scholar] [CrossRef] [PubMed]
- Koperna, T.; Tschabitscher, M.; Knosp, E. The termination of the vein of “Labbe” and its microsurgical significance. Acta Neurochir. 1992, 118, 172–175. [Google Scholar] [CrossRef] [PubMed]
- Tubbs, R.S.; Louis, R.G., Jr.; Song, Y.B.; Mortazavi, M.; Loukas, M.; Shoja, M.M.; Cohen-Gadol, A.A. External landmarks for identifying the drainage site of the vein of Labbe: Application to neurosurgical procedures. Br. J. Neurosurg. 2012, 26, 383–385. [Google Scholar] [CrossRef] [PubMed]
- Silva, P.S.; Vilarinho, A.; Carvalho, B.; Vaz, R. Anatomical variations of the vein of Labbe: An angiographic study. Surg. Radiol. Anat. 2014, 36, 769–773. [Google Scholar] [CrossRef]
- Lustig, L.R.; Jackler, R.K. The vulnerability of the vein of Labbe during combined craniotomies of the middle and posterior fossae. Skull Base Surg. 1998, 8, 1–9. [Google Scholar] [CrossRef]
- Żytkowski, A.; Tubbs, R.S.; Iwanaga, J.; Clarke, E.; Polguj, M.; Wysiadecki, G. Anatomical normality and variability: Historical perspective and methodological considerations. Transl. Res. Anat. 2021, 23, 100105. [Google Scholar] [CrossRef]
- Naidoo, J.; Harrichandparsad, R.; Lazarus, L. Anatomical variations of dominant anastomotic veins in the superficial cortical venous system. Transl. Res. Anat. 2022, 28, 100210. [Google Scholar] [CrossRef]
- Moraru, L.; Rusu, M.C.; Popescu, S.A. True terminal pentafurcation of the external carotid artery and terminal trifurcation of the contralateral one, occipitoauricular trunk, retropharyngeal internal carotid artery. Surg. Radiol. Anat. 2021, 43, 1895–1900. [Google Scholar] [CrossRef]
- Mizutani, K.; Miwa, T.; Akiyama, T.; Sakamoto, Y.; Fujiwara, H.; Yoshida, K. Fate of the three embryonic dural sinuses in infants: The primitive tentorial sinus, occipital sinus, and falcine sinus. Neuroradiology 2018, 60, 325–333. [Google Scholar] [CrossRef] [PubMed]
- Chung, J.I.; Weon, Y.C. Anatomic variations of the superficial middle cerebral vein: Embryologic aspects of the regressed embryonic tentorial sinus. Interv. Neuroradiol. 2005, 11, 115–122. [Google Scholar] [CrossRef]
- Suzuki, Y.; Matsumoto, K. Variations of the superficial middle cerebral vein: Classification using three-dimensional CT angiography. AJNR Am. J. Neuroradiol. 2000, 21, 932–938. [Google Scholar] [PubMed]
- San Millan Ruiz, D.; Fasel, J.H.; Rufenacht, D.A.; Gailloud, P. The sphenoparietal sinus of breschet: Does it exist? An anatomic study. AJNR Am. J. Neuroradiol. 2004, 25, 112–120. [Google Scholar] [PubMed]
- Matsushima, T.; Suzuki, S.O.; Fukui, M.; Rhoton, A.L., Jr.; de Oliveira, E.; Ono, M. Microsurgical anatomy of the tentorial sinuses. J. Neurosurg. 1989, 71, 923–928. [Google Scholar] [CrossRef]
- Shibao, S.; Toda, M.; Fujiwara, H.; Jinzaki, M.; Yoshida, K. Bridging vein and tentorial sinus in the subtemporal corridor during the anterior transpetrosal approach. Acta Neurochir. 2019, 161, 821–829. [Google Scholar] [CrossRef]
- Terbrugge, K.; Lasjaunias, P. Tentorial sinus. Radiologic and anatomic features of a case. Surg. Radiol. Anat. 1988, 10, 243–246. [Google Scholar] [CrossRef]
- Miabi, Z.; Midia, R.; Rohrer, S.E.; Hoeffner, E.G.; Vandorpe, R.; Berk, C.M.; Midia, M. Delineation of lateral tentorial sinus with contrast-enhanced MR imaging and its surgical implications. AJNR Am. J. Neuroradiol. 2004, 25, 1181–1188. [Google Scholar]
- Hyrtl, J. Der Sinus ophthalmo-petrosus. Wien. Med. Wochenschr. 1862, 19, 291–292. [Google Scholar]
- Minca, D.I.; Rusu, M.C. The Ophthalmopetrosal Sinus of Hyrtl: The Evidence of A Rare Variant. J. Craniofac. Surg. 2021, 32, 2551–2552. [Google Scholar] [CrossRef]
- Marsot-Dupuch, K.; Gayet-Delacroix, M.; Elmaleh-Berges, M.; Bonneville, F.; Lasjaunias, P. The petrosquamosal sinus: CT and MR findings of a rare emissary vein. AJNR Am. J. Neuroradiol. 2001, 22, 1186–1193. [Google Scholar] [PubMed]
- Sasaki, C.T.; Allen, W.E.; Spencer, D. Cerebral cortical veins in otologic surgery. Arch. Otolaryngol. 1977, 103, 730–734. [Google Scholar] [CrossRef] [PubMed]
- Fang, Q.; Jiang, A.; Tao, W.; Xin, L. Anatomic comparison of veins of Labbe between autopsy, digital subtraction angiography and computed tomographic venography. Biomed. Eng. Online 2017, 16, 84. [Google Scholar] [CrossRef] [PubMed]
- Sakata, K.; Al-Mefty, O.; Yamamoto, I. Venous consideration in petrosal approach: Microsurgical anatomy of the temporal bridging vein. Neurosurgery 2000, 47, 153–160, Discussion 60–61. [Google Scholar] [PubMed]
- Ahmad, W.; Siddique, K.; Din, I.U. Vein of Labbe Thrombosis-A missed culprit. J. Pak. Med. Assoc. 2020, 70, 1473–1474. [Google Scholar]
- Mageid, R.; Ding, Y.; Fu, P. Vein of Labbe thrombosis, a near-miss. Brain Circ. 2018, 4, 188–190. [Google Scholar]
- Gailloud, P.; Muster, M.; Khaw, N.; Martin, J.B.; Murphy, K.J.; Fasel, J.H.; Rufenacht, D.A. Anatomic relationship between arachnoid granulations in the transverse sinus and the termination of the vein of Labbe: An angiographic study. Neuroradiology 2001, 43, 139–143. [Google Scholar] [CrossRef]
- Castro-Afonso, L.H.; Trivelato, F.P.; Rezende, M.T.; Ulhoa, A.C.; Nakiri, G.S.; Monsignore, L.M.; Abud, D.G. The routes for embolization of dural carotid cavernous fistulas when the endovascular approach is indicated as a first-line strategy. Interv. Neuroradiol. 2019, 25, 66–70. [Google Scholar] [CrossRef]
- Konstas, A.A.; Song, A.; Song, J.; Thanos, A.; Ross, I.B. Embolization of a cavernous carotid fistula through the vein of Labbe: A new alternative transvenous access route. J. Neurointerv. Surg. 2018, 10, e11. [Google Scholar] [CrossRef]
- Kazumata, K.; Kamiyama, H.; Ishikawa, T.; Takizawa, K.; Maeda, T.; Makino, K.; Gotoh, S. Operative anatomy and classification of the sylvian veins for the distal transsylvian approach. Neurol. Med. Chir. 2003, 43, 427–433, discussion 34. [Google Scholar] [CrossRef]
- Imada, Y.; Kurisu, K.; Takumi, T.; Aoyama, H.; Sadatomo, T.; Migita, K.; Yuki, K. Morphological Pattern and Classification of the Superficial Middle Cerebral Vein by Cadaver Dissections: An Embryological Viewpoint. Neurol. Med. Chir. 2019, 59, 264–270. [Google Scholar] [CrossRef]
- Shibao, S.; Toda, M.; Orii, M.; Fujiwara, H.; Yoshida, K. Various patterns of the middle cerebral vein and preservation of venous drainage during the anterior transpetrosal approach. J. Neurosurg. 2016, 124, 432–439. [Google Scholar] [CrossRef] [Green Version]
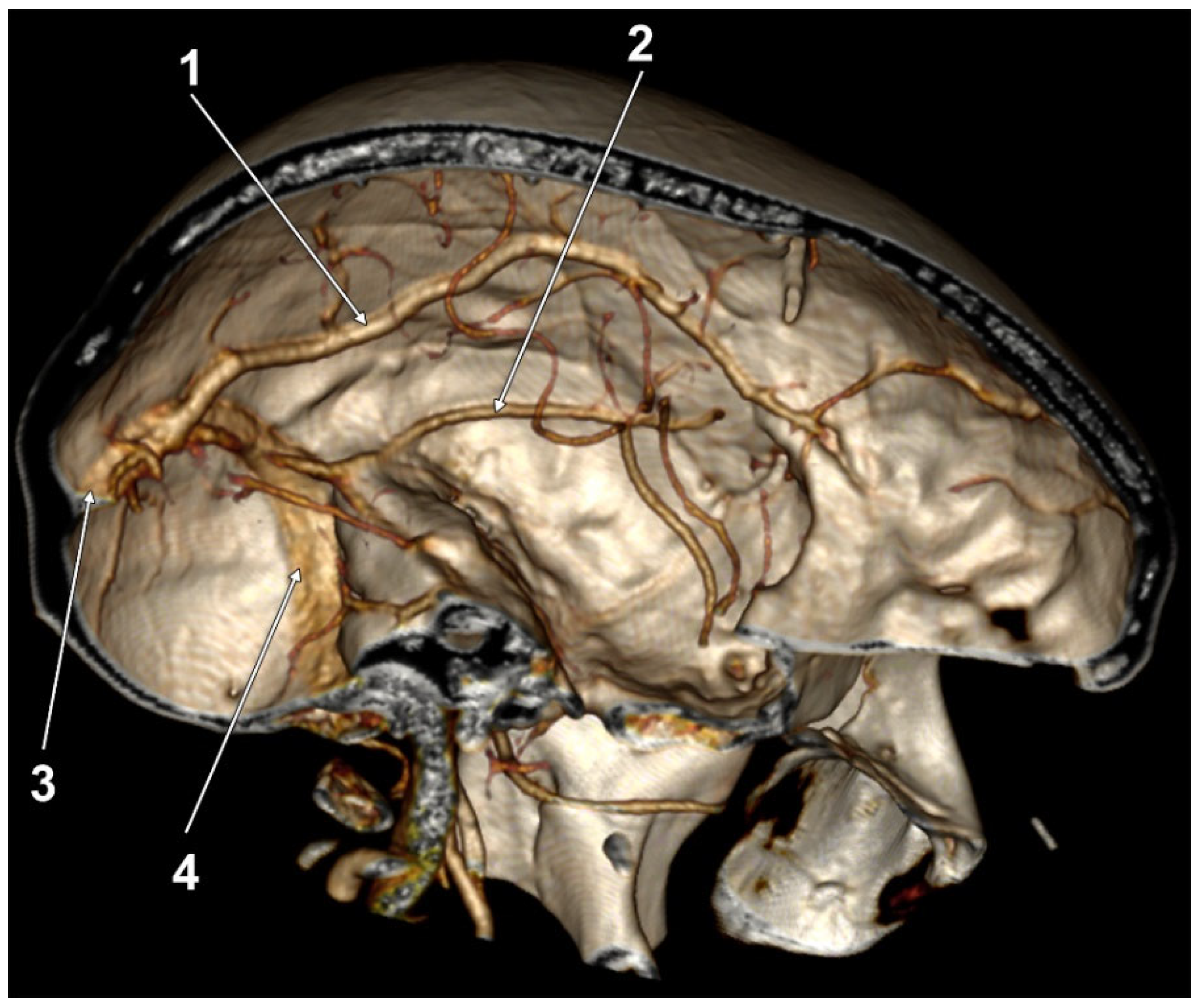
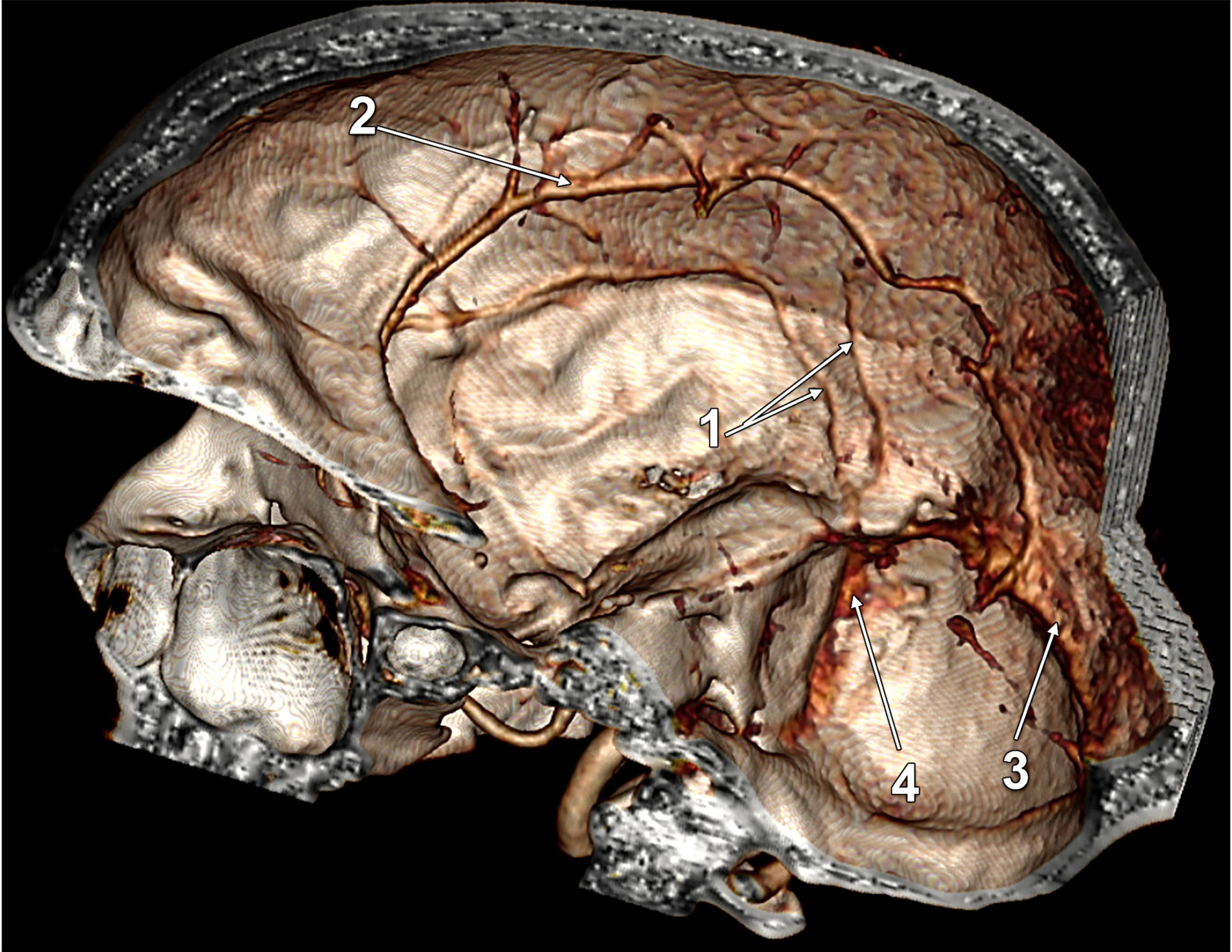
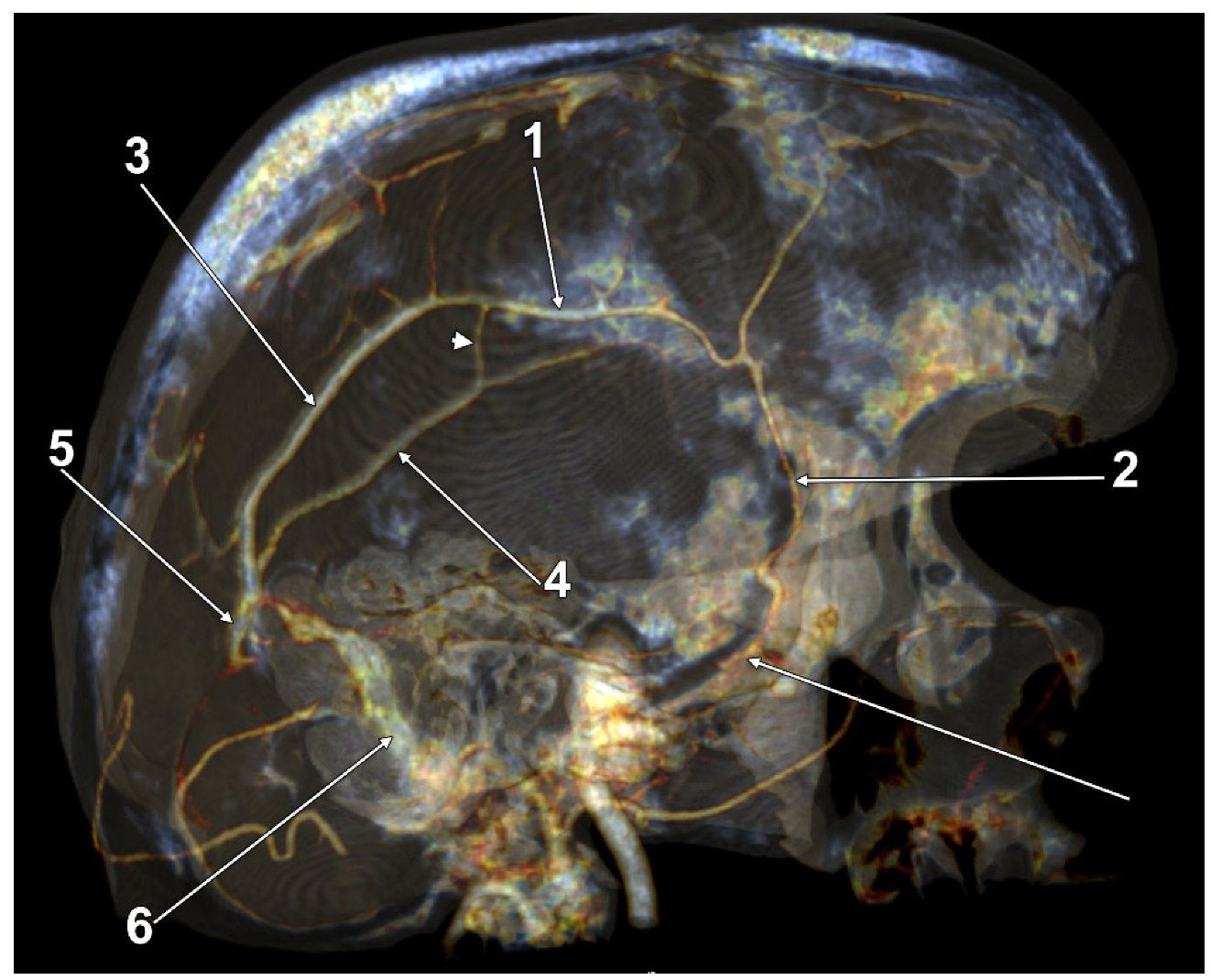
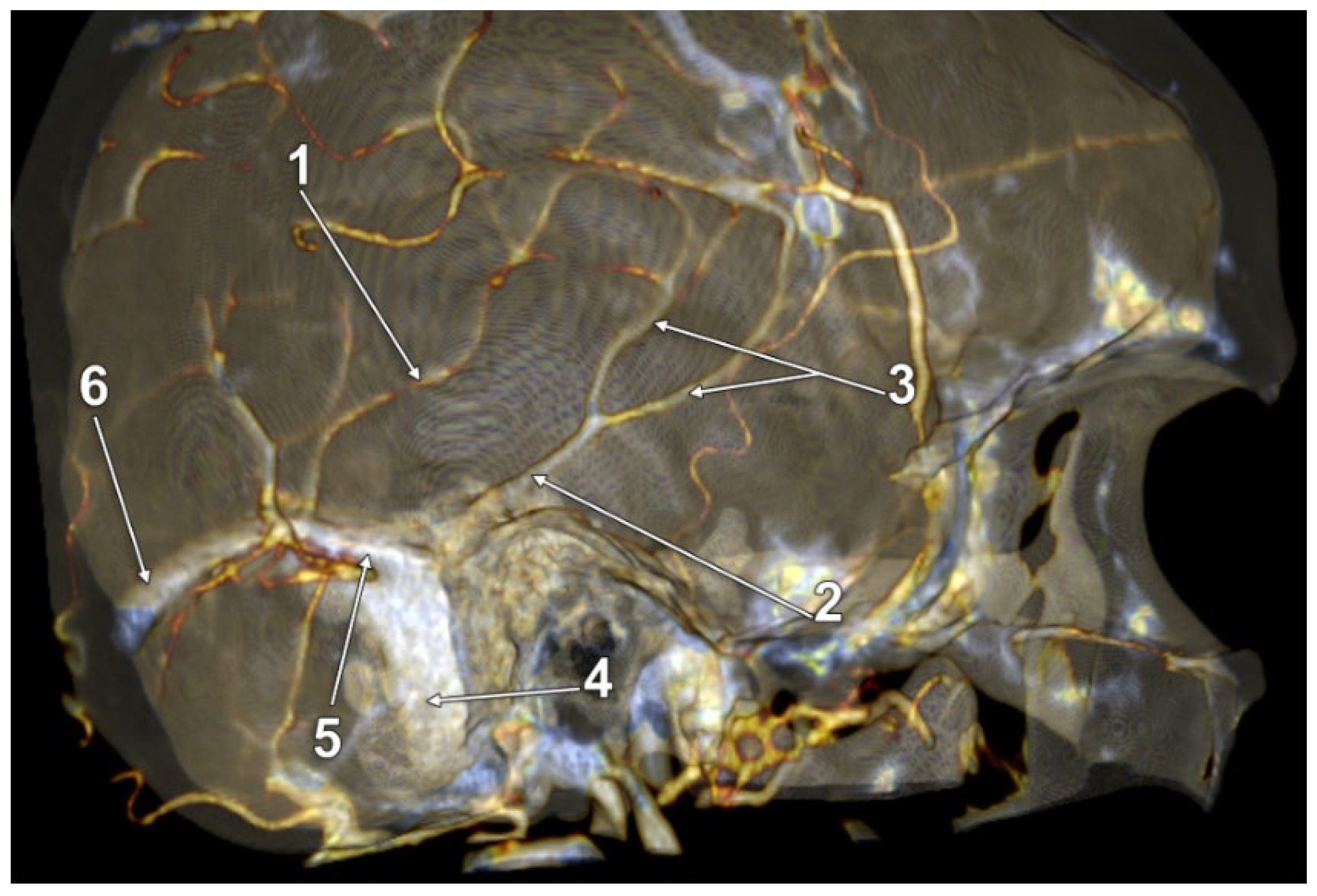
| Variant/Type | Pearson Chi2 Value | p Value (Two-Sided) |
|---|---|---|
| Right LV/0 | 0.5 | 0.479 |
| Left LV/0 | 0.81 | 0.368 |
| Right LV/1 | 0.01 | 0.921 |
| Left LV/1 | 1.303 | 0.254 |
| Right LV/2 | 0 | 1 |
| Left LV/2 | 2.381 | 0.123 |
| Right LV/3 | 2.903 | 0.088 |
| Left LV/3 | 0.230 | 0.631 |
| Gender | Right Side | Left Side |
|---|---|---|
| M | 2 + 2 p (Figure 6) | |
| M | 1 + 3 C | |
| M | 2 + 3 C (Figure 4) | |
| M | 2 + 3 C | |
| M | 2 + 3 C | |
| M | 3 + 3 p (Figure 7) | |
| F | 2 + 2 p | |
| F | 2 + 3 C (Figure 5) | |
| F | 2 + 3 C, 2 p | |
| F | 2 + 3 C | |
| F | 3 + 3 p |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mincă, D.I.; Rusu, M.C.; Rădoi, P.M.; Hostiuc, S.; Toader, C. A New Classification of the Anatomical Variations of Labbé’s Inferior Anastomotic Vein. Tomography 2022, 8, 2182-2192. https://doi.org/10.3390/tomography8050183
Mincă DI, Rusu MC, Rădoi PM, Hostiuc S, Toader C. A New Classification of the Anatomical Variations of Labbé’s Inferior Anastomotic Vein. Tomography. 2022; 8(5):2182-2192. https://doi.org/10.3390/tomography8050183
Chicago/Turabian StyleMincă, Dragoş Ionuţ, Mugurel Constantin Rusu, Petrinel Mugurel Rădoi, Sorin Hostiuc, and Corneliu Toader. 2022. "A New Classification of the Anatomical Variations of Labbé’s Inferior Anastomotic Vein" Tomography 8, no. 5: 2182-2192. https://doi.org/10.3390/tomography8050183
APA StyleMincă, D. I., Rusu, M. C., Rădoi, P. M., Hostiuc, S., & Toader, C. (2022). A New Classification of the Anatomical Variations of Labbé’s Inferior Anastomotic Vein. Tomography, 8(5), 2182-2192. https://doi.org/10.3390/tomography8050183








