Intracranial Hemorrhage from Dural Arteriovenous Fistulas: What Can We Find with CT Angiography?
Abstract
:1. Introduction
2. Materials and Methods
CT Angiography
3. Dimensional Digital Subtraction Angiography (3D DSA)
3.1. Analysis of CT Angiography and of 3D Rotational Angiography
3.2. Statistical Analysis
4. Results
5. Discussion
Limitations of the Study
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gandhi, D.; Chen, J.; Pearl, M.; Huang, J.; Gemmete, J.J.; Kathuria, S. Intracranial dural arteriovenous fistulas: Classification, imaging findings, and treatment. AJNR Am. J. Neuroradiol. 2012, 33, 1007–1013. [Google Scholar] [CrossRef] [Green Version]
- Kim, M.S.; Han, D.H.; Kwon, O.K.; Oh, C.W.; Han, M.H. Clinical characteristics of dural arteriovenous fistula. J. Clin. Neurosci. 2002, 9, 147–155. [Google Scholar] [CrossRef]
- Sarma, D.; ter Brugge, K. Management of intracranial dural arteriovenous shunts in adults. Eur. J. Radiol. 2003, 46, 206–220. [Google Scholar] [CrossRef]
- Cheng, K.M.; Chan, C.M.; Cheung, Y.L. Transvenous embolization of dural carotid-cavernous fistulas by multiple venous routes: A series of 27 cases. Acta Neurochir. 2003, 145, 17–29. [Google Scholar] [CrossRef]
- De Keukeleire, K.; Vanlangenhove, P.; Kalala Okito, J.P.; Hallaert, G.; Van Roost, D.; Defreyne, L. Transarterial embolization with ONYX for treatment of intracranial non-cavernous dural arteriovenous fistula with or without cortical venous reflux. J. Neurointerventional Surg. 2011, 3, 224–228. [Google Scholar] [CrossRef]
- Lawton, M.T.; Sanchez-Mejia, R.O.; Pham, D.; Tan, J.; Halbach, V.V. Tentorial dural arteriovenous fistulae: Operative strategies and microsurgical results for six types. Neurosurgery 2008, 62, 110–124, discussion 124–115. [Google Scholar] [CrossRef]
- van Dijk, J.M.; TerBrugge, K.G.; Willinsky, R.A.; Wallace, M.C. Multiplicity of dural arteriovenous fistulas. J. Neurosurg. 2002, 96, 76–78. [Google Scholar] [CrossRef]
- Cognard, C.; Januel, A.C.; Silva, N.A., Jr.; Tall, P. Endovascular treatment of intracranial dural arteriovenous fistulas with cortical venous drainage: New management using Onyx. AJNR. Am. J. Neuroradiol. 2008, 29, 235–241. [Google Scholar] [CrossRef] [Green Version]
- da Costa, L.B.; Terbrugge, K.; Farb, R.; Wallace, M.C. Surgical disconnection of cortical venous reflux as a treatment for Borden type II dural arteriovenous fistulae. Acta Neurochir. 2007, 149, 1103–1108, discussion 1108. [Google Scholar] [CrossRef]
- Nabors, M.W.; Azzam, C.J.; Albanna, F.J.; Gulya, A.J.; Davis, D.O.; Kobrine, A.I. Delayed postoperative dural arteriovenous malformations. Report of two cases. J. Neurosurg. 1987, 66, 768–772. [Google Scholar] [CrossRef]
- Houser, O.W.; Campbell, J.K.; Campbell, R.J.; Sundt, T.M., Jr. Arteriovenous malformation affecting the transverse dural venous sinus–an acquired lesion. Mayo Clin. Proc. 1979, 54, 651–661. [Google Scholar]
- Brown, R.D., Jr.; Wiebers, D.O.; Nichols, D.A. Intracranial dural arteriovenous fistulae: Angiographic predictors of intracranial hemorrhage and clinical outcome in nonsurgical patients. J. Neurosurg. 1994, 81, 531–538. [Google Scholar] [CrossRef] [PubMed]
- King, W.A.; Martin, N.A. Intracerebral hemorrhage due to dural arteriovenous malformations and fistulae. Neurosurg. Clin. N. Am. 1992, 3, 577–590. [Google Scholar] [CrossRef]
- Malik, G.M.; Pearce, J.E.; Ausman, J.I.; Mehta, B. Dural arteriovenous malformations and intracranial hemorrhage. Neurosurgery 1984, 15, 332–339. [Google Scholar] [CrossRef]
- Piippo, A.; Niemelä, M.; van Popta, J.; Kangasniemi, M.; Rinne, J.; Jääskeläinen, J.E.; Hernesniemi, J. Characteristics and long-term outcome of 251 patients with dural arteriovenous fistulas in a defined population. Clinical article. J. Neurosurg. 2013, 118, 923–934. [Google Scholar] [CrossRef] [PubMed]
- Gallego Ferrero, P.; Marco De Lucas, E.M.; Fernandez-Lobo, V.; Iturralde-Garriz, A.; Garcia Martinez, B.; Navasa Melado, J.M.; Gonzalez Mandly, A.; Santander, E.S. Correlation between CTA findings and angiographic characteristics of intracranial arteriovenous fistulas. In Proceedings of the 2017 European Congress of Radiology, Vienna, Austria, 1–5 March 2017. [Google Scholar] [CrossRef]
- Narvid, J.; Do, H.M.; Blevins, N.H.; Fischbein, N.J. CT angiography as a screening tool for dural arteriovenous fistula in patients with pulsatile tinnitus: Feasibility and test characteristics. AJNR Am. J. Neuroradiol. 2011, 32, 446–453. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saito, A.; Takahashi, N.; Furuno, Y.; Kamiyama, H.; Nishimura, S.; Midorikawa, H.; Nishijima, M. Multiple isolated sinus dural arteriovenous fistulas associated with antithrombin iii deficiency—Case report. Neurol. Med. Chir. 2008, 48, 455–459. [Google Scholar] [CrossRef] [Green Version]
- Satomi, J.; van Dijk, J.M.; Terbrugge, K.G.; Willinsky, R.A.; Wallace, M.C. Benign cranial dural arteriovenous fistulas: Outcome of conservative management based on the natural history of the lesion. J. Neurosurg. 2002, 97, 767–770. [Google Scholar] [CrossRef] [Green Version]
- Levrier, O.; Métellus, P.; Fuentes, S.; Manera, L.; Dufour, H.; Donnet, A.; Grisoli, F.; Bartoli, J.M.; Girard, N. Use of a self-expanding stent with balloon angioplasty in the treatment of dural arteriovenous fistulas involving the transverse and/or sigmoid sinus: Functional and neuroimaging-based outcome in 10 patients. J. Neurosurg. 2006, 104, 254–263. [Google Scholar] [CrossRef]
- Lucas Cde, P.; Zabramski, J.M. Dural arteriovenous fistula of the transverse-sigmoid sinus causing trigeminal neuralgia. Acta Neurochir. 2007, 149, 1249–1253, discussion 1253. [Google Scholar] [CrossRef]
- Agid, R.; Terbrugge, K.; Rodesch, G.; Andersson, T.; Söderman, M. Management strategies for anterior cranial fossa (ethmoidal) dural arteriovenous fistulas with an emphasis on endovascular treatment. J. Neurosurg. 2009, 110, 79–84. [Google Scholar] [CrossRef] [Green Version]
- van Rooij, W.J.; Sluzewski, M.; Beute, G.N. Dural arteriovenous fistulas with cortical venous drainage: Incidence, clinical presentation, and treatment. AJNR Am. J. Neuroradiol. 2007, 28, 651–655. [Google Scholar]
- Kasliwal, M.K.; Moftakhar, R.; O’Toole, J.E.; Lopes, D.K. High cervical spinal subdural hemorrhage as a harbinger of craniocervical arteriovenous fistula: An unusual clinical presentation. Spine J. 2015, 15, e13–e17. [Google Scholar] [CrossRef]
- Ma, C.; Lu, Q.; Shi, W.; Su, Z.; Zhao, Y.; Li, C.; Liu, Z. Diagnosis and treatment of a dural arteriovenous fistula presenting with progressive parkinsonism and dementia: A case report and literature review. Exp. Ther. Med. 2015, 9, 523–526. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hurst, R.W.; Bagley, L.J.; Galetta, S.; Glosser, G.; Lieberman, A.P.; Trojanowski, J.; Sinson, G.; Stecker, M.; Zager, E.; Raps, E.C.; et al. Dementia resulting from dural arteriovenous fistulas: The pathologic findings of venous hypertensive encephalopathy. AJNR Am. J. Neuroradiol. 1998, 19, 1267–1273. [Google Scholar] [PubMed]
- Hirono, N.; Yamadori, A.; Komiyama, M. Dural arteriovenous fistula: A cause of hypoperfusion-induced intellectual impairment. Eur. Neurol. 1993, 33, 5–8. [Google Scholar] [CrossRef] [PubMed]
- Hasumi, T.; Fukushima, T.; Haisa, T.; Yonemitsu, T.; Waragai, M. Focal dural arteriovenous fistula (DAVF) presenting with progressive cognitive impairment including amnesia and alexia. Intern. Med. 2007, 46, 1317–1320. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Noguchi, K.; Kuwayama, N.; Kubo, M.; Kamisaki, Y.; Kameda, K.; Tomizawa, G.; Kawabe, H.; Seto, H. Intracranial dural arteriovenous fistula with retrograde cortical venous drainage: Use of susceptibility-weighted imaging in combination with dynamic susceptibility contrast imaging. AJNR Am. J. Neuroradiol. 2010, 31, 1903–1910. [Google Scholar] [CrossRef] [Green Version]
- Kwon, B.J.; Han, M.H.; Kang, H.S.; Chang, K.H. MR imaging findings of intracranial dural arteriovenous fistulas: Relations with venous drainage patterns. AJNR Am. J. Neuroradiol. 2005, 26, 2500–2507. [Google Scholar]
- Willinsky, R.; Goyal, M.; terBrugge, K.; Montanera, W. Tortuous, engorged pialveins in intracranial dural arteriovenous fistulas: Correlations with presen-tation, location, and MR findings in 122 patients. AJNR Am. J. Neuroradiol. 1999, 20, 1031–1036. [Google Scholar] [PubMed]
- Daniels, D.J.; Vellimana, A.K.; Zipfel, G.J.; Lanzino, G. Intracranial hemorrhage from dural arteriovenous fistulas: Clinical features and outcome. Neurosurg. Focus 2013, 34, E15. [Google Scholar] [CrossRef]
- Pekkola, J.; Kangasniemi, M. Posterior fossa dural arteriovenous fistulas: Diagnosis and follow-up with time-resolved imaging of contrast kinetics (TRICKS) at 1.5T. Acta Radiol. 2011, 52, 442–447. [Google Scholar] [CrossRef] [PubMed]
- De Marco, J.K.; Dillon, W.P.; Halback, V.V.; Tsuruda, J.S. Dural arteriovenous fistulas:evaluation with MR imaging. Radiology 1990, 175, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Van Dijk, J.M.; Willinsky, R.A. Venous congestive encephalopathy related to cra-nial dural arteriovenous fistulas. Neuroimaging Clin. 2003, 13, 55–72. [Google Scholar] [CrossRef]
- Letourneau-Guillon, L.; Krings, T. Simultaneous arteriovenous shunting andvenous congestion identification in dural arteriovenous fistulas usingsusceptibility-weighted imaging: Initial experience. AJNR Am. J. Neuroradiol. 2012, 33, 301–307. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Geibprasert, S.; Pereira, V.; Krings, T.; Jiarakongmun, P.; Toulgoat, F.; Pongpech, S.; Lasjaunias, P. Dural arteriovenous shunts: A new classification of cran-iospinal epidural venous anatomical bases and clinical correlations. Stroke 2008, 39, 2783–2794. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tsai, L.K.; Jeng, J.S.; Liu, H.M.; Wang, H.J.; Yip, P.K. Intracranial dural arteriovenous fistu-las with or without cerebral sinus thrombosis: Analysis of 69 patients. J. Neurol. Neurosurg. Psychiatry 2004, 75, 1639–1641. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alatakis, S.; Koulouris, G.; Stuckey, S. CT-demonstrated transcalvarial chan-nels diagnostic of dural arteriovenous fistula. AJNR Am. J. Neuroradiol. 2005, 26, 2393–2396. [Google Scholar]
- Meckel, S.; Maier, M.; Ruiz, D.S.; Yilmaz, H.; Scheffler, K.; Radue, E.W.; Wetzel, S.G. MR angiography of dural arteriovenous fistulas: Diagnosis and follow-up after treatment using a time-resolved 3D contrast-enhanced technique. AJNR Am. J. Neuroradiol. 2007, 28, 877–884. [Google Scholar]
- Jang, J.; Schmitt, P.; Kim, B.Y.; Choi, H.S.; Jung, S.L.; Ahn, K.J.; Kim, I.; Paek, M.; Kim, B.S. Noncontrast- enhanced 4D MR angiography with STAR spin labeling and variable flip angle sampling: A feasibility study for the assessment of Dural Arteriovenous Fistula. Neuroradiology 2014, 56, 305–314. [Google Scholar] [CrossRef]
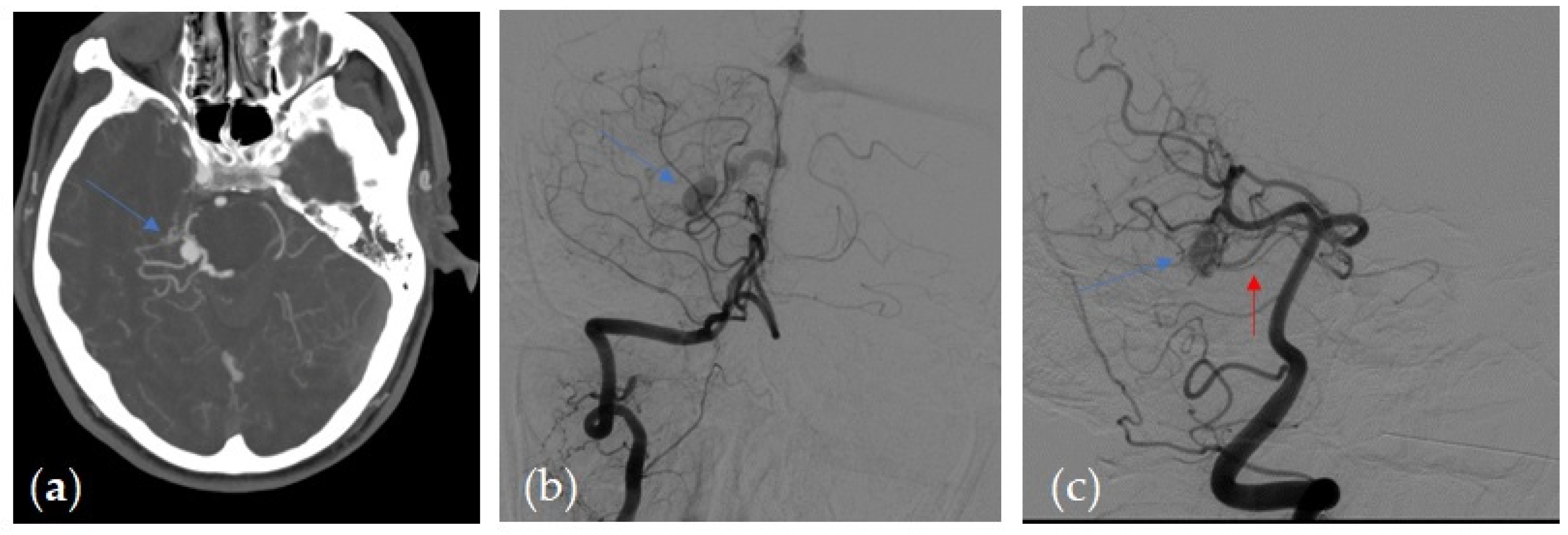
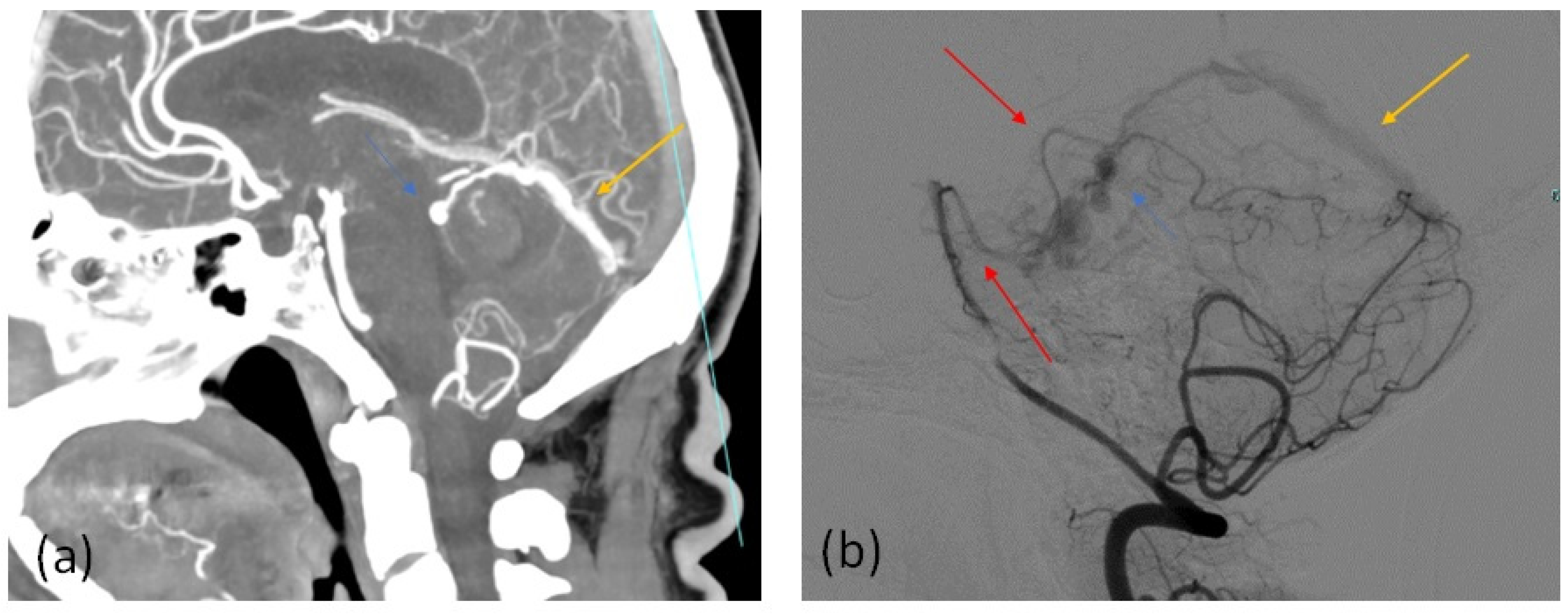
| Parameter | Value |
|---|---|
| No. of patients | 26 |
| Mean age in years | 55 |
| Male | 20 |
| Presenting symptom/sign: | |
| 19 |
| 15 |
| 5 |
| DAVF grade | |
| 26 |
| 10 |
| Location of fistula: | |
| 15 |
| 5 |
| 6 |
| Hemorrhage: | |
| 20 |
| 12 |
| 5 |
| Cognard Type | No. of Patients |
|---|---|
| I | 0 |
| IIa | 0 |
| IIa + b | 5 |
| III | 11 |
| IV | 10 |
| V | 0 |
| DAVF+ | DAVF− | |
|---|---|---|
| CTA+ | 16 | 0 |
| CTA− | 10 | 100 |
| Brain Angio CT Positive Findings | No. of Patients |
|---|---|
| Asymmetric and/or dilated feeding arteries | 7 |
| Numerous and engorged cortical veins | 15 |
| Sinus findings: | |
| 8 |
| 10 |
| 4 |
| Transcalvarial channels | 4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Negro, A.; Somma, F.; Piscitelli, V.; La Tessa, G.M.E.; Sicignano, C.; Fasano, F.; Tamburrini, S.; Vargas, O.; Pace, G.; Iannuzzi, M.; et al. Intracranial Hemorrhage from Dural Arteriovenous Fistulas: What Can We Find with CT Angiography? Tomography 2021, 7, 804-814. https://doi.org/10.3390/tomography7040068
Negro A, Somma F, Piscitelli V, La Tessa GME, Sicignano C, Fasano F, Tamburrini S, Vargas O, Pace G, Iannuzzi M, et al. Intracranial Hemorrhage from Dural Arteriovenous Fistulas: What Can We Find with CT Angiography? Tomography. 2021; 7(4):804-814. https://doi.org/10.3390/tomography7040068
Chicago/Turabian StyleNegro, Alberto, Francesco Somma, Valeria Piscitelli, Giuseppe Maria Ernesto La Tessa, Carmine Sicignano, Fabrizio Fasano, Stefania Tamburrini, Ottavia Vargas, Gianvito Pace, Michele Iannuzzi, and et al. 2021. "Intracranial Hemorrhage from Dural Arteriovenous Fistulas: What Can We Find with CT Angiography?" Tomography 7, no. 4: 804-814. https://doi.org/10.3390/tomography7040068
APA StyleNegro, A., Somma, F., Piscitelli, V., La Tessa, G. M. E., Sicignano, C., Fasano, F., Tamburrini, S., Vargas, O., Pace, G., Iannuzzi, M., Villa, A., Della Gatta, L., Chiaramonte, C., Caranci, F., Tortora, F., & D’Agostino, V. (2021). Intracranial Hemorrhage from Dural Arteriovenous Fistulas: What Can We Find with CT Angiography? Tomography, 7(4), 804-814. https://doi.org/10.3390/tomography7040068








