Imaging of Biliary Involvement in Sarcoidosis: Computed Tomography, Magnetic Resonance Cholangiopancreatography, and Gadolinium Ethoxybenzyl Diethylenetriamine Pentaacetic Acid-Enhanced Magnetic Resonance Imaging Findings
Abstract
1. Introduction
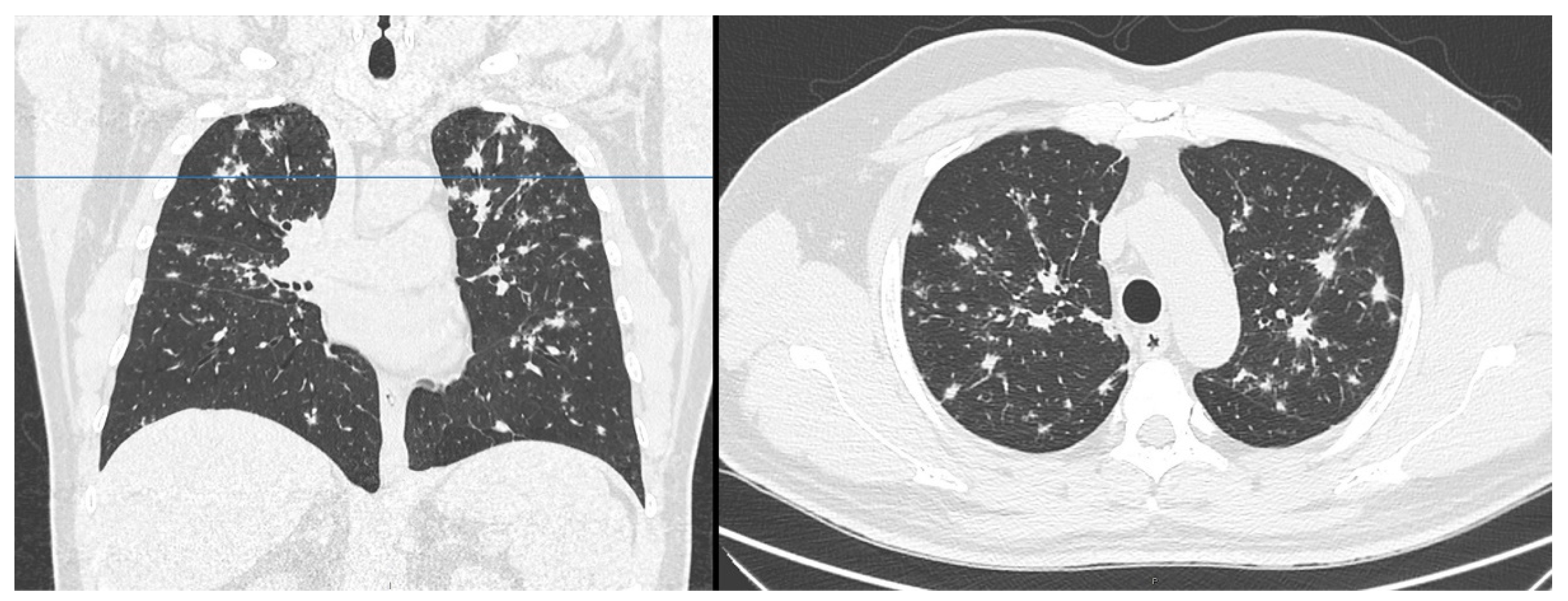
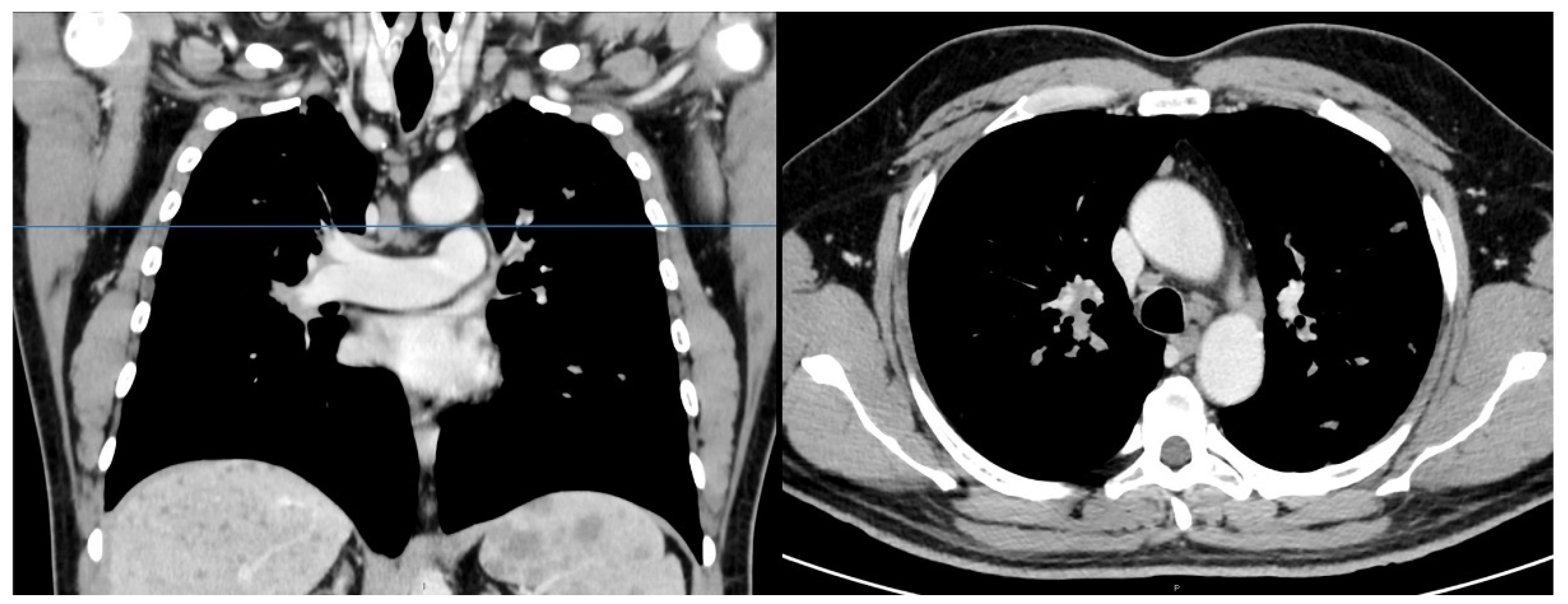
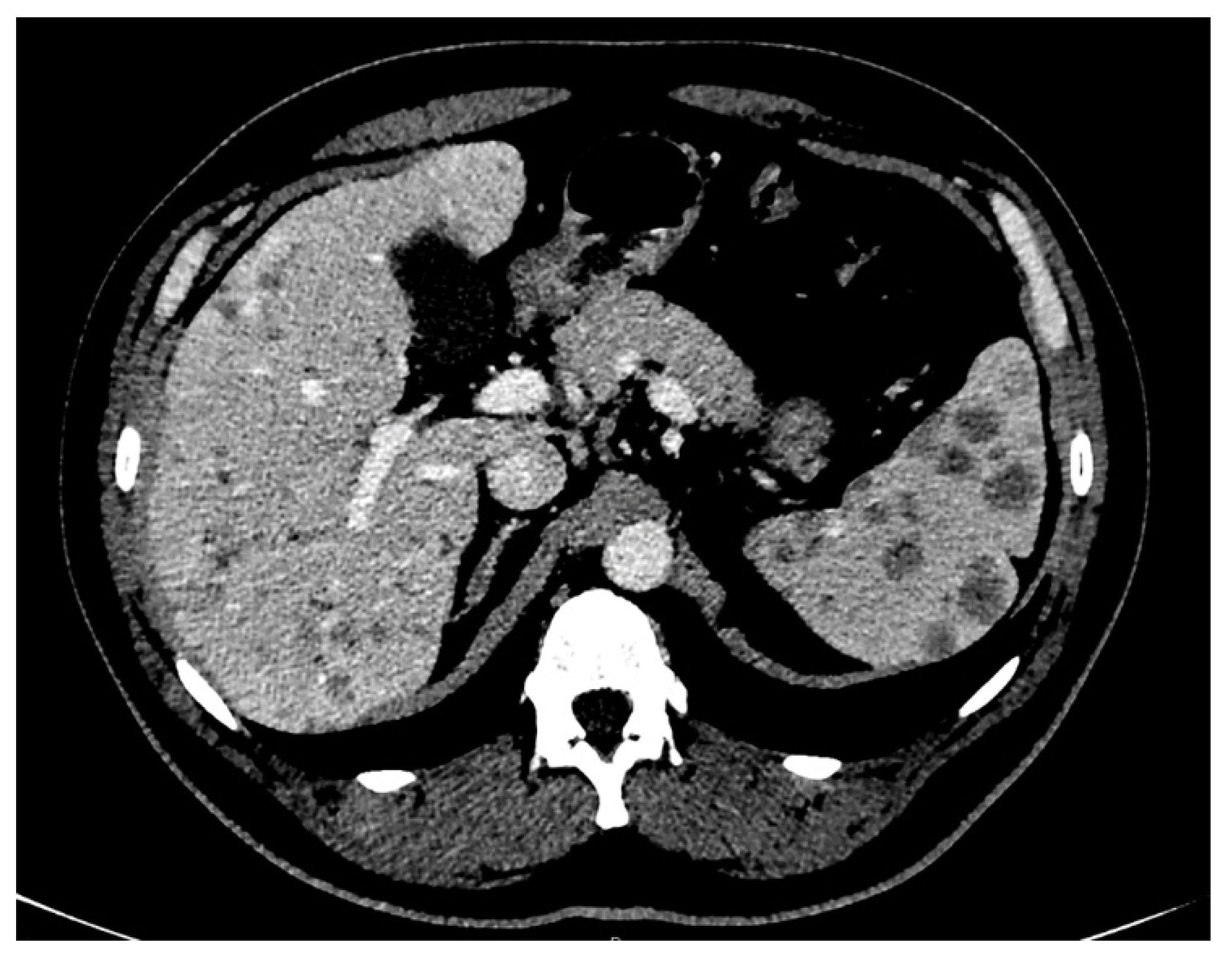
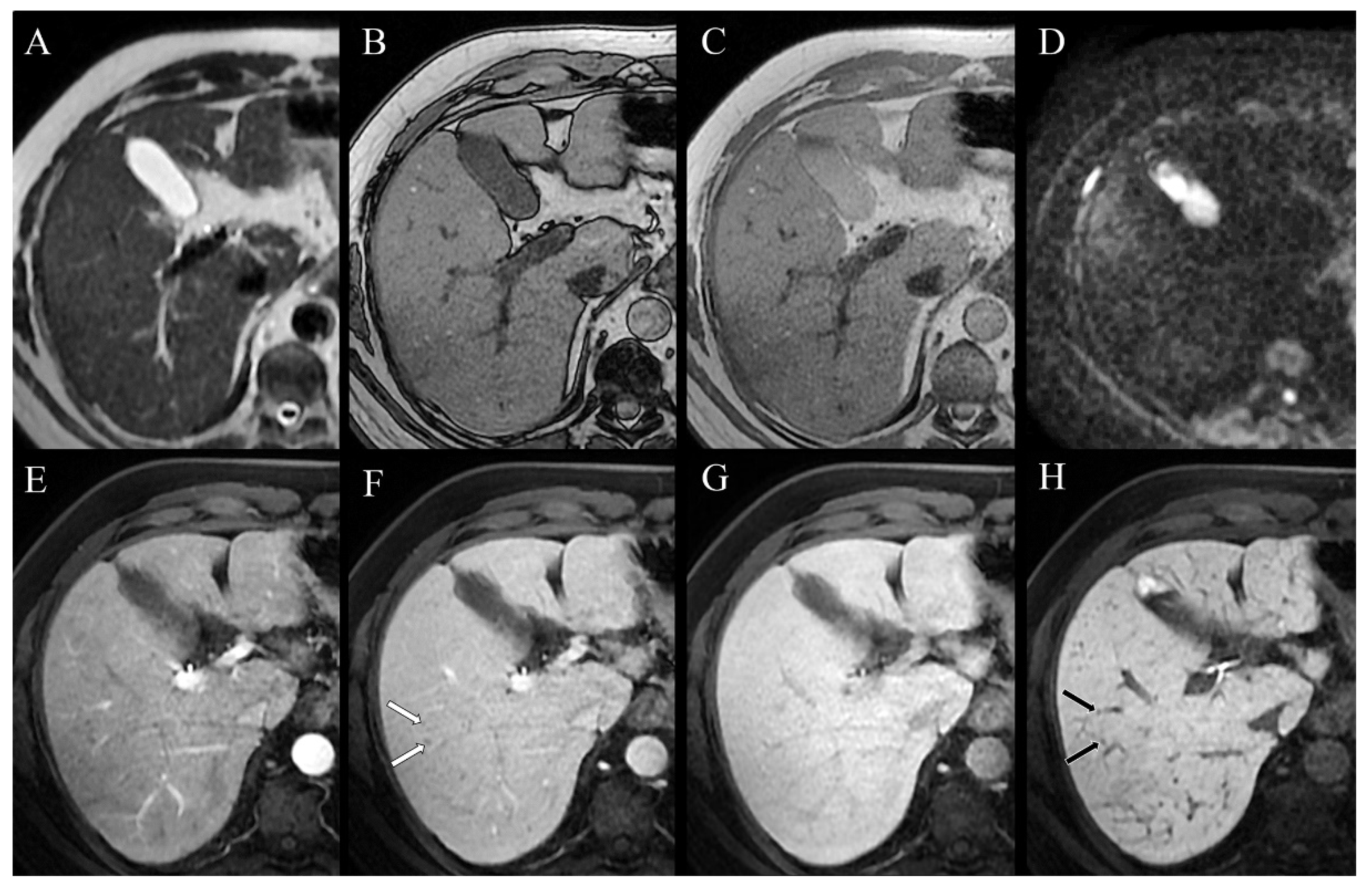
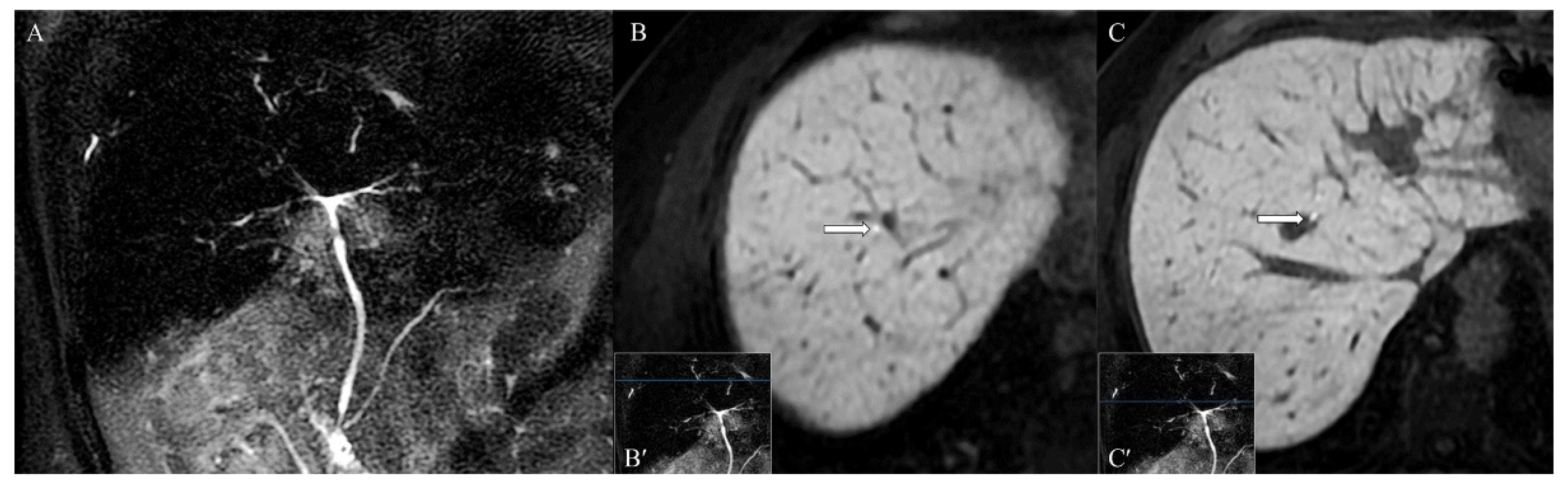
2. Case Presentation
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Arkema, E.V.; Cozier, Y.C. Epidemiology of sarcoidosis: Current findings and future directions. Ther. Adv. Chronic Dis. 2018, 9, 227–240. [Google Scholar] [CrossRef]
- Ungprasert, P.; Crowson, C.S.; Matteson, E.L. Influence of Gender on Epidemiology and Clinical Manifestations of Sarcoidosis: A Population-Based Retrospective Cohort Study 1976–2013. Lung 2017, 195, 87–91. [Google Scholar] [CrossRef]
- Baughman, R.P.; Field, S.; Costabel, U.; Crystal, R.G.; Culver, D.A.; Drent, M.; Judson, M.A.; Wolff, G. Sarcoidosis in America. Analysis Based on Health Care Use. Ann. Am. Thorac. Soc. 2016, 13, 1244–1252. [Google Scholar] [CrossRef]
- Arkema, E.V.; Grunewald, J.; Kullberg, S.; Eklund, A.; Askling, J. Sarcoidosis incidence and prevalence: A nationwide register-based assessment in Sweden. Eur. Respir. J. 2016, 48, 1690–1699. [Google Scholar] [CrossRef]
- Duchemann, B.; Annesi-Maesano, I.; Jacobe de Naurois, C.; Sanyal, S.; Brillet, P.Y.; Brauner, M.; Kambouchner, M.; Huynh, S.; Naccache, J.M.; Borie, R.; et al. Prevalence and incidence of interstitial lung diseases in a multi-ethnic county of Greater Paris. Eur. Respir. J. 2017, 50, 1602419. [Google Scholar] [CrossRef] [PubMed]
- Yoon, H.Y.; Kim, H.M.; Kim, Y.J.; Song, J.W. Prevalence and incidence of sarcoidosis in Korea: A nationwide population-based study. Respir. Res. 2018, 19, 158. [Google Scholar] [CrossRef]
- Gerke, A.K.; Hunninghake, G. The immunology of sarcoidosis. Clin. Chest Med. 2008, 29, 379–390. [Google Scholar] [CrossRef] [PubMed]
- Müller-Quernheim, J.; Schürmann, M.; Hofmann, S.; Gaede, K.I.; Fischer, A.; Prasse, A.; Zissel, G.; Schreiber, S. Genetics of sarcoidosis. Clin. Chest Med. 2008, 29, 391–414. [Google Scholar] [CrossRef] [PubMed]
- Newman, L.S.; Rose, C.S.; Bresnitz, E.A.; Rossman, M.D.; Barnard, J.; Frederick, M.; Terrin, M.L.; Weinberger, S.E.; Moller, D.R.; McLennan, G.; et al. A case control etiologic study of sarcoidosis: Environmental and occupational risk factors. Am. J. Respir. Crit. Care Med. 2004, 170, 1324–1330. [Google Scholar] [CrossRef]
- Baughman, R.P.; Teirstein, A.S.; Judson, M.A.; Rossman, M.D.; Yeager, H., Jr.; Bresnitz, E.A.; DePalo, L.; Hunninghake, G.; Iannuzzi, M.C.; Johns, C.J.; et al. Clinical characteristics of patients in a case control study of sarcoidosis. Am. J. Respir. Crit. Care Med. 2001, 164, 1885–1889. [Google Scholar] [CrossRef] [PubMed]
- Polverino, F.; Balestro, E.; Spagnolo, P. Clinical Presentations, Pathogenesis, and Therapy of Sarcoidosis: State of the Art. J. Clin. Med. 2020, 9, 2363. [Google Scholar] [CrossRef] [PubMed]
- De Cinque, A.; Corcioni, B.; Rossi, M.S.; Franceschelli, A.; Colombo, F.; Golfieri, R.; Renzulli, M.; Gaudiano, C. Case Report: Testicular Sarcoidosis: The Diagnostic Role of Contrast-Enhanced Ultrasound and Review of the Literature. Front. Med. 2021, 7, 610384. [Google Scholar] [CrossRef] [PubMed]
- Jain, R.; Yadav, D.; Puranik, N.; Guleria, R.; Jin, J.O. Sarcoidosis: Causes, Diagnosis, Clinical Features, and Treatments. J. Clin. Med. 2020, 9, 1081. [Google Scholar] [CrossRef]
- Rybicki, B.A.; Major, M.; Popovich, J., Jr.; Maliarik, M.J.; Iannuzzi, M.C. Racial differences in sarcoidosis incidence: A 5-year study in a health maintenance organization. Am. J. Epidemiol. 1997, 145, 234–241. [Google Scholar] [CrossRef]
- Diagnosis and Management of Sarcoidosis. Available online: https://www.aafp.org/afp/2016/0515/p840.html (accessed on 5 October 2019).
- Kumar, M.; Herrera, J.L. Sarcoidosis and the Liver. Clin. Liver Dis. 2019, 23, 331–343. [Google Scholar] [CrossRef]
- Dulai, P.S.; Rothstein, R.I. Disseminated sarcoidosis presenting as granulomatous gastritis: A clinical review of the gastrointestinal and hepatic manifestations of sarcoidosis. J. Clin. Gastroenterol. 2012, 46, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Nolan, J.P.; Klatskin, G. The fever of sarcoidosis. Ann. Intern. Med. 1964, 61, 455–461. [Google Scholar] [CrossRef]
- Tadros, M.; Forouhar, F.; Wu, G.Y. Hepatic Sarcoidosis. J. Clin. Transl. Hepatol. 2013, 1, 87–93. [Google Scholar] [CrossRef]
- Kennedy, P.T.; Zakaria, N.; Modawi, S.B.; Papadopoulou, A.M.; Murray-Lyon, I.; du Bois, R.M.; Jervoise, N.; Andreyev, H.; Devlin, J. Natural history of hepatic sarcoidosis and its response to treatment. Eur. J. Gastroenterol. Hepatol. 2006, 18, 721–726. [Google Scholar] [CrossRef]
- Karaosmanoğlu, A.D.; Onur, M.R.; Saini, S.; Taberi, A.; Karcaaltincaba, M. Imaging of hepatobiliary involvement in sarcoidosis. Abdom. Imaging 2015, 40, 3330–3337. [Google Scholar] [CrossRef]
- Jung, G.; Brill, N.; Poll, L.W.; Koch, J.A.; Wettstein, M. MRI of hepatic sarcoidosis: Large confluent lesions mimicking malignancy. AJR Am. J. Roentgenol. 2004, 183, 171–173. [Google Scholar] [CrossRef]
- Warshauer, D.M.; Lee, J.K. Imaging manifestations of abdominal sarcoidosis. AJR Am. J. Roentgenol. 2004, 182, 15–28. [Google Scholar] [CrossRef]
- Warshauer, D.M.; Dumbleton, S.A.; Molina, P.L.; Yankaskas, B.C.; Parker, L.A.; Woosley, J.T. Abdominal CT findings in sarcoidosis: Radiologic and clinical correlation. Radiology 1994, 192, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Fetzer, D.T.; Rees, M.A.; Dasyam, A.K.; Tublin, M.E. Hepatic sarcoidosis in patients presenting with liver dysfunction: Imaging appearance, pathological correlation and disease evolution. Eur. Radiol. 2016, 26, 3129–3137. [Google Scholar] [CrossRef] [PubMed]
- Warshauer, D.M.; Semelka, R.C.; Ascher, S.M. Nodular sarcoidosis of the liver and spleen: Appearance on MR images. J. Magn. Reson. Imaging 1994, 4, 553–557. [Google Scholar] [CrossRef]
- Wada, N.; Nouso, K.; Kariyama, K.; Wakuta, A.; Kishida, M.; Nishimura, M.; Higashi, T. Liver Sarcoidosis with Unique MRI Images Using Gadolinium Ethoxybenzyl Diethylenetriamine Pentaacetic Acid. Acta Med. Okayama 2015, 69, 307–311. [Google Scholar] [CrossRef] [PubMed]
- Gezer, N.S.; Başara, I.; Altay, C.; Harman, M.; Rocher, L.; Karabulut, N.; Seçil, M. Abdominal sarcoidosis: Cross-sectional imaging findings. Diagn. Interv. Radiol. 2015, 21, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Kessler, A.; Mitchell, D.G.; Israel, H.L.; Goldberg, B.B. Hepatic and splenic sarcoidosis: Ultrasound and MR imaging. Abdom. Imaging 1993, 18, 159–163. [Google Scholar] [CrossRef]
- Dourakis, S.P.; Cokkinos, D.D.; Soultati, A.S.; Alexopoulou, A.; Nezi, V.; Archimandritis, A.J. A case of liver sarcoidosis mimicking cirrhosis. Clin. Imaging 2007, 31, 47–49. [Google Scholar] [CrossRef]
- Farooq, P.D.; Potosky, D.R. The Klatskin Tumor That Wasn’t: An Unusual Presentation of Sarcoidosis. ACG Case Rep. J. 2016, 3, e141. [Google Scholar] [CrossRef]
- Tombazzi, C.; Waters, B.; Ismail, M.K.; Sylvestre, P.B.; Martinez-Hernandez, A.; Riely, C.A. Sarcoidosis mimicking primary sclerosing cholangitis requiring liver transplantation. Ann. Hepatol. 2008, 7, 83–86. [Google Scholar] [CrossRef] [PubMed]
- Nakanuma, Y.; Ohta, G.; Yamazaki, Y.; Doishita, K. Intrahepatic bile duct destruction in a patient with sarcoidosis and chronic intrahepatic cholestasis. Acta Pathol. Jpn. 1979, 29, 211–219. [Google Scholar] [CrossRef]
- Ilan, Y.; Rappaport, I.; Feigin, R.; Ben-Chetrit, E. Primary sclerosing cholangitis in sarcoidosis. J. Clin. Gastroenterol. 1993, 16, 326–328. [Google Scholar] [CrossRef]
- Maambo, E.; Brett, A.S.; Vasudeva, R.; Burns, R.G. Hepatobiliary sarcoidosis presenting as sclerosing cholangitis: Long-term follow-up. Dig. Dis. Sci. 2007, 52, 3363–3365. [Google Scholar] [CrossRef] [PubMed]
- Bloom, R.; Sybert, A.; Mascatello, V.J. Granulomatous biliary tract obstruction due to sarcoidosis. Report of a case and review of the literature. Am. Rev. Respir. Dis. 1978, 117, 783–787. [Google Scholar] [CrossRef] [PubMed]
- Baughman, R.P. Sarcoidosis. Usual and unusual manifestations. Chest 1988, 94, 165–170. [Google Scholar] [CrossRef]
- Kusielewicz, D.; Duchatelle, V.; Valeyre, D.; Battesti, J.P.; Vissuzaine, C.; Coste, T.; Rautureau, J. Ictère obstructif par sténose granulomateuse des voies biliaires extra-hépatiques au cours d’une sarcoïdose [Obstructive jaundice caused by granulomatous stenosis of the extrahepatic bile ducts in sarcoidosis]. Gastroenterol. Clin. Biol. 1988, 2, 664–667. [Google Scholar]
- Albu, E.; Saraiya, R.J.; Carvajal, S.J.; Sehonanda, A.; Balar, N.; Gerst, P.H. Sarcoidosis presenting as obstructive jaundice. Am. Surg. 1995, 61, 516–517. [Google Scholar] [PubMed]
- Rezeig, M.A.; Fashir, B.M. Biliary tract obstruction due to sarcoidosis: A case report. Am. J. Gastroenterol. 1997, 92, 527–528. [Google Scholar] [PubMed]
- Sotiropoulos, G.C.; Dahmen, U.; Dirsch, O.; Antoch, G.; Freudenberg, L.; Brokalaki, E.I.; Treichel, U.; Paul, A.; Broelsch, C.E. Biliary tract obstruction due to sarcoidosis. Endoscopy 2005, 37, 1262. [Google Scholar] [CrossRef][Green Version]
- Pungpapong, S.; Steers, J.L.; Wallace, M.B.; Krishna, M.; Keaveny, A.P. Hepatobiliary sarcoidosis mimicking Klatskin’s cholangiocarcinoma. Gastrointest. Endosc. 2006, 64, 124–125. [Google Scholar] [CrossRef] [PubMed]
- Buxbaum, J.; Papademetriou, M.; Klipfel, N.; Selby, R.; Fong, T.L.; Sharma, O. Biliary sarcoidosis: Early diagnosis minimizes the need for surgery. Am. J. Respir. Crit. Care Med. 2013, 187, 556–559. [Google Scholar] [CrossRef] [PubMed]
- Jebran, A.F.; Schmidt, W.E.; Kahraman, A.; Canbay, A.; Bulut, K. Sarcoidosis of the Intra- and Extrahepatic Bile Ducts with Concomitant Cholangitis in a Patient with Ulcerative Colitis. Case Rep. Gastroenterol. 2019, 13, 153–158. [Google Scholar] [CrossRef]
- Alam, I.; Levenson, S.D.; Ferrell, L.D.; Bass, N.M. Diffuse intrahepatic biliary strictures in sarcoidosis resembling sclerosing cholangitis. Case report and review of the literature. Dig. Dis. Sci. 1997, 42, 1295–1301. [Google Scholar] [CrossRef] [PubMed]
- Golfieri, R.; Garzillo, G.; Ascanio, S.; Renzulli, M. Focal lesions in the cirrhotic liver: Their pivotal role in gadoxetic acid-enhanced MRI and recognition by the Western guidelines. Dig. Dis. 2014, 32, 696–704. [Google Scholar] [CrossRef]
- Bonifazi, M.; Bravi, F.; Gasparini, S.; La Vecchia, C.; Gabrielli, A.; Wells, A.U.; Renzoni, E.A. Sarcoidosis and cancer risk: Systematic review and meta-analysis of observational studies. Chest 2015, 147, 778–791. [Google Scholar] [CrossRef]
- Masuda, K.; Takenaga, S.; Morikawa, K.; Kano, A.; Ojiri, H. Hepatic sarcoidosis with atypical radiological manifestations: A case report. Radiol. Case Rep. 2018, 13, 936–939. [Google Scholar] [CrossRef]
- Inoue, M.; Chiba, T.; Zen, Y.; Yokota, H.; Kanda, T.; Ogasawara, S.; Sugiyama, H.; Arai, M.; Kanai, F.; Ogawa, M.; et al. Hepatic sarcoidosis with an increased serum level of immunoglobulin G4. Intern. Med. 2012, 51, 3095–3098. [Google Scholar] [CrossRef]
- Yamada, S.; Mine, S.; Fujisaki, T.; Ohnari, N.; Eto, S.; Tanaka, Y. Hepatic sarcoidosis associated with chronic hepatitis C. J. Gastroenterol. 2002, 37, 564–570. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Renzulli, M.; Casavola, M.; Foà, A.; Pizzi, C.; Golfieri, R. Imaging of Biliary Involvement in Sarcoidosis: Computed Tomography, Magnetic Resonance Cholangiopancreatography, and Gadolinium Ethoxybenzyl Diethylenetriamine Pentaacetic Acid-Enhanced Magnetic Resonance Imaging Findings. Tomography 2021, 7, 783-791. https://doi.org/10.3390/tomography7040065
Renzulli M, Casavola M, Foà A, Pizzi C, Golfieri R. Imaging of Biliary Involvement in Sarcoidosis: Computed Tomography, Magnetic Resonance Cholangiopancreatography, and Gadolinium Ethoxybenzyl Diethylenetriamine Pentaacetic Acid-Enhanced Magnetic Resonance Imaging Findings. Tomography. 2021; 7(4):783-791. https://doi.org/10.3390/tomography7040065
Chicago/Turabian StyleRenzulli, Matteo, Mario Casavola, Alberto Foà, Carmine Pizzi, and Rita Golfieri. 2021. "Imaging of Biliary Involvement in Sarcoidosis: Computed Tomography, Magnetic Resonance Cholangiopancreatography, and Gadolinium Ethoxybenzyl Diethylenetriamine Pentaacetic Acid-Enhanced Magnetic Resonance Imaging Findings" Tomography 7, no. 4: 783-791. https://doi.org/10.3390/tomography7040065
APA StyleRenzulli, M., Casavola, M., Foà, A., Pizzi, C., & Golfieri, R. (2021). Imaging of Biliary Involvement in Sarcoidosis: Computed Tomography, Magnetic Resonance Cholangiopancreatography, and Gadolinium Ethoxybenzyl Diethylenetriamine Pentaacetic Acid-Enhanced Magnetic Resonance Imaging Findings. Tomography, 7(4), 783-791. https://doi.org/10.3390/tomography7040065







