Implementation of a Technique Based on Hounsfield Units and Hounsfield Density to Determine Kidney Stone Composition
Abstract
:1. Introduction
2. Materials and Methods
2.1. Selection of the Study Participants
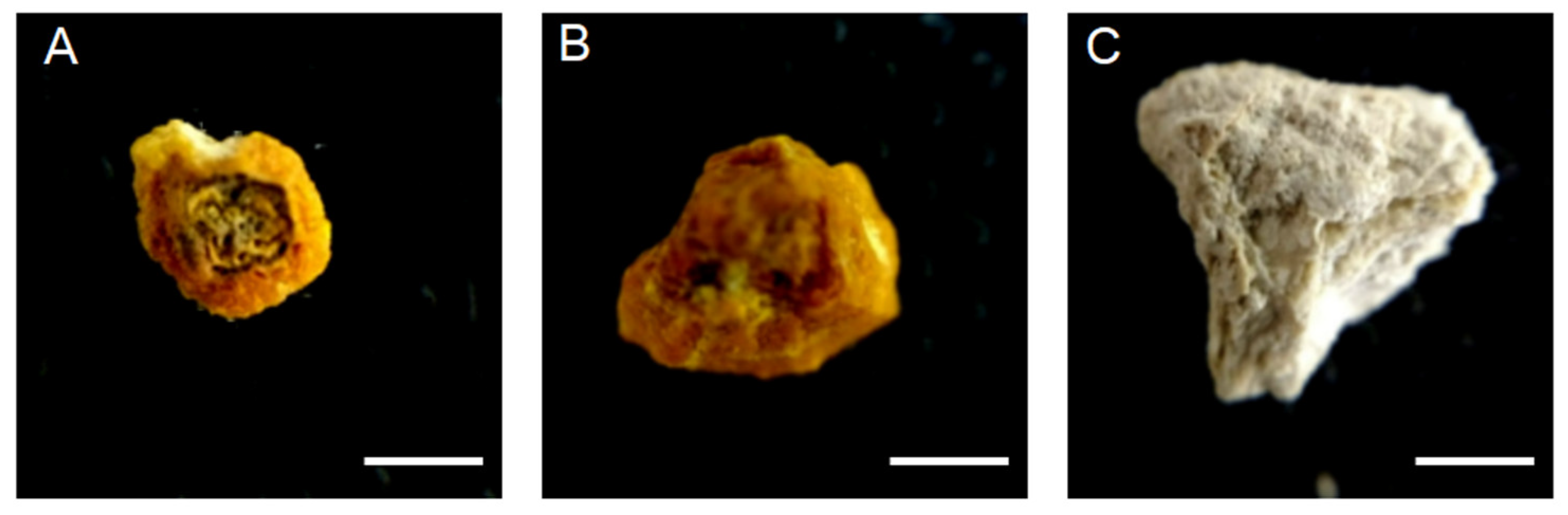
2.2. Chemical Determination of the Renal Stone
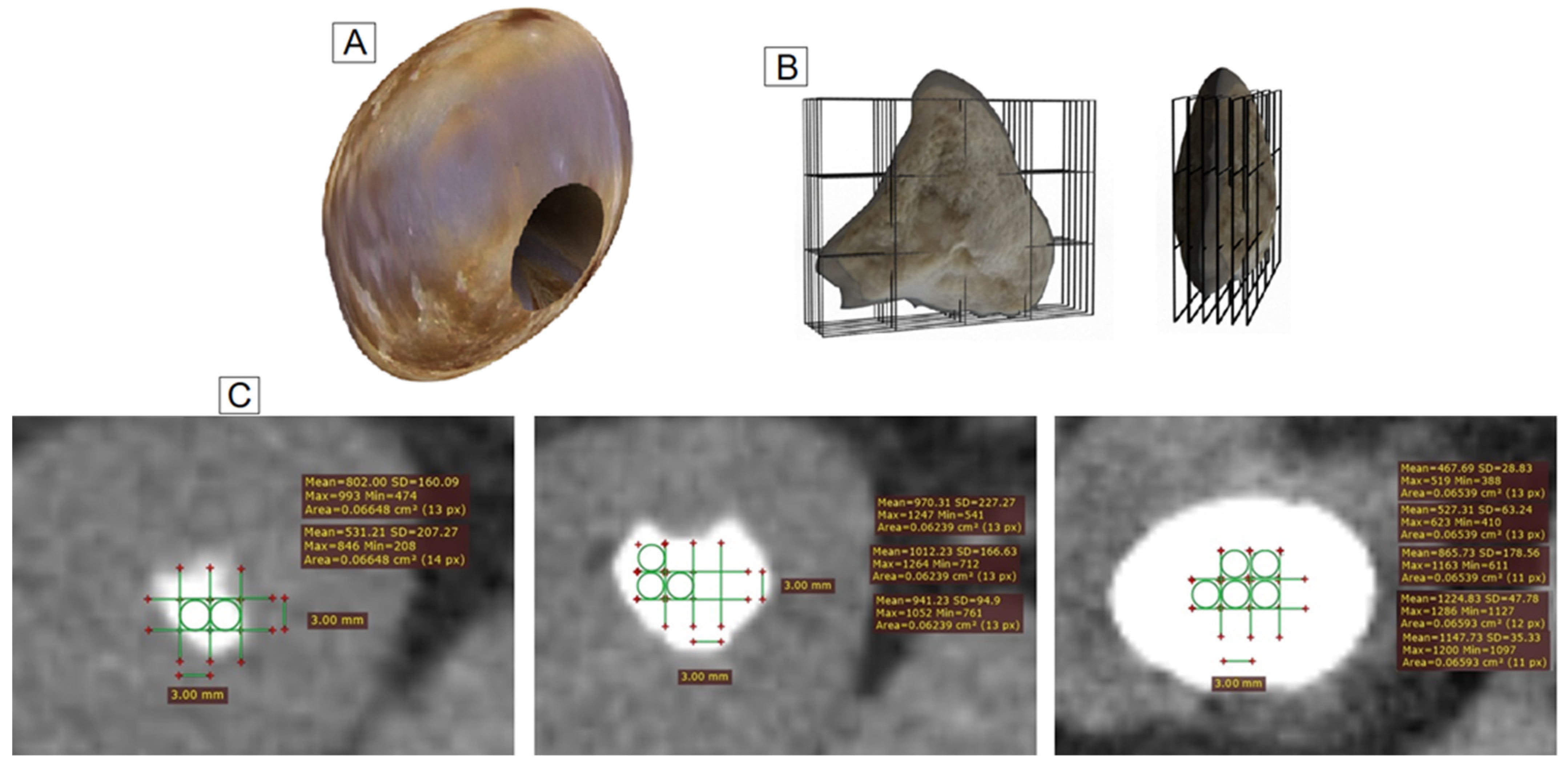
2.3. Identification of the Samples by Tomography
2.4. Statistical Analyses
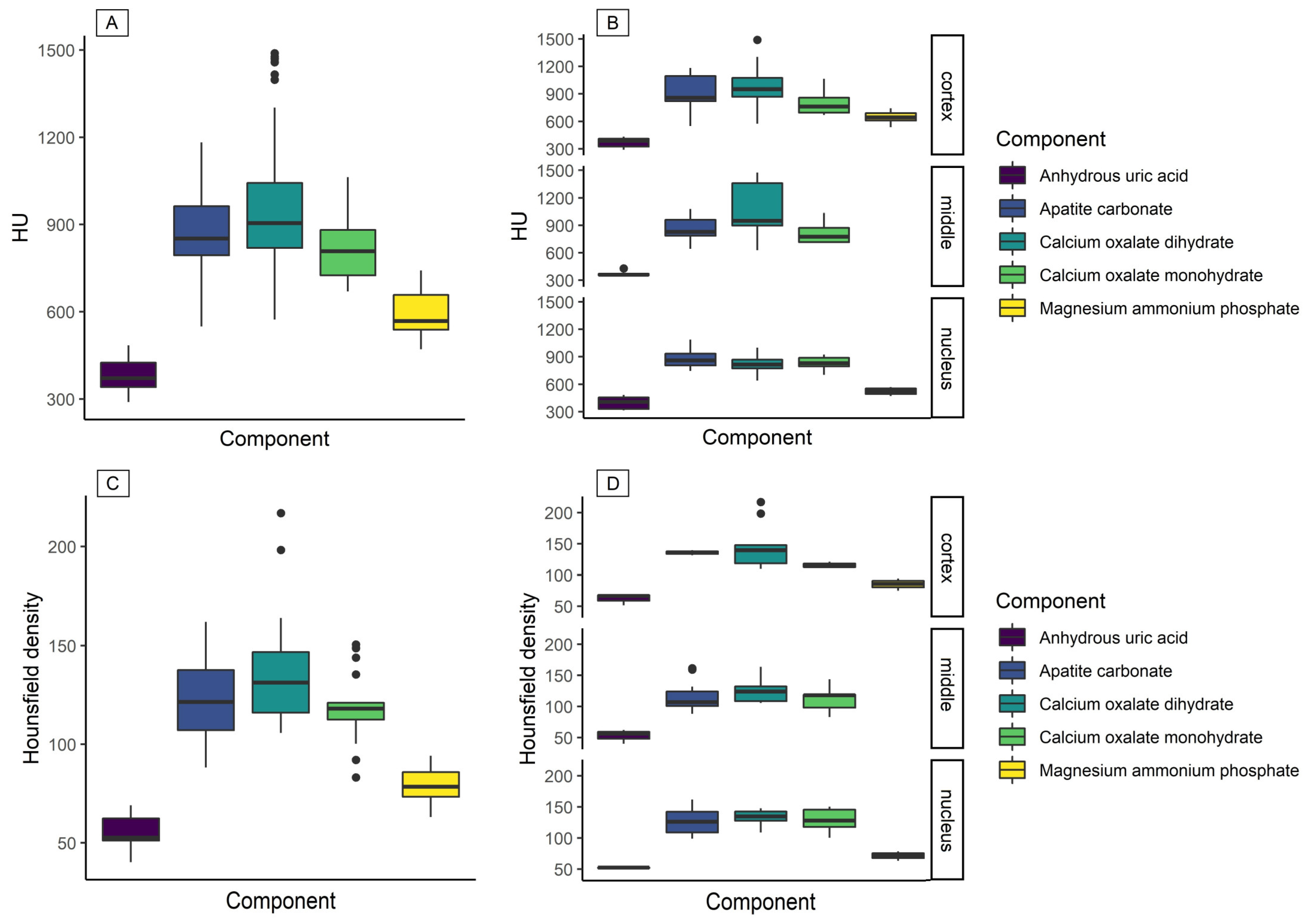
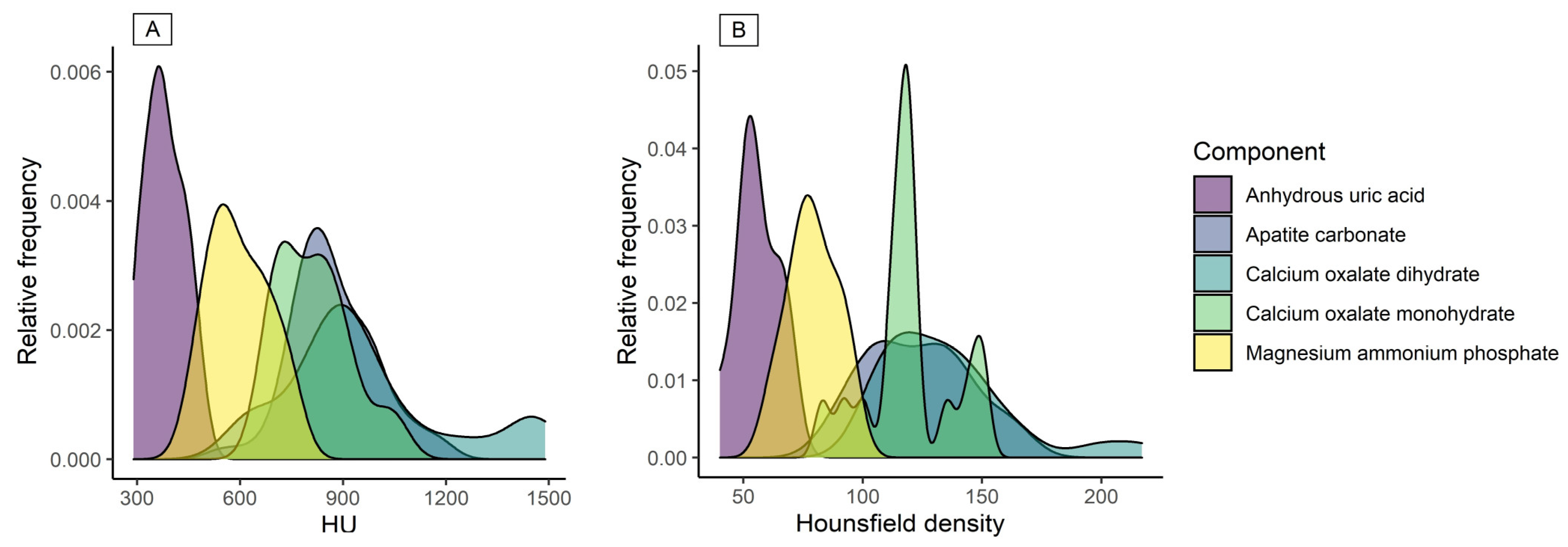
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Verkoelen, C.F.; Schepers, M.S. Changing concepts in the aetiology of renal stones. Curr. Opin. Urol. 2000, 10, 539–544. [Google Scholar] [CrossRef] [PubMed]
- Medina-Escobedo, M.; Zaidi, M.; Real-de Leon, E.; Orozco-Rivadeneyra, S. Urolithiasis prevalence and risk factors in Yucatan, Mexico. Salud Publica Mex. 2002, 44, 541–545. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Türk, C.; Knoll, T.; Petrik, A.; Sarica, K.; Skolarikos, A.; Straub, M.; Seitz, C. EAU guidelines on urolithiasis. Eur. Urol. 2014, 89, 1–32. [Google Scholar]
- Villanueva-Jorge, S.; Medina-Escobedo, M.; Díaz-Arce, A.; Soberanis-Martin, G. Excreción de oxalatos y citratos en pacientes adultos con litiasis urinaria. Bioquimia 2007, 32, 137–143. [Google Scholar]
- Assi, Z.; Platt, J.F.; Francis, I.R.; Cohan, R.H.; Korobkin, M. Sensitivity of CT Scout Radiography and Abdominal Radiography for Revealing Ureteral Calculi on Helical CT. Am. J. Roentgenol. 2000, 175, 333–337. [Google Scholar] [CrossRef] [PubMed]
- García, R.E.; Trejo, G.V. Lo que no debe faltar en una interpretación de…“litiasis renal”. Ann. Radiol. Mex. 2006, 5, 184–187. [Google Scholar]
- Cheng, P.M.; Moin, P.; Dunn, M.D.; Boswell, W.D.; Duddalwar, V.A. What the Radiologist Needs to Know About Urolithiasis: Part 2--CT Findings, Reporting, and Treatment. Am. J. Roentgenol. 2012, 198, W548–W554. [Google Scholar] [CrossRef] [PubMed]
- Shahnani, P.S.; Karami, M.; Astane, B.; Janghorbani, M. The comparative survey of Hounsfield units of stone composition in urolithiasis patients. J. Res. Med. Sci. 2014, 19, 650–653. [Google Scholar] [PubMed]
- Ito, H.; Kawahara, T.; Terao, H.; Ogawa, T.; Yao, M.; Kubota, Y.; Matsuzaki, J. Predictive Value of Attenuation Coefficients Measured as Hounsfield Units on Noncontrast Computed Tomography During Flexible Ureteroscopy with Holmium Laser Lithotripsy: A Single-Center Experience. J. Endourol. 2012, 26, 1125–1130. [Google Scholar] [CrossRef] [PubMed]
- Motley, G.; Dalrymple, N.; Keesling, C.; Fischer, J.; Harmon, W. Hounsfield unit density in the determination of urinary stone composition. Urology 2001, 58, 170–173. [Google Scholar] [CrossRef]
- Marchiñena, P.G.; Peres, N.B.; Liyo, J.; Ocantos, J.; Gonzalez, M.; Jurado, A.; Daels, F. CT SCAN as a predictor of composition and fragility of urinary lithiasis treated with extracorporeal shock wave lithotripsy in vitro. Arch. Esp. Urol. 2009, 62, 215–222. [Google Scholar]
- Cordova-Chavez, N.; Onofre-Castillo, J.; Mancilla-Serrato, A.; Santana-Vela, I.; Valdés-Martínez, F.; Torres-Gomez, E.; Cui-tuny-Romero, A. Use of Hounsfield units in predicting the chemical composition of kidney stones. Ann. Radiol. Mex. 2014, 13, 40–44. [Google Scholar]
- Zarse, C.A.; Hameed, T.A.; Jackson, M.E.; Pishchalnikov, Y.; Lingeman, J.E.; McAteer, J.A.; Williams, J.C. CT visible internal stone structure, but not Hounsfield unit value, of calcium oxalate monohydrate (COM) calculi predicts lithotripsy fragility in vitro. Urol. Res. 2007, 35, 201–206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gupta, N.P.; Ansari, M.S.; Kesarvani, P.; Kapoor, A.; Mukhopadhyay, S. Role of computed tomography with no contrast medium enhancement in predicting the outcome of extracorporeal shock wave lithotripsy for urinary calculi. BJU Int. 2005, 95, 1285–1288. [Google Scholar] [CrossRef] [PubMed]
- Gallioli, A.; De Lorenzis, E.; Boeri, L.; Delor, M.; Zanetti, S.P.; Longo, F.; Trinchieri, A.; Montanari, E. Clinical utility of computed tomography Hounsfield characterization for percutaneous nephrolithotomy: A cross-sectional study. BMC Urol. 2017, 17, 104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nalbandyan, V.B. X-ray diffraction analysis of urinary calculi: Need for heat treatment. Urol. Res. 2008, 36, 247–249. [Google Scholar] [CrossRef] [PubMed]
- Wilson, E.; Bushiri, M.J.; Vaidyan, V. Characterization and FTIR spectral studies of human urinary stones from Southern India. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2010, 77, 442–445. [Google Scholar] [CrossRef] [PubMed]
- Maurice-Estepa, L.; Levillain, P.; Lacour, B.; Daudon, M. Advantage of zero-crossing-point first-derivative spectrophotometry for the quantification of calcium oxalate crystalline phases by infrared spectrophotometry. Clin. Chim. Acta 2000, 298, 1–11. [Google Scholar] [CrossRef]
| HU | Hounsfield Density | ||||
|---|---|---|---|---|---|
| Compound | Compared with | t; d; p-Value | Compound | Compared with | t; d; p-Value |
| Anhydrous uric acid | Magnesium ammonium phosphate | 1.25; 0.52; 0.050 | Anhydrous uric acid | Magnesium ammonium phosphate | 1.30; 0.21; 0.050 |
| Calcium oxalate monohydrate | 3.74; 1.42; 0.008 | Calcium oxalate monohydrate | 3.10; 0.47; 0.034 | ||
| Calcium oxalate dihydrate | 3.15; 1.18; 0.031 | Calcium oxalate dihydrate | 3.50; 0.52; 0.014 | ||
| Apatite carbonate | 3.70; 1.38; 0.009 | Apatite carbonate | 2.97; 0.44; 0.046 | ||
| Magnesium ammonium phosphate | Calcium oxalate monohydrate | 1.26; 0.85; 0.058 | Magnesium ammonium phosphate | Calcium oxalate monohydrate | 1.55; 0.23; 0.050 |
| Calcium oxalate dihydrate | 1.64; 0.61; 0.051 | Calcium oxalate dihydrate | 1.92; 0.29; 0.031 | ||
| Apatite carbonate | 2.16; 0.81; 0.045 | Apatite carbonate | 1.37; 0.21; 0.064 | ||
| Calcium oxalate monohydrate | Calcium oxalate dihydrate | 0.86; 0.28; 0.906 | Calcium oxalate monohydrate | Calcium oxalate dihydrate | 0.44; 0.49; 0.058 |
| Apatite carbonate | 0.22; 0.07; 0.999 | Apatite carbonate | 0.31; 0.38; 0.045 | ||
| Calcium oxalate dihydrate | Apatite carbonate | 1.65; 0.61; 0.487 | Calcium oxalate dihydrate | Apatite carbonate | 0.71; 0.10; 0.931 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodríguez-Plata, I.T.; Medina-Escobedo, M.; Basulto-Martínez, M.; Avila-Nava, A.; Gutiérrez-Solis, A.L.; Méndez-Domínguez, N.; Lugo, R. Implementation of a Technique Based on Hounsfield Units and Hounsfield Density to Determine Kidney Stone Composition. Tomography 2021, 7, 606-613. https://doi.org/10.3390/tomography7040051
Rodríguez-Plata IT, Medina-Escobedo M, Basulto-Martínez M, Avila-Nava A, Gutiérrez-Solis AL, Méndez-Domínguez N, Lugo R. Implementation of a Technique Based on Hounsfield Units and Hounsfield Density to Determine Kidney Stone Composition. Tomography. 2021; 7(4):606-613. https://doi.org/10.3390/tomography7040051
Chicago/Turabian StyleRodríguez-Plata, Irvin Tadeo, Martha Medina-Escobedo, Mario Basulto-Martínez, Azalia Avila-Nava, Ana Ligia Gutiérrez-Solis, Nina Méndez-Domínguez, and Roberto Lugo. 2021. "Implementation of a Technique Based on Hounsfield Units and Hounsfield Density to Determine Kidney Stone Composition" Tomography 7, no. 4: 606-613. https://doi.org/10.3390/tomography7040051
APA StyleRodríguez-Plata, I. T., Medina-Escobedo, M., Basulto-Martínez, M., Avila-Nava, A., Gutiérrez-Solis, A. L., Méndez-Domínguez, N., & Lugo, R. (2021). Implementation of a Technique Based on Hounsfield Units and Hounsfield Density to Determine Kidney Stone Composition. Tomography, 7(4), 606-613. https://doi.org/10.3390/tomography7040051









