Semi-Automated 3D Volumetric Renal Measurements in Polycystic Kidney Disease Using b0-Images—A Feasibility Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Recruitment
2.2. MRI Examination
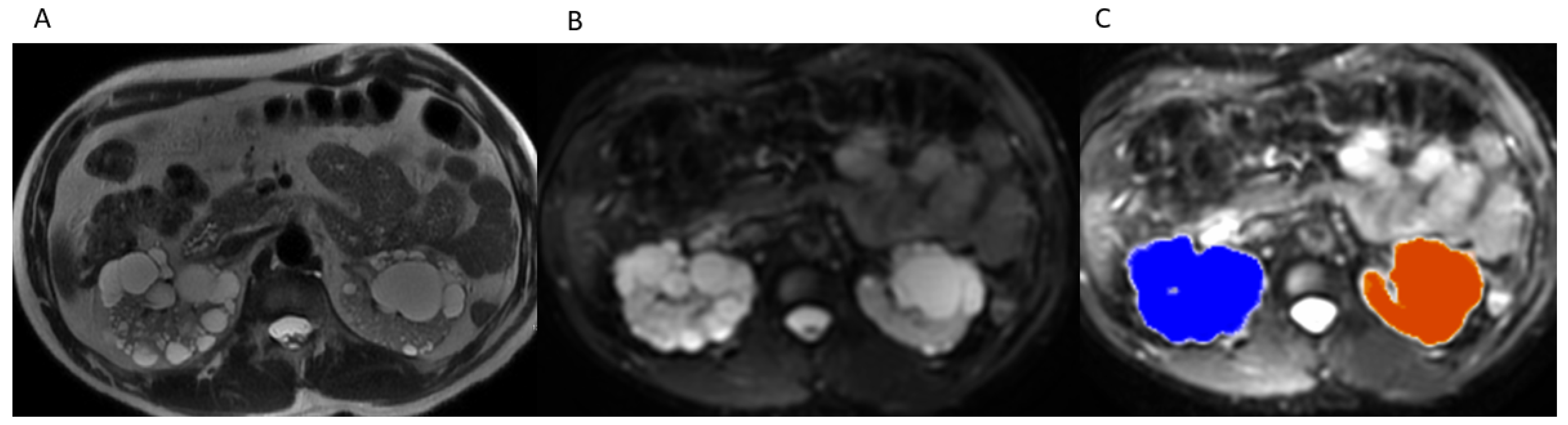
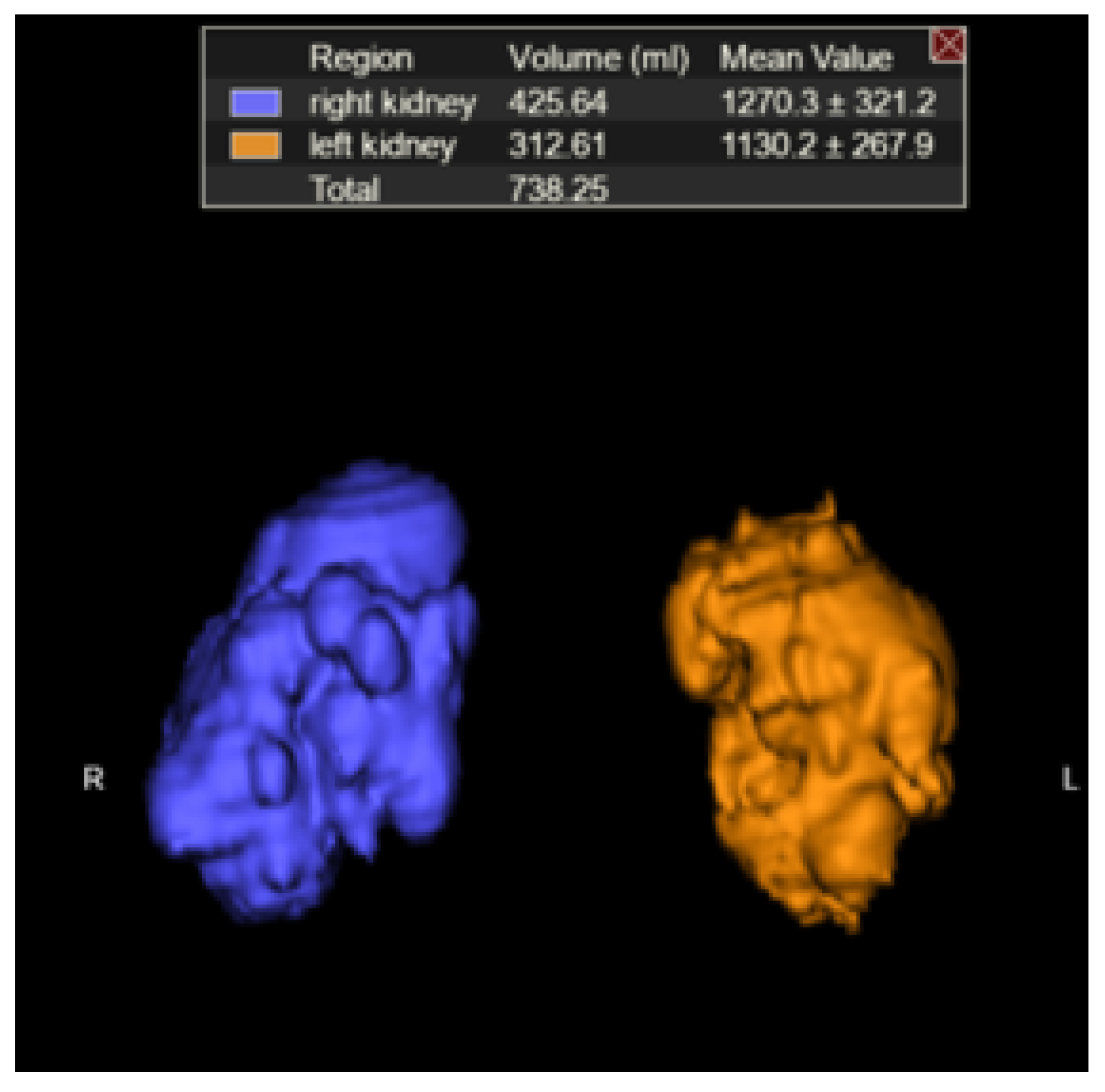
2.3. TKV Calculation
2.4. Statistical Methodology
2.5. Accuracy Testing
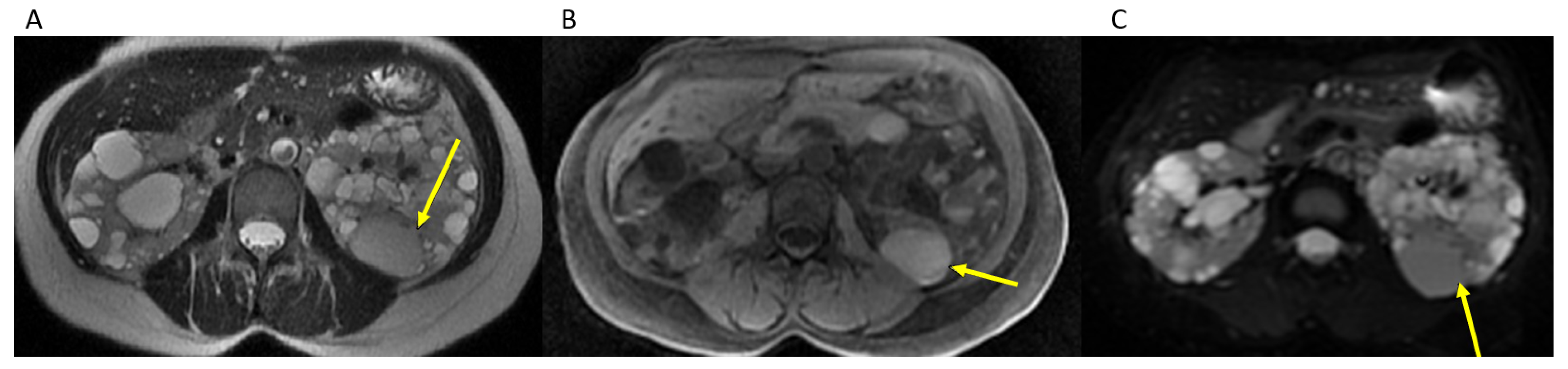
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- CDC. Table 95. Personal Health Care Expenditures, by Source of Funds and Type of Ex-penditure: United States, Selected Years 1960–2014. Available online: https://www.cdc.gov/nchs/data/hus/2015/095.pdf (accessed on 5 January 2018).
- CDC. Table 94. National Health Expenditures, Average Annual Percent Change, and per-Cent Distribution, by Type of Expenditure: United States, Selected Years 1960–2014. Available online: https://www.cdc.gov/nchs/data/hus/2015/094.pdf (accessed on 5 January 2018).
- CDC. Table 93. Gross Domestic Product, National Health Expenditures, Per Capita Amounts, Percent Distribution, and Average Annual Percent Change: United States, Selected Years 1960–2014. Available online: https://www.cdc.gov/nchs/data/hus/2015/093.pdf (accessed on 5 January 2018).
- Coresh, J.; Selvin, E.; Stevens, L.A.; Manzi, J.; Kusek, J.W.; Eggers, P.; Van Lente, F.; Levey, A.S. Prevalence of Chronic Kidney Disease in the United States. JAMA 2007, 298, 2038–2047. [Google Scholar] [CrossRef] [Green Version]
- Honeycutt, A.A.; Segel, J.; Zhuo, X.; Hoerger, T.J.; Imai, K.; Williams, D. Medical Costs of CKD in the Medicare Population. J. Am. Soc. Nephrol. 2013, 24, 1478–1483. [Google Scholar] [CrossRef]
- Hildebrandt, F. Genetic kidney diseases. Lancet 2010, 375, 1287–1295. [Google Scholar] [CrossRef] [Green Version]
- Chapman, A.B.; Bost, J.E.; Torres, V.E.; Guay-Woodford, L.; Bae, K.T.; Landsittel, D.; Li, J.; King, B.F.; Martin, D.; Wetzel, L.H.; et al. Kidney Volume and Functional Outcomes in Autosomal Dominant Polycystic Kidney Disease. Clin. J. Am. Soc. Nephrol. 2012, 7, 479–486. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alam, A.; Dahl, N.K.; Lipschutz, J.H.; Rossetti, S.; Smith, P.; Sapir, D.; Weinstein, J.; McFarlane, P.; Bichet, D.G. Total Kidney Volume in Autosomal Dominant Polycystic Kidney Disease: A Biomarker of Disease Progression and Therapeutic Efficacy. Am. J. Kidney Dis. 2015, 66, 564–576. [Google Scholar] [CrossRef] [PubMed]
- Bhutani, H.; Smith, V.; Rahbari-Oskoui, F.; Mittal, A.; Grantham, J.J.; Torres, V.E.; Mrug, M.; Bae, K.T.; Wu, Z.; Ge, Y.; et al. A comparison of ultrasound and magnetic resonance imaging shows kidney length predicts chronic kidney disease in autosomal dominant polycystic kidney disease. Kidney Int. 2015, 88, 146–151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grantham, J.J.; Torres, V.E.; Chapman, A.B.; Guay-Woodford, L.M.; Bae, K.T.; King, B.F.; Wetzel, L.H.; Baumgarten, D.A.; Kenney, P.J.; Harris, P.C.; et al. Volume Progression in Polycystic Kidney Disease. N. Engl. J. Med. 2006, 354, 2122–2130. [Google Scholar] [CrossRef] [Green Version]
- O’Neill, W.C.; Robbin, M.L.; Bae, K.T.; Grantham, J.J.; Chapman, A.B.; Guay-Woodford, L.M.; Torres, V.E.; King, B.F.; Wetzel, L.H.; Thompson, P.A.; et al. Sonographic assessment of the severity and progression of autosomal dominant polycystic kidney disease: The Consortium of Renal Imaging Studies in Polycystic Kidney Disease (CRISP). Am. J. Kidney Dis. 2005, 46, 1058–1064. [Google Scholar] [CrossRef]
- Bae, K.T.; Tao, C.; Wang, J.; Kaya, D.; Wu, Z.; Bae, J.T.; Chapman, A.B.; Torres, V.E.; Grantham, J.J.; Mrug, M.; et al. Novel Approach to Estimate Kidney and Cyst Vol-umes using Mid-Slice Magnetic Resonance Images in Polycystic Kidney Disease. Am. J. Nephrol. 2013, 38, 333–341. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, Y.; Ge, Y.; Tao, C.; Zhu, J.; Chapman, A.B.; Torres, V.E.; Yu, A.S.; Mrug, M.; Bennett, W.M.; Flessner, M.F.; et al. Automated Segmentation of Kidneys from MR Images in Patients with Autosomal Dominant Polycystic Kidney Disease. Clin. J. Am. Soc. Nephrol. 2016, 11, 576–584. [Google Scholar] [CrossRef] [Green Version]
- Bae, K.; Park, B.; Sun, H.; Wang, J.; Tao, C.; Chapman, A.B.; Torres, V.E.; Grantham, J.J.; Mrug, M.; Bennett, W.M.; et al. Segmentation of Individual Renal Cysts from MR Images in Patients with Autosomal Dominant Polycystic Kidney Disease. Clin. J. Am. Soc. Nephrol. 2013, 8, 1089–1097. [Google Scholar] [CrossRef] [Green Version]
- Sharma, K.; Caroli, A.; Van Quach, L.; Petzold, K.; Bozzetto, M.; Serra, A.L.; Remuzzi, G.; Remuzzi, A. Kidney volume measurement methods for clinical studies on autosomal dominant polycystic kidney disease. PLoS ONE 2017, 12, e0178488. [Google Scholar] [CrossRef] [Green Version]
- Tangri, N.; Hougen, I.; Alam, A.; Perrone, R.; McFarlane, P.; Pei, Y. Total Kidney Volume as a Biomarker of Disease Progression in Autosomal Dominant Polycystic Kidney Disease. Can. J. Kidney Health Dis. 2017, 4, 2054358117693355. [Google Scholar] [CrossRef] [Green Version]
- Bai, W.; Sinclair, M.; Tarroni, G.; Oktay, O.; Rajchl, M.; Vaillant, G.; Lee, A.M.; Aung, N.; Lukaschuk, E.; Sanghvi, M.M.; et al. Automated cardiovascular magnetic resonance image analysis with fully convulational networks. J. Cardiovasc. Magn. Reson. 2018, 20, 65. [Google Scholar] [CrossRef] [Green Version]
- Goyal, N.; Mor-Avi, V.; Volpato, V.; Narang, A.; Wang, S.; Salerno, M.; Lang, R.M.; Patel, A.R. Machine learning based quantification of ejection and filling parameters by fully automated dynamic measurement of left ventricular volumes from cardiac magnetic resonance images. Magn. Reson. Imaging 2020, 67, 28–32. [Google Scholar] [CrossRef]
- Schuster, A.; Lange, T.; Backhaus, S.J.; Strohmeyer, C.; Boom, P.C.; Matz, J.; Kowallick, J.T.; Lotz, J.; Steinmetz, M.; Kutty, S.; et al. Fully Automated Cardiac Assessment for Diagnostic and Prognostic Stratification Following Myocardial Infarction. J. Am. Heart Assoc. 2020, 9, e016612. [Google Scholar] [CrossRef]
- Kline, T.L.; Korfiatis, P.; Edwards, M.E.; Warner, J.D.; Irazabal, M.V.; King, B.F.; Torres, V.E.; Erickson, B.J. Automatic total kidney volume meas-urement on follow-up magnetic resonance images to facilitate monitoring of autoso-mal dominant polycystic kidney disease progression. Nephrol. Dial. Transpl. 2016, 31, 241–248. [Google Scholar] [CrossRef] [Green Version]
- van Gastel, M.D.A.; Messchendorp, A.L.; Kappert, P.; Kaatee, M.A.; de Jong, M.; Renken, R.J.; ter Horst, G.J.; Mahesh, S.V.K.; Gansevoort, R.T. T1 vs. T2 weighted magnetic resonance imaging to assess total kidney volume in patients with autosomal dominant polycystic kidney disease. Abdom. Radiol. 2017, 436, 1215–1222. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pattanayak, P.; Turkey, E.B.; Summers, R.M. Comparative Evaluation of Three Software Packages for Liver and Spleen Segmentation and Volumetry. Acad. Radiol. 2017, 24, 831–839. [Google Scholar] [CrossRef] [PubMed]
- Liss, M.A.; DeConde, R.; Caovan, D.; Hofler, J.; Gabe, M.; Palazzi, K.L.; Patel, N.D.; Lee, H.J.; Ideker, T.; Van Poppel, H.; et al. Parenchymal Volumetric Assessment as a Predictive Tool to Determine Renal Function Benefit of Nephron-Sparing Surgery Compared with Radical Nephrectomy. J. Endourol. 2016, 30, 114–121. [Google Scholar] [CrossRef] [Green Version]
- Koo, T.K.; Li, M.Y. A Guideline of Selecting and Reporting Intraclass Correlation Coefficients for Reliability Research. J. Chiropr. Med. 2016, 15, 155–163. [Google Scholar] [CrossRef] [Green Version]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017; Available online: https://www.R-project.org/ (accessed on 5 January 2018).
- Gamer, M.; Lemon, J.; Fellows, I.; Singh, P. Irr: Various Coefficients of Interrater Reliability and Agreement. R Package Version 0.84. 2012. Available online: https://CRAN.R-project.org/package=irr (accessed on 5 January 2018).
- Rahbari-Oskoui, F.; Mittal, A.; Mittal, P.; Chapman, A. Renal Relevant Radiology: Radiologic Imaging in Autosomal Dominant Polycystic Kidney Disease. Clin. J. Am. Soc. Nephrol. 2014, 9, 406–415. [Google Scholar] [CrossRef] [Green Version]
- Irazabal, M.V.; Blais, J.D.; Perrone, R.D.; Gansevoort, R.T.; Chapman, A.B.; Devuyst, O.; Higashihara, E.; Harris, P.C.; Zhou, W.; Ouyang, J.; et al. Prognostic Enrichment Design in Clinical Trials for Autosomal Dominant Polycystic Kidney Disease: The TEMPO 3:4 Clinical Trial. Kidney Int. Rep. 2016, 1, 213–220. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| RIGHT | LEFT | |
|---|---|---|
| ICC | 0.989 | 0.961 |
| 95% CI | [0.981, 0.994] | [0.936, 0.979] |
| P VALUE | <0.01 | <0.01 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roudenko, A.; Mahmood, S.; Du, L.; Gunio, D.; Barash, I.; Doo, F.; Slutzky, A.; Kukar, N.; Friedman, B.; Kagen, A. Semi-Automated 3D Volumetric Renal Measurements in Polycystic Kidney Disease Using b0-Images—A Feasibility Study. Tomography 2021, 7, 573-580. https://doi.org/10.3390/tomography7040049
Roudenko A, Mahmood S, Du L, Gunio D, Barash I, Doo F, Slutzky A, Kukar N, Friedman B, Kagen A. Semi-Automated 3D Volumetric Renal Measurements in Polycystic Kidney Disease Using b0-Images—A Feasibility Study. Tomography. 2021; 7(4):573-580. https://doi.org/10.3390/tomography7040049
Chicago/Turabian StyleRoudenko, Alexandra, Soran Mahmood, Linda Du, Drew Gunio, Irina Barash, Florence Doo, Alon Slutzky, Nina Kukar, Barak Friedman, and Alexander Kagen. 2021. "Semi-Automated 3D Volumetric Renal Measurements in Polycystic Kidney Disease Using b0-Images—A Feasibility Study" Tomography 7, no. 4: 573-580. https://doi.org/10.3390/tomography7040049
APA StyleRoudenko, A., Mahmood, S., Du, L., Gunio, D., Barash, I., Doo, F., Slutzky, A., Kukar, N., Friedman, B., & Kagen, A. (2021). Semi-Automated 3D Volumetric Renal Measurements in Polycystic Kidney Disease Using b0-Images—A Feasibility Study. Tomography, 7(4), 573-580. https://doi.org/10.3390/tomography7040049





