Cerebral Microvascular Perfusion Assessed in Elderly Adults by Spin-Echo Dynamic Susceptibility Contrast MRI at 7 Tesla
Abstract
1. Introduction
2. Methods
2.1. Study Subjects
2.2. Image Acquisition
2.3. Data Analysis
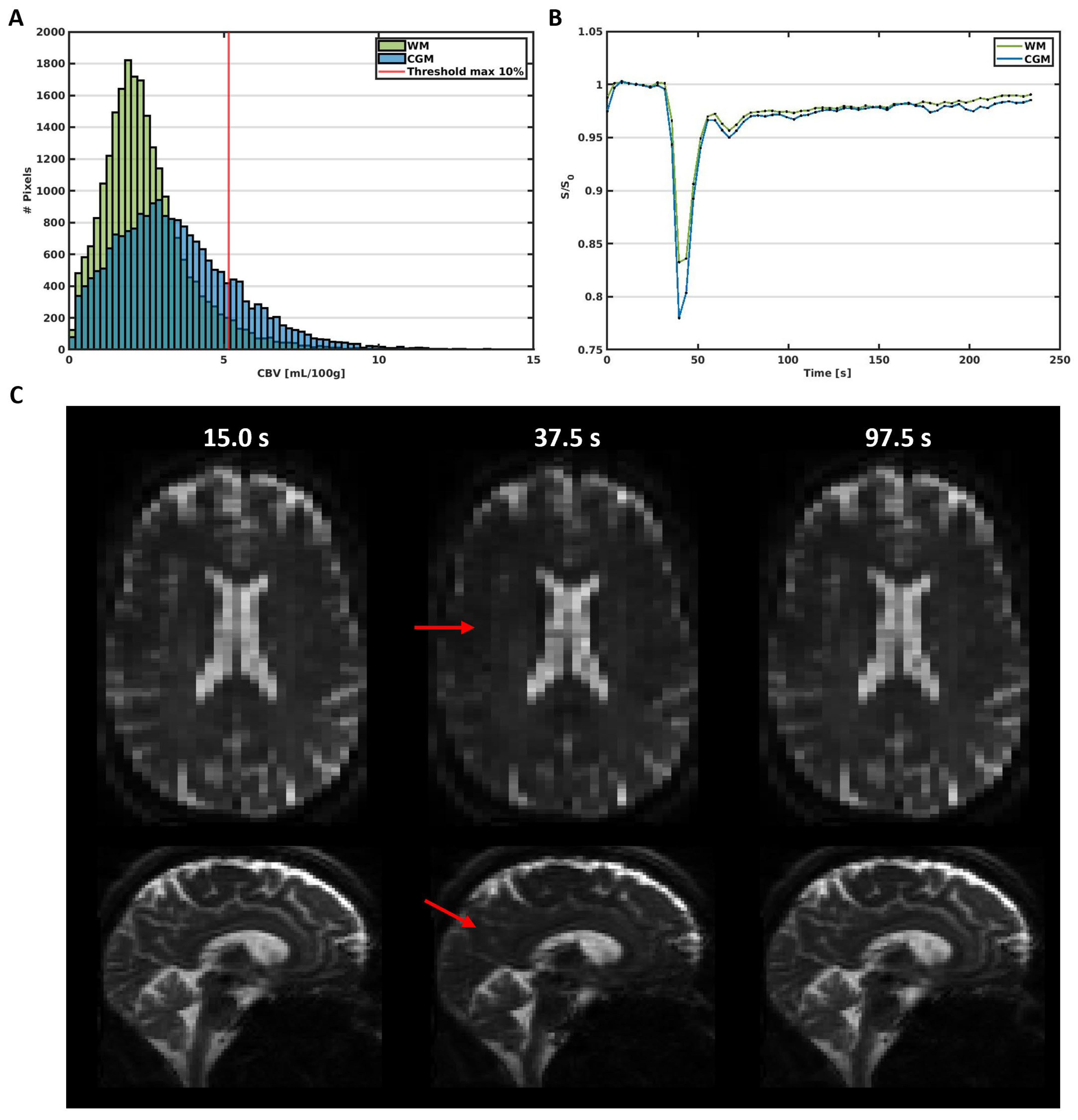
2.3.1. Segmentations
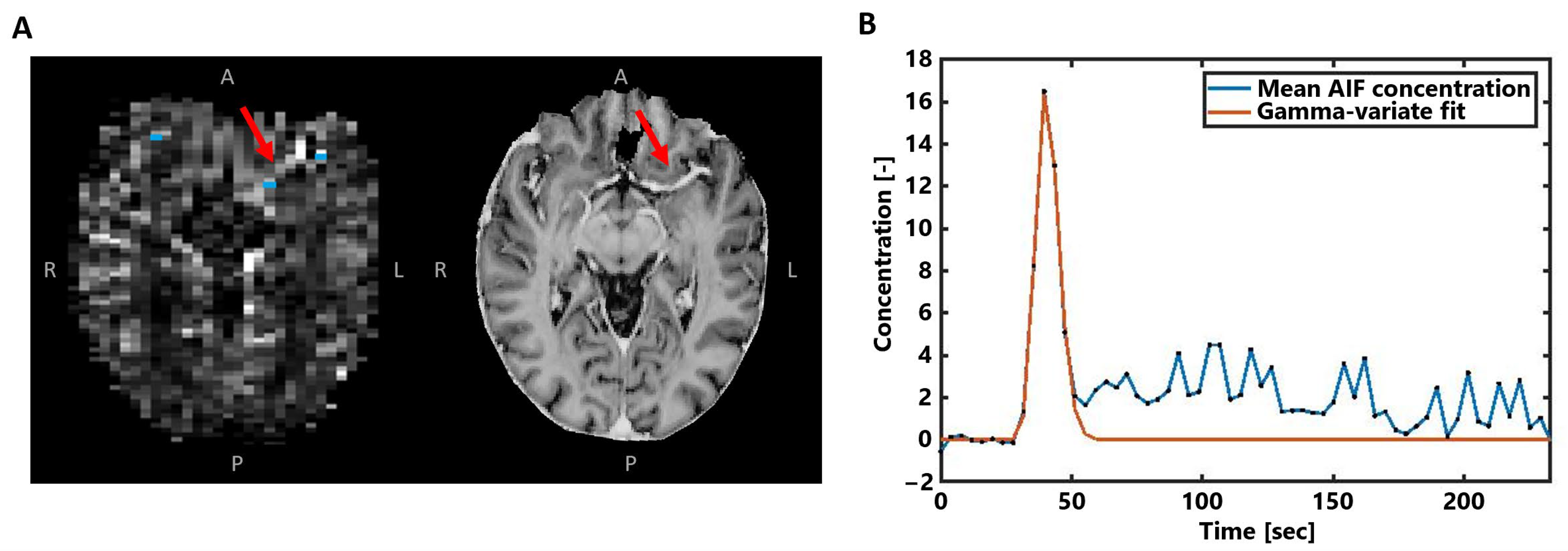
2.3.2. SE-EPI DSC-MRI
2.3.3. Statistics
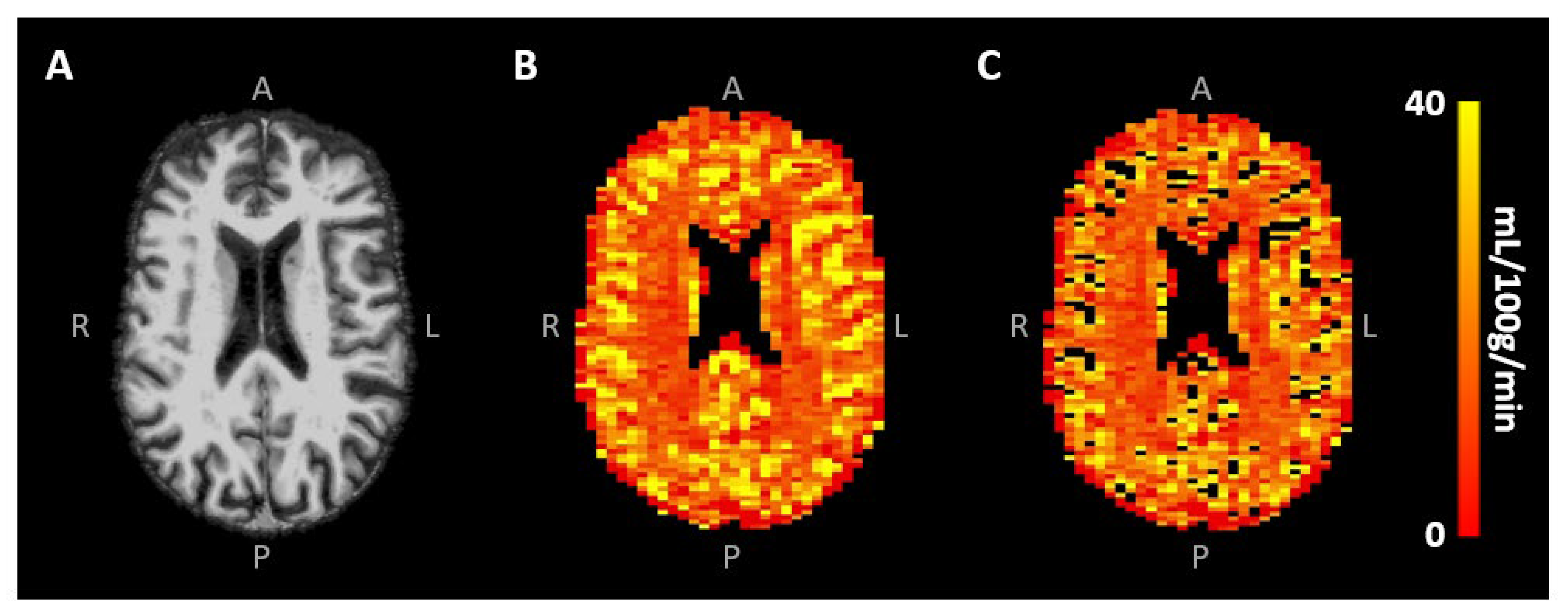
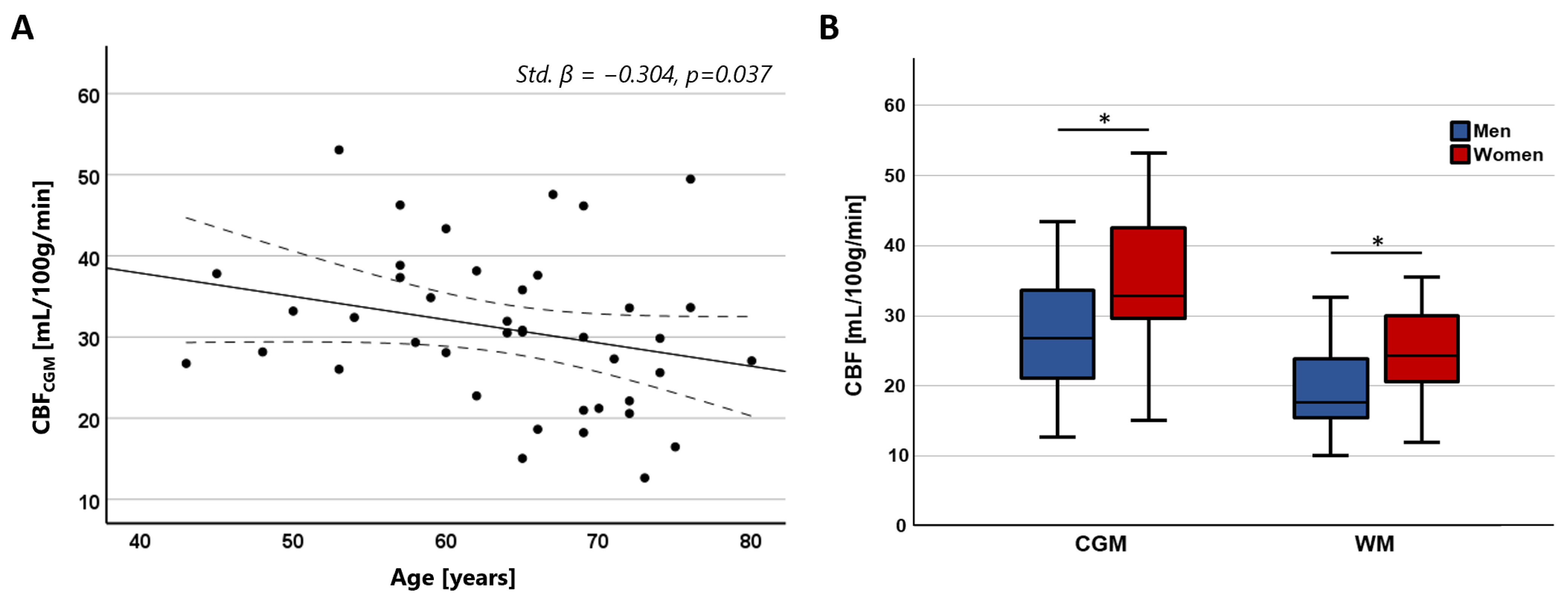
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shiroishi, M.S.; Castellazzi, G.; Boxerman, J.L.; D’Amore, F.; Essig, M.; Nguyen, T.B.; Provenzale, J.M.; Enterline, D.S.; Anzalone, N.; Dörfler, A.; et al. Principles of T2 *-weighted Dynamic Susceptibility Contrast MRI Technique in Brain Tumor Imaging. J. Magn. Reson. Imaging 2015, 41, 296–313. [Google Scholar] [CrossRef]
- Boxerman, J.L.; Quarles, C.C.; Hu, L.S.; Erickson, B.J.; Gerstner, E.R.; Smits, M.; Kaufmann, T.J.; Barboriak, D.P.; Huang, R.H.; Wick, W.; et al. Consensus Recommendations for a Dynamic Susceptibility Contrast MRI Protocol for Use in High-Grade Gliomas. Neuro. Oncol. 2020, 22, 1262–1275. [Google Scholar] [PubMed]
- Anzalone, N.; Castellano, A.; Cadioli, M.; Conte, G.M.; Cuccarini, V.; Bizzi, A.; Grimaldi, M.; Costa, A.; Grillea, G.; Vitali, P.; et al. Brain Gliomas: Multicenter Standardized Assessment of Dynamic Contrast-Enhanced and Dynamic Susceptibility Contrast MR Images. Radiology 2018, 287, 933–943. [Google Scholar] [PubMed]
- Patel, P.; Baradaran, H.; Delgado, D.; Askin, G.; Christos, P.; John Tsiouris, A.; Gupta, A. MR Perfusion-Weighted Imaging in the Evaluation of High-Grade Gliomas after Treatment: A Systematic Review and Meta-Analysis. Neuro. Oncol. 2017, 19, 118–127. [Google Scholar] [CrossRef] [PubMed]
- Quan, G.; Zhang, K.; Liu, Y.; Ren, J.-L.; Huang, D.; Wang, W.; Yuan, T. Role of Dynamic Susceptibility Contrast Perfusion MRI in Glioma Progression Evaluation. J. Oncol. 2021, 2021, 1696387. [Google Scholar] [CrossRef] [PubMed]
- Copen, W.A.; Schaefer, P.W.; Wu, O. MR Perfusion Imaging in Acute Ischemic Stroke. Neuroimaging Clin. N. Am. 2011, 21, 259–283. [Google Scholar] [CrossRef]
- Schellinger, P.D.; Bryan, R.N.; Caplan, L.R.; Detre, J.A.; Edelman, R.R.; Jaigobin, C.; Kidwell, C.S.; Mohr, J.P.; Sloan, M.; Sorensen, A.G.; et al. Evidence-Based Guideline: The Role of Diffusion and Perfusion MRI for the Diagnosis of Acute Ischemic Stroke. Neurology 2010, 75, 177–185. [Google Scholar] [CrossRef]
- Albers, G.W.; Thijs, V.N.; Wechsler, L.; Kemp, S.; Schlaug, G.; Skalabrin, E.; Bammer, R.; Kakuda, W.; Lansberg, M.G.; Shuaib, A.; et al. Magnetic Resonance Imaging Profiles Predict Clinical Response to Early Reperfusion: The Diffusion and Perfusion Imaging Evaluation for Understanding Stroke Evolution (DEFUSE) Study. Ann. Neurol. 2006, 60, 508–517. [Google Scholar]
- Nael, K.; Sakai, Y.; Khatri, P.; Prestigiacomo, C.J.; Puig, J.; Vagal, A. Imaging-Based Selection for Endovascular Treatment in Stroke. RadioGraphics 2019, 39, 1696–1713. [Google Scholar] [PubMed]
- Villringer, K.; Zimny, S.; Galinovic, I.; Nolte, C.H.; Fiebach, J.B.; Khalil, A.A. The Association Between Recanalization, Collateral Flow, and Reperfusion in Acute Stroke Patients: A Dynamic Susceptibility Contrast MRI Study. Front. Neurol. 2019, 10, 1147. [Google Scholar]
- Hauser, T.; Schönknecht, P.; Thomann, P.A.; Gerigk, L.; Schröder, J.; Henze, R.; Radbruch, A.; Essig, M. Regional Cerebral Perfusion Alterations in Patients with Mild Cognitive Impairment and Alzheimer Disease Using Dynamic Susceptibility Contrast MRI. Acad. Radiol. 2013, 20, 705–711. [Google Scholar]
- Østergaard, L. Principles of Cerebral Perfusion Imaging by Bolus Tracking. J. Magn. Reson. Imaging 2005, 22, 710–717. [Google Scholar] [CrossRef]
- Weisskoff, R.; Zuo, C.S.; Boxerman, J.L.; Rosen, B.R. Microscopic Susceptibility Variation and Transverse Relaxation: Theory and Experiment. Magn. Reson. Med. 1994, 31, 601–610. [Google Scholar] [CrossRef]
- Boxerman, J.L.; Hamberg, L.M.; Rosen, B.R.; Weisskoff, R.M. Mr Contrast Due to Intravascular Magnetic Susceptibility Perturbations. Magn. Reson. Med. 1995, 34, 555–566. [Google Scholar] [CrossRef] [PubMed]
- Kiselev, V.G. On the Theoretical Basis of Perfusion Measurements by Dynamic Susceptibility Contrast MRI. Magn. Reson. Med. 2001, 46, 1113–1122. [Google Scholar] [CrossRef] [PubMed]
- Simonsen, C.Z.; Østergaard, L.; Smith, D.F.; Vestergaard-Poulsen, P.; Gyldensted, C. Comparison of Gradient- and Spin-Echo Imaging: CBF, CBV, and MTT Measurements by Bolus Tracking. J. Magn. Reson. Imaging 2000, 12, 411–416. [Google Scholar]
- Rensma, S.P.; van Sloten, T.T.; Launer, L.J.; Stehouwer, C.D.A. Cerebral Small Vessel Disease and Risk of Incident Stroke, Dementia and Depression, and All-Cause Mortality: A Systematic Review and Meta-Analysis. Neurosci. Biobehav. Rev. 2018, 90, 164–173. [Google Scholar] [PubMed]
- Knutsson, L.; Xu, X.; Ståhlberg, F.; Barker, P.B.; Lind, E.; Sundgren, P.C.; van Zijl, P.C.M.; Wirestam, R. Dynamic Susceptibility Contrast MRI at 7 T: Tail-Scaling Analysis and Inferences About Field Strength Dependence. Tomography 2017, 3, 74–78. [Google Scholar] [CrossRef]
- Port, J.D.; Pomper, M.G. Quantification and Minimization of Magnetic Susceptibility Artifacts on GRE Images. J. Comput. Assist. Tomogr. 2000, 24, 958–964. [Google Scholar]
- Carroll, T.J.; Horowitz, S.; Shin, W.; Mouannes, J.; Sawlani, R.; Ali, S.; Raizer, J.; Futterer, S. Quantification of Cerebral Perfusion Using the “Bookend Technique”: An Evaluation in CNS Tumors. Magn. Reson. Imaging 2008, 26, 1352–1359. [Google Scholar] [CrossRef]
- Van Den Kerkhof, M.; Van Der Thiel, M.M.; Postma, A.A.; Van Oostenbrugge, R.J.; Kroon, A.A.; Jansen, J.F.A.; Backes, W.H. Hypertension Correlates with Stronger Blood Flow Pulsatility in Small Perforating Cerebral Arteries Assessed with 7 Tesla Magnetic Resonance Imaging. Hypertension 2023, 80, 802–810. [Google Scholar] [PubMed]
- Fazekas, F.; Chawluk, J.; Alavi, A.; Hurtig, H.; Zimmerman, R. MR Signal Abnormalities at 1.5 T in Alzheimer’s Dementia and Normal Aging. Am. J. Roentgenol. 1987, 149, 351–356. [Google Scholar] [CrossRef]
- Fischl, B.; Salat, D.H.; Busa, E.; Albert, M.; Dieterich, M.; Haselgrove, C.; Van Der Kouwe, A.; Killiany, R.; Kennedy, D.; Klaveness, S.; et al. Whole Brain Segmentation: Automated Labeling of Neuroanatomical Structures in the Human Brain. Neuron 2002, 33, 341–355. [Google Scholar] [CrossRef] [PubMed]
- Jenkinson, M.; Bannister, P.; Brady, M.; Smith, S. Improved Optimization for the Robust and Accurate Linear Registration and Motion Correction of Brain Images. Neuroimage 2002, 17, 825–841. [Google Scholar] [PubMed]
- Andersson, J.L.R.; Skare, S.; Ashburner, J. How to Correct Susceptibility Distortions in Spin-Echo Echo-Planar Images: Application to Diffusion Tensor Imaging. Neuroimage 2003, 20, 870–888. [Google Scholar] [PubMed]
- Østergaard, L.; Sorensen, A.G.; Kwong, K.K.; Weisskoff, R.M.; Gyldensted, C.; Rosen, B.R. High Resolution Measurement of Cerebral Blood Flow Using Intravascular Tracer Bolus Passages. Part II: Experimental Comparison and Preliminary Results. Magn. Reson. Med. 1996, 36, 726–736. [Google Scholar] [CrossRef]
- Boxerman, J.L.; Rosen, B.R.; Weisskoff, R.M. Signal-to-noise Analysis of Cerebral Blood Volume Maps from Dynamic NMR Imaging Studies. J. Magn. Reson. Imaging 1997, 7, 528–537. [Google Scholar] [CrossRef]
- Wu, O.; Østergaard, L.; Weisskoff, R.M.; Benner, T.; Rosen, B.R.; Sorensen, A.G. Tracer Arrival Timing-Insensitive Technique for Estimating Flow in MR Perfusion-Weighted Imaging Using Singular Value Decomposition with a Block-Circulant Deconvolution Matrix. Magn. Reson. Med. 2003, 50, 164–174. [Google Scholar]
- Leenders, K.L.; Perani, D.; Lammertsma, A.A.; Heather, J.D.; Buckingham, P.; Jones, T.; Healy, M.J.R.; Gibbs, J.M.; Wise, R.J.S.; Hatazawa, J.; et al. Cerebral Blood Flow, Blood Volume, and Oxygen Utilization. Brain 1990, 113, 27–47. [Google Scholar] [CrossRef]
- Wong, S.M.; Jansen, J.F.A.; Zhang, C.E.; Hoff, E.I.; Staals, J.; van Oostenbrugge, R.J.; Backes, W.H. Blood-Brain Barrier Impairment and Hypoperfusion Are Linked in Cerebral Small Vessel Disease. Neurology 2019, 92, e1669–e1677. [Google Scholar] [CrossRef]
- Grandin, C.B.; Bol, A.; Smith, A.M.; Michel, C.; Cosnard, G. Absolute CBF and CBV Measurements by MRI Bolus Tracking before and after Acetazolamide Challenge: Repeatabilily and Comparison with PET in Humans. Neuroimage 2005, 26, 525–535. [Google Scholar] [CrossRef]
- Bjørnerud, A.; Emblem, K.E. A Fully Automated Method for Quantitative Cerebral Hemodynamic Analysis Using DSC–MRI. J. Cereb. Blood Flow Metab. 2010, 30, 1066–1078. [Google Scholar]
- Wirestam, R.; Ryding, E.; Lindgren, A.; Geijer, B.; Østergaard, L.; Andersson, L.; Holtås, S.; Ståhlberg, F. Regional Cerebral Blood Flow Distributions in Normal Volunteers: Dynamic Susceptibility Contrast MRI Compared with 99mTc-HMPAO SPECT. J. Comput. Assist. Tomogr. 2000, 24, 526–530. [Google Scholar] [CrossRef] [PubMed]
- Ichise, M.; Golan, H.; Ballinger, J.R.; Vines, D.; Blackman, A.; Moldofsky, H. Regional Differences in Technetium-99m-ECD Clearance on Brain SPECT in Healthy Subjects. J. Nucl. Med. 1997, 38, 1253–1260. [Google Scholar]
- Liu, P.; Uh, J.; Devous, M.D.; Adinoff, B.; Lu, H. Comparison of Relative Cerebral Blood Flow Maps Using Pseudo-continuous Arterial Spin Labeling and Single Photon Emission Computed Tomography. NMR Biomed. 2012, 25, 779–786. [Google Scholar]
- Grüner, J.; Paamand, R.; Højgaard, L.; Law, I. Brain Perfusion CT Compared With15O-H2O-PET in Healthy Subjects. EJNMMI Res. 2011, 1, 28. [Google Scholar] [CrossRef]
- Zhang, K.; Herzog, H.; Mauler, J.; Filss, C.; Okell, T.W.; Kops, E.R.; Tellmann, L.; Fischer, T.; Brocke, B.; Sturm, W.; et al. Comparison of Cerebral Blood Flow Acquired by Simultaneous [15O]Water Positron Emission Tomography and Arterial Spin Labeling Magnetic Resonance Imaging. J. Cereb. Blood Flow Metab. 2014, 34, 1373–1380. [Google Scholar] [PubMed]
- Daniel, D.G.; Mathew, R.J.; Wilson, W.H. Sex Roles and Regional Cerebral Blood Flow. Psychiatry Res. 1989, 27, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Alisch, J.S.R.; Khattar, N.; Kim, R.W.; Cortina, L.E.; Rejimon, A.C.; Qian, W.; Ferrucci, L.; Resnick, S.M.; Spencer, R.G.; Bouhrara, M. Sex and age-related differences in cerebral blood flow investigated using pseudo-continuous arterial spin labeling magnetic resonance imaging. Aging 2021, 13, 4911–4925. [Google Scholar] [CrossRef]
- Aanerud, J.; Borghammer, P.; Rodell, A.; Jónsdottir, K.Y.; Gjedde, A. Sex Differences of Human Cortical Blood Flow and Energy Metabolism. J. Cereb. Blood Flow Metab. 2017, 37, 2433–2440. [Google Scholar] [CrossRef]
- Shin, W.; Horowitz, S.; Ragin, A.; Chen, Y.; Walker, M.; Carroll, T.J. Quantitative Cerebral Perfusion Using Dynamic Susceptibility Contrast MRI: Evaluation of Reproducibility and Age- and Gender-dependence with Fully Automatic Image Postprocessing Algorithm. Magn. Reson. Med. 2007, 58, 1232–1241. [Google Scholar] [PubMed]
- Parkes, L.M.; Rashid, W.; Chard, D.T.; Tofts, P.S. Normal Cerebral Perfusion Measurements Using Arterial Spin Labeling: Reproducibility, Stability, and Age and Gender Effects. Magn. Reson. Med. 2004, 51, 736–743. [Google Scholar] [CrossRef]
- Wenz, F.; Rempp, K.; Brix, G.; Knopp, M.V.; Gückel, F.; Hess, T.; van Kaick, G. Age Dependency of the Regional Cerebral Blood Volume (RCBV) Measured with Dynamic Susceptibility Contrast MR Imaging (DSC). Magn. Reson. Imaging 1996, 14, 157–162. [Google Scholar] [CrossRef]
- Lévy, S.; Roche, P.; Guye, M.; Callot, V. Feasibility of Human Spinal Cord Perfusion Mapping Using Dynamic Susceptibility Contrast Imaging at 7 T: Preliminary Results and Identified Guidelines. Magn. Reson. Med. 2021, 85, 1183–1194. [Google Scholar]
- Elschot, E.P.; Backes, W.H.; de Jong, J.J.A.; Drenthen, G.S.; Wong, S.M.; Staals, J.; Postma, A.A.; Rouhl, R.P.W.; van Oostenbrugge, R.J.; Jansen, J.F.A. Assessment of the Clinical Feasibility of Detecting Subtle Blood-Brain Barrier Leakage in Cerebral Small Vessel Disease Using Dynamic Susceptibility Contrast MRI. Magn. Reson. Imaging 2023, 102, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Boxerman, J.L.; Schmainda, K.M.; Weisskoff, R.M. Relative Cerebral Blood Volume Maps Corrected for Contrast Agent Extravasation Significantly Correlate with Glioma Tumor Grade, Whereas Uncorrected Maps Do Not. Ajnr. Am. J. Neuroradiol. 2006, 27, 859–867. [Google Scholar]
- Knutsson, L.; Ståhlberg, F.; Wirestam, R. Aspects on the Accuracy of Cerebral Perfusion Parameters Obtained by Dynamic Susceptibility Contrast MRI: A Simulation Study. Magn. Reson. Imaging 2004, 22, 789–798. [Google Scholar] [CrossRef]
- Ugurbil, K. Magnetic Resonance Imaging at Ultrahigh Fields. IEEE Trans. Biomed. Eng. 2014, 61, 1364–1379. [Google Scholar] [CrossRef] [PubMed]
- Johnson, P.M.; Recht, M.P.; Knoll, F. Improving the Speed of MRI with Artificial Intelligence. Semin. Musculoskelet. Radiol. 2020, 24, 012–020. [Google Scholar]

| Characteristic | N = 41 |
|---|---|
| Age (years), median (Q1–Q3) | 65 (58–72) |
| Male, n (%) | 21 (51) |
| Mean arterial pressure (mmHg), median (Q1–Q3) | 93.5 (84.8–99.3) |
| Fazekas score, 0/1/2/3, n (%) | 25 (61)/13 (31.7)/2 (4.9)/1 (2.4) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elschot, E.P.; Backes, W.H.; van den Kerkhof, M.; Postma, A.A.; Kroon, A.A.; Jansen, J.F.A. Cerebral Microvascular Perfusion Assessed in Elderly Adults by Spin-Echo Dynamic Susceptibility Contrast MRI at 7 Tesla. Tomography 2024, 10, 181-192. https://doi.org/10.3390/tomography10010014
Elschot EP, Backes WH, van den Kerkhof M, Postma AA, Kroon AA, Jansen JFA. Cerebral Microvascular Perfusion Assessed in Elderly Adults by Spin-Echo Dynamic Susceptibility Contrast MRI at 7 Tesla. Tomography. 2024; 10(1):181-192. https://doi.org/10.3390/tomography10010014
Chicago/Turabian StyleElschot, Elles P., Walter H. Backes, Marieke van den Kerkhof, Alida A. Postma, Abraham A. Kroon, and Jacobus F. A. Jansen. 2024. "Cerebral Microvascular Perfusion Assessed in Elderly Adults by Spin-Echo Dynamic Susceptibility Contrast MRI at 7 Tesla" Tomography 10, no. 1: 181-192. https://doi.org/10.3390/tomography10010014
APA StyleElschot, E. P., Backes, W. H., van den Kerkhof, M., Postma, A. A., Kroon, A. A., & Jansen, J. F. A. (2024). Cerebral Microvascular Perfusion Assessed in Elderly Adults by Spin-Echo Dynamic Susceptibility Contrast MRI at 7 Tesla. Tomography, 10(1), 181-192. https://doi.org/10.3390/tomography10010014





