The Friction Properties of Firebrat Scales
Abstract
1. Introduction
2. Materials and Methods
2.1. Animal Materials
2.2. Observations of Firebrats’ Surface
2.3. Chemical and Wettability Analysis of Firebrat Scale Surface
2.4. Friction Measurements on a Body Surface
2.5. Friction Measurements within a Scale
3. Results
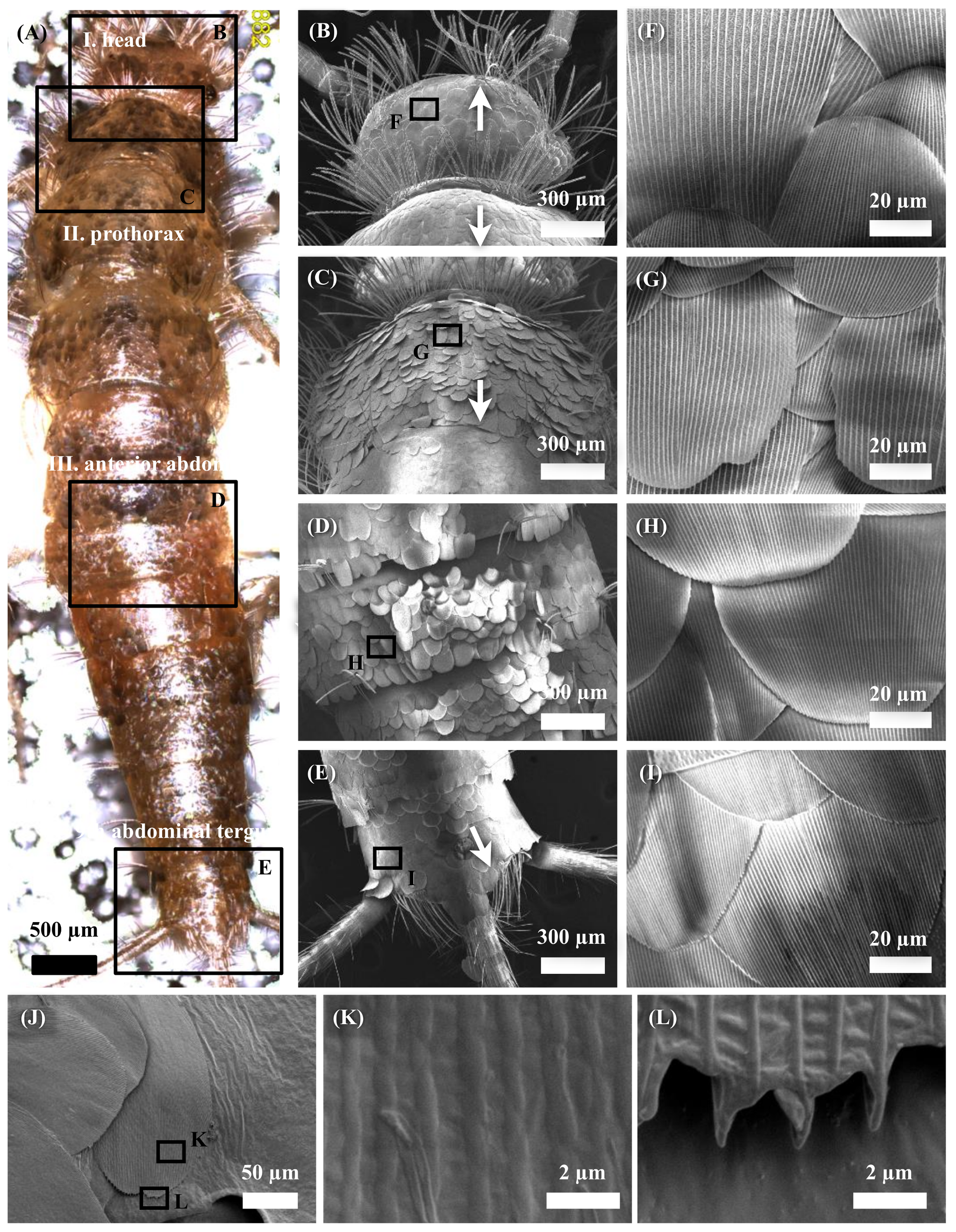
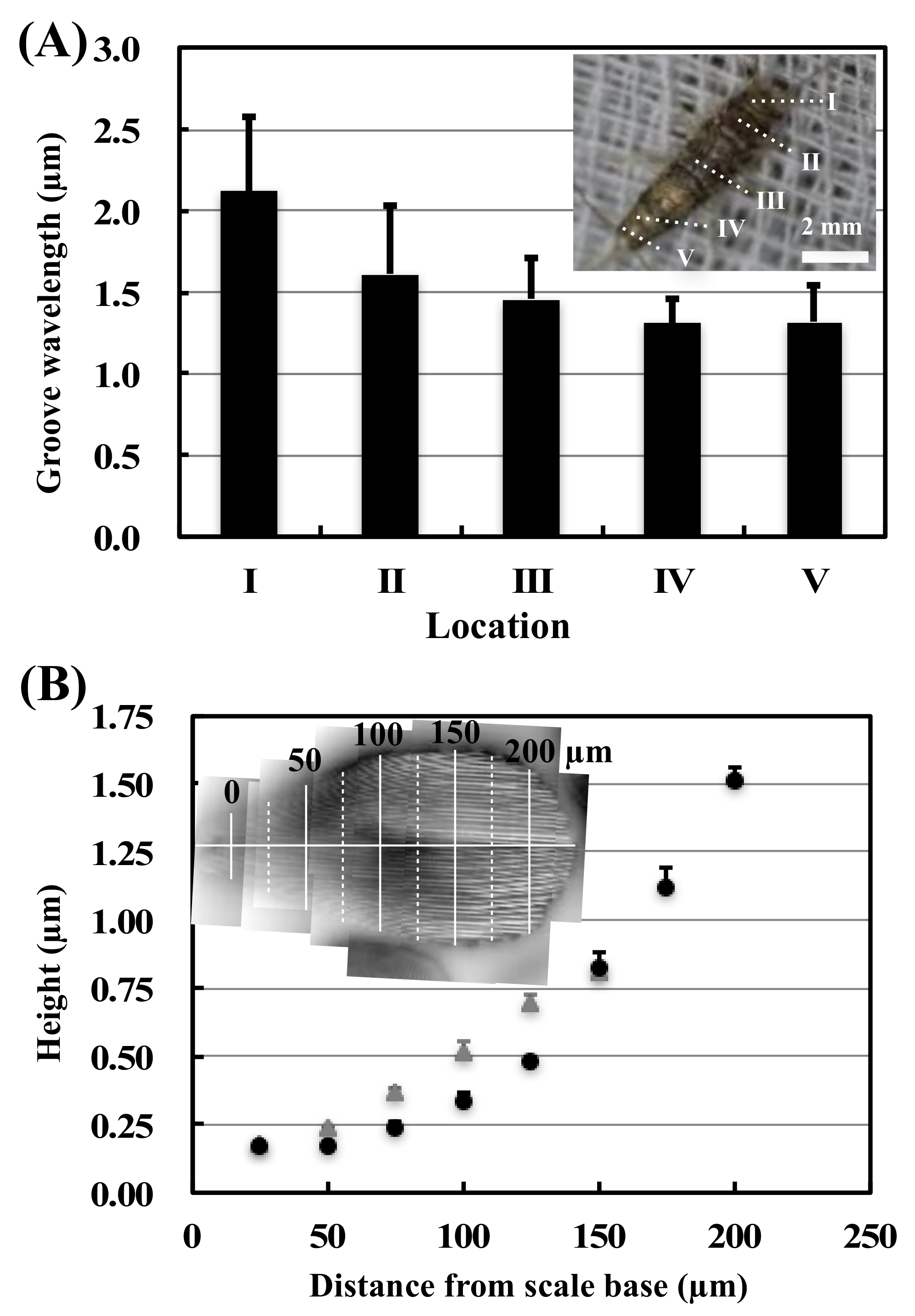
3.1. Surface Observations and Analysis of Firebrat Scales
3.2. Surface Chemical and Wettability Analysis of Firebrat Scales
3.3. Friction Measurements on a Body Surface–Needle Probe
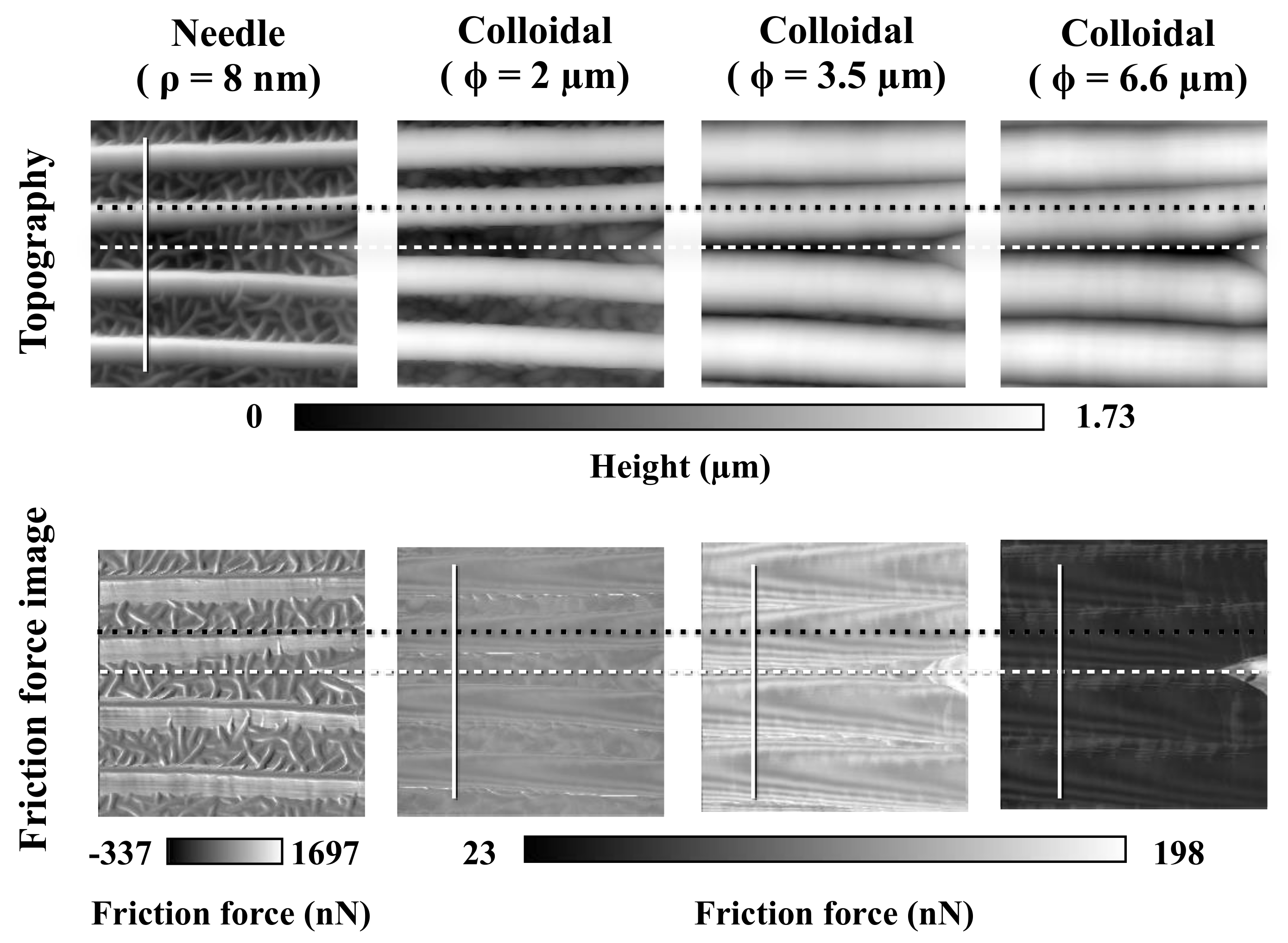
3.4. Friction Measurements on a Body Surface–Colloidal Probe
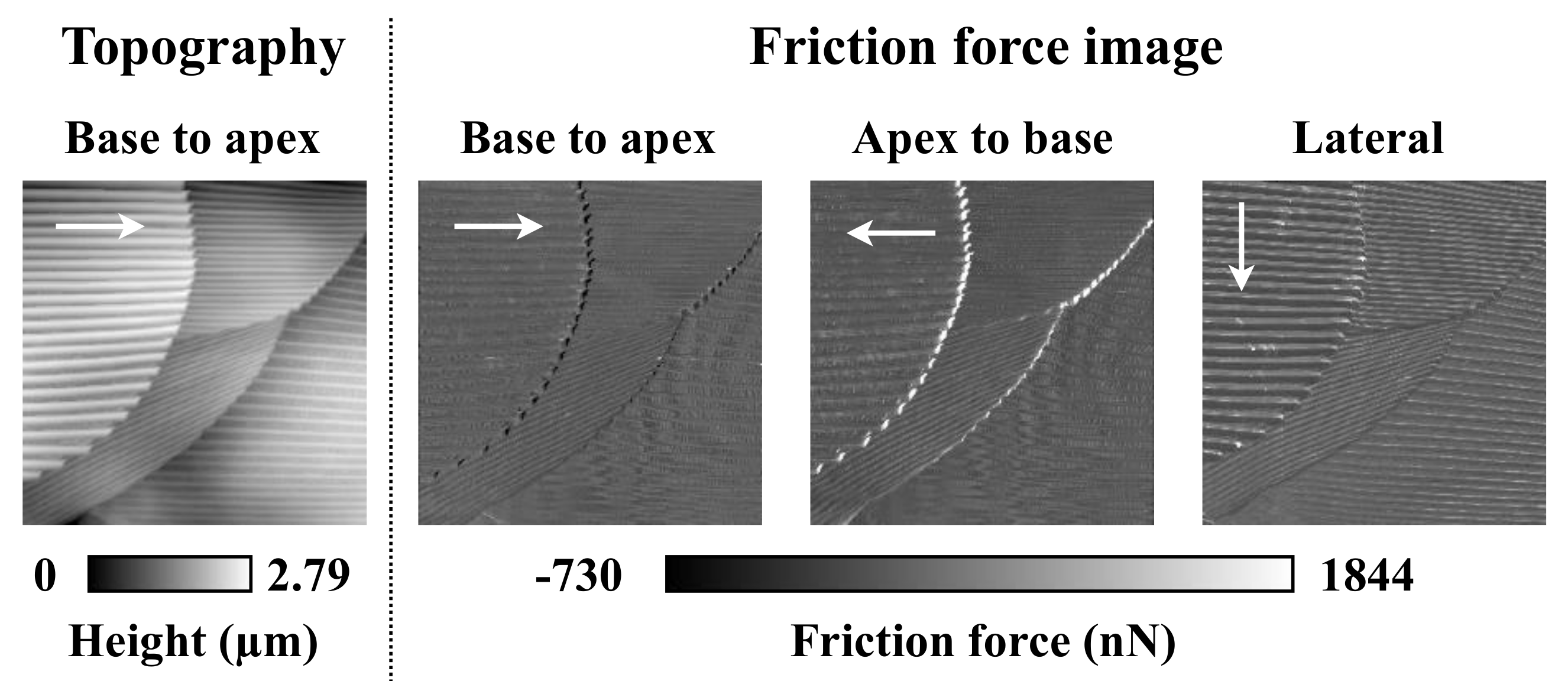
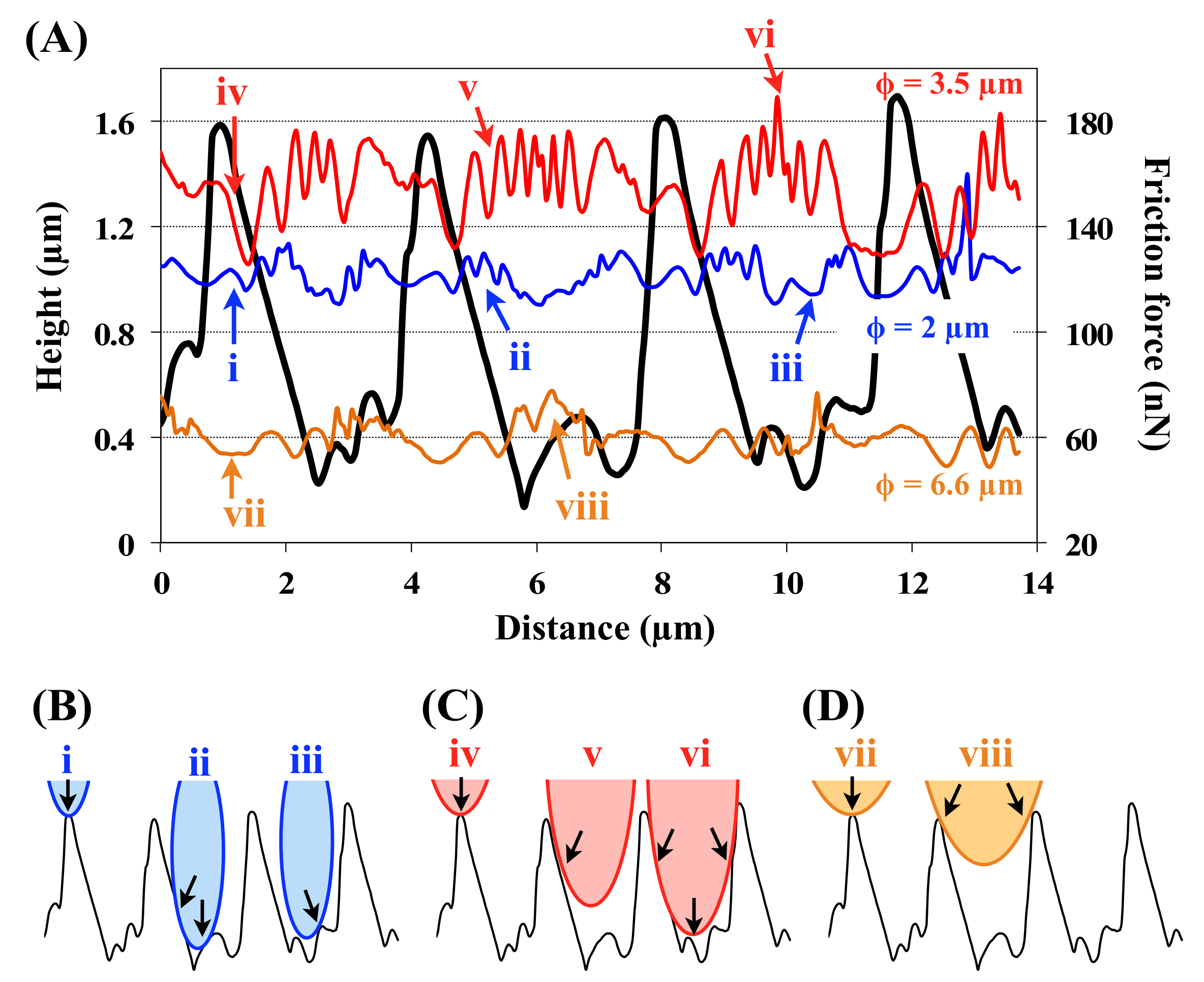
3.5. Friction Measurements within a Scale
4. Discussion
4.1. Observations and Analysis of Firebrat Scales
4.2. Meaning of Scale Growth Directions
4.3. Assumed Effect of Groove Structures on Scales
4.4. Considerations about Groove Wavelengths and Heights Unevenness
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nakada, M. Trends in engine technology and tribology. Tribol. Int. 1994, 27, 3–8. [Google Scholar] [CrossRef]
- Gong, J.; Iwasaki, Y.; Osada, Y.; Kurihara, K.; Hamai, Y. Friction of gels. 3. Friction on solid surfaces. J. Phys. Chem. B 1999, 103, 6001–6006. [Google Scholar] [CrossRef]
- Hörner, D. Recent trends in environmentally friendly lubricants. J. Synth. Lubr. 2002, 18, 327–347. [Google Scholar] [CrossRef]
- Wang, X.; Kato, K.; Adachi, K.; Aizawa, K. Loads carrying capacity map for the surface texture design of SiC thrust bearing sliding in water. Tribol. Int. 2003, 36, 189–197. [Google Scholar] [CrossRef]
- Miyake, K.; Nakano, M.; Korenaga, A.; Mano, H.; Ando, Y. Tribological properties of nanostripe surface structures—A design concept for improving tribological properties. J. Phys. D Appl. Phys. 2010, 43. [Google Scholar] [CrossRef]
- Sasaki, S.; Ogawa, H.; Korenaga, A.; Miyake, K.; Nakano, M.; Murakami, T. Effects of surface texture size on the tribological properties of slideways. Proc. Inst. Mech. Eng. Part J J. Eng. Tribol. 2010, 224, 885–890. [Google Scholar] [CrossRef]
- Overney, R.M.; Takano, H.; Fujihira, M.; Meyer, E.; Güntherodt, H.J. Wear, friction and sliding speed correlations on Langmuir–Blodgett films observed by atomic force microscopy. Thin Solid Films 1994, 240, 105–109. [Google Scholar] [CrossRef]
- Kobayashi, M.; Terayama, Y.; Hosaka, N.; Kaido, M.; Suzuki, A.; Yamada, N.; Torikai, N.; Ishihara, K.; Takahara, A. Friction behavior of high-density poly(2-methacryloyloxyethyl phosphorylcholine) brush in aqueous media. Soft Matter 2007, 3, 740–746. [Google Scholar] [CrossRef]
- Bhushan, B. Biomimetics: Lessons from nature—An overview. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2009, 367, 1445–1486. [Google Scholar] [CrossRef]
- Vincent, J.F.V.; Bogatyreva, O.A.; Bogatyrev, N.R.; Bowyer, A.; Pahl, A.K. Biomimetics: Its practice and theory. J. R. Soc. Interface 2006, 3, 471–482. [Google Scholar] [CrossRef]
- Koch, K.; Bhushan, B.; Barthlott, W. Multifunctional surface structures of plants: An inspiration for biomimetics. Prog. Mater. Sci. 2009, 54, 137–178. [Google Scholar] [CrossRef]
- Barthlott, W.; Neinhuis, C. Purity of the sacred lotus, or escape from contamination in biological surfaces. Planta 1997, 202, 1–8. [Google Scholar] [CrossRef]
- Clapham, P.B.; Hutley, M.C. Reduction of lens reflection by moth eye principle. Nature 1973, 244, 281–282. [Google Scholar] [CrossRef]
- Stavenga, D.G.; Foletti, S.; Palasantzas, G.; Arikawa, K. Light on the moth-eye corneal nipple array of butterflies. Proc. R. Soc. B Biol. Sci. 2006, 273, 661–667. [Google Scholar] [CrossRef] [PubMed]
- Ball, P. Engineering shark skin and other solutions. Nature 1999, 400, 507–509. [Google Scholar] [CrossRef]
- Bechert, D.W.; Bruse, M.; Hage, W.; Meyer, R. Fluid mechanics of biological surfaces and their technological application. Naturwissenschaften 2000, 87, 157–171. [Google Scholar] [CrossRef] [PubMed]
- Autumn, K.; Liang, Y.A.; Hsieh, S.T.; Zesch, W.; Chan, W.P.; Kenny, T.W.; Fearing, R.; Full, R.J. Adhesive force of a single gecko foot-hair. Nature 2000, 405, 681. [Google Scholar] [CrossRef]
- Berthe, R.A.; Westhoff, G.; Bleckmann, H.; Gorb, S.N. Surface structure and frictional properties of the skin of the Amazon tree boa Corallus hortulanus (Squamata, Boidae). J. Comp. Physiol. A Neuroethol. Sens. Neural Behav. Physiol. 2009, 195, 311–318. [Google Scholar] [CrossRef]
- Varenberg, M.; Gorb, S.N. Hexagonal surface micropattern for dry and wet friction. Adv. Mater. 2009, 21, 483–486. [Google Scholar] [CrossRef]
- Sweetman, H.L. Physical ecology of the firebrat, Thermobia domestica (Packard). Ecol. Monogr. 1938, 8, 285–311. [Google Scholar] [CrossRef]
- Watson, J.A.L. Moulting and reproduction in the adult firebrat, Thermobia domestica (Packard) (Thysanura, Lepismatidae)—II. The reproductive cycles. J. Insect Physiol. 1964, 10, 399–408. [Google Scholar] [CrossRef]
- Noble-Nesbitt, J. Water balance in the firebrat, Thermobia domestica (Packard). The site of uptake of water from the atmosphere. J. Exp. Biol. 1970, 52, 193–200. [Google Scholar]
- Ohde, T.; Masumoto, M.; Yaginuma, T.; Niimi, T. Embryonic RNAi analysis in the firebrat, Thermobia domestica: Distal-less is required to form caudal filament. J. Insect Biotechnol. Sericol. 2009, 78, 99–105. [Google Scholar] [CrossRef]
- Noble-Nesbitt, J. Water balance in the firebrat, Thermobia domestica (Packard). Exchanges of water with the atmosphere. J. Exp. Biol. 1969, 50, 745–769. [Google Scholar] [PubMed]
- Ducker, W.A.; Senden, T.J.; Pashley, R.M. Direct measurement of colloidal forces using an atomic force microscope. Nature 1991, 353, 239–241. [Google Scholar] [CrossRef]
- Francius, G.; Hemmerle, J.; Ohayon, J.; Schaaf, P.; Voegel, J.C.; Picart, C.; Senger, B. Effect of crosslinking on the elasticity of polyelectrolyte multilayer films measured by colloidal probe AFM. Microsc. Res. Tech. 2006, 69, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Zauscher, S.; Klingenberg, D.J. Friction between cellulose surfaces measured with colloidal probe microscopy. Colloids Surf. A Physicochem. Eng. Asp. 2001, 178, 213–229. [Google Scholar] [CrossRef]
- Kayvan Najarian, R.S. Biomedical Signal and Image Processing, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2012; 411p. [Google Scholar]
- Rehman, I.U.; Movasaghi, Z.; Rehman, S. Vibrational Spectroscopy for Tissue Analysis, 1st ed.; CRC Press: Boca Raton, FL, USA, 2012; 356p. [Google Scholar]
- Zhang, M.; Haga, A.; Sekiguchi, H.; Hirano, S. Structure of insect chitin isolated from beetle larva cuticle and silkworm (Bombyx mori) pupa exuvia. Int. J. Biol. Macromol. 2000, 27, 99–105. [Google Scholar] [CrossRef]
- Baio, J.E.; Spinner, M.; Jaye, C.; Fischer, D.A.; Gorb, S.N.; Weidner, T. Evidence of a molecular boundary lubricant at snakeskin surfaces. J. R. Soc. Interface 2015, 12. [Google Scholar] [CrossRef] [PubMed]
- Eisner, T.; Alsop, R.; Ettershank, G. Adhesiveness of Spider Silk. Science 1964, 146, 1058–1061. [Google Scholar] [CrossRef]
- Chang, C.; Wu, P.; Baker, R.E.; Maini, P.K.; Alibardi, L.; Chuong, C.-M. Reptile scale paradigm: Evo-devo, pattern formation and regeneration. Int. J. Dev. Biol. 2009, 53, 813–826. [Google Scholar] [CrossRef]
- Kinoshita, S.; Yoshioka, S.; Kawagoe, K. Mechanisms of structural colour in the Morpho butterfly: Cooperation of regularity and irregularity in an iridescent scale. Proc. R. Soc. Lond. B Biol. Sci. 2002, 269, 1417–1421. [Google Scholar] [CrossRef] [PubMed]
- Sloderbeck, P.E. Silverfish and Firebrats; Kansas State University: Manhattan, KS, USA, 2004. [Google Scholar]
- Tremblay, M.N.; Gries, G. Abiotic and biotic factors affect microhabitat selection by the firebrat, Thermobia domestica (Packard) (Thysanura: Lepismatidae). J. Insect Behav. 2006, 19, 321–335. [Google Scholar] [CrossRef]
- Yuan, S.; Huang, W.; Wang, X. Orientation effects of micro-grooves on sliding surfaces. Tribol. Int. 2011, 44, 1047–1054. [Google Scholar] [CrossRef]
- Xing, Y.; Deng, J.; Wu, Z.; Cheng, H. Effect of regular surface textures generated by laser on tribological behavior of Si3N4/TiC ceramic. Appl. Surf. Sci. 2013, 265, 823–832. [Google Scholar] [CrossRef]
- Labonte, D.; Federle, W. Scaling and biomechanics of surface attachment in climbing animals. Philos. Trans. R. Soc. B Biol. Sci. 2015, 370. [Google Scholar] [CrossRef] [PubMed]
- Buck, C.; Edwards, J.S. The effect of appendage and scale loss on instar duration in adult firebrats, Thermobia domestica (Thysanura). J. Exp. Biol. 1990, 151, 341–347. [Google Scholar]
- Xia, F.; Jiang, L. Bio-inspired, smart, multiscale interfacial materials. Adv. Mater. 2008, 20, 2842–2858. [Google Scholar] [CrossRef]
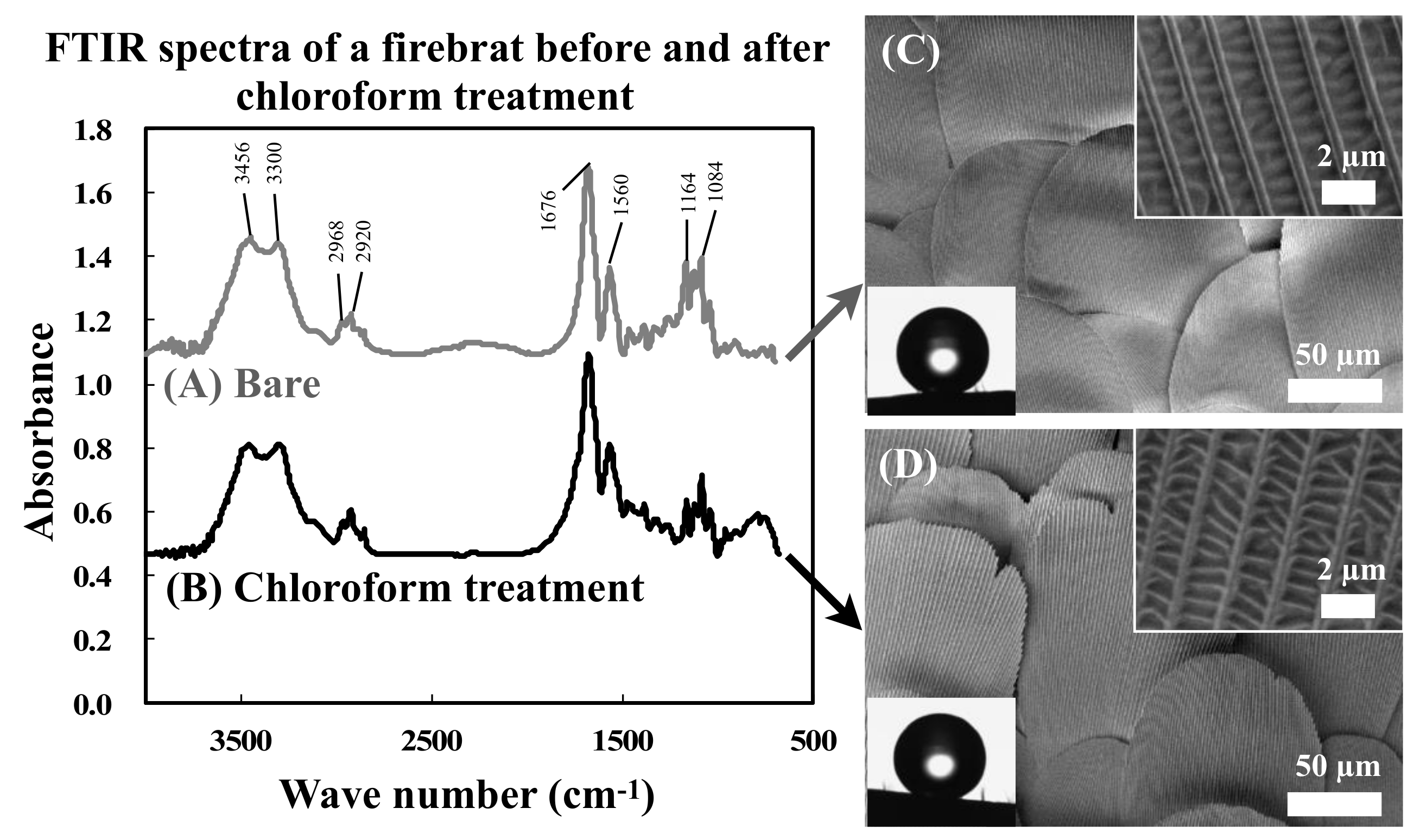

| Assignments | Wave Number (cm−1) | |
|---|---|---|
| Before Chloroform Treatment | After Chloroform Treatment | |
| ν (O–H) | 3456 | 3450 |
| ν (N–H) | 3300 | 3300 |
| ν (COCH3) | 2968 | 2966 |
| ν (C–H) | 2920 | 2922 |
| ν (C=O of N-acetyl group) | 1678 | 1678 |
| δ (N–H of N-acetyl group) | 1560 | 1562 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hirai, Y.; Okuda, N.; Saito, N.; Ogawa, T.; Machida, R.; Nomura, S.; Ôhara, M.; Haseyama, M.; Shimomura, M. The Friction Properties of Firebrat Scales. Biomimetics 2019, 4, 2. https://doi.org/10.3390/biomimetics4010002
Hirai Y, Okuda N, Saito N, Ogawa T, Machida R, Nomura S, Ôhara M, Haseyama M, Shimomura M. The Friction Properties of Firebrat Scales. Biomimetics. 2019; 4(1):2. https://doi.org/10.3390/biomimetics4010002
Chicago/Turabian StyleHirai, Yuji, Naoto Okuda, Naoki Saito, Takahiro Ogawa, Ryuichiro Machida, Shûhei Nomura, Masahiro Ôhara, Miki Haseyama, and Masatsugu Shimomura. 2019. "The Friction Properties of Firebrat Scales" Biomimetics 4, no. 1: 2. https://doi.org/10.3390/biomimetics4010002
APA StyleHirai, Y., Okuda, N., Saito, N., Ogawa, T., Machida, R., Nomura, S., Ôhara, M., Haseyama, M., & Shimomura, M. (2019). The Friction Properties of Firebrat Scales. Biomimetics, 4(1), 2. https://doi.org/10.3390/biomimetics4010002





