Synthesis of Optimized Molecularly Imprinted Polymers for the Isolation and Detection of Antidepressants via HPLC †
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Molecularly Imprinted Polymer Synthesis and Optimisation
2.3. Batch Rebinding Experiments Evaluated by UV–Vis Analysis
2.4. Batch Rebinding Experiments Evaluated by HPLC
3. Results and Discussion
3.1. Synthesis and Characterisation of Fluoxetine MIPs and NIPs
3.2. Monomer Optimization Study
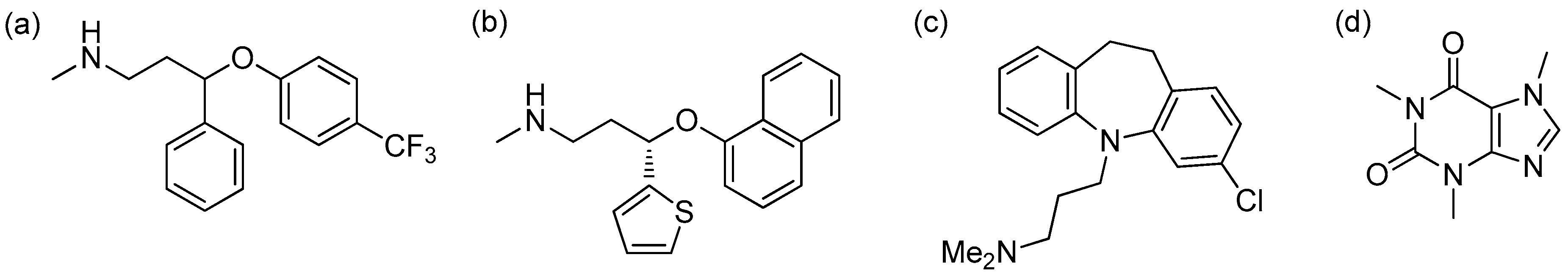
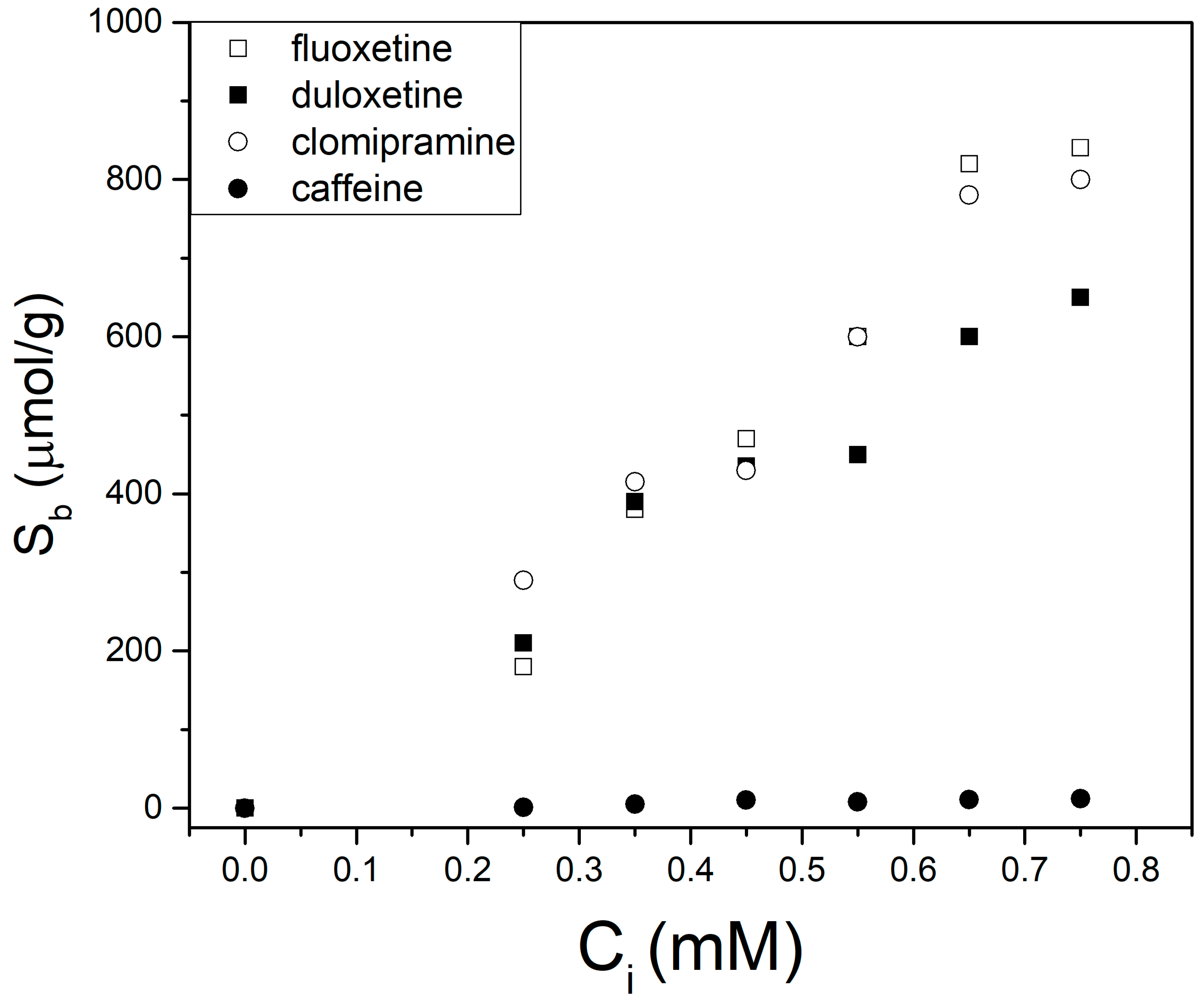
3.3. HPLC Analysis of SSRI Mixtures
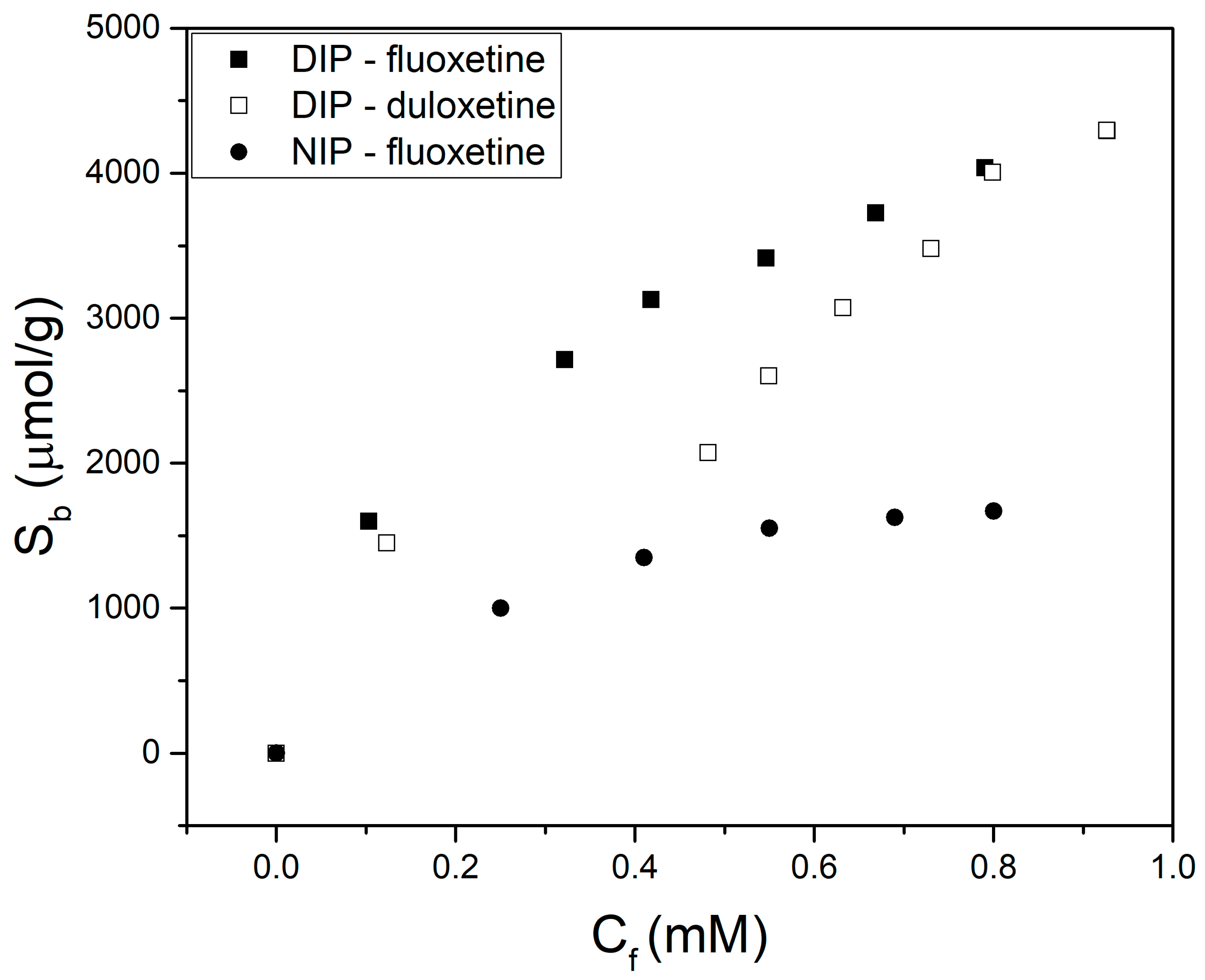
3.4. Dual Imprinted Polymer with Fluoxetine and Duloxetine
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bullard, I.; Prescribing and Medicines Team. Prescriptions Dispensed in the Community—Statistics for England, 2006–2016. Available online: https://digital.nhs.uk/data-and-information/publications/statistical/prescriptions-dispensed-in-the-community/prescriptions-dispensed-in-the-community-statistics-for-england-2006-2016-pas (accessed on 11 November 2018).
- Roberts, P.J. Fluoxetine. Drugs Future 1980, 5, 49. [Google Scholar] [CrossRef]
- Arroll, B.; Elley, C.R.; Fishman, T.; Goodyear-Smith, F.A.; Kenealy, T.; Blashki, G.; Kerse, N.; MacGillivray, S. Antidepressants versus placebo for depression in primary care. Cochrane Database Syst. Rev. 2009, 3, CD007954. [Google Scholar] [CrossRef] [PubMed]
- Fineberg, N.A.; Brown, A.; Reghunandanan, S.; Pampaloni, I. Evidence-based pharmacotherapy of obsessive-compulsive disorder. Int. J. Neuropsychopharmacol. 2012, 15, 1173–1191. [Google Scholar] [CrossRef] [PubMed]
- Flament, M.F.; Bissada, H.; Spettigue, W. Evidence-based pharmacotherapy of eating disorders. Int. J. Neuropsychopharmacol. 2012, 15, 189–207. [Google Scholar] [CrossRef] [PubMed]
- Batelaan, N.M.; Van Balkom, A.J.L.M.; Stein, D.J. Evidence-based pharmacotherapy of panic disorder: An update. Int. J. Neuropsychopharmacol. 2012, 15, 403–415. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Model Lists of Essential Medicines, 19th ed.; 2015; p. 51. [Google Scholar]
- Bound, J.P.; Voulvoulis, N. Household disposal of pharmaceuticals as a pathway for aquatic contamination in the United Kingdom. Environ. Health Perspect. 2005, 113, 1705–1711. [Google Scholar] [CrossRef] [PubMed]
- Weinberger, J.; Klaper, R. Environmental concentrations of the selective serotonin reuptake inhibitor fluoxetine impact specific behaviors involved in reproduction, feeding and predator avoidance in the fish Pimephales promelas (fathead minnow). Aquat. Toxicol. 2014, 151, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Brodin, T.; Piovano, S.; Fick, J.; Klaminder, J.; Heynen, M.; Jonsson, M. Ecological effects of pharmaceuticals in aquatic systems—Impacts through behavioural alterations. Philos. Trans. R. Soc. B Biol. Sci. 2014, 369, 20130580. [Google Scholar] [CrossRef] [PubMed]
- Chu, S.; Metcalfe, C.D. Analysis of paroxetine, fluoxetine and norfluoxetine in fish tissues using pressurized liquid extraction, mixed mode solid phase extraction cleanup and liquid chromatography–tandem mass spectrometry. J. Chromatogr. A 2007, 1163, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Brooks, B.W.; Chambliss, C.K.; Stanley, J.K.; Ramirez, A.; Banks, K.E.; Johnson, R.D.; Lewis, R.J. Determination of select antidepressants in fish from an effluent-dominated stream. Environ. Toxicol. Chem. 2005, 24, 464–469. [Google Scholar] [CrossRef] [PubMed]
- Queiroz, M.E.C.; Oliveira, E.B.; Breton, F.; Pawliszyn, J. Immunoaffinity in-tube solid phase microextraction coupled with liquid chromatography–mass spectrometry for analysis of fluoxetine in serum samples. J. Chromatogr. A 2007, 1174, 72–77. [Google Scholar] [CrossRef] [PubMed]
- Demeestere, K.; Petrović, M.; Gros, M.; Dewulf, J.; Van Langenhove, H.; Barceló, D. Trace analysis of antidepressants in environmental waters by molecularly imprinted polymer-based solid-phase extraction followed by ultra-performance liquid chromatography coupled to triple quadrupole mass spectrometry. Anal. Bioanal. Chem. 2010, 396, 825–837. [Google Scholar] [CrossRef] [PubMed]
- Crifasi, J.A.; Le, N.X.; Long, C. Simultaneous identification and quantitation of fluoxetine and its metabolite, norfluoxetine, in biological samples by GC–MS. J. Anal. Toxicol. 1997, 21, 415–419. [Google Scholar] [CrossRef] [PubMed]
- Shamsipur, M.; Dastjerdi, L.S.; Haghgoo, S.; Armspach, D.; Matt, D.; Aboul-Enein, H.Y. Chiral selectors for enantioresolution and quantitation of the antidepressant drug fluoxetine in pharmaceutical formulations by 19F NMR spectroscopic method. Anal. Chim. Acta 2007, 601, 130–138. [Google Scholar] [CrossRef] [PubMed]
- Kristoffersen, L.; Bugge, A.; Lundanes, E.; Slørdal, L. Simultaneous determination of citalopram, fluoxetine, paroxetine and their metabolites in plasma and whole blood by high-performance liquid chromatography with ultraviolet and fluorescence detection. J. Chromatogr. B Biomed. Sci. Appl. 1999, 734, 229–246. [Google Scholar] [CrossRef]
- Mosbach, K. Molecular imprinting. Trends Biochem. Sci. 1994, 19, 9–14. [Google Scholar] [CrossRef]
- Peeters, M.; Eersels, K.; Junkers, T.; Wagner, P. Molecularly imprinted polymers: Synthetic receptors for diagnostic medical devices. Synthetic receptors for diagnostic medical devices. In Molecularly Imprinted Catalysts: Principles, Syntheses, and Applications; Elsevier: Amsterdam, The Netherlands, 2015; pp. 253–271. ISBN 9780128013014. [Google Scholar]
- Nezhadali, A.; Motlagh, M.O.; Sadeghzadeh, S. Spectrophotometric determination of fluoxetine by molecularly imprinted polypyrrole and optimization by experimental design, artificial neural network and genetic algorithm. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2018, 190, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Barati, A.; Kazemi, E.; Dadfarnia, S.; Haji Shabani, A.M. Synthesis/characterization of molecular imprinted polymer based on magnetic chitosan/graphene oxide for selective separation/preconcentration of fluoxetine from environmental and biological samples. J. Ind. Eng. Chem. 2017, 46, 212–221. [Google Scholar] [CrossRef]
- Alizadeh, T.; Azizi, S. Graphene/graphite paste electrode incorporated with molecularly imprinted polymer nanoparticles as a novel sensor for differential pulse voltammetry determination of fluoxetine. Biosens. Bioelectron. 2016, 81, 198–206. [Google Scholar] [CrossRef] [PubMed]
- Buerge, I.J.; Poiger, T.; Müller, M.D.; Buser, H.-R. Caffeine, an anthropogenic marker for wastewater contamination of surface waters. Environ. Sci. Technol. 2003, 37, 691–700. [Google Scholar] [CrossRef] [PubMed]
- Casadio, S.; Lowdon, J.W.; Betlem, K.; Ueta, J.T.; Foster, C.W.; Cleij, T.J.; van Grinsven, B.; Sutcliffe, O.B.; Banks, C.E.; Peeters, M. Development of a novel flexible polymer-based biosensor platform for the thermal detection of noradrenaline in aqueous solutions. Chem. Eng. J. 2017, 315, 459–468. [Google Scholar] [CrossRef]
| Molecularly Imprinted Polymer | Monomer |
|---|---|
| MIP-1 | MAA |
| MIP-2 | IA |
| MIP-3 | HEMA |
| MIP-4 | Acrylamide |
| MIP-5 | Acrylonitrile |
| MIP-6 | MAA + IA (1:1) |
| MIP-7 | MAA + HEMA (1:1) |
| Polymer | Binding at Cf = 0.05 mM | Imprint Factor (Cf = 0.05 mM) | Binding at Cf = 0.2 mM (Sb/Cf) | Imprint Factor (Cf = 0.2 mM) |
|---|---|---|---|---|
| MIP-1 | 1240 | 2.1 | 1250 | 2.2 |
| NIP-1 | 600 | 575 | ||
| MIP-2 | 900 | 5.6 | 850 | 6.3 |
| NIP-2 | 160 | 135 | ||
| MIP-3 | 1320 | 2.8 | 1300 | 2.8 |
| NIP-3 | 480 | 465 | ||
| MIP-4 | 360 | 2.6 | 370 | 2.6 |
| NIP-4 | 140 | 145 | ||
| MIP-5 | 300 | 1.1 | 290 | 1.1 |
| NIP-5 | 260 | 255 | ||
| MIP-6 | 3600 | 7.2 | 3555 | 7.2 |
| NIP-6 | 500 | 495 | ||
| MIP-7 | 520 | 2.0 | 525 | 1.5 |
| NIP-7 | 320 | 340 |
| Compounds Mixture | IF for Fluoxetine | MIP/NIP Comparison Compounds |
|---|---|---|
| Fluoxetine:Duloxetine | 1.45 | 1.26 |
| Fluoxetine:Clomipramine | 1.58 | 1.37 |
| Fluoxetine:Caffeine | 2.19 | 0.71 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hudson, A.D.; Solà, R.; Ueta, J.T.; Battell, W.; Jamieson, O.; Dunbar, T.; Maciá, B.; Peeters, M. Synthesis of Optimized Molecularly Imprinted Polymers for the Isolation and Detection of Antidepressants via HPLC. Biomimetics 2019, 4, 18. https://doi.org/10.3390/biomimetics4010018
Hudson AD, Solà R, Ueta JT, Battell W, Jamieson O, Dunbar T, Maciá B, Peeters M. Synthesis of Optimized Molecularly Imprinted Polymers for the Isolation and Detection of Antidepressants via HPLC. Biomimetics. 2019; 4(1):18. https://doi.org/10.3390/biomimetics4010018
Chicago/Turabian StyleHudson, Alexander D., Ricard Solà, Jorge T. Ueta, William Battell, Oliver Jamieson, Thomas Dunbar, Beatriz Maciá, and Marloes Peeters. 2019. "Synthesis of Optimized Molecularly Imprinted Polymers for the Isolation and Detection of Antidepressants via HPLC" Biomimetics 4, no. 1: 18. https://doi.org/10.3390/biomimetics4010018
APA StyleHudson, A. D., Solà, R., Ueta, J. T., Battell, W., Jamieson, O., Dunbar, T., Maciá, B., & Peeters, M. (2019). Synthesis of Optimized Molecularly Imprinted Polymers for the Isolation and Detection of Antidepressants via HPLC. Biomimetics, 4(1), 18. https://doi.org/10.3390/biomimetics4010018






