Mitigating Food Protein Allergenicity with Biopolymers, Bioactive Compounds, and Enzymes
Abstract
1. Introduction
2. Current Research and Knowledge on Managing Allergic Reactions to Foods, Biopolymers, Bioactive Compounds, or Enzymes
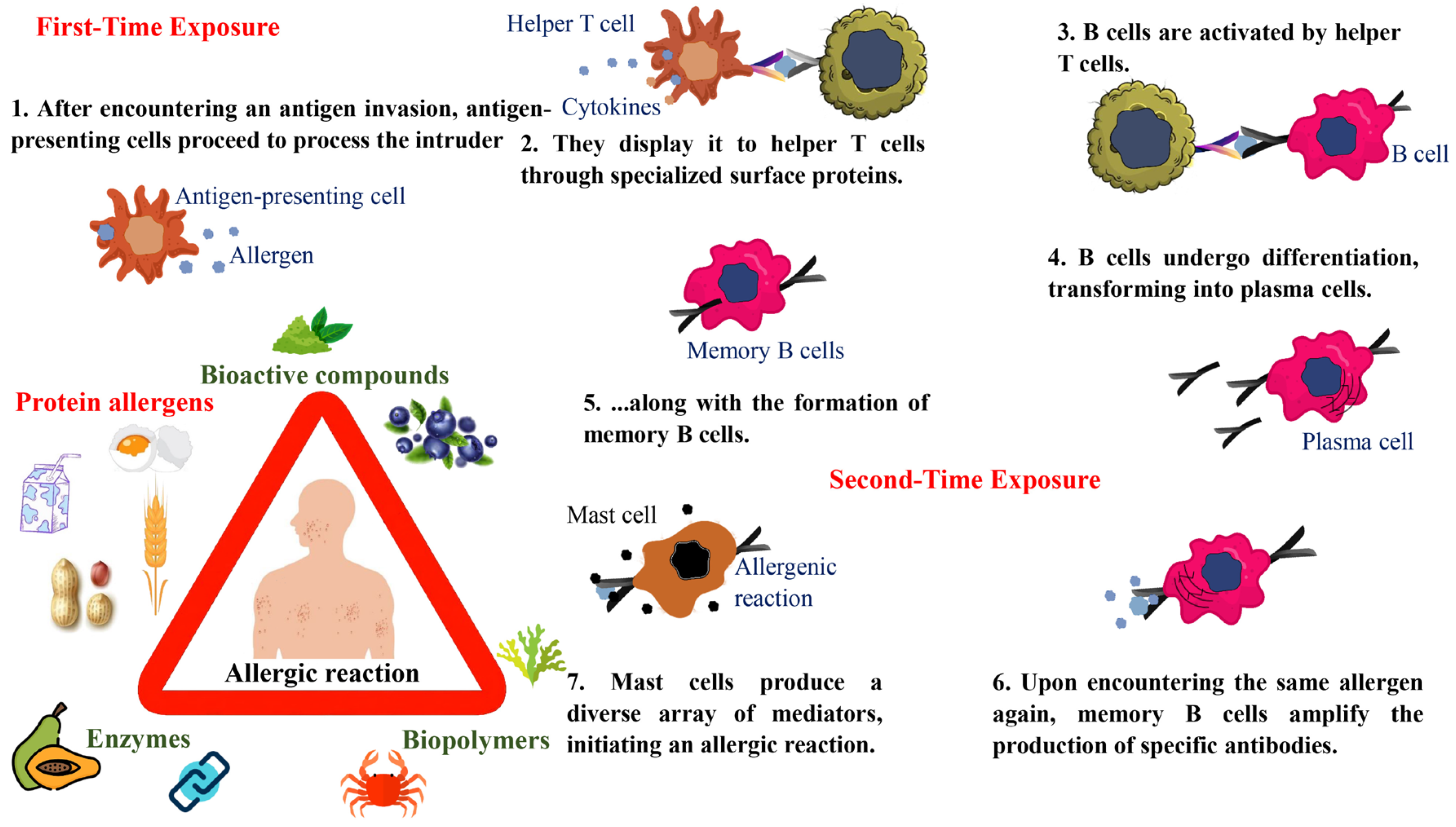
2.1. Managing Food Allergies: Mechanisms, Symptoms, and Identification
2.2. The Importance of Understanding the Molecular and Structural Characteristics of Food Allergens and the Compounds That Influence Them
2.3. Novel Approaches to Mitigating Allergic Reactions: Biopolymer–Allergen and Bioactive–Allergen Interactions and Enzymatic Strategies
2.3.1. Structural Modification of Allergen Proteins to Reduce Allergenicity
Biopolymer–Allergen Interactions
- Chitosan
- 2.
- Alginate (ALG)
- 3.
- Pectin
Protease–Enzyme Interactions
Transglutaminase (TG) Interactions
2.3.2. Interaction of Bioactive Compound with Allergen Proteins to Lessen Symptoms
Quercetin (QU)
Resveratrol
Curcumin (Cn)
Epigallocatechin Gallate (EGCG)
2.4. New Insights and Recommendations for Preventing Food Allergies
2.5. Future Prospects for Preventing Food Allergy
3. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- Sealey-Voyksner, J.A.; Khosla, C.; Voyksner, R.D.; Jorgenson, J.W. Novel aspects of quantitation of immunogenic wheat gluten peptides by liquid chromatography–mass spectrometry/mass spectrometry. J. Chromatogr. A 2010, 1217, 4167–4183. [Google Scholar] [CrossRef] [PubMed]
- Fox, A.; Brown, T.; Walsh, J.; Venter, C.; Meyer, R.; Nowak-Wegrzyn, A.; Levin, M.; Spawls, H.; Beatson, J.; Lovis, M.-T. An update to the milk allergy in primary care guideline. Clin. Transl. Allergy 2019, 9, 40. [Google Scholar] [CrossRef] [PubMed]
- Vereda, A.; van Hage, M.; Ahlstedt, S.; Ibañez, M.D.; Cuesta-Herranz, J.; van Odijk, J.; Wickman, M.; Sampson, H.A. Peanut allergy: Clinical and immunologic differences among patients from 3 different geographic regions. J. Allergy Clin. Immunol. 2011, 127, 603–607. [Google Scholar] [CrossRef] [PubMed]
- Patel, B.Y.; Volcheck, G.W. Food allergy: Common causes, diagnosis, and treatment. Mayo Clin. Proc. 2015, 90, 1411–1419. [Google Scholar] [CrossRef]
- Jensen-Jarolim, E. Gender effects in allergology–secondary publications and update. World Allergy Organ. J. 2017, 10, 47. [Google Scholar] [CrossRef]
- Center for Allergy and Asthma of Georgia. Do Men Suffer From Allergies More Than Women? 2021. Available online: https://www.caageorgia.com/about-us/blog/2021/may/do-men-suffer-from-allergies-more-than-women-/ (accessed on 4 May 2021).
- Wang, J.; Guo, X.; Chen, C.; Sun, S.; Liu, G.; Liu, M.; Hao, M.; Che, H. Gender differences in food allergy depend on the PPAR γ/NF-κB in the intestines of mice. Life Sci. 2021, 278, 119606. [Google Scholar] [CrossRef]
- Silverberg, J.I. Racial and ethnic disparities in atopic dermatitis. Curr. Dermatol. Rep. 2015, 4, 44–48. [Google Scholar] [CrossRef]
- Guilbert, T.; Zeiger, R.S.; Haselkorn, T.; Iqbal, A.; Alvarez, C.; Mink, D.R.; Chipps, B.E.; Szefler, S.J. Racial disparities in asthma-related health outcomes in children with severe/difficult-to-treat asthma. J. Allergy Clin. Immunol. Pract. 2019, 7, 568–577. [Google Scholar] [CrossRef]
- Sicherer, S.H.; Sampson, H.A. Food allergy: A review and update on epidemiology, pathogenesis, diagnosis, prevention, and management. J. Allergy Clin. Immunol. 2018, 141, 41–58. [Google Scholar] [CrossRef]
- Cox, L.; Nelson, H.; Lockey, R.; Calabria, C.; Chacko, T.; Finegold, I.; Nelson, M.; Weber, R.; Bernstein, D.I.; Blessing-Moore, J. Allergen immunotherapy: A practice parameter third update. J. Allergy Clin. Immunol. 2011, 127, S1–S55. [Google Scholar] [CrossRef]
- Fiocchi, A.; Pawankar, R.; Cuello-Garcia, C.; Ahn, K.; Al-Hammadi, S.; Agarwal, A.; Beyer, K.; Burks, W.; Canonica, G.W.; Ebisawa, M. World Allergy Organization-McMaster University guidelines for allergic disease prevention (GLAD-P): Probiotics. World Allergy Organ. J. 2015, 8, 4. [Google Scholar] [CrossRef] [PubMed]
- Alghamdi, R.; Alshaier, R.; Alotaibi, A.; Almutairi, A.; Alotaibi, G.; Faqeeh, A.; Almalki, A.; AbdulMajed, H. Immunotherapy effectiveness in treating peanut hypersensitivity: A systemic review. Cureus 2022, 14, e21832. [Google Scholar] [CrossRef] [PubMed]
- Grossman, M.R. Sesame: A Major Food Allergen in the United States. Eur. Food Feed L. Rev. 2022, 17, 159. [Google Scholar]
- Burks, A.W.; Sampson, H.A.; Plaut, M.; Lack, G.; Akdis, C.A. Treatment for food allergy. J. Allergy Clin. Immunol. 2018, 141, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Pi, X.; Yang, Y.; Sun, Y.; Cui, Q.; Wan, Y.; Fu, G.; Chen, H.; Cheng, J. Recent advances in alleviating food allergenicity through fermentation. Crit. Rev. Food Sci. Nutr. 2022, 62, 7255–7268. [Google Scholar] [CrossRef] [PubMed]
- Shriver, S.K.; Yang, W.W. Thermal and nonthermal methods for food allergen control. Food Eng. Rev. 2011, 3, 26–43. [Google Scholar] [CrossRef]
- Ekezie, F.-G.C.; Cheng, J.-H.; Sun, D.-W. Effects of nonthermal food processing technologies on food allergens: A review of recent research advances. Trends Food Sci. Technol. 2018, 74, 12–25. [Google Scholar] [CrossRef]
- Du Toit, G.; Roberts, G.; Sayre, P.H.; Bahnson, H.T.; Radulovic, S.; Santos, A.F.; Brough, H.A.; Phippard, D.; Basting, M.; Feeney, M. Randomized trial of peanut consumption in infants at risk for peanut allergy. N. Engl. J. Med. 2015, 372, 803–813. [Google Scholar] [CrossRef]
- Logan, K.; Bahnson, H.T.; Ylescupidez, A.; Beyer, K.; Bellach, J.; Campbell, D.E.; Craven, J.; Du Toit, G.; Clare Mills, E.; Perkin, M.R. Early introduction of peanut reduces peanut allergy across risk groups in pooled and causal inference analyses. Allergy 2023, 78, 1307–1318. [Google Scholar] [CrossRef]
- Lopata, A.L.; O’hehir, R.; Lehrer, S. Shellfish allergy. Clin. Exp. Allergy 2010, 40, 850–858. [Google Scholar] [CrossRef]
- McWilliam, V.; Koplin, J.; Lodge, C.; Tang, M.; Dharmage, S.; Allen, K. The prevalence of tree nut allergy: A systematic review. Curr. Allergy Asthma Rep. 2015, 15, 54. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.; Edwards-Salmon, S.; Vickery, B.P. Current and future treatments for peanut allergy. Clin. Exp. Allergy 2023, 53, 10–24. [Google Scholar] [CrossRef] [PubMed]
- Campbell, R.L.; Hagan, J.B.; Manivannan, V.; Decker, W.W.; Kanthala, A.R.; Bellolio, M.F.; Smith, V.D.; Li, J.T. Evaluation of national institute of allergy and infectious diseases/food allergy and anaphylaxis network criteria for the diagnosis of anaphylaxis in emergency department patients. J. Allergy Clin. Immunol. 2012, 129, 748–752. [Google Scholar] [CrossRef] [PubMed]
- Sabaghi, M.; Jamali, S.N. Advancements, challenges, and future prospects of nanobiosensors in food packaging for allergen detection. J. Food Meas. Charact. 2024, 18, 3444–3457. [Google Scholar] [CrossRef]
- Pasha, I.; Saeed, F.; Sultan, M.T.; Batool, R.; Aziz, M.; Ahmed, W. Wheat allergy and intolerence; Recent updates and perspectives. Crit. Rev. Food Sci. Nutr. 2016, 56, 13–24. [Google Scholar] [CrossRef]
- Kattan, J.D.; Cocco, R.R.; Järvinen, K.M. Milk and soy allergy. Pediatr. Clin. 2011, 58, 407–426. [Google Scholar] [CrossRef]
- Caubet, J.-C.; Wang, J. Current understanding of egg allergy. Pediatr. Clin. 2011, 58, 427–443. [Google Scholar] [CrossRef]
- Togias, A.; Cooper, S.F.; Acebal, M.L.; Assa’ad, A.; Baker, J.R.; Beck, L.A.; Block, J.; Byrd-Bredbenner, C.; Chan, E.S.; Eichenfield, L.F. Addendum guidelines for the prevention of peanut allergy in the United States: Report of the National Institute of Allergy and Infectious Diseases–sponsored expert panel. World Allergy Organ. J. 2017, 139, 29–44. [Google Scholar]
- Weiler, J.M.; Bonini, S.; Coifman, R.; Craig, T.; Delgado, L.; Capão-Filipe, M.; Passali, D.; Randolph, C.; Storms, W. American Academy of Allergy, Asthma & Immunology work group report: Exercise-induced asthma. J. Allergy Clin. Immunol. 2007, 119, 1349–1358. [Google Scholar]
- Hafsa, J.; Smach, M.A.; Mrid, R.B.; Sobeh, M.; Majdoub, H.; Yasri, A. Functional properties of chitosan derivatives obtained through Maillard reaction: A novel promising food preservative. Food Chem. 2021, 349, 129072. [Google Scholar] [CrossRef]
- Maleki, S.J.; Crespo, J.F.; Cabanillas, B. Anti-inflammatory effects of flavonoids. Food Chem. 2019, 299, 125124. [Google Scholar] [CrossRef] [PubMed]
- Kurup, V.P.; Barrios, C.S. Immunomodulatory effects of curcumin in allergy. Mol. Nutr. Food Res. 2008, 52, 1031–1039. [Google Scholar] [CrossRef] [PubMed]
- Pi, X.; Sun, Y.; Cheng, J.; Fu, G.; Guo, M. A review on polyphenols and their potential application to reduce food allergenicity. Crit. Rev. Food Sci. Nutr. 2022, 63, 10014–10031. [Google Scholar] [CrossRef]
- Maleki, S.J.; Viquez, O.; Jacks, T.; Dodo, H.; Champagne, E.T.; Chung, S.-Y.; Landry, S.J. The major peanut allergen, Ara h 2, functions as a trypsin inhibitor, and roasting enhances this function. J. Allergy Clin. Immunol. 2003, 112, 190–195. [Google Scholar] [CrossRef]
- Akkerdaas, J.; Totis, M.; Barnett, B.; Bell, E.; Davis, T.; Edrington, T.; Glenn, K.; Graser, G.; Herman, R.; Knulst, A. Protease resistance of food proteins: A mixed picture for predicting allergenicity but a useful tool for assessing exposure. Clin. Transl. Allergy 2018, 8, 30. [Google Scholar] [CrossRef]
- Sabaghi, M.; Maghsoudlou, Y.; Kashiri, M.; Shakeri, A. Evaluation of release mechanism of catechin from chitosan-polyvinyl alcohol film by exposure to gamma irradiation. Carbohydr. Polym. 2020, 230, 115589. [Google Scholar] [CrossRef]
- Roy, K.; Mao, H.-Q.; Huang, S.-K.; Leong, K.W. Oral gene delivery with chitosan–DNA nanoparticles generates immunologic protection in a murine model of peanut allergy. Nat. Med. 1999, 5, 387–391. [Google Scholar] [CrossRef]
- Jiang, T.; Ji, H.; Zhang, L.; Wang, Y.; Zhou, H. Chitosan oligosaccharide exerts anti-allergic effect against shrimp tropomyosin-induced food allergy by affecting Th1 and Th2 cytokines. Int. Arch. Allergy Immunol. 2019, 180, 10–16. [Google Scholar] [CrossRef]
- Li, F.; Wang, L.; Jin, X.-M.; Yan, C.-H.; Jiang, S.; Shen, X.-M. The immunologic effect of TGF-beta1 chitosan nanoparticle plasmids on ovalbumin-induced allergic BALB/c mice. Immunobiology 2009, 214, 87–99. [Google Scholar] [CrossRef]
- Fu, L.; Wang, C.; Wang, J.; Ni, S.; Wang, Y. Maillard reaction with ribose, galacto-oligosaccharide or chitosan-oligosaccharide reduced the allergenicity of shrimp tropomyosin by inducing conformational changes. Food Chem. 2019, 274, 789–795. [Google Scholar] [CrossRef]
- Maleki, S.J.; Chung, S.-Y.; Champagne, E.T.; Raufman, J.-P. The effects of roasting on the allergenic properties of peanut proteins. J. Allergy Clin. Immunol. 2000, 106, 763–768. [Google Scholar] [CrossRef] [PubMed]
- Usui, M.; Tamura, H.; Nakamura, K.; Ogawa, T.; Muroshita, M.; Azakami, H.; Kanuma, S.; Kato, A. Enhanced bactericidal action and masking of allergen structure of soy protein by attachment of chitosan through Maillard-type protein-polysaccharide conjugation. Food/Nahr. 2004, 48, 69–72. [Google Scholar] [CrossRef] [PubMed]
- Chung, M.J.; Park, J.K.; Park, Y.I. Anti-inflammatory effects of low-molecular weight chitosan oligosaccharides in IgE–antigen complex-stimulated RBL-2H3 cells and asthma model mice. Int. Immunopharmacol. 2012, 12, 453–459. [Google Scholar] [CrossRef]
- Yu, B.; Bi, D.; Yao, L.; Li, T.; Gu, L.; Xu, H.; Li, X.; Li, H.; Hu, Z.; Xu, X. The inhibitory activity of alginate against allergic reactions in an ovalbumin-induced mouse model. Food Funct. 2020, 11, 2704–2713. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Zeng, Q.; Zhang, Y.; Yin, Q.; Zhu, X.; Zhang, P.; Wang, C.; Liu, J. Effects of fucoidans and alginates from Sargassum graminifolium on allergic symptoms and intestinal microbiota in mice with OVA-induced food allergy. Food Funct. 2022, 13, 6702–6715. [Google Scholar] [CrossRef]
- Li, J.; Zou, C.; Liu, Y. Amelioration of ovalbumin-induced food allergy in mice by targeted rectal and colonic delivery of cyanidin-3-O-glucoside. Foods 2022, 11, 1542. [Google Scholar] [CrossRef]
- Peyron, S.; Mouécoucou, J.; Frémont, S.; Sanchez, C.; Gontard, N. Effects of heat treatment and pectin addition on β-lactoglobulin allergenicity. J. Agric. Food Chem. 2006, 54, 5643–5650. [Google Scholar] [CrossRef]
- Jiménez-Saiz, R.; López-Expósito, I.; Molina, E.; López-Fandiño, R. IgE-binding and in vitro gastrointestinal digestibility of egg allergens in the presence of polysaccharides. Food Hydrocoll. 2013, 30, 597–605. [Google Scholar] [CrossRef]
- Kopper, R.A.; Odum, N.J.; Sen, M.; Helm, R.M.; Stanley, J.S.; Burks, A.W. Peanut protein allergens: Gastric digestion is carried out exclusively by pepsin. J. Allergy Clin. Immunol. 2004, 114, 614–618. [Google Scholar] [CrossRef]
- Yu, J.; Mikiashvili, N. Effectiveness of different proteases in reducing allergen content and IgE-binding of raw peanuts. Food Chem. 2020, 307, 125565. [Google Scholar] [CrossRef]
- Piersma, S.R.; Gaspari, M.; Hefle, S.L.; Koppelman, S.J. Proteolytic processing of the peanut allergen Ara h 3. Mol. Nutr. Food Res. 2005, 49, 744–755. [Google Scholar] [CrossRef] [PubMed]
- Feng, N.; Zhang, H.; Li, Y.; Liu, Y.; Xu, L.; Wang, Y.; Fei, X.; Tian, J. A novel catalytic material for hydrolyzing cow’s milk allergenic proteins: Papain-Cu3 (PO4) 2· 3H2O-magnetic nanoflowers. Food Chem. 2020, 311, 125911. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Luo, Y.; Li, Z. Effects of pH, temperature, enzyme-to-substrate ratio and reaction time on the antigenicity of casein hydrolysates prepared by papain. Food Agric. Immunol. 2012, 23, 69–82. [Google Scholar] [CrossRef]
- Nath, A.; Ahmad, A.S.; Amankwaa, A.; Csehi, B.; Mednyánszky, Z.; Szerdahelyi, E.; Tóth, A.; Tormási, J.; Truong, D.H.; Abrankó, L. Hydrolysis of Soybean Milk Protein by Papain: Antioxidant, Anti-Angiotensin, Antigenic and Digestibility Perspectives. Bioengineering 2022, 9, 418. [Google Scholar] [CrossRef]
- Quintieri, L.; Monaci, L.; Baruzzi, F.; Giuffrida, M.G.; de Candia, S.; Caputo, L. Reduction of whey protein concentrate antigenicity by using a combined enzymatic digestion and ultrafiltration approach. J. Food Sci. Technol. 2017, 54, 1910–1916. [Google Scholar] [CrossRef]
- Meinlschmidt, P.; Schweiggert-Weisz, U.; Eisner, P. Soy protein hydrolysates fermentation: Effect of debittering and degradation of major soy allergens. LWT-Food Sci. Technol. 2016, 71, 202–212. [Google Scholar] [CrossRef]
- Medeiros, V.; Rainha, N.; Paiva, L.; Lima, E.; Baptista, J. Bovine milk formula based on partial hydrolysis of caseins by bromelain enzyme: Better digestibility and angiotensin-converting enzyme-inhibitory properties. Int. J. Food Prop. 2014, 17, 806–817. [Google Scholar] [CrossRef]
- Villas-Boas, M.B.; Benedé, S.; de Lima Zollner, R.; Netto, F.M.; Molina, E. Epitopes resistance to the simulated gastrointestinal digestion of β-lactoglobulin submitted to two-step enzymatic modification. Food Res. Int. 2015, 72, 191–197. [Google Scholar] [CrossRef]
- Arteaga, V.G.; Guardia, M.A.; Muranyi, I.; Eisner, P.; Schweiggert-Weisz, U. Effect of enzymatic hydrolysis on molecular weight distribution, techno-functional properties and sensory perception of pea protein isolates. Innov. Food Sci. Emerg. Technol. 2020, 65, 102449. [Google Scholar] [CrossRef]
- Watanabe, M.; Watanabe, J.; Sonoyama, K.; Tanabe, S. Novel method for producing hypoallergenic wheat flour by enzymatic fragmentation of the constituent allergens and its application to food processing. Biosci. Biotechnol. Biochem. 2000, 64, 2663–2667. [Google Scholar] [CrossRef]
- Fotschki, J.; Wróblewska, B.; Fotschki, B.; Kalicki, B.; Rigby, N.; Mackie, A. Microbial transglutaminase alters the immunogenic potential and cross-reactivity of horse and cow milk proteins. J. Dairy Sci. 2020, 103, 2153–2166. [Google Scholar] [CrossRef] [PubMed]
- Meng, S.; Tan, Y.; Chang, S.; Li, J.; Maleki, S.; Puppala, N. Peanut allergen reduction and functional property improvement by means of enzymatic hydrolysis and transglutaminase crosslinking. Food Chem. 2020, 302, 125186. [Google Scholar] [CrossRef] [PubMed]
- Leszczyńska, J.; Łącka, A.; Bryszewska, M. The use of transglutaminase in the reduction of immunoreactivity of wheat flour. Food Agric. Immunol. 2006, 17, 105–113. [Google Scholar] [CrossRef]
- Villas-Boas, M.B.; Vieira, K.P.; Trevizan, G.; de Lima Zollner, R.; Netto, F.M. The effect of transglutaminase-induced polymerization in the presence of cysteine on β-lactoglobulin antigenicity. Int. Dairy J. 2010, 20, 386–392. [Google Scholar] [CrossRef]
- Yuan, F.; Lv, L.; Li, Z.; Mi, N.; Chen, H.; Lin, H. Effect of transglutaminase-catalyzed glycosylation on the allergenicity and conformational structure of shrimp (Metapenaeus ensis) tropomyosin. Food Chem. 2017, 219, 215–222. [Google Scholar] [CrossRef]
- Tian, Y.; Liu, C.; Xue, W.; Wang, Z. Crosslinked recombinant-Ara h 1 catalyzed by microbial transglutaminase: Preparation, structural characterization and allergic assessment. Foods 2020, 9, 1508. [Google Scholar] [CrossRef]
- Zhang, T.; Hu, Z.; Cheng, Y.; Xu, H.; Velickovic, T.C.; He, K.; Sun, F.; He, Z.; Liu, Z.; Wu, X. Changes in Allergenicity of Ovalbumin in Vitro and in Vivo on Conjugation with Quercetin. J. Agric. Food Chem. 2020, 68, 4027–4035. [Google Scholar] [CrossRef]
- Wang, Q.; Tang, Y.; Yang, Y.; Zhao, J.; Zhang, Y.; Li, L.; Wang, Q.; Ming, J. Interaction between wheat gliadin and quercetin under different pH conditions analyzed by multi-spectroscopy methods. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2020, 229, 117937. [Google Scholar] [CrossRef]
- Shishehbor, F.; Behrou, L.; Ghaforian, B.M.; Namjouyan, F.; Latifi, S.-M. Quercetin effectively quells peanut-induced anaphylactic reactions in the peanut sensitized rats. IJAAI 2010, 9, 27–34. [Google Scholar]
- Yu, M.; Liu, H.; Shi, A.; Liu, L.; Wang, Q. Preparation of resveratrol-enriched and poor allergic protein peanut sprout from ultrasound treated peanut seeds. Ultrason. Sonochem. 2016, 28, 334–340. [Google Scholar] [CrossRef]
- Zhang, Y.-F.; Liu, Q.-M.; Gao, Y.-Y.; Liu, B.; Liu, H.; Cao, M.-J.; Yang, X.-W.; Liu, G.-M. Attenuation of allergic responses following treatment with resveratrol in anaphylactic models and IgE-mediated mast cells. Food Funct. 2019, 10, 2030–2039. [Google Scholar] [CrossRef] [PubMed]
- Okada, Y.; Oh-oka, K.; Nakamura, Y.; Ishimaru, K.; Matsuoka, S.; Okumura, K.; Ogawa, H.; Hisamoto, M.; Okuda, T.; Nakao, A. Dietary resveratrol prevents the development of food allergy in mice. PLoS ONE 2012, 7, e44338. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wang, B.; Luo, Y.; Zhang, Q.; Bian, Y.; Wang, R. Resveratrol-mediated SIRT1 activation attenuates ovalbumin-induced allergic rhinitis in mice. Mol. Immunol. 2020, 122, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Fuloria, S.; Mehta, J.; Chandel, A.; Sekar, M.; Rani, N.N.I.M.; Begum, M.Y.; Subramaniyan, V.; Chidambaram, K.; Thangavelu, L.; Nordin, R. A comprehensive review on the therapeutic potential of Curcuma longa Linn. in relation to its major active constituent curcumin. Front. Pharmacol. 2022, 13, 820806. [Google Scholar] [CrossRef]
- Kinney, S.R.; Carlson, L.; Ser-Dolansky, J.; Thompson, C.; Shah, S.; Gambrah, A.; Xing, W.; Schneider, S.S.; Mathias, C.B. Curcumin ingestion inhibits mastocytosis and suppresses intestinal anaphylaxis in a murine model of food allergy. PLoS ONE 2015, 10, e0132467. [Google Scholar] [CrossRef]
- Krajewski, D.; Polukort, S.H.; Gelzinis, J.; Rovatti, J.; Kaczenski, E.; Galinski, C.; Pantos, M.; Shah, N.N.; Schneider, S.S.; Kennedy, D.R. Protein disulfide isomerases regulate IgE-mediated mast cell responses and their inhibition confers protective effects during food allergy. Front. Immunol. 2020, 11, 606837. [Google Scholar] [CrossRef]
- Li, W.; Li, S.; Hu, Y.; Zhou, M.; Wang, C.; Li, D.; Li, D. Impact of hot alkali modification conditions on secondary structure of peanut protein and embedding rate of curcumin. Food Sci. Hum. Wellness 2019, 8, 283–291. [Google Scholar] [CrossRef]
- Li, X.; Li, M.; Zhang, T.; McClements, D.J.; Liu, X.; Wu, X.; Liu, F. Enzymatic and nonenzymatic conjugates of lactoferrin and (−)-epigallocatechin gallate: Formation, structure, functionality, and allergenicity. J. Agric. Food Chem. 2021, 69, 6291–6302. [Google Scholar] [CrossRef]
- Zhang, X.; Li, S.; Shao, X.; Li, M.; Hemar, Y. Probing the conjugation of epigallocatechin gallate with β-lactoglobulin and its in vivo desensitization efficiency. Food Funct. 2021, 12, 11343–11350. [Google Scholar] [CrossRef]
- Pi, X.; Liu, J.; Sun, Y.; Sun, X.; Sun, Z.; Cheng, J.; Guo, M. Investigation of the differences in the effect of (−)-epigallocatechin gallate and proanthocyanidins on the functionality and allergenicity of soybean protein isolate. Food Chem. X 2023, 17, 100566. [Google Scholar] [CrossRef]
- Fu, M.; Fu, S.; Ni, S.; Zou, L.; Liu, Y.; Hong, T. Anti-inflammatory effect of epigallocatechin gallate in a mouse model of ovalbumin-induced allergic rhinitis. Int. Immunopharmacol. 2017, 49, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.-D.; Huang, L.; Meng, L.; Lin, Y.-F.; Xu, X.; Dong, M.-S. Soy protein isolate-(-)-epigallocatechin gallate conjugate: Covalent binding sites identification and IgE binding ability evaluation. Food Chem. 2020, 333, 127400. [Google Scholar] [CrossRef]
- Xu, W.; Song, X.; Qu, Q.; Gong, Z.; Xiao, W. Synergistic effects of l-theanine and epigallocatechin gallate in alleviating ovalbumin allergy by regulating intestinal immunity through inhibition of mast cell degranulation. Food Funct. 2023, 14, 2059–2073. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Yu, X.; Tian, L.; Cong, Y.; Li, L. Therapeutic effects of epigallocatechin and epigallocatechin gallate on the allergic reaction of αs1-casein sensitized mice. Food Sci. Hum. Wellness 2023, 12, 882–888. [Google Scholar] [CrossRef]
| Biopolymer | Study/Experiment | Results | Target | Ref. |
|---|---|---|---|---|
| CS | Oral administration of CS DNA nanoparticles synthesized by combining plasmid DNA and CS in mice | Reduced levels of allergen-induced anaphylaxis, IgE, plasma histamine, and vascular leakage. | Peanut | [38] |
| CS | Treatment of a mouse model of food allergy with chitosan COS | Improved severe food allergy symptoms and reduced serum IgE and IgG1 levels, while increasing IgG2a levels. | Shrimp | [39] |
| CS | Oral administration of TGF-beta-expressing DNA vector in CS nanoparticles in mice | Sustained increase in TGF-beta protein in mouse intestinal tissue and improved food allergy symptoms induced by OVA. | Egg | [40] |
| CS | Use of Maillard reaction with reducing sugars to eliminate allergenicity of TM | All three sugars significantly reduced allergenicity, with COS showing the highest efficiency. | Shellfish | [41,42] |
| CS | Maillard-type soy protein–CS conjugate | Enhanced antimicrobial activity, improved emulsifying properties, and reduced allergenicity. | Soybean | [43] |
| CS | Anti-inflammatory effects of LM-COS in allergic inflammatory | LM-COS reduced levels of pro-inflammatory cytokines in OVA-sensitized/challenged allergic inflammatory model mice. | Egg | [44] |
| ALG | Use in a mouse model | ALG effectively reduced allergic reactions, inhibited mast cell degranulation, and improved intestinal epithelial villi integrity. | Egg | [45] |
| SG fucoidans and ALGs | Study on mice with food allergies | Reduced allergy symptoms, diarrhea, and jejunum injury. Down-regulated OVA-specific IgE and TNF-α levels. | Egg | [46] |
| C3G | Study on targeted rectal and colonic delivery via enteric sodium ALG in ameliorating OVA allergies | Enhanced intestinal microecological homeostasis, improved intestinal epithelial barrier function, and balanced Th1/Th2 immune responses. | Egg | [47] |
| Pectin | Effects of heat treatment and pectin addition on allergenicity of BLG | Heat denaturation increased protein’s susceptibility to proteolysis, reducing immunoreactivity. Pectin addition reduced accessibility to cleavage sites/epitope sequences via nonspecific interactions. | Milk | [48] |
| Pectin, gum arabic, and xylan | Effect of pectin, gum arabic, and xylan on reactivity of OVA and OM with human IgE | Polysaccharides increased reactivity of OVA and OM with human IgE while reducing susceptibility to digestion. Pectin showed highest resistance to digestion and could potentially hinder allergen’s reaction with human IgE. | Egg | [49] |
| Enzyme | Study/Experiment | Results | Target | Ref. |
|---|---|---|---|---|
| Pepsin | Investigated the gastric digestion of using in vitro assays. | Pepsin plays a crucial role in digestion, with implications for allergenic reactions. | Peanut | [50] |
| Papain | Evaluated the efficacy of different proteases. | IgE-binding, showing potential in reducing allergenicity. | Peanut | [51] |
| Trypsin | Proteolytic processing of Ara h 3 using trypsin, chymotrypsin, and pepsin. | Ara h 3 was cleaved by all tested proteases; some fragments retained IgE-binding, impacting allergenicity. | Peanut | [52] |
| Papain | Developed papain-Cu3(PO4)2·3H2O-magnetic nanoflowers. | Papain reduced IgE binding. | Milk | [53] |
| Papain | Casein hydrolysis under varying conditions. | Optimal conditions reduced protein allergies. | Milk | [54] |
| Papain | Effects on protein digestibility and antigenicity. | Reduced antigenicity and enhanced its health benefits. | Soybean | [55] |
| Papain | Combined enzymatic digestion and ultrafiltration | Papain significantly reduced the antigenicity. | Whey protein concentrate | [56] |
| Papain | Enzymatic Debittering and fermentation | led to reduced major allergen levels, and mitigated allergenicity | Soy protein | [57] |
| Bromelain | Analyzed enzymatic hydrolysis. | Improved digestibility and decreased allergenicity. | Milk Casein | [58] |
| Bromelain | Two-step enzymatic modification. | Reduced immunoreactivity and allergenicity. | BLG | [59] |
| Bromelain | Analyzed enzymatic hydrolysis. | Improved techno-functional properties and reduced allergenicity. | PPIs | [60] |
| Bromelain | Study on enzymatic fragmentation. | Reduced allergenicity, producing hypoallergenic flour. | wheat flour | [61] |
| TG | Enzymatic treatment on proteins. | Reduced immunoreactivity; lower IgE and IgG reactivity post digestion. | Milk (horse and cow) | [62] |
| TG | Enzymatic hydrolysis and crosslinking. | Reduced allergenicity; improved functional properties. | Peanut | [63] |
| TG | Enzymatic treatment on proteins. | Reduced immunoreactivity and altered protein structure; potential for improving safety in allergy management. | Wheat flour | [64] |
| TG | Enzyme-induced polymerization | Reduced BLG antigenicity; decreased IgE binding from allergic patient sera. | BLG | [65] |
| TG | Enzyme-catalyzed glycosylation | Induced structural unfolding of TM; reduced antigenicity and allergenicity. | Shrimp | [66] |
| TG | Crosslinking of recombinant Ara h 1. | Significant decrease in IgE-binding activity; potential candidate for immunotherapy. | Peanut | [67] |
| Bioactive Compound | Study/Experiment | Results | Target | Ref. |
|---|---|---|---|---|
| QU | Mouse model study | Conjugation with OVA reduced IgE-binding and release capacity of OVA, modulated Th1/Th2 immune response, and reduced levels of IgE, IgG1, IgG, plasma histamine, and mast cell protease-1 on sensitized mast cells. | Egg | [68] |
| QU | In vitro study | QU binds to G through different mechanisms at different pH levels, leading to changes in the structure of G and potentially reducing its allergenicity. | Wheat | [69] |
| QU | Rat model study | QU treatment completely abrogated anaphylactic reactions in sensitized rats. | Peanut | [70] |
| Resveratrol | In vitro study using immune cells | Resveratrol reduced the production of inflammatory molecules in response to peanut allergens. | Peanut | [71] |
| Resveratrol | In vitro study using human mast cells | Resveratrol treatment was able to attenuate anaphylactic responses and inhibit the activation of IgE-mediated mast cells, both of which are involved in allergic reactions. | Allergenic Foods | [72] |
| Resveratrol | Mouse model study | Resveratrol reduced allergic response, IgE antibodies, mast cell degranulation, and cytokines in resveratrol-fed mice. Resveratrol inhibited the activation of inflammatory cells and release of inflammatory molecules in response to allergens in human mast cells. | Egg | [73] |
| Polyphenols | Resveratrol as a therapeutic agent for allergic rhinitis in mice | Resveratrol reduced symptoms of allergic rhinitis, inhibited activation of inflammatory cells, and reduced pro-inflammatory cytokine expression in nasal tissues. | Egg | [74] |
| Cn | Mouse model study | Cn ingestion significantly inhibited mastocytosis and suppressed intestinal anaphylaxis in mice, as evidenced by a decrease in the number of mast cells and a reduction in the severity of allergic reactions. | Egg | [76] |
| Cn | In vitro study using human mast cells | Inhibiting PDIs reduced mast cell degranulation and cytokine production, leading to a decrease in allergic symptoms in mice. Cn was also found to inhibit PDIs and reduce allergic symptoms in mice. | Allergenic Foods | [77] |
| Cn | In vitro study | The embedding rate of Cn was highest under the conditions of 1% NaOH modification at 80 °C for 3 h. | Peanut | [78] |
| EGCG | In vitro study | Lactoferrin and EGCG are generally safe, but conjugation may increase the risk of allergic reactions. | Milk | [79] |
| EGCG | Mouse model study | EGCG- BLG conjugate significantly reduces allergic reactions in a mouse model of food allergy. | Milk | [80] |
| EGCG | In vitro study | Both EGCG and PACs can improve the functional properties of SPI while reducing its allergenicity. | SPI | [81] |
| EGCG | Mouse model study | EGCG showed anti-inflammatory effects in a mouse model of OVA -induced allergic rhinitis. | Egg | [82] |
| EGCG conjugate | Covalent binding sites identified and IgE-binding ability evaluated | Conjugation had reduced IgE-binding ability compared with unmodified SPI, suggesting reduced allergenicity. | SPI | [83] |
| EGCG | Synergistic effects in alleviating OVA allergy | Combination of l-theanine and EGCG reduced serum levels of OVA -specific IgE and reduced allergic reaction | Egg | [84] |
| EGC and EGCG | Therapeutic effects on the allergic reaction of αs1-casein sensitized mice | Both EGC and EGCG reduced serum levels of αs1-casein-specific IgE and reduced histamine release from mast cells in αs1-casein sensitized mice. | Milk | [85] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sabaghi, M.; Maleki, S.J. Mitigating Food Protein Allergenicity with Biopolymers, Bioactive Compounds, and Enzymes. Allergies 2024, 4, 234-253. https://doi.org/10.3390/allergies4040016
Sabaghi M, Maleki SJ. Mitigating Food Protein Allergenicity with Biopolymers, Bioactive Compounds, and Enzymes. Allergies. 2024; 4(4):234-253. https://doi.org/10.3390/allergies4040016
Chicago/Turabian StyleSabaghi, Moslem, and Soheila J. Maleki. 2024. "Mitigating Food Protein Allergenicity with Biopolymers, Bioactive Compounds, and Enzymes" Allergies 4, no. 4: 234-253. https://doi.org/10.3390/allergies4040016
APA StyleSabaghi, M., & Maleki, S. J. (2024). Mitigating Food Protein Allergenicity with Biopolymers, Bioactive Compounds, and Enzymes. Allergies, 4(4), 234-253. https://doi.org/10.3390/allergies4040016






