The Role of Medical History and Allergic Tests in the Analysis of Antibiotic Allergy in the Pediatric Population
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Sample
2.2. Exclusion Criteria
2.3. Allergy Testing
2.4. Statistical Analysis
2.5. Ethics
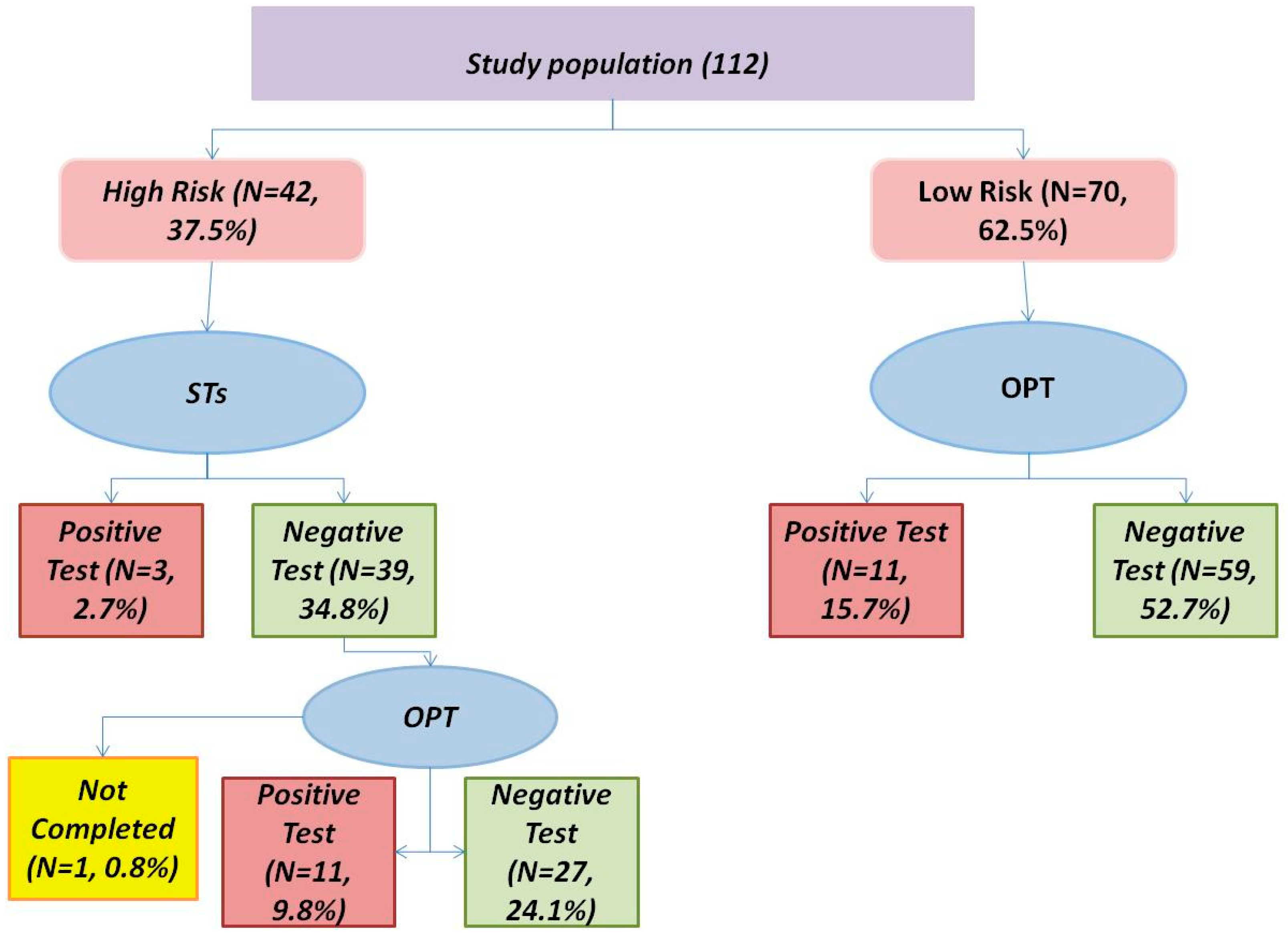
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sturkenboom, M.C.J.M.; Verhamme, K.M.C.; Nicolosi, A.; Murray, M.L.; Neubert, A.; Caudri, D.; Picelli, G.; Sen, E.F.; Giaquinto, C.; Cantarutti, L.; et al. Drug Use in Children: Cohort Study in Three European Countries. BMJ 2008, 337, a2245. [Google Scholar] [CrossRef]
- de Santa María, R.S.; Bogas, G.; Labella, M.; Ariza, A.; Salas, M.; Doña, I.; Torres, M.J. Approach for Delabeling Beta-Lactam Allergy in Children. Front. Allergy 2023, 4, 1298335. [Google Scholar] [CrossRef] [PubMed]
- Rimawi, R.H.; Cook, P.P.; Gooch, M.; Kabchi, B.; Ashraf, M.S.; Rimawi, B.H.; Gebregziabher, M.; Siraj, D.S. The Impact of Penicillin Skin Testing on Clinical Practice and Antimicrobial Stewardship. J. Hosp. Med. 2013, 8, 341–345. [Google Scholar] [CrossRef] [PubMed]
- Macy, E.; McCormick, T.A.; Adams, J.L.; Crawford, W.W.; Nguyen, M.T.; Hoang, L.; Eng, V.; Davis, A.C.; McGlynn, E.A. Association Between Removal of a Warning Against Cephalosporin Use in Patients With Penicillin Allergy and Antibiotic Prescribing. JAMA Netw. Open 2021, 4, 218367. [Google Scholar] [CrossRef] [PubMed]
- Macfadden, D.R.; Ladelfa, A.; Leen, J.; Gold, W.L.; Daneman, N.; Weber, E.; Al-Busaidi, I.; Petrescu, D.; Saltzman, I.; Devlin, M.; et al. Impact of Reported Beta-Lactam Allergy on Inpatient Outcomes: A Multicenter Prospective Cohort Study. Clin. Infect. Dis. 2016, 63, 904–910. [Google Scholar] [CrossRef] [PubMed]
- Jeffres, M.N.; Narayanan, P.P.; Shuster, J.E.; Schramm, G.E. Consequences of Avoiding β-Lactams in Patients with β-Lactam Allergies. J. Allergy Clin. Immunol. 2016, 137, 1148–1153. [Google Scholar] [CrossRef] [PubMed]
- Antibiotic Resistance. Available online: https://www.who.int/news-room/fact-sheets/detail/antibiotic-resistance (accessed on 26 November 2023).
- National Estimates for Antibiotic Resistance; CDC: Atlanta, GA, USA, 2021; Available online: https://www.cdc.gov/drugresistance/national-estimates.html (accessed on 27 November 2023).
- Gomes, C.V.; Bally, F. Duration of Antibiotic Therapy for Common Respiratory Infections in Adults. Rev. Med. Suisse 2023, 19, 1840–1843. [Google Scholar] [CrossRef] [PubMed]
- Murray, C.J.; Ikuta, K.S.; Sharara, F.; Swetschinski, L.; Robles Aguilar, G.; Gray, A.; Han, C.; Bisignano, C.; Rao, P.; Wool, E.; et al. Global Burden of Bacterial Antimicrobial Resistance in 2019: A Systematic Analysis. Lancet 2022, 399, 629–655. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Zembles, T.N.; Nimmer, M.; Brousseau, D.C.; Vyles, D. A Potential Cost Savings Analysis of a Penicillin De-Labeling Program. Front. Allergy 2023, 4, 1101321. [Google Scholar] [CrossRef]
- Au, L.Y.C.; Siu, A.M.; Yamamoto, L.G. Cost and Risk Analysis of Lifelong Penicillin Allergy. Clin. Pediatr. 2019, 58, 1309–1314. [Google Scholar] [CrossRef]
- Stone, C.A.; Trubiano, J.; Coleman, D.T.; Rukasin, C.R.F.; Phillips, E.J. The Challenge of De-Labeling Penicillin Allergy. Allergy 2020, 75, 273. [Google Scholar] [CrossRef]
- Vyles, D.; Chiu, A.; Simpson, P.; Nimmer, M.; Adams, J.; Brousseau, D.C. Parent-Reported Penicillin Allergy Symptoms in the Pediatric Emergency Department. Acad. Pediatr. 2017, 17, 251–255. [Google Scholar] [CrossRef] [PubMed]
- Rebelo Gomes, E.; Fonseca, J.; Araujo, L.; Demoly, P. Drug Allergy Claims in Children: From Self-Reporting to Confirmed Diagnosis. Clin. Exp. Allergy 2008, 38, 191–198. [Google Scholar] [CrossRef]
- Shenoy, E.S.; Macy, E.; Rowe, T.; Blumenthal, K.G. Evaluation and Management of Penicillin Allergy: A Review. JAMA 2019, 321, 188–199. [Google Scholar] [CrossRef] [PubMed]
- Keighley, C.L.; Saunderson, R.B.; Kok, J.; Dwyer, D.E. Viral Exanthems. Curr. Opin. Infect. Dis. 2015, 28, 139–150. [Google Scholar] [CrossRef] [PubMed]
- Di Lernia, V.; Mansouri, Y. Epstein-Barr Virus and Skin Manifestations in Childhood. Int. J. Dermatol. 2013, 52, 1177–1184. [Google Scholar] [CrossRef] [PubMed]
- Sousa-Pinto, B.; Tarrio, I.; Blumenthal, K.G.; Araújo, L.; Azevedo, L.F.; Delgado, L.; Fonseca, J.A. Accuracy of Penicillin Allergy Diagnostic Tests: A Systematic Review and Meta-Analysis. J. Allergy Clin. Immunol. 2021, 147, 296–308. [Google Scholar] [CrossRef] [PubMed]
- Romano, A.; Gaeta, F.; Valluzzi, R.L.; Caruso, C.; Rumi, G.; Bousquet, P.J. The Very Limited Usefulness of Skin Testing with Penicilloyl-Polylysine and the Minor Determinant Mixture in Evaluating Nonimmediate Reactions to Penicillins. Allergy 2010, 65, 1104–1107. [Google Scholar] [CrossRef] [PubMed]
- Padial, A.; Antunez, C.; Blanca-Lopez, N.; Fernandez, T.D.; Cornejo-Garcia, J.A.; Mayorga, C.; Torres, M.J.; Blanca, M. Non-Immediate Reactions to Beta-Lactams: Diagnostic Value of Skin Testing and Drug Provocation Test. Clin. Exp. Allergy 2008, 38, 822–828. [Google Scholar] [CrossRef]
- Demoly, P.; Adkinson, N.F.; Brockow, K.; Castells, M.; Chiriac, A.M.; Greenberger, P.A.; Khan, D.A.; Lang, D.M.; Park, H.S.; Pichler, W.; et al. International Consensus on Drug Allergy. Allergy 2014, 69, 420–437. [Google Scholar] [CrossRef]
- Norton, A.E.; Konvinse, K.; Phillips, E.J.; Broyles, A.D. Antibiotic Allergy in Pediatrics. Pediatrics 2018, 141, e20172497. [Google Scholar] [CrossRef] [PubMed]
- Mill, C.; Primeau, M.N.; Medoff, E.; Lejtenyi, C.; O’Keefe, A.; Netchiporouk, E.; Dery, A.; Ben-Shoshan, M. Assessing the Diagnostic Properties of a Graded Oral Provocation Challenge for the Diagnosis of Immediate and Nonimmediate Reactions to Amoxicillin in Children. JAMA Pediatr. 2016, 170, e160033. [Google Scholar] [CrossRef] [PubMed]
- Bircher, A.J.; Scherer Hofmeier, K. Drug Hypersensitivity Reactions: Inconsistency in the Use of the Classification of Immediate and Nonimmediate Reactions. J. Allergy Clin. Immunol. 2012, 129, 263–264. [Google Scholar] [CrossRef] [PubMed]
- Antunez, C.; Barbaud, A.; Gomez, E.; Audonnet, S.; Lopez, S.; Guéant-Rodriguez, R.M.; Aimone-Gastin, I.; Gomez, F.; Blanca, M.; Guéant, J.L. Recognition of Iodixanol by Dendritic Cells Increases the Cellular Response in Delayed Allergic Reactions to Contrast Media. Clin. Exp. Allergy 2011, 41, 657–664. [Google Scholar] [CrossRef] [PubMed]
- Johansson, S.G.O.; Bieber, T.; Dahl, R.; Friedmann, P.S.; Lanier, B.Q.; Lockey, R.F.; Motala, C.; Ortega Martell, J.A.; Platts-Mills, T.A.E.; Ring, J.; et al. Revised Nomenclature for Allergy for Global Use: Report of the Nomenclature Review Committee of the World Allergy Organization, October 2003. J. Allergy Clin. Immunol. 2004, 113, 832–836. [Google Scholar] [CrossRef]
- Brockow, K.; Garvey, L.H.; Aberer, W.; Atanaskovic-Markovic, M.; Barbaud, A.; Bilo, M.B.; Bircher, A.; Blanca, M.; Bonadonna, B.; Campi, P.; et al. Skin Test Concentrations for Systemically Administered Drugs—An ENDA/EAACI Drug Allergy Interest Group Position Paper. Allergy 2013, 68, 702–712. [Google Scholar] [CrossRef] [PubMed]
- Interpretation of Prick and Intradermal Skin Tests. Available online: https://www.aaaai.org/allergist-resources/ask-the-expert/answers/old-ask-the-experts/intradermal-skin (accessed on 27 November 2018).
- Blumenthal, K.G.; Peter, J.G.; Trubiano, J.A.; Phillips, E.J. Antibiotic Allergy. Lancet 2019, 393, 183–198. [Google Scholar] [CrossRef] [PubMed]
- Dyer, J.A. Childhood Viral Exanthems. Pediatr. Ann. 2007, 36, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Anci, E.; Braun, C.; Marinosci, A.; Rodieux, F.; Midun, E.; Torres, M.J.; Caubet, J.C. Viral Infections and Cutaneous Drug-Related Eruptions. Front. Pharmacol. 2020, 11, 586407. [Google Scholar] [CrossRef]
- Seitz, C.S.; Bröcker, E.B.; Trautmann, A. Diagnosis of Drug Hypersensitivity in Children and Adolescents: Discrepancy between Physician-Based Assessment and Results of Testing. Pediatr. Allergy Immunol. 2011, 22, 405–410. [Google Scholar] [CrossRef]
- Vezir, E.; Erkocoglu, M.; Civelek, E.; Kaya, A.; Azkur, D.; Akan, A.; Ozcan, C.; Toyran, M.; Ginis, T.; Misirlioglu, E.D.; et al. The Evaluation of Drug Provocation Tests in Pediatric Allergy Clinic: A Single Center Experience. Allergy Asthma Proc. 2014, 35, 156–162. [Google Scholar] [CrossRef]
- Arikoglu, T.; Aslan, G.; Batmaz, S.B.; Eskandari, G.; Helvaci, I.; Kuyucu, S. Diagnostic Evaluation and Risk Factors for Drug Allergies in Children: From Clinical History to Skin and Challenge Tests. Int. J. Clin. Pharm. 2015, 37, 583–591. [Google Scholar] [CrossRef]
- Demirhan, A.; Yildirim, D.D.; Arikoglu, T.; Ozhan, A.K.; Tokmeci, N.; Yuksek, B.C.; Kuyucu, S. A Combined Risk Modeling Strategy for Clinical Prediction of Beta-Lactam Allergies in Children. Allergy Asthma Proc. 2021, 42, E159–E166. [Google Scholar] [CrossRef]
- Ponvert, C.; Perrin, Y.; Bados-Albiero, A.; Le Bourgeois, M.; Karila, C.; Delacourt, C.; Scheinmann, P.; De Blic, J. Allergy to Betalactam Antibiotics in Children: Results of a 20-Year Study Based on Clinical History, Skin and Challenge Tests. Pediatr. Allergy Immunol. 2011, 22, 411–418. [Google Scholar] [CrossRef]
- Galli, S.J.; Tsai, M.; Piliponsky, A.M. The Development of Allergic Inflammation. Nature 2008, 454, 445. [Google Scholar] [CrossRef] [PubMed]
- Abbas, M.; Moussa, M.; Akel, H. Type I Hypersensitivity Reaction; StatPearls: Treasure Island, FL, USA, 2023. [Google Scholar]
- Apter, A.J.; Schelleman, H.; Walker, A.; Addya, K.; Rebbeck, T. Clinical and Genetic Risk Factors of Self-Reported Penicillin Allergy. J. Allergy Clin. Immunol. 2008, 122, 152–158. [Google Scholar] [CrossRef]
- Oussalah, A.; Mayorga, C.; Blanca, M.; Barbaud, A.; Nakonechna, A.; Cernadas, J.; Gotua, M.; Brockow, K.; Caubet, J.C.; Bircher, A.; et al. Genetic Variants Associated with Drugs-Induced Immediate Hypersensitivity Reactions: A PRISMA-Compliant Systematic Review. Allergy 2016, 71, 443–462. [Google Scholar] [CrossRef] [PubMed]
- Dias de Castro, E.; Carolino, F.; Carneiro-Leão, L.; Barbosa, J.; Ribeiro, L.; Cernadas, J.R. Allergy to Beta-Lactam Antibiotics in Children: Risk Factors for a Positive Diagnostic Work-Up. Allergol. Immunopathol. 2020, 48, 417–423. [Google Scholar] [CrossRef] [PubMed]
- Park, M.A.; Matesic, D.; Markus, P.J.; Li, J.T.C. Female Sex as a Risk Factor for Penicillin Allergy. Ann. Allergy Asthma Immunol. 2007, 99, 54–58. [Google Scholar] [CrossRef] [PubMed]
- Cornejo-García, J.A.; Guéant-Rodriguez, R.M.; Torres, M.J.; Blanca-Lopez, N.; Tramoy, D.; Romano, A.; Blanca, M.; Guéant, J.L. Biological and Genetic Determinants of Atopy Are Predictors of Immediate-Type Allergy to Betalactams, in Spain. Allergy 2012, 67, 1181–1185. [Google Scholar] [CrossRef]
- Ariza, A.; Fernández, T.D.; Mayorga, C.; Blanca, M.; Torres, M.J. Prediction of Hypersensitivity to Antibiotics: What Factors Need to Be Considered? Expert Rev. Clin. Immunol. 2013, 9, 1279–1288. [Google Scholar] [CrossRef] [PubMed]
- Faitelson, Y.; Boaz, M.; Dalal, I. Asthma, Family History of Drug Allergy, and Age Predict Amoxicillin Allergy in Children. J. Allergy Clin. Immunol. Pract. 2018, 6, 1363–1367. [Google Scholar] [CrossRef] [PubMed]
- Ben Romdhane, H.; Ben Fredj, N.; Ben Fadhel, N.; Chadli, Z.; Abderrahmen, A.; Boughattas, N.A.; Chaabane, A.; Aouam, K. Beta-Lactam Hypersensitivity in Children: Frequency and Risk Factors. Br. J. Clin. Pharmacol. 2023, 89, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Sipahi Cimen, S.; Hizli Demirkale, Z.; Yucel, E.; Ozceker, D.; Suleyman, A.; Sayili, U.; Tamay, Z.; Guler, N. Risk Factors of Challenge-Proven Beta-Lactam Allergy in Children with Immediate and Non-Immediate Mild Cutaneous Reactions. Int. Arch. Allergy Immunol. 2023, 184, 539–549. [Google Scholar] [CrossRef] [PubMed]
| Low Risk (N = 70) | High Risk (N = 42) | p Value | |
|---|---|---|---|
| Female sex | 32 (45.7%) | 15 (35.7%) | 0.299 |
| Current age | 6.27 ± 3.40 | 6.23 ± 3.62 | 0.951 |
| Age of episode | 3.93 ± 2.40 | 3.20 ± 2.54 | 0.138 |
| More frequently reported antibiotic (amoxicillin) | 35 (50.0%) | 19 (45.2%) | 0.730 |
| Route of drug administration (per os) | 70 (100%) | 41 (97.6%) | 0.195 |
| Reason for antibiotic prescription *: Respiratory infection | 59 (84.2%) | 41 (97.6%) | 0.027 |
| Urinary infection | 1 (1.4%) | 1 (2.4%) | 0.713 |
| Skin infection | 3 (4.3%) | 0 (0.0%) | 0.174 |
| Symptoms *: Maculopapular exanthema or urticaria | 70 (100%) | 42 (100%) | 1.000 |
| Angioedema | 4 (5.7%) | 2 (4.8%) | 0.828 |
| Duration of suspicious symptoms (<24 h) * | 10 (14.3%) | 11 (26.2%) | 0.118 |
| Treatment *: Discontinuation of antibiotic | 9 (12.9%) | 4 (9.5%) | 0.594 |
| Antihistamine drugs | 54 (77.1%) | 35 (83.3%) | 0.432 |
| Corticosteroids | 7 (10.0%) | 2 (4.8%) | 0.324 |
| Hospitalization | 7 (10.0%) | 7 (16.7%) | 0.302 |
| Allergy to other antibiotics | 13 (18.6%) | 10 (23.8%) | 0.506 |
| Food allergy | 10 (14.3%) | 5 (11.9%) | 0.720 |
| Asthma | 7 (10.0%) | 9 (21.4%) | 0.094 |
| Allergic rhinitis | 6 (8.6%) | 8 (19.0%) | 0.105 |
| Atopic dermatitis | 23 (32.9%) | 15 (35.7%) | 0.757 |
| Family history of antibiotic allergy | 5 (7.1%) | 7 (16.7) | 0.115 |
| Tested Negative (N = 86) | Tested Positive (N = 25) | p Value | |
|---|---|---|---|
| Female sex | 37 (43.0%) | 10 (40.0%) | 0.788 |
| Current age | 6.18 ± 3.58 | 6.36 ± 3.09 | 0.818 |
| Age of episode | 3.87 ± 2.69 | 2.94 ± 1.35 | 0.021 |
| Reason for antibiotic prescription: respiratory infection | 78 (90.7%) | 21 (84.0%) | 0.342 |
| Duration of suspicious symptoms (<24 h) | 14 (16.3%) | 7 (28.0%) | 0.188 |
| Allergy to another antibiotic | 14 (16.3%) | 8 (32.0%) | 0.83 |
| Food allergy | 8 (9.3%) | 7 (28.0%) | 0.016 |
| Asthma | 8 (9.3%) | 7 (28.0%) | 0.016 |
| Allergic rhinitis | 9 (10.5%) | 4 (16.0%) | 0.449 |
| Atopic dermatitis | 27 (31.4%) | 10 (40.0%) | 0.422 |
| Family history of antibiotic allergy | 9 (10.5%) | 3 (12.0%) | 0.828 |
| High-risk group | 27 (31.4%) | 14 (56.0%) | 0.025 |
| Univariate | Multivariate | |||
|---|---|---|---|---|
| OR (95%CI) | p Value | OR (95%CI) | p Value | |
| Female sex | 0.883 (0.356–2.187) | 0.788 | ||
| Current age | 1.015 (0.894–1.153) | 0.816 | ||
| Age of episode | 0.820 (0.648–1.038) | 0.100 | 0.872 (0.667–1.141) | 0.319 |
| Reason for antibiotic prescription: respiratory infection | 0.538 (0.148–1.963) | 0.348 | ||
| Duration of suspicious symptoms (<24 h) | 2.000 (0.704–5.681) | 0.193 | ||
| Allergy to another antibiotic | 2.420 (0.875–6.690) | 0.088 | 1.820 (0.573–5.784) | 0.310 |
| Food allergy | 3.792 (1.217–11.814) | 0.022 | 4.885 (1.377–17.336) | 0.014 |
| Asthma | 3.792 (1.217–11.814) | 0.022 | 4.028 (1.094–14.824) | 0.036 |
| Allergic rhinitis | 1.630 (0.456–5.819) | 0.452 | ||
| Atopic dermatitis | 1.457 (0.580–3.656) | 0.423 | ||
| Family history of antibiotic allergy | 1.167 (0.291–4.684) | 0.828 | ||
| High risk | 2.781 (1.118–6.920) | 0.028 | 2.583 (0.943–7.074) | 0.065 |
| Very high risk | 6.956 (1.772–27.302) | 0.005 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dimitroglou, M.; Moriki, D.; Sardeli, O.; Kartsiouni, E.; Koumpagioti, D.; Galani, A.; Papaevangelou, V.; Douros, K. The Role of Medical History and Allergic Tests in the Analysis of Antibiotic Allergy in the Pediatric Population. Allergies 2024, 4, 54-63. https://doi.org/10.3390/allergies4020005
Dimitroglou M, Moriki D, Sardeli O, Kartsiouni E, Koumpagioti D, Galani A, Papaevangelou V, Douros K. The Role of Medical History and Allergic Tests in the Analysis of Antibiotic Allergy in the Pediatric Population. Allergies. 2024; 4(2):54-63. https://doi.org/10.3390/allergies4020005
Chicago/Turabian StyleDimitroglou, Margarita, Dafni Moriki, Olympia Sardeli, Elpiniki Kartsiouni, Despoina Koumpagioti, Angeliki Galani, Vassiliki Papaevangelou, and Konstantinos Douros. 2024. "The Role of Medical History and Allergic Tests in the Analysis of Antibiotic Allergy in the Pediatric Population" Allergies 4, no. 2: 54-63. https://doi.org/10.3390/allergies4020005
APA StyleDimitroglou, M., Moriki, D., Sardeli, O., Kartsiouni, E., Koumpagioti, D., Galani, A., Papaevangelou, V., & Douros, K. (2024). The Role of Medical History and Allergic Tests in the Analysis of Antibiotic Allergy in the Pediatric Population. Allergies, 4(2), 54-63. https://doi.org/10.3390/allergies4020005






