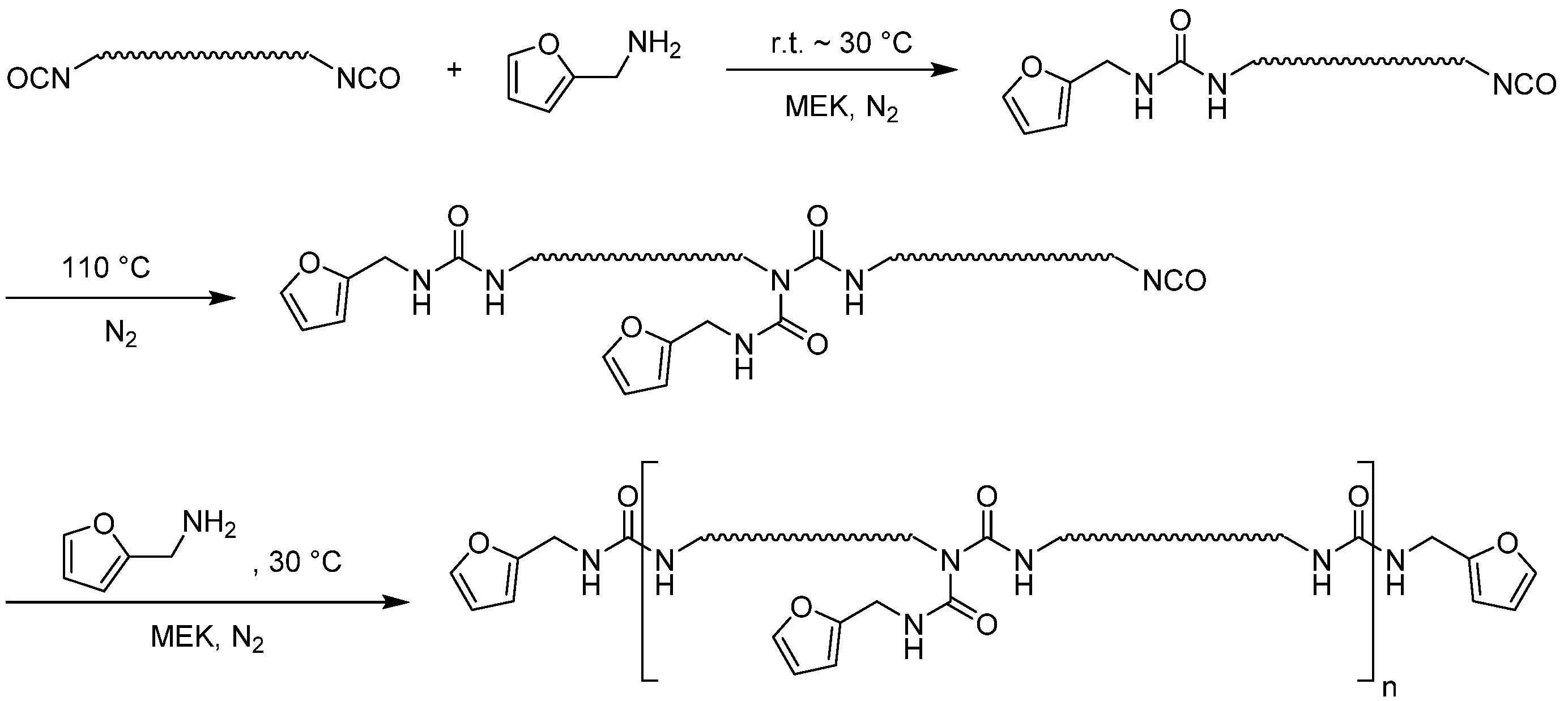
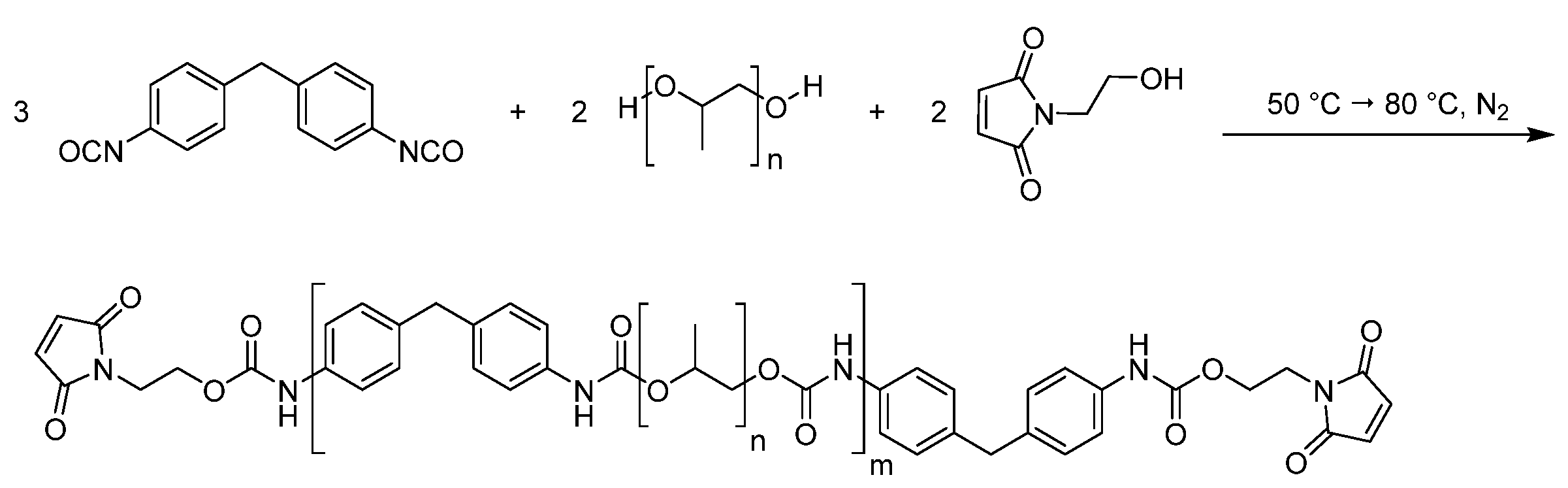
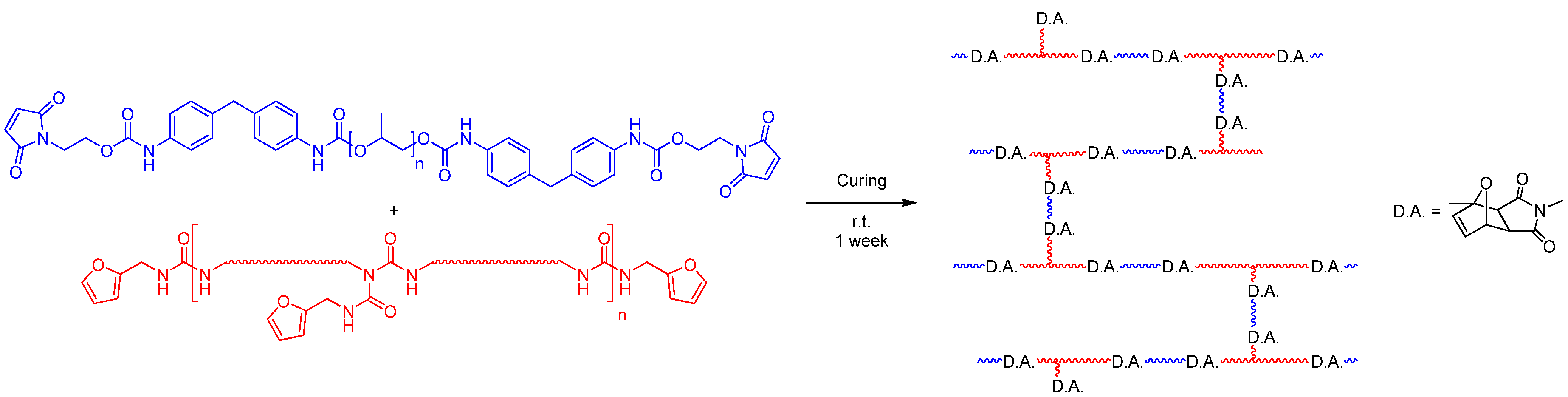
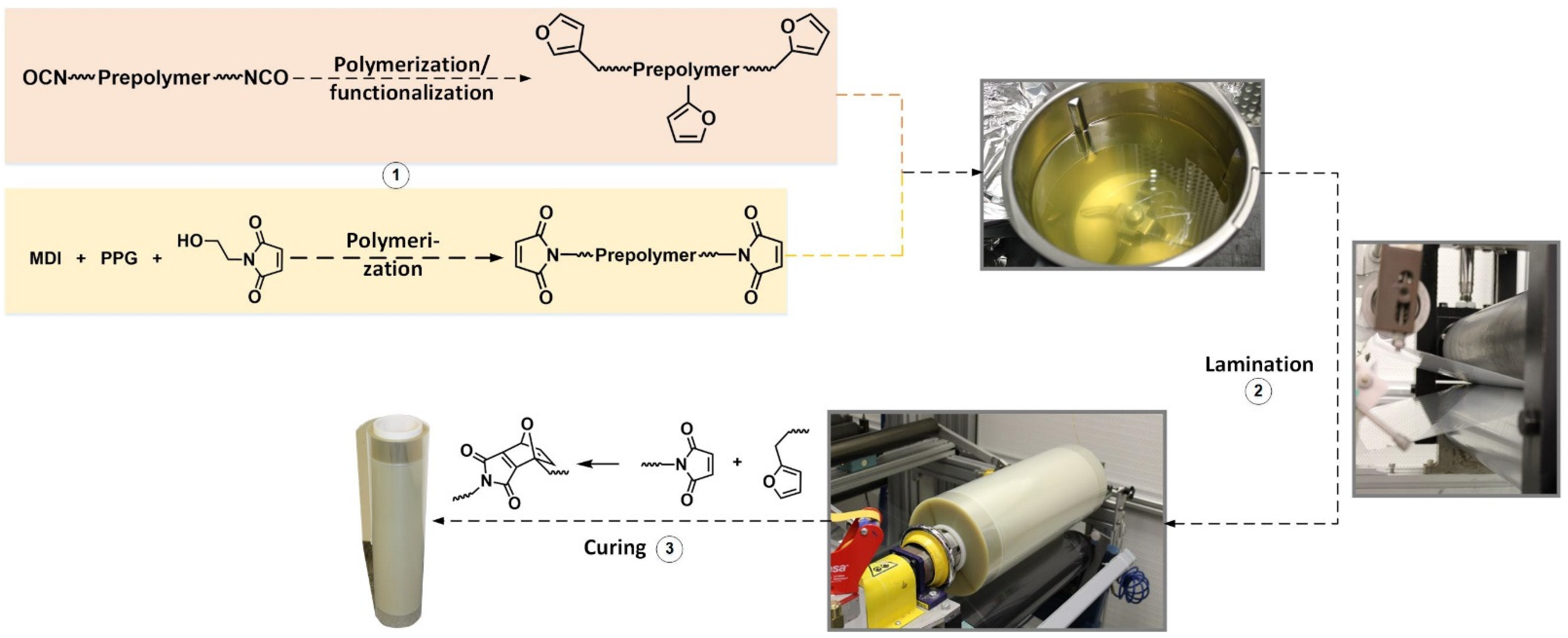
3.1. Preparation of the Laminate
As in a previous publication, the formulation was chosen in such a way that no carcinogenic, mutagenic, or reprotoxic chemicals were required to functionalize the prepolymers; moreover, no crosslinking molecule was necessary [
19]. This was achieved by the fact that the furan-functionalized prepolymer contains furan side chains and thus, on average, three functional groups. The maleimide prepolymer, on the other hand, is linear (see
Figure 6)). When formulating the two prepolymers in MEK, care was taken to ensure that the physical properties of the adhesive were similar to the parameters of commercial adhesives in order to guarantee an unproblematic and even application of the adhesive in the laminating process. Therefore, a solids content of 30% was selected, at which, according to ISO 2432-No4, the adhesive needed 19.6 s, which was in the same order of magnitude as the times commercial solvent-based packaging adhesives needed. On the rheometer, this corresponded to a viscosity of 40 Pa∙s at a shear rate of 50 1∙s
−1. The pot life of the adhesive was determined by the change in viscosity of the 30% solution over time due to the crosslinking of the adhesive to about 24 h.
To ensure that the stresses that affected the laminate in the laminating machine did not lead to problems during winding, care was taken that the adhesive was sufficiently tacky. Therefore, polypropylene glycol was used as the diol component for the maleimide prepolymer, which made the prepolymer a viscous liquid that led to tackiness of the formulation.
By processing this adhesive on the laminating line described in Materials and Methods, 300 linear meters of a PET//LDPE laminate could be produced. Using microtome sectioning, the thickness of the adhesive was determined to be 6.5 µm (compare
Figure 7), thus slightly exceeding the typical layer thicknesses of conventional packaging adhesives, which were around 2–5 µm [
27].
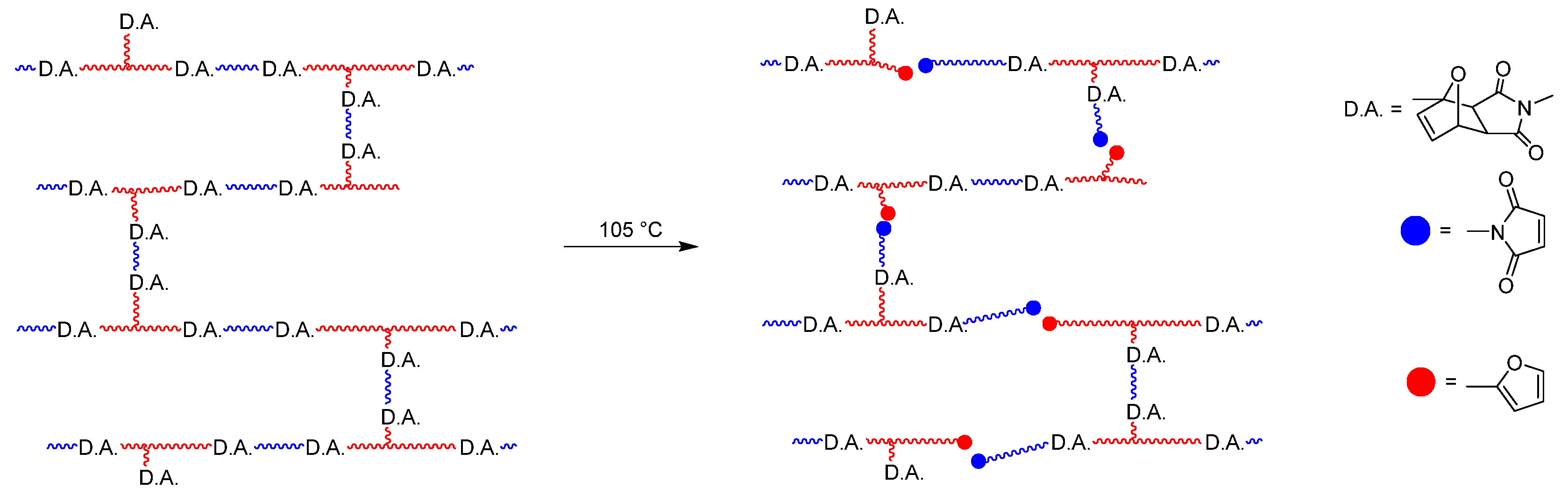
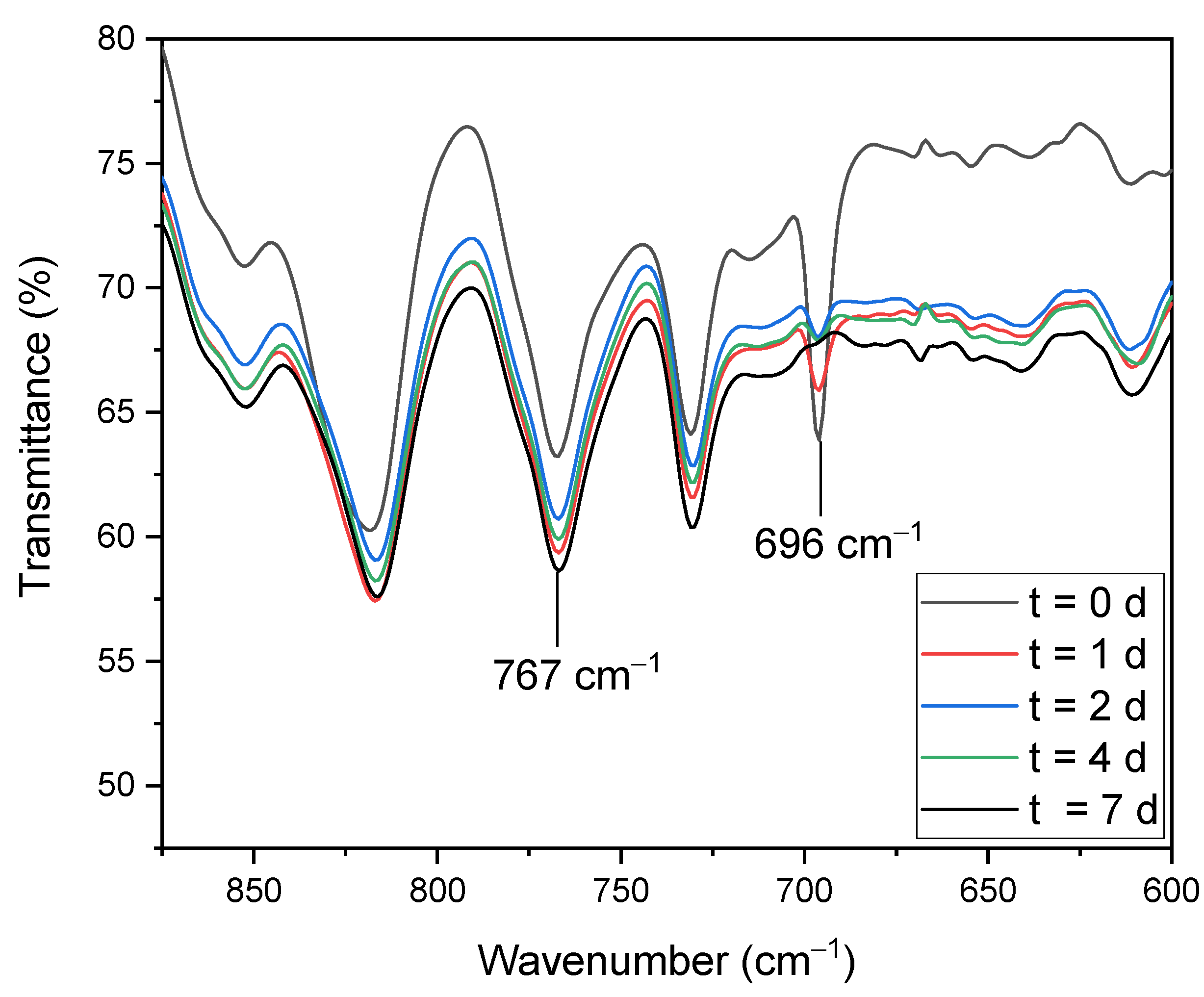
The curing process of the adhesive, the last step of the preparation of the laminate, was followed by infrared spectroscopy. Here, the decrease. of the ring deformation mode of the maleimide group at 696 cm
−1 could be followed [
28]. It was determined that 73.8% of the isocyanate groups had already reacted after 1 day, and 88.3% after 2 days. Since the isocyanate band disappeared completely after 7 days, it was assumed that after this time had elapsed, the Diels–Alder reaction was completed (see
Figure 8).
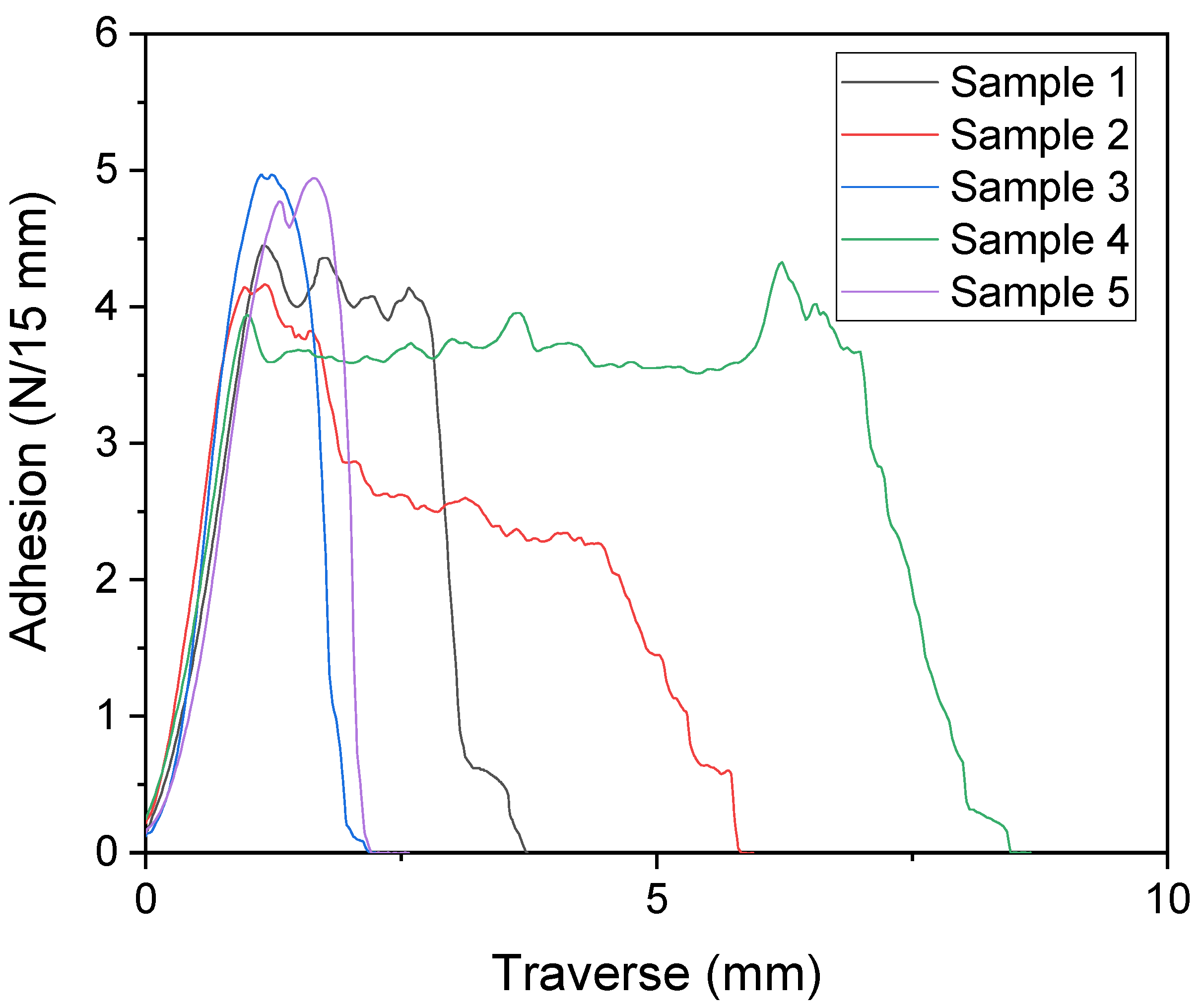
The bonding of the PET//LDPE laminate was tested using the T-peel test (see
Figure 9). It was found that each of the five test strips had a substrate failure of the PET, which implies that the bond was stronger than the cohesion of the PET. This means that the bonding in this case was stronger than in the previous publication [
19], although the same adhesive formulation and the same adhesive layer thickness were chosen. However, in contrast to manual lamination, machine lamination allowed an even application of the adhesive and lamination without any air bubbles. At 6.5 µm, the application of the adhesive was slightly thicker than in most commercial cases of packaging laminates, where the adhesive layer thickness is usually around 3 µm [
27]. With a lower adhesive layer thickness, a decrease in bond adhesion was also to be expected. However, since in many cases an adhesive strength of 2 N/15 mm would already be sufficient, it should also be possible to use a thinner adhesive layer.
3.2. Behavior of the Adhesive during the Sealing Process
Since a mere laminate does not yet constitute packaging, it must be converted into a packaging in a thermomechanical processing step such as sealing to form a pouch or thermoforming to form a tray. For the adhesive, however, this means that it has to withstand thermal stress. Therefore, the influence of short-term high-temperature exposure of sealing jaws on the crosslinking of the adhesive was examined here. The conditions chosen for this were a temperature of 160 °C with a sealing time of 1 s, which can be considered typical sealing conditions for LDPE [
29].
It can be seen that the ring deformation mode of the maleimide group at 696 cm
−1 reappeared (see
Figure S1). From the ratio of this absorption band, it was possible to estimate that about 62% of the Diels–Alder adducts had undergone the retro-Diels–Alder reaction. By recording another IR spectrum 10 days after the sample was sealed, it was shown that the adhesive was re-crosslinking. According to the literature, it would be expected that the re-crosslinking does not take place completely since residual amines can react with the maleimide groups at 70 °C and above in a Michael addition [
30,
31].
However, since the degree of crosslinking is determined via the absorption band of the maleimide group, this effect cannot be detected here. Since the residual content of furfurylamine was only about 15 ppm and the functionalization of the NCO groups was complete, it can be expected that the maleimide was not blocked in relevant amounts [
19].
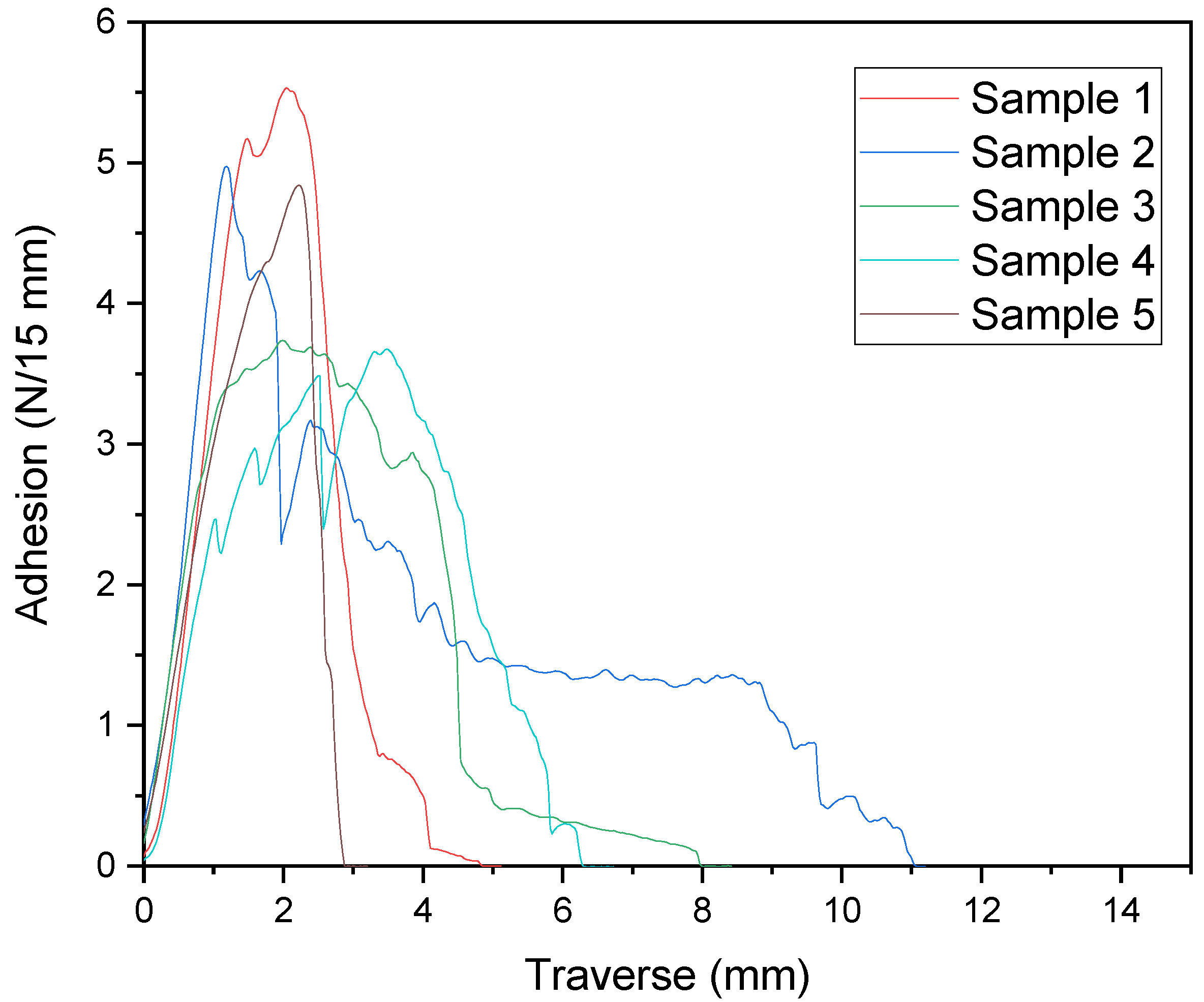
In order to test the influence of the partial opening of the Diels–Alder adducts by the temperature effect of the sealing test on the bond adhesion of the laminate, a T-peel test was performed directly after temperature exposure by the sealing jaws.
Figure 10 shows the measurement curves of the T-peel tests.
Here, too, as in the T-peel tests of the nonsealed samples, an immediate substrate failure of the PET film occurred. This means that even with a reduced crosslink density of the adhesive after the sealing treatment, there is still sufficient crosslink density in the adhesive to ensure cohesion in the adhesive. This shows that even directly after thermal processing of the packaging, laminate adhesion is still present, which is why there is no risk of unwanted delamination in this step.
3.3. Recycling of the PET//LDPE Laminate in a Small-Scale Technical Plant
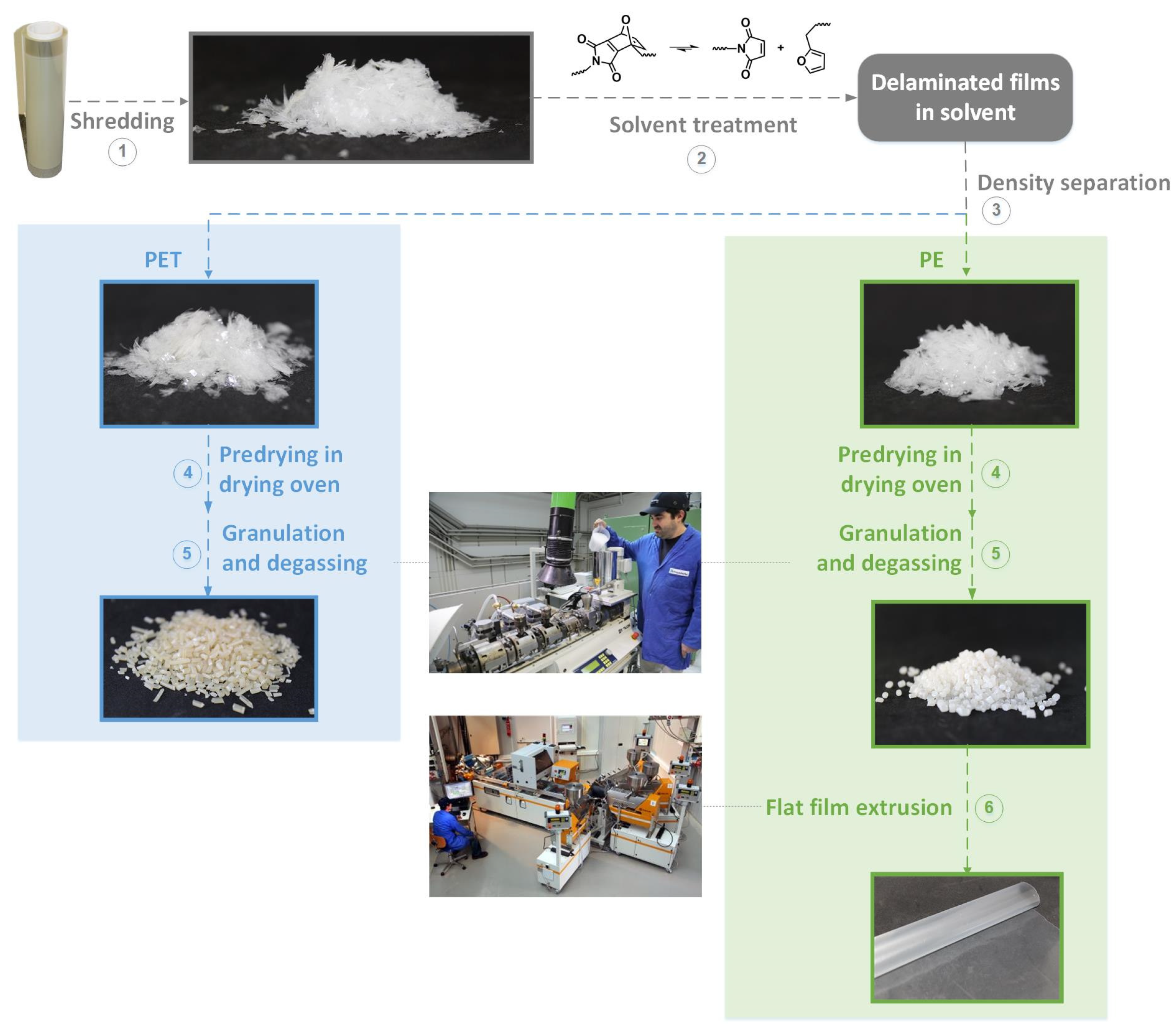
The individual steps of the recycling process are shown in
Figure 11 and include six steps: (1) shredding of the film, (2) solvent treatment, (3) density separation, (4) predrying of the material, (5) degassing and granulation, and (6) extrusion of the recycled film.
3.3.1. Step 1—Shredding
Shredding provided a size distribution of about 0.2 cm2 to about 0.7 cm2, which was a smaller size than in previous publications, in which the dimensions were 1 × 1 cm. This is because the material has to be fed into the extruder in step 5, and this is easier to carry out with smaller flake sizes. The smaller flake size also reduces the time needed for solvent treatment, as the dissolution of the adhesive proceeds via the cut edges of the flakes, and the area of the cut edges increases with decreasing flake size. However, the size distribution of the flakes influences the duration of delamination.
3.3.2. Step 2—Solvent Treatment of the Shredded Material
Step 2, the dissolving step itself was carried out for 20 min at 105 °C in a stainless steel vessel with a capacity of 100 L. The ratio between film and solvent was chosen so that the shredded films were completely covered with solvent.
In contrast to previous publications, in which laboratory-scale experiments were carried out, the solvent used in this pilot plant experiment was not dimethyl sulfoxide but an optimized proprietary CreaSolv® solvent from CreaCycle GmbH. Like DMSO, this solvent has the property of being able to dissolve the polymers of the adhesive, but it does not have the disadvantages associated with DMSO, such as the smell or the freezing point close to room temperature. Instead, it is characterized by a high flash point, which allows convenient and economical transport and storage, and a comparatively low evaporation enthalpy, which is favorable for solvent recycling.
The time required for delamination in the solvent was determined in advance by laboratory-scale tests in which the time required for the delamination of particularly large flakes was determined in a dissolution test. In order to estimate the influence of a treatment time of 20 min at 105 °C, a test strip of the cured adhesive was exposed to these conditions in the drying oven, and an IR spectrum was recorded immediately afterwards. Here, too, as after treatment with the sealing device, an increase in intensity of the maleimide absorption band can be observed. From the ratio of the maleimide absorption band to the band at 767 cm
−1 compared with the ratio of the two bands in the uncured adhesive, we found that 35% of all Diels–Alder adducts had undergone the retro reaction and were therefore present in their opened form (see
Figure S2). Since the adhesive formulation was chosen in such a way that each furan-functionalized prepolymer carried on average three functional groups, the opening of 35% of the adducts should also be sufficient to cancel the crosslinking of the adhesive. In this IR experiment, however, the adhesive was heated to 105 °C in the oven, whereas in the actual recycling experiment, the temperature effect would take place in the presence of a solvent. In the solvent, once an adduct is opened, the functional groups can be surrounded by a solvate shell and thus washed away from the interlayer of the composite, becoming less likely due to dilution. It is also conceivable that the kinetics of the retro-Diels–Alder reaction could be affected since it is known that the solvent has an influence on the kinetics of the Diels–Alder reaction [
17,
31,
32,
33,
34,
35]. However, Widstrom et al. [
36] performed studies on the kinetics of the retro-Diels–Alder reaction between a maleimide and furan in different solvents and could not demonstrate any influence. Therefore, it could be assumed that in the present case, there was also no influence on the kinetics of the retro-Diels–Alder reaction due to the solvent. However, it must be taken into account that the retro-Diels–Alder reaction does not necessarily take place in the solvated state, since the adhesive is crosslinked and thus initially not soluble, and is also embedded between layers of LDPE and PET. It can therefore be assumed that the retro-Diels–Alder reaction takes place to a relevant extent without interactions with the solvent or at most in a swollen state. A reliable statement on how the solvent influences the kinetics of the reaction and the mass transfer cannot be made without further investigations. The partial opening of the adducts can also be demonstrated by SEC measurements. The adhesive molecules in the recycling solvent directly after completion of the dissolution step show an elution peak in the SEC measurement, with Mw 44,470 g/mol. If the two prepolymers are freshly dissolved together and a SEC measurement is performed, an elution peak resulting from the superposition of the two prepolymers is shown, with Mw of 26,916 g/mol. This would indicate that more than one in three Diels–Alder adducts would be open. However, these two values can only be compared with a limited extent since a branched polymer is obtained due to the incomplete openings of the adducts, and branched molecules have smaller radii of gyration, and thus, smaller molar masses are determined than are actually present [
37]. Therefore, estimation by IR spectroscopy seems to provide a more reliable result.
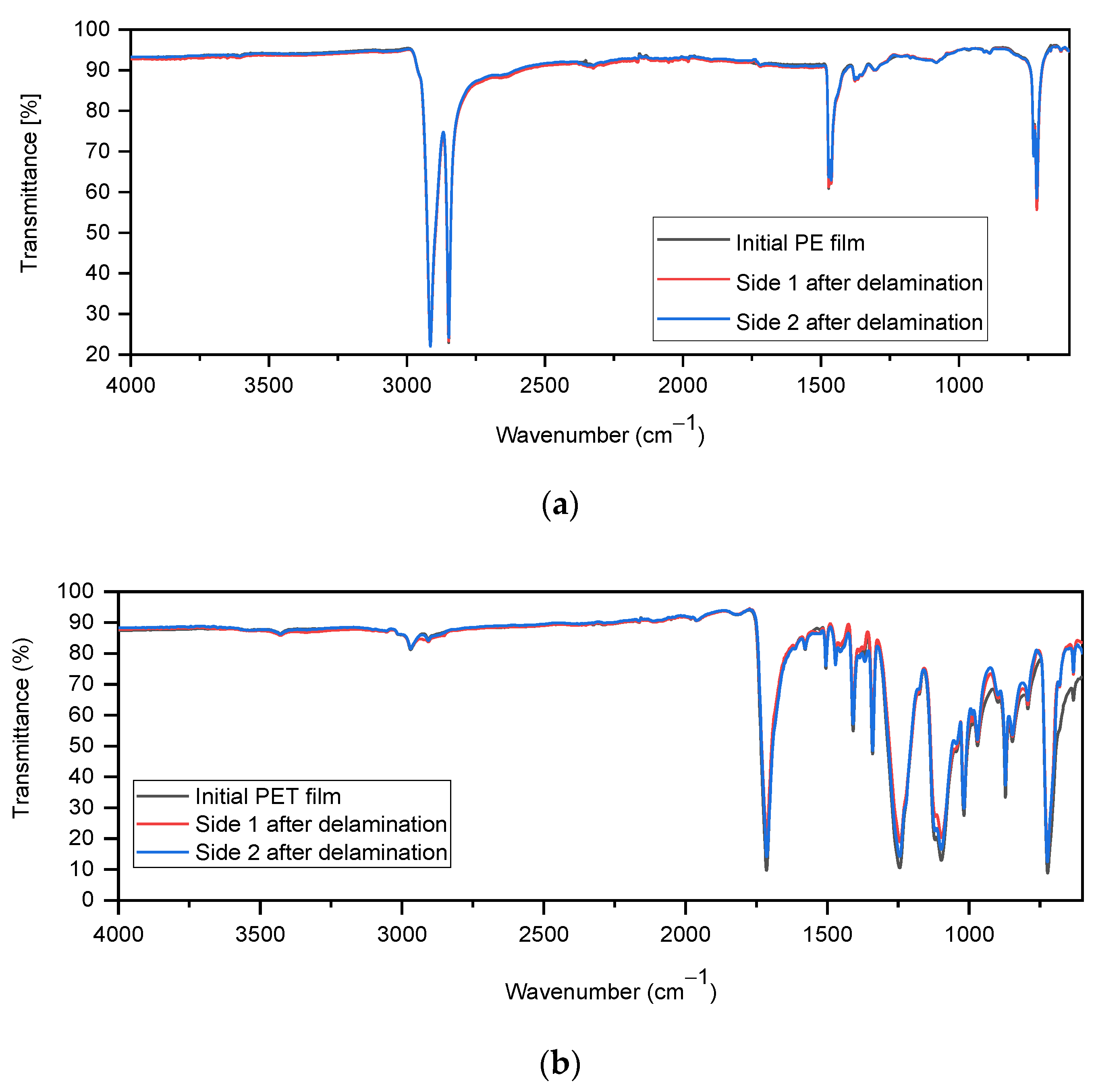
IR spectra were recorded on both sides of the dried flakes to check whether the adhesive could be completely removed. For this purpose, especially large flakes were selected from a sample of both fractions, as these have longer delamination times and should therefore be most likely to have adhesive residues on larger flakes.
Figure 12a shows a spectrum of the original LDPE film and also spectra taken on both sides of a delaminated LDPE film. It can be seen that on both sides of the recovered LDPE film, there is no evidence of adhesive even where the most intense bands of adhesive at 1711, 1531, and 1072 cm
−1 would be expected. The same can be observed with the spectra of PET (
Figure 12b). Therefore, it can be assumed that there is no residue of the adhesive on the surfaces of the films.
3.3.3. Step 3—Density Separation
Step 3 of the recycling process is the density separation. Therefore, subsequent to the solvent treatment, the flakes and the solvent were pumped into a barrel where a floating layer of LDPE and a sunken fraction of PET could be identified. Before the LDPE fraction was skimmed off with a sieve, the LDPE layer was stirred several times to give PET flakes, which might have been lying on the LDPE flakes and were thus prevented from sinking, the opportunity to sink. The sink fraction was then removed via an outlet at the bottom of the barrel and separated from the solvent. Both the floating and the sinking fractions were mechanically pressed out to reduce the solvent content.
In order to evaluate the effectiveness of the composite separation, the mass fractions of the floating, sinking, and solvent fractions; their experimentally determined solvent content; and the solids content calculated from them are given in
Table 4.
The solids content of the solvent can be attributed to the dissolved adhesive. The theoretical ratio of the fractions to each other was calculated on the basis of the layer thicknesses of PET, LDPE, and the adhesive and their densities. Comparing the ratio of the obtained quantity fractions with the theoretically expected ratio, it was noticeable that the actually obtained quantity of LDPE exceeded the theoretically expected one, while the actually obtained quantity of PET was lower than expected. This would indicate that a relatively high proportion of PET must be contained in the floating fraction.
To validate this, the LDPE content in both the floating and sinking fractions was dissolved out with a selective solvent. It was found that the floating fraction, which consisted of PE, still contained 3% PET. However, the PET fraction still contained 15% LDPE. This contradicts the estimation based on the theoretical mass fractions. The reason for the incorrect estimation could be, for example, that it is difficult to take a representative, homogeneous sample to determine the solvent content.
The reason for the remaining PET in the LDPE floating fraction could be air bubbles adhering to the PET or PET flakes lying on LDPE flakes. The LDPE content in PET can also be partly explained by the LDPE flakes being prevented from rising by the PET flakes. The handling of the density separation may also have caused LDPE to enter the PET fraction, because when the PET fraction was drained off at the bottom of the barrel, LDPE was also carried from the floating fraction into the sinking fraction. These two problems should be reduced by cyclonic separation equipment, which is specifically designed for density separation of materials. Nevertheless, it cannot be excluded that a certain amount of LDPE was not yet delaminated from the PET, which could mean that the duration of the solvent treatment would still have to be extended.
3.3.4. Step 4, 5, and 6—Drying Granulation and Flat Film Extrusion
The two fractions obtained are predried before they are further processed in the extruders, whereby a solids content of 94% could be achieved for the PE floating fraction and 97% for the PET sinking fraction. This reduces the influence of the solvent on the extrusion and enables a low residual solvent content. During the pelletizing of the LDPE, a melt screen could be installed in the extruder to remove the remaining PET. Since the LDPE has a lower melting point than PET, it was not possible to remove the LDPE residue during PET pelletizing.
To remove the solvent from both materials, a vacuum was applied in the extruder for degassing. In this way, LDPE granulate with a residual solvent content of 219 ppm and PET granulate with a residual solvent content of 4 ppm were obtained. The LDPE granulates were extruded into a film with a target thickness of 45 µm like the initial film.
3.3.5. Recovery of the Adhesive and the Solvent
A recovery of the adhesive from the solution would be theoretically conceivable and could be performed, for example, by precipitation from solution or by distillation. In practice, however, such a procedure would not be very practical. The reason for this is that the equilibrium of the Diels–Alder reaction at room temperature would shift on the side of the adduct, and the adhesive would thus crosslink again. If the process were to be used for post-consumer packaging, the solution would contain printing ink and other impurities, such as packaging residues in addition to the adhesive. This would make recovery of the adhesive even more impractical.
The adhesive solution obtained through the recycling process is also not suitable for the direct production of a new laminate for various reasons. One obvious reason is that a solids content of 0.6% is far too low, and as in the production of the laminate, a solids content of 20%–50% would rather be necessary. However, even if this solids content could be achieved by concentrating the solution, the solution would not be suitable for lamination, as low-boiling solvents such as ethyl acetate or MEK must be used for this purpose.
Therefore, the solution is distilled to recover the solvent, but the adhesive would be discarded as a distillation residue.
For the process to be both ecologically sound and economical, it is necessary to recycle the solvent by distillation. Because the solvent has a low enthalpy of vaporization and a low vapor pressure at room temperature, recovery of the solvent is possible with relatively low energy consumption and low solvent losses.
3.3.6. Characterization of the Recovered PE
Both the LDPE regranulate and the recycled film are colorless but exhibit a slight haze.
Figure 13 shows the IR spectra of LDPE after granulation and film extrusion with the spectrum of the initial film as a reference. It can be seen that some additional absorption bands appeared due to the extrusion processes. These were more intense in the spectrum of the recycled film than in the spectrum of the granulate since one more extrusion had to be performed for the film. At 1750 cm
−1, a carbonyl absorption band can be observed, which indicates the presence of ketones or aldehydes and is typical for oxidative degradation processes in LDPE [
38,
39,
40]. Furthermore, two additional absorption bands were found in the 1300–1000 cm
−1 region, indicating the presence of C–O groups, which is also typical for oxidative processes [
38,
39].
The thermal behavior of the virgin materialcompared with the recycled material was investigated by means of DSC measurements. The melting temperature in both cases was 109.74 °C, which shows that there was no effect on the melting temperature and the crystallization temperature also remained constant, which was 100.89 °C for the virgin material and 101.08 °C for the recycled material. This observation is consistent with the expectations, as it is described in the literature that no significant differences can be observed between the melting and crystallization temperatures of multiextruded LDPE and untreated LDPE [
26]. Jin et al. also describe that the percentage of crystallinity Xcr of the multiextruded LDPE remains basically constant for up to 40 extrusion cycles and only then starts to decrease [
26]. In the present case, the degree of crystallinity of the PE starting film was 38.97%, and that of the recycled film was 38.42%. There was, therefore, a decrease of 1.41% in the degree of crystallinity, which was probably due to unavoidable differences in the evaluation. The DSC measurement curves of LDPE and the data obtained from them are given in the
supporting information in Figure S3 and Table S1.
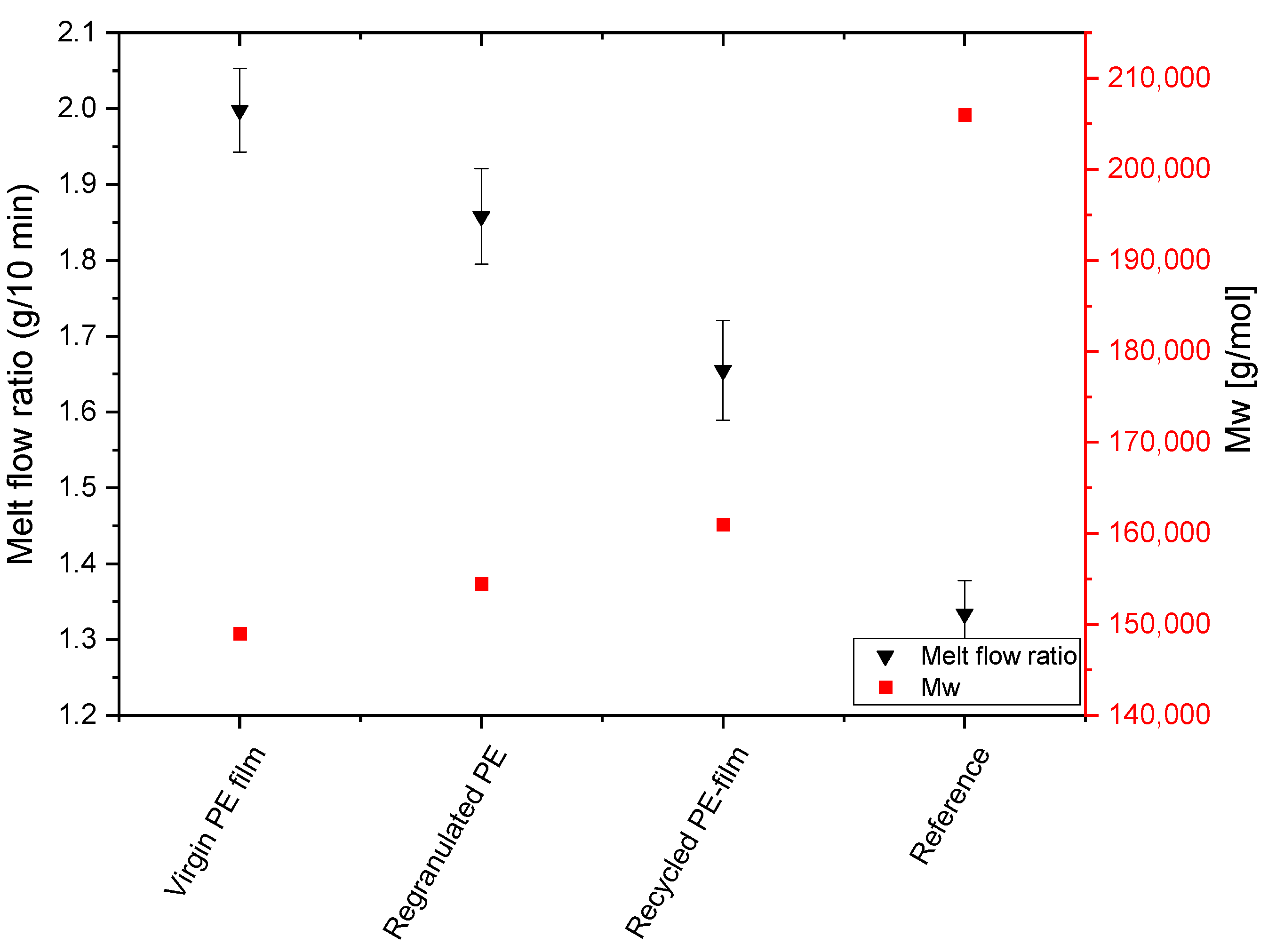
Figure 14 shows the influence of degassing/granulation and flat film extrusion on MFI and molecular weight. The literature reports that both chain scission and molecular enlargement occur simultaneously during thermal degradation of LDPE, but that crosslinking predominates over degradation, which is why extrusions lead to a decrease in MFI [
26,
41]. This is consistent with the values shown in
Figure 14, where a decrease in MFI is observed after both regranulation and extrusion of the recycled film. However, the MFI decreases reported in the literature and also the value of the reference obtained by granulating the commercial LDPE film not treated with solvent are much stronger than for the recyclate. This indicates that the residual solvent hinders the crosslinking that normally occurs during extrusion. The reason for this could be that the solvent molecules increase the distance between the molecular chains, making crosslinking more difficult. The results of the SEC measurement confirm the conclusion that extrusion in the presence of the solvent leads to a much weaker increase in molecular weight. On the other hand, with the reference where no solvent is present, the molecular weight increases strongly. This is also reflected in an increase in the polydispersity index. For the samples extruded in the presence of solvent molecules, the polydispersity index does not change significantly, from which it can be concluded that the comparatively high MFIs of the delaminated material do not result from chain degradation.
Table 5 shows the results of tensile testing of the recycled film and the virgin LDPE film used for lamination parallel to the extrusion direction.
It can be seen from the literature that due to crosslinking, an increase in tensile strength and ultimate tensile strength would be expected for recycled LDPE [
39,
42]. However, since the LDPE in the present case, as demonstrated by SEC, was only subjected to slight crosslinking, no increase in tensile strength could be observed. Instead, the measured sample exhibited a lower tensile strength and lower Young’s modulus than the virgin film. The reason for this could be a plasticizing effect of the residual solvent. The increase in free volume between the polymer chains due to plasticizing molecule leads to deformation at lower forces, making the material less elastic and thus having a lower Young’s modulus and also a lower tensile strength [
43,
44]. At the same time, the elongation of plasticized materials increases because the weakening of intermolecular interactions reduces the stiffness and increases the flexibility of the film [
45,
46,
47]. The typical behavior of plasticized polymers also includes an increase in elongation. This tendency can also be observed in the present case, although the high standard deviation does not allow any concrete statement on this. However, it must be taken into account here that the commercial film has a thickness of 45–47 µm, while the recycled film does not quite reach the target thickness of 45 µm and is rather between 42–43 µm thick. While this difference in thickness is taken into account in the tensile strength, this is not possible with elongation. With a film thickness of 45 µm of the recycled film, an even higher value for the elongation would therefore be expected [
48,
49].
The influence of the solvent on the quality of the LDPE can be reduced by reducing the solvent content prior to feeding into the extruder. This can be achieved by further optimizing the predrying or by carrying out an extraction of the solvent. The solvent content can also be further reduced by optimizing the degassing during granulation, thus further reducing its influence.
The properties of the recycled film should theoretically allow it to be used again in packaging applications. For food applications, it would first have to be checked whether the food law requirements can be met. According to EU Regulation 10/2011, this would involve guaranteeing that no more than 30 mg of solvent can pass into 1 kg of food, which is why it may only be possible to use the film behind a functional barrier [
50].
3.3.7. Characterization of the Recovered PET
The PET obtained was in the form of a predominantly transparent granule, which, however, exhibited a slight yellow coloration and also a whitish turbidity in some sections. Yellowing is more frequently observed in recycled PET and can be attributed to thermo-oxidative processes [
51,
52]. The whitish haze is due to residual LDPE that could not be removed during density separation. Since the melting point of LDPE is below that of PET, melt filtration is not possible in this case.
The influence of the recycling process on the molecular weight of PET is shown in
Table 6. Here, it can be seen that the solvent treatment and the predrying had no discernible influence on the molecular weight of the PET. During regranulation, however, a slight reduction in molecular weight took place. The determined weight-averaged molar mass of the regranulate was 36,653 g/mol and thus just below the assumed error range of 10% of the starting material, which has a Mw of 41,564 g/mol.
It is well known that the Mw of PET often decreases during extrusion in recycling processes. In the case of pure PET, this can occur due to the presence of water in the form of hydrolytic chain scission or thermal chain scission [
53]. One reason why only slight molar mass degradation occurred in the present case may be the thorough predrying of the material prior to extrusion or the maximum extrusion temperature of 260 °C, which is lower than the temperature of 280 °C usually mentioned as extrusion temperature for PET in the literature [
53].
The spectrum of the regranulated PET shows some differences from the spectrum of the virgin material. For one thing, changes can be observed in the C–H region between 3000–2800 cm
−1. In the regranulate, two newly appeared bands at 2907 cm
−1 and at 2853 cm
−1 can be seen, which can be attributed to the LDPE content in the PET fraction. On the other hand, the other PE-typical bands at 1463 and 718 cm
−1 are not found, probably because their intensity is lower and there are overlaps in the fingerprint region. The bands of PET in the C–H region shift slightly and change their intensities, which can be attributed to either chain scissions, conformational changes, or both [
54,
55]. The bands with wavelengths 1470, 1340, 1118, 970, and 845 cm
−1 significantly lost intensity somewhat, while the bands at 1370, 1044, and 898 cm
−1 significantly gained intensity. These bands are characteristic of the trans or gauche rotational conformers of the ethylene glycol moiety in PET [
55]. Since both conformers are found in the amorphous portion of the polymer, only the trans conformer is found in the crystalline phase. Since in the present case the absorption bands characteristic of the trans conformer decreased, this indicates that the regranulate has a higher amorphous fraction than the PET film used for laminate production. It is known from the literature that as a consequence of molar mass degradation during extrusion, an enrichment of trans conformers, and thus an increase in the overall degree of crystallinity, occurs [
54,
55,
56]. Therefore, the increase in gauche content occurring in this case after the extrusion step must have a cause other than chain degradation. Another possible explanation could be that the crystallization of PET is disturbed by the contamination with LDPE. It has been reported in the literature that this is possible even though the two polymers are immiscible with each other [
57].
However, this assumption can be ruled out on the basis of the DSC curves. The DSC measurement of the PET granulate shows that during the first heating process, a postcrystallization of the PET took place, which means that the conclusions drawn from the IR spectrum regarding the crystallinity were correct and the degree of crystallization of the regranulated PET was 13.91%.
During the second heating run, however, no postcrystallization of the PET could be detected. The degree of crystallization determined from the measurement curve of the second heating process was 34.09%, which was 2% higher compared with the degree of crystallinity of the virgin film, which was 33.21%.
This means that the LDPE content did not hinder the crystallization of the PET in general, but that the cooling process during the production of the granulate prevented the crystallization. The fact that, in addition, the melt temperatures for both virgin and recycled material were 253.73 °C and 253.81 °C, respectively, shows that molar mass degradation has no effect on the thermal properties of PET. Additionally, it is worth mentioning that the LDPE content of the granulate is not visible in the DSC curve, which means that the LDPE content cannot crystallize. The DSC curves of the PET and the measurement data determined from them can be found in the
Supplementary Information in Figures S4 and S5 and Table S2.
It should be possible to reduce the proportion of LDPE in PET using cyclonic separation, but it is nevertheless unlikely that the LDPE can be completely removed. Thus, in order to still be able to achieve good mechanical properties, the addition of compatibilizers might be necessary [
7,
58]. Such improved treatment or compatibilization of PET could then also lead to the possibility of reusing it in packaging applications. However, the food law restrictions already described for LDPE also apply here.