Tuning Nitrogen-Doped Carbon Electrodes via Synthesis Temperature Adjustment to Improve Sodium- and Lithium-Ion Storage
Abstract
1. Introduction
2. Materials and Methods
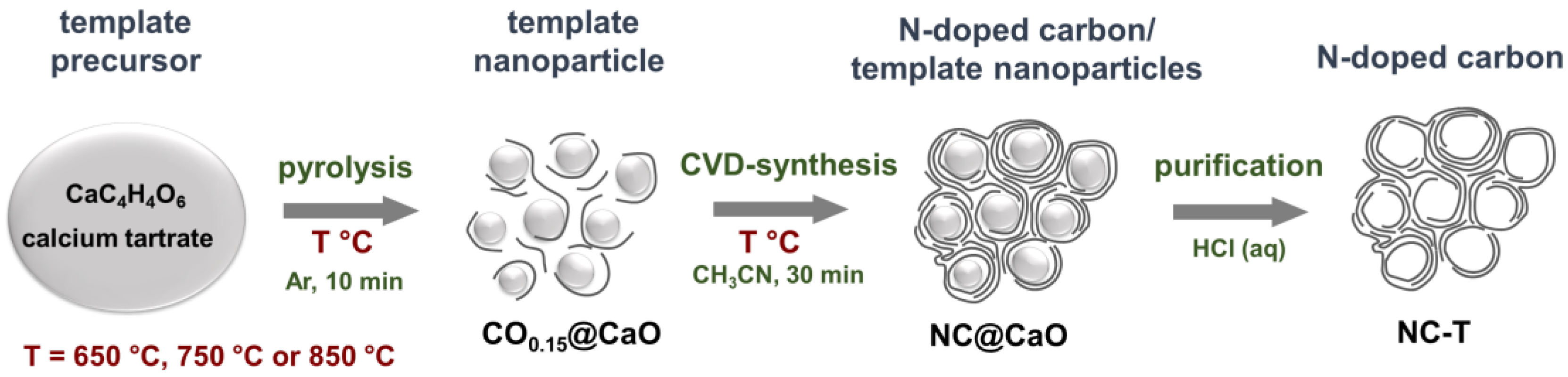
2.1. Synthesis
2.2. Characterization Instruments
2.3. Electrochemical Measurements
2.4. Quantum Chemical Calculations
3. Results
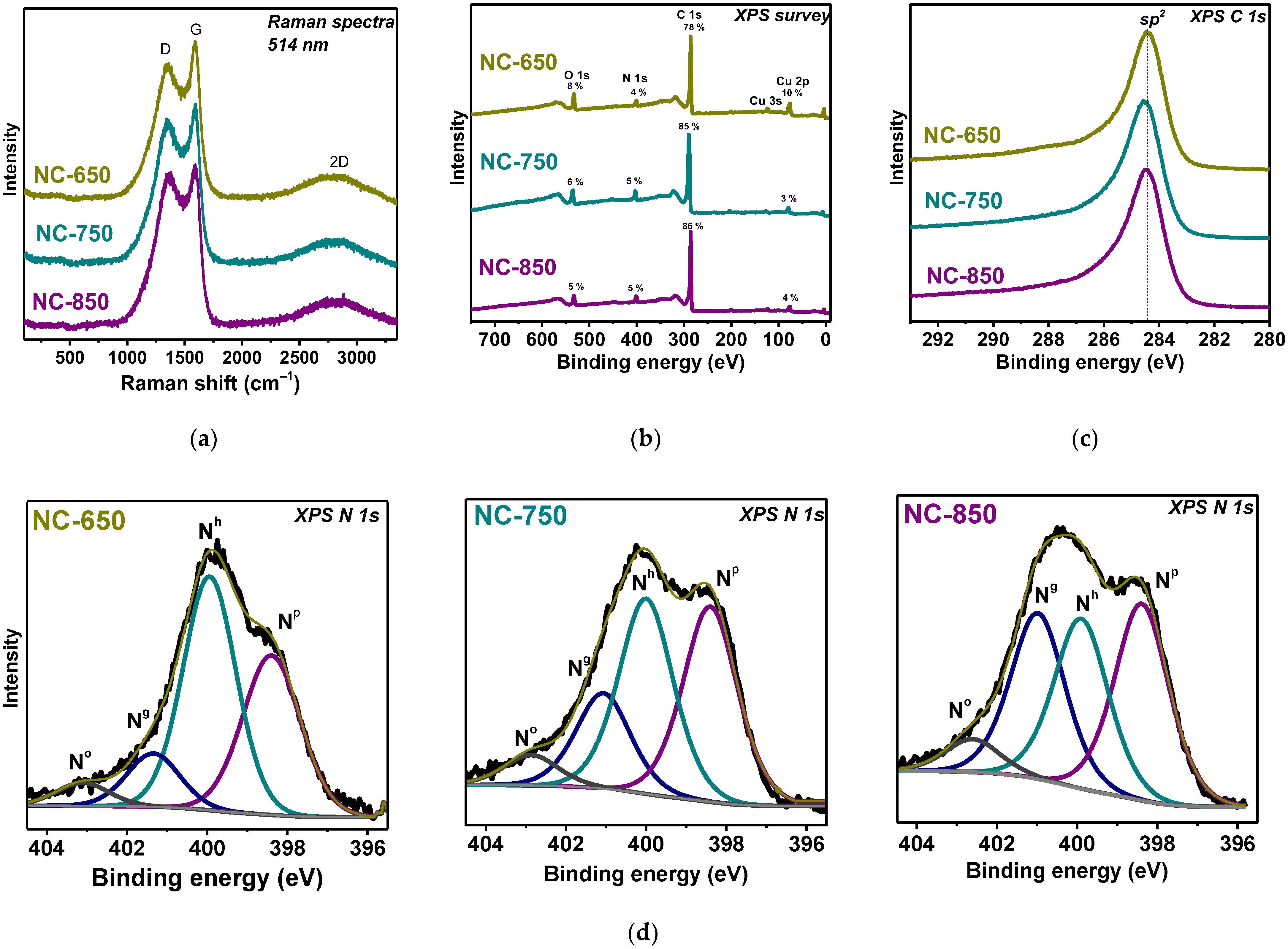
3.1. Structural Aspects
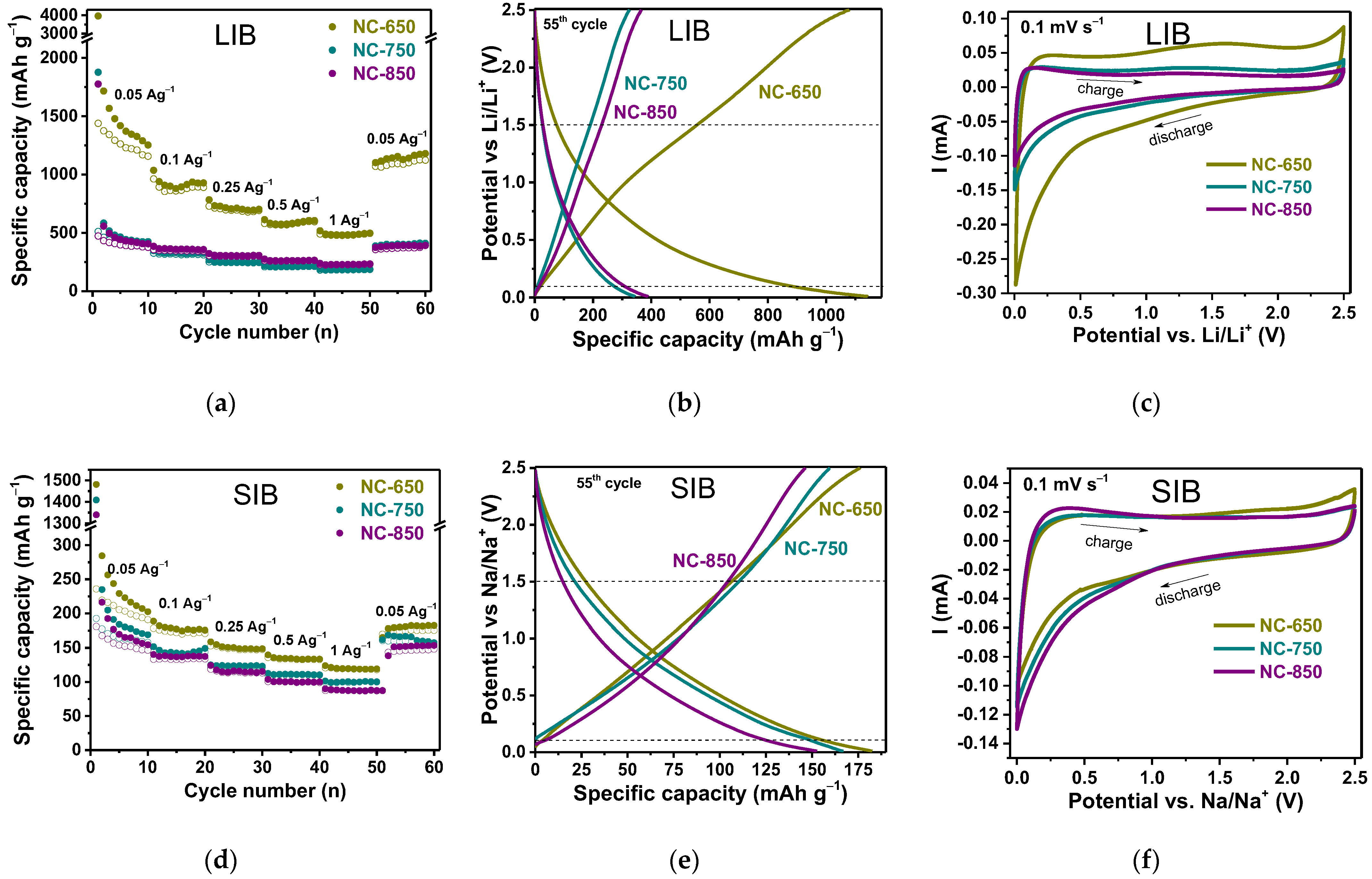
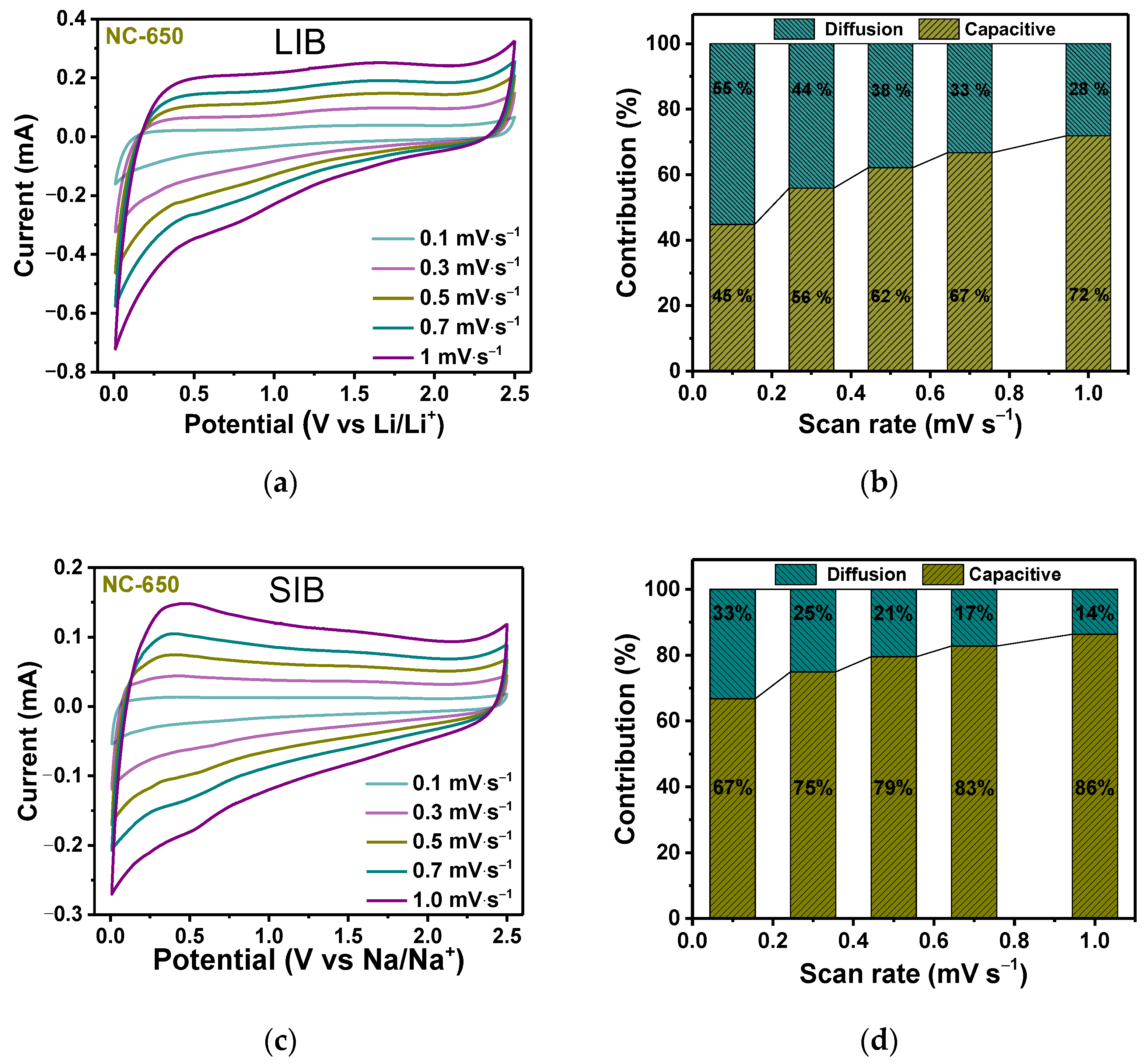
3.2. Electrochemical Performance
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cao, Y.; Xiao, L.; Sushko, M.L.; Wang, W.; Schwenzer, B.; Xiao, J.; Nie, Z.; Saraf, L.V.; Yang, Z.; Liu, J. Sodium Ion Insertion in Hollow Carbon Nanowires for Battery Applications. Nano Lett. 2012, 12, 3783–3787. [Google Scholar] [CrossRef]
- Olsson, E.; Cottom, J.; Au, H.; Guo, Z.; Jensen, A.C.S.; Alptekin, H.; Drew, A.J.; Titirici, M.M.; Cai, Q. Elucidating the Effect of Planar Graphitic Layers and Cylindrical Pores on the Storage and Diffusion of Li, Na, and K in Carbon Materials. Adv. Funct. Mater. 2020, 30, 1908209. [Google Scholar] [CrossRef]
- Karatrantos, A.; Cai, Q. Effects of Pore Size and Surface Charge on Na Ion Storage in Carbon Nanopores. Phys. Chem. Chem. Phys. 2016, 18, 30761–30769. [Google Scholar] [CrossRef]
- Datta, D.; Li, J.; Shenoy, V.B. Defective Graphene as a High-Capacity Anode Material for Na- and Ca-Ion Batteries. ACS Appl. Mater. Interfaces 2014, 6, 1788–1795. [Google Scholar] [CrossRef] [PubMed]
- Tsai, P.C.; Chung, S.C.; Lin, S.K.; Yamada, A. Ab Initio Study of Sodium Intercalation into Disordered Carbon. J. Mater. Chem. A 2015, 3, 9763–9768. [Google Scholar] [CrossRef]
- Matei Ghimbeu, C.; Górka, J.; Simone, V.; Simonin, L.; Martinet, S.; Vix-Guterl, C. Insights on the Na+ Ion Storage Mechanism in Hard Carbon: Discrimination between the Porosity, Surface Functional Groups and Defects. Nano Energy 2018, 44, 327–335. [Google Scholar] [CrossRef]
- Nie, W.; Cheng, H.; Liu, X.; Sun, Q.; Tian, F.; Yao, W.; Liang, S.; Lu, X.; Zhou, J. Surface Organic Nitrogen-Doping Disordered Biomass Carbon Materials with Superior Cycle Stability in the Sodium-Ion Batteries. J. Power Sources 2022, 522, 230994. [Google Scholar] [CrossRef]
- Liu, T.; Li, X. Biomass-Derived Nanostructured Porous Carbons for Sodium Ion Batteries: A Review. Mater. Technol. 2019, 34, 232–245. [Google Scholar] [CrossRef]
- Sun, N.; Liu, H.; Xu, B. Facile Synthesis of High Performance Hard Carbon Anode Materials for Sodium Ion Batteries. J. Mater. Chem. A 2015, 3, 20560–20566. [Google Scholar] [CrossRef]
- Liu, H.; Jia, M.; Yue, S.; Cao, B.; Zhu, Q.; Sun, N.; Xu, B. Creative Utilization of Natural Nanocomposites: Nitrogen-Rich Mesoporous Carbon for a High-Performance Sodium Ion Battery. J. Mater. Chem. A 2017, 5, 9572–9579. [Google Scholar] [CrossRef]
- Gaddam, R.R.; Farokh Niaei, A.H.; Hankel, M.; Searles, D.J.; Kumar, N.A.; Zhao, X.S. Capacitance-Enhanced Sodium-Ion Storage in Nitrogen-Rich Hard Carbon. J. Mater. Chem. A 2017, 5, 22186–22192. [Google Scholar] [CrossRef]
- Vu, N.H.; Le, H.T.T.; Hoang, V.H.; Dao, V.D.; Huu, H.T.; Jun, Y.S.; Im, W. Bin Highly N-Doped, H-Containing Mesoporous Carbon with Modulated Physicochemical Properties as High-Performance Anode Materials for Li-Ion and Na-Ion Batteries. J. Alloys Compd. 2021, 851, 156881. [Google Scholar] [CrossRef]
- Samdani, J.S.; Tran, T.N.; Kang, T.H.; Lee, B.J.; Jang, Y.H.; Yu, J.S.; Shanmugam, S. The Identification of Specific N-Configuration Responsible for Li-Ion Storage in N-Doped Porous Carbon Nanofibers: An Ex-Situ Study. J. Power Sources 2021, 483, 229174. [Google Scholar] [CrossRef]
- Feng, X.; Bai, Y.; Zheng, L.; Liu, M.; Li, Y.; Zhao, R.; Li, Y.; Wu, C. Effect of Different Nitrogen Configurations on Sodium Storage Properties of Carbon Anodes for Sodium Ion Batteries. ACS Appl. Mater. Interfaces 2021, 13, 56285–56295. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Shen, Z.; Chang, G.; Li, Z.; Zhao, H. Mechanochemistry Induced Pore Regulation and Pyridinic Nitrogen Doping in Anthracite Derived Carbon for Sodium Storage. Diam. Relat. Mater. 2022, 130, 109481. [Google Scholar] [CrossRef]
- Qu, Y.; Guo, M.; Zeng, F.; Zou, C.; Yuan, C.; Zhang, X.; Li, Q.; Lu, H. Synthesis of Nitrogen-Doped Porous Carbon Nanofibers as an Anode Material for High Performance Sodium-Ion Batteries. Solid State Ionics 2019, 337, 170–177. [Google Scholar] [CrossRef]
- Agrawal, A.; Janakiraman, S.; Biswas, K.; Venimadhav, A.; Srivastava, S.K.; Ghosh, S. Understanding the Improved Electrochemical Performance of Nitrogen-Doped Hard Carbons as an Anode for Sodium Ion Battery. Electrochim. Acta 2019, 317, 164–172. [Google Scholar] [CrossRef]
- Xu, K.; Li, Y.; Liu, Y.; Zhong, G.; Wang, C.; Su, W.; Li, X.; Yang, C. Na+-Storage Properties Derived from a High Pseudocapacitive Behavior for Nitrogen-Doped Porous Carbon Anode. Mater. Lett. 2020, 261, 127064. [Google Scholar] [CrossRef]
- Lee, H.W.; Moon, H.S.; Hur, J.; Kim, I.T.; Park, M.S.; Yun, J.M.; Kim, K.H.; Lee, S.G. Mechanism of Sodium Adsorption on N-Doped Graphene Nanoribbons for Sodium Ion Battery Applications: A Density Functional Theory Approach. Carbon 2017, 119, 492–501. [Google Scholar] [CrossRef]
- Kado, Y.; Soneda, Y.; Yoshizawa, N. Excellent Rate Capability of MgO-Templated Mesoporous Carbon as an Na-Ion Energy Storage Material. ECS Electrochem. Lett. 2014, 4, A22–A23. [Google Scholar] [CrossRef]
- Cao, B.; Liu, H.; Xu, B.; Lei, Y.; Chen, X.; Song, H. Mesoporous Soft Carbon as an Anode Material for Sodium Ion Batteries with Superior Rate and Cycling Performance. J. Mater. Chem. A 2016, 4, 6472–6478. [Google Scholar] [CrossRef]
- Hsieh, Y.Y.; Fang, Y.; Daum, J.; Kanakaraj, S.N.; Zhang, G.; Mishra, S.; Gbordzoe, S.; Shanov, V. Bio-Inspired, Nitrogen Doped CNT-Graphene Hybrid with Amphiphilic Properties as a Porous Current Collector for Lithium-Ion Batteries. Carbon 2019, 145, 677–689. [Google Scholar] [CrossRef]
- Wang, B.; Abdulla, W.A.; Wang, D.; Zhao, X.S. A Three-Dimensional Porous LiFePO4 Cathode Material Modified with a Nitrogen-Doped Graphene Aerogel for High-Power Lithium Ion Batteries. Energy Environ. Sci. 2015, 8, 869–875. [Google Scholar] [CrossRef]
- Ding, Y.L.; Kopold, P.; Hahn, K.; Van Aken, P.A.; Maier, J.; Yu, Y. Facile Solid-State Growth of 3D Well-Interconnected Nitrogen-Rich Carbon Nanotube-Graphene Hybrid Architectures for Lithium-Sulfur Batteries. Adv. Funct. Mater. 2016, 26, 1112–1119. [Google Scholar] [CrossRef]
- Fang, Y.; Hsieh, Y.Y.; Khosravifar, M.; Johnson, K.; Kwasi Adusei, P.; Kanakaraj, S.N.; Preisler, S.; Zhang, G.; Shanov, V. Lithiophilic Current Collector Based on Nitrogen Doped Carbon Nanotubes and Three-Dimensional Graphene for Long-Life Lithium Metal Batteries. Mater. Sci. Eng. B Solid-State Mater. Adv. Technol. 2021, 267, 115067. [Google Scholar] [CrossRef]
- Shlyakhova, E.V.; Bulusheva, L.G.; Kanygin, M.A.; Plyusnin, P.E.; Kovalenko, K.A.; Senkovskiy, B.V.; Okotrub, A.V. Synthesis of Nitrogen-Containing Porous Carbon Using Calcium Oxide Nanoparticles. Phys. Status Solidi Basic Res. 2014, 251, 2607–2612. [Google Scholar] [CrossRef]
- Lapteva, L.L.; Fedoseeva, Y.V.; Shlyakhova, E.V.; Makarova, A.A.; Bulusheva, L.G.; Okotrub, A.V. NEXAFS Spectroscopy Study of Lithium Interaction with Nitrogen Incorporated in Porous Graphitic Material. J. Mater. Sci. 2019, 54, 11168–11178. [Google Scholar] [CrossRef]
- Shlyakhova, E.V.; Okotrub, A.V.; Fedoseeva, Y.V.; Fedorovskaya, E.O.; Mel’gunova, E.A.; Mel’gunov, M.S.; Koroteev, V.O.; Makarova, A.A.; Zhou, J.; Song, H.; et al. Iron Induced Porosity of the Templated Carbon for Enhancement of Electrochemical Capacitance. Appl. Surf. Sci. 2021, 543, 148565. [Google Scholar] [CrossRef]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized Gradient Approximation Made Simple. Phys. Rev. Lett. 1996, 77, 3865–3868. [Google Scholar] [CrossRef]
- Grimme, S.; Antony, J.; Ehrlich, S.; Krieg, H. A Consistent and Accurate Ab Initio Parametrization of Density Functional Dispersion Correction (DFT-D) for the 94 Elements H-Pu. J. Chem. Phys. 2010, 132, 154104. [Google Scholar] [CrossRef]
- Fedoseeva, Y.V.; Shlyakhova, E.V.; Stolyarova, S.G.; Vorfolomeeva, A.A.; Grebenkina, M.A.; Makarova, A.A.; Shubin, Y.V.; Okotrub, A.V.; Bulusheva, L.G. Brominated Porous Nitrogen-Doped Carbon Materials for Sodium-Ion Storage. Batteries 2022, 8, 114. [Google Scholar] [CrossRef]
- Liu, W.; Low, N.; Feng, B.; Wang, G.; da Costa, J.C.D. Calcium Precursors for the Production of CaO Sorbents for Multicycle CO2 Capture. Environ. Sci. Technol. 2010, 44, 841–847. [Google Scholar] [CrossRef] [PubMed]
- Asanov, I.P.; Asanova, T.I.; Bulusheva, L.G.; Shlyakhova, E.V.; Okotrub, A.V.; Flahaut, E. Thermal Decomposition of Co-Doped Calcium Tartrate and Use of the Products for Catalytic Chemical Vapor Deposition Synthesis of Carbon Nanotubes. J. Phys. Chem. C 2012, 116, 343–351. [Google Scholar] [CrossRef]
- Nishchakova, A.D.; Grebenkina, M.A.; Shlyakhova, E.V.; Shubin, Y.V.; Kovalenko, K.A.; Asanov, I.P.; Fedoseeva, Y.V.; Makarova, A.A.; Okotrub, A.V.; Bulusheva, L.G. Porosity and Composition of Nitrogen-Doped Carbon Materials Templated by the Thermolysis Products of Calcium Tartrate and Their Performance in Electrochemical Capacitors. J. Alloys Compd. 2021, 858, 158259. [Google Scholar] [CrossRef]
- Han, M.L.; Wei, X.L.; Zhang, J.C.; Liu, Y.; Tang, X.; Li, P.; Liu, Z.Y. Influence of Structural Damage on Evaluation of Microscopic Pore Structure in Marine Continental Transitional Shale of the Southern North China Basin: A Method Based on the Low-Temperature N2 Adsorption Experiment. Pet. Sci. 2022, 19, 100–115. [Google Scholar] [CrossRef]
- Ferrari, A.C.; Meyer, J.C.; Scardaci, V.; Casiraghi, C.; Lazzeri, M.; Mauri, F.; Piscanec, S.; Jiang, D.; Novoselov, K.S.; Roth, S.; et al. Raman Spectrum of Graphene and Graphene Layers. Phys. Rev. Lett. 2006, 97, 187401. [Google Scholar] [CrossRef]
- Cançado, L.G.; Pimenta, M.A.; Neves, B.R.A.; Dantas, M.S.S.; Jorio, A. Influence of the Atomic Structure on the Raman Spectra of Graphite Edges. Phys. Rev. Lett. 2004, 93, 247401. [Google Scholar] [CrossRef]
- Hoekstra, J.; Beale, A.M.; Soulimani, F.; Versluijs-Helder, M.; Van De Kleut, D.; Koelewijn, J.M.; Geus, J.W.; Jenneskens, L.W. The Effect of Iron Catalyzed Graphitization on the Textural Properties of Carbonized Cellulose: Magnetically Separable Graphitic Carbon Bodies for Catalysis and Remediation. Carbon 2016, 107, 248–260. [Google Scholar] [CrossRef]
- Vasquez, R.P. Highly Oriented Pyrolytic Graphite by XPS. Surf. Sci. Spectra 1992, 1, 238–241. [Google Scholar] [CrossRef]
- Lapteva, L.L.; Fedoseeva, Y.V.; Gevko, P.N.; Smirnov, D.A.; Gusel’nikov, A.V.; Bulusheva, L.G.; Okotrub, A.V. X-ray Spectroscopy Study of Lithiated Graphite Obtained by Thermal Deposition of Lithium. J. Struct. Chem. 2017, 58, 1173–1179. [Google Scholar] [CrossRef]
- Arrigo, R.; Hävecker, M.; Wrabetz, S.; Blume, R.; Lerch, M.; McGregor, J.; Parrott, E.P.J.; Zeitler, J.A.; Gladden, L.F.; Knop-Gericke, A.; et al. Tuning the Acid/Base Properties of Nanocarbons by Functionalization via Amination. J. Am. Chem. Soc. 2010, 132, 9616–9630. [Google Scholar] [CrossRef] [PubMed]
- Fedoseeva, Y.V.; Pozdnyakov, G.A.; Okotrub, A.V.; Kanygin, M.A.; Nastaushev, Y.V.; Vilkov, O.Y.; Bulusheva, L.G. Effect of Substrate Temperature on the Structure of Amorphous Oxygenated Hydrocarbon Films Grown with a Pulsed Supersonic Methane Plasma Flow. Appl. Surf. Sci. 2016, 385, 464–471. [Google Scholar] [CrossRef]
- Desimoni, E.; Casella, G.I.; Cataldi, T.R.I.; Malitesta, C. A Comparison of Some Asymmetrical Line Shapes for XPS Data Analysis. J. Electron Spectros. Relat. Phenomena 1989, 49, 247–261. [Google Scholar] [CrossRef]
- Bulusheva, L.G.; Okotrub, A.V.; Fedoseeva, Y.V.; Kurenya, A.G.; Asanov, I.P.; Vilkov, O.Y.; Koós, A.A.; Grobert, N. Controlling Pyridinic, Pyrrolic, Graphitic, and Molecular Nitrogen in Multi-Wall Carbon Nanotubes Using Precursors with Different N/C Ratios in Aerosol Assisted Chemical Vapor Deposition. Phys. Chem. Chem. Phys. 2015, 17, 23741–23747. [Google Scholar] [CrossRef] [PubMed]
- Scardamaglia, M.; Struzzi, C.; Aparicio Rebollo, F.J.; De Marco, P.; Mudimela, P.R.; Colomer, J.F.; Amati, M.; Gregoratti, L.; Petaccia, L.; Snyders, R.; et al. Tuning Electronic Properties of Carbon Nanotubes by Nitrogen Grafting: Chemistry and Chemical Stability. Carbon 2015, 83, 118–127. [Google Scholar] [CrossRef]
- Jiang, Q.; Zhang, Z.; Yin, S.; Guo, Z.; Wang, S.; Feng, C. Biomass Carbon Micro/Nano-Structures Derived from Ramie Fibers and Corncobs as Anode Materials for Lithium-Ion and Sodium-Ion Batteries. Appl. Surf. Sci. 2016, 379, 73–82. [Google Scholar] [CrossRef]
- Saurel, D.; Orayech, B.; Xiao, B.; Carriazo, D.; Li, X.; Rojo, T. From Charge Storage Mechanism to Performance: A Roadmap toward High Specific Energy Sodium-Ion Batteries through Carbon Anode Optimization. Adv. Energy Mater. 2018, 8, 1703268. [Google Scholar] [CrossRef]
- Huang, S.; Li, Z.; Wang, B.; Zhang, J.; Peng, Z.; Qi, R.; Wang, J.; Zhao, Y. N-Doping and Defective Nanographitic Domain Coupled Hard Carbon Nanoshells for High. Adv. Funct. Mater. 2018, 28, 1706294. [Google Scholar] [CrossRef]
- Velez, V.; Ramos-Sánchez, G.; Lopez, B.; Lartundo-Rojas, L.; González, I.; Sierra, L. Synthesis of Novel Hard Mesoporous Carbons and Their Application as Anodes for Li and Na Ion Batteries. Carbon 2019, 147, 214–226. [Google Scholar] [CrossRef]
- Stevens, D.A.; Dahn, J.R. High Capacity Anode Materials for Rechargeable Sodium-ion Batteries. J. Electrochem. Soc. 2000, 147, 1271–1273. [Google Scholar] [CrossRef]
- Shi, W.; Zhang, Y.; Tian, Z.Q.; Pan, Z.; Key, J.; Shen, P.K. Low Temperature Synthesis of Polyhedral Hollow Porous Carbon with High Rate Capability and Long-Term Cycling Stability as Li-Ion and Na-Ion Battery Anode Material. J. Power Sources 2018, 398, 149–158. [Google Scholar] [CrossRef]
- Yu, K.; Wang, J.; Wang, X.; Liang, J.; Liang, C. Sustainable Application of Biomass By-Products: Corn Straw-Derived Porous Carbon Nanospheres Using as Anode Materials for Lithium Ion Batteries. Mater. Chem. Phys. 2020, 243, 122644. [Google Scholar] [CrossRef]
- Kuo, S.L.; Liu, W.R.; Kuo, C.P.; Wu, N.L.; Wu, H.C. Lithium Storage in Reduced Graphene Oxides. J. Power Sources 2013, 244, 552–556. [Google Scholar] [CrossRef]
- Chen, Y.; Shi, L.; Li, A.; Zhang, S.; Guo, M.; Chen, X.; Zhou, J.; Song, H. Capacity Enhancement of Porous Carbon Electrodes during Long-Term Cycling in Lithium-Ion Batteries. J. Electrochem. Soc. 2017, 164, A2000–A2006. [Google Scholar] [CrossRef]
- Wang, H.; Yu, W.; Shi, J.; Mao, N.; Chen, S.; Liu, W. Biomass Derived Hierarchical Porous Carbons as High-Performance Anodes for Sodium-Ion Batteries. Electrochim. Acta 2016, 188, 103–110. [Google Scholar] [CrossRef]
- Luo, X.F.; Yang, C.H.; Peng, Y.Y.; Pu, N.W.; Der Ger, M.; Te Hsieh, C.; Chang, J.K. Graphene Nanosheets, Carbon Nanotubes, Graphite, and Activated Carbon as Anode Materials for Sodium-Ion Batteries. J. Mater. Chem. A 2015, 3, 10320–10326. [Google Scholar] [CrossRef]
- Wang, C.; Appleby, A.J.; Little, F.E. Electrochemical Impedance Study of Initial Lithium Ion Intercalation into Graphite Powders. Electrochim. Acta 2001, 46, 1793–1813. [Google Scholar] [CrossRef]
- Vyroubal, P.; Kazda, T. Equivalent Circuit Model Parameters Extraction for Lithium Ion Batteries Using Electrochemical Impedance Spectroscopy. J. Energy Storage 2018, 15, 23–31. [Google Scholar] [CrossRef]
- Funabiki, A.; Inaba, M.; Ogumi, Z.A.c. Impedance Analysis of Electrochemical Lithium Intercalation into Highly Oriented Pyrolytic Graphite. J. Power Sources 1997, 68, 227–231. [Google Scholar] [CrossRef]
- Ho, C.; Raistrick, I.D.; Huggins, R.A. Application of A-C Techniques to the Study of Lithium Diffusion in Tungsten Trioxide Thin Films. J. Electrochem. Soc. 1980, 127, 343–350. [Google Scholar] [CrossRef]
- Guo, H.; Li, X.; Zhang, X.; Wang, H.; Wang, Z.; Peng, W. Diffusion Coefficient of Lithium in Artificial Graphite, Mesocarbon Microbeads, and Disordered Carbon. New Carbon Mater. 2007, 22, 7–10. [Google Scholar] [CrossRef]
- Chang, Y.C.; Sohn, H.J. Electrochemical Impedance Analysis for Lithium Ion Intercalation into Graphitized Carbons. J. Electrochem. Soc. 2000, 147, 50–58. [Google Scholar] [CrossRef]
- Tang, Z.Y.; Xue, J.J.; Liu, C.Y.; Zhuang, X.G. Determination of the Lithium Ion Diffusion Coefficient in Graphite Anode Material. J. Electrochem. Soc. 1991, 146, 8–14. [Google Scholar] [CrossRef]
- NuLi, Y.; Yang, J.; Jiang, Z. Intercalation of Lithium Ions into Bulk and Powder Highly Oriented Pyrolytic Graphite. J. Phys. Chem. Solids 2006, 67, 882–886. [Google Scholar] [CrossRef]
- Brühwiler, P.A.; Maxwell, A.J.; Puglia, C.; Nilsson, A.; Andersson, S.; Mårtensson, N. π* and σ* Excitons in C 1s Absorption of Graphite. Phys. Rev. Lett. 1995, 74, 614–617. [Google Scholar] [CrossRef]
- Zhang, L.; Li, X.; Augustsson, A.; Lee, C.M.; Rubensson, J.E.; Nordgren, J.; Ross, P.N.; Guo, J.H. Revealing the Electronic Structure of LiC6 by Soft X-ray Spectroscopy. Appl. Phys. Lett. 2017, 110, 104106. [Google Scholar] [CrossRef]
- Minasian, S.G.; Keith, J.M.; Batista, E.R.; Boland, K.S.; Kozimor, S.A.; Martin, R.L.; Shuh, D.K.; Tyliszczak, T.; Vernon, L.J. Carbon K-Edge X-ray Absorption Spectroscopy and Time-Dependent Density Functional Theory Examination of Metal-Carbon Bonding in Metallocene Dichlorides. J. Am. Chem. Soc. 2013, 135, 14731–14740. [Google Scholar] [CrossRef]
- Carlegrim, E.; Gao, B.; Kanciurzewska, A.; De Jong, M.P.; Wu, Z.; Luo, Y.; Fahlman, M. Near-Edge X-ray Absorption Studies of Na-Doped Tetracyanoethylene Films: A Model System for the V(TCNE)x Room-Temperature Molecular Magnet. Phys. Rev. B 2008, 77, 054420. [Google Scholar] [CrossRef]
- Bulusheva, L.G.; Stolyarova, S.G.; Chuvilin, A.L.; Shubin, Y.V.; Asanov, I.P.; Sorokin, A.M.; Mel’Gunov, M.S.; Zhang, S.; Dong, Y.; Chen, X.; et al. Creation of Nanosized Holes in Graphene Planes for Improvement of Rate Capability of Lithium-Ion Batteries. Nanotechnology 2018, 29, 134001. [Google Scholar] [CrossRef]
- Stolyarova, S.G.; Koroteev, V.O.; Shubin, Y.V.; Plyusnin, P.E.; Makarova, A.A.; Okotrub, A.V.; Bulusheva, L.G. Pressure-Assisted Interface Engineering in MoS2 Holey Graphene Hybrids for Improved. Energy Technol. 2019, 7, 1900659. [Google Scholar] [CrossRef]
- Bulushev, D.A.; Nishchakova, A.D.; Trubina, S.V.; Stonkus, O.A.; Asanov, I.P.; Okotrub, A.V.; Bulusheva, L.G. Ni-N4 Sites in a Single-Atom Ni Catalyst on N-Doped Carbon for Hydrogen Production from Formic Acid. J. Catal. 2021, 402, 264–274. [Google Scholar] [CrossRef]
- Bommier, C.; Ji, X.; Greaney, P.A. Electrochemical Properties and Theoretical Capacity for Sodium Storage in Hard Carbon: Insights from First Principles Calculations. Chem. Mater. 2019, 31, 658–677. [Google Scholar] [CrossRef]
- Liang, Z.; Fan, X.; Zheng, W.; Singh, D.J. Adsorption and Formation of Small Na Clusters on Pristine and Double-Vacancy Graphene for Anodes of Na-Ion Batteries. ACS Appl. Mater. Interfaces 2017, 9, 17076–17084. [Google Scholar] [CrossRef] [PubMed]


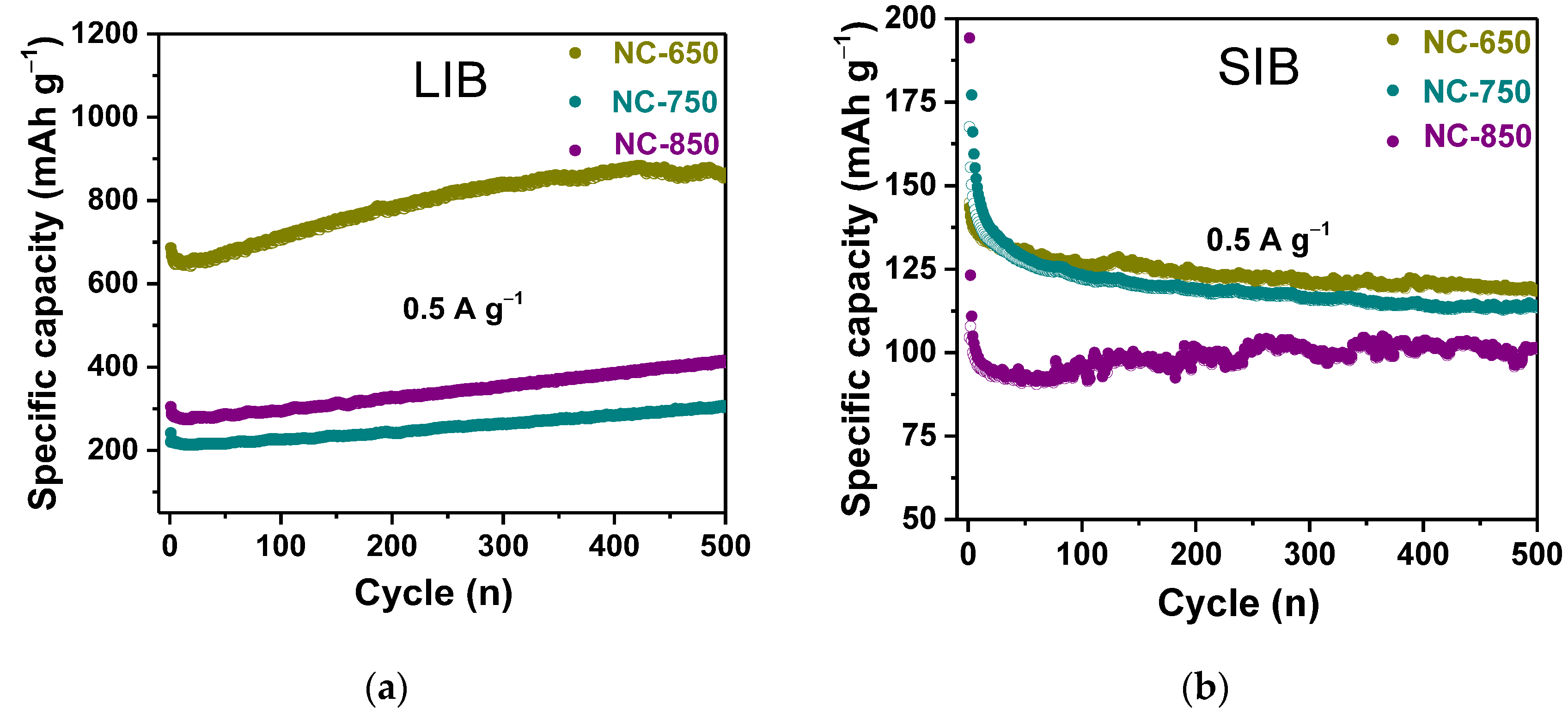
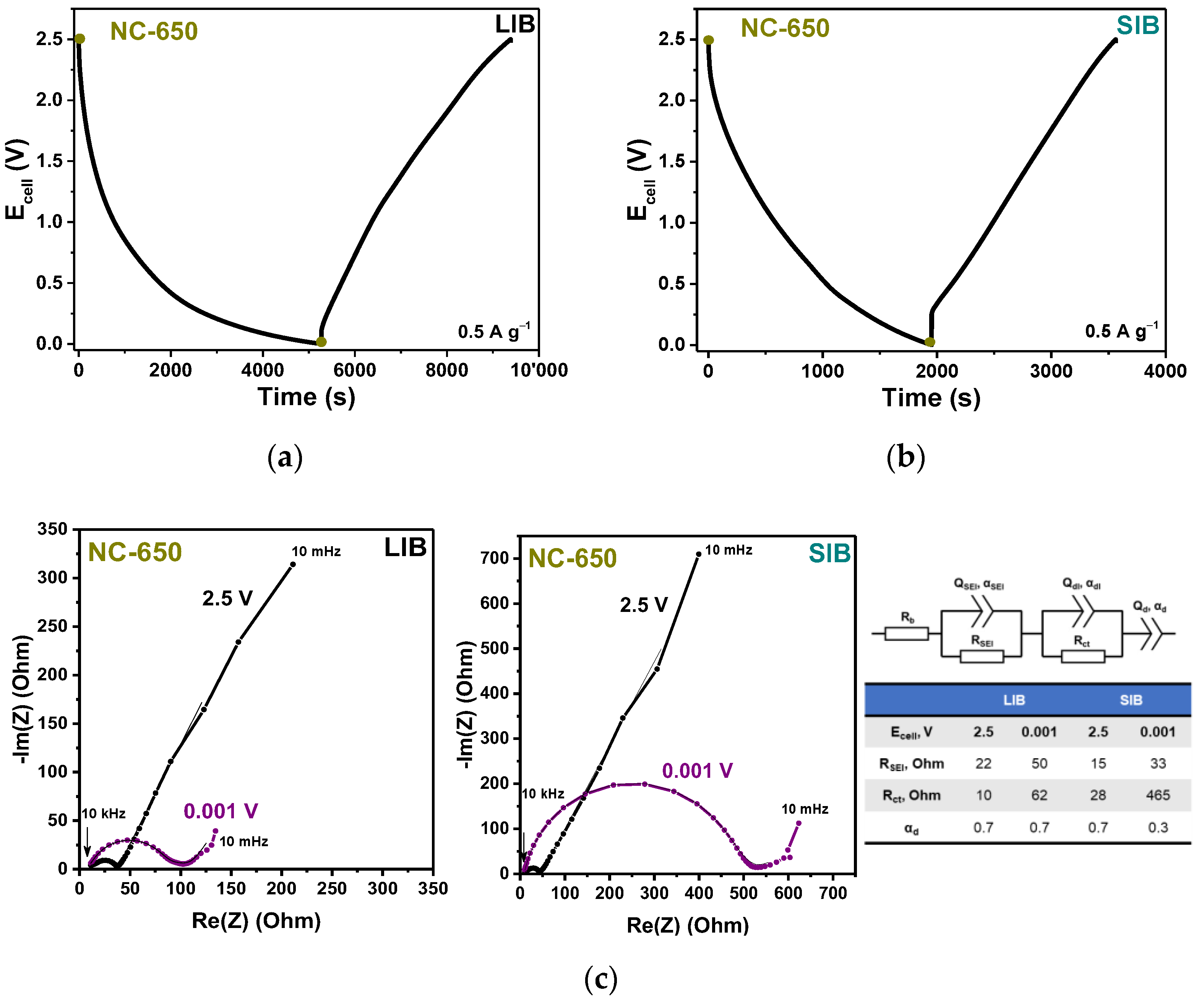
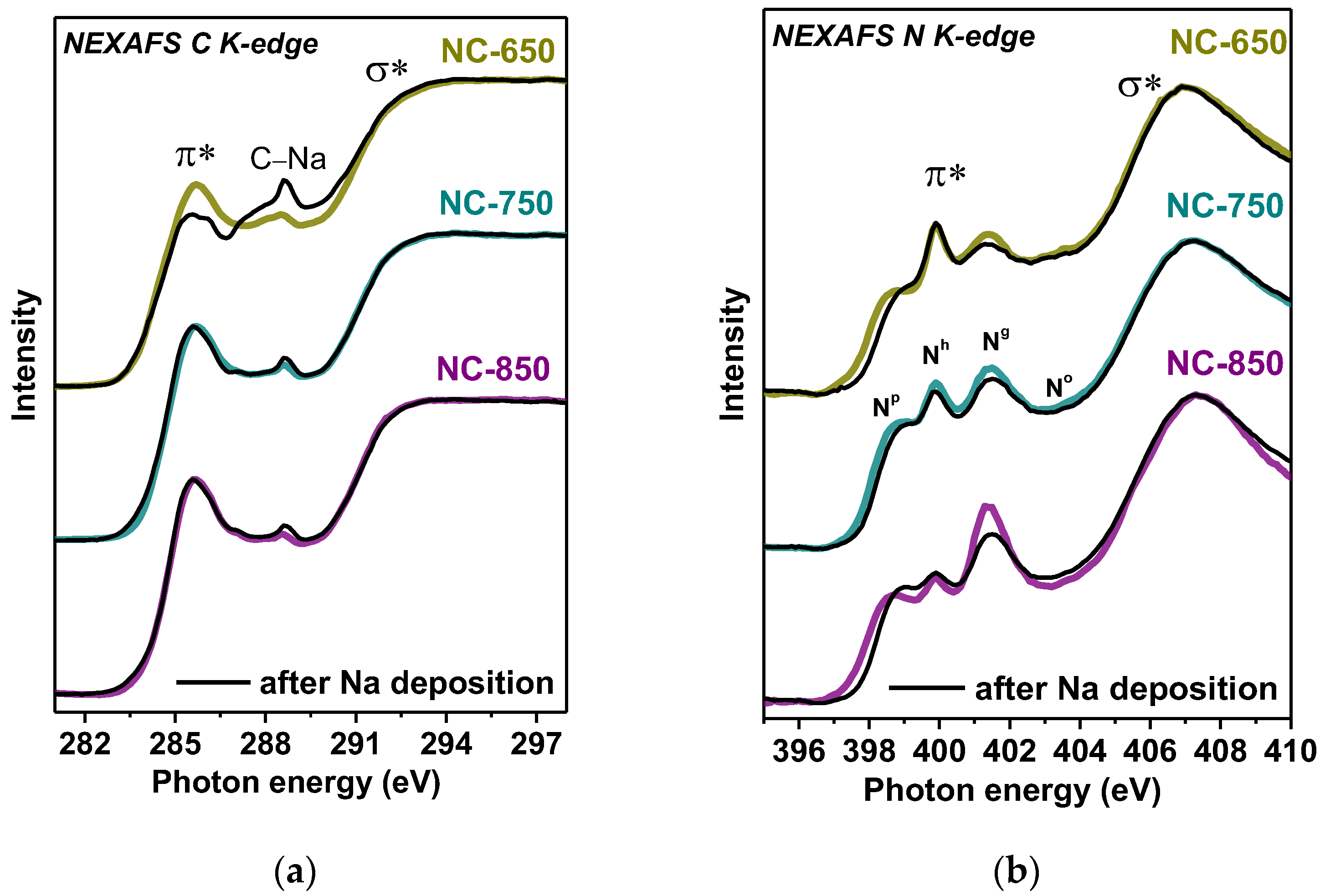
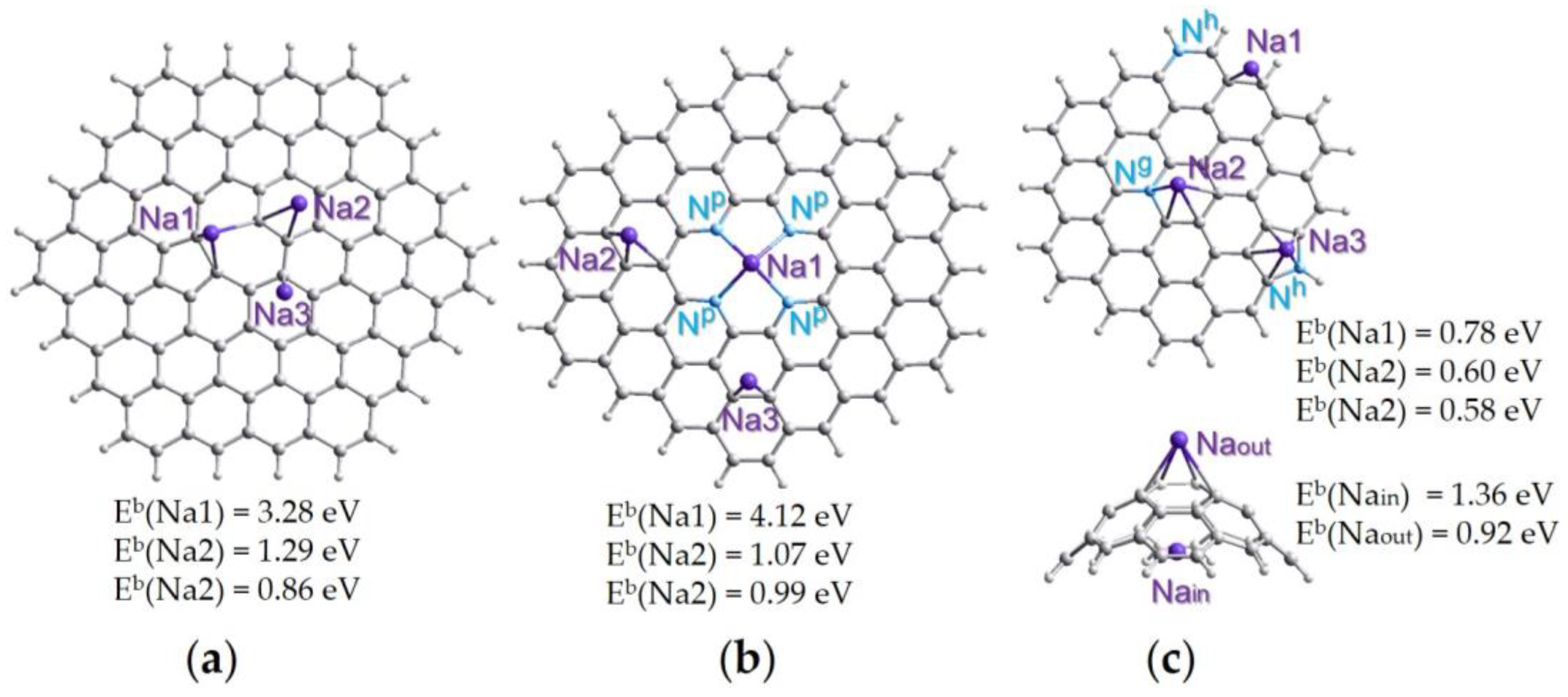
| Sample | Synthesis Temperature, °C | BET Specific Surface Area, m2·g−1 | Pore Volume, cm3·g−1 | Total Concentration of Nitrogen, at% | Concentration of Nitrogen Forms Np:Nh:Ng:No, % | LIB Capacity, mA·h·g−1 | SIB Capacity, mA·h·g−1 |
|---|---|---|---|---|---|---|---|
| NC-650 | 650 | 770 | 1.3 | 4 | 37:47:11:5 | 489 at 1 A·g−1 1158 at 0.05 A·g−1 | 120 at 1 A·g−1 182 at 0.05 A·g−1 |
| NC-750 | 750 | 527 | 1.1 | 6 | 38:38:18:6 | 180 at 1 A·g−1 405 at 0.05 A·g−1 | 102 at 1 A·g−1 165 at 0.05 A·g−1 |
| NC-850 | 850 | 358 | 0.9 | 6 | 35:30:29:6 | 223 at 1 A·g−1 399 at 0.05 A·g−1 | 86 at 1 A·g−1 150 at 0.05 A·g−1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fedoseeva, Y.V.; Shlyakhova, E.V.; Vorfolomeeva, A.A.; Grebenkina, M.A.; Sysoev, V.I.; Stolyarova, S.G.; Maksimovskiy, E.A.; Makarova, A.A.; Okotrub, A.V.; Bulusheva, L.G. Tuning Nitrogen-Doped Carbon Electrodes via Synthesis Temperature Adjustment to Improve Sodium- and Lithium-Ion Storage. Batteries 2023, 9, 45. https://doi.org/10.3390/batteries9010045
Fedoseeva YV, Shlyakhova EV, Vorfolomeeva AA, Grebenkina MA, Sysoev VI, Stolyarova SG, Maksimovskiy EA, Makarova AA, Okotrub AV, Bulusheva LG. Tuning Nitrogen-Doped Carbon Electrodes via Synthesis Temperature Adjustment to Improve Sodium- and Lithium-Ion Storage. Batteries. 2023; 9(1):45. https://doi.org/10.3390/batteries9010045
Chicago/Turabian StyleFedoseeva, Yuliya V., Elena V. Shlyakhova, Anna A. Vorfolomeeva, Mariya A. Grebenkina, Vitalii I. Sysoev, Svetlana G. Stolyarova, Evgeny A. Maksimovskiy, Anna A. Makarova, Alexander V. Okotrub, and Lyubov G. Bulusheva. 2023. "Tuning Nitrogen-Doped Carbon Electrodes via Synthesis Temperature Adjustment to Improve Sodium- and Lithium-Ion Storage" Batteries 9, no. 1: 45. https://doi.org/10.3390/batteries9010045
APA StyleFedoseeva, Y. V., Shlyakhova, E. V., Vorfolomeeva, A. A., Grebenkina, M. A., Sysoev, V. I., Stolyarova, S. G., Maksimovskiy, E. A., Makarova, A. A., Okotrub, A. V., & Bulusheva, L. G. (2023). Tuning Nitrogen-Doped Carbon Electrodes via Synthesis Temperature Adjustment to Improve Sodium- and Lithium-Ion Storage. Batteries, 9(1), 45. https://doi.org/10.3390/batteries9010045







