1. Context
An electrical energy storage systems (EESS) may be an electrochemical cell association in a battery [
1]. These cells can belong to different technologies and chemistries. The oldest are lead–acid cells. Then, there are the cells using alkaline metals. Finally, for a quarter of a century, lithium cells have been marketed. Lithium-ion cells are more stable than the first lithium cells and have higher energy and power densities than the lead–acid and nickel-based technologies [
2]. They also have a higher lifespan, which partly explains their strong growth. The lithium-ion battery market continues to grow, to the point that their unit manufacturing price decreases steadily because of the growing number of units produced [
3]. Naturally, in a battery, all cells belong to the same technology.
This paper presents three architectures that can be used in cell batteries: two classics, using permanent series and parallel connections between cells, and one innovative, described later and allowing different connections between the cells. On these architectures, different variants are tested: common solutions (balancing between cells, using of more cells than necessary to provide the nominal power), other conventional but not deployed in best industrial solutions (failure tolerance of some cells with over-solicitation of others, addition of redundant cells to replace the first failing cells) and a new variant (redundant cells management with a control law using cells fundamental states as a choice criterion). The performances of the architectures and their variants are compared by simulation under Matlab, by resorting to a cell model that including aging phenomena.
The cell main electrical characteristic is its maximum storable capacity
Q0. It is well known that this capacity decrease with aging. When a cell is new, this capacity is optimal. It is noted in this paper as
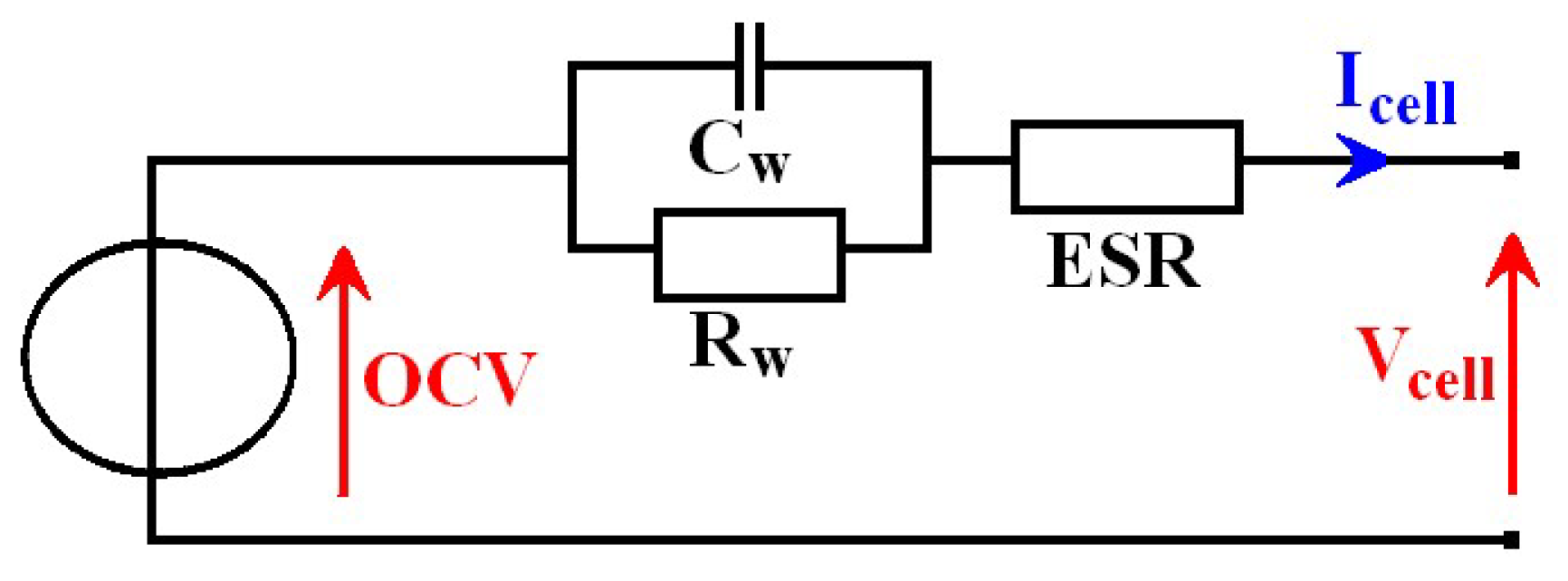
Q*. The battery physical behavior can be modeled by a second order Thevenin model scheme [
4] as seen in
Figure 1. In this, a cell is represented as a series association of an open-circuit voltage (
OCV), an equivalent series resistance (
ESR) and a parallel RC circuit. The
ESR corresponds to the battery terminals voltage
Vcell, only if it is measured without an external load connection and with the cell at rest for an adequate time. This time is necessary for balancing internal electrical charges (relaxation phenomenon). A double-layer capacitance (
Cw) and a charge transfer resistance (
Rw) complete the model by describing relaxation. Other internal phenomena such as a hysteresis voltage can also be modeled by additional subcircuits. Hysteresis consists in a shifting on the open-circuit voltage for the same contained charge between current direction, an upward offset with a negative current
Icell in charging phase and a downset offset with a positive current in the discharge phase.
To express physical state in which a cell is, two indicators are commonly used:
SoC and
SoH, respectively, the state of charge and the state of health [
5]. The
SoC describes, in percent, the amount of electrical charge
Q(t) it contains at t time, compared to its maximum storable capacity. The
SoC is determined by Equation (1). Moreover, when a cell ages, it cannot store as much electrical charge than when it is new. The maximum storable capacity
Q0 decreases over time continuously and gradually from its optimal capacity
Q*. So, a cell’s State of Health (
SoH) is defined by the ratio between these two capacity values, as shown by Equation (2). For applications requiring significant power more than a willingness to deliver energy,
SoH can also be defined relative to the
ESR [
6].
The ISO 12405-2 standard for electric vehicles [
7] specifies test procedures for lithium-ion batteries and electrically propelled road vehicles. It specifies that a cell enters the old age phase when its
SoH goes down to 80%. Manufacturers communicate a lifetime value for cells that incorporate, as specified in their datasheet, the two aging modes that a cell faces: an age-related calendar mode [
8] and a cyclic mode related to cell use [
9]. These two phenomena [
10] can be described by a combined evolution of a linear aging and an “
aging” power of time whose principles are described by Equation (3), where the aging parameter is between 0.5 and 2 and
A1 and
A2 are two constants.
Cyclic aging is aggravated mainly by three parameters: the operating temperature [
11], the amount of electrical charge extracted in a cycle [
12] and the current [
13]. Despite the cell manufacturing standardization, disparities can occur between cells from the same batch. These are reflected in an optimal capacity
Q* variability and in an equivalent series resistances
ESR variability [
14]. Thus, when cells are associated in an EESS, their disparities should lead to imbalances in currents, cell temperatures and then in their aging. Consequently, a battery may remain operational for a shorter time than a cell lifetime.
Operative dependability
Odep is defined as the time, expressed in hours or in number of cycles, during which a multi-cellular battery can meet the external load specifications [
15]. In other words,
Odep is the time before a downtime related to a full discharge or a too high age. That is to say, it contains enough operational cells to provide the requested power. For a single cell, this cessation comes from:
- −
a cell aging failure, resulting in a SoH of less than 0.8;
- −
a sudden random failure (open or short circuit);
- −
a complete discharge, SoC drops down to zero in operating phase.
For a battery,
Odep depends on the architecture (how cells are connected); if some cells are in redundancy for the current mission and if failed cells can be isolated or not. It is therefore necessary to monitor the cell states. In order to monitor cells, in particular to control end of charging and to prevent overheating, EESS cells are managed by a BMS (Battery Management System) [
16]. Today, multi-cellular EESS are constituted according to two conventional architectures (series-parallel and parallel-series) and sometimes include variants, as explained in the next section, to improve their availability.
From a formal point of view, the operative dependability
Odep is defined by the equation set (4), according to
SoC and
SoH cells.
In the next section, different architectures and possible variants to improve the battery operative dependability are presented. Then, part III presents simulations performed on each variant, whatever the cell technology, in order to determine the impact on operative dependability, for LiFePO4 cell example. The next part presents and compares the simulation results, especially for operative dependability.
2. Architectures and Variants
In the literature, to improve the operative dependability, various solutions for reconfiguration of the internal structure are proposed, such as the power tree solution presented in [
17] or the DESA architecture (Dependable, efficient, scalable architecture) [
18]. The first solution does not allow use of a battery at full power and the second only improves the operative dependability of a similar order to the amount of additional cell number (typically 50% with 50% of additional cells).
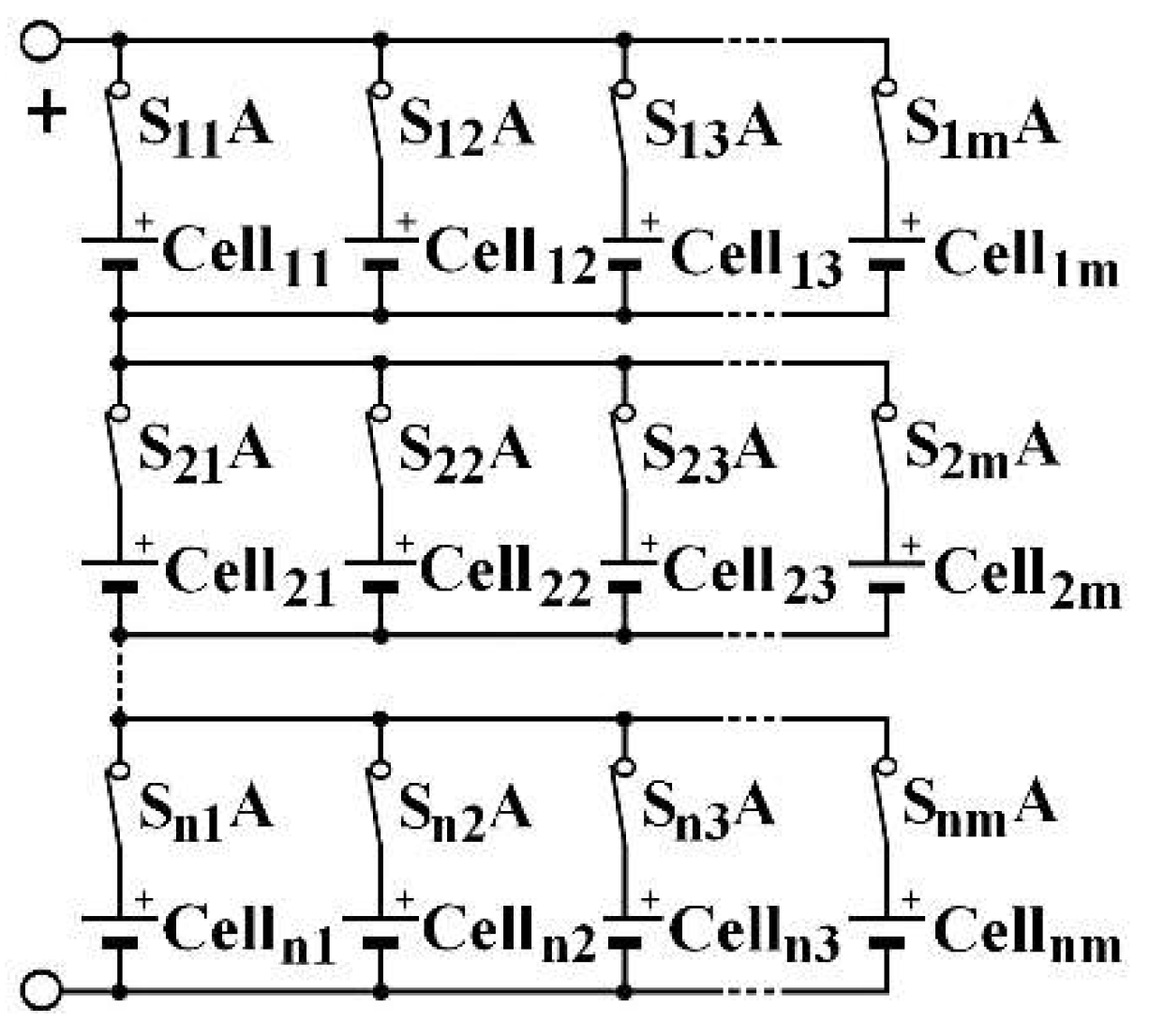
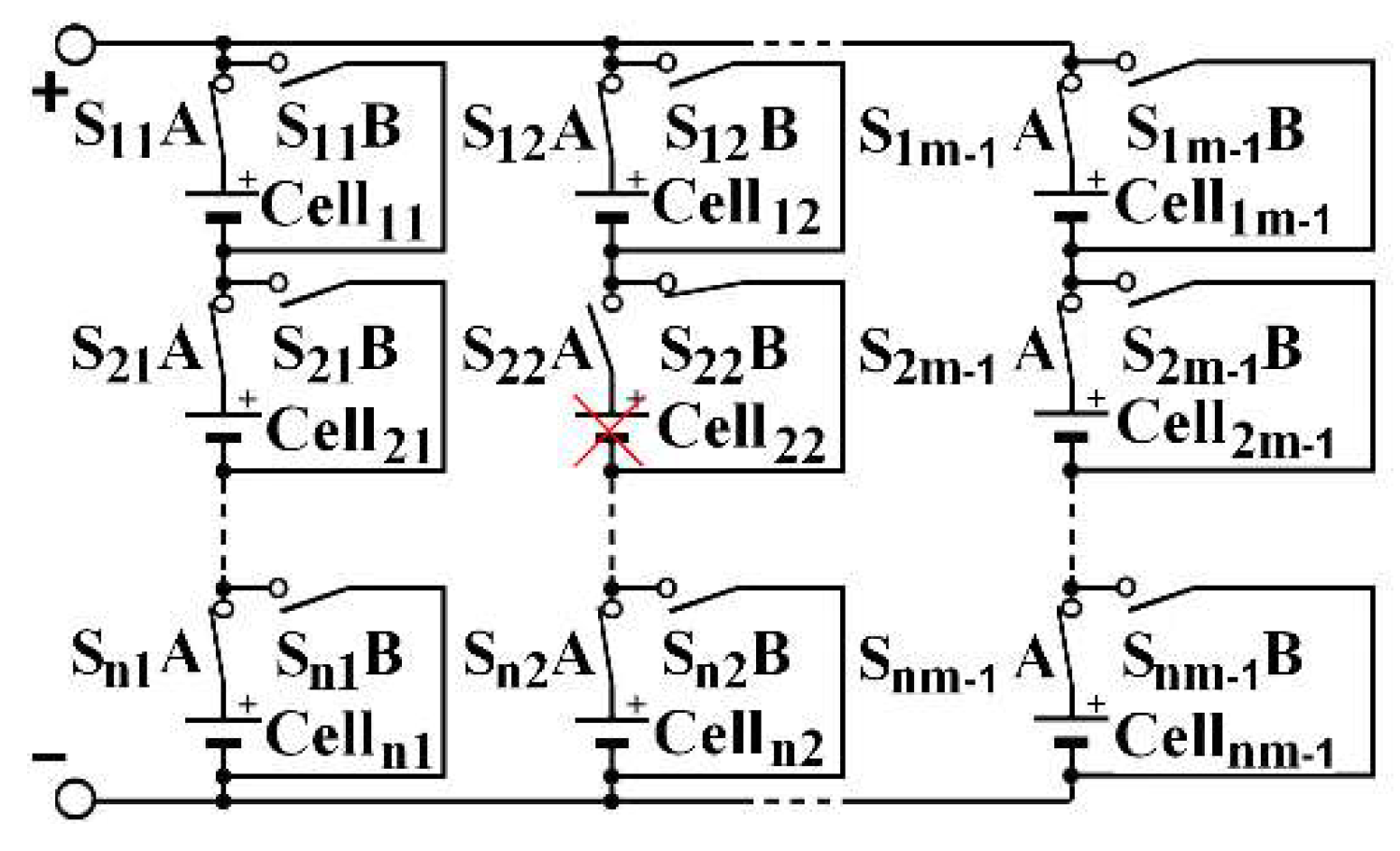
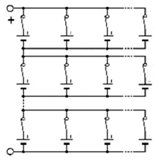
Three architectures are compared to determine which has the best operative dependability: two conventionally used and a new one. These architectures are reconfigurables by using a limited number of switches associated with a cell. To increase the battery voltage, the cells are associated in series. To increase the current, they are associated in parallel. Thus, batteries are generally associated in a SP (series-parallel) architecture, as described in
Figure 2, which presents a
n-rows
m-columns structure, noted (
n,
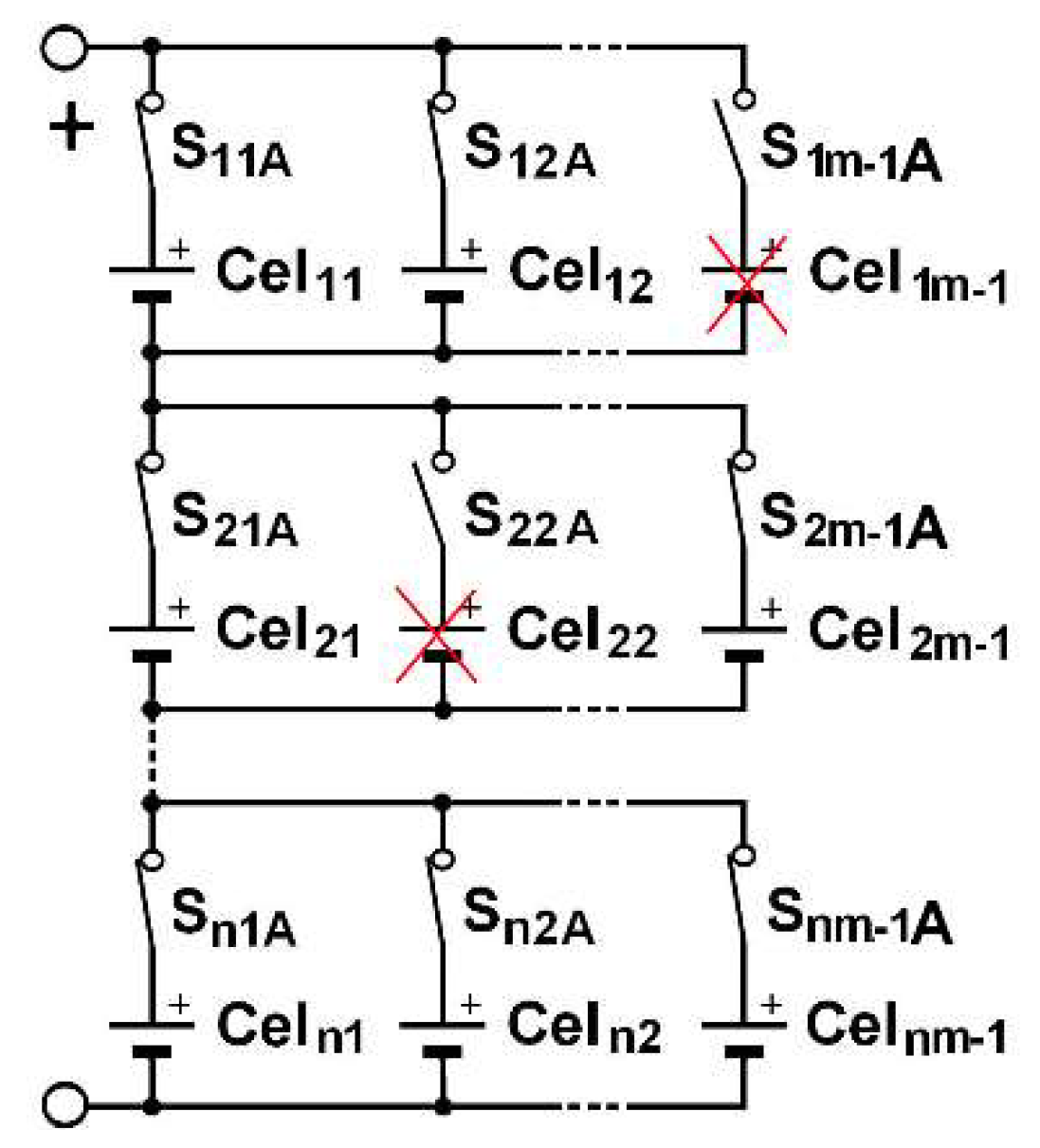
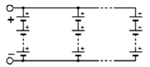
m). Switches allow to isolate a column, following one of its cell failure, for instance. By duality, the same structure can be inserted into a PS (parallel-series) architecture, as shown in
Figure 3. One switch for each cell is needed in order to isolate a failed cell. In an SP architecture, same-column cell voltage disparities can appear. In a PS architecture, the same-row cell currents can be different.
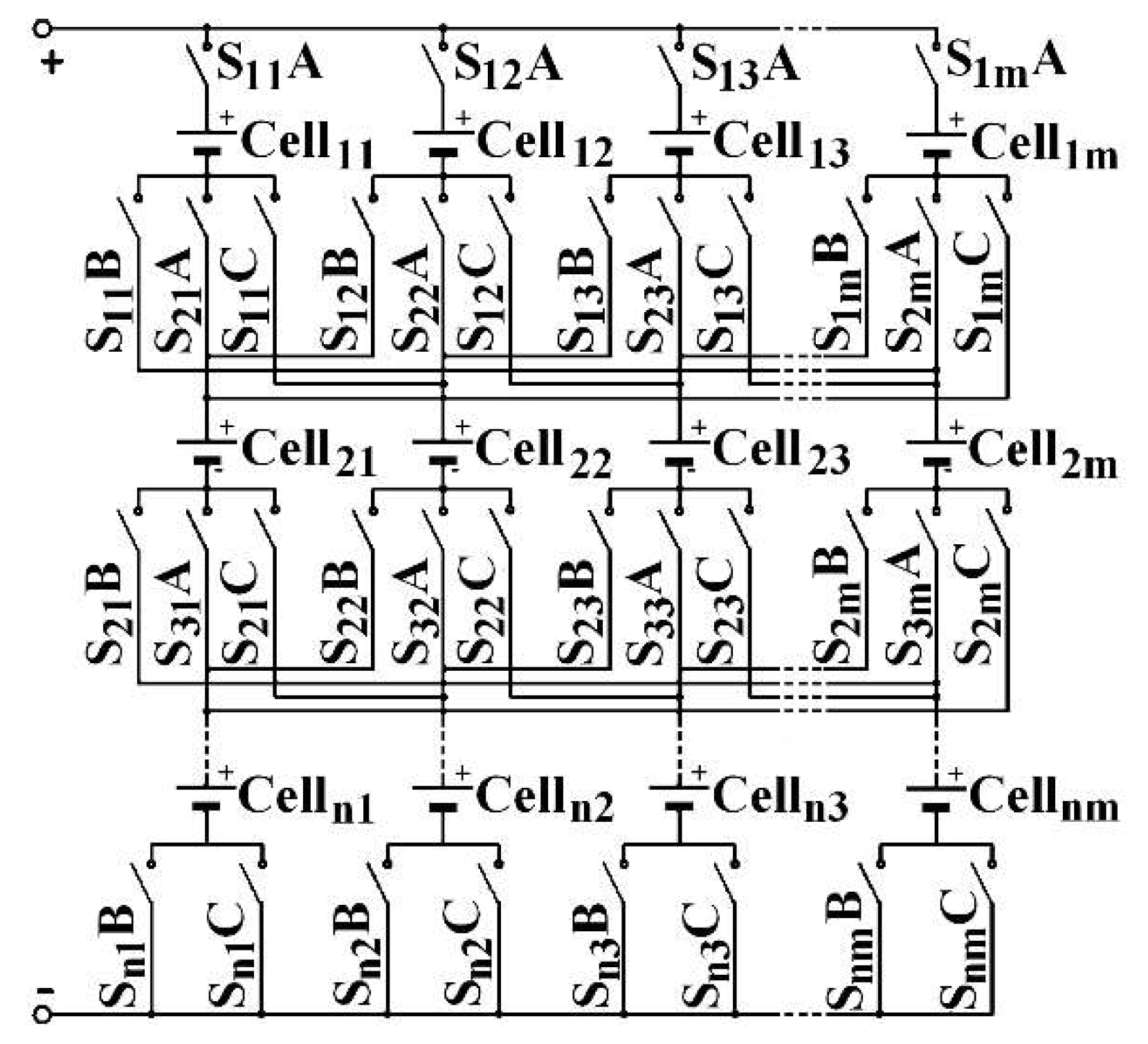
To deliver the same power, by keeping the same structure, another architecture is possible: the C3C [
19], depicted in
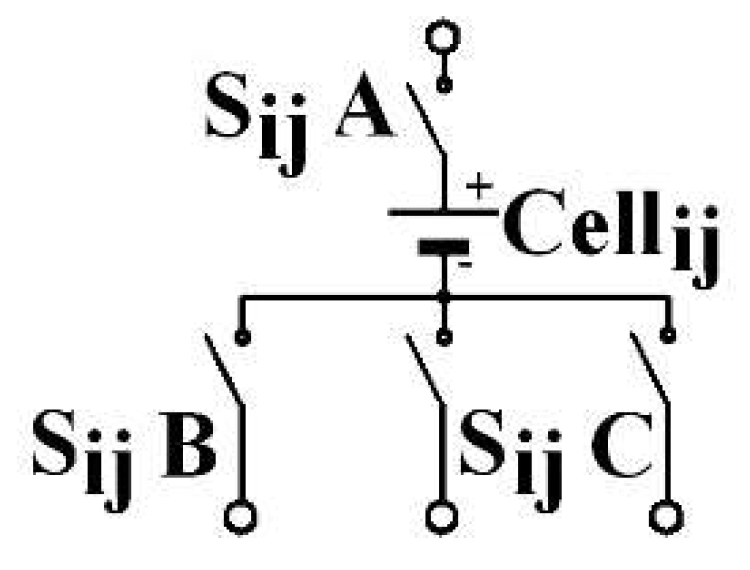
Figure 4. The architecture consists of an element association. Each C3C element comprises a cell and three switches, as shown by
Figure 5. Each includes an upstream connection and three downstream. The A indexed switch is placed on the upstream connection. Switch B is on the first downstream connection, C on the third. The middle connection is intended to be connected with the same-column downstream-row cell upstream connection. The C3C architecture combines the advantages of both conventional architectures, as PS’ self-balancing and allows a series association of some cells located on different columns. The current that leaves the Cell
i,j in the
ith row and the
jth column can be directed to one of the three following row cells: Cell
i+1,j−1, Cell
i+1,j, Cell
i+1,j+1, in a recharge phase, respectively by the switches S
i,jB, S
i+1,jA and S
i,jC or to one of the three upstream row cells: Cell
i−1,j−1, Cell
i−1,j, Cell
i−1,j+1, in a discharge phase. This is made by activating the appropriate switches, respectively S
i−1,j−1C, S
i,jA and S
i−1,j+1B. Thanks to the architecture, the
mth column cells are connected with those of the first column.
The C3C architecture involves two particularities. Firstly, to take advantage of its very large possible configuration number [
20], it must be managed by an algorithm that chooses the best cell combination, with a cell-aging-reducing aim. The cells selection parameters can be chosen to define the optimal combination. Only two are considered in this paper:
SoC and
SoH. Secondly, whatever the architecture, to increase battery reliability, a minimum amount of redundancy must be included [
21]. This minimal part consists of using a column (especially the
mth) as a redundant column. Thus, a (
n,
m) battery is calibrated to provide a nominal power
Pn given by Equation (5).
As a result, if the conventional architectures include a redundant column, they can also be managed by the same algorithm that chooses the best cell combination. The possible combination number is nevertheless much lower. Therefore, if the battery must supply its nominal current
Inom, depict in Equation (6), one cell in each row stays at rest. In PS and C3C architectures, this cell can be any of the
m cells of each row. In SP, all the same-column cells are placed at rest. To do this, it requires adding a switch in series with each cell in PS and one by column in SP, as shown in
Figure 2 and
Figure 3.
If SP and PS batteries include a redundant column, it becomes possible to use this redundancy classically. That is, the battery can work with its base cells, corresponding to the first (
m−1) columns, until one of them stops to be operational. It is thus replaced by the spare cell. In this variant, the
mth switches in the
Figure 3 are off in the beginning for a PS architecture. For an SP architecture, in
Figure 2, all cells in the
mth column are redundant.
Another variant can also be examined, tolerating a cell failure by replacing it with a short circuit in SP architecture and allowing the battery to continue to fulfill its mission, even if it requires the active cells to afford a greater current than their nominal current. To do so, two switches should be placed around each cell: one in series and one in parallel as S
i,jA is being open and S
i,jB closed when cell
i,j must be isolated, as depicted for cell
22, as shown by
Figure 6. In this example, cell
22 fails, the current of the second column passes through S
22B. The cell is marked with a red cross to symbolize its failure. This variant does not require redundancy cells. The same power as in Equation (5) is obtained with a (
n,
m−1) structure. In a PS architecture, the cell may just be disconnected by only one switch, as shown for cell
22 in
Figure 7, in which cell
1m−1 and cell
22 are faulty and marked with a red cross.
It is also possible to use a battery comprising m columns to provide a power corresponding to the nominal power of Equation (5). In this way, the battery has an over-capacity compared to the external load specifications.
Moreover, in order to reduce the disparities between the
SoCs when cells are associated in series, in an SP architecture, balancing circuits are often used. They enable to homogenize the electrical charges in all cells in the string [
22]. These balancing circuits are controlled by a BMS [
23]. They allow better stored energy use [
24] and a battery operative dependability improvement [
25,
26]. To evaluate balancing impact, basic SP of
Figure 2 (with (
m−1) columns) with and without balancing circuits are simulated. All compared batteries must provide the same Equation (5) power. So, this structure only includes (
m−1) columns. Several balancing techniques exist in an SP architecture:
- −
dissipative balancing [
27], consisting of balancing the electrical charges from below by removing excess energy by Joule effect;
- −
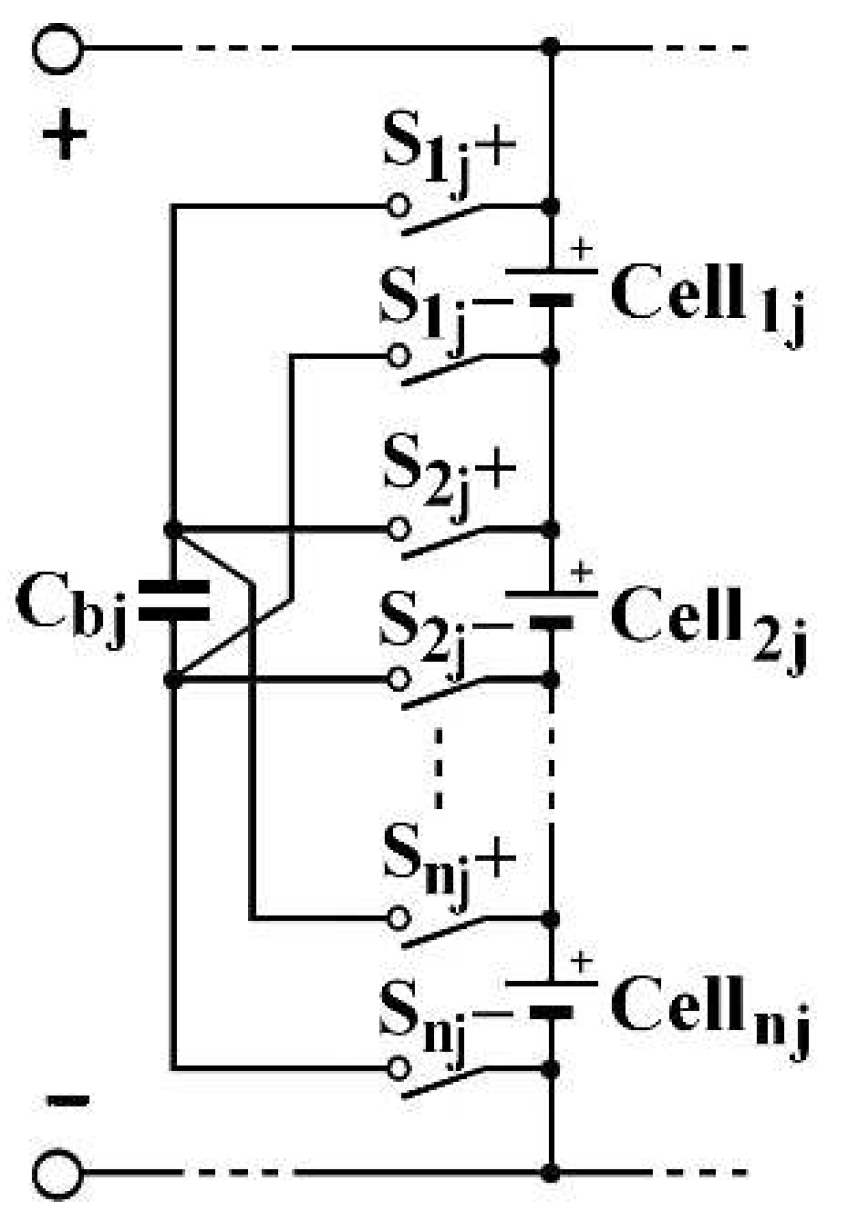
redistributive balancing to send excess energy from the most charging cell(s) to the least charging cell(s) in the same column [
28]. Its principle is described by the
Figure 8 scheme, relating to a single column j in an SP architecture. When Cell
a,j is more charged than Cell
b,j, the intermediate capacitor Cb
j, associate to this column j, is placed in parallel by switching on the S
a,j+ and S
a,j− switches. Then, these switches are switched off and the Sb
,j+ and Sb
,j− switches are switched on. At the end, Cell
aj electric charge have been reduced and Cell
aj one have been increased.
In this study, only redistributive balancing circuits from one cell to another are considered. This leads to add two switches by cell in SP schemes, each connecting a terminal of the cell to a terminal of an intermediate capacitor, used to temporarily store the energy to be transferred. By this way, the different variants combining architecture and improvement are listed in
Table 1. The structure column number m
c and the cell-associated switch number are also specified. For the basic PS and the over-capacity PS variants, the switches in
Figure 3 are not useful because cells are not managed individually. The cells are all active or all inactive. In the same way, those of
Figure 2 are not useful in the without-balancing SP variant. Apart from the variant reported without balancing, all other SPs include balancing circuits, which add two switches per cell. The variants deployed in present industrial solutions are PS-base, SP-base with or without balancing and over-capacity variants.
3. Simulations
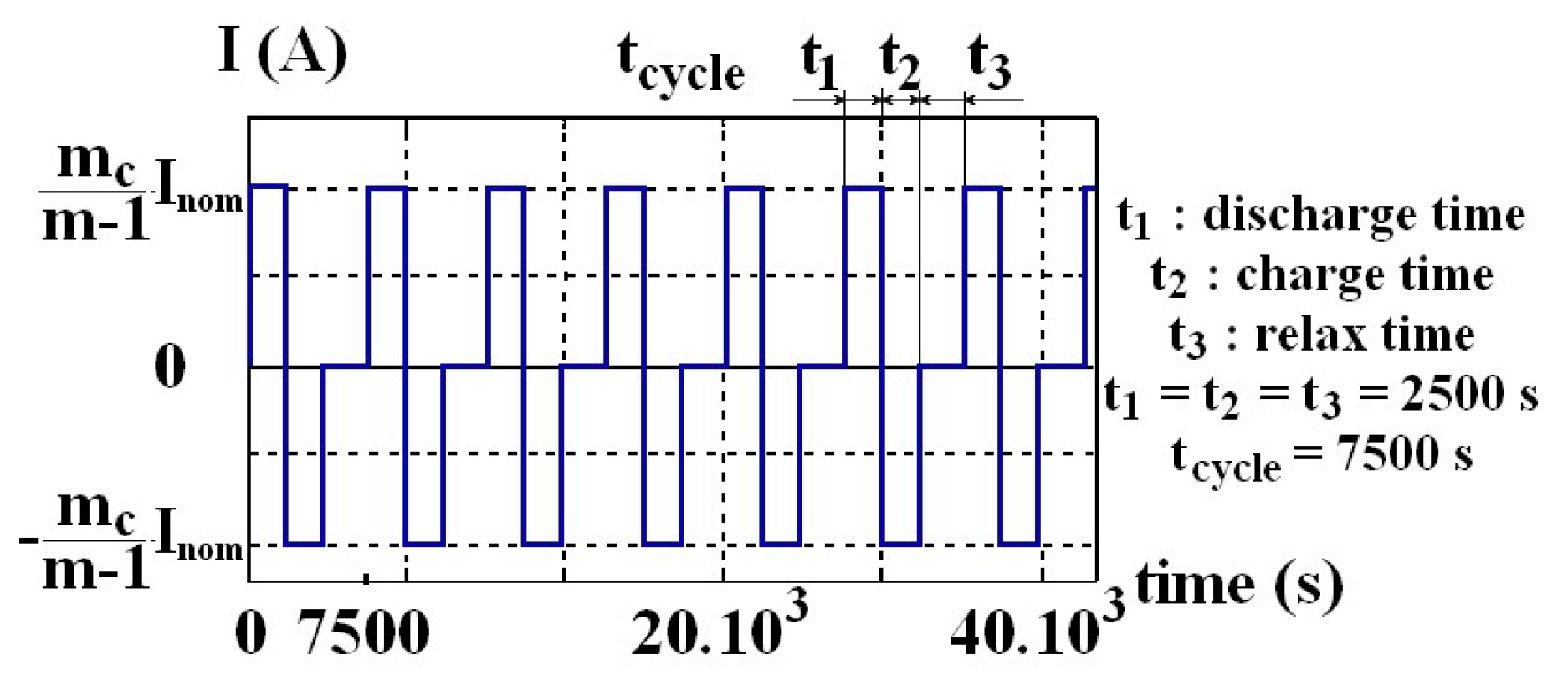
The different variants and architectures were modeled using Matlab. The program simulates cell associations according to each architecture for different (
n,
m) structures. For each variant, it submits the battery to regular cycling, leading the current shown in
Figure 9 for a single 10 Ah cell. The battery is initially full. This cycle consists of firstly, a discharge under a current I equal to the battery nominal current
Inom divided by the active column number m
c (that is to say (
m−1) or
m) for a duration of 2500 s. So, except for over-capacity variants,
I = 10 A in discharging phase. By this way, the battery is discharged by 70% of its initial capacity. This discharge value is relevant for quantifying cell aging. Indeed, it allows to stop the discharging phase before a complete discharge. The more
Qo(t) decreases, the more the
SoC at the end of discharging phase decreases. This is because a cell has to provide the same amount of energy. Then, the cells are recharged, for the same duration, to return to full charge. Finally, the battery is placed at rest for an identical duration, allowing the return to internal balance.
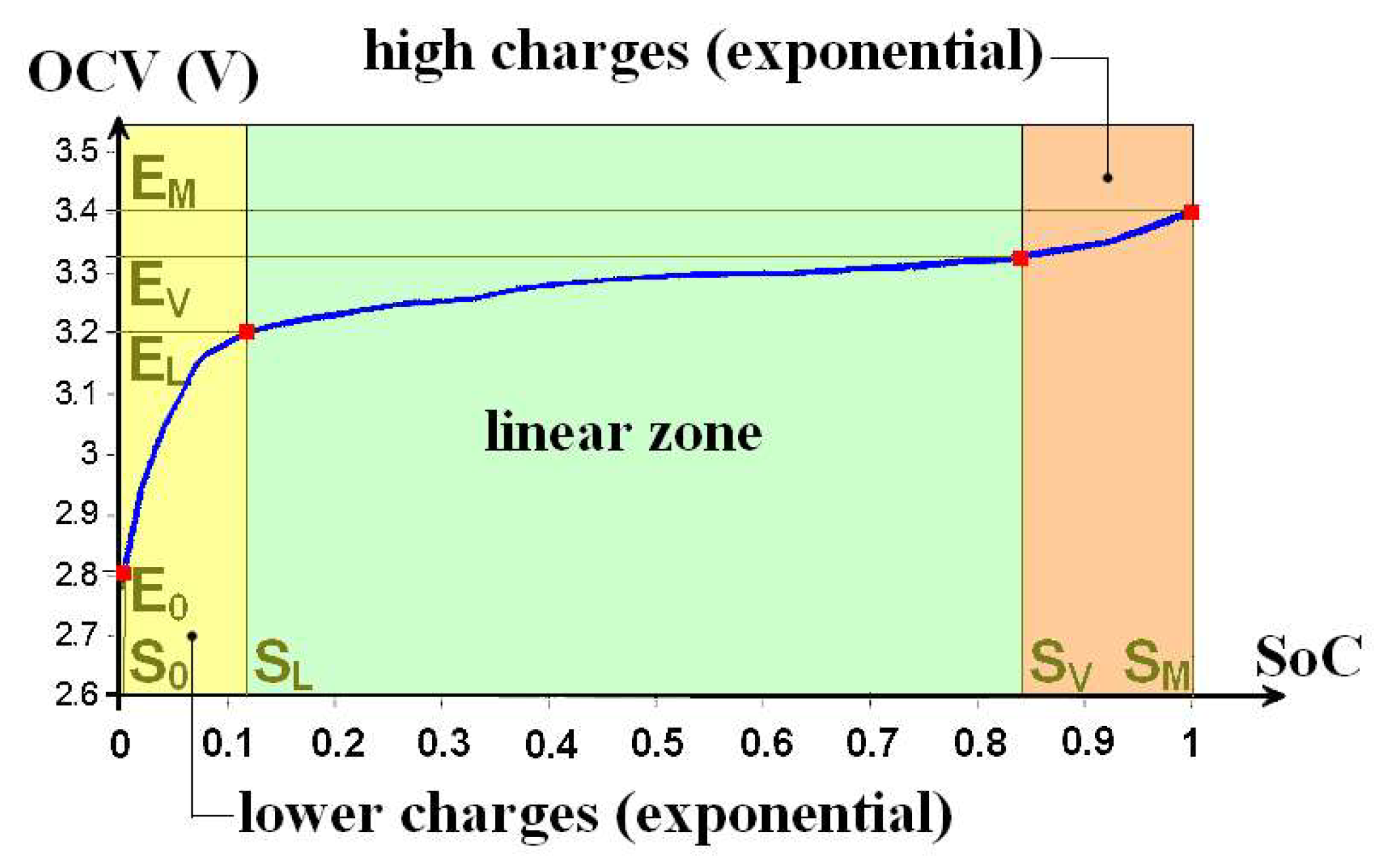
To model a cell, a characteristic equation that describes the
OCV evolution as a function of the
SoC is used. The typical shape of this curve is described for an amorphous iron phosphate (LiFePO
4) cell in
Figure 10. Batteries, with an amorphous iron phosphate positive electrode support high intensities in charge and discharge as well as fast charges. These cells have higher power density and lower fire risks than cells with a lithium cobalt oxide (LiCoO
2) positive electrode [
29]. On this curve, four points are identified by a red dot. These four coordinate points (
S0,
E0), (
SL,
EL), (
SV,
EV) and (
SM,
EM), respectively, delimit three sectors. The curve being continuous, it is so possible to perform partial regression. In the sector where the state of charge is low (
SoC <
SL) and
OCV low (
OCV <
EL), the curve can be described by an exponential function. It is the same for high states of charge (
SoC >
SV and
OCV >
EV). The central sector can be described by a linear function. Ultimately, the characteristic equation can be described by equation set (7) as a function of the
SoC (explain as a dummy variable
x).
ν and
ξ parameters are empirical.
ν is between 10 and 20. It corresponds to the rapid growth of the voltage when the
SoC approaches its maximum.
ξ parameter is its complement when the voltage collapses, the
SoC decreasing towards its minimum.
In these simulations, the cells had an initial capacity Q* = 10 Ah and an average ESR of 20 mΩ. The ambient temperature was set to 25 °C. Since the Q0(t) degradation is continuous and progressive, it is logical to consider that the SoH, which is a representation of this degradation, also decreases continuously. This degradation is proportional to the using conditions. At each cycle, a cell ages. This translates into an SoH decrease.
The cell temperature evolves as a function of the current flowing through it. By convection, radiation and conduction, this heat can spread to other cells. For simplicity, the used model does not integrate thermal coupling phenomena.
Finally, the initial conditions for each cell was given randomly around nominal a value with a variability of more or less 10%. The same Celli,j is used for all variants in all architectures, so as to ensure a true comparison of the variant intrinsic performance. A cell is considered faulty in two cases. First, when it is completely discharged whereas it should still supply energy (SoC = 0). Second, when it is too old and has SoH = 0.8. This corresponds, for a single cell, to the operative dependability definition given above. Depending on the architecture, the variant and the failed cell location(s), the battery may or may not continue to provide the requested power. If, at moment, it is no longer able to provide this power, the Odep worth.
Simulation was performed several times with different initial conditions. Only the mean values are reported here, even if the figures illustrating this demonstration are related to a single simulation, among all those performed.
From these different simulations, it is possible to extract operative dependability. Odep corresponds to the time when the battery stops fitting to the specifications: delivery of the battery nominal current Inom. According to the variant, some cells may have failed (by aging, random failure or complete discharge) before this time and be isolated or replaced by others. The simulations presented here were carried out for several structures: with n = 2 and m varying from 3 to 10 on the one hand, and on the other hand with m = 3 and n varying from 2 to 4.
4. Results and Comparisons
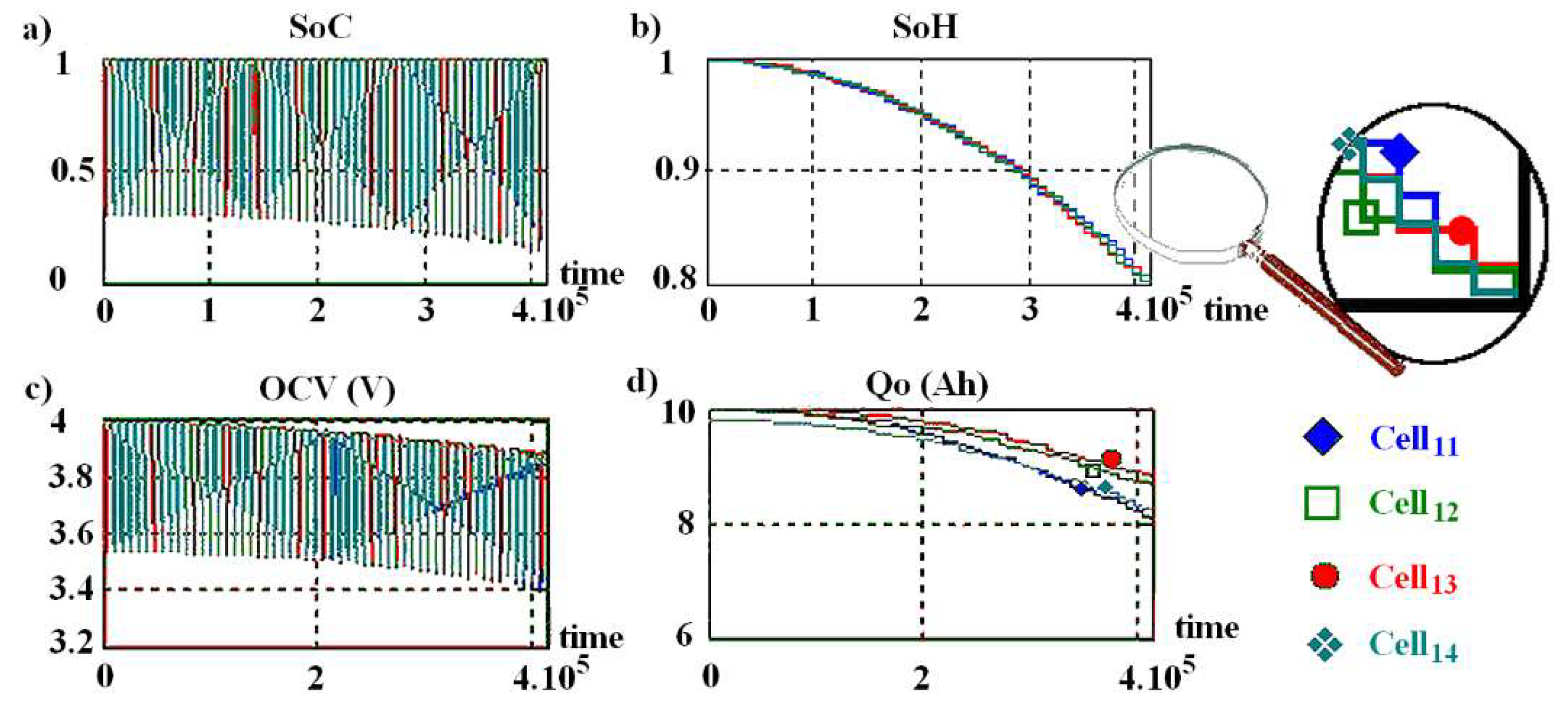
Results from a (3,4) structure simulation performed with a C3C architecture with
SoH-based optimization algorithm is illustrated in
Figure 11, which presents
SoC,
SoH,
OCV and
Q0 evolution. In this example, only the first-row cells are shown because it is in this row that the first two cells fail. Cell
14 (cyan curve) fails first.
Odep is limited by Cell
12 aging (green curve). This simulation was performed willingly in accelerated mode, with an announced cell lifetime of only 50 cycles. In order to read
SoC evolution on the curve, the aging parameter has been set to its maximum value (
aging = 2) to better differentiate the
SoH curves when the battery lifespan approaches. By using this variant,
aging control is visible on the curve b). All cells age together, their
SoH decreases together.
Table 2 records the average operative dependability obtained for the different structures. This average is given for cells with lifetimes of 1000 cycles.
In the same way that the reliability of a several identical all-necessary element system decreases if the element number increases, the more a battery contains cells, the lower the operative dependability is. For instance, for a basic PS architecture, with a
n = 2 structure,
Odep reduces when the column number j increases: respectively, to 772, 754, 750, 748, 747 and 674 cycles when j varies from 4 to 10. The lowest operative dependability variant is the PS architecture without redundant cell (basic PS). For instance,
Odep = 772 cycles for a (2,4) structure. Its performance serves as a reference value. Operative dependability for the variants are relative compared (base 100 for basic PS) in the bar graph in
Figure 12, drawn for a (3,4) structure. Thus, the performances can be grouped into three clusters: those that are between 100% and 120% of the basic PS operative dependability, those between 120% and 140%, and those higher [
30]. Among the less powerful variants are the classic solutions: balancing in SP architecture, redundancy, fault tolerance and over-capacity. In the second family are the
SoC-based optimization algorithm variants for the three architectures. Finally, those with the best operative dependability are those that use variant with
SoH optimization algorithm, regardless of the architecture. Cluster 3 variants show a different
Odep improvement by architecture, as summarized in
Table 3. On average, this improvement is close to 36%. Unlike the DESA architecture, whose management cannot be deployed in another architecture, with the optimization algorithm, the operative dependability improvement is greater than the share of redundant cells. For example, for a (2,4) structure,
Odep is improved by 50% for only 33% of redundant cells. In the same way, for a (2,10),
Odep is improved by almost 25% regardless of the architecture for 10% more cells.