Toxic Gas Emissions from Damaged Lithium Ion Batteries—Analysis and Safety Enhancement Solution
Abstract
:1. Introduction
2. Experimental
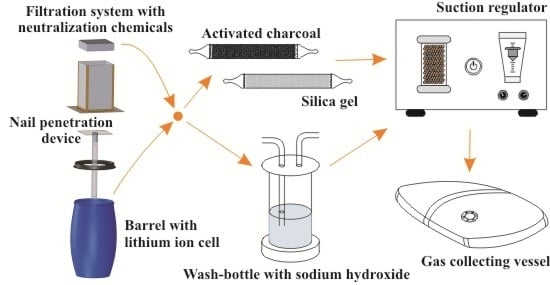
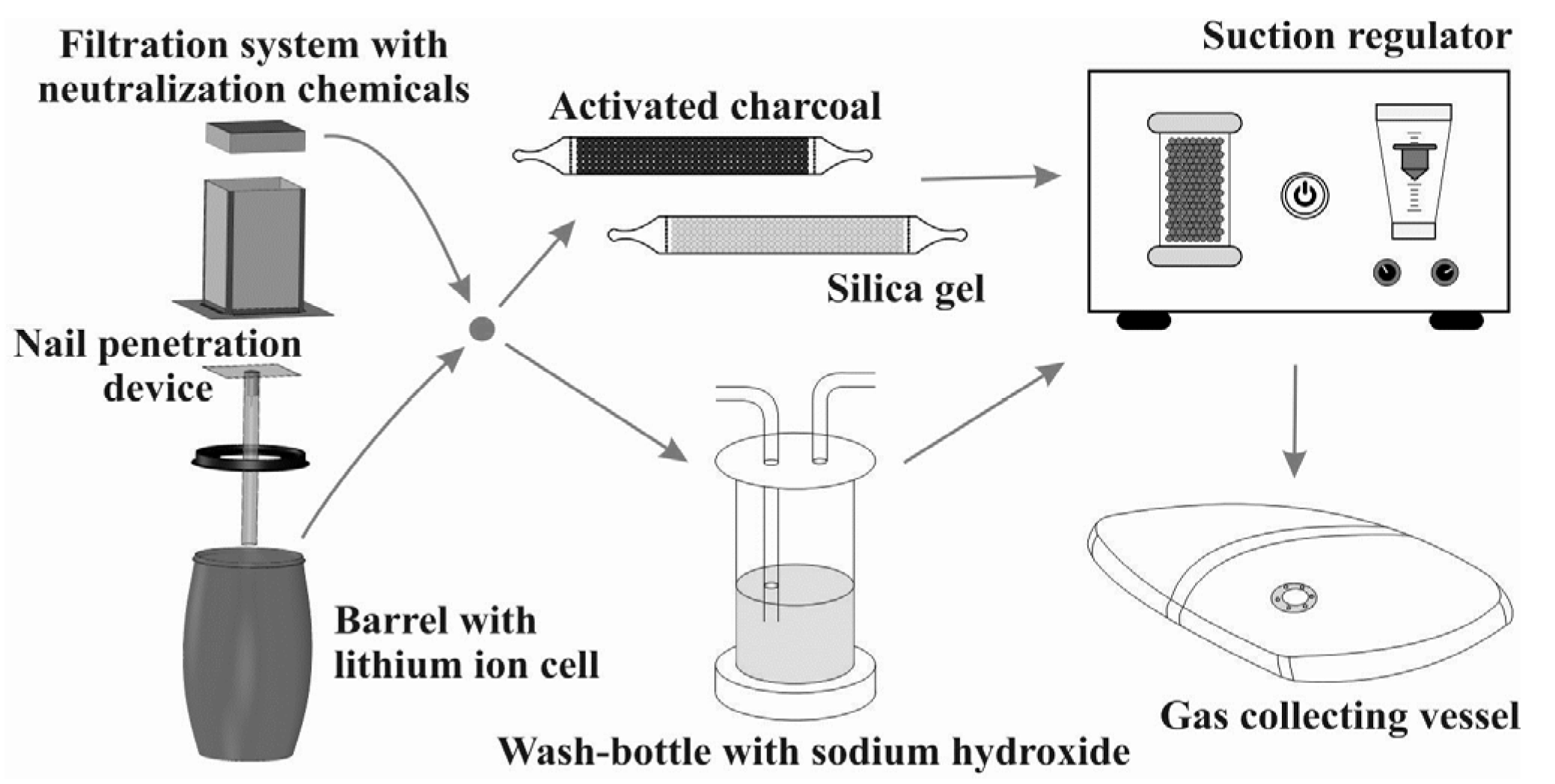
2.1. Measurement Setup
- (1)
- Catching fire: a single bare lithium ion cell is mounted within the barrel. The barrel is shut partly by a cover, which prevents the barrel from explosion but retains most of the emission gases. PTFE tubing leads the gas mixture to the analytics.
- (2)
- Fire prevention: conducted as Experiment 1, the cell under test is, additionally, wrapped within a specially-developed textile composite structure which is gas permeable but prevents flying sparks (FlotreX S500, Knein Technische Textilien GmbH, Herzogenrath-Merkstein, Germany). This cover material is made out of three layers: a fiberglass mesh, a finely-pored pure glass fleece, and a knitted fabric made of stainless steel-reinforced para-aramid fibers. Video “Experimental Setup Scenario 2” in Supplementary Materials presenting this particular experimental setup is available in the online version.
- (3)
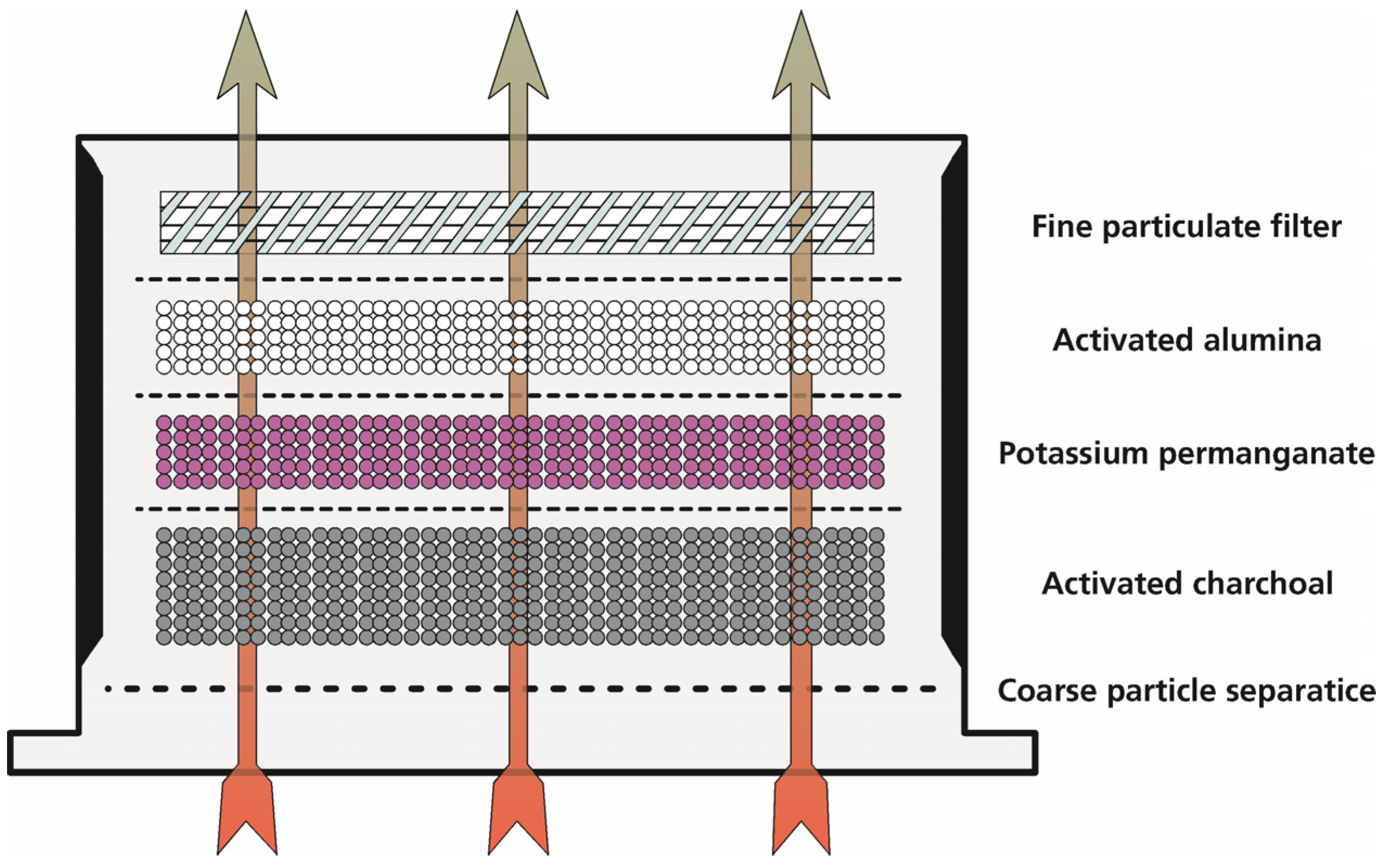
- Gas filtration: the experiment, as described under 2 is repeated using a tightly sealed barrel with a gas filtration unit mounted on top. The unit consists of five different stages: a particle filtration grid, three pellet layers of 18 dm3 activated charcoal, 9 dm3 potassium permanganate and 9 dm3 activated alumina, as well as a fine particle filter (CCP 610/210/510, SF 14, Viledon Freudenberg, Weinheim, Germany) (Figure 2). A gas bag is used to collect all gas emission behind the filtration; the PTFE tubing is inserted into this bag.
2.2. Analytical Methods
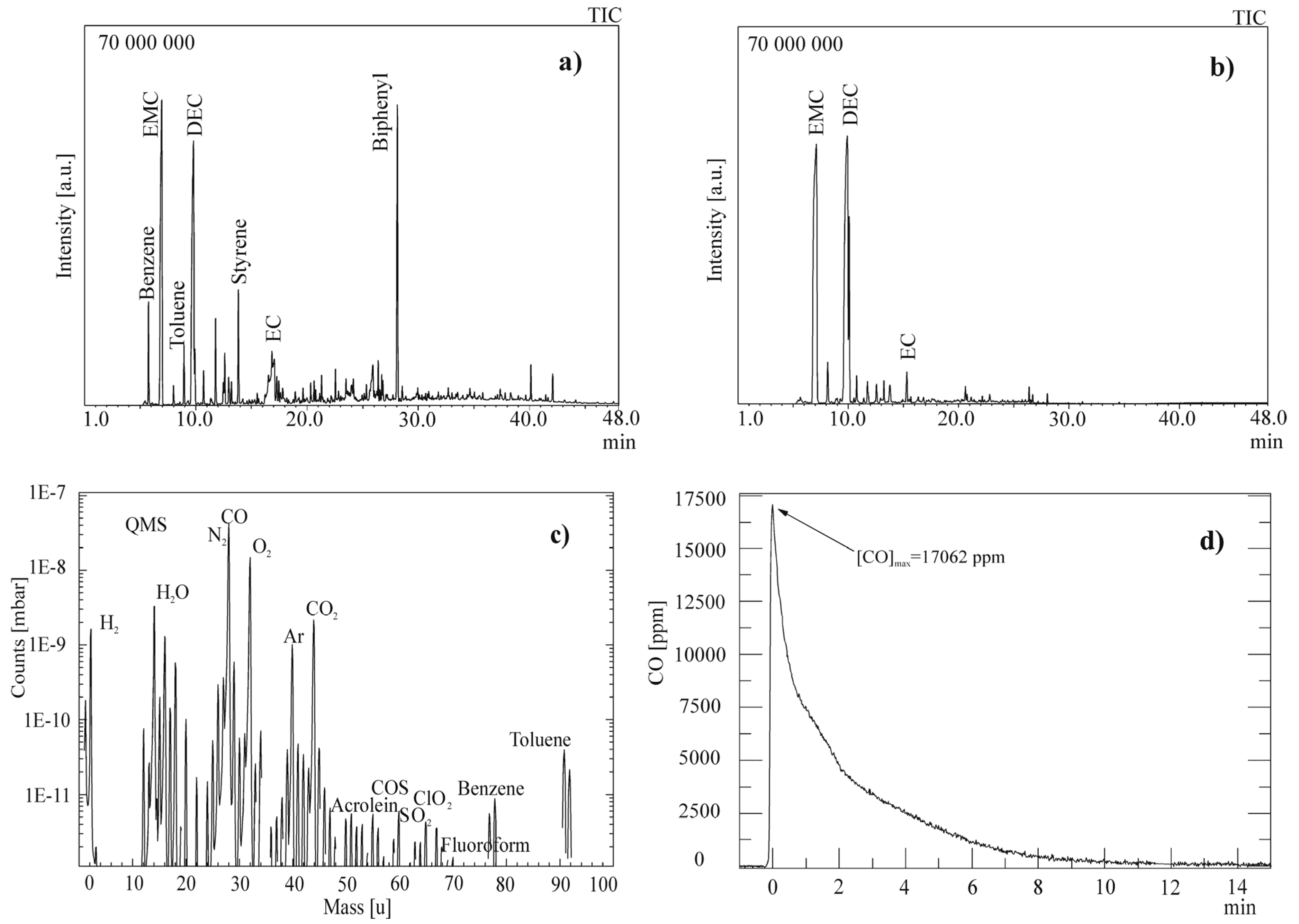
2.2.1. Gas Chromatography-Mass Spectrometry
2.2.2. Quadrupole Mass Spectrometry
2.2.3. HF Determination via Ion Chromatography
2.2.4. Quartz-Enhanced Photoacoustic Spectroscopy
3. Results and Discussion
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Leadbetter, J.; Swan, L.G. Selection of battery technology to support grid-integrated renewable electricity. J. Power Sources 2012, 216, 376–386. [Google Scholar] [CrossRef]
- Beaudin, M.; Zareipour, H.; Schellenberglabe, A.; Rosehart, W. Energy storage for mitigating the variability of renewable electricity sources: An updated review. Energy Sustain. Dev. 2010, 14, 302–314. [Google Scholar] [CrossRef]
- Rand, D.A.J.; Moseley, P.T. Energy Storage with Lead–Acid Batteries. In Electrochemical Energy Storage for Renewable Sources and Grid Balancing; Moseley, P.T., Garche, J., Eds.; Elsevier: Amsterdam, The Netherlands, 2015; Chapter 13; pp. 201–222. [Google Scholar]
- Kurzweil, P. Lithium Battery Energy Storage: State of the Art Including Lithium–Air and Lithium–Sulfur Systems. In Electrochemical Energy Storage for Renewable Sources and Grid Balancing; Moseley, P.T., Garche, J., Eds.; Elsevier: Amsterdam, The Netherlands, 2015; Chapter 16; pp. 269–307. [Google Scholar]
- Ren, G.; Ma, G.; Cong, N. Review of electrical energy storage system for vehicular applications. Renew. Sustain. Energy Rev. 2015, 41, 225–236. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, B.; Li, Q.; Cartmell, S.; Ferrara, S.; Deng, Z.D.; Xiao, J. Lithium and lithium ion batteries for applications in microelectronic devices: A review. J. Power Sources 2015, 286, 330–345. [Google Scholar] [CrossRef]
- Lisbona, D.; Snee, T. A review of hazards associated with primary lithium and lithium-ion batteries. Process Saf. Environ. Prot. 2011, 89, 434–442. [Google Scholar] [CrossRef]
- Wang, Q.; Ping, P.; Zhao, X.; Chu, G.; Sun, J.; Chen, C. Thermal runaway caused fire and explosion of lithium ion battery. J. Power Sources 2012, 208, 210–224. [Google Scholar] [CrossRef]
- Doughty, D.H.; Crafts, C.C. FreedomCAR Electrical Energy Storage System Abuse Test Manual for Electric and Hybrid Electric Vehicle Applications; Sandia Report SAND2005-3123; Sandia National Laboratories: Albuquerque, NM; Livermore, CA, USA, 2006.
- Abraham, D.P.; Roth, E.P.; Kostecki, R.; McCarthy, K.; MacLaren, S.; Doughty, D.H. Diagnostic examination of thermally abused high-power lithium-ion cells. J. Power Sources 2006, 161, 648–657. [Google Scholar] [CrossRef]
- Larsson, F.; Andersson, P.; Blomqvist, P.; Lorén, A.; Mellander, B.-E. Characteristics of lithium-ion batteries during fire tests. J. Power Sources 2014, 271, 414–420. [Google Scholar] [CrossRef]
- Ohsaki, T.; Kishi, T.; Kuboki, T.; Takami, N.; Shimura, N.; Sato, Y.; Sekino, M.; Satoh, A. Overcharge reaction of lithium-ion batteries. J. Power Sources 2005, 146, 97–100. [Google Scholar] [CrossRef]
- Stein, S.E. “Mass Spectra” in NIST Chemistry WebBook: NIST Standard Reference Database Number 69. Available online: http://webbook.nist.gov/ (accessed on 29 September 2015).
- Kosterev, A.A.; Bakhirkin, Y.A.; Curl, R.F.; Tittel, F.K. Quartz-enhanced photoacoustic spectroscopy. Opt. Lett. 2002, 27, 1902–1904. [Google Scholar] [CrossRef] [PubMed]
- Sigma-Aldrich. SDS Search and Product Safety Center. Available online: http://www.sigmaaldrich.com/safety-center.html (accessed on 31 August 2015).
- K.TeX—Knein Technische Textilien GmbH. Insulation Materials for Lithium Ion Batteries. Available online: http://www.ktex-gmbh.de/images/Anwendungen/Product-Portfolio-Insulation-Materials-for-Lithium-Ion-Batteries.pdf (accessed on 29 September 2015).
- Ballschmiter, K.; Bacher, R. Dioxine: Chemie, Analytik, Vorkommen, Umweltverhalten und Toxikologie der Halogenierten Dibenzo-p-Dioxine und Dibenzofurane; VCH: Weinheim, Germany; New York, NY, USA; Basel, Switzerland; Cambridge, UK; Tokyo, Japan, 1996. (In German) [Google Scholar]
- Curl, R.F.; Capasso, F.; Gmachl, C.; Kosterev, A.A.; McManus, B.; Lewicki, R.; Pusharsky, M.; Wysocki, G.; Tittel, F.K. Quantum cascade lasers in chemical physics. Chem. Phys. Lett. 2010, 487, 1–18. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Hydrogen Fluoride (as F). Available online: http://www.cdc.gov/niosh/idlh/7664393.html (accessed on 26 February 2016).
- Ingenieurbüro Oetzel. Aufnahmevermögen von Aktivkohle Gegenüber Chemischen Einzelverbindungen. Available online: http://www.umweltanalytik.com/daten/aktivkohle.htm (accessed on 26 February 2016). (In German)
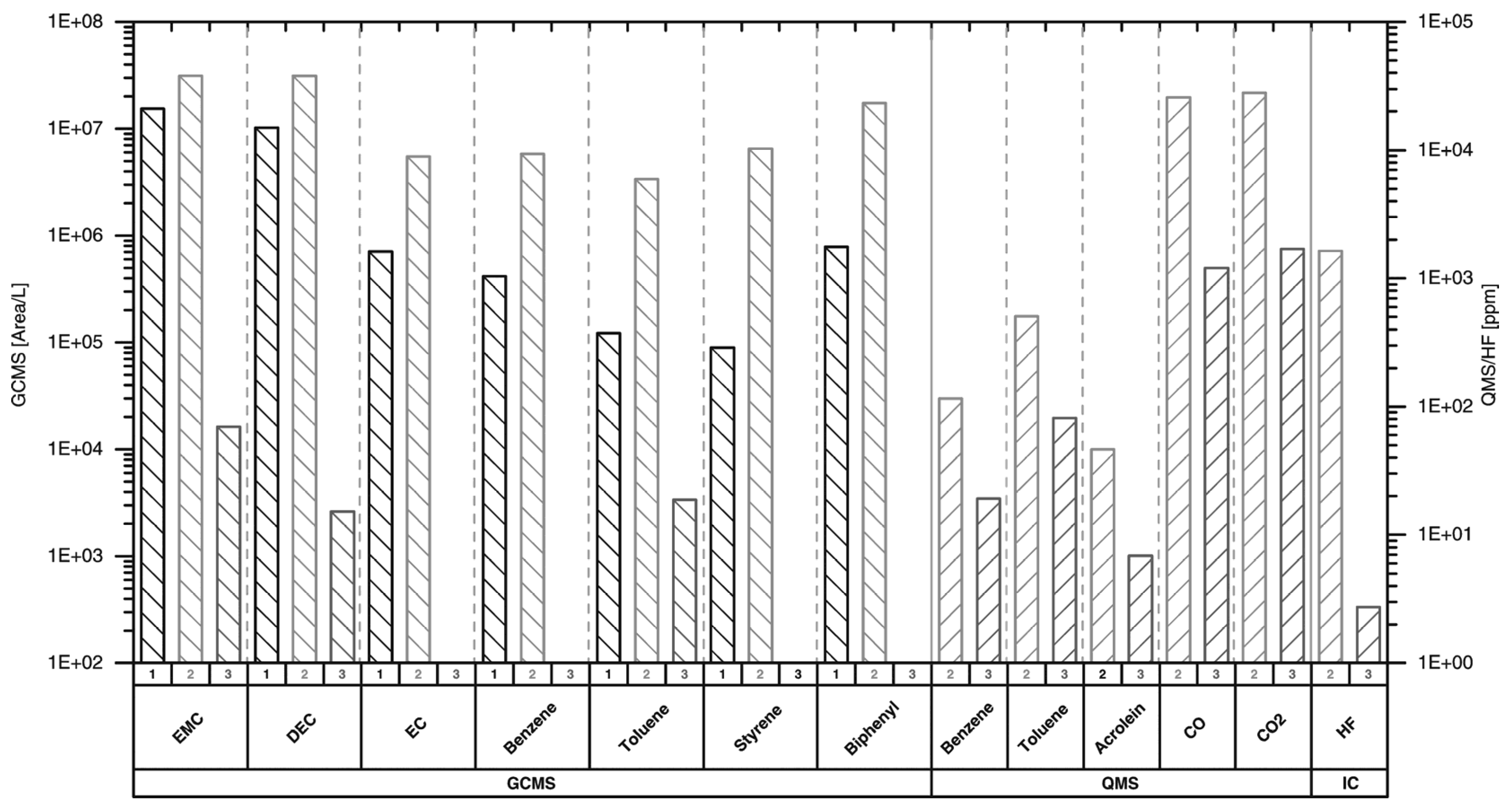
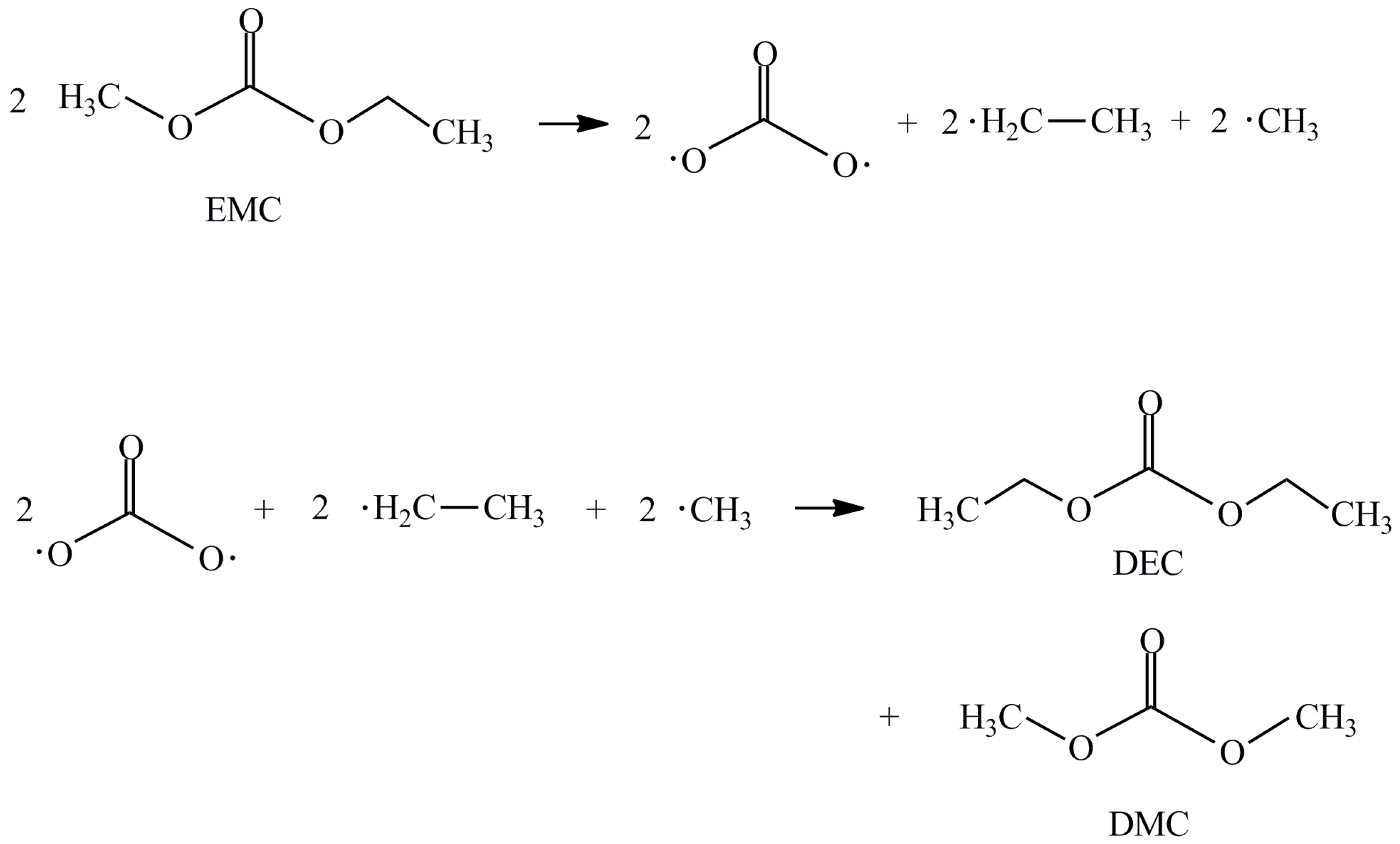
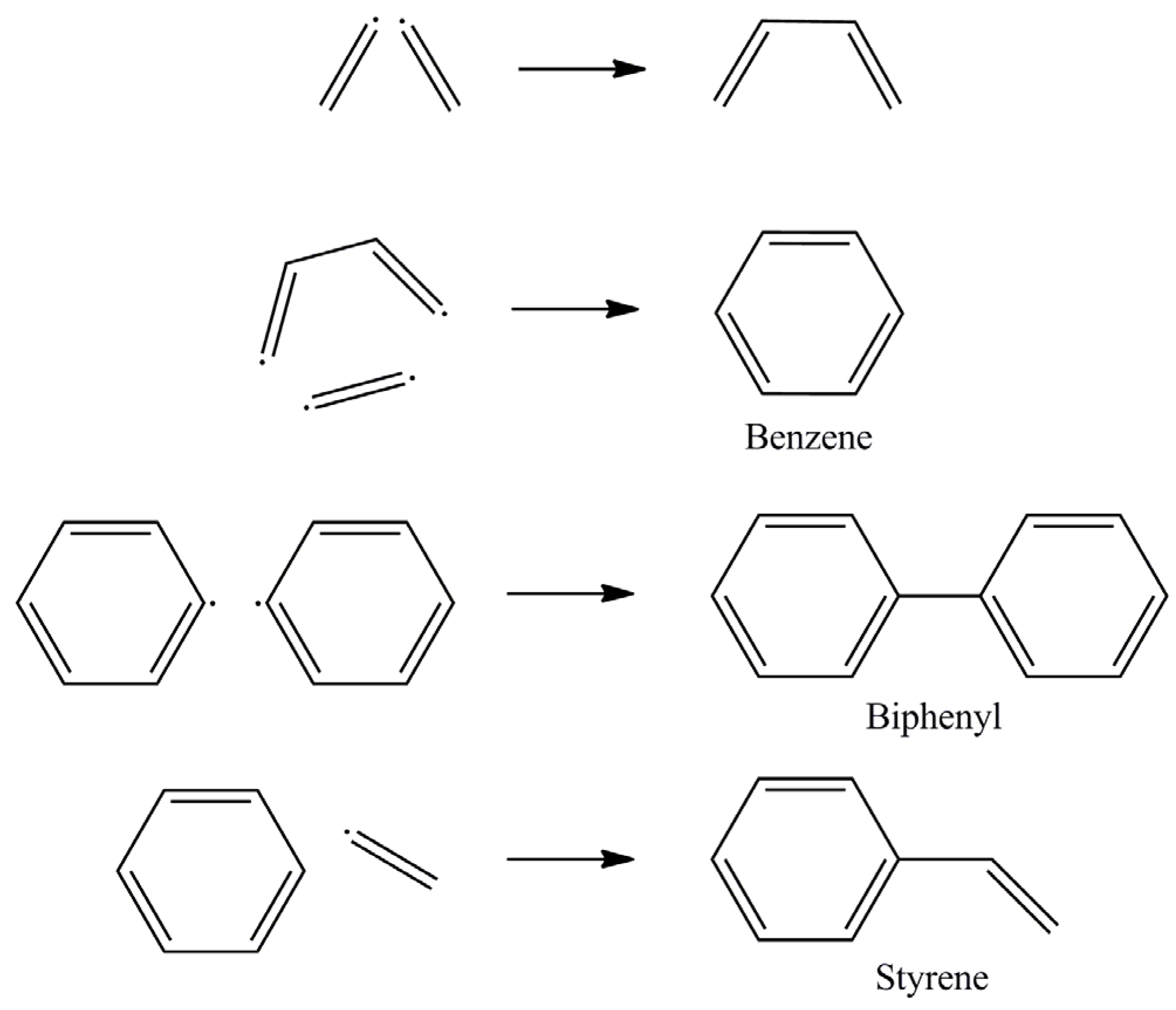
| Substance | Hazards according to EU Regulation (EG) Act 1272/2008 |
|---|---|
| EMC | Eye irritation; flammable liquid; H226; H315; H319; H335; Skin irritation, specific target organ toxicity-single exposure. |
| DEC | Eye irritation; flammable liquid; H226; H315; H319; H335; skin irritation; specific target organ toxicity-single exposure. |
| EC | Eye irritation; H315; H319; H335; skin irritation; specific target organ toxicity-single exposure. |
| Benzene | Aspiration hazard; carcinogenicity; eye irritation; H225; H304; H315; H319; H340; H350; H372; germ cell mutagenicity. |
| Toluene | Aspiration hazard; flammable liquid; H225; H304; H315; H336; H361d; H373; reproductive toxicity; skin irritation; specific target organ toxicity-repeated exposure. |
| Styrene | Acute toxicity; eye irritation; flammable liquid.; H226; H315; H319; H332; H361d; H372; Skin irritation; Specific target organ toxicity-repeated exposure. |
| Biphenyl | Aquatic acute toxicity; aquatic chronic toxicity; eye irritation; H315; H319; H335; H400; H410. |
| Acrolein | Acute toxicity; aquatic acute toxicity; aquatic chronic toxicity; carcinogenicity; corrosive to the respiratory tract; eye damage; flammable liquid; H225; H300; H300 + H330; H302;H311;H314;H317;H318;H330;H341; H351; H400; H410; germ cell mutagenicity; skin corrosion; skin sensitization. |
| CO | Acute toxicity; flammable gases; H220; H280; H331; H360DM H372M gases under pressure; reproductive toxicity; specific target organ toxicity-repeated exposure. |
| COS | Acute toxicity; eye irritation; flammable gases; H220; H280; H315; H319; H331; H335; Gases under pressure. |
| Hydrogen fluoride | Acute toxicity; corrosive to the respiratory tract; H300; H310; H314; H330; skin corrosion. |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nedjalkov, A.; Meyer, J.; Köhring, M.; Doering, A.; Angelmahr, M.; Dahle, S.; Sander, A.; Fischer, A.; Schade, W. Toxic Gas Emissions from Damaged Lithium Ion Batteries—Analysis and Safety Enhancement Solution. Batteries 2016, 2, 5. https://doi.org/10.3390/batteries2010005
Nedjalkov A, Meyer J, Köhring M, Doering A, Angelmahr M, Dahle S, Sander A, Fischer A, Schade W. Toxic Gas Emissions from Damaged Lithium Ion Batteries—Analysis and Safety Enhancement Solution. Batteries. 2016; 2(1):5. https://doi.org/10.3390/batteries2010005
Chicago/Turabian StyleNedjalkov, Antonio, Jan Meyer, Michael Köhring, Alexander Doering, Martin Angelmahr, Sebastian Dahle, Andreas Sander, Axel Fischer, and Wolfgang Schade. 2016. "Toxic Gas Emissions from Damaged Lithium Ion Batteries—Analysis and Safety Enhancement Solution" Batteries 2, no. 1: 5. https://doi.org/10.3390/batteries2010005
APA StyleNedjalkov, A., Meyer, J., Köhring, M., Doering, A., Angelmahr, M., Dahle, S., Sander, A., Fischer, A., & Schade, W. (2016). Toxic Gas Emissions from Damaged Lithium Ion Batteries—Analysis and Safety Enhancement Solution. Batteries, 2(1), 5. https://doi.org/10.3390/batteries2010005