Removal of Metal Ions via Adsorption Using Carbon Magnetic Nanocomposites: Optimization through Response Surface Methodology, Kinetic and Thermodynamic Studies
Abstract
1. Introduction
2. Experimental
2.1. Materials and Method
2.2. Characterization of NC
2.3. Adsorption and Desorption Experiments
2.4. Factorial Design
2.5. Statistical Analysis
3. Results and Discussions
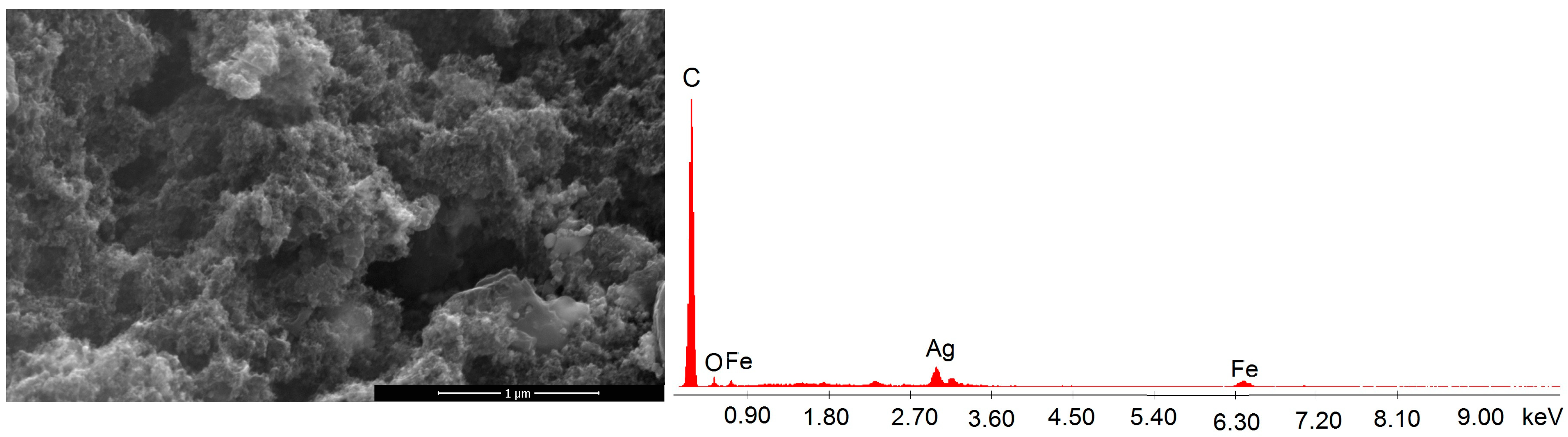
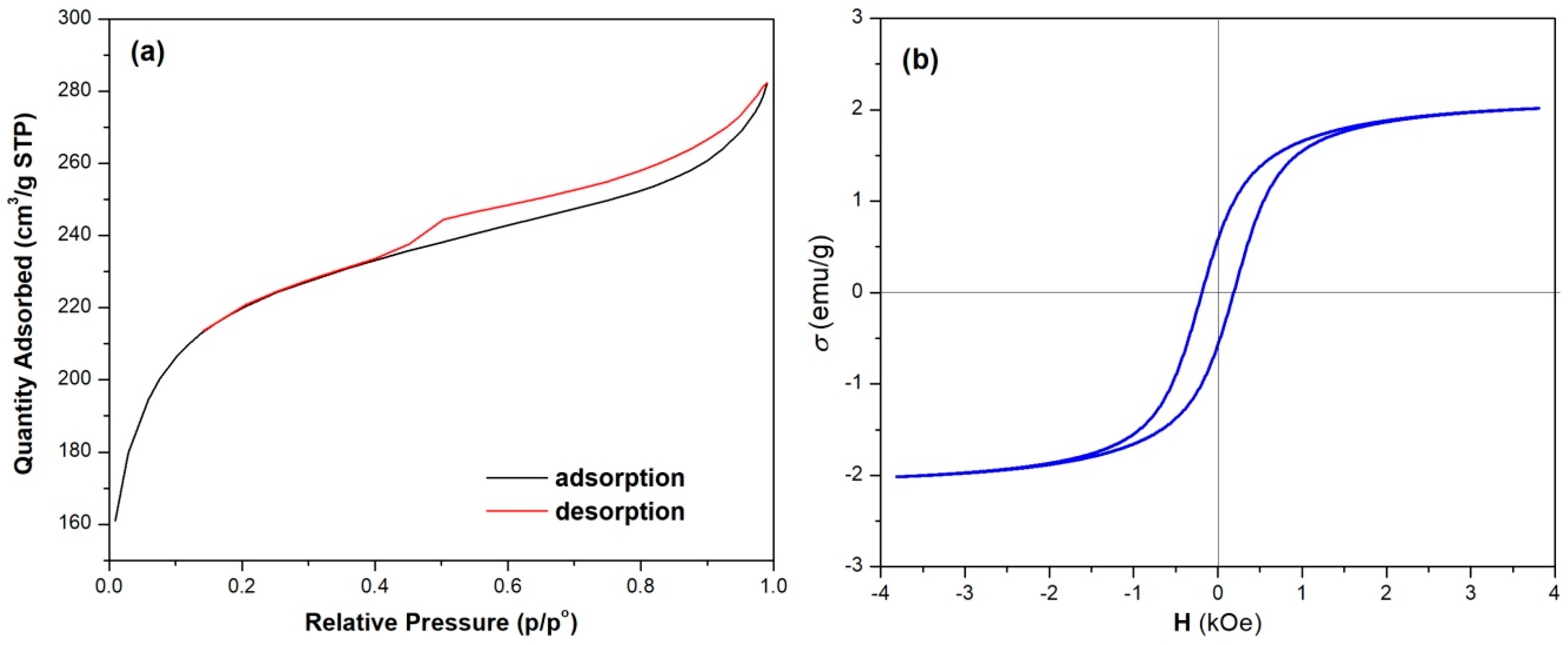
3.1. Characterization of NC Adsorbent
3.2. Factorial Design
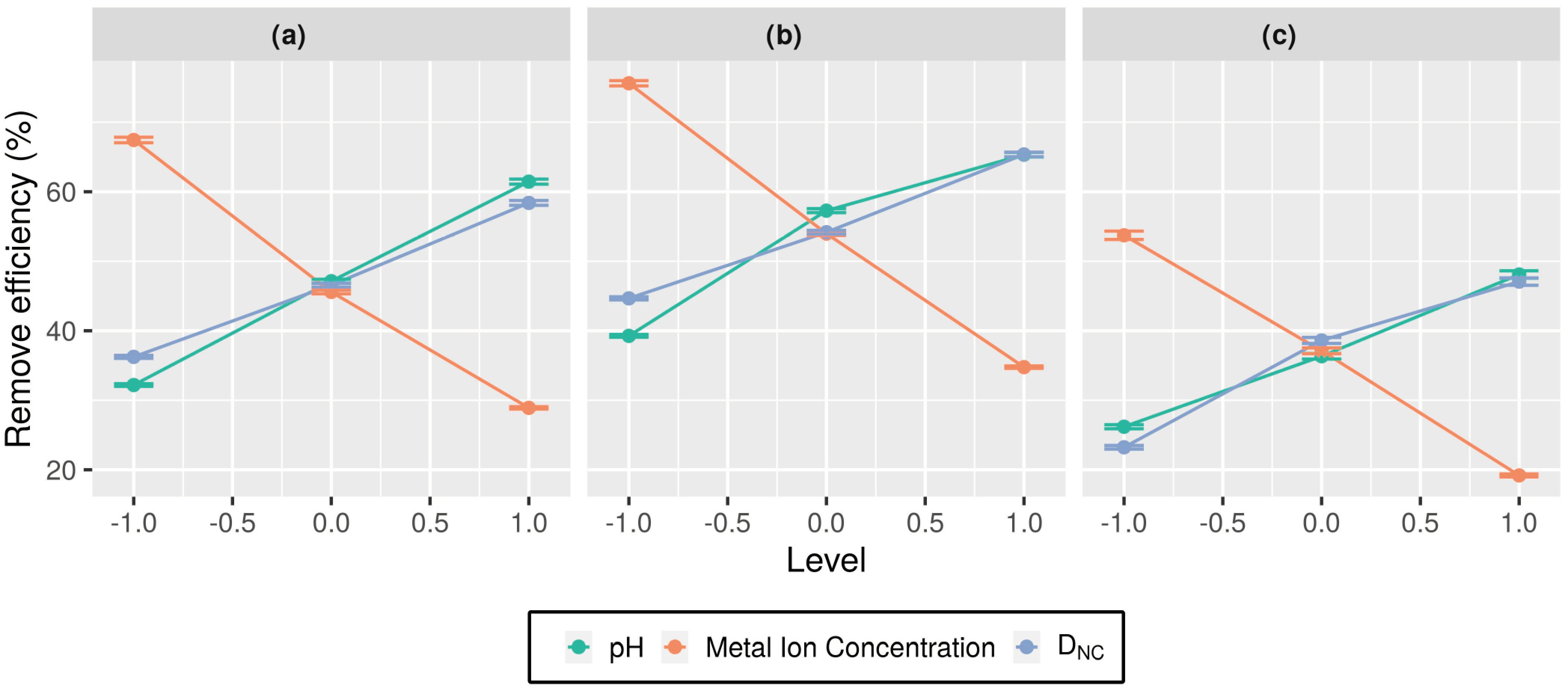
3.2.1. The Influence of the Main Effect
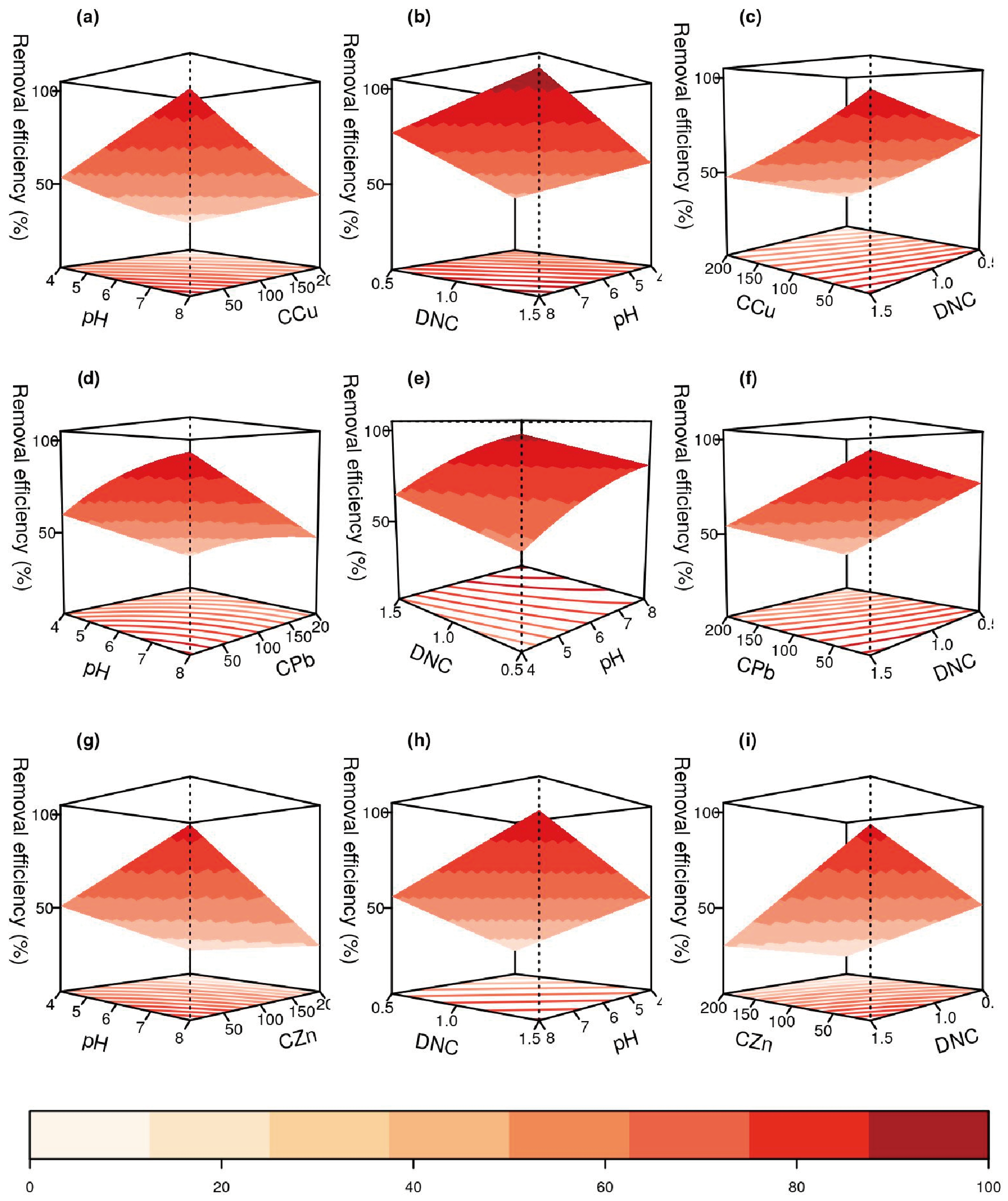
3.2.2. Model Generation
3.2.3. Optimization of Metal Ions’ Removal
3.3. Adsorption Experiments
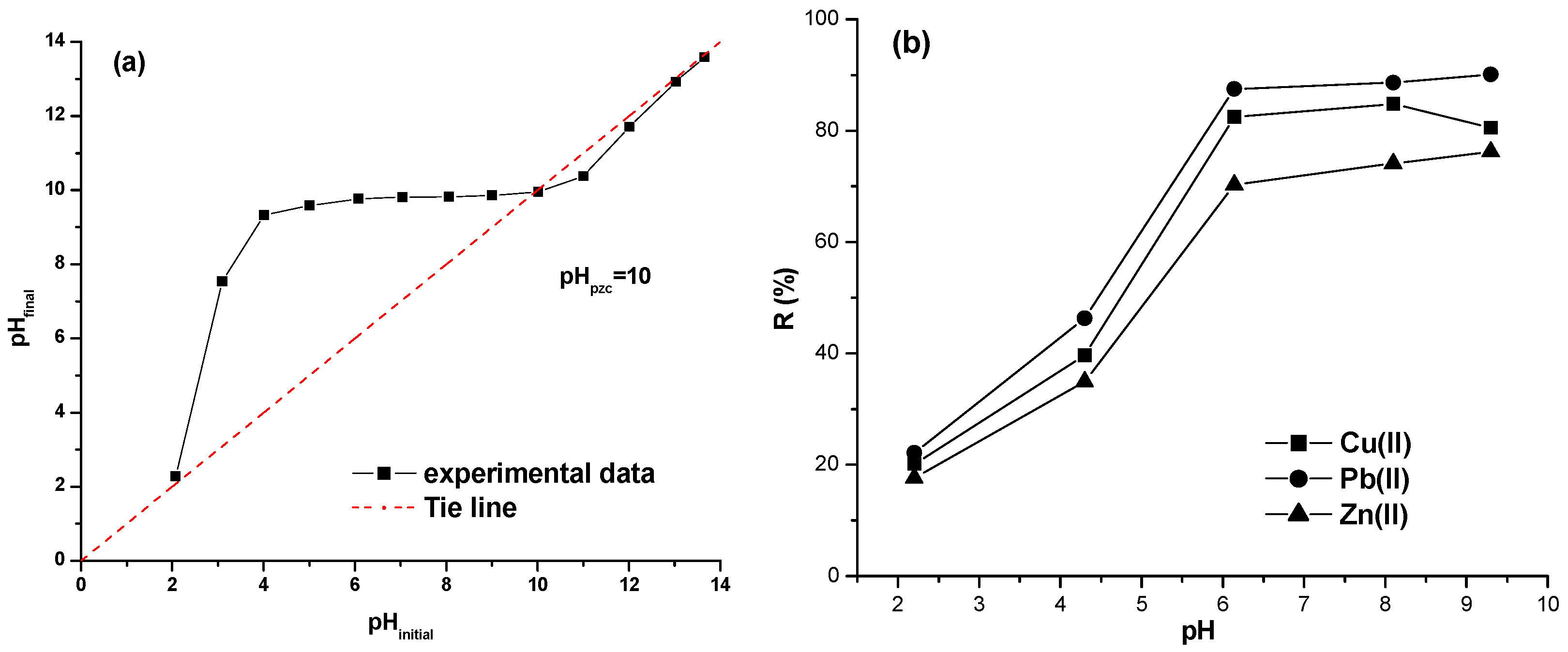
3.3.1. Influence of Solution pH
3.3.2. Kinetics Studies on Adsorption Process
3.3.3. Equilibrium Studies on Adsorption Process
3.3.4. Reusability Study
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yaseen, D.A.; Scholz, M. Textile dye wastewater characteristics and constituents of synthetic effluents: A critical review. Int. J. Environ. Sci. Technol. 2019, 16, 1193–1226. [Google Scholar] [CrossRef]
- Pattnaik, P.; Dangayach, G.S.; Bhardwaj, A.K. A review on the sustainability of textile industries wastewater with and without treatment, methodologies. Rev. Environ. Health 2018, 33, 163–203. [Google Scholar] [CrossRef]
- Liu, Y.-M.; Liu, D.-Y.; Zhang, W.; Chen, X.-X.; Zhao, Q.-Y.; Chen, X.-P.; Zou, C.-Q. Health risk assessment of heavy metals (Zn, Cu, Cd, Pb, As and Cr) in wheat grain receiving repeated Zn fertilizers. Environ. Pollut. 2020, 257, 113581. [Google Scholar] [CrossRef]
- Singh, R.; Bhateria, R. Optimization and experimental design of the Pb2+ adsorption process on a nano-Fe3O4-based adsorbent using the Response Surface Methodology. ACS Omega 2020, 5, 28305–28318. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Sharma, A.K.; Verma, R.; Chopade, R.L.; Pandit, P.P.; Nagar, V.; Aseri, V.; Choudhary, S.K.; Awasthi, G.; Awasthi, K.K.; et al. Heavy metal contamination of water and their toxic effect on living organisms. In Book the Toxicity of Environmental Pollutants; Dorta, D.J., Palma de Oliveira, D., Eds.; IntechOpen: London, UK, 2022. [Google Scholar] [CrossRef]
- Crichton, R.R. Chapter1: Metal toxicity—An introduction. In Metal Chelation in Medicine; Crichton, R.R., Ward, R.J., Hider, R.C., Eds.; RSC: London, UK, 2016; pp. 1–23. ISBN 978-1-78262-389-2. [Google Scholar]
- Sall, M.L.; Diaw, A.K.D.; Gningue-Sall, D.; Aaron, S.E.; Aaron, J.J. Toxic heavy metals: Impact on the environment and human health, and treatment with conducting organic polymers, a review. Environ. Sci. Pollut. Res. 2020, 27, 29927–29942. [Google Scholar] [CrossRef]
- Jain, R. Providing safe drinking water: A challenge for humanity. Clean Technol. Environ. Policy 2012, 14, 1–4. [Google Scholar] [CrossRef]
- Dinka, M.O. Safe drinking water: Concepts, benefits, principles and standards. In Water Challenges of an Urbanizing World; Glavan, M., Ed.; IntechOpen: London, UK, 2018. [Google Scholar] [CrossRef]
- Fu, F.; Wang, Q. Removal of heavy metal ions from wastewaters: A review. J. Environ. Manag. 2011, 92, 407–418. [Google Scholar] [CrossRef] [PubMed]
- Carolin, C.F.; Kumar, P.S.; Saravanan, A.; Joshiba, G.J.; Naushad, M. Efficient techniques for the removal of toxic heavy metals from aquatic environment: A review. J. Environ. Chem. Eng. 2017, 5, 2782–2799. [Google Scholar] [CrossRef]
- Qasem, N.A.A.; Mohammed, R.H.; Lawal, D.U. Removal of heavy metal ions from wastewater: A comprehensive and critical review. Npj Clean Water 2021, 4, 36. [Google Scholar] [CrossRef]
- Ince, M.; Ince, O.K. Wastewater Treatment; IntechOpen: London, UK, 2022. [Google Scholar]
- Arora, R. Adsorption of heavy metals—A review. Mater. Today: Proc. 2019, 18, 4745–4750. [Google Scholar] [CrossRef]
- Czikkely, M.; Neubauer, E.; Fekete, I.; Ymeri, P.; Fogarassy, C. Review of heavy metal adsorption processes by several organic matters from wastewaters. Water 2018, 10, 1377. [Google Scholar] [CrossRef]
- Liosis, C.; Papadopoulou, A.; Karvelas, E.; Karakasidis, T.E.; Sarris, I.E. Heavy metal adsorption using magnetic nanoparticles for water purification: A critical review. Materials 2021, 14, 7500. [Google Scholar] [CrossRef]
- Hussain, A.; Madan, S.; Madan, R. Removal of Heavy Metals from Wastewater by Adsorption. In Heavy Metals-Their Environmental Impacts and Mitigation; IntechOpen: London, UK, 2021. [Google Scholar]
- Sabzehmeidani, M.M.; Mahnaee, S.; Ghaedi, M.; Heidari, H.; Roy, V.A.L. Carbon based materials: A review of adsorbents for inorganic and organic compounds. Mater. Adv. 2021, 2, 598. [Google Scholar] [CrossRef]
- Muntean, S.G.; Nistor, M.A.; Ianoș, R.; Păcurariu, C.; Căpraru, A.; Surdu, V.A. Combustion synthesis of Fe3O4/Ag/C nanocomposite and application for dyes removal from multicomponent systems. Appl. Surf. Sci. 2019, 481, 825–837. [Google Scholar] [CrossRef]
- Ahmad, M.; Wang, J.; Xu, J.; Zhang, Q.; Zhang, B. Magnetic tubular carbon nanofibers as efficient Cu(II) ion adsorbent from wastewater. J. Clean. Prod. 2019, 252, 119825. [Google Scholar] [CrossRef]
- Peralta, M.E.; Nisticò, R.; Franzoso, F.; Magnacca, G.; Fernandez, L.; Parolo, M.E.; Leon, E.G.; Carlos, L. Highly efficient removal of heavy metals from waters by magnetic chitosan-based composite. Adsorption 2019, 25, 1337–1347. [Google Scholar] [CrossRef]
- Zeng, X.; Zhang, G.; Zhu, J.; Wu, Z. Adsorption of heavy metal ions in water by surface functionalized magnetic composites: A review. Environ. Sci. Water Res. Technol. 2022, 8, 907–925. [Google Scholar] [CrossRef]
- Sarma, G.K.; Sen Gupta, S.; Bhattacharyya, K.G. Nanomaterials as versatile adsorbents for heavy metal ions in water: A review. Environ. Sci. Pollut. Res. 2019, 26, 6245–6278. [Google Scholar] [CrossRef] [PubMed]
- Govan, J. Recent advances in magnetic nanoparticles and nanocomposites for the remediation of water resources. Magnetochem 2020, 6, 49. [Google Scholar] [CrossRef]
- Andelescu, A.; Nistor, M.A.; Muntean, S.G.; Rădulescu-Grad, M.E. Adsorption studies on copper, cadmium, and zinc ion removal from aqueous solution using magnetite/carbon nanocomposites. Sep. Sci. Technol. 2018, 53, 2352–2364. [Google Scholar] [CrossRef]
- Box, G.E.P.; Wilson, K.B. On the Experimental Attainment of Optimum Conditions. In Breakthroughs in Statistics; Springer Series in Statistics (Perspectives in Statistics); Kotz, S., Johnson, N.L., Eds.; Springer: New York, NY, USA, 1992. [Google Scholar]
- Myers, R.H.; Montgomery, D.C.; Vining, G.G.; Borror, C.N.; Kowalski, S.M. Response Surface Methodology: A retrospective and literature survey. J. Qual. Technol. 2004, 36, 53–77. [Google Scholar] [CrossRef]
- Asfaram, A.; Ghaedi, M.; Goudarzi, A.; Rajabi, M. Response surface methodology approach for optimization of simultaneous dyes and metal ions ultrasound-assisted adsorption onto Mn doped Fe3O4-NPs loaded on AC: Kinetic and isotherm study. Dalton Trans. 2015, 44, 14707–14723. [Google Scholar] [CrossRef]
- Brahmi, L.; Kaouah, F.; Boumaza, S.; Trari, M. Response surface methodology for the optimization of acid dye adsorption onto activated carbon prepared from wild date stones. Appl. Water Sci. 2019, 9, 171. [Google Scholar] [CrossRef]
- Lenth, R.V. Response-Surface Methods in R, using rsm. J. Stat. Softw. 2009, 32, 1–17. [Google Scholar] [CrossRef]
- Joos, A.; Rümenapp, C.; Wagner, F.E.; Gleich, B. Characterisation of iron oxide nanoparticles by Mössbauer spectroscopy at ambient temperature. J Magn. Magn. Mater. 2016, 399, 123–129. [Google Scholar] [CrossRef]
- Ianoş, R.; Tăculescu, E.A.; Păcurariu, C.; Lazău, I. Solution combustion synthesis and characterization of magnetite, Fe3O4, nanopowders. J. Am. Ceram. Soc. 2012, 95, 2236–2240. [Google Scholar] [CrossRef]
- Sing, K.S.W.; Everett, D.H.; Haul, R.A.W.; Moscou, L.; Pierotti, R.A.; Rouquerol, J.; Siemieniewska, T. Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity. Pure Appl. Chem. 1985, 57, 603. [Google Scholar] [CrossRef]
- Khan, M.N.; Sarwar, A. Determination of points of zero charge of natural and treated adsorbents. Surf. Rev. Lett. 2007, 14, 461–469. [Google Scholar] [CrossRef]
- Biswas, S.; Meikap, B.C.; Sen, T.K. Adsorptive removal of aqueous phase copper (Cu2+) and nickel (Ni2+) metal ions by synthesized biochar–biopolymeric hybrid adsorbents and process optimization by Response Surface Methodology (RSM). Water Air Soil Pollut. 2019, 230, 197. [Google Scholar] [CrossRef]
- Lagergren, S. Zur theorie der sogenannten adsorption geloster stoffe. K.-Sven. Vetensk. Handl. 1898, 24, 1–39. [Google Scholar]
- Ho, Y.; McKay, G. Pseudo-second order model for sorption processes. Process. Biochem. 1999, 34, 451–465. [Google Scholar] [CrossRef]
- Wu, F.-C.; Tseng, R.-L.; Juang, R.-S. Initial behavior of intraparticle diffusion model used in the description of adsorption kinetics. Chem. Eng. J. 2009, 153, 1–8. [Google Scholar] [CrossRef]
- Giraldo, L.; Erto, A.; Moreno-Piraján, J.C. Magnetite nanoparticles for removal of heavy metals from aqueous solutions: Synthesis and characterization. Adsorption 2013, 19, 465–474. [Google Scholar] [CrossRef]
- Dil, E.A.; Ghaedi, M.; Asfaram, A. The performance of nanorods material as adsorbent for removal of azo dyes and heavy metal ions: Application of ultrasound wave, optimization and modeling. Ultrason. Sonochem. 2017, 34, 792–802. [Google Scholar] [CrossRef]
- Kemik, Ö.F.; Ngwabebhoh, F.A.; Yildiz, U. A response surface modelling study for sorption of Cu2+, Ni2+, Zn2+ and Cd2+ using chemically modified poly(vinylpyrrolidone) and poly(vinylpyrrolidone-co-methylacrylate) hydrogels. Adsorp. Sci. Technol. 2017, 35, 263–283. [Google Scholar] [CrossRef]
- Freundlich, H. Uber die adsorption in losungen. Z. Phys. Chem. 1907, 57, 385–470. [Google Scholar] [CrossRef]
- Langmuir, I. The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef]
- Redlich, O.; Peterson, D.L. A useful adsorption isotherm. J. Phys. Chem. 1959, 63, 1024–1027. [Google Scholar] [CrossRef]
- Akaike, H. A new look at the statistical model identification. IEEE Trans. Automat. Contr. 1974, 19, 716–723. [Google Scholar] [CrossRef]
- Mayta-Armas, A.F.; Canchanya-Huaman, Y.; Pomalaya-Velasco, J.; Bendezú-Roca, Y.; Checca-Huaman, N.-R.; Ramos-Guivar, J.A. Enhanced Removal of As(V) and Pb(II) from Drinking and Irrigating Water Effluents Using Hydrothermally Synthesized Zeolite 5A. Water 2023, 15, 1892. [Google Scholar] [CrossRef]
- Kalantari, K.; Ahmad, M.B.; Masoumi, H.R.; Shameli, K.; Basri, M.; Khandanlou, R. Rapid adsorption of heavy metals by Fe3O4/talc nanocomposite and optimization study using response surface methodology. Int. J. Mol. Sci. 2014, 15, 12913–12927. [Google Scholar] [CrossRef]
- Khandanlou, R.; Ahmad, M.B.; Fard Masoumi, H.R.; Shameli, K.; Basri, M.; Kalantari, K. Rapid adsorption of copper(II) and lead(II) by rice straw/Fe3O4 nanocomposite: Optimization, equilibrium isotherms, and adsorption kinetics study. PLoS ONE 2015, 10, e0120264. [Google Scholar] [CrossRef]
- Ahmed, H.A.; Soliman, M.S.S.; Othman, S.A. Synthesis and characterization of magnetic nickel ferrite-modified montmorillonite nanocomposite for Cu(II) and Zn(II) ions removal from wastewater. Egypt. J. Chem. 2021, 64, 5627–5645. [Google Scholar] [CrossRef]
- Sulaiman, S.; Azis, R.S.; Ismail, I.; Man, H.C.; Yusof, K.F.M.; Abba, M.U.; Katibi, K.K. Adsorptive removal of copper (II) ions from aqueous solution using a magnetite nano-adsorbent from mill scale waste: Synthesis, characterization, adsorption and kinetic modelling studies. Nanoscale Res. Lett. 2021, 16, 168. [Google Scholar] [CrossRef]
- Qiu, X.; Li, N.; Yang, S.; Chen, D.; Xu, Q.; Li, H.; Lu, J. A new magnetic nanocomposite for selective detection and removal of trace copper ions from water. J. Mater. Chem. A 2015, 3, 1265–1271. [Google Scholar] [CrossRef]
- Gong, J.-L.; Wang, X.-Y.; Zeng, G.-M.; Chen, L.; Deng, J.-H.; Zhang, X.-R.; Niu, Q.-Y. copper(II) removal by pectin–iron oxide magnetic nanocomposite adsorbent. Chem. Eng. J. 2012, 185–186, 100–107. [Google Scholar] [CrossRef]
- Kataria, N.; Chauhan, A.K.; Garg, V.K.; Kumar, P. Sequestration of heavy metals from contaminated water using magnetic carbon nanocomposites. J. Hazard. Mater. Adv. 2022, 6, 100066. [Google Scholar] [CrossRef]
- Karimi, F.; Ayati, A.; Tanhaei, B.; Sanati, A.L.; Afshar, S.; Kardan, A.; Dabirifar, Z.; Karaman, C. Removal of metal ions using a new magnetic chitosan nano-bio-adsorbent; A powerful approach in water treatment. Environ. Res. 2022, 203, 111753. [Google Scholar] [CrossRef]
- Chen, D.; Awut, T.; Liu, B.; Ma, Y.; Wang, T.; Nurulla, I. Functionalized magnetic Fe3O4 nanoparticles for removal of heavy metal ions from aqueous solutions. E-Polymers 2016, 16, 313–322. [Google Scholar] [CrossRef]
- Nicola, R.; Costişor, O.; Ciopec, M.; Negrea, A.; Lazău, R.; Ianăşi, C.; Piciorus, E.M.; Len, A.; Almásy, L.; Szerb, E.I.; et al. Silica-coated magnetic nanocomposites for Pb2+ removal from aqueous solution. Appl. Sci. 2020, 10, 2726. [Google Scholar] [CrossRef]
- Alizadeh, K.; Khaledyan, E.; Mansourpanah, Y. Synthesis and application of amin-modified Fe3 O4@MCM-41 core-shell magnetic mesoporous for effective removal of Pb2+ ions from aqueous solutions and optimization with Response Surface Methodology. J. Water Environ. Nanotechnol. 2018, 3, 243–253. [Google Scholar]
- Ramos-Guivar, J.A.; Taipe, K.; Schettino, M.A., Jr.; Silva, E.; Morales Torres, M.A.; Passamani, E.C.; Litterst, F.J. Improved Removal Capacity and Equilibrium Time of Maghemite Nanoparticles Growth in Zeolite Type 5A for Pb(II) Adsorption. Nanomaterials 2020, 10, 1668. [Google Scholar] [CrossRef] [PubMed]
- Mirjavadi, E.S.; Tehrani, M.A.; Khadir, R.A. Effective adsorption of zinc on magnetic nanocomposite of Fe3O4/zeolite/cellulose nanofibers: Kinetic, equilibrium, and thermodynamic study. Environ. Sci. Pollut. Res. 2019, 26, 33478–33493. [Google Scholar] [CrossRef]
- Kowsari, M.; Sepehrian, H. Zinc adsorption properties of Alginate-SBA-15 nanocomposite. Int. J. Eng. Trans. A Basics 2015, 28, 1415. [Google Scholar]



| Factor | Variable | Unit | Level | ||
|---|---|---|---|---|---|
| −1 | 0 | +1 | |||
| X1 | pH | - | 4 | 6 | 8 |
| X2 | Initial metal concentration (Ccu; CPb; CZn) | mg/L | 10 | 105 | 200 |
| X3 | NC dosage (DNC) | g/L | 0.5 | 1 | 1.5 |
| Run | X1 * | X2 * | X3 * | YCu (%) | YPb (%) | YZn (%) |
|---|---|---|---|---|---|---|
| 1 | −1 | −1 | 0 | 49.30 | 56.32 | 34.90 |
| 2 | +1 | −1 | 0 | 84.79 | 88.65 | 74.14 |
| 3 | −1 | +1 | 0 | 18.47 | 21.34 | 15.42 |
| 4 | +1 | +1 | 0 | 38.16 | 43.56 | 29.34 |
| 5 | −1 | 0 | −1 | 19.88 | 29.14 | 17.48 |
| 6 | +1 | 0 | −1 | 50.21 | 52.65 | 28.64 |
| 7 | −1 | 0 | +1 | 41.12 | 50.26 | 36.97 |
| 8 | +1 | 0 | +1 | 72.65 | 76.45 | 60.25 |
| 9 | 0 | −1 | −1 | 57.90 | 72.14 | 36.70 |
| 10 | 0 | +1 | −1 | 16.98 | 18.72 | 10.12 |
| 11 | 0 | −1 | +1 | 77.80 | 88.31 | 69.20 |
| 12 | 0 | +1 | +1 | 42.00 | 30.45 | 21.87 |
| 13 | 0 | 0 | 0 | 45.22 | 57.21 | 38.90 |
| 14 | 0 | 0 | 0 | 44.43 | 58.01 | 37.66 |
| 15 | 0 | 0 | 0 | 45.58 | 58.12 | 39.89 |
| Pollutant | Coefficient | Std. Error | T Value | p-Value | Model Significance | |
|---|---|---|---|---|---|---|
| Cu(II) | (Intercept) | 46.61 | 0.52 | 90.39 | 1.25 × 10−14 | R2 = 0.9971 R2adj = 0.9955 F-value = 616.4 p-value = 3.99 × 10−11 |
| (X2)2 | 2.59 | 0.70 | 3.67 | 5.18 × 10−3 | ||
| X1 | 14.84 | 0.48 | 30.63 | 2.06 × 10−10 | ||
| X2 | −19.54 | 0.49 | −39.99 | 1.92 × 10−11 | ||
| X3 | 11.08 | 0.48 | 22.93 | 2.70 × 10−9 | ||
| X1:X2 | −3.95 | 0.68 | −5.78 | 2.65 × 10−4 | ||
| Pb(II) | (Intercept) | 57.28 | 0.55 | 104.13 | 8.08 × 10−14 | R2 = 0.997 R2adj = 0.994 F-value = 448.8 p-value = 1.15 × 10−9 |
| (X1)2 | −4.98 | 0.75 | −6.62 | 1.66 × 10−4 | ||
| X1 | 13.03 | 0.51 | 25.33 | 6.32 × 10−9 | ||
| X2 | −20.42 | 0.51 | −39.68 | 1.78 × 10−10 | ||
| X3 | 10.35 | 0.51 | 20.12 | 3.88 × 10−8 | ||
| X1:X2 | −2.53 | 0.73 | −3.47 | 8.40 × 10−3 | ||
| X2:X3 | 2.89 | 0.73 | 3.97 | 4.11 × 10−3 | ||
| Zn(II) | (Intercept) | 36.77 | 1.05 | 35.00 | 6.26 × 10−11 | R2 = 0.9696 R2adj = 0.9527 F-value = 57.41 p-value = 1.48 × 10−6 |
| X1 | 10.95 | 1.44 | 7.61 | 3.28 × 10−5 | ||
| X2 | −17.27 | 1.44 | −12.01 | 7.64 × 10−7 | ||
| X3 | 11.92 | 1.44 | 8.29 | 1.66 × 10−5 | ||
| X1:X2 | −6.33 | 2.03 | −3.11 | 1.25 × 10−2 | ||
| X2:X3 | −5.19 | 2.03 | −2.55 | 3.12 × 10−2 |
| Pollutant | Factor | Df | Sum Sq | Mean Sq | F Value | Pr (>F) |
|---|---|---|---|---|---|---|
| Cu(II) | (X2)2 | 1 | 11.2 | 11.23 | 6.0176 | 3.65 × 10−2 |
| X1,X2,X3 | 3 | 5678.7 | 1892.89 | 1014.1894 | 1.062 × 10−11 | |
| X1:X2 | 1 | 62.4 | 62.41 | 33.4386 | 2.6 × 10−4 | |
| Residuals | 10 | 16.8 | 1.87 | |||
| Lack of fit | 8 | 16.1 | 2.30 | 6.6491 | 13.69 × 10−2 | |
| Pure error | 2 | 0.7 | 0.35 | |||
| Pb(II) | (X1)2 | 1 | 92.7 | 92.73 | 43.779 | 1.6 × 10−4 |
| X1,X2,X3 | 3 | 5551.3 | 1850.43 | 873.645 | 2.11 × 10−10 | |
| X1:X2 | 1 | 25.6 | 25.55 | 12.064 | 8.4 × 10−3 | |
| X2:X3 | 1 | 33.4 | 33.41 | 15.773 | 4.1 × 10−3 | |
| Residuals | 8 | 16.9 | 2.12 | |||
| Lack of fit | 6 | 16.1 | 2.68 | 6.084 | 14.78 × 10−2 | |
| Pure error | 2 | 0.9 | 0.44 | |||
| Zn (II) | X1,X2,X3 | 3 | 4482.7 | 1494.24 | 90.287 | 4.90 × 10−7 |
| X1:X2 | 1 | 160.3 | 160.28 | 9.6844 | 1.25 × 10−2 | |
| X2:X3 | 1 | 107.6 | 107.64 | 6.504 | 31.2 × 10−3 | |
| Residuals | 9 | 148.9 | 16.55 | |||
| Lack of fit | 7 | 146.5 | 20.92 | 16.7584 | 5.75 × 10−2 | |
| Pure error | 2 | 2.5 | 1.25 |
| Pollutant | pH | Initial Concentration (mg/L) | Adsorbent Dose (g/L) | Removal Percentage (%) | |
|---|---|---|---|---|---|
| Predicted | Experimental | ||||
| Cu(II) | 7.0 | 10 | 1.25 | 83.37 | 82.88 |
| 6.0 | 100 | 1 | 46.61 | 42.79 | |
| 9.0 | 10 | 1 | 98.10 | 86.96 | |
| 6.5 | 100 | 1.5 | 60.64 | 58.65 | |
| 8.0 | 10 | 0.25 | 69.41 | 61.63 | |
| Pb(II) | 7.0 | 10 | 1.25 | 87.54 | 86.66 |
| 6.0 | 100 | 1 | 59.91 | 57.20 | |
| 9.0 | 10 | 1 | 89.80 | 88.93 | |
| 6.4 | 100 | 1.5 | 70.98 | 65.25 | |
| 8.0 | 10 | 0.25 | 77.08 | 73.54 | |
| Zn(II) | 7.0 | 30 | 1.25 | 71.02 | 69.75 |
| 6.0 | 100 | 1 | 39.12 | 38.90 | |
| 9.0 | 10 | 1 | 82.55 | 78.22 | |
| 6.4 | 100 | 1.5 | 52.13 | 45.62 | |
| 8.0 | 10 | 0.25 | 45.66 | 43.66 | |
| Pollutant | Conc (mg/L) | qe,exp (mg/g) | Pseudo-First-Order Model | Pseudo-Second-Order Model | Intraparticle Diffusion Model | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| qe,calc (mg/g) | k1 .103 (min−1) | R2 | qe,calc (mg/g) | k2 .104 (g/mg. min) | R2 | ki (mg/g. min0.5) | l | R2 | |||
| Cu(II) | 10 | 8.25 | 2.79 | 9.05 | 0.7092 | 8.77 | 64.77 | 0.9971 | 0.26 | 4.39 | 0.8955 |
| 30 | 20.33 | 5.78 | 10.66 | 0.4438 | 20.84 | 16.39 | 0.9976 | 0.32 | 15.39 | 0.9411 | |
| 50 | 31.29 | 9.46 | 18.40 | 0.8985 | 31.60 | 8.39 | 0.9962 | 0.73 | 20.35 | 0.8274 | |
| 100 | 45.22 | 20.83 | 13.91 | 0.5960 | 48.05 | 7.46 | 0.9989 | 0.96 | 32.36 | 0.8782 | |
| 150 | 57.28 | 15.05 | 35.10 | 0.4924 | 53.61 | 7.32 | 0.9914 | 0.73 | 45.65 | 0.6181 | |
| Pb(II) | 10 | 8.75 | 4.27 | 18.67 | 0.8608 | 7.11 | 93.71 | 0.9943 | 0.34 | 3.62 | 0.9824 |
| 30 | 22.41 | 6.76 | 18.91 | 0.7809 | 18.45 | 89.36 | 0.9983 | 0.63 | 12.61 | 0.9775 | |
| 50 | 33.42 | 11.29 | 23.21 | 0.6508 | 28.22 | 82.81 | 0.9984 | 0.97 | 18.57 | 0.9766 | |
| 100 | 57.21 | 15.85 | 15.45 | 0.9299 | 21.93 | 45.93 | 0.9871 | 1.62 | 32.55 | 0.9449 | |
| 150 | 63.45 | 25.47 | 44.10 | 0.9715 | 54.32 | 16.50 | 0.9983 | 2.02 | 32.74 | 0.9461 | |
| Zn(II) | 10 | 6.83 | 5.63 | 21.33 | 0.9876 | 7.43 | 62.79 | 0.9989 | 0.21 | 3.75 | 0.9926 |
| 30 | 17.32 | 10.23 | 14.56 | 0.8139 | 17.69 | 37.68 | 0.9951 | 0.43 | 10.58 | 0.9863 | |
| 50 | 23.96 | 18.83 | 20.46 | 0.9726 | 25.83 | 19.87 | 0.9992 | 0.62 | 14.59 | 0.9825 | |
| 100 | 38.90 | 35.75 | 18.49 | 0.9925 | 38.02 | 14.12 | 0.9977 | 1.15 | 21.31 | 0.9632 | |
| 150 | 43.80 | 31.45 | 21.67 | 0.9421 | 45.47 | 5.95 | 0.9997 | 0.66 | 33.57 | 0.9981 | |
| Isotherm | Equation * | Parameter | Value | ||
|---|---|---|---|---|---|
| Cu(II) | Pb(II) | Zn(II) | |||
| Freundlich | KF (mg/g(mg/L)−1/n) | 10.03 | 10.16 | 5.29 | |
| n | 2.333 | 2.339 | 2.16 | ||
| R2 | 0.9632 | 0.9634 | 0.9778 | ||
| χ2 | 26.33 | 26.52 | 6.87 | ||
| AIC | 12.87 | 12.92 | 4.81 | ||
| BIC | 12.46 | 12.50 | 4.40 | ||
| Langmuir | qm (mg/g) | 81.36 | 83.54 | 57.11 | |
| KL (L/mg) | 0.048 | 0.049 | 0.032 | ||
| R2 | 0.9851 | 0.9833 | 0.9908 | ||
| χ2 | 11.28 | 12.12 | 2.84 | ||
| AIC | 7.79 | 8.22 | −0.49 | ||
| BIC | 7.37 | 7.80 | −0.90 | ||
| Redlich–Peterson | KRP (L/g) | 9.21 | 12.27 | 7.49 | |
| αRP (mg/L)−β | 0.535 | 0.835 | 1.073 | ||
| β | 0.677 | 0.643 | 0.585 | ||
| R2 | 0.9728 | 0.9709 | 0.9827 | ||
| χ2 | 29.19 | 31.57 | 8.04 | ||
| AIC | 15.49 | 15.96 | 7.76 | ||
| BIC | 12.46 | 12.50 | 4.40 | ||
| Pollutant | Magnetic Nanocomposite | Dose (g) | pH | Temp (°C) | te (min) | qt (mg/g) | R (%) | Reference |
|---|---|---|---|---|---|---|---|---|
| Cu (II) | Fe3O4/talc nanocomposite | 0.12 | - | - | 2 | 100.92 | 72.15 | [47] |
| Rice straw/magnetic nanocomposites | 0.13 | - | - | 1 | 16.31 | 94.42 | [48] | |
| NiFe2O4/Mod MMT | 0.10 | 6 | 25 | 60 | 18.73 | 99.23 | [49] | |
| magnetite nano-adsorbent | 0.05 | 5.4 | 25 | 120 | 4.41 | 62.61 | [50] | |
| core–shell structured spherical magnetic nanocomposite | 0.03 | - | - | 20 | 35.71 | 80.00 | [51] | |
| Pectin–iron oxide magnetic nanocomposite | 0.02 | 5 | 25 | 1440 | 48.99 | - | [52] | |
| Fe3O4@Carbon nanocomposite | 0.80 | 6.5 | 27 | 120 | 48.08 | 92.00 | [53] | |
| magnetic chitosan/Al2O3/Fe3O4 | 1.00 | 5.3 | 15 | 300 | 31.65 | 93.69 | [54] | |
| NC | 1.00 | 5.8 | 25 | 10 | 81.36 | 90.13 | This work | |
| Pb(II) | Fe3O4 magnetite nanoparticles | 0.20 | 5.5 | 25 | 1440 | 37.26 | - | [39] |
| Fe3O4/talc nanocomposite | 0.12 | - | - | 2 | 74.62 | 91.35 | [47] | |
| rice straw/magnetic nanocomposites | 0.13 | - | - | 1 | 19.45 | 96.35 | [48] | |
| Fe3O4@carbon nanocomposite | 0.80 | 6.5 | 27 | 120 | 151.50 | 97.00 | [53] | |
| Fe3O4@SiO2-NH-pyd | 0.10 | 7 | 25 | 20 | 72.00 | 96.00 | [55] | |
| MNPs35@SiO2 | 0.10 | - | - | 90 | 17.10 | - | [56] | |
| Fe3O4@MCM-41-NH2 | 0.24 | - | 25 | - | 46.08 | 95.15 | [57] | |
| NPZEO3 | 0.50 | 5.5 | 27 | 5 | 252 | 99.90 | [58] | |
| NC | 1.00 | 5.9 | 25 | 10 | 83.48 | 80.32 | This work | |
| Zn(II) | magnetite/carbon nanocomposites | 1.00 | 6.1 | 25 | 240 | 48.45 | 76.90 | [25] |
| Fe3O4 magnetite nanoparticles | 0.20 | 5.5 | 25 | 1440 | 9.10 | - | [39] | |
| magnetic nickel ferrite-modified montmorillonite nanocomposite | 0.10 | 6 | 25 | 90 | 5.13 | 91.67 | [47] | |
| magnetic chitosan/Al2O3/Fe3O4 | 1.00 | 5.3 | 15 | 300 | 24.27 | 83.81 | [54] | |
| magnetic zeolite/cellulose nanofibers | 0.03 | 7 | 30 | 120 | 9.45 | 96.00 | [59] | |
| alginate-SBA-15 sorbent nanocomposite | 1.00 | - | - | 240 | 46.30 | - | [60] | |
| NC | 1.00 | 5.8 | 25 | 10 | 58.47 | 91.53 | This work |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muntean, S.G.; Halip, L.; Nistor, M.A.; Păcurariu, C. Removal of Metal Ions via Adsorption Using Carbon Magnetic Nanocomposites: Optimization through Response Surface Methodology, Kinetic and Thermodynamic Studies. Magnetochemistry 2023, 9, 163. https://doi.org/10.3390/magnetochemistry9070163
Muntean SG, Halip L, Nistor MA, Păcurariu C. Removal of Metal Ions via Adsorption Using Carbon Magnetic Nanocomposites: Optimization through Response Surface Methodology, Kinetic and Thermodynamic Studies. Magnetochemistry. 2023; 9(7):163. https://doi.org/10.3390/magnetochemistry9070163
Chicago/Turabian StyleMuntean, Simona Gabriela, Liliana Halip, Maria Andreea Nistor, and Cornelia Păcurariu. 2023. "Removal of Metal Ions via Adsorption Using Carbon Magnetic Nanocomposites: Optimization through Response Surface Methodology, Kinetic and Thermodynamic Studies" Magnetochemistry 9, no. 7: 163. https://doi.org/10.3390/magnetochemistry9070163
APA StyleMuntean, S. G., Halip, L., Nistor, M. A., & Păcurariu, C. (2023). Removal of Metal Ions via Adsorption Using Carbon Magnetic Nanocomposites: Optimization through Response Surface Methodology, Kinetic and Thermodynamic Studies. Magnetochemistry, 9(7), 163. https://doi.org/10.3390/magnetochemistry9070163






