Mössbauer Research and Magnetic Properties of Dispersed Microspheres from High-Calcium Fly Ash
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Characterization Methods
3. Results
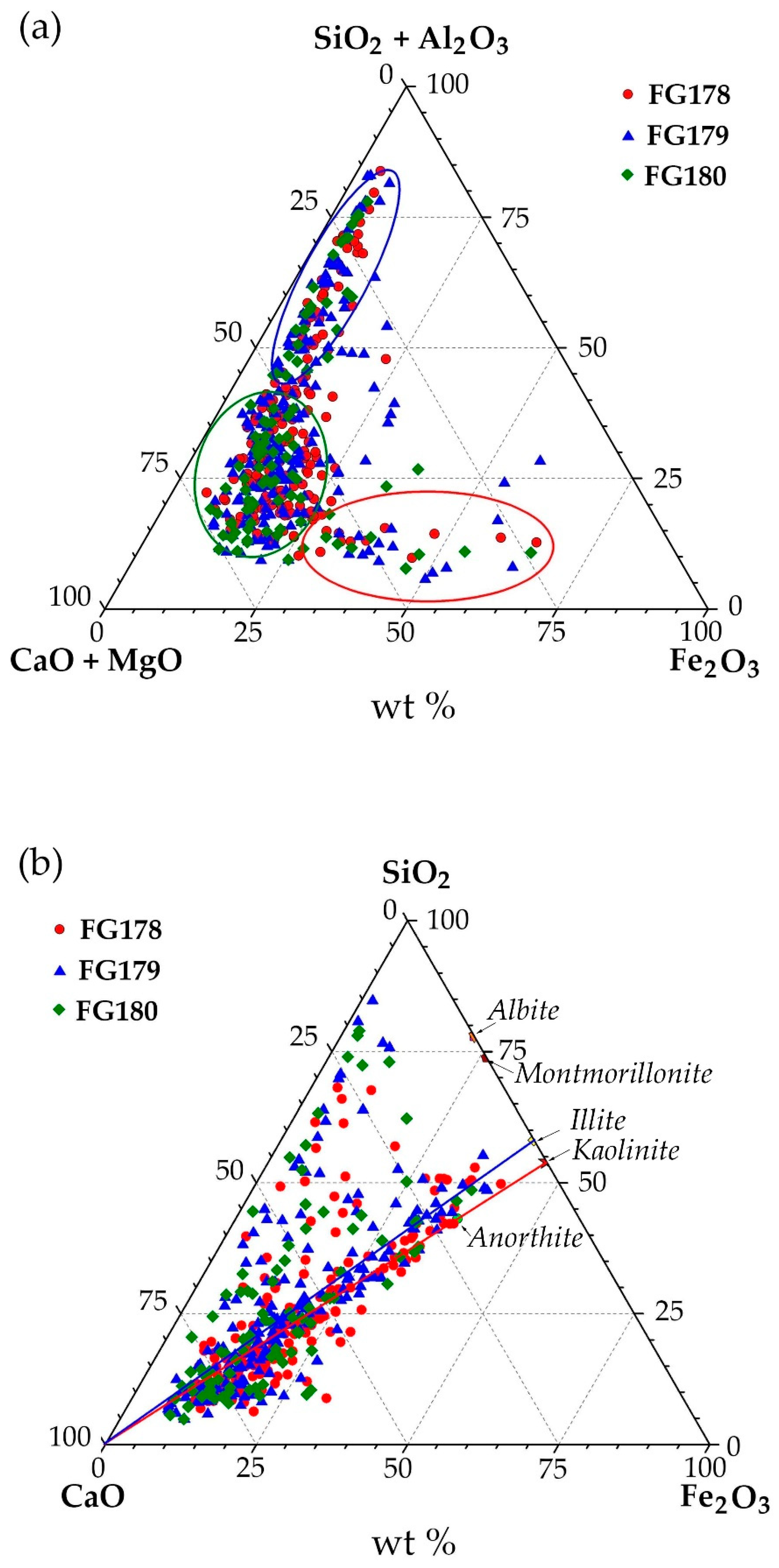
3.1. SEM-EDS Analysis of Narrow Fractions of Dispersed Microspheres
- Group 1 (shown in green) contains microspheres where CaO content is >40 wt % and the total content of SiO2 and Al2O3 is ≤35 wt %. Approximately 2/3 of investigated particles meet these composition criteria.
- Group 2 (shown in blue) consists of microspheres with elevated SiO2 and Al2O3 contents: the total content of these oxides in globules increases significantly (from 40 to 75 wt %), which is typical of 1/3 of all microspheres.
- Group 3 (shown in red) contains microspheres with high FeO content (from 30 to 60 wt %), which is typical of only 3% of the investigated globules.
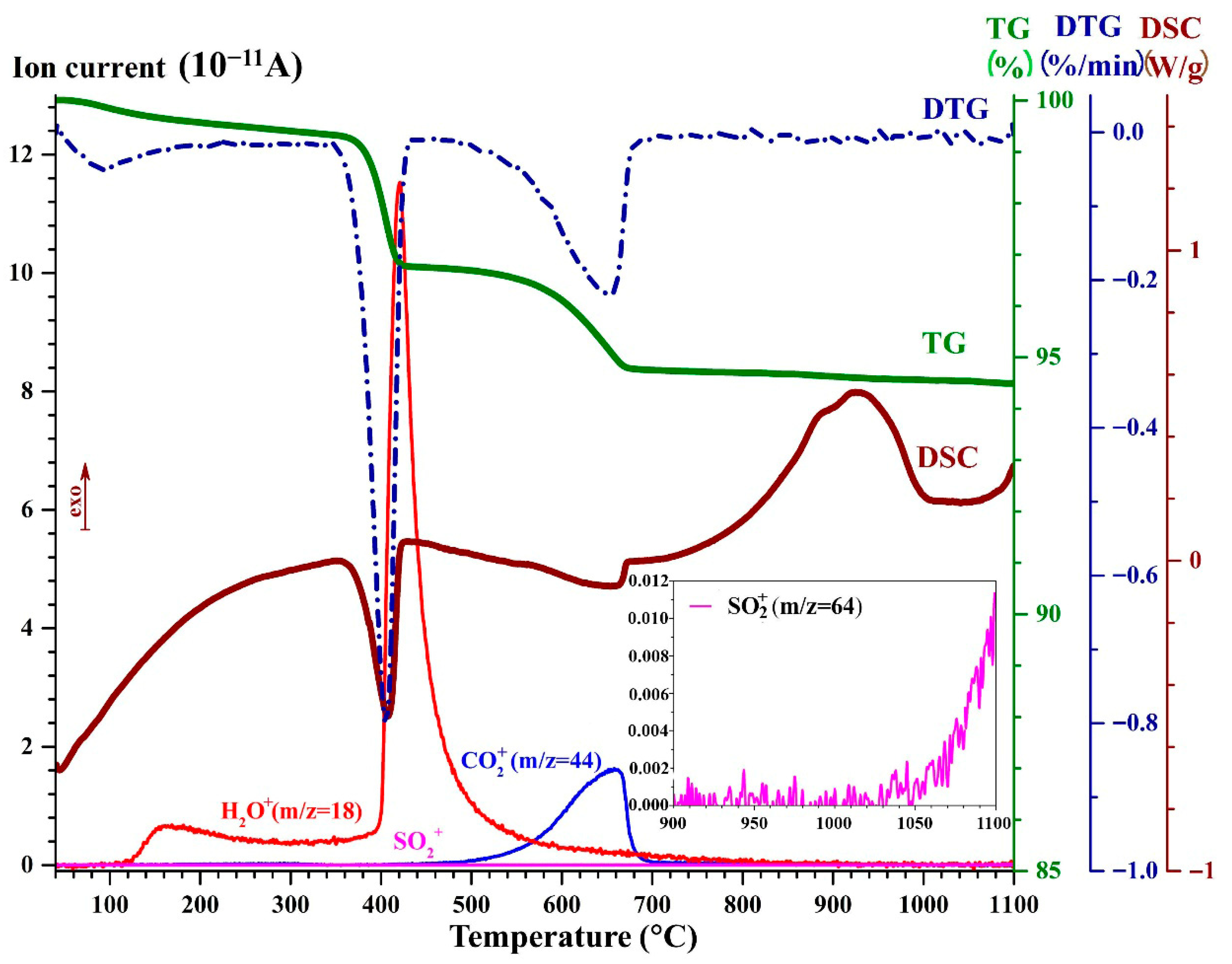
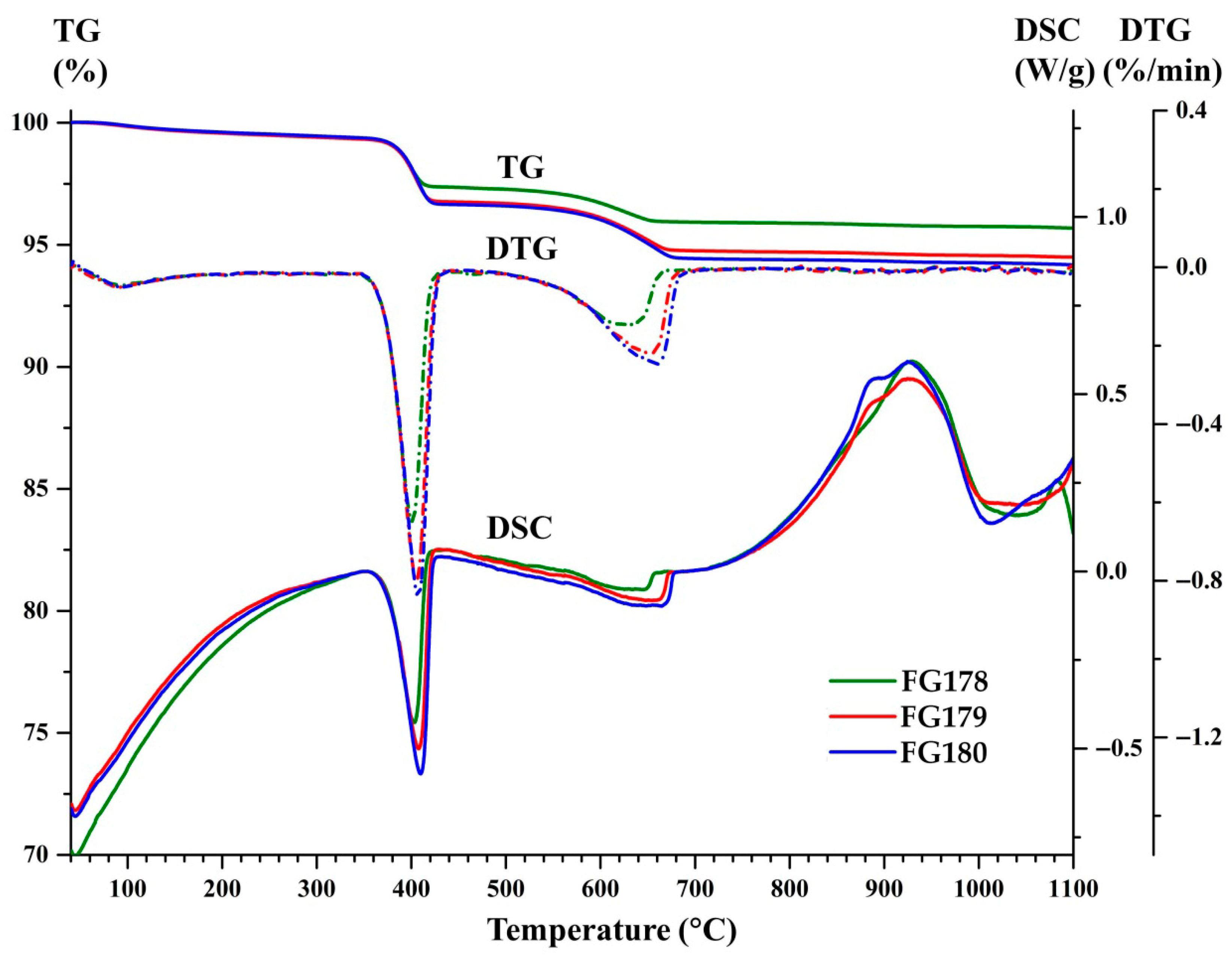
3.2. DSC-TG-DTG Analysis of the Narrow Fractions of Dispersed Microspheres
- dehydration in the temperature range of 40–244 °C, mainly due to thermal desorption of hygroscopic moisture, being accompanied by mass loss (0.46–0.50 wt.%) and an endothermic effect with maxima at 95 °C;
- dissociation of Ca(OH)2 in the temperature range of 244–437 °C being accompanied by mass loss (2.17–2.89 wt %) and an endothermic effect with maxima at 403–410 °C;
- dissociation of calcium carbonate (or solid solutions based on it with the net formula Ca(1−x−y)MgxFeyCO3) in the temperature range of 437–810 °C, being accompanied by mass loss (1.48–2.27 wt %) and an endothermic effect as a bimodal peak with the global maxima in the temperature range of 644–664 °C and local maxima, in the temperature range of 623–630 °C;
- crystallization of a new phase/phases (presumedly, wollastonite and/or pseudowollastonite with the net formula CaSiO3) in the temperature range of 810–1100 °C, being accompanied by exothermic effects as a bimodal peak with the global maxima in the temperature range of 930–935 °C and local maxima, in the temperature range of 854–886 °C.
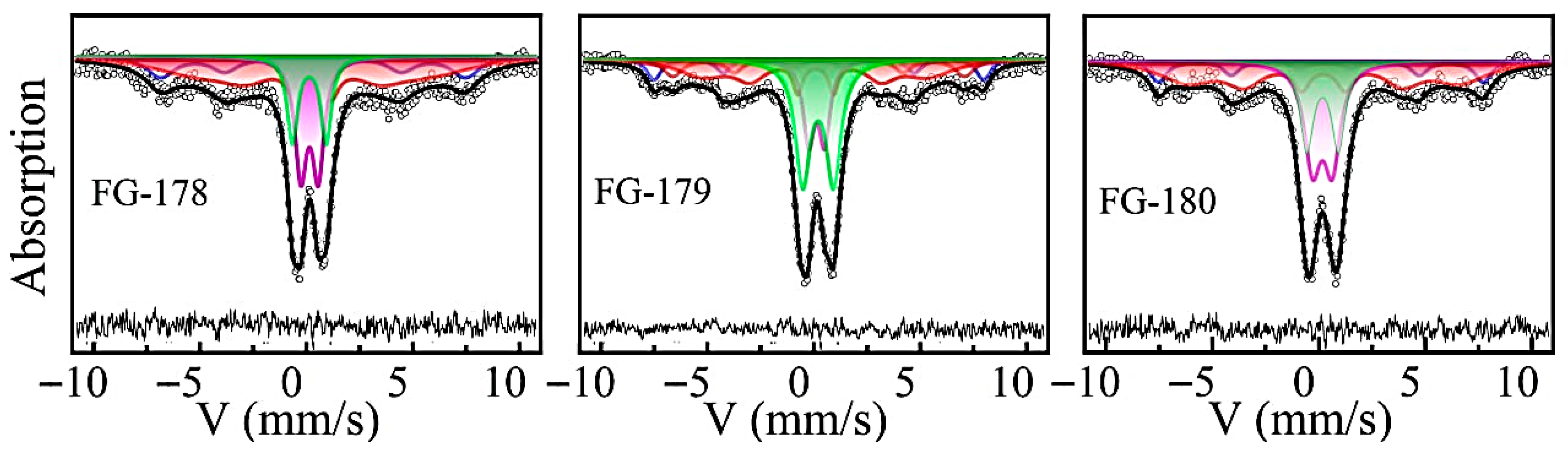
3.3. 57Fe Mössbauer Spectroscopy
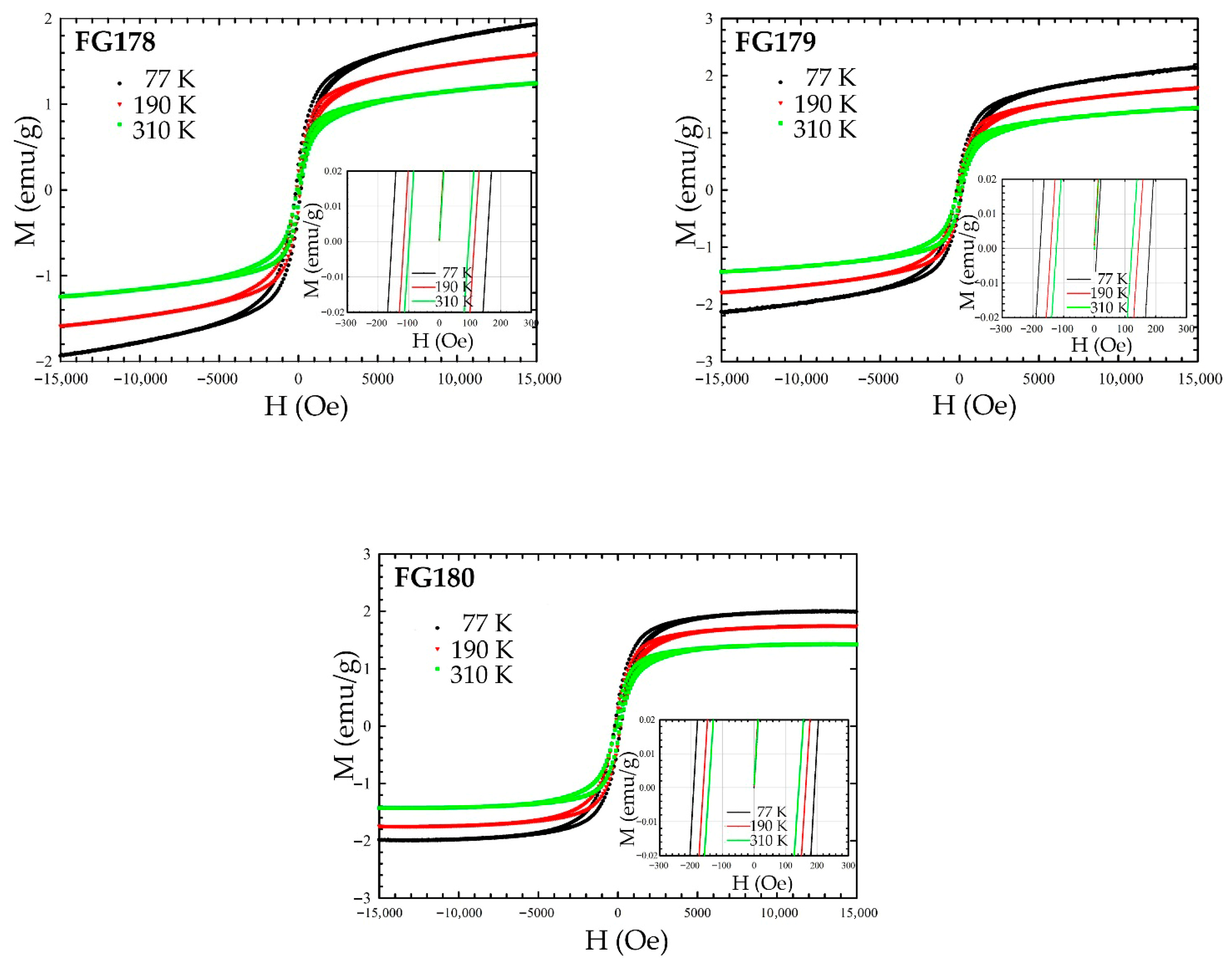
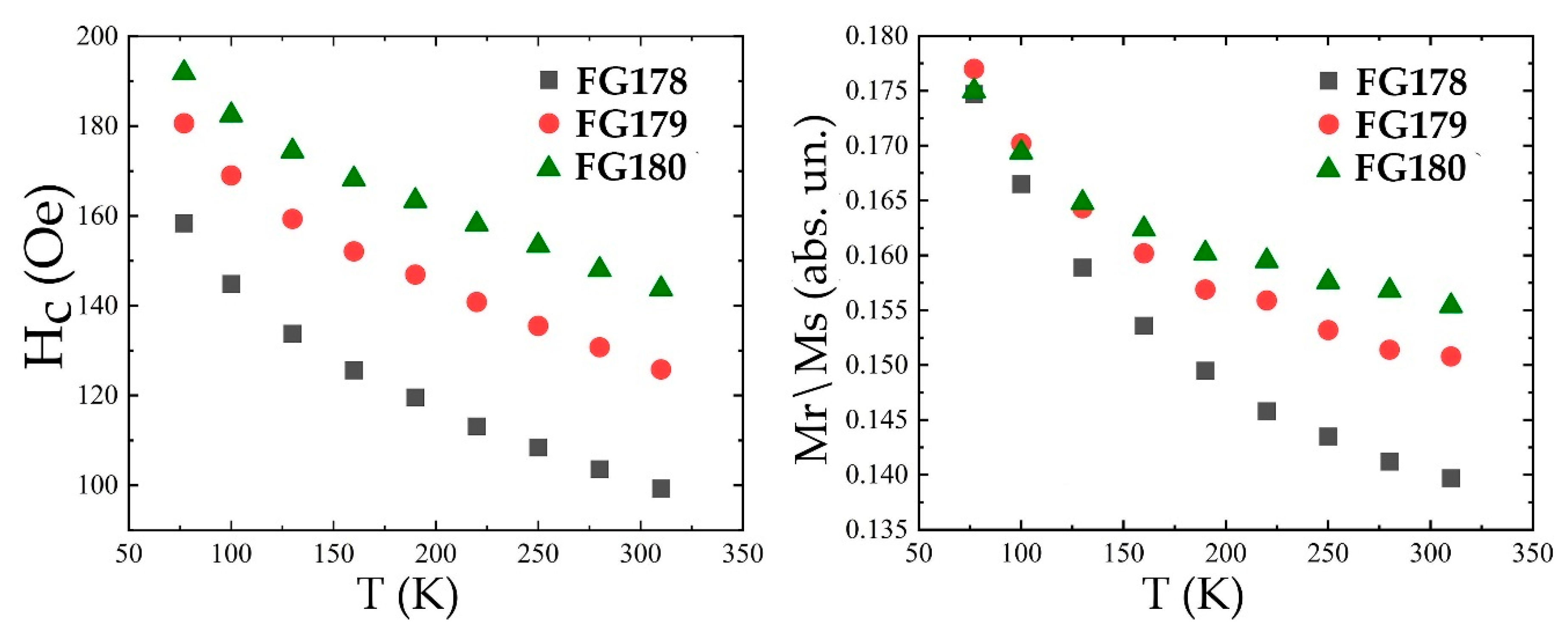
3.4. Magnetization Measurements
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Roy, W.R.; Thiery, R.G.; Schuller, R.M.; Subway, J.J. Coal Fly Ash: A Review of the Literature and Proposed Classification System with Emphasis on Environmental Impacts; State Geological Survey Division: Champaign, IL, USA, 1981; p. 69. [Google Scholar]
- Rodrigues, P.; Silvestre, J.D.; Flores-Colen, I.; Viegas, C.A.; Ahmed, H.H.; Kurda, R.; de Brito, J. Evaluation of the Ecotoxicological Potential of Fly Ash and Recycled Concrete Aggregates Use in Concrete. Appl. Sci. 2020, 10, 351. [Google Scholar] [CrossRef]
- Fomenko, E.V.; Anshits, N.N.; Solovyov, L.A.; Knyazev, Y.V.; Semenov, S.V.; Bayukov, O.A.; Anshits, A.G. Magnetic Fractions of PM2.5, PM2.5–10, and PM10 from Coal Fly Ash as Environmental Pollutants. ACS Omega 2021, 6, 20076–20085. [Google Scholar] [CrossRef] [PubMed]
- Gune, M.; Harshavardhana, B.G.; Balakrishna, K.; Udayashankar, H.N.; Shankar, R.; Manjunatha, B.R. Rock magnetic finger-printing of soil from a coal-fired thermal power plant. Environ. Monit. Assess. 2016, 188, 272. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.; Doh, S.-J.; Yu, Y. Anthropogenic contribution of magnetic particulates in urban roadside dust. Atmos. Environ. 2009, 43, 3137–3144. [Google Scholar] [CrossRef]
- Cohen, A.J.; Brauer, M.; Burnett, R.; Anderson, H.R.; Frostad, J.; Estep, K.; Balakrishnan, K.; Brunekreef, B.; Dandona, L.; Dandona, R.; et al. Estimates and 25-year trends of the global disease burden attributable to ambient air pollution: An analysis of data from the global burden of diseases study 2015. Lancet 2017, 389, 1907–1918. [Google Scholar] [CrossRef]
- Newell, K.; Kartsonaki, C.; Lam, K.B.H.; Kurmi, O.P. Cardiorespiratory health effects of particulate ambient air pollution exposure in low-income and middle-income countries: A systematic review and meta-analysis. Lancet Planet. Health 2017, 1, e368–e380. [Google Scholar] [CrossRef]
- Meo, S.A.; Salih, M.A.; Al-Hussain, F.; Alkhalifah, J.M.; Meo, A.S.; Akram, A. Environmental pollutants PM2.5, PM10, carbon monoxide (CO), nitrogen dioxide (NO2), sulfur dioxide (SO2), and Ozone (O3) impair human cognitive functions. Eur. Rev. Med. Pharmacol. Sci. 2024, 28, 789–796. [Google Scholar] [CrossRef]
- Anshits, A.G.; Bajukov, O.A.; Kondratenko, E.V.; Anshits, N.N.; Pletnev, O.V.; Rabchevskii, E.V. Catalytic Properties and Nature of Active Centers of Ferrospheres in Oxidative Coupling of Methane. Appl. Catal. A Gen. 2016, 524, 192–199. [Google Scholar] [CrossRef]
- Anshits, N.N.; Fedorchak, M.A.; Sharonova, O.M.; Kirik, N.P.; Shishkina, N.N.; Zhizhaev, A.M.; Anshits, A.G. Structure–Composition Relationship of Platelike Ferrospheres in Calcium-Rich Power Plant Ash. Inorg. Mat. 2018, 54, 466–472. [Google Scholar] [CrossRef]
- Goldman, A. Crystal Structure of Ferrites. In Handbook of Modern Ferromagnetic Materials; The Springer International Series in Engineering and Computer Science; Springer: Boston, MA, USA, 1999; Volume 505, pp. 207–227. [Google Scholar] [CrossRef]
- Ni, D.; Cava, R.J. Ferrites without iron as potential quantum materials. Prog. Solid State Chem. 2022, 66, 10034. [Google Scholar] [CrossRef]
- Yadav, N.; Ahmaruzzaman, M. Recent advancements in CaFe2O4-based composite: Properties. synthesis. and multiple applications. Energy Environ. 2023, 35, 0958305X2311554. [Google Scholar] [CrossRef]
- Taylor, H.F.W. Cement Chemistry, 2nd ed.; Academic Press: London, UK, 1997. [Google Scholar] [CrossRef]
- Antunes, I.; Ruivo, L.C.M.; Tarelho, L.A.C.; Yaremchenko, A.; Frade, J. Solid state synthesis of Ca2Fe2O5 by reactive firing of calcite and siderite. Ceram. Int. 2022, 48, 34025–34032. [Google Scholar] [CrossRef]
- Songvilay, M.; Petit, S.; Koza, M.; Rols, S.; Suard, E.; Skumryev, V.; Martin, C.; Damay, F. Disorder and magnetic excitations in CaCrxFe2−xO4 (x = 0.5). Phys. Rev. B 2020, 101, 014407. [Google Scholar] [CrossRef]
- Geller, S.; Grant, R.W.; Gonser, U. Crystal Chemistry and Magnetic Structures of Substituted Ca2[Fe](Fe)O5. Prog. Solid State Chem. 1971, 5, 1–26. [Google Scholar] [CrossRef]
- Itoh, S.; Tsubone, A.; Matsubae-Yokoyama, K.; Nakajima, K.; Nagasaka, T. New EAF Dust Treatment Process with the Aid of Strong Magnetic Field. ISIJ Int. 2008, 48, 1339–1344. [Google Scholar] [CrossRef]
- Xue, B.; Jia, L.; Zhang, F. Biodiesel production from soybean and Jatropha oils by magnetic CaFe2O4-Ca2Fe2O5-based catalyst. Energy 2014, 68, 584–591. [Google Scholar] [CrossRef]
- You, Z.; Ma, X.; Yu, Z.; Chen, C.; Dong, J.; Yue, W. Application of CaFe2O4/Ca2Fe2O5 to the fixation of heavy metals by co-combustion of coal and coconut shells: Experimental, simulation studies and environmental analysis. Ind. Crops Prod. 2024, 220, 119291. [Google Scholar] [CrossRef]
- Fomenko, E.V.; Akimochkina, G.V.; Anshits, A.G. Narrow dispersed fractions of high-calcium fly ash produced from the pulverized combustion of Irsha-Borodinsky coal. Therm. Eng. 2019, 66, 560–568. [Google Scholar] [CrossRef]
- GOST 5382-2019; Cements and Materials for Cement Production. Chemical Analysis Methods. Publishing House of Standards: Moscow, Russia, 2019. Available online: https://docs.cntd.ru/document/1200168999 (accessed on 20 July 2025). (In Russian)
- State Standard GOST 16190-70; Sorbents. Method of Bulk Density Determination. Publishing House of Standards: Moscow, Russia, 1970. Available online: https://docs.cntd.ru/document/1200018309 (accessed on 20 July 2025). (In Russian)
- Rietveld, H.M. A Profile Refinement Method for Nuclear and Magnetic Structures. J. Appl. Crystallogr. 1969, 2, 65–71. [Google Scholar] [CrossRef]
- Roberts, A.P.; Heslop, D.; Zhao, X.; Pike, C.R. Understanding fine magnetic particle systems through use of first-order reversal curve diagrams. Rev. Geophys. 2014, 52, 557–602. [Google Scholar] [CrossRef]
- Fomenko, E.V.; Akimochkina, G.V.; Anshits, A.G. Investigation of the composition of dispersed particles in fly ash and determination of the routes of PM10 formation from the mineral components of lignite. Chem. Sustain. Dev. 2024, 32, 860–868. [Google Scholar] [CrossRef]
- Kaminskas, R.; Kubiliūtė, R. The effect of coal ash on synthesis and properties of tricalcium silicate. Mater. Sci. (Medžiagotyra) 2010, 16, 73–79. [Google Scholar]
- Giergiczny, Z. Effect of some additives on the reactions in fly ASH-Ca(OH)2 system. J. Therm. Anal. Calorim. 2004, 76, 747–754. [Google Scholar] [CrossRef]
- Redhammer, G.J.; Amthauer, G.; Tippelt, G.; Lottermoser, W.; Roth, G. 57Fe Mössbauer Spectroscopic Investigations on the Brownmillerite Series Ca2(Fe2−xAlx)O5. AIP Conf. Proc. 2005, 765, 179–184. [Google Scholar] [CrossRef]
- Eibschütz, M.; Ganiel, U.; Shtrikman, S. Mössbauer and magnetic studies of dicalcium ferrite (Ca2Fe2O5). J. Mater. Sci. 1969, 4, 574–580. [Google Scholar] [CrossRef]
- Chakir, A.; Aqdim, S.; Mehdaoui, B.; El Bouari, A. Deciphering driven phase transitions: A study on the dielectric and electrical properties of Ca2Fe2O5. Eur. Phys. J. B 2024, 97, 40. [Google Scholar] [CrossRef]
- Redhammer, G.J.; Tippelt, G.; Roth, G.; Amthauer, G. Structural variations in the brownmillerite series Ca2(Fe2−xAlx)O5: Single-crystal X-ray diffraction at 25 °C and high-temperature X-ray powder diffraction (25 °C ≤ T ≤ 1000 °C). Am. Min. 2004, 89, 405–420. [Google Scholar] [CrossRef]
- Stöber, S.; Redhammer, G.; Schorr, S.; Prokhnenko, O.; Pöllmann, H. Structure refinements of members in the brownmillerite solid solution series Ca2Alx(Fe0.5Mn0.5)2−xO5+δ with 1/2 ≤ x ≤ 4/3. J. Solid State Chem. 2013, 197, 420–428. [Google Scholar] [CrossRef]
- Phan, T.L.; Tran, N.; Kim, D.H.; Tho, P.T.; Huy, B.T.; Dang, N.T.; Yang, D.S.; Lee, B.W. Electronic structure and magnetic properties of Al-doped Ca2Fe2O5 brownmillerite compounds. J. Am. Ceram. Soc. 2018, 101, 2181–2189. [Google Scholar] [CrossRef]
- Kharton, V.V.; Tsipis, E.V.; Kolotygin, V.A.; Avdeev, M.; Viskup, A.P.; Waerenborgh, J.C.; Frade, J.R. Mixed Conductivity and Stability of CaFe2O4–δ. J. Electrochem. Soc. 2008, 155, P13–P20. [Google Scholar] [CrossRef]
- Knyazev, Y.V.; Tarasov, A.S.; Platunov, M.S.; Trigub, A.L.; Bayukov, O.A.; Boronin, A.I.; Solovyov, L.A.; Rabchevskii, E.V.; Shishkina, N.N.; Anshits, A.G. Structural and electron transport properties of CaFe2O4 synthesized in air and in helium atmosphere. J. Alloys Compd. 2020, 20, 153073. [Google Scholar] [CrossRef]
- Gütlich, P.; Bill, E.; Trautwein, A.X. Mössbauer Spectroscopy and Transition Metal Chemistry: Fundamentals and Applications; Springer: Berlin/Heidelberg, Germany, 2010. [Google Scholar] [CrossRef]
- Preisach, F. Über die magnetische Nachwirkung. Z. Physik. 1935, 94, 277–302. (In Germany) [Google Scholar] [CrossRef]
- Egli, R.; Chen, A.P.; Winklhofer, M.; Kodama, K.P.; Horng, C.S. Detection of noninteracting single domain particles using first-order reversal curve diagrams. Geochem. Geophys. Geosyst. 2010, 11, Q01Z11. [Google Scholar] [CrossRef]
- Pike, C.R.; Roberts, A.P.; Dekkers, M.J.; Verosub, K.L. An investigation of multi-domain hysteresis mechanisms using FORC diagrams. Phys. Earth Planet. Inter. 2001, 126, 11–25. [Google Scholar] [CrossRef]
- Muxworthy, A.R.; Dunlop, D.J. First-order reversal curve (FORC) diagrams for pseudo-single-domain magnetites at high temperature. Earth Planet. Sci. Lett. 2002, 203, 369–382. [Google Scholar] [CrossRef]
- Smirnov, A.V. Low-temperature magnetic properties of magnetite using first-order reversal curve analysis: Implications for the pseudo-single domain state. Geochem. Geophys. Geosyst. 2006, 7, Q11011. [Google Scholar] [CrossRef]
- Muxworthy, A.R.; King, J.G.; Heslop, D. Assessing the ability of first-order reversal curve (FORC) diagrams to unravel complex magnetic signals. J. Geophys. Res. 2005, 110, B01105. [Google Scholar] [CrossRef]
- Punnoose, A.; Magnone, H.; Seehra, M.S.; Bonevich, J. Bulk to nanoscale magnetism and exchange bias in CuO nanoparticles. Phys. Rev. B 2001, 64, 174420. [Google Scholar] [CrossRef]
- Quickel, T.E.; Le, V.H.; Brezesinski, T.; Tolbert, S.H. On the Correlation between Nanoscale Structure and Magnetic Properties in Ordered Mesoporous Cobalt Ferrite (CoFe2O4) Thin Films. Nano Lett. 2010, 10, 2982–2988. [Google Scholar] [CrossRef]


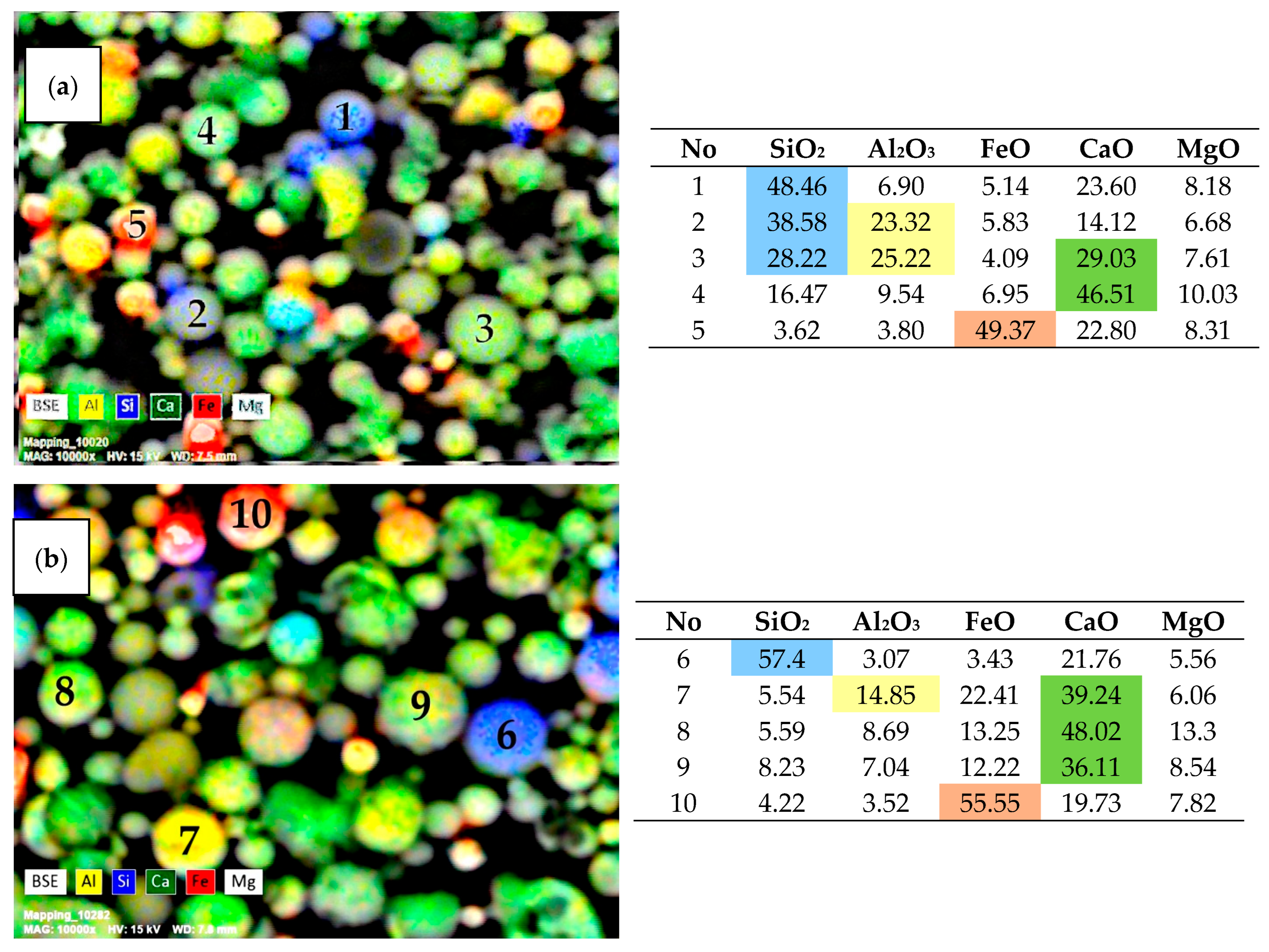
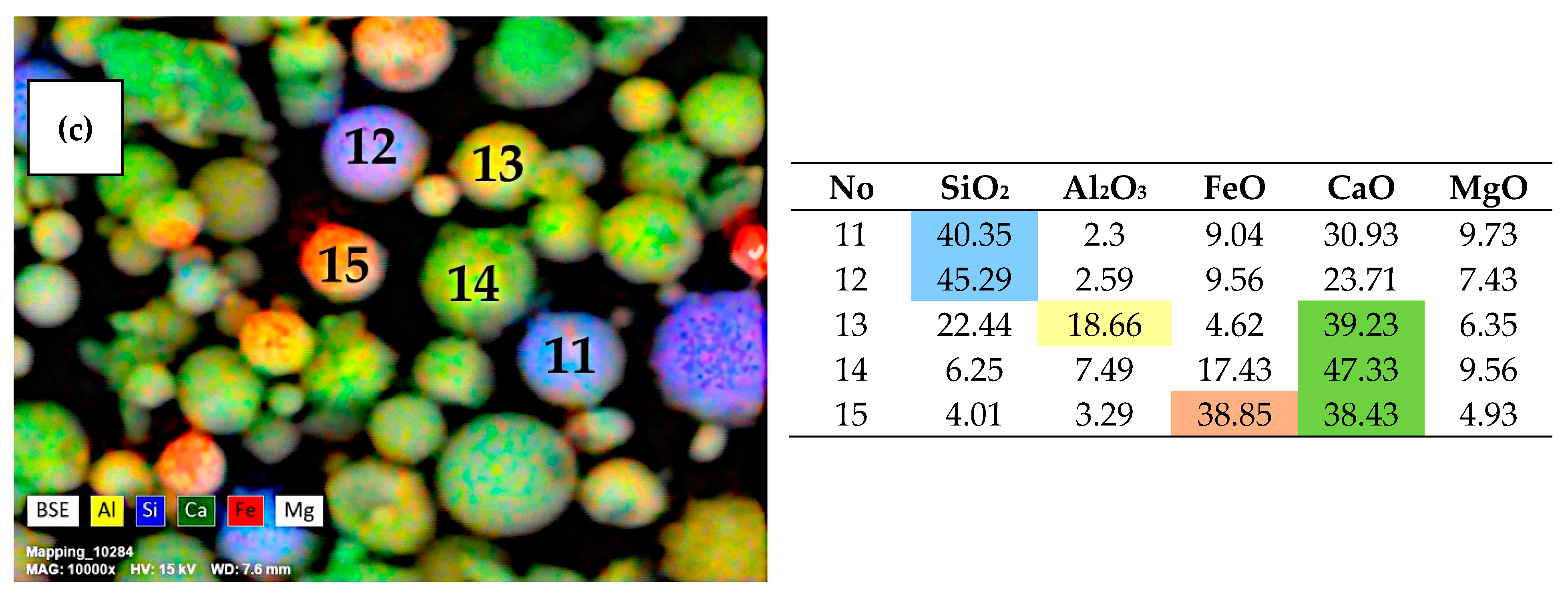


| Fraction | Bulk Density (g/cm3) | Particle Size Distribution (µm) | Chemical Composition (wt %) | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| dav | d10 | d50 | d90 | d99 | LOI | SiO2 | Al2O3 | Fe2O3 | CaO | MgO | Na2O | K2O | TiO2 | SO3 | ||
| FG178 | 0.89 | 1.6 | 0.5 | 1.3 | 3.1 | 5.4 | 5.30 | 13.98 | 9.17 | 13.96 | 38.50 | 8.20 | 0.32 | 0.18 | 0.32 | 9.60 |
| FG179 | 1.01 | 1.9 | 0.6 | 1.5 | 3.8 | 6.6 | 5.50 | 15.90 | 8.42 | 13.78 | 39.52 | 8.25 | 0.30 | 0.14 | 0.25 | 7.64 |
| FG180 | 1.12 | 2.5 | 0.7 | 2.0 | 4.9 | 8.4 | 10.80 | 15.88 | 7.99 | 13.96 | 38.60 | 7.82 | 0.27 | 0.19 | 0.10 | 4.62 |
| Fraction | Glass Phase | Ca4Al2Fe2O10 | Ca3Al2O6 | CaSO4 | CaCO3 | CaO | Ca(OH)2 | MgO | Quartz | Fe-Spinel |
|---|---|---|---|---|---|---|---|---|---|---|
| FG178 | 41.3 | 14.5 | 8.7 | 14.2 | 0.9 | 1.6 | 8.6 | 7.0 | 1.5 | 1.7 |
| FG179 | 40.7 | 12.9 | 10.2 | 11.2 | 0.7 | 2.0 | 11.5 | 6.8 | 2.0 | 2.0 |
| FG180 | 47.5 | 11.0 | 9.6 | 7.6 | 0.8 | 2.2 | 10.4 | 6.2 | 2.5 | 2.2 |
| Fraction | FG178 | FG179 | FG180 | ||
|---|---|---|---|---|---|
| TG data | |||||
| Mass loss, wt % | Dehydration | 40–244 °C | 0.47 | 0.50 | 0.46 |
| Ca(OH)2 = CaO + H2O | 244–437 °C | 2.17 | 2.74 | 2.89 | |
| *** (Ca,Mg,Fe)CO3 = (Ca,Mg,Fe)O + CO2 | 437–810 °C | 1.48 | 2.07 | 2.27 | |
| * Δmtotal | 40–1100 °C | 4.32 | 5.51 | 5.83 | |
| DSC data | |||||
| Ca(OH)2 = CaO + H2O (endothermic effect) | Tstart, °C | 373 | 374 | 375 | |
| Tmax, °C | 403 | 408 | 410 | ||
| Tfin, °C | 415 | 420 | 422 | ||
| *** (Ca,Mg,Fe)CO3 = (Ca,Mg,Fe)O + CO2 (endothermic effect) | Tstart, °C | 544 | 554 | 564 | |
| ** Tmax, °C | 623/644 | 624/657 | 630/664 | ||
| Tfin, °C | 657 | 671 | 677 | ||
| Crystallization of a new phase (exothermic effect) | Tstart, °C | 764 | 778 | 772 | |
| ** Tmax, °C | 854/930 | 885/925 | 886/925 | ||
| Tfin, °C | 1002 | 997 | 1003 | ||
| δ | Hhf | Δ | W34–16 | A | ||
|---|---|---|---|---|---|---|
| FG178 | 0.445 | 457 | −0.68 | 1.45–1.59 | 0.22 | [Fe3+] |
| 0.40 | 392 | −0.04 | 0.87–2.97 | 0.26 | [Fe3+] | |
| 0.184 | 271 | 0 | 1.00 | 0.12 | (Fe3+) | |
| 0.251 | – | 0.82 | 0.67 | 0.25 | CaFe2O4 | |
| 0.285 | – | 1.58 | 0.60 | 0.15 | Ca2Fe2−xAlxO5 | |
| FG179 | 0.31 | 480 | −0.18 | 0.80–0.80 | 0.12 | [Fe3+] |
| 0.35 | 425 | 0 | 0.79–1.07 | 0.20 | [Fe3+] | |
| 0.29 | 283 | −0.37 | 0.53–2.83 | 0.21 | (Fe3+) | |
| 0.23 | – | 0.79 | 0.66 | 0.26 | CaFe2O4 | |
| 0.27 | – | 1.57 | 0.70 | 0.21 | Ca2Fe2−xAlxO5 | |
| FG180 | 0.301 | 480 | −0.20 | 0.43–0.43 | 0.10 | [Fe3+] |
| 0.407 | 422 | 0.12 | 0.81–1.49 | 0.21 | [Fe3+] | |
| 0.270 | 293 | 0.08 | 2.584 | 0.20 | (Fe3+) | |
| 0.27 | – | 0.92 | 0.93 | 0.28 | CaFe2O4 | |
| 0.28 | – | 1.49 | 0.77 | 0.21 | Ca2Fe2−xAlxO5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fomenko, E.V.; Knyazev, Y.V.; Akimochkina, G.V.; Semenov, S.V.; Yumashev, V.V.; Solovyov, L.A.; Anshits, N.N.; Bayukov, O.A.; Anshits, A.G. Mössbauer Research and Magnetic Properties of Dispersed Microspheres from High-Calcium Fly Ash. Magnetochemistry 2025, 11, 72. https://doi.org/10.3390/magnetochemistry11090072
Fomenko EV, Knyazev YV, Akimochkina GV, Semenov SV, Yumashev VV, Solovyov LA, Anshits NN, Bayukov OA, Anshits AG. Mössbauer Research and Magnetic Properties of Dispersed Microspheres from High-Calcium Fly Ash. Magnetochemistry. 2025; 11(9):72. https://doi.org/10.3390/magnetochemistry11090072
Chicago/Turabian StyleFomenko, Elena V., Yuriy V. Knyazev, Galina V. Akimochkina, Sergey V. Semenov, Vladimir V. Yumashev, Leonid A. Solovyov, Natalia N. Anshits, Oleg A. Bayukov, and Alexander G. Anshits. 2025. "Mössbauer Research and Magnetic Properties of Dispersed Microspheres from High-Calcium Fly Ash" Magnetochemistry 11, no. 9: 72. https://doi.org/10.3390/magnetochemistry11090072
APA StyleFomenko, E. V., Knyazev, Y. V., Akimochkina, G. V., Semenov, S. V., Yumashev, V. V., Solovyov, L. A., Anshits, N. N., Bayukov, O. A., & Anshits, A. G. (2025). Mössbauer Research and Magnetic Properties of Dispersed Microspheres from High-Calcium Fly Ash. Magnetochemistry, 11(9), 72. https://doi.org/10.3390/magnetochemistry11090072






