ß-Farnesene Exogenous Application as a Novel Damage Induction Model to Fast Explore the Effectiveness of Postharvest Strategies: The Case Study of the ‘Rocha’ Pear DOP
Abstract
:1. Introduction
2. Materials and Methods
2.1. Material
2.2. Natural Extracts Screening and Selection
2.3. Fruit Material and Extract Material
2.4. Preparation of the Pectin-Based Coating and Fruit Coating
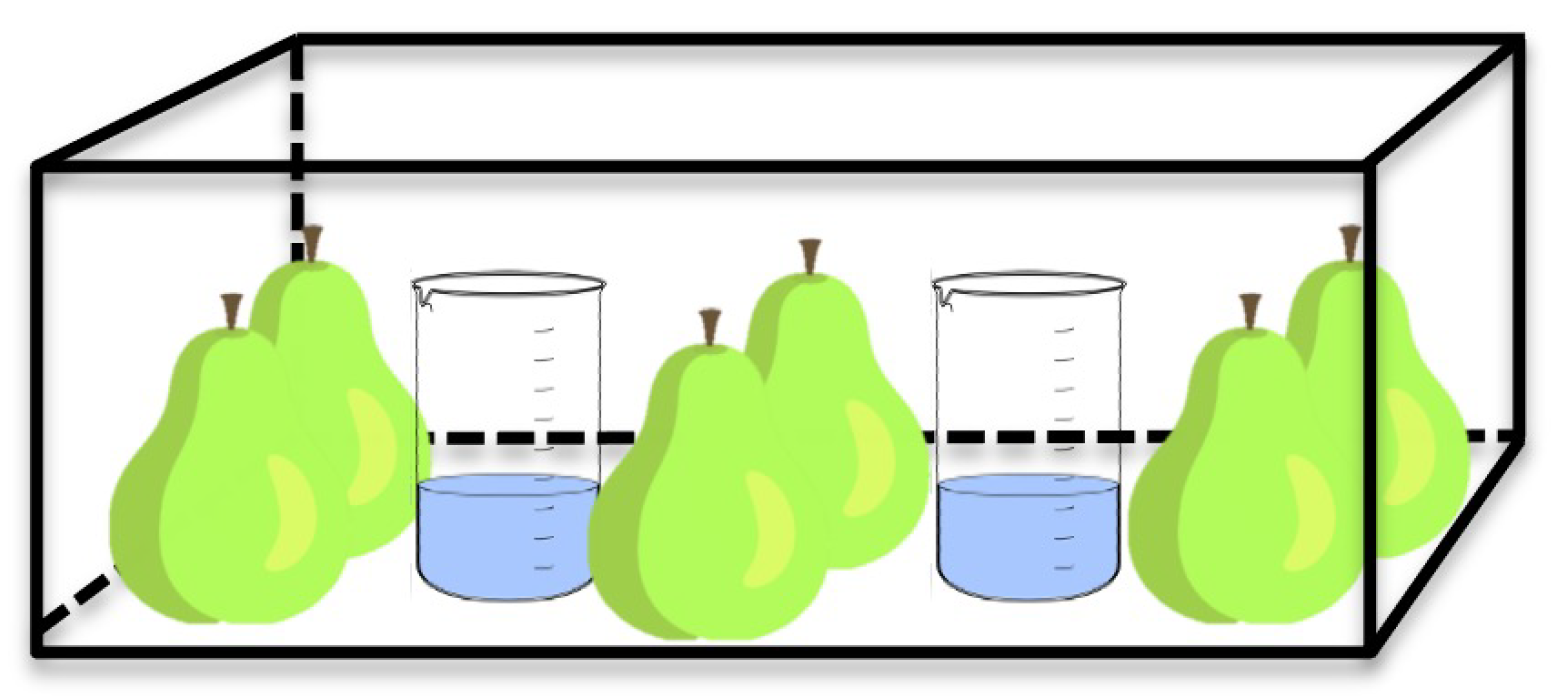
2.5. β-Farnesene-Atmosphere-Based Model
2.5.1. Model System Optimization
2.5.2. Model System Validation Using a Pilot Scale Storage of Pears
2.6. Determination of Pears Physicochemical Parameters
2.7. Antioxidant Capacity and Phenolic Content (Extraction and Quantification)
2.8. Determination of the PPO Enzyme Activity of Pears
2.9. Statistical Analysis
3. Results and Discussion
3.1. Selection of Natural-Based Extracts of Antioxidant Action
3.2. β-Farnesene-Atmohphere Model System Optimization and Evaluation
3.2.1. Visual Pear SC-Like Symptom Evaluation
3.2.2. Physicochemical Properties of Pears
Color
Firmness
Soluble Solids Content (SSC) and pH
SC Index
3.2.3. Antioxidant Activity of Pears
3.2.4. Oxidative Enzyme Activity of Pears
3.3. β-Farnesene-Atmosphere Model System Validation in Storage Conditions
3.3.1. Physicochemical Properties of Pears
Color
Firmness
SSC and PH
Scald Index
3.3.2. Antioxidant Activity of Pears
3.3.3. Oxidative Enzyme Activity of Pears
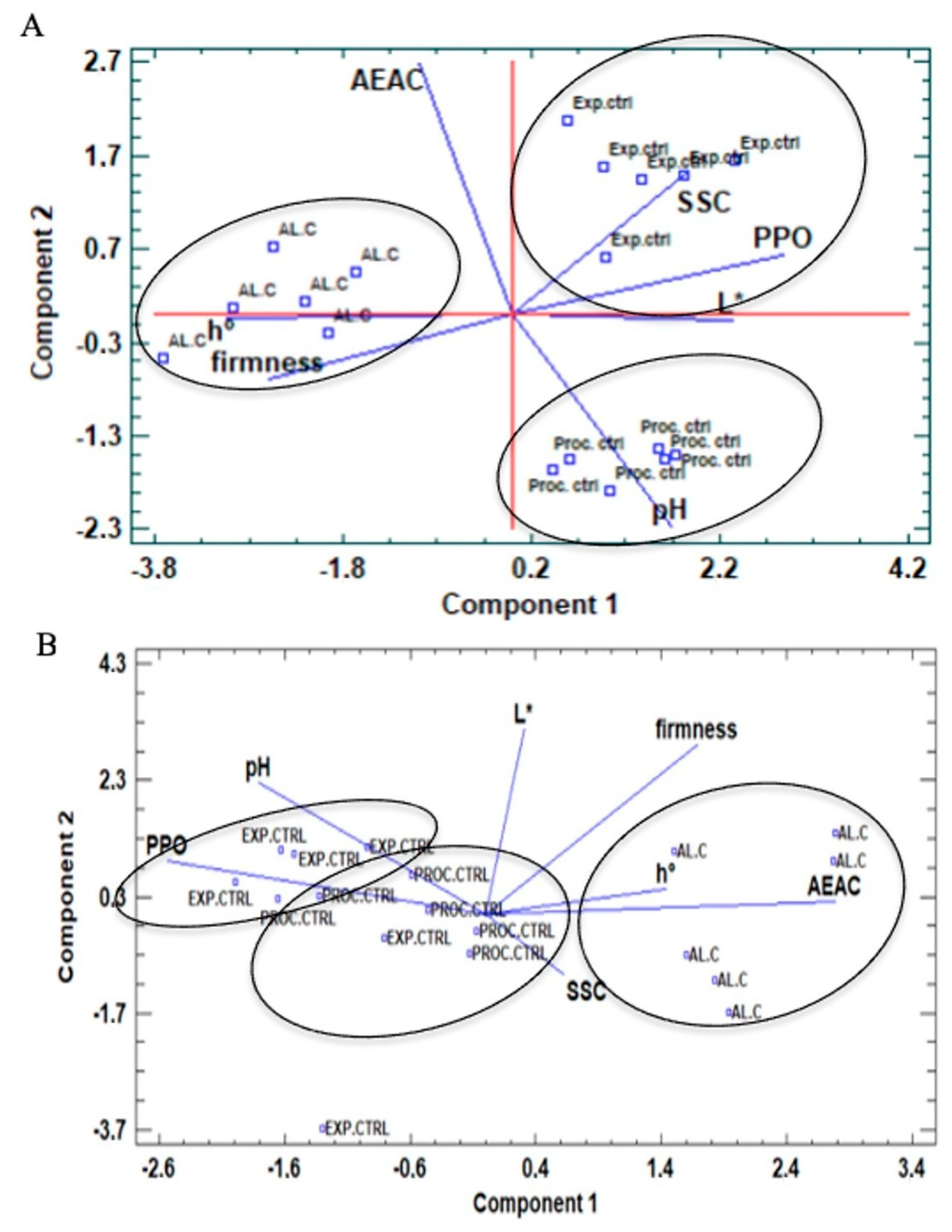
3.4. Principal Component Analysis of the Experiment
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- Saquet, A.A.; Almeida, D.P.F. Ripening physiology and biochemistry of ‘Rocha’ pear as affected by ethylene inhibition. Postharvest Biol. Technol. 2017, 125, 161–167. [Google Scholar] [CrossRef]
- Leisso, R.S.; Gapper, N.E.; Mattheis, J.P.; Sullivan, N.L.; Watkins, C.B.; Giovannoni, J.J.; Schaffer, R.J.; Johnston, J.W.; Hanrahan, I.; Hertog, M.L.A.T.M.; et al. Gene expression and metabolism preceding soft scald, a chilling injury of ‘Honeycrisp’ apple fruit. BMC Genom. 2016, 17, 798. [Google Scholar] [CrossRef] [Green Version]
- Deng, Z.; Jung, J.; Simonsen, J.; Wang, Y.; Zhao, Y. Cellulose Nanocrystal Reinforced Chitosan Coatings for Improving the Storability of Postharvest Pears Under Both Ambient and Cold Storages. J. Food Sci. 2017, 82, 453–462. [Google Scholar] [CrossRef] [PubMed]
- Karagiannis, E.; Tanou, G.; Scossa, F.; Samiotaki, M.; Michailidis, M.; Manioudaki, M.; Laurens, F.; Job, D.; Fernie, A.R.; Orsel, M.; et al. Systems-Based Approaches to Unravel Networks and Individual Elements Involved in Apple Superficial Scald. Front. Plant Sci. 2020, 11, 8. [Google Scholar] [CrossRef]
- Lurie, S.; Watkins, C.B. Superficial scald, its etiology and control. Postharvest Biol. Technol. 2012, 65, 44–60. [Google Scholar] [CrossRef]
- Fonseca, A.M.A.; Dias, C.; Amaro, A.L.; Isidoro, N.; Pintado, M.; Silvestre, A.J.D.; Rocha, S.M. The Impact of Plant-Based Coatings in “ROCHA” Pear Preservation during Cold Storage: A Metabolomic Approach. Foods 2020, 9, 1299. [Google Scholar] [CrossRef]
- Sarkar, D.; Ankolekar, C.; Greene, D.; Shetty, K. Natural preservatives for superficial scald reduction and enhancement of protective phenolic-linked antioxidant responses in apple during post-harvest storage. J. Food Sci. Technol. 2018, 55, 1767–1780. [Google Scholar] [CrossRef]
- Karagiannis, E.; Michailidis, M.; Tanou, G.; Samiotaki, M.; Karamanoli, K.; Avramidou, E.; Ganopoulos, I.; Madesis, P.; Molassiotis, A. Ethylene –dependent and –independent superficial scald resistance mechanisms in ‘Granny Smith’ apple fruit. Sci. Rep. 2018, 8, 11436. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pedreschi, R.; Franck, C.; Lammertyn, J.; Erban, A.; Kopka, J.; Hertog, M.; Verlinden, B.; Nicolaï, B. Metabolic profiling of ‘Conference’ pears under low oxygen stress. Postharvest Biol. Technol. 2009, 51, 123–130. [Google Scholar] [CrossRef]
- Li, Q.; Cheng, C.; Zhang, X.; Wang, C.; Yang, S. Preharvets bagging and postharvest calcium treatment affects superficial scald incidence and calcium nutrition during storage of ‘Chili’ pear (Pyrus bretschneideri) fruit. Postharvest Biol. Technol. 2020, 163, 111149. [Google Scholar] [CrossRef]
- Silva, F.J.P.; Gomes, M.H.; Fidalgo, F.; Rodrigues, J.A.; Almeida, D.P.F. Antioxidant properties and fruit quality during long-term storage of ‘Rocha’ pear: Effect of maturity and storage conditions. J. Food Qual. 2010, 33, 1–20. [Google Scholar] [CrossRef]
- Almeida, D.P.; Carvalho, R.; Dupille, E. Efficacy of 1-methylcyclopropene on the mitigation of storage disorders of “Rocha” pear under normal refrigerated and controlled atmospheres. Food Sci. Technol. Int. 2016, 22, 399–409. [Google Scholar] [CrossRef]
- Drzyzga, O. Diphenylamine and derivatives in the environment: A review. Chemosphere 2003, 53, 809–818. [Google Scholar] [CrossRef]
- Dias, C.; Amaro, A.L.; Salvador, Â.C.; Silvestre, A.J.D.; Rocha, S.M.; Isidoro, N.; Pintado, M. Strategies to preserve postharvest quality of horticultural crops and superficial scald control: From diphenylamine antioxidant usage to more recent approaches. Antioxidants 2020, 9, 356. [Google Scholar] [CrossRef]
- Rosenbloom, R.A.; Wang, W.; Zhao, Y. Delaying ripening of ‘Bartlett’ pears (Pyrus communis) during long-term simulated industrial cold storage: Mechanisms and validation of chitosan coatings with cellulose nanocrystals Pickering emulsions. LWT 2020, 122, 109053. [Google Scholar] [CrossRef]
- Panahirad, S.; Dadpour, M.; Peighambardoust, S.H.; Soltanzadeh, M.; Gullón, B.; Alirezalu, K.; Lorenzo, J.M. Applications of carboxymethyl cellulose- and pectin-based active edible coatings in preservation of fruits and vegetables: A review. Trends Food Sci. Technol. 2021, 110, 663–673. [Google Scholar] [CrossRef]
- Baraiya, N.S.; Rao, T.V.R.; Thakkar, V.R. Improvement of Postharvest Quality and Storability of Jamun Fruit (Syzygium cumini L. var. Paras) by Zein Coating Enriched with Antioxidants. Food Bioprocess Technol. 2015, 8, 2225–2234. [Google Scholar] [CrossRef]
- Poverenov, E.; Rutenberg, R.; Danino, S.; Horev, B.; Rodov, V. Gelatin-Chitosan Composite Films and Edible Coatings to Enhance the Quality of Food Products: Layer-by-Layer vs. Blended Formulations. Food Bioprocess Technol. 2014, 7, 3319–3327. [Google Scholar] [CrossRef]
- Gago, C.; Antão, R.; Dores, C.; Guerreiro, A.; Miguel, M.G.; Faleiro, M.L.; Figueiredo, A.C.; Antunes, M.D. The Effect of Nanocoatings Enriched with Essential Oils on “Rocha” Pear Long Storage. Foods 2020, 9, 240. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kähkönen, M.P.; Hopia, A.I.; Vuorela, H.J.; Rauha, J.P.; Pihlaja, K.; Kujala, T.S.; Heinonen, M. Antioxidant activity of plant extracts containing phenolic compounds. J. Agric. Food Chem. 1999, 47, 3954–3962. [Google Scholar] [CrossRef]
- Sharma, S.; Rao, T.V.R. Xanthan gum based edible coating enriched with cinnamic acid prevents browning and extends the shelf-life of fresh-cut pears. LWT-Food Sci. Technol. 2015, 62, 791–800. [Google Scholar] [CrossRef]
- Brooks, C.; Cooley, J.; Fisher, D. Nature and control of apple scald. J. Agric. Res. 1919, XVIII, 211–241. [Google Scholar]
- Bauchot, A.D.; Reid, S.J.; Ross, G.S.; Burmeister, D.M. Induction of apple scald by anaerobiosis has similar characteristics to naturally occurring superficial scald in ‘Granny Smith’ apple fruit. Postharvest Biol. Technol. 1999, 16, 9–14. [Google Scholar] [CrossRef]
- Dilley, D.R.; Dedolph, R.R.; Maclean, D.C.; Dewey, D.H. Apple Scald Induction by Anaerobiosis. Nature 1963, 200, 1229–1230. [Google Scholar] [CrossRef]
- Jaoui, M.; Lewandowski, M.; Offenberg, J.H.; Docherty, K.S.; Kleindienst, T.E. Ozonolysis of α/β-farnesene mixture: Analysis of gas-phase and particulate reaction products. Atmos. Environ. 2017, 169, 175–192. [Google Scholar] [CrossRef]
- Kumar, D.; Suryavanshi, P.; Padalia, R.C.; Chauhan, A.; Venkatesha, K.T.; Tiwari, A.K.; Singh, V.R.; Singh, S.; Upadhyay, R.K. Evaluation of harvesting time and standardization of distillation duration for higher essential oil content and quality in German chamomile (Chamomilla recutita L.). J. Spices Arom. Crops. 2020, 29, 140–147. [Google Scholar] [CrossRef]
- Jaoui, M.; Lewandowski, M.; Docherty, K.S.; Corse, E.W.; Lonneman, W.A.; Offenberg, J.H.; Kleindienst, T.E. Photooxidation of farnesene mixtures in the presence of NOx: Analysis of reaction products and their implication to ambient PM2.5. Atmos. Environ. 2016, 130, 190–201. [Google Scholar] [CrossRef]
- Dias, C.; Fonseca, A.M.A.; Amaro, A.L.; Vilas-Boas, A.A.; Oliveira, A.; Santos, S.A.O.; Silvestre, A.J.D.; Rocha, S.M.; Isidoro, N.; Pintado, M. Natural-Based Antioxidant Extracts as Potential Mitigators of Fruit Browning. Antioxidants 2020, 9, 715. [Google Scholar] [CrossRef] [PubMed]
- Oms-Oliu, G.; Soliva-Fortuny, R.; Martín-Belloso, O. Edible coatings with antibrowning agents to maintain sensory quality and antioxidant properties of fresh-cut pears. Postharvest Biol. Technol. 2008, 50, 87–94. [Google Scholar] [CrossRef]
- Lindo-García, V.; Giné-Bordonaba, J.; Leclerc, C.; Ubach, D.; Larrigaudière, C. The relationship between ethylene- and oxidative-related markers at harvest with the susceptibility of pears to develop superficial scald. Postharvest Biol. Technol. 2020, 163, 111135. [Google Scholar] [CrossRef]
- Salta, J.; Martins, A.; Santos, R.G.; Neng, N.R.; Nogueira, J.M.F.; Justino, J.; Rauter, A.P. Phenolic composition and antioxidant activity of Rocha pear and other pear cultivars—A comparative study. J. Funct. Foods 2010, 2, 153–157. [Google Scholar] [CrossRef]
- Gião, M.S.; González-Sanjosé, M.L.; Rivero-Pérez, M.D.; Pereira, C.I.; Pintado, M.E.; Malcata, X.F. Infusions of Portuguese medicinal plants: Dependence of final antioxidant capacity and phenol content on extraction features. J. Sci. Food Agric. 2007, 87, 2638–2647. [Google Scholar] [CrossRef] [PubMed]
- Galeazzi, M.A.M.; Sgarbieri, V.C. Substrate Specificity and Inhibition of Polyphenoloxidase (PPO) From a Dwarf Variety of Banana (Muss Cavendishii, L.). J. Food Sci. 1981, 46, 1404–1406. [Google Scholar] [CrossRef]
- Gatto, M.A.; Sergio, L.; Ippolito, A.; Di Venere, D. Phenolic extracts from wild edible plants to control postharvest diseases of sweet cherry fruit. Postharvest Biol. Technol. 2016, 120, 180–187. [Google Scholar] [CrossRef]
- Tenuta, M.C.; Deguin, B.; Loizzo, M.R.; Dugay, A.; Acquaviva, R.; Malfa, G.A.; Bonesi, M.; Bouzidi, C.; Tundis, R. Contribution of Flavonoids and Iridoids to the Hypoglycaemic, Antioxidant, and Nitric Oxide (NO) Inhibitory Activities of Arbutus unedo L. Antioxidants. 2020, 9, 184. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malheiro, R.; Sá, O.; Pereira, E.; Aguiar, C.; Baptista, P.; Pereira, J.A. Arbutus unedo L. leaves as source of phytochemicals with bioactive properties. Ind. Crops Prod. 2012, 37, 473–478. [Google Scholar] [CrossRef]
- Du, G.; Zhu, Y.; Wang, X.; Zhang, J.; Tian, C.; Liu, L.; Meng, Y.; Guo, Y. Phenolic composition of apple products and by-products based on cold pressing technology. J. Food Sci. Technol. 2019, 56, 1389–1397. [Google Scholar] [CrossRef]
- The Good Scents Company. (E)-Beta-Farnesene. Available online: www.thegoodscentscompany.com/data/rw1053891.html (accessed on 20 August 2021).
- Wang, Q.; Liu, H.; Zhang, M.; Liu, S.; Hao, Y.; Zhang, Y. MdMYC2 and MdERF3 Positively Co-Regulate α-Farnesene Biosynthesis in Apple. Front. Plant Sci. 2020, 11, 512844. [Google Scholar] [CrossRef]
- Guerreiro, A.C.; Gago, C.M.L.; Faleiro, M.L.; Miguel, M.G.C.; Antunes, M.D.C. The use of polysaccharide-based edible coatings enriched with essential oils to improve shelf-life of strawberries. Postharvest Biol. Technol. 2015, 110, 51–60. [Google Scholar] [CrossRef]
- Gago, C.M.L.; Miguel, M.G.; Cavaco, A.M.; Almeida, D.P.F.; Antunes, M.D.C. Combined effect of temperature and controlled atmosphere on storage and shelf-life of ‘Rocha’ pear treated with 1-methylcyclopropene. Food Sci. Technol. Int. 2015, 21, 94–103. [Google Scholar] [CrossRef]
- Hussain, P.R.; Meena, R.S.; Dar, M.A.; Wani, A.M. Carboxymethyl Cellulose Coating and Low-Dose Gamma Irradiation Improves Storage Quality and Shelf Life of Pear (Pyrus Communis L., Cv. Bartlett/William). J. Food Sci. 2010, 75, M586–M596. [Google Scholar] [CrossRef]
- Ali, U.; Kanwar, S.; Yadav, K.; Basu, S.; Mazumder, K. Effect of arabinoxylan and β-glucan stearic acid ester coatings on post-harvest quality of apple (Royal Delicious). Carbohydr. Polym. 2019, 209, 338–349. [Google Scholar] [CrossRef]
- Matabura, V.V. Modelling of firmness variability of Jonagold apple during postharvest storage. J. Food Sci. Technol. 2021, 89, 1–12. [Google Scholar] [CrossRef]
- Baraiya, N.S.; Rao, T.V.R.; Gol, N.B. Influence of Polysaccharide-Based Edible Coatings on the Shelf Life and Nutritional Quality of Tomato Fruit. Glob. Sci. Books 2012, 6, 22–27. [Google Scholar]
- Tesfay, S.Z.; Magwaza, L.S. Evaluating the efficacy of moringa leaf extract, chitosan and carboxymethyl cellulose as edible coatings for enhancing quality and extending postharvest life of avocado (Persea americana Mill.) fruit. Food Packag. Shelf Life 2017, 11, 40–48. [Google Scholar] [CrossRef]
- Yaman, Ö.; Bayoιndιrlι, L. Effects of an Edible Coating and Cold Storage on Shelf-life and Quality of Cherries. LWT-Food Sci. Technol. 2002, 35, 146–150. [Google Scholar] [CrossRef]
- Torres, C.A.; Sepulveda, G.; Mejía, N.; Defilippi, B.G.; Larrigaudière, C. Understanding the key preharvest factors determining ‘Packham’s Triumph’ pear heterogeneity and impact in superficial scald development and control. Postharvest Biol. Technol. 2021, 172, 1111399. [Google Scholar] [CrossRef]
- Ding, R.; Du, B.; Zhang, Y. Conjugated trienols and programmed cell death are more closely related to superficial scald than reactive oxygen species in apple fruit stored at low temperature. Sci. Hortic. 2019, 246, 597–603. [Google Scholar] [CrossRef]
- Correa-Betanzo, J.; Jacob, J.K.; Perez-Perez, C.; Paliyath, G. Effect of a sodium caseinate edible coating on berry cactus fruit (Myrtillocactus geometrizans) phytochemicals. Food Res. Int. 2011, 44, 1897–1904. [Google Scholar] [CrossRef]
- Deng, Y.; Wu, Y.; Li, Y. Effects of high O2 levels on post-harvest quality and shelf life of table grapes during long-term storage. Eur. Food Res. Technol. 2005, 221, 392–397. [Google Scholar] [CrossRef]
- Xiao, C.; Zhu, L.; Luo, W.; Song, X.; Deng, Y. Combined action of pure oxygen pretreatment and chitosan coating incorporated with rosemary extracts on the quality of fresh-cut pears. Food Chem. 2010, 121, 1003–1009. [Google Scholar] [CrossRef]
- Pastor, C.; Sánchez-González, L.; Marcilla, A.; Chiralt, A.; Cháfer, M.; González-Martínez, C. Quality and safety of table grapes coated with hydroxypropylmethylcellulose edible coatings containing propolis extract. Postharvest Biol. Technol. 2011, 60, 64–70. [Google Scholar] [CrossRef]
- Jung, J.; Deng, Z.; Zhao, Y. Mechanisms and performance of cellulose nanocrystals Pickering emulsion chitosan coatings for reducing ethylene production and physiological disorders in postharvest ‘Bartlett’ pears (Pyrus communis L.) during cold storage. Food Chem. 2020, 309, 125693. [Google Scholar] [CrossRef]
- Kowalczyk, D.; Kordowska-Wiater, M.; Zięba, E.; Baraniak, B. Effect of carboxymethylcellulose/candelilla wax coating containing potassium sorbate on microbiological and physicochemical attributes of pears. Sci. Hortic. 2017, 218, 326–333. [Google Scholar] [CrossRef]
- Leja, M.; Mareczek, A.; Ben, J. Antioxidant properties of two apple cultivars during long-term storage. Food Chem. 2003, 80, 303–307. [Google Scholar] [CrossRef]
- Ghasemnezhad, M.; Zareh, S.; Rassa, M.; Sajedi, R.H. Effect of chitosan coating on maintenance of aril quality, microbial population and PPO activity of pomegranate (Punica granatum L. cv. Tarom) at cold storage temperature. J. Sci. Food Agric. 2013, 93, 368–374. [Google Scholar] [CrossRef] [PubMed]
- Kou, X.; Wu, M.; Li, L.; Wang, S.; Xue, Z.; Liu, B.; Fei, Y. Effects of CaCl2 dipping and pullulan coating on the development of brown spot on “Huangguan” pears during cold storage. Postharvest Biol. Technol. 2015, 99, 63–72. [Google Scholar] [CrossRef]


| Quality Parameters | 0 d | 15 d | ||
|---|---|---|---|---|
| Lightness (L*) | − | Experimental control | 64.58 ± 0.95 a | 71.72 ± 2.27 A |
| Procedural control | 63.21 ± 2.45 a | 67.38 ± 3.33 B | ||
| AL.C | 66.13 ± 5.20 a | 68.87 ± 1.36 B | ||
| + | Experimental control | 60.73 ± 2.91 a | 79.47 ± 3.36 C | |
| Procedural control | 65.64 ± 1.97 a | 72.46 ± 3.26 A | ||
| AL.C | 64.66 ± 5.57 a | 67.87 ± 3.36 B | ||
| Hue angle (h°) | − | Experimental control | 106.74 ± 3.07 a | 87.20 ± 0.80 A |
| Procedural control | 103.91 ± 3.30 ab | 93.81 ± 1.50 C | ||
| AL.C | 103.83 ± 3.42 ab | 97.76 ± 4.39 D | ||
| + | Experimental control | 104.97 ± 4.66 ab | 82.88 ± 1.45 E | |
| Procedural control | 105.35 ± 2.47 ab | 88.03 ± 0.88 A | ||
| AL.C | 102.21 ± 1.80 b | 103.82 ± 4.40 B | ||
| Firmness (N) | − | Experimental control | 33.26 ± 1.67 a | 6.28 ± 0.29 A |
| Procedural control | 34.24 ± 1.77 a | 8.04 ± 0.39 B | ||
| AL.C | 35.90 ± 5.30 a | 11.58 ± 2.26 C | ||
| + | Experimental control | 36.40 ± 3.24 a | 6.38 ± 0.78 A | |
| Procedural control | 33.85 ± 1.67 a | 7.65 ± 0.39 AB | ||
| AL.C | 36.59 ± 3.04 a | 9.81 ± 1.47 D | ||
| SSC (%) | − | Experimental control | 12.13 ± 1.26 a | 14.15 ± 0.59 AC |
| Procedural control | 12.42 ± 0.43 a | 13.62 ± 0.45 AB | ||
| AL.C | 13.85 ± 0.55 b | 13.38 ± 0.37 B | ||
| + | Experimental control | 13.42 ± 0.45 b | 14.27 ± 0.60 C | |
| Procedural control | 12.45 ± 0.31 a | 13.40 ± 0.32 B | ||
| AL.C | 12.52 ± 0.38 a | 13.02 ± 0.74 B | ||
| pH | − | Experimental control | 5.66 ± 0.17 ac | 4.79 ± 0.05 A |
| Procedural control | 5.67 ± 0.22 ac | 5.48 ± 0.16 B | ||
| AL.C | 5.88 ± 0.17 b | 5.14 ± 0.24 C | ||
| + | Experimental control | 5.96 ± 0.15 b | 4.79 ± 0.17 A | |
| Procedural control | 5.82 ± 0.13 ab | 5.17 ± 0.09 C | ||
| AL.C | 5.60 ± 0.10 c | 4.72 ± 0.10 A | ||
| SC incidence (%) | − | Experimental control | 0 | 0 |
| Procedural control | 0 | 0 | ||
| AL.C | 0 | 0 | ||
| + | Experimental control | 0 | 100 | |
| Procedural control | 0 | 83.30 | ||
| AL.C | 0 | 16.67 | ||
| AEAC (g kg−1) | − | Experimental control | 0.64 ± 0.08 a | 0.56 ± 0.02 A |
| Procedural control | 0.61 ± 0.06 a | 0.19 ± 0.04 B | ||
| AL.C | 0.67 ± 0.09 a | 0.60 ± 0.01 C | ||
| + | Experimental control | 0.60 ± 0.10 a | 0.69 ± 0.01 D | |
| Procedural control | 0.62 ± 0.04 a | 0.23 ± 0.03 B | ||
| AL.C | 0.66 ± 0.03 a | 0.62 ± 0.04 C | ||
| PPO (U.mgprotein−1) | − | Experimental control | 2.95 ± 0.66 a | 3.11 ± 0.43 A |
| Procedural control | 2.72 ± 0.34 ac | 2.75 ± 0.38 AC | ||
| AL.C | 3.52 ± 0.48 b | 2.28 ± 0.34 B | ||
| + | Experimental control | 2.64 ± 0.45 ac | 2.95 ± 0.43 AC | |
| Procedural control | 2.41 ± 0.11 c | 2.59 ± 0.23 BC | ||
| AL.C | 2.67 ± 0.42 ac | 1.67 ± 0.19 D |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dias, C.; Amaro, A.; Fonseca, A.; Ferrante, A.; Silvestre, A.; Rocha, S.M.; Isidoro, N.; Pintado, M. ß-Farnesene Exogenous Application as a Novel Damage Induction Model to Fast Explore the Effectiveness of Postharvest Strategies: The Case Study of the ‘Rocha’ Pear DOP. Horticulturae 2022, 8, 93. https://doi.org/10.3390/horticulturae8020093
Dias C, Amaro A, Fonseca A, Ferrante A, Silvestre A, Rocha SM, Isidoro N, Pintado M. ß-Farnesene Exogenous Application as a Novel Damage Induction Model to Fast Explore the Effectiveness of Postharvest Strategies: The Case Study of the ‘Rocha’ Pear DOP. Horticulturae. 2022; 8(2):93. https://doi.org/10.3390/horticulturae8020093
Chicago/Turabian StyleDias, Cindy, Ana Amaro, Alexandre Fonseca, António Ferrante, Armando Silvestre, Sílvia M. Rocha, Nélson Isidoro, and Manuela Pintado. 2022. "ß-Farnesene Exogenous Application as a Novel Damage Induction Model to Fast Explore the Effectiveness of Postharvest Strategies: The Case Study of the ‘Rocha’ Pear DOP" Horticulturae 8, no. 2: 93. https://doi.org/10.3390/horticulturae8020093
APA StyleDias, C., Amaro, A., Fonseca, A., Ferrante, A., Silvestre, A., Rocha, S. M., Isidoro, N., & Pintado, M. (2022). ß-Farnesene Exogenous Application as a Novel Damage Induction Model to Fast Explore the Effectiveness of Postharvest Strategies: The Case Study of the ‘Rocha’ Pear DOP. Horticulturae, 8(2), 93. https://doi.org/10.3390/horticulturae8020093












