Optimization of Ultrasound-Assisted Extraction (UAE) for Simultaneous Determination of Individual Phenolic Compounds in 15 Dried Edible Flowers
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Plant Material
2.3. Ultrasound-Assisted Extraction (UAE)
2.4. Analysis of the Phenolic Compounds by UPLC-PDA
2.5. Experimental Design
2.6. Kinetics Study
2.7. Method Validation
3. Result and Discussion
3.1. Determination of Individual Phenolic Compounds
3.2. Solvent Screening
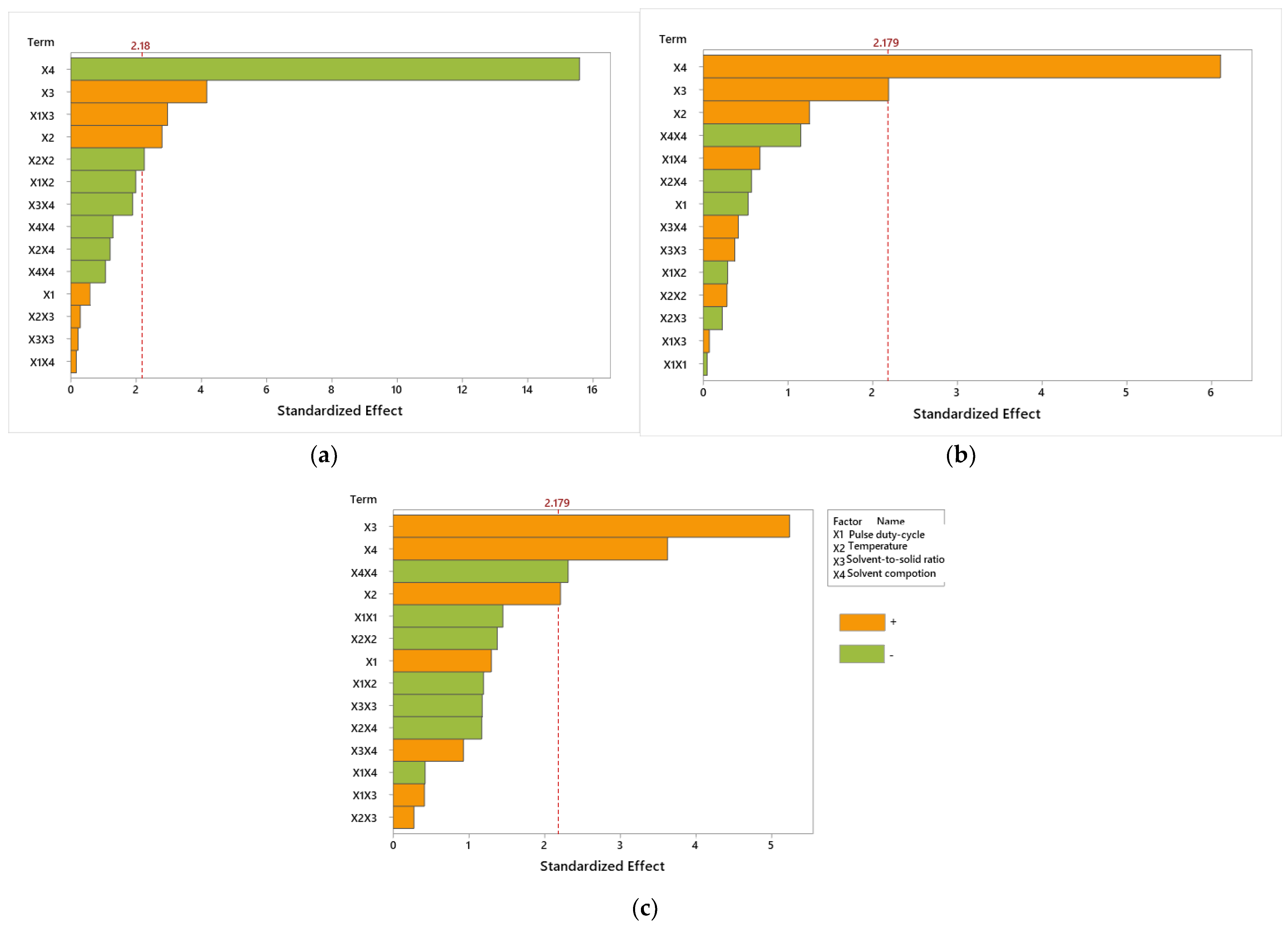
3.3. Optimization of UAE Method
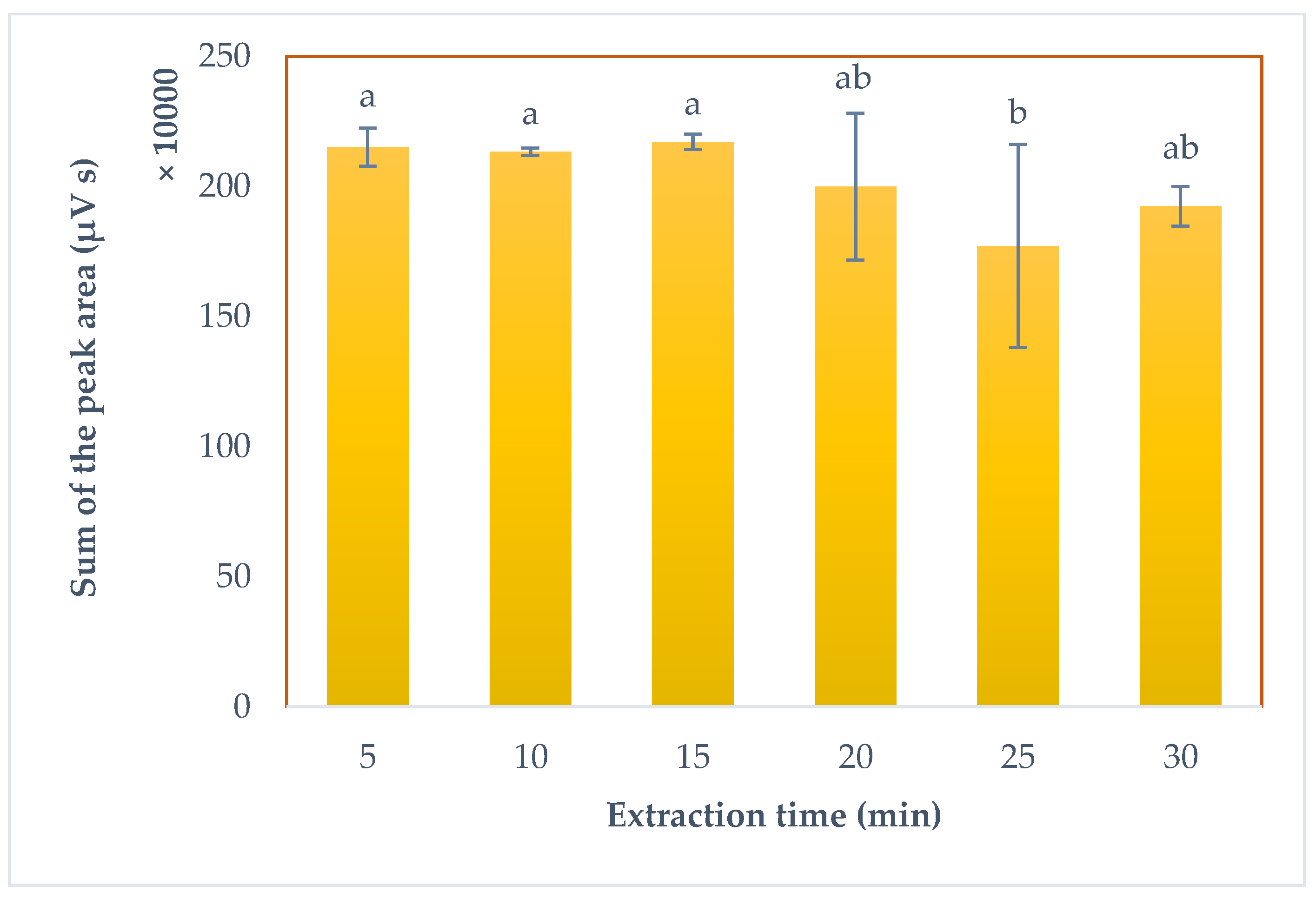
3.4. Assessment of the Extraction Time
3.5. Precision and Accuracy
3.6. Real Sample Application
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Takahashi, J.A.; Rezende, F.A.G.G.; Moura, M.A.F.; Dominguete, L.C.B.; Sande, D. Edible Flowers: Bioactive Profile and Its Potential to Be Used in Food Development. Food Res. Int. 2020, 129, 108868. [Google Scholar] [CrossRef] [PubMed]
- Etheridge, C.J.; Derbyshire, E. Herbal Infusions and Health: A Review of Findings from Human Studies, Mechanisms and Future Research Directions. Nutr. Food Sci. 2020, 50, 969–985. [Google Scholar] [CrossRef]
- Chen, N.H.; Wei, S. Factors Influencing Consumers’ Attitudes towards the Consumption of Edible Flowers. Food Qual. Prefer. 2017, 56, 93–100. [Google Scholar] [CrossRef]
- Lu, B.; Li, M.; Yin, R. Phytochemical Content, Health Benefits, and Toxicology of Common Edible Flowers: A Review (2000–2015). Crit. Rev. Food Sci. Nutr. 2016, 56, S130–S148. [Google Scholar] [CrossRef] [PubMed]
- Pires, T.C.S.P.; Dias, M.I.; Barros, L.; Calhelha, R.C.; Alves, M.J.; Oliveira, M.B.P.P.; Santos-Buelga, C.; Ferreira, I.C.F.R. Edible Flowers as Sources of Phenolic Compounds with Bioactive Potential. Food Res. Int. 2018, 105, 580–588. [Google Scholar] [CrossRef]
- Kumari, P.; Ujala; Bhargava, B. Phytochemicals from Edible Flowers: Opening a New Arena for Healthy Lifestyle. J. Funct. Foods 2021, 78, 104375. [Google Scholar] [CrossRef]
- Khan, M.K.; Abert-Vian, M.; Fabiano-Tixier, A.S.; Dangles, O.; Chemat, F. Ultrasound-Assisted Extraction of Polyphenols (Flavanone Glycosides) from Orange (Citrus Sinensis L.) Peel. Food Chem. 2010, 119, 851–858. [Google Scholar] [CrossRef]
- Pinela, J.; Prieto, M.A.; Pereira, E.; Jabeur, I.; Barreiro, M.F.; Barros, L.; Ferreira, I.C.F.R. Optimization of Heat- and Ultrasound-Assisted Extraction of Anthocyanins from Hibiscus Sabdariffa Calyces for Natural Food Colorants. Food Chem. 2019, 275, 309–321. [Google Scholar] [CrossRef]
- Setyaningsih, W.; Saputro, I.E.; Palma, M.; Barroso, C.G. Stability of 40 Phenolic Compounds during Ultrasound-Assisted Extractions (UAE). In Proceedings of the AIP Conference Proceedings, Yogyakarta, Indonesia, 11–13 November 2016; Volume 1755, p. 080009. [Google Scholar]
- Mehmood, A.; Ishaq, M.; Zhao, L.; Yaqoob, S.; Safdar, B.; Nadeem, M.; Munir, M.; Wang, C. Impact of Ultrasound and Conventional Extraction Techniques on Bioactive Compounds and Biological Activities of Blue Butterfly Pea Flower (Clitoria Ternatea L.). Ultrason. Sonochem. 2019, 51, 12–19. [Google Scholar] [CrossRef]
- Yu, J.; Lou, Q.; Zheng, X.; Cui, Z.; Fu, J. Sequential Combination of Microwave- and Ultrasound-Assisted Extraction of Total Flavonoids from Osmanthus Fragrans Lour. Flowers. Molecules 2017, 22, 2216. [Google Scholar] [CrossRef]
- Aourach, M.; González-De-peredo, A.V.; Vázquez-Espinosa, M.; Essalmani, H.; Palma, M.; Barbero, G.F. Optimization and Comparison of Ultrasound and Microwave-Assisted Extraction of Phenolic Compounds from Cotton-Lavender (Santolina Chamaecyparissus L.). Agronomy 2021, 11, 84. [Google Scholar] [CrossRef]
- Leliana, L.; Setyaningsih, W.; Palma, M.; Supriyadi, S.; Santoso, U. Optimization of Ultrasound-Assisted Extraction from Young Coconut Mesocarp in the Rapid Extraction of Phenolic Compounds and Antioxidant Activity. Agronomy 2022, 12, 2798. [Google Scholar] [CrossRef]
- Carrera, C.; Ruiz-Rodríguez, A.; Palma, M.; Barroso, C.G. Ultrasound Assisted Extraction of Phenolic Compounds from Grapes. Anal. Chim. Acta 2012, 732, 100–104. [Google Scholar] [CrossRef]
- Chemat, F.; Zill-E-Huma; Khan, M.K. Applications of Ultrasound in Food Technology: Processing, Preservation and Extraction. Ultrason. Sonochem. 2011, 18, 813–835. [Google Scholar] [CrossRef] [PubMed]
- Berger, P.D.; Maurer, R.E.; Celli, G.B. Experimental Design: With Applications in Management, Engineering and the Sciences, 2nd ed.; Springer International Publishing: Cham, Switzerland, 2018; pp. 1–639. ISBN 9783319645834. [Google Scholar]
- ICH. ICH Topic Q2 (R1) Validation of Analytical Procedures: Text and Methodology; European Medicines Agency: Amsterdam, The Netherlands, 1995. [Google Scholar]
- Lin, L.Z.; Harnly, J.M. Identification of the Phenolic Components of Chrysanthemum Flower (Chrysanthemum Morifolium Ramat). Food Chem. 2010, 120, 319–326. [Google Scholar] [CrossRef]
- Ramírez-Bolaños, S.; Pérez-Jiménez, J.; Díaz, S.; Robaina, L. A Potential of Banana Flower and Pseudo-Stem as Novel Ingredients Rich in Phenolic Compounds. Int. J. Food Sci. Technol. 2021, 56, 5601–5608. [Google Scholar] [CrossRef]
- Liu, F.; Wang, M.; Wang, M. Phenolic Compounds and Antioxidant Activities of Flowers, Leaves and Fruits of Five Crabapple Cultivars (Malus Mill. Species). Sci. Hortic. 2018, 235, 460–467. [Google Scholar] [CrossRef]
- Barros, L.; Dueñas, M.; Carvalho, A.M.; Ferreira, I.C.F.R.; Santos-Buelga, C. Characterization of Phenolic Compounds in Flowers of Wild Medicinal Plants from Northeastern Portugal. Food Chem. Toxicol. 2012, 50, 1576–1582. [Google Scholar] [CrossRef]
- Zardo, I.; de Espíndola Sobczyk, A.; Marczak, L.D.F.; Sarkis, J. Optimization of Ultrasound Assisted Extraction of Phenolic Compounds from Sunflower Seed Cake Using Response Surface Methodology. Waste Biomass Valorization 2019, 10, 33–44. [Google Scholar] [CrossRef]
- Kumar, K.; Srivastav, S.; Sharanagat, V.S. Ultrasound Assisted Extraction (UAE) of Bioactive Compounds from Fruit and Vegetable Processing by-Products: A Review. Ultrason. Sonochem. 2021, 70, 105325. [Google Scholar] [CrossRef] [PubMed]
- Dzah, C.S.; Duan, Y.; Zhang, H.; Wen, C.; Zhang, J.; Chen, G.; Ma, H. The Effects of Ultrasound Assisted Extraction on Yield, Antioxidant, Anticancer and Antimicrobial Activity of Polyphenol Extracts: A Review. Food Biosci. 2020, 35, 100547. [Google Scholar] [CrossRef]
- Fernández-Barbero, G.; Pinedo, C.; Espada-Bellido, E.; Ferreiro-González, M.; Carrera, C.; Palma, M.; García-Barroso, C. Optimization of Ultrasound-Assisted Extraction of Bioactive Compounds from Jabuticaba (Myrciaria Cauliflora) Fruit through a Box-Behnken Experimental Design. Food Sci. Technol. 2019, 39, 1018–1029. [Google Scholar] [CrossRef]
- Setyaningsih, W.; Saputro, I.E.; Carrera, C.A.; Palma, M. Optimisation of an Ultrasound-Assisted Extraction Method for the Simultaneous Determination of Phenolics in Rice Grains. Food Chem. 2019, 288, 221–227. [Google Scholar] [CrossRef]
- Espada-Bellido, E.; Ferreiro-González, M.; Carrera, C.; Palma, M.; Barroso, C.G.; Barbero, G.F. Optimization of the Ultrasound-Assisted Extraction of Anthocyanins and Total Phenolic Compounds in Mulberry (Morus Nigra) Pulp. Food Chem. 2017, 219, 23–32. [Google Scholar] [CrossRef]
- Brahmi, F.; Blando, F.; Sellami, R.; Mehdi, S.; De Bellis, L.; Negro, C.; Haddadi-Guemghar, H.; Madani, K.; Makhlouf-Boulekbache, L. Optimization of the Conditions for Ultrasound-Assisted Extraction of Phenolic Compounds from Opuntia Ficus-Indica [L.] Mill. Flowers and Comparison with Conventional Procedures. Ind. Crops Prod. 2022, 184, 114977. [Google Scholar] [CrossRef]
- Tao, Y.; Wu, D.; Zhang, Q.A.; Sun, D.W. Ultrasound-Assisted Extraction of Phenolics from Wine Lees: Modeling, Optimization and Stability of Extracts during Storage. Ultrason. Sonochem. 2014, 21, 706–715. [Google Scholar] [CrossRef] [PubMed]
- AOAC. Appendix F: Guidelines for Standard Method Performance Requirements; AOAC International: Rockville, MA, USA, 2016. [Google Scholar]
- Li, A.N.; Li, S.; Li, H.B.; Xu, D.P.; Xu, X.R.; Chen, F. Total Phenolic Contents and Antioxidant Capacities of 51 Edible and Wild Flowers. J. Funct. Foods 2014, 6, 319–330. [Google Scholar] [CrossRef]
- Yun, J.M.; Kang, D.W. Antioxidant Activity and Qualitative and Quantitative HPLC Analyses of Five Types of Apple Blossoms Prepared by Two Different Drying Methods. Korean J. Food Preserv. 2021, 28, 780–789. [Google Scholar] [CrossRef]
- Schmitzer, V.; Veberic, R.; Osterc, G.; Stampar, F. Color and Phenolic Content Changes during Flower Development in Groundcover Rose. J. Am. Soc. Hortic. Sci. 2010, 135, 195–202. [Google Scholar] [CrossRef]
| Run | Factors | Responses (%) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| X1 | X2 | X3 | X4 | Benzoic Acid Derivatives | Cinnamic Acid Derivatives | Flavonoids | ||||
| Observed | Error | Observed | Error | Observed | Error | |||||
| 1 | −1 | −1 | 0 | 0 | 58.71 | 5.11 | 71.01 | 1.11 | 73.29 | 3.22 |
| 2 | 1 | −1 | 0 | 0 | 71.45 | 1.83 | 77.91 | 0.42 | 89.37 | 2.68 |
| 3 | −1 | 1 | 0 | 0 | 73.50 | 1.59 | 85.81 | 3.73 | 87.44 | 2.96 |
| 4 | 1 | 1 | 0 | 0 | 72.09 | 4.74 | 85.57 | 3.17 | 89.61 | 2.40 |
| 5 | 0 | 0 | −1 | −1 | 79.17 | 1.00 | 54.48 | 18.34 | 71.94 | 0.95 |
| 6 | 0 | 0 | 1 | −1 | 100.00 | 4.94 | 69.98 | 23.90 | 89.43 | 7.19 |
| 7 | 0 | 0 | −1 | 1 | 50.17 | 8.41 | 74.23 | 11.97 | 71.68 | 8.12 |
| 8 | 0 | 0 | 1 | 1 | 57.47 | 1.59 | 100.00 | 4.79 | 100.00 | 1.01 |
| 9 | −1 | 0 | 0 | −1 | 86.65 | 0.71 | 55.70 | 8.66 | 79.05 | 7.91 |
| 10 | 1 | 0 | 0 | −1 | 84.32 | 2.68 | 35.68 | 23.71 | 73.75 | 7.88 |
| 11 | −1 | 0 | 0 | 1 | 55.04 | 2.95 | 99.61 | 15.28 | 97.57 | 11.02 |
| 12 | 1 | 0 | 0 | 1 | 53.96 | 2.44 | 96.15 | 2.36 | 87.36 | 2.70 |
| 13 | 0 | −1 | −1 | 0 | 63.54 | 0.02 | 68.27 | 1.93 | 78.39 | 5.82 |
| 14 | 0 | 1 | −1 | 0 | 65.84 | 3.53 | 79.56 | 1.12 | 82.24 | 2.92 |
| 15 | 0 | −1 | 1 | 0 | 72.73 | 2.34 | 86.56 | 1.45 | 91.10 | 1.15 |
| 16 | 0 | 1 | 1 | 0 | 77.11 | 0.94 | 92.30 | 0.90 | 98.16 | 0.95 |
| 17 | −1 | 0 | −1 | 0 | 77.05 | 6.46 | 61.37 | 11.86 | 71.67 | 5.49 |
| 18 | 1 | 0 | −1 | 0 | 66.22 | 5.01 | 80.25 | 10.66 | 80.95 | 4.08 |
| 19 | −1 | 0 | 1 | 0 | 67.89 | 3.57 | 71.05 | 15.72 | 84.80 | 6.82 |
| 20 | 1 | 0 | 1 | 0 | 78.12 | 4.90 | 91.72 | 3.07 | 98.90 | 1.16 |
| 21 | 0 | −1 | 0 | −1 | 78.19 | 1.64 | 36.10 | 15.66 | 65.55 | 6.00 |
| 22 | 0 | 1 | 0 | −1 | 88.66 | 0.99 | 50.14 | 14.63 | 81.99 | 2.35 |
| 23 | 0 | −1 | 0 | 1 | 53.37 | 2.95 | 96.31 | 3.29 | 87.65 | 1.19 |
| 24 | 0 | 1 | 0 | 1 | 55.23 | 3.66 | 96.32 | 1.24 | 90.48 | 1.29 |
| 25 | 0 | 0 | 0 | 0 | 73.45 | 0.74 | 67.82 | 12.08 | 94.24 | 2.19 |
| 26 | 0 | 0 | 0 | 0 | 74.11 | 0.15 | 88.21 | 14.37 | 93.40 | 1.28 |
| 27 | 0 | 0 | 0 | 0 | 74.44 | 0.59 | 86.90 | 12.67 | 89.03 | 3.46 |
| Phenolic Compounds | Low Range (0.5–10 ppm) | High Range (10–50 ppm) | LOD (ppm) | LOQ (ppm) | ||
|---|---|---|---|---|---|---|
| Linear Equation | R² | Linear Equation | R² | |||
| 2,4,6-Trihydroxybenzoic acid | 0.951 | 0.985 | 1.35 | 4.10 | ||
| Protocatechuic acid | 0.999 | 0.999 | 0.15 | 0.46 | ||
| Protocatechuic aldehyde | 0.999 | 0.991 | 0.14 | 0.44 | ||
| p-Hydroxybenzoic acid | 0.999 | 0.999 | 0.16 | 0.50 | ||
| Caffeic acid | 0.995 | 0.997 | 0.24 | 0.74 | ||
| Vanillic acid | 0.997 | 0.998 | 0.22 | 0.66 | ||
| Epicatechin | 0.985 | 0.995 | 0.53 | 1.61 | ||
| p-Coumaric | 0.993 | 0.998 | 0.36 | 1.10 | ||
| Ferulic acid | 0.992 | 0.995 | 0.38 | 1.15 | ||
| Quercetin-3-rutinose | 0.999 | 0.997 | 0.16 | 0.49 | ||
| Iso-ferulic acid | 0.997 | 0.992 | 0.26 | 0.78 | ||
| Quercetin 3 glucose | 0.997 | 0.992 | 0.22 | 0.66 | ||
| Phenolic Compounds | Precision CV (%) | Recovery (%) | |
|---|---|---|---|
| Repeatability | Intermediate Precision | ||
| 2,4,6-Trihydroxybenzoic acid | 0.57 | 1.32 | 91.94 ± 1.04 |
| Protocatechuic acid | 2.50 | 1.15 | 100.00 ± 0.00 |
| Protocatechuic aldehyde | 2.95 | 2.42 | 84.9 ± 13.10 |
| p-Hydroxybenzoic acid | 5.00 | 3.08 | 93.61 ± 11.06 |
| Caffeic acid | 1.50 | 1.86 | 83.82 ± 0.61 |
| Vanillic acid | 1.45 | 2.23 | 93.23 ± 11.72 |
| Epicatechin | 4.63 | 6.66 | 94.12 ± 10.18 |
| p-Coumaric | 0.61 | 1.23 | 84.41 ± 0.21 |
| Ferulic acid | 0.95 | 3.96 | 87.04 ± 11.88 |
| Quercetin-3-rutinose | 1.12 | 3.94 | 82.07 ± 1.49 |
| Iso-ferulic acid | 5.82 | 5.22 | 99.34 ± 1.14 |
| Quercetin 3-glucose | 4.11 | 4.17 | 82.01 ± 1.58 |
| Phenolic Compounds | Phenolic Compounds from Extracted Edible Flowers (µg g−1) | |||||
|---|---|---|---|---|---|---|
 Calendula officinalis |  Dianthus caryophyllus |  Lilium bulbiferum |  Chrysanthemum morifolium |  Osmanthus fragrans |  Prunus persica | |
| 2,4,6-Trihydroxybenzoic acid | 265.69 ± 2.86 | <LOD | <LOD | 317.51 ± 3.93 | 765.58 ± 13.17 | 700.66 ± 14.36 |
| Protocatechuic acid | <LOD | <LOD | 52.39 ± 2.56 | 30.63 ± 0.36 | 19.06 ± 1.90 * | 64.95 ± 2.14 |
| Protocatechuic aldehyde | <LOD | <LOD | <LOD | <LOD | 38.66 ± 2.95 | 32.81 ± 1.79 |
| p-Hydroxybenzoic acid | 36.42 ± 3.56 | 73.09 ± 0.34 | 61.4 ± 0.86 | <LOD | <LOD | 27.28 ± 1.95 * |
| Caffeic acid | 102.17 ± 2.81 | 148.47 ± 1.50 | 570.15 ± 11.47 | 73.05 ± 1.98 | 86.77 ± 1.36 | 70.74 ± 1.46 |
| Vanillic acid | 9.08 ± 1.26 * | 228.95 ± 2.17 | 29.64 ± 0.77 * | 4.77 ± 1.23 * | <LOD | 30.83 ± 1.55 * |
| Epicatechin | 302.72 ± 1.81 | <LOD | <LOD | <LOD | 409.78 ± 4.07 | <LOD |
| p-Coumaric | 8.78 ± 0.10 * | 300.52 ± 2.56 | 138.43 ± 3.76 | 28.82 ± 0.50 * | 291.68 ± 9.76 | 130.17 ± 15.01 |
| Ferulic acid | 11.06 ± 0.41 * | 10.13 ± 0.87 * | 95.49 ± 2.70 | <LOD | 376.01 ± 23.25 | 265.34 ± 21.61 |
| Quercetin-3-rutinose | 244.06 ± 0.48 | <LOD | 94.79 ± 2.29 | 485.21 ± 3.87 | <LOD | <LOD |
| Iso-ferulic acid | <LOD | <LOD | 15.77 ± 0.70 * | 519.58 ± 6.64 | 53.8 ±1.99 | <LOD |
| Quercetin-3-glucose | 2377.99 ± 139.00 | <LOD | <LOD | 951.39 ± 15.65 | <LOD | 1215.06 ± 118.79 |
| Total phenolic compounds | 3357.97 ± 148.89 | 761.16 ± 3.09 | 1058.06 ± 18.21 | 2410.97 ± 21.63 | 2041.33 ± 22.50 | 2537.84 ± 173.68 |
| Phenolic Compounds | Phenolic Compounds from Extracted Edible Flowers (µg g−1) | |||||
|---|---|---|---|---|---|---|
 Jasminum sambac |  Clitoria ternatea |  Rosa gallica (bud) |  Rose mengyin (bud) |  Malva sylvestris |  Hibiscus sabdariffa | |
| 2,4,6-Trihydroxybenzoic acid | 880.23 ± 8.33 | 641.88 ± 12.47 | 110.16 ± 10.49 * | 151.48 ± 11.66 * | 181.77 ± 6.68 * | <LOD |
| Protocatechuic acid | <LOD | 39.45 ± 1.75 | 46.97 ± 7.15 | 195.8 ± 4.59 | 31.72 ± 0.53 | <LOD |
| Protocatechuic aldehyde | <LOD | <LOD | <LOD | <LOD | <LOD | <LOD |
| p-Hydroxybenzoic acid | <LOD | <LOD | 12.51 ± 0.6 * | <LOD | 33.74 ± 2.11 | <LOD |
| Caffeic acid | <LOD | 23.92 ± 0.81 * | <LOD | <LOD | <LOD | 72.88 ± 1.11 |
| Vanillic acid | 5.19 ± 0.92 * | 22.62 ± 0.51 * | <LOD | <LOD | <LOD | 13.25 ± 1.27 * |
| Epicatechin | 63.69 ± 1.53 | <LOD | 182.49 ± 11 | <LOD | 89.69 ± 12.31 | 263.24 ± 11.02 |
| p-Coumaric | 17.07 ± 0.42 * | 16.82 ± 1.20 * | <LOD | <LOD | 460.57 ± 12.27 | 33.04 ± 0.59 * |
| Ferulic acid | <LOD | <LOD | <LOD | 41.47 ± 2.88 * | 445.62 ± 16.5 | <LOD |
| Quercetin-3-rutinose | 472.54 ± 14.76 | 492.29 ± 22.19 | 791.78 ± 30.82 | 1193.39 ± 125.49 | 396.41 ± 10.67 | 24.57 ± 0.95 * |
| Iso-ferulic acid | <LOD | <LOD | <LOD | <LOD | <LOD | <LOD |
| Quercetin-3-glucose | 639.9 ± 18.07 | 174.21 ± 3.61 | 1158.57 ± 41.22 | 2154.83 ± 160.92 | 381.36 ± 7.45 | <LOD |
| Total phenolic compounds | 2078.62 ± 35.95 | 1411.19 ± 37.14 | 2302.48 ± 64.81 | 3736.97 ± 265.48 | 2020.87 ± 47.12 | 406.98 ± 13.17 |
| Phenolic Compounds | Phenolic Compounds from Extracted Edible Flowers (µg g−1) | ||
|---|---|---|---|
 Chrysanthemum morifolium (bud) |  Malus sp. |  Paeonia suffruticosa (bud) | |
| 2,4,6-Trihydroxybenzoic acid | 1208.72 ± 7.6 | <LOD | 852.61 ± 38.92 |
| Protocatechuic acid | 37.18 ± 1.23 | <LOD | 65.64 ± 2.77 |
| Protocatechuic aldehyde | <LOD | <LOD | <LOD |
| p-Hydroxybenzoic acid | <LOD | 30.97 ± 2.72 | 161.74 ± 3.13 |
| Caffeic acid | <LOD | 111.26 ± 2.4 | <LOD |
| Vanillic acid | <LOD | <LOD | <LOD |
| Epicatechin | <LOD | <LOD | 1101.82 ± 18.81 |
| p-Coumaric | 24.27 ± 1.29 * | 155.89 ± 1.97 | 31.86 ± 0.97 * |
| Ferulic acid | 43.64 ± 1.57 * | 51.55 ± 2.09 * | 54.54 ± 0.55 |
| Quercetin-3-rutinose | 1035.07 ± 36.45 | 409.24 ± 20.7 | <LOD |
| Iso-ferulic acid | 1272.76 ± 33.89 | <LOD | <LOD |
| Quercetin-3-glucose | 2277.83 ± 50.29 | 189.22 ± 2.94 | 4261.23 ± 222.58 |
| Total phenolic compounds | 5899.48 ± 99.26 | 948.16 ± 26.42 | 6529.45 ± 191.95 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Briliantama, A.; Oktaviani, N.M.D.; Rahmawati, S.; Setyaningsih, W.; Palma, M. Optimization of Ultrasound-Assisted Extraction (UAE) for Simultaneous Determination of Individual Phenolic Compounds in 15 Dried Edible Flowers. Horticulturae 2022, 8, 1216. https://doi.org/10.3390/horticulturae8121216
Briliantama A, Oktaviani NMD, Rahmawati S, Setyaningsih W, Palma M. Optimization of Ultrasound-Assisted Extraction (UAE) for Simultaneous Determination of Individual Phenolic Compounds in 15 Dried Edible Flowers. Horticulturae. 2022; 8(12):1216. https://doi.org/10.3390/horticulturae8121216
Chicago/Turabian StyleBriliantama, Asadin, Nurul Mutmainah Diah Oktaviani, Sitti Rahmawati, Widiastuti Setyaningsih, and Miguel Palma. 2022. "Optimization of Ultrasound-Assisted Extraction (UAE) for Simultaneous Determination of Individual Phenolic Compounds in 15 Dried Edible Flowers" Horticulturae 8, no. 12: 1216. https://doi.org/10.3390/horticulturae8121216
APA StyleBriliantama, A., Oktaviani, N. M. D., Rahmawati, S., Setyaningsih, W., & Palma, M. (2022). Optimization of Ultrasound-Assisted Extraction (UAE) for Simultaneous Determination of Individual Phenolic Compounds in 15 Dried Edible Flowers. Horticulturae, 8(12), 1216. https://doi.org/10.3390/horticulturae8121216










