Firmness and Cell Wall Changes during Maturation of ‘Arbequina’ Olive Fruit: The Impact of Irrigation
Abstract
:| “Olea prima arborum omnium est”. |
| Lucius Iunius Moderatus, a.k.a. Columella |
| De Arboribus |
1. Introduction
2. Materials and Methods
2.1. Plant Material and Assessment of Fruit Firmness
2.2. Extraction, Fractionation and Analysis of Cell Wall Materials
2.3. Cell Wall-Related Enzyme Activities
2.4. Antioxidant Properties
2.5. Statistical Analysis
3. Results
3.1. Physical and Chemical Characteristics
3.2. Cell Wall Composition and Ripening-Related Changes
3.3. Cell Wall-Modifying Enzyme Activities
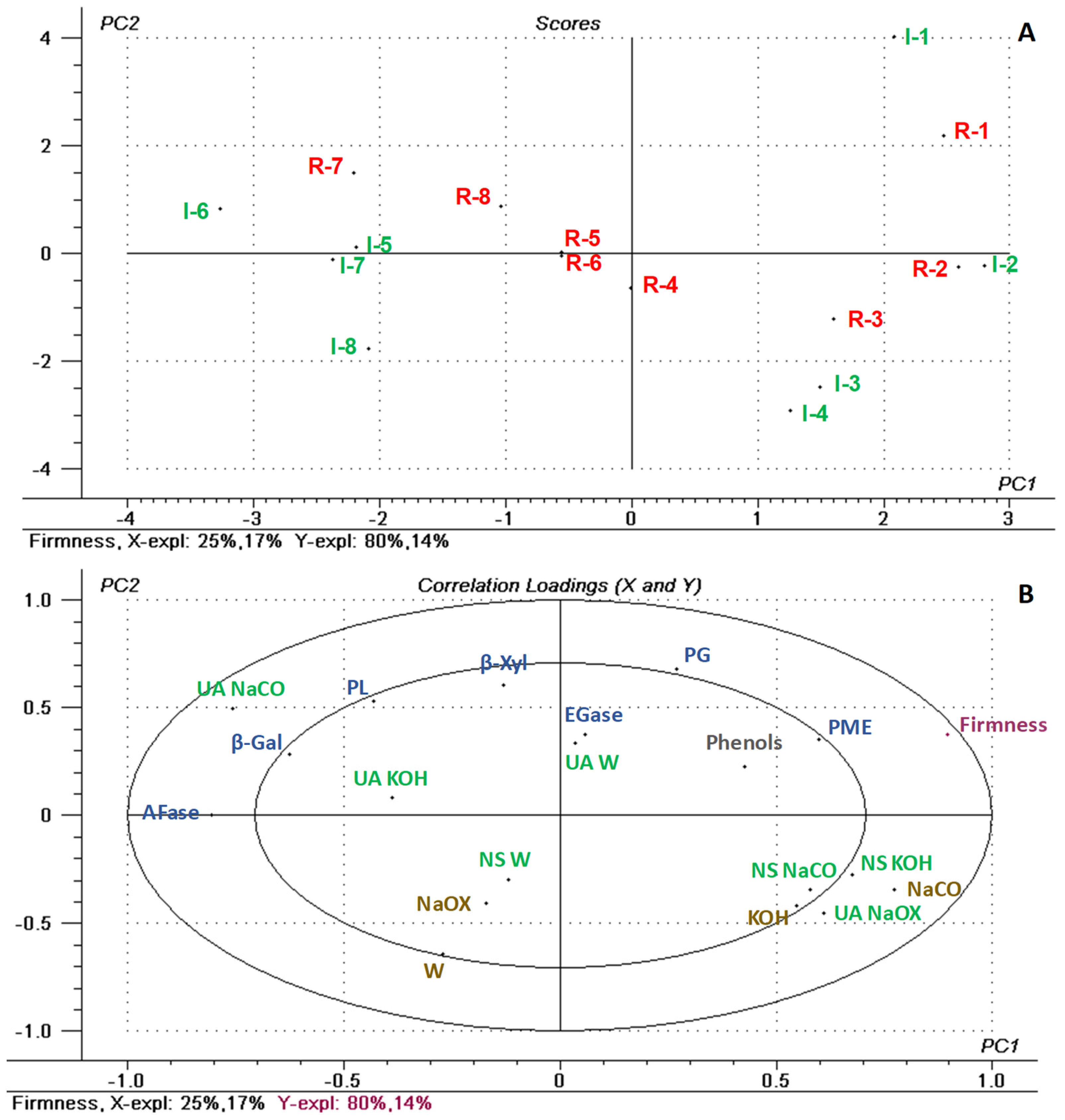
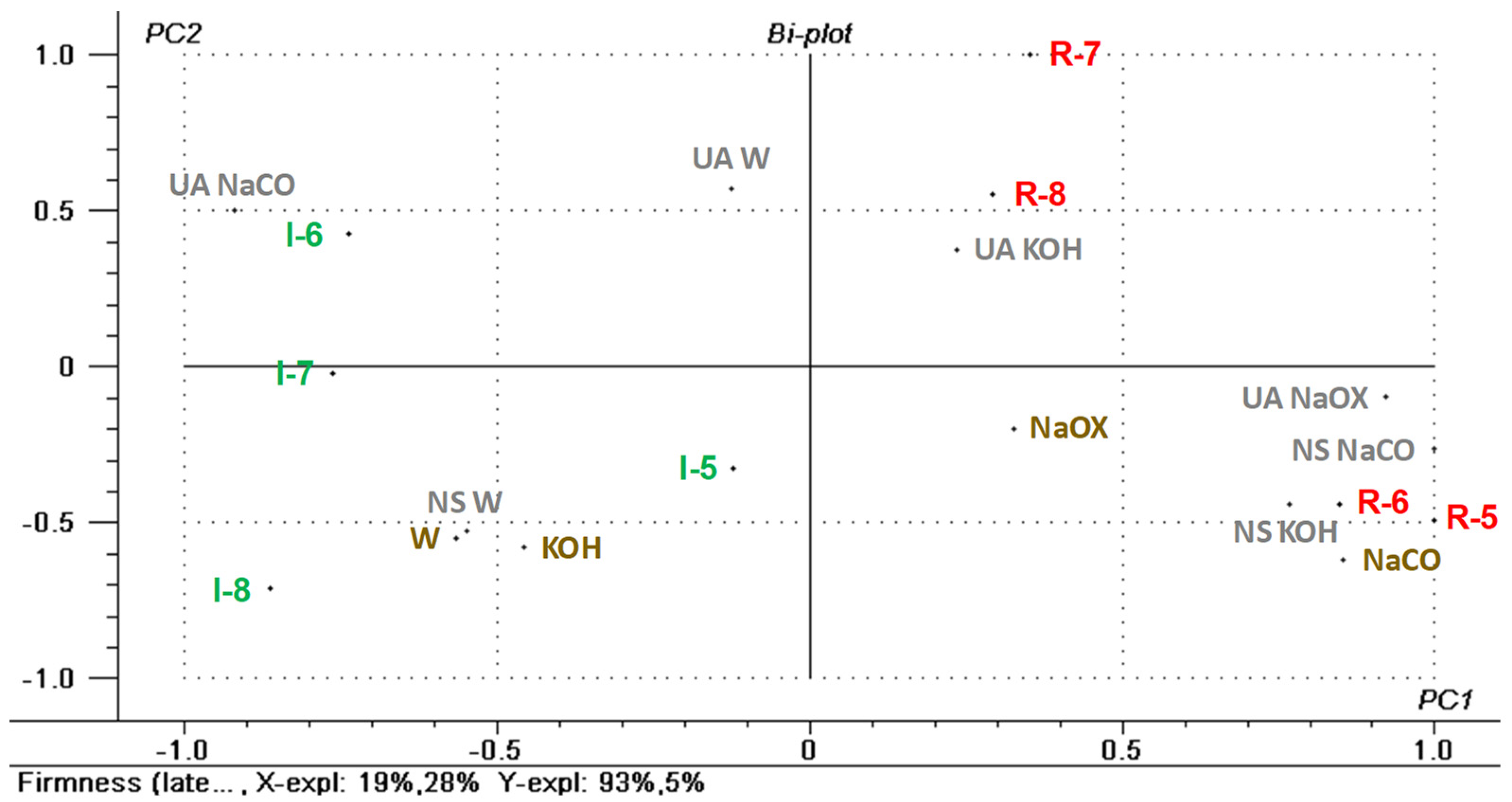
3.4. Regression Model for Fruit Firmness
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Waterman, E.; Lockwood, B. Active components and clinical applications of olive oil. Altern. Med. Rev. 2007, 12, 331–342. [Google Scholar] [PubMed]
- Connor, D.J.; Gómez-del-Campo, M.; Rousseaux, M.C.; Searles, P.S. Structure, management and productivity of hedgerow olive orchards: A review. Sci. Hortic. 2014, 169, 71–93. [Google Scholar] [CrossRef]
- Goulao, L.F.; Oliveira, C.M. Cell wall modifications during fruit ripening: When a fruit is not the fruit. Trends Food Sci. Technol. 2008, 19, 4–25. [Google Scholar] [CrossRef]
- Marsilio, V.; Lanza, B.; Campestre, C.; De Angelis, M. Oven-dried table olives: Textural properties as related to pectic composition. J. Sci. Food Agric. 2000, 80, 1271–1276. [Google Scholar] [CrossRef]
- González-Cabrera, M.; Domingues-Vidal, A.; Ayora-Cañada, M.J. Hyperspectral FTIR imaging of olive fruit for understanding ripening processes. Postharvest Biol. Technol. 2018, 145, 74–82. [Google Scholar] [CrossRef]
- Dumville, J.C.; Fry, S.C. Solubilisation of tomato fruit pectins by ascorbate: A possible non-enzymic mechanism of fruit softening. Planta 2003, 217, 951–961. [Google Scholar] [CrossRef]
- Cheng, G.; Duan, X.; Shi, J.; Lu, W.; Luo, Y.; Jiang, W.; Jiang, Y. Effects of reactive oxygen species on cellular wall disassembly of banana fruit during ripening. Food Chem. 2008, 109, 319–324. [Google Scholar] [CrossRef]
- Duan, X.; Zhang, H.; Zhang, D.; Sheng, J.; Lin, H.; Jiang, Y. Role of hydroxyl radical in modification of cell wall polysaccharides and aril breakdown during senescence of harvested longan fruit. Food Chem. 2011, 128, 203–207. [Google Scholar] [CrossRef]
- Belge, B.; Comabella, E.; Graell, J.; Lara, I. Post-storage cell wall metabolism in two sweet cherry (Prunus avium L.) cultivars displaying different postharvest performance. Food Sci. Technol. Int. 2015, 21, 416–427. [Google Scholar] [CrossRef]
- Vierhuis, E.; Schols, H.A.; Beldman, G.; Voragen, A.G.J. Isolation and characterisation of cell wall material from olive fruits (Olea europaea cv. Koroneiki) at different ripening stages. Carbohydr. Polym. 2000, 43, 11–21. [Google Scholar] [CrossRef]
- Jiménez, A.; Rodríguez, R.; Fernández-Caro, I.; Guillén, R.; Fernández-Bolaños, J.; Heredia, A. Olive fruit cell wall: Degradation of pectic polysaccharides during ripening. J. Agric. Food Chem. 2001, 49, 409–415. [Google Scholar] [CrossRef] [PubMed]
- Mafra, I.; Lanza, B.; Reis, A.; Marsilio, V.; Campestre, C.; De Angelis, M.; Coimbra, M.A. Effect of ripening on texture, microstructure and cell wall polysaccharide composition of olive fruit (Olea europaea). Physiol. Plant. 2001, 111, 439–447. [Google Scholar] [CrossRef]
- Fernández-Bolaños, J.; Heredia, J.; Vioque, B.; Castellano, J.M.; Guillén, R. Changes in cell-wall-degrading enzyme activities in stored olives in relation to respiration and ethylene production—Influence of exogenous ethylene. Eur. Food Res. Technol. 1997, 204, 293–299. [Google Scholar] [CrossRef]
- Parra, R.; Paredes, M.A.; Sánchez-Calle, I.M.; Gómez-Jiménez, M.C. Comparative transcriptional profiling analysis of olive ripe-fruit pericarp and abscission zone tissues shows expression differences and distinct patterns of transcriptional regulation. BMC Genom. 2013, 14, 866–886. [Google Scholar] [CrossRef]
- Lara, I.; Albrecht, R.; Comabella, E.; Riederer, M.; Graell, J. Cell-wall metabolism of ‘Arbequina’ olive fruit picked at different maturity stages. Acta Hortic. 2018, 1199, 133–138. [Google Scholar] [CrossRef]
- Diarte, C.; Iglesias, I.; Romero, A.; Casero, T.; Ninot, A.; Gatius, F.; Graell, J.; Lara, I. Ripening-related cell wall modifications in olive (Olea europaea L.) fruit: A survey of nine genotypes. Food Chem. 2021, 338, 127754. [Google Scholar] [CrossRef]
- Motilva, M.J.; Tovar, M.J.; Romero, M.P.; Alegre, A.; Girona, J. Influence of regulated deficit irrigation strategies applied to olive trees (Arbequina cultivar) on oil yield and oil composition during the tree fruit ripening. J. Sci. Food Agric. 2000, 80, 2037–2043. [Google Scholar] [CrossRef]
- Dabbou, S.; Chehab, H.; Faten, B.; Dabbou, S.; Esposto, S.; Selvaggini, R.; Taticchi, A.; Servili, M.; Montedoro, G.F.; Hammami, M. Effect of three irrigation regimes on Arbequina olive oil produced under Tunisian growing conditions. Agric. Water Manag. 2010, 97, 763–768. [Google Scholar] [CrossRef]
- García, J.M.; Cuevas, M.V.; Fernández, J.E. Production and oil quality in ‘Arbequina’ olive (Olea europaea L.) trees under two deficit irrigation strategies. Irrig. Sci. 2013, 31, 359–370. [Google Scholar] [CrossRef]
- Petridis, A.; Therios, I.; Samouris, G.; Koundouras, S.; Giannakoula, A. Effect of water deficit on leaf phenolic composition, gas exchange, oxidative damage and antioxidant activity of four Greek olives (Olea europaea L.) cultivars. Plant Physiol. Biochem. 2012, 60, 1–11. [Google Scholar] [CrossRef]
- Mechri, B.; Tekaya, M.; Hammami, M.; Chehab, H. Effects of drought stress on phenolic accumulation in greenhouse-grown olive trees (Olea europaea). Biochem. Syst. Ecol. 2020, 92, 104112. [Google Scholar] [CrossRef]
- Voragen, F.G.J.; Timmers, J.P.J.; Linssen, J.P.H.; Schols, H.A.; Pilnik, W. Methods of analysis for cell-wall polysaccharides of fruit and vegetables. Z. Lebensm. Unters. Forch. 1983, 177, 251–256. [Google Scholar] [CrossRef]
- Lefever, G.; Vieuille, M.; Delage, N.; d’Harlingue, A.; de Monteclerc, J.; Bompeix, G. Characterization of cell wall enzyme activities, pectin composition, and technological criteria of strawberry cultivars (Fragaria x ananassa Duch). Food Chem. Toxicol. 2004, 69, 221–226. [Google Scholar] [CrossRef]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Blumenkrantz, N.; Asboe-Hansen, G. New method for quantitative determination of uronic acids. Anal. Biochem. 1973, 54, 484–489. [Google Scholar] [CrossRef]
- Klavons, J.A.; Bennet, R.D. Determination of methanol using alcohol oxidase and its application to methyl ester content of pectins. J. Agric. Food Chem. 1986, 34, 597–599. [Google Scholar] [CrossRef]
- Ortiz, A.; Graell, J.; Lara, I. Preharvest calcium applications inhibit some cell wall-modifying enzyme activities and delay cell wall disassembly at commercial harvest of ‘Fuji Kiku-8′ apples. Postharvest Biol. Technol. 2011, 62, 161–167. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Lara, I.; Camats, J.A.; Comabella, E.; Ortiz, A. Eating quality and health-promoting properties of two sweet cherry (Prunus avium L.) cultivars stored in passive modified atmosphere. Food Sci. Technol. Int. 2015, 21, 133–144. [Google Scholar] [CrossRef]
- Gillespie, K.M.; Ainsworth, E.A. Measurement of reduced, oxidized and total ascorbate content in plants. Nat. Protoc. 2007, 2, 871–874. [Google Scholar] [CrossRef]
- Jemai, H.; Bouaziz, M.; Sayadi, S. Phenolic composition, sugar content and antioxidant activity of Tunisian sweet olive cultivar with regard to fruit ripening. J. Agric. Food Chem. 2009, 57, 2961–2968. [Google Scholar] [CrossRef] [PubMed]
- Micheli, F. Pectin methylesterases: Cell wall enzymes with important roles in plant physiology. Trends Plant Sci. 2001, 6, 414–419. [Google Scholar] [CrossRef]

| Picking | Date | Irrigation Regime | Weight (g) | Length (mm) | Diameter (mm) | F:S Ratio * | Anthocyanin (mg g−1 DW) | Phenols (mg g−1 DW) | RSA * (%) | AA * (nmol g−1 DW) | DHA * (nmol g−1 DW) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Sep 18 | Irrigated | 1.23 | e A | 14.18 | d A | 12.66 | f A | 3.80 | c A | 0.4 | e A | 19.8 | a B | 95.0 | a A | 0.10 | bc B | 0.15 | a B |
| 2 | Oct 2 | 1.62 | cd A | 15.41 | bc A | 14.00 | cde A | 4.57 | b A | 0.4 | de A | 11.1 | d B | 83.7 | b A | 0.10 | bc A | 0.13 | ab A | |
| 3 | Oct 16 | 1.81 | bc A | 16.01 | a A | 14.77 | a A | 5.55 | a A | 0.3 | e B | 16.0 | b B | 95.5 | a A | 0.07 | d B | 0.06 | d B | |
| 4 | Oct 30 | 1.86 | ab A | 15.60 | abc A | 14.08 | cd A | 5.58 | a A | 0.6 | cd B | 13.0 | c B | 80.7 | bc B | 0.11 | b A | 0.08 | c B | |
| 5 | Nov 13 | 2.03 | a A | 15.90 | ab A | 14.52 | ab A | 4.69 | b A | 0.7 | c B | 11.3 | d B | 72.6 | c B | 0.11 | b A | 0.05 | de B | |
| 6 | Nov 28 | 1.84 | ab A | 15.48 | bc A | 14.21 | bc A | 4.46 | b A | 0.8 | bc A | 9.4 | e B | 38.0 | e B | 0.09 | c A | 0.04 | e B | |
| 7 | Dec 11 | 1.58 | d A | 15.20 | c A | 13.68 | de A | 4.55 | b A | 1.0 | b A | 14.4 | bc B | 53.7 | d A | 0.10 | bc A | 0.06 | d B | |
| 8 | Jan 15 | 1.59 | d A | 15.40 | c A | 13.61 | e A | 3.61 | c A | 3.1 | a A | 14.5 | bc A | 74.1 | c A | 0.16 | a B | 0.12 | b A | |
| 1 | Sep 18 | Non-irrigated | 1.08 | d B | 12.90 | c B | 11.63 | e B | 3.19 | e B | 0.2 | f B | 25.3 | a A | 97.0 | a A | 0.13 | b A | 0.21 | a A |
| 2 | Oct 2 | 1.38 | b A | 13.54 | b B | 12.12 | cd B | 3.66 | cd B | 0.2 | ef B | 17.0 | bc A | 94.0 | a A | 0.07 | de B | 0.10 | d A | |
| 3 | Oct 16 | 1.23 | c B | 13.32 | bc B | 11.86 | de B | 3.63 | cd B | 0.4 | de A | 24.5 | a A | 88.9 | ab A | 0.08 | d A | 0.09 | d A | |
| 4 | Oct 30 | 1.42 | b B | 13.73 | b B | 12.53 | bc B | 4.48 | a B | 1.0 | b A | 24.2 | a A | 96.3 | a A | 0.13 | b A | 0.15 | b A | |
| 5 | Nov 13 | 1.52 | a B | 14.59 | a B | 12.96 | ab B | 3.86 | bc A | 1.0 | b A | 19.2 | b A | 82.1 | bc A | 0.11 | c A | 0.10 | d A | |
| 6 | Nov 28 | 1.28 | c B | 14.49 | a B | 12.69 | ab B | 3.41 | de B | 0.4 | d B | 16.4 | bc A | 58.7 | e A | 0.06 | e B | 0.07 | e A | |
| 7 | Dec 11 | 1.56 | a A | 14.40 | a B | 12.59 | abc B | 4.02 | b B | 0.8 | c A | 18.2 | b A | 66.4 | de A | 0.10 | c A | 0.10 | d A | |
| 8 | Jan 15 | 1.54 | a A | 14.30 | a B | 13.04 | a A | 3.46 | de A | 2.9 | a A | 14.1 | c A | 73.7 | cd A | 0.19 | a A | 0.12 | c A | |
| Picking | Date | Irrigation Regime | AIR (g 100 g−1 FW) | d.e. (%) | AIR Fractions (g 100 g−1 AIR) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Wsf * | NaOxsf * | Na2CO3sf * | KOHsf * | Insoluble Residue | |||||||||||
| 1 | Sep 18 | Irrigated | 9.79 | 75.26 | b A | 1.52 | ef A | 7.89 | c B | 1.33 | d B | 1.53 | bc B | 87.7 | a A |
| 2 | Oct 2 | 5.78 | 61.41 | c A | 0.86 | f B | 7.91 | c A | 3.52 | b A | 1.20 | bc A | 86.5 | b A | |
| 3 | Oct 16 | 6.05 | 56.09 | cd A | 5.31 | ab A | 8.76 | a A | 3.98 | a A | 3.58 | a A | 78.3 | e B | |
| 4 | Oct 30 | 4.85 | 50.57 | d A | 3.85 | c A | 8.64 | ab A | 2.59 | c A | 2.99 | a A | 82.1 | d B | |
| 5 | Nov 13 | 9.25 | 88.43 | a A | 2.06 | de B | 8.64 | ab A | 1.13 | d B | 1.66 | b A | 86.6 | b A | |
| 6 | Nov 28 | 13.19 | 84.73 | ab A | 2.46 | d A | 8.31 | bc A | 1.00 | d B | 0.93 | c A | 87.3 | ab A | |
| 7 | Dec 11 | 13.87 | 49.54 | de A | 5.45 | a A | 8.11 | c A | 1.26 | d A | 0.93 | c A | 84.2 | c B | |
| 8 | Jan 15 | 14.50 | 39.88 | e A | 4.59 | bc A | 8.11 | c A | 1.40 | d A | 1.40 | bc A | 84.5 | c B | |
| 1 | Sep 18 | Non-irrigated | 12.00 | 58.85 | bc A | 1.59 | d A | 8.37 | ab A | 1.99 | c A | 2.92 | a A | 85.2 | b B |
| 2 | Oct 2 | 8.82 | 57.88 | bc A | 1.33 | d A | 7.85 | bc A | 3.32 | a A | 1.33 | bc A | 86.2 | b A | |
| 3 | Oct 16 | 8.21 | 69.49 | b A | 3.86 | a A | 8.18 | abc A | 2.79 | b B | 3.26 | a A | 81.9 | c A | |
| 4 | Oct 30 | 8.57 | 42.67 | d A | 2.79 | b B | 8.63 | a A | 1.79 | c B | 1.53 | b B | 85.3 | b A | |
| 5 | Nov 13 | 9.42 | 92.43 | a A | 3.79 | a A | 8.26 | abc A | 1.73 | c A | 0.93 | cd B | 85.4 | b A | |
| 6 | Nov 28 | 11.81 | 90.08 | a A | 3.13 | b A | 8.59 | a A | 2.00 | c A | 1.07 | bcd A | 85.3 | b A | |
| 7 | Dec 11 | 16.15 | 53.42 | cd A | 1.33 | d B | 8.42 | ab A | 0.93 | d A | 0.80 | cd A | 88.5 | a A | |
| 8 | Jan 15 | 16.06 | 21.12 | e B | 2.12 | c B | 7.70 | c B | 1.20 | d A | 0.66 | d B | 88.3 | a A | |
| Picking | Date | Irrigation Regime | Uronic Acids (g 100−1 g) | Neutral Sugars (g 100−1 g) | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AIR | Wsf | NaOxsf | Na2CO3sf | KOHsf | AIR | Wsf | Na2CO3 | KOHsf | ||||||||||||
| 1 | Sep 18 | Irrigated | 6.40 | c A | 11.45 | b A | 4.15 | c A | 20.77 | c A | 3.80 | c A | 3.53 | b B | 19.87 | b A | 3.82 | de B | 27.67 | c B |
| 2 | Oct 2 | 11.16 | b A | 18.98 | a A | 4.20 | c A | 8.56 | g B | 5.62 | a A | 3.51 | b B | 14.24 | d B | 15.69 | a A | 35.62 | a B | |
| 3 | Oct 16 | 12.38 | a A | 8.61 | c A | 6.72 | a A | 11.88 | f A | 3.04 | d A | 3.32 | b B | 17.61 | c A | 9.52 | b B | 36.70 | a A | |
| 4 | Oct 30 | 10.61 | b A | 7.01 | e B | 6.22 | b A | 8.69 | g B | 4.06 | c B | 8.91 | a A | 18.62 | bc B | 15.20 | a A | 32.70 | b B | |
| 5 | Nov 13 | 4.60 | d A | 7.75 | d A | 3.44 | d B | 19.48 | d A | 4.93 | b A | 4.25 | ab A | 20.18 | b A | 4.58 | d B | 24.81 | e B | |
| 6 | Nov 28 | 4.61 | d A | 7.78 | d A | 2.52 | e A | 27.94 | a A | 4.00 | c A | 2.19 | b A | 13.65 | d B | nd | 20.26 | f B | ||
| 7 | Dec 11 | 4.14 | de A | 11.45 | b B | 1.80 | f B | 21.76 | b A | 4.07 | c B | 2.91 | b A | 10.03 | e A | 1.93 | e A | 25.54 | de A | |
| 8 | Jan 15 | 3.24 | e A | 11.48 | b A | 2.01 | f B | 16.74 | e A | 4.70 | b A | 3.54 | b A | 54.12 | a A | 6.62 | c B | 26.87 | cd A | |
| 1 | Sep 18 | Non-irrigated | 6.73 | b A | 11.13 | b A | 2.91 | c B | 12.43 | cd B | 2.52 | g B | 6.50 | b A | 16.79 | c B | 12.78 | ab A | 35.14 | b A |
| 2 | Oct 2 | 7.62 | a B | 8.04 | c B | 4.05 | b A | 10.72 | e A | 3.08 | e B | 10.76 | a A | 18.88 | bc A | 4.86 | de B | 40.64 | a A | |
| 3 | Oct 16 | 7.58 | a B | 7.98 | c B | 4.58 | a B | 11.67 | de A | 2.80 | f B | 10.63 | a A | 17.58 | bc A | 10.77 | b A | 29.88 | c B | |
| 4 | Oct 30 | 6.63 | b B | 7.50 | cd A | 4.20 | b B | 13.38 | c A | 6.21 | b A | 4.18 | c A | 20.97 | a A | 8.02 | c B | 34.65 | b A | |
| 5 | Nov 13 | 5.43 | c A | 7.58 | cd A | 4.06 | b A | 16.37 | b B | 5.29 | c A | 5.97 | bc A | 17.21 | bc B | 7.04 | cd A | 41.96 | a A | |
| 6 | Nov 28 | 5.01 | c A | 7.26 | d A | 2.34 | d A | 11.22 | e B | 3.31 | d B | 2.19 | d A | 18.10 | bc A | 14.16 | a A | 25.66 | d A | |
| 7 | Dec 11 | 3.47 | d B | 18.95 | a A | 2.87 | c A | 22.30 | a A | 8.65 | a A | 1.14 | d B | 11.70 | e A | 3.63 | e A | 25.25 | d A | |
| 8 | Jan 15 | 2.98 | d A | 11.46 | b A | 2.34 | d A | 16.97 | b A | 3.27 | de B | 1.05 | d B | 14.05 | d B | 11.58 | ab A | 19.82 | e B | |
| Non-Pectolytic | Pectolytic | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Picking | Date | Irrigation Regime | β-Xyl | EGase | Backbone-Acting | Side Chain-Acting | ||||||||||
| PME | PG | PL | AFase | β-Gal | ||||||||||||
| 1 | Sep 18 | Irrigated | 0.041 | a A | 0.787 | a A | 1246.23 | a A | 5.802 | a A | 3.949 | b A | 0.013 | e B | 0.129 | c A |
| 2 | Oct 2 | 0.015 | d A | 0.276 | d A | 1100.28 | b A | 0.544 | d B | 2.329 | d A | 0.023 | d B | 0.051 | d A | |
| 3 | Oct 16 | 0.016 | d B | 0.328 | c A | 327.54 | c A | 0.892 | c A | 2.139 | d A | 0.020 | d B | 0.005 | e B | |
| 4 | Oct 30 | 0.011 | e B | 0.432 | b A | 299.33 | c A | 1.922 | b A | 1.022 | e B | 0.033 | c B | 0.164 | bc A | |
| 5 | Nov 13 | 0.030 | b B | 0.401 | b A | 151.31 | d A | 0.779 | c A | 4.661 | a A | 0.057 | b B | 0.235 | b A | |
| 6 | Nov 28 | 0.017 | d B | 0.422 | b B | 310.32 | c B | 0.842 | c B | 3.081 | c B | 0.071 | a A | 0.331 | a A | |
| 7 | Dec 11 | 0.014 | de B | 0.192 | e B | 186.01 | d B | 0.303 | e B | 4.132 | b A | 0.073 | a B | 0.105 | cd B | |
| 8 | Jan 15 | 0.026 | c B | 0.394 | b A | 205.76 | d A | 0.770 | c B | 2.223 | d A | 0.077 | a B | 0.091 | cd A | |
| 1 | Sep 18 | Non-irrigated | 0.031 | c B | 0.448 | bc B | 249.57 | d B | 2.265 | ab B | 2.909 | c B | 0.032 | d A | 0.121 | c A |
| 2 | Oct 2 | 0.012 | e B | 0.447 | c A | 1093.25 | a A | 1.106 | d A | 2.476 | d A | 0.035 | d A | 0.027 | e A | |
| 3 | Oct 16 | 0.033 | bc A | 0.407 | cd A | 259.03 | d B | 0.716 | d B | 0.930 | e B | 0.061 | c A | 0.043 | e A | |
| 4 | Oct 30 | 0.020 | d A | 0.255 | e B | 130.16 | e B | 0.764 | d B | 2.528 | d A | 0.057 | c A | 0.114 | c A | |
| 5 | Nov 13 | 0.038 | a A | 0.341 | de B | 122.92 | e B | 0.804 | d A | 3.933 | b A | 0.065 | c A | 0.159 | a B | |
| 6 | Nov 28 | 0.029 | c A | 0.545 | ab A | 485.76 | b A | 1.382 | cd A | 4.998 | a A | 0.074 | b A | 0.132 | bc B | |
| 7 | Dec 11 | 0.038 | a A | 0.624 | a A | 344.62 | c A | 2.010 | bc A | 3.652 | b B | 0.096 | a A | 0.151 | ab A | |
| 8 | Jan 15 | 0.036 | ab A | 0.352 | cde B | 99.48 | e B | 3.028 | a A | 2.249 | d A | 0.095 | a A | 0.084 | d A | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Diarte, C.; Iglesias, A.; Graell, J.; Lara, I. Firmness and Cell Wall Changes during Maturation of ‘Arbequina’ Olive Fruit: The Impact of Irrigation. Horticulturae 2022, 8, 872. https://doi.org/10.3390/horticulturae8100872
Diarte C, Iglesias A, Graell J, Lara I. Firmness and Cell Wall Changes during Maturation of ‘Arbequina’ Olive Fruit: The Impact of Irrigation. Horticulturae. 2022; 8(10):872. https://doi.org/10.3390/horticulturae8100872
Chicago/Turabian StyleDiarte, Clara, Anna Iglesias, Jordi Graell, and Isabel Lara. 2022. "Firmness and Cell Wall Changes during Maturation of ‘Arbequina’ Olive Fruit: The Impact of Irrigation" Horticulturae 8, no. 10: 872. https://doi.org/10.3390/horticulturae8100872
APA StyleDiarte, C., Iglesias, A., Graell, J., & Lara, I. (2022). Firmness and Cell Wall Changes during Maturation of ‘Arbequina’ Olive Fruit: The Impact of Irrigation. Horticulturae, 8(10), 872. https://doi.org/10.3390/horticulturae8100872







