Remobilization of Storage Nitrogen in Young Pear Trees Grafted onto Vigorous Rootstocks (Pyrus betulifolia)
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Site and Materials
2.2. Experimental Design
2.3. Measurements
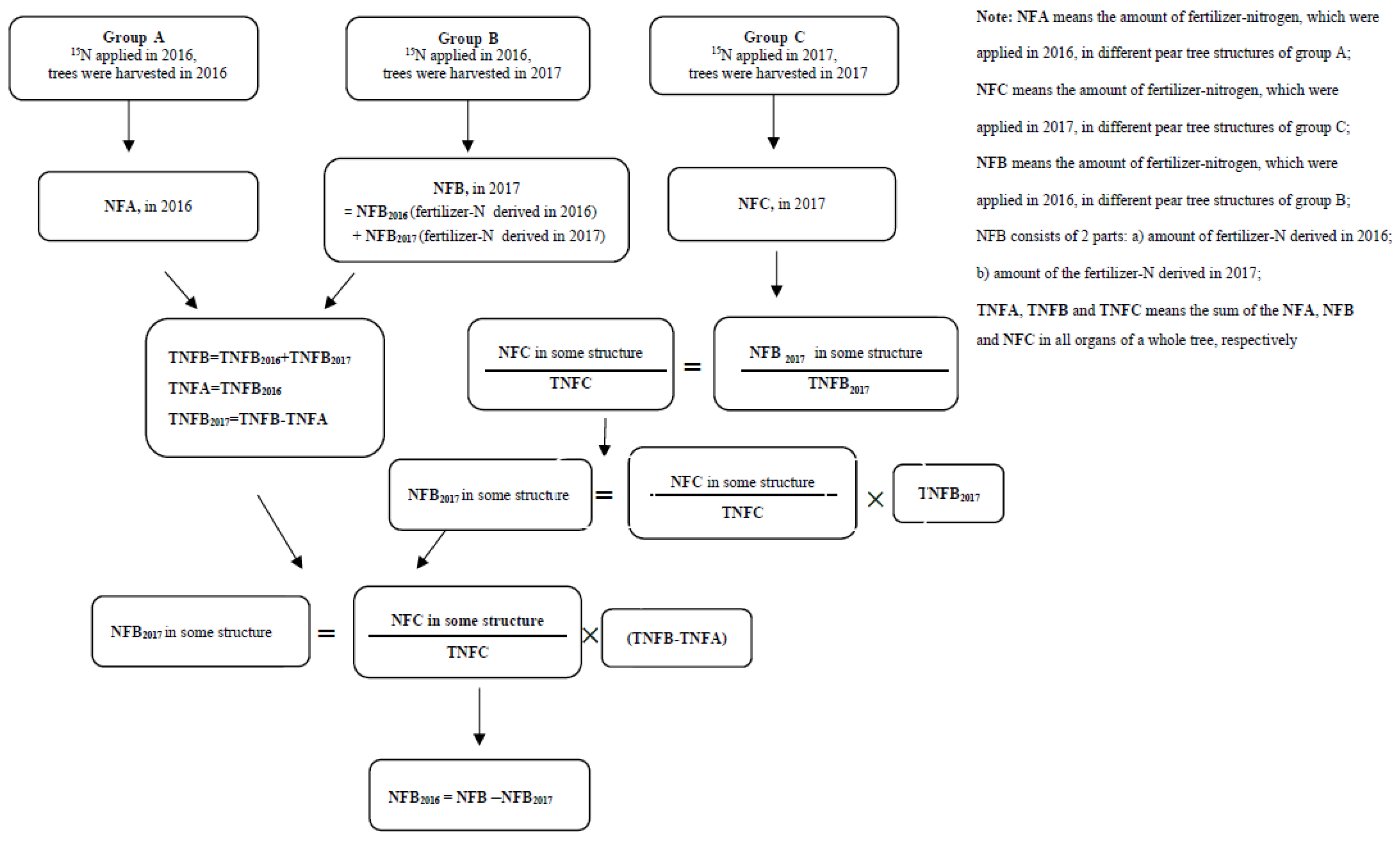
2.4. Calculations and Data Analysis
2.5. Statistical Analysis
3. Results
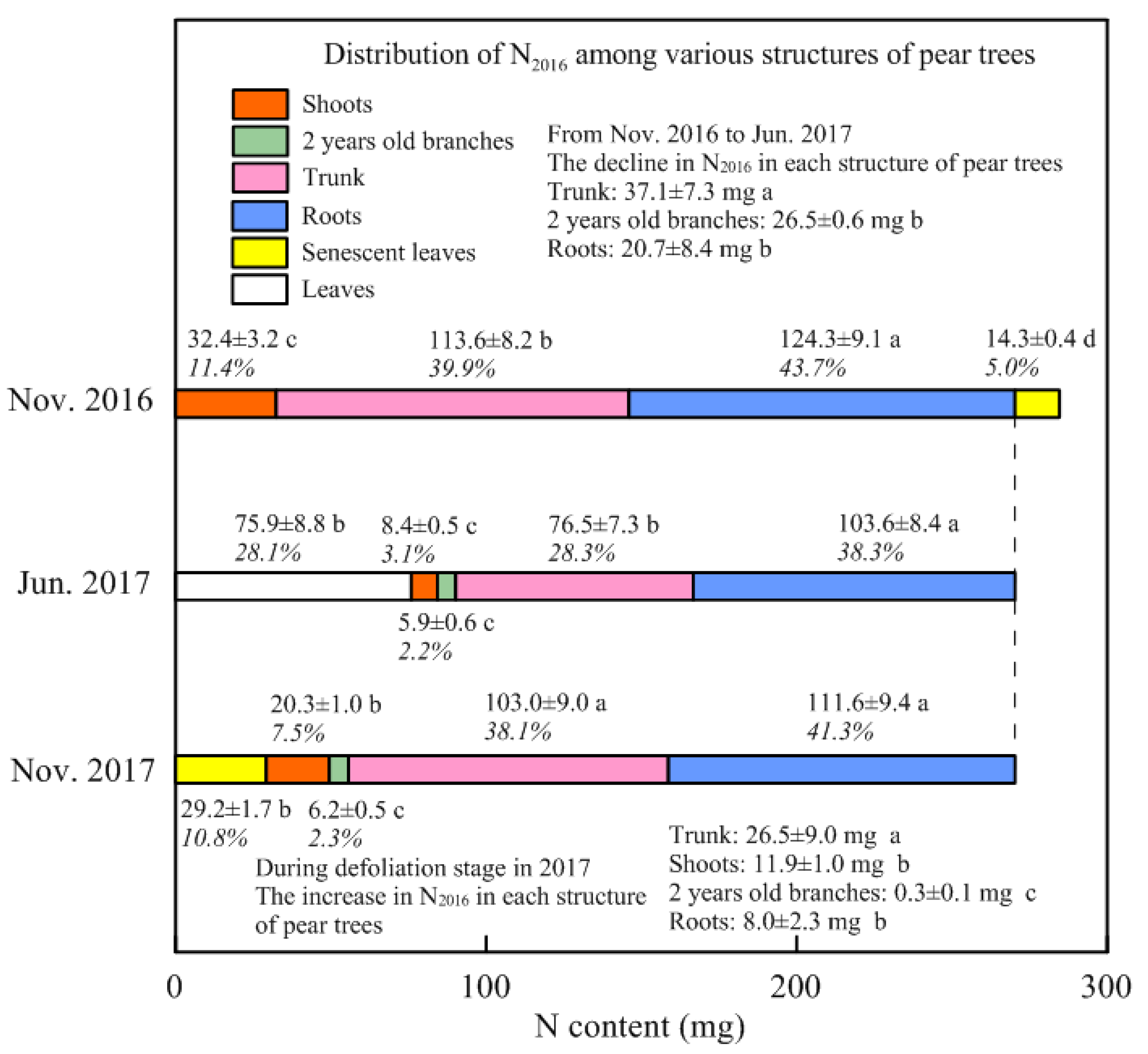
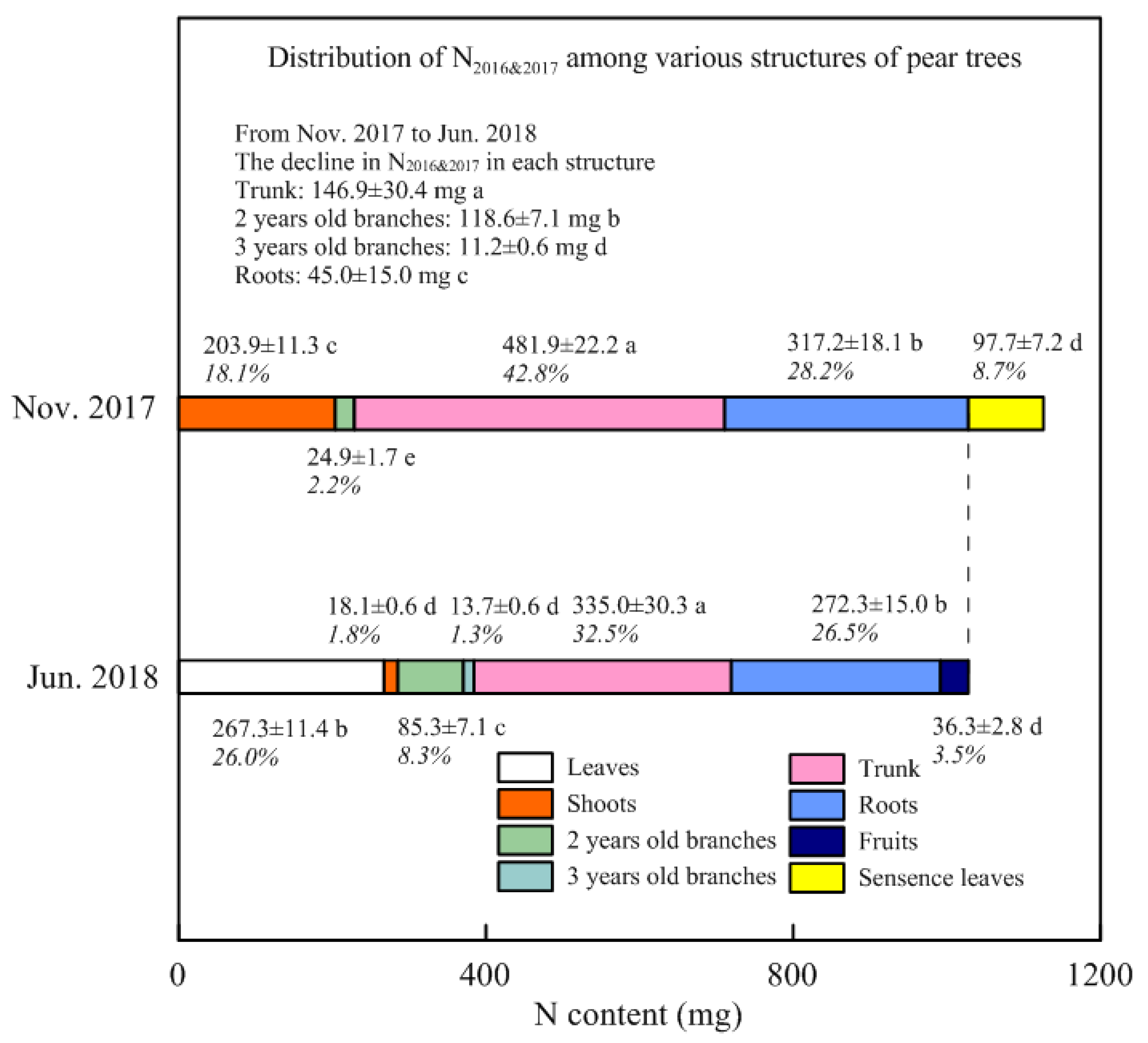
3.1. The N Content and Concentration in Each Structure of Young Pear Trees
3.2. Stored N Remobilized in Spring and Withdrawn from Senescent Leaves in Autumn
4. Discussion
4.1. N Absorbed from Soil and Fertilizer
4.2. Remobilization of the Stored N during Spring
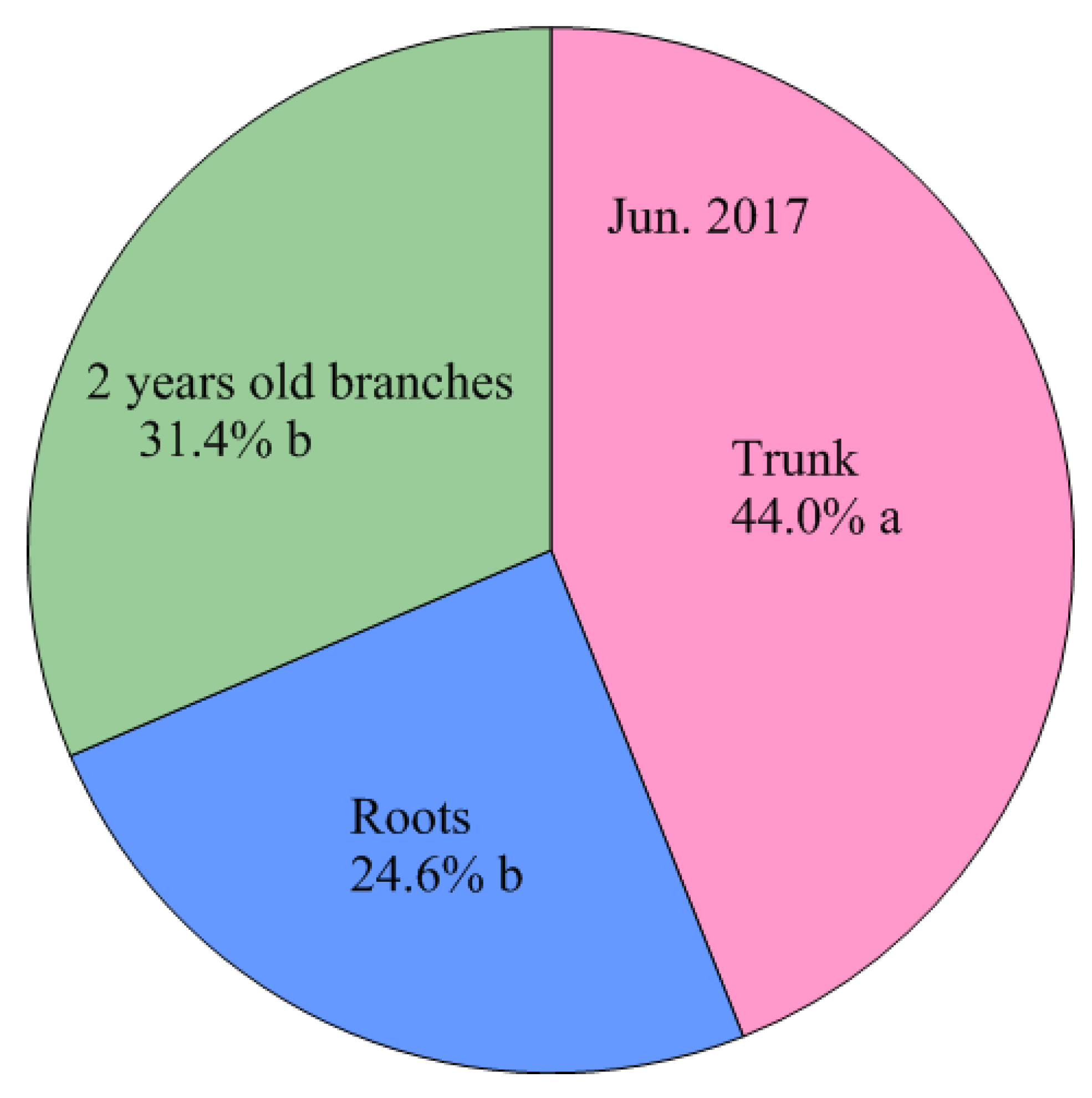
4.3. Storage N Derived from Perennial Structures in Spring
4.4. Redistribution of the Stored N Withdrawn from Senescent Leaves
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Thitithanakul, S.; Pétel, G.; Chalot, M.; Beaujard, F. Supplying nitrate before bud break induces pronounced changes in nitrogen nutrition and growth of young poplars. Funct. Plant Biol. 2012, 39, 795–803. [Google Scholar] [CrossRef] [PubMed]
- Roccuzzo, G.; Scandellari, F.; Allegra, M.; Torrisi, B.; Stagno, F.; Mimmo, T.; Zanotelli, D.; Gioacchini, P.; Millard, P.; Tagliavini, M. Seasonal dynamics of root uptake and spring remobilisation of nitrogen in field grown orange trees. Sci. Hortic. 2017, 226, 223–230. [Google Scholar] [CrossRef]
- Maity, A.; Babu, K.D.; Sarkar, A. Guidelines for fertilizer use in pomegranate orchards based on seasonal uptake and partitioning of nutrients. Sci. Hortic. 2019, 252, 138–148. [Google Scholar] [CrossRef]
- Zarate-Valdez, J.L.; Muhammad, S.; Saa, S.; Lampinen, B.D.; Brown, P.H. Light interception, leaf nitrogen and yield prediction in almonds: A case study. Eur. J. Agron. 2015, 66, 1–7. [Google Scholar] [CrossRef]
- Sanchez, E.E.; Righetti, T.L.; Sugar, D.; Lombard, P.B. Seasonal differences, soil texture and uptake of newly absorbed nitrogen in field-grown pear trees. J. Hortic. Sci. 1990, 65, 395–400. [Google Scholar] [CrossRef]
- Tagliavini, M.; Millard, P.; Quartieri, M.; Marangoni, B. Timing of nitrogen uptake affects winter storage and spring remobilisation of nitrogen in nectarine (Prunus persica var. nectarina) trees. Plant Soil 1999, 211, 149–153. [Google Scholar] [CrossRef]
- Malaguti, D.; Tagliavini, M.; Masia, A.; Marangoni, B.; Gioacchini, P.; Vittori Antisari, L.; Millard, P. Dynamic of root uptake of ammonium and nitrate by pear (Pyrus communis L.) trees after growth resumption in spring. In Plant Nutrition—Food Security and Sustainability of Agro-Ecosystems; Springer: Berlin/Heidelberg, Germany, 2001; pp. 124–125. [Google Scholar]
- Sanchez, E.E.; Righetti, T.L. Tree nitrogen status and leaf canopy position influence postharvest nitrogen accumulation and efflux from pear leaves. J. Am. Soc. Hortic. Sci. 1990, 115, 934–937. [Google Scholar] [CrossRef]
- Niederholzer, F.J.A.; de Jong, T.M.; Saenz, J.L.; Muraoka, T.T.; Weinbaum, S.A. Effectiveness of fall versus spring soil fertilization of field-grown peach trees. J. Am. Soc. Hortic. Sci. 2001, 125, 644–648. [Google Scholar] [CrossRef]
- Neto, C.; Carranca, C.; Clemente, J.; de Varennes, A. Nitrogen distribution, remobilization and re-cycling in young orchard of non-bearing ‘Rocha’ pear trees. Sci. Hortic. 2008, 118, 299–307. [Google Scholar] [CrossRef]
- Arrobas, M.; Ferreira, I.Q.; Freitas, S.; Verdial, J.; Rodrigues, M.A. Guidelines for fertilizer use in vineyards based on nutrient content of grapevine parts. Sci. Hortic. 2014, 172, 191–198. [Google Scholar] [CrossRef]
- Jordan, M.O.; Wendler, R.; Millard, P. Autumnal N storage determines the spring growth, N uptake and N internal cycling of young peach trees. Trees 2012, 26, 393–404. [Google Scholar] [CrossRef]
- Pescie, M.A.; Borda, M.P.; Ortiz, D.P.; Landriscini, M.R.; Lavado, R.S. Absorption, distribution and accumulation of nitrogen applied at different phenological stages in southern highbush blueberry (Vaccinium corymbosum interspecific hybrid). Sci. Hortic. 2018, 230, 11–17. [Google Scholar] [CrossRef]
- Vajari, M.A.; Moghadam, J.F.; Eshghi, S. Influence of late season foliar application of urea, boric acid and zinc sulfate on nitrogenous compounds concentration in the bud and flower of Hayward kiwifruit. Sci. Hortic. 2018, 242, 137–145. [Google Scholar] [CrossRef]
- Vajari, M.A.; Eshghi, S.; Moghadam, J.F.; Gharaghani, A. Late season mineral foliar application improves nutritional reserves and flowering of kiwifruit. Sci. Hortic. 2018, 232, 22–28. [Google Scholar] [CrossRef]
- Neilsen, D.; Millard, P.; Neilsen, G.H.; Hogue, E.J. Sources of N used for leaf growth in a high density apple (Malus domestica) orchard irrigated with ammonium nitrate solution. Tree Physiol. 1997, 17, 733–739. [Google Scholar] [CrossRef] [PubMed]
- Cheng, L.; Fuchigami, L.H. Growth of young apple trees in relation to reserve nitrogen and carbohydrates. Tree Physiol. 2002, 22, 1297–1303. [Google Scholar] [CrossRef] [PubMed]
- Weinbaum, S.; Kessel, C.V. Quantitative estimates of uptake and internal cycling of 14N-labeled fertilizer in mature walnut trees. Tree Physiol. 1998, 18, 795–801. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.K.; Lim, C.S.; Kang, S.M.; Cho, J.L. Root storage of nitrogen applied in autumn and its remobilization to new growth in spring of persimmon trees (Diospyros kaki cv. Fuyu). Sci. Hortic. 2009, 119, 193–196. [Google Scholar] [CrossRef]
- Taylor, B.K. Storage and mobilization of nitrogen in fruit trees: A review. J. Aust. Inst. Agric. Sci. 1967, 33, 23–29. [Google Scholar]
- May, L.H. The nitrogen nutrition of the peach tree II—Storage and mobilization of nitrogen in mature trees. Aust. J. Biol. Sci. 1967, 20, 389–411. [Google Scholar]
- Millard, P.; Proe, M.F. Leaf demography and the seasonal internal cycling of nitrogen in sycamore (Acer pseudoplatanus L.) seedlings in relation to nitrogen supply. New Phytol. 1991, 17, 587–596. [Google Scholar] [CrossRef]
- Toselli, M.; Flore, J.A.; Zavalloni, C.; Marangoni, B. Nitrogen partitioning in apple trees as affected by application time. Horttechnology 2000, 10, 136–140. [Google Scholar] [CrossRef]
- Frak, E.; Millard, P.; Le Roux, X.; Guillaumie, S.; Wendler, R. Coupling sap flow velocity and amino acid concentrations as an alternative method to 15N labeling for quantifying nitrogen remobilization by walnut trees. Plant Physiol. 2002, 130, 1043–1053. [Google Scholar] [CrossRef] [PubMed]
- Grassi, G.; Millard, P.; Wendler, R.; Minotta, G.; Tagliavini, M. Measurement of xylem sap aminoacid concentrations in conjunction with whole tree transpiration estimates spring N remobilization by cherry (Prunus avium L.) trees. Plant Cell Environ. 2002, 25, 1689–1699. [Google Scholar] [CrossRef]
- San-Martino, L.; Sozzi, G.O.; San-Martino, S.; Lavado, R.S. Isotopically-labelled nitrogen uptake and partitioning in sweet cherry as influenced by timing of fertilizer application. Sci. Hortic. 2010, 126, 42–49. [Google Scholar] [CrossRef]
- Gomez, L.; Vercambre, G.; Jordan, M.O. Spatial-temporal management of nitrogen and carbon on the peach tree (Prunus persicae L. Batsch.). Sci. Hortic. 2020, 273, 109613. [Google Scholar] [CrossRef]
- Millard, P.; Neilsen, G.H. The influence of nitrogen supply on the uptake and remobilization of stored N for the seasonal growth of apple Trees. Ann. Bot. 1989, 63, 301–309. [Google Scholar] [CrossRef]
- Bravo, K.; Marcolini, G.; Sorrenti, G.; Baldi, E.; Quartieri, M.; Toselli, M. Effect of time of application on nitrogen uptake, partitioning, and remobilization in walnut trees. J. Plant Nutr. 2017, 40, 719–725. [Google Scholar] [CrossRef]
- Sanchez, E.E.; Righetti, T.L.; Sugar, D.; Lombard, P.B. Recycling of nitrogen in field-grown ‘Comice’ pears. J. Hortic. Sci. 1991, 66, 479–486. [Google Scholar] [CrossRef]
- Sanchez, E.E.; Righetti, T.L.; Sugar, D.; Lombard, P.B. Effects of timing of nitrogen application on nitrogen partitioning between vegetative, reproductive and structural components of mature ‘Comice’ pears. J. Hortic. Sci. 1992, 67, 51–58. [Google Scholar] [CrossRef]
- Tagliavini, M.; Quartieri, M.; Millard, P. Remobilised nitrogen and root uptake of nitrate for spring leaf growth, flowers and developing fruits of pear (Pyrus communis L.) trees. Plant Soil 1997, 195, 137–142. [Google Scholar] [CrossRef]
- Khemira, H.; Azarenko, A.N.; Sugar, D.; Righetti, T.L. Postharvest nitrogen application effect on ovule longevity of ‘Comice’ pear trees. J. Plant Nutr. 1998, 21, 405–411. [Google Scholar] [CrossRef]
- Quartieri, M.; Millard, P.; Tagliavini, M. Storage and remobilisation of nitrogen by pear (Pyrus communis L.) trees as affected by timing of N supply. Eur. J. Agron. 2002, 17, 105–110. [Google Scholar] [CrossRef]
- Zhao, Y.; Sun, M.; Liang, Z.; Li, H.; Yu, F.; Liu, S. Analysis of contrast iron chlorosis tolerance in the pear cv. ‘Huangguan’ grafted onto Pyrus betulifolia and quince A grown in calcareous soils. Sci. Hortic. 2020, 271, 109–488. [Google Scholar] [CrossRef]
- Jiang, H.; Li, H.; Zhao, M.; Mei, X.; Kang, Y.; Dong, C.; Xu, Y. Strategies for timing nitrogen fertilization of pear trees based on the distribution, storage, and remobilization of 15N from seasonal application of (15NH4)2SO. J. Integr. Agric. 2020, 19, 1340–1353. [Google Scholar] [CrossRef]
- Oland, K. Changes in the content of dry matter and major nutrient element of apple foliage during senescence and abscission. Physiol. Plant. 1963, 16, 682–692. [Google Scholar]
- Deng, X.; Weinbaum, S.A.; Dejong, T.M.; Muraoka, T.T. Utilization of nitrogen from storage and current-year uptake in walnut spurs during the spring flush of growth. Physiol. Plant. 1989, 75, 492–498. [Google Scholar] [CrossRef]
- Millard, P. Ecophysiology of the internal cycling of nitrogen for tree growth. J. Plant Nutr. Soil Sci. 1996, 159, 1–10. [Google Scholar] [CrossRef]
- Götz, K.P.; Naher, J.; Fettke, J.; Chmielewski, F.M. Changes of proteins during dormancy and bud development of sweet cherry (Prunus avium L.). Sci. Hortic. 2018, 239, 41–49. [Google Scholar] [CrossRef]
- Carranca, C.; de Varennes, A.; Rolston, D.E. Variation in N-recovery of winter wheat under Mediterranean conditions studied with 15N-labelled fertilizers. Eur. J. Agron. 1999, 11, 145–155. [Google Scholar] [CrossRef]
- Muñoz, N.; Guerri, J.; Legaz, F.; Primo Millo, E. Seasonal uptake of 15N-nitrate and distribution of absorbed nitrogen in peach trees. Plant Soil 1993, 150, 263–269. [Google Scholar] [CrossRef]
- Wu, Y.; Sun, M.; Liu, J.; Wang, W.; Liu, S. Fertilizer and soil nitrogen utilization of pear trees as affected by the timing of split fertilizer application in rain-fed orchard. Sci. Hortic. 2019, 252, 363–369. [Google Scholar] [CrossRef]
- Mason-Jones, K.; Vrehen, P.; Koper, K.; Wang, J.; van der Putten, W.H.; Veen, G.F. Short-term temperature history affects mineralization of fresh litter and extant soil organic matter, irrespective of agricultural management. Soil Biol. Biochem. 2020, 150, 107985. [Google Scholar] [CrossRef]
- Bradford, M.A.; Wieder, W.R.; Bonan, G.B.; Fierer, N.; Raymond, P.A.; Crowther, T.W. Managing uncertainty in soil carbon feedbacks to climate change. Nat. Clim. Chang. 2016, 6, 751–758. [Google Scholar] [CrossRef]
- Lopez-Sangil, L.; Hartley, I.P.; Rovira, P.; Casals, P.; Sayer, E.J. Drying and rewetting conditions differentially affect the mineralization of fresh plant litter and extant soil organic matter. Soil Biol. Biochem. 2018, 124, 81–89. [Google Scholar] [CrossRef]
- Tromp, J. Nutrient reserves in roots of fruit trees, in particular carbohydrates and nitrogen. Plant Soil 1983, 71, 401–413. [Google Scholar] [CrossRef]
| Group | 2016 | 2017 | 2018 |
|---|---|---|---|
| A | 10 g/tree ○ + 5 g/tree ● | - | - |
| B | 10 g/tree ○ + 5 g/tree ● | 20 g/tree ○ | - |
| C | 15 g/tree ○ | 15 g/tree ○ + 5 g/tree ● | - |
| D | 10 g/tree ○ + 5 g/tree ● | 15 g/tree ○ + 5 g/tree ● | - |
| E | 10 g/tree ○ + 5 g/tree ● | 15 g/tree ○ + 5 g/tree ● | 30 g/tree ○ |
| F | 15 g/tree ○ | 20 g/tree ○ | 25 g/tree ○ + 5 g/tree ● |
| Group | 2016 | 2017 | 2018 | |
|---|---|---|---|---|
| Nov. | Jun. | Nov. | Jun. | |
| A | 6 trees | - | ||
| B | 6 trees | 6 trees | ||
| C | 6 trees | 6 trees | ||
| D | 6 trees | 6 trees | ||
| E | - | 6 trees | ||
| F | - | 6 trees | ||
| Years | Organs | N Content (mg) | Dry Mass Weight (g) | N Concentration (%) |
|---|---|---|---|---|
| Mar. 2016 Nursery | Trunk | 278.7 ± 14.8 c E | 43.2 ± 1.4 b D | 0.64 ± 0.02 b C |
| Roots | 485.0 ± 15.8 b D | 42.3 ± 1.1 b D | 1.14 ± 0.06 a B | |
| Total | 763.7 ± 11.7 a E | 85.6 ± 2.3 a E | ||
| Nov. 2016 | Leaves | 89.6 ± 10.4 d D | 6.9 ± 0.8 d D | 1.30 ± 0.01 a D |
| Shoots | 168.4 ± 20.0 d D | 13.2 ± 1.3 c E | 1.27 ± 0.04 a A | |
| Trunk | 668.8 ± 39.0 c D | 65.8 ± 4.6 b D | 1.01 ± 0.03 c A | |
| Roots | 789.6 ± 45.6 b C | 64.6 ± 3.8 b CD | 1.22 ± 0.03 b A | |
| Total | 1716.4 ± 86.2 a D | 150.5 ± 8.2 a D | ||
| Jun. 2017 | Leaves | 2286.3 ± 178.2 b B | 86.8 ± 6.7 c B | 2.64 ± 0.01 a B |
| Shoots | 345.3 ± 27.2 d C | 38.1 ± 3.2 d D | 0.91 ± 0.08 c D | |
| 2 years old branches | 129.3 ± 4.6 d D | 16.3 ± 0.5 d E | 0.79 ± 0.01 d E | |
| Trunk | 1035.3 ± 80.9 c C | 168.7 ± 12.7 b C | 0.61 ± 0.01 e C | |
| Root | 1042.5 ± 67.0 c C | 84.6 ± 5.0 c C | 1.23 ± 0.01 b A | |
| Total | 4838.7 ± 332.6 a C | 394.5 ± 26.2 a C | ||
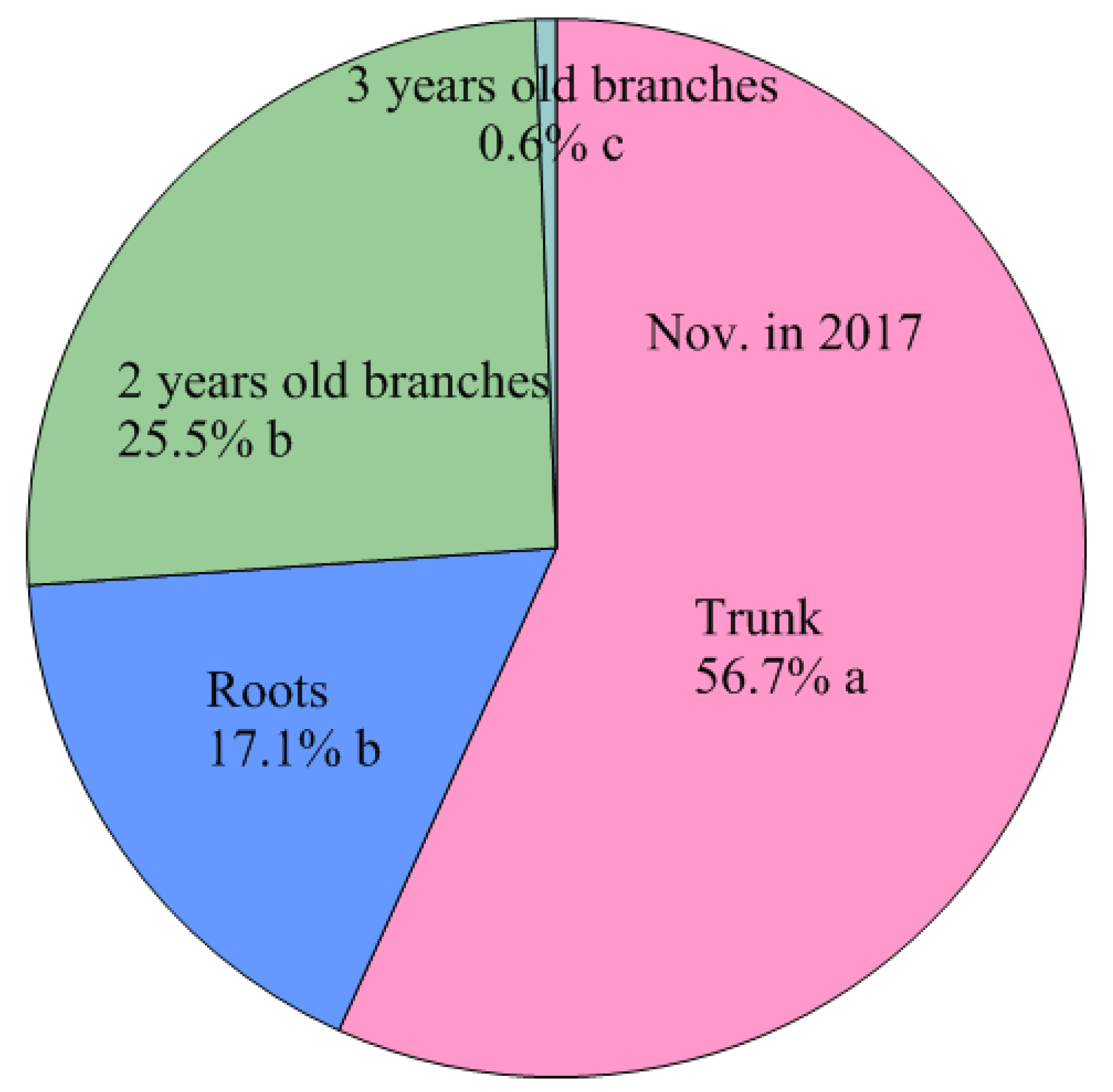
| Nov. 2017 | Leaves | 738.3 ± 66.4 e C | 50.6 ± 5.2 e C | 1.46 ± 0.02 a C |
| Shoots | 1554.6 ± 149.6 d A | 140.8 ± 15.8 d B | 1.11 ± 0.03 c B | |
| 2 years old branches | 180.2 ± 13.6 f D | 17.7 ± 1.6 e E | 1.02 ± 0.05 d C | |
| Trunk | 3832.6 ± 203.1 b B | 455.6 ± 34.0 b B | 0.84 ± 0.02 e B | |
| Roots | 3211.6 ± 174.3 c B | 253.2 ± 20.6 c B | 1.27 ± 0.07 b A | |
| Total | 9517.3 ± 584.4 a B | 917.9 ± 74.5 a B | ||
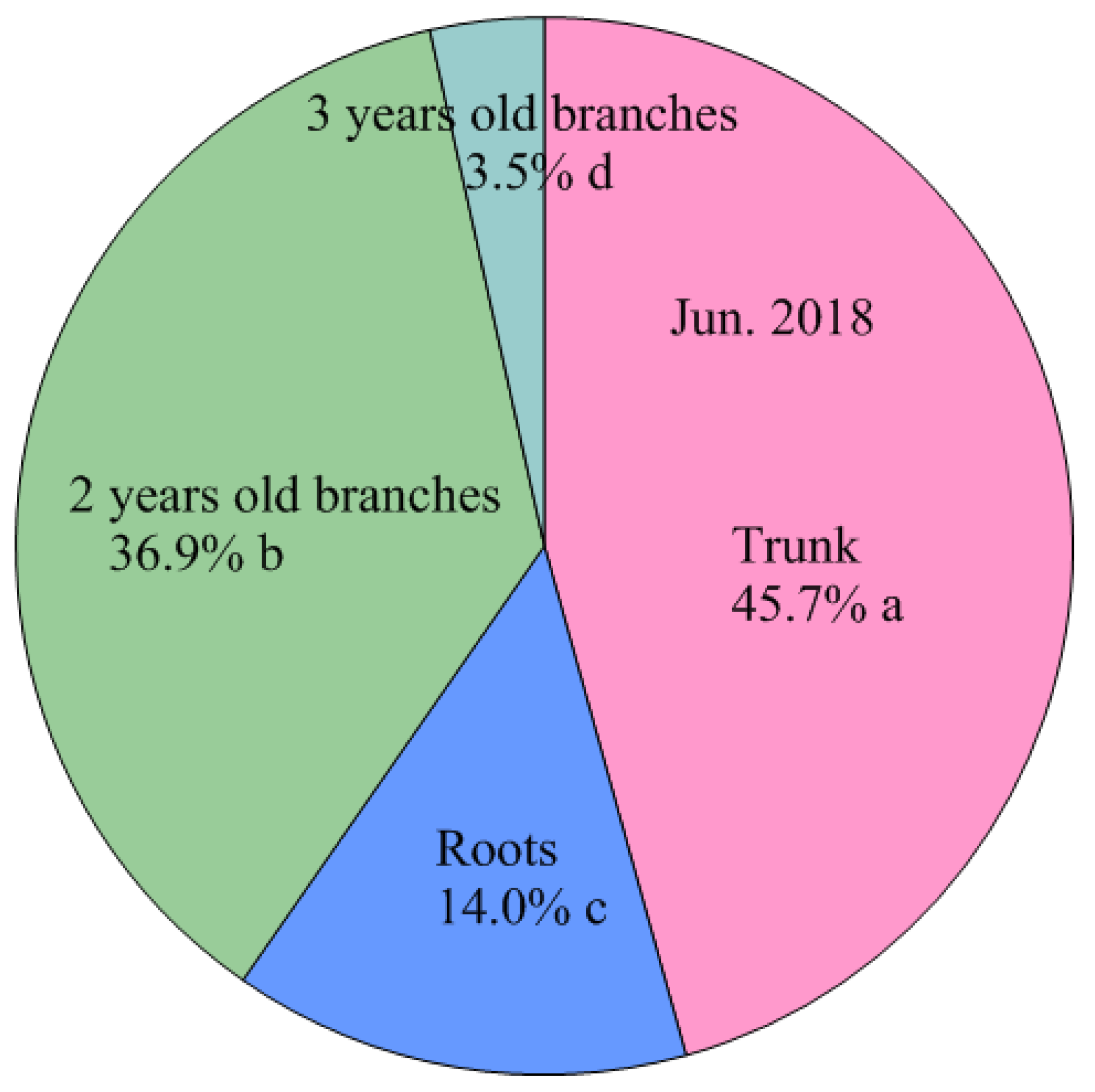
| Jun. 2018 | Leaves | 8099.7 ± 541.4 b A | 295.0 ± 24.7 d A | 2.75 ± 0.07 a A |
| Shoots | 1150.5 ± 124.5 ef B | 117.7 ± 12.3 f C | 0.98 ± 0.02 d C | |
| 2 years old branches | 1456.3 ± 108.5 e A | 208.7 ± 16.3 e A | 0.70 ± 0.01 f F | |
| 3 years old branches | 248.9 ± 22.9 g CD | 23.0 ± 2.1 g D E | 1.08 ± 0.01 c B | |
| Trunk | 3263.5 ± 194.1 d A | 632.1 ± 32.0 b A | 0.52 ± 0.02 g D | |
| Roots | 4914.6 ± 311.8 c A | 406.7 ± 27.7 c A | 1.21 ± 0.01 b AB | |
| Fruits | 465.5 ± 41.9 fg | 51.1 ± 4.8 fg | 0.91 ± 0.01 e | |
| Total | 19599.0 ± 1177.7 a A | 1734.3 ± 108.9 a A |
| Time | Increment of Total N (mg) | Increment of N Uptake from Fertilizer (mg) | Increment of N Uptake from Soil (mg) |
|---|---|---|---|
| Mar. 2016 to Nov.2016 | 953 ± 93 d | 285 ± 11 c | 668 ± 88 d |
| Nov.2016 to Jun. 2017 | 3122 ± 333 c | 743 ± 57 b | 2379 ± 287 c |
| Jun. 2017 to Nov. 2017 | 4679 ± 584 b | 855 ± 39 b | 3823 ± 550 b |
| Nov. 2017 to Jun. 2018 | 10082 ± 1178 a | 2128 ± 160 a | 7954 ± 1030 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, Y.; Sun, M.; Qi, Y.; Liu, S. Remobilization of Storage Nitrogen in Young Pear Trees Grafted onto Vigorous Rootstocks (Pyrus betulifolia). Horticulturae 2021, 7, 148. https://doi.org/10.3390/horticulturae7060148
Wu Y, Sun M, Qi Y, Liu S. Remobilization of Storage Nitrogen in Young Pear Trees Grafted onto Vigorous Rootstocks (Pyrus betulifolia). Horticulturae. 2021; 7(6):148. https://doi.org/10.3390/horticulturae7060148
Chicago/Turabian StyleWu, Yang, Mingde Sun, Yuanyong Qi, and Songzhong Liu. 2021. "Remobilization of Storage Nitrogen in Young Pear Trees Grafted onto Vigorous Rootstocks (Pyrus betulifolia)" Horticulturae 7, no. 6: 148. https://doi.org/10.3390/horticulturae7060148
APA StyleWu, Y., Sun, M., Qi, Y., & Liu, S. (2021). Remobilization of Storage Nitrogen in Young Pear Trees Grafted onto Vigorous Rootstocks (Pyrus betulifolia). Horticulturae, 7(6), 148. https://doi.org/10.3390/horticulturae7060148





