Synergistic Effect of Preharvest Spray Application of Natural Elicitors on Storage Life and Bioactive Compounds of Date Palm (Phoenix dactylifera L., cv. Khesab)
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Sampling
2.2. Physiochemical Analysis
2.2.1. Fruit Characteristics
2.2.2. Fruit Ripening and Decay Percentage
2.2.3. Total Soluble Solids (TSS)
2.2.4. Fruit Surface Color
2.3. Determination of Total Microbial Load
2.4. Extraction of Bioactive Compounds
2.4.1. Phytochemical Analysis
Total Phenolic Content (TPC)
Total Tannin Content (TTC)
Total Flavonoid Content (TFC)
2.4.2. Antioxidant Activities
DPPH Radical Scavenging Activity
ABTS Radical Scavenging Activity
2.5. Statistical Analysis
3. Results and Discussion
3.1. Physical Quality Characteristics of Fruit at Harvest
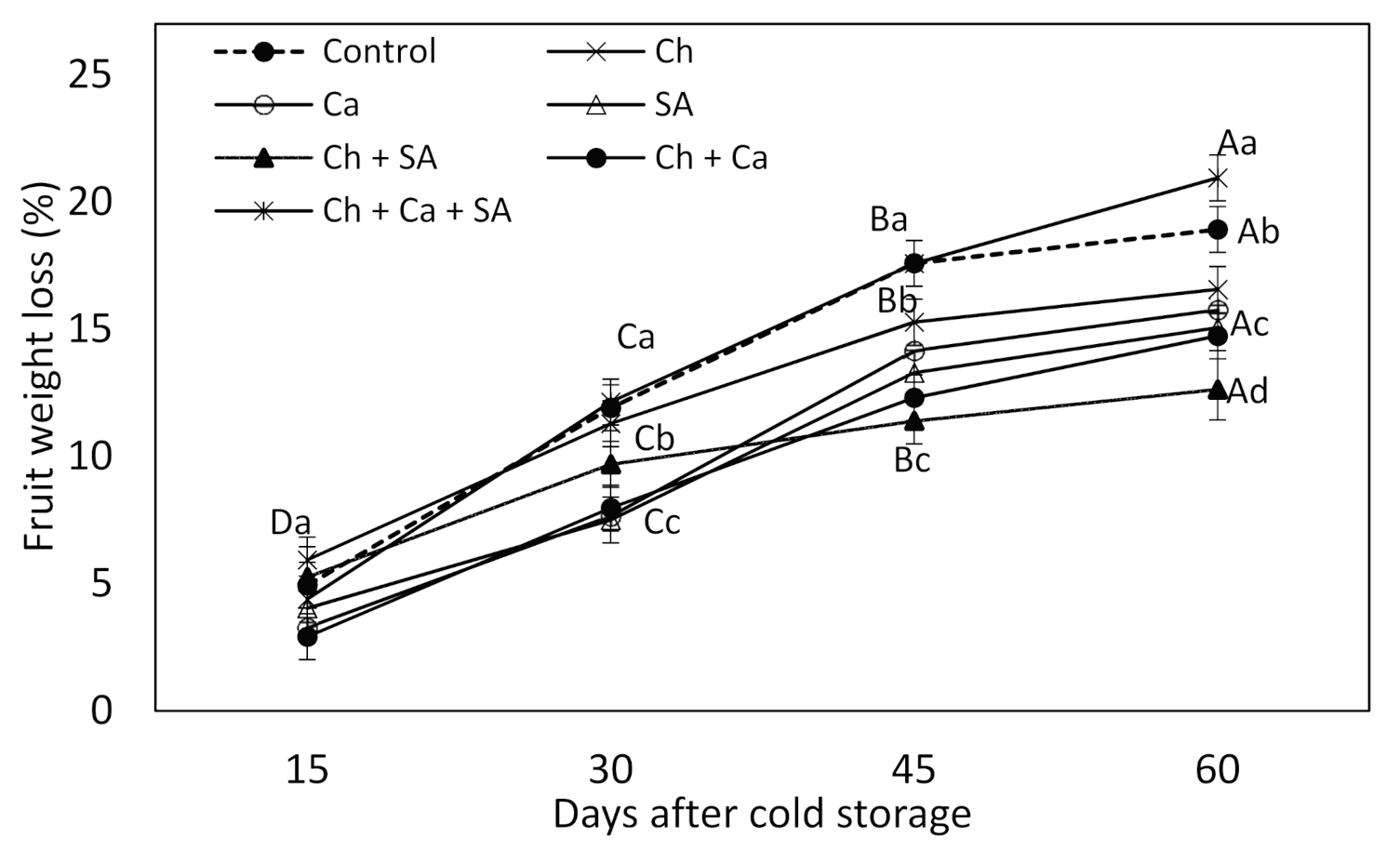
3.2. Fruit Weight Loss during Cold Storage
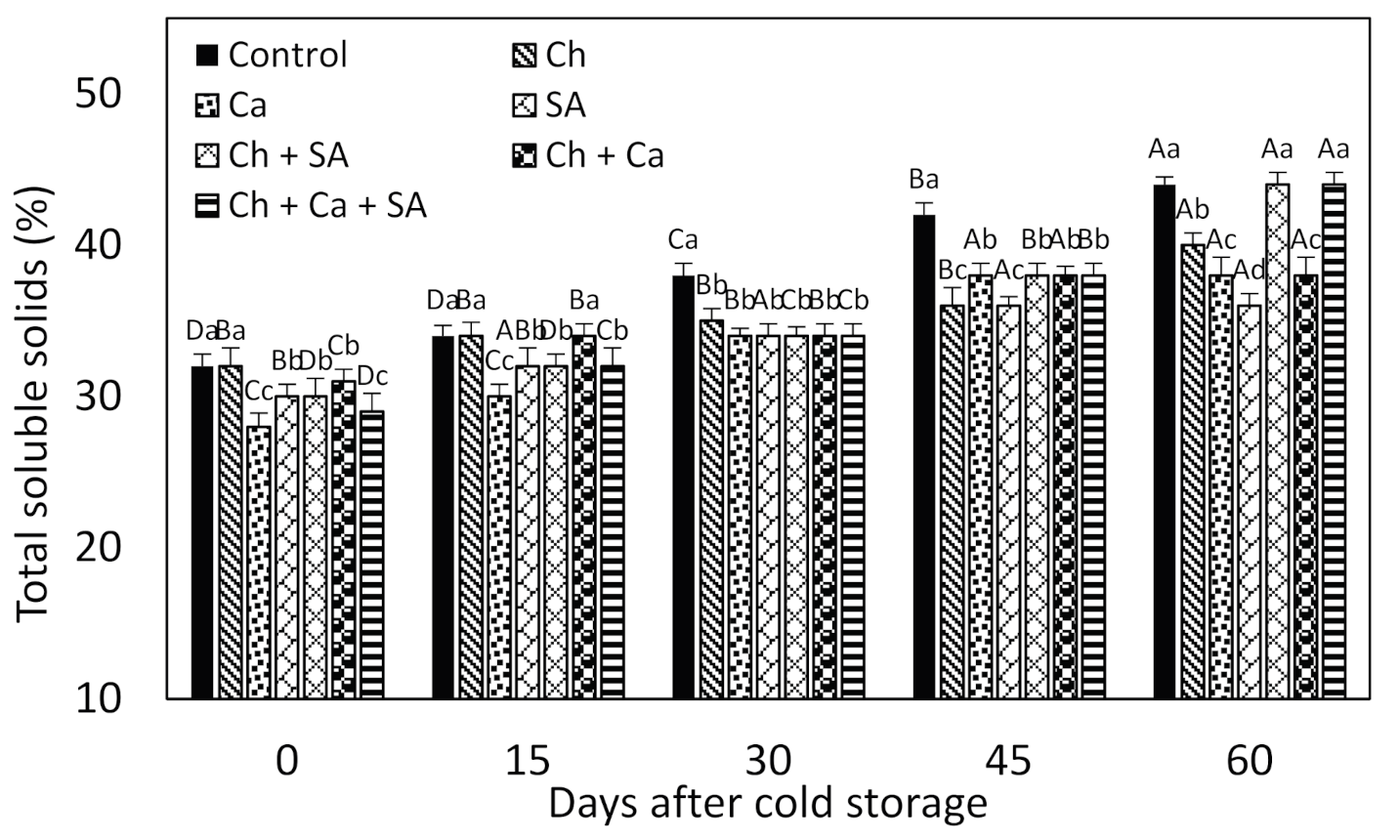
3.3. Total Soluble Solids (TSS)
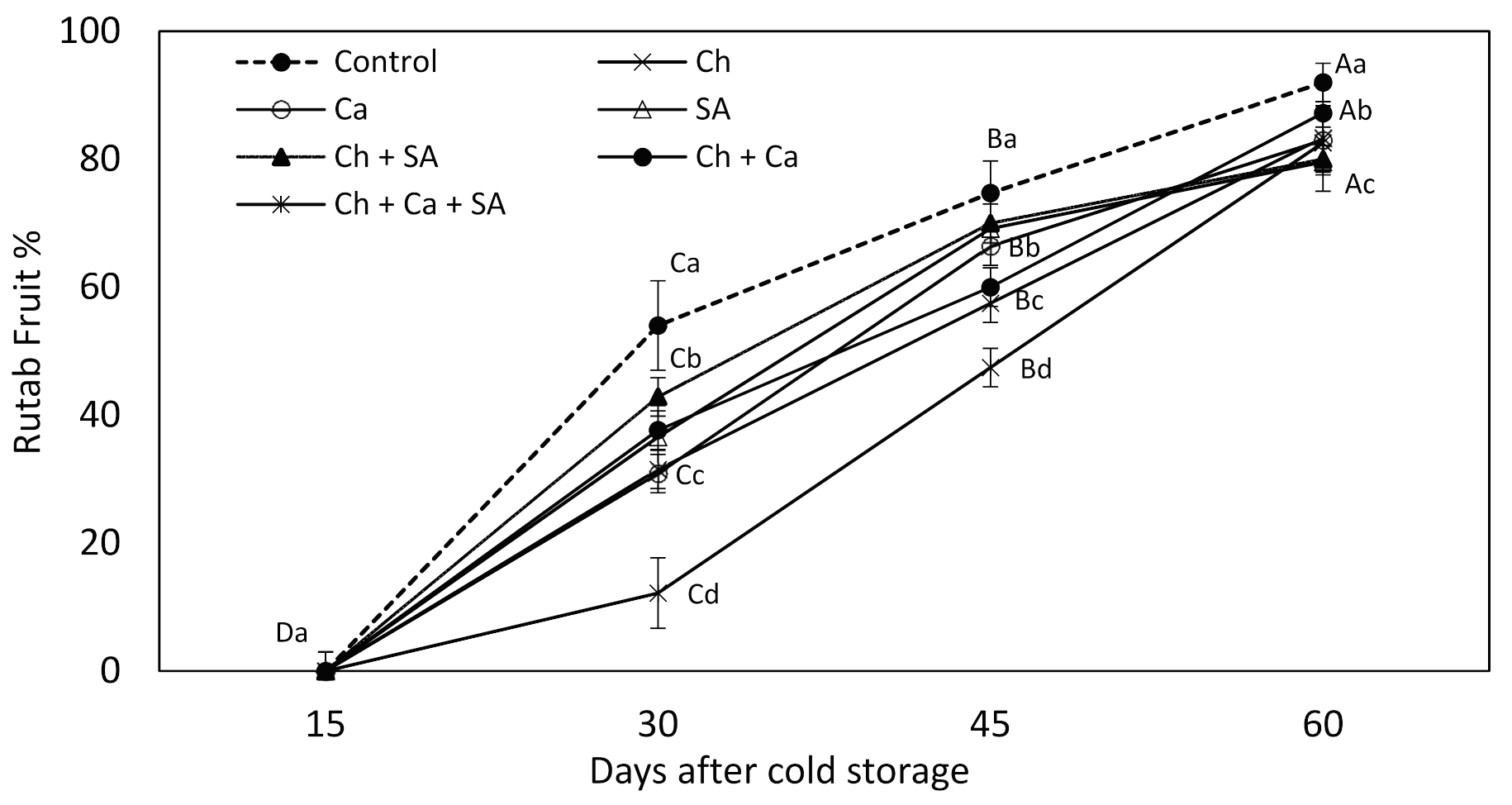
3.4. Fruit Ripening
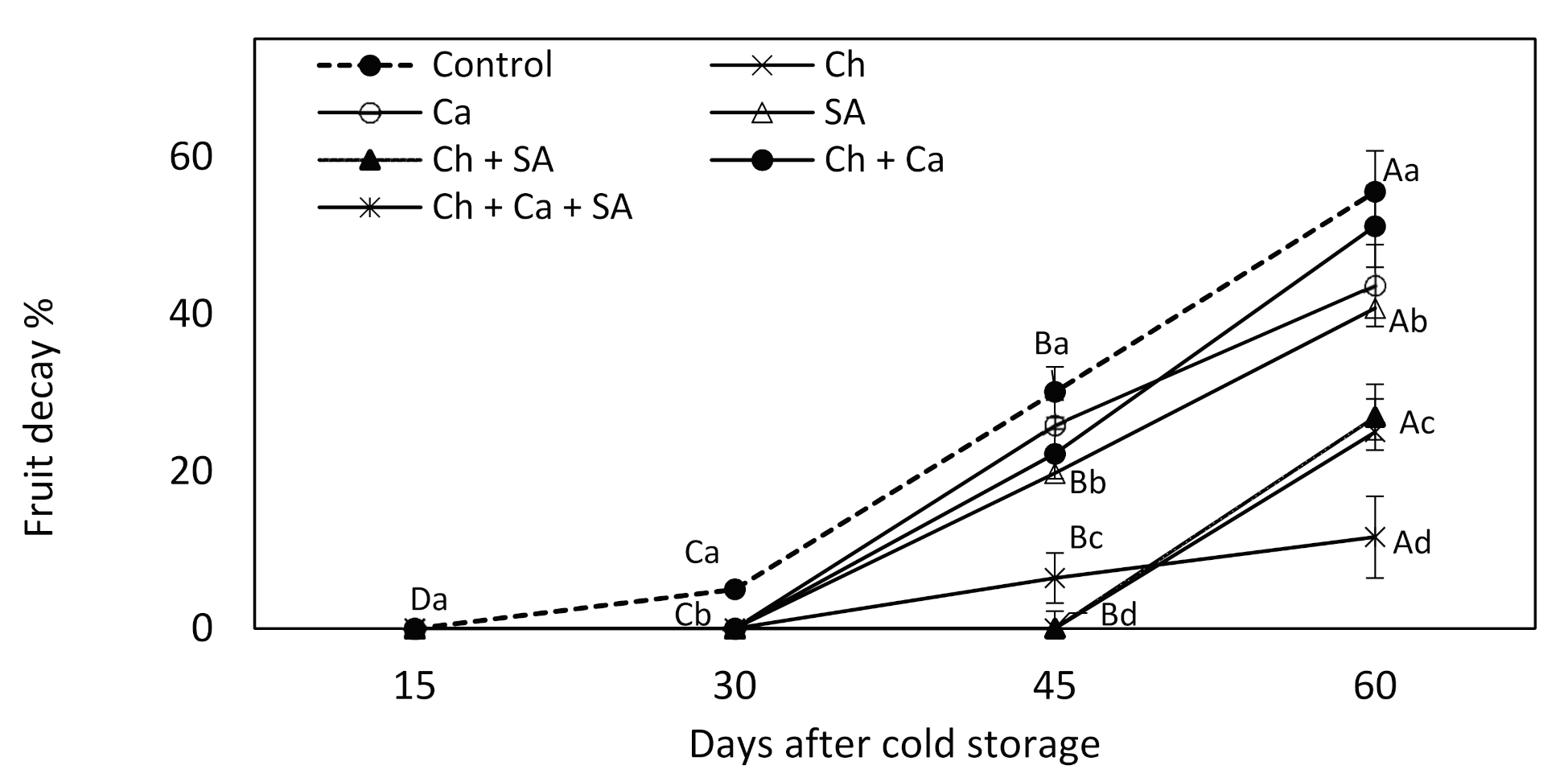
3.5. Fruit Decay during Storage
3.6. Color Attributes
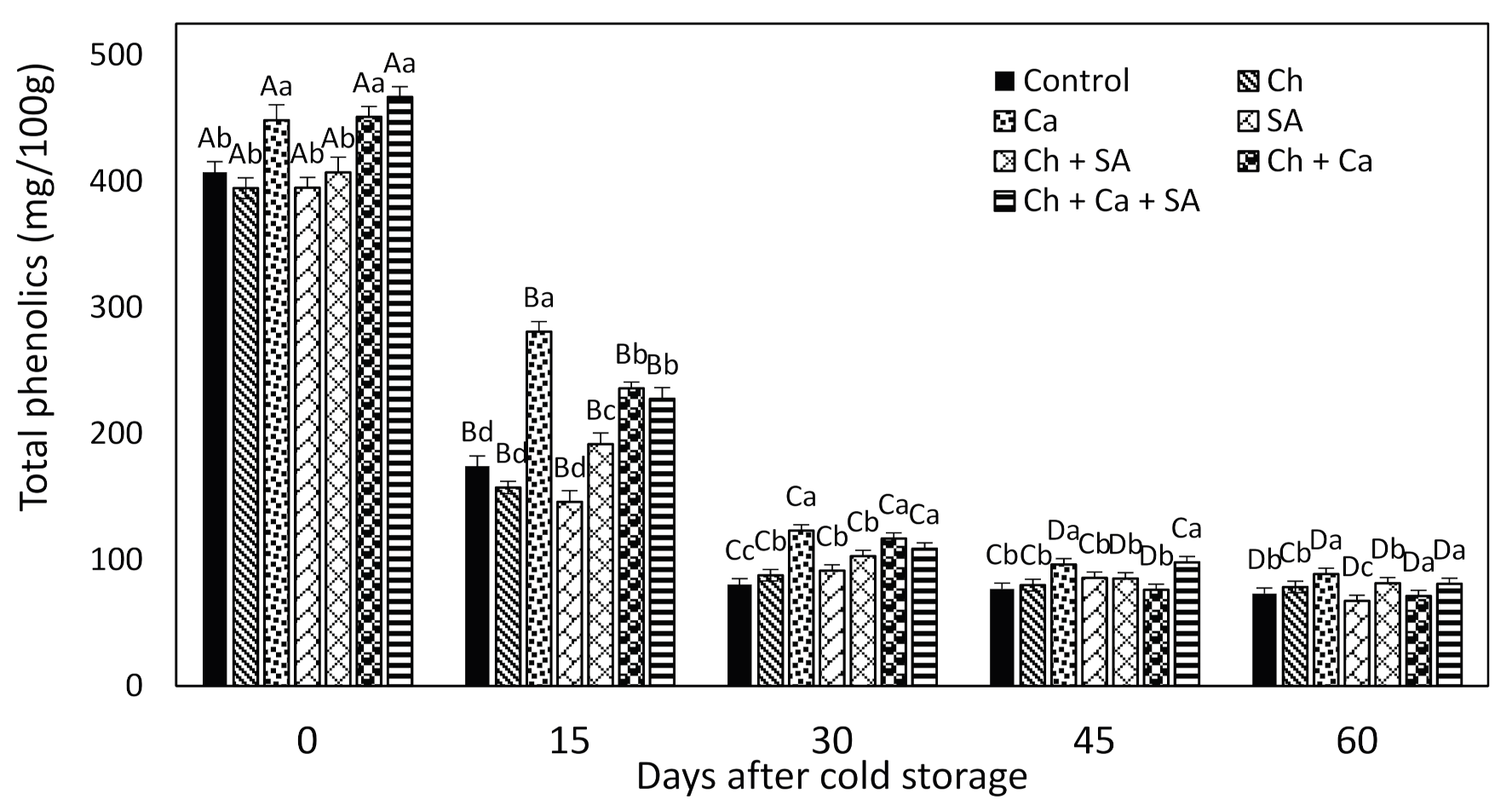
3.7. Total Phenolic Content of Fruit at Harvest and during Storage
3.8. Total Tannin, Flavonoids, and Antioxidants Concentrations at Harvest
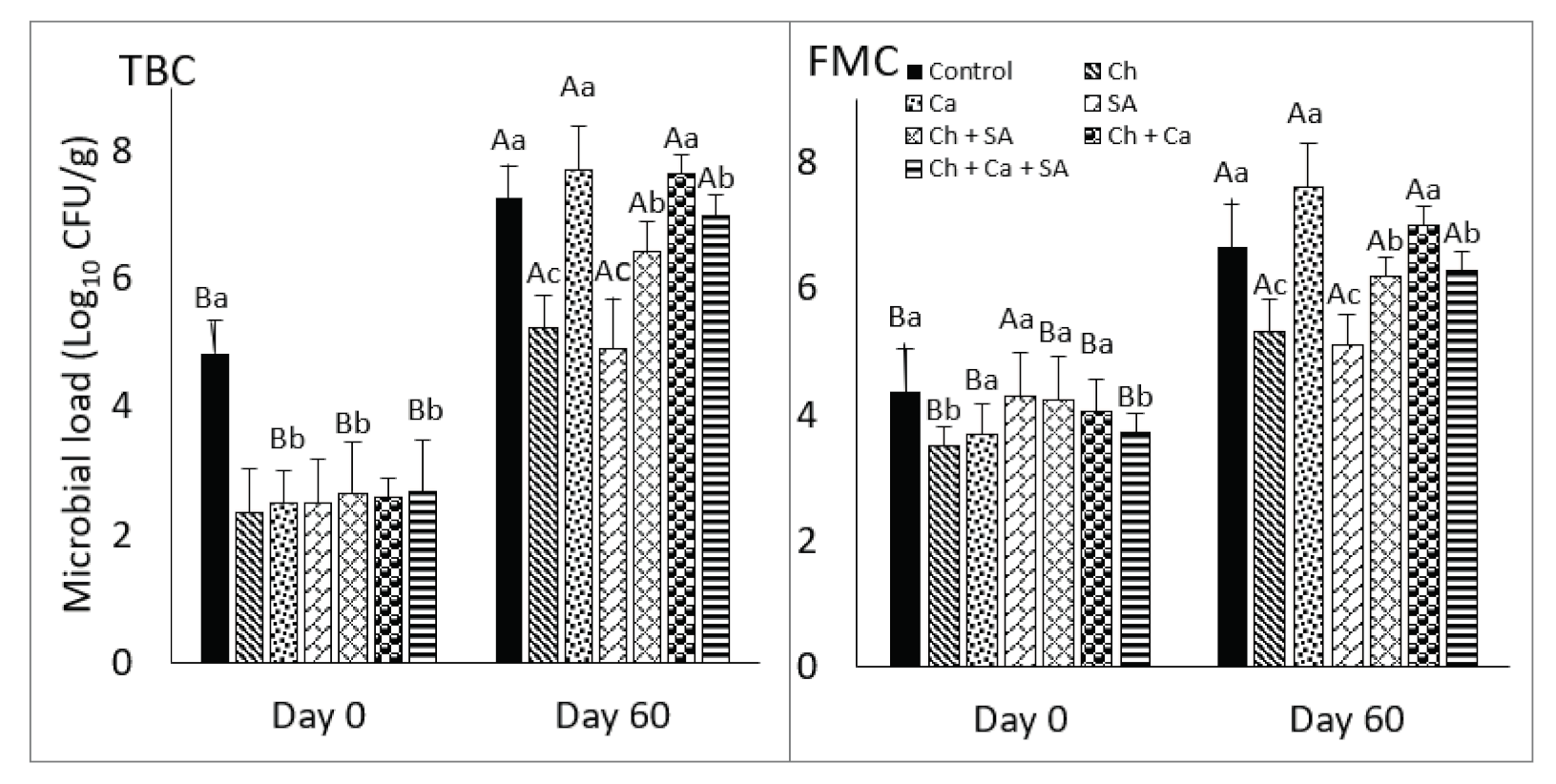
3.9. Microbiological Quality of Fruit
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mattar, M.A.; Soliman, S.S.; Al-Obeed, R.S. Effects of various quantities of three irrigation water types on yield and fruit quality of ‘Succary’ date palm. Agronomy 2021, 11, 796. [Google Scholar] [CrossRef]
- Gantait, S.; El-Dawayati, M.M.; Panigrahi, J.; Labrooy, C.; Verma, S.K. The retrospect and prospect of the applications of biotechnology in Phoenix dactylifera L. Appl. Microbiol. Biotechnol. 2018, 102, 8229–8259. [Google Scholar] [CrossRef] [PubMed]
- Al-Alawi, R.A.; Al-Mashiqri, J.H.; Al-Nadabi, J.S.M.; Al-Shihi, B.I.; Baqi, Y. Date Palm Tree (Phoenix dactylifera L.): Natural products and therapeutic options. Front. Plant Sci. 2017, 8, 845. [Google Scholar] [CrossRef] [PubMed]
- Al-Mssallem, M.Q.; Alqurashi, R.M.; Al-Khayri, J.M. Bioactive compounds of date palm (Phoenix dactylifera L.). In Bioactive Compounds in Underutilized Fruits and Nuts; Murthy, H.N., Bapat, V.A., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 91–105. [Google Scholar]
- Bentrad, N.; Hamida-Ferhat, A. Date palm fruit (Phoenix dactylifera): Nutritional values and potential benefits on health. In The Mediterranean Diet, 2nd ed.; Preedy, V.R., Watson, R.R., Eds.; Academic Press: Cambridge, MA, USA, 2020; pp. 239–255. [Google Scholar]
- Vayalil, P.K. Bioactive compounds, nutritional and functional properties of date fruit. In Dates: Postharvest Science, Processing Technology and Health Benefits; Siddiq, M., Salah, S.M., Kader, A.A., Eds.; John Wiley & Sons Ltd: Chichester, UK, 2013; pp. 285–303. [Google Scholar]
- Al-Qurashi, A.D.; Awad, M.A. Quality characteristics of bisir ‘Barhee’ dates during cold storage as affected by postharvest dipping in gibberellic acid, naphthaleneacetic acid and benzyladenine. Fruits 2011, 66, 343–352. [Google Scholar] [CrossRef]
- Awad, M.A.; Al-Qurashi, A.D.; Mohamed, S.A. Biochemical changes in fruit of an early and a late date palm cultivar during development and ripening. Int. J. Fruit Sci. 2011, 11, 167–183. [Google Scholar] [CrossRef]
- Abu-Shama, H.S.; Abou-Zaid, F.O.F.; El-Sayed, E.Z. Effect of using edible coatings on fruit quality of Barhi date cultivar. Sci. Hortic. 2020, 265, 109262. [Google Scholar] [CrossRef]
- Mohamed, S.A.; Awad, M.A.; Al-Qurashi, A.D. Antioxidant activity, antioxidant compounds, antioxidant and hydrolytic enzymes activities of ‘Barhee’ dates at harvest and during storage as affected by pre-harvest spray of some growth regulators. Sci. Hortic. 2014, 167, 91–99. [Google Scholar] [CrossRef]
- Alsawmahi, O.N.; Al-Juhaimi, F.; Alhamdan, A.M.; Ghafoor, K.; Adiamo, O.Q.; Mohamed Ahmed, I.A.; Hassan, B.H.; Ehmed, K.A.; Babiker, E.E.; Abdelkarim, D. Phenolic, tannin, antioxidant, color, and sensory attributes of Barhi date (Phoenix dactylifera) fruit stored in modified atmosphere packages. J. Food Biochem. 2018, 42, e12576. [Google Scholar] [CrossRef]
- Aleid, S.; Saikhan, M. Effect of permeable modified atmosphere packaging on quality and shelf life of fresh Khenaizy dates stored at low temperature. J. Food Nutr. Res. 2017, 5, 503–509. [Google Scholar]
- Alhamdan, A.M.; Elkhair, D.O.; Ehmed, K.A. Modeling of respiration rate of fresh date fruits (Barhi cultivar) under aerobic conditions. J. Adv. Agric. Technol. 2015, 2, 120–124. [Google Scholar] [CrossRef]
- Baloch, M.K.; Saleem, S.A.; Baloch, A.K.; Baloch, W.A. Impact of controlled atmosphere on the stability of Dhakki dates. LWT Food Sci. Tech. 2006, 39, 671–676. [Google Scholar] [CrossRef]
- Baenas, N.; García-Viguera, C.; Moreno, D.A. Elicitation: A tool for enriching the bioactive composition of foods. Molecules 2014, 19, 13541–13563. [Google Scholar] [CrossRef]
- Moreno-Escamilla, J.O.; Alvarez-Parrilla, E.; de la Rosa, L.A.; Núñez-Gastélum, J.A.; González-Aguilar, G.A.; Rodrigo-García, J. Effect of elicitors in the nutritional and sensorial quality of fruits and vegetables. In Preharvest Modulation of Postharvest Fruit and Vegetable Quality; Siddiqui, M.W., Ed.; Academic Press: Cambridge, MA, USA, 2018; pp. 71–91. [Google Scholar]
- Rahman, M.; Mukta, J.A.; Sabir, A.A.; Gupta, D.R.; Mohi-Ud-Din, M.; Hasanuzzaman, M.; Miah, M.G.; Rahman, M.; Islam, M.T. Chitosan biopolymer promotes yield and stimulates accumulation of antioxidants in strawberry fruit. PLoS ONE 2018, 13, e0203769. [Google Scholar] [CrossRef]
- Romanazzi, G.; Feliziani, E.; Santini, M.; Landi, L. Effectiveness of postharvest treatment with chitosan and other resistance inducers in the control of storage decay of strawberry. Postharvest Biol. Technol. 2013, 75, 24–27. [Google Scholar] [CrossRef]
- Romanazzi, G.; Feliziani, E.; Sivakumar, D. Chitosan, a biopolymer with triple action on postharvest decay of fruit and vegetables: Eliciting, antimicrobial and film-forming properties. Front. Microbiol. 2018, 9, 2745. [Google Scholar] [CrossRef]
- Shen, Y.; Yang, H. Effect of preharvest chitosan-g-salicylic acid treatment on postharvest table grape quality, shelf life, and resistance to Botrytis cinerea-induced spoilage. Sci. Hortic. 2017, 224, 367–373. [Google Scholar] [CrossRef]
- Chen, Y.; Sun, J.; Lin, H.; Lin, M.; Lin, Y.; Wang, H.; Hung, Y.C. Salicylic acid treatment suppresses Phomopsis longanae Chi-induced disease development of postharvest longan fruit by modulating membrane lipid metabolism. Postharvest Biol. Technol. 2020, 164, 111168. [Google Scholar] [CrossRef]
- Gomes, E.P.; Vanz Borges, C.; Monteiro, G.C.; Filiol Belin, M.A.; Minatel, I.O.; Pimentel Junior, A.; Tecchio, M.A.; Lima, G.P.P. Preharvest salicylic acid treatments improve phenolic compounds and biogenic amines in ‘Niagara Rosada’ table grape. Postharvest Biol. Technol. 2021, 176, 111505. [Google Scholar] [CrossRef]
- Luo, Z.; Chen, C.; Xie, J. Effect of salicylic acid treatment on alleviating postharvest chilling injury of ‘Qingnai’ plum fruit. Postharvest Biol. Technol. 2011, 62, 115–120. [Google Scholar] [CrossRef]
- Sayyari, M.; Babalar, M.; Kalantari, S.; Serrano, M.; Valero, D. Effect of salicylic acid treatment on reducing chilling injury in stored pomegranates. Postharvest Biol. Technol. 2009, 53, 152–154. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, K.; Zhang, S.; Ferguson, I. The role of salicylic acid in postharvest ripening of kiwifruit. Postharvest Biol. Technol. 2003, 28, 67–74. [Google Scholar] [CrossRef]
- Atia, A.; Abdelkarim, D.; Younis, M.; Alhamdan, A. Effects of calcium chloride and salicylic acid postharvest treatments on the quality of Khalal Barhi dates at different ripening levels during cold storage. J. Food Meas. Charact. 2018, 12, 1156–1166. [Google Scholar] [CrossRef]
- Irfan, P.K.; Vanjakshi, V.; Prakash, M.N.K.; Ravi, R.; Kudachikar, V.B. Calcium chloride extends the keeping quality of fig fruit (Ficus carica L.) during storage and shelf-life. Postharvest Biol. Technol. 2013, 82, 70–75. [Google Scholar] [CrossRef]
- Sohail, M.; Ayub, M.; Khalil, S.A.; Zeb, A.; Ullah, F.; Afridi, S.R.; Ullah, R. Effect of calcium chloride treatment on post harvest quality of peach fruit during cold storage. Int. Food Res. J. 2015, 22, 2225–2229. [Google Scholar]
- Ruiz-García, Y.; Gómez-Plaza, E. Elicitors: A tool for improving fruit phenolic content. Agriculture 2013, 3, 33–52. [Google Scholar] [CrossRef]
- Awad, M.A.; Al-Qurashi, A.D. Gibberellic acid spray and bunch bagging increase bunch weight and improve fruit quality of ‘Barhee’ date palm cultivar under hot arid conditions. Sci. Hortic. 2012, 138, 96–100. [Google Scholar] [CrossRef]
- Rastegar, S.; Rahemi, M.; Baghizadeh, A.; Gholami, M. Enzyme activity and biochemical changes of three date palm cultivars with different softening pattern during ripening. Food Chem. 2012, 134, 1279–1286. [Google Scholar] [CrossRef]
- Kumar, D.; Mishra, D.S.; Chakraborty, B.; Kumar, P. Pericarp browning and quality management of litchi fruit by antioxidants and salicylic acid during ambient storage. J. Food Sci. 2013, 50, 797–802. [Google Scholar] [CrossRef]
- Ferrer, A.; Remón, S.; Negueruela, A.I.; Oria, R. Changes during the ripening of the very late season Spanish peach cultivar Calanda: Feasibility of using CIELAB coordinates as maturity indices. Sci. Hortic. 2005, 105, 435–446. [Google Scholar] [CrossRef]
- Maskan, M. Kinetics of colour change of kiwifruits during hot air and microwave drying. J. Food Eng. 2001, 48, 169–175. [Google Scholar] [CrossRef]
- Feliziani, E.; Landi, L.; Romanazzi, G. Preharvest treatments with chitosan and other alternatives to conventional fungicides to control postharvest decay of strawberry. Carbohydr. Polym. 2015, 132, 111–117. [Google Scholar] [CrossRef]
- Velioglu, Y.S.; Mazza, G.; Gao, L.; Oomah, B.D. Antioxidant activity and total phenolics in selected fruits, vegetables, and grain products. J. Agric. Food Chem. 1998, 46, 4113–4117. [Google Scholar] [CrossRef]
- Bentebba, F.Z.; Zineb, G.; Saidi, M.; Bensaci, C. Effects of development and ripening stage on phytochemical compositions, antioxidant and antibacterial activities of date palm fruits. Asian J. Chem. 2020, 32, 415–419. [Google Scholar] [CrossRef]
- Kim, D.O.; Chun, O.K.; Kim, Y.J.; Moon, H.Y.; Lee, C.Y. Quantification of polyphenolics and their antioxidant capacity in fresh plums. J. Agric. Food Chem. 2003, 51, 6509–6515. [Google Scholar] [CrossRef]
- Abd Elwahab, S.A.; Allatif, A.M.A.; Farid, M.A.; Soliman, S.M. Effect of safe post-harvest alternatives on quality and storage life of “barhi” date palm. Plant Arch. 2019, 19, 3937–3945. [Google Scholar]
- Kassem, H.; Alobeed, R.; Ahmed, M. Extending harvest season, improving fruit quality and shelf life of ‘Barhee’ date palm by preharvest sprays. Acta Hortic. 2010, 882, 147–154. [Google Scholar] [CrossRef]
- Shiri, M.A.; Bakhshi, D.; Ghasemnezhad, M.; Dadi, M.; Papachatzis, A.; Kalorizou, H. forestry. Chitosan coating improves the shelf life and postharvest quality of table grape (Vitis vinifera) cultivar Shahroudi. Turkish J. Agric. 2013, 37, 148–156. [Google Scholar]
- Petriccione, M.; De Sanctis, F.; Pasquariello, M.S.; Mastrobuoni, F.; Rega, P.; Scortichini, M.; Mencarelli, F. The effect of chitosan coating on the quality and nutraceutical traits of sweet cherry during postharvest life. Food and Bioproc. Technol. 2015, 8, 394–408. [Google Scholar] [CrossRef]
- Petriccione, M.; Mastrobuoni, F.; Pasquariello, M.S.; Zampella, L.; Nobis, E.; Capriolo, G.; Scortichini, M. Effect of chitosan coating on the postharvest quality and antioxidant enzyme system response of strawberry fruit during cold storage. Foods 2015, 4, 501–523. [Google Scholar] [CrossRef]
- Atia, A.; Abdelkarim, D.; Younis, M.; Alhamdan, A.; Engineering, B. Effects of pre-storage dipping in calcium chloride and salicylic acid on the quality attributes of stored Khalal Barhi dates. Int. J. Agric. 2020, 13, 206–212. [Google Scholar] [CrossRef]
- Ennab, H.A.; El-Shemy, M.A.; Alam-Eldein, S.M. Salicylic acid and putrescine to reduce post-harvest storage problems and maintain quality of Murcott Mandarin fruit. Agronomy 2020, 10, 115. [Google Scholar] [CrossRef]
- Alqarni, S.; Bazzi, M.D. Evaluating the Phoenix dactylifera L. Biochemical changes associated with its maturation. J. Agric. Sci. 2019, 11, 614–624. [Google Scholar] [CrossRef]
- Hazbavi, I.; Khoshtaghaza, M.H.; Mostaan, A.; Banakar, A. Effect of postharvest hot-water and heat treatment on quality of date palm (cv. Stamaran). J. Saudi Soc. Agric. Sci. 2015, 14, 153–159. [Google Scholar] [CrossRef]
- Pila, N.; Gol, N.B.; Rao, T.V.R. Effect of post harvest treatments on physicochemical characteristics and shelf life of tomato ( Lycopersicon esculentum Mill ) fruits during storage. Am. Eurasian J. Agric. Environ. Sci. 2010, 9, 470–479. [Google Scholar]
- Shafiee, M.; Taghavi, T.S.; Babalar, M. Addition of salicylic acid to nutrient solution combined with postharvest treatments (hot water, salicylic acid, and calcium dipping) improved postharvest fruit quality of strawberry. Sci. Hortic. 2010, 124, 40–45. [Google Scholar] [CrossRef]
- Tian, S.; Wan, Y.; Qin, G.; Xu, Y. Induction of defense responses against Alternaria rot by different elicitors in harvested pear fruit. Appl. Microbiol. Biotechnol. 2006, 70, 729–734. [Google Scholar] [CrossRef]
- Wang, Y.Y.; Li, B.Q.; Qin, G.Z.; Li, L.; Tian, S.P. Defense response of tomato fruit at different maturity stages to salicylic acid and ethephon. Sci. Hortic. 2011, 129, 183–188. [Google Scholar] [CrossRef]
- FernÁndez-VÁzquez, R.; Stinco, C.M.; MelÉndez-MartÍnez, A.J.; Heredia, F.J.; Vicario, I.M. Visual and instrumental evaluation of orange juice color: A consumers’preference study. J. Sens. Stud. 2011, 26, 436–444. [Google Scholar] [CrossRef]
- Molamohammadi, H.; Pakkish, Z.; Akhavan, H.R.; Saffari, V.R. Effect of salicylic acid incorporated chitosan coating on shelf life extension of fresh In-Hull Pistachio fruit. Food Bioproc. Technol. 2020, 13, 121–131. [Google Scholar] [CrossRef]
- Awad, M.A.; Al-Qurashi, A.D.; Mohamed, S.A. Antioxidant capacity, antioxidant compounds and antioxidant enzyme activities in five date cultivars during development and ripening. Sci. Hortic. 2011, 129, 688–693. [Google Scholar] [CrossRef]
- Mortazavi, S.M.H.; Azizollahi, F.; Moallemi, N. Some quality attributes and biochemical properties of nine iranian date (Phoenix dactylifera L.) cultivars at different stages of fruit development. Int. J. Hort. Sci. Technol 2015, 2, 161–171. [Google Scholar]
- Wang, L.; Chen, S.; Kong, W.; Li, S.; Archbold, D.D. Salicylic acid pretreatment alleviates chilling injury and affects the antioxidant system and heat shock proteins of peaches during cold storage. Postharvest Biol. Technol. 2006, 41, 244–251. [Google Scholar] [CrossRef]
- Sathiyabama, M.; Akila, G.; Charles, R.E. Chitosan-induced defence responses in tomato plants against early blight disease caused by Alternaria solani (Ellis and Martin) Sorauer. Arch. Phytopathol. Plant Protect. 2014, 47, 1963–1973. [Google Scholar] [CrossRef]
| Treatment | Chemical | Application |
|---|---|---|
| Control | Water | Water |
| Ch | Chitosan | 1% |
| Ca | Calcium chloride | 2 mM |
| SA | Salicylic acid | 3% |
| Ch+SA | Chitosan+Salicylic acid | 1:1, v/v |
| Ch+Ca | Chitosan+Calcium chloride | 1:1, v/v |
| Ch+Ca+SA | Chitosan+Calcium chloride+Salicylic acid | 1:1:1, v/v/v |
| Fruit Weight (g) | Fruit Length (mm) | Fruit Width (mm) | TPC (mg/100 g) | TTC (mg/100 g) | TFC (mg/100 g) | IC50 (mg/mL) | ABTS (mg/g) | |
|---|---|---|---|---|---|---|---|---|
| Control | 10.2 ± 0.11 b | 35.0 ± 0.38 b | 23.5 ± 0.24 b | 407.4 ± 12.2 c | 73.9 ± 0.75 c | 71.5 ± 0.34 bc | 1.7 ± 0.03 b | 1.9 ± 0.07 b |
| Ch | 10.9 ± 0.22 b | 35.2 ± 0.36 b | 24.1 ± 0.27 ab | 394.6 ± 8.89 c | 42.0 ± 0.43 e | 87.8 ± 0.42 b | 3.3 ± 0.10 a | 1.6 ± 0.11 c |
| Ca | 10.0 ± 0.22 b | 34.7 ± 0.38 b | 23.9 ± 0.23 b | 448.5 ± 11.3 b | 61.9 ± 0.68 d | 67.9 ± 0.22 c | 1.7 ± 0.06 b | 1.5 ± 0.09 c |
| SA | 11.8 ± 0.25 a | 36.7 ± 0.40 a | 24.2 ± 0.25 ab | 394.9 ± 10.7 c | 47.8 ± 0.26 e | 66.4 ± 0.31 c | 2.1 ± 0.07 b | 1.3 ± 0.17 d |
| Ch + SA | 12.4 ± 0.46 a | 37.2 ± 0.48 a | 25.6 ± 0.32 a | 407.2 ± 13.2 c | 59.5 ± 0.45 d | 86.6 ± 0.49 b | 1.8 ± 0.05 ab | 1.9 ± 0.18 b |
| Ch + Ca | 10.8 ± 0.39 b | 34.9 ± 0.51 b | 24.2 ± 0.34 ab | 451.1 ± 13.4 b | 80.7 ± 0.64 b | 82.9 ± 0.54 b | 1.8 ± 0.04 ab | 1.2 ± 0.12 d |
| Ch + Ca + SA | 10.6 ± 0.16 b | 35.2 ± 0.33 b | 24.2 ± 0.21 ab | 486.8 ± 12.7 a | 116.2 ± 0.73 a | 91.4 ± 0.41 a | 1.5 ± 0.04 b | 3.2 ± 0.13 a |
| Storage Time | L* | h0 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Control | Ch | Ca | SA | Ch+SA | Ch+Ca | Ch+Ca+SA | Control | Ch | Ca | SA | Ch+SA | Ch+Ca | Ch+Ca+SA | |
| Day 0 | 33.1 ± 0.44b | 33.1 ± 0.36b | 32.6 ± 0.58b | 31.2 ± 0.52b | 33.4 ± 0.83b | 37.6 ± 0.67a | 37.7 ± 0.86a | 27.7 ± 0.34a | 26.0 ± 0.36b | 24.9 ± 0.38c | 23.8 ± 0.37c | 27.3 ± 0.47a | 24.2 ± 0.32c | 27.4 ± 0.41a |
| Day 15 | 27.4 ± 0.54b | 29.5 ± 0.38a | 29.8 ± 0.64a | 27.9 ± 0.51b | 29.6 ± 0.54a | 29.6 ± 0.70a | 30.8 ± 0.91a | 28.9 ± 0.46b | 30.3 ± 0.45a | 26.8 ± 0.31d | 25.4 ± 0.33d | 27.8 ± 0.39c | 26.1 ± 0.46d | 29.9 ± 0.49a |
| Day 30 | 25.2 ± 0.59b | 26.7 ± 0.64b | 28.4 ± 0.85a | 26.0 ± 0.91b | 28.5 ± 0.78a | 27.1 ± 0.94a | 27.4 ± 0.89a | 38.1 ± 0.51a | 35.9 ± 0.53b | 29.9 ± 0.45d | 28.8 ± 0.44d | 34.2 ± 0.84c | 34.5 ± 0.91c | 38.7 ± 0.81a |
| Day 45 | 22.0 ± 0.32c | 23.4 ± 0.43b | 25.2 ± 0.69a | 22.9 ± 0.47bc | 23.2 ± 0.60b | 24.6 ± 0.48a | 24.1 ± 0.44a | 38.0 ± 0.53b | 38.3 ± 0.67b | 35.8 ± 0.74c | 34.4 ± 0.68c | 37.5 ± 0.73b | 38.3 ± 0.94b | 40.6 ± 0.90a |
| Day 60 | 21.2 ± 0.52b | 21.7 ± 0.43b | 23.7 ± 0.88a | 21.2 ± 0.44b | 21.7 ± 0.36b | 23.3 ± 0.67a | 22.2 ± 0.41b | 41.9 ± 0.46a | 41.2 ± 0.66a | 37.1 ± 0.62c | 36.8 ± 0.41c | 39.4 ± 0.45b | 39.4 ± 0.82b | 42.2 ± 0.44a |
| C* | ΔE | |||||||||||||
| Control | Ch | Ca | SA | Ch+SA | Ch+Ca | Ch+Ca+SA | Control | Ch | Ca | SA | Ch+SA | Ch+Ca | Ch+Ca+SA | |
| Day 0 | 30.7 ± 0.47bc | 31.8 ± 0.82b | 29.9 ± 0.39c | 28.7 ± 0.38c | 32.8 ± 0.76b | 32.6 ± 0.56b | 34.3 ± 0.714a | 0e | 1.4 ± 0.06d | 1.7 ± 0.04d | 3.4 ± 0.13b | 2.3 ± 0.07c | 5.3 ± 0.10a | 5.9 ± 0.16a |
| Day 15 | 27.4 ± 0.56c | 29.8 ± 0.46b | 29.4 ± 0.44b | 28.1 ± 0.41bc | 32.1 ± 0.54a | 27.6 ± 0.42c | 28.6 ± 0.43b | 6.7 ± 0.14a | 3.9 ± 0.08bc | 3.5 ± 0.08c | 5.8 ± 0.15b | 3.7 ± 0.09c | 4.7 ± 0.08b | 3.3 ± 0.05c |
| Day 30 | 10.4 ± 0.23d | 12.8 ± 0.21c | 15.6 ± 0.24b | 13.1 ± 0.25bc | 18.4 ± 0.23a | 11.8 ± 0.18c | 14.4 ± 0.22b | 21.9 ± 0.36a | 19.1 ± 0.33b | 15.8 ± 0.15c | 18.9 ± 0.31b | 13.4 ± 0.23d | 19.9 ± 0.34b | 17.7 ± 0.24b |
| Day 45 | 6.6 ± 0.12c | 7.5 ± 0.11b | 8.7 ± 0.13a | 6.1 ± 0.11c | 7.3 ± 0.12b | 5.8 ± 0.14c | 8.9 ± 0.12a | 26.5 ± 034a | 25.3 ± 0.46a | 23.5 ± 0.37b | 26.6 ± 0.41a | 25.5 ± 0.53a | 26.4 ± 0.43a | 23.8 ± 0.35b |
| Day 60 | 5.2 ± 0.14a | 5.7 ± 0.9a | 5.8 ± 0.11a | 3.7 ± 0.08b | 4.6 ± 0.10a | 4.9 ± 0.05a | 4.9 ± 0.09a | 28.2 ± 0.57a | 27.5 ± 0.72b | 26.6 ± 0.51b | 29.5 ± 0.64a | 28.5 ± 0.69a | 28.8 ± 0.47a | 27.7 ± 0.42ab |
| Traits | TSS | TPC | Rutab % | Decay % | Weight Loss % | L* | ΔE | C* |
|---|---|---|---|---|---|---|---|---|
| TPC | −0.73 *** | |||||||
| Rutab % | 0.74 *** | −0.92 *** | ||||||
| Decay % | 0.61 *** | −0.42 *** | 0.56 *** | |||||
| Weight loss % | 0.52 *** | −0.77 *** | 0.65 *** | 0.43 *** | ||||
| L* | −0.67 *** | 0.85 *** | −0.91 *** | −0.39 *** | −0.60 *** | |||
| ΔE | 0.68 *** | −0.81 *** | 0.88 *** | 0.47 *** | 0.55 *** | −0.96 *** | ||
| C* | −0.63 *** | 0.72 *** | −0.85 *** | −0.49 *** | −0.52 *** | 0.93 *** | −0.94 *** | |
| h o | −0.67 *** | 0.81 *** | −0.89 *** | −0.55 *** | −0.63 *** | 0.92 *** | −0.90 *** | 0.91 *** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmed, Z.F.R.; Alblooshi, S.S.N.A.; Kaur, N.; Maqsood, S.; Schmeda-Hirschmann, G. Synergistic Effect of Preharvest Spray Application of Natural Elicitors on Storage Life and Bioactive Compounds of Date Palm (Phoenix dactylifera L., cv. Khesab). Horticulturae 2021, 7, 145. https://doi.org/10.3390/horticulturae7060145
Ahmed ZFR, Alblooshi SSNA, Kaur N, Maqsood S, Schmeda-Hirschmann G. Synergistic Effect of Preharvest Spray Application of Natural Elicitors on Storage Life and Bioactive Compounds of Date Palm (Phoenix dactylifera L., cv. Khesab). Horticulturae. 2021; 7(6):145. https://doi.org/10.3390/horticulturae7060145
Chicago/Turabian StyleAhmed, Zienab F. R., Shamsa S. N. A. Alblooshi, Navjot Kaur, Sajid Maqsood, and Guillermo Schmeda-Hirschmann. 2021. "Synergistic Effect of Preharvest Spray Application of Natural Elicitors on Storage Life and Bioactive Compounds of Date Palm (Phoenix dactylifera L., cv. Khesab)" Horticulturae 7, no. 6: 145. https://doi.org/10.3390/horticulturae7060145
APA StyleAhmed, Z. F. R., Alblooshi, S. S. N. A., Kaur, N., Maqsood, S., & Schmeda-Hirschmann, G. (2021). Synergistic Effect of Preharvest Spray Application of Natural Elicitors on Storage Life and Bioactive Compounds of Date Palm (Phoenix dactylifera L., cv. Khesab). Horticulturae, 7(6), 145. https://doi.org/10.3390/horticulturae7060145







