Living Mulch with Selected Herbs for Soil Management in Organic Apple Orchards
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Sites and Management Practices
2.2. Weed Biodiversity Assessment
2.3. Root Density and Morphological Traits
2.4. Leaf Chlorophyll Content and Nutrient Analysis
2.5. Statistical Analysis
3. Results
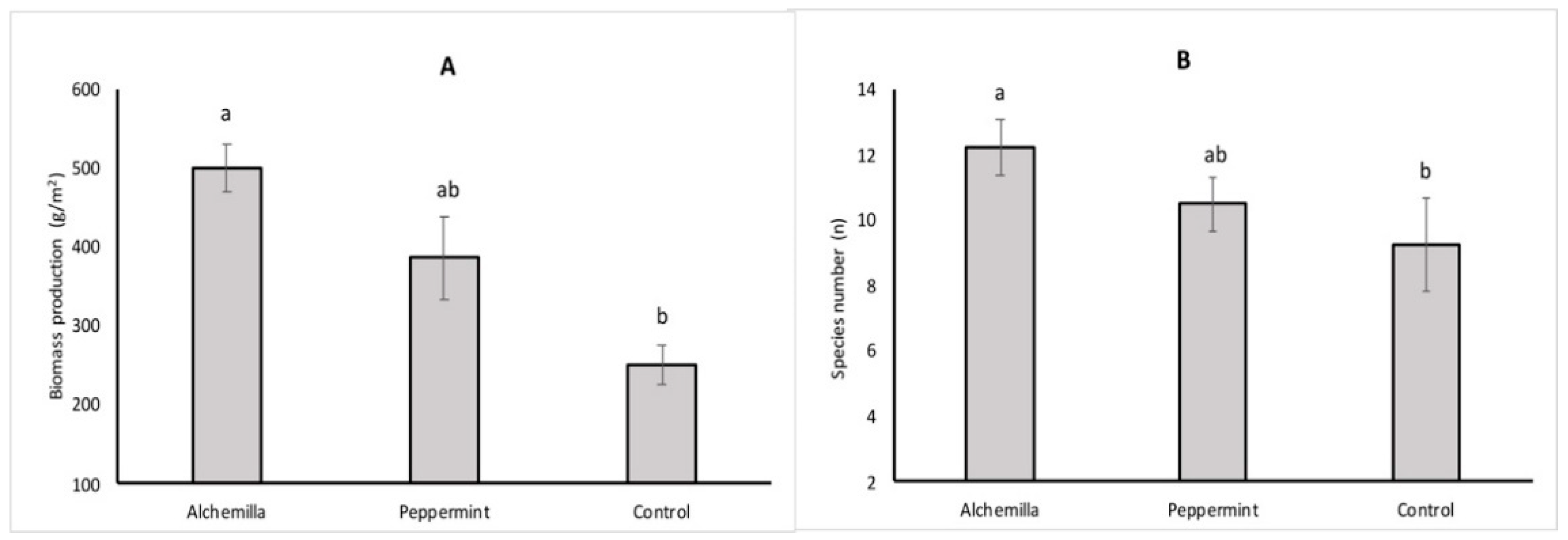
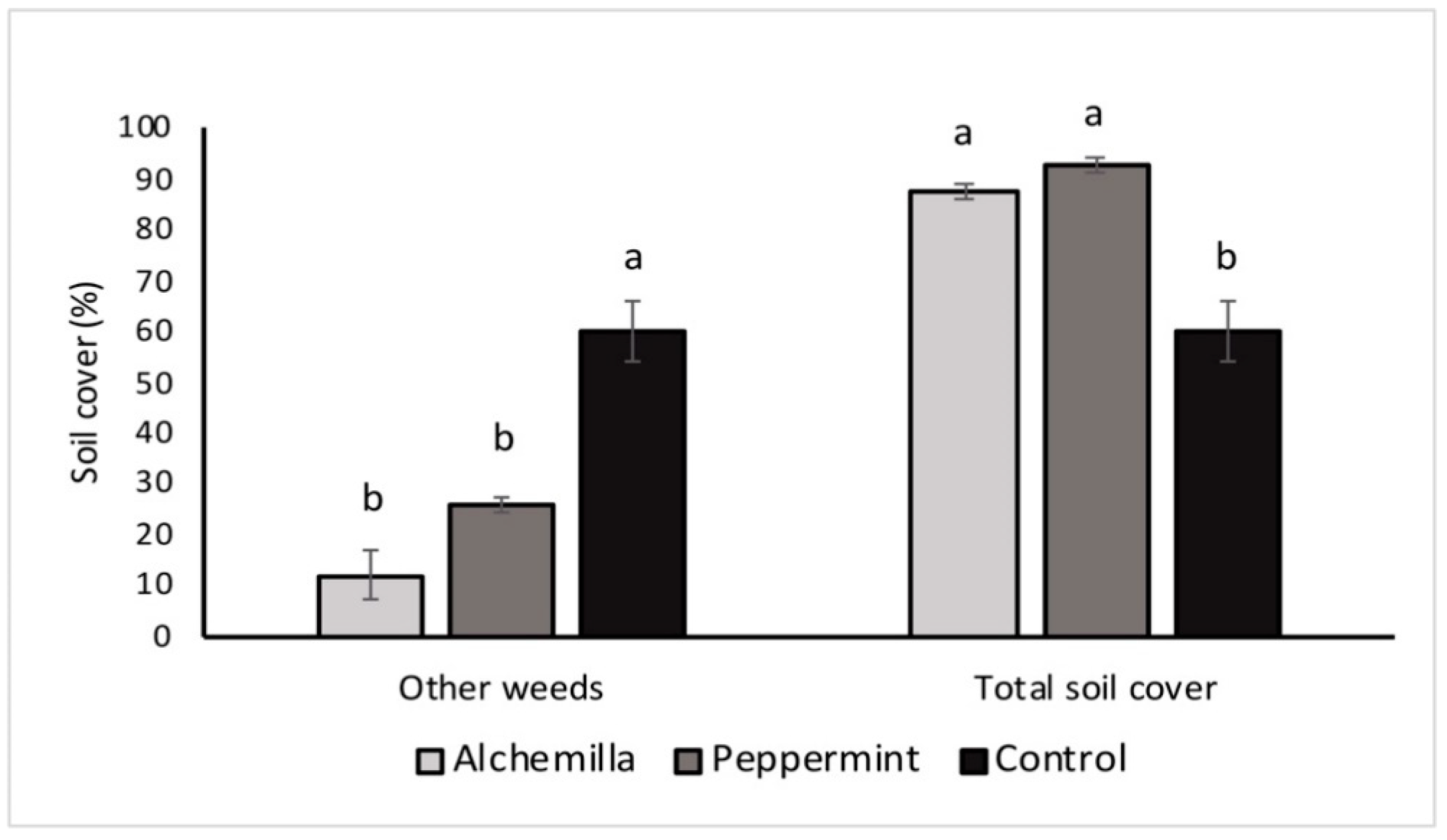
3.1. Weed Biodiversity
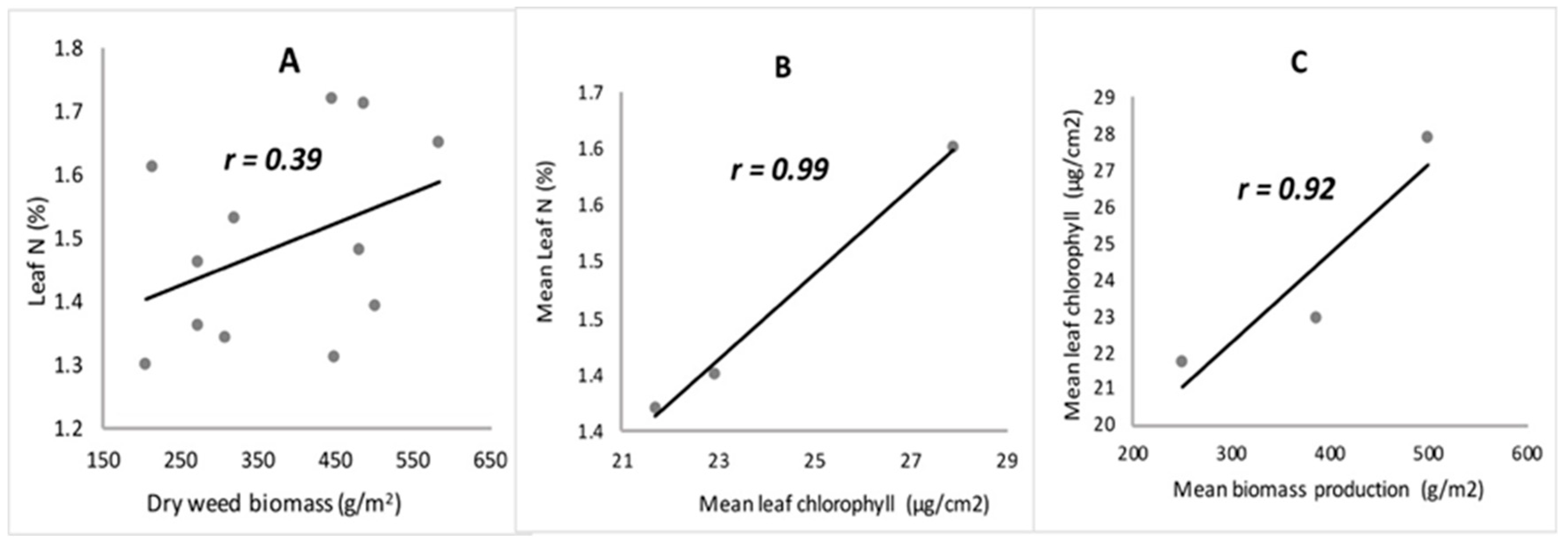
3.2. Leaf Nutrients Analysis and Chlorophyll Content
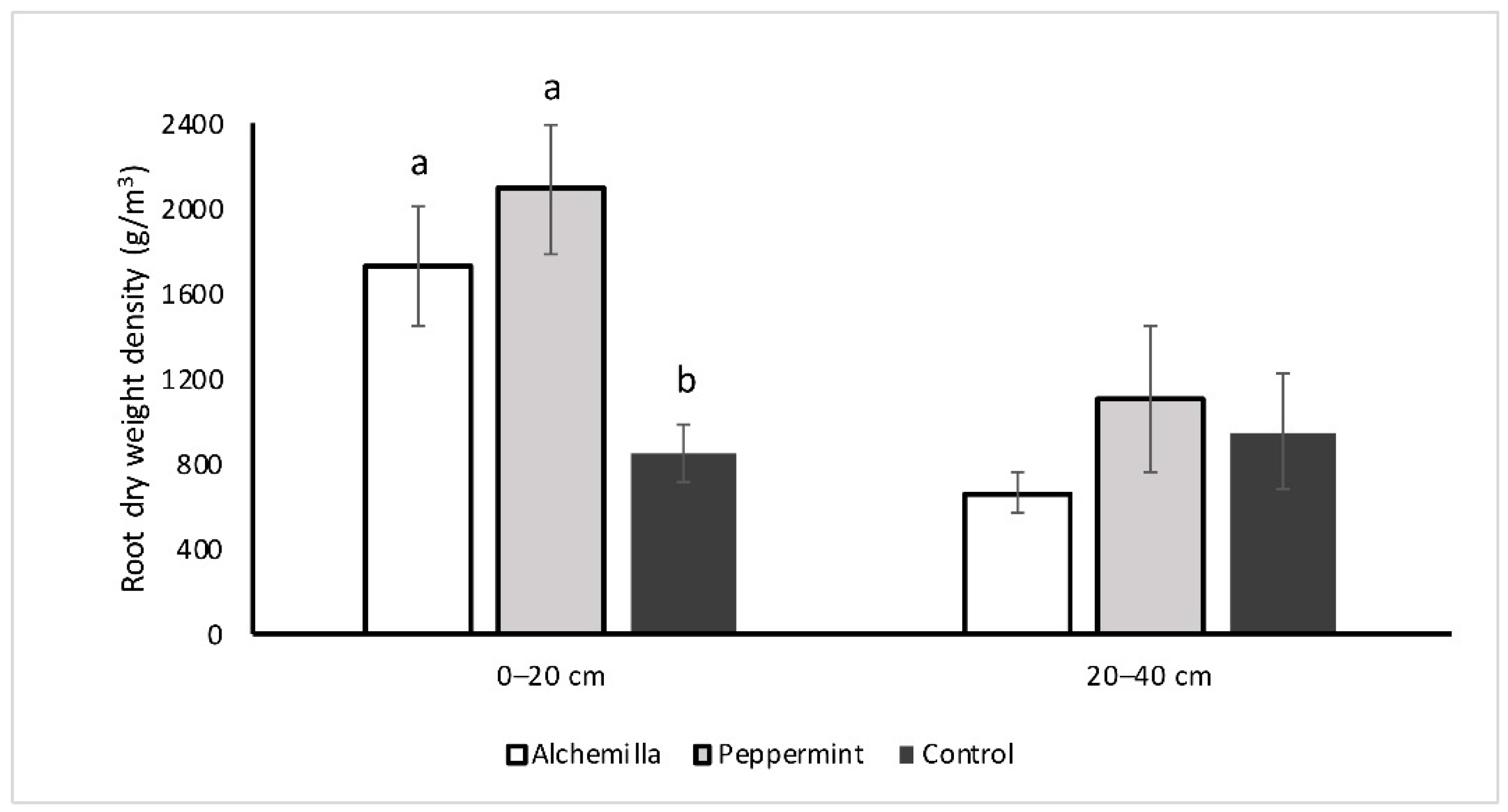
3.3. Root Morphological Parameters
3.3.1. Analysis of Overall Soil Root Community
3.3.2. Apple Root Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Weibel, F. Soil management and in-row weed control in organic apple production. Compact Fruit Tree 2002, 35, 118–121. [Google Scholar]
- Marsh, K.B.; Daly, M.J.; McCarthy, T.P. The effect of understorey management on soil fertility, tree nutrition, fruit production and apple fruit quality. Biol. Agric. Hort. 1996, 13, 161–173. [Google Scholar] [CrossRef]
- Yao, S.; Merwin, I.A.; Bird, G.W.; Abawi, G.S.; Thies, J.E. Orchard floor management practices that maintain vegetative or biomass groundcover stimulate soil microbial activity and alter soil microbial community composition. Plant Soil 2005, 271, 377–389. [Google Scholar] [CrossRef]
- Merwin, I.A.; Ray, J.A. Orchard groundcover management systems affect meadow vole populations and damage to apple trees. HortScience 1999, 34, 271–274. [Google Scholar] [CrossRef]
- Granatstein, D.; Mullinix, K. Mulching Options for Northwest Organic and Conventional Orchards. HortScience 2008, 43, 45–50. [Google Scholar] [CrossRef]
- Granatstein, D.; Sanchez, E. Research Knowledge and Needs for Orchard Floor Management in Organic Tree Fruit Systems. Int. J. Fruit Sci. 2009, 9, 257–281. [Google Scholar] [CrossRef]
- Granatstein, D.; Wiman, M.; Kirby, E.; Mullinix, K. Sustainability Trade-Offs in Organic Orchard Floor Management. Acta Hortic. 2010, 873, 115–122. [Google Scholar] [CrossRef]
- Mia, M.J.; Massetani, F.; Murri, G.; Facchi, J.; Monaci, E.; Amadio, L.; Neri, D. Integrated Weed Management in High Density Fruit Orchards. Agronomy 2020, 10, 1492. [Google Scholar] [CrossRef]
- Jiang, G.; Liang, X.; Li, L.; Li, Y.; Wu, G.; Meng, J.; Li, C.; Guo, L.; Cheng, D.; Yu, X.; et al. Biodiversity Management of Organic Orchard Enhances Both Ecological and Economic Profitability. PeerJ 2016, 4, e2137. [Google Scholar] [CrossRef]
- Yu, C.; Hu, X.M.; Deng, W.; Li, Y.; Xiong, C.; Ye, C.H.; Han, G.M.; Li, X. Changes in Soil Microbial Community Structure and Functional Diversity in the Rhizosphere Surrounding Mulberry Subjected to Long-Term Fertilization. Appl. Soil Ecol. 2015, 86, 30–40. [Google Scholar] [CrossRef]
- Hammermeister, A.M. Organic Weed Management in Perennial Fruits. Sci. Hortic. (Amst.) 2016, 208, 28–42. [Google Scholar] [CrossRef]
- Granatstein, D.; Andrews, P.; Groff, A. Productivity, Economics, and Fruit and Soil Quality of Weed Management Systems in Commercial Organic Orchards in Washington State, USA. Org. Agric. 2014, 4, 197–207. [Google Scholar] [CrossRef]
- Schmid, A.; Weibel, F. Das Sandwich-System–ein Verfahren zur herbizidfreien Baumstreifenbe- wirtschaftung? [The Sandwich System, a procedure for herbicide free in-row weed control?]. Obstbau 2000, 25, 214–217. [Google Scholar]
- Weibel, F.; Häseli, A. Organic apple production. In Apples: Botany, Production, and Uses; Ferree, D.C., Warrington, I.J., Eds.; CABI Publisher: Cambridge, UK, 2003; pp. 551–583. [Google Scholar]
- Mia, M.J.; Massetani, F.; Murri, G.; Neri, D. Sustainable Alternatives to Chemicals for Weed Control in the Orchard—A Review. Hortic. Sci. 2020, 47, 1–12. [Google Scholar] [CrossRef]
- Lemessa, F.; Wakjira, M. Cover Crops as a Means of Ecological Weed Management in Agroecosystems. J. Crop Sci. Biotechnol. 2015, 133–145. [Google Scholar] [CrossRef]
- Bàrberi, P.; Bocci, G.; Carlesi, S.; Armengot, L.; Blanco-Moreno, J.M.; Sans, F.X. Linking Species Traits to Agroecosystem Services: A Functional Analysis of Weed Communities. Weed Res. 2018, 58, 76–88. [Google Scholar] [CrossRef]
- Muscas, E.; Cocco, A.; Mercenaro, L.; Cabras, M.; Lentini, A.; Porqueddu, C.; Nieddu, G. Effects of Vineyard Floor Cover Crops on Grapevine Vigor, Yield, and Fruit Quality, and the Development of the Vine Mealybug under a Mediterranean Climate. Agric. Ecosyst. Environ. 2017, 237, 203–212. [Google Scholar] [CrossRef]
- Kremer, R.J.; Hezel, L.F.; Veum, K.S. Soil Health Improvement in an Organic Orchard Production System in Northwest Missouri. In Proceedings of the Organic Agriculture Research Symposium, LaCrosse, WI, USA, 25–26 February 2015; pp. 1–4. [Google Scholar]
- Mia, M.J.; Monaci, E.; Murri, G.; Massetani, F.; Facchi, J.; Neri, D. Soil Nitrogen and Weed Biodiversity: An Assessment under Two Orchard Floor Management Practices in a Nitrogen Vulnerable Zone in Italy. Horticulturae 2020, 6, 96. [Google Scholar] [CrossRef]
- Neri, D. Organic Soil Management to Prevent Soil Sickness during Integrated Fruit Production. IOBC WPRS Bull. 2013, 91, 87–99. [Google Scholar]
- Polverigiani, S.; Kelderer, M.; Neri, D.; Ancona, B. Growth of ‘M9’ Apple Root in Five Central Europe Replanted Soils. Plant Root 2014, 55–63. [Google Scholar] [CrossRef]
- Polverigiani, S.; Franzina, M.; Neri, D. Short Communication E Ff Ect of Soil Condition on Apple Root Development and Plant Resilience in Intensive Orchards. Appl. Soil Ecol. 2018, 123, 787–792. [Google Scholar] [CrossRef]
- Kumar, B.M.; Jose, S. Phenotypic plasticity of roots in mixed tree species agroforestry systems: Review with examples from peninsular India. Agroforest Syst. 2018, 92, 59–69. [Google Scholar] [CrossRef]
- Polverigiani, S.; Massetani, F.; Tarragoni, A.; Neri, D. Apricot Root Development and Morphology as Influenced by Mulching and Multispecies Ground Cover. Acta Hortic. 2013, 1001, 353–360. [Google Scholar] [CrossRef]
- Wardle, D.A.; Yeates, G.W.; Bonner, K.I.; Nicholson, K.S.; Watson, R.N. Impacts of Ground Vegetation Management Strategies in a Kiwifruit Orchard on the Composition and Functioning of the Soil Biota. Soil Biol. Biochem. 2001, 33, 893–905. [Google Scholar] [CrossRef]
- Hoagland, L.; Carpenter-Boggs, L.; Granatstein, D.; Mazzola, M.; Smith, J.; Peryea, F.; Reganold, J.P. Orchard Floor Management Effects on Nitrogen Fertility and Soil Biological Activity in a Newly Established Organic Apple Orchard. Biol. Fertil. Soils 2008, 45, 11–18. [Google Scholar] [CrossRef]
- Granatstein, D.; Kirby, E.; VanWechel, L. Availability of Mulch Material for Orchards in Central Washington—2002 Center for Sustaining Agriculture and Natural Resources, WSU, Wenatchee, WA, 24 April 2007. Available online: http://tfrec.cahnrs.wsu.edu/organicag/wp-content/uploads/sites/9/2016/12/Organic-OrFM-AvailMulch.pdf (accessed on 10 January 2021).
- Boroja, T.; Mihailović, V.; Katanić, J.; Pan, S.P.; Nikles, S.; Imbimbo, P.; Monti, D.M.; Stanković, N.; Stanković, M.S.; Bauer, R. The Biological Activities of Roots and Aerial Parts of Alchemilla Vulgaris L. S. Afr. J. Bot. 2018, 116, 175–184. [Google Scholar] [CrossRef]
- Balakrishnan, A. Therapeutic uses of peppermint—A review. J. Pharm. Sci. 2015, 7, 474–476. [Google Scholar]
- Domaradzki, K.; Badowski, M.; Filipiak, K.; Franek, M.; Gołębiowska, H.; Kieloch, R.; Kucharski, M.; Rola, H.; Rola, J.; Sadowski, J.; et al. Metodyka doświadczeń biologicznej oceny herbicydów, bioregulatorów i adiuwantów. CZ Dosw. Polowe 2001, 1, 167. [Google Scholar]
- Böhm, W. Methods of Studying Root Systems; Springer: Berlin/Heidelberg, Germany, 1979. [Google Scholar] [CrossRef]
- Frasier, I.; Noellemeyer, E.; Fernández, R.; Quiroga, A. Direct field method for root biomass quantification in agroecosystems. MethodsX 2016, 3, 513–519. [Google Scholar] [CrossRef]
- Cerovic, Z.G.; Masdoumier, G.; Ghozlen, N.B.; Latouche, G. A New Optical Leaf-Clip Meter for Simultaneous Non-Destructive Assessment of Leaf Chlorophyll and Epidermal Flavonoids. Physiol. Plant. 2012, 146, 251–260. [Google Scholar] [CrossRef]
- Kowalczyk, K.; Gajc-Wolska, J.; Mirgos, M.; Geszprych, A.; Kowalczyk, W.; Sieczko, L.; Niedzińska, M.; Gajewski, M. Mineral nutrients needs of cucumber and its yield in protected winter cultivation, with HPS and LED supplementary lighting. Sci. Hortic. (Amst.) 2020, 265, 265. [Google Scholar] [CrossRef]
- Steinmaus, S.; Elmore, C.L.; Smith, R.J.; Donaldson, D.; Weber, E.A.; Roncoroni, J.A.; Miller, P.R.M. Mulched cover crops as an alternative to conventional weed management systems in vineyards. Weed Res. 2008, 48, 273–281. [Google Scholar] [CrossRef]
- Tursun, N.; Işık, D.G.; Demir, Z.; Jabran, K. Use of Living, Mowed, and Soil-Incorporated Cover Crops for Weed Control in Apricot Orchards. Agronomy 2018, 8, 150. [Google Scholar] [CrossRef]
- Qasem, J.R. Applied Allelopathy in Weed Management: An Update. In Allelopathy; Cheema, Z., Farooq, M., Wahid, A., Eds.; Springer: Berlin/Heidelberg, Germany, 2013. [Google Scholar] [CrossRef]
- Cheng, F.; Cheng, Z. Research progress on the use of plant allelopathy in agriculture and the physiological and ecological mechanisms of allelopathy. Front. Plant Sci. 2015, 6, 1020. [Google Scholar] [CrossRef]
- Rohloff, J. Monoterpene Composition of Essential Oil from Peppermint (Mentha × piperita L.) with Regard to Leaf Position Using Solid-Phase Microextraction and Gas Chromatography/Mass Spectrometry Analysis. J. Agric. Food Chem. 1999, 47, 3782–3786. [Google Scholar] [CrossRef] [PubMed]
- Campiglia, E.; Mancinelli, R.; Cavalieri, A.; Caporali, F. Use of essential oils of cinnamon (Cinnamomum zeylanicum L.), lavender (Lavandula spp.) and peppermint (Mentha × piperita L.) for weed control. Ital. J. Agron. 2007, 2, 171–175. [Google Scholar] [CrossRef]
- Mahdavikia, F.; Saharkhiz, M.J. Phytotoxic activity of essential oil and water extract of peppermint (Mentha × piperita L. Cv. Mitcham). J. Appl. Res. Med. Aromat. Plants 2015, 2, 146–153. [Google Scholar] [CrossRef]
- Sarheed, M.M.; Rajabi, F.; Kunert, M.; Boland, W.; Wetters, S.; Miadowitz, K.; Kaz’mierczak, A.; Sahi, V.P.; Nick, P. Cellular Base of Mint Allelopathy: Menthone Affects Plant Microtubules. Front. Plant Sci. 2020, 11, 546345. [Google Scholar] [CrossRef]
- Anthony, R.G.; Waldin, T.R.; Ray, J.A.; Bright, S.W.J.; Hussey, P.J. Herbicide resistance caused by spontaneous mutation of the cytoskeletal protein tubulin. Nature 1998, 3936682, 260–263. [Google Scholar] [CrossRef]
- Eom, S.H.; Senesac, A.F.; Tsontakis-bradley, I.; Weston, L.A. Evaluation of Herbaceous Perennials as Weed Suppressive Groundcovers for Use Along Roadsides or in Landscapes. J. Environ. Hort. 2005, 23, 198–203. [Google Scholar] [CrossRef]
- Costa, A.G.; Bertolucci, S.K.V.; Chagas, J.H.; Ferraz, E.O.; Pinto, J.E.B.P. Biomass production, yield and chemical composition of peppermint essential oil using different organic fertilizer sources. Ciência Agrotecnol. 2013, 37, 202–210. [Google Scholar] [CrossRef]
- Jeliazkova, E.A.; Zheljazkov, V.D.; Craker, L.E.; Yankov, B.; Georgieva, T. NPK fertilizer and yields of peppermint, Mentha × piperita. Acta Hortic. 1999, 502, 231–236. [Google Scholar] [CrossRef]
- Lawrence, B.M. Mint: The Genus Mentha; CRC Press: Boca Raton, FL, USA, 2006. [Google Scholar]
- Vitkova, A.; Tashev, A.; Gavrilova, A. Alchemilla mollis (Rosaceae)—A critically endangered species in Bulgaria. Phytol. Balc. 2011, 17, 123–128. [Google Scholar]
- Liebman, M.; Davis, A.S. Integration of soil, crop, and weed management in low-external-input farming systems. Weed Res. 2000, 40, 27–47. [Google Scholar] [CrossRef]
- Schütt, M.; Borken, W.; Spott, O.; Stange, C.F.; Matzner, E. Temperature Sensitivity of C and N Mineralization in Temperate Forest Soils at Low Temperatures. Soil Biol. Biochem. 2013, 69, 320–327. [Google Scholar] [CrossRef]
- Vokou, D.; Chalkos, D.; Karamanlidou, G.; Yiangou, M. Activation of soil respiration and shift of the microbial population balance in soil as a response to Lavandula stoechas essential oil. J. Chem. Ecol. 2002, 28, 755–768. [Google Scholar] [CrossRef] [PubMed]
- Kadoglidou, K.; Lagopodi, A.; Karamanoli, K.; Vokou, D.; Bardas, G.A.; Menexes, G.; Constantinidou, H.-I.A. Inhibitory and stimulatory effects of essential oils and individual monoterpenoids on growth and sporulation of four soil-borne fungal isolates of Aspergillus terreus, Fusarium oxysporum, Penicillium expansum and Verticillium dahlia. Eur. J. Plant Pathol. 2011, 130, 297–309. [Google Scholar] [CrossRef]
- Konstantinou, S.; Monokrousos, N.; Kapagianni, P.; Menkissoglu-Spiroudi, U.; Gwynn-Jones, D.; Stamou, G.P.; Papatheodorou, E.M. Instantaneous responses of microbial communities to stress in soils pretreated with Mentha spicata essential oil and/or inoculated with arbuscular mycorrhizal fungus. Ecol. Res. 2019, 34, 701–710. [Google Scholar] [CrossRef]
- Vokou, D.; Liotiri, S. Stimulation of soil microbial activity by essential oils. Chemoecology 1999, 9, 41–45. [Google Scholar] [CrossRef]
- Mariotte, P.; Vandenberghe, C.; Meugnier, C.; Rossi, P.; Bardgett, R.D.; Buttler, A. Subordinate plant species impact on soil microbial communities and ecosystem functioning in grasslands: Findings from a removal experiment. Perspect. Plant Ecol. Evol. Syst. 2013, 15, 77–85. [Google Scholar] [CrossRef]
- Qu, Q.; Zhang, Z.; Peijnenburg, W.J.G.M.; Liu, W.; Lu, T.; Hu, B.; Chen, J.; Chen, J.; Lin, Z.; Qian, H. Rhizosphere microbiome assembly and its impact on plant growth. J. Agric. Food Chem. 2020, 68, 5024–5038. [Google Scholar] [CrossRef] [PubMed]
- Bardi, L.; Malusá, E. Drought and nutritional stresses in plant: Alleviating role of rhizospheric microorganisms. In Abiotic Stress: New Research; Haryana, N., Punj, S., Eds.; Nova Science Publishers, Inc.: Hauppauge, NY, USA, 2012; pp. 1–57. [Google Scholar]
- Żelazny, W.R.; Licznar-Małańczuk, M. Soil quality and tree status in a twelve-year-old apple orchard under three mulch-based floor management systems. Soil Tillage Res. 2018, 180, 250–258. [Google Scholar] [CrossRef]
- Vassileva, M.; Flor-Peregrin, E.; Malusà, E.; Vassilev, N. Towards better understanding of the interactions and efficient application of plant beneficial prebiotics, probiotics, postbiotics and synbiotics. Front. Plant. Sci. 2020, 11, 1068. [Google Scholar] [CrossRef] [PubMed]

| EPPO Code | Scientific Name | Treatments | ||
|---|---|---|---|---|
| A. vulgaris | M. piperita | Control | ||
| AGRRE | Agropyron repens L. | ✓ | ✓ | ✓ |
| CAPBP | Capsella bursa-pastoris L. | ✓ | ✓ | |
| ECHCG | Echinochloa crus-galli L. | ✓ | ✓ | |
| EQUAR | Equisetum arvense L. | ✓ | ✓ | ✓ |
| ERICA | Erigeron canadensis L. | ✓ | ✓ | ✓ |
| EROCI | Erodium cicutarium L. | ✓ | ✓ | |
| GASPA | Galinsoga parviflora Cav. | ✓ | ✓ | |
| HRYRA | Hypochaeris radicata L. | ✓ | ✓ | ✓ |
| LAMPU | Lamium purpureum L. | ✓ | ||
| POAPR | Poa pratensis L. | ✓ | ✓ | ✓ |
| RUMSS | Rumex sp. | ✓ | ✓ | ✓ |
| STEME | Stellaria media L. | ✓ | ✓ | ✓ |
| TAROF | Taraxacum officinale Weber | ✓ | ✓ | ✓ |
| TRFAR | Trifolium arvense L. | ✓ | ✓ | ✓ |
| VERPE | Veronica persica Poiret | ✓ | ||
| VIOAR | Viola arvensis L. | ✓ | ✓ | ✓ |
| Parameters | Chlorophyll (µg/cm²) | N (%) | P (%) | K (%) | Ca (%) | Mg (%) |
|---|---|---|---|---|---|---|
| Treatment | ||||||
| Alchemilla | 27.9 ± 0.66 a | 1.56 ± 0.06 | 0.15 ± 0.01 | 1.68 ± 0.08 | 2.12 ± 0.02a | 0.29 ± 0.01a |
| Peppermint | 22.9 ± 0.44 b | 1.42 ± 0.05 | 0.13 ± 0.01 | 1.42 ± 0.06 | 2.0 ± 0.07ab | 0.29 ± 0.01 a |
| Control | 21.7 ± 0.50 b | 1.37 ± 0.04 | 0.14 ± 0.01 | 1.54 ± 0.06 | 1.61 ± 0.07 b | 0.23 ± 0.01 b |
| p-value | 0.0001 | 0.066 | 0.503 | 0.247 | 0.0150 | 0.0084 |
| Parameters | Root Dry Weight Density (g/m3) | Root Length (cm) | Root Surface Area (cm2) | Root Diameter (mm) | Root Volume (cm3) |
|---|---|---|---|---|---|
| Treatment | |||||
| Alchemilla | 1191.0 ab | 700.0 | 156.0 ab | 0.90 | 3.2 |
| Peppermint | 1592.4 a | 619.9 | 167.5 a | 0.90 | 3.8 |
| Control | 895.9 b | 546.5 | 121.1 b | 0.91 | 2.9 |
| P–value | 0.03 | 0.29 | 0.04 | 0.99 | 0.29 |
| Soil depth | |||||
| 0–20 cm | 1551.6 a | 885.9 a | 205.8 a | 0.86 | 4.2 a |
| 20–40 cm | 901.2 b | 358.4 b | 90.7 b | 0.94 | 2.4 b |
| p-value | 0.002 | 0.0001 | 0.0001 | 0.454 | 0.0002 |
| Interaction | |||||
| Treatment* Soil depth (p–value) | 0.042 | 0.640 | 0.724 | 0.515 | 0.224 |
| Parameters | Root Dry Weight Density (g/m3) | Root Length (cm) | Root Surface Area (cm2) | Root Diameter (mm) | Root Volume (cm3) |
|---|---|---|---|---|---|
| Treatment | |||||
| Alchemilla | 556.8 | 186.3 | 35.3 | 0.40 | 0.61 |
| Peppermint | 624.3 | 104.9 | 23.3 | 0.51 | 0.50 |
| Control | 425.3 | 69.2 | 13.0 | 0.42 | 0.41 |
| p-value | 0.788 | 0.189 | 0.189 | 0.837 | 0.770 |
| Soil depth | |||||
| 0–20 cm | 508 | 165.2 | 30.4 | 0.30 | 0.49 |
| 20–40 cm | 563 | 75.1 | 17.3 | 0.60 | 0.51 |
| p-value | 0.819 | 0.094 | 0.191 | 0.109 | 0.921 |
| Interaction | |||||
| Treatment*Soil depth (p-value) | 0.068 | 0.113 | 0.038 | 0.350 | 0.043 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mia, M.J.; Furmanczyk, E.M.; Golian, J.; Kwiatkowska, J.; Malusá, E.; Neri, D. Living Mulch with Selected Herbs for Soil Management in Organic Apple Orchards. Horticulturae 2021, 7, 59. https://doi.org/10.3390/horticulturae7030059
Mia MJ, Furmanczyk EM, Golian J, Kwiatkowska J, Malusá E, Neri D. Living Mulch with Selected Herbs for Soil Management in Organic Apple Orchards. Horticulturae. 2021; 7(3):59. https://doi.org/10.3390/horticulturae7030059
Chicago/Turabian StyleMia, Md Jebu, Ewa M. Furmanczyk, Joanna Golian, Joanna Kwiatkowska, Eligio Malusá, and Davide Neri. 2021. "Living Mulch with Selected Herbs for Soil Management in Organic Apple Orchards" Horticulturae 7, no. 3: 59. https://doi.org/10.3390/horticulturae7030059
APA StyleMia, M. J., Furmanczyk, E. M., Golian, J., Kwiatkowska, J., Malusá, E., & Neri, D. (2021). Living Mulch with Selected Herbs for Soil Management in Organic Apple Orchards. Horticulturae, 7(3), 59. https://doi.org/10.3390/horticulturae7030059









