Chemical Composition and Anti-Microbial Activity of Hog Plum (Spondias mombin L.) Peel Oil Extracted from Different Regions of Tropical Climates
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Collection
2.2. Preparation of Hog Plum Peel Powder
2.3. Extraction of Essential Oil
2.4. Gas Chromatography and Mass Spectrometry (GC-MS) Analysis
2.5. Determination of Antimicrobial Activity
2.6. Statistical Analysis
3. Results and Discussion
3.1. Yield
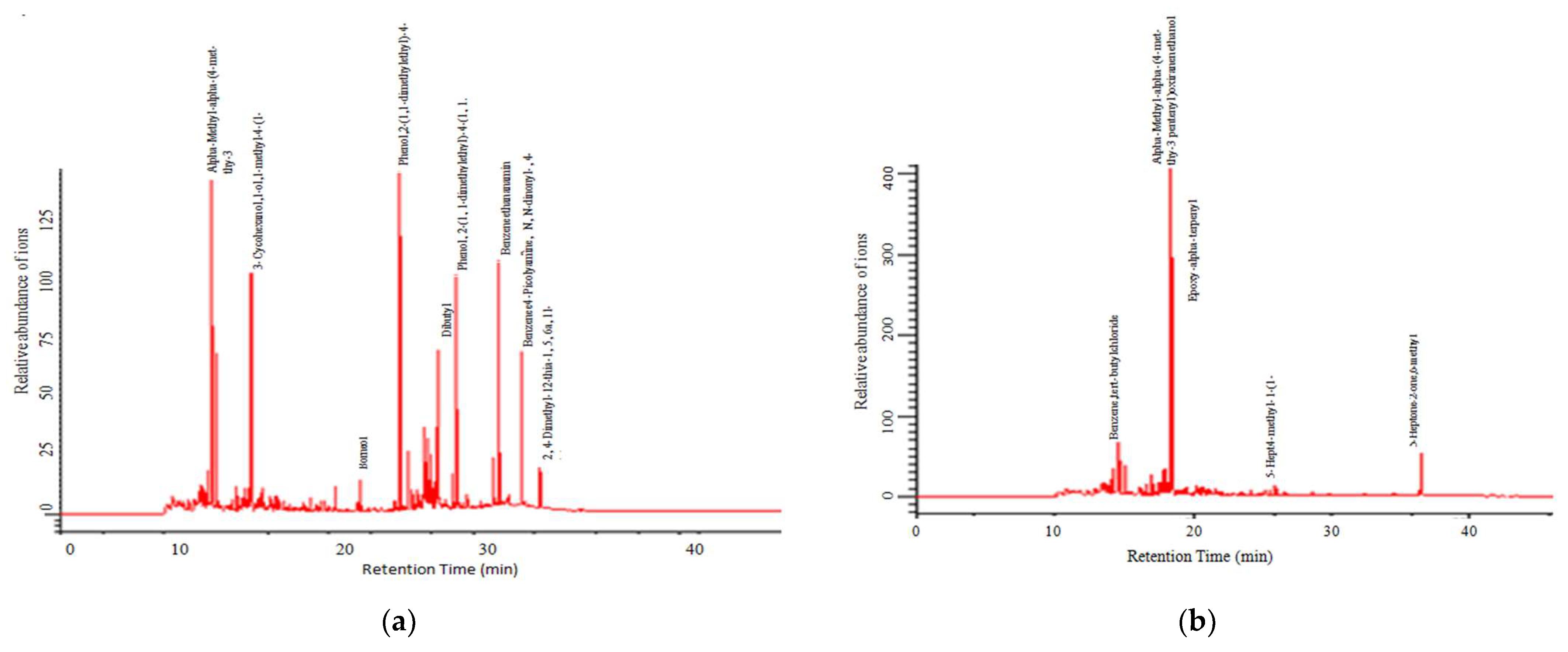
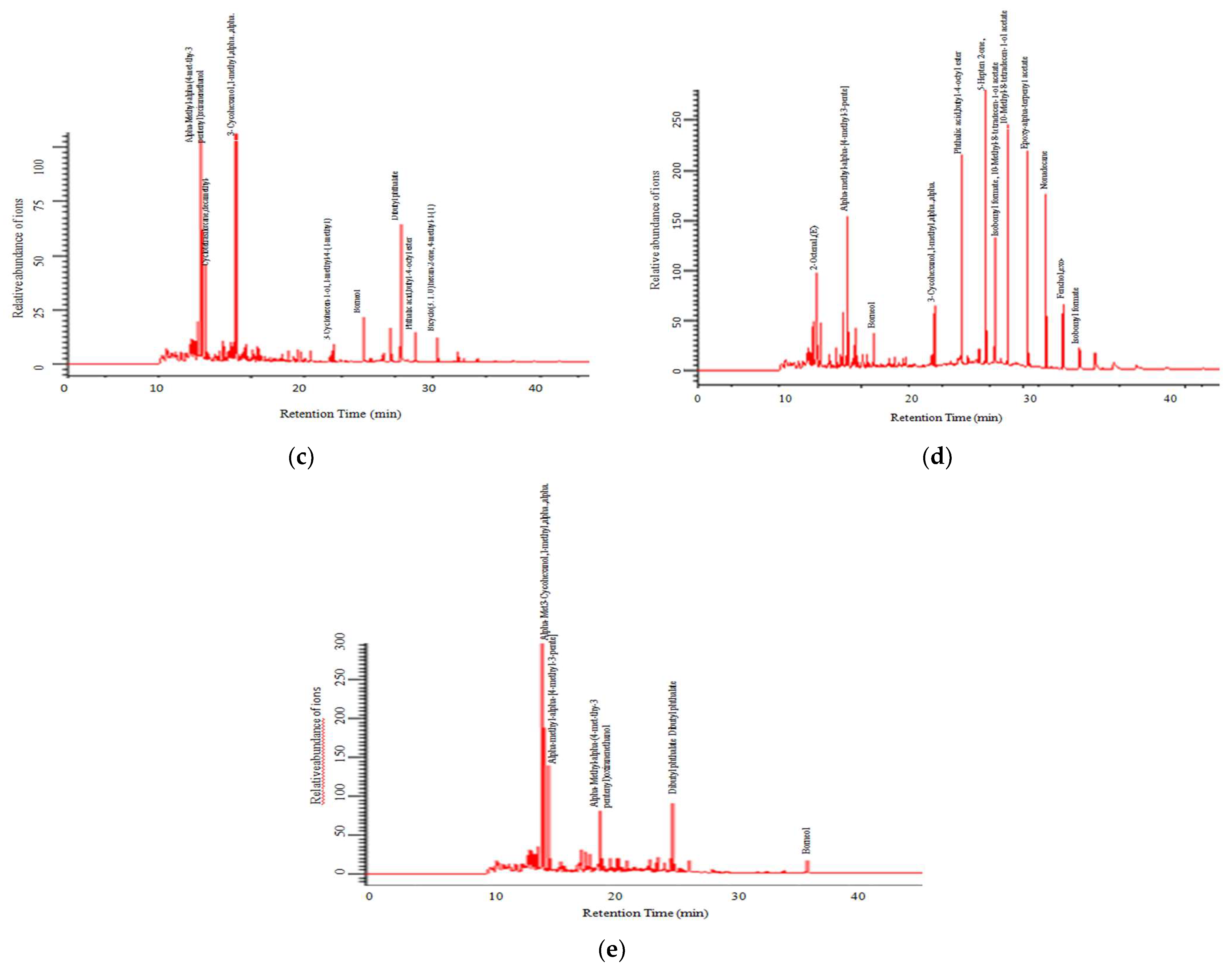
3.2. Chemical Composition of Volatile Oil
3.3. Anti-Bacterial Activities of Essential Oil
3.4. Anti-Fungal Activity of Essential Oil
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Siddiqu, A.; Nasrin, S.; Moonmoon, M.; Islam, M.A.; Khatun, M.M. Bacterial assessment of street-vended hog plum (Spondias mombin) and its public health importance. Bangladesh Vet. 2015, 32, 19–26. [Google Scholar] [CrossRef] [Green Version]
- Bhuiyan, M.H.R. Pickle and chutney development from fresh Hog Plum (Spondias dulcis). J. Environ. Sci. Nat. Resource 2012, 5, 67–72. [Google Scholar] [CrossRef]
- Bangladesh Bureau of Statistics (BBS). Yearbook of Agricultural Statistics-2017. Stat. Div., Min. Plann., Govt. Peoples Repub. Bangladesh, Dhaka, Bangladesh. 2018. Available online: http://bbs.portal.gov.bd/sites/default/files/files/bbs.portal.gov.bd/page/1b1eb817_9325_4354_a756_3d18412203e2/Yearbook-2017-Final-05-05-2018.pdf (accessed on 1 August 2021).
- Akter, S.; Shahriar, S.M.S.; Akter, F.; Morshed, S.; Islam, M.N. Study on chemical composition of fresh Mymensingh and Barishal hog-plum (Spondius mangifera) and developed leather and jelly and sensory evaluation. J. Environ. Sci. Nat. Resource 2012, 5, 29–36. [Google Scholar] [CrossRef] [Green Version]
- Rymbai, H.; Deshmukh, N.A.; Jha, V.A.; Verma, V.K.; War, F.G.; Paul, D.; Patel, R.S.; Roy, A.R. Indian Hog Plum. Breed. Underutilized Fruit Crop 2016, 13, 183–190. [Google Scholar]
- Miller, A. Wild Crop Relatives: Genomic and Breeding Resources: Tropical and subtropical Fruits; Kole, C., Ed.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 203–212. [Google Scholar] [CrossRef]
- Chacko, C.M.; Estherlydia, D. Antimicrobial evaluation of jams made from indigenous fruit peels. Int. J. Adv. Res. 2014, 2, 202–207. [Google Scholar]
- Sharmeen, J.B.; Mahomoodally, F.M.; Zengin, G.; Maggi, F. Essential Oils as Natural Sources of Fragrance Compounds for Cosmetics and Cosmeceuticals. Molecules 2021, 26, 666. [Google Scholar] [CrossRef] [PubMed]
- Ni, Z.-J.; Wang, X.; Shen, Y.; Thakur, K.; Han, J.; Zhang, J.-G.; Hu, F.; Wei, Z.-J. Recent updates on the chemistry, bioactivities, mode of action, and industrial applications of plant essential oils. Trends Food Sci. Technol. 2021, 110, 78–89. [Google Scholar] [CrossRef]
- Beltrán Sanahuja, A.; Valdés García, A. New Trends in the Use of Volatile Compounds in Food Packaging. Polymers 2021, 13, 1053. [Google Scholar] [CrossRef] [PubMed]
- Sarrou, E.; Chatzopoulou, P.; Dimassi-Theriou, K.; Therios, I. Volatile Constituents and Antioxidant Activity of Peel, Flowers and Leaf Oils of Citrus aurantium L. Growing in Greece. Molecules 2013, 18, 10639–10647. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bora, H.; Kamle, M.; Mahato, D.K.; Tiwari, P.; Kumar, P. Citrus Essential Oils (CEOs) and Their Applications in Food: An Overview. Plants 2020, 9, 357. [Google Scholar] [CrossRef] [Green Version]
- Raveau, R.; Fontaine, J.; Lounès-Hadj Sahraoui, A. Essential Oils as Potential Alternative Biocontrol Products against Plant Pathogens and Weeds: A Review. Foods 2020, 9, 365. [Google Scholar] [CrossRef] [Green Version]
- Lin, X.; Cao, S.; Sun, J.; Lu, D.; Zhong, B.; Chun, J. The Chemical Compositions, and Antibacterial and Antioxidant Activities of Four Types of Citrus Essential Oils. Molecules 2021, 26, 3412. [Google Scholar] [CrossRef]
- López-Camacho, P.Y.; Martínez-Espinosa, J.C.; Basurto-Islas, G.; Torres-Zarraga, A.; Márquez-Villa, J.M.; Macías-Alonso, M.; Marrero, J.G. Spondias mombin Seed Oil Compounds Identification by Raman Spectroscopy and NMR. Appl. Sci. 2021, 11, 2886. [Google Scholar] [CrossRef]
- An, N.T.G.; Huong, L.T.; Satyal, P.; Tai, T.A.; Dai, D.N.; Hung, N.H.; Ngoc, N.T.B.; Setzer, W.N. Mosquito Larvicidal Activity, antimicrobial activity, and chemical compositions of essential oils from four species of myrtaceae from central vietnam. Plants 2020, 9, 544. [Google Scholar] [CrossRef] [Green Version]
- Syafiq, R.; Sapuan, S.M.; Zuhri, M.Y.M.; Ilyas, R.A.; Nazrin, A.; Sherwani, S.F.K.; Khalina, A. Antimicrobial Activities of Starch-Based Biopolymers and Biocomposites Incorporated with Plant Essential Oils: A Review. Polymers 2020, 12, 2403. [Google Scholar] [CrossRef] [PubMed]
- Klūga, A.; Terentjeva, M.; Vukovic, N.L.; Kačániová, M. Antimicrobial Activity and Chemical Composition of Essential Oils against Pathogenic Microorganisms of Freshwater Fish. Plants 2021, 10, 1265. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.M.; Wills, R.B.H.; Bowyer, M.C.; Golding, J.B.; Kirkman, T.; Pristijono, P. Efficacy of Orange Essential Oil and Citral after Exposure to UV-C Irradiation to Inhibit Penicillium digitatum in Navel Oranges. Horticulturae 2020, 6, 102. [Google Scholar] [CrossRef]
- Alonso-Gato, M.; Astray, G.; Mejuto, J.C.; Simal-Gandara, J. Essential Oils as Antimicrobials in Crop Protection. Antibiotics 2021, 10, 34. [Google Scholar] [CrossRef]
- Cristofoli, N.L.; Lima, C.A.R.; Vieira, M.M.C.; Andrade, K.S.; Ferreira, S.R.S. Antioxidant and antimicrobial potential of cajazeira leaves (Spondias mombin) extracts. Sep. Sci. Technol. 2019, 54, 580–590. [Google Scholar] [CrossRef]
- Bora, P.S.; Narain, N.; Holschuh, H.J.; da S. Vasconcelos, M.A. Changes in physical and chemical composition during maturation of yellow mombin (Spondias mombin) fruits. Food Chem. 1991, 41, 341–348. [Google Scholar] [CrossRef]
- Samuggam, S.; Chinni, S.V.; Mutusamy, P.; Gopinath, S.C.B.; Anbu, P.; Venugopal, V.; Reddy, L.V.; Enugutti, B. Green Synthesis and Characterization of Silver Nanoparticles Using Spondias mombin Extract and Their Antimicrobial Activity against Biofilm-Producing Bacteria. Molecules 2021, 26, 2681. [Google Scholar] [CrossRef]
- Elufioye, T.O.; Obuotor, E.M.; Agbedahunsi, J.M.; Adesanya, S.A. Anticholinesterase constituents from the leaves of Spondias mombin L. (Anacardiaceae). Biologics 2017, 11, 107–114. [Google Scholar] [CrossRef] [Green Version]
- Enogieru, A.B.; Momodu, O.I. African Medicinal Plants Useful for Cognition and Memory: Therapeutic Implications for Alzheimer’s Disease. Bot. Rev. 2021, 87, 107–134. [Google Scholar] [CrossRef]
- Brito, S.A.; de Almeida, C.L.F.; de Santana, T.I.; da Silva Oliveira, A.R.; do Nascimento Figueiredo, J.C.B.; Souza, I.T.; de Almeida, L.L.; da Silva, M.V.; Borges, A.S.; de Medeiros, J.W.; et al. Antiulcer Activity and Potential Mechanism of Action of the Leaves of Spondias mombin L. Oxid. Med. Cell. Longev. 2018, 2018, 1731459. [Google Scholar] [CrossRef] [Green Version]
- Muhammad, A.; Rahman, M.; Kabir, A.N.M.; Kabir, S.; Hossain, M. Antibacterial and cytotoxic activities of Spondias pinnata (Linn. f.) Kurz fruit extract. Nat. Prod. Resource 2011, 2, 265–267. [Google Scholar]
- da Silva, A.R.A.; de Morais, S.M.; Mendes Marques, M.M.; de Oliveira, D.F.; Barros, C.C.; de Almeida, R.R.; Vieira, Í.G.P.; Guedes, M.I.F. Chemical composition, antioxidant and antibacterial activities of two Spondias species from Northeastern Brazil. Pharm. Biol. 2012, 50, 740–746. [Google Scholar] [CrossRef] [PubMed]
- Determination of Essential oils in Vegetable drugs. European Pharmacopoeia, 4th ed.; European Directorate for the Quality of Medicines, Council of Europe Editions: Strasbourg, France, 2002; pp. 183–184.
- Petretto, G.L.; Maldini, M.; Addis, R.; Chessa, M.; Foddai, M.; Rourke, J.P.; Pintore, G. Variability of chemical composition and antioxidant activity of essential oils between Myrtus communis var. Leucocarpa DC and var. Melanocarpa DC. Food Chem. 2016, 197, 124–131. [Google Scholar] [CrossRef]
- Mitropoulou, G.; Fitsiou, E.; Stavropoulou, E.; Papavassilopoulou, E.; Vamvakias, M.; Pappa, A.; Oreopoulou, A.; Kourkoutas, Y. Composition, antimicrobial, antioxidant, and antiproliferative activity of Origanum dictamnus (dittany) essential oil. Microbial Ecol. Health Dis. 2015, 26, 26543. [Google Scholar] [CrossRef]
- Panda, B.K.; Patra, V.J.; Mishra, U.S.; Kar, S.; Panda, B.R.; Hati, M.R. Analgesic activities of the stem bark extract of Spondias pinata (Linn. f) Kurz. J. Pharm. Res. 2009, 2, 825–827. [Google Scholar]
- Boukaew, S.; Prasertsan, P.; Sattayasamitsathit, S. Evaluation of antifungal activity of essential oils against aflatoxigenic Aspergillus flavus and their allelopathic activity from fumigation to protect maize seeds during storage. Ind. Crops Prod. 2017, 97, 558–566. [Google Scholar] [CrossRef]
- Laohaprasit, N.; Kukreja, R.K.; Arunrat, A. Extraction of volatile compounds from ‘Nam Dok Mai’ and ‘Maha Chanok’ mangoes. Int. Food Res. J. 2012, 19, 1445. [Google Scholar]
- Bourgou, S.; Rahali, S.F.Z.; Ourghemmi, I.; Saïdani Tounsi, M.S. Changes of Peel Essential Oil Composition of Four Tunisian Citrus during Fruit Maturation. Sci. World J. 2012, 2012, 528593. [Google Scholar] [CrossRef] [Green Version]
- Asekun, O.T.; Grieron, D.S.; Afolayan, A.S. Influence of drying methods on the chemical composition and yield of the essential oil of Leonotis leonurus. J. Sci. Res. 2006, 10, 61–64. [Google Scholar]
- de Lima, E.Q.; de Oliveira, E.; de Brito, H.R. Extraction and characterization of the essential oils from Spondias mombin L.(Caj), Spondias purpurea L.(Ciriguela) and Spondia ssp. (Cajarana do serto). Afr. J. Agric. Res. 2016, 11, 105–116. [Google Scholar]
- González-Mas, M.C.; Rambla, J.L.; López-Gresa, M.P.; Blázquez, M.A.; Granell, A. Volatile Compounds in Citrus Essential Oils: A Comprehensive Review. Front. Plant Sci. 2019, 10, 12. [Google Scholar] [CrossRef]
- Quintana, S.E.; Salas, S.; García-Zapateiro, L.A. Bioactive compounds of mango (Mangifera indica): A review of extraction technologies and chemical constituents. J. Sci. Food Agric. 2021. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wen, H.; Chen, J.; Peng, Z.; Shi, M.; Chen, M.; Yuan, Z.; Liu, Y.; Zhang, H.; Xu, J. Volatile Compounds in Fruit Peels as Novel Biomarkers for the Identification of Four Citrus Species. Molecules 2019, 24, 4550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scurria, A.; Sciortino, M.; Presentato, A.; Lino, C.; Piacenza, E.; Albanese, L.; Zabini, F.; Meneguzzo, F.; Nuzzo, D.; Pagliaro, M.; et al. Volatile Compounds of Lemon and Grapefruit IntegroPectin. Molecules 2021, 26, 51. [Google Scholar] [CrossRef]
- Yang, S.; Hao, N.; Meng, Z.; Li, Y.; Zhao, Z. Identification, Comparison and Classification of Volatile Compounds in Peels of 40 Apple Cultivars by HS–SPME with GC–MS. Foods 2021, 10, 1051. [Google Scholar] [CrossRef]
- Aguilar-Hernández, M.G.; Sánchez-Bravo, P.; Hernández, F.; Carbonell-Barrachina, Á.A.; Pastor-Pérez, J.J.; Legua, P. Determination of the Volatile Profile of Lemon Peel Oils as Affected by Rootstock. Foods 2020, 9, 241. [Google Scholar] [CrossRef] [Green Version]
- Afolabi, T.A.; Onadeji, R.S.; Ogunkunle, O.A.; Bamiro, F.O. Comparative analysis of the nutritional quality of browse leaves (Spondias mombin and (Albizia saman) and tuber peels (yam and cassava) used as ruminant feeds. IFE J. Sci. 2012, 14, 337–344. [Google Scholar]
- Miller, A.B.; Cates, R.G.; Lawrence, M.; Soria, J.A.F.; Espinoza, L.V.; Martinez, J.V.; Arbizú, D.A. The antibacterial and antifungal activity of essential oils extracted from Guatemalan medicinal plants. Pharm. Biol. 2015, 53, 548–554. [Google Scholar] [CrossRef] [Green Version]
- Viuda-Martos, M.; Ruiz-Navajas, Y.; Fernández-López, J.; Pérez-Álvarez, J. Antifungal activity of lemon (Citrus lemon L.), mandarin (Citrus reticulata L.), grapefruit (Citrus paradisi L.) and orange (Citrus sinensis L.) essential oils. Food Control 2008, 19, 1130–1138. [Google Scholar] [CrossRef]
- Kalemba, D.A.A.K.; Kunicka, A. Antibacterial and antifungal properties of essential oils. Curr. Med. Chem. 2003, 10, 813–829. [Google Scholar] [CrossRef]
- Shukla, R.; Kumar, A.; Singh, P.; Dubey, N.K. Efficacy of Lippia alba (Mill.) N.E. Brown essential oil and its monoterpene aldehyde constituents against fungi isolated from some edible legume seeds and aflatoxin B1 production. Int. J. Food Microbiol. 2009, 135, 165–170. [Google Scholar] [CrossRef] [PubMed]
- Duru, M.; Cakir, A.; Kordali, S.; Zengin, H.; Harmandar, M.; Izumi, S.; Hirata, T. Chemical composition and antifungal properties of essential oils of three Pistacia species. Fitoterapia 2003, 74, 170–176. [Google Scholar] [CrossRef]
- Badr, M.M.; Badawy, M.E.I.; Taktak, N.E.M. Characterization, antimicrobial activity, and antioxidant activity of the nanoemulsions of Lavandula spica essential oil and its main monoterpenes. J. Drug Deliv. Sci. Technol. 2021, 65, 102732. [Google Scholar] [CrossRef]
- de Lira, M.H.P.; de Andrade Júnior, F.P.; Moraes, G.F.Q.; de Silva Macena, G.; de Oliveira Pereira, F.; Lima, I.O. Antimicrobial activity of geraniol: An integrative review. J. Essential Oil Res. 2020, 32, 187–197. [Google Scholar] [CrossRef]
- Ebani, V.V.; Bertelloni, F.; Najar, B.; Nardoni, S.; Pistelli, L.; Mancianti, F. Antimicrobial Activity of Essential Oils against Staphylococcus and Malassezia Strains Isolated from Canine Dermatitis. Microorganisms 2020, 8, 252. [Google Scholar] [CrossRef] [Green Version]
- Alabi, O.A.; Haruna, M.T.; Anokwuru, C.P.; Jegede, T.; Abia, H.; Okegbe, V.U.; Esan, B.E. Comparative studies on antimicrobial properties of extracts of fresh and dried leaves of Carica papaya (L.) on clinical bacterial and fungal isolates. Adv. Appl. Sci. Res. 2012, 3, 3107–3114. [Google Scholar]
- Bajpai, V.K.; Rahman, A.; Kang, S.C. Chemical composition and anti-fungal properties of the essential oil and crude extracts of Metasequoia glyptostroboides Miki ex Hu. Ind. Crop. Prod. 2007, 26, 28–35. [Google Scholar] [CrossRef]
- Nazzaro, F.; Fratianni, F.; Coppola, R.; de Feo, V. Essential Oils and Antifungal Activity. Pharmaceut 2017, 10, 86. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El-Mohamedy, R.S.R. Plant Essential Oils for Controlling Plant Pathogenic Fungi. In Volatiles and Food Security: Role of Volatiles in Agro-Ecosystems; Choudhary, D.K., Sharma, A.K., Agarwal, P., Varma, A., Tuteja, N., Eds.; Springer: Singapore, 2017; pp. 171–198. [Google Scholar] [CrossRef]
- Jing, L.; Lei, Z.; Li, L.; Xie, R.; Xi, W.; Guan, Y.; Sumner, L.W.; Zhou, Z. Antifungal Activity of Citrus Essential Oils. J. Agric. Food Chem. 2014, 62, 2011–3033. [Google Scholar] [CrossRef] [PubMed]
- Wojtunik-Kulesza, K.A.; Kasprzak, K.; Oniszczuk, T.; Oniszczuk, A. Natural Monoterpenes: Much More than Only a Scent. Chem. Biodivers. 2019, 16, e1900434. [Google Scholar] [CrossRef] [PubMed]
- LU, S.M.; SHI, Y.C.; YANG, Y. Research Progress on Extraction and Refining of Citrus Essential Oil. Food Ferment Technol. 2012, 48, 1–5. [Google Scholar]
| Regional Sample | Peel Mass (g) | Oil Mass (g) | Yield (%) |
|---|---|---|---|
| Barishal | 20 | 0.15 | 75 |
| Dinajpur | 20 | 0.14 | 70 |
| Mymensingh | 20 | 0.136 | 68 |
| Rajshahi | 20 | 0.118 | 59 |
| Pabna | 20 | 0.094 | 47 |
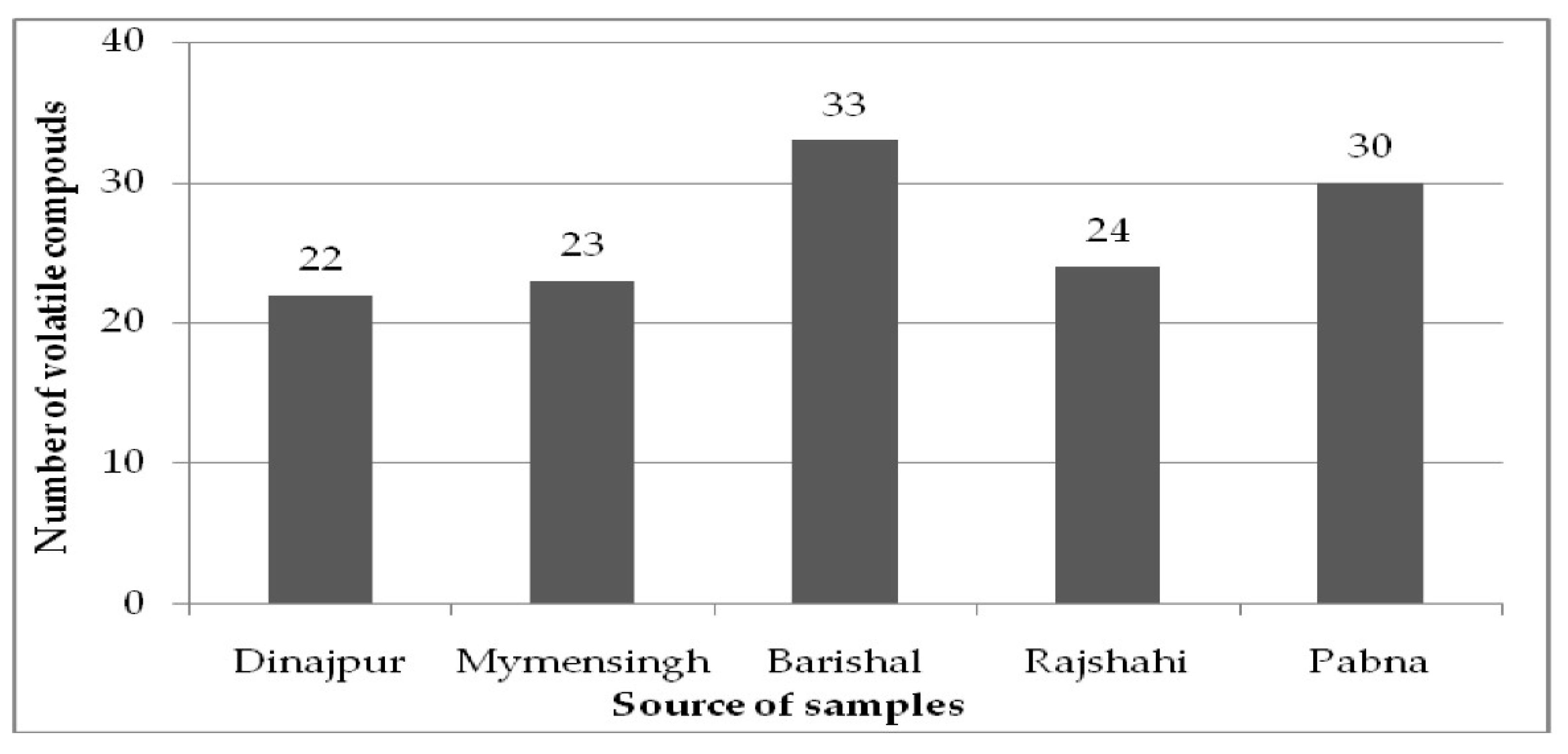
| Sl. No. | Chemical Compound | Amounts (%) | ||||
|---|---|---|---|---|---|---|
| Barishal | Dinajpur | Mymensingh | Rajshahi | Pabna | ||
| 1 | Propen 1, 2, 3 trichloro benzene | 0.24 | - | - | - | - |
| 2 | Epoxycarane-3, 2 | 0.85 | - | - | - | - |
| 3 | Chloride-butyl-benzene, tert | 0.47 | - | - | - | - |
| 4 | Pentenyl-3-thy-met-4-alpha-methyl-alpha-ethanol-oxiranen | 29.04 | 21.57 | 28.52 | 9.27 | 11.45 |
| 5 | Acetate, -benzenethanol, alpha, dimethyl | 0.14 | - | - | - | - |
| 6 | Methyl-nonyne, 7-1 | 0.12 | - | - | - | - |
| 7 | (Fenchol)-trimethyl-ol, 1, 3, 3-2-bichlo(2.2.1)hepton | 0.17 | - | - | - | - |
| 8 | (Methylethyl-1)-4-methyl-oxabicyclo(4.1.6) hepton, 1-7- | 1.37 | - | - | - | - |
| 9 | Nonennal, (E)-6 | 0.64 | - | - | - | - |
| 10 | (Methylyethyl-1)-4-methyl-oxabicyclo, 1-7 | 1.37 | 0.56 | - | - | 2.13 |
| 11 | 3,1-Cycloheptadiene | 0.44 | - | - | - | - |
| 12 | (Yl-buten 3-1)-bicyclo (2.2.1) hepton, 2 | 1.44 | - | - | - | - |
| 13 | 10-Methyl-8-ol acetate-1-tetradecen | 0.49 | - | - | - | - |
| 14 | 4,1-Cyclohexadiene-(methyl-1)-methanol,4-1 | 0.49 | - | - | - | 0.39 |
| 15 | Exo-2-hydroxycineole | 0.36 | - | - | - | - |
| 16 | 5-Heptone-methyl-one, 6-2 ester | 3.24 | - | - | - | - |
| 17 | (Ethyl-methyl-1)-benzaldehyde,4 | 0.28 | - | - | 0.04 | 0.01 |
| 18 | Methyl-bromo-ene,7-7-pentadec | 0.26 | - | - | - | - |
| 19 | (Ethyl-methyl-1)-1-methyl-ene,4-2-bicyclo(3.1.0)hexan- | 0.92 | - | - | - | - |
| 20 | (Ethyl-methyl-1)-5-methyl-phenol,2 | 0.73 | 0.29 | 0.14 | 0.04 | 0.31 |
| 21 | Terpenyl acetate-alpha-epoxy | 0.83 | - | 0.12 | 1.36 | 0.61 |
| 22 | Hylidenne-methanol,2-6, bicyclo (3.1.0) hexane | 0.53 | - | - | - | - |
| 23 | 9-E-8-Methyl-ol, acetate-2-tridecel | 0.71 | - | - | - | - |
| 24 | 15,12,9-bis (a)-octadecatrienoicacid, 2,3 | 0.09 | - | - | - | - |
| 25 | Diene, 2-[8, (7) 1]-menpha-p-2r,4r-hydroperoxide | 0.58 | - | - | - | - |
| 26 | 8-Hydroxycarvctancetone | 0.08 | - | - | - | - |
| 27 | 2,5-Dihydro-3,4-furanacetic acid | 0.38 | - | - | - | - |
| 28 | 3-Buten-2-one-4[2,6,6-trimethyl-1-cyclohexen] | 0.11 | - | - | - | - |
| 29 | 2-(3,4-Dibromo-4-methyl cyclohexyl) propanol | 0.20 | - | - | - | - |
| 30 | Phenol, 2,4-bis (1.1 dimethylehyl) | 0.06 | 0.68 | 0.46 | - | 0.30 |
| 31 | Nonadecane | 0.11 | - | - | 0.65 | - |
| 32 | Hydroxylamine-O-decylamine | 0.09 | - | - | - | - |
| 33 | Phthalic acid, isobutylnonyl ester | 0.96 | - | 0.45 | 0.46 | - |
| 34 | Cycohexanol-3 | - | 8.59 | 10.85 | 5.80 | 25.00 |
| 35 | Cycohexanol, 5-methyl-2(1-methylethen) | - | 0.80 | - | - | - |
| 36 | Isopulegol acetate | - | 0.50 | - | - | 1.91 |
| 37 | Borneol | - | 0.82 | - | 2.35 | 4.96 |
| 38 | 3-Acetoxy-p-menthane-3-one | - | 0.41 | - | - | - |
| 39 | 2-Methyl-3-(1-methylethyl cyclohexanol) | - | 0.18 | - | 0.89 | - |
| 40 | Carbamic acid, N-(1,1-ethyl bistrifluoron) | - | 0.82 | - | - | - |
| 41 | (3,3.1, 1)-4-Dimethylethyl-1,1-phenol | - | 8.00 | - | - | - |
| 42 | Picolyamine | - | 10.49 | - | - | - |
| 43 | 3,4-Methyl-dimethyl 2,3-butyryl benzoate | - | 0.57 | - | - | - |
| 44 | Phthalicacid, 2-acetylphenyl heptyl ester | - | 0.44 | - | - | - |
| 45 | 2-Phenylquinazolin-4-ol | - | 1.38 | - | - | - |
| 46 | Dibutyl phthalate | - | 4.32 | 6.85 | - | 3.03 |
| 47 | 4-Hydroxy-penzeneethanamine | - | 7.09 | - | - | - |
| 48 | 2,4-Dimethyl-5,6,11,12 tetraaza | - | 2.71 | - | - | - |
| 49 | 4-Oxo-1,2,3,4,7,12-octahydropy- | - | 1.46 | - | - | - |
| 50 | 2,2-(Dimethyl-1,1-bisene-6-methylethyl-phenol | - | 3.84 | - | - | - |
| 51 | 4-(1-methylethyl) benzenmethanol | - | 0.29 | 0.29 | - | - |
| 52 | Cyclotetrasiloxane | - | - | 1.94 | - | - |
| 53 | Benzene, 1-methyl-4(1-methylethenyl) | - | - | 0.07 | - | - |
| 54 | 1,7-Octaden-3-ol, 2,6-dimethylamin | - | - | 0.07 | - | - |
| 55 | Decamethylcyclopentasiloxane | - | - | 0.31 | - | - |
| 56 | 1,3,3-Trimethylbicyclo[2.2.1]-heptan-2-ol propanoate | - | - | 0.47 | - | - |
| 57 | 3-Cyclohexen-1-ol | - | - | 0.32 | 0.73 | 3.57 |
| 58 | Isopuleggol acetate | - | - | 1.87 | 0.24 | 1.22 |
| 59 | Borneal | - | - | 0.78 | - | - |
| 60 | Bicyclo (3.1.0) hexan 4-methyl-1 | - | - | 1.30 | - | - |
| 61 | P-menth-2-en-7-ol, trans | - | - | 0.81 | - | - |
| 62 | Nonynoic acid, 7-methyl ester | - | - | 0.34 | - | - |
| 63 | 5,7-Dodecadiyl, 1,12-diol | - | - | 0.32 | - | - |
| 64 | O-decyl-hyroxylamine | - | - | 0.21 | - | - |
| 65 | Hyroxylamine, O-decyl- | - | - | 0.28 | - | - |
| 66 | Acetic acid, Chloro-albydrate | - | - | 0.24 | - | - |
| 67 | (E)-2-Octenal | - | - | - | 0.03 | - |
| 68 | 7-methyl-1-Nonyne | - | - | - | 3.52 | - |
| 69 | Exo-Fenchol | - | - | - | 0.11 | - |
| 70 | 2,4-Pentadien-1-ol,3-pentyl-, 2Z | - | - | - | 0.73 | - |
| 71 | Isobornyl formate | - | - | - | 0.15 | - |
| 72 | Sanitolina alcohol | - | - | - | 0.78 | - |
| 73 | 1,3,3,-Trimethyl-2-oxabicyclo[2,2,2] octan-6-ol | - | - | - | 0.18 | - |
| 74 | 2-Octen-1-ol, 3,7 –dimethyl –isobutyrate | - | - | - | 0.12 | 0.06 |
| 75 | (E)-8-Methyaltetradec 1-ol acetate | - | - | - | 0.37 | - |
| 76 | 10 Methyl-8-tetradecen-1-ol acetate | - | - | - | 1.69 | - |
| 77 | Phthalic acid, 4-butyl-octyl ester | - | - | - | 0.69 | - |
| 78 | Hydroxylamine | - | - | - | 4.63 | - |
| 79 | 2-Methyl-2-propenyl benzene | - | - | - | 0.17 | - |
| 80 | 1-Pentadecyne | - | - | - | - | 0.56 |
| 81 | Bicyclo (1,2,2) heptan-2-ol, 1,3,3 trimewthyl- | - | - | - | - | 0.53 |
| 82 | (E)—6 Nonenal | - | - | - | - | 0.77 |
| 83 | Beta-cisterpineol | - | - | - | - | 0.56 |
| 84 | Trifluoro-epiiso-bomeol | - | - | - | - | 0.21 |
| 85 | Santolina alcohol | - | - | - | - | 1.46 |
| 86 | 1,5,5-Trimethyl—6-methylene cyclohexen | - | - | - | - | 0.30 |
| 87 | E-3 Bicyclo[2.1.1] trihepten | - | - | - | - | 0.91 |
| 88 | Trans-m-2,8-mentha –dienol | - | - | - | - | 0.22 |
| 89 | Bicyclo [3.1.0] hexane 2-one 5,5,6-trimethyl- | - | - | - | - | 0.96 |
| 90 | Bicyclo [3.1.0] hexane, 6-(1-isopropylidene) | - | - | - | - | 1.19 |
| 91 | 1-Methyl-3-(1-methylethenyl) cyclohexane | - | - | - | - | 0.67 |
| 92 | Eugenol | - | - | - | - | 0.92 |
| 93 | 2,4,4-Trimethyl-1,5-dienyl cyclohexane | - | - | - | - | 0.16 |
| 94 | 4-Hexyl 2,5-dihydro-3-acetic acid | - | - | - | - | 0.15 |
| 95 | 1,2-Benzenedicarboxylic acid, butyl 2-methylpropyl ester | - | - | - | - | 0.78 |
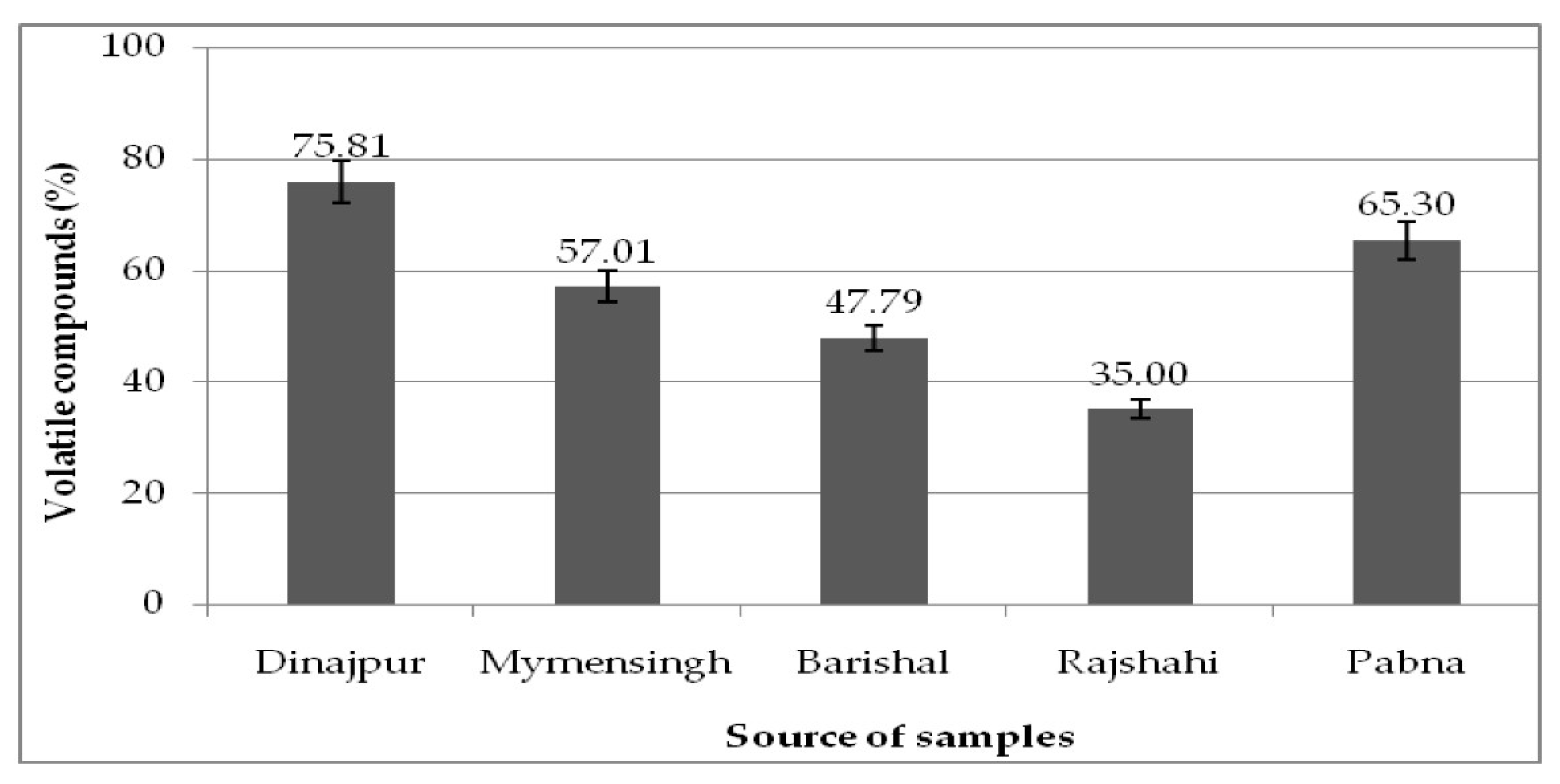
| Not identified (N.I) | 51.72 | 23.90 | 42.23 | 64.38 | 34.42 | |
| Identified | 47.79 | 75.81 | 57.01 | 35.00 | 65.30 | |
| Total (%) | 99.51 | 99.23 | 99.24 | 99.38 | 99.72 | |
| SN | Bacterial Strains | Diameter of Zone of Inhibition (mm) * | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| BS | DS | MS | RS | PS | |||||||
| Cultures (5 μL/Petridish) | Standard (Ciprocin) (10 μg/disc) | Cultures (5 μL/Petridish) | Standard (Ciprocin) (10 μg/disc) | Cultures (5 μL/Petridish) | Standard (Ciprocin) (10 μg/disc) | Cultures (5 μL/Petridis) | Standard (Ciprocin) (10 μg/disc) | Cultures (5 μL/Petridish) | Standard (Ciprocin) (10 μg/disc) | ||
| 1 | Salmonella spp. | 12.00 ± 0.17 | 23.00 ± 0.58 | 11.50 ± 0.01 | 22.5 ± 0.007 | 10.87 ± 0.015 | 21.63 ± 0.01 | 10.50 ± 0.007 | 21.50 ± 0.007 | 12.80 ± 0.03 | 22.00 ± 0.20 |
| 2 | Staphylococcus aureus | 14.00 ± 0.17 | 22.00 ± 0.27 | 13.83 ± 0.04 | 21.96 ± 0.0004 | 13.13 ± 0.001 | 21.00 ± 0.007 | 13.40 ± 0.190 | 21.40 ± 0.009 | 13.90 ± 0.0001 | 21.87 ± 0.05 |
| 3 | Escherichia coli | 13.00 ± 0.29 | 22.00 ± 0.22 | 12.7 ± 0.015 | 21.26 ± 0.019 | 12.37 ± 0.01 | 20.67 ± 0.01 | 13.17 ± 0.001 | 20.70 ± 0.02 | 13.40 ± 0.005 | 22.00 ± 0.67 |
| SN | Fungal Strains | Diameter of Zone of Inhibition (mm) * | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| BS | DS | MS | RS | PS | |||||||
| Cultures (5 μL/Petridish) | Standard (Fluconozole) (10 μg/disc) | Cultures (5 μL/Petridish) | Standard (Fluconozole) (10 μg/disc) | Cultures (5 μL/Petridish) | Standard (Fluconozole) (10 μg/disc) | Cultures (5 μL/Petridish) | Standard (Fluconozole) (10 μg/disc) | Cultures (5 μL/Petridish) | Standard (Fluconozole) (10 μg/disc) | ||
| 1 | A. niger | 11.33 ± 0.17 | 19.13 ± 0.35 | 11.27 ± 0.02 | 19.43 ± 0.01 | 11.07 ± 0.0002 | 18.90 ± 0.0002 | 11.63 ± 0.0003 | 18.9 ± 0.020 | 11.10 ± 0.0005 | 19.73 ± 0.0005 |
| 2 | P. oxalicum | 13.17 ± 0.17 | 20.15 ± 0.77 | 12.93 ± 0.02 | 20.15 ± 0.77 | 12.87 ± 0.0004 | 19.87 ± 0.004 | 12.93 ± 0.002 | 19.3 ± 0.003 | 13.67 ± 1.97 | 20.37 ± 0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Plabon, M.E.A.; Mondal, S.C.; Or Rashid, M.M.; Chowdhury, M.K.; Saeid, A.; Althobaiti, F.; Dessok, E.S.; Rehmani, M.I.A.; Mustafa, S.K.; Islam, M.S. Chemical Composition and Anti-Microbial Activity of Hog Plum (Spondias mombin L.) Peel Oil Extracted from Different Regions of Tropical Climates. Horticulturae 2021, 7, 428. https://doi.org/10.3390/horticulturae7110428
Plabon MEA, Mondal SC, Or Rashid MM, Chowdhury MK, Saeid A, Althobaiti F, Dessok ES, Rehmani MIA, Mustafa SK, Islam MS. Chemical Composition and Anti-Microbial Activity of Hog Plum (Spondias mombin L.) Peel Oil Extracted from Different Regions of Tropical Climates. Horticulturae. 2021; 7(11):428. https://doi.org/10.3390/horticulturae7110428
Chicago/Turabian StylePlabon, Md. Emrul Ahsan, Shakti Chandra Mondal, Md. Mamun Or Rashid, M. Kaium Chowdhury, Abu Saeid, Fayez Althobaiti, Eldessoky S. Dessok, Muhammad Ishaq Asif Rehmani, Syed Khalid Mustafa, and Mohammad Sohidul Islam. 2021. "Chemical Composition and Anti-Microbial Activity of Hog Plum (Spondias mombin L.) Peel Oil Extracted from Different Regions of Tropical Climates" Horticulturae 7, no. 11: 428. https://doi.org/10.3390/horticulturae7110428
APA StylePlabon, M. E. A., Mondal, S. C., Or Rashid, M. M., Chowdhury, M. K., Saeid, A., Althobaiti, F., Dessok, E. S., Rehmani, M. I. A., Mustafa, S. K., & Islam, M. S. (2021). Chemical Composition and Anti-Microbial Activity of Hog Plum (Spondias mombin L.) Peel Oil Extracted from Different Regions of Tropical Climates. Horticulturae, 7(11), 428. https://doi.org/10.3390/horticulturae7110428








