Evaluation of Two Wild Populations of Hedge Mustard (Sisymbrium officinale (L.) Scop.) as a Potential Leafy Vegetable
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Non-Destructive Analyses
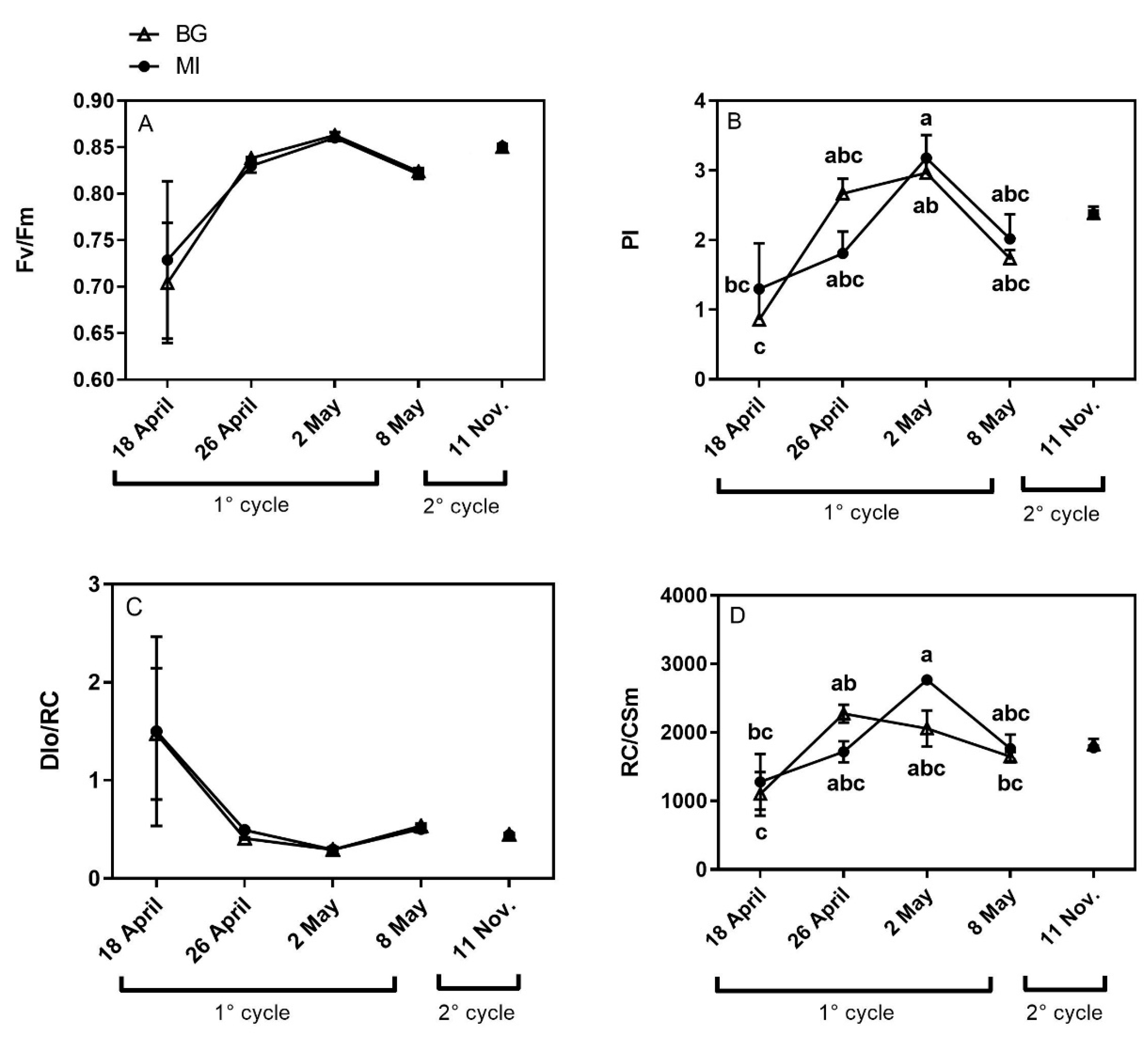
Chlorophyll a Fluorescence
2.3. Destructive Analyses
2.3.1. Chlorophyll and Carotenoids
2.3.2. Phenolic Index, Total Phenols, and Anthocyanins
2.3.3. Ascorbic Acid Determination
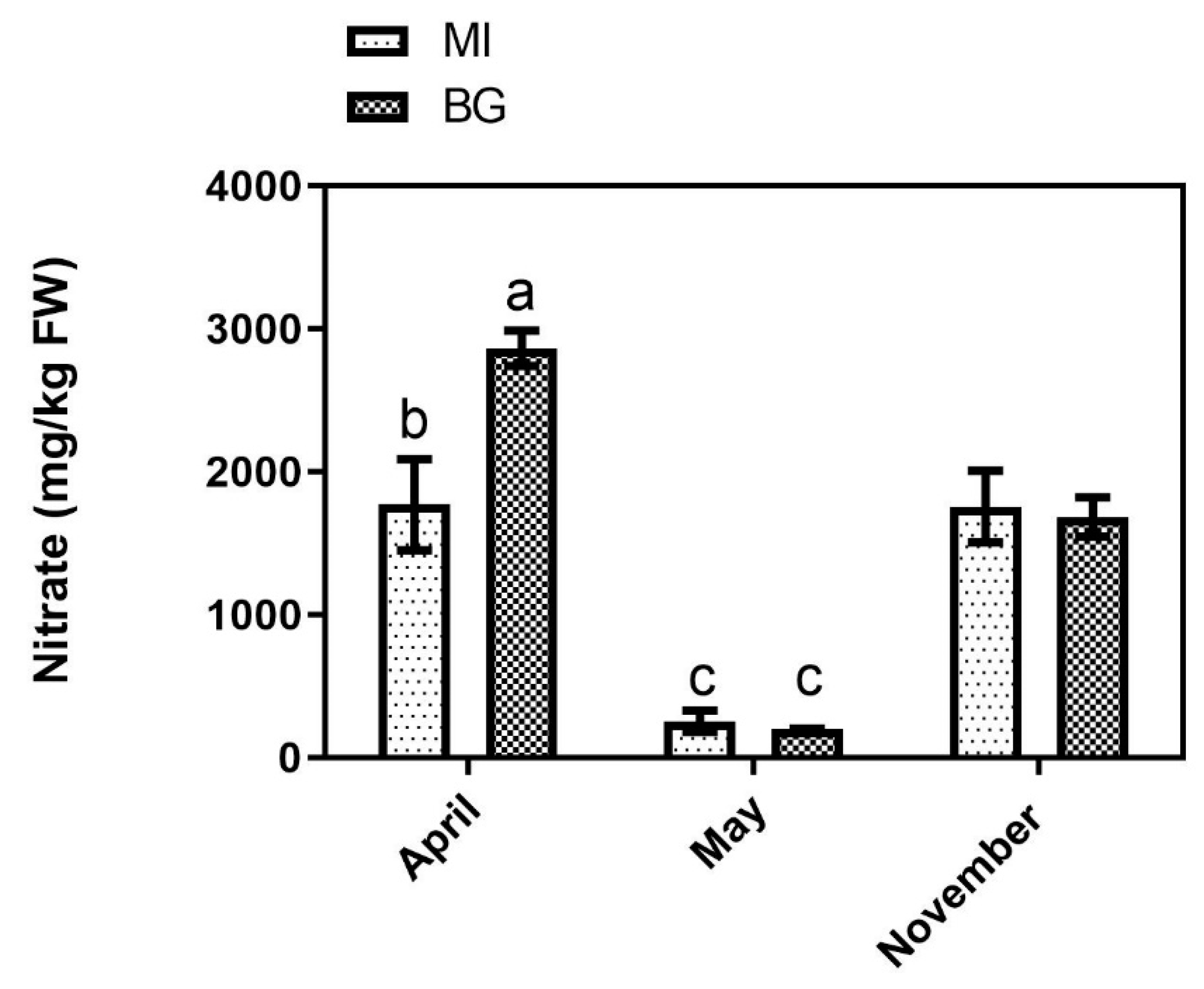
2.3.4. Nitrate Determination
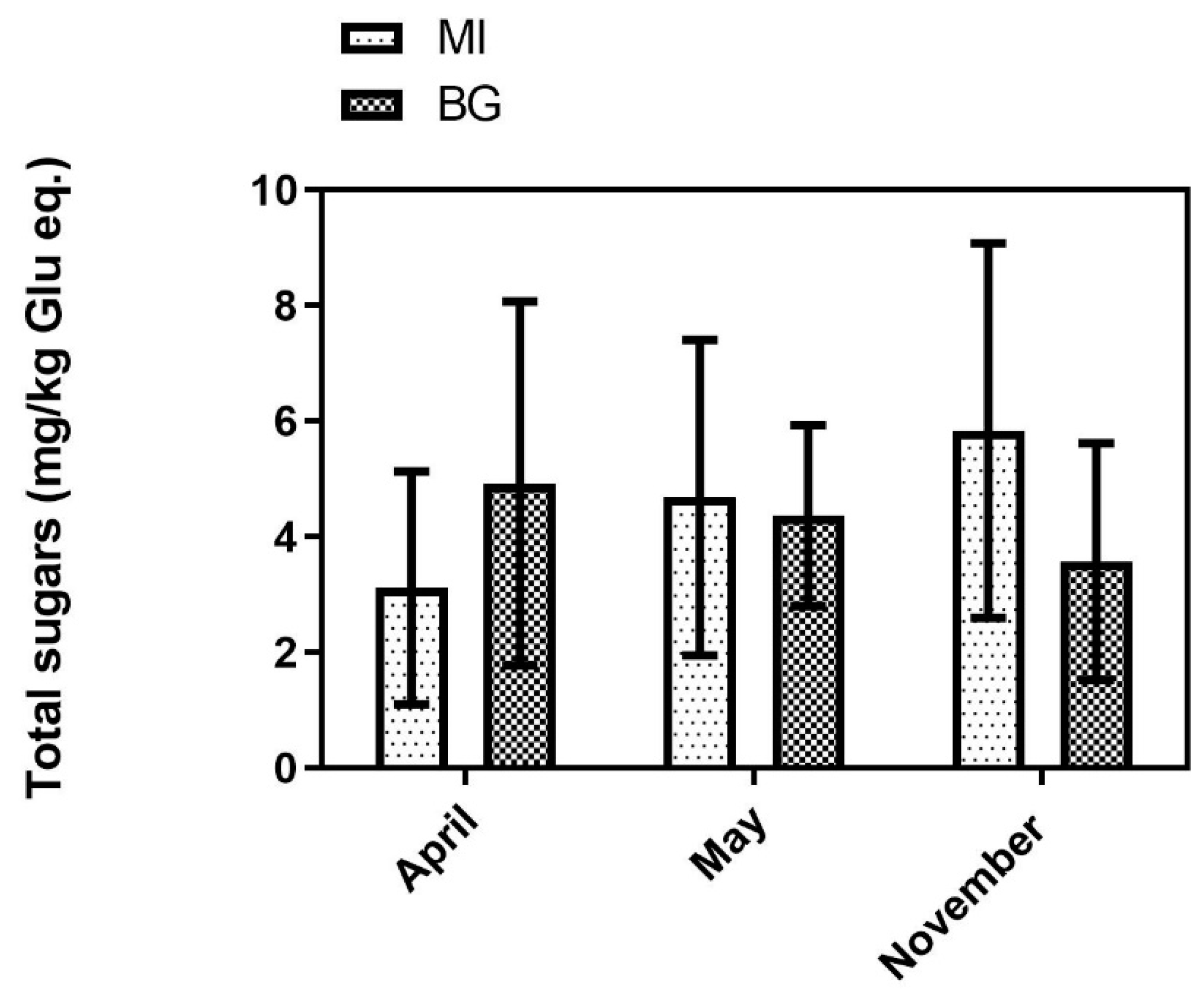
2.3.5. Total Sugar Determination
2.4. Statistical Analyses
3. Results
3.1. Total Chlorophylls, Carotenoids, Phenols, and Anthocyanins
3.2. Chlorophyll a Fluorescence Measurements
3.3. Nitrate and Total Sugars
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Pignatti, S. Flora d’Italia; Edizioni Edagricole: Bologna, Italy, 1982; Volume 2, p. 377. [Google Scholar]
- Bouwmeester, H.J.; Karseen, C.M. Annual changes in dormancy and germination in seeds of Sisymbriumofficinale (L.) Scop. New Phytol. 1993, 124, 179–191. [Google Scholar] [CrossRef]
- Politi, M.; Braca, A.; Altinier, G.; Sosa, S.; Ndjoko, K.; Wolfender, J.L.; Hostettmann, K.; Jimenez-Barbero, J. Different approaches to study the traditional remedy of “hierba del canto” Sisymbriumofficinale (L.) Scop. Bol. Latinoam. Caribe Plantasmedicinales Aromàt. 2008, 7, 30–37. [Google Scholar]
- Blažević, I.; Radonić, A.; Mastelić, J.; Zekić, M.; Skočibusić, M.; Maravić, A. Hedge Mustard (Sisymbriumofficinale): Chemical diversity of volatiles and their antimicrobial activity. Chem. Biodivers. 2010, 7, 2023–2034. [Google Scholar] [CrossRef] [PubMed]
- Di Sotto, A.; Vitalone, A.; Nicoletti, M.; Piccin, A.; Mazzanti, G. Pharmacological and phytochemicalstudy on Sisymbriumofficinale Scop. Extract. J. Ethnopharmacol. 2010, 127, 731–736. [Google Scholar] [CrossRef] [PubMed]
- EMA—European Medicines Agency. Assessment report on Sisymbriumofficinale (L.) Scop., Herba; EMA: London, UK, 15 January 2014. [Google Scholar]
- Calcinoni, O. Sisymbrium “Singers’ Plant” Efficacy in Reducing Perceived Vocal Tract Disability. J. Otolaryngol. Ent Res. 2017, 8, 00243. [Google Scholar] [CrossRef]
- Carnat, A.; Fraisse, D.; Carnat, A.P.; Groubert, A.; Lamaison, J.L. Normalization of hedge mustard, Sisymbriumofficinale L. Ann. Pharm. Fr. 1998, 56, 36–39. [Google Scholar]
- Pardossi, A. Orticoltura. Principi e Pratica; Edizioni Edagricole: Bologna, Italy, 2018; pp. 261–276. [Google Scholar]
- Lichtenthaler, H.K. Chlorophylls and carotenoids: Pigmentsof photosynthetic membranes. Methods Enzymol. 1987, 148, 350–382. [Google Scholar]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventos, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Methods Enzymol. 1999, 299, 152–178. [Google Scholar]
- Giusti, M.M.; Rodríguez-Saona, L.E.; Wrolstad, R.E. Molar absorptivity and color characteristics of acylated and non-acylated pelargonidin-based anthocyanins. J. Agric. Food Chem. 1999, 47, 4631–4637. [Google Scholar] [CrossRef] [PubMed]
- Rizzolo, A.; Brambilla, A.; Valsecchi, S.; Eccher-Zerbini, P. Evaluation of sampling and extraction procedures for the analysis of ascorbic acid from pear fruit tissue. Food Chem. 2002, 77, 257–262. [Google Scholar] [CrossRef]
- Cataldo, C.A.; Maroon, M.; Schrader, L.E.; Youngs, V.L. Rapid colorimetric determination of nitrate in plant tissue by titration of salicylic acid. Commun. Soil Sci. Plant Anal. 1975, 6, 71–80. [Google Scholar] [CrossRef]
- Yemm, E.W.; Willis, A.J. The estimation of carbohydrates in plant extracts by anthrone. Biochem. J. 1954, 57, 508–514. [Google Scholar] [CrossRef] [PubMed]
- Branca, F. Studies on some wild Brassicaceae species utilizable as vegetables in the Mediterranean areas. Plant Genet. Resour. Newsl. 1995, 104, 6–9. [Google Scholar]
- Branca, F.; Li, G.; Goyal, S.; Quiros, C.F. Survey of aliphatic glucosinolates in Sicilian wild and cultivated Brassicaceae. Phytochemistry 2002, 59, 717–724. [Google Scholar] [CrossRef]
- Albrecht, J. Ascorbic Acid Content And Retention In Lettuce1. J. Food Qual. 1993, 16, 311–316. [Google Scholar] [CrossRef]
- Žnidarčič, D.; Ban, D.; Šircelj, H. Carotenoid and chlorophyll composition of commonly consumed leafy vegetables in Mediterranean countries. Food Chem. 2011, 129, 1164–1168. [Google Scholar] [CrossRef]
- Ferrante, A.; Maggiore, T. Chlorophyll a fluorescence measurements to evaluate storage time and temperature of Valeriana leafy vegetables. Postharvest Biol. Technol. 2007, 45, 73–80. [Google Scholar] [CrossRef]
- Fallovo, C.; Rouphael, Y.; Rea, E.; Battistelli, A.; Colla, G. Nutrient solution concentration and growing season affect yield and quality of Lactuca sativa L. var. acephala in floating raft culture. J. Sci. Food Agric. 2009, 89, 1682–1689. [Google Scholar] [CrossRef]
- Ferrante, A.; Incrocci, L.; Maggini, R.; Serra, G.; Tognoni, F. Colour changes of fresh-cut leafy vegetables during storage. J. Food Agric. Environ. 2004, 2, 40–44. [Google Scholar]
- Colonna, E.; Rouphael, Y.; Barbieri, G.; De Pascale, S. Nutritional quality of ten leafy vegetables harvested at two light intensities. Food Chem. 2016, 199, 702–710. [Google Scholar] [CrossRef]
- Bulgari, R.; Cola, G.; Ferrante, A.; Franzoni, G.; Mariani, L.; Martinetti, L. Micrometeorological environment in traditional and photovoltaic greenhouses and effects on growth and quality of tomato (Solanum lycopersicum L.). Ital. J. Agrometeorol. 2015, 2, 27–38. [Google Scholar]
- Maxwell, K.; Johnson, G.N. Chlorophyll fluorescence—A practical guide. EXBOTJ 2000, 51, 659–668. [Google Scholar] [CrossRef]
- Alberici, A.; Quattrini, E.; Penati, M.; Martinetti, L.; Gallina, P.M.; Ferrante, A.; Schiavi, M. Effect of the Reduction of Nutrient Solution Concentration on Leafy Vegetables Quality Grown in Floating System. Acta Hortic. 2008, 801, 1167–1176. [Google Scholar] [CrossRef]
- EFSA—European Food Safety Authority. Nitrate in Vegetables. Scientific Opinion of the Panel on Contaminants in the Food Chain; Question N. EFSA-Q-2006-071; EFSA: Parma, Italy, 2008; Volume 689, pp. 1–79. [Google Scholar]
- European Commission. Maximum Levels for Nitrates in Foodstuffs; Regulation (EU) N. 1258/2011 Amending Regulation (EC) N. 1881/2006; Official Journal European Union: Brussel, Belgium, 2011; pp. 15–17. [Google Scholar]
- Hanafy Ahmed, A.H.; Khalil, M.K.; Farrag, A.M. Nitrate accumulation, growth, yield and chemical composition of Rocket (Eruca vesicaria subsp. sativa) plant as affected by NPK fertilization, kinetin and salicylic acid. Ann. Agric. Sci. Cairo 2002, 47, 1–26. [Google Scholar]
- Ferrante, A.; Incrocci, L.; Maggini, R.; Serra, G.; Tognoni, F. Preharvest and postharvest strategies for reducing nitrate content in rocket (Eruca sativa L.). Acta Hortic. 2003, 628, 153–159. [Google Scholar] [CrossRef]
- Tannenbaum, S.R.; Wishnok, J.S.; Leaf, C.D. Inhibition of nitrosamine formation by ascorbic acid. Am. J. Clin. Nutr. 1991, 53, 247S–250S. [Google Scholar] [CrossRef]
- Shad, A.A.; Shah, H.U.; Bakht, J. Ethnobotanical assessment and nutritive potential of wild food plants. J. Anim. Plant Sci. 2013, 23, 92–97. [Google Scholar]
| Cycle | Wild Population | Fresh Weight (g/plant) | Dry Matter (%) | |||
|---|---|---|---|---|---|---|
| I | May | MI | 2.41 | ns | 10.64 | ns |
| BG | 4.00 | 10.76 | ||||
| II | November | MI | 2.26 | ns | 8.07 | ns |
| BG | 2.45 | 7.87 |
| Cycle | Wild Population | Chl a (mg/g FW) | Chl b (mg/g FW) | Total Carotenoids (mg/g FW) | ||||
|---|---|---|---|---|---|---|---|---|
| I | April | MI | 0.63 b | 0.19 b | 0.120 | |||
| BG | 0.93 ab | 0.37 ab | 0.143 | ns | ||||
| May | MI | 1.22 a | 0.42 a | 0.215 | ||||
| BG | 0.79 ab | 0.27 ab | 0.157 | ns | ||||
| II | November | MI | 1.56 | ns | 0.51 | ns | 0.382 | ns |
| BG | 1.44 | 0.47 | 0.379 |
| Cycle | Wild Population | ASA (mg/100 g FW) | Phenolic Index (ABS320nm/g FW) | Total Phenols | Anthocyanins (mg/100 g FW) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| I | April | MI | - | 27.45 | 1.13 | 32.13 ab | ||||
| BG | - | 26.72 | ns | 1.09 | ns | 26.38 b | ||||
| May | MI | - | 19.05 | 0.75 | 14.88 c | |||||
| BG | - | 32.60 | ns | 1.35 | ns | 38.79 a | ||||
| II | November | MI | 54.45 | * | 42.51 | * | 1.62 | ns | 29.53 | ns |
| BG | 34.57 | 55.44 | 2.37 | 31.51 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guarise, M.; Borgonovo, G.; Bassoli, A.; Ferrante, A. Evaluation of Two Wild Populations of Hedge Mustard (Sisymbrium officinale (L.) Scop.) as a Potential Leafy Vegetable. Horticulturae 2019, 5, 13. https://doi.org/10.3390/horticulturae5010013
Guarise M, Borgonovo G, Bassoli A, Ferrante A. Evaluation of Two Wild Populations of Hedge Mustard (Sisymbrium officinale (L.) Scop.) as a Potential Leafy Vegetable. Horticulturae. 2019; 5(1):13. https://doi.org/10.3390/horticulturae5010013
Chicago/Turabian StyleGuarise, Marta, Gigliola Borgonovo, Angela Bassoli, and Antonio Ferrante. 2019. "Evaluation of Two Wild Populations of Hedge Mustard (Sisymbrium officinale (L.) Scop.) as a Potential Leafy Vegetable" Horticulturae 5, no. 1: 13. https://doi.org/10.3390/horticulturae5010013
APA StyleGuarise, M., Borgonovo, G., Bassoli, A., & Ferrante, A. (2019). Evaluation of Two Wild Populations of Hedge Mustard (Sisymbrium officinale (L.) Scop.) as a Potential Leafy Vegetable. Horticulturae, 5(1), 13. https://doi.org/10.3390/horticulturae5010013








