Implications of Smallholder Farm Production Diversity for Household Food Consumption Diversity: Insights from Diverse Agro-Ecological and Market Access Contexts in Rural Tanzania
Abstract
1. Introduction
2. Literature Review
2.1. Farm Production Diversity in Smallholder Agriculture
2.2. Linking Production Diversity to Consumption Diversity
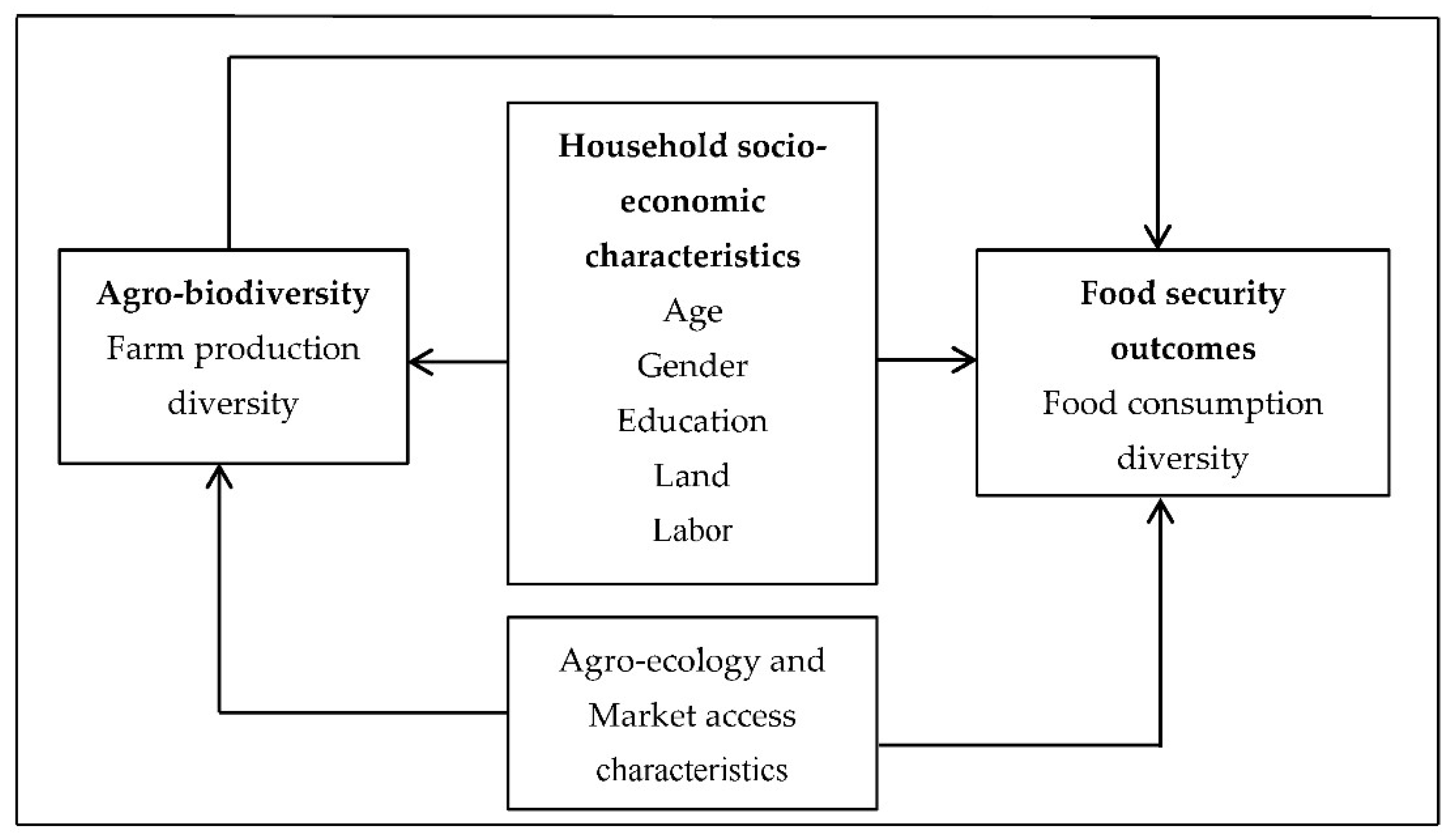
3. Materials and Methods
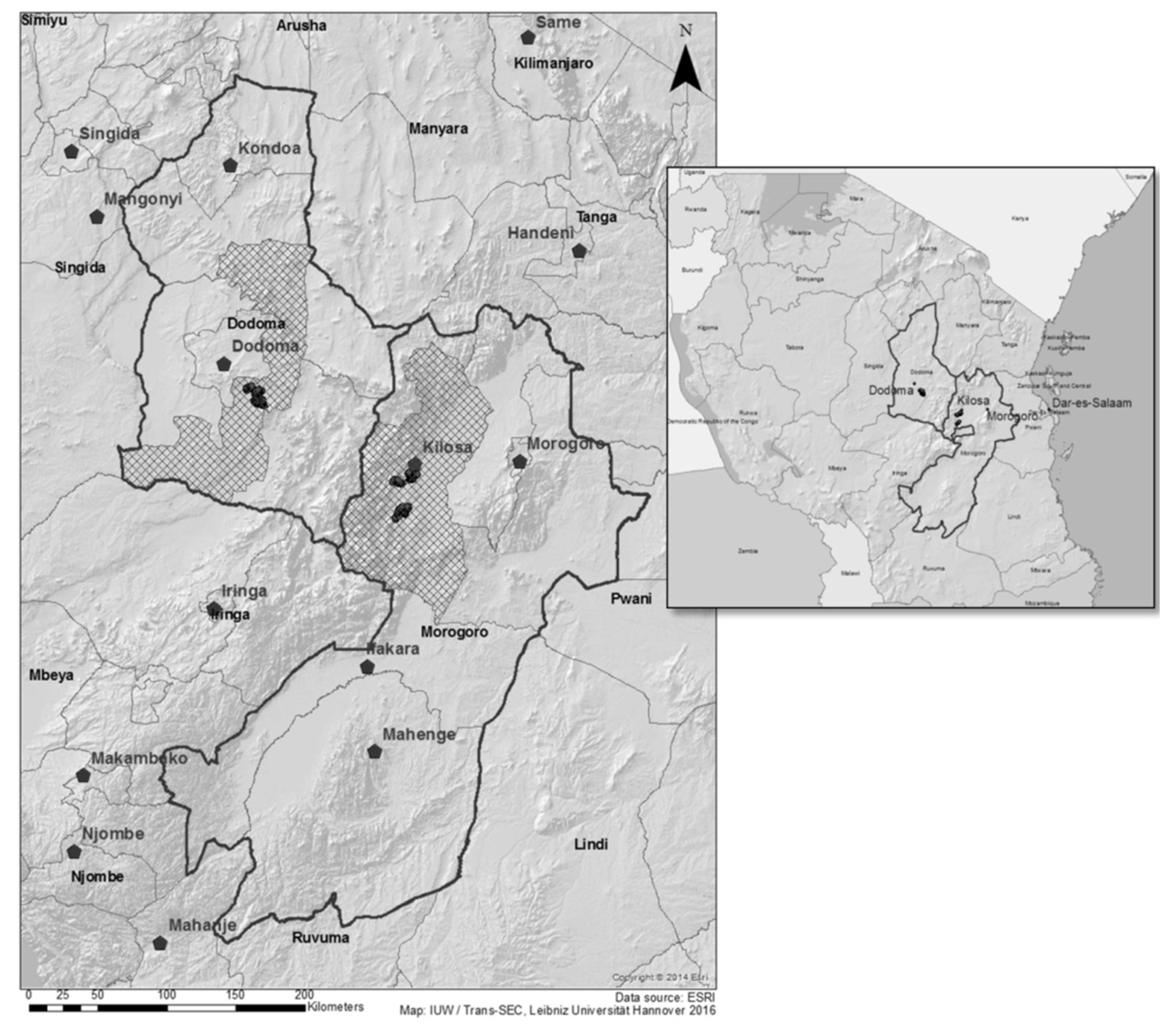
3.1. Study Area and Data
3.2. Data Collection
3.3. Measures of Diversity
3.4. Empirical Strategy
3.4.1. Analyzing the Determinants of Farm Production Diversity
3.4.2. Analyzing the Influence of Farm Production Diversity on Consumption Diversity
4. Results
4.1. Descriptive Results
4.1.1. Sample Characteristics
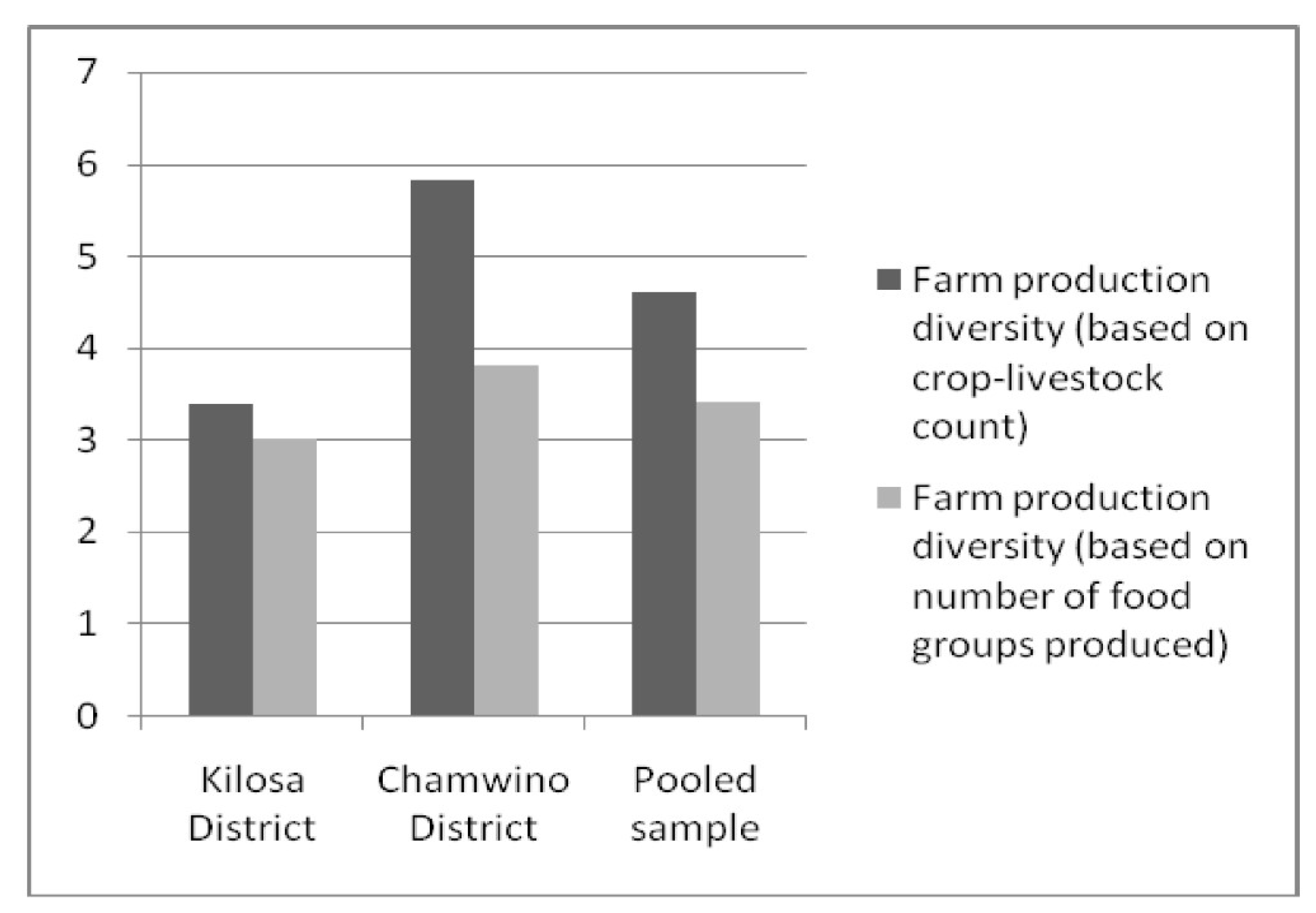
4.1.2. Comparison of Farm Production Diversity by Agro-Ecology and Market Access
4.1.3. Comparison of Food Consumption Diversity in Kilosa and Chamwino Districts
4.2. Determinants of Farm Production Diversity
4.3. The Role of Farm Production Diversity on Household Food Consumption Diversity
5. Discussion
5.1. The Nature and Drivers of Farm Production Diversity
5.2. The Influence of Farm Production Diversity on Food Consumption Diversity
5.3. The Role of Market Access in Food Consumption Diversity
5.4. Farm Production Diversity and Seasonal Food Consumption
5.5. Limitations
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Variable | HDDS | HDDS (Planting) | HDDS (Pre-Harvest) | HDDS (Post-Harvest) | FVS |
|---|---|---|---|---|---|
| Crop-livestock count | 0.032 *** | 0.016 *** | 0.018 *** | 0.011 ** | 0.051 *** |
| (0.006) | (0.006) | (0.006) | (0.005) | (0.009) | |
| Age of HH head (years) | −0.004 *** | −0.003 *** | −0.003 *** | −0.002 *** | −0.006 *** |
| (0.001) | (0.001) | (0.001) | (0.001) | (0.001) | |
| Gender of HH head (Male = 1) | −0.022 | 0.044 | −0.039 | −0.027 | −0.030 |
| (0.035) | (0.036) | (0.033) | (0.027) | (0.045) | |
| Education of HH head (School years) | 0.003 | −0.005 | 0.001 | 0.001 | 0.004 |
| (0.004) | (0.004) | (0.004) | (0.003) | (0.006) | |
| Land size owned (ha.) | 0.001 | 0.008 | 0.005 | −0.001 | −0.003 |
| (0.008) | (0.006) | (0.006) | (0.005) | (0.010) | |
| Livestock owned (TLU) | 0.005 | 0.011 *** | 0.007 * | 0.007 ** | 0.003 |
| (0.004) | (0.004) | (0.004) | (0.003) | (0.007) | |
| Labor (Worker equivalents) | −0.001 | 0.000 | −0.001 | 0.008 | 0.009 |
| (0.009) | (0.009) | (0.010) | (0.007) | (0.013) | |
| Per capita per month: Low-middle | 0.025 | 0.051 | 0.002 | 0.003 | 0.021 |
| (0.040) | (0.049) | (0.043) | (0.033) | (0.054) | |
| Per capita per month: Middle | 0.020 | 0.163 *** | 0.075 * | 0.033 | 0.041 |
| (0.045) | (0.049) | (0.044) | (0.034) | (0.057) | |
| Per capita per month: High-middle | 0.124 *** | 0.209 *** | 0.124 *** | 0.075 ** | 0.157 *** |
| (0.044) | (0.046) | (0.044) | (0.033) | (0.055) | |
| Per capita per month: High | 0.114 ** | 0.225 *** | 0.147 *** | 0.073 ** | 0.161 ** |
| (0.048) | (0.052) | (0.052) | (0.036) | (0.066) | |
| Share of home consumption | −0.006 | −0.007 | 0.008 | −0.001 | −0.002 |
| (0.033) | (0.034) | (0.033) | (0.026) | (0.043) | |
| Access to market information (Yes = 1) | 0.123 *** | 0.124 *** | 0.147 *** | 0.079 *** | 0.131 *** |
| (0.029) | (0.029) | (0.029) | (0.023) | (0.037) | |
| Distance to nearest paved road | 0.000 | −0.005 | 0.003 | −0.005 | 0.003 |
| (0.009) | (0.009) | (0.009) | (0.008) | (0.012) | |
| Access to off-farm employment (Yes = 1) | −0.012 | 0.044 | −0.005 | −0.006 | 0.005 |
| (0.029) | (0.029) | (0.028) | (0.022) | (0.038) | |
| Access to non-farm self-employment (Yes = 1) | 0.044 | 0.039 | 0.014 | 0.056 *** | 0.031 |
| (0.028) | (0.027) | (0.027) | (0.020) | (0.038) | |
| Household asset holding (asset score) | 0.000 *** | 0.000 ** | 0.000 ** | 0.000 | 0.000 *** |
| (0.000) | (0.000) | (0.000) | (0.000) | (0.000) | |
| Household resides in Ilolo village | 0.048 | −0.001 | 0.059 | −0.024 | 0.065 |
| (0.054) | (0.055) | (0.054) | (0.047) | (0.078) | |
| Household resides in Ndebwe village | 0.035 | 0.026 | 0.082 ** | 0.023 | 0.045 |
| (0.035) | (0.036) | (0.034) | (0.028) | (0.045) | |
| Constant | 1.556 *** | 1.597 *** | 1.605 *** | 1.934 *** | 1.908 *** |
| (0.132) | (0.132) | (0.126) | (0.104) | (0.175) | |
| ln(alpha) | −3.673 | ||||
| (0.369) | |||||
| Alpha | 0.785 | ||||
| (0.095) | |||||
| Observations | 449 | 449 | 449 | 449 | 449 |
| Wald chi2 | 166.31 | 130.43 | 117.44 | 72.48 | 127.74 |
| Probability > chi2 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Pseudo R2 | 0.034 | 0.032 | 0.027 | 0.013 | 0.052 |
| HDDS | HDDS (Planting) | HDDS (Pre-Harvest) | HDDS (Post-Harvest) | FVS | |
|---|---|---|---|---|---|
| Number of food groups produced | 0.048 *** | 0.015 | 0.030 ** | 0.014 | 0.061 *** |
| (0.013) | (0.014) | (0.014) | (0.012) | (0.016) | |
| Age of HH head (years) | −0.004 *** | −0.003 *** | −0.002 * | −0.002 ** | −0.005 *** |
| (0.001) | (0.001) | (0.001) | (0.001) | (0.001) | |
| Gender of HH head (Male = 1) | 0.001 | 0.068 | −0.025 | −0.025 | −0.026 |
| (0.036) | (0.043) | (0.041) | (0.034) | (0.046) | |
| Education of HH head (School years) | 0.001 | −0.006 | 0.001 | 0.003 | 0.003 |
| (0.004) | (0.005) | (0.005) | (0.004) | (0.006) | |
| Land size owned (ha.) | 0.007 | 0.016 * | 0.011 | 0.001 | 0.010 |
| (0.007) | (0.008) | (0.008) | (0.007) | (0.010) | |
| Livestock owned (TLU) | 0.007 | 0.021 *** | 0.015 *** | 0.013 *** | 0.007 |
| (0.006) | (0.005) | (0.005) | (0.004) | (0.007) | |
| Labor (Worker equivalents) | −0.004 | −0.005 | −0.012 | 0.010 | 0.013 |
| (0.009) | (0.011) | (0.012) | (0.009) | (0.013) | |
| Per capita per month: Low-middle | −0.004 | 0.027 | −0.024 | 0.012 | 0.034 |
| (0.041) | (0.054) | (0.052) | (0.040) | (0.055) | |
| Per capita per month: Middle | 0.042 | 0.171 *** | 0.081 | 0.054 | 0.060 |
| (0.045) | (0.055) | (0.052) | (0.042) | (0.058) | |
| Per capita per month: High-middle | 0.124 *** | 0.211 *** | 0.143 *** | 0.112 *** | 0.167 *** |
| (0.045) | (0.054) | (0.054) | (0.042) | (0.056) | |
| Per capita per month: High | 0.116 ** | 0.239 *** | 0.166 *** | 0.100 ** | 0.197 *** |
| (0.050) | (0.060) | (0.064) | (0.047) | (0.068) | |
| Share of home consumption | −0.001 | −0.050 | −0.003 | −0.029 | −0.023 |
| (0.033) | (0.040) | (0.040) | (0.032) | (0.044) | |
| Access to market information (Yes = 1) | 0.125 *** | 0.169 *** | 0.177 *** | 0.093 *** | 0.141 *** |
| (0.030) | (0.035) | (0.035) | (0.028) | (0.038) | |
| Distance to nearest paved road | −0.006 | −0.012 | −0.004 | −0.013 | −0.000 |
| (0.009) | (0.010) | (0.011) | (0.010) | (0.013) | |
| Access to off-farm employment (Yes = 1) | −0.000 | 0.067 * | 0.009 | −0.012 | 0.014 |
| (0.030) | (0.035) | (0.035) | (0.028) | (0.039) | |
| Access to non-farm self-employment (Yes = 1) | 0.038 | 0.028 | −0.003 | 0.073 *** | 0.034 |
| (0.030) | (0.033) | (0.034) | (0.027) | (0.039) | |
| Household asset holding (asset score) | 0.000 *** | 0.000 *** | 0.000 *** | 0.000 | 0.000 ** |
| (0.000) | (0.000) | (0.000) | (0.000) | (0.000) | |
| Household resides in Ilolo village | 0.048 | −0.073 | 0.014 | −0.086 | 0.081 |
| (0.055) | (0.066) | (0.069) | (0.057) | (0.079) | |
| Household resides in Ndebwe village | 0.055 | −0.016 | 0.061 | 0.007 | 0.046 |
| (0.035) | (0.042) | (0.042) | (0.035) | (0.046) | |
| Constant | 1.286 *** | 1.489 *** | 1.407 *** | 1.785 *** | 1.933 *** |
| (0.136) | (0.160) | (0.163) | (0.129) | (0.181) | |
| ln(alpha) | −3.447 | ||||
| (0.308) | |||||
| Alpha | 0.823 | ||||
| (0.075) | |||||
| Observations | 449 | 449 | 449 | 449 | 449 |
| Wald chi2 | 139.84 | 162.66 | 135.11 | 86.41 | 107.96 |
| Probability > chi2 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Pseudo R2 | 0.027 | 0.038 | 0.031 | 0.017 | 0.044 |
| HDDS | HDDS (Planting) | HDDS (Pre-Harvest) | HDDS (Post-Harvest) | FVS | |
|---|---|---|---|---|---|
| Crop-livestock count | 0.001 | 0.016 ** | 0.008 | 0.006 | 0.008 |
| (0.007) | (0.005) | (0.005) | (0.005) | (0.008) | |
| Age of HH head (years) | −0.002 ** | −0.001 | −0.000 | −0.000 | −0.002 ** |
| (0.001) | (0.001) | (0.001) | (0.001) | (0.001) | |
| Gender of HH head (Male = 1) | 0.008 | 0.007 | −0.001 | −0.023 | 0.015 |
| (0.032) | (0.029) | (0.028) | (0.024) | (0.039) | |
| Education of HH head (School years) | 0.003 | 0.002 | 0.004 | 0.004 | 0.001 |
| (0.004) | (0.003) | (0.003) | (0.003) | (0.005) | |
| Land size owned (ha.) | 0.008 | 0.014 *** | 0.010 ** | 0.012 *** | 0.007 |
| (0.007) | (0.005) | (0.005) | (0.004) | (0.008) | |
| Livestock owned (TLU) | −0.002 | 0.000 | 0.000 | 0.000 | −0.004 ** |
| (0.001) | (0.000) | (0.000) | (0.000) | (0.002) | |
| Labor (Worker equivalents) | 0.007 | −0.001 | 0.005 | 0.006 | 0.012 |
| (0.009) | (0.008) | (0.006) | (0.005) | (0.011) | |
| Per capita per month: Low-middle | 0.051 | −0.041 | −0.025 | −0.019 | 0.092 * |
| (0.042) | (0.035) | (0.037) | (0.030) | (0.053) | |
| Per capita per month: Middle | 0.097 ** | 0.020 | 0.057 * | 0.036 | 0.135 *** |
| (0.039) | (0.030) | (0.030) | (0.026) | (0.051) | |
| Per capita per month: High-middle | 0.117 *** | −0.010 | 0.028 | 0.016 | 0.167 *** |
| (0.038) | (0.030) | (0.029) | (0.025) | (0.049) | |
| Per capita per month: High | 0.154 *** | 0.040 | 0.053 ** | 0.038 * | 0.192 *** |
| (0.034) | (0.028) | (0.027) | (0.022) | (0.044) | |
| Share of home consumption | −0.036 | −0.036 | −0.028 | −0.034 | −0.048 |
| (0.031) | (0.026) | (0.027) | (0.023) | (0.039) | |
| Access to market information (Yes = 1) | 0.063 ** | 0.039 * | 0.024 | 0.038 * | 0.071 ** |
| (0.027) | (0.023) | (0.023) | (0.021) | (0.033) | |
| Distance to nearest paved road | −0.033 *** | −0.015 ** | −0.009 | −0.013 ** | −0.042 *** |
| (0.007) | (0.006) | (0.006) | (0.005) | (0.009) | |
| Access to off-farm employment (Yes = 1) | −0.065 ** | −0.051 ** | −0.029 | 0.017 | −0.102 *** |
| (0.029) | (0.024) | (0.024) | (0.018) | (0.036) | |
| Access to non-farm self-employment (Yes = 1) | 0.051 * | 0.027 | 0.044 ** | 0.039 ** | 0.075 ** |
| (0.026) | (0.021) | (0.019) | (0.017) | (0.034) | |
| Household asset holding (asset score) | −0.000 | −0.000 | −0.000 | −0.000 | −0.000 |
| (0.000) | (0.000) | (0.000) | (0.000) | (0.000) | |
| Household resides in Ilakala village | −0.013 | 0.004 | 0.010 | −0.012 | −0.018 |
| (0.028) | (0.024) | (0.024) | (0.022) | (0.033) | |
| Household resides in Nyali village | 0.007 | 0.064 *** | 0.049 ** | 0.040 * | 0.036 |
| (0.030) | (0.025) | (0.025) | (0.023) | (0.037) | |
| Constant | 1.970 *** | 1.983 *** | 1.951 *** | 2.010 *** | 2.335 *** |
| (0.077) | (0.072) | (0.069) | (0.063) | (0.099) | |
| Observations | 450 | 450 | 450 | 450 | 450 |
| Wald chi2 | 119.35 | 56.28 | 45.14 | 48.03 | 119.33 |
| Probability > chi2 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Pseudo R2 | 0.021 | 0.009 | 0.006 | 0.005 | 0.039 |
| HDDS | HDDS (Planting) | HDDS (Pre-Harvest) | HDDS (Post-Harvest) | FVS | |
|---|---|---|---|---|---|
| Number of food groups produced | 0.009 | 0.014 | 0.006 | 0.004 | 0.015 |
| (0.009) | (0.010) | (0.010) | (0.009) | (0.011) | |
| Age of HH head (years) | −0.002 ** | −0.001 | −0.001 | −0.000 | −0.002 ** |
| (0.001) | (0.001) | (0.001) | (0.001) | (0.001) | |
| Gender of HH head (Male = 1) | 0.012 | 0.021 | 0.003 | −0.028 | 0.013 |
| (0.035) | (0.036) | (0.035) | (0.031) | (0.040) | |
| Education of HH head (School years) | 0.003 | 0.003 | 0.006 | 0.005 | 0.001 |
| (0.004) | (0.004) | (0.004) | (0.004) | (0.005) | |
| Land size owned (ha.) | 0.009 | 0.018 ** | 0.013 * | 0.016 *** | 0.008 |
| (0.008) | (0.007) | (0.007) | (0.006) | (0.010) | |
| Livestock owned (TLU) | −0.002 * | 0.002 ** | 0.001 * | 0.001 ** | −0.003 |
| (0.001) | (0.001) | (0.001) | (0.001) | (0.003) | |
| Labor (Worker equivalents) | 0.000 | −0.007 | 0.001 | 0.001 | 0.012 |
| (0.010) | (0.011) | (0.009) | (0.008) | (0.011) | |
| Per capita per month: Low-middle | 0.084 * | −0.035 | −0.003 | −0.003 | 0.091 * |
| (0.046) | (0.045) | (0.046) | (0.040) | (0.053) | |
| Per capita per month: Middle | 0.120 *** | 0.037 | 0.088 ** | 0.056 | 0.135 *** |
| (0.044) | (0.041) | (0.041) | (0.036) | (0.050) | |
| Per capita per month: High-middle | 0.147 *** | −0.024 | 0.030 | 0.015 | 0.167 *** |
| (0.045) | (0.043) | (0.040) | (0.037) | (0.050) | |
| Per capita per month: High | 0.172 *** | 0.049 | 0.064 * | 0.040 | 0.192 *** |
| (0.040) | (0.040) | (0.038) | (0.034) | (0.048) | |
| Share of home consumption | −0.025 | −0.071 ** | −0.053 | −0.065 ** | −0.053 |
| (0.034) | (0.036) | (0.035) | (0.031) | (0.039) | |
| Access to market information (Yes = 1) | 0.047 | 0.048 | 0.034 | 0.050 * | 0.074 ** |
| (0.030) | (0.031) | (0.030) | (0.028) | (0.034) | |
| Distance to nearest paved road | −0.029 *** | −0.021 ** | −0.014 * | −0.020 *** | −0.043 *** |
| (0.009) | (0.008) | (0.008) | (0.007) | (0.010) | |
| Access to off-farm employment (Yes = 1) | −0.082 ** | −0.082 ** | −0.038 | 0.014 | −0.102 *** |
| (0.034) | (0.032) | (0.032) | (0.026) | (0.039) | |
| Access to non-farm self-employment (Yes = 1) | 0.041 | 0.038 | 0.061 ** | 0.067 *** | 0.072 * |
| (0.029) | (0.030) | (0.029) | (0.026) | (0.040) | |
| Household asset holding (asset score) | 0.000 | 0.000 | 0.000 | −0.000 | −0.000 |
| (0.000) | (0.000) | (0.000) | (0.000) | (0.000) | |
| Household resides in Ilakala village | −0.035 | −0.007 | 0.004 | −0.026 | −0.018 |
| (0.031) | (0.033) | (0.032) | (0.029) | (0.037) | |
| Household resides in Nyali village | −0.041 | 0.079 ** | 0.063 * | 0.047 | 0.040 |
| (0.034) | (0.034) | (0.035) | (0.032) | (0.041) | |
| Constant | 1.638 *** | 1.793 *** | 1.757 *** | 1.826 *** | 2.321 *** |
| (0.084) | (0.093) | (0.090) | (0.082) | (0.096) | |
| Observations | 450 | 450 | 450 | 450 | 450 |
| Wald chi2 | 104.02 | 59.67 | 48.72 | 56.58 | 93.54 |
| Probability > chi2 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Pseudo R2 | 0.019 | 0.013 | 0.009 | 0.009 | 0.040 |
References
- Herrero, M.; Thornton, P.K.; Notenbaert, A.M.; Wood, S.; Msangi, S.; Freeman, H.A.; Bossio, D.; Dixon, J.; Peters, M.; van de Steeg, J.; et al. Smart investments in sustainable food production: Revisiting mixed crop-livestock systems. Science 2010, 327, 822–825. [Google Scholar] [CrossRef] [PubMed]
- International Fund for Agricultural Development (IFAD); United Nations Environment Program (UNEP). Smallholders, Food Security and the Environment; IFAD (International Fund for Agricultural Development): Rome, Italy; Nairobi, Kenya, 2013; Available online: https://www.ifad.org/documents/10180/666cac24-14b6-43c2-876d-9c2d1f01d5dd (accessed on 25 January 2018).
- Jones, A.D.; Shrinivas, A.; Bezner-Kerr, R. Farm production diversity is associated with greater household dietary diversity in Malawi: Findings from nationally representative data. Food Policy 2014, 46, 1–12. [Google Scholar] [CrossRef]
- Godfray, H.C.J.; Beddington, J.R.; Crute, I.R.; Haddad, L.; Lawrence, D.; Muir, J.F.; Pretty, J.; Robinson, S.; Thomas, S.M.; Toulmin, C. Food security: The challenge of feeding 9 billion people. Science 2010, 327, 812–818. [Google Scholar] [CrossRef] [PubMed]
- Dorward, A. The short- and medium- term impacts of rises in staple food prices. Food Secur. 2012, 4, 633–645. [Google Scholar] [CrossRef]
- International Food Policy Research Institute (IFPRI). Actions and Accountability to Accelerate the World’s Progress on Nutrition. Global Nutrition Report; International Food Policy Research Institute: Washington, DC, USA, 2014; Available online: http://ebrary.ifpri.org/cdm/ref/collection/p15738coll2/id/128484 (accessed on 13 December 2017).
- Burlingame, B.; Dernini, S. Sustainable diets and biodiversity: Directions and solutions for policy, research and action. In Proceedings of the International Scientific Symposium on Biodiversity and Sustainable Diets: United Against Hunger, Rome, Italy, 3–5 November 2010; Food and Agriculture Organization: Rome, Italy, 2012. [Google Scholar]
- Pellegrini, L.; Tasciotti, L. Crop diversification, dietary diversity and agricultural income: Empirical evidence from eight developing countries. Can. J. Dev. Stud. 2014, 35, 211–227. [Google Scholar] [CrossRef]
- Sibhatu, K.T.; Krishna, V.V.; Qaim, M. Production diversity and dietary diversity in smallholder farm households. Proc. Natl. Acad. Sci. USA 2015, 112, 10657–10662. [Google Scholar] [CrossRef] [PubMed]
- Powell, B.; Thilsted, S.H.; Ickowitz, A.; Termote, C.; Sunderland, T.; Herforth, A. Improving diets with wild and cultivated biodiversity from across the landscape. Food Secur. 2015, 7, 535–554. [Google Scholar] [CrossRef]
- KC, K.; Pant, L.; Fraser, E.; Shrestha, P.; Shrestha, D.; Lama, A. Assessing links between crop diversity and food self-sufficiency in three agro-ecological regional of Nepal. Reg. Environ. Chang. 2015, 16, 1239–1251. [Google Scholar] [CrossRef]
- Fanzo, J.; Hunter, D.; Borelli, T.; Mattei, F. Diversifying Food and Diets: Using Agricultural Diversity to Improve Nutrition and Health; Routledge: London, UK, 2013; pp. 1–13. ISBN 978-1-84971-457-0. [Google Scholar]
- Remans, R.; Flynn, D.; DeClerck, F.; Diru, W.; Fanzo, J.; Gaynor, K.; Lambrecht, I.; Mudiope, J.; Mutuo, P.K.; Nkhoma, P.; et al. Assessing Nutritional Diversity of Cropping Systems in African Villages. PLoS ONE 2011, 6, e21235. [Google Scholar] [CrossRef] [PubMed]
- Kumar, N.; Harris, J.; Rawat, R. If They Grow It, Will They Eat and Grow? Evidence from Zambia on Agricultural Diversity and Child Undernutrition. J. Dev. Stud. 2015, 51, 1060–1077. [Google Scholar] [CrossRef]
- Ruel, M.T. Operationalizing dietary diversity: A review of measurement issues and research priorities. J. Nutr. 2003, 133, 3911S–3926S. [Google Scholar] [CrossRef] [PubMed]
- Vaitla, B.; Devereux, S.; Swan, S.H. Seasonal hunger: A neglected problem with proven solutions. PLoS Med. 2009, 6, e1000101. [Google Scholar] [CrossRef] [PubMed]
- Bacon, C.M.; Sundstrom, W.A.; Gómez, M.E.; Méndez, E.V.; Santos, R.; Goldoftas, B.; Dougherty, I. Explaining the ‘hungry farmer paradox’: Smallholders and fair trade cooperatives navigate seasonality and change in Nicaragua’s corn and coffee markets. Glob. Environ. Chang. 2014, 25, 133–149. [Google Scholar] [CrossRef]
- Herforth, A. Promotion of Traditional African Vegetables in Kenya and Tanzania: A Case Study of an Intervention Representing Emerging Imperatives in Global Nutrition. Ph.D. Thesis, Cornell University, Ithaca, NY, USA, 2010. [Google Scholar]
- Tittonell, P.; Muriuki, A.W.; Shepherd, K.D.; Mugendi, D.; Kaizzi, K.C.; Okeyo, J.; Verchot, L.; Coe, R.; Vanlauwe, B. The diversity of rural livelihoods and their influence on soil fertility in agricultural systems of East Africa—A typology of smallholder farms. Agric. Syst. 2010, 103, 83–97. [Google Scholar] [CrossRef]
- Barrett, C.; Reardon, R.; Webb, P. Non-farm income diversification and household livelihood strategies in rural Africa: Concepts, dynamics, and policy implications. Food Policy 2001, 26, 315–331. [Google Scholar] [CrossRef]
- Benin, S.; Smale, M.; Pender, J.; Gebremedhin, B.; Ehui, S. The economic determinants of cereal crop diversity on farms in the Ethiopian highlands. Agric. Econ. 2004, 31, 197–208. [Google Scholar] [CrossRef]
- Di Falco, S.; Bezabih, M.; Yesuf, M. Seeds for livelihood: Crop biodiversity and food production in Ethiopia. Ecol. Econ. 2010, 69, 1695–1702. [Google Scholar] [CrossRef]
- Benin, S.; Smale, M.; Pender, J. Explaining the diversity of cereal crops and varieties grown on household farms in the highlands of northern Ethiopia. In Valuing Crop Biodiversity: On-Farm Genetic Resources and Economic Change; Smale, M., Ed.; CABI Publishing: Wallingford, UK, 2005; pp. 78–96. ISBN 9780851990835. [Google Scholar]
- Radchenko, N.; Corral, P. Agricultural Commercialization and Food Security in Rural Economies: Malawian Experience. J. Dev. Stud. 2018, 54, 256–270. [Google Scholar] [CrossRef]
- Hazell, P. Managing drought risks in the low-rainfall areas of the Middle East and North Africa. In Case Studies in Food Policy for Developing Countries; Pinstrup-Andersen, P., Cheng, F., Eds.; Cornell University Press: Ithaca, NY, USA, 2007; p. 10. [Google Scholar]
- Hunter, D.; Fanzo, J. Agricultural biodiversity, diverse diets and improving nutrition. In Diversifying Food and Diets: Using Agricultural Biodiversity to Improve Nutrition and Health; Issues in Agricultural Biodiversity; Fanzo, J., Hunter, D., Borelli, T., Mattei, F., Eds.; Earthscan: London, UK, 2013; pp. 1–13. [Google Scholar]
- Oyarzun, P.J.; Borja, R.M.; Sherwood, S.; Parra, V. Making sense of agro-biodiversity, diet, and intensification of smallholder family farming in the highland Andes of Ecuador Ecol. Food Nutr. 2013, 52, 515–541. [Google Scholar] [CrossRef]
- Ng’endo, M.; Bhagwat, S.; Keding, G.B. Influence of Seasonal On-Farm Diversity on Dietary Diversity: A Case Study of Smallholder Farming Households in Western Kenya. Ecol. Food Nutr. 2016, 55, 403–427. [Google Scholar] [CrossRef] [PubMed]
- National Bureau of Statistics (NBS). Environment Statistics 2015; United Republic of Tanzania: Dar es Salaam, Tanzania, 2015. [Google Scholar]
- United States Agency for International Development (USAID). Preliminary Rural Livelihood Zoning: Tanzania, A Special Report by the Famine Early Warning System Network (FEWS NET). Dar es Salaam. 2008. Available online: http://fews.net/sites/default/files/documents/reports/tz_zonedescriptions_en.pdf (accessed on 12 January 2018).
- United Republic of Tanzania. National Sample Census of Agriculture 2007/2008 Morogoro Region Report; United Republic of Tanzania: Dar es Salaam, Tanzania, 2012. [Google Scholar]
- National Sample Census of Agriculture 2007/2008 Dodoma Region Report; United Republic of Tanzania: Dar es Salaam, Tanzania, 2012.
- Trans-Sec Research Project, Innovating Strategies to Safeguard Food Security Using Technology and Knowledge Transfer: A People-Centred Approach. 2016. Available online: http://www.trans-sec.org (accessed on 10 January 2018).
- Last, L.; Arndorfer, M.; Balázs, K.; Dennis, P.; Dyman, T.; Fjellstad, W.; Friedel, J.K.; Herzog, F.; Jeanneret, P.; Lüscher, G.; et al. Indicators for the on-farm assessment of crop cultivar and livestock breed diversity: A survey-based participatory approach. Biodivers. Coserv. 2014, 23, 3051–3071. [Google Scholar] [CrossRef]
- Berti, P.R. Relationship between production diversity and dietary diversity depends on how number of foods is counted. Proc. Natl. Acad. Sci. USA 2015, 112, e5656. [Google Scholar] [CrossRef] [PubMed]
- Sibhatu, K.T.; Qaim, M. Farm production diversity and dietary quality: Linkages and measurement issues. Food Secur. 2018, 10, 47–59. [Google Scholar] [CrossRef]
- Di Falco, S.; Chavas, J.P. On crop biodiversity, risk exposure, and food security in the highlands of Ethiopia. Am. J. Agric. Econ. 2009, 91, 599–611. [Google Scholar] [CrossRef]
- Swindale, A.; Bilinsky, P. Household Dietary Diversity Score (HDDS) for Measurement of Household Food Access: Indicator Guide (v.2); Food and Nutrition Technical Assistance (FANTA) Project; Academy for Educational Development: Washington, DC, USA, 2006; Available online: https://www.fantaproject.org/sites/default/files/resources/HDDS_v2_Sep06_0.pdf (accessed on 13 January 2018).
- Food and Agriculture Organization (FAO). Guidelines for Measuring Household and Individual Dietary Diversity; Food and Agriculture Organization of the United Nations: Rome, Italy, 2012; Available online: http://www.fao.org/docrep/014/i1983e/i1983e00.htm (accessed on 16 June 2018).
- Hatley, A.; Torheim, L.E.; Oshaug, A. Food variety—A good indicator of nutritional adequacy of the diet? A case study from an urban area in Mali, West Africa. Eur. J. Clin. Nutr. 1998, 52, 891–898. [Google Scholar] [CrossRef]
- Greene, W. Functional forms for the negative binomial model for count data. Econ. Lett. 2008, 99, 585–590. [Google Scholar] [CrossRef]
- Mnenwa, R.; Maliti, E. A Comparative Analysis of Poverty Incidence in Farming Systems of Tanzania. Special paper 10/4. REPOA. Dar-es-Salaam. 2010. Available online: http://www.repoa.or.tz/documents_storage/publications/10-4%20web.pdf (accessed on 20 January 2018).
- Porter, C. Shocks, consumption and income diversification in rural ethiopia. J. Dev. Stud. 2012, 48, 1209–1222. [Google Scholar] [CrossRef]

| Morogoro (Kilosa District—Rural) | Dodoma (Chamwino District—Rural) | |
|---|---|---|
| Agro-ecology | Semi-humid (Rainfall 600–800 mm) | Semi-arid (Rainfall 350–500 mm) |
| Agricultural potential | Relatively good | Relatively poor |
| Access to major markets | Relatively good | Relatively poor |
| Cropping system | Cereals and legumes (Maize, Rice, Peas and Sesame) Fruit and vegetables (Tomatoes, Okras, Eggplants, Onions, Cabbage, Chilies, Amaranths and Pumpkins) | Drought resistant cereals, legumes and seeds (Sorghum, Millet, Groundnuts and Sunflower) Fruit and vegetables (Tomatoes, Onions, Spinach, Grapes, Pawpaws) |
| Livestock system | Little livestock keeping (poultry, goats) | Heavy integration of livestock (Cattle, goat, poultry) |
| Variable | Kilosa District—Semi Humid with Better Market Access (n = 450) | Chamwino District—Semi Arid with Poor Market Access (n = 449) | Pooled Sample |
|---|---|---|---|
| Mean (SD) | Mean (SD) | Mean (SD) | |
| Household characteristics | |||
| Age of HH head (years) | 48.20 (17.28) | 49.10 (16.94) | 48.65 (17.11) |
| Gender of HH head (Male = 1) | 0.81 (0.39) | 0.77 (0.42) | 0.79 (0.41) |
| Education of HH head (School years) | 4.89 (3.30) | 3.96 (3.48) | 4.42 (3.42) |
| Labor (Worker equivalents) | 2.84 (1.43) | 3.19 (1.49) | 3.01 (1.47) |
| Access to off-farm employment (Yes = 1) | 0.20 (0.40) | 0.47 (0.50) | 0.33 (0.47) |
| Access to non-farm self-employment (Yes = 1) | 0.16 (0.37) | 0.35 (0.48) | 0.25 (0.44) |
| Non-food expenditure (Per capita/month-PPP $) | 34.11 (34.97) | 23.49 (20.31) | 28.81 (29.07) |
| Food expenditure (Per capita/ month PPP $) | 13.65 (19.18) | 9.94 (11.33) | 11.81 (15.86) |
| Share of home consumption from total output | 0.45 (0.38) | 0.68 (0.42) | 0.57 (0.42) |
| Distance to nearest paved road (Km) | 1.94 (1.16) | 10.18 (2.74) | 6.15 (4.72) |
| Farm characteristics | |||
| Land size owned (ha) | 1.47 (1.56) | 1.95 (1.91) | 1.71 (1.76) |
| Number of plots cultivated | 2.2 (0.7) | 3.2 (1.3) | 2.6 (1.11) |
| Livestock owned (Tropical Livestock Unit) | 0.53 (6.06) | 1.26 (2.70) | 0.90 (4.71) |
| Number of crops cultivated | 2.66 (1.28) | 4.47 (1.80) | 3.56 (1.81) |
| Food Consumption Diversity Indicator | Kilosa | Chamwino | ||||||
|---|---|---|---|---|---|---|---|---|
| Low Production Diversity (n = 133) | High Production Diversity (n = 317) | Low Production Diversity (n = 213) | High Production Diversity (236) | |||||
| Mean | SD | Mean | SD | Mean | SD | Mean | SD | |
| HDDS | 7.32 | 1.94 | 7.32 | 1.78 | 5.15 | 1.79 | 6.25 *** | 1.73 |
| HDDS (Planting) | 7.41 | 1.66 | 7.71 *** | 1.41 | 5.59 | 1.97 | 6.54 *** | 1.79 |
| HDDS (Pre-harvest) | 7.53 | 1.63 | 7.82 *** | 1.41 | 5.71 | 2.01 | 6.57 *** | 1.66 |
| HDDS (post-harvest) | 7.82 | 1.44 | 7.95 ** | 1.29 | 6.77 | 1.76 | 7.38 *** | 1.53 |
| Food Variety Score (FVS) | 10.81 | 3.45 | 11.00 | 3.36 | 7.80 | 3.61 | 10.14 *** | 3.68 |
| Variable | (1) Kilosa | (2) Chamwino | (3) Pooled | |||
|---|---|---|---|---|---|---|
| Crop-Livestock Count | Number of Food Groups Produced | Crop-Livestock Count | Number of Food Groups Produced | Crop-Livestock Count | Number of Food Groups Produced | |
| Age of HH head (years) | 0.003 * | 0.002 | 0.002 * | 0.001 | 0.002 *** | 0.002 ** |
| (0.001) | (0.001) | (0.001) | (0.001) | (0.001) | (0.001) | |
| Gender of HH head (Male = 1) | 0.057 | 0.087 | 0.088 * | 0.037 | 0.087 ** | 0.066 * |
| (0.062) | (0.064) | (0.047) | (0.040) | (0.038) | (0.035) | |
| Education of HH head (School years) | 0.012 * | 0.008 | 0.003 | 0.005 | 0.006 | 0.007 |
| (0.007) | (0.007) | (0.006) | (0.005) | (0.004) | (0.004) | |
| Risk attitude (scale: 1–10) | 0.001 | −0.004 | 0.019 *** | 0.010 ** | 0.015 *** | 0.005 |
| (0.010) | (0.009) | (0.006) | (0.005) | (0.005) | (0.004) | |
| Land size owned (ha) | 0.027 | 0.011 | 0.059 *** | 0.038 *** | 0.051 *** | 0.028 *** |
| (0.019) | (0.016) | (0.008) | (0.006) | (0.007) | (0.006) | |
| Labor (Worker equivalents) | 0.040 *** | 0.028 * | 0.038 *** | 0.033 *** | 0.038 *** | 0.030 *** |
| (0.013) | (0.014) | (0.011) | (0.010) | (0.009) | (0.008) | |
| Access to off-farm employment (Yes = 1) | −0.085 | −0.043 | 0.045 | 0.042 | 0.004 | 0.005 |
| (0.056) | (0.059) | (0.037) | (0.031) | (0.030) | (0.028) | |
| Access to non-farm self-employment (Yes = 1) | 0.105 * | 0.136 ** | 0.049 | 0.042 | 0.068 ** | 0.076 *** |
| (0.058) | (0.056) | (0.037) | (0.031) | (0.032) | (0.028) | |
| Distance to nearest paved road (Km) | 0.024 * | 0.032 ** | 0.000 | 0.011 | 0.012 | 0.025 *** |
| (0.014) | (0.014) | (0.013) | (0.011) | (0.010) | (0.009) | |
| Access to credit (Yes = 1) | 0.144 * | 0.132 ** | 0.165 *** | 0.109 *** | 0.150 *** | 0.103 *** |
| (0.074) | (0.061) | (0.045) | (0.041) | (0.037) | (0.033) | |
| Access to market information (Yes = 1) | 0.005 | 0.002 | 0.000 | 0.002 | 0.001 | 0.002 |
| (0.004) | (0.005) | (0.002) | (0.001) | (0.001) | (0.001) | |
| Agricultural shocks (Yes = 1) | −0.110 * | −0.177 *** | −0.047 | −0.027 | −0.067 ** | −0.072 ** |
| (0.058) | (0.068) | (0.037) | (0.032) | (0.031) | (0.029) | |
| Household asset holding (asset score) | 0.000 | −0.000 | 0.000 * | 0.000 *** | 0.000 ** | 0.000 |
| (0.000) | (0.000) | (0.000) | (0.000) | (0.000) | (0.000) | |
| Household resides in Ilolo village | 0.075 | 0.031 | 0.124 ** | 0.086 | ||
| (0.077) | (0.068) | (0.063) | (0.058) | |||
| Household resides in Ndebwe village | 0.001 | −0.009 | 0.009 | 0.005 | ||
| (0.042) | (0.037) | (0.042) | (0.037) | |||
| Household resides in Changarawe village | −0.102 * | −0.047 | −0.376 *** | 0.036 | ||
| (0.055) | (0.050) | (0.114) | (0.105) | |||
| Household resides in Ilakala village | −0.291 *** | 0.074 | ||||
| (0.110) | (0.102) | |||||
| Household resides in Nyali village | −0.150 *** | −0.176 *** | −0.403 *** | −0.073 | ||
| (0.056) | (0.056) | (0.095) | (0.089) | |||
| Constant | 0.854 *** | 0.817 *** | 1.127 *** | 0.800 *** | 1.041 *** | 0.681 *** |
| (0.142) | (0.134) | (0.166) | (0.156) | (0.130) | (0.128) | |
| Observations | 450 | 450 | 449 | 449 | 899 | 899 |
| Wald chi2 | 80.79 | 49.70 | 201.86 | 135.46 | 690.71 | 239.01 |
| Probability > chi2 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Pseudo R2 | 0.023 | 0.016 | 0.060 | 0.024 | 0.107 | 0.030 |
| HDDS | HDDS (Planting) | HDDS (Pre-Harvest) | HDDS (Post-Harvest) | FVS | |
|---|---|---|---|---|---|
| Crop-livestock count | 0.022 *** | 0.019 *** | 0.015 *** | 0.010 *** | 0.037 *** |
| (0.004) | (0.004) | (0.004) | (0.003) | (0.006) | |
| Age of HH head (years) | −0.002 *** | −0.002 *** | −0.001 ** | −0.001 *** | −0.004 *** |
| (0.001) | (0.001) | (0.001) | (0.000) | (0.001) | |
| Gender of HH head (Male = 1) | −0.003 | 0.033 | −0.012 | −0.024 | −0.001 |
| (0.024) | (0.023) | (0.022) | (0.018) | (0.029) | |
| Education of HH head (School years) | 0.004 | −0.001 | 0.003 | 0.002 | 0.004 |
| (0.003) | (0.003) | (0.003) | (0.002) | (0.004) | |
| Land size owned (ha.) | 0.006 | 0.010 ** | 0.006 | 0.006 | 0.004 |
| (0.005) | (0.004) | (0.004) | (0.003) | (0.007) | |
| Livestock owned (TLU) | −0.001 | 0.001 | 0.001 | 0.001 | −0.003 |
| (0.001) | (0.001) | (0.001) | (0.001) | (0.003) | |
| Labor (Worker equivalents) | 0.002 | −0.001 | 0.001 | 0.008 * | 0.009 |
| (0.006) | (0.006) | (0.006) | (0.004) | (0.008) | |
| Food consumption expenditure quintile | |||||
| Per capita per month: Low-middle | 0.044 | 0.010 | −0.008 | −0.005 | 0.062 * |
| (0.029) | (0.030) | (0.029) | (0.022) | (0.037) | |
| Per capita per month: Middle | 0.063 ** | 0.094 *** | 0.072 *** | 0.038 * | 0.088 ** |
| (0.029) | (0.028) | (0.026) | (0.021) | (0.036) | |
| Per capita per month: High-middle | 0.118 *** | 0.098 *** | 0.075 *** | 0.046 ** | 0.158 *** |
| (0.029) | (0.028) | (0.026) | (0.021) | (0.036) | |
| Per capita per month: High | 0.142 *** | 0.126 *** | 0.095 *** | 0.054 *** | 0.179 *** |
| (0.028) | (0.027) | (0.026) | (0.020) | (0.037) | |
| Share of home consumption | −0.031 | −0.018 | −0.016 | −0.015 | −0.030 |
| (0.022) | (0.021) | (0.020) | (0.017) | (0.028) | |
| Access to market information (Yes = 1) | 0.101 *** | 0.084 *** | 0.095 *** | 0.062 *** | 0.108 *** |
| (0.020) | (0.019) | (0.018) | (0.015) | (0.024) | |
| Distance to nearest paved road | −0.027 *** | −0.021 *** | −0.021 *** | −0.012 *** | −0.027 *** |
| (0.003) | (0.003) | (0.003) | (0.002) | (0.004) | |
| Access to off-farm employment (Yes = 1) | −0.036 * | −0.006 | −0.024 | −0.000 | −0.039 |
| (0.020) | (0.019) | (0.019) | (0.015) | (0.026) | |
| Access to non-farm self-employment (Yes = 1) | 0.046 ** | 0.033 * | 0.022 | 0.047 *** | 0.049 * |
| (0.020) | (0.018) | (0.018) | (0.014) | (0.026) | |
| Household asset holding (asset score) | 0.000 | 0.000 | 0.000 * | 0.000 | 0.000 * |
| (0.000) | (0.000) | (0.000) | (0.000) | (0.000) | |
| Household resides in Ilakala village | −0.018 | 0.026 | 0.032 | −0.006 | −0.037 |
| (0.026) | (0.023) | (0.023) | (0.020) | (0.036) | |
| Household resides in Nyali village | 0.010 | 0.102 *** | 0.097 *** | 0.044 ** | 0.023 |
| (0.026) | (0.022) | (0.022) | (0.018) | (0.035) | |
| Household resides in Ilolo village | −0.100 *** | −0.090 *** | −0.079 *** | −0.054 ** | −0.089 ** |
| (0.028) | (0.028) | (0.029) | (0.022) | (0.036) | |
| Household resides in Ndebwe village | 0.021 | 0.012 | 0.052 * | 0.019 | 0.043 |
| (0.034) | (0.034) | (0.032) | (0.027) | (0.039) | |
| Constant | 1.903 *** | 1.864 *** | 1.903 *** | 2.004 *** | 2.256 *** |
| (0.060) | (0.057) | (0.055) | (0.046) | (0.072) | |
| ln(alpha) | −4.945 | ||||
| (0.726) | |||||
| Alpha | 0.802 | ||||
| (0.132) | |||||
| Observations | 899 | 899 | 899 | 899 | 899 |
| Wald chi2 | 456.17 | 338.94 | 321.60 | 153.50 | 250.30 |
| Probability > chi2 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Pseudo R2 | 0.044 | 0.035 | 0.032 | 0.013 | 0.051 |
| HDDS | HDDS (Planting) | HDDS (Pre-Harvest) | HDDS (Post-Harvest) | FVS | |
|---|---|---|---|---|---|
| Number of food groups produced | 0.030 *** | 0.025 *** | 0.023 *** | 0.012 * | 0.041 *** |
| (0.007) | (0.008) | (0.008) | (0.007) | (0.009) | |
| Age of HH head (years) | −0.002 *** | −0.002 ** | −0.001 * | −0.001 * | −0.003 *** |
| (0.001) | (0.001) | (0.001) | (0.001) | (0.001) | |
| Gender of HH head (Male = 1) | 0.011 | 0.053 * | −0.002 | −0.027 | −0.002 |
| (0.026) | (0.029) | (0.027) | (0.023) | (0.030) | |
| Education of HH head (School years) | 0.003 | −0.001 | 0.005 | 0.004 | 0.004 |
| (0.003) | (0.003) | (0.003) | (0.003) | (0.004) | |
| Land size owned (ha.) | 0.010 * | 0.018 *** | 0.012 ** | 0.009 * | 0.012 * |
| (0.005) | (0.006) | (0.005) | (0.005) | (0.007) | |
| Livestock owned (TLU) | −0.001 | 0.003 * | 0.003 * | 0.003 * | −0.002 |
| (0.001) | (0.002) | (0.001) | (0.002) | (0.002) | |
| Labor (Worker equivalents) | −0.002 | −0.007 | −0.005 | 0.007 | 0.011 |
| (0.007) | (0.008) | (0.007) | (0.006) | (0.008) | |
| Food consumption expenditure quintile | |||||
| Per capita per month: Low-middle | 0.044 | −0.001 | −0.014 | 0.004 | 0.068 * |
| (0.030) | (0.036) | (0.035) | (0.028) | (0.037) | |
| Per capita per month: Middle | 0.086 *** | 0.105 *** | 0.092 *** | 0.058 ** | 0.099 *** |
| (0.031) | (0.034) | (0.033) | (0.027) | (0.037) | |
| Per capita per month: High-middle | 0.135 *** | 0.090 ** | 0.084 ** | 0.063 ** | 0.166 *** |
| (0.032) | (0.035) | (0.034) | (0.028) | (0.036) | |
| Per capita per month: High | 0.151 *** | 0.134 *** | 0.105 *** | 0.066 ** | 0.192 *** |
| (0.031) | (0.035) | (0.034) | (0.027) | (0.038) | |
| Share of home consumption | −0.024 | −0.054 ** | −0.035 | −0.042 * | −0.043 |
| (0.023) | (0.026) | (0.026) | (0.022) | (0.028) | |
| Access to market information (Yes = 1) | 0.094 *** | 0.108 *** | 0.116 *** | 0.074 *** | 0.114 *** |
| (0.021) | (0.024) | (0.023) | (0.020) | (0.024) | |
| Distance to nearest paved road | −0.027 *** | −0.020 *** | −0.022 *** | −0.013 *** | −0.023 *** |
| (0.003) | (0.003) | (0.003) | (0.003) | (0.004) | |
| Access to off-farm employment (Yes = 1) | −0.041 * | −0.016 | −0.027 | −0.009 | −0.038 |
| (0.022) | (0.024) | (0.024) | (0.020) | (0.026) | |
| Access to non-farm self-employment (Yes = 1) | 0.036 * | 0.032 | 0.020 | 0.069 *** | 0.050 * |
| (0.022) | (0.024) | (0.023) | (0.019) | (0.027) | |
| Household asset holding (asset score) | 0.000 ** | 0.000 ** | 0.000 ** | 0.000 | 0.000 * |
| (0.000) | (0.000) | (0.000) | (0.000) | (0.000) | |
| Household resides in Ilakala village | −0.040 | −0.004 | 0.013 | −0.030 | −0.042 |
| (0.029) | (0.031) | (0.031) | (0.027) | (0.036) | |
| Household resides in Nyali village | −0.027 | 0.097 *** | 0.101 *** | 0.035 | 0.020 |
| (0.029) | (0.029) | (0.028) | (0.025) | (0.035) | |
| Household resides in Ilolo village | −0.074 ** | −0.102 *** | −0.088 *** | −0.067 ** | −0.041 |
| (0.030) | (0.033) | (0.033) | (0.028) | (0.035) | |
| Household resides in Ndebwe village | 0.043 | −0.015 | 0.035 | 0.012 | 0.052 |
| (0.034) | (0.040) | (0.039) | (0.033) | (0.039) | |
| Constant | 1.570 *** | 1.642 *** | 1.665 *** | 1.789 *** | 2.225 *** |
| (0.065) | (0.073) | (0.071) | (0.060) | (0.074) | |
| ln(alpha) | −4.697 | ||||
| (0.579) | |||||
| Alpha | 0.398 | ||||
| (0.067) | |||||
| Observations | 899 | 899 | 899 | 899 | 899 |
| Wald chi2 | 411.99 | 337.48 | 311.61 | 151.31 | 231.70 |
| Probability > chi2 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Pseudo R2 | 0.035 | 0.039 | 0.037 | 0.016 | 0.047 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kissoly, L.; Faße, A.; Grote, U. Implications of Smallholder Farm Production Diversity for Household Food Consumption Diversity: Insights from Diverse Agro-Ecological and Market Access Contexts in Rural Tanzania. Horticulturae 2018, 4, 14. https://doi.org/10.3390/horticulturae4030014
Kissoly L, Faße A, Grote U. Implications of Smallholder Farm Production Diversity for Household Food Consumption Diversity: Insights from Diverse Agro-Ecological and Market Access Contexts in Rural Tanzania. Horticulturae. 2018; 4(3):14. https://doi.org/10.3390/horticulturae4030014
Chicago/Turabian StyleKissoly, Luitfred, Anja Faße, and Ulrike Grote. 2018. "Implications of Smallholder Farm Production Diversity for Household Food Consumption Diversity: Insights from Diverse Agro-Ecological and Market Access Contexts in Rural Tanzania" Horticulturae 4, no. 3: 14. https://doi.org/10.3390/horticulturae4030014
APA StyleKissoly, L., Faße, A., & Grote, U. (2018). Implications of Smallholder Farm Production Diversity for Household Food Consumption Diversity: Insights from Diverse Agro-Ecological and Market Access Contexts in Rural Tanzania. Horticulturae, 4(3), 14. https://doi.org/10.3390/horticulturae4030014





