1. Introduction
Hydroponics, or soilless cultivation, has become established as a sustainable alternative for vegetable production. However, its current development requires incorporating Industry 4.0 technologies, such as the Internet of Things (IoT) and artificial intelligence, to address the economic, social, and automation challenges inherent in these systems [
1]. This technological integration is particularly relevant in controlled-environment production systems, which aim to optimize growth parameters and identify suitable cultivation conditions. Among these variables, light is the fundamental energy source, essential for plant growth and development [
2,
3]. Artificial light sources have long been used to replace sunlight and optimize crop yields. In this regard, lighting that targets specific regions of the UV and visible spectrum is currently used in growth chambers, greenhouses, and vertical farming to produce fresher, more standardized, and healthier fruits and vegetables [
4]. Photosynthesis uses photosynthetically active radiation (PAR), which corresponds to the visible light spectrum (400–700 nm); it is estimated that the light energy absorbed in this range represents between 60% and 65% of the energy that triggers physiological responses [
5]. Consequently, lighting is one of the most critical variables in controlled environments. Its precise management—including intensity, spectral quality, wavelength, and daily light integral (DLI)—directly influences the morphology, physiology, chemical composition, and sensory properties of crops [
1,
6,
7,
8].
Regarding spectral quality, LED light sources can emit monochromatic radiation. The literature indicates that the combination of blue light (400–495 nm) and red light (620–750 nm) optimizes plant growth. Positive results have also been obtained by adding green light in the 500–600 nm spectrum to this mixture [
9]. While red light is essential for cell division, it affects chlorophyll content, promotes stem elongation, enhances root absorption, and promotes hypocotyl elongation and flowering. Studies have shown that blue light is fundamental for chloroplast development, stomatal opening, and carotenoid synthesis [
10]. The combination of blue and red light can prolong the flowering period and improve the quality of floral organs [
10]. Studies suggest that different red-to-blue ratios (e.g., 7:1 or 3:1) can significantly improve biomass and leaf morphology in vertical farming systems [
5,
11]. The light spectrum produced by combining far-red, deep-red, and blue LEDs results in higher lettuce growth rates and nutrient uptake [
12]. Furthermore, the photoperiod, which corresponds to the amount of light and darkness in a 24 h daily cycle [
13], and daily light integral (DLI) are key factors. Although photoperiod coordinates physiological development with the seasons, in controlled environments, it is adjusted to balance plant needs with energy efficiency. Photoperiods of 12 to 16 h and DLI ranges of 8.5 to 11.5 mol m
−2 day
−1 are commonly used [
8]. It is worth noting that artificial lighting can represent between 20% and 40% of total energy production costs [
14], which highlights the practical importance of identifying biologically effective lighting strategies for controlled-environment production systems.
The species under study is arugula (
Eruca sativa Mill.), a member of the Brassicaceae family. Native to Mediterranean and Western Asia, it was commercially cultivated in the 1990s. In addition to its culinary uses, it has medicinal applications due to its high content of glucosinolates and antioxidants with anticancer properties [
15,
16]. It is also notable for its vitamin C and flavonoid content, whose concentrations are directly related to the plant’s antioxidant capacity [
17,
18]. Previous research on arugula cultivation in controlled environments recommends parameters such as a 16 h photoperiod, a DLI of 14 mol m
−2 day
−1, and a photosynthetic photon flux density (PPFD) of 150–250 μmol·m
−2·s
−1 [
1,
11]. However, despite these general recommendations, there is still limited information on how lighting configurations used in vertical NFT systems influence crop performance and quality-related traits when spectral composition and lamp-to-canopy distance vary simultaneously, together with the resulting radiation environment at canopy level.
Therefore, the objective of this study was to compare four practical LED lighting configurations defined by spectral composition and lamp-to-canopy distance, and characterized by their resulting PPFD and DLI, in terms of their effects on the agronomic performance and selected quality traits of arugula (Eruca sativa Mill.) grown in a Nutrient Film Technique (NFT) system under controlled conditions. This study was not designed to isolate spectral quality as an independent factor or to quantify energy-use efficiency.
2. Materials and Methods
2.1. Experimental Site
In 2025, two experiments were conducted in a controlled-environment vertical farming unit at the Soilless Cultivation and Vertical Agriculture Laboratory, Faculty of Agricultural Sciences, University of Talca, Talca Campus, Maule Region, Chile. The unit was developed and implemented as part of an FIC-funded project to design and validate vertical agriculture modules for continuous hydroponic production of leafy vegetables under local conditions. In this context, the infrastructure provided a suitable experimental platform for evaluating lighting strategies in a vertical NFT system under controlled environmental conditions. The facility is located at an altitude of 111 m a.s.l., at 35°24′26″ S latitude and 71°38′10″ W longitude.
2.2. Plant Material
The species studied was arugula (Eruca sativa Mill.) var. Roquet. Seedlings were produced in rigid trays containing 162 cells (15 cm3 cell−1) filled with a peat–perlite substrate mixture (2:1, v/v). When plants reached 4–5 true leaves, they were transplanted into the final cultivation system using the Nutrient Film Technique (NFT). The first experiment was conducted from 17 March to 8 April 2025, and plants were harvested after 22 days of cultivation. The second experiment was conducted in April, from 23 April to 14 May 2025, with plants also harvested after 22 days of cultivation.
2.3. Plant Growing Conditions
The controlled environment was established in a repurposed shipping container, conditioned and insulated to support plant cultivation. The container measured 12 m in length, 2.5 m in width, and 2.2 m high. It was equipped with a CO2 injection system consisting of a cylinder connected to a sensor-controlled regulator that periodically released CO2 into the environment, maintaining concentrations between 500 and 1000 ppm. In addition, the container was fitted with two air-conditioning units to maintain the temperature within the optimal range of the species. Air circulation was provided by two fans located at opposite ends of the container, programmed to recirculate the air continuously within the system. The fans were activated every 15 min and operated for 5 min. The start time and daily duration of CO2 enrichment, as well as quantitative ventilation and air exchange parameters, were not recorded during the experiments and therefore could not be specified.
2.4. Cultivation System
Plants were cultivated inside the container using a vertical Nutrient Film Technique (NFT) system [
19]. The system consisted of paired shelving units (60 cm deep, 90 cm wide, and 2 m high), each with four vertical levels. Each level contained four rectangular growing channels (110 cm long, 9 cm wide, and 5 cm high) connected to a 20 L reservoir containing the nutrient solution, which was continuously recirculated using a pump. This configuration allowed each growing rack to operate as four independent NFT systems. In addition, each level of the four shelving pairs was equipped with an LED lighting system (Shenzhen, China) to enable the different light treatments applied in the experiment, as already described (
Table 1). Each NFT unit (i.e., each shelf level) accommodated 22 plants, resulting in a total of 88 plants per growing rack.
2.5. Environmental Conditions and Nutrient Solution Management
Environmental conditions and nutrient solution parameters were monitored throughout the cultivation period. During the first experiment, the air temperature inside the container averaged 13.7 °C to 20.1 °C. In the second experiment, average air temperatures ranged between 17.6 °C (minimum) and 20.4 °C (maximum) (
Figure 1a). In both experiments, lower temperatures were generally recorded during the dark period, whereas higher temperatures occurred during the light period. Relative humidity (RH) averaged during the first experiment ranged from 60.9% (minimum) to 77.1% (maximum), whereas in the second experiment it ranged from 53.2% (minimum) to 86.6% (maximum) (
Figure 1b).
The nutrient solution and plant water consumption were also monitored during the experiments. Reservoir volumes were periodically replenished to their initial levels, allowing estimation of plant water consumption. The nutrient solution formulation followed that described by Cooper [
20], with electrical conductivity maintained between 1.5 and 1.6 dS m
−1. The pH was adjusted with 5% phosphoric acid and maintained between 5.8 and 6.0.
2.6. Evaluation and Characterization of the Light Spectrum
To establish the lighting treatments described in
Table 1, commercially available LED sources were used as artificial lighting inputs within the vertical cultivation system. The white-light treatment was provided by 18 W Megabright LED lamps (Shenzhen, China), whereas the red–blue lighting treatment was generated using commercially available red and blue LED strips with nominal powers of 4.8 W for red light and 14 W for blue light, supplemented with a white LED lamp (
Table 1 and
Table 2). These lighting sources were selected because they represented commercially available LED sources commonly used in controlled-environment horticulture consistent with the technological approach of the production unit used in this study.
The LED sources were installed above the growing channels and arranged according to the spectral composition and height defined for each treatment. In this way, the experimental setup combined variations in light quality and vertical distance from the crop canopy, enabling evaluation of how these factors modified the radiation environment experienced by the plants. The spatial arrangement of the lamps was carefully adjusted to ensure that each treatment produced a distinct lighting condition in terms of spectral distribution and irradiance at the crop level.
To characterize the spectral emission of each light source, spectral distribution was measured using a spectrometer (SS-110, Apogee Instruments, Logan, UT, USA). These measurements allowed identification of the relative contribution of each wavelength region within the emitted spectrum. The spectrometer was used according to the manufacturer’s specifications. In addition to spectral characterization, photosynthetic photon flux density (PPFD) was measured at canopy level using a quantum sensor (MQ-200, Apogee Instruments, Logan, UT, USA), corresponding to the crop light interception zone. For each treatment, PPFD measurements were taken at three representative points per shelf level and averaged. The instruments were used according to the manufacturer’s calibration and operating specifications. These measurements were used to quantify photosynthetically active radiation effectively reaching the crop under each lighting treatment. Because the distance between the light source and the crop differed among treatments, these measurements were essential to characterize the actual radiation environment experienced by the plants. Thus, the combined evaluation of spectral distribution and PPFD provided a more complete characterization of the light environment to which arugula plants were exposed. Spectral characterization was performed at the emitter level and not at the canopy level for each treatment.
The spectral profiles and PPFD values obtained for each treatment are presented in
Figure 2 and
Figure 3. Together, these measurements supported treatment differentiation and provided context for interpreting plant responses under the evaluated lighting conditions.
2.7. Lighting Treatments
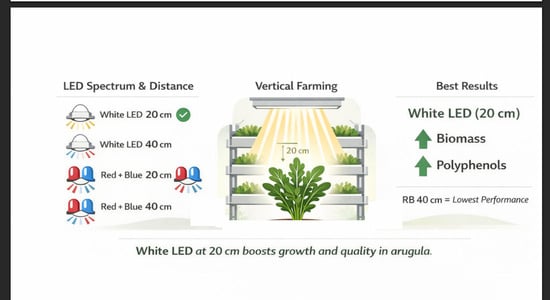
Four lighting treatments were established by combining two spectral conditions with two vertical distances between the light source and the plant canopy. The spectral treatments consisted of white LED light alone and a red–blue LED combination in a 3:1 ratio, supplemented with white LED light. Each of these spectral conditions was evaluated at two distances from the crop canopy, 20 cm and 40 cm, generating four treatment combinations in total.
All treatments were applied under a fixed photoperiod of 16 h light per day during the cultivation period. At the crop level, light intensity ranged from 193.46 to 257.90 μmol m
−2 s
−1, corresponding to a daily light integral (DLI) of 11.14 to 14.85 mol m
−2 d
−1 (
Table 1). These light levels were selected to provide a suitable radiation environment for leafy vegetable growth under controlled environment conditions while preserving the contrast among spectral and positional treatments.
The treatment structure allowed the simultaneous evaluation of two relevant lighting factors in vertical cultivation systems: spectral composition and the distance between the lighting source and the crop canopy. While spectral quality may influence photosynthetic activity, morphology, and the accumulation of functional compounds, the positioning of the light source may modify the plants’ effective light interception. Thus, the experimental design was intended to assess the combined effect of light quality and lamp distance on the agronomic and quality responses of arugula grown under a vertical NFT production system. Therefore, the experimental design does not allow a direct comparison of spectral quality independently of lamp-to-canopy distance, and the observed responses should be interpreted as the result of combined lighting configurations rather than isolated spectral effects.
2.8. Measurements of Plant Growth Parameters
Plant growth was evaluated throughout the cultivation period by tracking leaf development and recording morphological and biomass variables at harvest (22 days after transplanting). The number of leaves per plant was monitored periodically as an indicator of vegetative growth. At harvest, plants within each experimental unit were measured individually, and the values were averaged at the shelf level prior to statistical analysis.
The growth variables measured in each trial included total plant fresh weight (g plant−1), leaf fresh weight (g plant−1), root fresh weight (g plant−1), total number of leaves per plant, leaf length (cm), and leaf width (cm). These variables were used to assess the effects of lighting treatments on plant growth, biomass partitioning, and leaf expansion, which are key indicators of performance in leafy vegetables grown under controlled conditions.
2.9. Determination of Total Polyphenols, Antioxidant Capacity, and Moisture
At harvest, samples from each replicate were collected and sent to the Centro de Estudios de Alimentos Procesados (CEAP), Chemistry Laboratory (Chile) for quality analysis. Total polyphenol content was determined using the Folin–Ciocalteu method, following Singleton et al. [
21], and expressed as mg gallic acid equivalents (GAE) per 100 g fresh weight. Briefly, 2 g of fresh tissue were mixed with 10 mL of extraction solvent, and absorbance was measured at 762 nm and 37 °C using a multimode microplate reader (Synergy HTX, BioTek Instruments, Winooski, VT, USA).
Antioxidant capacity was determined by the oxygen radical absorbance capacity (ORAC) method and expressed as μmol Trolox equivalents (TE) 100 g
−1 fresh weight (FW), following AOAC method 2012.23 [
22]. For this analysis, 0.2 g of fresh tissue was extracted with 20 mL of solvent, and fluorescence readings were obtained in microplates using fluorescein in the same multimode reader.
Moisture content was determined by gravimetric analysis in a conventional oven at 105 °C and expressed as a percentage of fresh weight (%
w/
w) [
23].
2.10. Relative Chlorophyll Index
Leaf chlorophyll status was estimated using a relative chlorophyll index. Measurements were taken on five plants per replicate. For each plant, the first fully expanded leaf was selected, and readings were obtained from the central portion of the lamina using a portable chlorophyll meter (GYJ-B model, Hangzhou, China). Results were expressed in SPAD units. This parameter was used as a non-destructive indicator of leaf greenness and potential variation in photosynthetic status under different lighting treatments. To minimize variability, both leaf developmental stage and measurement position were standardized.
2.11. Determination of Nitrate
Nitrate concentration was determined in leaf tissue at harvest. For each sample, 2 g of arugula leaves were weighed, finely chopped, and homogenized with 2 mL of distilled water to obtain a plant extract. An aliquot of 1 mL of the extract was then analyzed using a portable nitrate meter (Horiba LAQUA Twin, Kioto, Japan). Nitrate content was expressed as mg NO3− kg−1 FW. This measurement was included because nitrate accumulation is an important quality attribute in leafy vegetables and can be affected by environmental conditions, particularly light. Therefore, determining nitrate levels provided additional information on the effects of the evaluated treatments on product quality at harvest.
2.12. Experimental Design and Statistical Analysis
The experiment was established under a randomized block design, in which each block corresponded to a shelf within the vertical cultivation system. Four replicates were assigned to each treatment, and each shelf level was considered an experimental unit. Plant-level measurements obtained within each shelf level were averaged, and these experimental-unit means were used for the analysis of variance (ANOVA). This design was adopted to account for potential positional variation within the cultivation structure while maintaining adequate comparability among lighting treatments.
The data were analyzed by analysis of variance (ANOVA) using Statgraphics 19-X64 software. When significant treatment effects were detected, mean comparisons were performed using Tukey’s honestly significant difference (HSD) test at p ≤ 0.05. Pearson’s correlation coefficients were also calculated to assess relationships among the measured variables and to examine associations between growth and quality traits. Correlation analysis was performed using replicate-level data (n = 16), considering each experimental unit (shelf-level mean) as an independent observation across all treatments.
3. Results and Discussion
3.1. Fresh Biomass Production
In the first experiment, plants achieved greater fresh biomass under the LW20 treatment, averaging 42.6 g plant
−1, which was significantly higher than that obtained under the other lighting treatments. In contrast, RB20 and RB40 were associated with the lowest fresh biomass values (
Figure 4a). However, because these treatments also differed in PPFD, DLI, and lamp-to-canopy distance, this response cannot be attributed exclusively to spectral composition. Similar trends were observed for leaf and root biomass in both experiments (
Figure 4b).
The higher biomass observed under the LW20 configuration may be associated with the overall radiation environment of this treatment, including spectral distribution, photosynthetic photon flux density (PPFD), and daily light integral (DLI). However, these results should be interpreted cautiously, as the experimental design does not allow for the isolation of spectral effects from lamp-to-canopy distance or associated differences in light intensity. Therefore, the observed responses reflect the combined influence of multiple lighting factors rather than the effect of spectral composition alone.
Previous studies have reported that arugula cultivated under an intensity of 350 μmol m
−2 s
−1 with a 12 h photoperiod reached an average fresh weight of 30.06 g [
2], which is lower than the values obtained in the present study (42.6 and 70.9 g plant
−1) under the LW20 configuration, with an intensity of 258 μmol m
−2 s
−1 and a 16 h photoperiod. This is consistent with previous findings indicating that higher light intensity and DLI are associated with increased plant fresh weight [
2].
Light quality has been reported to influence the efficiency with which plants convert light energy into chemical energy [
24]. However, in the present study, the evaluated treatments differed simultaneously in spectral composition, lamp-to-canopy distance, and the resulting radiation environment. Under these conditions, RB20 and RB40 were associated with lower fresh biomass production per plant, as well as reduced shoot and root biomass, relative to the other evaluated configurations.
Recent evidence suggests that crop response to LED lighting depends on the interaction among spectral composition, light intensity, and photoperiod, rather than on spectrum alone [
2,
5,
25]. In this context, the present results provide additional evidence under vertical NFT cultivation, showing that LW20 was associated with the highest biomass under the tested conditions.
These results differ from those reported by [
5], who observed a positive effect of red-dominant LED lighting on biomass accumulation in hydroponically grown arugula, particularly under a 7:1 red-to-blue ratio. This discrepancy may be attributed to differences in the overall radiation environment and experimental design, rather than to spectral composition alone.
3.2. Number of Leaves
The number of leaves per plant ranged from 11 to 18 in the first experiment, with the highest values observed under LW treatments and the lowest under RB lighting configurations (
Figure 5). A similar pattern was observed in the second experiment, where LW20 again showed the highest leaf number (21.5), while RB treatments showed the lowest values (11.9 and 12.7). The greater leaf number observed in Experiment 2 may be related to differences in plant establishment conditions. Previous studies have reported that leaf development in arugula is influenced by light intensity and environmental conditions [
26]. However, unlike previous findings [
27], where no differences among light treatments were observed, the present study showed clear differences between LW and RB configurations.
3.3. Length and Width of Leaves
In Experiment 1, both variables showed significant differences among treatments (
Table 3). The greatest leaf length and width were obtained under the LW40 treatment (13.0 and 4.9 cm leaf
−1, respectively), whereas the smallest values were recorded under RB40 (10.9 and 3.7 cm leaf
−1, respectively). In Experiment 2 (
Table 4), no significant differences were observed, with leaf length ranging from 13.2 to 14.3 cm leaf
−1 and width from 4.7 to 5.1 cm leaf
−1.
These results indicate that leaf morphology responses are influenced by the interaction between lighting conditions and the resulting radiation environment. In Experiment 1, leaves under the RB40 treatment tended to be smaller, which may be associated with the combined lighting conditions generated by each treatment, including spectral composition, lamp-to-canopy distance, PPFD, and DLI. Previous studies have shown that leaf morphology in arugula can vary depending on the combination of light spectrum, intensity, and photoperiod. For instance, Ref. [
5] reported that red–blue LED combinations promoted favorable leaf growth responses in hydroponic arugula. However, this trend was not observed under the lighting configurations evaluated in the present study. These findings suggest that arugula responses to LED lighting are context-dependent and may vary according to the combined effects of spectral composition and other radiation-related factors.
3.4. Vegetative Development
Vegetative development was assessed by monitoring the number of leaves per plant over time (
Figure 6). Significant differences among treatments were observed in both experiments. In Experiment 1, these differences were evident throughout the evaluation period, whereas in Experiment 2 they became apparent only toward the end of the cultivation cycle. At the final evaluation (day 22), the LW20 treatment showed the highest leaf number (more than 10 leaves per plant), while RB20 exhibited the lowest values (approximately 8 leaves per plant). Overall, LW20 consistently produced the highest leaf number, whereas RB treatments generally resulted in lower values.
3.5. Chlorophyll Content
In Experiment 1, the highest relative chlorophyll index (48.9 SPAD units) was recorded under the LW40 treatment. In contrast, the red–blue LED combinations resulted in lower SPAD values (46.2 and 45.6 SPAD units). In Experiment 2, no significant differences were observed among treatments, although the lowest SPAD value was recorded under the RB20 treatment (41.8 SPAD units) (
Figure 7). Because chlorophyll status was estimated indirectly using SPAD readings, these results should be interpreted as relative differences in leaf greenness rather than as direct measurements of chlorophyll a, chlorophyll b, total chlorophyll, or the chlorophyll a/b ratio.
Adequate light intensity promotes photosynthesis in arugula leaves, resulting in greater accumulation of photosynthetic compounds [
28]. The influence of light intensity on chlorophyll content has also been reported by [
2], who evaluated arugula grown under different light conditions in a controlled environment. In that study, plants exposed to a light intensity of 200 μmol·m
−2·s
−1 with a photoperiod of 6 h exhibited significantly higher chlorophyll content than those maintained in complete darkness. Insufficient light exposure limits chlorophyll synthesis, leading to leaf yellowing and eventual senescence [
8]. Additionally, the daily light integral (DLI) has been reported to influence chlorophyll concentration [
23].
In the present study, the observed differences in relative chlorophyll index may be related to differences in light availability among treatments. Previous studies have reported variable responses depending on the spectral composition of LED lighting. For example, in hydroponically grown arugula cultivated on shelves under different LED lighting combinations [
5], red light had a significant positive effect on chlorophyll content, particularly under a red-to-blue ratio of 7:1. Similarly, [
24] reported the highest chlorophyll content under a red–green–blue combination (R7:G0:B3) across different light intensities and photoperiods. However, other studies have found no significant differences in chlorophyll content among LED treatments [
27].
In contrast, the results of the present study indicate that treatments combining red and blue LED light did not result in the highest chlorophyll index values. In Experiment 1, the highest SPAD value was observed under the LW40 treatment, whereas in Experiment 2, no significant differences among treatments were detected. These results indicate that chlorophyll responses were not consistent across experiments and may have been influenced by differences in the overall radiation environment and growing conditions. Because chlorophyll status was estimated indirectly using SPAD readings and direct pigment quantification was not performed, these results should be interpreted as relative differences in leaf greenness rather than as direct measurements of chlorophyll a, chlorophyll b, total chlorophyll, or the chlorophyll a/b ratio.
3.6. Nitrate Content
Nitrate concentration is an important quality parameter in leafy vegetables, as excessive nitrate accumulation is associated with food safety concerns and may reduce product quality due to its toxic and carcinogenic effects in humans [
29]. The concentration of this compound is regulated in some countries under legislation such as the European Commission Regulation [
30]. Arugula is recognized as one of the leafy vegetables with the highest nitrate accumulation capacity [
25], making nitrate content a relevant quality indicator in this species [
2]. In the present study, after 22 days of cultivation, nitrate content did not differ significantly among treatments, with values ranging from 3575 to 4100 mg kg
−1 FW (
Table 5). These values are comparable with [
2], who evaluated arugula under different light conditions and observed nitrate concentrations ranging from 2591.6 to 3826.18 mg kg
−1 FW during the first 20 days of cultivation. In their study, the highest nitrate levels were observed in plants grown in complete darkness, whereas the lowest concentrations occurred under a light intensity of 200 μmol m
−2 s
−1 and a photoperiod of 12 h, suggesting that adequate light conditions may favor nitrate reduction in hydroponically grown arugula.
From a physiological perspective, nitrate accumulation depends on the balance between nitrate uptake and assimilation. Under low light, photosynthetic activity declines, limiting the supply of reducing power and carbon skeletons needed for nitrate reduction and amino acid synthesis. Consequently, nitrate tends to accumulate in leaf tissues. Previous studies in arugula have shown that both light intensity and spectral composition can influence this process [
2,
24]. Researchers [
24] reported greater nitrate accumulation under lighting regimes with lower red-light contribution, whereas other authors [
25] found higher nitrate concentrations in leaves exposed to white light than under other LED treatments. Although nitrate content did not differ significantly among treatments in the present study, the values obtained remained below the maximum levels established by [
30], which sets limits of 6000 to 7000 mg kg
−1 FW for arugula depending on harvest period. Therefore, under all lighting conditions evaluated, nitrate concentration remained within the acceptable food safety range for leafy vegetables. The absence of significant treatment differences may be explained by the relatively similar light intensity across treatments, which remained close to 200 μmol m
−2 s
−1, reducing the likelihood of strong contrasts in nitrate assimilation responses. In a study conducted under different arugula production systems, nitrate concentrations ranged from 4546 to 7873 mg kg
−1 FW [
29].
3.7. Moisture, Total Polyphenols, and Antioxidant Activity
The results of the total polyphenol analysis (
Table 6) show that the highest content was obtained in the LW20 treatment, with 38.4 mg GAE 100 g
−1 FW, and the lowest in the combined RB20 treatment, with 26.2 mg GAE 100 g
−1 FW. The literature indicates that phenolic compounds in plants are among the most antioxidant-active, are involved in defense mechanisms, and are regulated by light quality [
25]. Regarding antioxidant capacity, no significant differences were observed among the different treatments, with values ranging from 1215.4 to 1849.7 μmol TE 100 g
−1 FW (
Table 6).
A previous study evaluated the antioxidant capacity of several leafy vegetable species from the Brassicaceae family used as baby leaf greens [
16]. Among these species, arugula showed high antioxidant activity and contained 132.3 mg of total polyphenols per 100 g fresh weight, with kaempferol as the main flavonoid, accounting for 78.8% of total flavonoids. According to those authors, the relationship between total polyphenol content and antioxidant capacity is generally positive; however, specific compounds such as kaempferol may show relatively low antioxidant activity despite their abundance. In addition, Ref. [
25] reported that wild arugula grown under red LED light showed higher antioxidant capacity than plants grown under white LED light. In contrast, in the present study, no significant differences in antioxidant capacity were observed among the evaluated lighting treatments.
Among the evaluated quality-related parameters, total polyphenols showed the clearest response to lighting treatments, whereas antioxidant capacity and moisture were not significantly affected.
3.8. Correlation Between Total Phenol Content and Antioxidant Capacity
A positive correlation was observed between total polyphenol content and antioxidant capacity (r = 0.64), based on replicate-level observations (n = 16). This value indicates a moderate positive association between both variables [
31]. In general, as polyphenol concentration increased, antioxidant capacity also tended to increase (
Figure 8). This trend is consistent with previous findings [
16], suggesting that phenolic compounds contribute to antioxidant activity in leafy vegetables. However, other bioactive compounds present in arugula, such as glucosinolates, may also contribute to antioxidant capacity, which may explain why the correlation was not stronger.
3.9. Water Consumption
Plant water consumption was estimated by replenishing the volume of water consumed from each reservoir. Average consumption was 0.51 L plant
−1 day
−1 during Experiment 1 and 0.61 L plant
−1 day
−1 during Experiment 2. In both experiments, the cultivation period lasted 22 days. In the second experiment, the highest water consumption was observed under the LW treatments, whereas the lowest was recorded in plants grown under the RB20 treatment (
Figure 9). This pattern was consistent with the greater fresh biomass production observed under the LW treatments and may reflect higher transpiration rates and metabolic activity associated with increased plant growth.
4. Conclusions
Under the conditions of this study, arugula performance in a vertical NFT cultivation system differed among the evaluated lighting configurations. The LW20 treatment was associated with the highest fresh biomass production and greater accumulation of total polyphenols, whereas the red–blue treatments generally resulted in lower biomass values. Among the evaluated quality-related parameters, total polyphenols showed the clearest response to lighting conditions, while antioxidant capacity, moisture, nitrate concentration, and relative chlorophyll index were not significantly affected.
These results indicate that plant responses were influenced by the overall radiation environment generated by each lighting configuration, including spectral composition, photosynthetic photon flux density (PPFD), and daily light integral (DLI). However, because these factors were not independently controlled, the observed effects should be interpreted as the result of combined lighting conditions rather than isolated spectral effects.
Overall, the LW20 configuration was the most favorable among the four evaluated lighting treatments under the tested conditions. Nevertheless, no quantitative assessment of energy consumption or resource use efficiency was conducted in this study. Future research should focus on isolating individual lighting factors and integrating biological responses with energy efficiency metrics to optimize lighting strategies in vertical farming systems.