Effects of Selenate Application on Growth, Nutrient Bioaccumulation, and Bioactive Compounds in Broccoli (Brassica oleracea var. italica L.)
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Site Conditions
2.2. Soil Characteristics and Conditioning
2.3. Experimental Design and Crop Management
2.4. Plant Material Analysis
2.5. Antioxidant Activity
2.6. Total Phenol Concentration (TPC)
2.7. Statistical Analysis
3. Results
3.1. Effect on Plant Growth Parameters
3.2. Effect on Nutritional Parameters
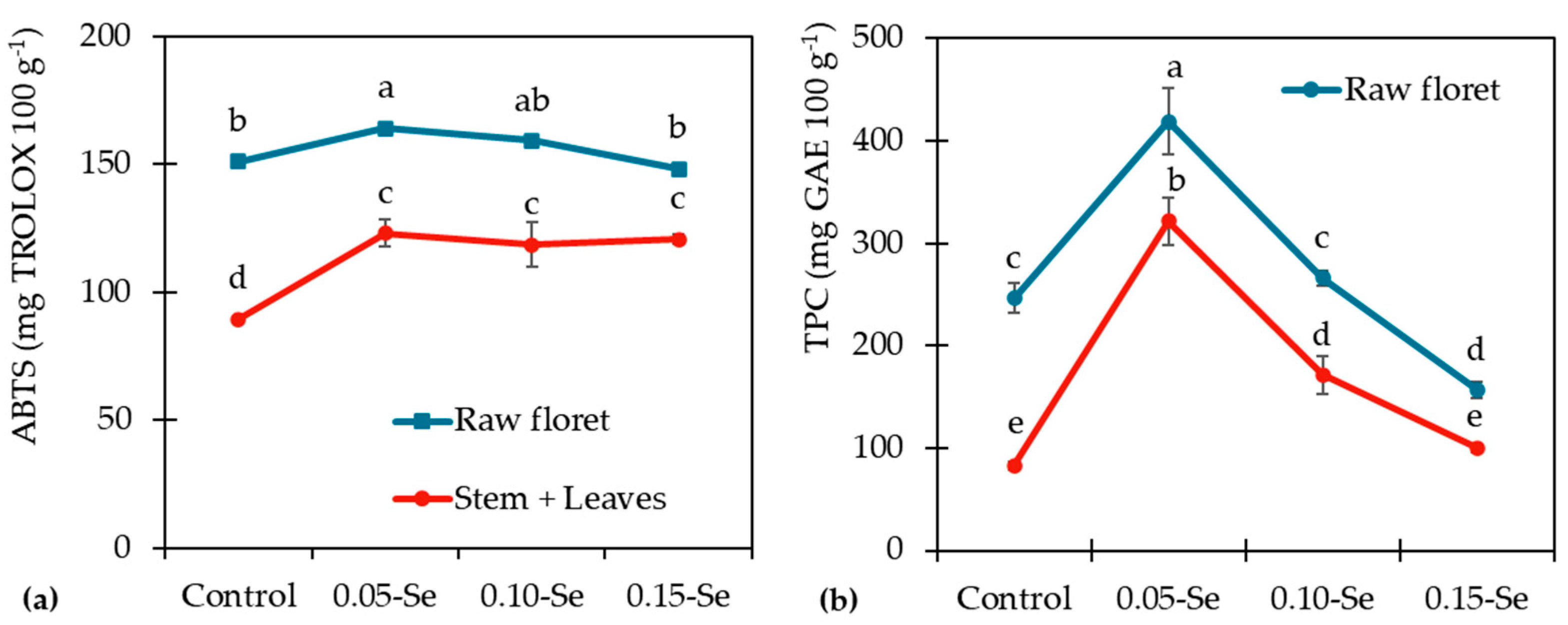
3.3. Effect on Phytic Acid, Antioxidant Activity and Polyphenol Concentration
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Roman, M.; Jitaru, P.; Barbante, C. Selenium biochemistry and its role for human health. Metallomics 2014, 6, 25–54. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Yang, J.; Kronzucker, H.J.; Shi, W. Selenium Biofortification and Interaction With Other Elements in Plants: A Review. Front. Plant Sci. 2020, 11, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Peeler, J.C.; Weerapana, E. Chemical Biology Approaches to Interrogate the Selenoproteome. Acc. Chem. Res. 2019, 52, 2832–2840. [Google Scholar] [CrossRef] [PubMed]
- Kieliszek, M.; Błazejak, S. Current knowledge on the importance of selenium in food for living organisms: A review. Molecules 2016, 21, 609. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Li, X.; Wei, Y. Selenium and Selenoproteins in Health. Biomolecules 2023, 13, 799. [Google Scholar] [CrossRef] [PubMed]
- Monsen, E.R. Dietary reference intakes for the antioxidant nutrients: Vitamin C, vitamin E, selenium, and carotenoids. J. Acad. Nutr. Diet. 2000, 100, 637. [Google Scholar]
- Radomska, D.; Czarnomysy, R.; Radomski, D.; Bielawska, A.; Bielawski, K. Selenium as a bioactive micronutrient in the human diet and its cancer chemopreventive activity. Nutrients 2021, 13, 1649. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Chen, B.; Huang, Y.; Li, J.; Cao, D.; Chen, Z.; Li, J.; Ran, B.; Yang, J.; Wang, R.; et al. Selenium intake and multiple health-related outcomes: An umbrella review of meta-analyses. Front. Nutr. 2023, 10, 1–16. [Google Scholar]
- Stoffaneller, R.; Morse, N.L. A Review of Dietary Selenium Intake and Selenium Status in Europe and the Middle East. Nutrients 2015, 7, 1494–1537. [Google Scholar] [CrossRef]
- Ligowe, I.S.; Phiri, F.P.; Ander, E.L.; Bailey, E.H.; Chilimba, A.D.C.; Gashu, D.; Joy, E.J.M.; Lark, R.M.; Kabambe, V.; Kalimbira, A.A.; et al. Selenium deficiency risks in sub-Saharan African food systems and their geospatial linkages. Proc. Nutr. Soc. 2020, 79, 457–467. [Google Scholar] [CrossRef]
- Zhang, L.; Guo, Y.; Liang, K.; Hu, Z.; Sun, X.; Fang, Y.; Mei, X.; Yin, H.; Liu, X.; Lu, B. Determination of Selenium in Common and Selenium-Rich Rice from Different Areas in China and Assessment of Their Dietary Intake. Int. J. Environ. Res. Public Health 2020, 17, 4596. [Google Scholar] [CrossRef] [PubMed]
- Olza, J.; Aranceta-Bartrina, J.; González-Gross, M.; Ortega, R.M.; Serra-Majem, L.; Varela-Moreiras, G.; Gil, Á. Reported dietary intake and food sources of zinc, selenium, and vitamins a, e and c in the spanish population: Findings from the anibes study. Nutrients 2017, 9, 697. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.; Bailey, E.H.; Arshad, M.; Ahmed, S.; Watts, M.J.; Stewart, A.G.; Young, S.D. Environmental and human iodine and selenium status: Lessons from Gilgit-Baltistan, North-East Pakistan. Environ. Geochem. Health 2021, 43, 4665–4686. [Google Scholar] [CrossRef] [PubMed]
- Belay, A.; Joy, E.J.M.; Chagumaira, C.; Zerfu, D.; Ander, E.L.; Young, S.D.; Bailey, E.H.; Lark, R.M.; Broadley, M.R.; Gashu, D. Selenium Deficiency Is Widespread and Spatially Dependent in Ethiopia. Nutrients 2020, 12, 1565. [Google Scholar] [CrossRef] [PubMed]
- Kamchen, C.M.; de Oliveira, F.L.; de Souza, T.R.; Vieira, B.S.; Telles, B.; Morzelle, M.C. Biofortification with selenium as an alternative to increase the total phenolic compounds in brassicas: A systematic review and meta-analysis. J. Sci. Food Agric. 2024, 104, 1234–1243. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.; Bañuelos, G.S.; Zhao, J.; Wang, Z.; Farooq, M.R.; Yang, Y.; Song, J.; Zhang, Z.; Chen, Y.; Yin, X.; et al. Comprehensive evaluation of factors influencing selenium fertilization biofortification. J. Sci. Food Agric. 2024, 104, 6100–6107. [Google Scholar] [CrossRef] [PubMed]
- Gao, C.; Xiao, M.; Gong, Z.; Song, L.; Wang, H.; Ludlow, R.A.; Li, W.; Li, P.; Liu, W.; Jia, C.; et al. Selenium Biofortification and Increased Nutritional Quality in Alfalfa (Medicago sativa L.) Using Foliar Application of Selenium-Rich Nutrient Solution. J. Soil Sci. Plant Nutr. 2023, 23, 3600–3611. [Google Scholar] [CrossRef]
- Đulović, A.; Usanović, K.; Kukoč Modun, L.; Blažević, I. Selenium Biofortification Effect on Glucosinolate Content of Brassica oleracea var. italic and Eruca vesicaria. Molecules 2023, 28, 7203. [Google Scholar] [CrossRef]
- Schiavon, M.; Nardi, S.; dalla Vecchia, F.; Ertani, A. Selenium biofortification in the 21st century: Status and challenges for healthy human nutrition. Plant Soil 2020, 453, 245–270. [Google Scholar] [CrossRef]
- Bouis, H.E.; Saltzman, A. Improving nutrition through biofortification: A review of evidence from HarvestPlus, 2003 through 2016. Glob. Food Sec. 2017, 12, 49–58. [Google Scholar] [CrossRef]
- Radawiec, A.; Szulc, W.; Rutkowska, B. Selenium biofortification of wheat as a strategy to improve human nutrition. Agriculture 2021, 11, 144. [Google Scholar] [CrossRef]
- Poblaciones, M.J.; Rengel, Z. The effect of processing on Pisum sativum L. biofortified with sodium selenate. J. Plant Nutr. Soil Sci. 2018, 181, 932–937. [Google Scholar] [CrossRef]
- Trod, B.S.; Buttarelli, M.S.; Stoffel, M.M.; Céccoli, G.; Olivella, L.; Barengo, P.B.; Llugany, M.; Guevara, M.G.; Muñoz, F.F.; Daurelio, L.D. Postharvest commercial quality improvement of broccoli (Brassica oleracea L.) after foliar biofortification with selenium. Crop Sci. 2022, 63, 784–800. [Google Scholar] [CrossRef]
- D’Amato, R.; Regni, L.; Falcinelli, B.; Mattioli, S.; Benincasa, P.; Dal Bosco, A.; Pacheco, P.; Proietti, P.; Troni, E.; Santi, C.; et al. Current Knowledge on Selenium Biofortification to Improve the Nutraceutical Profile of Food: A Comprehensive Review. J. Agric. Food Chem. 2020, 68, 4075–4097. [Google Scholar] [CrossRef]
- Groth, S.; Budke, C.; Neugart, S.; Ackermann, S.; Kappenstein, F.S.; Daum, D.; Rohn, S. Influence of a selenium biofortification on antioxidant properties and phenolic compounds of apples (Malus domestica). Antioxidants 2020, 9, 187. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhao, Z.; Zhang, X.; Zhang, W.; Huang, L.; Zhang, Z.; Yuan, L.; Liu, X. Effects of foliar application of selenate and selenite at different growth stages on Selenium accumulation and speciation in potato (Solanum tuberosum L.). Food Chem. 2019, 286, 550–556. [Google Scholar] [CrossRef] [PubMed]
- Meucci, A.; Shiriaev, A.; Rosellini, I.; Malorgio, F.; Pezzarossa, B. Se-Enrichment Pattern, Composition, and Aroma Profile of Ripe Tomatoes after Sodium Selenate Foliar Spraying Performed at Different Plant Developmental Stages. Plants 2021, 10, 1050. [Google Scholar] [CrossRef] [PubMed]
- Tarafder, S.K.; Biswas, M.; Sarker, U.; Ercisli, S.; Okcu, Z.; Marc, R.A.; Golokhvast, K.S. Influence of foliar spray and post-harvest treatments on head yield, shelf-life, and physicochemical qualities of broccoli. Front. Nutr. 2023, 10, 1057084. [Google Scholar] [CrossRef] [PubMed]
- Treccarichi, S.; Cali, R.; Amari, M.; Mortada, A.; Felis, M.D.; Achkar, N.; Branca, F. New tools for organic plant nutrition: Microbial and aminoacid treatments for organic seed production of broccoli (Brassica oleracea var italica Plenk.). In Proceedings of the XXXI International Horticultural Congress (IHC2022): International Symposium on Quality Seeds and Transplants for Horticultural Crops and Restorative Species, Angers, France, 14–20 August 2022; International Society for Horticultural Science (ISHS): Leuven, Belgium, 2023; pp. 81–90. [Google Scholar]
- Li, H.; Xia, Y.; Liu, H.Y.; Guo, H.; He, X.Q.; Liu, Y.; Wu, D.T.; Mai, Y.H.; Li, H.B.; Zou, L.; et al. Nutritional values, beneficial effects, and food applications of broccoli (Brassica oleracea var. italica Plenck). Trends Food Sci. Technol. 2022, 119, 288–308. [Google Scholar] [CrossRef]
- Šola, I.; Davosir, D.; Kokić, E.; Zekirovski, J. Effect of Hot- and Cold-Water Treatment on Broccoli Bioactive Compounds, Oxidative Stress Parameters and Biological Effects of Their Extracts. Plants 2023, 12, 1135. [Google Scholar] [CrossRef]
- Li, Z.; Zheng, S.; Liu, Y.; Fang, Z.; Yang, L.; Zhuang, M.; Zhang, Y.; Lv, H.; Wang, Y.; Xu, D. Characterization of glucosinolates in 80 broccoli genotypes and different organs using UHPLC-Triple-TOF-MS method. Food Chem. 2021, 334, 127519. [Google Scholar] [CrossRef] [PubMed]
- Korzekwa, K. Building a Better Broccoli. CSA News 2023, 68, 8–13. [Google Scholar] [CrossRef]
- Asare, O.E.; Bhujel, K.N.; Cizkova, H.; Rajchl, A. Fortification of fruit products—A review. Czech J. Food Sci. 2022, 40, 259–272. [Google Scholar] [CrossRef]
- Muñoz, F.F.; Stoffel, M.M.; Céccoli, G.; Trod, B.S.; Daurelio, L.D.; Bouzo, C.A.; Guevara, M.G. Improving the foliar biofortification of broccoli with selenium without commercial quality losses. Crop Sci. 2021, 61, 4218–4228. [Google Scholar] [CrossRef]
- Abdalla, Z.; El-Bassiony, A.E.-M.; El-Ramady, H.; El-Sawy, S.; Shedeed, S. Broccoli Biofortification Using Biological Nano- and Mineral Fertilizers of Selenium: A Comparative Study under Soil Nutrient Deficiency Stress. Egypt. J. Soil Sci. 2023, 63, 57–66. [Google Scholar] [CrossRef]
- Bachiega, P.; Salgado, J.M.; De Carvalho, J.E.; Ruiz, A.L.T.G.; Schwarz, K.; Tezotto, T.; Morzelle, M.C. Antioxidant and antiproliferative activities in different maturation stages of broccoli (Brassica oleracea Italica) biofortified with selenium. Food Chem. 2016, 190, 771–776. [Google Scholar] [CrossRef] [PubMed]
- Feizollahi, E.; Mirmahdi, R.S.; Zoghi, A.; Zijlstra, R.T.; Roopesh, M.S.; Vasanthan, T. Review of the beneficial and anti-nutritional qualities of phytic acid, and procedures for removing it from food products. Food Res. Int. 2021, 143, 110284. [Google Scholar] [CrossRef]
- Faizal, F.I.; Ahmad, N.H.; Yaacob, J.S.; Halim-Lim, S.A.; Abd Rahim, M.H. Food processing to reduce antinutrients in plant-based foods. Int. Food Res. J. 2023, 30, 25–45. [Google Scholar] [CrossRef]
- Ogbede, S.C.; Saidu, A.N.; Kabiru, A.Y.; Busari, M.B. Nutrient And Anti-Nutrient Compositions Of Brassica Oleracae Var. Capitata L. IOSR J. Pharm. 2015, 5, 2319–4219. [Google Scholar]
- Cominelli, E.; Pilu, R.; Sparvoli, F. Phytic acid and mineral biofortification strategies: From plant science to breeding and biotechnological approaches. Plants 2020, 9, 553. [Google Scholar] [CrossRef]
- Poblaciones, M.J.; Broadley, M.R. Foliar selenium biofortification of broccolini: Effects on plant growth and mineral accumulation. J. Hortic. Sci. Biotechnol. 2022, 97, 730–738. [Google Scholar] [CrossRef]
- Zhao, F.; McGrath, S.P. Extractable sulphate and organic sulphur in soils and their availability to plants. Plant Soil 1994, 164, 243–250. [Google Scholar] [CrossRef]
- Poblaciones, M.J.; Rodrigo, S.; Santamaria, O.; Chen, Y.; McGrath, S.P. Selenium accumulation and speciation in biofortified chickpea (Cicer arietinum L.) under Mediterranean conditions. J. Sci. Food Agric. 2014, 94, 1101–1106. [Google Scholar] [CrossRef]
- Gomez-Coronado, F.; Poblaciones, M.J.; Almeida, A.S.; Cakmak, I. Combined Zinc and Nitrogen Fertilization in Different Bread Wheat Genotypes Grown under Mediterranean Conditions. Cereal Res. Commun. 2017, 45, 154–165. [Google Scholar] [CrossRef][Green Version]
- Teixeira, D.M.; Canelas, V.C.; Canto, A.M.D.; Teixeira, J.M.; Dias, C.B. HPLC-DAD Quantification of Phenolic Compounds Contributing to the Antioxidant Activity of Maclura pomifera, Ficus carica and Ficus elastica Extracts. Anal. Lett. 2009, 42, 2986–3003. [Google Scholar] [CrossRef]
- Cano, A.; Acosta, M.; Arnao, M.B. A method to measure antioxidant activity in organic media: Application to lipophilic vitamins. Redox Rep. 2000, 5, 365–370. [Google Scholar] [CrossRef] [PubMed]
- Casquete, R.; Castro, S.M.; Martín, A.; Ruiz-Moyano, S.; Saraiva, J.A.; Córdoba, M.G.; Teixeira, P. Evaluation of the effect of high pressure on total phenolic content, antioxidant and antimicrobial activity of citrus peels. Innov. Food Sci. Emerg. Technol. 2015, 31, 37–44. [Google Scholar] [CrossRef]
- Bouranis, D.L.; Stylianidis, G.P.; Manta, V.; Karousis, E.N.; Tzanaki, A.; Dimitriadi, D.; Bouzas, E.A.; Siyiannis, V.F.; Constantinou-Kokotou, V.; Chorianopoulou, S.N.; et al. Floret Biofortification of Broccoli Using Amino Acids Coupled with Selenium under Different Surfactants: A Case Study of Cultivating Functional Foods. Plants 2023, 12, 1272. [Google Scholar] [CrossRef] [PubMed]
- Šindelářová, K.; Száková, J.; Tremlová, J.; Mestek, O.; Praus, L.; Kana, A.; Najmanová, J.; Tlustoš, P. The response of broccoli (Brassica oleracea convar. italica) varieties on foliar application of selenium: Uptake, translocation, and speciation. Food Addit. Contam. Part A 2015, 32, 2027–2038. [Google Scholar]
- Hasanuzzaman, M.; Bhuyan, M.H.M.B.; Raza, A.; Hawrylak-Nowak, B.; Matraszek-Gawron, R.; Al Mahmud, J.; Nahar, K.; Fujita, M. Selenium in plants: Boon or bane? Environ. Exp. Bot. 2020, 178, 104170. [Google Scholar] [CrossRef]
- Gui, J.Y.; Rao, S.; Huang, X.; Liu, X.; Cheng, S.; Xu, F. Interaction between selenium and essential micronutrient elements in plants: A systematic review. Sci. Total Environ. 2022, 853, 158673. [Google Scholar] [CrossRef] [PubMed]
- Cunha, M.L.O.; de Oliveira, L.C.A.; Silva, V.M.; Montanha, G.S.; dos Reis, A.R. Selenium increases photosynthetic capacity, daidzein biosynthesis, nodulation and yield of peanuts plants (Arachis hypogaea L.). Plant Physiol. Biochem. 2022, 190, 231–239. [Google Scholar] [CrossRef]
- Raza, M.A.S.; Aslam, M.U.; Valipour, M.; Iqbal, R.; Haider, I.; Mustafa, A.E.-Z.M.A.; Elshikh, M.S.; Ali, I.; Roy, R.; Elshamly, A.M.S. Seed priming with selenium improves growth and yield of quinoa plants suffering drought. Sci. Rep. 2024, 14, 886. [Google Scholar] [CrossRef]
- Kleiber, T.; Krzesiński, W.; Przygocka-Cyna, K.; Spiżewski, T. Alleviation Effect of Selenium on Manganese Stress of Plants. Ecol. Chem. Eng. S 2018, 25, 143–152. [Google Scholar] [CrossRef]
- Lanza, M.G.D.B.; Reis, A.R. dos Roles of selenium in mineral plant nutrition: ROS scavenging responses against abiotic stresses. Plant Physiol. Biochem. 2021, 164, 27–43. [Google Scholar] [CrossRef] [PubMed]
- Saeedi, M.; Soltani, F.; Babalar, M.; Izadpanah, F.; Wiesner-Reinhold, M.; Baldermann, S. Selenium Fortification Alters the Growth, Antioxidant Characteristics and Secondary Metabolite Profiles of Cauliflower (Brassica oleracea var. botrytis) Cultivars in Hydroponic Culture. Plants 2021, 10, 1537. [Google Scholar] [CrossRef] [PubMed]
- Golubkina, N.; Antoshkina, M.; Bondareva, L.; Sekara, A.; Campagna, E.; Caruso, G. Effect of Foliar Application of Sodium Selenate on Mineral Relationships in Brassicaceae Crops. Horticulturae 2023, 9, 535. [Google Scholar] [CrossRef]
- Rao, S.; Gou, Y.; Yu, T.; Cong, X.; Gui, J.; Zhu, Z.; Zhang, W.; Liao, Y.; Ye, J.; Cheng, S.; et al. Effects of selenate on Se, flavonoid, and glucosinolate in broccoli florets by combined transcriptome and metabolome analyses. Food Res. Int. 2021, 146, 110463. [Google Scholar] [CrossRef]
- Golubkina, N.; Moldovan, A.; Kekina, H.; Kharchenko, V.; Sekara, A.; Vasileva, V.; Skrypnik, L.; Tallarita, A.; Caruso, G. Joint biofortification of plants with selenium and iodine: New field of discoveries. Plants 2021, 10, 1352. [Google Scholar] [CrossRef]
- do Nascimento da Silva, E.; Cadore, S. Bioavailability Assessment of Copper, Iron, Manganese, Molybdenum, Selenium, and Zinc from Selenium-Enriched Lettuce. J. Food Sci. 2019, 84, 2840–2846. [Google Scholar] [CrossRef]
- Rivera-Martin, A.; Broadley, M.R.; Poblaciones, M.J. Soil and foliar zinc application to biofortify broccoli (Brassica oleracea var. Italica L.): Effects on the zinc concentration and bioavailability. Plant Soil Environ. 2020, 66, 113–118. [Google Scholar] [CrossRef]
- Page, V.; Feller, U. Heavy metals in crop plants: Transport and redistribution processes on the whole plant level. Agronomy 2015, 5, 447–463. [Google Scholar] [CrossRef]
- Liu, M.; Zhang, L.; Ser, S.L.; Cumming, J.R.; Ku, K.-M. Comparative Phytonutrient Analysis of Broccoli By-Products: The Potentials for Broccoli By-Product Utilization. Molecules 2018, 23, 900. [Google Scholar] [CrossRef] [PubMed]
- Majkowska-gadomska, J.; Kaliniewicz, Z.; Francke, A.; Sałata, A. An Evaluation of the Biometric Parameters and Chemical Composition of the Florets, Leaves, and Stalks of Broccoli Plants Grown in Different Soil Types. Appl. Sci. 2024, 14, 4411. [Google Scholar] [CrossRef]
- Nagraj, G.S.; Chouksey, A.; Jaiswal, S.; Jaiswal, A.K. Broccoli. In Nutritional Composition and Antioxidant Properties of Fruits and Vegetables; Academic Press: Cambridge, MA, USA, 2020; pp. 5–17. ISBN 9780128127803. [Google Scholar]
- Wani, S.H.; Gaikwad, K.; Razzaq, A.; Samantara, K.; Kumar, M.; Govindan, V. Improving Zinc and Iron Biofortification in Wheat through Genomics Approaches. Mol. Biol. Rep. 2022, 49, 8007–8023. [Google Scholar] [CrossRef] [PubMed]
- Sarkhel, S.; Roy, A. Phytic acid and its reduction in pulse matrix: Structure–function relationship owing to bioavailability enhancement of micronutrients. J. Food Process Eng. 2022, 45, e14030. [Google Scholar] [CrossRef]
- Morris, E.R.; Ellis, R. Usefulness of the dietary phytic acid/zinc molar ratio as an index of zinc bioavailability to rats and humans. Biol. Trace Elem. Res. 1989, 19, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Hallberg, L.; Brune, M.; Rossander, L. Iron absorption in man: Ascorbic acid and dose-dependent inhibition by phytate. Am. J. Clin. Nutr. 1989, 49, 140–144. [Google Scholar] [CrossRef] [PubMed]
- Evans, W.J.; Martin, C.J. Interactions of Mg(II), Co(II), Ni(II), and Zn(II) with phytic acid. VIII. A calorimetric study. J. Inorg. Biochem. 1988, 32, 259–268. [Google Scholar] [CrossRef]
- Gibson, R.S. The role of diet- and host-related factors in nutrient bioavailability and thus in nutrient-based dietary requirement estimates. Food Nutr. Bull. 2007, 28, S77–S100. [Google Scholar] [CrossRef]
- Mezzetti, B.; Biondi, F.; Balducci, F.; Capocasa, F.; Mei, E.; Vagnoni, M.; Visciglio, M.; Mazzoni, L. Variation of Nutritional Quality Depending on Harvested Plant Portion of Broccoli and Black Cabbage. Appl. Sci. 2022, 12, 6668. [Google Scholar] [CrossRef]
- Poblaciones, M.J.; García-Latorre, C.; Martinez, M.; Velazquez, R. Effects of soil and foliar applications of iron with or without nitrogen on production and nutritional quality of broccoli (Brassica oleracea var. italica). Crop Pasture Sci. 2023, 75, 23208. [Google Scholar] [CrossRef]
- Abdalla, M.A.; Wick, J.E.; Famuyide, I.M.; McGaw, L.J.; Mühling, K.H. Selenium enrichment of green and red lettuce and the induction of radical scavenging potential. Horticulturae 2021, 7, 488. [Google Scholar] [CrossRef]
- Fernández-León, M.F.; Fernández-León, A.M.; Lozano, M.; Ayuso, M.C.; González-Gómez, D. Altered commercial controlled atmosphere storage conditions for ‘Parhenon’ broccoli plants (Brassica oleracea L. var. italica). Influence on the outer quality parameters and on the health-promoting compounds. LWT—Food Sci. Technol. 2013, 50, 665–672. [Google Scholar] [CrossRef]


| Se Treatment | Stem+Leaves Weight (g) | Stem+Leaves Height (cm) | Floret Weight (g) | D (cm) | d (cm) |
|---|---|---|---|---|---|
| Control | 280 ± 23 a | 28.3 ± 0.2 c | 86.4 ± 3.0 a | 8.4 ± 0.2 a | 7.3 ± 0.1 a |
| 0.05-Se | 311 ± 13 a | 29.3 ± 1.0 bc | 92.8 ± 3.2 a | 8.8 ± 0.2 a | 7.2 ± 0.2 a |
| 0.10-Se | 282 ± 21 a | 32.0 ± 0.8 a | 97.8 ± 5.9 a | 8.7 ± 0.2 a | 8.0 ± 0.0 a |
| 0.15-Se | 300 ± 11 a | 31.2 ± 0.7 ab | 98.8 ± 10.4 a | 8.8 ± 0.3 a | 7.8 ± 0.6 a |
| df | 3 | 3 | 3 | 3 | 3 |
| F-value | 0.72 | 5.35 * | 0.79 | 0.8 | 1.59 |
| Source | Plant Fraction (P) | Se Treatment (T) | P × T |
|---|---|---|---|
| df | 2 | 3 | 6 |
| Ca | 366.28 *** | 0.24 | 0.10 |
| K | 50.72 *** | 0.44 | 0.58 |
| Na | 29.68 *** | 1.63 | 1.78 |
| Mg | 91.97 *** | 0.32 | 0.30 |
| P | 87.74 *** | 4.68 | 0.71 |
| S | 147.31 *** | 1.26 | 0.90 |
| Co | 269.13 *** | 1.39 | 0.30 |
| Cu | 55.86 *** | 0.41 | 2.66 |
| Fe | 12.46 ** | 0.83 | 0.77 |
| Mn | 4.42 | 0.49 | 0.60 |
| Se | 52.61 *** | 147.04 *** | 5.23 ** |
| Zn | 2.81 | 1.17 | 0.98 |
| df | 1 | 3 | 3 |
| Phytic acid | 150.99 *** | 104.55 *** | 132.00 *** |
| PA:Ca | 553.14 *** | 48.10 *** | 44.74 *** |
| PA:Mg | 144.03 *** | 52.71 *** | 44.05 *** |
| PA:Fe | 9.04 * | 6.55 * | 9.21 * |
| PA:Se | 58 ** | 223.19 *** | 111.83 *** |
| PA:Zn | 13.31* | 15.44 ** | 5.99 * |
| ABTS | 263.7 *** | 14.73 *** | 7.16 ** |
| DPPH | 877.82 *** | 18.21 *** | 0.63 |
| TPC | 72.97 *** | 77.98 *** | 3.38 * |
| Parameter | Unit | Stem+Leaves | Raw Floret | Boiled Floret |
|---|---|---|---|---|
| Ca | g kg−1 | 13.2 ± 0.7 a | 2.6 ± 0.1 b | 2.6 ± 0.1 b |
| K | 17.1 ± 0.7 b | 23.8 ± 0.5 a | 18.5 ± 0.3 b | |
| Na | 618 ± 35 a | 439 ± 20 b | 381 ± 19 b | |
| Mg | 1.71 ± 0.06 a | 1.31 ± 0.04 b | 1.02 ± 0.03 c | |
| P | 3.1 ± 0.0 b | 4.6 ± 0.1 a | 4.4 ± 0.1 a | |
| S | 2.7 ± 0.1 c | 6.8 ± 0.2 a | 4.9 ± 0.1 b | |
| Co | mg kg−1 | 0.13 ± 0.00 c | 0.26 ± 0.01 a | 0.17 ± 0.01 b |
| Cu | 1.0 ± 0.2 c | 3.0 ± 0.2 a | 2.3 ± 0.1 b | |
| Fe | 24.9 ± 1.9 b | 38.6 ± 3.3 a | 26.5 ± 1.2 b | |
| Mn | 17.4 ± 0.7 a | 16.6 ± 0.6 a | 14.5 ± 0.4 a | |
| Se | 11.5 ± 2.9 b | 33.9 ± 8.4 a | 29.0 ± 7.6 a | |
| Zn | 14.8 ± 6.8 a | 27.2 ± 1.3 a | 17.4 ± 0.6 a | |
| Phytic acid | g kg−1 | 1.6 ± 0.1 b | 3.4 ± 0.7 a | - |
| ABTS | mg Trolox 100 g−1 | 113 + 5 b | 156 + 2 a | - |
| DPPH | 419 ± 14 b | 741 ± 14 a | - | |
| TPC | mg GAE 100 g−1 | 169 ± 29 b | 272 ± 30 a | - |
| Parameter | Unit | Control | 0.05-Se | 0.10-Se | 0.15-Se |
|---|---|---|---|---|---|
| Ca | g kg−1 | 6.3 ± 0.6 a | 6.2 ± 0.6 a | 5.7 ± 0.6 a | 6.2 ± 0.7 a |
| K | 19.2 ± 0.4 a | 19.9 ± 0.4 a | 19.7 ± 0.3 a | 20.4 ± 0.4 a | |
| Na | 421 ± 9 a | 475 ± 12 a | 504 ± 16 a | 516 ± 20 a | |
| Mg | 1.34 ± 0.04 a | 1.35 ± 0.03 a | 1.31 ± 0.04 a | 1.39 ± 0.04 a | |
| P | 3.8 ± 0.1 a | 4.2 ± 0.1 a | 3.9 ± 0.1 a | 4.2 ± 0.1 a | |
| S | 4.7 ± 0.2 a | 4.8 ± 0.2 a | 4.7 ± 0.2 a | 5.1 ± 0.2 a | |
| Co | mg kg−1 | 0.19 ± 0.01 a | 0.17 ± 0.01 a | 0.20 ± 0.01 a | 0.20 ± 0.01 a |
| Cu | 2.2 ± 0.2 a | 2.2 ± 0.1 a | 1.9 ± 0.1 a | 2.2 ± 0.1 a | |
| Fe | 32.0 ± 1.4 a | 27.1 ± 0.5 a | 28.8 ± 1.0 a | 32.2 ± 1.3 a | |
| Mn | 16.1 ± 0.4 a | 16.1 ± 0.2 a | 15.4 ± 0.3 a | 17.0 ± 0.2 a | |
| Se | 0.3 ± 0.0 d | 14.0 ± 0.6 c | 31.0 ± 1.7 b | 54.0 ± 2.7 a | |
| Zn | 17.8 ± 1.2 a | 26.4 ± 2.7 a | 16.6 ± 0.8 | 18.3 ± 0.9 a | |
| Phytic acid | g kg−1 | 4.5 ± 1.3 a | 1.9 ± 0.2 b | 1.6 ± 0.2 b | 1.9 ± 0.1 b |
| ABTS | mg Trolox 100 g−1 | 120 ± 14 c | 144 ± 10 a | 139 ± 10 ab | 135 ± 6 b |
| DPPH | 511 ± 73 b | 600 ± 71 a | 600 ± 78 a | 610 ± 70 a | |
| TPC | mg GAE 100 g−1 | 165 ± 37 c | 370 ± 28 a | 219 ± 23 b | 129 ± 13 c |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Poblaciones, M.J.; García-Latorre, C.; Velazquez, R.; Broadley, M.R. Effects of Selenate Application on Growth, Nutrient Bioaccumulation, and Bioactive Compounds in Broccoli (Brassica oleracea var. italica L.). Horticulturae 2024, 10, 808. https://doi.org/10.3390/horticulturae10080808
Poblaciones MJ, García-Latorre C, Velazquez R, Broadley MR. Effects of Selenate Application on Growth, Nutrient Bioaccumulation, and Bioactive Compounds in Broccoli (Brassica oleracea var. italica L.). Horticulturae. 2024; 10(8):808. https://doi.org/10.3390/horticulturae10080808
Chicago/Turabian StylePoblaciones, Maria J., Carlos García-Latorre, Rocio Velazquez, and Martin R. Broadley. 2024. "Effects of Selenate Application on Growth, Nutrient Bioaccumulation, and Bioactive Compounds in Broccoli (Brassica oleracea var. italica L.)" Horticulturae 10, no. 8: 808. https://doi.org/10.3390/horticulturae10080808
APA StylePoblaciones, M. J., García-Latorre, C., Velazquez, R., & Broadley, M. R. (2024). Effects of Selenate Application on Growth, Nutrient Bioaccumulation, and Bioactive Compounds in Broccoli (Brassica oleracea var. italica L.). Horticulturae, 10(8), 808. https://doi.org/10.3390/horticulturae10080808






